Contents
- What is Addison’s disease
What is Addison’s disease
Addison’s disease is a rare endocrine disorder that occurs when the adrenal glands do not produce enough of their hormones – cortisol (a “stress” hormone) and aldosterone and androgens (the other hormones made by the adrenal glands) 1. Addison’s disease can be caused by damage to the adrenal glands, autoimmune conditions, and certain genetic conditions. Some of the symptoms include changes in blood pressure, chronic diarrhea, darkening of the skin, paleness, extreme weakness, fatigue, nausea and vomiting, salt craving, and weight loss. Treatment with replacement corticosteroids usually controls the symptoms.
Addison’s disease, the common term for primary adrenal insufficiency, occurs when the adrenal glands are damaged and cannot produce enough of the adrenal hormone cortisol. The adrenal hormone aldosterone and androgens may also be lacking. Addison’s disease is seen in all age groups and affects male and female equally. Addison’s disease affects 110 to 144 of every 1 million people in developed countries 2.
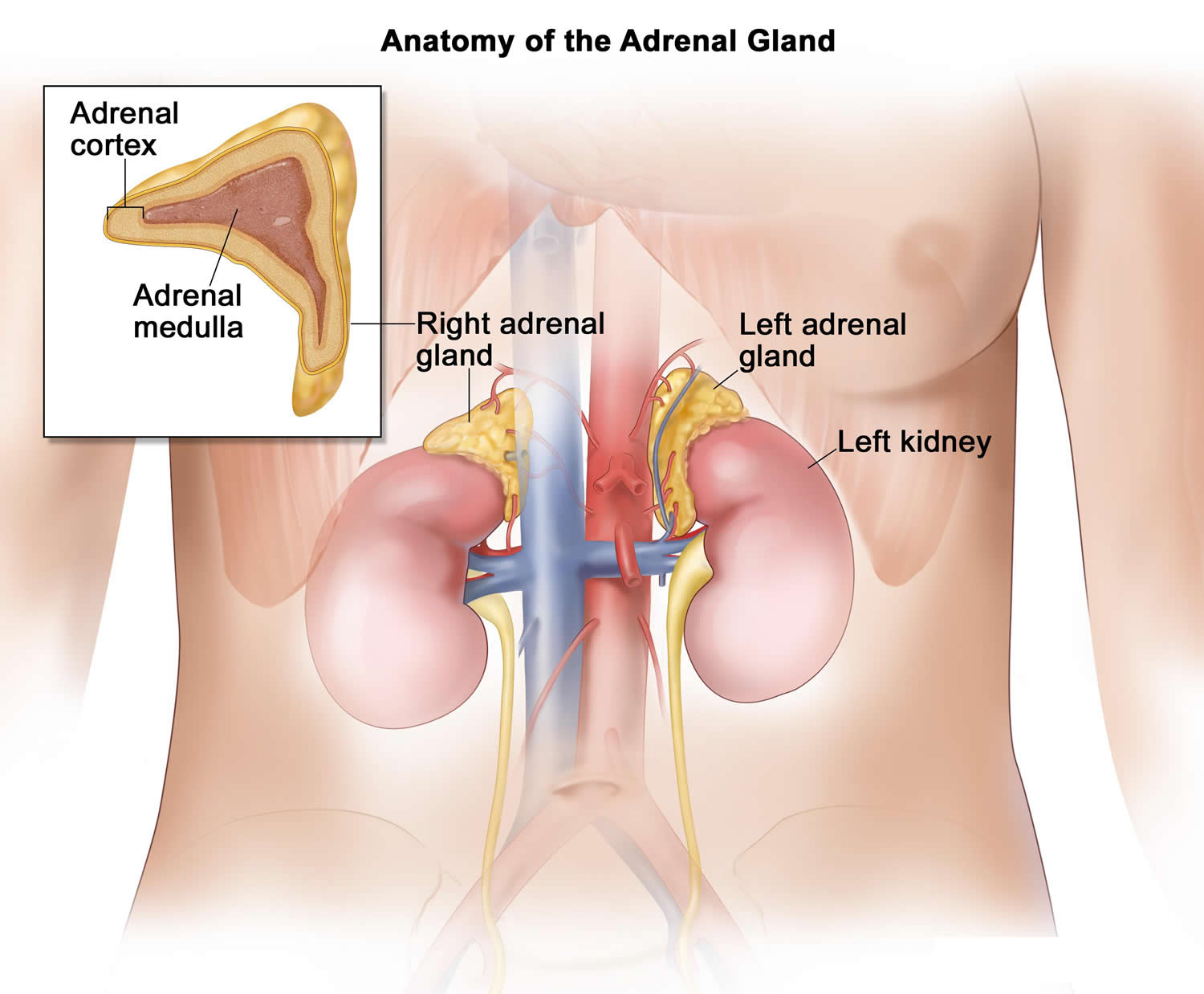
Figure 1. Adrenal gland anatomy
This disease is named after Thomas Addison, who first described patients affected by this disorder in 1855, in the book titled “On the constitutional and local effects of the disease of supra renal capsule” 3. Addison’s disease can present as a life-threatening crisis, because it is frequently unrecognized in its early stages. The basis of Addison’s disease has dramatically changed from an infectious cause to autoimmune pathology since its initial description. However, tuberculosis is still the predominant cause of Addison’s disease in developing countries 4.
Your adrenal glands are composed of two sections (see Figure 1 above). The interior (medulla) produces adrenaline-like hormones. The outer layer (cortex) produces a group of hormones called corticosteroids, which include glucocorticoids, mineralocorticoids and male sex hormones (androgens).
Some of the hormones the cortex produces are essential for life — the glucocorticoids (Cortisol) and the mineralocorticoids (Aldosterone).
What do adrenal hormones do ?
Adrenal hormones, such as cortisol and aldosterone, play key roles in the functioning of the human body, such as regulating blood pressure; metabolism, the way the body uses digested food for energy; and the body’s response to stress. In addition, the body uses the adrenal hormone dehydroepiandrosterone (DHEA) to make androgens and estrogens, the male and female sex hormones.
Cortisol
Cortisol belongs to the class of hormones called glucocorticoids, which affect almost every organ and tissue in the body. Cortisol’s most important job is to help the body respond to stress. Among its many tasks, cortisol helps:
- maintain blood pressure and heart and blood vessel function
- slow the immune system’s inflammatory response—how the body recognizes and defends itself against bacteria, viruses, and substances that appear foreign and harmful
- regulate metabolism
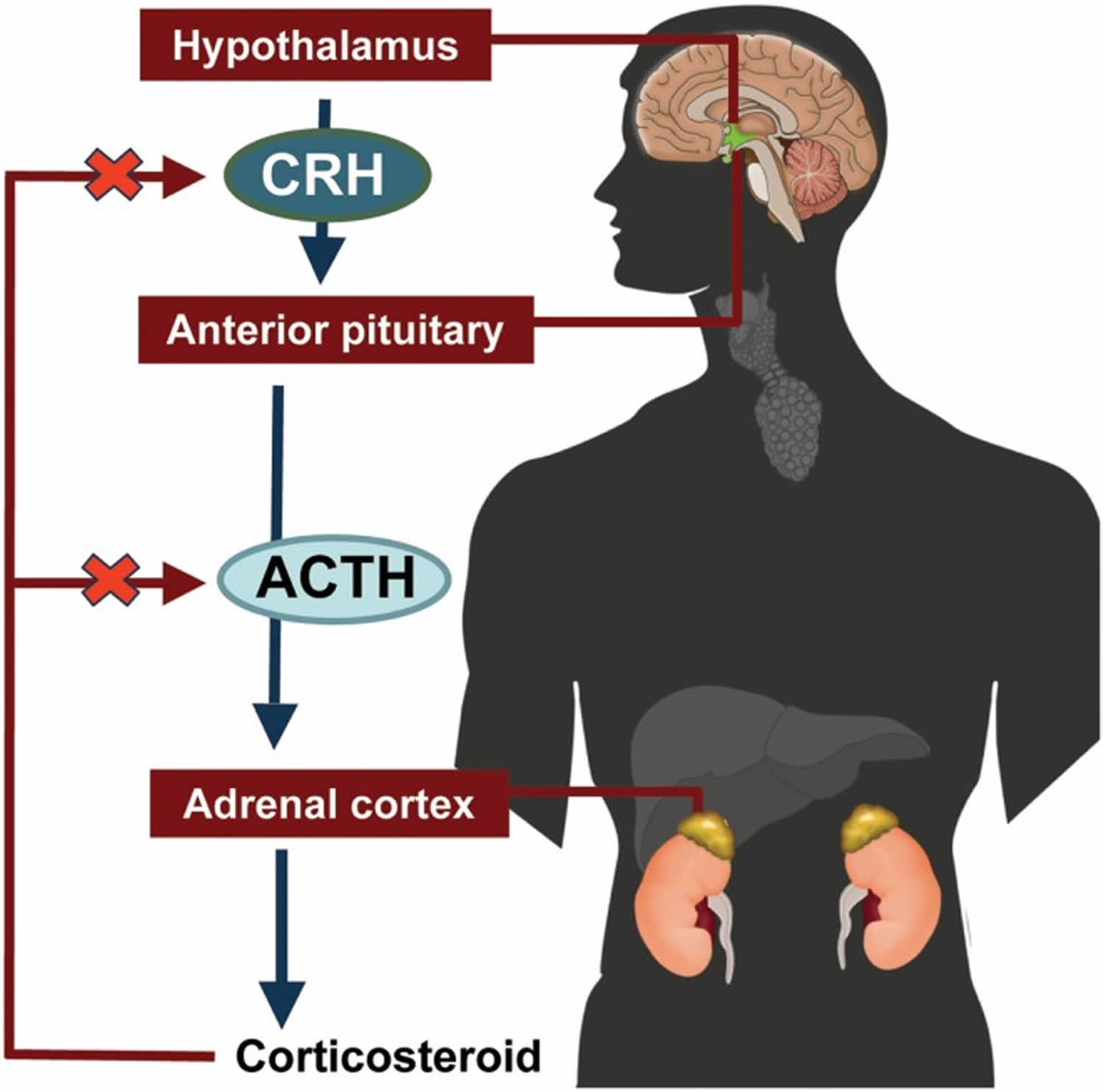
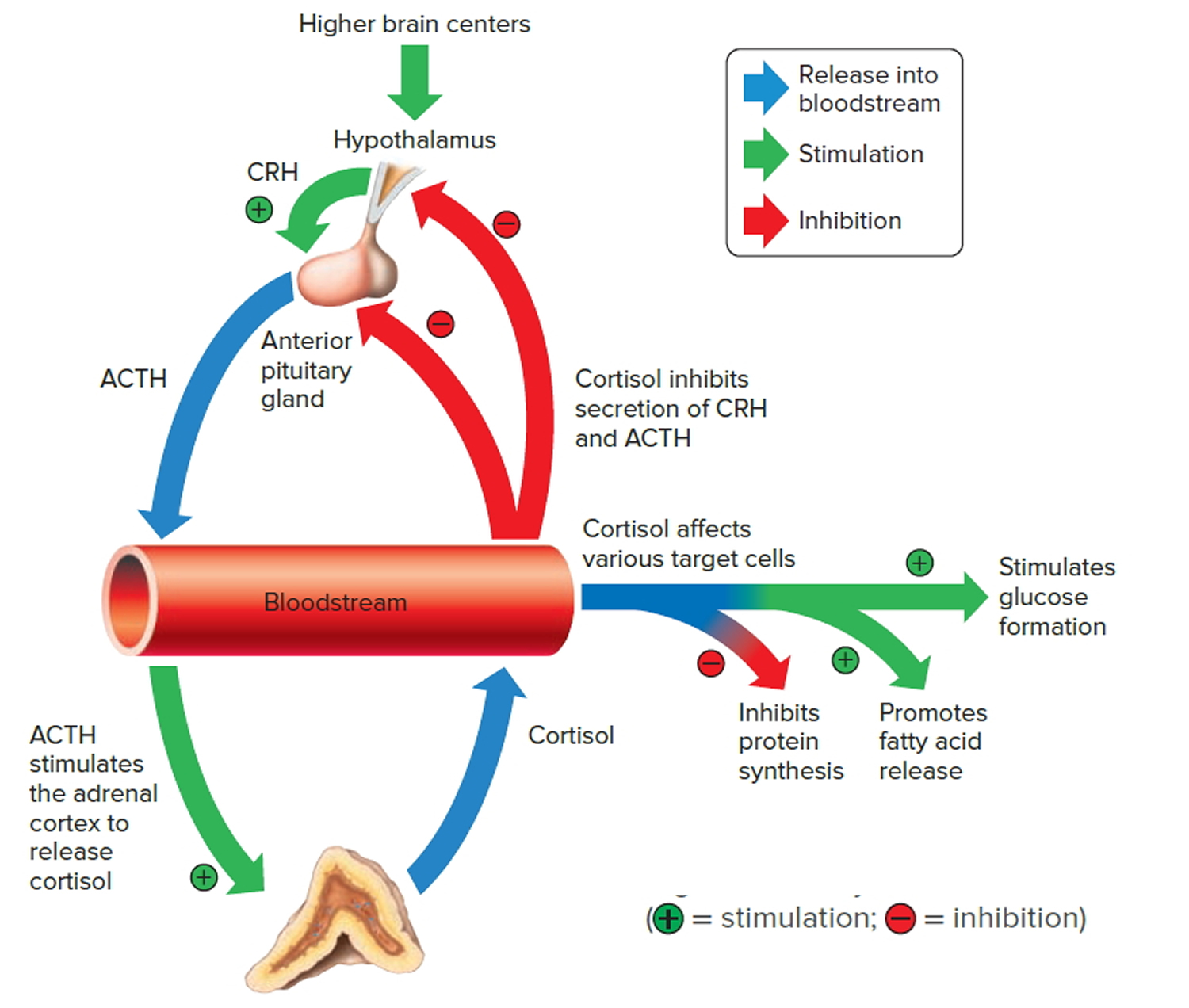
The amount of cortisol produced by the adrenal glands is precisely balanced. Like many other hormones, cortisol is regulated by the hypothalamus, which is a part of the brain, and the pituitary gland. First, the hypothalamus releases a “trigger” hormone called corticotropin-releasing hormone (CRH), which signals the pituitary gland to send out adrenocorticotropic hormone (ACTH). ACTH stimulates the adrenal glands to produce cortisol. Cortisol then signals back to both the pituitary gland and hypothalamus to decrease these trigger hormones.
Figure 2. ACTH and Cortisol production and control by the hypothalamus (the Hypothalamic-Pituitary-Adrenal axis)
 [Source 5]
[Source 5]
Figure 3. Negative feedback regulates cortisol secretion
Aldosterone
Aldosterone belongs to the class of hormones called mineralocorticoids, also produced by the adrenal glands. Aldosterone helps maintain blood pressure and the balance of sodium and potassium in the blood. When aldosterone production falls too low, the body loses too much sodium and retains too much potassium.
The decrease of sodium in the blood can lead to a drop in both blood volume—the amount of fluid in the blood—and blood pressure. Too little sodium in the body also can cause a condition called hyponatremia. Symptoms of hyponatremia include feeling confused and fatigued and having muscle twitches and seizures.
Too much potassium in the body can lead to a condition called hyperkalemia. Hyperkalemia may have no symptoms; however, it can cause irregular heartbeat, nausea, and a slow, weak, or an irregular pulse.
Dehydroepiandrosterone
Dehydroepiandrosterone is another hormone produced by your adrenal glands. The body uses DHEA to make the sex hormones, androgen and estrogen. With adrenal insufficiency, the adrenal glands may not make enough DHEA. Healthy men derive most androgens from the testes. Healthy women and adolescent girls get most of their estrogens from the ovaries. However, women and adolescent girls may have various symptoms from DHEA insufficiency, such as loss of pubic hair, dry skin, a reduced interest in sex, and depression.
Addison’s disease causes
Addison’s disease results when your adrenal glands are damaged, producing insufficient amounts of the hormone cortisol and often aldosterone as well. These glands are located just above your kidneys.
The failure of your adrenal glands to produce adrenocortical hormones is most commonly the result of the body attacking itself (autoimmune disease). An auto-immune disorder in which the body’s immune system makes antibodies which attack the cells of the adrenal cortex and slowly destroys them. This can take months to years. For unknown reasons, your immune system views the adrenal cortex as foreign, something to attack and destroy.
Other causes of adrenal gland failure may include:
- Tuberculosis. Tuberculosis was the leading cause of Addison disease up until the middle of the 20th century, when antibiotics were introduced that successfully treated tuberculosis. Tuberculosis (TB) is the most common cause of Addison’s disease worldwide, but it’s rare in the US. Tuberculosis (TB) is a bacterial infection that mostly affects the lungs but can also spread to other parts of your body. It can cause Addison’s disease if it damages your adrenal glands.
- Infections (bacterial, fungal, tuberculosis) of the adrenal glands e.g. CMV virus
- Cancer that spreads to the adrenal glands
- Surgical removal of the adrenal glands
- Amyloidosis—protein build up in organs (very rare)
- Bleeding into the adrenal glands, which may present as adrenal crisis without any preceding symptoms.
Primary Adrenal Insufficiency: Addison’s Disease
The outer layer of the adrenal glands is called the adrenal cortex. If the cortex is damaged, it may not be able to produce enough cortisol.
A common cause of primary adrenal insufficiency is an autoimmune disease that causes the immune system to attack healthy tissues. In the case of Addison’s disease, the immune system turns against the adrenal gland(s). There are some very rare syndromes (several diseases that occur together) that can cause autoimmune adrenal insufficiency.
Autoimmune Disorders
Up to 80 percent of Addison’s disease cases are caused by an autoimmune disorder, which is when the body’s immune system attacks the body’s own cells and organs 6. In autoimmune Addison’s, which mainly occurs in middle-aged females, the immune system gradually destroys the adrenal cortex—the outer layer of the adrenal glands 6.
Primary adrenal insufficiency occurs when at least 90 percent of the adrenal cortex has been destroyed 2. As a result, both cortisol and aldosterone are often lacking. Sometimes only the adrenal glands are affected. Sometimes other endocrine glands are affected as well, as in polyendocrine deficiency syndrome.
Autoimmune Polyendocrine Syndrome
Autoimmune polyendocrine syndrome is a rare cause of Addison’s disease. Sometimes referred to as multiple endocrine deficiency syndrome, polyendocrine deficiency syndrome is classified into type 1 and type 2.
Autoimmune polyendocrine syndrome type 1
Type 1 is inherited and occurs in children. Symptoms start during childhood. Almost any organ can be affected by autoimmune damage. Fortunately it is extremely rare—only several hundred cases have been reported worldwide. Conditions associated with autoimmune polyendocrine syndrome type 1 include:
In addition to adrenal insufficiency, these children may have:
- Underactive parathyroid glands, which are four pea-sized glands located on or near the thyroid gland in the neck; they produce a hormone that helps maintain the correct balance of calcium in the body.
- Delayed or slow sexual development.
- Vitamin B12 malabsorption / deficiency (pernicious anemia), a severe type of anemia; anemia is a condition in which red blood cells are fewer than normal, which means less oxygen is carried to the body’s cells. With most types of anemia, red blood cells are smaller than normal; however, in pernicious anemia, the cells are bigger than normal.
- Candidiasis (chronic yeast infection).
- Chronic hepatitis, a liver disease.
Autoimmune polyendocrine syndrome Type 2
Researchers think type 2, which is sometimes called Schmidt’s syndrome, is also inherited. Type 2 usually affects young adults, symptoms primarily develop in adults aged 18 to 30 and may include:
- Addison’s disease
- Underactive or overactive thyroid function
- Delayed or slow sexual development
- Diabetes, in which a person has high blood glucose, also called high blood sugar or hyperglycemia
- White skin patches, a loss of pigment on areas of the skin (vitiligo)
- Celiac disease.
Secondary adrenal insufficiency
Adrenal insufficiency can also occur if your pituitary gland is diseased. The pituitary gland makes a hormone called adrenocorticotropic hormone (ACTH), which stimulates the adrenal cortex to produce its hormones. Inadequate production of ACTH can lead to insufficient production of hormones normally produced by your adrenal glands, even though your adrenal glands aren’t damaged. Secondary adrenal insufficiency can occur if your pituitary gland becomes damaged – for example, because of a tumour on the pituitary gland (pituitary adenoma). Doctors call this condition secondary adrenal insufficiency and is a separate condition to Addison’s disease.
Another more common cause of secondary adrenal insufficiency occurs when people who take corticosteroids for treatment of chronic conditions, such as asthma or arthritis, abruptly stop taking the corticosteroids.
Stoppage of Corticosteroid Medication
A temporary form of secondary adrenal insufficiency may occur when a person who has been taking a synthetic glucocorticoid hormone, called a corticosteroid, for a long time stops taking the medication. Corticosteroids are often prescribed to treat inflammatory illnesses such as rheumatoid arthritis, asthma, and ulcerative colitis. In this case, the prescription doses often cause higher levels than those normally achieved by the glucocorticoid hormones created by the body. When a person takes corticosteroids for prolonged periods, the adrenal glands produce less of their natural hormones. Once the prescription doses of corticosteroid are stopped, the adrenal glands may be slow to restart their production of the body’s glucocorticoids. To give the adrenal glands time to regain function and prevent adrenal insufficiency, prescription corticosteroid doses should be reduced gradually over a period of weeks or even months. Even with gradual reduction, the adrenal glands might not begin to function normally for some time, so a person who has recently stopped taking prescription corticosteroids should be watched carefully for symptoms of secondary adrenal insufficiency.
Surgical Removal of Pituitary Tumors
Another cause of secondary adrenal insufficiency is surgical removal of the usually noncancerous, ACTH-producing tumors of the pituitary gland that cause Cushing’s syndrome. Cushing’s syndrome is a hormonal disorder caused by prolonged exposure of the body’s tissues to high levels of the hormone cortisol. When the tumors are removed, the source of extra ACTH is suddenly gone and a replacement hormone must be taken until the body’s adrenal glands are able to resume their normal production of cortisol. The adrenal glands might not begin to function normally for some time, so a person who has had an ACTH-producing tumor removed and is going off of his or her prescription corticosteroid replacement hormone should be watched carefully for symptoms of adrenal insufficiency.
Changes in the Pituitary Gland
Less commonly, secondary adrenal insufficiency occurs when the pituitary gland either decreases in size or stops producing ACTH. These events can result from
- tumors or an infection in the pituitary
- loss of blood flow to the pituitary
- radiation for the treatment of pituitary or nearby tumors
- surgical removal of parts of the hypothalamus
- surgical removal of the pituitary
Infections
Tuberculosis (TB), an infection that can destroy the adrenal glands, accounts for 10 to 15 percent of Addison’s disease cases in developed countries. When primary adrenal insufficiency was first identified by Dr. Thomas Addison in 1849, tuberculosis (TB) was the most common cause of the disease. As tuberculosis (TB) treatment improved, the incidence of Addison’s disease due to tuberculosis (TB) of the adrenal glands greatly decreased. However, recent reports show an increase in Addison’s disease from infections such as tuberculosis (TB) and cytomegalovirus. Cytomegalovirus is a common virus that does not cause symptoms in healthy people; however, it does affect babies in the womb and people who have a weakened immune system—mostly due to HIV/AIDS. Other bacterial infections, such as Neisseria meningitidis, which is a cause of meningitis, and fungal infections can also lead to Addison’s disease.
Other Causes
Less common causes of Addison’s disease are:
- Spread of cancer to the adrenal glands
- Amyloidosis, a serious, though rare, group of diseases that occurs when abnormal proteins, called amyloids, build up in the blood and are deposited in tissues and organs
- Surgical removal of both adrenal glands (adrenalectomy) – for example, to remove a tumor
- Bleeding into the adrenal glands, which may present as adrenal crisis without any preceding symptoms. Very heavy bleeding into the adrenal glands, sometimes associated with meningitis or other types of severe sepsis.
- Genetic defects including abnormal adrenal gland development, an inability of the adrenal glands to respond to ACTH, or a defect in adrenal hormone production
- Medication-related causes, such as from anti-fungal medications and the anesthetic etomidate, which may be used when a person undergoes an emergency
- Intubation—the placement of a flexible, plastic tube through the mouth and into the trachea, or windpipe, to assist with breathing.
- Adrenoleukodystrophy – a rare, life-limiting inherited condition that affects the adrenal glands and nerve cells in the brain, and is mostly seen in young boys
- Certain treatments needed for Cushing’s syndrome – a collection of symptoms caused by very high levels of cortisol in the body
Addison’s disease symptoms
Addison’s disease symptoms usually develop slowly, often over several months, and may include:
- Extreme fatigue, chronic, or long lasting, fatigue
- Weight loss
- Loss of appetite or decreased appetite
- Darkening of your skin (hyperpigmentation)
- Low blood pressure that drops further when a person stands up, causing dizziness or fainting
- Salt craving
- Low blood sugar (hypoglycemia)
- Nausea, diarrhea or vomiting
- Abdominal pain
- Muscle or joint pains
- Muscle weakness
- Irritability
- Depression
- Body hair loss or sexual dysfunction in women
- Headache
- Sweating
- Irregular or absent menstrual periods
- In women, loss of interest in sex.
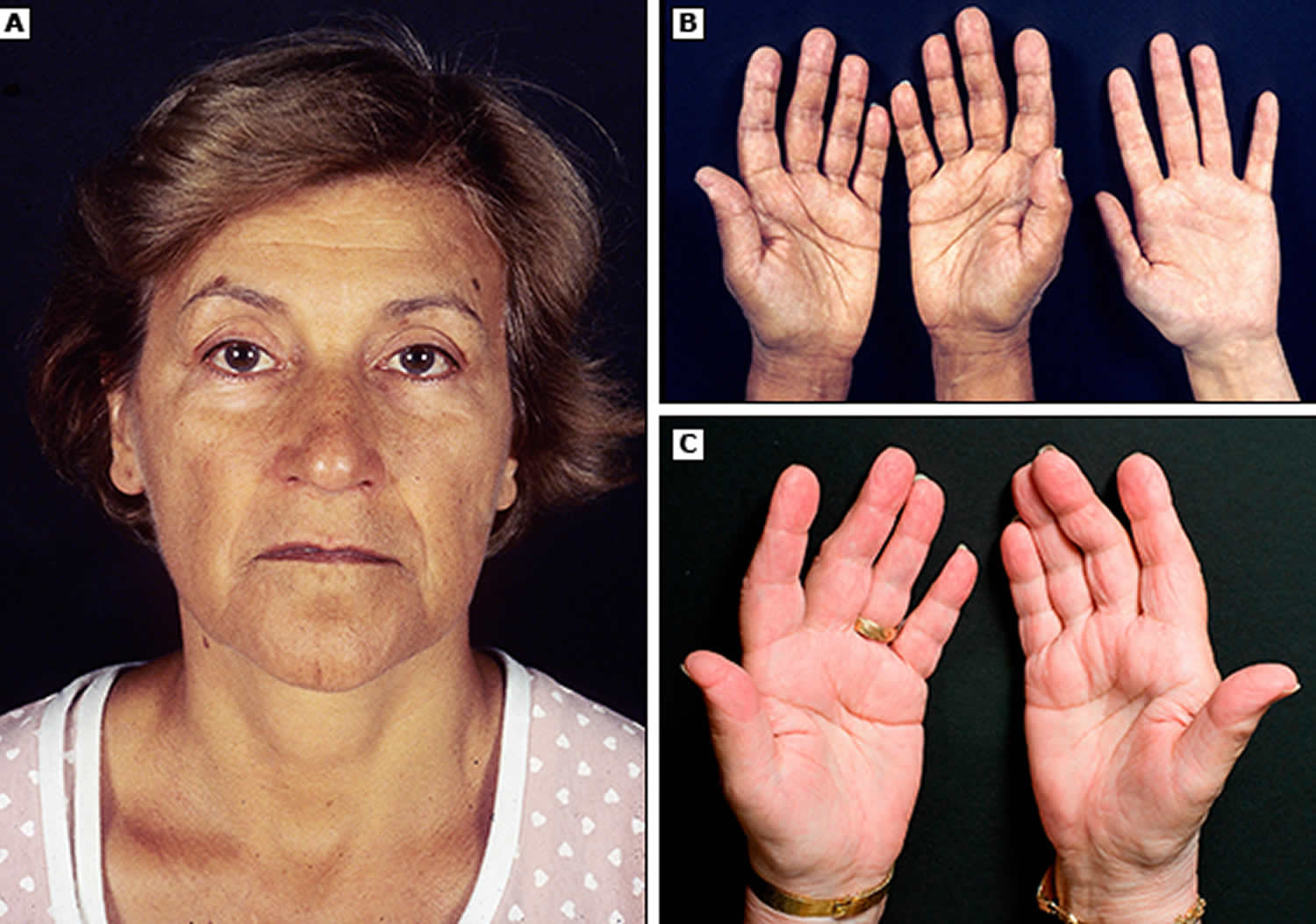
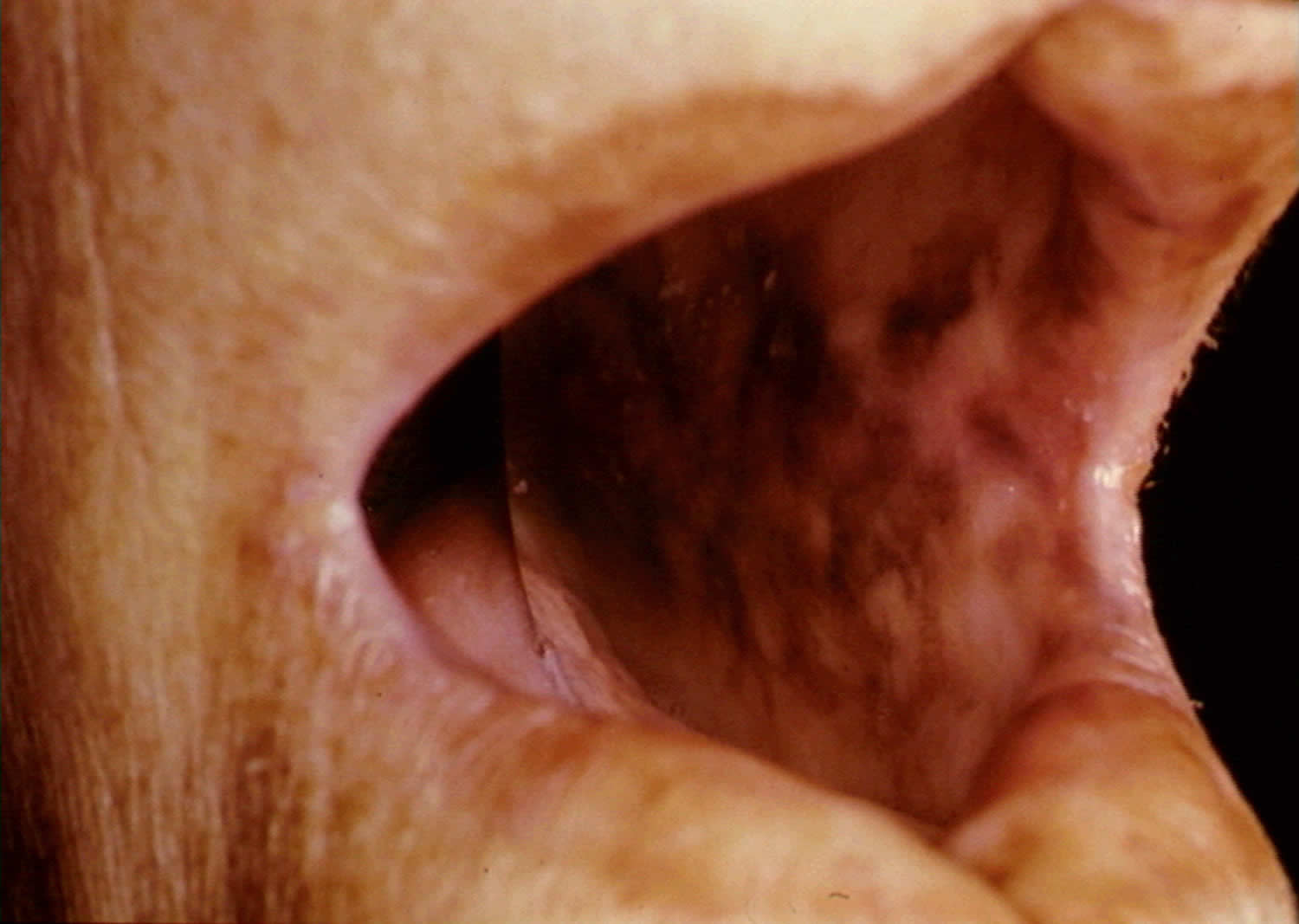
Hyperpigmentation, or darkening of the skin, can occur in Addison’s disease. This darkening is most visible on scars; skin folds; pressure points such as the elbows, knees, knuckles, and toes; lips; and mucous membranes such as the lining of the cheek.
The slowly progressing symptoms of adrenal insufficiency are often ignored until a stressful event, such as surgery, a severe injury, an illness, or pregnancy, causes them to worsen.
Figure 1. Hyperpigmentation in the hand
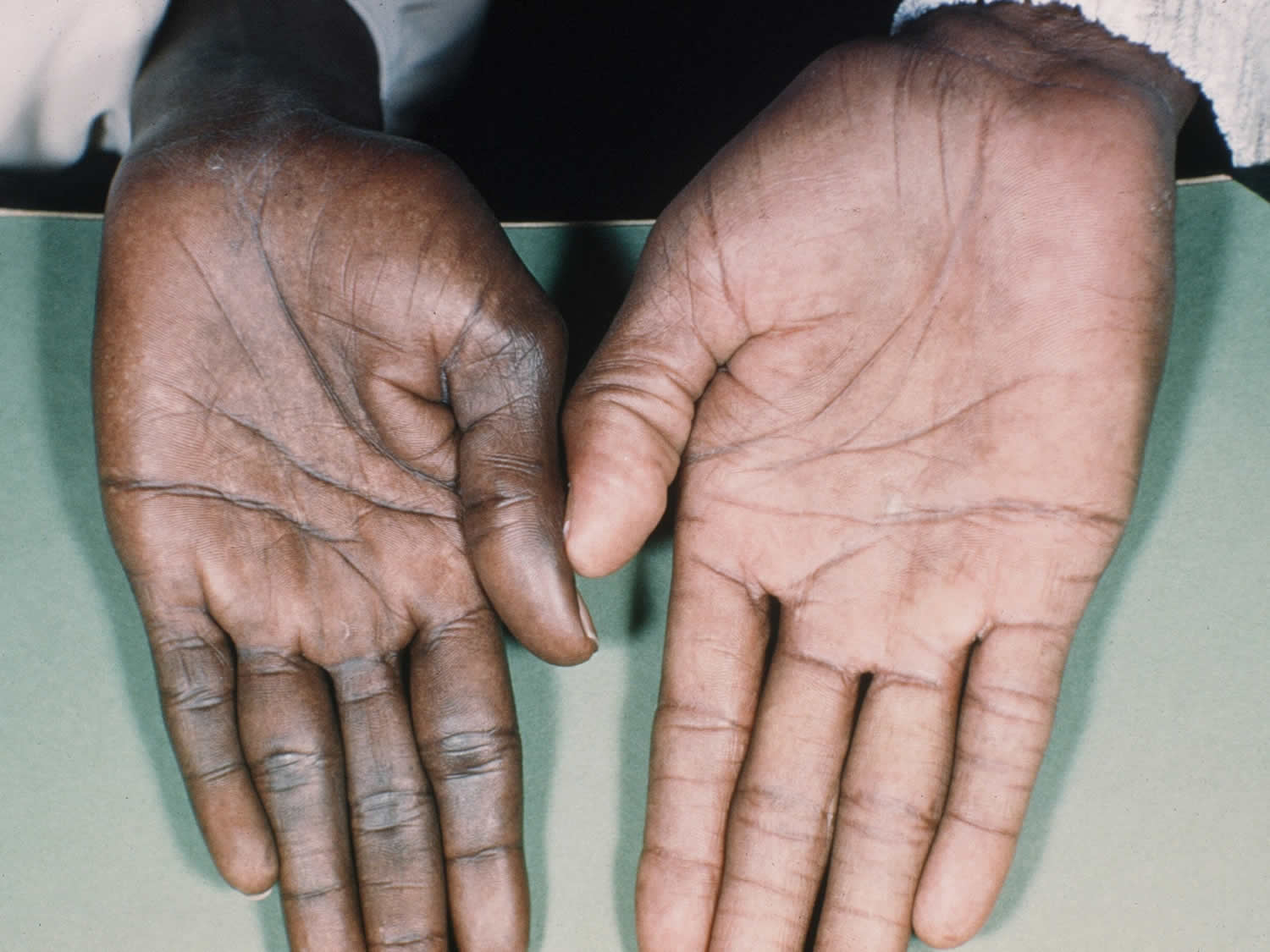
Figure 2. Hyperpigmentation in the mouth
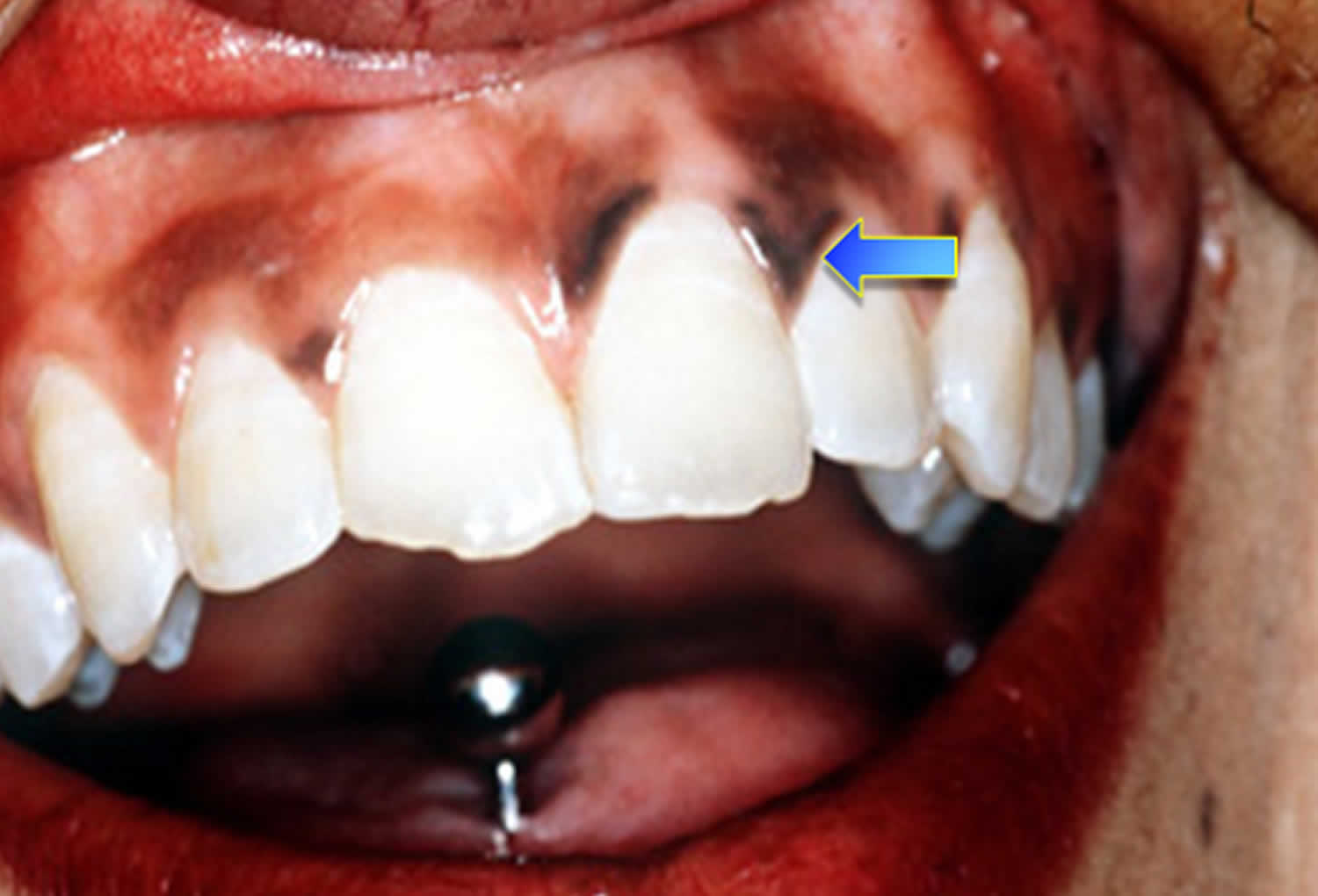
Figure 3. Hyperpigmentation in the mouth
Acute adrenal failure (Addisonian crisis)
Sudden, severe worsening of adrenal insufficiency symptoms is called adrenal crisis. If the person has Addison’s disease, this worsening can also be called an Addisonian crisis. In most cases, symptoms of adrenal insufficiency become serious enough that people seek medical treatment before an adrenal crisis occurs. However, sometimes symptoms appear for the first time during an adrenal crisis.
In acute adrenal failure (Addisonian crisis), the signs and symptoms may also include:
- Sudden, severe pain in your lower back, abdomen or legs
- Severe vomiting and diarrhea, leading to dehydration
- Low blood pressure
- Loss of consciousness
- High potassium (hyperkalemia) and low sodium (hyponatremia)
If not treated, an adrenal crisis can cause death.
Get Treatment for Adrenal Crisis Right Away – If not treated, an adrenal crisis can cause death.
Addisonian crisis requires immediate medical care. Treatment typically includes intravenous injections of:
- Hydrocortisone
- Saline solution
- Sugar (dextrose)
How is adrenal crisis treated ?
Adrenal crisis is treated with adrenal hormones. People with adrenal crisis need immediate treatment. Any delay can cause death. When people with adrenal crisis are vomiting or unconscious and cannot take their medication, the hormones can be given as an injection.
A person with adrenal insufficiency should carry a corticosteroid injection at all times and make sure that others know how and when to administer the injection, in case the person becomes unconscious.
The dose of corticosteroid needed may vary with a person’s age or size. For example, a child younger than 2 years of age can receive 25 milligrams (mg), a child between 2 and 8 years of age can receive 50 mg, and a child older than 8 years should receive the adult dose of 100 mg.
How can a person prevent adrenal crisis ?
The following steps can help a person prevent adrenal crisis:
- Ask a health care provider about possibly having a shortage of adrenal hormones, if always feeling tired, weak, or losing weight.
- Learn how to increase the dose of corticosteroid for adrenal insufficiency when ill. Ask a health care provider for written instructions for sick days. First discuss the decision to increase the dose with the health care provider when ill.
- When very ill, especially if vomiting and not able to take pills, seek emergency medical care immediately.
How can someone with adrenal insufficiency prepare in case of an emergency ?
People with adrenal insufficiency should always carry identification stating their condition, “adrenal insufficiency,” in case of an emergency. A card or medical alert tag should notify emergency health care providers of the need to inject corticosteroids if the person is found severely injured or unable to answer questions.
The card or tag should also include the name and telephone number of the person’s health care provider and the name and telephone number of a friend or family member to be notified. People with adrenal insufficiency should always carry a needle, a syringe, and an injectable form of corticosteroids for emergencies.
What problems can occur with adrenal insufficiency ?
Problems can occur in people with adrenal insufficiency who are undergoing surgery, suffer a severe injury, have an illness, or are pregnant. These conditions place additional stress on the body, and people with adrenal insufficiency may need additional treatment to respond and recover.
Surgery
People with adrenal insufficiency who need any type of surgery requiring general anesthesia must be treated with IV corticosteroids and saline. IV treatment begins before surgery and continues until the patient is fully awake after surgery and is able to take medication by mouth. The “stress” dosage is adjusted as the patient recovers until the regular, presurgery dose is reached.
In addition, people who are not currently taking corticosteroids, yet have taken long-term corticosteroids in the past year, should tell their health care provider before surgery. These people may have sufficient ACTH for normal events; however, they may need IV treatment for the stress of surgery.
Severe Injury
Patients who suffer severe injury may need a higher, “stress” dosage of corticosteroids immediately following the injury and during recovery. Often, these stress doses must be given intravenously. Once the patient recovers from the injury, dosing is returned to regular, pre-injury levels.
Illness
During an illness, a person taking corticosteroids orally may take an adjusted dose to mimic the normal response of the adrenal glands to this stress on the body. Significant fever or injury may require a triple dose. Once the person recovers from the illness, dosing is then returned to regular, pre-illness levels. People with adrenal insufficiency should know how to increase medication during such periods of stress, as advised by their health care provider. Immediate medical attention is needed if severe infections, vomiting, or diarrhea occur. These conditions can lead to an adrenal crisis.
Pregnancy
Women with adrenal insufficiency who become pregnant are treated with the same hormone therapy taken prior to pregnancy. However, if nausea and vomiting in early pregnancy interfere with taking medication orally, injections of corticosteroids may be necessary. During delivery, treatment is similar to that of people needing surgery. Following delivery, the dose is gradually lessened, and the regular dose is reached about 10 days after childbirth.
Addison’s disease diagnosis
In its early stages, adrenal insufficiency can be difficult to diagnose. A health care provider may suspect it after reviewing a person’s medical history and symptoms.
A diagnosis of adrenal insufficiency is confirmed through hormonal blood and urine tests. A health care provider uses these tests first to determine whether cortisol levels are too low and then to establish the cause. Imaging studies of the adrenal and pituitary glands can be useful in helping to establish the cause.
Your doctor will talk to you first about your medical history and your signs and symptoms. If your doctor thinks that you may have Addison’s disease, you may undergo some of the following tests:
- Blood test. Measuring your blood levels of sodium, potassium, cortisol and ACTH gives your doctor an initial indication of whether adrenal insufficiency may be causing your signs and symptoms. A blood test can also measure antibodies associated with autoimmune Addison’s disease.
- Thyroid function test. Your thyroid gland may also be tested to see if it’s working properly. Your thyroid gland is found in your neck. It produces hormones that control your body’s growth and metabolism. People with Addison’s disease often have an underactive thyroid gland (hypothyroidism), where the thyroid gland does not produce enough hormones. By testing the levels of certain hormones in your blood, your endocrinologist (a specialist in hormone conditions) can determine whether you have hypothyroidism.
- ACTH stimulation test. The ACTH stimulation test is the most commonly used test for diagnosing adrenal insufficiency. In this test, the patient is given an intravenous (IV) injection of synthetic ACTH, and samples of blood, urine, or both are taken before and after the injection. The cortisol levels in the blood and urine samples are measured in a lab. The normal response after an ACTH injection is a rise in blood and urine cortisol levels. People with Addison’s disease or longstanding secondary adrenal insufficiency have little or no increase in cortisol levels.Both low- and high-dose ACTH stimulation tests may be used depending on the suspected cause of adrenal insufficiency. For example, if secondary adrenal insufficiency is mild or has only recently occurred, the adrenal glands may still respond to ACTH because they have not yet shut down their own production of hormone. Some studies have suggested a low dose—1 microgram (mcg)—may be more effective in detecting secondary adrenal insufficiency because the low dose is still enough to raise cortisol levels in healthy people, yet not in people with mild or recent secondary adrenal insufficiency. However, recent research has shown that a significant proportion of healthy children and adults can fail the low-dose test, which may lead to unnecessary treatment. Therefore, some health care providers favor using a 250 mcg ACTH test for more accurate results.
- CRH stimulation test. When the response to the ACTH test is abnormal, a CRH (Corticotropin-releasing hormone) stimulation test can help determine the cause of adrenal insufficiency. In this test, the patient is given an IV injection of synthetic CRH (Corticotropin-releasing hormone) and blood is taken before and 30, 60, 90, and 120 minutes after the injection. The cortisol levels in the blood samples are measured in a lab. People with Addison’s disease respond by producing high levels of ACTH, yet no cortisol. People with secondary adrenal insufficiency do not produce ACTH or have a delayed response. CRH will not stimulate ACTH secretion if the pituitary is damaged, so no ACTH response points to the pituitary as the cause. A delayed ACTH response points to the hypothalamus as the cause.
- Insulin-induced hypoglycemia test. Occasionally, doctors suggest this test if pituitary disease is a possible cause of adrenal insufficiency (secondary adrenal insufficiency). The test involves checking your blood sugar (blood glucose) and cortisol levels at various intervals after an injection of insulin. In healthy people, glucose levels fall and cortisol levels increase.
- Ultrasound of the abdomen. Ultrasound uses a device, called a transducer, that bounces safe, painless sound waves off organs to create an image of their structure. A specially trained technician performs the procedure in a health care provider’s office, an outpatient center, or a hospital, and a radiologist—a doctor who specializes in medical imaging—interprets the images; a patient does not need anesthesia. The images can show abnormalities in the adrenal glands, such as enlargement or small size, nodules, or signs of calcium deposits, which may indicate bleeding.
- Tuberculin skin test. A tuberculin skin test measures how a patient’s immune system reacts to the bacteria that cause tuberculosis. A small needle is used to put some testing material, called tuberculin, under the skin. A nurse or lab technician performs the test in a health care provider’s office; a patient does not need anesthesia. In 2 to 3 days, the patient returns to the health care provider, who will check to see if the patient had a reaction to the test. The test can show if adrenal insufficiency could be related to tuberculosis.To test whether a person has TB infection, which is when TB bacteria live in the body without making the person sick, a special TB blood test is used. To test whether a person has TB disease, which is when TB bacteria are actively attacking a person’s lungs and making the person sick, other tests such as a chest x-ray and a sample of sputum—phlegm that is coughed up from deep in the lungs—may be needed.
Antibody blood tests. A blood test involves drawing blood at a health care provider’s office or a commercial facility and sending the sample to a lab for analysis. The blood test can detect antibodies—proteins made by the immune system to protect the body from foreign substances—associated with autoimmune Addison’s disease.
After secondary adrenal insufficiency is diagnosed, health care providers may use the following tests to obtain a detailed view of the pituitary gland and assess how it is functioning:
- Imaging tests. Your doctor may have you undergo a computerized tomography (CT) scan of your abdomen to check the size of your adrenal glands and look for other abnormalities that may give insight to the cause of the adrenal insufficiency. Your doctor may also suggest an MRI scan of your pituitary gland if testing indicates you might have secondary adrenal insufficiency.
Addison’s disease treatment
All treatment for Addison’s disease involves hormone replacement therapy, you’ll need to take daily prescription hormones to replace the lost hormones to correct the levels of steroid hormones your body isn’t producing.
This can include hydrocortisone, prednisone, or cortisone acetate. If your body is not making enough of the hormone aldosterone, your doctor may prescribe fludrocortisone. These medicines are taken every day by mouth (in pill form). This should help you to live an active life, although many people find they still need to manage their fatigue.
Your doctor may also recommend that you take a medicine called dehydroepiandrosterone. Some women who have Addison’s disease find that taking this medicine improves their mood and sex drive.
The dose of each medication is adjusted to meet the needs of the patient.
In some cases, the underlying causes of Addison’s disease can be treated. For example, tuberculosis (TB) is treated with a course of antibiotics over a period of at least 6 months.
However, most cases are caused by a problem with the immune system that cannot be cured.
If you are experiencing an Addisonian crisis, you need immediate medical care. The treatment typically consists of intravenous (IV) injections of hydrocortisone, saline (salt water), and dextrose (sugar). These injections help restore blood pressure, blood sugar, and potassium levels to normal.
During adrenal crisis, low blood pressure, low blood glucose, low blood sodium, and high blood levels of potassium can be life threatening. Standard therapy involves immediate IV injections of corticosteroids and large volumes of IV saline solution with dextrose, a type of sugar. This treatment usually brings rapid improvement. When the patient can take liquids and medications by mouth, the amount of corticosteroids is decreased until a dose that maintains normal hormone levels is reached. If aldosterone is deficient, the person will need to regularly take oral doses of fludrocortisone acetate.
Researchers have found that using replacement therapy for DHEA in adolescent girls who have secondary adrenal insufficiency and low levels of DHEA can improve pubic hair development and psychological stress. Further studies are needed before routine supplementation recommendations can be made.
Some options for treatment include:
- Oral corticosteroids. Hydrocortisone (Cortef), prednisone or cortisone acetate may be used to replace cortisol. Your doctor may prescribe fludrocortisone to replace aldosterone.
- Corticosteroid injections. If you’re ill with vomiting and can’t retain oral medications, injections may be needed.
Cortisol is replaced with a corticosteroid, such as hydrocortisone, prednisone, or dexamethasone, taken orally one to three times each day, depending on which medication is chosen.
If aldosterone is also deficient, it is replaced with oral doses of a mineralocorticoid hormone, called fludrocortisone acetate (Florinef), taken once or twice daily. People with secondary adrenal insufficiency normally maintain aldosterone production, so they do not require aldosterone replacement therapy.
An ample amount of sodium is recommended, especially during heavy exercise, when the weather is hot or if you have gastrointestinal upsets, such as diarrhea. Your doctor will also suggest a temporary increase in your dosage if you’re facing a stressful situation, such as an operation, an infection or a minor illness.
Adjusting your medication
At certain times, your medication may need to be adjusted to account for any additional strain on your body. For example, you may need to increase the dosage of your medication if you experience any of the following:
- an illness or infection – particularly if you have a high temperature of 38C or above
- an accident, such as a car crash
- an operation, dental or medical procedure – such as a tooth filling or endoscopy
- strenuous exercise that’s not usually part of your daily life
This will help your body cope with the additional stress. Your endocrinologist will monitor your dosage and advise about any changes.
Over time, as you get used to the condition and learn what can trigger your symptoms, you may learn how to adjust your medication yourself. However, always consult your doctor or specialist if you’re unsure.
Emergency treatment
You and a partner or family member may be trained to administer an injection of hydrocortisone in an emergency.
This could be necessary if you go into shock after an injury, or if you experience vomiting or diarrhea and are unable to keep down oral medication. This may occur if you’re pregnant and have morning sickness. Your endocrinologist will discuss with you when an injection might be necessary.
If you need to administer emergency hydrocortisone, always call your doctor immediately afterwards. Check what out-of-hours services are available in your local area, in case the emergency is outside normal working hours.
You can also register yourself with your local ambulance service, so they have a record of your requirement for a steroid injection or tablets, if you need their assistance.
Treating adrenal crisis
Adrenal crisis, or Addisonian crisis, needs urgent medical attention. Dial 999 for an ambulance if you or someone you know are experiencing adrenal crisis.
Signs of an adrenal crisis include:
- severe dehydration
- pale, cold, clammy skin
- sweating
- rapid, shallow breathing
- dizziness
- severe vomiting and diarrhea
- severe muscle weakness
- headache
- severe drowsiness or loss of consciousness
In hospital, you’ll be given lots of fluid through a vein in your arm to rehydrate you. This will contain a mixture of salts and sugars (sodium, glucose and dextrose) to replace what your body is lacking. You’ll also be injected with hydrocortisone to replace the missing cortisol hormone.
Any underlying causes of the adrenal crisis, such as an infection, will also be treated.
Addison’s disease diet
Some people with Addison’s disease who are aldosterone deficient can benefit from following a diet rich in sodium. A health care provider or a dietitian can give specific recommendations on appropriate sodium sources and daily sodium guidelines if necessary.
Corticosteroid treatment is linked to an increased risk of osteoporosis—a condition in which the bones become less dense and more likely to fracture. People who take corticosteroids should protect their bone health by consuming enough dietary calcium and vitamin D. A health care provider or a dietitian can give specific recommendations on appropriate daily calcium intake based upon age and suggest the best types of calcium supplements, if necessary.
Coping and support
These steps may help you cope better with a medical emergency if you have Addison’s disease:
- Carry a medical alert card and bracelet at all times. In the event you’re incapacitated, emergency medical personnel know what kind of care you need.
- Keep extra medication handy. Because missing even one day of therapy may be dangerous, it’s a good idea to keep a small supply of medication at work, at a vacation home and in your travel bag, in the event you forget to take your pills. Also, have your doctor prescribe a needle, syringe and injectable form of corticosteroids to have with you in case of an emergency.
- Stay in contact with your doctor. Keep an ongoing relationship with your doctor to make sure that the doses of replacement hormones are adequate, but not excessive. If you’re having persistent problems with your medications, you may need adjustments in the doses or timing of the medications.
Living with Addison’s disease
Many people with Addison’s disease find that taking their medication enables them to continue with their normal diet and exercise routines.
However, bouts of fatigue are also common, and it can take some time to learn how to manage these periods of low energy.
Some people find that needing to take regular doses of medication is restrictive and affects their daily life or emotional health. Missing a dose of medication, or taking it late, can also lead to exhaustion or insomnia.
Some people can develop associated health conditions, such as diabetes or an underactive thyroid, which require extra treatment and management.
You’ll usually need to have appointments with an endocrinologist every 6 to 12 months so they can review your progress and adjust your medication dose, if necessary. Your doctor can provide support and repeat prescriptions in between these visits.
Failing to take your medication could lead to a serious condition called an adrenal crisis, so you must:
- remember to collect your repeat prescriptions
- keep spare medication as necessary – for example, in the car or at work, and always carry some spare medication with you
- take your medication every day at the right time
- pack extra medication if you’re going away – usually double what you would normally need, plus your injection kit
- carry your medication in your hand luggage if you are traveling by plane, with a note from your doctor explaining why it is necessary
You could also inform close friends or colleagues of your condition. Tell them about the signs of adrenal crisis and what they should do if you experience one.
Medical alert bracelets
It’s also a good idea to wear a medical alert bracelet or necklace that informs people you have Addison’s disease.
After a serious accident, such as a car crash, a healthy person produces more cortisol. This helps you cope with the stressful situation and additional strain on your body that results from serious injury. As your body cannot produce cortisol, you’ll need a hydrocortisone injection to replace it and prevent an adrenal crisis.
Wearing a medical alert bracelet will inform any medical staff treating you about your condition and what medication you require.
Medical alert bracelets or necklaces are pieces of jewellery engraved with your medical condition and an emergency contact number. They are available from a number of retailers.
- Adrenal Insufficiency. https://www.hormone.org/diseases-and-conditions/adrenal/adrenal-insufficiency[↩]
- Betterle C, Morlin L. Autoimmune Addison’s disease. In: Ghizzoni L, Cappa M, Chrousos G, Loche S, Maghnie M, eds. Pediatric Adrenal Diseases. Endocrine Development. Vol. 20. Padova, Italy: Karger Publishers; 2011: 161–172.[↩][↩]
- The conquest of Addison’s disease. Hiatt JR, Hiatt N. Am J Surg. 1997 Sep; 174(3):280-3.[↩]
- Stewart PM, Krone NP. The adrenal cortex. In: Kronenburg HM, Melmed S, Polonsky KS, Reed Larson P, editors. Williams Textbook of Endocrinology. 12th ed. Philadelphia PA: Saunders Elsevier; 2011. pp. 515–20. Ch. 15.[↩]
- Ross AP, Ben-Zacharia A, Harris C, Smrtka J. Multiple Sclerosis, Relapses, and the Mechanism of Action of Adrenocorticotropic Hormone. Frontiers in Neurology. 2013;4:21. doi:10.3389/fneur.2013.00021. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3591751/[↩]
- Neary N, Nieman L. Adrenal insufficiency: etiology, diagnosis and treatment. Current Opinion in Endocrinology, Diabetes and Obesity. 2010;(3):217–223.[↩][↩]