Contents
What is autonomic neuropathy
Autonomic neuropathy occurs when the nerves that control involuntary bodily functions are damaged. This may affect blood pressure, temperature control, digestion, bladder function and even sexual function.
The nerve damage interferes with the messages sent between the brain and other organs and areas of the autonomic nervous system, such as the heart, blood vessels and sweat glands.
While diabetes is generally the most common cause of autonomic neuropathy, other health conditions — even an infection — may be to blame. Some medications also may cause nerve damage. Symptoms and treatment will vary based on which nerves are damaged.
The Autonomic Nervous System
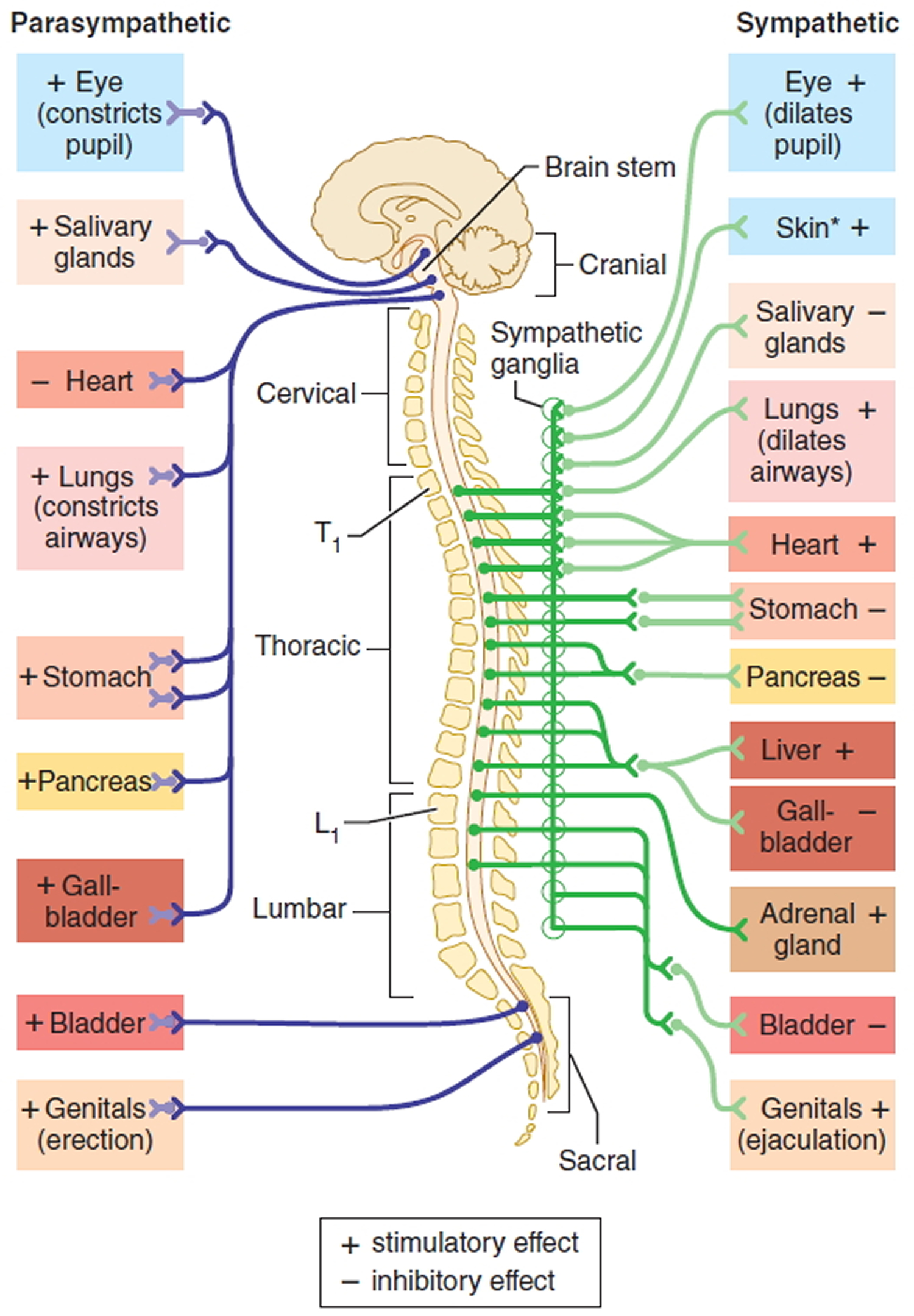
The autonomic nervous system is the part of the nervous system that regulates cardiac muscle, smooth muscle, and glands. These tissues are often referred to as visceral effectors because they are usually associated with the viscera (internal organs) of the body. The term autonomic is derived from the Latin words auto- = self and -nomic = law because the autonomic nervous system was once thought to be self-governing.
The autonomic nervous system consists of autonomic motor neurons that regulate visceral activities by either increasing (exciting) or decreasing (inhibiting) ongoing activities in their eff ector tissues (cardiac muscle, smooth muscle, and glands). Changes in the diameter of the pupils, dilation and constriction of blood vessels, and adjustment of the rate and force of the heartbeat are examples of autonomic motor responses. Unlike skeletal muscle, tissues innervated by the autonomic nervous system oft en function to some extent even if their nerve supply is damaged. The heart continues to beat when it is removed for transplantation into another person, smooth muscle in the lining of the gastrointestinal tract contracts rhythmically on its own, and glands produce some secretions in the absence of autonomic nervous system control.
The autonomic nervous system consists of two main division (branches):
- the sympathetic nervous system and
- the parasympathetic nervous system.
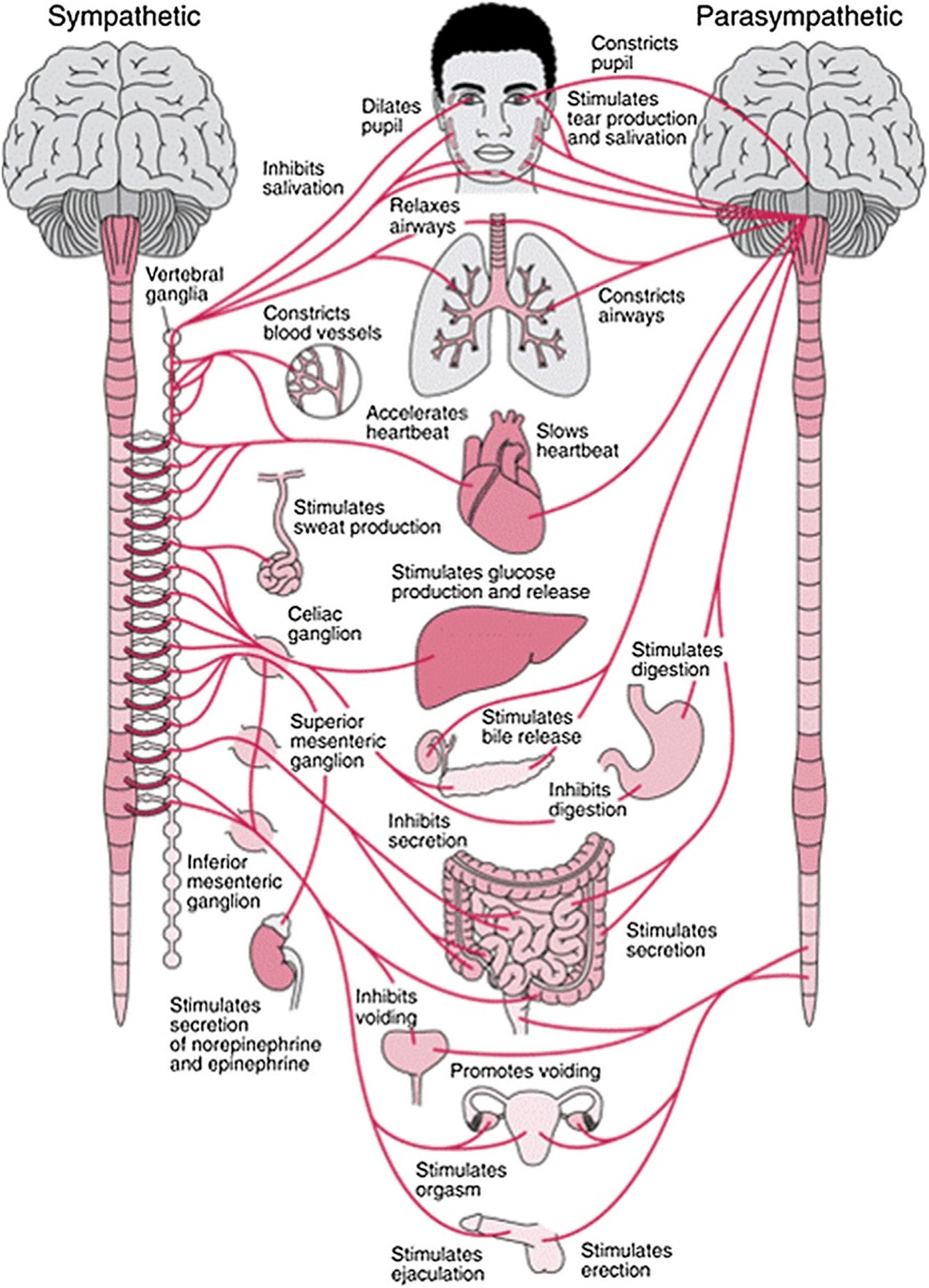
Most organs receive nerves from both of these divisions, an arrangement known as dual innervation. In general, one division stimulates the organ to increase its activity (excitation), and the other division decreases the organ’s activity (inhibition). For example, neurons of the sympathetic nervous system increase heart rate, and neurons of the parasympathetic nervous system slow it down. The sympathetic nervous system promotes the fight-or-flight response, which prepares the body for emergency situations. By contrast, the parasympathetic nervous system enhances rest-and-digest activities, which conserve and restore body energy during times of rest or digesting a meal. Although both the sympathetic and parasympathetic divisions are concerned with maintaining health, they do so in dramatically different ways.
The autonomic nervous system is also comprised of a third division known as the enteric nervous system. The enteric nervous system consists of millions of neurons in plexuses that extend most of the length of the gastrointestinal tract. Its operation is involuntary. Although the neurons of the enteric nervous system can function autonomously, they can also be regulated by the other divisions of the autonomic nervous system. The enteric nervous system contains sensory neurons, interneurons, and motor neurons. Enteric sensory neurons monitor chemical changes within the gastrointestinal tract as well as the stretching of its walls. Enteric interneurons integrate information from the sensory neurons and provide input to motor neurons. Enteric motor neurons govern contraction of gastrointestinal tract smooth muscle and secretion of gastrointestinal tract glands.
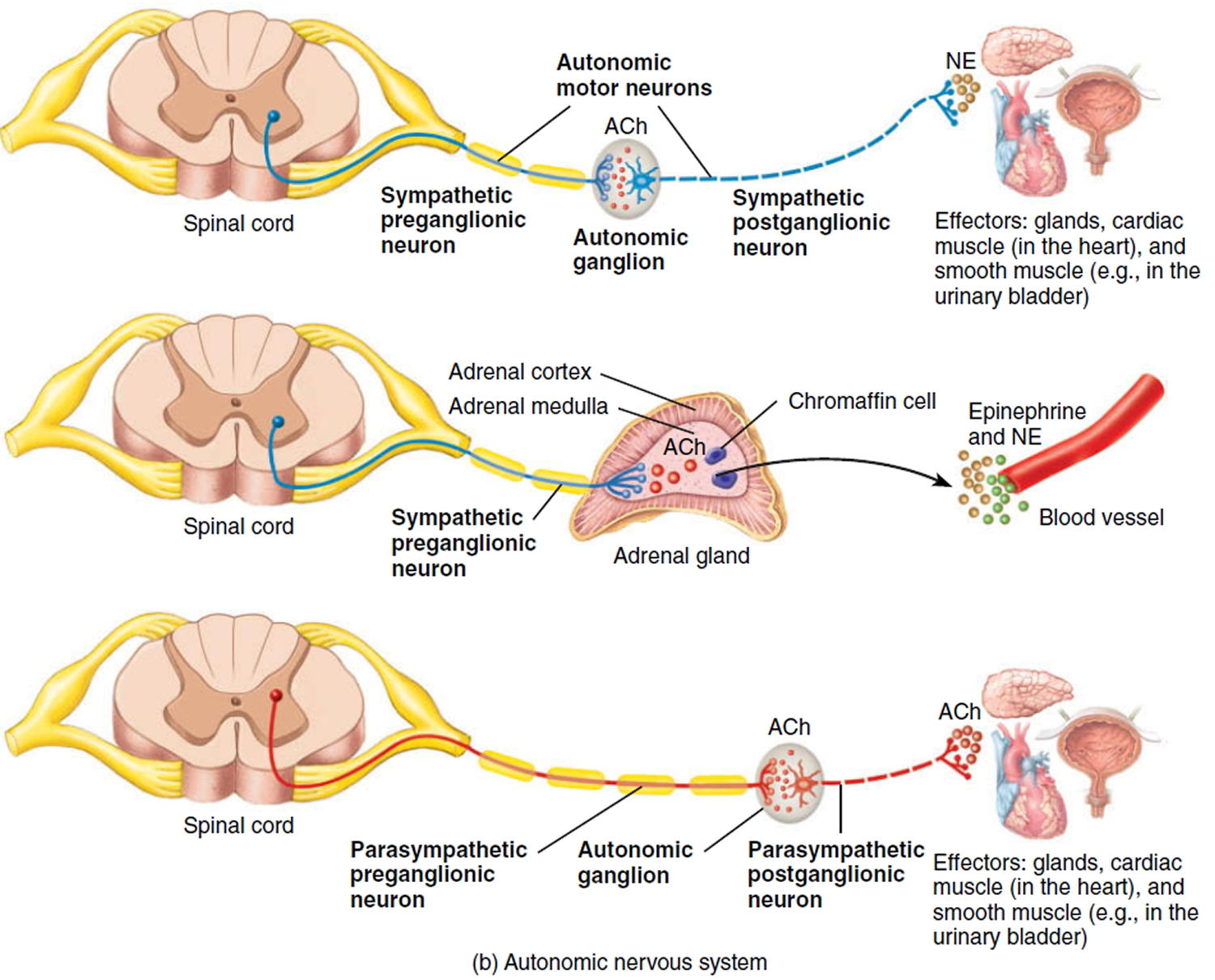
Most autonomic motor pathways consist of two motor neurons in series; that is, one following the other. The first neuron (preganglionic neuron) has its cell body in the central nervous system; its myelinated axon extends from the central nervous system to an autonomic ganglion. The cell body of the second neuron (postganglionic neuron) is also in that same autonomic ganglion; its unmyelinated axon extends directly from the ganglion to the eff ector (smooth muscle, cardiac muscle, or a gland). Alternatively, in some autonomic pathways, the first motor neuron extends to specialized cells called chromaffin cells in the adrenal medullae (inner portion of the adrenal glands) rather than an autonomic ganglion. Chromaffin cells secrete the neurotransmitters epinephrine and norepinephrine (NE). Autonomic motor neurons release either Acetylcholine (ACh) or norepinephrine (NE).
Figure 1. Autonomic nervous system
Figure 2. Autonomic nervous system
 Note: Acetylcholine (ACh) and Norepinephrine (NE)
Note: Acetylcholine (ACh) and Norepinephrine (NE)
The autonomic nervous system usually operates without conscious control. For example, you probably cannot voluntarily slow down your heart rate; instead, your heart rate is subconsciously regulated. For this reason, some autonomic responses are the basis for polygraph (“lie detector”) tests. However, practitioners of yoga or other techniques of meditation may learn how to regulate at least some of their autonomic activities through long practice. Biofeedback, in which monitoring devices display information about a body function such as heart rate or blood pressure, enhances the ability to learn such conscious control.
The autonomic nervous system can also receive sensory input from sensory neurons associated with interoceptors, sensory receptors located in blood vessels, visceral organs, muscles, and the nervous system that monitor conditions in the internal environment. Examples of interoceptors are chemoreceptors that monitor blood CO2 level and mechanoreceptors that detect the degree of stretch in the walls of organs or blood vessels. Unlike those triggered by a flower’s perfume, a beautiful painting, or a delicious meal, these sensory signals are not consciously perceived most of the time, although intense activation of interoceptors may produce conscious sensations. Two examples of perceived visceral sensations are pain sensations from damaged viscera and angina pectoris (chest pain) from inadequate blood flow to the heart. Signals from the somatic senses and special senses, acting via the limbic system, also influence responses of autonomic motor neurons. Seeing a bike about to hit you, hearing squealing brakes of a nearby car, or being grabbed by an attacker would all increase the rate and force of your heartbeat.
Cardiac autonomic neuropathy
Cardiac autonomic neuropathy is defined as the impairment of cardiovascular autonomic control in patients with established diabetes mellitus following the exclusion of other causes 1. Cardiac autonomic neuropathy, especially at the early stages, can be sub-clinical and thus as the disease progresses, it becomes clinically evident.
The prevalence of cardiac autonomic neuropathy varies between 1%-90% in patients with type 1 diabetes mellitus and 20%-73% in patients with type1 diabetes mellitus 2. This huge variation in cardiac autonomic neuropathy prevalence is due to the inconsistency in the criteria used to diagnose cardiac autonomic neuropathy and significant differences in the study populations, particularly in relation to cardiac autonomic neuropathy risk factors (such as age, gender and diabetes mellitus duration amongst others).
Longitudinal studies have shown a 5-year mortality rate of 16%–50% in both type 1 diabetes mellitus and type 2 diabetes mellitus once cardiac autonomic neuropathy had been diagnosed, with most of these attributed to sudden cardiac death 3. This large range was due to differences in study population and methodologies used to diagnose cardiac autonomic neuropathy. These include changes in the perception of myocardial ischemia, altered hemodynamic response to stress on the cardiovascular system (eg, surgery, infection, or anesthesia), QT-interval dispersion leading to cardiac arrhythmias, variations in autonomic cardiac innervation balance and focal regions of sympathetic denervation, and reinnervation of the myocardium 4.
A meta-analysis of 15 studies including studies with a baseline assessment of heart rate variability using one or more tests and follow-up mortality data showed that the pooled estimated relative mortality risk was 3.45 when cardiac autonomic neuropathy was defined as the presence of two or more abnormalities in cardiac autonomic function. The ACCORD trial also showed cardiac autonomic neuropathy to be an independent predictor of all-cause mortality and cardiovascular disease mortality after a mean follow-up of 3.5 years, while the EURODIAB IDDM 5 complication study demonstrated that cardiac autonomic neuropathy had the strongest association with mortality when compared to other risk factors.
Cardiac autonomic neuropathy has been detected at time of diagnosis of diabetes in patients with either type 1 diabetes mellitus or type 2 diabetes mellitus irrespective of age, suggesting that cardiac autonomic neuropathy presentation is not limited by age or type of diabetes and can occur before diabetes mellitus is evident clinically 6. However, the duration of diabetes is an independent factor for developing cardiac autonomic neuropathy irrespective to diabetes type 7. Cardiac autonomic neuropathy is detected in about 7% of both type 1 diabetes mellitus and type 2 diabetes mellitus at the time of initial diagnosis 8 and it is estimated that the risk for developing cardiac autonomic neuropathy increases annually by approximately 6% and 2% in patients with type 1 diabetes mellitus and type 2 diabetes mellitus respectively 9.
Poor glycaemic control is a major risk factor for cardiac autonomic neuropathy progression 10. In the Diabetes Control and Complications Trial 11, intensive glycemic control resulted in a 50% decrease in cardiac autonomic neuropathy incidence over the 6.5 years follow-up period. This protective effect persisted 14 years after the end of the study despite the disappearance of HbA1c differences that were achieved between the groups during the randomized phase of trial 9. Similarly, cardiac autonomic neuropathy has been shown to be associated with conventional cardiovascular disease risk factors, such as hypertension, smoking, hyperlipidaemia and obesity 12. In the Steno-2 trial 13 of patients with type 2 diabetes mellitus and microalbuminuria, intensive pharmacological intervention targeting hypertension, hyperlipidemia and microalbuminuria combined with behavioral treatment (exercise, diet and smoking cessation) reduced the risk of autonomic neuropathy over the course of a 7.8 years follow-up. After a mean of 5.5 years following the end of the study, the same protective effect against the development of autonomic neuropathy persisted. There was also reduction in the risk for developing cardiovascular disease and overall mortality 14 in this study.
Moreover, in a large cohort of more than 1000 patients with type 2 diabetes mellitus the incidence of cardiac autonomic neuropathy over a 7.5 years follow-up correlated with age and microvascular disease 15. Diabetic nephropathy (including microalbuminura), diabetic retinopathy and diabetic polyneuropathy have been widely identified as clinical predictors of cardiac autonomic neuropathy 16, which is not surprising as diabetic microvascular complications share common mechanisms and risk factors. The impact of gender on cardiac autonomic neuropathy is controversial. In a multi-center, cross sectional study of 3250 patients with diabetes mellitus, cardiac autonomic neuropathy prevalence was no different between men and women (35% male vs 37% female) 17. However, in the action to control cardiovascular risk in diabetes trial including more than 8000 patients with type 2 diabetes mellitus cardiac autonomic neuropathy was more prevalent in women (2.6% in men vs 4.7% in women for moderate severity cardiac autonomic neuropathy and 1.4% in men vs 2.2% in women for severe cardiac autonomic neuropathy, for all three definitions of cardiac autonomic neuropathy in the study) 18.
Ethnicity has also been postulated to be a risk factor for cardiac autonomic neuropathy as South Asians seem to have lower rates of peripheral neuropathy than White Europeans with diabetes mellitus 19. More specifically, the prevalence of small fiber neuropathy was significantly lower in Indian Asians than in Europeans (32% vs 43% respectively) and mean nerve conduction velocity Z scores (measuring large fibre neuropathy) were superior in Asians compared to Europeans (mean ± SD 0.07 ± -0.62 vs -0.11 ± 0.60,). However, using heart rate variability (HRV) spectral analysis as well as frequency and time domain analysis showed no difference in cardiac autonomic neuropathy prevalence between South Asians and white Europeans.
Cardiac autonomic neuropathy pathogenesis
The exact pathogenesis of cardiac autonomic neuropathy is complex, remains unclear and still under much debate. Most of the proposed mechanisms of neuronal injury are based on models of somatic rather than autonomic neuropathy. Although many of these mechanisms might be shared between autonomic and somatic neuropathies, differences do exist as shown by the Steno-2 trial 13 in which the multi-factorial intervention (including intensive metabolic control and lifestyle changes) slowed down the progression of autonomic but not somatic neuropathy.
Hyperglycemia
Hyperglycemia can activate multiple pathways involved in the pathogenesis of cardiac autonomic neuropathy. Of these pathways, the majority are related to the metabolic and/or oxidative state of neuronal cells. Although these alone can cause excessive damage, when combined they result in mitochondrial dysfunction and formation of reactive oxygen species 20. Oxidative stress can induce DNA damage, leading to activation of PARP and inhibition of GAPDH 21. This in turn can activate multiple pathways, including the polyol pathway, the hexosamine pathway, as well as activation of PKC and increased production of advanced glycation end products, which in turn correlate with the severity of autonomic nerve abnormalities in patients with diabetes mellitus 22.
Recent studies have supported the theory that hyperglycemia also impairs neuronal regeneration, and suggested that an increase in plasma superoxide generation may be able to predict decline in cardiac nerve function, and in particular may be the main determinant in early cardiac autonomic dysfunction 23. In relation to cardiac autonomic neuropathy, increased reactive oxygen species is thought to depress autonomic ganglion synaptic transmission, contributing to increased risk of fatal cardiac arrhythmias, as well as to sudden death after myocardial infarction due to posttranslational protein modifications 24.
Autoimmunity
Cardiac autonomic neuropathy might occur as a result of autoimmune autonomic ganglionopathy. This is where autonomic failure occurs in the presence of antibodies to the nicotinic acetylcholine receptor of autonomic ganglia, leading to severe autonomic manifestations, e.g., orthostatic intolerance, syncope, constipation, gastroparesis, urinary retention, dry mouth, dry eyes, anhidrosis, and cognitive impairment 25. However, the role of autoimmunity in patients with diabetes and cardiac autonomic neuropathy remains controversial 26. A recent small cross-sectional study did show that the presence of IgG antibodies was associated with autonomic neuropathy 27. However, there remains no clear consensus on the role of autoimmunity in cardiac autonomic neuropathy progression, and most studies showing a positive association have been in patients with type 1 diabetes mellitus. Therefore, whether autoimmunity contributes to cardiac autonomic neuropathy in patients with type 2diabetes mellitus is still not clear 28.
Genetics
Several genes have been linked to the development and progression of diabetic polyneuropathy and cardiac autonomic neuropathy, among which are TCF7L2, APOE, and ACE 29. Ciccacci et al 30 showed an association between miRI146a and miR27a single-nucleotide polymorphisms and cardiac autonomic neuropathy susceptibility. However, a twin study by Osztovits et al 31 found that genetic factors did not have a substantial influence on cardiovascular autonomic function, and the authors went on to say that environmental factors played a bigger role.
Obstructive sleep apnea
Obstructive sleep apnea is known to be common in patients with diabetes mellitus and believed to be associated with diabetic peripheral neuropathy in patients with type 2 diabetes mellitus 32. While obesity is a major risk factor for obstructive sleep apnea, Janovsky et al 33 showed that obstructive sleep apnea was common in lean patients with type 1 diabetes mellitus and that obstructive sleep apnea can be associated with cardiac autonomic neuropathy. Unpublished data showed that obstructive sleep apnea was associated with weaker sympathetic and parasympathetic tone in patients with type 2 diabetes mellitus and that patients who were compliant with obstructive sleep apnea treatment (continuous positive airway pressure) had improvements in several autonomic parameters over the follow-up period 34.
The pathophysiology and relationship between obstructive sleep apnea and cardiac autonomic neuropathy might differ between patients with type 1 diabetes mellitus and type 2 diabetes mellitus. The intermittent hypoxia that occurs with obstructive sleep apnea could result in increased reactive oxygen species and impaired microvascular function, leading to cardiac autonomic neuropathy 35. On the other hand, the relationship may be bidirectional, with cardiac autonomic neuropathy causing changes in respiratory drive and upper-airway tone, increasing the risk of obstructive sleep apnea.
Inflammation
Inflammation plays an important role in the pathogenesis of diabetes and its related micro- and macrovascular complications. cardiac autonomic neuropathy has been associated with increased inflammatory markers such as CRP, IL6, and TNFα, and adipose-tissue inflammation 36. However, the direction of the relationship between inflammation and cardiac autonomic neuropathy is not clear and could be bidirectional 36. Vinik et al 36 suggested that the inflammatory response is controlled by a neural circuit in which the afferent arc consists of nerves that sense injury and transmits the information via the vagus nerve to the brain stem, which in turn activates the cholinergic anti-inflammatory pathways modulating the response. Increased activity in the vagus nerve to the spleen reduces the innate immune system’s response to damage-associated molecular patterns and suppresses inflammation. Therefore, changes in vagal activity due to autonomic neuropathy might have an impact on inflammatory responses.
Cardiac autonomic neuropathy signs and symptoms
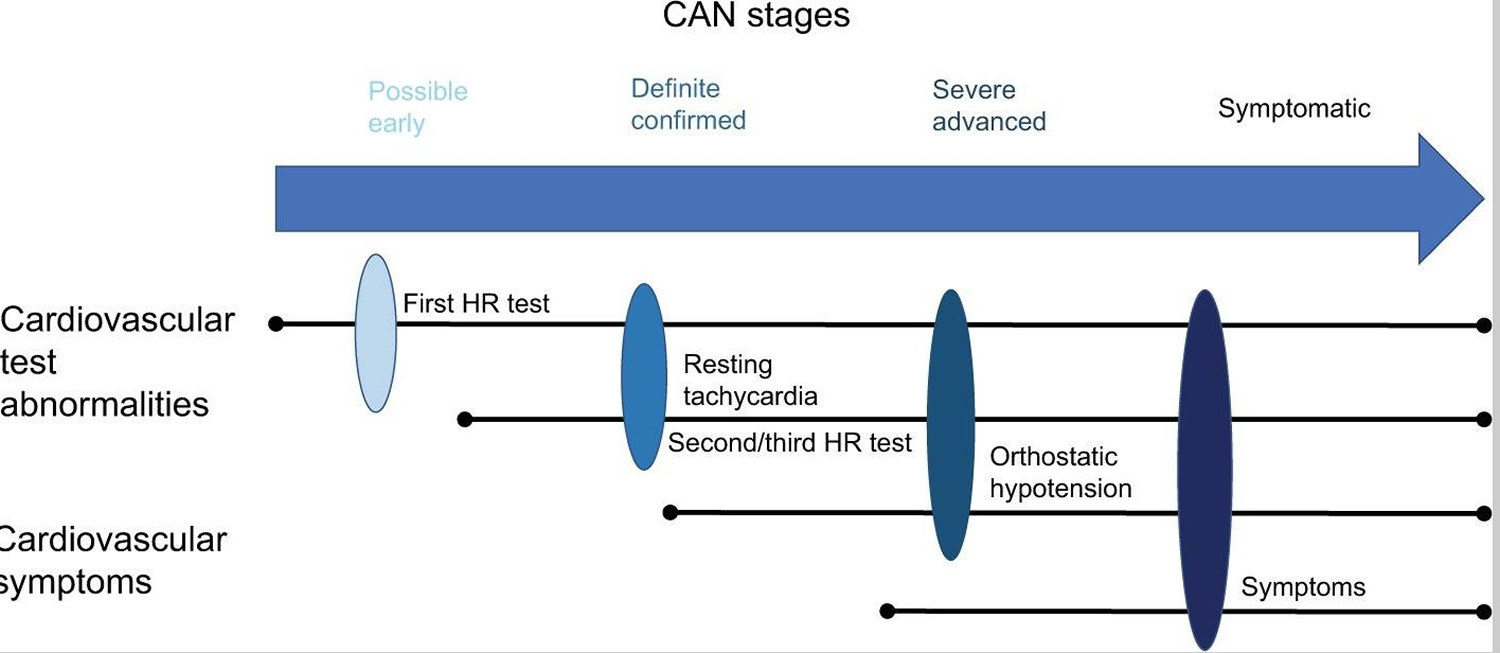
Cardiac autonomic neuropathy only becomes symptomatic in the later stages of the disease as it advances (Figure 3). The denervation of both the autonomic and peripheral nervous systems occurs in an ascending length-dependent manner 3. Therefore, the vagus nerve is usually the first nerve to be affected in cardiac autonomic neuropathy, resulting in patients presenting with symptoms of sympathetic predominance. However, this is initially limited to baroreceptor abnormalities and changes in heart-rate variability (HRV), but as the disease progresses cardiac involvement becomes more evident and symptomatic.
Figure 3. Cardiac autonomic neuropathy stages
 CAN = Cardiac Autonomic Neuropathy
CAN = Cardiac Autonomic Neuropathy
Notes: CAN symptoms can include reduced exercise tolerance, silent ischemia, interoperative complications, and lower-limb complications.
[Source 37]Resting tachycardia
One of the earliest signs of cardiac autonomic neuropathy is asymptomatic abnormalities in heart rate, which later progresses to resting tachycardia (90–130 bpm) 37. A fixed and unresponsive heart rate to breathing is associated with complete cardiac denervation and severe cardiac autonomic neuropathy 37. Therefore, resting heart rate can be used as a diagnostic and prognostic tool in patients with diabetes mellitus after excluding other causes of tachycardia 38. Furthermore, a study of 11,400 type 2 diabetes mellitus patients concluded that resting tachycardia is associated with an increased risk of death and cardiovascular complications. However, it was unclear whether this increased risk was a direct result of a higher resting heart rate or just a marker of other factors that may determine a poor outcome 39.
Reduced exercise tolerance
Parasympathetic denervation and sympathetic predominance are known to impair exercise tolerance by reducing heart rate and blood pressure response to activity, as well as blunting the appropriate increases in cardiac output. Patients with cardiac autonomic neuropathy who are undertaking an exercise program should be warned that heart rate cannot be used as an indicator of exercise intensity 37.
Orthostatic hypotension
Orthostatic hypotension is defined as a reduction in systolic BP >20 mmHg or diastolic BP >10 mmHg following a postural change from supine to standing, and is deemed to be a late sign in cardiac autonomic neuropathy 40. In diabetes, it is thought to be the result of efferent sympathetic vasomotor denervation leading to a defective reflex arc, culminating in an inadequate heart rate response and peripheral vasodilation when a person stands 41. Symptoms can include faintness, dizziness, and light-headness, and in severe cases a syncopal episode 42. Several medications given to patients with diabetes can aggravate these symptoms, including diuretics, vasodilators, tricyclic antidepressants, and insulin 40.
QT prolongation
QT prolongation is associated with cardiac arrhythmias and sudden death 43. Its pathogenesis is multifactorial, but includes an imbalance in cardiac sympathetic innervation and left-ventricular hypertrophy, as is often seen in cardiac autonomic neuropathy 44.
Silent ischemia and cardiovascular disease
Silent coronary artery disease has been reported with varied prevalence (12%–34%) in patients with diabetes mellitus, and is dependent on age, diabetes duration, and the presence of other risk factors 45. In the DIAD study, cardiac autonomic neuropathy (based on Valsalva ratio) was strongly associated with silent myocardial ischemia independently of traditional cardiovascular disease risk factors 46. cardiac autonomic neuropathy was also associated with a prolonged subjective angina threshold, meaning that electrocardiography changes occur prior to the onset of angina pectoris, and thus cardiac autonomic neuropathy patients may be more susceptible to silent myocardial ischemia and/or infarction 47.
A study by Valensi et al 48 showed that cardiac autonomic neuropathy was a better predictor of a major cardiac event than silent ischemia, but that patients with cardiac autonomic neuropathy and associated silent ischemia had the highest risk. A meta-analysis of 12 cross-sectional studies showed that cardiac autonomic neuropathy was associated with silent ischemia in patients with diabetes mellitus; the Mantel–Haenszel test estimated the prevalence risk ratio was 1.96 for patients with cardiac autonomic neuropathy vs no cardiac autonomic neuropathy when cardiac autonomic neuropathy was defined using two or more measures 40. The pathophysiology linking these two conditions is still unclear, with several proposed mechanisms, including reduced pain threshold, changes to the myocardial autonomic pathways or ischemic processes, and a causative relationship with both conditions being the product of coronary artery disease present in diabetes 3.
In the EURODIAB study, cardiac autonomic neuropathy was independently associated with cardiovascular disease in patients with type 1 diabetes mellitus 49. Similarly, the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications studies 50 showed that cardiac autonomic neuropathy predicted long-term cardiovascular disease events during follow-up. cardiac autonomic neuropathy was also found to be a predictor of cerebrovascular disease over 5–10 years of follow-up in patients with type 2 diabetes mellitus 51.
Cardiomyopathy
Diabetic cardiomyopathy results in systolic and diastolic dysfunction in the absence of structural and valvular cardiac disease, hypertension, or coronary vessel disease 52. The perceived mechanisms include left-ventricular hypertrophy, increased oxidative stress, altered substrate utilization, and mitochondrial dysfunction 53. The combination of sympathetic predominance and autonomic myocardial denervation leads to reduced coronary blood flow and thus diastolic and eventually systolic dysfunction 54.
Peri- and intraoperative complications
Patients with cardiac autonomic neuropathy have a greater risk of anesthetic-related complications. Sufferers of cardiac autonomic neuropathy may have varied hemodynamic response to induction and tracheal intubation, leading to intraoperative hypotension, which is thought to be due to an inability to vasoconstrict correctly after the use of vasodilatory agents, such as anesthesia.91,92 Furthermore, patients with cardiac autonomic neuropathy have been shown to have a two- to three-fold increase in perioperative morbidity and mortality as a result of severe intraoperative hypothermia 55. It is thus vital that all patients with a diabetes diagnosis are subject to a thorough preoperative assessment to minimize any of these complications perioperatively 55.
Lower-limb complications
Sudomotor dysfunction, which can lead to foot ulceration, is due to both peripheral neuropathy and autonomic neuropathy 3. A study over 5 years of 595 patients with type 2 diabetes mellitus showed that the development of foot ulcers was independently associated with cardiac autonomic neuropathy after adjustment for possible confounding factors, normal vs definite cardiac autonomic neuropathy 56. In addition, a study by Chahal et al 57 suggested that there may be an association between sudomotor dysfunction and peripheral artery disease, which can contribute to foot ulceration and lower-limb amputations. Autonomic dysfunction has been suggested to play an important role in the pathogenesis of Charcot neuroarthropathy in addition to periphery sensory and motor neuropathy 3.
Chronic kidney disease
A recent study of 755 patients showed that cardiac autonomic neuropathy was an independent predictor for developing chronic kidney disease in patients with type 2 diabetes mellitus after a 9.6-year follow-up 58. Cardiac autonomic neuropathy was also an independent predictor of estimated glomerular filtration-rate decline in patients with type 2 diabetes mellitus in another study 59. Similarly, in patients with type 1 diabetes mellitus, cardiac autonomic neuropathy predicted the development of albuminuria and estimated glomerular filtration-rate decline over a 14-year period 60.
Anemia
Anemia is positively associated with cardiac autonomic neuropathy in patients with type 2 diabetes mellitus, despite adjustment for sex, age, smoking habits, body-mass index, ALT, hyperlipidemia, hypertension, diabetes mellitus duration, HbA1c, retinopathy, and nephropathy 61.
Cardiac autonomic neuropathy treatment
Early determination of cardiac autonomic neuropathy is vital to the success of therapeutic input, as it has been suggested that cardiovascular denervation may be reversible if diagnosed soon after onset 62. The aim of cardiac autonomic neuropathy treatment is symptom control or slowing progression. Current methods use a combination of nonpharmacological and pharmacological approaches, including lifestyle modification, intensive glycemic control, and treating underlying risk factors, such as hyperlipidemia and hypertension.
Despite similarities in the pathogenesis of cardiac autonomic neuropathy and diabetic peripheral neuropathy, they are not the same disease. The STENO-2 trial showed improvements in cardiac autonomic neuropathy, but not diabetic peripheral neuropathy 63. In addition, cardiac autonomic neuropathy and diabetic peripheral neuropathy do not always coexist, despite similar pathogenesis, suggesting that these two conditions are not the same. It has also been shown that the ethnic differences seen in diabetic peripheral neuropathy prevalence between South Asians and white Caucasians are not seen in cardiac autonomic neuropathy. This could contribute to the differences observed between cardiac autonomic neuropathy and diabetic peripheral neuropathy responses to treatment. However, when considering treatment, it is important to consider the treatment modality and how responses were measured. For example, an improvement in the gold standard for diabetic peripheral neuropathy – nerve-conduction studies – does not necessarily correlate with an improvement in cardiac autonomic neuropathy and vice versa, whereas if corneal confocal microscopy, were used as a measure of diabetic peripheral neuropathy, it may lead to a different result. Indeed, following islet-cell transplant, corneal confocal microscopy shows improvements in corneal nerves 64.
Lifestyle modifications
Increased physical activity has been demonstrated to improve heart-rate variability and lower resting heart rate 65. Even simple activity, such as dog walking, could improve parasympathetic activity 66. The Diabetes Prevention Program demonstrated that lifestyle modifications were superior to metformin or placebo in regard to improvement in autonomic dysfunction (assessed with heart-rate variability and QT indices) 67. A review by Voulgari et al concluded that moderate endurance and aerobic exercise improved heart-rate variability and parasympathetic dominance in patients with type 1 diabetes mellitus or type 2 diabetes mellitus 68. Esposito et al 69 suggested that improvement in autonomic function following physical activity may be related to improvements in tissue hypoxia.
Intensive glycemic control
The DCCT trial 70 showed that intensive glycemic control reduced cardiac autonomic neuropathy incidence by 50% in type 1 diabetes mellitus over a 6.5-year follow-up when compared to conventional therapy (7% vs 14%). These beneficial effects of intensive glycemic control in DCCT persisted during long-term follow-up in the EDIC study 71, and thus early intervention with intensive therapy in patients with type 1 diabetes mellitus may help reduce the progression and development of cardiac autonomic neuropathy. Additionally, the SEARCH cardiovascularD study looked at subclinical autonomic dysfunction in 354 young type 1 diabetes mellitus patients. Dysfunction was assessed with heart-rate variability testing and the presence of parasympathetic loss with sympathetic override, and HbA1c >7.5% was independently associated with the presence of subclinical cardiac autonomic neuropathy when compared to a control group without diabetes mellitus 72.
The effects of intensive glycemic control on cardiac autonomic neuropathy in patients with type 2 diabetes mellitus is still unclear. The Veterans Affairs Cooperative Study 73 suggested no impact of intensive glycemic control on cardiac autonomic neuropathy. In another randomized controlled trial, intensive glycemic control in newly diagnosed type 2 diabetes mellitus in primary care did not have an impact on cardiac autonomic neuropathy prevalence at 6-year follow-up 74. Conversely, the STENO-2 trial demonstrated that intensive multifactorial treatment (including behavior modification and intensive therapy targeting hyperglycemia and cardiovascularD risk factors) lowered progression to autonomic neuropathy (based on heart-rate variability during paced breathing and orthostatic hypotension) in type 2 diabetes mellitus 75 these benefits were sustained at the 2-year follow-up 63.
Treatment of orthostatic hypotension
Management of orthostatic hypotension in cardiac autonomic neuropathy is complex, and the condition is difficult to treat 76. Nonpharmacological interventions include increasing water consumption, avoiding sudden changes in body posture, reducing physical maneuvers that increase intra-abdominal and intrathoracic pressure, use of stockings of the lower extremities, and eating smaller and more frequent meals 76. Additionally, certain medications have been associated with orthostatic hypotension and should thus be stopped in patients who are symptomatic, including tricyclic antidepressants, diuretics, and α-adrenoreceptor antagonists 76. If these methods are unsuccessful, then pharmacological methods should be considered on a risk–benefit basis: weighing up an increase in standing BP against prevention of supine hypotension 3.
Midodrine is a peripheral selective α1-adrenergic agonist that causes peripheral vasoconstriction of arterioles and veins and is the only Food and Drug Administration-approved drug, hence its common use 3. Other pharmacological agents used in the treatment of orthostatic hypotension include fludrocortisone, which is a synthetic mineralocorticoid, octreotide, which inhibits the release of vasoactive peptides in the gastrointestinal tract, leading to an increase in splanchnic vasoconstriction and a rise in mean arterial BP, erythropoietin, which increases intravascular volume and blood viscosity, and pyridostigmine, which is a cholinesterase inhibitor 77.
Autonomic neuropathy causes
Many health conditions can cause autonomic neuropathy. It may also be a side effect of treatments for other diseases, such as cancer. Some common causes of autonomic neuropathy include:
- Abnormal protein buildup in organs (amyloidosis), which affects the organs and the nervous system.
- Autoimmune diseases, in which your immune system attacks and damages parts of your body, including your nerves. Examples include Sjogren’s syndrome, systemic lupus erythematosus, rheumatoid arthritis and celiac disease. Guillain-Barre syndrome is an autoimmune disease that happens rapidly and can affect autonomic nerves. Autonomic neuropathy may also be caused by an abnormal attack by the immune system that occurs as a result of some cancers (paraneoplastic syndrome).
- Diabetes, which is the most common cause of autonomic neuropathy, can gradually cause nerve damage throughout the body.
- Injury to nerves caused by surgery or radiation to the neck.
- Treatment with certain medications, including some drugs used in cancer chemotherapy.
- Other chronic illnesses, such as Parkinson’s disease, multiple sclerosis and some types of dementia.
- Certain infectious diseases. Some viruses and bacteria, such as botulism, Lyme disease and HIV, can cause autonomic neuropathy.
- Inherited disorders. Certain hereditary disorders can cause autonomic neuropathy.
Risk factors for autonomic neuropathy
Factors that may increase your risk of autonomic neuropathy include:
- Diabetes. Diabetes, especially when poorly controlled, increases your risk of autonomic neuropathy and other nerve damage. You’re at greatest risk if you have had the disease for more than 25 years and have difficulty controlling your blood sugar, according to the National Institute of Diabetes and Digestive and Kidney Diseases. Additionally, people with diabetes who are overweight or have high blood pressure or high cholesterol have a higher risk of autonomic neuropathy.
- Other diseases. Amyloidosis, porphyria, hypothyroidism and cancer (usually due to side effects from treatment) may also increase the risk of autonomic neuropathy.
Autonomic neuropathy prevention
While certain inherited diseases that put you at risk of developing autonomic neuropathy can’t be prevented, you can slow the onset or progression of symptoms by taking good care of your health in general and managing your medical conditions.
Follow your doctor’s advice on healthy living to control diseases and conditions, which may include these recommendations:
- Control your blood sugar if you have diabetes.
- Seek treatment for alcoholism.
- Get appropriate treatment for any autoimmune disease.
- Take steps to prevent or control high blood pressure.
- Achieve and maintain a healthy weight.
- Stop smoking.
- Exercise regularly.
Autonomic neuropathy symptoms
Signs and symptoms of autonomic neuropathy vary based on the nerves affected. They may include:
- Dizziness and fainting when standing caused by a sudden drop in blood pressure.
- Urinary problems, such as difficulty starting urination, incontinence, difficulty sensing a full bladder and inability to completely empty the bladder, which can lead to urinary tract infections.
- Sexual difficulties, including problems achieving or maintaining an erection (erectile dysfunction) or ejaculation problems in men and vaginal dryness, low libido and difficulty reaching orgasm in women.
- Difficulty digesting food, such as feeling full after a few bites of food, loss of appetite, diarrhea, constipation, abdominal bloating, nausea, vomiting, difficulty swallowing and heartburn, all due to changes in digestive function.
- Sweating abnormalities, such as sweating too much or too little, which affects the ability to regulate body temperature.
- Sluggish pupil reaction, making it difficult to adjust from light to dark and seeing well when driving at night.
- Exercise intolerance, which may occur if your heart rate stays the same instead of adjusting in response to your activity level.
Autonomic neuropathy diagnosis
Autonomic neuropathy is a possible complication of a number of diseases, and the tests you’ll need often depend on your symptoms and risk factors for autonomic neuropathy.
When you have known risk factors for autonomic neuropathy
If you have conditions that increase your risk of autonomic neuropathy (such as diabetes) and have symptoms of the condition, extensive testing may not be necessary. Your doctor may perform a physical exam and ask about your symptoms.
If you are undergoing cancer treatment with a drug known to cause nerve damage, your doctor will check for signs of neuropathy.
When you don’t have risk factors for autonomic neuropathy
If you have symptoms of autonomic neuropathy but don’t have risk factors, the diagnosis may be more involved. Your doctor will probably review your medical history, discuss your symptoms and do a physical exam.
Your doctor may perform tests to evaluate autonomic functions, which may include:
- Breathing tests. These tests measure how your heart rate and blood pressure respond during exercises such as forcefully exhaling (Valsalva maneuver).
- Tilt-table test. This test monitors the response of blood pressure and heart rate to changes in posture and position, simulating what occurs when you stand up after lying down. You lie flat on a table, which is then tilted to raise the upper part of your body. Normally, your body narrows blood vessels and increases heart rate to compensate for the drop in blood pressure. This response may be slowed or abnormal if you have autonomic neuropathy. A simpler way test for this response involves standing for a minute, then squatting for a minute and then standing again while blood pressure and heart rate are monitored.
- Gastrointestinal tests. Gastric-emptying tests are the most common tests to check for digestive abnormalities such as slow digestion and delayed emptying of the stomach (gastroparesis). These tests are usually done by a doctor who specializes in digestive disorders (gastroenterologist).
- Quantitative sudomotor axon reflex test. This test evaluates how the nerves that regulate your sweat glands respond to stimulation. A small electrical current passes through four capsules placed on your forearm, foot and leg, while a computer analyzes the response of your nerves and sweat glands. You may feel warmth or a tingling sensation during the test.
- Thermoregulatory sweat test. During this test, you’re coated with a powder that changes color when you sweat. While lying in a chamber with slowly increasing temperature, digital photos document the results as you begin to sweat. Your sweat pattern may help confirm a diagnosis of autonomic neuropathy or suggest other causes for decreased or increased sweating.
- Urinalysis and bladder function (urodynamic) tests. If you have bladder or urinary symptoms, a series of urine tests can evaluate bladder function.
- Ultrasound. If you have bladder symptoms, your doctor may do an ultrasound in which high-frequency sound waves create an image of the bladder and other parts of the urinary tract.
Autonomic neuropathy treatment
Treatment of autonomic neuropathy includes:
- Treating the underlying disease. The first goal of treating autonomic neuropathy is to manage the disease or condition damaging your nerves. For example, if the underlying cause is diabetes, you’ll need to tightly control blood sugar to prevent autonomic neuropathy from progressing.
- Managing specific symptoms. Some treatments can relieve the symptoms of autonomic neuropathy. Treatment is based on what part of your body is most affected by nerve damage.
Digestive (gastrointestinal) symptoms
Your doctor may recommend:
- Modifying your diet. You may need to increase dietary fiber and fluids. Fiber supplements, such as Metamucil or Citrucel, also may help. Slowly increase fiber to avoid gas and bloating.
- Medication to help your stomach empty. A prescription drug called metoclopramide (Reglan) helps your stomach empty faster by increasing the contractions of the digestive tract. This medication may cause drowsiness, and its effectiveness wears off over time.
- Medications to ease constipation. Over-the-counter laxatives may help ease constipation. Ask your doctor how often you should use these medications. Increasing dietary fiber also may help relieve constipation.
- Medications to ease diarrhea. Antibiotics can help treat diarrhea by preventing excess bacterial growth in the intestines. Medications usually used to treat high blood pressure and cholesterol may also be prescribed for managing diarrhea.
- Antidepressants. Tricyclic antidepressants, such as imipramine (Tofranil), can help treat nerve-related abdominal pain. Dry mouth and urine retention are possible side effects of these medications.
Urinary symptoms
Your doctor may suggest:
- Retraining your bladder. Following a schedule of when to drink fluids and when to urinate can help increase your bladder’s capacity and retrain your bladder to empty completely at the appropriate times.
- Medication to help empty the bladder. Bethanechol is a medication that helps ensure complete emptying of the bladder. Possible side effects include headache, abdominal cramping, bloating, nausea and flushing.
- Urinary assistance (catheterization). During this procedure, a tube is guided through your urethra to empty your bladder.
- Medications that decrease overactive bladder. These include tolterodine (Detrol) or oxybutynin (Ditropan XL). Possible side effects include dry mouth, headache, fatigue, constipation and abdominal pain.
Sexual dysfunction
For men with erectile dysfunction, your doctor may recommend:
- Medications that enable erections. Drugs such as sildenafil (Viagra), vardenafil (Levitra) or tadalafil (Cialis) can help you achieve and maintain an erection. Possible side effects include mild headache, flushing, upset stomach and changes in color vision. If you have a history of heart disease, arrhythmia, stroke or high blood pressure, use these medications with caution and medical discretion. Also avoid taking these medications if you are taking any type of organic nitrates. Seek immediate medical assistance if you have an erection that lasts longer than four hours.
- An external vacuum pump. This device helps pull blood into the penis using a hand pump. A tension ring helps keep the blood in place, maintaining the erection for up to 30 minutes.
For women with sexual symptoms, your doctor may recommend:
- Vaginal lubricants. Vaginal lubricants may decrease dryness and make sexual intercourse more comfortable and enjoyable.
Heart rhythm and blood pressure symptoms
Autonomic neuropathy can cause a number of heart rate and blood pressure problems. Your doctor may prescribe:
- Medications that help raise your blood pressure. If you feel faint or dizzy when you stand up, your doctor may suggest a drug called fludrocortisone. This medication helps your body retain salt, which helps regulate your blood pressure. Other drugs that can help raise your blood pressure include midodrine and pyridostigmine (Mestinon). Midodrine may cause high blood pressure when lying down.
- Medication that helps regulate your heart rate. A class of medications called beta blockers helps to regulate your heart rate if it goes too high with an activity level.
- A high-salt, high-fluid diet. If your blood pressure drops when you stand up, a high-salt, high fluid diet may help maintain your blood pressure. This is generally only recommended for very severe cases of blood pressure problems, as this treatment may cause blood pressure that is too high or swelling of the feet, ankles or legs.
Sweating
If you experience excessive sweating, your doctor may prescribe:
- A medication that decreases perspiration. The drug glycopyrrolate (Robinul, Robinul Forte) can decrease sweating. Side effects may include diarrhea, dry mouth, urinary retention, blurred vision, changes in heart rate, headaches, loss of taste and drowsiness. Glycopyrrolate may also increase the risk of heat-related illness (such as heatstroke) from a reduced ability to sweat.
Home remedies
- Posture changes. Stand up slowly, in stages, to decrease dizziness. Sit with your legs dangling over the side of the bed for a few minutes before getting out of bed. Flex your feet and grip your hands for a few seconds before standing up, to increase blood flow. Once standing, try tensing your leg muscles while crossing one leg over the other a few times to increase blood pressure.
- Elevate the bed. If you have low blood pressure, it may also help to raise the head of your bed by about 4 inches by placing blocks or risers under the legs at the head of the bed.
- Digestion. Eat small, frequent meals to combat digestive problems. Increase fluids, and opt for low-fat, high-fiber foods, which may improve digestion. You may also want to try restricting foods that contain lactose and gluten.
- Diabetes management. Try to keep your blood sugar as close to normal as possible. Tight blood sugar control can help lessen symptoms and help to prevent or delay the onset of new problems.
Alternative medicine
Several alternative medicine treatments may help people with autonomic neuropathy. Remember to discuss any new treatments with your doctor to ensure that they won’t interfere with treatments you’re already receiving or cause you any harm.
Alpha-lipoic acid
Preliminary research suggests this antioxidant may be helpful in slowing or even reversing neuropathy that’s causing blood pressure or heart rate problems, but more study is needed.
Acupuncture
This therapy, which uses numerous thin needles placed in specific points in the body, may help treat slow stomach emptying. More studies are needed to confirm what acupuncture’s role is in treating autonomic neuropathy.
Electrical nerve stimulation
Some studies have found that this therapy, which uses low-energy electrical waves transmitted through electrodes placed on the skin, may help ease pain associated with diabetic neuropathy.
Coping and support
Living with a chronic condition presents daily challenges. Some of these suggestions may make it easier for you to cope:
- Set priorities. Accomplish the most important tasks, such as paying bills or grocery shopping, and save less important tasks for another day. Stay active, but don’t overdo it.
- Seek and accept help from friends and family. Having a support system and a positive attitude can help you cope with the challenges you face. Ask for or accept help when you need it. Don’t shut yourself off from loved ones.
- Talk to a counselor or therapist. Depression and impotence are possible complications of autonomic neuropathy. Seek help from a counselor or therapist in addition to your primary care doctor to discuss possible treatments.
- Consider joining a support group. Ask your doctor about support groups in your area. If there isn’t a specific group for people with neuropathies, you may find that there’s a support group for your underlying condition, such as diabetes. Some people find it helpful to talk to other people who truly understand what they’re going through. Support group members can offer camaraderie, as well as tips or tricks to make living with autonomic neuropathy easier.
- Cardiovascular autonomic neuropathy in diabetes: clinical impact, assessment, diagnosis, and management. Spallone V, Ziegler D, Freeman R, Bernardi L, Frontoni S, Pop-Busui R, Stevens M, Kempler P, Hilsted J, Tesfaye S, Low P, Valensi P, Toronto Consensus Panel on Diabetic Neuropathy. Diabetes Metab Res Rev. 2011 Oct; 27(7):639-53. https://deepblue.lib.umich.edu/bitstream/handle/2027.42/86954/1239_ftp.pdf[↩]
- The International Diabetes Federation. https://www.idf.org/[↩]
- Dimitropoulos G, Tahrani AA, Stevens MJ. Cardiac autonomic neuropathy in patients with diabetes mellitus. World J Diabetes. 2014;5(1):17–39. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3932425/[↩][↩][↩][↩][↩][↩][↩]
- Freeman R. Diabetic autonomic neuropathy. Handb Clin Neurol. 2014;126:63–79. https://www.ncbi.nlm.nih.gov/pubmed/25410215[↩]
- Soedamah-Muthu SS, Chaturvedi N, Witte DR, et al. Relationship between risk factors and mortality in type 1 diabetic patients in Europe: the EURODIAB Prospective Complications Study (PCS) Diabetes Care. 2008;31(7):1360–1366. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2453640/[↩]
- Javorka K, Javorková J, Petrásková M, Tonhajzerová I, Buchanec J, Chromá O. Heart rate variability and cardiovascular tests in young patients with diabetes mellitus type 1. J Pediatr Endocrinol Metab. 1999;12:423–431. https://www.ncbi.nlm.nih.gov/pubmed/10821222[↩]
- Spallone V, Ziegler D, Freeman R, Bernardi L, Frontoni S, Pop-Busui R, Stevens M, Kempler P, Hilsted J, Tesfaye S, Low P, Valensi P; on behalf of the Toronto Consensus Panel on Diabetic Neuropathy*. Cardiovascular autonomic neuropathy in diabetes: clinical impact, assessment, diagnosis, and management. Diabetes Metab Res Rev. 2011:Jun 22; Epub ahead of print. https://www.ncbi.nlm.nih.gov/pubmed/21695768[↩]
- Vinik AI, Freeman R, Erbas T. Diabetic autonomic neuropathy. Semin Neurol. 2003;23:365–372. https://www.ncbi.nlm.nih.gov/pubmed/15088257[↩]
- Pop-Busui R, Low PA, Waberski BH, Martin CL, Albers JW, Feldman EL, Sommer C, Cleary PA, Lachin JM, Herman WH. Effects of prior intensive insulin therapy on cardiac autonomic nervous system function in type 1 diabetes mellitus: the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study (DCCT/EDIC) Circulation. 2009;119:2886–2893. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2757005/[↩][↩]
- Ziegler D. Diabetic cardiovascular autonomic neuropathy: prognosis, diagnosis and treatment. Diabetes Metab Rev. 1994;10:339–383. https://www.ncbi.nlm.nih.gov/pubmed/7796704[↩]
- The effect of intensive diabetes therapy on measures of autonomic nervous system function in the Diabetes Control and Complications Trial (DCCT) Diabetologia. 1998;41:416–423. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2635092/[↩]
- Boulton AJ, Vinik AI, Arezzo JC, Bril V, Feldman EL, Freeman R, Malik RA, Maser RE, Sosenko JM, Ziegler D. Diabetic neuropathies: a statement by the American Diabetes Association. Diabetes Care. 2005;28:956–962. https://www.ncbi.nlm.nih.gov/pubmed/15793206[↩]
- Gaede P, Vedel P, Larsen N, Jensen GV, Parving HH, Pedersen O. Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N Engl J Med. 2003;348:383–393. http://www.nejm.org/doi/full/10.1056/NEJMoa021778[↩][↩]
- Gaede P, Lund-Andersen H, Parving HH, Pedersen O. Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl J Med. 2008;358:580–591. http://www.nejm.org/doi/full/10.1056/NEJMoa0706245[↩]
- Ko SH, Park SA, Cho JH, Song KH, Yoon KH, Cha BY, Son HY, Yoo KD, Moon KW, Park YM, et al. Progression of cardiovascular autonomic dysfunction in patients with type 2 diabetes: a 7-year follow-up study. Diabetes Care. 2008;31:1832–1836. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2518354/[↩]
- Low PA, Benrud-Larson LM, Sletten DM, Opfer-Gehrking TL, Weigand SD, O’Brien PC, Suarez GA, Dyck PJ. Autonomic symptoms and diabetic neuropathy: a population-based study. Diabetes Care. 2004;27:2942–2947. https://www.ncbi.nlm.nih.gov/pubmed/15562211[↩]
- Kempler P, Tesfaye S, Chaturvedi N, Stevens LK, Webb DJ, Eaton S, Kerényi Z, Tamás G, Ward JD, Fuller JH. Autonomic neuropathy is associated with increased cardiovascular risk factors: the EURODIAB IDDM Complications Study. Diabet Med. 2002;19:900–909. https://www.ncbi.nlm.nih.gov/pubmed/12421426[↩]
- Pop-Busui R, Evans GW, Gerstein HC, Fonseca V, Fleg JL, Hoogwerf BJ, Genuth S, Grimm RH, Corson MA, Prineas R. Effects of cardiac autonomic dysfunction on mortality risk in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial. Diabetes Care. 2010;33:1578–1584. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2890362/[↩]
- Abbott CA, Chaturvedi N, Malik RA, Salgami E, Yates AP, Pemberton PW, Boulton AJ. Explanations for the lower rates of diabetic neuropathy in Indian Asians versus Europeans. Diabetes Care. 2010;33:1325–1330. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2875448/[↩]
- Edwards JL, Vincent AM, Cheng HT, Feldman EL. Diabetic neuropathy: mechanisms to management. Pharmacol Ther. 2008;120(1):1–34. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4007052/[↩]
- Verrotti A, Prezioso G, Scattoni R, Chiarelli F. Autonomic neuropathy in diabetes mellitus. Front Endocrinol (Lausanne) 2014;5:205. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4249492/[↩]
- Edwards JL, Vincent AM, Cheng HT, Feldman EL. Diabetic neuropathy: mechanisms to management. Pharmacol Ther. 2008;120(1):1–34. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4007052[↩]
- Polydefkis M, Griffin JW, McArthur J. New insights into diabetic polyneuropathy. JAMA. 2003;290(10):1371–6. https://www.ncbi.nlm.nih.gov/pubmed/12966130[↩]
- Shah MS, Brownlee M. Molecular and cellular mechanisms of cardiovascular disorders in diabetes. Circ Res. 2016;118(11):1808–1829. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4888901/[↩]
- Gibbons C, Centi J, Vernino S, Freeman R. Autoimmune autonomic ganglionopathy with reversible cognitive impairment. Arch Neurol. 2012;69(4):461–466. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3359761/[↩]
- Skärstrand H, Dahlin LB, Lernmark A, Vaziri-Sani F. Neuropeptide Y autoantibodies in patients with long-term type 1 and type 2 diabetes and neuropathy. J Diabetes Complications. 2013;27(6):609–617. https://www.ncbi.nlm.nih.gov/pubmed/23910631[↩]
- Berntorp K, Frid A, Alm R, Fredrikson GN, Sjöberg K, Ohlsson B. Antibodies against gonadotropin-releasing hormone (GnRH) in patients with diabetes mellitus is associated with lower body weight and autonomic neuropathy. BMC Res Notes. 2013;6:329. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3751565/[↩]
- Zanone MM, Raviolo A, Coppo E, et al. Association of autoimmunity to autonomic nervous structures with nerve function in patients with type 1 diabetes: a 16-year prospective study. Diabetes Care. 2014;37(4):1108–1115. https://www.ncbi.nlm.nih.gov/pubmed/24550215[↩]
- Politi C, Ciccacci C, D’Amato C, Novelli G, Borgiani P, Spallone V. Recent advances in exploring the genetic susceptibility to diabetic neuropathy. Diabetes Res Clin Pract. 2016;120:198–208. https://www.ncbi.nlm.nih.gov/pubmed/27596057[↩]
- Ciccacci C, Morganti R, Di Fusco D, et al. Common polymorphisms in MIR146a, MIR128a and MIR27a genes contribute to neuropathy susceptibility in type 2 diabetes. Acta Diabetol. 2014;51(4):663–671. https://www.ncbi.nlm.nih.gov/pubmed/24682535[↩]
- Osztovits J, Horváth T, Littvay L, et al. Effects of genetic vs. environmental factors on cardiovascular autonomic function: a twin study. Diabet Med. 2011;28(10):1241–8. https://www.ncbi.nlm.nih.gov/pubmed/21679234[↩]
- Tahrani AA. Obstructive sleep apnoea in diabetes: does it matter? Diab Vasc Dis Res. 2017 Jun 1; Epub. https://www.ncbi.nlm.nih.gov/pubmed/28610436[↩]
- Janovsky CC, Rolim LC, De Sá JR, et al. Cardiovascular autonomic neuropathy contributes to sleep apnea in young and lean type 1 diabetes mellitus patients. Front Endocrinol (Lausanne) 2014;5:119. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4127968/[↩]
- Stevens M, Ali A, Dubb K, Begum S, Piya M, Tahrani A. Obstructive sleep apnoea is associated with cardiac autonomic abnormalities in patients with type 2 diabetes. https://insights.ovid.com/diabetic-medicine/diame/2014/03/001/obstructive-sleep-apnoea-associated-cardiac/110/00003135[↩]
- Tahrani AA, Ali A, Raymond NT, et al. Obstructive Sleep Apnea and Diabetic Neuropathy: A Novel Association in Patients with Type 2 Diabetes. American Journal of Respiratory and Critical Care Medicine. 2012;186(5):434-441. doi:10.1164/rccm.201112-2135OC. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3443800/[↩]
- Vinik AI, Erbas T, Casellini CM. Diabetic cardiac autonomic neuropathy, inflammation and cardiovascular disease. J Diabetes Investig. 2013;4(1):4–18. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3580884/[↩][↩][↩]
- Vinik AI, Erbas T, Casellini CM. Diabetic cardiac autonomic neuropathy, inflammation and cardiovascular disease. Journal of Diabetes Investigation. 2013;4(1):4-18. doi:10.1111/jdi.12042. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3580884/[↩][↩][↩][↩]
- Dimitropoulos G, Tahrani AA, Stevens MJ. Cardiac autonomic neuropathy in patients with diabetes mellitus. World Journal of Diabetes. 2014;5(1):17-39. doi:10.4239/wjd.v5.i1.17. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3932425/[↩]
- Hillis G, Woodward M, Rodgers A, et al. Resting heart rate and the risk of death and cardiovascular complications in patients with type 2 diabetes mellitus. Diabetologia. 2012;55(5):1283–1290. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4170780/[↩]
- Vinik AI, Ziegler D. Diabetic cardiovascular autonomic neuropathy. Circulation. 2007;115(3):387–397. http://circ.ahajournals.org/content/115/3/387.long[↩][↩][↩]
- Low PA, Benrud-Larson LM, Sletten DM, et al. Autonomic symptoms and diabetic neuropathy: a population-based study. Diabetes Care. 2004;27(12):2942–2947. https://www.ncbi.nlm.nih.gov/pubmed/15562211[↩]
- Pop-Busui R. What do we know and we do not know about cardiovascular autonomic neuropathy in diabetes. J Cardiovasc Transl Res. 2012;5(4):463–478. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3634565/[↩]
- Ninkovic VM, Ninkovic SM, Miloradovic V, et al. Prevalence and risk factors for prolonged QT interval and QT dispersion in patients with type 2 diabetes. Acta Diabetol. 2016;53(5):737–744. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5014905/[↩]
- Veglio M, Chinaglia A, Cavallo-Perin P. QT interval, cardiovascular risk factors and risk of death in diabetes. J Endocrinol Invest. 2004;27(2):175–181. https://www.ncbi.nlm.nih.gov/pubmed/15129815[↩]
- Sargin H, Ozisik M, Ozisik NC, et al. The prevalence of silent ischemia in Turkish patients with type 2 diabetes mellitus. Tohoku J Exp Med. 2005;205(4):351–355. https://www.ncbi.nlm.nih.gov/pubmed/15750331[↩]
- Wackers FJ, Young LH, Inzucchi SE, et al. Detection of silent myocardial ischemia in asymptomatic diabetic subjects: the DIAD study. Diabetes Care. 2004;27(8):1954–1961. https://www.ncbi.nlm.nih.gov/pubmed/15277423[↩]
- Ambepityia G, Kopelman PG, Ingram D, Swash M, Mills PG, Timmis AD. Exertional myocardial ischemia in diabetes: a quantitative analysis of anginal perceptual threshold and the influence of autonomic function. J Am Coll Cardiol. 1990;15(1):72–77. https://www.ncbi.nlm.nih.gov/pubmed/2295745[↩]
- Valensi P, Sachs RN, Harfouche B, et al. Predictive value of cardiac autonomic neuropathy in diabetic patients with or without silent myocardial ischemia. Diabetes Care. 2001;24(2):339–343. https://www.ncbi.nlm.nih.gov/pubmed/11213889[↩]
- Witte DR, Tesfaye S, Chaturvedi N, et al. Risk factors for cardiac autonomic neuropathy in type 1 diabetes mellitus. Diabetologia. 2005;48(1):164–171. https://www.ncbi.nlm.nih.gov/pubmed/15619072[↩]
- Pop-Busui R, Braffett BH, Zinman B, et al. Cardiovascular autonomic neuropathy and cardiovascular outcomes in the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) study. Diabetes Care. 2017;40(1):94–100. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5180458/[↩]
- Töyry JP, Niskanen LK, Länsimies EA, Partanen KP, Uusitupa MI. Autonomic neuropathy predicts the development of stroke in patients with non-insulin-dependent diabetes mellitus. Stroke. 1996;27(8):1316–1318. http://stroke.ahajournals.org/content/27/8/1316.long[↩]
- Hayat SA, Patel B, Khattar RS, Malik RA. Diabetic cardiomyopathy: mechanisms, diagnosis and treatment. Clin Sci. 2004;107(6):539–557. https://www.ncbi.nlm.nih.gov/pubmed/15341511[↩]
- Boudina S, Abel ED. Diabetic cardiomyopathy, causes and effects. Rev Endocr Metab Disord. 2010;11(1):31–39. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2914514/[↩]
- Vinik AI, Erbas T. Diabetic autonomic neuropathy. Handb Clin Neurol. 2013;117:279–294. https://www.ncbi.nlm.nih.gov/pubmed/24095132[↩]
- Oakley I, Emond L. Diabetic cardiac autonomic neuropathy and anesthetic management: review of the literature. AANA J. 2011;79(6):473–479. https://www.ncbi.nlm.nih.gov/pubmed/22400413[↩][↩]
- Yun JS, Cha SA, Lim TS, et al. Cardiovascular autonomic dysfunction predicts diabetic foot ulcers in patients with type 2 diabetes without diabetic polyneuropathy. Medicine (Baltimore) 2016;95(12):e3128. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4998383/[↩]
- Chahal S, Vohra K, Syngle A. Association of sudomotor function with peripheral artery disease in type 2 diabetes. Neurol Sci. 2017;38(1):151–156. https://www.ncbi.nlm.nih.gov/pubmed/27783183[↩]
- Yun JS, Ahn YB, Song KH, et al. The association between abnormal heart rate variability and new onset of chronic kidney disease in patients with type 2 diabetes: a ten-year follow-up study. Diabetes Res Clin Pract. 2015;108(1):31–37. https://www.ncbi.nlm.nih.gov/pubmed/25656759[↩]
- Tahrani AA, Dubb K, Raymond NT, et al. Cardiac autonomic neuropathy predicts renal function decline in patients with type 2 diabetes: a cohort study. Diabetologia. 2014;57(6):1249–1256. https://www.ncbi.nlm.nih.gov/pubmed/24623102[↩]
- Forsén A, Kangro M, Sterner G, et al. A 14-year prospective study of autonomic nerve function in type 1 diabetic patients: association with nephropathy. Diabet Med. 2004;21(8):852–858. https://www.ncbi.nlm.nih.gov/pubmed/15270788[↩]
- Chung JO, Park SY, Cho DH, Chung DJ, Chung MY. Anemia, bilirubin, and cardiovascular autonomic neuropathy in patients with type 2 diabetes. Medicine (Baltimore) 2017;96(15):e6586. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5403087/[↩]
- Howorka K, Pumprla J, Haber P, Koller-Strametz J, Mondrzyk J, Schabmann A. Effects of physical training on heart rate variability in diabetic patients with various degrees of cardiovascular autonomic neuropathy. Cardiovasc Res. 1997;34(1):206–214. https://www.ncbi.nlm.nih.gov/pubmed/9217892[↩]
- Gæde P, Oellgaard J, Carstensen B, et al. Years of life gained by multifactorial intervention in patients with type 2 diabetes mellitus and microalbuminuria: 21 years follow-up on the Steno-2 randomised trial. Diabetologia. 2016;59(11):2298–2307. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5506099/[↩][↩]
- Tavakoli M, Begum P, McLaughlin J, Malik R. Corneal confocal microscopy for the diagnosis of diabetic autonomic neuropathy. Muscle Nerve. 2015;52(3):363–370. https://www.ncbi.nlm.nih.gov/pubmed/25556884[↩]
- Soares-Miranda L, Sandercock G, Vale S, et al. Metabolic syndrome, physical activity and cardiac autonomic function. Diabetes Metab Res Rev. 2012;28(4):363–369. https://www.ncbi.nlm.nih.gov/pubmed/22238216[↩]
- Motooka M, Koike H, Yokoyama T, Kennedy NL. Effect of dog-walking on autonomic nervous activity in senior citizens. Med J Aust. 2006;184(2):60–63. https://www.ncbi.nlm.nih.gov/pubmed/16411869[↩]
- Carnethon MR, Prineas RJ, Temprosa M, et al. The association among autonomic nervous system function, incident diabetes, and intervention arm in the Diabetes Prevention Program. Diabetes Care. 2006;29(4):914–919. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1751934/[↩]
- Voulgari C, Pagoni S, Vinik A, Poirier P. Exercise improves cardiac autonomic function in obesity and diabetes. Metab Clin Exp. 2013;62(5):609–621. https://www.ncbi.nlm.nih.gov/pubmed/23084034[↩]
- Esposito P, Mereu R, De Barbieri G, et al. Trained breathing-induced oxygenation acutely reverses cardiovascular autonomic dysfunction in patients with type 2 diabetes and renal disease. Acta Diabetol. 2016;53(2):217–226. https://www.ncbi.nlm.nih.gov/pubmed/25956276[↩]
- Ceriello A, Esposito K, Ihnat M, Thorpe J, Giugliano D. Long-term glycemic control influences the long-lasting effect of hyperglycemia on endothelial function in type 1 diabetes. J Clin Endocrinol Metab. 2009;94(8):2751–2756. https://www.ncbi.nlm.nih.gov/pubmed/19491221[↩]
- Pop-Busui R, Low PA, Waberski BH, et al. Effects of prior intensive insulin therapy on cardiac autonomic nervous system function in type 1 diabetes mellitus: the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study (DCCT/EDIC) Circulation. 2009;119(22):2886–2893. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2757005/[↩]
- Jaiswal M, Urbina EM, Wadwa RP, et al. Reduced heart rate variability among youth with type 1 diabetes: the SEARCH CVD study. Diabetes Care. 2013;36(1):157–162. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3526238/[↩]
- Charles M, Fleischer J, Witte DR, et al. Impact of early detection and treatment of diabetes on the 6-year prevalence of cardiac autonomic neuropathy in people with screen-detected diabetes: ADDITION-Denmark, a cluster-randomised study. Diabetologia. 2013;56(1):101–108. https://www.ncbi.nlm.nih.gov/pubmed/23064291[↩]
- Azad N, Emanuele NV, Abraira C, et al. The effects of intensive glycemic control on neuropathy in the VA cooperative study on type II diabetes mellitus (VA CSDM) J Diabetes Complications. 1999;13(5–6):307–313. https://www.ncbi.nlm.nih.gov/pubmed/10765007[↩]
- Gæde P, Vedel P, Parving HH, Pedersen O. Intensified multifactorial intervention in patients with type 2 diabetes mellitus and microalbuminuria: the Steno type 2 randomised study. Lancet. 1999;353(9153):617–622. https://www.ncbi.nlm.nih.gov/pubmed/10030326[↩]
- Balcıoğlu AS, Müderrisoğlu H. Diabetes and cardiac autonomic neuropathy: clinical manifestations, cardiovascular consequences, diagnosis and treatment. World J Diabetes. 2015;6(1):80–91. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4317320/[↩][↩][↩]
- Shibao C, Lipsitz LA, Biaggioni I. ASH position paper: evaluation and treatment of orthostatic hypotension. J Clin Hypertens (Greenwich) 2013;15(3):147–153. https://www.ncbi.nlm.nih.gov/pubmed/23458585[↩]