Contents
What is a bone bruise
Bone bruises also called bone contusions 1, subchondral edema or trabecular microfractures (not visible on plain x-ray images) 2.
In 1988, Wilson et al 3 introduced the term “bone marrow edema” in describing a group of patients with traumatic debilitating knee and hip pain. On T2-weighted magnetic resonance (MR) images, they recognized an ill-defined hyperintensity in the bone marrow where standard (x-ray images) radiographs showed nonspecific osteopenia or normal findings. This discrepancy in the imaging studies was labeled bone marrow edema owing to the “lack of a better term and to emphasize the generic character of the condition.” The proposed pathogenesis of these marrow changes asserts that increased blood pooling, edema, reactive hyperemia, and possible microfracture of the trabecular subchondral bone alter the marrow signal intensity 4. In time, “bone marrow edema” earned the name “bone bruise” to reflect its traumatic nature 5.
Moreover, the term bone bruise should be applied exclusively to subchondral lesions that exhibit the typical magnetic resonance (MR) findings. These lesions are acute traumatic noncystic areas of bone marrow edema, and they can be distinguished from cystic lesions and atraumatic presentations 6. There are many causes may result in bone marrow lesions (Table 1) that look like bone bruises on magnetic resonance (MR) images. However, bone bruises (acute traumatic noncystic bone marrow edema) are often caused by falls, sports injuries, car accidents, or blows received from other people or objects. Besides pure acute traumatic noncystic bone marrow edema causes, bone bruise may result from repetitive or chronic trauma in sports activities.
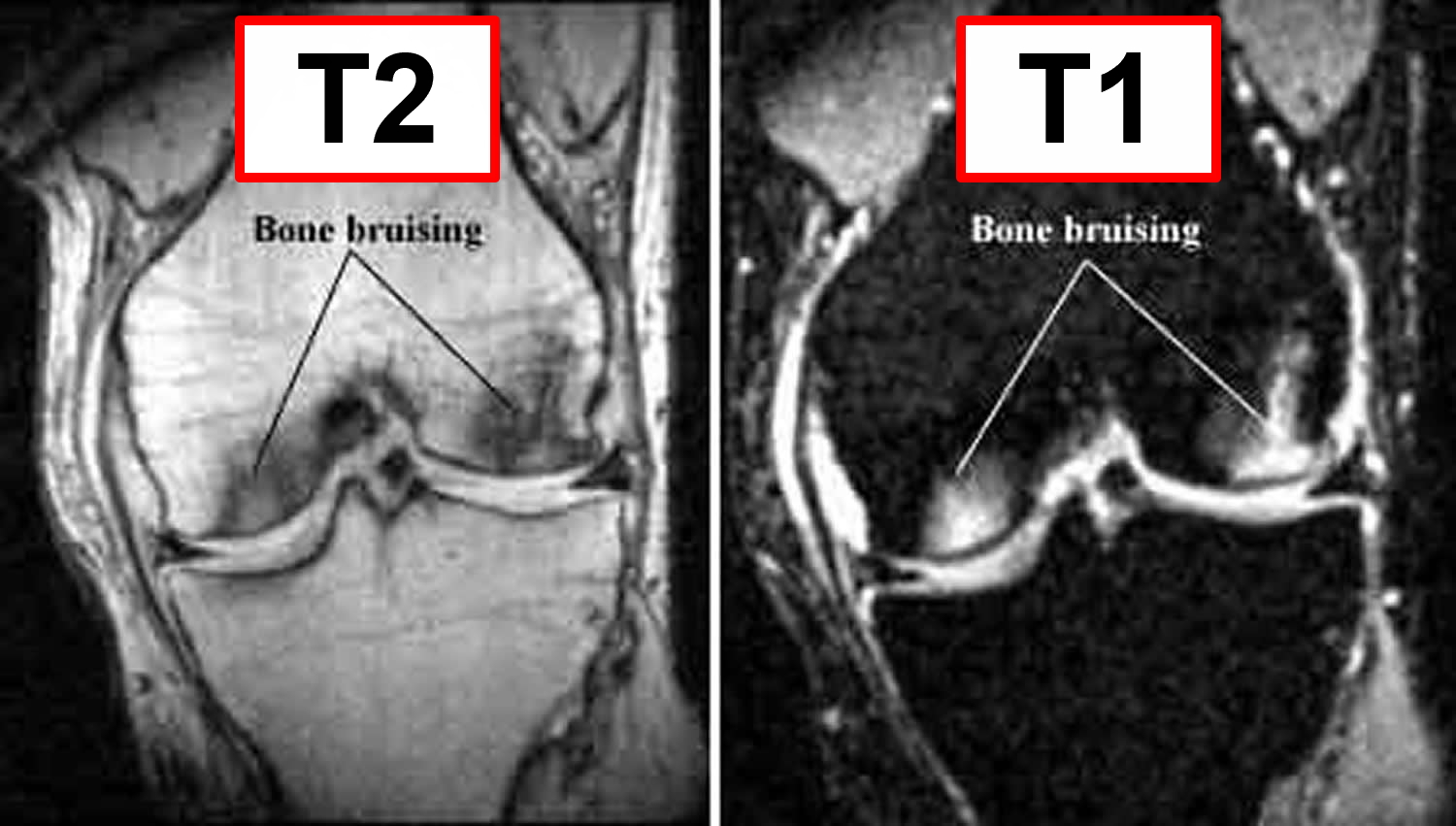
A bone bruise is best diagnosed by correlating the increased signal intensity seen on T2-weighted MR images with the decreased signal intensity seen on Tl-weighted MR images. T2-weighted MR images reflect shifts in free water (i.e. edema, hemorrhage, and inflammatory response) and therefore are useful for determining the acuity of injury. Tl-weighted MR images demonstrate fat content of the bone marrow and offer less resolution of the bone bruise, but are useful in concert with T2-weighted images in determining location and morphology of the injury (see Figure 6).
Most of what we know about the incidence and prevalence of bone bruises comes from studies involving the knee, in particular after anterior cruciate ligament (ACL) tear.
Vellet’s study 7 of 120 patients with acute traumatic haemarthroses (bleeding into the joint space) of the knee identified bone bruising in 72% of patients. Bone bruises are seen in 80% of patients with an acute ACL injury 8. The phenomenon is also seen in conjunction with meniscal, posterior cruciate and collateral ligament injuries of the knee.
Other anatomical sites where bone bruises have been identified include the hip, ankle, elbow, foot and spine. As they can only be detected on expensive MRI scanning, it is likely that most of these bone bruises go unrecognized.
Though occult bone lesions may occur wherever there is direct trauma to bone, most knee and ankle sprains are the result of forceful twisting injuries. It is thought that the bone bruises are caused as adjacent bones are forcefully impacted (‘kissing contusions’). In ACL tears, bone bruises are mainly seen in the lateral tibial plateau (outer top edge/surface of tibia) and femoral condyle (lower end of the femur), which is consistent with the valgus (outward force) mechanism of injury.
Why some people with the same injury get bone bruises and others don’t can be put down to variations in the exact mechanism and force of each individual injury.
Table 1. Differential diagnosis of bone marrow lesions
| Traumatic | Contusions (subchondral impaction) |
| Fractures (chondral, subchondral, osteochondral, insufficiency, stress) | |
| Spontaneous osteonecrosis of the knee | |
| Overuse (repetitive microtrauma) | |
| Nontraumatic | Vascular (periarticular bone infarction) |
| Osteoarthritis associated | |
| Transient bone marrow edema syndrome | |
| Osteochondritis dissecans | |
| Reactive inflammatory (polyarthritis, reactive arthritis, bacterial arthritis, osteomyelitis) | |
| Tumor / malignant marrow infiltration | Lymphoma, multiple myeloma, etc |
| Other | Postoperative, subchondral cysts, red marrow |
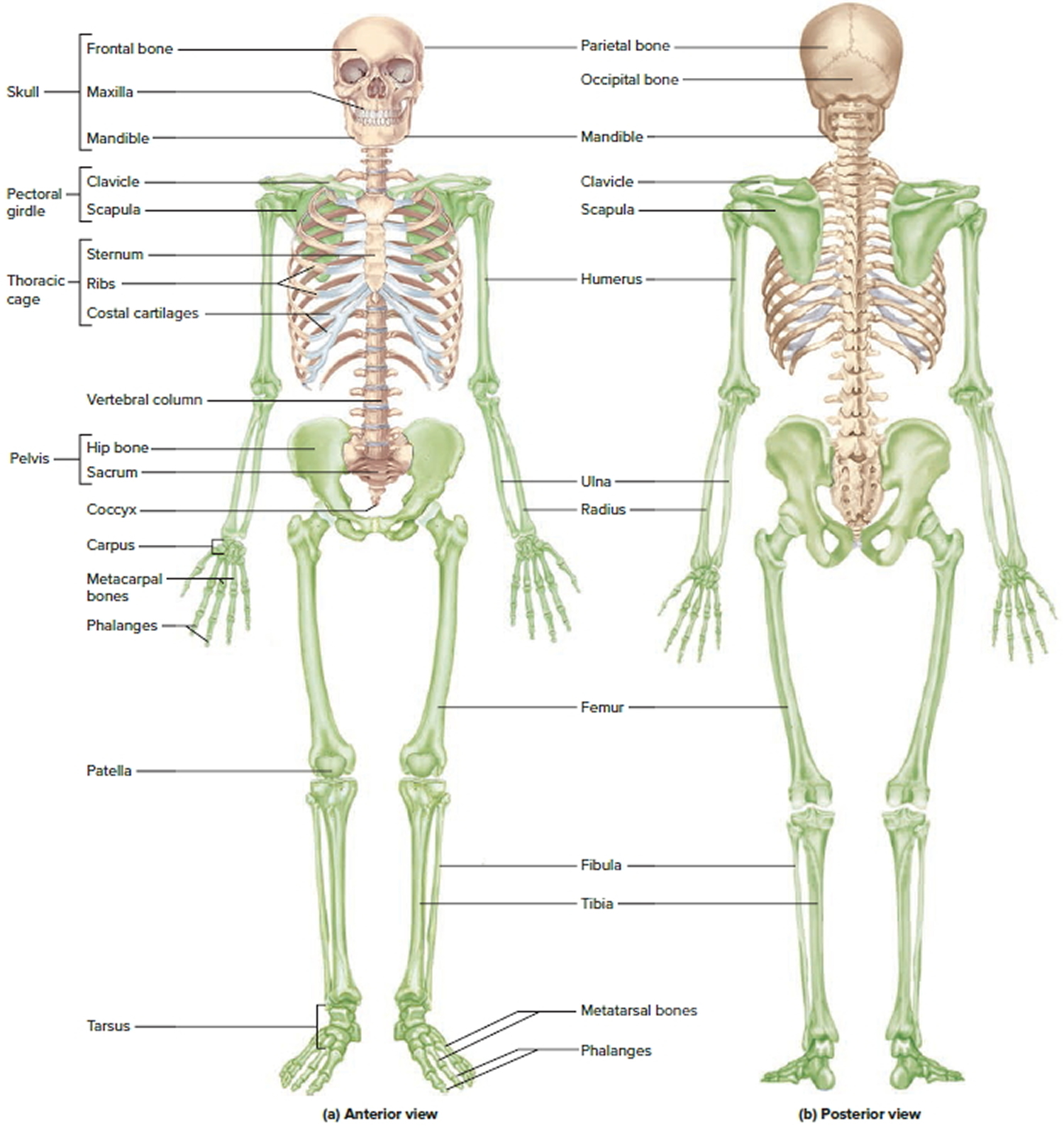
Figure 1. Human skeleton
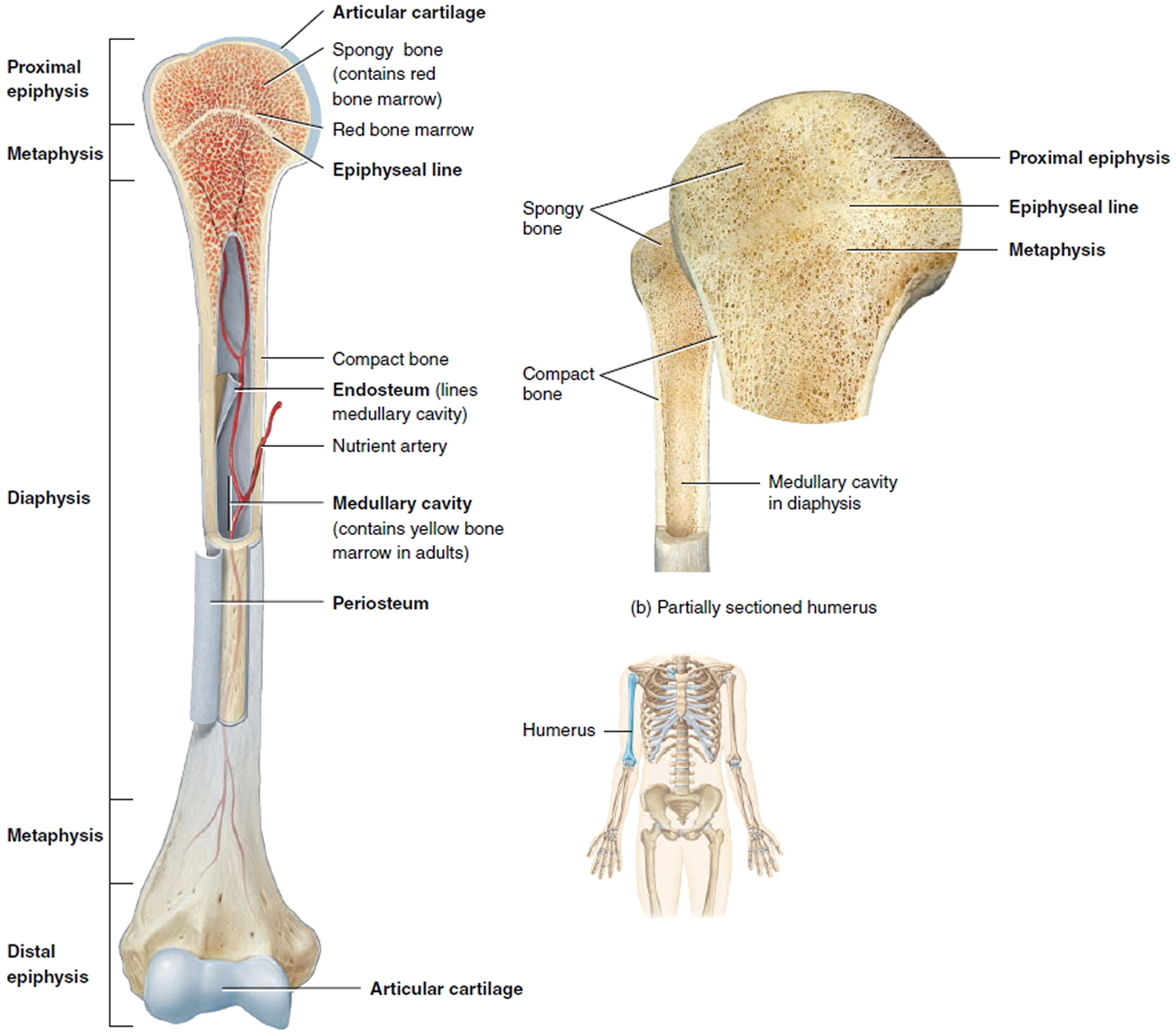
Figure 2. Parts of a long bone
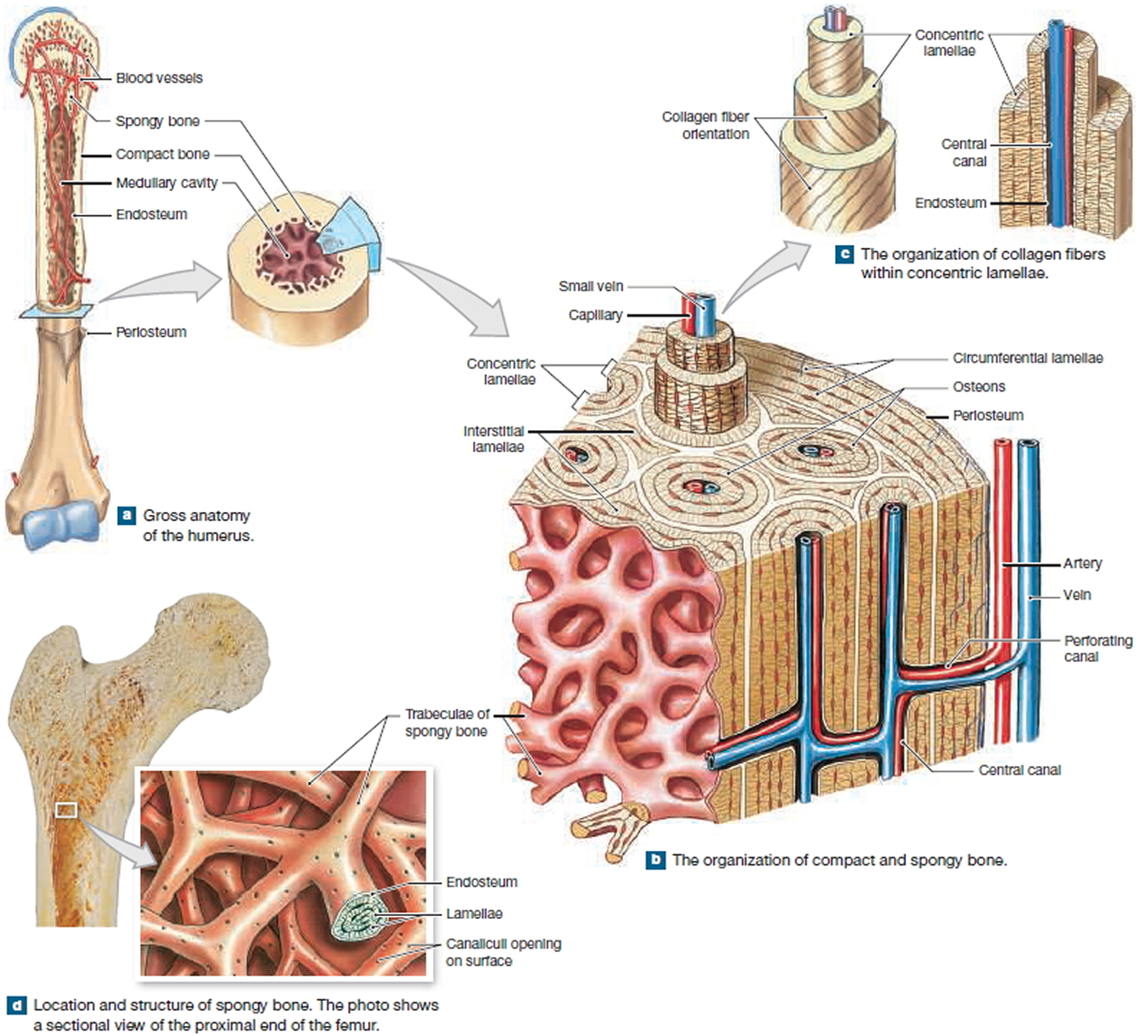
Figure 3. Internal organization of bones
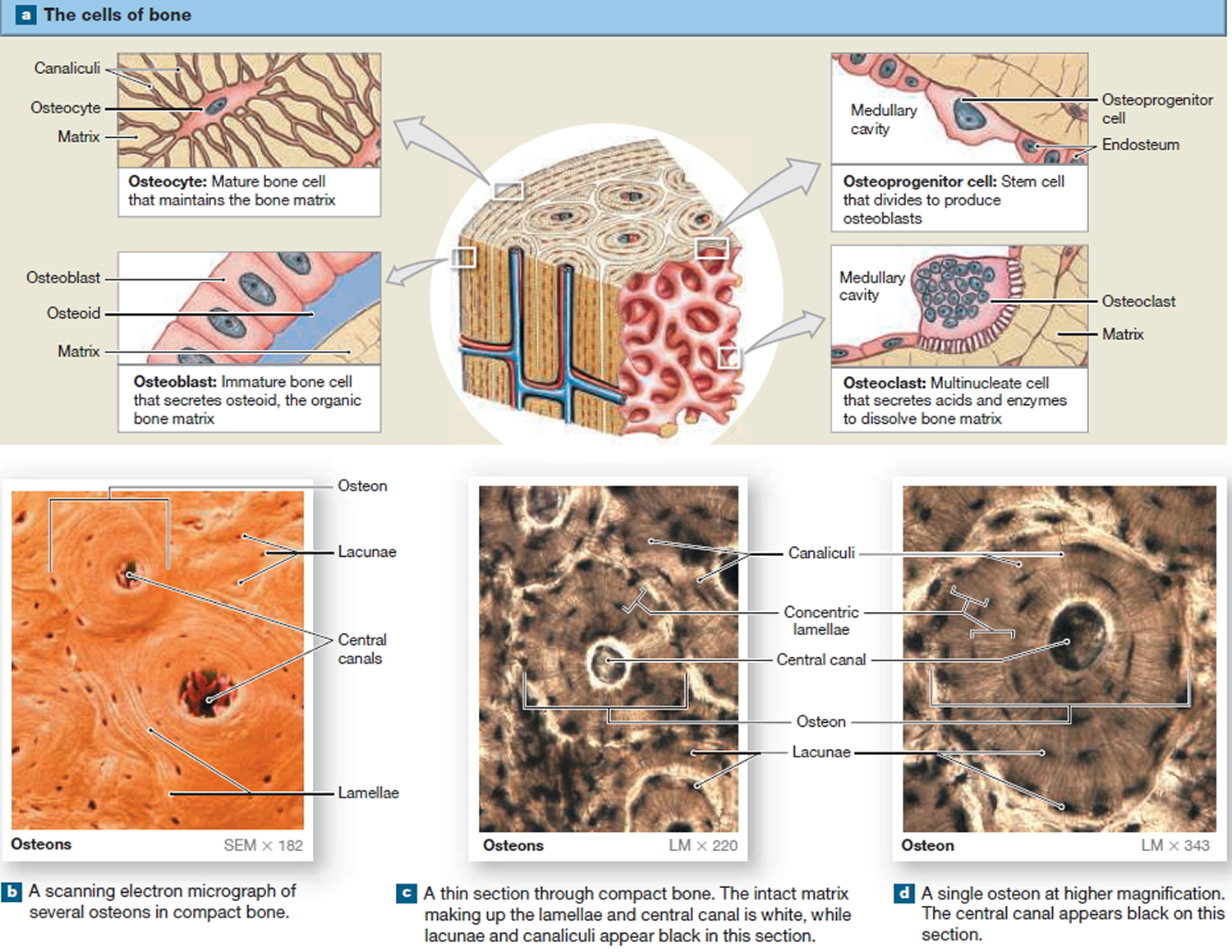
Figure 4. Microscopic Structure of a Typical Bone
Traumatic Bone Bruises
The subcortical marrow cavity consists of cancellous bone that usually demonstrates fatty marrow at all ages. The normal marrow signal on MRI parallels that of subcutaneous fat-high on conventional T1-weighted and intermediate on T2-weighted spin echo sequences. A typical bone bruise appears as an area of signal loss on T1 images and high signal intensity on T2 images, as a result of increased water content in the injured marrow. Further information can be gained with Short T1 Inversion Recovery (“STIR”) imaging when signal from normal medullary fat is markedly suppressed and bone bruises show increased intensity (Figure 6). Distinction between bone bruising and marrow oedema syndromes is primarily based on a clinical history of trauma, as the radiological features are largely indistinguishable.
Types of Bone Bruise
Several classification systems have been proposed. Most authors agree on the differentiation between reticular and geographic/demarcated pattern. Others stress the importance of the location – subchondral vs at distance of joint space. Costa-Paz et al 9 proposes the following classification:
- Type 1: Diffuse often reticular alterations of the medullary component, distant from the subjacent articular surface.
- Type 2 a and b: Localised geographic signal (mostly convex margins towards normal marrow) with contiguity to articular surface. In a type 2 lesion compared to type 1, the impact is more focally concentrated.
- Type 3: Slight disruption or depression of the normal contour of the cortical surface/subchondral lamella, often associated with type 2 lesion (small osteochondral fractures).
Another classification for the 3 different kinds of bone bruises 10, 11:
- Sub-periosteal hematoma: This is a concentrated collection of blood underneath the periosteal of the bone. It will appear mostly after a direct high-force trauma on the bone. This type is most common in the lower extremities.
- Inter-osseous bruising: This is a damage of the bone marrow. The blood supply within the bone is damaged, and this causes internal bleedings. The trigger to this type of bone bruise is a repetitive high compressive force on the bone (extreme pressure on regular base). The areas most affected are the knee and the ankle from professional athletes, such as foot-, basketball players and runners.
- Sub-chondral lesion: This type will occur beneath the cartilage layer of a joint. The main trigger is an extreme compressive force that literally crushes the cells, that results in a separation of the cartilage (or ligament) and the underlying bone, plus bleeding when the energy of the impact extends into the bone.
The other trigger is a shearing force, it sustains from a rotational mechanism such as twisting and translational forces. These will also cause that the cartilage tissue will be stripped away and exposing the underlying bone. It results in the same injury as a compressive force injury but this is another source of the injury. This type is seen more frequently in foot- and basketball players.
For the three types: an extreme compressive force can include jumping or the impact from running on hard surfaces.
Figure 5. Bone bruise knee MRI
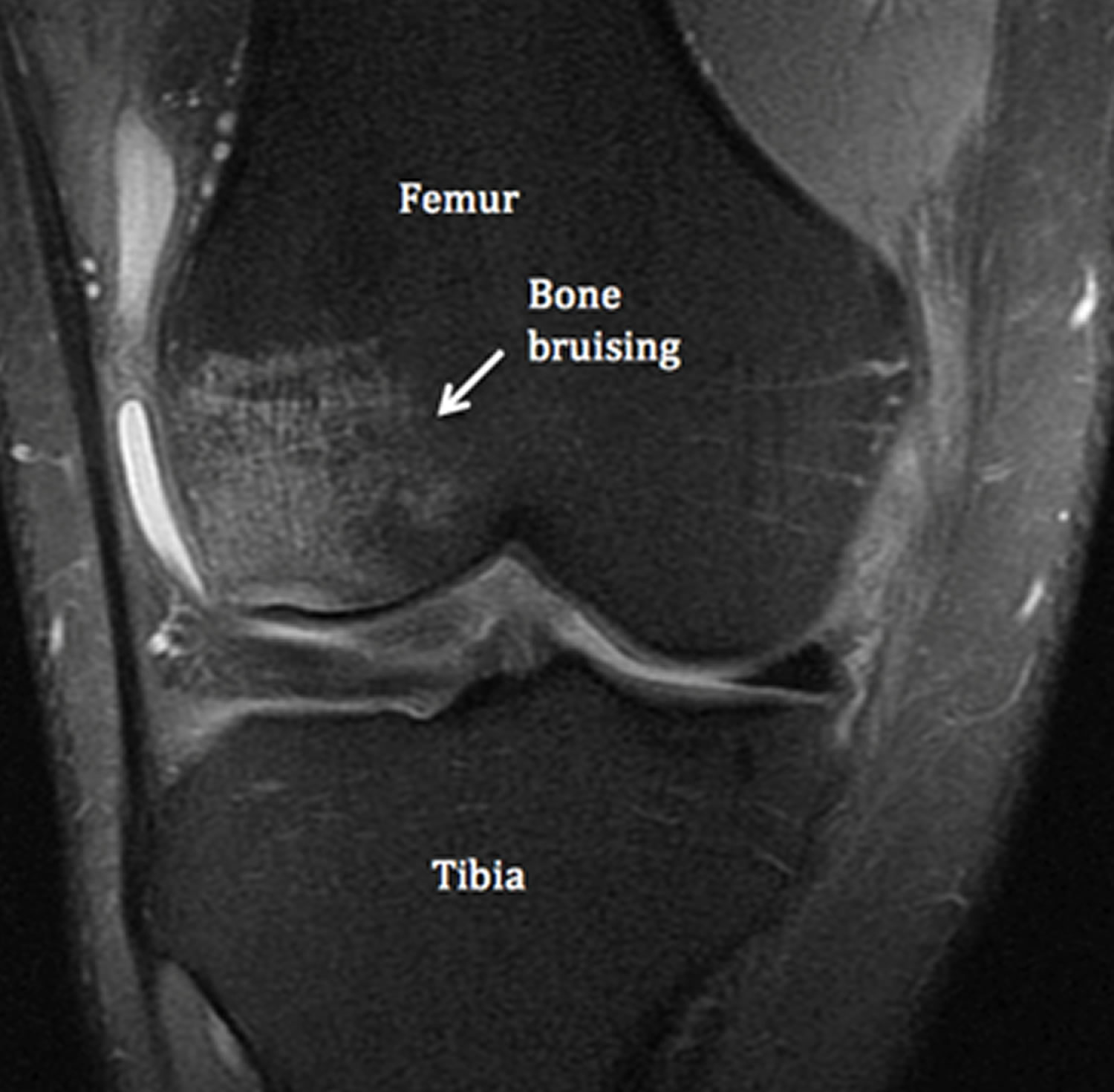
Figure 6. Knee bone bruise (T1 and T2 weighted MRI)
Bone bruise diagnosis
Difficulty arises in identifying clinical signs and symptoms directly attributable to the bone bruising, because of the spectra of associated injuries. However, patients with contusions appear to have a more protracted clinical recovery, with greater effusions and pain scores at matched time intervals and a slower return of motion 12.
Arthroscopic evidence of damage to the joint surface overlying bone bruises is not universally supported in the literature. Several authors found no arthroscopic evidence of osteochondral lesions corresponding anatomically with bone bruises in the acute phase 13, 14. Coen described normal joint appearance but “dimpling” of the cartilage over geographic femoral bruises when probed 15. Several authors describe articular lesions later at the time of anterior cruciate ligament reconstruction 16, 17. Speer found a small incidence of fissuring (6%) overlying lateral femoral condyle and posterolateral tibial plateau contusions 18. Although Spindler found 46% of patients had articular lesions, many did not correlate with bone bruise. The only significant relationship was in the lateral femoral condyle where 40% of bone bruises had an overlying lesion 17. In contrast, Johnson consistently found evidence of articular cartilage injury over femoral condyle bone bruises, varying from subtle indentation when probed, to severe fibrillation, fissuring or overt chondral fracture 19.
Some interesting histological information has arisen from biopsy at varying time periods 20. In acute lesions, Rangger found microfractures of the trabecular bone, oedema and bleeding in the fatty marrow 21. In Johnson’s series, all patients had articular cartilage and subchondral bone changes at ACL reconstruction. Chondrocytes in the superficial zone of the articular cartilage showed different stages of degeneration, and loss of matrix proteogylcan and variable osteocyte necrosis in the underlying subchondral bone was noted 19. Fang supported the evidence for proteoglycan loss and also found a 10-fold increase in matrix protein degradation products in the synovial fluid from injured compared with uninjured knees 20. This clinical data supports previous animal studies suggesting blunt trauma to articular cartilage produces profound changes in its histologic, biochemical and ultrastructural characteristics in the absence of surface disruption 22 and lends scientific evidence to the notion that bone bruising may be a precursor of posttraumatic arthritis.
Bone bruise healing time
Few studies to date address resolution of bone contusions or long-term sequelae. The reported time for the resolution of the bone bruising is variable, ranging from as early as three weeks to two years. Vellet demonstrated complete resolution of MRI bone bruises at 6-12 months but osteochondral sequelae in 67% of lateral femoral condyle lesions 14. Bretlau recently reported persistent bone bruising in 69% and 12% of patients rescanned at 4 and 12 months respectively 23. Miller, in contrast, suggested the majority of lesions resolved in 2-4 months but his study involved patients with isolated medial collateral ligament injury, the benign nature of which may have influenced the rate of recovery 24. The radiological outcome of bone bruise is that the bone bruise resolves within 2–4 months 25, 26, with a good clinical outcome, provided no other articular structures have been injured 27. A recent systematic review of the literature 28 identified 266 articles but only included 13 as quality studies. The percentage of complete healing of bone bruises on MR scanning ranged from 88 % after 11-16 months of follow-up in one study to 100 % after 5-12 months of follow-up in another. This variability may be attributed to several factors, such as the severity of injury, extent of bone bruising or other associated internal knee derangement. Further large prospective studies are required to validate those factors affecting the radiological evolution of bone bruise. The commonest finding was an overt cartilage defect (48%), but features of osteosclerosis, cartilage thinning and osteochondral defects were also seen. No articular defects occurred over associated reticular bruises in the posterolateral tibial plateau. Two year follow-up studies suggest that 10-15% of patients have persistent bone marrow edema at 2 years and up to one third have some evidence of subchrondral osteonecrosis or articular cartilage degeneration 29, 30.
Another study looked at vertebral spine bone bruise 2, suggested that bone bruises within a thoracic or lumbar vertebral body in adult patients do not cause significant vertebral wedging and so are not, in isolation, a risk factor for post-traumatic deformity.
In bone bruise, the mean interval from the onset of symptoms until complete clinical resolution ranges from 4 to 24 months, with an average of 6 months. All patients with bone bruise recover completely without intervention 31.
The natural history of bone bruise is not well known as well as whether they predispose to premature osteoarthritis.
Many studies have shown that bone bruising may have deleterious effect on the overlying articular cartilage, although this concept is not generally accepted. The pathophysiological mechanisms by which the cartilage can undergo this degenerative process may be multifactorial. The initial blunt trauma might exceed a suprephysiological threshold and lead to progressive chondral damage. Additionally, the bone lesion might heal into stiffer construction than the previous normal bone. The decrease compliance might then generates greater load in the articular cartilage, leading to a progressive cartilage degeneration.
What does a bone bruise feel like
It is very difficult to identify clinical signs and symptoms directly attributable to the underlying bone bruise, because there are usually associated soft tissue changes.
In a prospective study of 95 patients with inversion injuries of the ankle and no plain fracture on plain radiographs, Alanen et al found an incidence of bone bruises of 27 percent. Most of the bone bruises were located in the talus, typically in the medial part. The authors further found no statistical difference in the time to return to work, limitation to walking or physical activity and clinical outcome at 3 months in the two groups with and without bone bruise. This findings were in line with previous study by Zanetti et al.
In another study involving the knee joint and bone bruise, bone bruise did not have any effect on the function, symptoms and activity at 6 months, despite patients initially presenting with bone bruise at the time of MRI had a significant higher level of symptoms, functional deficit and decrease in activity.
How are Bruised Bones treated ?
A bone bruise treatment may consist of the following methods: ice therapy, elevation, medication and/or rest.
- Ice Therapy – An effective way to treat a bone bruise is to apply a pack of frozen vegetables or an ice pack to the injured bone. Ice therapy can ease the pain and reduce the inflammation in the affected area. Apply ice to the injured bone for 15 to 20 minute intervals, several times a day. Make sure to place a towel or washcloth over the affected area before you place the ice on it. Placing ice directly on the skin can cause frostbite.
- Elevation – Another beneficial bone bruise treatment is elevation. The first step when treating a bone bruise is to raise the affected area above the heart. This step prevents and/or limits fluid accumulation in the affected area, therefore reducing the risk of a bone bruise. Once the fluid has been restricted or reduced, the body is more efficiently able to repair the injured bone. It is important to elevate the affected area (injured bone) for 15 to 20 minutes, several times a day.
- Rest – One of the easiest and most effective ways to help a bone bruise heal is to rest it. Staying off the injured bone can help it heal. Walking and/or moving the damaged bone not only causes extra stress and pressure in the affected area, it also delays the healing process. In fact, excess stress and pressure can cause the blood vessels in the affected area to break, leading to blood accumulation, pain and/or bone damage. A physician may suggest the use of an assistive walking device such as: a walker, cane or crutches until the bone properly heals. Physical activity should also be limited or restricted while the bruised bone is trying to heal.
- Medication – Bone bruises can be treated with medications when natural remedies do not work. Medications like topical pain-relieving creams and gels and over-the-counter pain- relieving medications like Tylenol, Bayer and Excedrin have the ability to ease pain, reduce inflammation and aid in the healing process. Many medications, especially non-steroidal anti-inflammatory drugs, can change the way the brain interprets and responds to inflammation and pain, therefore accelerating the healing process.
Summary
As a recently recognised entity, the natural history of bone bruising is unknown 32. If bone bruise represents trabecular microfracture, as the histological evidence to date suggests, one can reasonably expect bony healing of the subchrondral lesion. Certainly, there is a consensus that most bone bruises heal in the short term. It has been proposed however, that increased stiffness of the healed bone may decrease the potential for the joint to dissipate load by deformation and this may also increase shear-stress at the bone cartilage interface, precipitating cartilage degeneration 33. However, it is also likely that the initial trauma insults the cartilage microstructure in its own right and the relative influence of the underlying bone bruise on chondral degeneration is still open to question. Further understanding of articular cartilage microstructure in the acute phase is needed, as evidence for chondral injury to date is based on macroscopic appearances and histology at the time of anterior cruciate ligament (ACL) reconstruction.
The high prevalence of bone bruises with ACL rupture has raised questions about its prognostic implications for this injured population. Posttraumatic arthritis is an established complication of nonoperative treatment of ACL rupture 34. Some radiological degeneration is seen in up to 80% of patients as early as 3 years post injury 35. McDaniel and Dameron found 37% of patients had profound degeneration at 14 years 36. Whilst associated meniscal injury was often considered the determining factor, several authors have shown similar rates of long-term degeneration, regardless of initial meniscal damage 37. Two possible explanations exist. Chronic instability may provoke secondary injury to menisci seen to be intact at the time of ACL rupture 38. Alternatively, initial damage to the articular cartilage may be the predominant prognostic factor and, if so, bone bruising may be the missing link. Sherman’s observation that knees with concomitant medial collateral ligament injury degenerated significantly earlier than those with ACL/ meniscal tear patterns could be explained by a more traumatic impact to the lateral compartment with extensive bone contusions.
- X-ray (Radiography) – Bone. https://www.radiologyinfo.org/en/info.cfm?pg=bonerad[↩]
- Teli M, de Roeck N, Horowitz MD, Saifuddin A, Green R, Noordeen H. Radiographic outcome of vertebral bone bruise associated with fracture of the thoracic and lumbar spine in adults. European Spine Journal. 2005;14(6):541-545. doi:10.1007/s00586-004-0786-1. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3489230/[↩][↩]
- Transient osteoporosis: transient bone marrow edema? Wilson AJ, Murphy WA, Hardy DC, Totty WG. Radiology. 1988 Jun; 167(3):757-60. http://pubs.rsna.org/doi/pdf/10.1148/radiology.167.3.3363136[↩]
- Occult cartilage and bone injuries of the knee: detection, classification, and assessment with MR imaging. Mink JH, Deutsch AL. Radiology. 1989 Mar; 170(3 Pt 1):823-9. https://www.ncbi.nlm.nih.gov/pubmed/2916038/[↩]
- DeAngelis JP, Spindler KP. Traumatic Bone Bruises in the Athlete’s Knee. Sports Health. 2010;2(5):398-402. doi:10.1177/1941738110377745. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3445054/[↩]
- MRI-detected subchondral bone marrow signal alterations of the knee joint: terminology, imaging appearance, relevance and radiological differential diagnosis. Roemer FW, Frobell R, Hunter DJ, Crema MD, Fischer W, Bohndorf K, Guermazi A. Osteoarthritis Cartilage. 2009 Sep; 17(9):1115-31. https://www.ncbi.nlm.nih.gov/pubmed/19358902/[↩][↩]
- Vellet AD, Marks PH, Fowler PJ, Munro TG (1991) ‘Occult posttraumatic osteochondral lesions of the knee: Prevalence, classification and short-term sequelae evaluated with MR imaging’ Radiology178: 271-276.[↩]
- Spindler KP, Schils JP, Bergfeld JA et al (1993) ‘Prospective study of osseous, articular and meniscal lesions in recent anterior cruciate ligament tears by magnetic resonance imaging and arthroscopy’ Am J Sports Med 21(4): 551-6.[↩]
- Costa-Paz M, Musculo DL, Ayerza M et al (2001) ‘Magnetic resonance imaging follow-up study of bone bruises associated with anterior cruciate ligament ruptures’ Arthroscopy17: 445-449.[↩]
- V. Mandalia, J.H.L. Henson. Traumatic bone bruising – A review article, European Journal of Radiology 2008; 67; 54–61[↩]
- V. Mandalia, A.J.B. Fogg, R. Chari, J. Murray, A. Beale, J.H.L. Henson. Bone bruising of the knee. Clinical Radiology 2005; 60, 627–636.[↩]
- Johnson DL, Bealle DP, Brand JC et al. The effect of a geographic lateral bone bruise on knee inflammation after acute anterior cruciate ligament rupture. Am J Sports Med. 2000 Mar;28(2):152-5.[↩]
- Graf BK, Cook DA, De Smet AA et al. “Bone bruises” on magnetic resonance imaging evaluation of anterior cruciate ligament injuries. Am J Sports Med 1993; 21(2):220-3.[↩]
- Vellet AD, Marks PH, Fowler PJ et al. Occult post-traumatic osteochondral lesions: Prevalence, classification and short-term sequelae evaluated with MR imaging. Radiology 1991; 178:271-6.[↩][↩]
- Coen MJ, Caborn DN, Johnson DL. The Dimpling phenomenon: articular cartilage injury overlying an occult osteochondral lesion at the time of anterior cruciate ligament reconstruction. Arthroscopy 1996 Aug;12(4):502-5.[↩]
- Speer KP, Warren RF, Wickiewicz TL et al. Observations on the injury mechanism of anterior cruciate ligament tears in skiers. Am J Sports Med 1995; 23(1):77-81.[↩]
- Spindler KP, Schils JP, Bergfeld JA et al. Prospective study of osseous, articular and meniscal lesions in recent anterior cruciate ligament tears by magnetic resonance imaging and arthroscopy. Am J Sports Med 1993; 21(4):551-6.[↩][↩]
- Speer KP, Warren RF, Wickiewicz TL et al. Observations on the injury mechanism of anterior cruciate ligament (ACL) tears in skiers. Am J Sports Med 1995; 23(1):77-81.[↩]
- Johnson D, Urban WP, Caborn DNM et al. Articular cartilage changes seen with magnetic resonance imaging detected bone bruises associated with acute anterior cruciate ligament rupture. Am J Sports Med 1998; 26(3):409-14.[↩][↩]
- Fang C, Johnson D, Leslie MP et al. Tissue distribution and measurement of cartilage oligomeric matrix protein in patients with magnetic resonance imaging-detected bone bruises after acute anterior cruciate ligament tears. J Orthop Res. 2001 Jul;19(4):634-41.[↩][↩]
- Rangger C, Kathrein A, Freund MC et al. Bone bruise of the knee: histology and cryosections in 5 cases. Acta Orthop Scand. 1998 Jun;69(3):291-4.[↩]
- Thompson RC Jr, Oegema TR Jr, Lewis JL et al. Osteoarthritic changes after acute transarticular load. J Bone Joint Surg 1991; 73A:990-1001.[↩]
- Bretlau T, Tuxoe J, Larsen L et al. Bone bruise in the acutely injured knee. Knee Surg Sports Traumatol Arthrosc. 2002 Mar;10(2):96-101.[↩]
- Miller MD, Osborne JR, Gordon WT et al. The Natural History of bone bruises. A prospective study of magnetic resonance imaging-detected trabecular microfractures in patients with isolated medial collateral ligament injuries. Am J Sports Med 1998; 26(1):15-19.[↩]
- Magnetic resonance imaging of the musculoskeletal system. Part 4. The knee. Crotty JM, Monu JU, Pope TL Jr. Clin Orthop Relat Res. 1996 Sep; (330):288-303. https://www.ncbi.nlm.nih.gov/pubmed/8804304/[↩]
- The natural history of bone bruises. A prospective study of magnetic resonance imaging-detected trabecular microfractures in patients with isolated medial collateral ligament injuries. Miller MD, Osborne JR, Gordon WT, Hinkin DT, Brinker MR. Am J Sports Med. 1998 Jan-Feb; 26(1):15-9. https://www.ncbi.nlm.nih.gov/pubmed/9474396/[↩]
- Clinical outcome of isolated subcortical trabecular fractures (bone bruise) detected on magnetic resonance imaging in knees. Wright RW, Phaneuf MA, Limbird TJ, Spindler KP. Am J Sports Med. 2000 Sep-Oct; 28(5):663-7. https://www.ncbi.nlm.nih.gov/pubmed/11032221/[↩]
- Boks SS, Vroegindeweij D, Koes BW, Hunink MG, Bierma-Zienstra SM (2006) ‘Follow-up of occult bone lesions detected at MR imaging: Systematic Review’ Radiology238: 853-62.[↩]
- Costa-Paz M, Muscolo DL, Ayerza M et al. Magnetic resonance imaging follow-up study of bone bruises associated with anterior cruciate ligament ruptures. Arthroscopy. 2001 May;17(5):445-9.[↩]
- Roemer FW, Bohndorf K. Long-term osseous sequelae after acute trauma of the knee joint evaluated by MRI. Skeletal Radiol 2002, 31:615-623.[↩]
- Clinical outcome of isolated subcortical trabecular fractures (bone bruise) detected on magnetic resonance imaging in knees. Am J Sports Med. 2000 Sep-Oct;28(5):663-7. https://www.ncbi.nlm.nih.gov/pubmed/11032221/[↩]
- Bone Bruising and Bone Marrow Edema Syndromes: Incidental Radiological Findings or Harbingers of Future Joint Degeneration? https://www.isakos.com/innovations/niall[↩]
- Radin EL, Rose RM. Role of subchrondral bone in the initiation and progression of cartilage damage. Clin Orthop 1986;213:34-40.[↩]
- Sherman MF, Warren RF, Marshall JL. A Clinical and radiographical analysis of 127 anterior cruciate insufficient knees. Clin Orthop 1988; 227:229-37.[↩]
- Kannus P, Jarvinen M. Conservatively treated tears of the anterior cruciate ligament-long-term results. J Bone Joint Surg 1987; 69A(7):1007-12.[↩]
- McDaniel WJ, Dameron TB. The untreated anterior cruciate ligament rupture. Clin Orthop 1983;(172):158-63.[↩]
- Noyes FR, Mooar PA, Matthews DS et al. The symptomatic anterior cruciate deficient knee-Part 1: The long-term functional disability in athletically active individuals. J Bone Joint Surg 1983; 65A(2):154-162.[↩]
- Hirshman HP, Daniel DM, Miyasaka K. The fate of unoperated knee ligament injuries. Knee ligaments: structure, function,injury and repair (eds). New York. Raven Press 1990; 481-503.[↩]