Contents
What is a brain aneurysm
A brain aneurysm also referred to as a cerebral aneurysm or intracranial aneurysm, is an abnormal bulge or “ballooning” in the wall of an artery in the brain. They are sometimes called berry aneurysms because they are often the size of a small berry. Most brain aneurysms produce no symptoms until they become large, begin to leak blood, or burst.
The blood flow within the artery in the brain pounds against the thinned portion of the artery wall and aneurysms form silently from wear and tear on the arteries. As the artery wall becomes gradually thinner from the dilation, the blood flow causes the weakened wall to swell outward. This pressure may cause the aneurysm to rupture and allow blood to escape into the space around the brain. A ruptured brain aneurysm commonly requires advanced surgical treatment.
If a brain aneurysm presses on nerves in your brain, it can cause signs and symptoms. These can include:
- A droopy eyelid
- Double vision or other changes in vision
- Pain above or behind the eye
- A dilated pupil
- Numbness or weakness on one side of the face or body
Treatment depends on the size and location of the aneurysm, whether it is infected, and whether it has burst. If a brain aneurysm bursts, symptoms can include a sudden, severe headache, nausea and vomiting, stiff neck, loss of consciousness, and signs of a stroke. Any of these symptoms requires immediate medical attention.
Seek Medical Attention Immediately If You Are Experiencing Some Or All Of These Symptoms:
- Sudden severe headache, the worst headache of your life
- Loss of consciousness
- Nausea/Vomiting
- Stiff Neck
- Sudden blurred or double vision
- Sudden pain above/behind the eye or difficulty seeing
- Sudden change in mental status/awareness
- Sudden trouble walking or dizziness
- Sudden weakness and numbness
- Sensitivity to light (photophobia)
- Seizure
- Drooping eyelid
If you begin to experience symptoms of a ruptured aneurysm, call your local emergency number for an ambulance. Do NOT allow a family member to take you in a private vehicle to the hospital. This is a high stress situation that may require the paramedics to execute lifesaving procedures in the emergency vehicle. Patients who present to the emergency room with the “worst headache of their life” need a thorough neurologic evaluation which includes diagnostic testing such as a CT scan and/or lumbar puncture to evaluate for subarachnoid hemorrhage (SAH).
Brain Aneurysm Statistics and Facts 1
- An estimated 6 million people in the United States have an unruptured brain aneurysm or 1 in 50 people.
- The annual rate of rupture is approximately 8 – 10 per 100,000 people or about 30,000 people in the United States suffer a brain aneurysm rupture. There is a brain aneurysm rupturing every 18 minutes. Ruptured brain aneurysms are fatal in about 40% of cases. Of those who survive, about 66% suffer some permanent neurological deficit.
- Approximately 15% of patients with aneurysmal subarachnoid hemorrhage (SAH) die before reaching the hospital. Most of the deaths from subarachnoid hemorrhage are due to rapid and massive brain injury from the initial bleeding which is not correctable by medical and surgical interventions.
- 4 out of 7 people who recover from a ruptured brain aneurysm will have disabilities.
- Brain aneurysms are most prevalent in people ages 35 – 60, but can occur in children as well. The median age when aneurysmal hemorrhagic stroke occurs is 50 years old and there are typically no warning signs. Most aneurysms develop after the age of 40.
- Most aneurysms are small, about 1/8 inch to nearly one inch, and an estimated 50 to 80 percent of all aneurysms do not rupture during the course of a person’s lifetime. Aneurysms larger than one inch are referred to as “giant” aneurysms and can pose a particularly high risk and can be difficult to treat.
- Women, more than men, suffer from brain aneurysms at a ratio of 3:2.
- African-Americans at twice the rate of rupture of whites (a 2.1:1 ratio)
- Hispanics at nearly twice the rate of rupture of whites (a 1.67:1 ratio)
- Ruptured brain aneurysms account for 3 – 5% of all new strokes.
- Subarachnoid hemorrhage (SAH) is one of the most feared causes of acute headache upon presentation to the emergency department. Headache accounts for 1 – 2% of the emergency room visits and up to 4% of visits to the primary care offices. Among all the patients who present to the emergency room with headaches, approximately 1% has subarachnoid hemorrhage. One study put the figure at 4%.
- Accurate early diagnosis is critical, as the initial hemorrhage may be fatal, may result in devastating neurologic outcomes, or may produce minor symptoms. Despite widespread neuroimaging availability, misdiagnosis or delays in diagnosis occurs in up to 25% of patients with subarachnoid hemorrhage (SAH) when initially presenting for medical treatment. Failure to do a scan results in 73% of these misdiagnoses. This makes SAH a low-frequency, high-risk disease.
- There are almost 500,000 deaths worldwide each year caused by brain aneurysms and half the victims are younger than 50.
- The cost of a brain aneurysm treated by clipping via open brain surgery more than doubles in cost after the aneurysm has ruptured. The cost of a brain aneurysm treated by coiling, which is less invasive and is done through a catheter, increases by about 70% after the aneurysm has ruptured.
- 10 – 15% of patients diagnosed with a brain aneurysm will harbor more than one aneurysm.
High Risk Groups For Aneurysm
Certain people have a higher risk for brain aneurysms than the general population. These include individuals with a strong family history (more than one first degree relative with brain aneurysm), or a history of certain conditions such as polycystic kidney disease or collagen vascular diseases. If you are such an individual, screening for brain aneurysms using imaging such as magnetic resonance angiogram, may be advisable. You should discuss this with your primary care physician.
What are the two types of brain aneurysms?
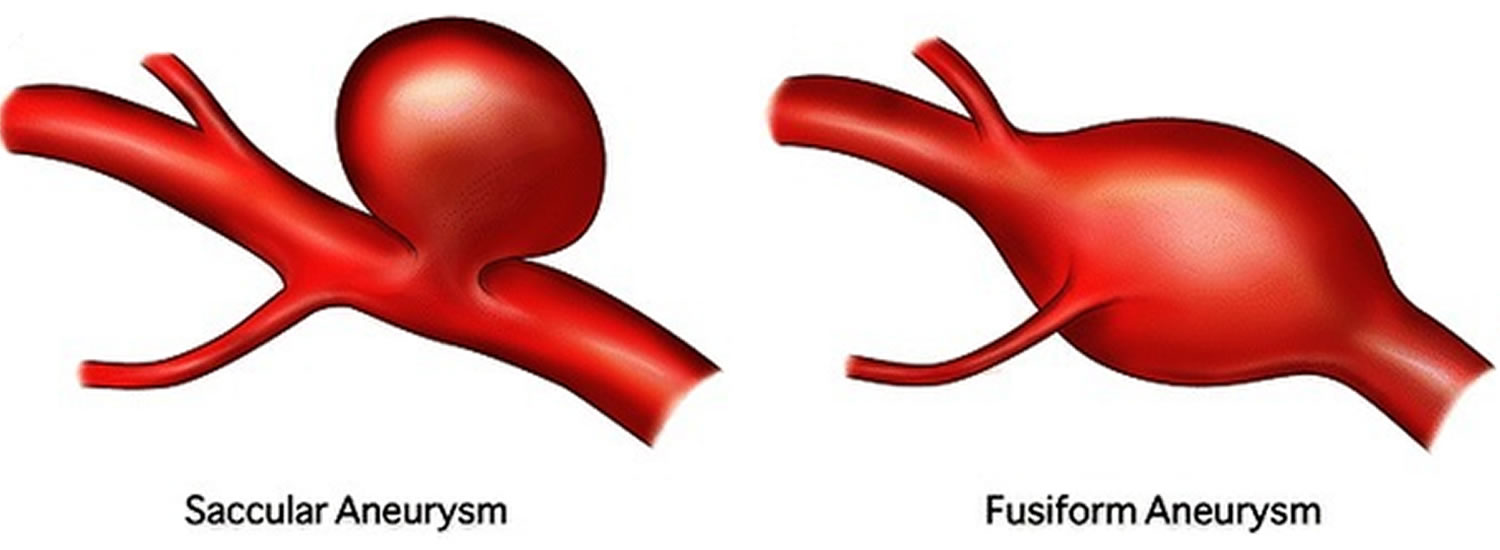
Saccular Aneurysm
A saccular aneurysm is the most common type of aneurysm and account for 80% to 90% of all brain aneurysms and are the most common cause of non-traumatic subarachnoid hemorrhage (SAH). It is also known as a “berry” aneurysm because of its shape. The berry aneurysm looks like a sac or berry forming at the bifuraction or the “Y” segment of arteries. It has a neck and stem. These small, berry-like projections occur at arterial bifurcations and branches of the large arteries at the base of the brain, known as the Circle of Willis.
Fusiform aneurysm
The fusiform aneurysm is a less common type of aneurysm. It looks like an outpouching of an arterial wall on both sides of the artery or like a blood vessel that is expanded in all directions. The fusiform aneurysm does not have a stem and it seldom ruptures.
Figure 1. Types of brain aneurysm
Brain Blood Supply
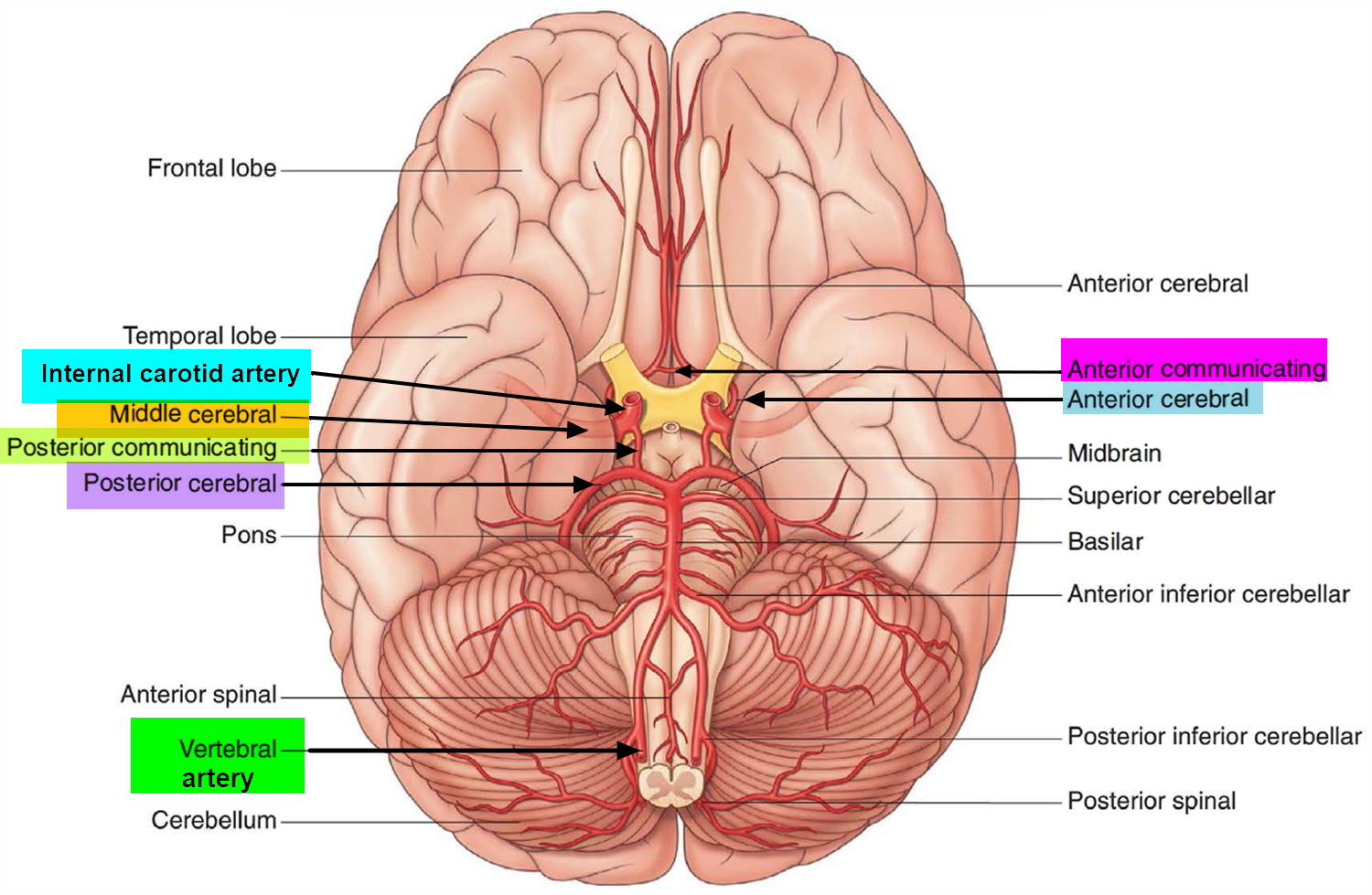
To understand aneurysms, it is helpful to understand the circulatory system of the brain. The heart pumps oxygen- and nutrient-laden blood to the brain, face, and scalp via two major sets of vessels: the internal carotid arteries and the vertebral arteries. The jugular and other veins bring blood out of the brain.
The carotid arteries run along the front of the neck – one on the left and one on the right. They are what you feel when you take your pulse just under your jaw. The carotid arteries split into external and internal arteries near the top of the neck.
The external carotid arteries supply blood to the face and scalp. The internal carotid arteries supply blood to the front (anterior) three-fifths of cerebrum, except for parts of the temporal and occipital lobes.
The vertebral arteries travel along the spinal column and cannot be felt from the outside. They join to form a single basilar artery (hence the name vertebrobasilar arteries) near the brain stem at the base of the skull. The arteries supply blood to the posterior two-fifths of the cerebrum, part of the cerebellum, and the brain stem.
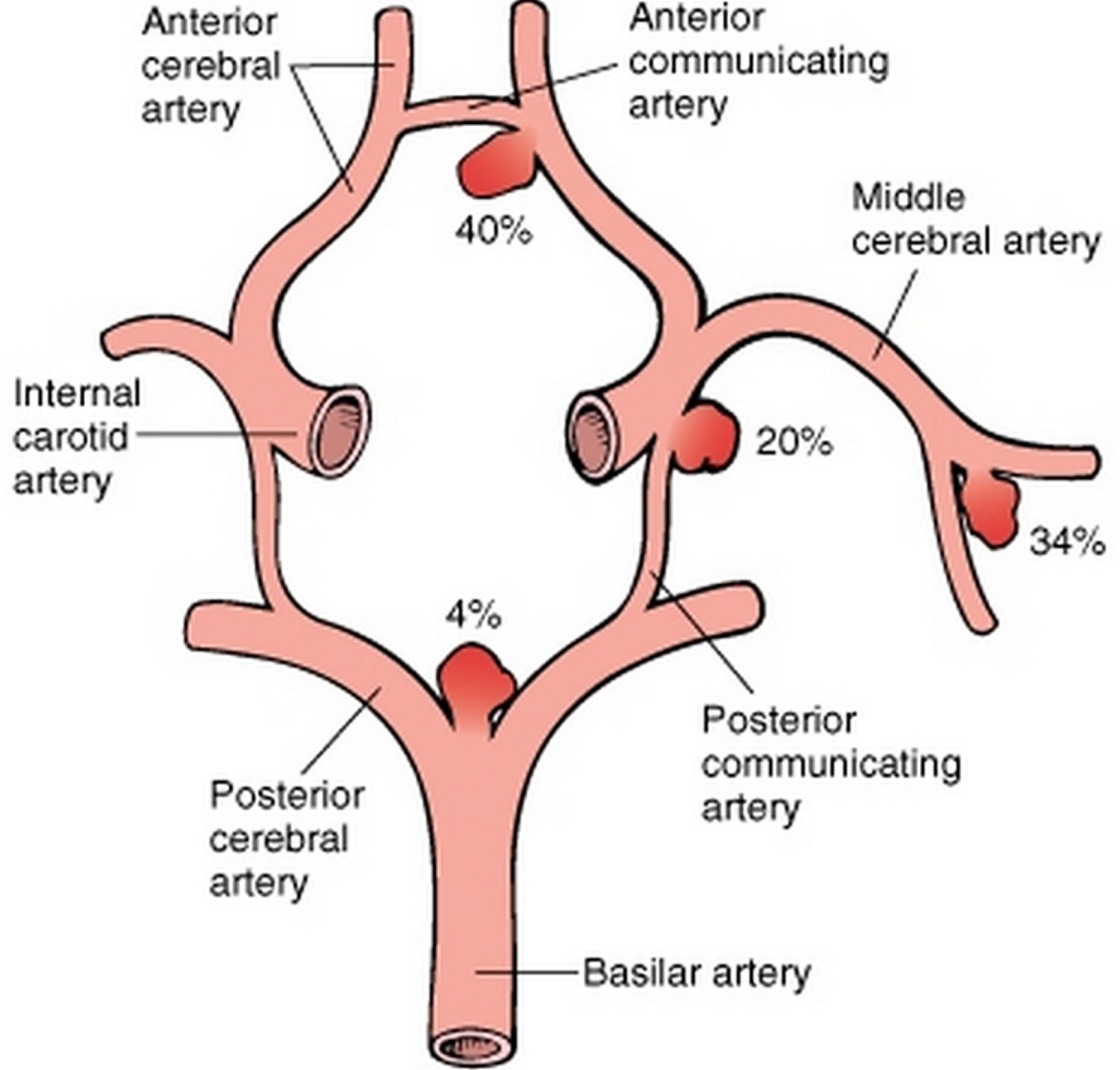
Because the brain relies on only two sets of major arteries for its blood supply, it is very important that these arteries are healthy. These arteries that conduct blood to the brain — the internal-carotid and vertebral arteries — connect through the Circle of Willis, which loops around the brainstem at the base of the brain (see Figure 2). From this circle, other arteries — the anterior cerebral artery, the middle cerebral artery and the posterior cerebral artery — arise and travel to all parts of the brain. Brain aneurysms tend to occur at the junctions between the arteries that make up the Circle of Willis.
Figure 2. Brain blood supply (bottom view)
Figure 3. Saccular aneurysms involving the Circle of Willis
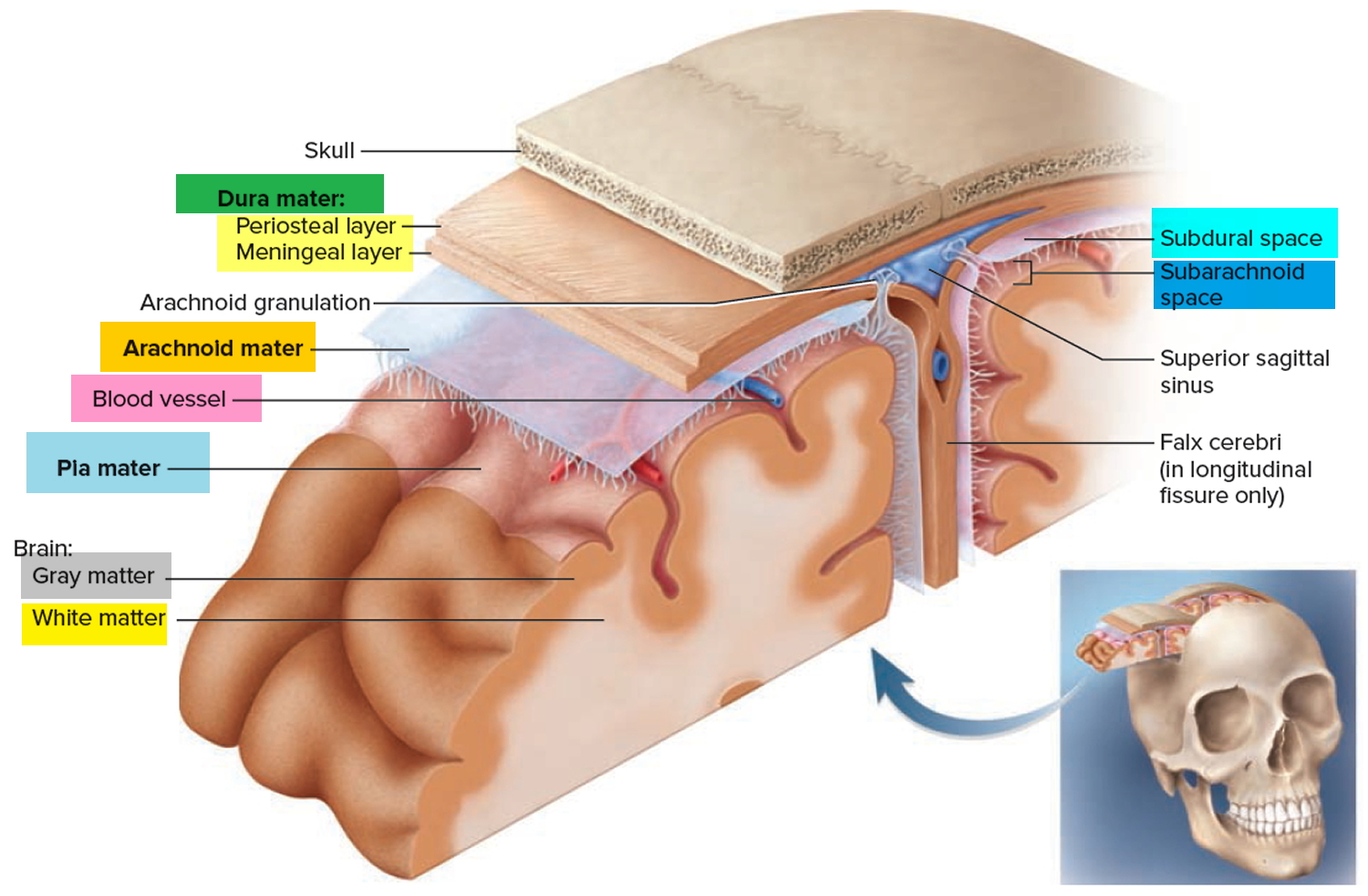
Figure 4. Meninges of the brain
Brain aneurysm warning signs
Unruptured brain aneurysms are typically completely asymptomatic 2. These brain aneurysms are typically small in size, usually less than one half inch in diameter. However, large unruptured aneurysms can occasionally press on the brain or the nerves stemming out of the brain and may result in various neurological symptoms. Any individual experiencing some or all of the following symptoms, regardless of age, should undergo immediate and careful evaluation by a physician.
- Localized Headache
- Dilated pupils
- Blurred or double vision
- Pain above and behind eye
- Weakness and numbness
- Difficulty speaking
Ruptured brain aneurysms usually result in a subarachnoid hemorrhage (SAH), which is defined as bleeding into the subarachnoid space. When blood escapes into the space around the brain, it can cause sudden symptoms.
Discovery Of An Unruptured Aneurysm
Brain aneurysms are discovered more and more frequently in people undergoing brain imaging, such as magnetic resonance imaging (MRI), for other reasons, such as evaluation of headaches, after head trauma or in work-up of other neurological symptoms. Often in these situations, the aneurysm itself is an incidental finding, unrelated to the symptoms that prompted the imaging. However, once diagnosed, it is important to consult with a neurosurgeon to review the findings, determine if further imaging is needed, and discuss management options.
These options can include:
- Observation with follow-up imaging at future intervals to monitor for any change or growth of the aneurysm
- Aneurysm treatment with either surgical clipping or endovascular coiling.
- It is important to discuss and explore these options with a physician specializing in aneurysm management.
Brain aneurysm complications
When a brain aneurysm ruptures, the bleeding usually lasts only a few seconds. The blood can cause direct damage to surrounding cells, and the bleeding can damage or kill other cells. It also increases pressure inside the skull.
If the pressure becomes too elevated, the blood and oxygen supply to the brain may be disrupted to the point that loss of consciousness or even death may occur.
Complications that can develop after the rupture of an aneurysm include:
Rebleeding
- Of the 18,000 persons who survive the initial rupture of an aneurysm annually, 3,000 either die or are disabled from rebleeding.
- Some believe the incidence of rebleeding is as high as 30%.
- The highest incidence occurs in the first 2 weeks after initial hemorrhage.
- Peaks in the incidence of rebleeding occur in the first 24 to 28 hours and at 7 to 10 days.
- Rebleeding within the first 24 to 48 hours is the leading cause of death in persons surviving the initial bleed.
- Approximately 70% of patients who rebleed will die.
The onset of rebleeding is usually accompanied by sudden severe headache, often associated with severe nausea and vomiting; a decrease in or loss of consciousness; and new neurological deficits. Death may occur. Rebleeding can be confirmed by a CT scan or a sudden spike in intracranial pressure with new blood seen in the bag if a ventricular drain is in place. Early treatment, with either surgical or endovascular methods, of the aneurysm is the most effective means of preventing rebleeding.
Cerebral Vasospasm
- Of the 18,000 persons annually who survive initial aneurysmal rupture, 3,000 either die or are disabled from cerebral vasospasm.
- Vasospasm occurs in approximately 30% of patients.
- By definition, cerebral vasospasm is narrowing of a cerebral blood vessel and causes reduced blood flow distally, which may lead to delayed ischemic deficit and cerebral infarction if left untreated.
- Besides the damage done by the initial subarachnoid hemorrhage (SAH), brain damage produced by vasospasm is an important cause of morbidity and mortality after hemorrhage, with 14% to 36% of patients suffering disability and death.
- Since improved treatment of aneurysmal subarachnoid hemorrhage has occurred with early and improved microsurgery, new endovascular techniques and better post operative care and monitoring, vasospasm has significantly decreased as the cause of death over the last ten years (from 35% in the seventies to less than 10% at this time).
- The present rescue therapies, which include ‘triple H therapy’ HHH, (hypertension/hypervolemia/ hemodilution), interventional procedures such as balloon angioplasty, intra-arterial nicardipine and other vasodilators, are associated with significant morbidity, and are labor intensive and expensive.120 A drug that would prevent delayed ischemic effects and minimize the amount of rescue therapy and optimize late outcome is desirable. When the patient’s condition deteriorates 3 to 14 days after SAH, vasospasm should be considered as the possible cause. A CT scan should be performed immediately to rule out hydrocephalus, infarction, or rebleeding.
- Vasospasm can decrease cerebral perfusion to an area, causing ischemia and perhaps infarction, and can lead to further deterioration of neurological function.
- Vasospasm may be differentiated as either angiographic or symptomatic.
- Angiographic vasospasm refers to narrowing of a cerebral arterial territory, as noted on angiography, without clinical symptoms.
- Symptomatic vasospasm is the clinical syndrome of delayed cerebral ischemia associated with angiographically documented narrowing of a major cerebral arterial territory and transcranial doppler elevation of a specific arterial territory.
- Vasospasm develops 3 to 14 days after SAH (peaking at 7 to 10 days), although the onset may be delayed up to 21 days.
Hydrocephalus
Hydrocephalus is a condition in which there is either an obstruction to the flow of cerebrospinal fluid (CSF) within the ventricular system or subarachnoid space (noncommunicating hydrocephalus) either due to intraventricular mass lesions or to external compression or a problem with reabsorption of cerebrospinal fluid (communicating hydrocephalus). The type of hydrocephalus that occurs with SAH is communicating hydrocephalus. Hydrocephalus can be classified as acute, subacute, or delayed. The profiles for each are different and are briefly discussed here. With SAH, hydrocephalus develops as a result of blood in the cerebrospinal fluid, which plugs the arachnoid villi, thus interfering with the reabsorption of cerebrospinal fluid. Diagnosis is established on the basis of CT findings, which will reveal dilated ventricles with blood within the ventricles.
The following summarizes the signs and symptoms of the three types of hydrocephalus, as well as the appropriate treatment for each.
Acute hydrocephalus
- Occurs within the first 24 hours after hemorrhage
- Occurs in up to 20% to 67% of affected patients within 3 days following SAH
- Associated with intraventricular hemorrhage or excessive blood in the basal cisterns of posterior fossa
- Characterized by the abrupt onset of stupor or persistence of coma
- Management: immediate ventriculostomy to drain the CSF periodically, especially when intracranial pressure is elevated above a predetermined level such as 20 mm Hg
Subacute hydrocephalus
- Occurs within the first few days to 7 days after hemorrhage
- Associated with blood in the CSF secondary to SAH
- Characterized by drowsiness, the onset of which is gradual, although an abrupt onset is possible
- Management: ventriculostomy, or serial lumbar puncture or lumbar drainage of CSF
Delayed hydrocephalus
- Occurs 10 or more days after hemorrhage
- Associated with blood in the CSF secondary to SAH
- Characterized by a gradual onset of symptoms when the patient is recovering from surgery; symptoms include gait difficulty, behavioral changes (dull, quiet, and blunted animation)
- Management: surgical placement of a ventriculoperitoneal shunt
Because signs and symptoms of hydrocephalus are nonspecific, changes in responsiveness may be attributed to other problems, thus delaying appropriate treatment.
Seizures
The frequency of seizures following SAH is not known with certainty. In the early period, seizures occur between 16% and 90%. Risk factors for seizures in the early period after SAH include previous history of hypertension, CT-documented presence of focal intraparenchymal blood, occurrence of a cerebral infarction, middle cerebral aneurysm location, and duration of coma after SAH. Seizures generally occur within 18 months (if they do occur) and may be generalized, focal, or complex. On the basis of available data, many treat all aneurysmal SAH patients with anticonvulsants. If the hemorrhage is mild, anticonvulsants are tapered after 1 month. If the hemorrhage is more severe and if intraparenchymal brain injury has occurred, extended therapy and EEG monitoring are employed.
Brain aneurysm causes
The causes of brain aneurysm are unknown, but a range of factors may increase your risk.
Risk Factors for brain aneurysm
Risk factors that doctors and researchers believe contribute to the formation of brain aneurysms:
- Smoking
- High blood pressure or hypertension
- Heavy alcohol consumption
- Congenital resulting from inborn abnormality in artery wall
- Family history of brain aneurysms
- Age over 40
- Gender, women compared with men have an increased incidence of aneurysms at a ratio of 3:2
- Other disorders: Ehlers-Danlos Syndrome, Polycystic Kidney Disease, Marfan Syndrome, and Fibromuscular Dysplasia (FMD)
- Presence of an arteriovenous malformation (AVM)
- Drug use, particularly cocaine
- Blood infections (mycotic aneurysm)
- Tumors
- Traumatic head injury (dissecting aneurysm)
Risk factors that doctors and researchers believe contribute to the rupture of brain aneurysms:
- Smoking
- High blood pressure or hypertension
- African-Americans at twice the rate of rupture of whites (a 2.1:1 ratio)
- Hispanics at nearly twice the rate of rupture of whites (a 1.67:1 ratio)
Risk factors present at birth
Selected conditions that date to birth can be associated with an elevated risk of developing a brain aneurysm. These include:
- Inherited connective tissue disorders, such as Ehlers-Danlos syndrome, that weaken blood vessels
- Polycystic kidney disease, an inherited disorder that results in fluid-filled sacs in the kidneys and usually increases blood pressure
- Abnormally narrow aorta (coarctation of the aorta), the large blood vessel that delivers oxygen-rich blood from the heart to the body
- Cerebral arteriovenous malformation (brain AVM), an abnormal connection between arteries and veins in the brain that interrupts the normal flow of blood between them
- Family history of brain aneurysm, particularly a first-degree relative, such as a parent, brother, sister, or child.
Family history of brain aneurysm
Familial intracranial aneurysms are generally defined as the presence of two or more family members among first- and second-degree relatives with proven aneurysmal SAH (subarachnoid hemorrhage) or incidental aneurysms.
- The incidence of familial aneurysms among SAH patients is 6% to 20%.
- Familial intracranial aneurysm is defined as two or more blood relatives who possess intracranial aneurysms.
- The familial occurrence suggests a genetic component and the possibility of a genetically determined defect of the arterial wall.
- Several studies suggest that individuals with familial intracranial aneurysms are more likely to have multiple aneurysms and that these aneurysms are more likely to rupture at a smaller size than those patients with an isolated aneurysm.
- Treatment considerations are different for patients with familial aneurysms than for patients with an unruptured isolated aneurysm.
- The Familial Intracranial Aneurysm Study has recruited a number of families and their goal is to identify genes that underlie the development and rupture of intracranial aneurysms.
- The National Institute of Neurological Diseases’ funded study is the largest genetic linkage study to date. The study includes 26 clinical centers that have broad experience in clinical management and imaging patients with intracranial aneurysms. The study will recruit 475 families with affected sib pairs or with multiple affected relatives through retrospective and prospective screening of potential subjects with an intracranial aneurysm.
- An important aspect of the genetic study is the study of environmental factors in disease risk such as smoking. Nearly 80% of patients with an Intracranial Aneurysm (brain aneurysm) have a history of smoking at some time in their life. Not all individuals who smoke develop IA. Smoking may increase the risk of IA in for individual with specific genotypes at IA susceptibility loci.
Screening for brain aneurysms
The use of imaging tests to screen for unruptured brain aneurysms is generally not recommended. However, you may want to discuss with your doctor the potential benefit of a screening test if you have:
- Risk factors for developing brain aneurysms include cigarette use, disorders of the body’s structural proteins (Ehlers-Danlos syndrome, Marfan syndrome), fibromuscular dysplasia, chronic hypertension, use of cocaine, crack or amphetamines and polycystic kidney disease.
- A parent or sibling who has had a brain aneurysm, particularly if you have two such first-degree family members with brain aneurysms
- A congenital disorder that increases your risk of a brain aneurysm e.g. Ehlers-Danlos Syndrome, Polycystic Kidney Disease, Marfan Syndrome, and Fibromuscular Dysplasia (FMD)
Screening Recommendations
Screening in the setting of a family history of fusiform aneurysm:
In the case of a first degree relative with a known intracranial fusiform aneurysm, it is unclear if screening is recommended, but there are several reports of fusiform aneurysms occurring in a family. Some doctors would recommend screening.
Screening in the setting of a family history of saccular aneurysm:
If one member of the family is known to be affected with saccular aneurysm, then screening of other family members is not clearly recommended since the occurrence of an aneurysm with screening is approximately the same or only slightly higher than the general population. This is a somewhat controversial issue, and family members will understandably not infrequently ask to be screened, and screening with either brain MRA or brain CTA could readily be performed if that would be their preference. It is important that the patient understands the potential implications of screening should an aneurysm or some other unexpected finding be detected (i.e., life and health insurance issues, etc.). Even in the setting of only one affected member of the family, some doctors do recommend screening of first degree relatives (siblings, parents, children), and there are some data, (unpublished at this point) which suggest that the occurrence in these first degree relatives may be higher than previously thought.
If two or more members of the family are affected with brain aneurysms, then aneurysm screening (with brain MRA or brain CTA) is usually recommended for at least the first degree relatives over the age of 25 of those affected. Our recommendation is for any individual who has two or more relatives (e.g. mother, father, brother, sister, aunt, uncle, grandparent, cousins) with a brain aneurysm be screened for aneurysms starting in their twenties and then every 5 to 10 years thereafter. It is thought that patients with a family history of aneurysm may develop them at a younger age in subsequent generations. Those with a family history of aneurysms are more likely to have multiple aneurysms and those aneurysms are more likely to rupture at a smaller size. The risk of aneurysm detection in these family members is increased in women, those with hypertension, and in cigarette smokers. If your family has a history of aneurysms, it is imperative for you to be screened by MRA or CTA when you are in your twenties. Early detection saves lives as aneurysms most often times can be treated.
Two quick and safe ways to screen for aneurysms include MRI with MRA (Magnetic Resonance Imaging with angiography) and CT with CTA (Computed Tomography with Angiography). Images that are obtained during these studies will reliably detect aneurysms as small as 2 mm. There are advantages and disadvantages of each of these types of studies.
MRA images are generated as a result of disturbances in a strong magnetic field. Excellent pictures of the brain itself are obtained and reasonably good pictures of the major arteries are as well. This is a good way to do an initial “screen”. These are very safe tests as no radiation is used, but the quality and detail of the images are not as good as CTA or catheter angiography. In addition, it might take as long as 40 minutes for a patient to complete one of these studies, and patients who are claustrophobic frequently need to be sedated, as the confines of the machine induce a sense of claustrophobia.
CTA images are created by injecting an iodine-based dye into the vein of the arm. As it passes from the vein to the heart and then pumped to the brain, X-rays are passed through the head and images are created. This is a very fast test that takes only a few minutes to perform and the quality and detail of the images are excellent. The down side is that it does expose the patient to X-ray radiation and iodine which in some patients can lead to an allergic reaction. Usually, CTA is reserved as a follow-up study to a MRA study if an aneurysm is detected and there is a need to collect more information on the aneurysm. This additional information allows for a more detailed conversation with the physician on the need for treatment and what type of treatment options are available.
Catheter-based angiography is not a good initial test for screening as the small risk of this procedure does not justify its use when MRA and CTA are effective in this role.
Brain aneurysm signs and symptoms
Ruptured brain aneurysm
A sudden, severe headache is the key symptom of a ruptured aneurysm. This headache is often described as the “worst headache” ever experienced.
Common signs and symptoms of a ruptured aneurysm include:
- Sudden, extremely severe headache
- Nausea and vomiting
- Stiff neck
- Blurred or double vision
- Sensitivity to light
- Seizure
- A drooping eyelid
- Loss of consciousness
- Confusion
‘Leaking’ brain aneurysm
In some cases, an aneurysm may leak a slight amount of blood. This leaking (sentinel bleed) may cause only a:
- Sudden, extremely severe headache
A more severe rupture often follows leaking.
Unruptured Brain Aneurysms
An unruptured brain aneurysm may produce no symptoms, particularly if it’s small. However, a larger unruptured aneurysm may press on brain tissues and nerves, possibly causing:
- Pain above and behind one eye
- A dilated pupil
- Change in vision or double vision
- Numbness of one side of the face.
Subarachnoid Hemorrhage
When a brain aneurysm ruptures, it causes bleeding into the compartment surrounding the brain, the subarachnoid space and is therefore also known as a subarachnoid hemorrhage (SAH). Often the aneurysm heals over, bleeding stops, and the person survives. In more serious cases, the bleeding may cause brain damage with paralysis or coma. In the most severe cases, the bleeding leads to death.
What Happens to the Brain When an Aneurysm Bleeds?
In most cases, after a rupture the bleeding quickly stops. Blood in the cerebrospinal fluid (CSF) increases the pressure on the brain.
Damage to Brain Cells
Blood from an aneurysm can leak into the CSF (cerebrospinal fluid) in the space around the brain (subarachnoid space). The pool of blood forms a clot. Blood can irritate, damage, or destroy nearby brain cells. This may cause problems with body functions or mental skills.
Fluid Buildup in the Brain
Blood from a torn aneurysm can block CSF circulation. This can lead to fluid buildup and increased pressure on the brain. Because blood is sprayed around the base of the brain, the possibility of fluid buildup exists, causing hydrocephalus. The open spaces in the brain, called ventricles, may enlarge. It can make a patient lethargic, confused, or incontinent. To stop fluid buildup, a drain may be placed in the ventricles. The tube is called a ventriculostomy, and often drains into a bag at the patient’s bedside. This removes leaked blood and trapped CSF.
Vasospasm
The blood around the base of the brain can also produce a problem called vasospasm. Vasospasm typically develops 5-8 days after the initial hemorrhage. Narrowing of the blood vessels can occur and at times not enough blood is supplied to the brain and a stroke may result. To treat vasospasm, blood pressure is often elevated with medicines. Certain medications are also given to try to ease vasospasm. Finally, catheters can be introduced inside the artery in an attempt to use balloons or medications delivered to the vessel directly to open up these narrowed vessels. Vasospasm does relax over several days.
Outlook
Subarachnoid hemorrhage (SAH) survivors usually have a much longer recovery time than unruptured aneurysm patients, as well as more serious deficits. Symptoms are proportional to the degree of hemorrhage. Patients who are comatose or semi-comatose after a hemorrhage have longer recoveries and have more significant neurocognitive problems as compared to patients with smaller hemorrhages or unruptured aneurysms.
Pediatric Brain Aneurysms
Intracranial Aneurysms In Children (<18 Years Old) Are Rare
- Reported prevalence ranging from .5% to 4.6%
- Epidemiology poorly understood
- Pediatric brain aneurysms occur more often in male patients
- Male/ female occurrence is 1.8: 1
- Subarachnoid hemorrhage was the most common presentation in 80% of the children
- The most common overall location was the internal carotid artery terminus, which was the location in 26% of the cases
- Only 17% occurred in the posterior circulation
- One fifth of all aneurysms were giant lesions
- The sex predominance (male/ female 1.8:1) may suggest the existence of differences in pathogenesis of aneurysm formation in the pediatric patient
- One interpretation is that congenital factors present in all aneurysm patients may be expressed more in boys, but environmental factors may contribute to the increased incidence in girls
- Aneurysms in the pediatric population are four times as likely to present with subarachnoid hemorrhage versus without subarachnoid hemorrhage
- Pediatric aneurysms are twice as likely to be of a better grade
- Of the published series of pediatric aneurysms, almost all the earliest series before 1981 were comprised of subarachnoid hemorrhage patients. Unruptured aneurysms were detected more often thereafter, a reflection in the improvement of neuroimaging techniques.
- Posterior circulation aneurysms accounted for 17% of all pediatric aneurysms.
Brain aneurysm diagnosis
If you experience a sudden, severe headache or other symptoms possibly related to a ruptured aneurysm, you’ll be given a test or series of tests to determine whether you’ve had bleeding into the space between your brain and surrounding tissues (subarachnoid hemorrhage) or possibly another type of stroke.
If bleeding has occurred, your emergency care team will determine whether the cause is a ruptured aneurysm.
If you show symptoms of an unruptured brain aneurysm — such as pain behind the eye, changes in vision or double vision — you will also undergo some tests to identify the offending aneurysm.
Diagnostic tests include:
- Computerized tomography (CT). A CT scan, a specialized X-ray exam, is usually the first test used to determine if you have bleeding in the brain. The test produces images that are 2-D “slices” of the brain. With this test, you may also receive an injection of a dye that makes it easier to observe blood flow in the brain and may indicate the presence of an aneurysm. This variation of the test is called CT angiography.
- Cerebrospinal fluid test. If you’ve had a subarachnoid hemorrhage (SAH), there will most likely be red blood cells in the fluid surrounding your brain and spine (cerebrospinal fluid). Your doctor will order a test of the cerebrospinal fluid if you have symptoms of a ruptured aneurysm but a CT scan hasn’t shown evidence of bleeding. The procedure to draw cerebrospinal fluid from your back with a needle is called a lumbar puncture (spinal tap).
- Magnetic resonance imaging (MRI). An MRI uses a magnetic field and radio waves to create detailed images of the brain, either 2-D slices or 3-D images. A type of MRI that assesses the arteries in detail (MRI angiography) may detect the presence of an aneurysm.
- Cerebral angiogram. During this procedure, also called a cerebral arteriogram, your doctor inserts a thin, flexible tube (catheter) into a large artery — usually in your groin — and threads it past your heart to the arteries in your brain. A special dye injected into the catheter travels to arteries throughout your brain. A series of X-ray images can then reveal details about the conditions of your arteries and detect an aneurysm. This test is more invasive than others and is usually used when other diagnostic tests don’t provide enough information.
Brain aneurysm treatment
Treatment for brain aneurysms is more promising than it was several years ago. There are more effective and less invasive treatment options for patients, who in years past, would have been told they had inoperable aneurysms. Doctors consider several factors when deciding which treatment option is best for a particular patient. Some factors include patient age, size of aneurysm and location of aneurysm.
Brain aneurysm surgery
There are two common treatment options for a ruptured brain aneurysm.
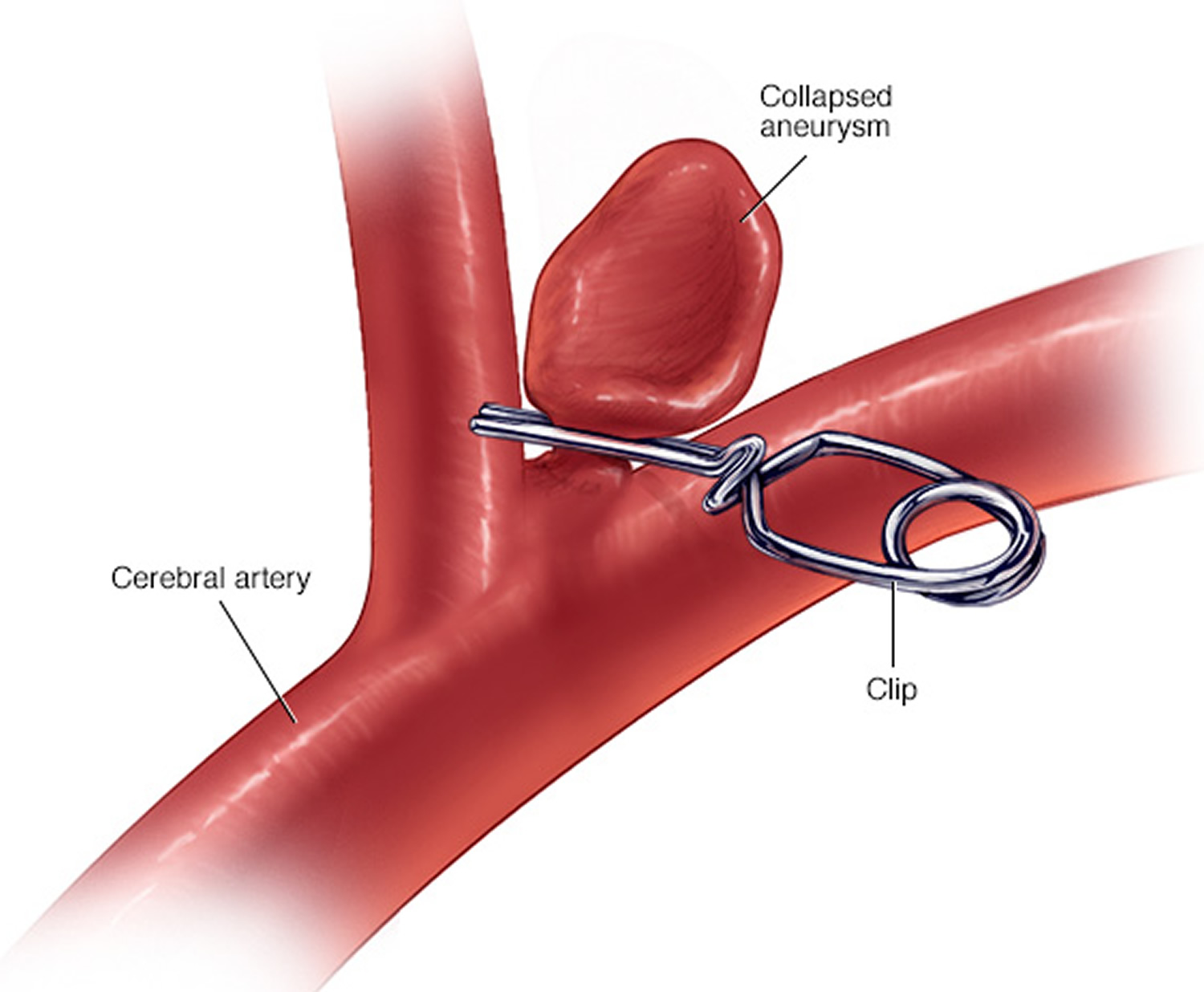
- Surgical clipping is a procedure to close off an aneurysm. The neurosurgeon removes a section of your skull to access the aneurysm and locates the blood vessel that feeds the aneurysm. Then he or she places a tiny metal clip on the neck of the aneurysm to stop blood flow to it.
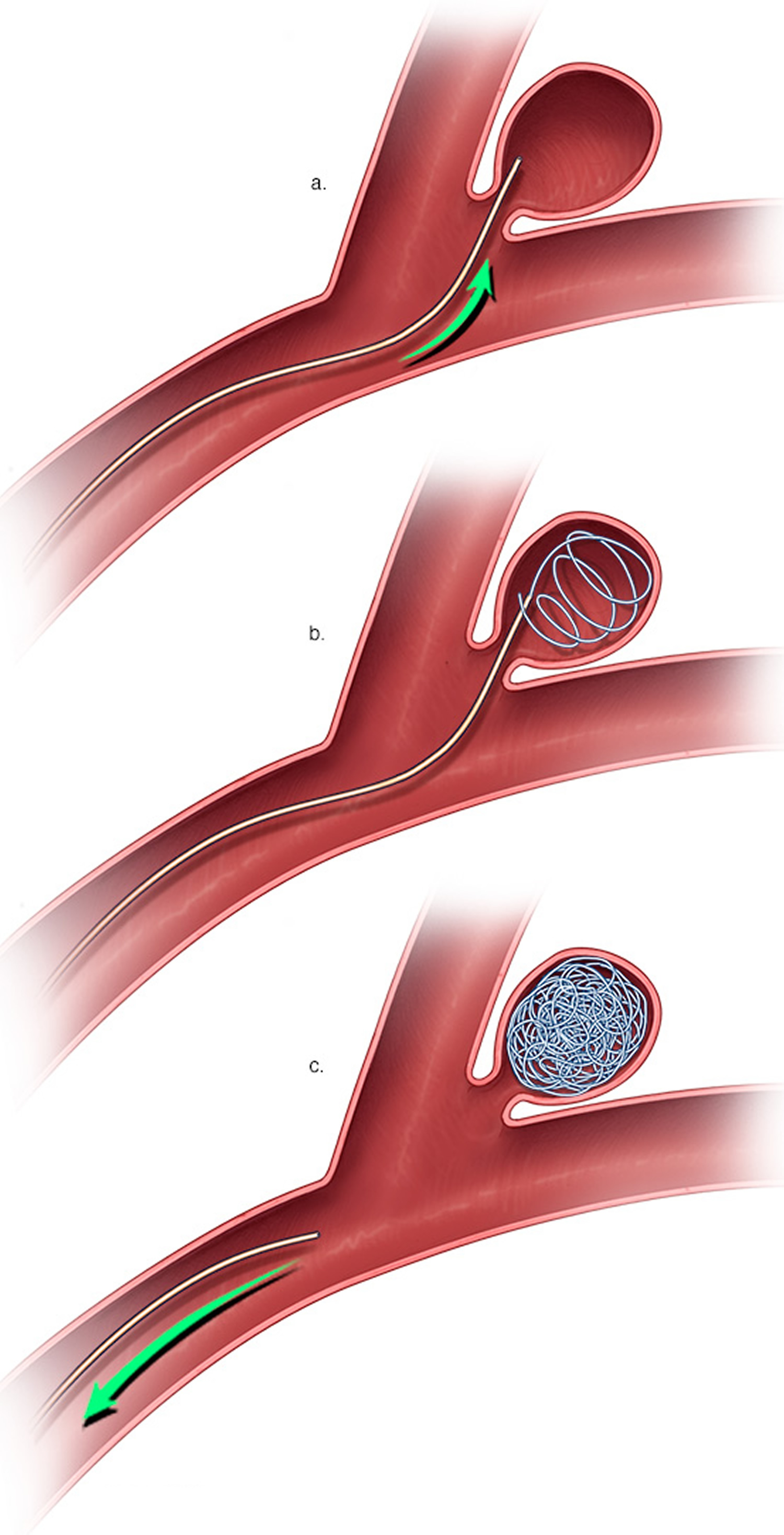
- Endovascular coiling is a less invasive procedure than surgical clipping. The surgeon inserts a hollow plastic tube (catheter) into an artery, usually in your groin, and threads it through your body to the aneurysm. He or she then uses a guide wire to push a soft platinum wire through the catheter and into the aneurysm. The wire coils up inside the aneurysm, disrupts the blood flow and essentially seals off the aneurysm from the artery.
Both procedures pose potential risks, particularly bleeding in the brain or loss of blood flow to the brain. The endovascular coil is less invasive and may be initially safer, but it may have a slightly higher risk of need for a repeat procedure in the future due to reopening of the aneurysm.
Figure 5. Brain aneurysm surgical clipping
Figure 6. Brain aneurysm endovascular coiling
Flow diverters
Newer treatments available for brain aneurysm include flow diverters, tubular stent-like implants that work by diverting blood flow away from an aneurysm sac. The diversion stops blood movement within the aneurysm and so stimulates the body to heal the site, encouraging reconstruction of the parent artery. Flow diverters may be particularly useful in larger aneurysms that can’t be safely treated with other options.
Your neurosurgeon or interventional neuroradiologist, in collaboration with your neurologist, will make a recommendation based on the size, location and overall appearance of the brain aneurysm, your ability to undergo a procedure, and other factors.
Other treatments (ruptured aneurysms)
Other treatments for ruptured brain aneurysms are aimed at relieving symptoms and managing complications.
- Pain relievers, such as acetaminophen (Tylenol, others), may be used to treat headache pain.
- Calcium channel blockers prevent calcium from entering cells of the blood vessel walls. These medications may lessen the erratic narrowing of blood vessels (vasospasm) that may be a complication of a ruptured aneurysm. One of these medications, nimodipine (Nymalize, Nimotop), has been shown to reduce the risk of delayed brain injury caused by insufficient blood flow after subarachnoid hemorrhage from a ruptured aneurysm.
- Interventions to prevent stroke from insufficient blood flow include intravenous injections of a drug called a vasopressor, which elevates blood pressure to overcome the resistance of narrowed blood vessels. An alternative intervention to prevent stroke is angioplasty. In this procedure, a surgeon uses a catheter to inflate a tiny balloon that expands a narrowed blood vessel in the brain. A drug known as a vasodilator also may be used to expand blood vessels in the affected area.
- Anti-seizure medications may be used to treat seizures related to a ruptured aneurysm. These medications include levetiracetam (Keppra), phenytoin (Dilantin, Phenytek, others), valproic acid (Depakene) and others. Their use has been debated by several experts, and is generally subject to caregiver discretion, based on the medical needs of each patient.
- Ventricular or lumbar draining catheters and shunt surgery can lessen pressure on the brain from excess cerebrospinal fluid (hydrocephalus) associated with a ruptured aneurysm. A catheter may be placed in the spaces filled with fluid inside of the brain (ventricles) or surrounding your brain and spinal cord to drain the excess fluid into an external bag. Sometimes it may then be necessary to introduce a shunt system — which consists of a flexible silicone rubber tube (shunt) and a valve — that creates a drainage channel starting in your brain and ending in your abdominal cavity.
- Rehabilitative therapy. Damage to the brain from a subarachnoid hemorrhage may result in the need for physical, speech and occupational therapy to relearn skills.
Treating unruptured brain aneurysms
Not all brain aneurysms rupture. Doctors are now able to detect unruptured brain aneurysms with an increased frequency because of the growing availability of non-invasive imaging methods such as magnetic resonance imaging/magnetic resonance angiogram. An unruptured brain aneurysm may or may not cause symptoms. As opposed to ruptured aneurysms which require urgent treatment in almost all cases, unruptured aneurysms may require treatment or may be followed with serial imaging studies in some cases. Things doctors consider in deciding whether or not to treat an unruptured aneurysm:
- Risk of hemorrhage — Is it probable or not that the aneurysm will rupture?
- Size and location
- Age and health of patient
- Family history — Is there a family history? Have any of those aneurysms ruptured?
- Surgical/endovascular risks
- Patient preference
Like ruptured aneurysms, unruptured aneurysms may be treated with either endovascular coiling or a flow diverter or open surgical clipping – to seal off an unruptured brain aneurysm and help prevent a future rupture. However, in some unruptured aneurysms, the known risks of the procedures may outweigh the potential benefit. On the other hand, if the unruptured aneurysm is treated successfully, the recovery period is generally shorter than that following treatment of a ruptured aneurysm. Although, survivors of unruptured brain aneurysm treatment may suffer many of the same physical and emotional symptoms as a survivor of a rupture, they will have a shorter hospital stay, require less rehabilitative therapy, and return to work more quickly.
A neurologist, in collaboration with a neurosurgeon or interventional neuroradiologist, can help you determine whether the treatment is appropriate for you.
Factors to consider in making treatment recommendations include:
- The size, location and overall appearance of the aneurysm
- Your age and general health
- Neurological condition of patient
- Other medical conditions
- Previous history of SAH or familial aneurysm
- Family history of ruptured aneurysm
- Congenital conditions that increase the risk of a ruptured aneurysm
If you have high blood pressure, talk to your doctor about medication to manage the condition. If you have a brain aneurysm, proper control of blood pressure may lower the risk of rupture.
In addition, if you smoke cigarettes, talk with your provider about strategies to stop smoking since cigarette smoking is a risk factor for formation, growth and rupture of the aneurysm.
Lifestyle changes to lower your risk
If you have an unruptured brain aneurysm, you may lower the risk of its rupture by making these lifestyle changes:
- Don’t smoke or use recreational drugs. If you smoke or use recreational drugs, talk to your doctor about strategies or an appropriate treatment program to help you quit.
- Eat a healthy diet and exercise. Changes in diet and exercise can help lower blood pressure. Talk to your doctor about changes appropriate for you.
Post Treatment and Outcome
The recovery following treatment of an intracranial aneurysm is dependent on many factors, including whether the aneurysm has bled and the type of treatment for the aneurysm.
If an aneurysm presents with a subarachnoid hemorrhage, the length of hospitalization and recovery are dictated by the severity of the hemorrhage and not by the treatment modality. Following a subarachnoid hemorrhage, most patients will remain hospitalized for a minimum of two weeks during which time they will be monitored for the development of cerebral vasospasm and other complications of the hemorrhage including hydrocephalus. If the patient develops any of these complications or has a neurological deficit from the initial hemorrhage, variable periods of rehabilitation on an inpatient or outpatient basis may be necessary. Under the best of circumstances many patients may resume all of their previous activities within a period of several weeks without any specific limitations on their activities.
For patients with unruptured aneurysms, the hospitalization and recovery is typically much smoother and more predictable. In this case, the modality of treatment does influence the hospitalization and recovery. Endovascular therapy, being less invasive, is associated with a shorter hospitalization and more rapid return to previous activities. Following a successful endovascular procedure, most patients are observed in the intensive care unit overnight and discharged from the hospital the following day if there are no complications. These patients may resume all of their prior activities without any specific restrictions within a matter of days. This includes going back to work, driving and flying in airplanes.
Following a craniotomy for clip ligation of an aneurysm, most patients will spend a night in the intensive care unit and be moved out to a private room the following day. Typically, patients are discharged from the hospital a couple of days after discharge from the intensive care unit able to perform all the activities of daily living. It generally takes 4 to 6 weeks to recover from any major operation including a craniotomy for aneurysm. During this time the patient is able to care for themselves, but typically does not engage in aggressive physical activity. Patients are usually able to a care for themselves and stay alone with gradually increasing activity including walking, household chores and work that does not involve significant physical exertion. After approximately 4 to 6 weeks these activities are liberalized and patients are encouraged to resume all of their previous activities without any specific restrictions. It is by increasing activity at this time that patients more quickly resume their strength and endurance.
Because of the higher risk of recurrence of aneurysms following endovascular therapy, some routine maintenance is required and patients will typically return to their physician for follow-up angiography at variable periods of time to monitor for any evidence of recurrence. Following surgical clip ligation of the aneurysm, there typically is no routine maintenance of the aneurysm, particularly if intraoperative imaging documented complete obliteration of the aneurysm. For younger patients or those with a strong family history of aneurysms who may be at higher risk of the development of de novo aneurysms some routine monitoring may be recommended.