Contents
- What is gardasil
- What kinds of problems does HPV infection cause?
- Why does my child need HPV vaccine?
- How do people get an HPV infection?
- How common is HPV and the health problems caused by HPV?
- How do I know if I have HPV?
- How can I avoid HPV and the health problems it can cause?
- Who should get HPV vaccine?
- Who else should get the HPV vaccine?
- When should my child be vaccinated?
- How well does HPV vaccine work?
- Does HPV vaccination offer similar protection from cervical cancer in all racial/ethnic groups?
- Where can I find HPV Vaccines?
- How can someone get help paying for HPV vaccine?
- Gardasil Vaccine Recommendations
- Is Gardasil safe?
- Gardasil vaccine schedule
- Gardasil shot contraindications and precautions
- Gardasil vaccine side effects
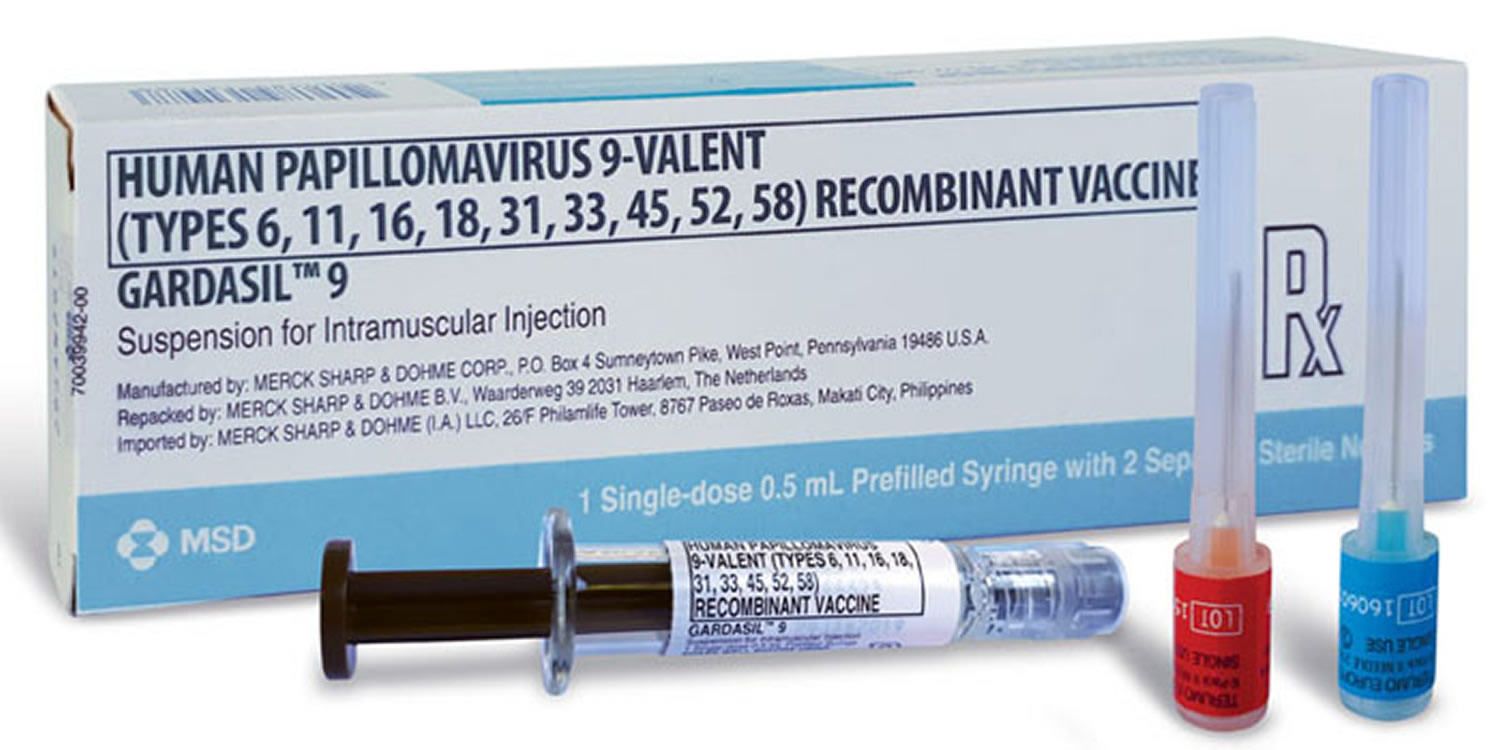
What is gardasil
Gardasil (HPV quadrivalent vaccine) and Gardasil 9 (HPV 9-valent vaccine) are used in both females and males. Another form of HPV vaccine (Cervarix – a bivalent HPV vaccine) is used only in females.
Human papillomavirus (HPV) is the most common sexually transmitted infection in the United States. Human papillomavirus (HPV) can cause genital warts, cancer of the cervix, anal cancer, and various cancers of the vulva or vagina; cancers of the penis in men; and cancers of the anus and back of the throat, including the base of the tongue and tonsils (oropharynx), in both women and men. Some health effects caused by HPV can be prevented by the HPV vaccines like Gardasil.
Nearly 80 million people—about one in four—are currently infected with HPV in the United States. About 14 million people, including teens, become infected with HPV each year.
Over 30,000 people in the United States each year are affected by a cancer caused by HPV infection. While there is screening available for cervical cancer for women, there is no screening for the other cancers caused by HPV infection, like cancers of the mouth/throat, anus/rectum, penis, vagina, or vulva.
Cervical cancer remains the most common cancer in Eastern and Central Africa with an estimated with estimated age standardized rates of 42.7 and 30.6 respectively compared with 2–5 in Western Europe and Australia 1). Mortality is highest in Eastern Africa at 27 per 10,000 compared to less than 2 per 100,000 in Western Asia/Europe, Australia and New Zealand 2). Cervical cancer is the 4th commonest cancer affecting women worldwide with over 260,000 deaths reported in 2012 3).
HPV vaccination provides safe, effective, and lasting protection against the HPV infections that most commonly cause cancer and is supplemented by cervical cancer screening later in life. This is because HPV vaccines do not protect against all high-risk and potential high-risk HPV types.
The quadrivalent (Gardasil 4) HPV vaccine protecting against human papilloma virus (HPV) types 16/18/6/11 and the bivalent (Cervirax) vaccine protecting against infection with HPV-16/18 were both originally used as a three dose schedule. A third vaccine, the nonavalent HPV (Gardasil 9) vaccine protecting against HPV types 6/11/16/18/31/33/45/52/58 was approved by Food and Drug Administration (FDA) in 2014 4).
Three HPV vaccines have been licensed by the U.S. Food and Drug Administration (FDA):
- Gardasil (Merck) is a quadrivalent HPV vaccine (4vHPV) that protects against HPV types 6, 11, 16, and 18.
- Gardasil-9 (Merck) is a nine-valent HPV vaccine (9vHPV) that protects against HPV types 6, 11, 16, 18, 31, 45, 52, and 58.
- Cervirax is a bivalent HPV vaccine (2vHPV) that protects against HPV types 16 and 18
All three HPV vaccines protect against the two HPV types, 16 and 18, that cause most HPV cancers.
Gardasil is used in girls and young women ages 9 through 26 to prevent cervical/vaginal/anal cancers caused by certain types of HPV.
Gardasil is also used in boys and young men ages 9 through 26 to prevent anal cancer or genital warts caused by certain types of HPV.
You may receive Gardasil even if you have already had genital warts, or had a positive HPV test or abnormal pap smear in the past. However, Gardasil vaccine will not treat active genital warts or HPV-related cancers, and it will not cure HPV infection.
Gardasil only prevents diseases caused by HPV types 6, 11, 16, and 18. It will not prevent diseases caused by other types of HPV.
The Centers for Disease Control and Prevention (CDC) recommends HPV vaccine for all boys and girls ages 11 or 12 years old. The vaccine is also recommended in teenage boys and girls who have not already received the vaccine or have not completed all booster shots.
Like any vaccine, the Gardasil and Gardasil 9 may not provide protection from disease in every person.
Some people should NOT get Gardasil and Gardasil 9 vaccine
- Anyone who has had a severe (life-threatening) allergic reaction to a dose of HPV vaccine should not get another dose.
- Anyone who has a severe (life threatening) allergy to any component of HPV vaccine should not get the vaccine.
- Tell your doctor if you have any severe allergies that you know of, including a severe allergy to yeast.
- HPV vaccine is not recommended for pregnant women. If you learn that you were pregnant when you were vaccinated, there is no reason to expect any problems for you or your baby. Any woman who learns she was pregnant when she got HPV vaccine is encouraged to contact the manufacturer’s registry for HPV vaccination during pregnancy. Women who are breastfeeding may be vaccinated.
- If you have a mild illness, such as a cold, you can probably get the vaccine today. If you are moderately or severely ill, you should probably wait until you recover. Your doctor can advise you.
What kinds of problems does HPV infection cause?
Most people with HPV never develop symptoms or health problems. Most HPV infections (9 out of 10) go away by themselves within two years. But, sometimes, HPV infections will last longer, and can cause certain cancers and other diseases. HPV infections can cause:
- cancers of the cervix, vagina, and vulva in women;
- cancers of the penis in men; and
- cancers of the anus and back of the throat, including the base of the tongue and tonsils (oropharynx), in both women and men.
Every year in the United States, HPV causes 32,500 cancers in men and women.
Why does my child need HPV vaccine?
HPV vaccine is important because it protects against cancers caused by human papillomavirus (HPV) infection. HPV is a very common virus; nearly 80 million people—about one in four—are currently infected in the United States. About 14 million people, including teens, become infected with HPV each year.
Most people with HPV never develop symptoms or health problems. Most HPV infections (9 out of 10) go away by themselves within two years. But, sometimes, HPV infections will last longer, and can cause certain cancers and other diseases. HPV infection can cause:
- cancers of the cervix, vagina, and vulva in women;
- cancers of the penis in men; and
- cancers of the anus and back of the throat, including the base of the tongue and tonsils (oropharynx), in both women and men.
Every year in the United States, HPV causes 32,500 cancers in men and women. HPV vaccination can prevent most of the cancers (about 30,000) from ever developing.
How do people get an HPV infection?
People get HPV from another person during intimate sexual contact. Most of the time, people get HPV from having vaginal and/or anal sex. Men and women can also get HPV from having oral sex or other sex play. A person can get HPV even if their partner doesn’t have any signs or symptoms of HPV infection. A person can have HPV even if years have passed since he or she had sexual contact with an infected person. Most people do not realize they are infected. They also don’t know that they may be passing HPV to their sex partner(s). It is possible for someone to get more than one type of HPV.
It’s not very common, but sometimes a pregnant woman with HPV can pass it to her baby during delivery. The child might develop recurrent respiratory papillomatosis, a rare but dangerous condition where warts caused by HPV (similar to genital warts) grow inside the throat.
There haven’t been any documented cases of people getting HPV from surfaces in the environment, such as toilet seats. However, someone could be exposed to HPV from objects (toys) shared during sexual activity if the object has been used by an infected person.
How common is HPV and the health problems caused by HPV?
HPV (the virus): About 79 million Americans are currently infected with HPV. About 14 million people become newly infected each year. HPV is so common that almost every person who is sexually-active will get HPV at some time in their life if they don’t get the HPV vaccine.
Health problems related to HPV include genital warts and cervical cancer.
Genital warts: Before HPV vaccines were introduced, roughly 340,000 to 360,000 women and men were affected by genital warts caused by HPV every year.* Also, about one in 100 sexually active adults in the U.S. has genital warts at any given time.
Cervical cancer: Every year, nearly 12,000 women living in the U.S. will be diagnosed with cervical cancer, and more than 4,000 women die from cervical cancer—even with screening and treatment.
There are other conditions and cancers caused by HPV that occur in people living in the United States. Every year, approximately 19,400 women and 12,100 men are affected by cancers caused by HPV.
*These figures only look at the number of people who sought care for genital warts. This could be an underestimate of the actual number of people who get genital warts.
How do I know if I have HPV?
There is no test to find out a person’s “HPV status.” Also, there is no approved HPV test to find HPV in the mouth or throat.
There are HPV tests that can be used to screen for cervical cancer. These tests are only recommended for screening in women aged 30 years and older. HPV tests are not recommended to screen men, adolescents, or women under the age of 30 years.
Most people with HPV do not know they are infected and never develop symptoms or health problems from it. Some people find out they have HPV when they get genital warts. Women may find out they have HPV when they get an abnormal Pap test result (during cervical cancer screening). Others may only find out once they’ve developed more serious problems from HPV, such as cancers.
How can I avoid HPV and the health problems it can cause?
You can do several things to lower your chances of getting HPV.
Get vaccinated. The HPV vaccine is safe and effective. It can protect against diseases (including cancers) caused by HPV when given in the recommended age groups. CDC recommends 11 to 12 year olds get two doses of HPV vaccine to protect against cancers caused by HPV 5).
Get screened for cervical cancer. Routine screening for women aged 21 to 65 years old can prevent cervical cancer.
If you are sexually active:
- Use latex condoms the right way every time you have sex. This can lower your chances of getting HPV. But HPV can infect areas not covered by a condom – so condoms may not fully protect against getting HPV;
- Be in a mutually monogamous relationship – or have sex only with someone who only has sex with you.
Who should get HPV vaccine?
All girls and boys who are 11 or 12 years old should get the recommended series of Gardasil vaccine. The vaccination series can be started at age 9 years. Teen boys and girls who did not get vaccinated when they were younger should get it now. Gardasil vaccine is recommended for young women through age 26, and young men through age 21. Gardasil vaccine is also recommended for the following people, if they did not get vaccinated when they were younger:
- young men who have sex with men, including young men who identify as gay or bisexual or who intend to have sex with men through age 26;
- young adults who are transgender through age 26; and
- young adults with certain immunocompromising conditions (including HIV) through age 26.
Who else should get the HPV vaccine?
Teen boys and girls who did not start or finish the HPV vaccine series when they were younger should get it now.
HPV vaccine is recommended for young women through age 26, and young men through age 21. HPV vaccine is also recommended for the following people, if they did not get vaccinated when they were younger:
- young men who have sex with men, including young men who identify as gay or bisexual or who intend to have sex with men through age 26;
- young adults who are transgender through age 26; and
- young adults with certain immunocompromising conditions (including HIV) through age 26.
When should my child be vaccinated?
In this video, a family physician explains his decision, as a doctor and a parent, to make sure each of his children received HPV vaccine at age 11 or 12. HPV vaccine is cancer prevention. Ask about it for your child.
All kids who are 11 or 12 years old should get two shots of HPV vaccine six to twelve months apart. Adolescents who receive their two shots less than five months apart will require a third dose of HPV vaccine.
If your teen hasn’t gotten the vaccine yet, talk to their doctor or nurse about getting it for them as soon as possible. If your child is older than 14 years, three shots will need to be given over 6 months. Also, three doses are still recommended for people with certain immunocompromising conditions aged 9 through 26 years.
How well does HPV vaccine work?
Gardasil HPV vaccines work extremely well. Clinical trials showed HPV vaccines provide close to 100% protection against cervical precancers and genital warts. Since the first HPV vaccine was recommended in 2006, there has been a 64% reduction in vaccine-type HPV infections among teen girls in the United States. Studies have shown that fewer teens are getting genital warts and cervical precancers are decreasing. In other countries, such as Australia, where HPV vaccination coverage is higher than in the United States, large decreases have been observed in these HPV-associated outcomes. HPV vaccines offer long-lasting protection against HPV infection and HPV disease. There has been no evidence to suggest that HPV vaccine loses any ability to provide protection over time. Data are available for about 10 years of follow-up after vaccination.
Like all vaccines, HPV vaccine is monitored on an ongoing basis to make sure it remains safe and effective. If it turns out that protection from HPV vaccine is not long-lasting, then the Advisory Committee on Immunization Practices would review the data and determine whether a booster dose would be recommended.
Even if it has been months or years since the last shot, the HPV vaccine series should be completed—but they do not need to restart the series.
HPV vaccine is recommended based on age, not sexual experience. Even if someone has already had sex, they should still get HPV vaccine. Even though a person’s first HPV infection usually happens during one of the first few sexual experiences, a person might not be exposed to all of the HPV types that are covered by HPV vaccines.
Does HPV vaccination offer similar protection from cervical cancer in all racial/ethnic groups?
Yes. Several different HPV types cause cervical cancer. HPV vaccines are designed to prevent the HPV types that cause most cervical cancers, so HPV vaccination will provide high protection for all racial/ethnic groups.
All three licensed HPV vaccines protect against types 16 and 18, which cause the majority of cervical cancers across racial/ethnic groups (67% of the cervical cancers among whites, 68% among blacks, and 64% among Hispanics). The 9-valent Gardasil vaccine protects against seven HPV types that cause about 80% of cervical cancer among all racial/ethnic groups in the United States.
Teens and young adults who haven’t completed the HPV vaccine series should make an appointment today to get vaccinated. To protect against cervical cancer, women age 21–65 years should get screened for cervical cancer at regular intervals and get follow-up care as recommended by their doctor or nurse.
Where can I find HPV Vaccines?
HPV vaccine may be available at private doctor offices, community health clinics, school-based health centers, and health departments.
If your doctor does not stock HPV vaccine, ask for a referral. If you don’t have a regular source of health care, federally funded health centers can provide services. Locate one near you here (https://www.vaccines.gov/getting/where/index.html).
How can someone get help paying for HPV vaccine?
The Vaccines for Children program helps families of eligible children who might not otherwise have access to vaccines. The program provides vaccines at no cost to children ages 18 years and younger who are uninsured, Medicaid-eligible, or American Indian/Alaska Native.
Children through 18 years of age who meet at least one of the following criteria are eligible to receive Vaccines for Children program vaccine:
- Medicaid eligible: A child who is eligible for the Medicaid program. (For the purposes of the VFC program, the terms “Medicaid-eligible” and “Medicaid-enrolled” are equivalent and refer to children who have health insurance covered by a state Medicaid program)
- Uninsured: A child who has no health insurance coverage
- American Indian or Alaska Native: As defined by the Indian Health Care Improvement Act (25 U.S.C. 1603)
- Underinsured: Underinsured means the child has health insurance, but it:
- Doesn’t cover vaccines, or
- Doesn’t cover certain vaccines, or
- Covers vaccines but has a fixed dollar limit or cap for vaccines. Once that fixed dollar amount is reached, a child is then eligible.
- Underinsured children are eligible to receive vaccines only at Federally Qualified Health Centers (FQHC) or Rural Health Clinics (RHC). An Federally Qualified Health Center is a type of provider that meets certain criteria under Medicare and Medicaid programs. To locate an Federally Qualified Health Center or Rural Health Clinic, contact the state Vaccines for Children program coordinator (https://www.cdc.gov/vaccines/imz-managers/awardee-imz-websites.html).
Children whose health insurance covers the cost of vaccinations are not eligible for Vaccines for Children program vaccines, even when a claim for the cost of the vaccine and its administration would be denied for payment by the insurance carrier because the plan’s deductible had not been met.
Gardasil Vaccine Recommendations
- The Centers for Disease Control and Prevention (CDC) recommends Gardasil HPV vaccine for girls and boys at ages 11 or 12 years to protect against cancers caused by HPV infections. (Vaccination can be started at age 9.)
- The Advisory Committee on Immunization Practices (ACIP) also recommends Gardasil vaccination for females aged 13 through 26 years and males aged 13 through 21 years who are not adequately vaccinated previously.
- Gardasil vaccination is also recommended through age 26 years for gay, bisexual, and other men who have sex with men, transgender people, and for immunocompromised persons (including those with HIV infection) not adequately vaccinated previously.
Ideally, adolescents should be vaccinated before they are exposed to HPV. However, people who have already been infected with one or more HPV types can still get protection from other HPV types in the Gardasil vaccine.
Why is HPV vaccine recommended at age 11 or 12 years?
For HPV vaccine to be most effective, the HPV vaccine series should be given prior to exposure to HPV. There is no reason to wait to vaccinate until teens reach puberty or start having sex. Preteens should receive all recommended doses of the HPV vaccine series long before they begin any type of sexual activity.
Why is HPV vaccine not mandatory for school entry?
Each state determines which vaccines are required for school entry. Many factors are taken into consideration before requiring any vaccine for school entry, including: community support for the requirement, financial resources needed to implement the requirement, burden on school personnel for enforcing the requirement, vaccine supply, and current vaccination coverage levels.
Since almost every state requires Tdap (tetanus, diphtheria, and acellular pertussis vaccine) for middle school entry, parents can use this visit to the doctor to get the first HPV and quadrivalent meningococcal conjugate vaccines for their preteen at the same time.
Is Gardasil safe?
Gardasil HPV vaccines are very safe. Scientific research shows the benefits of Gardasil vaccination far outweigh the potential risks. Like all medical interventions, vaccines can have some side effects.
All vaccines used in the United States, including Gardasil vaccines, are required to go through years of extensive safety testing before they are licensed by the U.S. Food and Drug Administration (FDA). During clinical trials conducted before they were licensed:
- 9-valent Gardasil vaccine was studied in more than 15,000 males and females
- Quadrivalent Gardasil vaccine was studied in more than 29,000 males and females
- Bivalent Cervarix HPV vaccine was studied in more than 30,000 females
- Each HPV vaccine was found to be safe and effective.
How do you know that the HPV vaccine is safe?
The United States currently has the safest, most effective vaccine supply in history. Years of testing are required by law to ensure the safety of vaccines before they are made available for use in the United States. This process can take ten years or longer. Once a vaccine is in use, CDC and the Food and Drug Administration (FDA) monitor any associated side effects or possible side effects (adverse events) through the Vaccine Adverse Event Reporting System and other vaccine safety systems.
All three HPV vaccines—Cervarix®, Gardasil®, and Gardasil® 9—went through years of extensive safety testing before they were licensed by FDA. Cervarix® was studied in clinical trials with more than 30,000 females. Gardasil® trials included more than 29,000 females and males, and Gardasil® 9 trials included more than 15,000 females and males. No serious safety concerns were identified in these clinical trials. FDA only licenses a vaccine if it is safe, effective, and the benefits outweigh the risks. CDC and FDA continue to monitor HPV vaccines to make sure they are safe and beneficial for the public.
Gardasil vaccine schedule
Two doses of Gardasil vaccine are recommended for most persons starting the series before their 15th birthday.
- The second dose of Gardasil vaccine should be given 6 to 12 months after the first dose.
- Adolescents who receive 2 doses less than 5 months apart will require a third dose of Gardasil vaccine.
Three doses of Gardasil vaccine are recommended for teens and young adults who start the series at ages 15 through 26 years, and for immunocompromised persons.
- The recommended 3-dose schedule is 0, 1–2 and 6 months.
- Three doses are recommended for immunocompromised persons (including those with HIV infection) aged 9–26 years.
Why are two doses of Gardasil vaccine recommended for 9–14 year olds, while older adolescents need three doses?
Since 2006, HPV vaccines have been recommended in a three-dose series given over six months. In 2016, CDC changed the recommendation to two doses for persons starting the series before their 15th birthday. The second dose of HPV vaccine should be given six to twelve months after the first dose. Adolescents who receive their two doses less than five months apart will require a third dose of HPV vaccine.
Teens and young adults who start the series at ages 15 through 26 years still need three doses of HPV vaccine Also, three doses are still recommended for people with certain immunocompromising conditions aged 9 through 26 years.
CDC makes recommendations based on the best available scientific evidence. Studies have shown that two doses of HPV vaccine given at least six months apart to adolescents at age 9–14 years worked as well or better than three doses given to older adolescents and young adults. Studies have not been done to show this for adolescents starting the series at age 15 years or older.
Gardasil shot contraindications and precautions
A severe allergic reaction (e.g., anaphylaxis) to a vaccine component or following a prior dose of Gardasil vaccine is a contraindication to receipt of Gardasil vaccine.
- Anaphylactic allergy to latex is a contraindication to bivalent Gardasil vaccine in a prefilled syringe since the tip cap might contain natural rubber latex.
- Quadrivalent and 9-valent Gardasil vaccines are produced in Saccharomyces cerevisiae (baker’s yeast) and are contraindicated for persons with a history of immediate hypersensitivity to yeast.
- A moderate or severe acute illness is a precaution to vaccination, and vaccination should be deferred until symptoms of the acute illness improve.
- A minor acute illness (e.g., diarrhea or mild upper respiratory tract infection, with or without fever) is not a reason to defer vaccination.
Gardasil and Pregnancy
Gardasil vaccine is NOT recommended for use during pregnancy.
- Gardasil vaccine has not been causally associated with adverse pregnancy outcomes or with adverse effects on the developing fetus, but data on vaccination during pregnancy are limited.
- Women known to be pregnant should delay initiation of the Gardasil vaccine series until after the pregnancy.
- Pregnancy testing before vaccination is not needed. However, if a woman is found to be pregnant after initiation of the vaccination series, the remainder of the series should be delayed until after she is no longer pregnant. No intervention is indicated.
Gardasil vaccine side effects
Vaccines, like any medicine, can have side effects. Many people who get Gardasil HPV vaccine have no side effects at all. Some people report having very mild side effects, like a sore arm. The most common side effects are usually mild.
A clinical study 6) evaluated the safety of Gardasil 9 in 12- through 26-year-old girls and women who had previously been vaccinated with three doses of Gardasil. The time interval between the last injection of Gardasil and the first injection of Gardasil 9 ranged from approximately 12 to 36 months. Individuals were administered Gardasil 9 or saline placebo and safety was evaluated using VRC-aided surveillance for 14 days after each injection of Gardasil 9 or saline placebo in these individuals. The individuals who were monitored included 608 individuals who received Gardasil 9 and 305 individuals who received saline placebo. Few (0.5%) individuals who received Gardasil 9 discontinued due to adverse reactions. The vaccine-related adverse experiences that were observed among recipients of Gardasil 9 at a frequency of at least 1.0% and also at a greater frequency than that observed among saline placebo recipients. Overall the safety profile was similar between individuals vaccinated with Gardasil 9 who were previously vaccinated with Gardasil and those who were naïve to HPV vaccination with the exception of numerically higher rates of injection-site swelling and erythema among individuals who were previously vaccinated with Gardasil.
Common side effects of HPV vaccine include:
- The most common adverse reactions reported during clinical trials of HPV vaccines were local reactions at the site of injection – pain, redness, or swelling in the arm where the shot was given.
- In prelicensure clinical trials, local reactions, such as pain, redness, or swelling, were reported by 20% to 90% of recipients.
- A fever of 100°F (37.8 °C) during the 15 days after vaccination was reported in 10% to 13% of HPV vaccine recipients. A similar proportion of placebo recipients reported an elevated temperature.
- A variety of systemic adverse reactions have been reported by vaccine recipients, including nausea, dizziness, myalgia (muscle pain) and malaise. However, these symptoms occurred with equal frequency among both HPV vaccine and placebo recipients.
- Local reactions generally increased in frequency with increasing doses. However, reports of fever did not increase significantly with increasing doses.
- No serious adverse events have been associated with either any HPV vaccine. Ongoing monitoring is conducted by CDC and the Food and Drug Administration.
Brief fainting spells (syncope) and related symptoms (such as jerking movements) can occur after any medical procedure, including vaccination. Adolescents should be seated or lying down during vaccination and remain in that position for 15 minutes after vaccination. This is to prevent any injuries that could occur from a fall from fainting.
On very rare occasions, severe (anaphylactic) allergic reactions may occur after vaccination. People with severe allergies to any component of a vaccine should not receive that vaccine.
HPV vaccine does not cause HPV infection or cancer. HPV vaccine is made from one protein from the virus, and is not infectious, meaning that it cannot cause HPV infection or cancer. Not receiving HPV vaccine at the recommended ages can leave one vulnerable to cancers caused by HPV.
There are no data that suggest getting HPV vaccine will have an effect on future fertility for women. In fact, getting vaccinated and protecting against HPV-related cancers can help women and families have healthy pregnancies and healthy babies.
Not getting HPV vaccine leaves people vulnerable to HPV infection and related cancers. Treatments for cancers and precancers might include surgery, chemotherapy, and/or radiation, which might cause pregnancy complications or leave someone unable to have children.
Based on the review of available information by FDA and CDC, Gardasil continues to be safe and effective, and its benefits continue to outweigh its risks 7).
CDC has not changed its recommendations for use of Gardasil 8). FDA has not made any changes to the prescribing information for how the vaccine is used 9). In addition, FDA routinely reviews manufacturing information, and has not identified any issues affecting the safety, purity and potency of Gardasil.
Public health and safety are priorities for FDA and CDC. As with all licensed vaccines, they will continue to closely monitor the safety of Gardasil. FDA and CDC continue to find that Gardasil is a safe and effective vaccine that will potentially benefit the health of millions of women by providing protection against the types of HPV in the vaccine that cause cervical, vulvar and vaginal cancer, genital warts, and other HPV-related genital diseases in females 10).
Gardasil risks
With any medicine, including vaccines, there is a chance of side effects. These are usually mild and go away on their own, but serious reactions are also possible.
Most people who get HPV vaccine do not have any serious problems with it.
Mild or moderate problems following HPV vaccine:
Reactions in the arm where the shot was given:
- Soreness (about 9 people in 10)
- Redness or swelling (about 1 person in 3)
Fever:
- Mild (100°F or 37.8 °C) (about 1 person in 10)
- Moderate (102°F or 38.9 °C) (about 1 person in 65)
Other problems:
- Headache (about 1 person in 3)
Problems that could happen after any injected vaccine:
- People sometimes faint after a medical procedure, including vaccination. Sitting or lying down for about 15 minutes can help prevent fainting and injuries caused by a fall. Tell your doctor if you feel dizzy, or have vision changes or ringing in the ears.
- Some people get severe pain in the shoulder and have difficulty moving the arm where a shot was given. This happens very rarely.
- Any medication can cause a severe allergic reaction. Such reactions from a vaccine are very rare, estimated at about 1 in a million doses, and would happen within a few minutes to a few hours after the vaccination.
As with any medicine, there is a very remote chance of a vaccine causing a serious injury or death.
What if there is a serious reaction?
If you think it is a severe allergic reaction or other emergency that can’t wait, call your local emergency number or get to the nearest hospital. Otherwise, call your doctor.
Afterward, the reaction should be reported to the Vaccine Adverse Event Reporting System (VAERS). Your doctor should file this report, or you can do it yourself through the VAERS website (https://vaers.hhs.gov/).
Gardasil vaccine deaths
Across the clinical studies, ten deaths occurred (five each in the Gardasil 9 and Gardasil groups); none were assessed as vaccine-related 11). Causes of death in the Gardasil 9 group included one automobile accident, one suicide, one case of acute lymphocytic leukemia, one case of hypovolemic septic shock, and one unexplained sudden death 678 days following the last dose of Gardasil 9. Causes of death in the Gardasil control group included one automobile accident, one airplane crash, one cerebral hemorrhage, one gunshot wound, and one stomach adenocarcinom 12).
References [ + ]