Contents
What is hemangioma
A hemangioma is a benign benign overgrowth of vascular tissue on the skin. Hemangioma (also called angioma) is due to proliferating endothelial cells; these are the cells that line the inside of a blood vessel.
The hemangioma may be filled with oxygenated blood and red, or deoxygenated blood and purple. The later may be called a venous hemangioma.
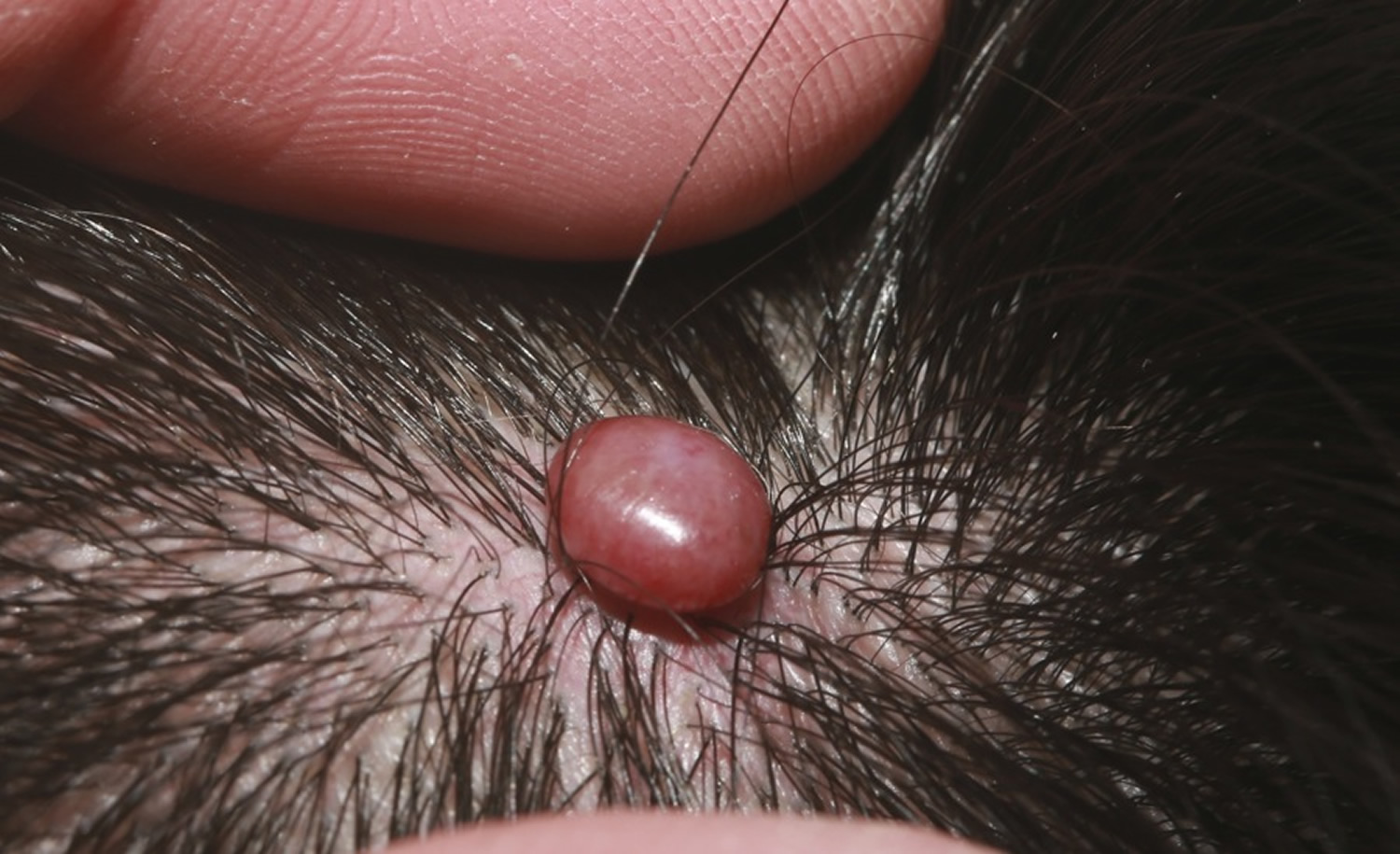
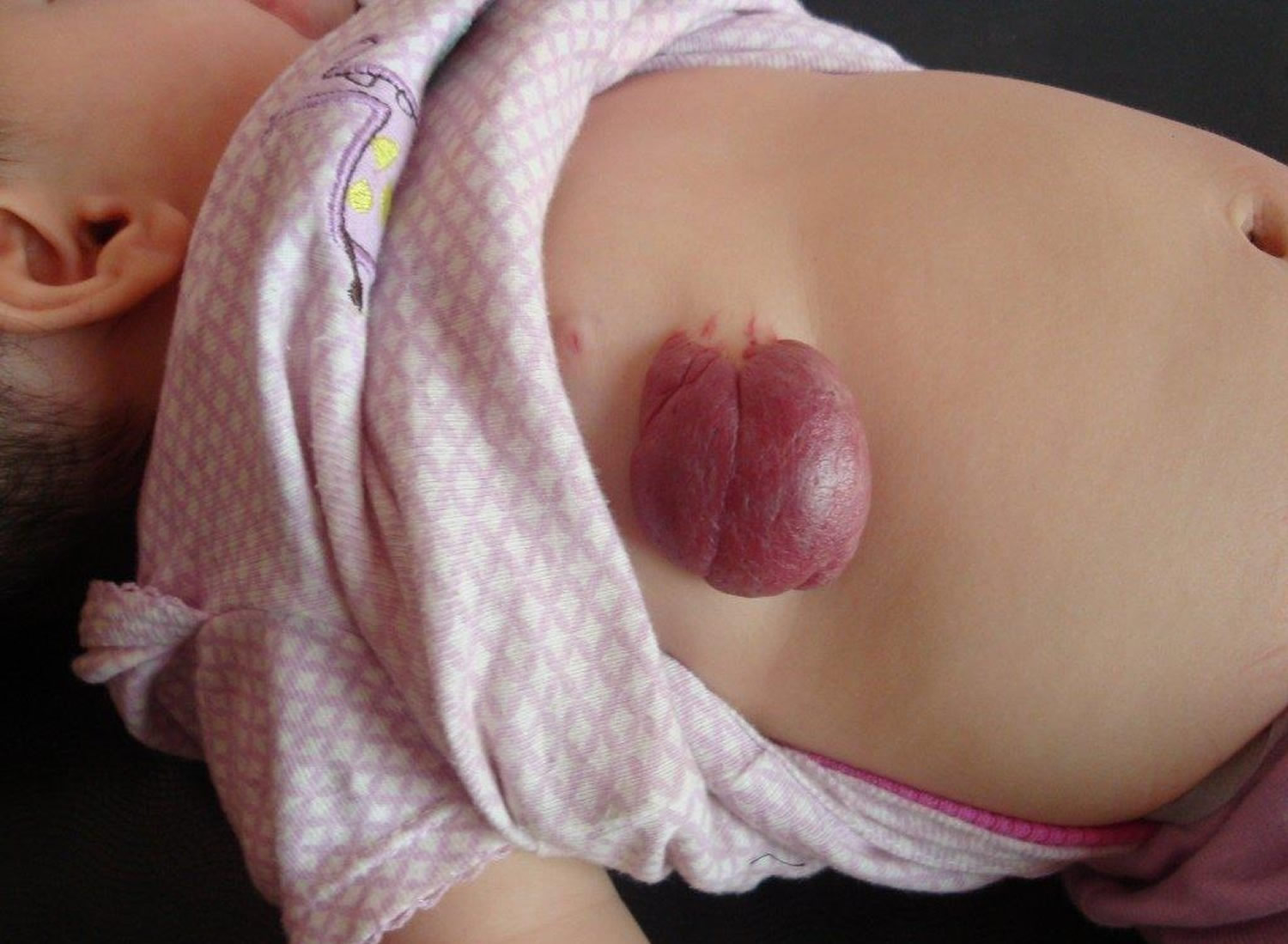
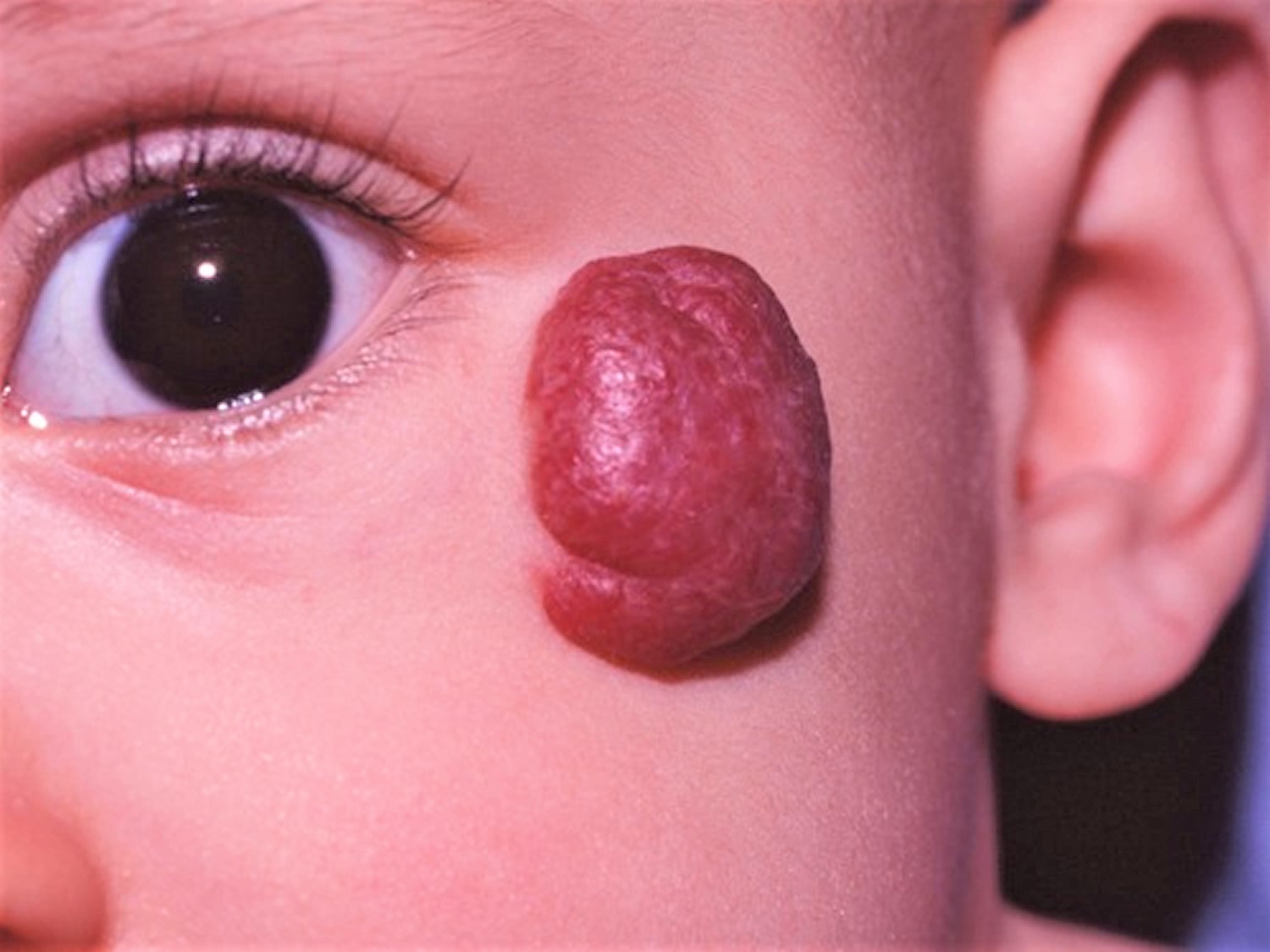
A red to purple vascular skin lump is typical of a hemangioma of skin (see Figures 1 and 2). Compressing the lesion and squeezing out blood can help confirm the diagnosis. In discriminating a venous hemangioma from a pigmented lesion, the presence of purple color is key.
Hemangiomas are usually painless, red to blue colored lesions on the skin, lips, or inside the mouth. They are often soft to the touch. Most often they are flush with the skin or slightly elevated, but sometimes they grow from a stalk. Superficial lesions may bleed or turn into sores, particularly if bumped or injured.
Deep hemangiomas in muscle may cause pain, as well as swelling around the hemangioma that increases with activity.
Hemangiomas in bones may cause pain and enlargement of the bone.
Figure 1. Hemangioma of skin
Figure 2. Hemangioma
Hemangioma causes
Different types of hemangioma are associated with different causes, although exactly why hemangiomas develop is not well understood. For example, infantile hemangiomas are caused by errors in the development of the vascular system that occur during fetal development, but in many cases, the event that caused the error cannot be identified. Some hemangiomas develop after an injury, but whether an injury actually can cause a hemangioma has not been proven. Some hemangiomas develop with pregnancy and go away afterward. Some hemangiomas are associated with genetic abnormalities (for example, cavernous hemangiomas in von Hippel-Lindau disease).
There has been no proven connection between the development of hemangiomas and any particular occupation or exposure to chemicals or radiation. In addition, no known food, medication, or activity during pregnancy is known to cause a hemangioma in an infant.
Hemangioma diagnosis
Hemangiomas can be confused with other vascular malformations. Because treatment may vary considerably depending on whether the lesion is a hemangioma or other type of malformation, a thorough doctor’s examination is recommended.
Before a physical examination, your doctor will talk with you about your general health and current condition — or that of your child’s, if you are a parent or family member. He or she will want to get a good history of the lesion, particularly how long the mass has been there and whether it has changed at all. Your doctor will want to know what symptoms — such as pain — are associated with the lesion, and when the symptoms began.
During the physical examination, your doctor will inspect and palpate any mass, noting exactly where it is located, what it feels like, and perhaps any surrounding changes in the skin.
Tests
Although doctors can often diagnose hemangiomas based on medical history and physical examination alone, imaging tests can be helpful.
X-rays and computed tomography (CT) scans. Although these tests create better pictures of dense structures like bone, plain x-rays and CT scans may show a cavernous hemangioma if it has calcifications. These calcifications are called phleboliths.
Magnetic resonance imaging (MRI) scans. These scans can create clear images of soft structures like a hemangioma. Hemangiomas in an MRI scan are often described as a “bag of worms.”
Angiogram. In this test, dye is injected into the surrounding blood stream making the hemangioma show up in an x-ray image.
The imaging and tissue tests help your doctor differentiate hemangioma from other types of vascular malformations and soft-tissue tumors.
It is very important in planning treatment to differentiate hemangioma from more aggressive, cancerous vascular tumors like angiosarcoma.
Biopsy. It may be difficult to distinguish hemangiomas from other tumors, and a biopsy is sometimes necessary to confirm a diagnosis of hemangioma. In a biopsy, a tissue sample of the tumor is taken and examined under a microscope. Looking at the tissue under a microscope may be the best way to tell if the tumor is indeed a hemangioma and what sub-type it is.
During the biopsy, your doctor may give you a local anesthetic to numb the area and take a sample using a needle. Biopsies can also be performed as a small operation.
Blood tests. If there are multiple tumors or your symptoms are concerning for a specific disease pattern, your doctor may recommend blood tests for genetic analysis.
Hemangioma treatment
Treatment varies slightly by hemangioma subtype. Most hemangiomas are benign and need not be treated. Flat or only slightly raised lesions often are not bothersome. Simple cautery after lidocaine (local anesthetic) infiltration may be done. Shave excision with histologic examination should be considered if the diagnosis is in question as amelanotic melanoma may occasionally appear similarly.
Hemangiomas usually do not respond to cryotherapy. Pedunculated lesions, like the one pictured above (Figure 1), are easily snipped after infiltration of the underlying skin with lidocaine.
Nonsurgical Treatment
Observation. Although a hemangioma may not require any treatment, it is important to visit your doctor regularly to monitor any changes in the tumor.
Anti-inflammatory medication. If a tumor is growing near vital structures, such as the nose, lips, or eyelids, you doctor may recommend steroid medication. Steroids are often used to slow down the growth of the tumor. The drugs may be injected directly into the hemangioma, or given orally (in pill form).
Compression. Intermittent pneumatic compression is a treatment that uses inflatable sleeves or leggings to apply pressure to the tumor. It can be useful to decrease the swelling associated with a hemangioma. It will not, however, make the hemangioma go away.
Embolization. In this procedure, the blood supply to the tumor is closed off. This is a minimally invasive procedure where small particles are injected into the blood vessels to block them off. Sclerotherapy is a similar procedure where chemical agents are used to close off the vessels. These procedures can be very helpful in shrinking the tumor and decreasing pain. Often, however, the tumor will regrow its blood supply over time after these procedures. Embolization is also sometimes used prior to surgery to reduce the risk of heavy blood loss.
Surgical Treatment
Surgery may be recommended with a cavernous hemangioma if the lesion is destroying the healthy tissues surrounding it. In some cases, a hemangioma can cause painful symptoms severe enough to consider surgical treatment.
Procedure. The procedure to surgically remove a hemangioma is called an excision. General anesthesia is used to put you to sleep, then your doctor will make an incision in your skin and cut the tumor out.
Recovery. You will have a few stitches that your doctor will remove within a few weeks. Your doctor will wrap the area with a tight, compressive bandage and provide you with specific instructions about activity restrictions to guide your recovery.
Complications. The most common complication of surgery to remove a hemangioma is hemorrhage (blood loss). In addition, hemangiomas have a high tendency to come back after surgery, depending upon the type and location of the tumor.
Capillary hemangioma
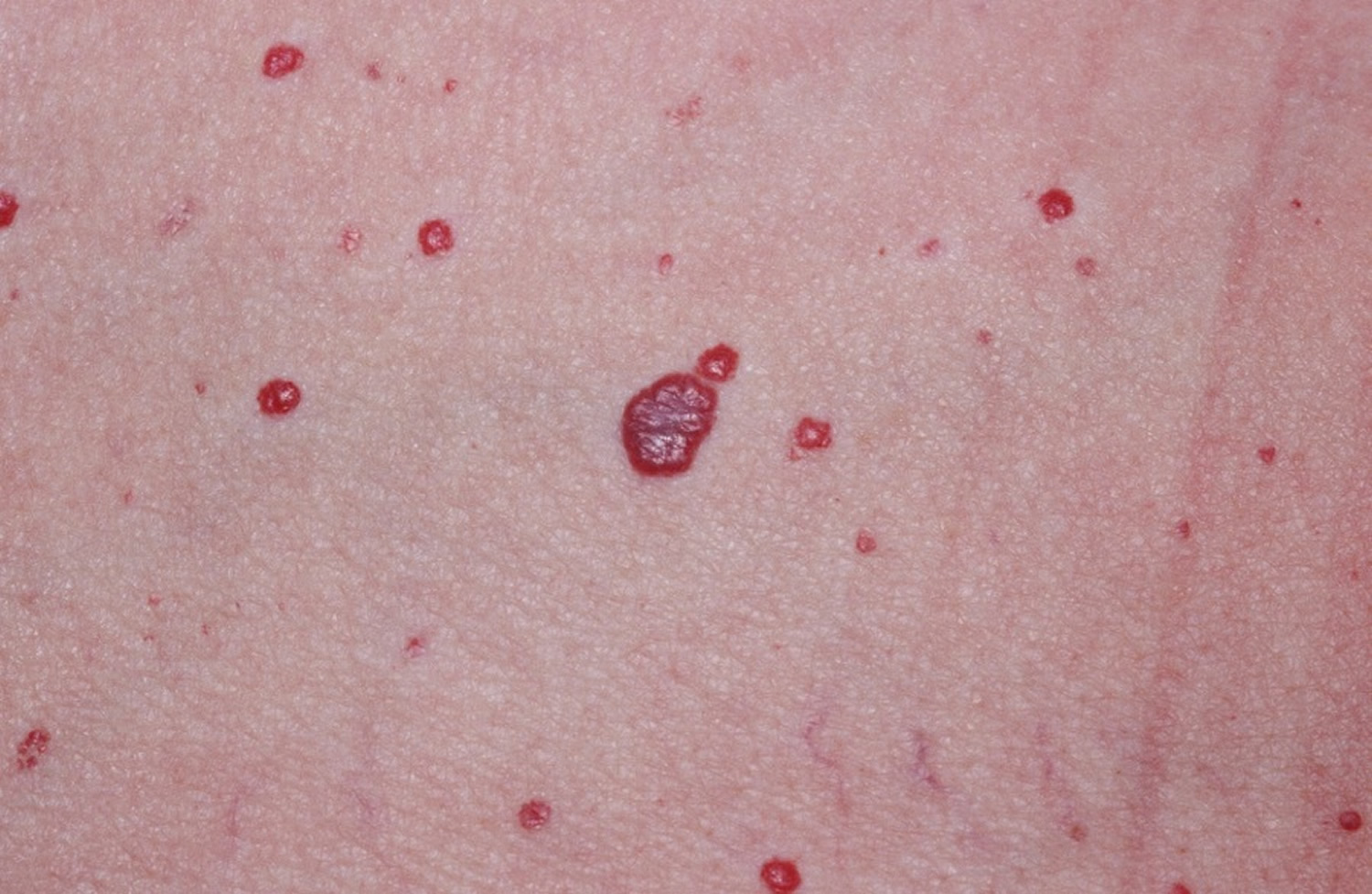
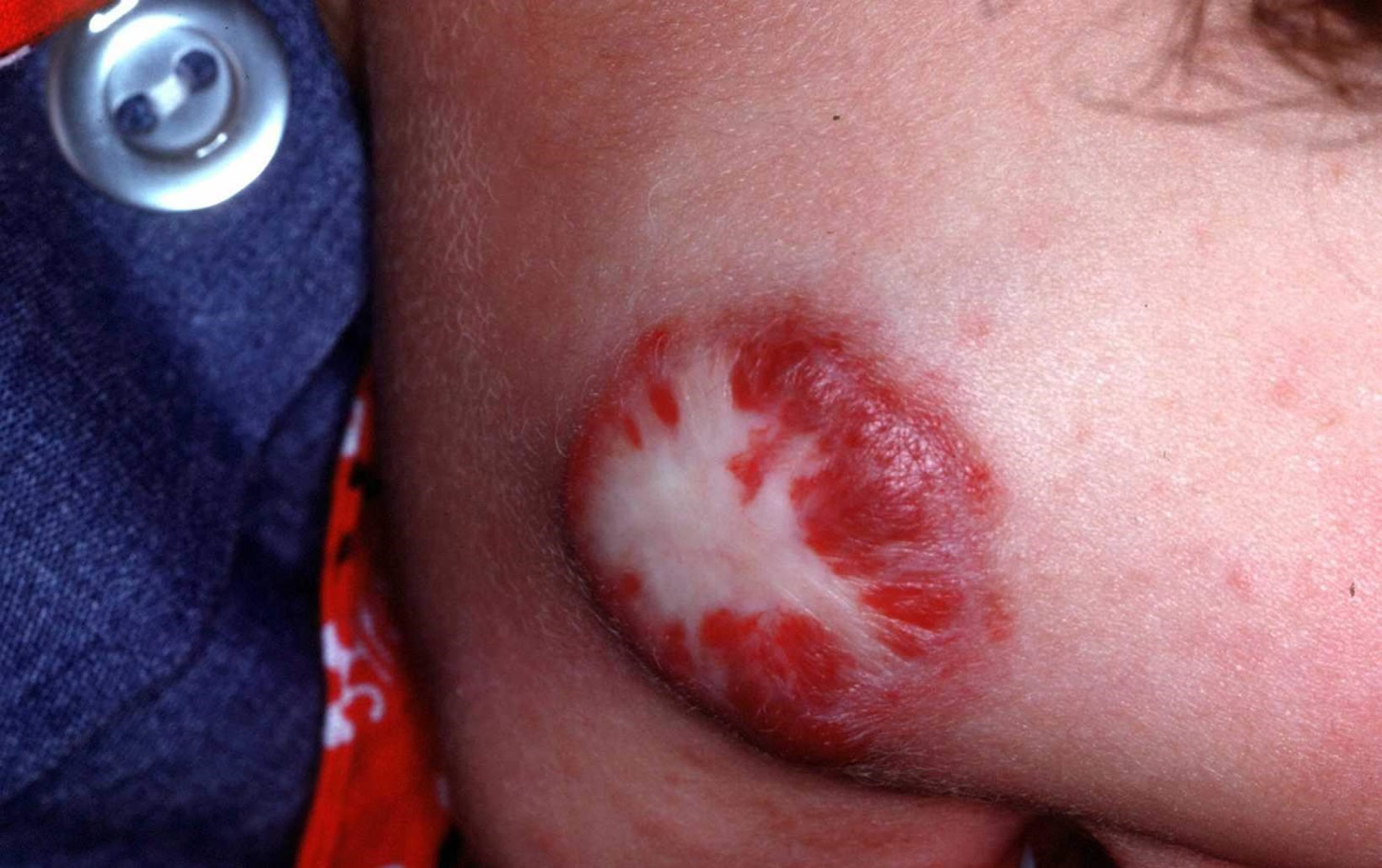
The cherry hemangioma or capillary hemangioma is an extremely common, benign vascular bleb on the skin of most humans. Capillary hemangioma (cherry hemangioma) usually appear as small, dome-shaped or slightly raised papules (Figure 3). Sometimes, they appear almost “petechial” as 1-2 mm. red macules. Most hemangiomas are cherry red, but some have enough deoxygenated venous blood to appear purple.
Capillary hemangiomas may not be present at birth, but appear within the first 6 months of life. They usually begin to decrease in size between 12 and 15 months of age. Most regress nearly completely by 5 or 6 years of age. Capillary hemangiomas are more common in premature infants and in girls.
- Most adults over 30 have one or more red vascular “blood blisters” on the trunk.
- In one study of adults 30-39 years of age, 90% of the men and 65% of the women had at least one cherry hemangioma.
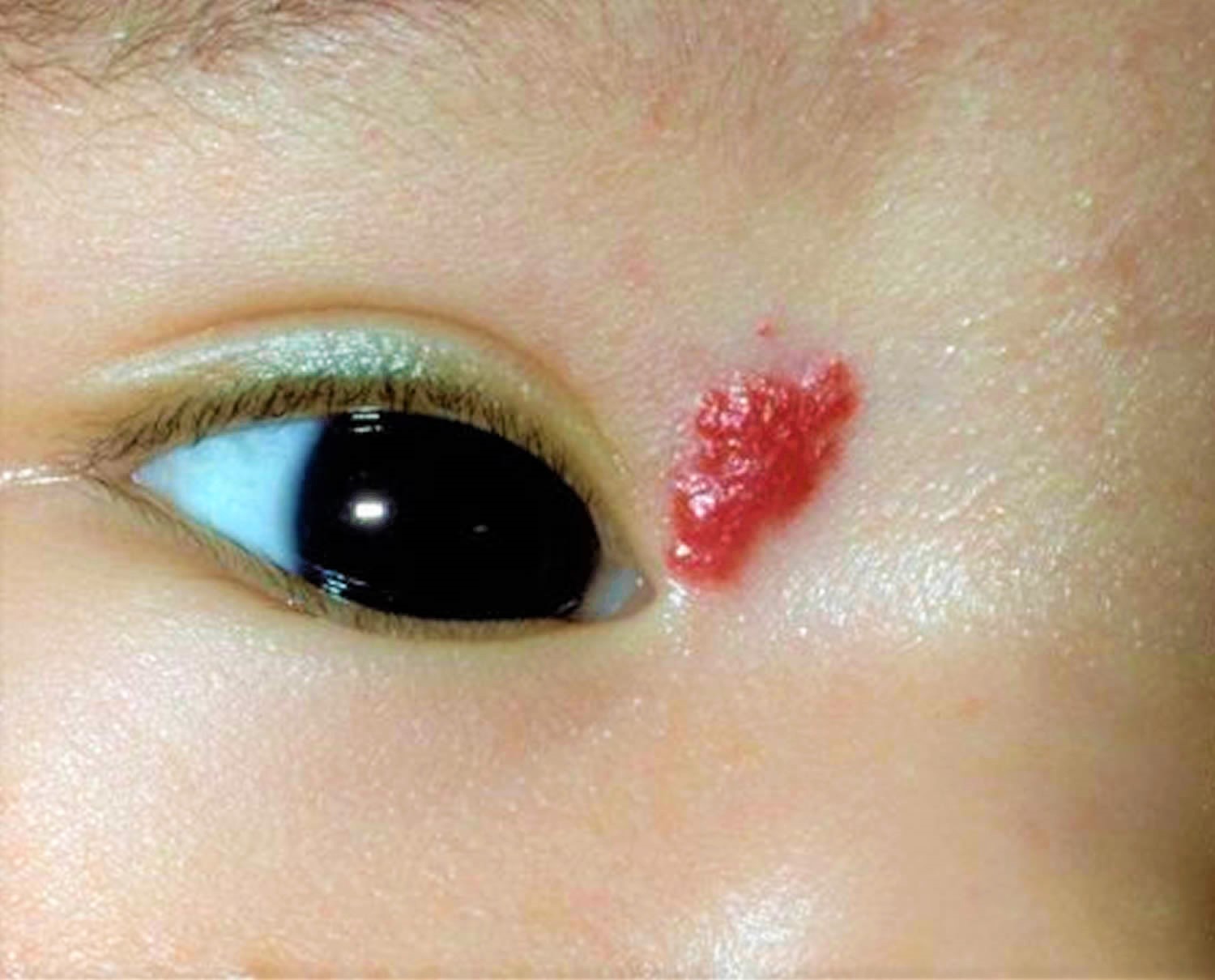
Capillary hemangiomas can be found anywhere on the body. Eye involvement includes eyelids, the eye surface called the conjunctiva, and the eye socket or orbit.
A capillary hemangioma in the eye socket (the area around the eye called the orbit) can put pressure on the eye and result in amblyopia or strabismus. It can also press on the optic nerve which may cause optic nerve atrophy. Any of these conditions may damage vision. However, the majority of capillary hemangiomas around the eye do not require treatment. They are simply monitored for the development of vision problems. Treatment is needed only if there is a threat to vision.
Capillary Hemangiomas of the eyelid can cause amblyopia (decreased vision) by two mechanisms. First, if the lesion exerts pressure on the surface of the eye, this causes astigmatism and the baby may develop refractive amblyopia. Second, if the lesion causes the eyelid to droop significantly (ptosis) to the extent that it blocks vision in the eye, this can result in occlusion amblyopia.
If amblyopia develops in the affected eye, it can be treated with glasses and/or occlusion therapy using patches or atropine drops.
How are capillary hemangiomas treated?
Capillary hemangiomas can be treated in a variety of ways depending on the location of the hemangioma, its severity, and whether or not it is causing vision problems.
Propranolol is an effective medication that is used in the treatment of hemangiomas. Usually it is taken orally but in some cases it can be applied topically if the hemangioma is very small and thin. Since propranolol can affect heart rate and blood pressure, careful monitoring at the beginning of treatment is sometimes required and may include a brief admission to the hospital.
Steroid medications can also be used to treat hemangiomas. Steroids can stop the progression of hemangiomas by causing the blood vessels to shrink. Depending on the size and location of the hemangioma, steroids may be prescribed by mouth, injected directly into the hemangioma, or applied to the surface of the hemangioma. Steroid medications can have undesirable side effects including delayed physical growth, cataract, glaucoma and central retinal artery occlusion.
Laser treatments can sometimes be used on superficial hemangiomas to prevent growth, diminish their size, or lighten their color.
Traditional surgery to remove hemangiomas around the eye is generally reserved for small, well-defined hemangiomas that are located under the skin surface.
How long does it take for the capillary hemangioma to go away?
Capillary hemangiomas typically take several years to regress. The involved skin may retain a red color or may be slightly puckered in appearance, or may look perfectly normal depending upon how completely the hemangioma resolves.
Figure 3. Capillary hemangioma (cherry hemangioma)
Cavernous hemangioma
A cavernous hemangioma is a cluster of abnormal blood vessels, usually found in the brain and spinal cord. They’re sometimes known as cavernous angiomas, cavernoma or cerebral cavernous malformation.
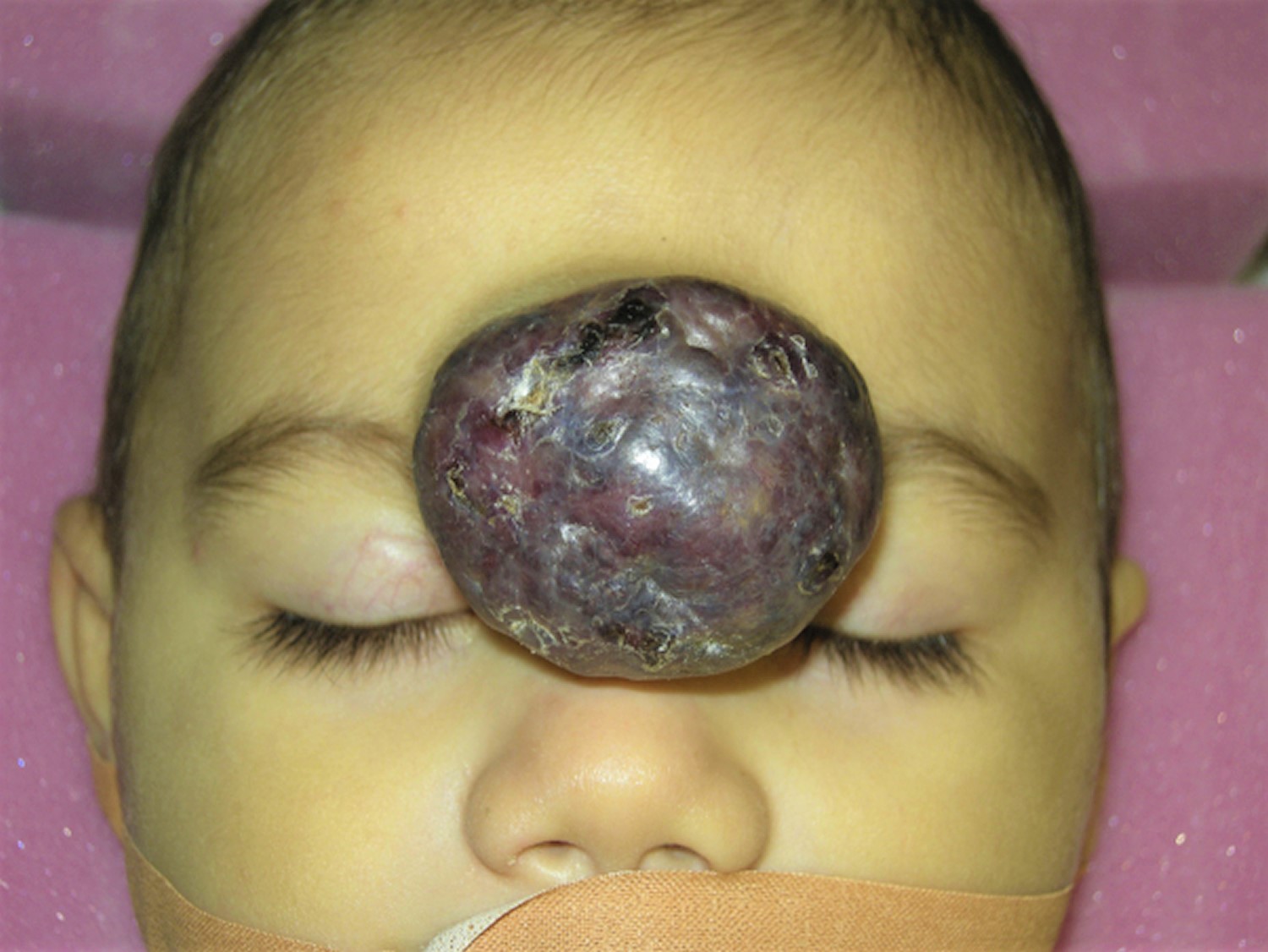
A typical cavernous hemangioma looks like a raspberry. It’s filled with blood that flows slowly through vessels that are like “caverns”. A cavernous hemangioma can vary in size from a few millimeters to several centimeters across.
It’s estimated about 1 in every 600 people in the US has a cavernous hemangioma that doesn’t cause symptoms.
Every year, around 1 person in every 400,000 in the US is diagnosed with a cavernous hemangioma that has caused symptoms.
If symptoms do occur, most people will develop them by the time they reach their 30s.
Cavernous hemangioma causes
In most cases, there’s no clear reason why a person develops a cavernous hemangioma. The condition can sometimes run in families – less than 50% of cases are thought to be genetic.
However, in most cases cavernous hemangiomas occur randomly. Genetic testing can be carried out to determine whether a cavernous hemangioma is genetic or whether it’s occurred randomly.
Some cavernous hemangioma cases have also been linked to radiation exposure, such as previously having radiotherapy to the brain, usually as a child.
Symptoms of cavernous hemangioma
A cavernous hemangioma often doesn’t cause symptoms, but when symptoms do occur they can include:
- bleeding (hemorrhage)
- fits (seizures)
- headaches
- neurological problems, such as dizziness, slurred speech (dysarthria), double vision, balance problems and tremor
- weakness, numbness, tiredness, memory problems and difficulty concentrating
- a type of stroke called a hemorrhagic stroke
The severity and duration of symptoms can vary depending on the type of cavernous hemangioma and where it’s located.
Problems can occur if the cavernous hemangioma bleeds or presses on certain areas of the brain. The cells lining a cavernous hemangioma are often thinner than those that line normal blood vessels, which means they’re prone to leaking blood.
In most cases, bleeding is small – usually around half a teaspoonful of blood – and may not cause other symptoms. But severe hemorrhages can be life threatening and may lead to longlasting problems.
You should seek medical help as soon as possible if you experience any of the above symptoms for the first time.
What are the chances of a cavernous hemangioma bleeding?
The risk of having a hemorrhage varies from person to person, depending on whether you have experienced any bleeding before.
If you haven’t had any bleeding before, it’s estimated you have a less than 1% chance of experiencing a hemorrhage each year.
If your cavernous hemangioma has bled previously, your risk of having another hemorrhage is somewhere between 4% and 25% each year.
However, this risk decreases progressively over time if you don’t experience any further bleeds, and eventually returns to the same level as that of people who haven’t had any bleeding before.
Your level of risk will be one of the main factors taken into consideration when deciding if you would benefit from treatment.
Cavernous hemangioma diagnosis
Magnetic resonance imaging (MRI) scans are mainly used to diagnose cavernous hemangiomas. As symptoms aren’t always evident, many people are only diagnosed with a cavernous hemangioma after having an MRI scan for another reason.
A computerised tomography (CT) scan or angiography can also be used to diagnose cavernous hemangioma, but they’re not as reliable as an MRI scan.
Monitoring your symptoms
Any symptoms you have may come and go as the cavernous hemangioma bleeds and then reabsorbs blood. It’s important to closely monitor your symptoms, as any new symptoms might be a sign of a hemorrhage.
Your doctor can advise you about what to do if you experience any new or worsening symptoms. They may also recommend having a further brain scan.
MRI and CT scans can be used to detect bleeding on the brain, although they can’t necessarily identify cavernous hemangiomas that are at an increased risk of bleeding.
This is because the features of a cavernous hemangioma that can be seen on a brain scan, such as an increase in size, don’t appear to be directly linked to the likelihood of bleeding.
Although cavernous hemangiomas can get bigger, large cavernous hemangiomas aren’t any more likely to bleed than smaller ones.
Treating cavernous hemangioma
The recommended treatment for cavernous hemangioma will vary depending on an individual’s circumstances and factors such as size, location and number.
Some cavernous hemangioma symptoms, such as headaches and seizures, can be controlled with medication.
However, more invasive treatment may sometimes be offered to reduce the risk of future hemorrhages. The decision to have such treatment is made on a case-by-case basis in discussion with your doctor.
Types of treatment offered to reduce the risk of hemorrhages include:
- neurosurgery – carried out under general anaesthetic to remove the cavernous hemangioma
- stereotactic radiosurgery – where a single, concentrated dose of radiation is aimed directly at the cavernous hemangioma, causing it to become thickened and scarred
In most cases, neurosurgery is preferred to stereotactic radiosurgery because the effectiveness of radiosurgery in preventing hemorrhages is unknown.
Stereotactic radiosurgery is usually only considered if the position of the cavernous hemangioma makes neurosurgery too difficult or dangerous.
Risks of invasive treatment include stroke and death, although the exact risks depend on the location of the cavernous hemangioma. You should discuss the possible risks of treatment with your doctor beforehand.
Hemangioma liver
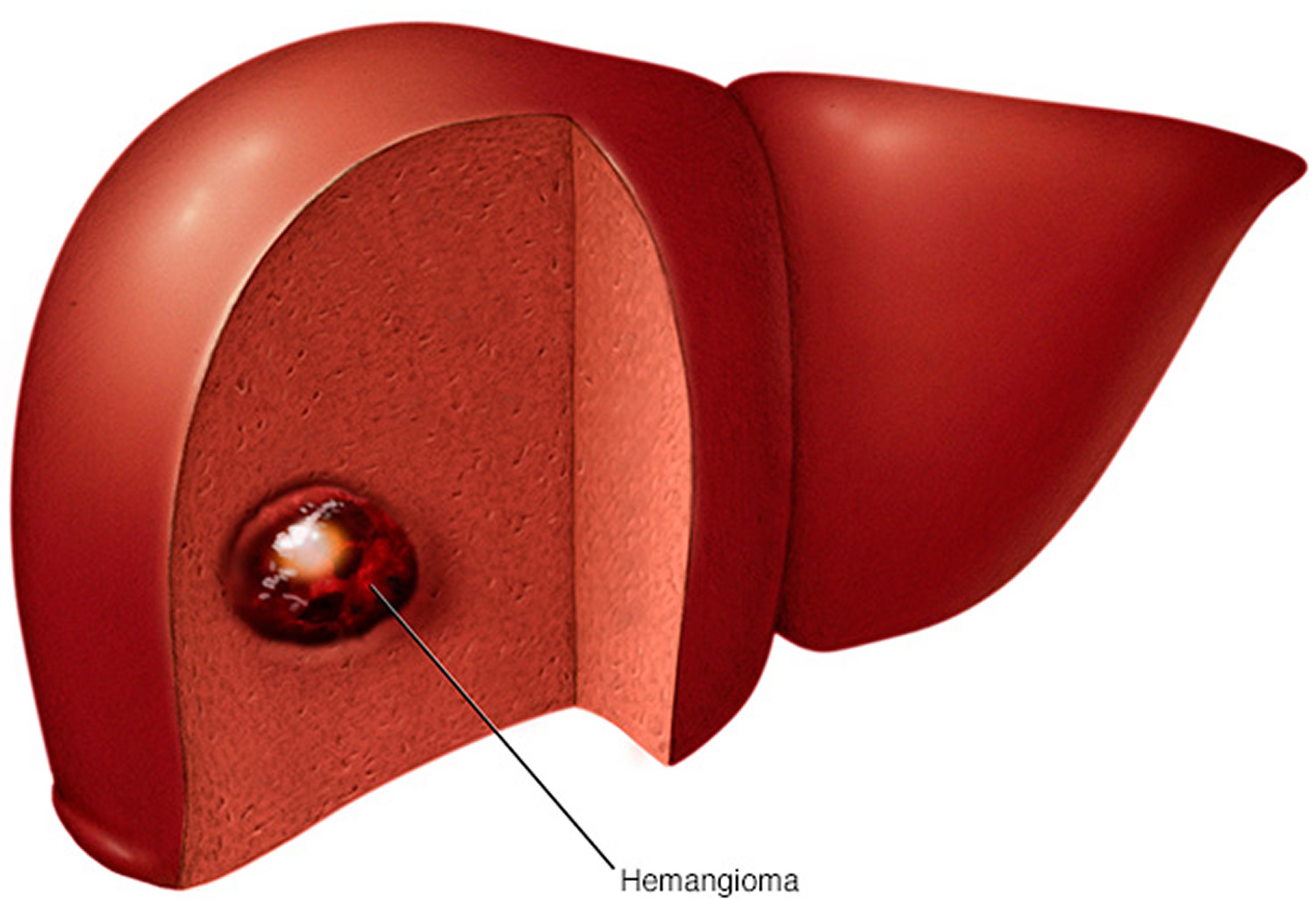
A liver hemangioma (also called hepatic hemangioma) is a noncancerous (benign) mass in the liver. A liver hemangioma is made up of a tangle of blood vessels.
Most cases of liver hemangiomas are discovered during a test or procedure for some other condition. People who have a liver hemangioma rarely experience signs and symptoms and don’t need treatment.
It may be unsettling to know you have a mass in your liver, even if it’s a benign mass. There’s no evidence that an untreated liver hemangioma can lead to liver cancer.
Figure 4. Liver hemangioma
Liver hemangioma symptoms
In most cases, a liver hemangioma doesn’t cause any signs or symptoms.
When a liver hemangioma causes signs and symptoms, they may include:
- Pain in the upper right abdomen
- Feeling full after eating only a small amount of food
- Nausea
- Vomiting
However, these symptoms are nonspecific and may be due to something else, even if you have a liver hemangioma.
What cause liver hemangioma?
It’s not clear what causes a liver hemangioma to form. Doctors believe liver hemangiomas are congenital — meaning that you’re born with them.
A liver hemangioma usually occurs as a single abnormal collection of blood vessels that is less than about 1.5 inches (about 4 centimeters) wide. Occasionally liver hemangiomas can be larger or occur in multiples. Large hemangiomas can occur in young children but this is very rare.
In most people, a liver hemangioma will never grow and never cause any signs and symptoms. But in a small number of people, a liver hemangioma will grow to cause complications and require treatment. It’s not clear why this happens.
Risk factors for liver hemangioma
Factors that can increase the risk that a liver hemangioma will cause signs and symptoms include:
- Your age. A liver hemangioma can be diagnosed at any age, but it’s most commonly diagnosed in people ages 30 to 50.
- Your sex. Women are more likely to be diagnosed with a liver hemangioma than men are.
- Pregnancy. Women who have been pregnant are more likely to be diagnosed with a liver hemangioma than women who have never been pregnant. It’s believed the hormone estrogen, which rises during pregnancy, may play a role in liver hemangioma growth.
- Hormone replacement therapy. Women who used hormone replacement therapy for menopausal symptoms may be more likely to be diagnosed with a liver hemangioma than women who did not.
Liver hemangioma complications
Women who have been diagnosed with liver hemangiomas face a risk of complications if they become pregnant. The female hormone estrogen, which increases during pregnancy, is believed to cause some liver hemangiomas to grow larger.
Rarely, growing hemangioma can cause signs and symptoms that may require treatment, including pain in the upper right quadrant of the abdomen, abdominal bloating or nausea. Having a liver hemangioma doesn’t mean you can’t become pregnant. However, discussing the possible complications with your doctor can help you make a more informed choice.
Medications that affect hormone levels in your body, such as birth control pills, could cause complications if you’ve been diagnosed with a liver hemangioma. But this is controversial. If you’re considering this type of medication, discuss the benefits and risks with your doctor.
Liver hemangiomas diagnosis
Tests and procedures used to diagnose liver hemangiomas include:
- Ultrasound
- Computerized tomography scan
- Magnetic resonance imaging
Other tests and procedures may be used depending on your situation.
Treatment for liver hemangiomas
If your liver hemangioma is small and doesn’t cause any signs or symptoms, you won’t need treatment. In most cases a liver hemangioma will never grow and will never cause problems. Your doctor may schedule follow-up exams to check your liver hemangioma periodically for growth if the hemangioma is large.
Liver hemangioma treatment depends on the location and size of the hemangioma, whether you have more than one hemangioma, your overall health, and your preferences.
Treatment options may include:
- Surgery to remove the liver hemangioma. If the hemangioma can be easily separated from the liver, your doctor may recommend surgery to remove the mass.
- Surgery to remove part of the liver, including the hemangioma. In some cases, surgeons may need to remove a portion of your liver along with the hemangioma.
- Procedures to stop blood flow to the hemangioma. Without a blood supply, the hemangioma may stop growing or shrink. Two ways to stop the blood flow are tying off the main artery (hepatic artery ligation) or injecting medication into the artery to block it (arterial embolization). Healthy liver tissue is unharmed because it can draw blood from other nearby vessels.
- Liver transplant surgery. In the unlikely event that you have a large hemangioma or multiple hemangiomas that can’t be treated by other means, your doctor may recommend surgery to remove your liver and replace it with a liver from a donor.
- Radiation therapy. Radiation therapy uses powerful energy beams, such as X-rays, to damage the cells of the hemangioma. This treatment is rarely used because of the availability of safer and more effective treatments.
Hemangioma spine
Spinal hemangiomas (also called vertebral hemangiomas) are common, benign vascular lesions that involve the bony portion of vertebral bodies and are generally asymptomatic 1. First described by Perman, in 1926, followed by Bailey and Bucy in 1930, spinal hemangiomas are common benign lesions of the spinal column with an estimated incidence of 10–12% based on large autopsy series and reviews of spine radiographs 1. Although typically incidental findings, they are symptomatic in 0.9 to 1.2% of adults 2. This figure is much lower in the pediatric population with only five cases reported in the literature. Rarely, spinal hemangiomas can become aggressive and present with predominantly epidural extension – extra-osseous extension, bone expansion, disturbance of blood flow, and occasionally compression fractures and thereby referred to as aggressive hemangiomas, mimicking other neoplasms 3. Approximately 45% of aggressive hemangiomas are associated with neurologic deficits, the others only characterized by pain 4.
Aggressive vertebral hemangiomas most often occur between T3 and T9 vertebral segments 5. They generally occupy the entire vertebral body, extend into the neural arch, expand the osseous margins, and contain a soft tissue component 5. Cord compression and subsequent myelopathy may result from either encroachment of extradural soft tissue, pathologic fracture, or hemorrhage 6. Vertebral hemangiomas may become symptomatic during pregnancy due to increased intraabdominal pressure and vascular redistribution of flow in the vertebral venous plexus when the uterus enlarges 7.
The histologic pattern of spinal hemangiomas is characterized by the proliferation of anomalous thin-walled blood vessels and sinuses lined by endothelium between the thickened, vertically oriented trabeculae of bone. The dilated vascular channels are set in a stroma of fat. The ratio of fat to vascular tissue wrapped between the pillars of bone determines the signal intensity on MR images 8.
Table 1: Summary table for aggressive hemangioma of spine (vertebral hemangioma)
| Etiology | Debated. Likely multifactorial. |
| Incidence | 10–12% of adult population |
| Gender ratio | Slightly more common in women |
| Age predilection | Middle aged adults |
| Risk factors | Rarely associated with chromosomal abnormalities |
| Treatment | Embolization, Sclerotherapy, Radiation therapy, Surgery, and Vertebroplasty |
| Prognosis | Variable, depending on degree of spinal cord involvement. |
| Findings on imaging |
|
Treatment options for symptomatic or aggressive hemangiomas without cord compression include vertebroplasty 7, embolization, and sclerotherapy 9. In a recent case series of four patients, Urrutia et al. demonstrated that surgical resection is a relatively safe method for treatment of compressive vertebral hemangiomas 10. Radiation therapy may be used in those cases of subtotal resection 11. Adjuvant radiotherapy can reduce the recurrence of cavernous vertebral hemangiomas (Surgical treatment of compressive spinal hemangioma. A case series of three patients and literature review. Orthopäde (2017). https://doi.org/10.1007/s00132-017-3503-6)).
Infantile hemangioma
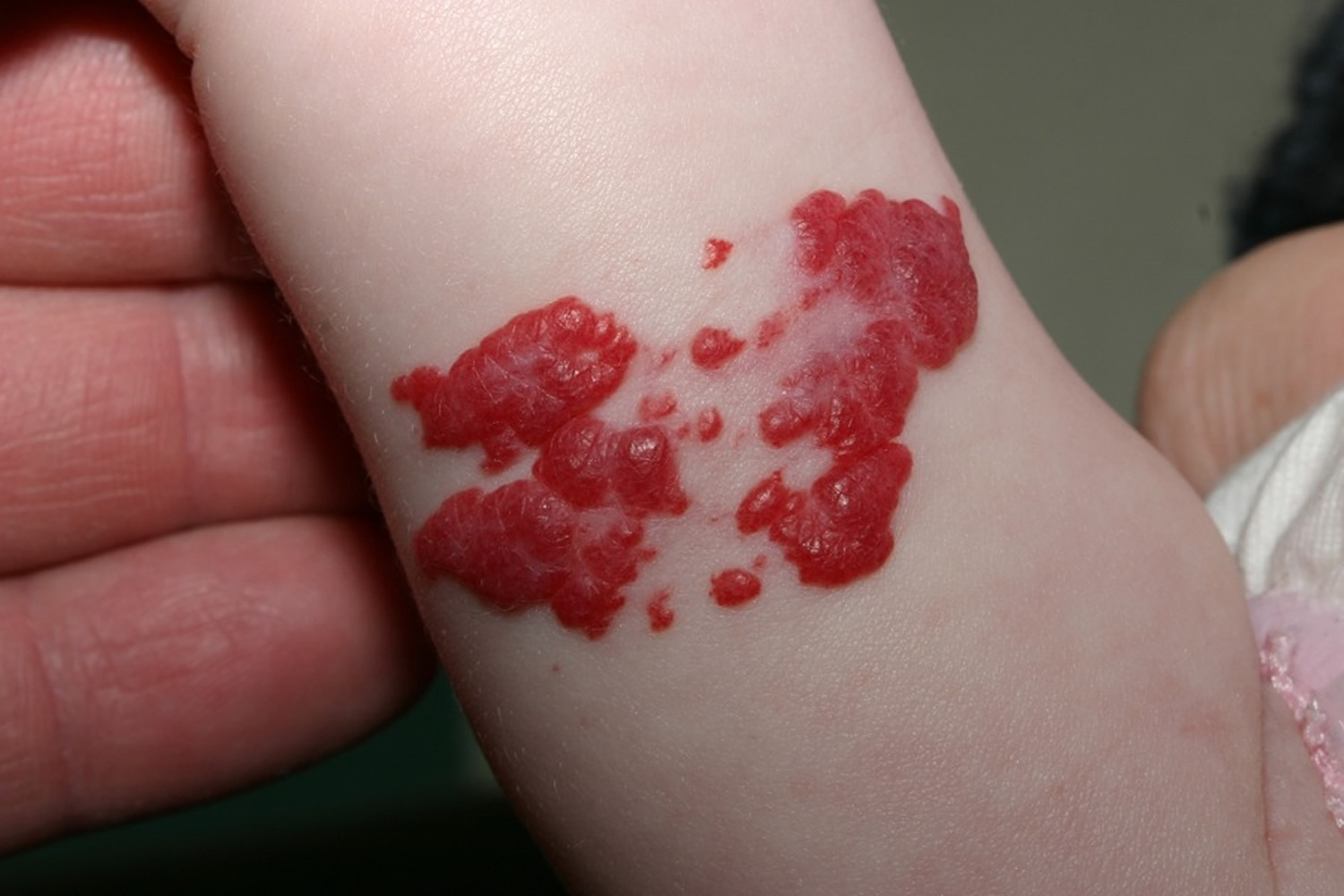
Infantile hemangioma is a vascular proliferation occurring in infancy. Infantile hemangiomas usually present during the first month of life. They may be present at birth, but more commonly the skin at birth shows a mild abnormality (e.g. a barely visible, white, anemic stain, a faint, telangiectatic lesion, a blue spot mimicking a bruise). Over the ensuing weeks and months, the lesion enlarges. One study showed that 80% of the growth occurs in the first 3 months with the greatest in the second month of life. Infantile hemangioma lesions may ulcerate. Although the hemangiomas in baby usually decrease in size between ages 2 and 10 years, a single study found that most of these infantile hemangiomas do not regress after age 3.5 years.
Ulceration may occur in approximately 5% of infantile hemaingioma, especially when the lesion is located in the lips or anogenital area. A white discoloration of skin may presage ulceration. Even though the lesions is highly vascularized it may not do a good job of oxygenating the overlying skin. Ulcerated hemangiomas may bleed. Compression is usually sufficient. Rarely, serious, life-threatening hemorrhage can occur with large hemangiomas.
Routine cleansing followed by a topical barrier cream, e.g. zinc oxide-containing is recommended. Healing should occur within 2-3 weeks. Topical timolol helps heal the lesion (see hemangioma, infantile) [JAAD 2016;74;567]. It may be prescribed as 1-2 drops of timolol 0.5% BID. It topical timolol is not effective, oral propranolol or nadolol may be employed. Pain control is often needed. e.g. acetaminophen, ibuprofen, topical lidocaine, oral opiate-derivatives such as morphine. Topical antibiotics (and if needed oral) are sometimes given if infection is suspected/documented. Significant bleeding usually does not occur, but if it does, compression is recommended as first line treatment.
For ulcerated hemangiomas on the extremities, saline or Burrow’s compressed followed by an antibiotic ointment, adherent pad and a compressive wrap (e.g. Coban) has been recommended as being able to heal most ulcers within 2 weeks.
Oranje et al 12 described the treatment of 8 infants with ulcerated hemangiomas with a polyurethane film (Opsite Flexigrid). For the first 3 days, the films were changed 3-4 times per day. After that, the films were changed once daily. If the films were removed with a vertical action away from the skin, pain was minimal. The wound was rinsed with saline at each change. The authors noted that most patients experienced prompt pain relief with initiation of this therapy. Healing within 1-2 months was observed. The authors recommend this approach as initial therapy for ulcerative hemangiomas in children.
Combigan
Combigan which is the combination of (topical brimonidine 0.2% timolol 0.5%) is FDA approved for glaucoma. It is not approved for patients less than 2 years of age. There is one case report of an ulcerated hemangioma of the lip that was treated with application of two drops from a fingertip to the lesion twice daily. Within 6 weeks, the ulcerated hemangioma dramatically improved, reepithelialized, flattened, and faded 13.
The package insert for Combigan states: “COMBIGAN® is contraindicated in children under the age of 2 years. During post-marketing surveillance, apnea, bradycardia, coma, hypotension, hypothermia, hypotonia, lethargy, pallor, respiratory depression, and somnolence have been reported in infants receiving brimonidine. The safety and effectiveness of brimonidine tartrate and timolol maleate have not been studied in children below the age of 2 years. ”
Figure 5. Infantile hemangioma
In general terms, infantile hemangiomas may be divided into superficial, deep or or mixed lesions.
Infantile hemangiomas may be localized (confined to a small area) or segmental (involving a larger neuroectodermal unit).
- The superficial infantile haemangiomas is also called capillary hemangioma, capillary nevus, strawberry hemangioma, strawberry nevus, and haemangioma simplex. The blood vessels in uppermost layers of the skin are dilated.
- Deep infantile haemangiomas are also called cavernous hemangiomas and are more deeply set in the dermis and subcutis. They appear as a bluish soft to firm swelling.
- Both types of hemangiomas may occur together in mixed angiomatous naevi. A strawberry nevus overlies a bluish swelling.
Figure 6. Strawberry hemangioma
Segmental haemangiomas are more serious than localized hemangiomas.
- They occur at a younger age and grow up to ten times larger
- They are consequently more unsightly
- Other congenital anomalies may be associated with facial segmental haemangiomas (PHACE syndrome). These includ posterior fossa abnormalities, hemangiomas, arterial abnormalities, cardiac abnormalities, and eye abnormalities.
- Likewise, segmental infantile haemangiomas involving the perineum may be associated with pelvic congenital anomolies, the PELVIS syndrome (perineal hemangioma with any of the following: external genital malformations, lipomyelomeningocele, vesicorenal abnormalities, imperforate anus, or skin tag)
Many parents worry that the infantile hemangioma will “pop like a balloon” resulting in uncontrolled bleeding. Thankfully, most hemangioma bleeding is slow, minor, and can be controlled with 10 to 15 minutes of direct pressure.
Which babies get hemangiomas?
Ten percent of babies develop one or more hemangiomas. Localized hemangiomas are more common if the baby has a low birthweight, for example in the following circumstances.
- Females
- White skin
- Premature
- Multiple birth (twins)
- Advanced maternal age
- Family history of infantile hemangioma
Hypoxia (inadequate oxygen to the skin) is now considered the likely reason for the proliferation of blood vessels. Endothelial progenitor cells (EPCs) circulate in a fetus and cause new blood vessels to form in response to hypoxia. Normally, endothelial progenitor cells have gone by the time a baby is born, but they may still be present in low birthweight or premature babies. As the endothelial progenitor cells disappear later in life, the haemangioma may regress.
Segmental hemangiomas are thought to arise early in gestation (6-8 weeks) as a developmental error.
Females and preterm infants are preferentially affected. There are rare families where hemangiomas are more common, with an autosomal dominant inheritance pattern being seen. Occasionally, hemangiomas may be associated with other malformations, e.g. a large facial hemangioma with the Dandy-Walker malformation, a plaque-like hemangioma spanning the midline of the sacrum with a tethered cord, or a hemangioma over the neck and airway involvement.
- Approximately 4% of infants have these vascular growths.
- Risk factors include female sex, white non-Hispanic race, prematurity, low birth weight, multiple gestation, gestational diabetes, and preeclampsia.
- The incidence appears to be increasing in the US over the last 3 decades, probably related to more infants being born with lower birth weight and decreased gestational age 14.
- Most rapid growth is in the second month of life.
- Certain locations may signal underlying abnormality (e.g facial and seblgmental–PHACE syndrome, sacral–Lumbar Syndrome)
- Two studies have found that 92% completely involuted by age 4.
- Familial clustering does occur with an autosomal dominant inheritance pattern with incomplete penetrance or through maternal transmission 15.
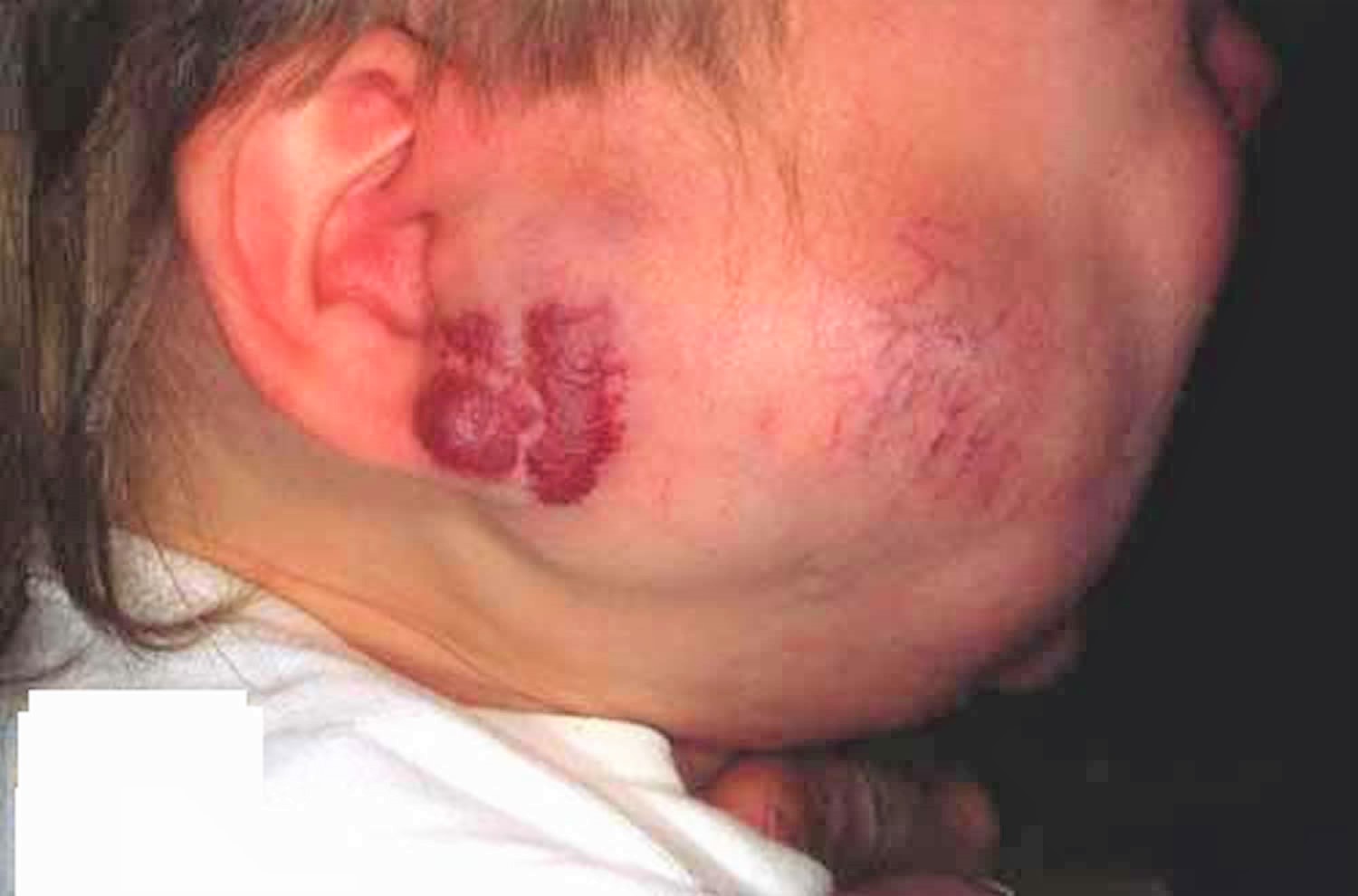
Figure 7. Cavernous haemangiomas (mixed type)
A hemangioma differs from a vascular malformation in that hemangioma proliferates, typically for the first 3-9 months of life and rarely beyond a year and a half. Congenital hemangiomas include RICH (rapidly involuting congenital hemangiomas) and NICH (non-involuting congenital hemangiomas). Rapidly involuting congenital hemangiomas (RICH) is a vascular papulonodule that is fully formed at birth. Rapidly involuting congenital hemangiomas (RICH) may be a few to many centimeters in diameter. The head and neck are the most common sites. Ulceration, complicated by life-threatening hemorrhage may rarely occur 16. Rarely, a RICH may be complicated by may present with transient thrombocytopenia and cardiac overload 17. Rapidly involuting congenital hemangiomas (RICH) lesions may rapidly involute within weeks or may do so over many months–up to 16 months. Rarely, lesions involute in utero. No treatment is needed for RICH, but surgery may be done for persistent, redundant skin. RICH has excellent response to prednisone 18. Some RICH only partially involute, leaving a PICH (partially involuting hemangioma) 19 or a NICH (non-involuting congenital hemangiomas) (J Am Acad Dermatol. 2014;70:899-903)).
Partially involuting hemangioma (PICH) is a type of congenital hemangiomas that start off involuting, but then stop. Partially involuting hemangiomas (PICHs) tend to stabilize after 12-30 months of life 20.
Non-involuting congenital hemangiomas (NICHs) never involutes. Two types of NICH have been described. A patch type, characterized by a flat or slightly atrophic surface and a nodular/plaque type (see Figure 9 below). The surface in both the nodular and patch types may have a hypopigmented color with telangiectasias throughout. Most NICH (non-involuting congenital hemangiomas) do not require treatment. Surgical resection is an option in selected cases 21, but may be complicated by incomplete excision as well as intraoperative and postoperative bleeding. Other described treatment options include sclerothreapy with surgery 22, endovascular particulate embolization, tissue cauterization, vascular laser therapy and cryotherapy to control recurrent episodes of oral bleeding.
Figure 8. Hemangioma baby
Rhabdomyosarcomas, infantile myofibromatosis, etc. can resemble hemangiomas but are more firm. Any hemangioma that seems excessively firm deserves careful evaluation and biopsy if necessary. Rarely, a hemangioma may be so flat at birth as to resemble a port wine stain. Deep hemangiomas can be a challenge diagnostically and imaging may be very helpful.
Vascular lesions full size at birth may represent RICH, NICH, partially involuting hemangiomas and vascular malformations.
Glucose transporter 1 (GLUT-1) is found normally in placental blood vessels and blood-brain barrier blood vessels. 97% of Hemangiomas test positive for GLUT-1. RICH and NICH area glut-1 negative.
Typical infantile hemangiomas, even in the midline on scalp or upper back usually do not need imaging. However, a midline and sacral/lumbar hemangioma does need an Magnetic Resonance Imaging (MRI) to rule out Lumbar syndrome.
The acronym of LUMBAR syndrome has been used to describe the association of a lower-body hemangioma with other cutaneous defects 23.
- Lower body hemangioma (most commonly lower back or buttocks)
- Urogenital abnormalities (e.g. bladder, vagina) or Ulceration
- Myelopathy (malformation of the spinal cord, e.g. tethered cord)
- Bone abnormalities (e.g. deformity of the foot)
- Anorectal or Arterial abnormalities
- Renal disorders (e.g. one kidney instead of two)
A recent prospective study demonstrated that isolated cutaneous infantile hemangiomas of the lumbosacral region have one of the highest risks (relative risk of 438) of associated spinal dysraphism, including in decreasing order of frequency, tethered cord, spinal lipoma, intraspinal hemangioma, and sinus tract 24.
Figure 9. Noninvoluting congenital hemangioma (NICH)
Infantile hemangioma diagnosis
Infantile hemangiomas are usually diagnosed clinically and no investigations are necessary for the majority of superficial lesions.
Deep infantile hemangiomas or segmental hemangiomas are routinely investigated with ultrasound scanning. An ultrasound scan is also often performed when there is uncertainty about the diagnosis or whether underlying tissues are affected. Characteristically, a hemangioma has a firm lobular structure with vessels separating the lobules.
It may also be necessary to perform Magnetic Resonance Imaging (MRI) or angiography to help plan treatment. Children with complex lesions are best assessed by a panel of experts, including paediatrician, dermatologist, radiologist, ophthalmologist, vascular and plastic surgeons.
Haemangiomas arising over the lower part of the spine are sometimes a marker for occult spinal dysraphism (spina bifida), when spinal imaging may be appropriate.
Infantile haemangioma treatment
Because infantile hemangiomas are likely to improve or regress completely with time, there is no need for specific treatment in most cases. Treatment should be considered in the following circumstances.
- Very large and unsightly lesions
- Ulcerating hemangiomas (up to 5-25% of lesions)
- Lesions that impair vision, hearing, breathing or feeding
- If they fail to resolve by school age
- Hemangiomas that have a steep or stepped border, thick pebbled surface or combined superficial and deep components
The baby is best assessed early i.e. before the rapidly growing phase ideally under the age of 5 to 7 weeks of age. If the lesion obstructs vision, it may prevent the development of normal sight.
Infantile hemangioma treatment options
- Observation.
- Topical timolol maleate 0.5% gel bid x 24 weeks effective in reducing size of hemangiomas.
- Propranolol can be used for more aggressive therapy.
- Itraconazole
- Laser
In the perfect situation, all patients with an infantile hemangioma or suspected infantile hemangioma would be evaluated as early as possible by a pediatric dermatologist or other clinician familiar with the use of topical timolol, oral propranolol and pulsed dye laser (PDL). Clearly, these therapies are more efficacious the sooner they are employed. Waiting until the infantile hemangioma has undergone the “growth spurt” (usually at 5-7 weeks of life) makes both topical timolol and laser less effective. Topical timolol is readily available, efficacious and with few side effects than pulsed dye laser. It may be employed for most uncomplicated infantile hemangioma. In selected cases, pulsed dye laser therapy can be very beneficial, but side effect and logistics are issues (e.g. will general anesthesia be needed? Pain control during the procedure, etc.) Lesions on the face may require close follow up (weekly?) to assess response to therapy, growth and any potential complications.
Alarming Hemangiomas
Referral to a pediatric dermatologist may be considered for the “alarming” hemangiomas (those that interfere with vital functions such as eating, breathing, seeing), and those larger ones on the face, perianal area or lumbosacral area or those associated with other congenital abnormalities. Nasal hemangiomas develop the most complications, e.g., airway obstruction, ulceration, visual obstruction, ocular compression and infection. Involvement of the beard area and neck should prompt airway evaluation to exclude subglottic infantile hemangiomas which can cause life-threatening airway obstruction 25. Systemic propranolol may be indicated. Sometimes, infants are seen too late, e.g. at 12 weeks of age, when most of the growth has already occurred. In that case, a short course e.g. 2-3 months of propranolol, may considered to see if there is any effect.
Observation
Two studies have found that 92% of infantile hemangiomas completely involute by age 4. Even though the infantile hemangioma may have involuted, that does not mean that normal skin remains. In one study of 184 hemangiomas 26, resolution to normal skin only occurred in 45%. Common sequelae included telangiectasias (84%), fibrofatty tissue (47%), and anetodermic skin (33%). Combined hemangiomas with a superficial component and a step border were associated with more sequelae. Once resolution has occurred, the skin should be evaluated for cosmesis, and redundant skin excised if needed. Active treatment with propranolol in various studies has shown complete regression without sequelae after 6 months of treatment in 60% of cases. Thus, consideration of propranolol or other active therapy should be considered, especially in larger lesions.
Topical Timolol
One drop of topical timolol 0.5% liquid (eyedrops) or gel forming solution may be applied to the infantile hemangioma twice daily. Starting as early as possible is key. Both the liquid and gel are equally efficacious 27, but the liquid is not as convenient as it tends to run off the hemangioma onto normal skin. Measured blood levels of timolol are either undetectable or very low in most cases.
In one double blind placebo controlled trial of infants aged 5-24 weeks, timolol appeared most effective in the management of infantile hemangiomas with a volume of 100 mm³ or less. No episodes of bradycardia nor hypotension were seen. A key point is that the effect may not visible for 4 months especially for thicker lesions. The mean duration of therapy in one study was 9.5 months. Make sure to follow closely to make sure not proliferating rapidly (and might need oral propranolol). Initiation of therapy within 3 months of life and superficial lesions respond best.
In preterm infants, those with ulceration, large surface area or mucosal lesions, the concern of systemic effect similar to oral propranolol has been raised. Close monitoring of temperature, blood pressure, and heart rate in premature and low-birthweight infants with infantile hemangiomas at initiation of and during therapy with topical timolol is recommended 28.
Timolol is particularly useful for periorbital hemangiomas. There is little concern if it smears into the eye as it has been used for treatment of glaucoma in infants for decades.
If PHACES is a possibility (see PHACE syndrome below), timolol treatment should not be initiated because of concern about cardiovascular effects.
Propranolol
The FDA approved the first and only beta-blocker formulation in March 2014 for the treatment of proliferating infantile hemangioma requiring systemic therapy. Hemangeol™, was studied in infants aged 5 weeks to 5 months. In a RDBPCT, propranolol 3 mg/kg/day for 6 months demonstrated a 60.4% success rate (complete or near complete resolution) vs. 3.6% in the placebo group. Side effects include drop in blood pressure (usually mild), sleep disturbances, and hypoglycemia. Treatment may need 10-12 months. Many experts in clinical practice use 2-2.5 mg/kg/day and that is sufficient. Premature babies are more likely to have issues with hypotension, so for them a lower dose is preferred, e.g. 1-1.5 mg/kg/day. A question was asked of pediatric experts, “Do you admit the patient to start propranolol?” Most only for specific situations, e.g. parents not reliable, very complicated/severe case.
Hypoglycemia, though rare, can induce seizures. Temporarily suspending treatment with propranolol may be considered in infants with restricted oral intake, e.g. during a respiratory illness.
Rebound of hemangiomas treated with propranolol occurred in 25% of cases in one review of nearly 1000.
Concern has been raised about potential CNS (central nervous system) side effects and the use of propranolol 29. Unfortunately, studies in infants are lacking.
Propranolol in one small study of 6 children had no effect 30. A comparative study of atenolol vs propranolol in 23 patients, no significant difference was seen in results or side effects 31. In a comparative study against captopril, propranolol was found superior 32.
Nadolol
Nadolol is a nonselective beta-blocker with no intrinsic sympathomimetic activity, and in contrast to propranolol, it has little myocardial depressant activity, wider therapeutic uses in childhood, and a longer half-life of 12–24 hours. A twice daily dosage is recommended in children younger than 22 months. The longer half-life improves adherence and decreases rebound. In contrast to propranolol, nadolol cannot cross the blood–brain barrier, decreasing adverse events such as nightmares and concerns about longterm memory loss. Patients with proliferative infantile hemangioma, treated with oral nadolol for 6 months, experienced almost complete involution of their tumor, which was significantly different from patients treated with propranolol 33.
Itraconazole
In an article 34 shows that about 70% (12/17) of infants experienced 80-100% improvement in their infantile hemangioma with oral itraconazole 5 mg/kg/day given for an average of 8.8 weeks. Liver tests should be monitored but in his series they have been normal. 30% of patients have mild diarrhea.
Laser
One study showed a high clearance rate with just one treatment using the 650 microsecond, pulsed 1064-nm Nd:YAG laser, spot size 2-3 mm and fluence of 42-64 joules 35. The best results were in patients treated in the first 6 weeks of life. Over 75% of hemangiomas were either completely or almost completely cleared over the 1-year follow up period.
Imiquimod
Imiquimod has been used for infantile hemangiomas 36. In a comparison study of imiquimod 5% every other day vs timolol three times daily 37, timolol was superior, especially in terms of earlier onset and fewer side effects.
PHACE Syndrome
PHACE Syndrome is the uncommon association between large infantile hemangiomas, usually of the face, and birth defects of the brain, heart, eyes, skin and/or arteries.
PHACE syndrome is the association of a large facial hemangioma with a posterior fossa malformation and various other abnormalities.
PHACE Syndrome (PHACE) is an acronym that stands for the medical names of the parts of the body it often impacts:
- P Posterior fossa malformations
- H Hemangiomas, typically facial, large, segmental and prone to ulceration
- A Arterial Abnormalities
- C Coarctation of the Aorta and Cardiac Defects
- E Eye abnormalities.
- Sometimes an “S” is added to PHACE making the acronym PHACES; with the “S” standing for “Sternal defects” and/or “Supraumbilical raphe.”
It is not uncommon to have infantile hemangioma of the brain in PHACE approximately 15-18% of those with PHACE will have intracranial hemangioma.
30-35% of those with PHACE will have malformations of the brain(structural brain abnormalities) — mainly of the back of the brain called the posterior fossa, or the space at the base of the skull containing the brainstem and cerebellum.
Developmental anomalies of the pituitary gland occur in 15-18% of those with PHACE syndrome. This could lead to growth or endocrine problems.
80-90% of PHACE patients have Arterial Anomalies. This is the most variable and complicated abnormality in PHACE. Each child seems to have their own unique changes in the arteries of the chest, neck and brain. These abnormalities are believed to be the cause of migraine-like headaches.
Complications from severe infantile hemangiomas in PHACE are becoming increasingly less common due to treatment with oral beta blockers. Long term affects can include scarring, impaired vision and damage to hearing.
30% of PHACE patients have the Cardiovascular anomalies which can include coarctation of the aortic arch, aortic arch anomalies and subclavian artery anomalies.
12-15% of PHACE patients have developmental abnormalities of the Eye. Most of the time they occur in the posterior segment of the eye.
Some children can have hearing problems and endocrine problems such as thyroid anomalies for problems with growth hormone.
PHACE syndrome criteria 38
- The presence of a plaque-like facial hemangioma, plus either
- 1 major criterion (posterior fossa anomalies, dysplasia of the large cerebral arteries, aortic arch anomaly, eye posterior segment anomaly, or sternal defect) or
- 2 minor criteria (callosal agenesis, pituitary malformation, ventricular septal defect, eye anterior segment anomaly, hypopituitarism, or ectopic thyroid, among others).
If in addition, there is sternal clefting and⁄or supraumbilical raphe, the term PHACES is used. Babies with PHACE syndrome are at risk for stroke. Test should include MRI with and with contrast as well as MRA (MR angiogram).
The hemangiomas are facial, typically large, segmental, prone to ulceration, and generally require treatment. Enamel hypoplasia may be a feature of PHACE syndrome when an intraoral hemangioma is present 39. Abortive hemangiomas have also been part of PHACE syndrome. One study of PHACE syndrome found that most children did not have significant neurodevelopmental deficits. A subset of patients had delays in multiple areas 40.
There has been a great deal of research to understand the cause of PHACE Syndrome. The abnormalities associated with this syndrome are thought to be due to errors that occur very early during development. Unfortunately, why the errors occur, or the exact cause is still unknown. PHACE has a shared biology of other vascular anomalies. There may be a genetic component involved and studies are underway to investigate this idea. No familial cases have been identified to date. Research is ongoing to find the cause of all vascular anomalies including PHACE Syndrome.
Diagnosis of PHACE Syndrome
If the medical history and the actual exam of the hemangioma look typical for PHACE Syndrome, more tests are ordered to confirm the diagnosis. These tests may include:
- Ultrasound
- Magnetic resonance imaging (MRI)
- Magnetic resonance angiography of the brain (MRA)
- Echocardiogram of the heart
- Eye exam by an eye doctor
- Other tests may be needed for diagnosis and treatment
Possible Complications of PHACE Syndrome
As the hemangioma(s) grow, the skin surface of the hemangioma can break down (ulceration), distort facial features or get in the way of other vital functions, such as breathing, vision, and hearing. Other complications will depend on what other structures are involved. These could include cardiac problems, developmental delay, seizures, headaches, and abnormal muscle tone if the brain is involved.
Treatment for PHACE Syndrome
A multidisciplinary team is needed for PHACE syndrome treatment. For the hemangioma, oral propranolol is used, but be cautious as oral propranolol may increase risk of stroke. Use three times daily (not twice daily) and lowest possible dose of propranolol (e.g. < 2 mg/kg/day). In urgent cases, if you don’t have MRI/MRA data, can get an echocardiogram to rule out coarctation of the aorta which is a contraindication for propranolol and then start low dose propranolol. The combination of prednisolone and propranolol has been advocated 41.
Management of PHACE Syndrome
PHACE syndrome needs to be managed by a multidisciplinary team of experts familiar with the treatment of hemangiomas and special considerations that arise when a child has PHACE. Additional specialties such as cardiology, ophthalmology, neurology, and neurosurgery may need to be involved.
This team of experts needs to follow the infant initially to manage acute issues and complications but it is very important for this same team to follow the patient long term to evaluate development and any long term issues, pay close attention to how these children develop throughout the school age period.
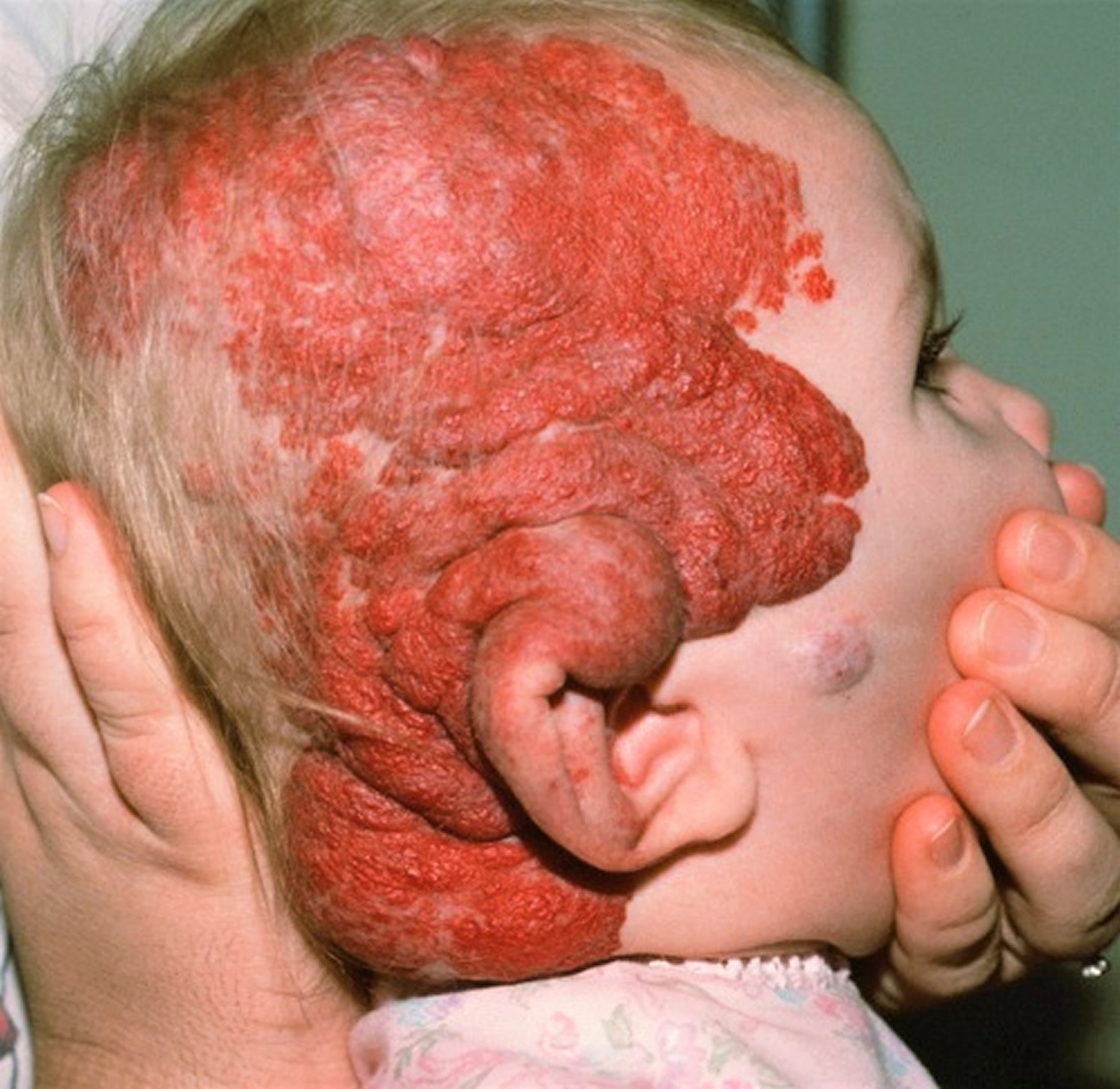
Figure 10. Large segmental infantile hemangioma involving the right side of the face and forehead (PHACE syndrome. MRI is needed to exclude brain involvement)
- Schrock WB, Wetzel RJ, Tanner SC, Khan MA. Aggressive hemangioma of the thoracic spine. Journal of Radiology Case Reports. 2011;5(10):7-13. doi:10.3941/jrcr.v5i10.828. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3303463/[↩][↩][↩]
- Cheung NK, Doorenbosch X, Christie JG. Rapid onset aggressive vertebral haemangioma. Childs Nerv Syst. 2011 Mar;27:469–72. https://www.ncbi.nlm.nih.gov/pubmed/21246374[↩]
- Spinal hemangioma mimicking a dumbbell-shaped schwannoma: Case report and review of the literature. Huang, Kevin T. et al. J Clin Neurosci. 2018 Feb 1. pii: S0967-5868(17)31293-6. doi: 10.1016/j.jocn.2018.01.037[↩]
- Pastushyn A, Slin’ko EI, Mirzoyeva GM. Vertebral hemangiomas: diagnosis, management, natural history and clinicopathological correlates in 86 patients. Surg Neurol. 1998 Dec;50:535–547. https://www.ncbi.nlm.nih.gov/pubmed/9870814[↩]
- Laredo JD, Reizine D, Bard M, Merland JJ. Vertebral hemangiomas: radiologic evaluation. Radiology. 1986 Oct;161:183–189. https://www.ncbi.nlm.nih.gov/pubmed/3763864[↩][↩]
- Dickerman RD, Bennett MT. Acute spinal cord compression caused by vertebral hemangioma. Spine J. 2005 Sep-Oct;5:582–584. author reply 584. https://www.ncbi.nlm.nih.gov/pubmed/16153588[↩]
- Guarnieri G, Ambrosanio G, Vassallo P, et al. Vertebroplasty as treatment of aggressive and symptomatic vertebral hemangiomas: up to 4 years of follow up. Neuroradiology. 2009 Jul;51:471–476. https://www.ncbi.nlm.nih.gov/pubmed/19300988[↩][↩]
- Laredo JD, Assouline E, Gelbert F, Wybier M, Merland JJ, Tubiana JM. Vertebral hemangiomas: fat content as a sign of aggressiveness. Radiology. 1990 Nov;177:467–472. https://www.ncbi.nlm.nih.gov/pubmed/2217787[↩]
- Doppman JL, Oldfield EH, Heiss JD. Symptomatic vertebral hemangiomas: treatment by means of direct intralesional injection of ethanol. Radiology. 2000 Feb;214:341–348. https://www.ncbi.nlm.nih.gov/pubmed/10671579[↩]
- Urrutia J, Postigo R, Larrondo R, Martin AS. Clinical and imaging findings in patients with aggressive spinal hemangioma requiring surgical treatment. J Clin Neurosci. 2011 Feb;18:209–12. https://www.ncbi.nlm.nih.gov/pubmed/21177111[↩]
- Fox MW, Onofrio BM. The natural history and management of symptomatic and asymptomatic vertebral hemangiomas. J Neurosurg. 1993 Jan;78:36–45. https://www.ncbi.nlm.nih.gov/pubmed/8416240[↩]
- Dermatology 2000;200:31–34[↩]
- Pediatric Dermatology 2014;31;754[↩]
- J Am Acad Dermatol. 2016;74;120[↩]
- Pediatric 2016;138[↩]
- JAMA Derm 2015;151;422[↩]
- Ped Derm 2014;31:402[↩]
- Rapidly involuting congenital hemangioma: case report and review of the literature. John C Browning MD, Denise W Metry MD. Dermatology Online Journal 14 (4): 11[↩]
- JAAD 2014;70:899-903[↩]
- Partially involuting congenital hemangiomas: a report of 8 cases and review of the literature. J Am Acad Dermatol. 2014 Jan;70(1):75-9. doi: 10.1016/j.jaad.2013.09.018. Epub 2013 Oct 29. http://www.jaad.org/article/S0190-9622(13)00969-9/fulltext[↩]
- J Am Acad Dermatol. 2014;70:899-903[↩]
- Phlebology 2014;29 :4–8[↩]
- J Am Acad Dermatol. 2015;73;258.[↩]
- Pediatr Radiol. 2012 Mar;42(3):315-20[↩]
- JEADV 2016;30;2056[↩]
- JAMA Derm 2016;152;1239[↩]
- PD 2017;34;492[↩]
- Pediatric Dermatology 2016;33;405[↩]
- Br J Dermatol 2015 Jan 01;172(1)13-23[↩]
- Pediatric Dermatology March 2014[↩]
- JAAD 2014 :70:1045[↩]
- JAAD 2016;74;499[↩]
- Br J Dermatol. 2013 Jan;168(1):222-4[↩]
- J Dermatol. 2015;42:202[↩]
- Dermatology News June 2016, p. 31[↩]
- Clin Exp Dermatol. 2013 Dec;38(8):845-50[↩]
- Dermatology. 2015;230(2):150-5[↩]
- Mussa A, Baldassarre G, Rosaia De Santis L, Gastaldi R, Corrias A, Silengo MC. Four new cases of PHACES syndrome: Variable phenotypic expression and endocrine features. Acta Paediatr 2008;97:1729-33. http://onlinelibrary.wiley.com/wol1/doi/10.1111/j.1651-2227.2008.01032.x/full[↩]
- Pediatric Dermatology 2014;31;455–458[↩]
- Pediatric Dermatology 2016;33;415[↩]
- BJD 2015;173;242[↩]