Contents
- What is hemolytic uremic syndrome
What is hemolytic uremic syndrome
Hemolytic uremic syndrome (HUS) is a a kidney condition that happens when red blood cells are destroyed and block the kidneys’ filtering system. The damaged red blood cells clog the filtering system in the kidneys, which can lead to life-threatening kidney failure.
Red blood cells contain hemoglobin—an iron-rich protein that gives blood its red color and carries oxygen from the lungs to all parts of the body.
When the kidneys and glomeruli—the tiny units within the kidneys where blood is filtered—become clogged with the damaged red blood cells, they are unable to do their jobs. If the kidneys stop functioning, a child can develop acute kidney injury—the sudden and temporary loss of kidney function. Hemolytic uremic syndrome (HUS) is the most common cause of acute kidney failure in children.
Hemolytic uremic syndrome affects males and females in equal numbers. Some studies have suggested that the disorder affects females more severely than males. Hemolytic uremic syndrome can affect children or adults, but is more common in children under 10, especially children between 7 months and 6 years of age. Hemolytic uremic syndrome is estimated to occur in 1-3 per 100,000 people in the general population. The incidence rate of E. coli infection in North America is estimated to be 8 in 100,000 people in the general population. Fortunately, only 5 to 15 percent of individuals infected with E. coli progress to develop hemolytic uremic syndrome.
Children who are more likely to develop hemolytic uremic syndrome include those who:
- are younger than age 5 and have been diagnosed with an E. coli O157:H7 infection
- have a weakened immune system
- have a family history of inherited hemolytic uremic syndrome
The risk of developing hemolytic uremic syndrome is highest for:
- Children under 5 years of age
- People over 75
- People with certain genetic changes that make them more susceptible.
The treatment of hemolytic uremic syndrome is aimed at managing existing symptoms and preventing further complications. Early diagnosis is essential for appropriate acute, aggressive care. Intravenous volume expansion may decrease the frequency of oligoanuric renal failure in patients with E. coli 0157:H7 gastroenteritis who are at risk for progressing to hemolytic uremic syndrome. Treatment requires the coordinated efforts of a team of specialists. Pediatricians, kidney specialists (nephrologists), intensive care physicians, nurses, nutritionists and social workers need to work in teams to treat patients.
Specific treatment includes control of hypertension and seizures.
See your doctor immediately if you or your child experiences bloody diarrhea or several days of diarrhea followed by:
- Decreased urine output
- Unexplained bruises
- Unusual bleeding
- Extreme fatigue
- Swelling, most often in the legs, feet, or ankles and less often in the hands or face (edema)
Seek emergency care if you or your child doesn’t urinate for 12 hours or more.
What are the kidneys and what do they do?
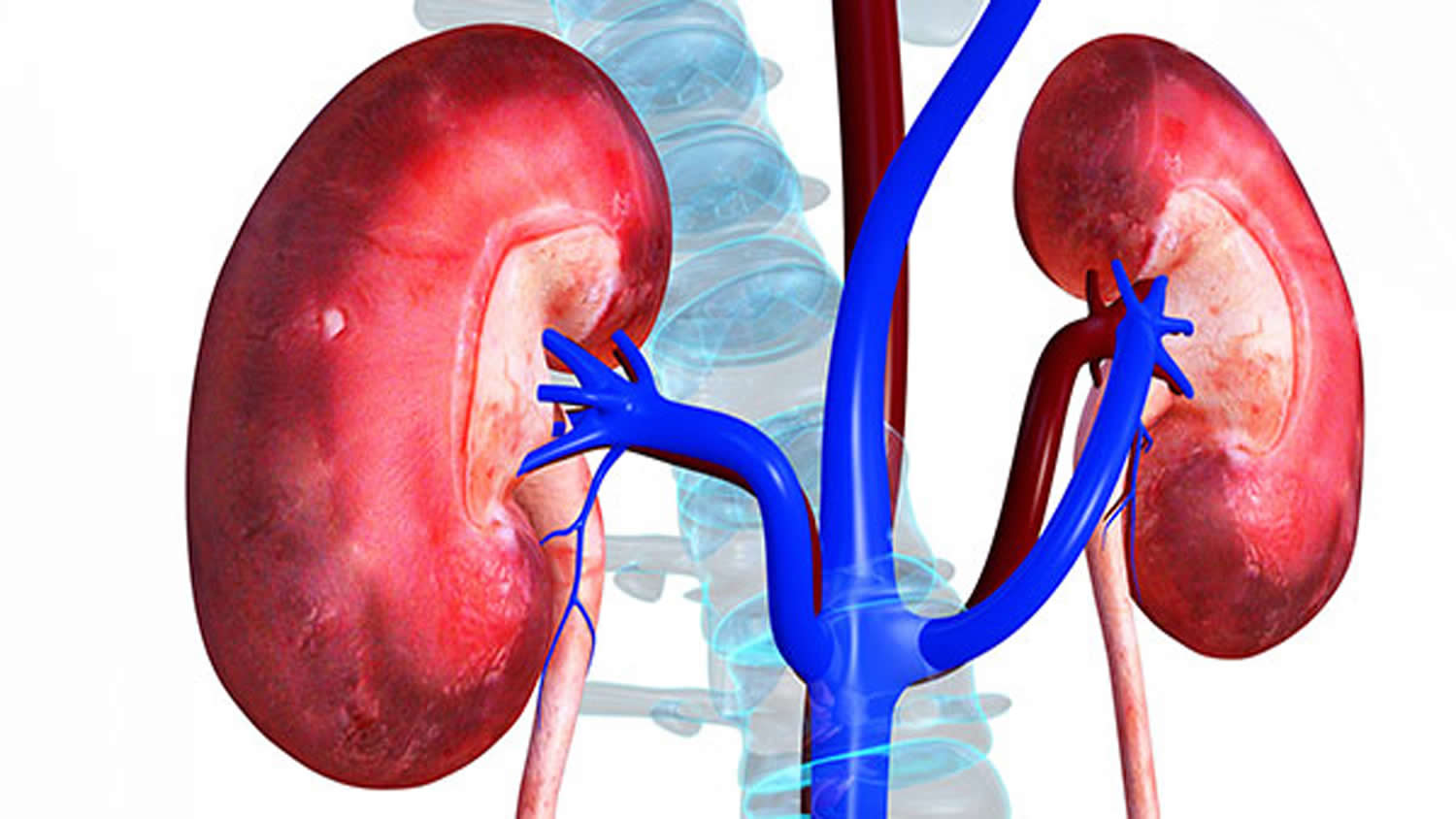
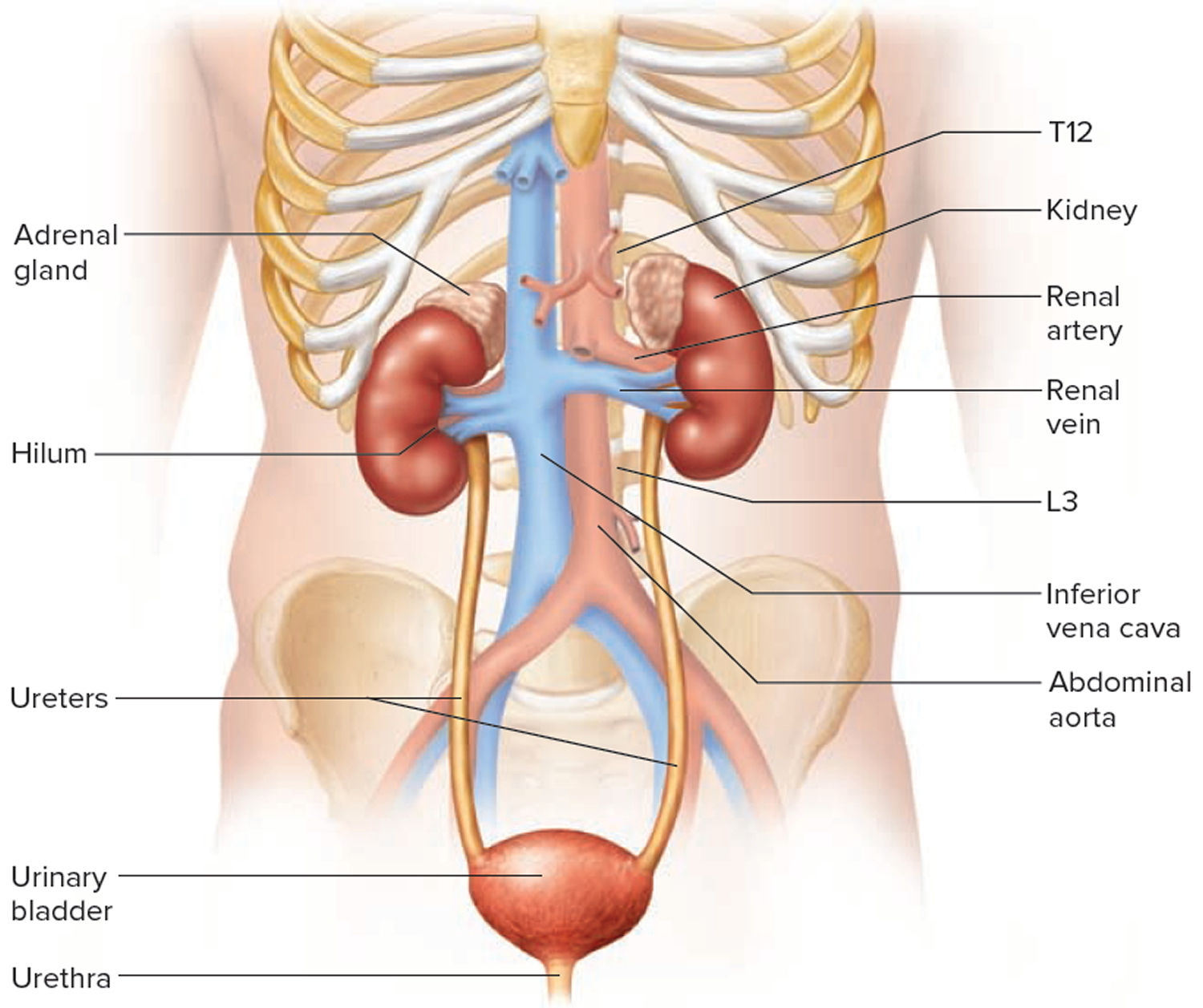
The kidneys are two bean-shaped organs, each about the size of a fist. They are located just below the rib cage, one on each side of the spine.
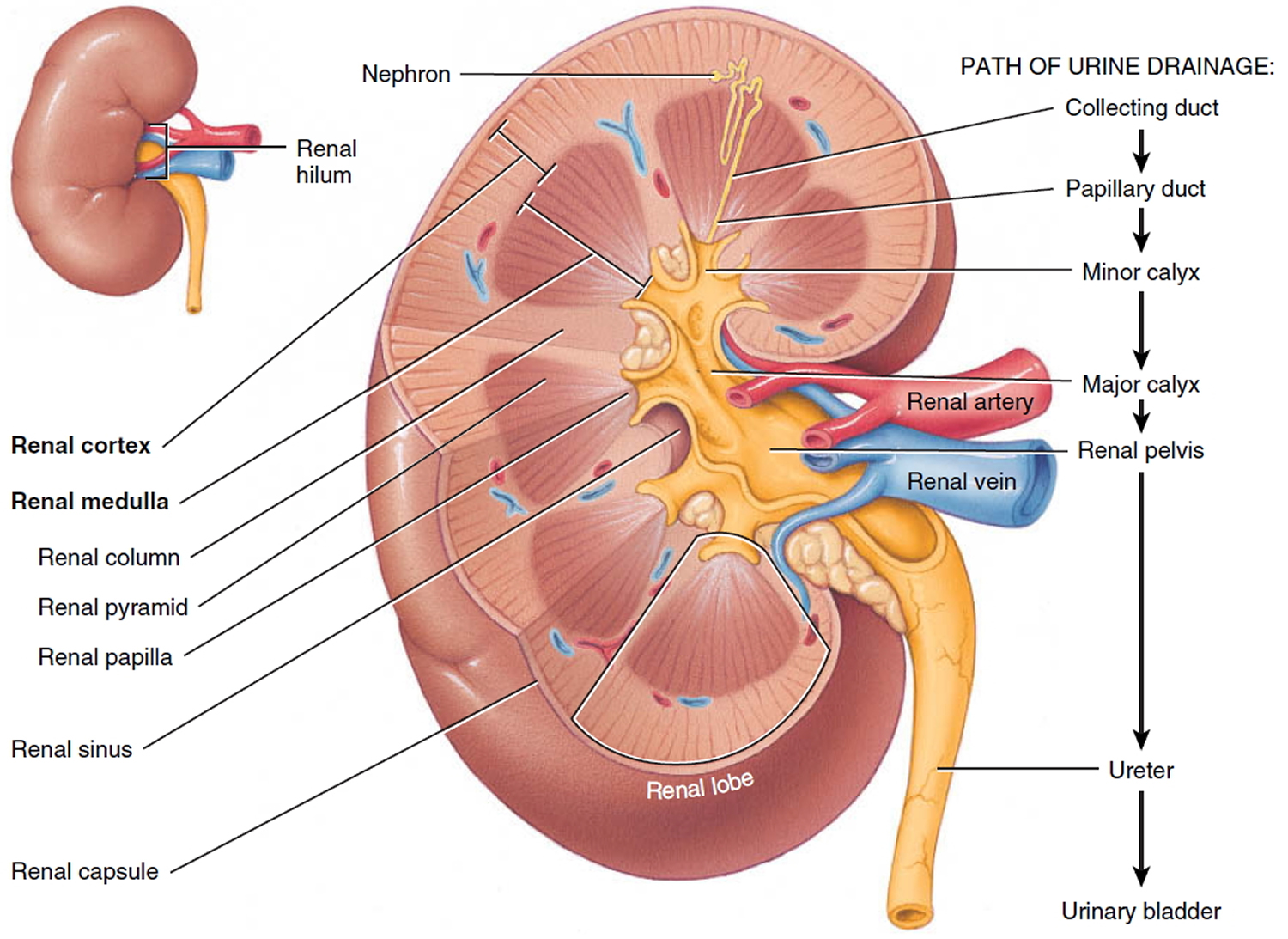
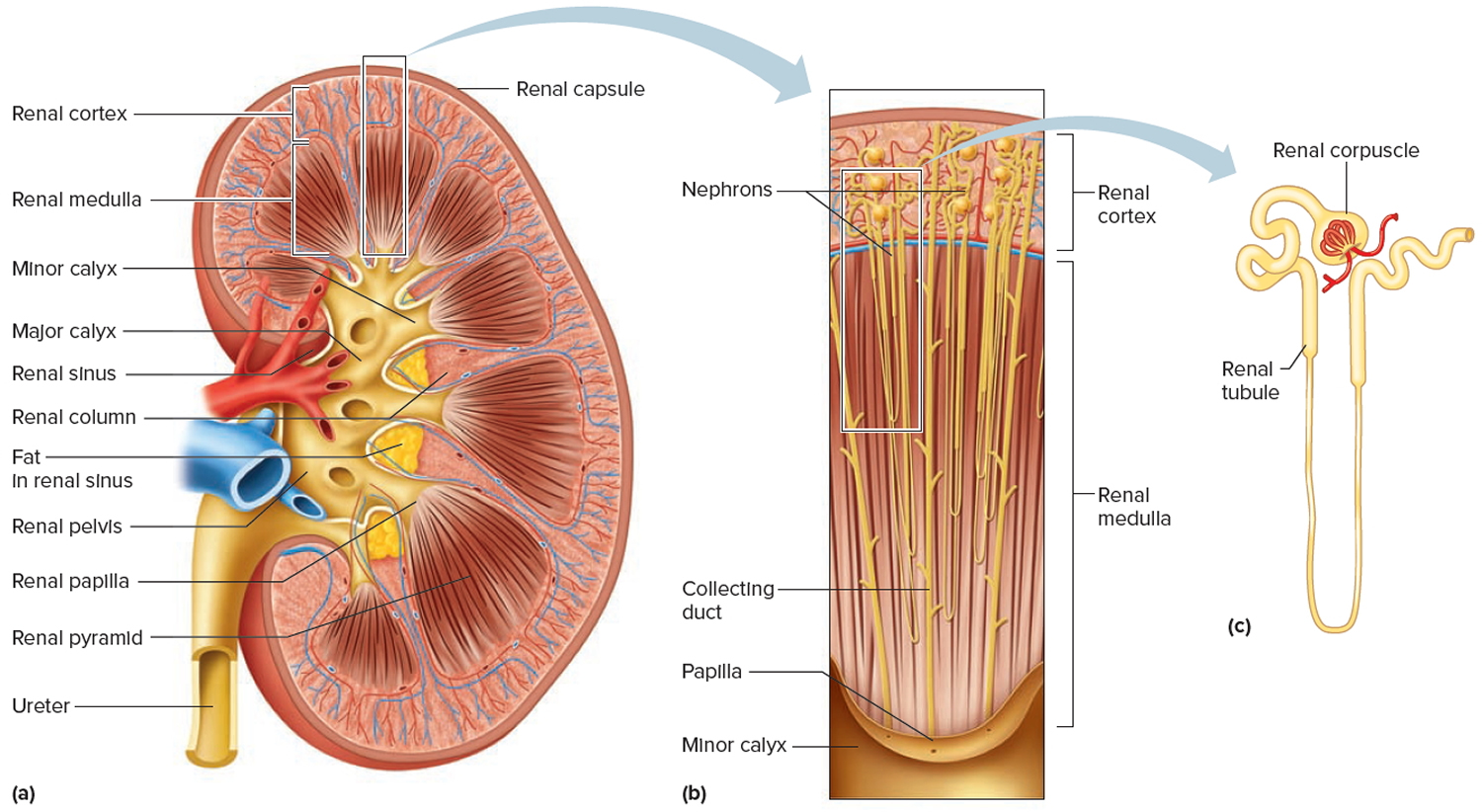
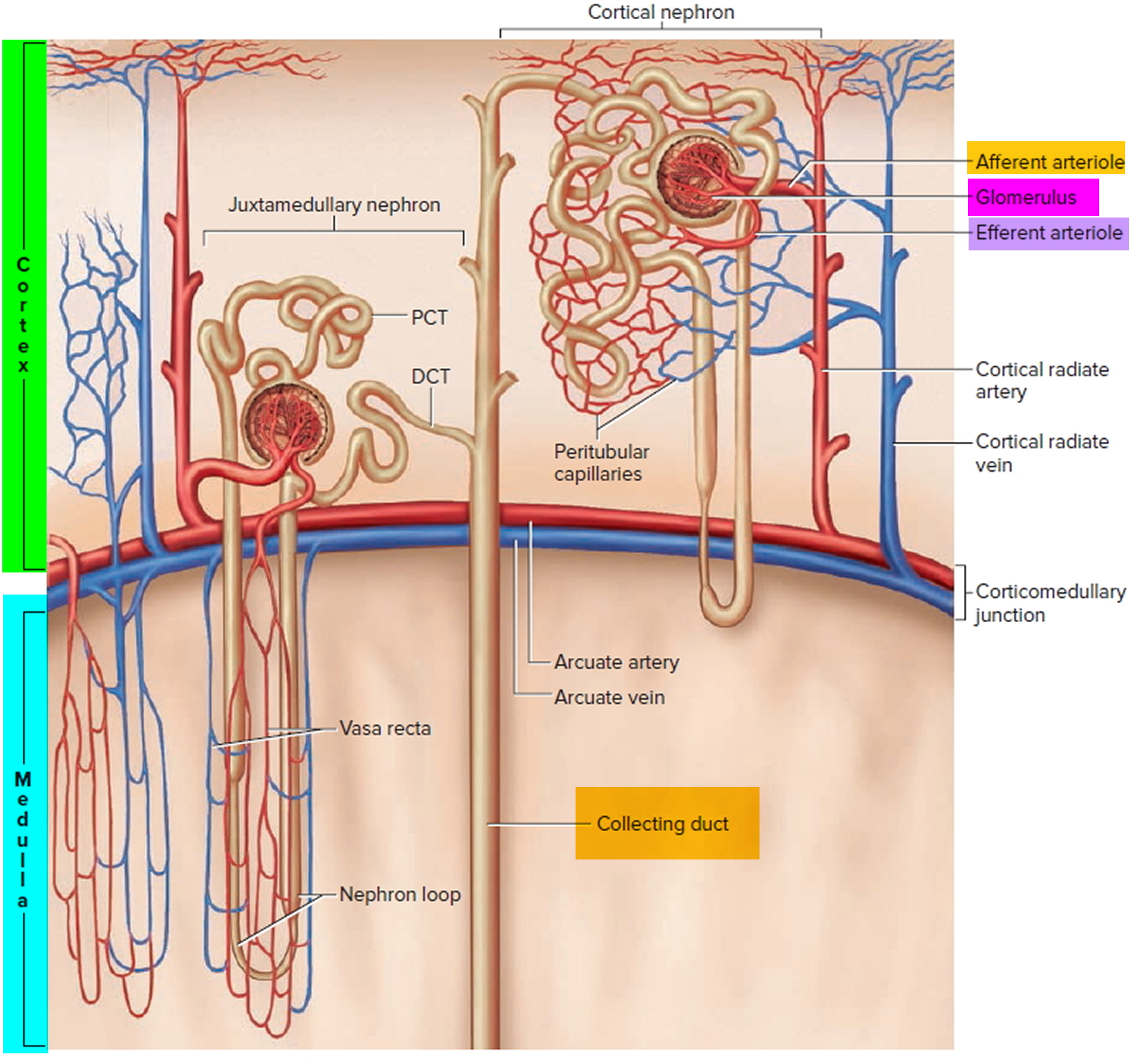
A frontal section through the kidney reveals two distinct regions: a superficial, light red region called the renal cortex and a deep, darker reddish-brown inner region called the renal medulla (medulla = inner portion) (Figures 2 and 3). The renal medulla consists of several cone-shaped renal pyramids. The base (wider end) of each pyramid faces the renal cortex, and its apex (narrower end), called a renal papilla, points toward the renal hilum. The renal cortex is the smooth-textured area extending from the renal capsule to the bases of the renal pyramids and into the spaces between them. It is divided into an outer cortical zone and an inner juxtamedullary zone. Those portions of the renal cortex that extend between renal pyramids are called renal columns.
Together, the renal cortex and renal pyramids of the renal medulla constitute the parenchyma or functional portion of the kidney. Within the parenchyma are the functional units of the kidney—about 1 million microscopic structures called nephrons (Figures 3 and 4). Filtrate (filtered fluid) formed by the nephrons drains into large papillary ducts, which extend through the renal papillae of the pyramids. The papillary ducts drain into cuplike structures called minor and major calyces. Each kidney has 8 to 18 minor calyces and 2 or 3 major calyces. A minor calyx receives filtrate from the papillary ducts of one renal papilla and delivers it to a major calyx. Once the filtrate enters the calyces it becomes urine because no further reabsorption can occur. The reason for this is that the simple epithelium of the nephron and ducts becomes transitional epithelium in the calyces. From the major calyces, urine drains into a single large cavity called the renal pelvis and then out through the ureter to the urinary bladder.
Every day, the two kidneys filter about 120 to 150 quarts of blood to produce about 1 to 2 quarts of urine, composed of wastes and extra fluid. Children produce less urine than adults and the amount produced depends on their age. The urine flows from the kidneys to the bladder through tubes called ureters. The bladder stores urine. When the bladder empties, urine flows out of the body through a tube called the urethra, located at the bottom of the bladder.
Figure 1. Kidney location
Figure 2. Kidney anatomy
Figure 3. Kidney structure
Figure 4. Microcirculation of the kidney
Hemolytic uremic syndrome complications
Most children who develop hemolytic uremic syndrome and its complications recover without permanent damage to their health.
However, children with hemolytic uremic syndrome may have serious and sometimes life-threatening complications, including:
- Acute kidney injury
- Kidney failure, which can be sudden (acute) or develop over time (chronic)
- High blood pressure
- Blood-clotting problems that can lead to bleeding
- Seizures
- Heart problems
- Chronic, or long lasting, kidney disease
- Stroke
- Coma
- Intestinal problems, such as inflammatory colitis
Most individuals with hemolytic uremic syndrome completely recover from the renal failure. However, approximately 10 percent of affected individuals may develop chronic renal failure. This is extremely uncommon after recovery from an episode of hemolytic uremic syndrome. Those who have severe permanent kidney damage with proteinuria, elevated blood pressures and an increase in serum creatinine concentrations may have progressive loss of kidney function approximately five to 10 years after the acute episode. A small percentage of patients will require chronic dialysis and kidney transplantation.
Some individuals with hemolytic uremic syndrome develop symptoms associated with the central nervous system including (as mentioned above) the sudden onset of lethargy and irritability and seizures. Additional CNS symptoms include an impaired ability to control voluntary movements (ataxia), weakness on one side of the body (hemiparesis), confusion, and coma.
The pancreas may become involved in hemolytic uremic syndrome. The pancreas is an important organ located behind the stomach that secretes enzymes that aid in digestion. The pancreas also secretes insulin, which helps break down sugar. Pancreatic involvement is usually mild, but fluid accumulation in the pancreas (pseudocysts) and destruction of pancreatic tissue (necrosis) can occur. In about 3 percent of patients, insulin-dependent diabetes can develop. A small number of patients may develop gall bladder stones.
Hemolytic uremic syndrome prognosis (outlook)
The outcomes of hemolytic uremic syndrome have improved, and the acute mortality rate in children is 1-4%. About 70% of patients recover completely from the acute episode and the remainder has varying degrees of sequelae. Very few retrospective studies have reviewed these patients over long periods. The kidneys bear the brunt of the long-term damage: proteinuria (15-30% of cases); hypertension (5-15%); chronic kidney disease (9-18%); and end stage kidney disease (ESKD) (3%). A small number have extra-renal sequelae such as colonic strictures, cholelithiasis, diabetes mellitus, or brain injury. Most renal sequelae are minor abnormalities such as treatable hypertension and/or variable proteinuria. Most of the patients who progress to end stage kidney disease do not recover normal renal function after the acute episode. Length of anuria [no urine] (more than 10-14 days) and prolonged dialysis are the most important risk factors for a poor acute and long-term renal outcome. After the acute episode all patients must be followed for at least five years, and severely affected patients should be followed indefinitely if there is proteinuria, hypertension or a reduced glomerular filtration rate (GFR).
Patients whose disease has progressed to end stage renal disease are candidates for kidney transplant. End stage renal disease is a condition in which the kidneys permanently fail to work, but rarely occurs in hemolytic uremic syndrome. Kidney transplants for children with hemolytic uremic syndrome are successful and the disease almost never recurs.
Hemolytic uremic syndrome causes
The most common cause of hemolytic uremic syndrome (HUS) in children is an Escherichia coli (E. coli) bacteria that produce certain toxins (shiga toxin-producing E. coli or STEC). One such strain of E. coli is known as E. coli O157:H7. Other strains of E. coli have also been linked to hemolytic uremic syndrome (HUS). The O157:H7 strain of E. coli produces a poison known as Shiga toxin or verotoxin that is absorbed through the intestines. Verotoxin damages specific cells (endothelial cells) that line the inner walls of the blood vessels, particularly those of the glomeruli [filtering bodies] in the kidneys. Damage to these blood vessels (microangiopathy) leads to complications such as anemia, thrombocytopenia, acute renal failure, and the other symptoms and findings associated with hemolytic uremic syndrome. For example, microangiopathic hemolytic anemia occurs when red blood cells are destroyed or damaged as they pass through the small damaged blood vessels. Circulating platelets are consumed in the small clots in these microscopic blood vessels resulting in thrombocytopenia and the abnormal accumulation of platelets within narrowed blood vessels, causing the formation of small blood clots (microthrombi). As a result, blood flow to organs such as the kidneys, brain, and pancreas variably decreases leading to multiple organ dysfunction or failure.
Your digestive system is made up of the gastrointestinal, or GI, tract—a series of hollow organs joined in a long, twisting tube from the mouth to the anus—and other organs that help the body break down and absorb food.
Escherichia coli (E. coli) bacteria refers to a group of bacteria normally found in the intestines of healthy humans and animals. Most of the hundreds of types of E. coli are normal and harmless and are an important part of digestion. But some strains of E. coli, usually E. coli O157:H7 but also E. coli 0104:H4— including those that cause hemolytic uremic syndrome — are responsible for serious foodborne infections. If a child becomes infected with the O157:H7 strain of E. coli, the bacteria will lodge in the digestive tract and produce toxins that can enter the bloodstream. The toxins travel through the bloodstream and can destroy the red blood cells. More recently large numbers of adults were affected by hemolytic uremic syndrome in Europe [E. coli serotype 0104:H4] and there were 4320 people with bloody diarrhea, 850 people with hemolytic uremic syndrome and 82 deaths. The onset of hemolytic uremic syndrome is preceded by an illness characterized by vomiting, abdominal pain, fever, and, usually, bloody diarrhea.
Escherichia coli (E. coli) bacteria may reside in the intestinal tract of domestic animals, mainly cattle, and may be transmitted to humans through the consumption of unpasteurized milk or infected, undercooked meat or poultry. Cases have been reported in which hemolytic uremic syndrome occurred after the consumption of unpasteurized or otherwise untreated apple juice or cider. Epidemics have followed ingestion of contaminated lettuce, spinach or bean sprouts. It is important to note that transmission may be the result of person-to-person contact within a family or at a kindergarten or an infected wading pool.
Although most people with hemolytic uremic syndrome have an associated E. coli infection, other related Shiga-toxin-producing bacteria, such as Shigella dysenteriae type I, have caused hemolytic uremic syndrome.
E.coli O157:H7 can be found in:
- undercooked meat, most often ground beef
- contaminated meat or produce
- unpasteurized, or raw, milk
- unwashed, contaminated raw fruits and vegetables
- contaminated juice
- swimming pools or lakes contaminated with feces
Sometimes, E. coli infection is spread through close contact with an infected person, such as within a family or at a day care center.
Most people who are infected with E. coli, even the more dangerous strains, don’t develop hemolytic uremic syndrome.
Other less common causes, sometimes called atypical hemolytic uremic syndrome, can include:
- taking certain medications, such as quinine sulfate (Qualaquin), some chemotherapy medications, medications containing the immunosuppressant cyclosporine (Neoral, Sandimmune, Gengraf) and anti-platelet medications
- having other viral or bacterial infections, such as HIV/AIDS or an infection with the pneumococcal bacteria
- inheriting a certain type of hemolytic uremic syndrome that runs in families
- rarely, pregnancy
Susceptibility to an uncommon type of hemolytic uremic syndrome — known as atypical hemolytic uremic syndrome, primary hemolytic uremic syndrome or complement-mediated hemolytic uremic syndrome — can be passed down genetically to children. People who have inherited the mutated gene that causes atypical hemolytic uremic syndrome won’t necessarily develop the condition. The mutated gene might be activated after an upper respiratory or abdominal infection.
Atypical hemolytic uremic syndrome
Atypical hemolytic uremic syndrome is an extremely rare disease chronic disease in which uncontrolled complement activation causes blood clots (thrombotic microangiopathy) in small blood vessels throughout the body and altered kidney function 1. Blood clots affects various organs, including the kidneys, heart, lungs, brain, and gastrointestinal systems. The complement system is part of the human immune system, which normally helps (or complements) your ability to fight illness by attacking any foreign or invading cells. Controlled by a group of genes, your complement system is usually regulated by proteins that prevent it from becoming overactive. In atypical hemolytic uremic, certain complement proteins are missing or not working properly, prompting the medical terms “complement dysregulation” or “complement-mediated” disease.
People with atypical hemolytic uremic syndrome are born with this genetic disease and have a lifelong risk of having their genetic condition suddenly become active with life-threatening complications. Atypical hemolytic uremic syndrome occurs because of a patient’s abnormal genes at birth, and while certain conditions such as bacterial or viral exposure might be among suspected triggers, genetic mutations are the underlying cause whether the mutations are identified by genetic screening or whether the genetic tests are inconclusive. Atypical hemolytic uremic syndrome is unpredictable and varies greatly in episode length, frequency of events, and severity from patient to patient. Some atypical hemolytic uremic syndrome patients will have intermittent signs and symptoms, while others have chronic symptoms on a daily basis. Some atypical hemolytic uremic syndrome events occur with rapid and devastating consequences. Historically, due to limited treatment options, the outlook for patients was poor as life-threatening thromboses (blood clots) could recur and could be fatal.
When abnormal blood clots to form in small blood vessels in the kidneys, they stop the kidneys from processing waste products from the blood and excrete them into the urine (acute kidney failure), a condition known as uremia. These clots can cause serious medical problems if they restrict or block blood flow, including hemolytic anemia, low platelet count (thrombocytopenia), and kidney failure. Atypical hemolytic uremic syndrome can occur at any age and is often caused by a combination of environmental and genetic factors. Genetic factors involve genes that code for proteins that help control the complement system (part of your body’s immune system). Environmental factors include certain medications (such as anticancer drugs), chronic diseases (e.g., systemic sclerosis and malignant hypertension), viral or bacterial infections, cancers, organ transplantation, and pregnancy. In about 60% of atypical hemolytic uremic syndrome, a gene mutation may be identified. Almost two thirds of the cases of atypical hemolytic uremic syndrome, related to an inactivating mutation of the proteins inhibiting the alternative pathway (H factor, I factor, membrane cofactor protein—MCP or CD46—and thrombomodulin) or a gain-of-function mutation of the pathway activating factors (C3 or B Factor) 2. The formation of anti-H factor IgG antibodies is associated with genetic rearrangement in the proteins related to Factor H (CFHR1). Polymorphisms of risk and variants in these genes determine the penetrance of the disease in mutation carriers 3. Mutations in these genes increase the likelihood (predisposition) to developing atypical hemolytic uremic syndrome, rather than directly causing the disease. Most cases are sporadic. In familiar cases, predisposition to atypical hemolytic uremic syndrome is inherited in an autosomal dominant or an autosomal recessive pattern of inheritance.
Atypical hemolytic uremic syndrome is a distinctly different illness from the more common disorder known as typical hemolytic uremic syndrome (HUS), which is caused by E.coli-producing Shiga toxins (Stx hemolytic uremic syndrome) and is generally foodborne. Most cases of atypical hemolytic uremic syndrome are genetic, although some may be acquired due to autoantibodies (autoimmune disorder) or occur for unknown reasons (idiopathic). Atypical hemolytic uremic syndrome may become chronic, and affected individuals may experience repeated episodes of the disorder. Unlike individuals with typical hemolytic uremic syndrome (HUS), who usually recover from the life-threatening initial episode and usually respond well to supportive treatment, individuals with atypical hemolytic uremic syndrome are much more likely to develop chronic serious complications such as severe high blood pressure (hypertension) and kidney (renal) failure. The signs and symptoms of atypical hemolytic uremic syndrome result from the formation of tiny blood clots (microthrombi) in various small blood vessels of the body. These clots reduce or prevent proper blood flow to various organs of the body, especially the kidneys. Atypical hemolytic uremic syndrome is a complex disorder and multiple factors, including certain genetic, environmental and immunologic factors, all play a role in its development.
Genetic screening can be conducted to identify the patient’s mutation, but approximately 30% to 50% of atypical hemolytic uremic syndrome patients will not have an identified genetic mutation (such as factor H, factor I, membrane cofactor protein, etc), and a diagnosis of atypical hemolytic uremic syndrome is not dependent on results of genetic tests.
A disease is classified as “rare” if it affects fewer than 500 patients per million population, but atypical hemolytic uremic syndrome bears the “ultra rare” designation reserved for diseases that affect 1 to 2 cases per million people 4. To put this into perspective, according to the United States Census Bureau as of April 2012, with America’s population of 312.8 million people, the number of patients with atypical hemolytic uremic syndrome would be fewer than 625; with the world population estimated to be 7 billion, the number of patients with atypical hemolytic uremic syndrome worldwide is estimated to be fewer than 140,000.
Atypical hemolytic-uremic syndrome causes
Atypical hemolytic-uremic syndrome often results from a combination of environmental and genetic factors. Mutations in at least seven genes appear to increase the risk of developing the disorder. Mutations in a gene called CFH are most common; they have been found in about 30 percent of all cases of atypical hemolytic-uremic syndrome. Mutations in the other genes have each been identified in a smaller percentage of cases.
The genes associated with atypical hemolytic-uremic syndrome provide instructions for making proteins involved in a part of the body’s immune response known as the complement system. This system is a group of proteins that work together to destroy foreign invaders (such as bacteria and viruses), trigger inflammation, and remove debris from cells and tissues. The complement system must be carefully regulated so it targets only unwanted materials and does not attack the body’s healthy cells. The regulatory proteins associated with atypical hemolytic-uremic syndrome protect healthy cells by preventing activation of the complement system when it is not needed.
Mutations in the genes associated with atypical hemolytic-uremic syndrome lead to uncontrolled activation of the complement system. The overactive system attacks cells that line blood vessels in the kidneys, causing inflammation and the formation of abnormal clots. These abnormalities lead to kidney damage and, in many cases, kidney failure and end stage kidney disease.
Although gene mutations increase the risk of atypical hemolytic-uremic syndrome, studies suggest that they are often not sufficient to cause the disease. In people with certain genetic changes, the signs and symptoms of the disorder may be triggered by factors including certain medications (such as anticancer drugs), chronic diseases, viral or bacterial infections, cancers, organ transplantation, or pregnancy.
Some people with atypical hemolytic-uremic syndrome do not have any known genetic changes or environmental triggers for the disease. In these cases, the disorder is described as idiopathic.
Most cases of atypical hemolytic-uremic syndrome are sporadic, which means that they occur in people with no apparent history of the disorder in their family. Less than 20 percent of all cases have been reported to run in families. When the disorder is familial, it can have an autosomal dominant or an autosomal recessive pattern of inheritance.
Autosomal dominant inheritance means one copy of an altered gene in each cell is sufficient to increase the risk of the disorder. In some cases, an affected person inherits the mutation from one affected parent. However, most people with the autosomal dominant form of atypical hemolytic-uremic syndrome have no history of the disorder in their family. Because not everyone who inherits a gene mutation will develop the signs and symptoms of the disease, an affected individual may have unaffected relatives who carry a copy of the mutation.
Autosomal recessive inheritance means both copies of a gene in each cell have mutations. The parents of an individual with an autosomal recessive condition each carry one copy of the mutated gene, but they typically do not show signs and symptoms of the condition.
Hemolytic uremic syndrome prevention
Meat or produce contaminated with E. coli won’t necessarily look, feel or smell bad. To protect against E. coli infection and other foodborne illnesses:
- Avoid unpasteurized milk, juice and cider.
- Wash hands well before eating and after using the restroom and changing diapers.
- Clean utensils and food surfaces often.
- Cook meat to an internal temperature of at least 160 degrees Fahrenheit (71.1 ° C).
- Defrost meat in the microwave or refrigerator.
- Keep raw foods separate from ready-to-eat foods.
- Don’t place cooked meat on plates previously contaminated by raw meat.
- Store meat below produce in the refrigerator to reduce the risk of liquids such as blood dripping on produce.
- Avoid unclean swimming areas. Don’t swim if you have diarrhea.
When a child is taking medications that may cause hemolytic uremic syndrome, it is important that the parent or caretaker watch for symptoms and report any changes in the child’s condition to the health care provider as soon as possible.
Hemolytic uremic syndrome symptoms
The symptoms and severity of hemolytic uremic syndrome vary greatly from one person to another. The disorder can be mild or it can progress to cause life-threatening complications. Most children with hemolytic uremic syndrome recover without permanent damage; however, about a small percent recover with complications.
A child with hemolytic uremic syndrome may develop signs and symptoms similar to those seen with gastroenteritis—an inflammation of the lining of the stomach, small intestine, and large intestine— such as:
- vomiting
- bloody diarrhea
- abdominal pain
- fever and chills
- headache
As the infection progresses, the toxins released in the intestine begin to destroy red blood cells. When the red blood cells are destroyed, the child may experience the signs and symptoms of anemia—a condition in which red blood cells are fewer or smaller than normal, which prevents the body’s cells from getting enough oxygen.
Approximately 3 to 10 days after the development of gastroenteritis, additional symptoms appear including sudden onset of paleness (pallor), irritability, weakness, diminished excretion of urine (oliguria) or no urine (anuria) and lack of energy (lethargy). In some cases, seizures also occur during this initial phase.
Signs and symptoms of anemia may include:
- fatigue, or feeling tired
- weakness
- fainting
- paleness
As the damaged red blood cells clog the glomeruli, the kidneys may become damaged and make less urine. When damaged, the kidneys work harder to remove wastes and extra fluid from the blood, sometimes leading to acute kidney injury.
Some infants may also develop small, unexplained bruises, small red or purple, pinhead-sized spots on the skin or mucous membranes (petechiae), and, rarely, bleeding from the nose or mouth.
When hemolytic uremic syndrome causes acute kidney injury, a child may have the following signs and symptoms:
- edema—swelling, most often in the legs, feet, or ankles and less often in the hands or face
- albuminuria—when a child’s urine has high levels of albumin, the main protein in the blood
- decreased urine output
- hypoalbuminemia—when a child’s blood has low levels of albumin
- blood in the urine
Almost all affected persons have kidney injury and more than half of the children with hemolytic uremic syndrome develop impaired kidney (renal) function that may progress to renal failure and require dialysis. Hemolytic uremic syndrome is the most common cause of acute renal failure in young children that is not related to complications of treatment of procedures. Renal failure is characterized by an inability of the kidneys to process waste products from the blood and excrete them in the urine, regulate the balance of salt and water in the body, excrete potassium, acids (metabolic acidosis) and phosphorus, and perform other vital functions such as control of blood pressure and production of erythropoietin and calcitriol. Acute renal failure may result in diminished amounts of urine [oliguria, anuria]; blood in the urine (hematuria); excess concentrations of protein in the urine (proteinuria), high blood pressure (hypertension); an abnormal accumulation of fluid between layers of tissue under the skin (edema); In some cases, acute renal failure may lead to life-threatening complications such as severe acidosis, high potassium and very low sodium levels.
Hemolytic uremic syndrome diagnosis
A health care provider diagnoses hemolytic uremic syndrome with:
- a medical and family history
- a physical exam
- urine tests
- a blood test
- a stool test
- kidney biopsy
Medical and Family History
Taking a medical and family history is one of the first things a health care provider may do to help diagnose hemolytic uremic syndrome.
Physical Exam
A physical exam may help diagnose hemolytic uremic syndrome. During a physical exam, a health care provider most often
- examines a child’s body
- taps on specific areas of the child’s body
Urine Tests
A health care provider may order the following urine tests to help determine if a child has kidney damage from hemolytic uremic syndrome.
- Dipstick test for albumin. A dipstick test performed on a urine sample can detect the presence of albumin in the urine, which could mean kidney damage. The child or caretaker collects a urine sample in a special container in a health care provider’s office or a commercial facility. For the test, a nurse or technician places a strip of chemically treated paper, called a dipstick, into the child’s urine sample. Patches on the dipstick change color when albumin is present in the urine.
- Urine albumin-to-creatinine ratio. A health care provider uses this measurement to estimate the amount of albumin passed into the urine over a 24-hour period. The child provides a urine sample during an appointment with the health care provider. Creatinine is a waste product that is filtered in the kidneys and passed in the urine. A high urine albumin-to-creatinine ratio indicates that the kidneys are leaking large amounts of albumin into the urine.
Blood Test
A blood test involves drawing blood at a health care provider’s office or a commercial facility and sending the sample to a lab for analysis. A health care provider will test the blood sample to:
- estimate how much blood the kidneys filter each minute, called the estimated glomerular filtration rate, or eGFR. The test results help the health care provider determine the amount of kidney damage from hemolytic uremic syndrome.
- check red blood cell and platelet levels.
- check for liver and kidney function.
- assess protein levels in the blood.
Stool Test
A stool test is the analysis of a sample of stool. The health care provider will give the child’s parent or caretaker a container for catching and storing the stool. The parent or caretaker returns the sample to the health care provider or a commercial facility that will send the sample to a lab for analysis. Stool tests can show the presence of E. coli O157:H7.
Kidney Biopsy
Biopsy is a procedure that involves taking a small piece of kidney tissue for examination with a microscope. A health care provider performs the biopsy in an outpatient center or a hospital. The health care provider will give the child light sedation and local anesthetic; however, in some cases, the child will require general anesthesia. A pathologist—a doctor who specializes in diagnosing diseases—examines the tissue in a lab. The pathologist looks for signs of kidney disease and infection. The test can help diagnose hemolytic uremic syndrome.
Hemolytic uremic syndrome treatment
Hemolytic uremic syndrome requires treatment in the hospital. To ease signs and symptoms and to prevent further problems, treatment might include:
- Fluid replacement. Lost fluid and electrolytes must be carefully replaced because the kidneys aren’t removing fluids and waste as efficiently as normal.
- Red blood cell transfusions. Red blood cells, transfused through an intravenous (IV) needle, can help reverse signs and symptoms of hemolytic uremic syndrome including chills, fatigue, shortness of breath, rapid heart rate, yellow skin and dark urine.
- Platelet transfusions. IV transfusions of platelets can help your blood clot more normally if you’re bleeding or bruising easily.
- Plasma exchange. Plasma is the part of blood that supports the circulation of blood cells and platelets. Sometimes a machine is used to clear the blood of its own plasma and replace it with fresh or frozen donor plasma.
- Kidney dialysis. Sometimes dialysis is needed to filter waste and excess fluid from the blood. Dialysis is usually a temporary treatment until the kidneys begin functioning adequately again. But if you have significant kidney damage, you might need long-term dialysis.
Monitoring fluid and electrolyte balance is essential to prevent fluid overload and dangerous complications such as elevated potassium and acid levels. In order to maintain proper fluid and electrolyte levels, intravenous fluid and/or nutritional supplementation may be required. Some affected individuals with kidney impairment may require treatment that involves using a special medical procedure to remove wastes from the blood (dialysis) until the kidneys can recover and function on their own.
Most infants and young children with hemolytic uremic syndrome tend to recover with immediate, appropriate, aggressive supportive therapy. Recovery time may be longer in affected adults, since kidney complications tend to be more extensive. Long-term follow-up and observation are usually recommended to monitor for potential chronic kidney disease and hypertension.
Antibiotic therapy for the E. coli gastroenteritis should be avoided in hemolytic uremic syndrome because antibiotics may increase the release of toxins into the intestine. However large, randomized clinical trials have not been performed to confirm these findings.
If you have lasting kidney damage from hemolytic uremic syndrome, your doctor might recommend a medication to lower your blood pressure, to prevent or delay further kidney damage. Your doctor might also recommend follow-up visits to evaluate your kidney function every year for five years.
Eating, Diet, and Nutrition
At the beginning of the illness, children with hemolytic uremic syndrome may need intravenous (IV) nutrition or supplements to help maintain fluid balance in the body. Some children may need to follow a low-salt diet to help prevent swelling and high blood pressure.
Health care providers will encourage children with hemolytic uremic syndrome to eat when they are hungry. Most children who completely recover and do not have permanent kidney damage can return to their usual diet.
Atypical hemolytic uremic syndrome
This uncommon type of hemolytic uremic syndrome is generally treated with plasma exchange. Your doctor might also recommend monoclonal antibody eculizumab (Soliris) — that can prevent the continued destruction of healthy cells. To prevent serious infection, you or your child might have a meningococcal vaccine before receiving this medication.
The administration of eculizumab is associated with improvement in the kidney function and lower rate of recurrence of thrombotic microangiopathy as compared to traditional plasmapheresis and plasma infusion therapies 5. In the post-kidney transplantation period, atypical hemolytic uremic syndrome is even more challenging, with increased mortality and high rates of recurrence of the disease, ranging from 60 to 90% in the first year 6.
The risk of recurrence of atypical hemolytic uremic syndrome in the kidney graft is correlated with the type of mutation. The kidney transplantation is highly complex in the patients suffering from atypical hemolytic uremic syndrome, since 50 to 80% of the patients with atypical hemolytic uremic syndrome may experience thrombotic microangiopathy in the kidney graft, with graft survival of 51% in five years 6. The kidney transplantation recipients are exposed to the risk of MAT by factors directly injuring the endothelium such as immunosuppressive drugs (calcineurin inhibitors and mTOR inhibitors), ischemia-reperfusion injury, rejection and post-transplantation infections 1. The use of related live donor in the patients with atypical hemolytic uremic syndrome is contraindicated due to the risk of mutations or polymorphisms that are still undetectable 7. There are reports of atypical hemolytic uremic syndrome occurring in previously asymptomatic donors after donation, potentially triggered by the surgical procedure (complement amplifying condition) 8. The use of eculizumab in kidney transplantation has been chosen as a question of priority, deserving investigation in an initiative of cooperation between patient associations and the global registry of atypical hemolytic uremic syndrome 9. Older studies suggest the combination of kidney and liver transplantation, aiming to avoid relapses in patients with certain dysfunctions of complement factors 10. Eculizumab, a terminal complement inhibitor, is approved for the treatment of atypical hemolytic uremic syndrome to control the thrombotic microangiopathy manifestations and to avoid the relapses of the disease. In the post-kidney transplantation, many studies have already described the same effect 11. However, long-term studies combining eculizumab to immunosuppressive agents used in post-kidney transplantation are lacking 12. The objective was to assess the long-term outcome of post-transplantation relapsed thrombotic microangiopathy and the incidence of adverse events in a cohort of patients primarily treated with eculizumab for atypical hemolytic uremic syndrome.
- Nester CM, Barbour T, de Cordoba SR, Dragon-Durey MA, Fremeaux-Bacchi V, Goodship TH, et al. Atypical aHUS: State of the art. Mol Immunol. Mol Immunol. 2015. September;67(1):31–42. doi: 10.1016/j.molimm.2015.03.246 https://www.ncbi.nlm.nih.gov/pubmed/25843230[↩][↩]
- De Andrade LGM, Contti MM, Nga HS, et al. Long-term outcomes of the Atypical Hemolytic Uremic Syndrome after kidney transplantation treated with eculizumab as first choice. Stepkowski S, ed. PLoS ONE. 2017;12(11):e0188155. doi:10.1371/journal.pone.0188155. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5685617/[↩]
- Noris M, Remuzzi G. Managing and preventing atypical hemolytic uremic syndrome recurrence after kidney transplantation. Curr Opin Nephrol Hypertens. 2013. November;22(6):704–12. doi: 10.1097/MNH.0b013e328365b3fe https://www.ncbi.nlm.nih.gov/pubmed/24076560[↩]
- Eculizumab for atypical hemolytic-uremic syndrome. Nürnberger J, Philipp T, Witzke O, Opazo Saez A, Vester U, Baba HA, Kribben A, Zimmerhackl LB, Janecke AR, Nagel M, Kirschfink M. N Engl J Med. 2009 Jan 29; 360(5):542-4. https://www.ncbi.nlm.nih.gov/pubmed/19179328/[↩]
- Legendre CM, Licht C, Muus P, Greenbaum LA, Babu S, Bedrosian C, et al. Terminal Complement Inhibitor Eculizumab in Atypical Hemolytic–Uremic Syndrome. N Engl J Med. 2013. June 6;368(23):2169–81. doi: 10.1056/NEJMoa1208981 http://www.nejm.org/doi/10.1056/NEJMoa1208981[↩]
- Le Quintrec M, Zuber J, Moulin B, Kamar N, Jablonski M, Lionet A, et al. Complement genes strongly predict recurrence and graft outcome in adult renal transplant recipients with atypical hemolytic and uremic syndrome. Am J Transplant. 2013. March;13(3):663–75. doi: 10.1111/ajt.12077 https://www.ncbi.nlm.nih.gov/pubmed/23356914[↩][↩]
- Barbour T, Johnson S, Cohney S, Hughes P. Thrombotic microangiopathy and associated renal disorders. Nephrol Dial Transplant. 2012. July;27(7):2673–85. doi: 10.1093/ndt/gfs279 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3398067/[↩]
- Fehrman-Ekholm I, Wadström J, Alkas J, Elinder C. Eculizumab Prevented Recurrence of Atypical Hemolytic Uremic Syndrome in a Kidney Donor after a Third Kidney Transplantation. Austin J Nephrol Hypertens. 2014;1(4): 1019. ISSN:964 2381–8[↩]
- Woodward L, Johnson S, Walle JV, Beck J, Gasteyger C, Licht C, et al. An innovative and collaborative partnership between patients with rare disease and industry-supported registries: the Global aHUS Registry. Orphanet J Rare Dis. 2016. November 21;11(1):154 doi: 10.1186/s13023-016-0537-5 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5117495/[↩]
- Saland JM, Ruggenenti P, Remuzzi G. Liver-kidney transplantation to cure atypical hemolytic uremic syndrome. J Am Soc Nephrol. 2009. May;20(5):940–9. doi: 10.1681/ASN.2008080906 http://jasn.asnjournals.org/content/20/5/940.long[↩]
- Palma LM, Langman CB. Critical appraisal of eculizumab for atypical hemolytic uremic syndrome. J Blood Med. 2016. April 12;7:39–72. doi: 10.2147/JBM.S36249 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4835139/[↩]
- Barnett AN, Asgari E, Chowdhury P, Sacks SH, Dorling A, Mamode N. The use of eculizumab in renal transplantation Clin Transplant. 2013. May-Jun;27(3):E216–29. doi: 10.1111/ctr.12102 https://www.ncbi.nlm.nih.gov/pubmed/23516966[↩]