Contents
- What is hemorrhagic fever
- Crimean Congo hemorrhagic fever
- Dengue hemorrhagic fever
- Ebola hemorrhagic fever
- Korean hemorrhagic fever
- How do humans get hemorrhagic fever with renal syndrome?
- Which rodents carry the hantaviruses that cause hemorrhagic fever with renal syndrome in humans?
- What are the symptoms of hemorrhagic fever with renal syndrome?
- How is hemorrhagic fever with renal syndrome diagnosed?
- How is hemorrhagic fever with renal syndrome treated?
- Is hemorrhagic fever with renal syndrome ever fatal?
- Marburg hemorrhagic fever
What is hemorrhagic fever
Viral hemorrhagic fever refers to a group of diseases characterized by an acute febrile syndrome with vascular damages, hemorrhagic manifestations and high mortality rates caused by several families of viruses that affect humans and animals and high mortality rates 1.
Viral hemorrhagic fevers that have the potential for human-to-human transmission and onset of large nosocomial (originating in a hospital) outbreaks include Crimean–Congo haemorrhagic fever, Ebola haemorrhagic fever, Marburg haemorrhagic fever and Lassa fever 2. Nosocomial outbreaks of viral hemorrhagic fevers are increasingly reported nowadays, which likely reflects the dynamics of emergence of viral hemorrhagic fevers. Such outbreaks are associated with an enormous impact in terms of human lives and costs for the management of cases, contact tracing and containment. Surveillance, diagnostic capacity, infection control and the overall preparedness level for management of a hospital-based viral hemorrhagic fever event are very limited in most endemic countries. These diseases are typically endemic in certain geographical regions and sometimes cause major outbreaks.
Some viral hemorrhagic fever agents could cause an outbreak of a febrile illness in 2 to 21 days after infection. Specific signs and symptoms vary according to the infecting virus, but initial signs and symptoms often include noticeable fever, fatigue, dizziness, muscle aches, loss of strength, vomiting, and diarrhea 3. Coagulation defects can lead to more severe clinical symptoms that include bleeding under the skin, causing petechiae, and sometimes conjunctivitis in the eye. Bleeding may also occur in internal organs and from orifices. Despite widespread bleeding, blood loss is rarely the cause of the death 3. Viral hemorrhagic fevers may also induce thoracic manifestations, including pleural effusion, pneumonia, pulmonary hemorrhage and hemoptysis, acute respiratory distress syndrome, and uncommon manifestations such as acute kidney injury 4.
Viral hemorrhagic fevers are caused by viruses of five distinct families: Arenaviridae, Bunyaviridae, Filoviridae, Flaviviridae, and Paramyxoviridae 5.
Each of these families share a number of features:
- They are all RNA viruses, and are all covered, or enveloped, in a fatty (lipid) coating.
- Their survival is dependent on an animal or insect host, called the natural reservoir.
- The viruses are geographically restricted to the areas where their host species live.
- Humans are not the natural reservoir for any of these viruses. Humans are infected when they come into contact with infected hosts. However, with some viruses, after the accidental transmission from the host, humans can transmit the virus to one another.
- Human cases or outbreaks of hemorrhagic fevers caused by these viruses occur sporadically and irregularly. The occurrence of outbreaks cannot be easily predicted.
- With a few noteworthy exceptions, there is no cure or established drug treatment for viral hemorrhagic fevers.
In rare cases, other viral and bacterial infections can cause a hemorrhagic fever; scrub typhus is a good example.
The viral hemorrhagic fevers include infections caused by viruses of the families:
- Flaviviridae (dengue fever, yellow fever, Omsk hemorrhagic fever, Kyasanur Forest disease, Alkhurma hemorrhagic fever),
- Bunyaviridae (Crimean-Congo hemorrhagic fever, Rift Valley fever, and Hantavirus diseases),
- Arenaviridae (Argentine, Bolivian, Brazilian, and Venezuelan hemorrhagic fevers and Lassa fever), and
- Filoviridae (Ebola and Marburg hemorrhagic fevers) 6.
These diseases are all caused by RNA viruses enveloped in a lipid bilayer coating derived from the host cell membrane 3. The persistence of these viruses in nature depends on a natural reservoir host, which is an animal or an insect. Some of these viruses may be transmitted from person to person (Lassa, Crimean Congo hemorrhagic fever, Ebola, Marburg, and several other viruses) by direct contact with blood or other body fluids of infected patients 3.
Confirmation of viral etiology is guided by the epidemiological setting simultaneously with research for other microbial or parasitic agents causing hemorrhagic fever syndromes. These diseases are typically endemic in certain geographical regions and sometimes cause major outbreaks 6.
Crimean Congo hemorrhagic fever
Crimean-Congo hemorrhagic fever is caused by infection with a tick-borne virus (Nairovirus) in the family Bunyaviridae 7. The disease was first characterized in the Crimea in 1944 and given the name Crimean hemorrhagic fever. It was then later recognized in 1969 as the cause of illness in the Congo, thus resulting in the current name of the disease.
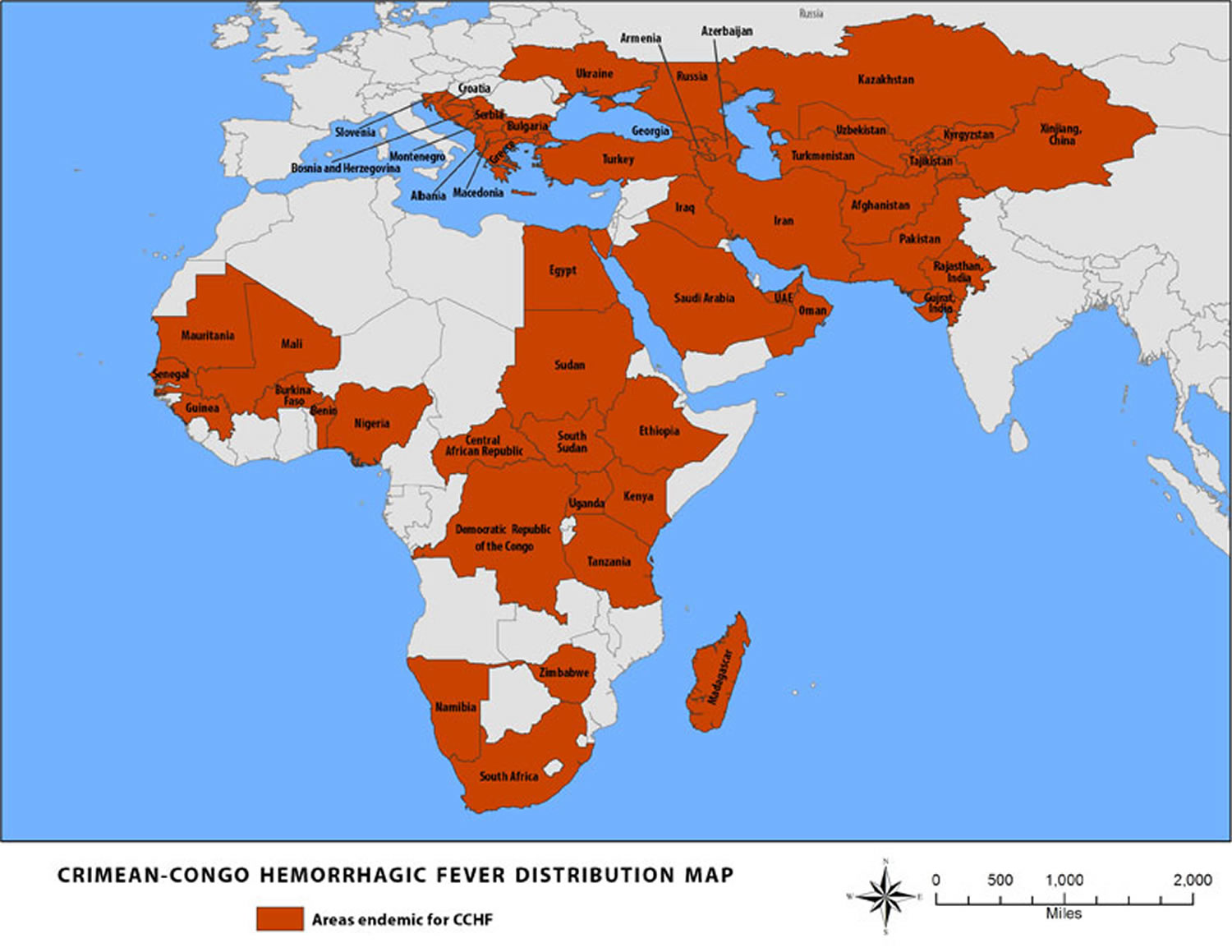
Crimean-Congo hemorrhagic fever is found in Eastern Europe, particularly in the former Soviet Union, throughout the Mediterranean, in northwestern China, central Asia, southern Europe, Africa, the Middle East, and the Indian subcontinent.
Figure 1. Crimean-Congo hemorrhagic fever outbreak distribution map
 [Source 8]
[Source 8]Crimean-Congo hemorrhagic fever Transmission
Ixodid (hard) ticks, especially those of the genus, Hyalomma, are both a reservoir and a vector for the Crimean-Congo hemorrhagic fever virus. Numerous wild and domestic animals, such as cattle, goats, sheep and hares, serve as amplifying hosts for the virus. Transmission to humans occurs through contact with infected ticks or animal blood. Crimean-Congo hemorrhagic fever can be transmitted from one infected human to another by contact with infectious blood or body fluids. Documented spread of Crimean-Congo hemorrhagic fever has also occurred in hospitals due to improper sterilization of medical equipment, reuse of injection needles, and contamination of medical supplies.
Figure 2. Ixodid (hard) ticks
Risk of Exposure
Animal herders, livestock workers, and slaughterhouse workers in endemic areas are at risk of Crimean-Congo hemorrhagic fever. Healthcare workers in endemic areas are at risk of infection through unprotected contact with infectious blood and body fluids. Individuals and international travelers with contact to livestock in endemic regions may also be exposed.
Crimean-Congo hemorrhagic fever prevention
Agricultural workers and others working with animals should use insect repellent on exposed skin and clothing. Insect repellants containing DEET (N, N-diethyl-m-toluamide) are the most effective in warding off ticks. Wearing gloves and other protective clothing is recommended. Individuals should also avoid contact with the blood and body fluids of livestock or humans who show symptoms of infection. It is important for healthcare workers to use proper infection control precautions to prevent occupational exposure.
An inactivated, mouse-brain derived vaccine against Crimean-Congo hemorrhagic fever has been developed and is used on a small scale in Eastern Europe. However, there is no safe and effective vaccine currently available for human use. Further research is needed to develop these potential vaccines as well as determine the efficacy of different treatment options including ribavirin and other antiviral drugs.
Crimean-Congo hemorrhagic fever Signs and Symptoms
The onset of Crimean-Congo hemorrhagic fever is sudden, with initial signs and symptoms including headache, high fever, back pain, joint pain, stomach pain, and vomiting. Red eyes, a flushed face, a red throat, and petechiae (red spots) on the palate are common. Symptoms may also include jaundice, and in severe cases, changes in mood and sensory perception.
As the illness progresses, large areas of severe bruising, severe nosebleeds, and uncontrolled bleeding at injection sites can be seen, beginning on about the fourth day of illness and lasting for about two weeks. In documented outbreaks of Crimean-Congo hemorrhagic fever, fatality rates in hospitalized patients have ranged from 9% to as high as 50%.
The long-term effects of Crimean-Congo hemorrhagic fever infection have not been studied well enough in survivors to determine whether or not specific complications exist. However, recovery is slow.
Crimean-Congo hemorrhagic fever diagnosis
Laboratory tests that are used to diagnose Crimean-Congo hemorrhagic fever include antigen-capture enzyme-linked immunosorbent assay (ELISA), real time polymerase chain reaction (RT-PCR), virus isolation attempts, and detection of antibody by ELISA (IgG and IgM). Laboratory diagnosis of a patient with a clinical history compatible with Crimean-Congo hemorrhagic fever can be made during the acute phase of the disease by using the combination of detection of the viral antigen (ELISA antigen capture), viral RNA sequence (RT-PCR) in the blood or in tissues collected from a fatal case and virus isolation. Immunohistochemical staining can also show evidence of viral antigen in formalin-fixed tissues. Later in the course of the disease, in people surviving, antibodies can be found in the blood. But antigen, viral RNA and virus are no more present and detectable.
Crimean-Congo hemorrhagic fever treatment
Treatment for Crimean-Congo hemorrhagic fever is primarily supportive. Care should include careful attention to fluid balance and correction of electrolyte abnormalities, oxygenation and hemodynamic support, and appropriate treatment of secondary infections. The virus is sensitive in vitro to the antiviral drug ribavirin. It has been used in the treatment of Crimean-Congo hemorrhagic fever patients reportedly with some benefit.
Crimean-Congo hemorrhagic fever recovery
The long-term effects of Crimean-Congo hemorrhagic fever infection have not been studied well enough in survivors to determine whether or not specific complications exist. However, recovery is slow.
Dengue hemorrhagic fever
Dengue is the most common arbovirus infection that belongs to the family Flaviviridae, genus Flavivirus, and can be transmitted by the bite of infective female mosquitoes of the species Aedes aegypti and, to a lesser extent, Aedes albopictus 9. There are four serotypes dengue virus (DENV1–4), but infection with one of them does not provide cross-protective immunity against the other serotypes 10 and sequential infections put people at greater risk for dengue hemorraghic fever and dengue shock syndrome 11.
Dengue haemorrhagic fever is a rare, severe – sometimes fatal – form of dengue fever, which is most often seen in children under 15 years. Initially it presents in the same way as classical dengue fever, but after a few days there is rapid deterioration and collapse.
In dengue haemorrhagic fever, large bruises often appear on the skin and there can be bleeding from the nose, gums or bowel. Urgent treatment is needed.
Figure 3. Aedes aegypti mosquito
Figure 4. Aedes albopictus mosquito
The four dengue viruses originated in monkeys and independently jumped to humans in Africa or Southeast Asia between 100 and 800 years ago 11. Dengue remained a relatively minor, geographically restricted disease until the middle of the 20th century. The disruption of the second world war – in particular the coincidental transport of Aedes mosquitoes around the world in cargo – are thought to have played a crucial role in the dissemination of the viruses. Dengue hemorrhagic fever was first documented only in the 1950s during epidemics in the Philippines and Thailand. It was not until 1981 that large numbers of Dengue hemorrhagic fever cases began to appear in the Carribean and Latin America, where highly effective Aedes control programs had been in place until the early 1970s.
Dengue most commonly occurs in tropical and subtropical areas of the world, including the Asia-Pacific, Central American and Caribbean regions.
After an incubation period of two to five days, dengue virus may cause a mild flu-like illness or quickly progresses to serious dengue hemorrhagic fever–dengue shock syndrome 12.
Alarmingly, the incidence of dengue fever increased dramatically between 1990 and 2013, from 8.3 million cases in 1990 to 58.4 million cases in 2013 13.
With more than one-third of the world’s population living in areas at risk for infection, dengue virus is a leading cause of illness and death in the tropics and subtropics. As many as 400 million people are infected yearly. There are not yet any vaccines to prevent infection with dengue virus and the most effective protective measures are those that avoid mosquito bites. When infected, early recognition and prompt supportive treatment can substantially lower the risk of medical complications and death.
Dengue has emerged as a worldwide problem only since the 1950s. Although dengue rarely occurs in the continental United States, it is endemic in Puerto Rico and in many popular tourist destinations in Latin America, Southeast Asia and the Pacific islands.
Transmission of the Dengue Virus
Dengue is transmitted between people by the mosquitoes Aedes aegypti and Aedes albopictus, which are found throughout the world. Insects that transmit disease are vectors. Symptoms of infection usually begin 4 – 7 days after the mosquito bite and typically last 3 – 10 days. In order for transmission to occur the mosquito must feed on a person during a 5- day period when large amounts of virus are in the blood; this period usually begins a little before the person become symptomatic. Some people never have significant symptoms but can still infect mosquitoes. After entering the mosquito in the blood meal, the virus will require an additional 8-12 days incubation before it can then be transmitted to another human. The mosquito remains infected for the remainder of its life, which might be days or a few weeks.
In rare cases dengue can be transmitted in organ transplants or blood transfusions from infected donors, and there is evidence of transmission from an infected pregnant mother to her fetus. But in the vast majority of infections, a mosquito bite is responsible.
In many parts of the tropics and subtropics, dengue is endemic, that is, it occurs every year, usually during a season when Aedes mosquito populations are high, often when rainfall is optimal for breeding. These areas are, however, additionally at periodic risk for epidemic dengue, when large numbers of people become infected during a short period. Dengue epidemics require a coincidence of large numbers of vector mosquitoes, large numbers of people with no immunity to one of the four virus types (DENV 1, DENV 2, DENV 3, DENV 4), and the opportunity for contact between the two. Although Aedes are common in the southern U. S., dengue is endemic in northern Mexico, and the U.S. population has no immunity, the lack of dengue transmission in the continental U.S. is primarily because contact between people and the vectors is too infrequent to sustain transmission.
Dengue in the United States
Nearly all dengue cases reported in the 48 continental states were acquired elsewhere by travelers or immigrants. Travel Associated Dengue Infections – United States, 2001- 2004 14 and Imported Dengue – United States, 1999 and 2000 15. Because contact between Aedes and people is infrequent in the continental U.S., these imported cases rarely result in secondary transmission. The last reported continental dengue outbreak was in south Texas in 2005 16. A small dengue outbreak occurred in Hawaii in 2001.
Most dengue cases in U.S. citizens occur in those inhabitants of Puerto Rico, the U.S. Virgin Islands, Samoa and Guam, which are endemic for the virus. Dengue and Dengue hemorrhagic fever have been a particular challenge in Puerto Rico, where outbreaks have been reported since 1915 and large island-wide epidemics have been documented since the late 1960s. The most recent island-wide epidemic occurred in 2007, when more than 10,000 cases were diagnosed. In Puerto Rico, and most of the Caribbean Basin, the principle dengue vector Aedes aegypti is abundant year-round. Dengue transmission in the Puerto Rico follows a seasonal pattern. Low transmission season begins in March and lasts until June, and high transmission begins in August until November.
Global Dengue
Today about 2.5 billion people, or 40% of the world’s population, live in areas where there is a risk of dengue transmission. Dengue is endemic in at least 100 countries in Asia, the Pacific, the Americas, Africa, and the Caribbean. The World Health Organization (WHO) estimates that 50 to 100 million infections occur yearly, including 500,000 Dengue hemorrhagic fever cases and 22,000 deaths, mostly among children.
Dengue Entomology & Ecology
Aedes aegypti, the principal mosquito vector of dengue viruses is an insect closely associated with humans and their dwellings. People not only provide the mosquitoes with blood meals but also water-holding containers in and around the home needed to complete their development. The mosquito lays her eggs on the sides of containers with water and eggs hatch into larvae after a rain or flooding. A larva changes into a pupa in about a week and into a mosquito in two days. Aedes aegypti main aquatic habitats; from tree cavities to toilets and learn about the mosquitoes life cycle. People also furnish shelter as Aedes aegypti preferentially rests in darker cool areas, such as closets leading to their ability to bite indoors.
Aquatic habitats are containers in which eggs develop into adult Aedes aegypti mosquitoes. Mosquitoes that transmit dengue lay eggs on the walls of water-filled containers in the house and patio. The eggs hatch when submerged in water and can survive for months. Mosquitoes can lay dozens of eggs up to 5 times during their lifetime.
There is a great variety of man-made containers on backyards or patios that collect rain water or that are filled with water by people where dengue vectors thrive. Disposing of unused containers, placing useful containers under a roof or protected with tight covers, and frequently changing the water of animal drinking pans and flower pots will greatly reduce the risk of dengue infections. Water storage containers should be kept clean and sealed so mosquitoes cannot use them as aquatic habitats.
It is very difficult to control or eliminate Aedes aegypti mosquitoes because they have adaptations to the environment that make them highly resilient, or with the ability to rapidly bounce back to initial numbers after disturbances resulting from natural phenomena (e.g., droughts) or human interventions (e.g., control measures). One such adaptation is the ability of the eggs to withstand desiccation (drying) and to survive without water for several months on the inner walls of containers. For example, if we were to eliminate all larvae, pupae, and adult Aedes aegypti at once from a site, its population could recover two weeks later as a result of egg hatching following rainfall or the addition of water to containers harboring eggs.
It is likely that Aedes aegypti is continually responding or adapting to environmental change. For example, it was recently found that Aedes aegypti is able to undergo immature development in broken or open septic tanks in Puerto Rico, resulting in the production of hundreds or thousands of Aedes aegypti adults per day.
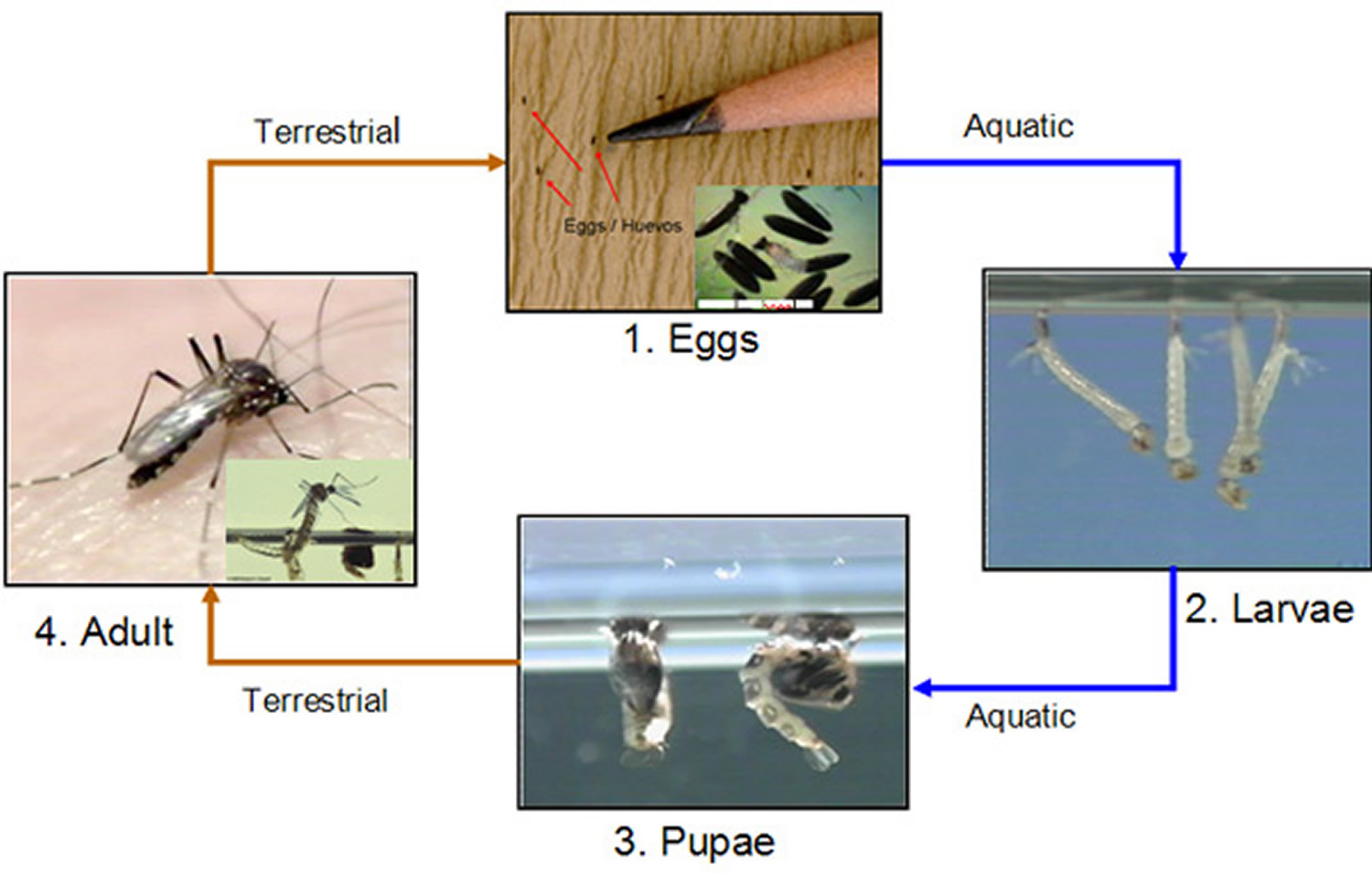
Dengue Mosquito Life-Cycle
Aedes aegypti and other mosquitoes have a complex life-cycle with dramatic changes in shape, function, and habitat. Female mosquitoes lay their eggs on the inner, wet walls of containers with water. Larvae hatch (Figure 5 – 1) when water inundates the eggs as a result of rains or the addition of water by people. In the following days, the larvae (Figure 5 – 2) will feed on microorganisms and particulate organic matter, shedding their skins three times to be able to grow from first to fourth instars. When the larva has acquired enough energy and size and is in the fourth instar, metamorphosis is triggered, changing the larva into a pupa (Figure 5 – 3). Pupae do not feed; they just change in form until the body of the adult, flying mosquito is formed. Then, the newly formed adult emerges from the water after breaking the pupal skin (Figure 5 – 4). The entire life cycle lasts 8-10 days at room temperature, depending on the level of feeding. Thus, there is an aquatic phase (larvae, pupae) and a terrestrial phase (eggs, adults) in the Aedes aegypti life-cycle.
It is this life-cycle complexity that makes it rather difficult to understand where the mosquitoes come from. Similar complex life-cycles with aquatic and terrestrial forms are observed in amphibians.
There is a very important adaptation of dengue vectors that makes controlling their populations a difficult task. Their eggs can withstand desiccation for several months, which means that even if all larvae, pupae, and adults were eliminated at some point in time, repopulation will occur as soon as the eggs in the containers are flooded with water. Unfortunately, there is no effective way to control the eggs in containers.
Figure 5. Dengue Mosquito Life-Cycle
Preventing Dengue
There’s currently no widely available vaccine for dengue. You can prevent it by avoiding being bitten by mosquitoes.
The following can reduce your risk of being bitten:
- use insect repellent – products containing 50% DEET are most effective, but lower concentrations (15-30% DEET) should be used in children, and alternatives to DEET should be used in children younger than two months
- wear loose but protective clothing – mosquitoes can bite through tight-fitting clothes; trousers, long-sleeved shirts, and socks and shoes (not sandals) are ideal
- wear light-colored clothing that covers as much of the body as possible, especially around dawn and dusk when mosquitoes are usually most active. However, note that dengue mosquitoes can bite at any time.
- kill dengue mosquitoes indoors with surface insecticide. (Dengue mosquitoes typically hide in dark places in and under the house.)
- sleep under a mosquito net – ideally one that has been treated with insecticide
- be aware of your environment – mosquitoes that spread dengue breed in standing water in urban areas
It’s a good idea to speak to your doctor, practice nurse or a travel clinic before traveling to get specific advice about what you can do to avoid dengue and other travel illnesses.
Symptoms of Dengue
Dengue fever usually affects older children and adults. Symptoms of dengue usually develop suddenly 3 to 14 days after bitten by the infecting mosquito.
Dengue cannot be spread directly from person to person. However, if a mosquito bites a person with dengue, it can pick up the virus and transmit it to other people.
Many people with dengue have no symptoms. When symptoms occur, they typically include:
- sudden onset of fever (lasting 3 to 7 days)
- a high temperature (fever), which can reach 40° C (104° F) or higher
- a severe headache
- pain behind the eyes
- muscle and joint pain – particularly affecting the ankles, knees and elbows
- feeling or being sick
- a widespread red rash
- loss of appetite, vomiting and diarrhea
- a metallic taste in the mouth
- minor bleeding from the nose and gums
- extreme fatigue
The symptoms normally pass in about a week, although you may feel tired and slightly unwell for several weeks afterwards.
In rare cases severe dengue – Dengue hemorrhagic fever can develop after the initial symptoms.
Dengue hemorrhagic fever
Dengue hemorrhagic fever is characterized by a fever that lasts from 2 to 7 days, with general signs and symptoms consistent with dengue fever. When the fever declines, warning signs may develop. This marks the beginning of a 24 to 48 hour period when the smallest blood vessels (capillaries) become excessively permeable (“leaky”), allowing the fluid component to escape from the blood vessels into the peritoneum (causing ascites) and pleural cavity (leading to pleural effusions). This may lead to failure of the circulatory system and shock, and possibly death without prompt, appropriate treatment. In addition, the patient with Dengue hemorrhagic fever has a low platelet count and hemorrhagic manifestations, tendency to bruise easily or have other types of skin hemorrhages, bleeding nose or gums, and possibly internal bleeding.
Signs of severe dengue can include:
- severe tummy (abdominal) pain
- a swollen tummy
- persistent vomiting and vomiting blood
- bleeding gums or bleeding under the skin
- breathing difficulties or fast breathing
- cold, clammy skin
- a weak but fast pulse
- drowsiness or loss of consciousness
Go IMMEDIATELY to an emergency room or the closest health care provider if any of the above warning signs.
Diagnosis and treatment of dengue
There’s no cure or specific treatment for dengue. Treatment involves relieving your symptoms while the infection runs its course.
There is no specific treatment for dengue fever. Non-specific treatments include:
- rest;
- use insect repellent so you don’t spread the disease;
- drink plenty of fluids; and
- take acetaminophen to control fever and pain (you should not take aspirin or non-steroidal anti-inflammatory medicines as they increase the risk of bleeding).
In general, most people recover fully from dengue fever within a few weeks, but it can occasionally take several months. Recovery can be complicated by depression and fatigue.
As with dengue, there is no specific medication for Dengue hemorrhagic fever . If a clinical diagnosis is made early, a health care provider can effectively treat Dengue hemorrhagic fever using fluid replacement therapy. Adequate management of Dengue hemorrhagic fever generally requires hospitalization.
Ebola hemorrhagic fever
Ebola virus disease, previously called Ebola Hemorrhagic fever, is a serious disease caused by infection with the Ebola virus.
The disease was first discovered in 1976 near the Ebola river (now the Democratic Republic of the Congo) and outbreaks continue to occur in Africa. Some of the areas that have seen Ebola virus disease outbreaks include: the Democratic Republic of the Congo (DRC), Gabon, South Sudan, Ivory Coast, Uganda, Republic of the Congo (ROC) and imported cases in South Africa.
The most recent outbreak began in March 2014 with cases first reported in Guinea, followed by the surrounding regions of Liberia, Sierra Leone and Nigeria. This outbreak is the first in West Africa and is considered to be the largest in Ebola history, with more cases and deaths in this outbreak than all the others combined.
Occurring at the same time as the outbreak in West Africa, there is an Ebola virus outbreak in the Democratic Republic of the Congo. This is believed to be unrelated to the outbreak in West Africa.
Ebola virus disease occurs at irregular intervals in time (sporadic) and hence the number of cases that occur over time (incidence) varies depending on time as well as different areas or countries.
There have been confirmed cases in Senegal (1 case), Spain (1 case) and the United States (2 cases) related to patients traveling from West Africa.
The natural source of infection of the Ebola virus remains unknown, however, fruit bats are believed to be the most likely source. Occasionally, the Ebola virus can infect and cause disease in animals such as monkeys, gorillas and chimpanzees.
Risk Factors for Ebola virus disease
There are several factors that may increase the chances of an individual becoming infected with the Ebola virus. These are largely related to that person’s risk of becoming exposed to the virus. They include:
- People who are living in or traveling to affected areas of Africa, although this risk is extremely low unless there has been direct exposure to the bodily fluids of an infected person or animal;
- Caring for those infected with Ebola virus, especially in areas with inadequate infection control; and/or
- Coming into contact with the body of an infected person or animal after death, including the handling of bushmeat.
Progression of Ebola Virus Disease
The manner in which the Ebola virus first infects a human at the start of an outbreak is unknown, however, researchers believe that first human becomes infected through contact with an infected animal. The natural source of the Ebola virus has not yet been identified, although the most likely host is thought to be bats. Humans can also be exposed from coming into contact with infected dead animals such as the handling of bushmeat.
Once infected, the time interval between infection and onset of symptoms (incubation period) is between 2 and 21 days. Humans infected with the virus are not considered infectious until they develop symptoms.
Ebola virus disease transmission
Because the natural reservoir host of Ebola viruses has not yet been identified, the way in which the virus first appears in a human at the start of an outbreak is unknown. However, scientists believe that the first patient becomes infected through contact with an infected animal, such as a fruit bat or primate (apes and monkeys), which is called a spillover event. Person-to-person transmission follows and can lead to large numbers of affected people. In some past Ebola outbreaks, primates were also affected by Ebola and multiple spillover events occurred when people touched or ate infected primates.
When an infection occurs in humans, the virus can be spread to others through direct contact (through broken skin or mucous membranes in, for example, the eyes, nose, or mouth) with:
- blood or body fluids (including but not limited to urine, saliva, sweat, feces, vomit, breast milk, and semen) of a person who is sick with or has died from Ebola,
- objects (like needles and syringes) that have been contaminated with body fluids from a person who is sick with Ebola or the body of a person who has died from Ebola,
- infected fruit bats or primates (apes and monkeys), and
- possibly from contact with semen from a man who has recovered from Ebola (for example, by having oral, vaginal, or anal sex)
Ebola is not spread through the air, by water, or in general, by food. However, in Africa, Ebola may be spread as a result of handling bushmeat (wild animals hunted for food) and contact with infected bats. There is no evidence that mosquitoes or other insects can transmit Ebola virus. Only a few species of mammals (e.g., humans, bats, monkeys, and apes) have shown the ability to become infected with and spread Ebola virus.
Healthcare providers caring for Ebola patients and family and friends in close contact with Ebola patients are at the highest risk of getting sick because they may come in contact with infected blood or body fluids.
During outbreaks of Ebola, the disease can spread quickly within healthcare settings (such as a clinic or hospital). Exposure to Ebola can occur in healthcare settings where hospital staff are not wearing appropriate personal protective equipment.
Dedicated medical equipment (preferably disposable, when possible) should be used by healthcare personnel providing patient care. Proper cleaning and disposal of instruments, such as needles and syringes, also are important. If instruments are not disposable, they must be sterilized before being used again. Without adequate sterilization of instruments, virus transmission can continue and amplify an outbreak.
Ebola virus has been found in the semen of some men who have recovered from Ebola. It is possible that Ebola could be spread through sex or other contact with semen. It is not known how long Ebola might be found in the semen of male Ebola survivors. The time it takes for Ebola to leave the semen is different for each man. Based on the results from limited studies conducted to date, it appears that the amount of virus decreases over time and eventually leaves the semen. Until more information is known, avoid contact with semen from a man who has had Ebola. It is not known if Ebola can be spread through sex or other contact with vaginal fluids from a woman who has had Ebola.
Ebola virus disease Prevention
There is no FDA-approved vaccine available for Ebola.
If you travel to or are in an area affected by an Ebola outbreak, make sure to do the following:
- Practice careful hygiene. For example, wash your hands with soap and water or an alcohol-based hand sanitizer and avoid contact with blood and body fluids (such as urine, feces, saliva, sweat, urine, vomit, breast milk, semen, and vaginal fluids).
- Do not handle items that may have come in contact with an infected person’s blood or body fluids (such as clothes, bedding, needles, and medical equipment).
- Avoid funeral or burial rituals that require handling the body of someone who has died from Ebola.
- Avoid contact with bats and nonhuman primates or blood, fluids, and raw meat prepared from these animals.
- Avoid facilities in West Africa where Ebola patients are being treated. The U.S. embassy or consulate is often able to provide advice on facilities.
- Avoid contact with semen from a man who has had Ebola until you know Ebola is gone from his semen.
- After you return, monitor your health for 21 days and seek medical care immediately if you develop symptoms of Ebola.
Healthcare workers who may be exposed to people with Ebola should follow these steps:
- Wear appropriate personal protective equipment.
- Practice proper infection control and sterilization measures. For more information, see U.S. Healthcare Workers and Settings 17.
- Isolate patients with Ebola from other patients.
- Avoid direct, unprotected contact with the bodies of people who have died from Ebola.
- Notify health officials if you have had direct contact with the blood or body fluids, such as but not limited to, feces, saliva, urine, vomit, and semen of a person who is sick with Ebola. The virus can enter the body through broken skin or unprotected mucous membranes in, for example, the eyes, nose, or mouth.
Ebola virus disease signs and symptoms
Symptoms may appear anywhere from 2 to 21 days after exposure to Ebola, but the average is 8 to 10 days.
Early signs of infection are often non-specific and resemble a flu-like illness including:
- Fever
- Neurological complaints such as confusion
- Fatigue,
- Headache, and/or
- Aching muscles.
Later signs of infection include:
- Gastrointestinal complaints such as vomiting and/or diarrhea;on and/or coma;
- Cardiovascular symptoms including changes in blood pressure and swelling of the limbs;
- Respiratory symptoms including sore throat and cough; and/or
- Rash.
Recovery from Ebola depends on good supportive clinical care and the patient’s immune response. People who recover from Ebola infection develop antibodies that last for at least 10 years.
How is Ebola Virus Disease diagnosed?
Diagnosing Ebola in a person who has been infected for only a few days is difficult because the early symptoms, such as fever, are nonspecific to Ebola infection and often are seen in patients with more common diseases, such as malaria and typhoid fever.
Ebola disease is diagnosed by a series of test that are performed depending on the patients symptoms and if there is a history to support likely exposure to the virus as discussed in risk factors.
However, a person should be isolated and public health authorities notified if they have the early symptoms of Ebola and have had contact with:
- blood or body fluids from a person sick with or who has died from Ebola,
- objects that have been contaminated with the blood or body fluids of a person sick with or who has died from Ebola,
- infected fruit bats and primates (apes and monkeys), or
- semen from a man who has recovered from Ebola
Samples from the patient can then be collected and tested to confirm infection.
Ebola virus is detected in blood only after onset of symptoms, most notably fever, which accompany the rise in circulating virus within the patient’s body. It may take up to three days after symptoms start for the virus to reach detectable levels. Laboratory tests used in diagnosis include:
Samples that may be taken for testing include:
- Blood samples;
- Throat swabs; and/or
- Urine.
A number of different scientific tests can then be done on these samples to diagnose Ebola virus disease. The most common method involves looking for the genetic material of the virus in the samples provided.
| Timeline of Infection | Diagnostic tests available |
|---|---|
| Within a few days after symptoms begin |
|
| Later in disease course or after recovery |
|
| Retrospectively in deceased patients |
|
The examination findings will vary depending on the stage of disease at the time of examination.
Early in the course of the disease your Doctor may examine your:
- Temperature to see if you have a fever;
- Eyes to see if they are bloodshot;
- Skin for evidence of a rash (especially on the torso, more evident on lighter skin and usually shows from around day 5 of infection); and/or
- Mouth for evidence of a sore throat.
As the disease progresses, examination may show:
- Changes to facial expressions
- Bleeding
- Signs of shock: increased heart rate, low blood pressure, reduced urine production
- Signs of fluid on the lungs (pulmonary edema)
- Reduced consciousness
How is Ebola virus disease treated?
Currently there is no specific treatment for Ebola virus disease, however, there are a number of potential treatments currently being investigated. Experimental vaccines and treatments for Ebola are under development, but they have not yet been fully tested for safety or effectiveness.
Supportive care involves the use of:
- fluid and electrolyte replacement / balance;
- maintaining oxygen status;
- maintaining blood pressure;
- treatment of complicating infections; and
- treatment of specific symptoms as they appear;
Recovery from Ebola depends on good supportive care and the patient’s immune response. People who recover from Ebola infection develop antibodies that last for at least 10 years, possibly longer. It is not known if people who recover are immune for life or if they can become infected with a different species of Ebola. Some people who have recovered from Ebola have developed long-term complications, such as joint and vision problems.
Even after recovery, Ebola might be found in some body fluids, including semen. The time it takes for Ebola to leave the semen is different for each man. For some men who survived Ebola, the virus left their semen in three months. For other men, the virus did not leave their semen for more than nine months. Based on the results from limited studies conducted to date, it appears that the amount of virus decreases over time and eventually leaves the semen.
Korean hemorrhagic fever
Hemorrhagic fever with renal syndrome is a group of clinically similar illnesses caused by hantaviruses from the family Bunyaviridae. Hemorrhagic fever with renal syndrome includes diseases such as Korean hemorrhagic fever, epidemic hemorrhagic fever, and nephropathia epidemica. The viruses that cause hemorrhagic fever with renal syndrome include Hantaan, Dobrava, Saaremaa, Seoul, and Puumala.
Hemorrhagic fever with renal syndrome is found throughout the world. Haantan virus is widely distributed in eastern Asia, particularly in China, Russia, and Korea. Puumala virus is found in Scandinavia, western Europe, and western Russia. Dobrava virus is found primarily in the Balkans, and Seoul virus is found worldwide. Saaremaa is found in central Europe and Scandinavia. In the Americas, hantaviruses cause a different disease known as hantavirus pulmonary syndrome.
Hantaviruses, members of the family Bunyaviridae, genus Hantavirus, cause 2 human zoonoses, hemorrhagic fever with renal syndrome in Asia and Europe and hantavirus pulmonary syndrome in North and South America 18. In their natural hosts, rodents of the families Muridae and Cricetidae, hantaviruses cause chronic infection with no apparent harm 19. Hemorrhagic fever with renal syndrome has been recognized as a serious public health problem in China since 1955 20. The disease is caused mainly by the Hantaan virus, transmitted by the striped field mouse (Apodemus agrarius), and Seoul virus, transmitted by the brown Norway rat (Rattus norvegicus) 21.
Seoul virus is a member of the hantavirus family of rodent-borne viruses. This family also includes Sin Nombre virus, which is the most common hantavirus causing disease in the United States. Most hantaviruses have only one or two rodent species as their natural host. For Seoul virus, the natural host is the Norway rat (Rattus norvegicus) and the black rat (Rattus rattus), whereas for Sin Nombre it’s the deer mouse (Peromyscus maniculatus). In the rodents that carry them, these viruses don’t cause disease, but do cause life-long infection and shedding of the virus. These viruses can occasionally spill over into other species of rodents, but they don’t cause chronic virus infections and shedding.
The severe disease associated with Sin Nombre virus infections is called hantavirus pulmonary syndrome. Most hantavirus pulmonary syndrome infections lead to fever and body aches, progressing to severe breathing difficulties that frequently require hospitalization. Death occurs in approximately 38% of cases (or 38 of every 100 patients). In contrast, the severe disease associated with Seoul virus is called hemorrhagic fever with renal syndrome. Most people who get infected with Seoul virus experience mild or even no symptoms. However, in the severe form of the disease, patients can exhibit bleeding and kidney involvement, and death occurs in approximately 1-2% of cases (or 1-2 of every 100 sick individuals). Because there is presently no effective treatment for Seoul virus infection, preventing infections in people is important.
People can become infected with the Seoul virus after coming in contact with urine, droppings, or saliva of infected rodents. When fresh rodent urine, droppings, or nesting materials are stirred up (for example, when vacuuming or sweeping), tiny particles containing the virus get into the air. This process is known as “aerosolization”. You may become infected when you breathe in these contaminated materials. You may also become infected when the urine or these other materials containing the virus get directly into a cut or other broken skin or into your eyes, nose, or mouth. In addition, people who work with live rodents can get the Seoul virus through bites from infected animals. Seoul virus is shed in the urine, feces, and saliva of recently infected rats. Rats can become infected with Seoul virus through wounding or biting other rats and after coming in contact with the urine and feces of infected rats.
Seoul virus is not known to be spread from person to person.
How do humans get hemorrhagic fever with renal syndrome?
Hantaviruses are carried and transmitted by rodents. People can become infected with these viruses and develop hemorrhagic fever with renal syndrome after exposure to aerosolized urine, droppings, or saliva of infected rodents or after exposure to dust from their nests. Transmission may also occur when infected urine or these other materials are directly introduced into broken skin or onto the mucous membranes of the eyes, nose, or mouth. In addition, individuals who work with live rodents can be exposed to hantaviruses through rodent bites from infected animals. Transmission from one human to another may occur, but is extremely rare.
Which rodents carry the hantaviruses that cause hemorrhagic fever with renal syndrome in humans?
Rodents are the natural reservoir for hantaviruses. Known carriers include the striped field mouse (Apodemus agrarius), the reservoir for both the Saaremaa and Hantaan virus; the brown or Norway rat (Rattus norvegicus), the reservoir for Seoul virus; the bank vole (Clethrionomys glareolus), the reservoir for Puumala virus; and the yellow-necked field mouse (Apodemus flavicollis), which carries Dobrava virus.
What are the symptoms of hemorrhagic fever with renal syndrome?
Symptoms of hemorrhagic fever with renal syndrome usually develop within 1 to 2 weeks after exposure to infectious material, but in rare cases, they may take up to 8 weeks to develop. Initial symptoms begin suddenly and include intense headaches, back and abdominal pain, fever, chills, nausea, and blurred vision. Individuals may have flushing of the face, inflammation or redness of the eyes, or a rash. Later symptoms can include low blood pressure, acute shock, vascular leakage, and acute kidney failure, which can cause severe fluid overload. The severity of the disease varies depending upon the virus causing the infection. Hantaan and Dobrava virus infections usually cause severe symptoms, while Seoul, Saaremaa, and Puumala virus infections are usually more moderate. Complete recovery can take weeks or months.
How is hemorrhagic fever with renal syndrome diagnosed?
Several laboratory tests are used to confirm a diagnosis of hemorrhagic fever with renal syndrome in patients with a clinical history compatible with the disease. Such patients are determined to have hemorrhagic fever with renal syndrome if they have serologic test results positive for hantavirus infection, evidence of hantavirus antigen in tissue by immunohistochemical staining and microscope examination, or evidence of hantavirus RNA sequences in blood or tissue.
How is hemorrhagic fever with renal syndrome treated?
Supportive therapy is the mainstay of care for patients with hantavirus infections. Care includes careful management of the patient’s fluid (hydration) and electrolyte (e.g., sodium, potassium, chloride) levels, maintenance of correct oxygen and blood pressure levels, and appropriate treatment of any secondary infections. Dialysis may be required to correct severe fluid overload. Intravenous ribavirin, an antiviral drug, has been shown to decrease illness and death associated with hemorrhagic fever with renal syndrome if used very early in the disease.
Is hemorrhagic fever with renal syndrome ever fatal?
Depending upon which virus is causing the hemorrhagic fever with renal syndrome, death occurs in less than 1% to as many as 15% of patients. Fatality ranges from 5-15% for hemorrhagic fever with renal syndrome caused by Hantaan virus, and it is less than 1% for disease caused by Puumala virus.
How is hemorrhagic fever with renal syndrome prevented?
Rodent control is the primary strategy for preventing hantavirus infections. Rodent populations near human communities should be controlled, and rodents should be excluded from homes. Individuals should avoid contact with rodent urine, droppings, saliva, and nesting materials, and the safety measures described below should be followed when cleaning rodent-infested areas.
Marburg hemorrhagic fever
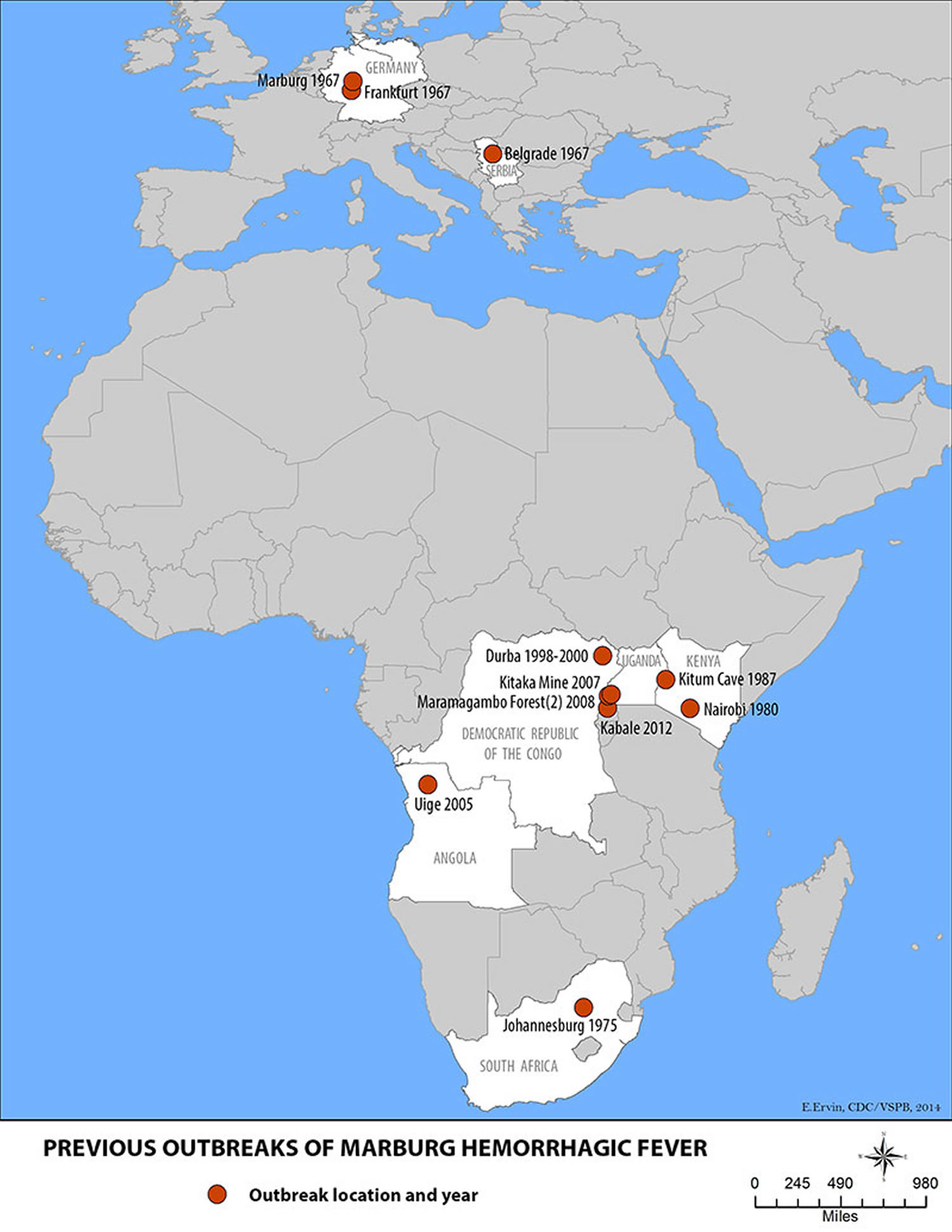
Marburg virus was first recognized in 1967, when outbreaks of hemorrhagic fever occurred simultaneously in laboratories in Marburg and Frankfurt, Germany and in Belgrade, Yugoslavia (now Serbia). Thirty-one people became ill, initially laboratory workers followed by several medical personnel and family members who had cared for them. Seven deaths were reported. The first people infected had been exposed to imported African green monkeys or their tissues while conducting research. One additional case was diagnosed retrospectively.
The reservoir host of Marburg virus is the African fruit bat, Rousettus aegyptiacus. Fruit bats infected with Marburg virus do not to show obvious signs of illness. Primates (including humans) can become infected with Marburg virus, and may develop serious disease with high mortality. Further study is needed to determine if other species may also host the virus.
This Rousettus bat is a sighted, cave-dwelling bat widely distributed across Africa. Given the fruit bat’s wide distribution, more areas are potentially at risk for outbreaks of Marburg hemorrhagic fever than previously suspected. The virus is not known to be native to other continents, such as North America.
Marburg hemorrhagic fever typically appears in sporadic outbreaks throughout Africa; laboratory confirmed cases have been reported in Uganda, Zimbabwe, the Democratic Republic of the Congo, Kenya, Angola, and South Africa. Many of the outbreaks started with male mine workers working in bat-infested mines. The virus is then transmitted within their communities through cultural practices, under-protected family care settings, and under-protected health care staff. It is possible that sporadic, isolated cases occur as well, but go unrecognized.
Cases of Marburg hemorrhagic fever have occurred outside Africa, such as during the 1967 outbreak, but are infrequent. In 2008, a Dutch tourist developed Marburg hemorrhagic fever after returning to the Netherlands from Uganda, and subsequently died. Also in 2008, an American traveler developed Marburg hemorrhagic fever after returning to the US from Uganda and recovered. Both travelers had visited a well-known cave inhabited by fruit bats in a national park.
Figure 6. Marburg hemorrhagic fever outbreak distribution map
 [Source 22]
[Source 22]
Marburg hemorrhagic fever transmission
It is unknown how Marburg virus first transmits from its animal host to humans; however, for the 2 cases in tourists visiting Uganda in 2008, unprotected contact with infected bat feces or aerosols are the most likely routes of infection.
After this initial crossover of virus from host animal to humans, transmission occurs through person-to-person contact. This may happen in several ways: direct contact to droplets of body fluids from infected persons, or contact with equipment and other objects contaminated with infectious blood or tissues.
In previous outbreaks, persons who have handled infected non-human primates or have come in direct contact with their fluids or cell cultures have become infected. Spread of the virus between humans has occurred in close environments and direct contacts. A common example is through caregivers in the home or in a hospital (nosocomial transmission).
Marburg hemorrhagic fever risk of exposure
People who have close contact with African fruit bats, humans patients, or non-human primates infected with Marburg virus are at risk.
Historically, the people at highest risk include family members and hospital staff who care for patients infected with Marburg virus and have not used proper barrier nursing techniques. Particular occupations, such as veterinarians and laboratory or quarantine facility workers who handle non-human primates from Africa, may also be at increased risk of exposure to Marburg virus.
Exposure risk can be higher for travelers visiting endemic regions in Africa, including Uganda and other parts of central Africa, and have contact with fruit bats, or enter caves or mines inhabited by fruit bats.
Marburg hemorrhagic fever prevention
Preventive measures against Marburg virus infection are not well defined, as transmission from wildlife to humans remains an area of ongoing research. However, avoiding fruit bats, and sick non-human primates in central Africa, is one way to protect against infection.
Measures for prevention of secondary, or person-to-person, transmission are similar to those used for other hemorrhagic fevers. If a patient is either suspected or confirmed to have Marburg hemorrhagic fever, barrier nursing techniques should be used to prevent direct physical contact with the patient. These precautions include wearing of protective gowns, gloves, and masks; placing the infected individual in strict isolation; and sterilization or proper disposal of needles, equipment, and patient excretions.
In conjunction with the World Health Organization, CDC has developed practical, hospital-based guidelines, titled: Infection Control for Viral Haemorrhagic Fevers In the African Health Care Setting 23. The manual can help health-care facilities recognize cases and prevent further hospital-based disease transmission using locally available materials and few financial resources.
Marburg hemorrhagic fever is a very rare human disease. However, when it occurs, it has the potential to spread to other people, especially health care staff and family members who care for the patient. Therefore, increasing awareness in communities and among health-care providers of the clinical symptoms of patients with Marburg hemorrhagic fever is critical. Better awareness can lead to earlier and stronger precautions against the spread of Marburg virus in both family members and health-care providers. Improving the use of diagnostic tools is another priority. With modern means of transportation that give access even to remote areas, it is possible to obtain rapid testing of samples in disease control centers equipped with Biosafety Level 4 laboratories in order to confirm or rule out Marburg virus infection.
Marburg hemorrhagic fever signs and symptoms
After an incubation period of 5-10 days, symptom onset is sudden and marked by fever, chills, headache, and myalgia. Around the fifth day after the onset of symptoms, a maculopapular rash, most prominent on the trunk (chest, back, stomach), may occur. Nausea, vomiting, chest pain, a sore throat, abdominal pain, and diarrhea may then appear. Symptoms become increasingly severe and can include jaundice, inflammation of the pancreas, severe weight loss, delirium, shock, liver failure, massive hemorrhaging, and multi-organ dysfunction.
Because many of the signs and symptoms of Marburg hemorrhagic fever are similar to those of other infectious diseases such as malaria or typhoid fever, clinical diagnosis of the disease can be difficult, especially if only a single case is involved.
The case-fatality rate for Marburg hemorrhagic fever is between 23-90%.
Marburg hemorrhagic fever diagnosis
Many of the signs and symptoms of Marburg hemorrhagic fever are similar to those of other more frequent infectious diseases, such as malaria or typhoid fever, making diagnosis of the disease difficult. This is especially true if only a single case is involved.
However, if a person has the early symptoms of Marburg hemorrhagic fever and there is reason to believe that Marburg hemorrhagic fever should be considered, the patient should be isolated and public health professionals notified. Samples from the patient can then be collected and tested to confirm infection.
Antigen-capture enzyme-linked immunosorbent assay (ELISA) testing, polymerase chain reaction (PCR), and IgM-capture ELISA can be used to confirm a case of Marburg hemorrhagic fever within a few days of symptom onset. Virus isolation may also be performed but should only be done in a high containment laboratory with good laboratory practices. The IgG-capture ELISA is appropriate for testing persons later in the course of disease or after recovery. In deceased patients, immunohistochemistry, virus isolation, or PCR of blood or tissue specimens may be used to diagnose Marburg hemorrhagic fever retrospectively.
Marburg hemorrhagic fever treatment
There is no specific treatment for Marburg hemorrhagic fever. Supportive hospital therapy should be utilized, which includes balancing the patient’s fluids and electrolytes, maintaining oxygen status and blood pressure, replacing lost blood and clotting factors, and treatment for any complicating infections.
Experimental treatments are validated in non-human primates models, but have never been tried in humans.
- Viral haemorrhagic fevers in healthcare settings. Ftika L, Maltezou HC. J Hosp Infect. 2013 Mar; 83(3):185-92. https://www.ncbi.nlm.nih.gov/pubmed/23333147/[↩]
- Viral haemorrhagic fevers in healthcare settings. Journal of Hospital Infection , Volume 83 , Issue 3 , 185 – 192. http://www.journalofhospitalinfection.com/article/S0195-6701(12)00395-7/fulltext[↩]
- Bray M. and Schaechter M., Hemorrhagic Fever Viruses, in Encyclopedia of Microbiology (Third Edition) 2009, Academic Press: Oxford: p. 339–353.[↩][↩][↩][↩]
- Viral hemorrhagic fever-induced acute kidney injury. Lima EQ, Nogueira ML. Semin Nephrol. 2008 Jul; 28(4):409-15. https://www.ncbi.nlm.nih.gov/pubmed/18620963/[↩]
- Viral Hemorrhagic Fevers (VHFs). https://www.cdc.gov/vhf/virus-families/index.html[↩]
- Zeller H. and Georges-Courbot M.C., Les fievres hemorragiques virales. Antibiotiques, 2006. 8(4): p. 215–220.[↩][↩]
- Crimean-Congo Hemorrhagic Fever (CCHF). https://www.cdc.gov/vhf/crimean-congo/[↩]
- Crimean-Congo Hemorrhagic Fever (CCHF) Outbreak Distribution Map. https://www.cdc.gov/vhf/crimean-congo/outbreaks/distribution-map.html[↩]
- Gubler D.J., Dengue and dengue hemorrhagic fever. Seminars in Pediatric Infectious Diseases, 1997. 8(1): p. 3–9.[↩]
- Gubler D.J., Epidemic Dengue and Dengue Hemorrhagic Fever: a Global Public Health Problem in the 21st Century, in Emerging Infections 1 1998, American Society of Microbiology.[↩]
- Dengue Epidemiology. https://www.cdc.gov/dengue/epidemiology/index.html[↩][↩]
- Yacoub S. and Wills B., Dengue: an update for clinicians working in non-endemic areas. Clinical Medicine, 2015. 15(1): p. 82–85. Clin Med (Lond). 2015 Feb;15(1):82-5. doi: 10.7861/clinmedicine.15-1-82. http://www.clinmed.rcpjournal.org/content/15/1/82.long[↩]
- Stanaway J.D., et al. , The global burden of dengue: an analysis from the Global Burden of Disease Study 2013. The Lancet Infectious Diseases, 2016. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5012511/[↩]
- Travel-Associated Dengue Infections, United States, 2001-2004. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5422a2.htm[↩]
- Imported Dengue, United States, 1999 and 2000. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5113a5.htm[↩]
- Dengue Hemorrhagic Fever, U.S.-Mexico Border, 2005. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5631a1.htm[↩]
- Ebola (Ebola Virus Disease) U.S. Healthcare Workers and Settings. https://www.cdc.gov/vhf/ebola/healthcare-us/index.html[↩]
- Schmaljohn C, Hjelle B. Hantaviruses: a global disease problem. Emerging Infectious Diseases. 1997;3(2):95-104. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2627612/pdf/9204290.pdf[↩]
- Netski D, Thran BH, St Jeor SC Sin Nombre virus pathogenesis in Peromyscus maniculatus. J Virol 1999;73:585–91 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC103864/[↩]
- Zhang YZ, Xiao DL, Wang Y, Wang HX, Sun L, Tao XX, et al. The epidemic characteristics and preventive measures of hemorrhagic fever with syndromes in China [in Chinese] Zhonghua Liu Xing Bing Xue Za Zhi. 2004;25:466–9 https://www.ncbi.nlm.nih.gov/pubmed/15231118[↩]
- Song G Epidemiological progresses of hemorrhagic fever with renal syndrome in China. Chin Med J (Engl) 1999;112:472–7 https://www.ncbi.nlm.nih.gov/pubmed/11593522[↩]
- Marburg hemorrhagic fever (Marburg HF) Outbreak Distribution Map. https://www.cdc.gov/vhf/marburg/outbreaks/distribution-map.html[↩]
- Infection Control for Viral Haemorrhagic Fevers in the African Health Care Setting. https://www.cdc.gov/vhf/abroad/vhf-manual.html[↩]