Contents
What is hormone replacement therapy
Hormone replacement therapy is now replaced with “hormone therapy”, is another umbrella term your healthcare provider might use that refers to either estrogen therapy or estrogen-progestogen therapy. The term “hormone replacement therapy” is no longer used by the Food and Drug Administration (FDA) or the North American Menopause Society because the goal of hormone therapy is to provide the amount of hormones required to relieve symptoms, not “replace” the amount produced before menopause. It is considered normal to have low estrogen and progesterone after menopause. Menopause is not a deficiency disease.
There are three benchmark stages of natural menopause:
- Perimenopause (or the menopause transition) is the span of time between the start of symptoms (such as erratic periods) and 1 year after the final menstrual period.
- Menopause is confirmed 1 year (12 months) after the final menstrual period.
- Postmenopause is all the years beyond menopause.
Menopause is the time in a woman’s life when she naturally stops having menstrual periods. Menopause marks the end of the reproductive years. The average age of menopause for women in the United States is 51 years.
Most women enter a transitional phase in the years leading up to menopause called perimenopause. Perimenopause is a time of gradual change in the levels of estrogen, a hormone that helps control the menstrual cycle. Changing estrogen levels can bring on symptoms such as hot flashes and sleep changes. To manage these symptoms, some women may choose to take hormone therapy.
Hormone therapies are the prescription drugs used most often to treat hot flashes and genitourinary syndrome of menopause, which includes vaginal dryness, after menopause. Hormone therapy can help relieve the symptoms of perimenopause and menopause. Hormone therapy means taking estrogen and, if you have never had a hysterectomy and still have a uterus, progestin. Progestin is a form of progesterone. Taking progestin helps reduce the risk of cancer of the uterus that occurs when estrogen is used alone. If you do not have a uterus, estrogen is given without progestin. Estrogen plus progestin sometimes is called “combined hormone therapy” or simply “hormone therapy.” Estrogen-only therapy sometimes is called “estrogen therapy.”
There are two basic types of hormone (replacement) therapy:
- Estrogen therapy means estrogen-only therapy. Estrogen is the hormone that provides the most menopausal symptom relief. Estrogen therapy is prescribed for women without a uterus due to a hysterectomy.
- Estrogen-Progestogen therapy means combined estrogen plus progestogen therapy. Progestogen is added to estrogen therapy to protect women with a uterus against uterine (endometrial) cancer from estrogen alone.
Your Body’s Hormones
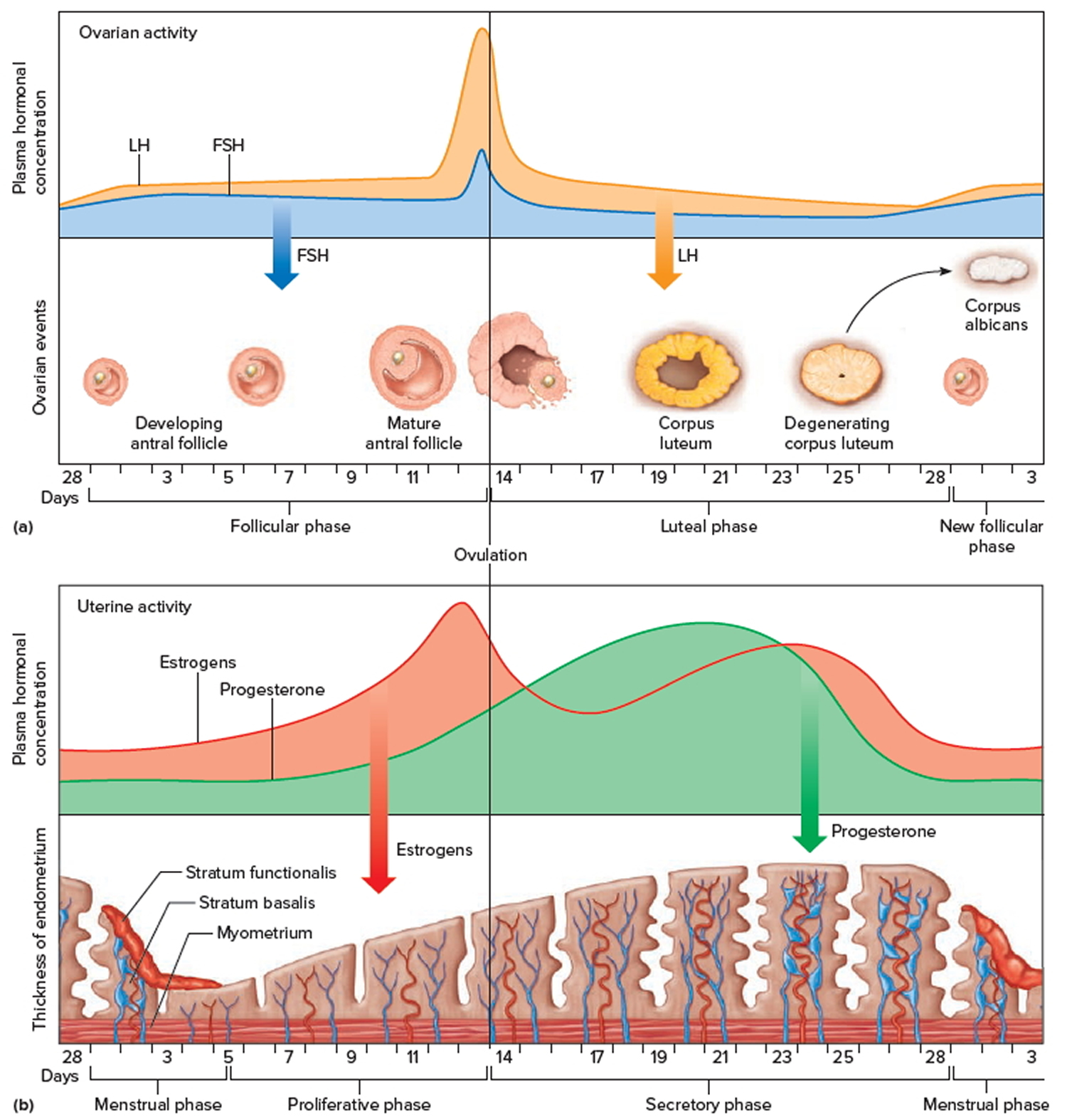
During your childbearing years, monthly changes in the production of two hormones—estrogen and progesterone—control your menstrual cycle. These hormones are made by the ovaries. Estrogen causes the endometrium (the lining of the uterus) to grow and thicken to prepare for a possible pregnancy. On about day 14 of your menstrual cycle, an egg is released from one of the ovaries, a process called ovulation. If the egg is not fertilized, no pregnancy occurs. This causes the levels of estrogen and progesterone to decrease, which signals the uterus to shed its lining. This shedding is your monthly period.
Figure 1. Ovarian activity during the Menstrual cycle
Note: Major events in the female menstrual cycle. (a) Plasma hormonal concentrations of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) affect follicle maturation in the ovaries. (b) Plasma hormonal concentrations of estrogen and progesterone influence changes in the uterine lining.
Estrogen
This is the “female hormone” that promotes the development and maintenance of female sex characteristics for purposes of reproduction. The 3 main estrogen types called estrone, 17beta-estradiol (most biologically active), and estriol (highest in pregnancy) all decrease at menopause, and that decrease can result in menopause symptoms such as hot flashes and vaginal dryness.
What is Estrogen therapy
Various estrogens can be used by women in different ways (pills; skin patches and gels; vaginal creams, rings, and tablets) for the treatment of distressing menopause-related conditions. Therapy with estrogen alone is generally appropriate only for women who have had a hysterectomy and do not need any uterine protection in the form of progestogens (either as natural progesterone or synthetic progestin) to prevent uterine cancer. (See more below)
“Systemic” (meaning throughout the body) oral and skin preparations of estrogen therapy are government approved in the United States and Canada for the treatment of moderate to severe hot flashes and vaginal atrophy. Most of these products are also approved for lowering the risk of osteoporosis if used long term. “Local” low-dose vaginal estrogen therapy is effective (and approved) for vaginal atrophy only.
Estrogen therapy has been widely studied and used for more than 50 years by millions of women. Systemic estrogen therapy is associated with side effects, such as an increased risk of stroke, blood clots, and possibly breast cancer if used long term. Estrogen therapy should be used at the lowest effective dose consistent with a woman’s treatment goals.
Progesterone
Often called the “nurturing hormone,” progesterone signals the uterus to prepare a lining of tissue for a fertilized egg. It also acts to maintain pregnancy and promote development of mammary glands (breasts). In women having periods, progesterone is produced by the ovary only after ovulation (or the release of an egg). If the egg is not fertilized, levels of progesterone fall and menstruation results. The end of ovulation at menopause means the end of progesterone production as well.
What is Progestogen therapy
Progestogen therapy is an umbrella term used to describe therapy that aims to mimic the effects of the hormone progesterone. Natural progesterone and synthetic progestins with progesterone-like activity are all progestogens. These hormones have sometimes been used alone during perimenopause to treat symptoms such as hot flashes when a woman cannot use estrogen, but their most common use is to protect against uterine cancer associated with estrogen therapy.
What is Estrogen-Progestogen therapy
Women with a uterus who wish to use estrogen for symptom relief should combine it with a progestogen to protect the lining of the uterus (endometrium). Estrogen stimulates the uterine lining and causes it to thicken, increasing risk for endometrial cancer (cancer of the lining of the uterus). Progestogen is used to decrease the risk caused from estrogen therapy, but does not protect against the type of uterine cancer that that is unrelated to estrogen. Estrogen-progestogen therapy is associated with side effects similar to estrogen therapy and should be used at the lowest effective dose consistent with treatment goals. The risk of breast cancer appears to be higher with estrogen-progestogen therapy, especially when used long-term (more than 5 yrs).
Androgens
Androgens — Often called “male hormones,” androgens are also produced in the female body as testosterone and dehydroepiandrosterone (DHEA), among others, but in much lower quantities than in men. Insufficient androgen levels at any age are thought to contribute to fatigue, mood changes, and lowered sex drive. There is no dramatic change in androgen levels at menopause; androgen production seems to be affected more by aging, although women who have their ovaries removed (surgical menopause) sometimes experience a sharper drop in their levels of testosterone. Interestingly, testosterone has been found to increase again in older women.
What is Androgen therapy ?
Some studies have shown a beneficial effect of androgen therapy on women’s sex drive. There are no government-approved androgen products available for women in the US or Canada although a number of testosterone products for women are currently under development and study. Some testosterone products approved for men are prescribed for women (called “off-label” use) but in much lower doses than used for men. DHEA is available over the counter in the US but not Canada where a prescription is required. Custom-compounded androgen products are also available through prescription. There are many uncertainties about the role of androgens in female health, and while the risks and side effects are rare if the level is kept within the female normal range, high doses may cause side effects and may not improve sex drive., Further study is needed to determine the effectiveness and safety of long-term androgen use by women.
There are two general ways to take hormone therapy
- Systemic products circulate throughout the bloodstream and to all parts of the body. They are available as an oral tablet, patch, gel, emulsion, spray, or injection and can be used for hot flashes and night sweats, vaginal symptoms, and osteoporosis.
- Local (nonsystemic) products affect only a specific or localized area of the body. They are available as a cream, ring, or tablet and can be used for vaginal symptoms.
Systemic Hormone Therapy
Hormone therapy can be either “systemic” or “local.” These two terms describe where and how the hormones act in the body. With systemic therapy, the hormones are released into your bloodstream and travel to the organs and tissues where they are needed. Systemic forms of estrogen include pills, skin patches, and gels and sprays that are applied to the skin. If progestin is prescribed, it can be given separately or combined with estrogen in the same pill or in a patch.
For hot flashes, hormones are given in pills, patches, sprays, gels, emulsion or injection that deliver hormones throughout the body—known as “systemic” therapy.
For women taking estrogen-only therapy, estrogen may be taken every day or every few days, depending on the way the estrogen is given. For women taking combined therapy, there are two types of regimens:
- Cyclic therapy: Estrogen is taken every day, and progestin is added for several days each month or for several days every 3 months or 4 months.
- Continuous therapy: Estrogen and progestin are taken every day.
Systemic hormones include estrogens—either the same or similar to the estrogens the body produces naturally—and progestogens, which include progesterone—the progestogen the body produces naturally—or a similar compound. Another approach to systemic hormone therapy is a pill that combines conjugated estrogens (those in the brand Premarin) and a compound known as a “SERM” (selective estrogen receptor modulator) that protects the uterus but is not a progestogen. Women who have had a hysterectomy (had their uterus or womb removed) can use estrogen alone to control their hot flashes. Women who still have a uterus or womb need to take a progestogen in addition to estrogen or the estrogen-SERM combination to protect against uterine cancer. Systemic hormones are very effective for hot flashes and have other benefits, such as protecting your bones. They also carry risks, such as blood clots and breast cancer. The breast cancer risk usually doesn’t rise until after about 5 years with estrogen-progestogen therapy or after 7 years with estrogen alone.
Local Hormone Therapy
Women who only have vaginal dryness may be prescribed “local” estrogen therapy in the form of a vaginal ring, tablet, or cream. These forms release small doses of estrogen into the vaginal tissue. The estrogen helps restore the thickness and elasticity to the vaginal lining while relieving dryness and irritation.
For genitourinary symptoms, hormones are given in creams, pills, or rings that are inserted into the vagina. (An approved pill is also available to treat genitourinary symptoms that is not considered a hormone but does affect estrogen receptors, mostly in and around the vagina.)
Vaginal estrogen therapy for genitourinary syndrome of menopause after menopause is administered in the vagina and is effective for both moisturizing and rebuilding tissue. Very little goes into blood circulation, so the risks are far lower.
You should discuss your individual risks and preferences with your healthcare team to determine whether hormone therapy or alternatives, including FDA-approved nonhormonal therapies, are right for you.
Current hormone therapy practice
Begin hormone therapy with the lowest effective dose for the shortest amount of time consistent with their individual goals 1. The benefit-risk ratio is favorable for women who initiate hormone therapy close to menopause (ages 50-59, typically) but becomes riskier with time since menopause and advancing age.
Women with early menopause before age 40 without a history of breast cancer risk can take hormone therapy until the typical age of menopause at 51 if there is no reason not to take it.
Clinicians will recommend an individualized plan for each woman. There is no “one size fits all” therapy.
Benefits of hormone therapy
Literally hundreds of clinical studies have provided evidence that systemic hormone therapy (estrogen with or without progestogen) effectively helps such conditions as hot flashes, vaginal dryness, night sweats, and bone loss. These benefits can lead to improved sleep, and sexual relations, and quality of life.
- The primary indications for hormone therapy are hot flashes, night sweats, vaginal dryness, and prevention of osteoporosis.
Hormone replacement therapy side effects
In 2002, a study that was part of the Women’s Health Initiative (WHI), funded by the National Institutes of Health, was stopped early because participants who received a certain kind of estrogen with progesterone were found to have a significantly higher risk of stroke, heart attacks, breast cancer, dementia, urinary incontinence, and gallbladder disease.
This study raised significant concerns at the time and left many women wary of using hormones.
However, research reported since then found that younger women may be at less risk and have more potential benefits than was suggested by the WHI study. The negative effects of the WHI hormone treatments mostly affected women who were over age 60 and post-menopausal. Newer versions of treatments developed since 2002 may reduce the risks of using hormones for women experiencing the menopausal transition, but studies are needed to evaluate the long-term safety of these newer treatments.
As a result of the Women’s Health Initiative (WHI) trial in 2002, the US Food & Drug Administration and Health Canada require all estrogen-containing prescription therapies to carry a “black box” warning in their prescribing information about the adverse risks of hormone therapy. Although only two products were studied in the WHI, Premarin and Prempro, the risks of all hormone therapy products, including “natural” bioidentical and compounded hormones, should be assumed to be similar until evidence shows otherwise.
In order to minimize serious health risks, hormone therapy is recommended at the lowest effective dose for the shortest time period and in consultation with a doctor. The real concern about hormone safety is with long-term use of systemic estrogen therapy or estrogen-progestogen therapy.
- Both estrogen therapy and estrogen with progestogen therapy increase the risk of blood clots in the legs (deep vein thrombosis) and lungs, similar to birth control pills, patches, and vaginal rings. Although the risks of blood clots and strokes increase with either type of hormone therapy, the risk is rare in the 50 to 59 age group. Forms of therapy not taken by mouth (patches, sprays, rings, and others) have less risk of causing DVT than those taken by mouth.
- An increased risk in breast cancer is seen with 5 or more years of continuous estrogen-progestin therapy, possibly earlier. To put the risk into numbers, if 10,000 women took estrogen-progestin therapy for a year, it would result in up to about 8 more cases of breast cancer per year than if they had not taken hormone therapy. The risk decreases after hormone therapy is stopped. Use of estrogen alone for an average of 7 years in the Women’s Health Initiative trial did not increase the risk of breast cancer. Currently, it is recommended that women with a history of hormone-sensitive breast cancer try non-hormonal therapies first for the treatment of menopausal symptoms.
- Estrogen therapy causes the lining of the uterus to grow and can increase the risk of uterine cancer. Adding progestin decreases the risk of uterine cancer.
- Combined hormone therapy is linked to a small increased risk of heart attack. This risk may be related to age, existing medical conditions, and when a woman starts taking hormone therapy. Some research suggests that for women who start combined therapy within 10 years of menopause and who are younger than 60 years, combined therapy may protect against heart attacks. However, combined hormone therapy should not be used solely to protect against heart disease.
- Combined hormone therapy may cause vaginal spotting. Some women may have heavier bleeding like that of a menstrual period. If you are postmenopausal, it is important to tell your health care provider if you have bleeding. Although it is often an expected side effect of hormone therapy, it also can be a sign of endometrial cancer. All bleeding after menopause should be evaluated. Other side effects reported by women who take hormone therapy include fluid retention and breast soreness. This soreness usually lasts for a short time.
- Ovarian cancer. Risk factors for ovarian cancer are harder to study because it is a less common cancer. Even when something increases the risk of developing ovarian cancer, the risk of actually getting this cancer is still likely to be low. The WHI did not find a real difference in ovarian cancer risk with estrogen-progestin therapy. Although there were more cases of ovarian cancer in the women on EPT, this may have been due to chance because of the small number of women who were affected with this cancer. However, a recent analysis combined the results of more than 50 studies, including randomized controlled trials and observational studies. This analysis found that women who took estrogen and progestin (progesterone) after menopause did have an increased risk of getting ovarian cancer. The risk was highest for women taking hormones, and decreased over time after the hormones were stopped. To put the risk into numbers, if 1,000 women who were 50 years old took hormones for menopause for 5 years, one extra ovarian cancer would be expected to develop.
- There is a small increased risk of gallbladder disease associated with estrogen therapy with or without progestin. The risk is greatest with forms of therapy taken by mouth.
- Colorectal cancer. In the WHI study of estrogen-progestin therapy, the results were mixed. Women who took estrogen-progestin therapy had a lower risk of getting colorectal cancer at all, but the cancers they got were more advanced (more likely to have spread to lymph nodes or distant sites) than the cancers in the women not taking hormones. Some observational studies have found a lower risk of colorectal cancer in women taking estrogen-progestin therapy, but some did not. So far, though, observational studies have not linked estrogen-progestin therapy with a higher risk of colorectal cancer.
- Lung cancer. Estrogen-progestin therapy is not linked to a higher risk of getting lung cancer, but it is linked to a higher risk of dying from lung cancer.
- Skin cancer. Estrogen-progestin therapy is not linked to a higher risk of any type of skin cancer (including both melanoma and other types of skin cancer).
- Some women should not use hormones for their hot flashes. You should not take hormones for menopausal symptoms if:
- You have had certain kinds of cancers, like breast cancer or uterine cancer
- You have had a stroke or heart attack, or you have a strong family history of stroke or heart disease
- You have had blood clots
- You have had problems with vaginal bleeding or have a bleeding disorder
- You have liver disease
- You think you are pregnant or may become pregnant
- You have had allergic reactions to hormone medications
Talk with your doctor to find out if taking hormones to treat your symptoms is right for you.
Estrogen therapy and cancer risk
Endometrial cancer
In women who still have a uterus, using systemic estrogen therapy has been shown to increase the risk of endometrial cancer (cancer of the lining of the uterus – the endometrium). The risk remains higher than average even after estrogen therapy is no longer used. Although most studies that showed an increased risk were of women taking estrogen as a pill, women using a patch or high-dose vaginal ring can also expect to have an increased risk of endometrial cancer.
Because of this increased cancer risk, women who have gone through menopause and who still have a uterus are given a progestin along with estrogen. Studies have shown that estrogen-progestin therapy does not increase the risk for endometrial cancer.
Long-term use of vaginal creams, rings, or tablets containing topical estrogen doses may also increase the levels of estrogen in the body. It’s not clear if this leads to health risks, but the amounts of hormone are much smaller than systemic therapies.
Breast cancer
Estrogen therapy is not linked to a higher risk of breast cancer. In fact, certain groups of women taking estrogen therapy, such as women who had no family history of breast cancer and those who had no history of benign breast disease, had a slightly lower risk of breast cancer.
Ovarian cancer
The WHI study of estrogen therapy did not report any results about ovarian cancer.
However, a recent analysis combined the results of more than 50 studies, including randomized controlled trials and observational studies. This analysis found that women who took estrogen after menopause did have an increased risk of getting ovarian cancer. The risk was highest for women currently taking estrogen, and decreased over time after estrogen was stopped.
To put the risk into numbers, if 1,000 women who were 50 years old took estrogen for menopause for 5 years, one extra ovarian cancer would be expected to develop.
Observational studies have shown that women who take estrogen therapy have a higher risk for ovarian cancer compared with women who take no hormones after menopause. The overall risk remains low, but it does increase the longer a woman uses estrogen therapy. The risk of ovarian cancer goes down after a woman stops taking the hormone.
Colorectal cancer
In the WHI study, estrogen therapy did not seem to have any effect on the risk of colorectal cancer.
Observational studies have found a lower risk of colorectal cancer in women who have used estrogen therapy for many years.
Lung cancer
Estrogen therapy does not seem to have any effect on the risk of lung cancer.
Skin cancer
Estrogen therapy is not linked to a higher risk of any type of skin cancer (including both melanoma and other types of skin cancer).
Weighing Benefits & Risks of Hormone Therapy
The decision to use estrogen, alone (estrogen therapy) or with a progestin therapy (estrogen-progestin therapy), after menopause should be made by each woman and her doctor after weighing the possible risks and benefits.
In general, hormone therapy use should be limited to the treatment of menopausal symptoms at the lowest effective dose for the shortest amount of time possible. Continued use should be reevaluated on a yearly basis. Some women may require longer therapy because of persistent symptoms.
Benefits
Hormone therapy has the following benefits:
- Systemic estrogen therapy (with or without progestin) has been shown to be the best treatment for the relief of hot flashes and night sweats.
- Systemic and local types of estrogen therapy relieve vaginal dryness.
- Systemic estrogen protects against the bone loss that occurs early in menopause and helps prevent hip and spine fractures.
- Combined estrogen and progestin therapy may reduce the risk of colon cancer.
There is no single way to ensure the best possible quality of life around menopause and beyond. Each woman is unique and must weigh her discomfort against her fear of treatment. Risk is defined as the possibility or chance of harm; it does not indicate that harm will occur. Generally, hormone therapy risks are lower in younger women than originally reported in all women ages 50 to 70 combined. It is now believed that women taking estrogen alone—women who have had their uterus removed by a hysterectomy—have a more favorable benefit-risk profile than those taking estrogen-progestogen therapy. This is especially true for younger menopausal women (in their 50s or within 10 years of menopause) than for older women.
Medical professionals have modified their views about the role of hormones as more research has been conducted. Experts agree that there is much they still have to learn. Although recent studies such as the Women’s Health Initiative (WHI) have provided some clarity for large populations, they don’t necessarily address all of the issues an individual woman faces. Only she, with the counsel of her healthcare providers, can do that.
Many factors will be part of a woman’s decision to use a particular hormone product—her age, her risks, her preferences, available treatment options, and the cost of the product. Do her potential benefits outweigh her potential risks? Only after examining and understanding her own situation and after a thorough consultation with her clinician can a woman make the best treatment choice. As new therapies and guidelines are available, and as a woman’s body changes over time, reevaluation and adjustments should be made.
Reducing the cancer risks of hormone therapy
If you and your doctor decide that menopausal hormone therapy is the best way to treat symptoms or problems caused by menopause, keep in mind that it is medicine and like any other medicine it’s best to use it at the lowest dose needed for as short a time as possible. And just as you would if you were taking another type of medicine, you need to see your doctor regularly. Your doctor can see how well the treatment is working, monitor you for side effects, and let you know what other treatments are available for your symptoms.
All women should report any vaginal bleeding that happens after menopause to their doctors right away – it may be a symptom of endometrial cancer. A woman who takes estrogen-progestin therapy does not have a higher risk of endometrial cancer, but she can still get it.
Women using vaginal cream, rings, or tablets containing only estrogen should talk to their doctors about follow-up and the possible need for progestin treatment.
For women who have had a hysterectomy (surgery to remove the uterus), a progestin does not need to be a part of hormone therapy because there’s no risk of endometrial cancer. Adding a progestin does raise the risk of breast cancer, so estrogen therapy is a better option for women without a uterus.
What is Bioidentical hormone therapy ?
The term “bioidentical hormone therapy” began as a marketing term for custom-compounded hormones that are the same chemical and molecular structure as hormones that are produced by the human female. Some people use the term to mean compounded hormones (meaning custom mixed), but bioidentical hormones do not have to be custom-compounded (meaning custom mixed) hormones. There are many well-tested, FDA-approved hormone therapy products that meet this definition and are commercially available from retail pharmacies in a variety of doses that will allow you and your doctor to customize your therapy to meet your needs. Furthermore, the FDA does not recognize the term “bioidentical hormone therapy” as an acceptable scientific term and will not use that term in their drug labeling.
The FDA has created a page called Bio-Identicals: Sorting Myths from Facts 2, specifically to address the misleading and false information by marketers of bioidentical hormone replacement therapy. FDA is concerned that claims like these: “A natural, safer alternative to dangerous prescription drugs”; “Can slim you down by reducing hormonal imbalances”; “Prevents Alzheimer’s disease and senility”, mislead women, giving them a false sense of assurance about using potentially dangerous hormone products. The FDA is providing the facts about “bioidentical hormone therapy” drugs and the uncertainties surrounding their safety and effectiveness so that women and their doctors can make informed decisions about their use. FDA has not approved compounded “bioidentical hormone therapy” drugs and cannot assure their safety or effectiveness.
FDA is taking action against pharmacies that make false and misleading claims about “bioidentical hormone therapy” drugs and is encouraging consumers to become informed about these products and their risks. Here is some information to help sort the myths from the facts:
Myth: “Bio-identical” hormones are safer and more effective than FDA-approved MHT drugs.
Fact: FDA is not aware of any credible scientific evidence to support claims made regarding the safety and effectiveness of compounded “bioidentical hormone therapy” drugs. “They are not safer just because they are ‘natural,'” says Kathleen Uhl, M.D., Director of FDA’s Office of Women’s Health.
Drugs that are approved by FDA must undergo the agency’s rigorous evaluation process, which scrutinizes everything about the drug to ensure its safety and effectiveness—from early testing, to the design and results of large clinical trials, to the severity of side effects, to the conditions under which the drug is manufactured. FDA-approved menopausal hormone therapy drugs have undergone this process and met all federal standards for approval. No compounded “bioidentical hormone therapy” drug has met these standards.
Pharmacies that compound these “bioidentical hormone therapy” drugs may not follow good drug manufacturing requirements that apply to commercial drug manufacturers. Compounding pharmacies custom-mix these products according to a health care professional’s order. The mix contains not only the active hormone, but other inactive ingredients that help hold a pill together or give a cream, lotion, or gel its form and thickness so that it can be applied to the body. It is unknown whether these mixtures, which are not FDA-approved, are properly absorbed or provide the appropriate levels of hormones needed in the body. It is also unknown whether the amount of drug delivered is consistent from pill to pill or each time a cream or gel is applied.
Myth: “Bio-identical” hormone products can prevent or cure heart disease, Alzheimer’s disease, and breast cancer.
Fact: Compounded “bioidentical hormone therapy” drugs have not been shown to prevent or cure any of these diseases. In fact, like FDA-approved menopausal hormone therapy drugs, they may increase the risk of heart disease, breast cancer, and dementia in some women. (See www.nhlbi.nih.gov/whi/index.html for information on the Women’s Health Initiative, a large, long-term study that tested the effects of FDA-approved menopausal hormone therapy drugs.) No large, long-term study has been done to determine the adverse effects of “bio-identical” hormones.
Myth: “Bio-identical” hormone products that contain estriol, a weak form of estrogen, are safer than FDA-approved estrogen products.
Fact: FDA has not approved any drug containing estriol. The safety and effectiveness of estriol are unknown. “No data have been submitted to FDA that demonstrate that estriol is safe and effective,” according to Daniel Shames, M.D., a senior official in the FDA office that oversees reproductive products.
Myth: If “bio-identical” products were unsafe, there would be a lot of reports of bad side effects.
Fact: “Bio-identical” products are typically compounded in pharmacies. “Unlike commercial drug manufacturers, pharmacies aren’t required to report adverse events associated with compounded drugs,” says Steve Silverman, Assistant Director of the Office of Compliance in FDA’s Center for Drug Evaluation and Research. “Also, while some health risks associated with ‘bioidentical hormone therapy’ drugs may arise after a relatively short period of use, others may not occur for many years. One of the big problems is that we just don’t know what risks are associated with these so-called ‘bio-identicals.'”
Myth: A pharmacy can make a “bioidentical hormone therapy” drug just for you based on hormone levels in a saliva sample.
Fact: “Advertisements that a drug can be created ‘just for you’ based on saliva testing are appealing,” says Uhl, “but unrealistic.” Hormone levels in saliva do not accurately reflect the amount of hormones a woman has in her body for the purpose of adjusting hormone therapy dose levels. A woman’s hormone levels change throughout the day, and from day to day. FDA-approved tests can tell a woman’s hormone level in a specific body fluid, such as saliva, blood, or urine, at that particular point in time. “These tests are useful to tell if a woman is menopausal or not,” says Uhl, “but they have not been shown to be useful for adjusting hormone therapy dosages.”
Myth: FDA wants all compounded hormone therapies off the market.
Fact: “We are not trying to pull all compounded hormone therapies off the market,” says Silverman. “We believe that, like all traditionally compounded drugs, a woman should be able to get a compounded hormone therapy drug when her physician decides that it will best serve her specific medical needs. But we also want women to be informed and careful about choosing products that have not been proven safe and effective. And pharmacies cannot promote compounded drugs with false or misleading claims.”
In addition, FDA has not approved any drug containing the hormone estriol. Pharmacies should not compound drugs containing estriol unless the prescriber has a valid investigational new drug (IND) application. INDs provide benefits that include allowing physicians to treat individual patients with drugs that are not FDA-approved, while also providing additional safeguards for patients.
Myth: All women who take FDA-approved menopausal hormone therapy drugs are going to get blood clots, heart attacks, strokes, breast cancer, or gall bladder disease.
Fact: Like all medicines, hormone therapy has risks and benefits. For some women, hormone therapy may increase their chances of getting these conditions. However, there are no convincing data that there is less risk of developing a blood clot, heart attack, stroke, breast cancer, or gall bladder disease with a “bioidentical hormone therapy” product. Women should talk to their health care professional about taking hormones. If you decide to use menopausal hormone therapy drugs for menopause
- use at the lowest dose that helps
- use for the shortest time needed
If you are taking a compounded “bioidentical hormone therapy” drug now, talk to your health care professional about treatment options to determine if compounded drugs are the best option for your particular medical needs.
Despite many marketing claims, there is no scientific evidence that custom-compounded bioidentical hormone therapy is safer or more effective than the many government-approved therapies mentioned above. Studies indicate that compounded products are not supervised as closely as FDA-approved hormone products.
- Here is a list of FDA approved prescription hormone products for menopausal symptoms in the US and Canada that meet the above definition of bioidentical hormones: http://www.menopause.org/docs/default-source/professional/nams-ht-tables.pdf?sfvrsn=18.pdf
The concern about the use of bioidentical hormone therapy is really about custom-compounded recipes prepared by a pharmacist following an individual prescriber’s order for a specific patient. Compounders often rely on salivary and blood tests to “assess” your hormone levels to mix their recipes, but these tests are meaningless for midlife women because hormone levels vary from day to day and even from hour to hour.
In addition, these medications do not have FDA approval because individually mixed recipes have not been tested for safety and effectiveness or to prove that the active ingredients are absorbed appropriately or provide predictable levels in blood and tissue. Furthermore, there is no scientific evidence that these compounded medications are safer or more effective than government-approved hormones.
In fact, they may not even contain the prescribed amounts of hormones and that can be dangerous. For example, when the progesterone level is too low, you are not protected against endometrial (uterine) cancer. When estrogen levels are too high, there can be overstimulation of the endometrium and breast tissue, putting you at risk of endometrial cancer and possibly breast cancer. A recent investigative report from More magazine showed that many custom-compounded hormone therapy preparations have too little progesterone and too much estrogen.
Natural hormone replacement therapy
Many approaches have been promoted as aids in managing the symptoms of menopause, but few of them have scientific evidence to back up the claims. Some complementary and alternative treatments that have been or are being studied include:
Plant estrogens (phytoestrogens). These estrogens occur naturally in certain foods. There are two main types of phytoestrogens — isoflavones and lignans. Isoflavones are found in soybeans, lentils, chickpeas and other legumes. Lignans occur in flaxseed, whole grains, and some fruits and vegetables.
Whether the estrogens in these foods can relieve hot flashes and other menopausal symptoms remains to be proved, but most studies have found them ineffective. Isoflavones have some weak estrogen-like effects, so if you’ve had breast cancer, talk to your doctor before supplementing your diet with isoflavone pills.
The herb sage is thought to contain compounds with estrogen-like effects, and there’s good evidence that it can effectively manage menopause symptoms. The herb and its oils should be avoided in people who are allergic, and in pregnant or breast-feeding women. Use carefully in people with high blood pressure or epilepsy.
At this time, it is unknown whether herbs or other “natural” products are helpful or safe. The benefits and risks are still being studied.
Bioidentical hormones. These hormones come from plant sources. The term “bioidentical” implies the hormones in the product are chemically identical to those your body produces. However, though there are some commercially available bioidentical hormones approved by the Food and Drug Administration (FDA), many preparations are compounded — mixed in a pharmacy according to a doctor’s prescription — and aren’t regulated by the FDA, so quality and risks could vary. There’s also no scientific evidence that bioidentical hormones work any better than traditional hormone therapy in easing menopause symptoms.
Black cohosh. Black cohosh (Actaea racemosa, Cimicifuga racemosa) – this herb has received quite a bit of scientific attention for its possible effects on hot flashes. Studies of its effectiveness in reducing hot flashes have produced mixed results. However, some women report that it has helped them. Recent research suggests that black cohosh does not act like estrogen, as once thought. This reduces concerns about its effect on hormone-sensitive tissue (eg, uterus, breast). Black cohosh has had a good safety record over a number of years. But there’s little evidence that black cohosh is effective, and there have been reports linking black cohosh to liver problems and this connection continues to be studied.
Red Clover. Red Clover (Trifolium pratense) In five controlled studies, no consistent or conclusive evidence was found that red clover leaf extract reduces hot flashes. As with black cohosh, however, some women claim that red clover has helped them. Studies report few side effects and no serious health problems with use. But studies in animals have raised concerns that red clover might have harmful effects on hormone-sensitive tissue.
Dong Quai. Dong quai (Angelica sinensis) has been used in Traditional Chinese Medicine to treat gynecologic conditions for more than 1,200 years. Yet only one randomized clinical study of dong quai has been conducted to determine its effects on hot flashes, and this botanical therapy was not found to be useful in reducing them. Some experts on Chinese medicine point out that the preparation studied was not the same as they use in practice. Dong quai should never be used by women with fibroids or blood-clotting problems such as hemophilia, or by women taking drugs that affect clotting such as warfarin (Coumadin) as bleeding complications can result.
Ginseng (Panax ginseng or Panax quinquefolius). Research has shown that ginseng may help with some menopausal symptoms, such as mood symptoms and sleep disturbances, and with one’s overall sense of well-being. However, it has not been found to be helpful for hot flashes.
Kava (Piper methysticum). Kava may decrease anxiety, but there is no evidence that it decreases hot flashes. It is important to note that kava has been associated with liver disease. The FDA has issued a warning to patients and providers about kava because of its potential to damage the liver. Because of this concern, Health Canada does not allow kava to be sold in Canada.
Evening Primrose Oil (Oenothera biennis). This botanical is also promoted to relieve hot flashes. However, the only randomized, placebo-controlled study (in only 56 women) found no benefit over placebo (mock medication). Reported side effects include inflammation, problems with blood clotting and the immune system, nausea, and diarrhea. It has been shown to induce seizures in patients diagnosed with schizophrenia who are taking antipsychotic medication. Evening primrose oil should not be used with anticoagulants or phenothiazines (a type of psychotherapeutic agent).
Yoga. There’s no evidence to support the practice of yoga in reducing menopausal symptoms. But, balance exercises such as yoga or tai chi can improve strength and coordination and may help prevent falls that could lead to broken bones. Check with your doctor before starting balance exercises. Consider taking a class to learn how to perform postures and proper breathing techniques.
Acupuncture. Acupuncture may have some temporary benefit in helping to reduce hot flashes, but in research hasn’t shown significant or consistent improvements. More research is needed.
Hypnosis. Hypnotherapy may decrease the incidence of hot flashes for some menopausal women, according to research from the National Center for Complementary and Integrative Health. Hypnotherapy also helped improve sleep and decreased interference in daily life, according to the study.
You may have heard of or tried other dietary supplements, such as DHEA, evening primrose oil and wild yam (natural progesterone cream). Scientific evidence on effectiveness is lacking, and some of these products may be harmful.
Talk with your doctor before taking any herbal or dietary supplements for menopausal symptoms. The FDA does not regulate herbal products, and some can be dangerous or interact with other medications you take, putting your health at risk.
Hormone replacement therapy for men
Aging is closely associated with a progressive decline in muscle mass 3, strength 4, and aerobic exercise capacity 5, and an increase in body fat. Although these changes could be considered as physiological, they have a detrimental effect and contribute to the incidence of frailty, metabolic disorders, and cardiovascular morbidity and mortality of older men 6. Both muscle power and aerobic capacity are major determinants of physical performance, and this association has been confirmed in several studies.
Thus, the decline of muscle mass and strength, a universal process of aging [for which the term sarcopenia has been coined by Rosenberg 7], has been linked with falls, fractures, and higher mortality rates 8.
The GH (growth hormone)/IGF-I (insulin like growth factor) axis and testosterone levels (especially biologically available testosterone) have all been reported to decline with aging in such a way that older men may be considered partially growth hormone and testosterone deficient 9. Both growth hormone and testosterone are powerful anabolic agents that promote nitrogen retention, increase muscle mass and bone mass, and promote muscle protein synthesis 10. The aging-associated decline in growth hormone and/or testosterone secretion may contribute to the detrimental aspects of aging 11. Replacement therapy with growth hormone and testosterone, respectively, in growth hormone deficiency and hypogonadal adults improves and reverses most of these detrimental changes 12. Thus, it was reasoned that treatment with growth hormone and testosterone may confer clinical benefits in older men, and indeed, Rudman et al. 13 in his pioneering study showed that this could happen, but he studied a highly selected group of subjects, and although pivotal, the applicability of his findings remains controversial 20 yr later.
Aging is associated with several functional changes of the endocrine system. Daily production of growth hormone starts to decrease from the third decade of life by almost 14% for each passing decade, with a marked attenuation of growth hormone secretory pulse amplitude but not frequency 14. Insulin like growth factor-I levels decrease in parallel with the reduction of growth hormone secretion, and 30% of older people could be considered growth hormone deficiency in that their insulin like growth factor-I levels are lower than the lower limit of the young adult normal range 15. In a similar but less dramatic fashion, testosterone levels decline with increasing age, and this has been seen in both cross-sectional and longitudinal studies 16. In the recently updated Baltimore Longitudinal Study, the incidence of hypogonadism, defined as total T levels at or below 11.2 nmol/liter, increased from 20% in those aged 60 to 70 yr to more than 50% in those aged over 80 yr 17.
Aging is also associated with a progressive decrease in exercise capacity that occurs regardless of physical activity and accelerates with each successive decade 18. The mechanism behind this is unclear, but one possible explanation is accumulating oxidative damage because both mitochondria DNA abundance and ATP production have been shown to decline with aging 19. This, in association with the increase in fatigability that occurs with aging 20, may contribute to reduced physical activity commonly observed in older people 21. Restricted physical activity is a hallmark of aging and is closely associated with progression to frailty and disability 22. It is of great importance for two main reasons. First, by reducing energy expenditure and more specifically exercise energy expenditure 23 and without appropriate dietary change, it may facilitate the accumulation of total fat, visceral fat and body fat 24, all being strongly associated with an adverse metabolic profile, insulin resistance, and cardiovascular morbidity and mortality 25. Second, and most importantly, restricted physical activity may further compromise the already impaired muscle adaptation to habitual activity and training observed in older men 26.
Thus, it appears that both exercise capacity and the anabolic hormone profile necessary for muscle tissue integrity are compromised in older men. Exercise improves muscle function and exercise capacity in healthy older men when a resistance-training program of high intensity and sufficient duration is undertaken 27. The hypertrophic response of muscle to training in older men is blunted when compared with younger counterparts, and this has been attributed (at least in part) to the deficient anabolic hormone profile and locally expressed milieu 28. Recent evidence suggests that in healthy young men, muscle protein synthesis starts to increase in response to intensities of resistance exercise as low as 20% of 1 repetition maximum (1RM). This underlines the importance of exercise and leisure time physical activity in maintaining normal muscle tissue homeostasis 29.
Growth hormone, the most abundant pituitary hormone, is a single chain polypeptide of 191 amino acids, which is secreted in a pulsatile fashion by the somatotropic cells in the anterior pituitary gland and whose secretion is directly controlled by hypothalamic and peripheral factors acting on the somatotrophs 30. Three hypothalamic peptides in a fine coordinated interplay regulate pulsatile growth hormone secretion: hypothalamic GHRH 31, which stimulates growth hormone secretion; somatostatin, which inhibits growth hormone secretion; and ghrelin, recently discovered as the endogenous ligand of previously identified growth hormone secretagogue receptor and suggested to be a powerful regulator of growth hormone secretion in humans 32. The growth hormone secretagogue receptor is distinct from the growth hormone–releasing hormone( GHRH) receptor 32. Ghrelin is secreted by the stomach but is also expressed in many other tissues, including the pituitary, and was suggested to be a powerful regulator of growth hormone secretion in men in experimental settings 33. It was proposed that ghrelin facilitates a periodic secretory burst of growth hormone by inhibiting nocturnal somatostatin, but so far the exact role under physiological conditions has not been established 34. Thus, Avram et al. 34 have examined ghrelin secretory dynamics over 48 h in the fed and fasted state using frequent (every 10 min) sampling and found no change in ghrelin levels despite a clear secretory burst of growth hormone, whereas Nass et al. 35, using a similar technique, concluded differently in that they found evidence that ghrelin amplified the growth hormone pulses.
Growth hormone has direct effects, but many of its actions are mediated through circulating and locally expressed IGF-I 36. Circulating IGF-I is largely (∼70%) derived from the liver in response to pituitary growth hormone, whereas IGF-I in turn appears to play an active role in regulating growth hormone secretion through a negative feedback mechanism because infusion of IGF-I rapidly suppresses growth hormone pulsatile secretion in humans 37.
Growth hormone secretion and aging
Integrated daily GH (IDGH) secretion and IGF-I production decline progressively during adult life 38. Consequently, more than 30% of older men have IGF-I levels lower than the young adult reference range 39. GH is secreted almost exclusively through the 10 to 20 daily recorded secretory bursts, with the highest pulses occurring during the period of deep sleep, so that more than 70% of daily GH is secreted during the night 30.
Aging is associated with a significant alteration of GH secretion patterns so that most of the GH is secreted during the day instead and is associated with a steep decline of integrated daily growth hormone (IDGH) secretion 40. Accordingly, it has been demonstrated that for each decade of increasing age, integrated daily growth hormone (IDGH) secretion falls by 14%, and in a 70-yr-old man, on average GH secretion has declined by more than 70% 41.
Physiology of aging in the male gonadal axis
GnRH (gonadotropin-releasing hormone) is secreted into the hypophyseal portal system in a pulsatile fashion, which in turn elicits pulsatile secretion of LH (Luteinizing hormone) and FSH (Follicle-stimulating hormone) by the gonadotrophs of the anterior pituitary. The pulsatile release of GnRH is essential for the pulsatile secretion of LH and FSH because continuous administration of GnRH inhibits gonadotropin release 42. LH in turn interacts with cell membrane receptors on Leydig cells in the testis to stimulate, via a series of intermediate steps, testosterone synthesis 43. Testosterone then directly or indirectly, after conversion to estrogens, exerts a negative feedback at the level of both the pituitary and hypothalamus and thus modulates the pulse generator of GnRH and gonadotroph secretion 44.
Testosterone in plasma is bound strongly to SHBG [Sex hormone-binding globulin] (60%) and to a lesser degree loosely to albumin, and only 1 to 2% of Testosterone circulates freely 45. Of note, SHBG-bound testosterone is not biologically active. SHBG levels increase with age, thus resulting in lower levels of bioavailable testosterone (BioTestosterone) 46. Aging is associated with a progressive decline of daily testosterone secretion rates and thus reduced plasma testosterone levels. Both primary and secondary hypogonadism have been suggested as possible causes for the decline of testosterone secretion observed in older men 47. Indeed, the reduced responsiveness of the testis to stimulation by human chorionic gonadotropin, clomiphene, or more recently to pulsatile LH drive, proved a reduced capacity of older men to increase testosterone concentration when compared with young men 48. In a recent study, a GnRH antagonist (ganirelix) was administered to block endogenous LH. Older men then had a reduced capacity to stimulate testosterone secretion after pulsatile exogenous LH compared with young men 49, which in turn denotes reduced Leydig cell secretory capacity. On the other hand, it has been shown repeatedly that the pituitary of older men responds to acute or even prolonged (up to 14 d) pulsatile stimulation by GnRH 50. Thus, Mulligan et al. 47, using discrete pulse detection algorithms to analyze the LH concentration series and mathematical deconvolution analysis of the LH pulses, demonstrated that 14 d of pulsatile GnRH administration restored normal pituitary 24-h LH release with normal pulsatile pattern in older men. The authors concluded, in view of their lower T levels, that a combined defect of GnRH release and Leydig cell responsiveness could underlie the lower testosterone levels in older men.
LH pulse amplitude was reported to decline with age and to be the main determinant of lower testosterone concentration commonly seen in aging 51. A series of elaborate studies from the same group revealed the attenuated capacity of hypothalamic GnRH release mechanism, which results in low-amplitude, high-frequency pulses and consequent decreased T levels. This was achieved by selectively blocking the negative feedback on the hypothalamus (either with an aromatase inhibitor or by the administration of ketoconazole) and quantifying the LH response with deconvolution analysis 52.
This review 53 showed that both testosterone and growth hormone are powerful anabolic agents. The authors believe that these two hormones are the most important agents that, in combination with exercise, normally regulate body composition in adult men. The evidence shows that the anabolic effects of testosterone and growth hormone are dose- and time-dependent. In studies of hormone replacement, there is clear evidence that testosterone delivered into the systemic circulation via injections, or more recently by gels and patches, is more effective than oral preparations. Hormone replacement with testosterone may also be safer when given systemically. The data also show that testosterone combined with GH is a more effective anabolic treatment regime than either alone. By combining GH and testosterone, a given anabolic effect is achieved with a smaller dose of each compared with when GH and testosterone are given alone. There may also be an extra effect that may not be achievable with either alone. This also has considerable implications in terms of avoiding side effects and achieving long-term safety.
The evidence reviewed indicates unequivocally that it is possible to influence at least some components of this aging process favorably by hormone replacement with GH and testosterone. The favorable effects are seen most easily in terms of changes in body composition than in physical function.
The published data also indicate that the therapeutic interventions with replacement hormone replacement therapy with testosterone and growth hormone in healthy older men is safe, at least over an interval of 3 yr (testosterone) and 6 months (growth hormone and testosterone), providing that moderate doses are used. Safety beyond that time scale will require longer studies than are available today. Although incomplete, the evidence available is generally positive and should encourage rather than discourage future clinical research.
Growth hormone Side Effects
The very long-term effects of GH administration in healthy older men are currently unknown. Some epidemiological studies have shown an association between serum IGF-I level and the occurrence of prostate and breast cancer in the normal population.
On one hand, in a nested case-control study from the Physician’s Health Study, a positive association was observed between a single serum IGF-I level and the risk of prostate cancer development after 5 or more years 54. A similar analysis of the Nurses’ Health Study showed that IGF-I levels could predict breast cancer in premenopausal but not postmenopausal women 55. Consequent to the coverage of these results in the lay press, there has been concern that GH therapy and its attendant increase in IGF-I could lead to the development of malignancies. This is a statistical association only and as such does not provide any evidence of causality. Because the direct evidence does not indicate GH to be carcinogenic, this statistical link could be what is recognized in the statistical world as a “spurious relationship” or spurious correlation 56.
Older men are more susceptible to GH-related adverse effects, and earlier studies that have administered GH in doses comparable to young growth hormone deficiency adults have reported a high incidence of adverse events. Adverse effects appeared to occur early during the study period and were similar to those observed in growth hormone deficiency patients, with fluid retention (varying degrees of pitting leg edema and carpal tunnel syndrome) and arthralgia involving small hand joints being most prevalent. Although most of the symptoms reported were mild and subsided or even disappeared after GH dose reduction, it becomes apparent that the incidence of adverse effects is still somewhat higher than that reported in young growth hormone deficiency patients. The symptoms recorded were largely those predictably attributable to GH action (effects), such as those consequential to sodium retention (ankle edema and carpal tunnel) and arthralgias (growing pains), rather than unexpected side effects. These usually subside spontaneously over 1 or 2 wk or in response to a dose reduction and are in reality indicators of overdosage rather than side effects. In modern regimes of GH treatment developed through experience, the starting dose is always low, and the dose is slowly escalated based on the subject’s well-being and the measured IGF-I responses. Individual people differ substantially in their sensitivity to GH (just as they do to insulin), and fixed doses based on body weight (originating from pediatric experience) are now outdated.
Growth hormone therapy could be a cause of insulin resistance, as has been shown in studies with growth hormone deficiency adults 57 and has been recorded in older men 58. This is an unwanted and possibly avoidable adverse event 59 because older men are already at higher risk for cardiovascular disease 60. It seems that insulin sensitivity may improve after 6 to 12 months of GH treatment, and 5 or 7 yr of GH therapy do not adversely affect insulin sensitivity 61. It has been suggested that the initial deterioration in insulin sensitivity is due to increased free fatty acid oxidation because of GH-induced lipolysis, which adversely affects glucose disposal in muscle 62. An inverse relationship between circulating free fatty acid concentrations and insulin sensitivity in growth hormone deficiency adults has been confirmed in several studies using acipimox, a blocker of free fatty acid release 63.
Another rare complication reported after GH treatment is gynecomastia or nipple tenderness, mainly in older men, which was reported in some studies 64, 58, 59 but not in others 65, 66.
Testosterone Side Effects
One systematic review recently reported the adverse effects of testosterone treatment in 51 studies where testosterone was administered to men with a wide range of conditions and low or low-normal testosterone levels 67. Testosterone treatment was associated with a significant increase in hemoglobin and hematocrit and a decrease in HDL-“good” cholesterol levels, but no treatment effect was reported on prostate specific antigen (PSA) levels, prostate cancer, composite prostate outcome, cardiovascular events, or overall mortality. Interestingly, testosterone does not appear to affect adversely the lipid profile in healthy older men.
Another meta-analysis of 19 studies of middle-aged and older men reported that the testosterone-treated men were four times more likely to have a hematocrit higher than 50% and a higher combined rate of all prostate events when compared with placebo. The individual rates of prostate cancer, increments in PSA levels, and prostate biopsy events did not differ, however, when compared with placebo 68. Testosterone has been known to stimulate erythropoiesis, possibly by stimulating erythropoietin production 69, whereas suppression by testosterone of serum hepcidin (an iron regulatory peptide) may also contribute to this 70. Polycythemia has been reported to occur in healthy older men mainly after intramuscular and oral testosterone administration 71, but usually not after transdermal testosterone 72. Nevertheless, it is important to monitor the hematocrit at regular intervals to avoid this potentially serious adverse event.
Pharmacological doses of testosterone may induce or worsen sleep apnea in healthy older men 73; this appears to be an uncommon side effect. Snyder et al. 74 could not detect any change in the respiratory distress index after 36 months of transdermal testosterone.
Finally, an increase in blood pressure and clinically significant edema may seldom occur after testosterone administration in healthy older men 75, but these are potentially serious adverse events that appear to occur most frequently in older patients with preexisting cardiovascular and pulmonary diseases. Indeed, it was necessary to interrupt a recent study where testosterone was prescribed in frail older men because of a high incidence of serious cardiovascular and pulmonary adverse events.
In conclusion, testosterone treatment in healthy older men in near physiological doses does not appear to incur serious adverse events, although long-term safety has not been established, and regular monitoring of PSA and hematocrit levels is required.
- Hormone Therapy: Benefits & Risks. http://www.menopause.org/for-women/menopauseflashes/menopause-symptoms-and-treatments/hormone-therapy-benefits-risks[↩]
- Bio-Identicals: Sorting Myths from Facts. U.S. Food and Drug Administration. https://www.fda.gov/forconsumers/consumerupdates/ucm049311.htm[↩]
- Cress ME , Meyer M. 2003. Maximal voluntary and functional performance levels needed for independence in adults aged 65 to 97 years. Phys Ther 83:37–48. https://www.ncbi.nlm.nih.gov/pubmed/12495411[↩]
- Goodpaster BH , Park SW , Harris TB , Kritchevsky SB , Nevitt M , Schwartz AV , Simonsick EM , Tylavsky FA , Visser M , Newman AB. 2006. The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J Gerontol A Biol Sci Med Sci 61:1059–1064. https://www.ncbi.nlm.nih.gov/pubmed/17077199[↩]
- Fleg JL , Morrell CH , Bos AG , Brant LJ , Talbot LA , Wright JG , Lakatta EG. 2005. Accelerated longitudinal decline of aerobic capacity in healthy older adults. Circulation 112:674–682. http://circ.ahajournals.org/content/112/5/674.long[↩]
- Calle EE , Thun MJ , Petrelli JM , Rodriguez C , Heath CW. 1999. Body-mass index and mortality in a prospective cohort of U.S. adults. N Engl J Med 341:1097–1105. http://www.nejm.org/doi/full/10.1056/NEJM199910073411501[↩]
- Rosenberg IH. 1989. Summary comments. Am J Clin Nutr 50:1231–1233[↩]
- Skeletal muscle strength as a predictor of all-cause mortality in healthy men. Metter EJ, Talbot LA, Schrager M, Conwit R. J Gerontol A Biol Sci Med Sci. 2002 Oct; 57(10):B359-65. https://www.ncbi.nlm.nih.gov/pubmed/12242311[↩]
- Harman SM , Metter EJ , Tobin JD , Pearson J , Blackman MR. 2001. Longitudinal effects of aging on serum total and free testosterone levels in healthy men. Baltimore Longitudinal Study of Aging. J Clin Endocrinol Metab 86:724–731. https://www.ncbi.nlm.nih.gov/pubmed/11158037[↩]
- Bhasin S , Storer TW , Berman N , Callegari C , Clevenger B , Phillips J , Bunnell TJ , Tricker R , Shirazi A , Casaburi R. 1996. The effects of supraphysiologic doses of testosterone on muscle size and strength in normal men. N Engl J Med 335:1–7. http://www.nejm.org/doi/full/10.1056/NEJM199607043350101[↩]
- Snyder PJ , Peachey H , Hannoush P , Berlin JA , Loh L , Lenrow DA , Holmes JH , Dlewati A , Santanna J , Rosen CJ , Strom BL. 1999. Effect of testosterone treatment on body composition and muscle strength in men over 65 years of age. J Clin Endocrinol Metab 84:2647–2653. https://www.ncbi.nlm.nih.gov/pubmed/10443654[↩]
- Snyder PJ , Peachey H , Berlin JA , Hannoush P , Haddad G , Dlewati A , Santanna J , Loh L , Lenrow DA , Holmes JH , Kapoor SC , Atkinson LE , Strom BL. 2000. Effects of testosterone replacement in hypogonadal men. J Clin Endocrinol Metab 85:2670–2677. https://www.ncbi.nlm.nih.gov/pubmed/10946864[↩]
- Rudman D , Feller AG , Nagraj HS , Gergans GA , Lalitha PY , Goldberg AF , Schlenker RA , Cohn L , Rudman IW , Mattson DE. 1990. Effects of human growth hormone in men over 60 years old. N Engl J Med 323:1–6. http://www.nejm.org/doi/full/10.1056/NEJM199007053230101[↩]
- Differential impact of age, sex steroid hormones, and obesity on basal versus pulsatile growth hormone secretion in men as assessed in an ultrasensitive chemiluminescence assay. Veldhuis JD, Liem AY, South S, Weltman A, Weltman J, Clemmons DA, Abbott R, Mulligan T, Johnson ML, Pincus S. J Clin Endocrinol Metab. 1995 Nov; 80(11):3209-22. https://www.ncbi.nlm.nih.gov/pubmed/7593428/[↩]
- Age-related changes of serum sex hormones, insulin-like growth factor-1 and sex-hormone binding globulin levels in men: cross-sectional data from a healthy male cohort. Leifke E, Gorenoi V, Wichers C, Von Zur Mühlen A, Von Büren E, Brabant G. Clin Endocrinol (Oxf). 2000 Dec; 53(6):689-95. https://www.ncbi.nlm.nih.gov/pubmed/11155090/[↩]
- The decline of serum testosterone levels in community-dwelling men over 70 years of age: descriptive data and predictors of longitudinal changes. Lapauw B, Goemaere S, Zmierczak H, Van Pottelbergh I, Mahmoud A, Taes Y, De Bacquer D, Vansteelandt S, Kaufman JM. Eur J Endocrinol. 2008 Oct; 159(4):459-68. https://www.ncbi.nlm.nih.gov/pubmed/18593825/[↩]
- Longitudinal effects of aging on serum total and free testosterone levels in healthy men. Baltimore Longitudinal Study of Aging. Harman SM, Metter EJ, Tobin JD, Pearson J, Blackman MR, Baltimore Longitudinal Study of Aging. J Clin Endocrinol Metab. 2001 Feb; 86(2):724-31. https://www.ncbi.nlm.nih.gov/pubmed/11158037/[↩]
- Accelerated longitudinal decline of aerobic capacity in healthy older adults. Fleg JL, Morrell CH, Bos AG, Brant LJ, Talbot LA, Wright JG, Lakatta EG. Circulation. 2005 Aug 2; 112(5):674-82. http://circ.ahajournals.org/content/112/5/674.long[↩]
- Decline in skeletal muscle mitochondrial function with aging in humans. Short KR, Bigelow ML, Kahl J, Singh R, Coenen-Schimke J, Raghavakaimal S, Nair KS. Proc Natl Acad Sci U S A. 2005 Apr 12; 102(15):5618-23. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC556267/[↩]
- Crespo CJ , Keteyian SJ , Heath GW , Sempos CT. 1996. Leisure-time physical activity among US adults. Results from the Third National Health and Nutrition Examination Survey. Arch Intern Med 156:93–98. https://www.ncbi.nlm.nih.gov/pubmed/8526703[↩]
- [↩]
- Gill TM , Allore H , Guo Z. 2003. Restricted activity and functional decline among community-living older persons. Arch Intern Med 163:1317–1322. https://www.ncbi.nlm.nih.gov/pubmed/12796067[↩]
- Frisard MI, Fabre JM, Russell RD, et al. Physical Activity Level and Physical Functionality in Nonagenarians Compared to Individuals Aged 60–74 Years. The journals of gerontology Series A, Biological sciences and medical sciences. 2007;62(7):783-788. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2724866[↩]
- Kehayias JJ , Fiatarone MA , Zhuang H , Roubenoff R. 1997. Total body potassium and body fat: relevance to aging. Am J Clin Nutr 66:904–910. https://www.ncbi.nlm.nih.gov/pubmed/9322566[↩]
- Vega GL , Adams-Huet B , Peshock R , Willett D , Shah B , Grundy SM. 2006. Influence of body fat content and distribution on variation in metabolic risk. J Clin Endocrinol Metab 91:4459–4466. https://www.ncbi.nlm.nih.gov/pubmed/16926254[↩]
- Petrella JK , Kim JS , Cross JM , Kosek DJ , Bamman MM. 2006. Efficacy of myonuclear addition may explain differential myofiber growth among resistance-trained young and older men and women. Am J Physiol Endocrinol Metab 291:E937–E946. http://ajpendo.physiology.org/content/291/5/E937.long[↩]
- Exercise training and nutritional supplementation for physical frailty in very elderly people. Fiatarone MA, O’Neill EF, Ryan ND, Clements KM, Solares GR, Nelson ME, Roberts SB, Kehayias JJ, Lipsitz LA, Evans WJ. N Engl J Med. 1994 Jun 23; 330(25):1769-75. http://www.nejm.org/doi/full/10.1056/NEJM199406233302501[↩]
- Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Conboy IM, Conboy MJ, Wagers AJ, Girma ER, Weissman IL, Rando TA. Nature. 2005 Feb 17; 433(7027):760-4. https://www.ncbi.nlm.nih.gov/pubmed/15716955/[↩]
- Age-related differences in the dose-response relationship of muscle protein synthesis to resistance exercise in young and old men. Kumar V, Selby A, Rankin D, Patel R, Atherton P, Hildebrandt W, Williams J, Smith K, Seynnes O, Hiscock N, Rennie MJ. J Physiol. 2009 Jan 15; 587(1):211-7. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2670034/[↩]
- Pathophysiology of the neuroregulation of growth hormone secretion in experimental animals and the human. Giustina A, Veldhuis JD. Endocr Rev. 1998 Dec; 19(6):717-97. https://www.ncbi.nlm.nih.gov/pubmed/9861545/[↩][↩]
- growth hormone–releasing hormone[↩]
- Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K. Nature. 1999 Dec 9; 402(6762):656-60. https://www.ncbi.nlm.nih.gov/pubmed/10604470/[↩][↩]
- Ghrelin potentiates growth hormone secretion driven by putative somatostatin withdrawal and resists inhibition by human corticotropin-releasing hormone. Veldhuis JD, Iranmanesh A, Mielke K, Miles JM, Carpenter PC, Bowers CY. J Clin Endocrinol Metab. 2006 Jun; 91(6):2441-6. https://www.ncbi.nlm.nih.gov/pubmed/16537682/[↩]
- Endogenous circulating ghrelin does not mediate growth hormone rhythmicity or response to fasting. Avram AM, Jaffe CA, Symons KV, Barkan AL. J Clin Endocrinol Metab. 2005 May; 90(5):2982-7. https://www.ncbi.nlm.nih.gov/pubmed/15713719/[↩][↩]
- Evidence for acyl-ghrelin modulation of growth hormone release in the fed state. Nass R, Farhy LS, Liu J, Prudom CE, Johnson ML, Veldhuis P, Pezzoli SS, Oliveri MC, Gaylinn BD, Geysen HM, Thorner MO. J Clin Endocrinol Metab. 2008 May; 93(5):1988-94. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2386275/[↩]
- The somatomedin hypothesis 2007: 50 years later. Kaplan SA, Cohen P. J Clin Endocrinol Metab. 2007 Dec; 92(12):4529-35. https://www.ncbi.nlm.nih.gov/pubmed/17986643/[↩]
- A low dose euglycemic infusion of recombinant human insulin-like growth factor I rapidly suppresses fasting-enhanced pulsatile growth hormone secretion in humans. Hartman ML, Clayton PE, Johnson ML, Celniker A, Perlman AJ, Alberti KG, Thorner MO. J Clin Invest. 1993 Jun; 91(6):2453-62. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC443305/[↩]
- Abdominal adiposity and physical fitness are major determinants of the age associated decline in stimulated GH secretion in healthy adults. Vahl N, Jørgensen JO, Jurik AG, Christiansen JS. J Clin Endocrinol Metab. 1996 Jun; 81(6):2209-15. https://www.ncbi.nlm.nih.gov/pubmed/8964853[↩]
- Serum free IGF-I, total IGF-I, IGFBP-1 and IGFBP-3 levels in an elderly population: relation to age and sex steroid levels. Janssen JA, Stolk RP, Pols HA, Grobbee DE, de Jong FH, Lamberts SW. Clin Endocrinol (Oxf). 1998 Apr; 48(4):471-8. https://www.ncbi.nlm.nih.gov/pubmed/9640414/[↩]
- Effects of sex and age on the 24-hour profile of growth hormone secretion in man: importance of endogenous estradiol concentrations. Ho KY, Evans WS, Blizzard RM, Veldhuis JD, Merriam GR, Samojlik E, Furlanetto R, Rogol AD, Kaiser DL, Thorner MO. J Clin Endocrinol Metab. 1987 Jan; 64(1):51-8. https://www.ncbi.nlm.nih.gov/pubmed/3782436/[↩]
- Age and relative adiposity are specific negative determinants of the frequency and amplitude of growth hormone (GH) secretory bursts and the half-life of endogenous GH in healthy men. Iranmanesh A, Lizarralde G, Veldhuis JD. J Clin Endocrinol Metab. 1991 Nov; 73(5):1081-8. https://www.ncbi.nlm.nih.gov/pubmed/1939523/[↩]
- Hypophysial responses to continuous and intermittent delivery of hypopthalamic gonadotropin-releasing hormone. Belchetz PE, Plant TM, Nakai Y, Keogh EJ, Knobil E. Science. 1978 Nov 10; 202(4368):631-3. https://www.ncbi.nlm.nih.gov/pubmed/100883/[↩]
- The molecular basis of gonadotropin-releasing hormone action. Conn PM. Endocr Rev. 1986 Feb; 7(1):3-10. https://www.ncbi.nlm.nih.gov/pubmed/3007080/[↩]
- Aromatization mediates testosterone’s short-term feedback restraint of 24-hour endogenously driven and acute exogenous gonadotropin-releasing hormone-stimulated luteinizing hormone and follicle-stimulating hormone secretion in young men. Schnorr JA, Bray MJ, Veldhuis JD. J Clin Endocrinol Metab. 2001 Jun; 86(6):2600-6. https://www.ncbi.nlm.nih.gov/pubmed/11397860/[↩]
- Studies on the binding of testosterone to human plasma. Vermeulen A, Verdonck L. Steroids. 1968 May; 11(5):609-35. https://www.ncbi.nlm.nih.gov/pubmed/4172115/[↩]
- Age-related changes in serum testosterone and sex hormone binding globulin in Australian men: longitudinal analyses of two geographically separate regional cohorts. Liu PY, Beilin J, Meier C, Nguyen TV, Center JR, Leedman PJ, Seibel MJ, Eisman JA, Handelsman DJ. J Clin Endocrinol Metab. 2007 Sep; 92(9):3599-603. https://www.ncbi.nlm.nih.gov/pubmed/17595245/[↩]
- Two-week pulsatile gonadotropin releasing hormone infusion unmasks dual (hypothalamic and Leydig cell) defects in the healthy aging male gonadotropic axis. Mulligan T, Iranmanesh A, Kerzner R, Demers LW, Veldhuis JD. Eur J Endocrinol. 1999 Sep; 141(3):257-66. https://www.ncbi.nlm.nih.gov/pubmed/10474123/[↩][↩]
- Reproductive hormones in aging men. I. Measurement of sex steroids, basal luteinizing hormone, and Leydig cell response to human chorionic gonadotropin. Harman SM, Tsitouras PD. J Clin Endocrinol Metab. 1980 Jul; 51(1):35-40. https://www.ncbi.nlm.nih.gov/pubmed/7189758/[↩]
- Age attenuates testosterone secretion driven by amplitude-varying pulses of recombinant human luteinizing hormone during acute gonadotrope inhibition in healthy men. Takahashi PY, Votruba P, Abu-Rub M, Mielke K, Veldhuis JD. J Clin Endocrinol Metab. 2007 Sep; 92(9):3626-32. https://www.ncbi.nlm.nih.gov/pubmed/17579202/[↩]
- Influence of age on the responsiveness of the gonadotrophs to luteinizing hormone-releasing hormone in males. Kaufman JM, Giri M, Deslypere JM, Thomas G, Vermeulen A. J Clin Endocrinol Metab. 1991 Jun; 72(6):1255-60. https://www.ncbi.nlm.nih.gov/pubmed/2026746/[↩]
- Attenuation of luteinizing hormone secretory burst amplitude as a proximate basis for the hypoandrogenism of healthy aging in men. Veldhuis JD, Urban RJ, Lizarralde G, Johnson ML, Iranmanesh A. J Clin Endocrinol Metab. 1992 Sep; 75(3):707-13. https://www.ncbi.nlm.nih.gov/pubmed/1517359/[↩]
- Short-term aromatase-enzyme blockade unmasks impaired feedback adaptations in luteinizing hormone and testosterone secretion in older men. Veldhuis JD, Iranmanesh A. J Clin Endocrinol Metab. 2005 Jan; 90(1):211-8. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1315304/[↩]
- Giannoulis MG, Martin FC, Nair KS, Umpleby AM, Sonksen P. Hormone Replacement Therapy and Physical Function in Healthy Older Men. Time to Talk Hormones? Endocrine Reviews. 2012;33(3):314-377. doi:10.1210/er.2012-1002. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5393154/[↩]
- Plasma insulin-like growth factor-I and prostate cancer risk: a prospective study. Chan JM, Stampfer MJ, Giovannucci E, Gann PH, Ma J, Wilkinson P, Hennekens CH, Pollak M. Science. 1998 Jan 23; 279(5350):563-6. https://www.ncbi.nlm.nih.gov/pubmed/9438850/[↩]
- Circulating concentrations of insulin-like growth factor-I and risk of breast cancer. Hankinson SE, Willett WC, Colditz GA, Hunter DJ, Michaud DS, Deroo B, Rosner B, Speizer FE, Pollak M. Lancet. 1998 May 9; 351(9113):1393-6. https://www.ncbi.nlm.nih.gov/pubmed/9593409/[↩]
- Simon HA. 1954. Spurious correlation: A causal interpretation. Journal of the American Statistical Association 49:467–479[↩]
- Defects of insulin action and skeletal muscle glucose metabolism in growth hormone-deficient adults persist after 24 months of recombinant human growth hormone therapy. Christopher M, Hew FL, Oakley M, Rantzau C, Alford F. J Clin Endocrinol Metab. 1998 May; 83(5):1668-81. https://www.ncbi.nlm.nih.gov/pubmed/9589675/[↩]
- Carpal tunnel syndrome and gynaecomastia during growth hormone treatment of elderly men with low circulating IGF-I concentrations. Cohn L, Feller AG, Draper MW, Rudman IW, Rudman D. Clin Endocrinol (Oxf). 1993 Oct; 39(4):417-25. https://www.ncbi.nlm.nih.gov/pubmed/8287568/[↩][↩]
- Testosterone and growth hormone improve body composition and muscle performance in older men. Sattler FR, Castaneda-Sceppa C, Binder EF, Schroeder ET, Wang Y, Bhasin S, Kawakubo M, Stewart Y, Yarasheski KE, Ulloor J, Colletti P, Roubenoff R, Azen SP. J Clin Endocrinol Metab. 2009 Jun; 94(6):1991-2001. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2690426/[↩][↩]
- Banting lecture 1988. Role of insulin resistance in human disease. Reaven GM. Diabetes. 1988 Dec; 37(12):1595-607. https://www.ncbi.nlm.nih.gov/pubmed/3056758/[↩]
- Jorgensen JO , Vahl N , Nyholm B , Juul A , Moller N , Schmitz O , Skakkebaek NE , Christ , Christiansen JS. 1996. Substrate metabolism and insulin sensitivity following long-term growth hormone (GH) replacement therapy in GH-deficient adults. Endocrinol Metab 3:281–286[↩]
- The glucose fatty-acid cycle. Its role in insulin sensitivity and the metabolic disturbances of diabetes mellitus. RANDLE PJ, GARLAND PB, HALES CN, NEWSHOLME EA. Lancet. 1963 Apr 13; 1(7285):785-9. https://www.ncbi.nlm.nih.gov/pubmed/13990765[↩]
- Growth hormone replacement therapy and insulin sensitivity. Svensson J, Bengtsson BA. J Clin Endocrinol Metab. 2003 Apr; 88(4):1453-4. https://www.ncbi.nlm.nih.gov/pubmed/12679421/[↩]
- Growth hormone replacement in healthy older men improves body composition but not functional ability. Papadakis MA, Grady D, Black D, Tierney MJ, Gooding GA, Schambelan M, Grunfeld C. Ann Intern Med. 1996 Apr 15; 124(8):708-16. https://www.ncbi.nlm.nih.gov/pubmed/8633830/[↩]
- The effects of growth hormone and/or testosterone in healthy elderly men: a randomized controlled trial. Giannoulis MG, Sonksen PH, Umpleby M, Breen L, Pentecost C, Whyte M, McMillan CV, Bradley C, Martin FC. J Clin Endocrinol Metab. 2006 Feb; 91(2):477-84. https://www.ncbi.nlm.nih.gov/pubmed/16332938/[↩]
- Growth hormone and sex steroid administration in healthy aged women and men: a randomized controlled trial. Blackman MR, Sorkin JD, Münzer T, Bellantoni MF, Busby-Whitehead J, Stevens TE, Jayme J, O’Connor KG, Christmas C, Tobin JD, Stewart KJ, Cottrell E, St Clair C, Pabst KM, Harman SM. JAMA. 2002 Nov 13; 288(18):2282-92. https://www.ncbi.nlm.nih.gov/pubmed/12425705/[↩]
- Clinical review 1: Adverse effects of testosterone therapy in adult men: a systematic review and meta-analysis. Fernández-Balsells MM, Murad MH, Lane M, Lampropulos JF, Albuquerque F, Mullan RJ, Agrwal N, Elamin MB, Gallegos-Orozco JF, Wang AT, Erwin PJ, Bhasin S, Montori VM. J Clin Endocrinol Metab. 2010 Jun; 95(6):2560-75. https://www.ncbi.nlm.nih.gov/pubmed/20525906/[↩]
- Adverse events associated with testosterone replacement in middle-aged and older men: a meta-analysis of randomized, placebo-controlled trials. Calof OM, Singh AB, Lee ML, Kenny AM, Urban RJ, Tenover JL, Bhasin S. J Gerontol A Biol Sci Med Sci. 2005 Nov; 60(11):1451-7. https://www.ncbi.nlm.nih.gov/pubmed/16339333/[↩]
- Effects of graded doses of testosterone on erythropoiesis in healthy young and older men. Coviello AD, Kaplan B, Lakshman KM, Chen T, Singh AB, Bhasin S. J Clin Endocrinol Metab. 2008 Mar; 93(3):914-9. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2266950/[↩]
- Testosterone suppresses hepcidin in men: a potential mechanism for testosterone-induced erythrocytosis. Bachman E, Feng R, Travison T, Li M, Olbina G, Ostland V, Ulloor J, Zhang A, Basaria S, Ganz T, Westerman M, Bhasin S. J Clin Endocrinol Metab. 2010 Oct; 95(10):4743-7. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3050108/[↩]
- Difficulties in measuring the effect of testosterone replacement therapy on muscle function in older men. Clague JE, Wu FC, Horan MA. Int J Androl. 1999 Aug; 22(4):261-5. https://www.ncbi.nlm.nih.gov/pubmed/10442299/[↩]
- Effects of transdermal testosterone on bone and muscle in older men with low bioavailable testosterone levels. Kenny AM, Prestwood KM, Gruman CA, Marcello KM, Raisz LG. J Gerontol A Biol Sci Med Sci. 2001 May; 56(5):M266-72. https://www.ncbi.nlm.nih.gov/pubmed/11320105/[↩]
- The short-term effects of high-dose testosterone on sleep, breathing, and function in older men. Liu PY, Yee B, Wishart SM, Jimenez M, Jung DG, Grunstein RR, Handelsman DJ. J Clin Endocrinol Metab. 2003 Aug; 88(8):3605-13. https://www.ncbi.nlm.nih.gov/pubmed/12915643/[↩]
- Effect of testosterone treatment on bone mineral density in men over 65 years of age. Snyder PJ, Peachey H, Hannoush P, Berlin JA, Loh L, Holmes JH, Dlewati A, Staley J, Santanna J, Kapoor SC, Attie MF, Haddad JG Jr, Strom BL. J Clin Endocrinol Metab. 1999 Jun; 84(6):1966-72. https://www.ncbi.nlm.nih.gov/pubmed/10372695/[↩]
- Risks of testosterone-replacement therapy and recommendations for monitoring. Rhoden EL, Morgentaler A. N Engl J Med. 2004 Jan 29; 350(5):482-92. https://www.ncbi.nlm.nih.gov/pubmed/14749457/[↩]