Contents
What is hypopituitarism
Hypopituitarism (also called pituitary insufficiency) is a rare condition in which your pituitary gland either fails to produce one or more of its hormones or doesn’t produce enough of them. Your body can’t work properly when important glands, such as your thyroid gland and adrenal gland, don’t get the hormones they need from your pituitary gland. The pituitary gland controls a system of hormones in your body that regulate growth, metabolism, the stress response, and functions of the sex organs via the thyroid gland, adrenal gland, ovaries, and testes. Hypopituitarism can develop very slowly, over several months or even over several years.
In hypopituitarism, you have a short supply of one or more of these pituitary hormones. This deficiency can affect any number of your body’s routine functions, such as growth, blood pressure and reproduction.
You’ll likely need medications for the rest of your life to treat hypopituitarism, but your symptoms can be controlled.
The pituitary gland
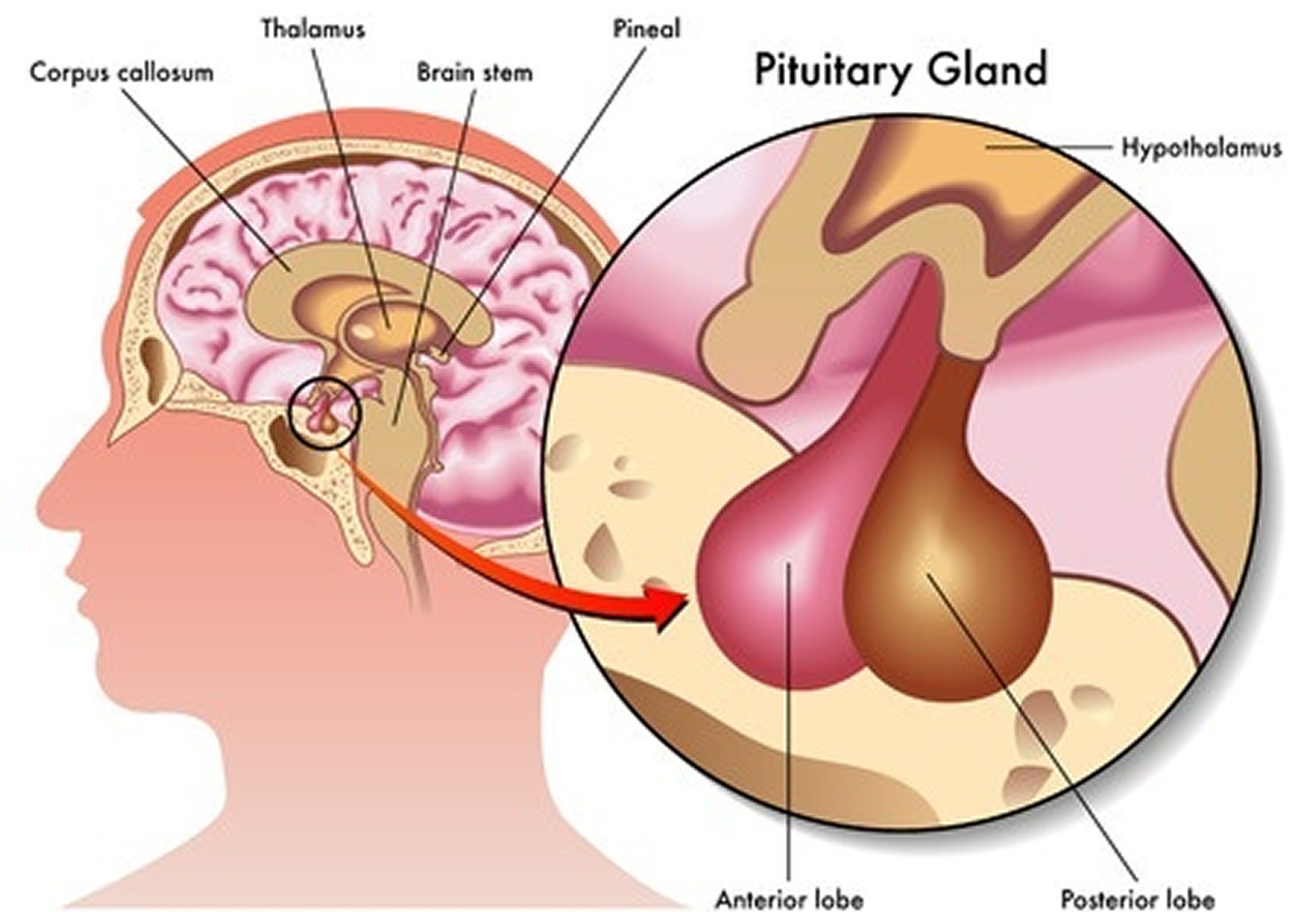
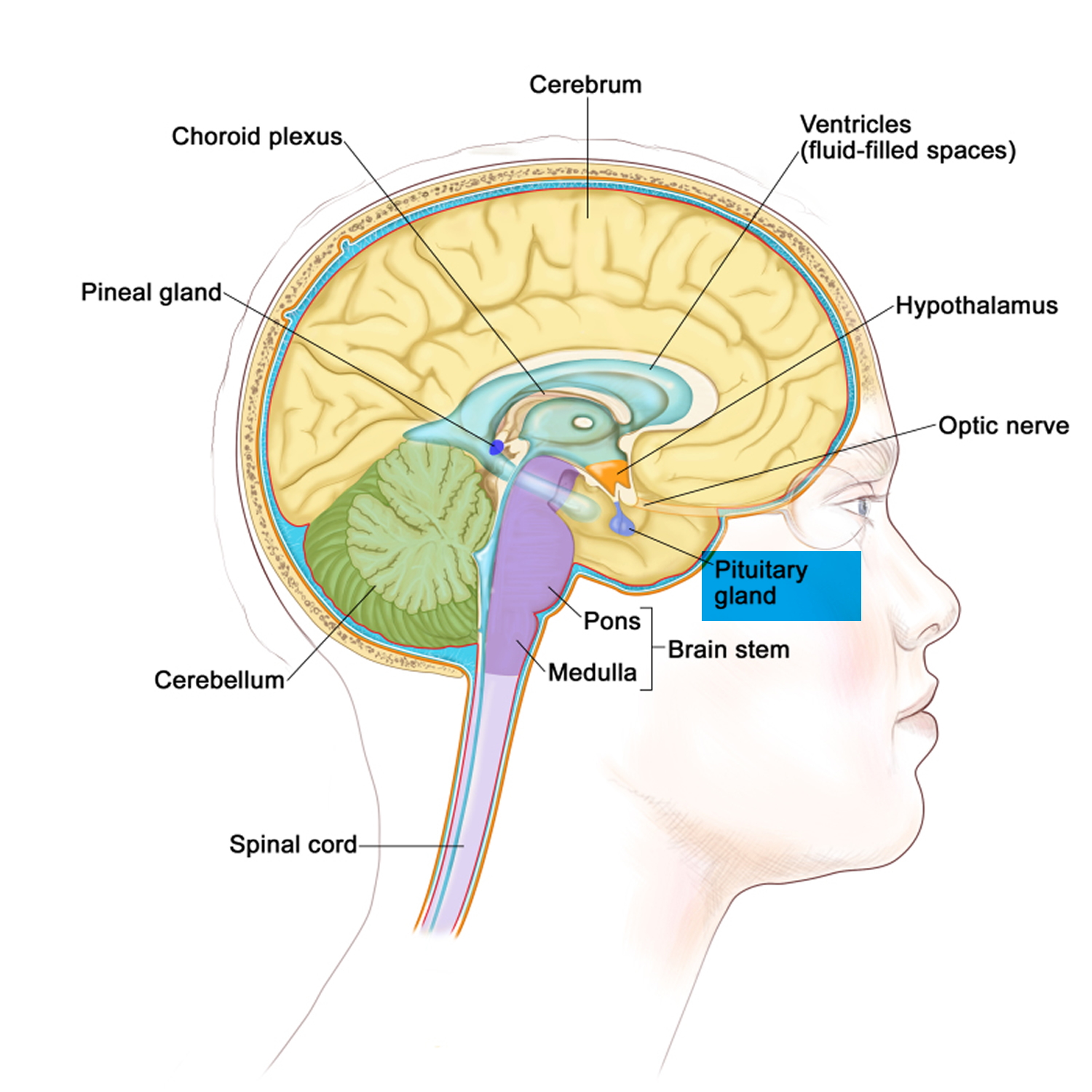
The pituitary gland (hypophysis) is located at the base of the brain, where a pituitary stalk (infundibulum) attaches it to the hypothalamus. The gland is about 1 centimeter in diameter and weighs about 0.5 to 1 g and is divided into anterior pituitary or anterior lobe, and a posterior pituitary, or posterior lobe. The pituitary stalk (infundibulum) contains both blood vessels and nerves. Despite its size, this gland secretes hormones that influence nearly every part of your body.
Figure 1. The pituitary gland location
Figure 2. Pituitary gland
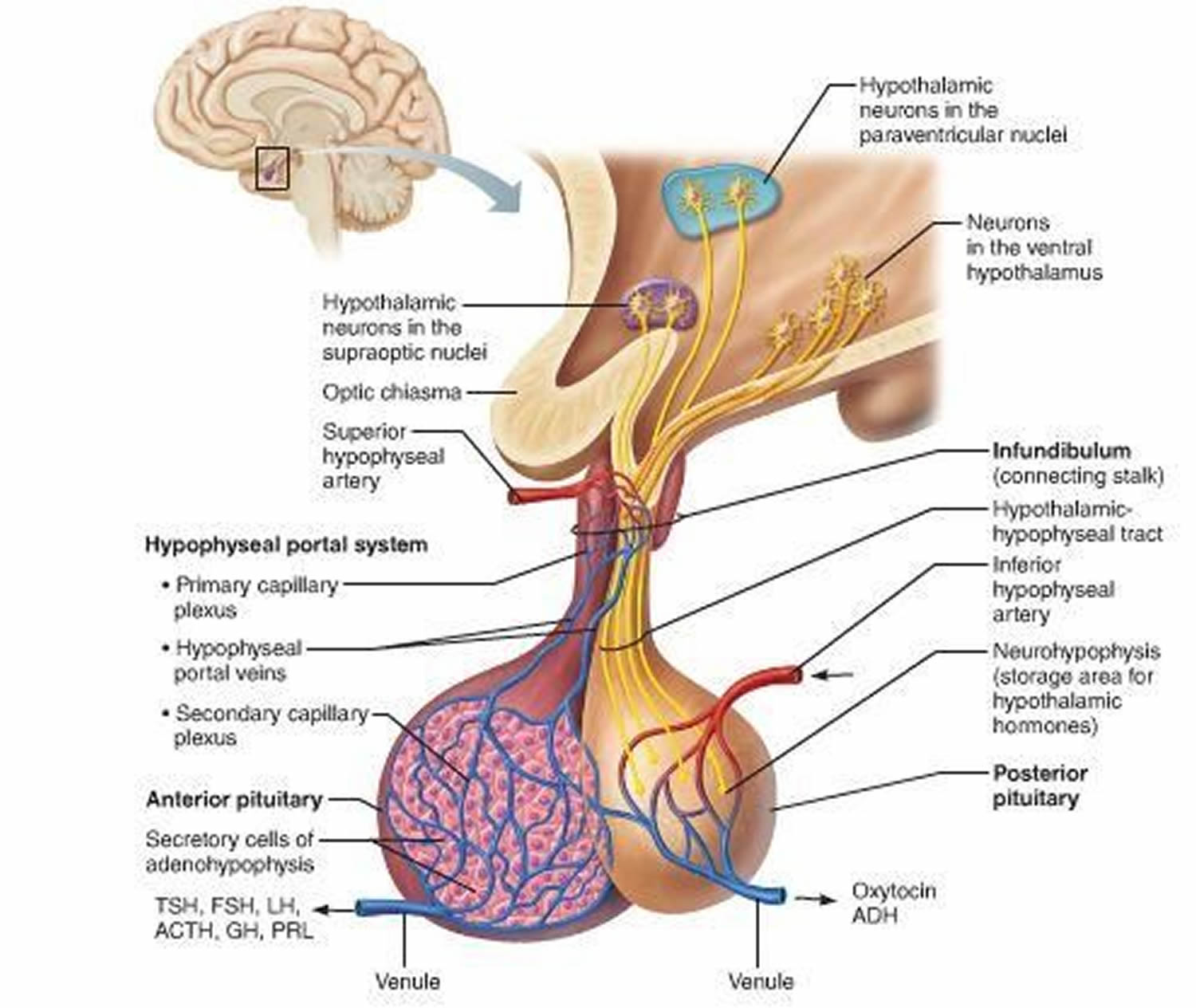
Figure 3. The hypothalamus and pituitary gland (anterior and posterior) endocrine pathways and target organs
Pituitary gland function 1
Anterior Pituitary Function and Hormones
The anterior pituitary is enclosed in a capsule of dense connective tissue. It consists largely of epithelial tissue organized in blocks around many thin-walled blood vessels. The cells of the anterior pituitary lobe (which constitutes 80% of the pituitary by weight) synthesize and release several hormones necessary for normal growth and development and also stimulate the activity of several target glands.
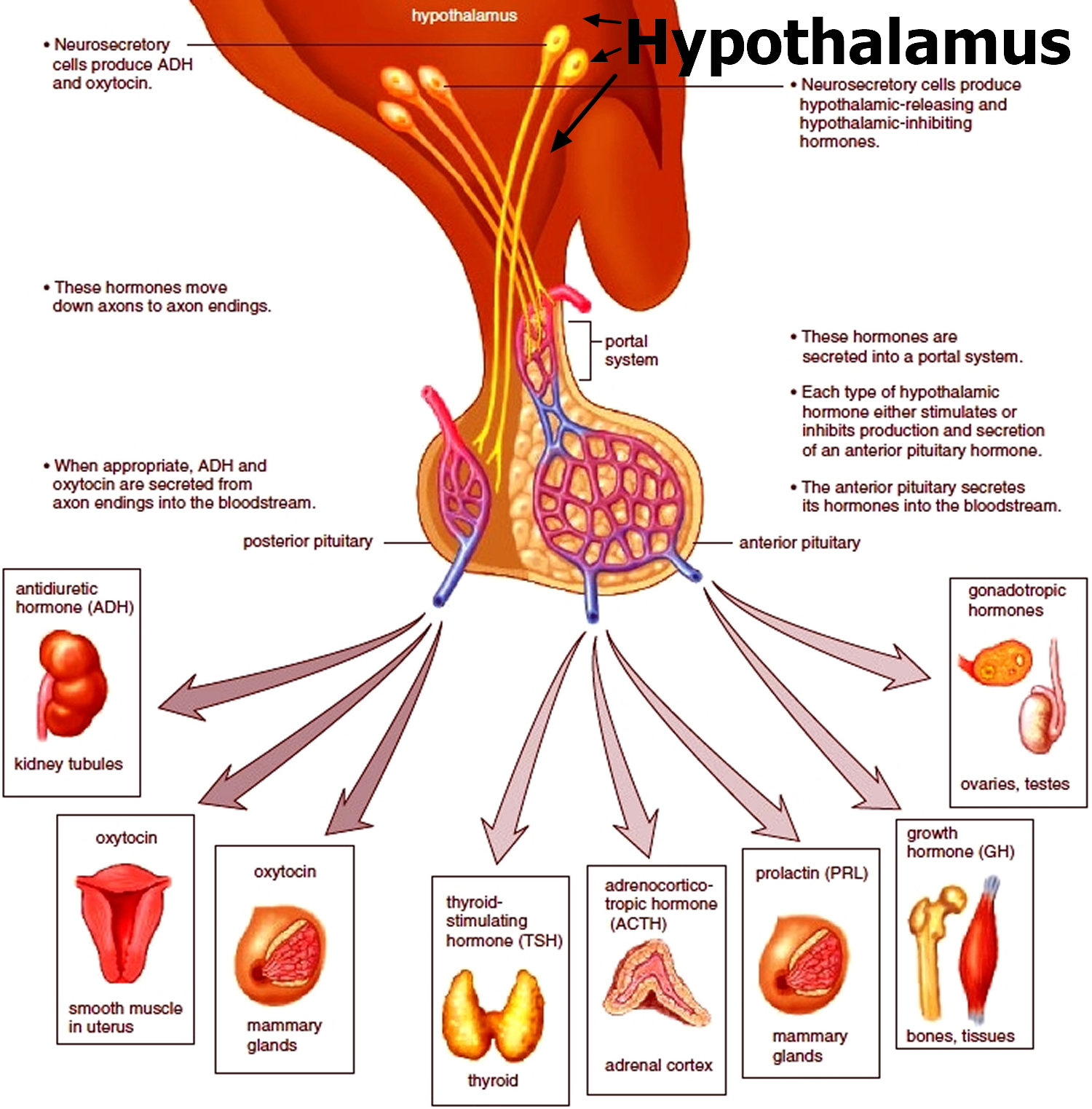
Anterior pituitary hormones are regulated by hypothalamic releasing and inhibitory hormones and by negative feedback of the target glandular hormones at the pituitary and hypothalamic levels (Table 1). Among pituitary hormones, only the secretion of prolactin is increased in the absence of hypothalamic influence, because it is mainly under tonic suppression by dopamine, the main inhibitory factor. Antidiuretic hormone (ADH, vasopressin) is produced by the supraoptic and paraventricular nuclei of the hypothalamus and travels in the axons through the pituitary stalk to the posterior pituitary gland.
Table 1. Relationship Among Hypothalamic, Pituitary, and Feedback Hormones and Target Glands
| Target Gland | Hypothalamic Regulatory Hormone | Pituitary Hormone | Feedback Hormone |
|---|---|---|---|
| Thyroid gland | TRH | TSH | T4, T3 |
| Gonad | LHRH (luteinizing hormone-releasing hormone) | LH | E2, T |
| Gonad | LHRH (luteinizing hormone-releasing hormone) | FSH | Inhibin, E2, T |
| Many organs | GHRH, SMS | GH | IGF-1 |
| Breast | PIF | Prolactin | ? |
| Adrenal | CRH, ADH | ACTH | Cortisol |
Note: ACTH, adrenocorticotropic hormone; ADH, antidiuretic hormone; CRH, corticotropin-releasing hormone; E2, estradiol; GHRH, growth hormone-releasing hormone; IGF-1, insulin-like growth factor 1; LHRH, luteinizing hormone-releasing hormone; PIF, prolactin release inhibitory factor; SMS, somatostatin; T, testosterone; T3, triiodothyronine; T4, thyroxine; TRH, thyrotropin-releasing hormone.
- Adrenocorticotropic hormone (ACTH)
Adrenocorticotropic hormone (ACTH) is also known as corticotropin. Corticotropin-releasing hormone (CRH) is the primary stimulator of adrenocorticotropic hormone (ACTH) release, but vasopressin plays a role during stress. Adrenocorticotropic hormone (ACTH) induces the adrenal cortex to release cortisol and several weak androgens, such as dehydroepiandrosterone (DHEA). Circulating cortisoland other corticosteroids (including exogenous corticosteroids) inhibit the release of CRH and ACTH. The CRH-ACTH-cortisol axis is a central component of the response to stress. Without adrenocorticotropic hormone (ACTH), the adrenal cortex atrophies and cortisol release virtually ceases.
- Thyroid-stimulating hormone (TSH)
Thyroid-stimulating hormone (TSH) regulates the structure and function of the thyroid gland and stimulates synthesis and release of thyroid hormones. Thyroid-stimulating hormone (TSH) synthesis and release are stimulated by the hypothalamic hormone thyrotropin-releasing hormone (TRH) and suppressed (by negative feedback) by circulating thyroid hormones.
- Luteinizing hormone (LH) and follicle-stimulating hormone (FSH)
Luteinizing hormone (LH) and follicle-stimulating hormone (FSH) control the production of the sex hormones. Synthesis and release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) are stimulated mainly by gonadotropin-releasing hormone (GnRH) and suppressed by estrogen and testosterone. One factor controlling GnRH release is kisspeptin, a hypothalamic peptide that is triggered by increased leptin levels at puberty. Two gonadal hormones, activin and inhibin, affect only FSH; activin is stimulative, and inhibin is inhibitory.
In women, luteinizing hormone (LH) and follicle-stimulating hormone (FSH) stimulate ovarian follicular development and ovulation. In men, FSH acts on Sertoli cells and is essential for spermatogenesis; LH acts on Leydig cells of the testes to stimulate testosterone biosynthesis.
- Growth hormone (GH)
Growth hormone (GH) stimulates somatic growth and regulates metabolism. Growth hormone–releasing hormone (GHRH) is the major stimulator and somatostatin is the major inhibitor of the synthesis and release of growth hormone. Growth hormone (GH) controls synthesis of insulin-like growth factor 1 (IGF-1, also called somatomedin-C), which largely controls growth. Although IGF-1 is produced by many tissues, the liver is the major source. A variant of IGF-1 occurs in muscle, where it plays a role in enhancing muscle strength. It is less under control of growth hormone than is the liver variant.
The metabolic effects of growth hormone are biphasic. Growth hormone initially exerts insulin-like effects, increasing glucose uptake in muscle and fat, stimulating amino acid uptake and protein synthesis in liver and muscle, and inhibiting lipolysis in adipose tissue. Several hours later, more profound anti–insulin-like metabolic effects occur. They include inhibition of glucose uptake and use, causing blood glucose and lipolysis to increase, which increases plasma free fatty acids. Growth hormone levels increase during fasting, maintaining blood glucose levels and mobilizing fat as an alternative metabolic fuel. Production of growth hormone decreases with aging. Ghrelin, a hormone produced in the fundus of the stomach, promotes growth hormone release from the pituitary, increases food intake, and improves memory.
Insufficient secretion of growth hormone (GH) during childhood limits growth, causing pituitary dwarfism. Body parts are normally proportioned, and mental development is normal—the individual is just very small. Typically, hormone therapy can stimulate some growth. Oversecretion of growth hormone during childhood causes gigantism, in which height may exceed 8 feet. This rare condition is usually a result of a pituitary gland tumor, which may also cause oversecretion of other pituitary hormones. As a result, a person with gigantism often has several metabolic disturbances. Acromegaly is the overproduction of growth hormone in adulthood. The many symptoms attesting to the wide effects of this hormone include enlargement of the heart, the bones, the thyroid gland, facial features, the hands, the feet, and the head. Early symptoms include headache, joint pain, fatigue, and depression.
- Prolactin
Prolactin is produced in cells called lactotrophs that constitute about 30% of the cells of the anterior pituitary. The pituitary doubles in size during pregnancy, largely because of hyperplasia and hypertrophy of lactotrophs. In humans, the major function of prolactin is stimulating milk production. Also, prolactin release occurs during sexual activity and stress. Prolactin may be a sensitive indicator of pituitary dysfunction; prolactin is the hormone most frequently produced in excess by pituitary tumors, and it may be one of the hormones to become deficient from infiltrative disease or tumor compression of the pituitary.
- Other hormones
Several other hormones are produced by the anterior pituitary. These include pro-opiomelanocortin (POMC, which gives rise to ACTH), alpha- and beta-melanocyte-stimulating hormone (MSH), beta-lipotropin (β-LPH), the enkephalins, and the endorphins. Pro-opiomelanocortin (POMC) and melanocyte-stimulating hormone (MSH) can cause hyperpigmentation of the skin and are only significant clinically in disorders in which ACTH levels are markedly elevated (eg, Addison disease, Nelson syndrome). The function of β-LPH is unknown. Enkephalins and endorphins are endogenous opioids that bind to and activate opioid receptors throughout the brain.
Posterior Pituitary Function
The posterior pituitary consists mostly of axons and neuroglia, unlike the anterior pituitary, which is composed primarily of glandular epithelial cells. Neuroglia support the axons, which originate from neurons in the hypothalamus. The secretions of these neurons function not as neurotransmitters, but as hormones.
The posterior pituitary releases vasopressin (also called arginine vasopressin or antidiuretic hormone [ADH]) and oxytocin. These hormones are transported down axons through the pituitary stalk to the posterior pituitary lobe, and are stored in vesicles (secretory granules) near the ends of the axons. Impulses from the hypothalamus release the hormones into the blood and have half-lives of about 10 min. Thus, though synthesized in the hypothalamus, antidiuretic hormone (ADH) and oxytocin are considered posterior pituitary hormones because they enter the bloodstream from the posterior pituitary gland.
- Antidiuretic Hormone (ADH)
A diuretic is a chemical that increases urine production, whereas an antidiuretic decreases urine formation. Antidiuretic hormone (ADH) produces an antidiuretic effect by reducing the volume of water the kidneys excrete. In this way, antidiuretic hormone helps regulate the water concentration of body fluids. Antidiuretic hormone (ADH) acts primarily to promote water conservation by the kidneys by increasing the permeability of the distal tubular epithelium to water. At high concentrations, antidiuretic hormone (ADH) also causes vasoconstriction. Like aldosterone, antidiuretic hormone plays an important role in maintaining fluid homeostasis and vascular and cellular hydration. The main stimulus for antidiuretic hormone release is increased osmotic pressure of water in the body, which is sensed by osmoreceptors in the hypothalamus. The other major stimulus is volume depletion, which is sensed by baroreceptors in the left atrium, pulmonary veins, carotid sinus, and aortic arch, and then transmitted to the brain through the vagus and glossopharyngeal nerves. Other stimulants for antidiuretic hormone release include pain, stress, emesis, hypoxia, exercise, hypoglycemia, cholinergic agonists, beta-blockers, angiotensin, and prostaglandins. Inhibitors of antidiuretic hormone release include alcohol, alpha-blockers, and glucocorticoids.
A lack of antidiuretic hormone causes central diabetes insipidus. An inability of the kidneys to respond normally to antidiuretic hormone causes nephrogenic diabetes insipidus. Removal of the pituitary gland usually does not result in permanent diabetes insipidus because some of the remaining hypothalamic neurons produce small amounts of vasopressin.
Copeptin is coproduced with antidiuretic hormone in the posterior pituitary. Measuring it may be useful in distinguishing the cause of hyponatremia.
- Oxytocin
Oxytocin has 2 major targets:
- Myoepithelial cells of the breast, which surround the alveoli of the mammary gland
- Smooth muscle cells of the uterus
Suckling stimulates the production of oxytocin, which causes the myoepithelial cells to contract. This contraction causes milk to move from the alveoli to large sinuses for ejection (ie, the milk letdown reflex of nursing mothers). Oxytocin stimulates contraction of uterine smooth muscle cells, and uterine sensitivity to oxytocin increases throughout pregnancy. However, plasma levels do not increase sharply during parturition, and the role of oxytocin in the initiation of labor is unclear.
In males, oxytocin may play a role in the sexual response, including erection of the penis and movement of sperm. In addition, oxytocin is an antidiuretic, but it is much weaker than antidiuretic hormone (ADH). Men have extremely low levels of oxytocin.
Hypopituitarism causes
Hypopituitarism may be the result of inherited disorders, but more often it’s acquired. Hypopituitarism frequently is triggered by a tumor of the pituitary gland. As a pituitary tumor increases in size, it can compress and damage pituitary tissue, interfering with hormone production. A tumor can also compress the optic nerves, causing visual disturbances.
The cause of hypopituitarism can also be other diseases and events that damage the pituitary.
Hypopituitarism can be caused by:
- Tumors of the pituitary gland or hypothalamus (e.g., pituitary adenomas, which are usually benign, meaning not cancer)
- Radiation therapy to the brain for a tumor, which can destroy pituitary gland tissue
- Chemotherapy
- Brain surgery
- Traumatic brain injury, such as with a head injury from an accident
- Severe bleeding in the brain or severe blood loss during childbirth. The loss of blood leads to tissue death in the pituitary gland. This condition is called Sheehan syndrome.
- Autoimmune condition that causes inflammation of the pituitary (lymphocytic hypophysitis)
- Subarachnoid hemorrhage (from a burst aneurysm)
- Stroke
- Death of an area of tissue in the pituitary gland (pituitary apoplexy)
- Tuberculosis
- Infections or inflammation of the brain and the tissues that support the brain, such as meningitis
- Certain conditions present at birth
- Infiltrative diseases, such as sarcoidosis, which is an inflammatory disease occurring in various organs;
- Langerhans cell histiocytosis (histiocytosis X), in which abnormal cells cause scarring in numerous parts of the body, such as the lungs and bones;
- Hemochromatosis, which causes excess iron deposition in the liver and other tissues
- Severe loss of blood during childbirth, which may cause damage to the front part of the pituitary gland (Sheehan syndrome or postpartum pituitary necrosis)
- Genetic mutations resulting in impaired pituitary hormone production
- Certain medicines can also suppress pituitary function. The most common drugs are glucocorticoids (such as prednisone and dexamethasone), which are taken for inflammatory and immune conditions. Drugs used to treat prostate cancer can also lead to low pituitary function.
Diseases of the hypothalamus, a portion of the brain situated just above the pituitary, also can cause hypopituitarism. The hypothalamus produces hormones of its own that directly affect the activity of the pituitary.
Sometimes, the cause of hypopituitarism is unknown.
Hypopituitarism life expectancy
Hypopituitarism is usually permanent. It requires lifelong treatment with one or more medicines. But you can expect a normal life span, as long as you regularly take the medications recommended by your doctor.
Hypopituitarism possible complications
Side effects of medicines to treat hypopituitarism can develop. However, do not stop any medicine on your own without talking with your health care provider first.
Hypopituitarism symptoms
Hypopituitarism is often progressive. Although the signs and symptoms can occur suddenly, they more often develop gradually. They are sometimes subtle and may be overlooked for months or even years.
Signs and symptoms of hypopituitarism vary, depending on which pituitary hormones are deficient and how severe the deficiency is.
Symptoms can include one or more of the following:
- Stomach pain, decreased appetite, nausea and vomiting, and constipation
- Excessive thirst and urination
- Fatigue and/or weakness
- Dizziness or fainting
- Anemia, meaning weakness from not having enough red blood cells
- Headache and dizziness
- Low blood pressure
- Low blood sugar
- Sensitivity to cold
- Vision problems
- Hoarseness or changing voice
- Joint stiffness
- Weight loss or weight gain
- Stiffness in the joints
- Hypophysitis (inflammation of the pituitary gland)
- Histiocytosis
- Hypopituitarism symptoms in women: loss of armpit or pubic hair, decreased sex drive, infertility, problems with breast feeding, irregular or no menstrual periods, and hot flashes
- Hypopituitarism symptoms in men: loss of hair (on the face, or in the armpits or pubic area), decreased sex drive, infertility, erectile dysfunction
- Hypopituitarism symptoms in children, problems with growth (including height – short stature) and sexual development. Short height (less than 5 feet or 1.5 meters) if onset is during a growth period
Symptoms may develop slowly and may vary greatly, depending upon:
- The number of hormones that are missing and the organs they affect
- The severity of the disorder
Hypopituitarism dwarfism
Growth hormone deficiency means the pituitary gland does not make enough growth hormone. Children with physical defects of the face and skull, such as cleft lip or cleft palate, may have decreased growth hormone level.
Most of the time, the cause of growth hormone deficiency is unknown.
Slow growth may first be noticed in infancy and continue through childhood. The pediatrician will most often draw the child’s growth curve on a growth chart. Children with growth hormone deficiency have a slow or flat rate of growth. The slow growth may not show up until a child is 2 or 3 years old.
The child will be much shorter than most children of the same age and gender. The child will still have normal body proportions, but may be chubby. The child’s face often looks younger than other children of the same age. The child will have normal intelligence in most cases.
In older children, puberty may come late or may not come at all, depending on the cause.
Hypopituitarism dwarfism prognosis
The earlier the condition is treated, the better the chance that a child will grow to near-normal adult height. Many children gain 4 or more inches (about 10 centimeters) during the first year, and 3 or more inches (about 7.6 centimeters) during the next 2 years. The rate of growth then slowly decreases.
Growth hormone therapy does not work for all children.
Left untreated, growth hormone deficiency may lead to short stature and delayed puberty.
Growth hormone deficiency can occur with deficiencies of other hormones such as those that control:
- Production of thyroid hormones
- Water balance in the body
- Production of male and female sex hormones
- The adrenal glands and their production of cortisol, DHEA, and other hormones
Hypopituitarism dwarfism prevention
Most cases are not preventable.
Review your child’s growth chart with the pediatrician at each checkup. If there is concern about your child’s growth rate, evaluation by a specialist is recommended.
Hypopituitarism dwarfism diagnosis
A physical exam, including weight, height, and body proportions, will show signs of slowed growth. The child will not follow the normal growth curves.
A hand x-ray can determine bone age. Normally, the size and shape of bones change as a person grows. These changes can be seen on an x-ray and they most often follow a pattern as a child grows older.
Testing is most often done after the pediatrician has looked into other causes of poor growth. Tests that may be done include:
- Insulin-like growth factor 1 (IGF-1) and insulin-like growth factor binding protein 3 (IGFBP3). These are substances that growth hormones cause the body to make. Tests can measure these growth factors. Accurate growth hormone deficiency testing involves a stimulation test. This test takes several hours.
- MRI of the head can show the hypothalamus and pituitary glands.
- Tests to measure other hormone levels may be done, because a lack of growth hormone may not be the only problem.
Hypopituitarism dwarfism treatment
Treatment involves growth hormone shots (injections) given at home. The shots are most often given once a day. Older children can often learn how to give themselves the shot.
Treatment with growth hormone is long-term, often lasting for several years. During this time, the child needs to be seen regularly by the pediatrician to ensure the treatment is working. If needed, the provider will change the dosage of the medicine.
Serious side effects of growth hormone treatment are rare. Common side effects include:
- Headache
- Fluid retention
- Muscle and joint aches
- Slippage of the hip bones
Table 2. Long-term benefits and risks of growth hormone replacement therapy in adult patients with growth hormone deficiency.
| Patient Data | Benefits | Risks or Drawbacks |
|---|---|---|
| Body composition | Reduction in fat mass Increase in lean mass Increase in muscle strength | Increase in BMI Increased waist circumference Increase of waist-hip index |
| Bone metabolism | Increase in bone mineral density | Effect on the incidence of fractures not clearly shown |
| Health-related quality of life | Improvement in quality of life questionnaires Greater benefit in patients with low quality of life at baseline | No improvement in all dimensions Probable absence of effect in patients with normal quality of life |
| Cardiovascular risk markers | Increase in HDL-chol Reduction of total and LDL-chol Diastolic blood pressure reduction Reduction of CRP Reduction of carotid intima-media thickness | Reduced insulin sensitivity Increase in fasting glucose and insulin Trend to the increase in the prevalence of metabolic syndrome Increase in lipoprotein (a) |
| Cardiovascular disease | Reduction in the incidence rate of myocardial infarction | Trend to increase in cerebrovascular disease |
| Neoplasms | No increase in the rate of recurrence or progression of hypothalamic-pituitary tumors No increase in overall risk of neoplasia in adults with GHD | Tendency to increase risk of second malignancy in childhood cancer survivors treated with GH in childhood There are subgroups with increased risk of certain neoplasia in adults who were treated with GH in childhood |
| Mortality | Tendency to decrease the global and cardiovascular mortality of hypopituitarism | Persistence of higher mortality than the general population in some studies |
Note: BMI, body mass index; chol, cholesterol; HDL, high density lipoprotein; LDL, low density lipoprotein; CRP, C-reactive protein; GHD, deficiency of growth hormone.
[Source 2]Hypopituitarism diagnosis
If your doctor suspects a pituitary disorder, he or she will likely order several tests to check levels of various hormones in your body. Your doctor may also want to check for hypopituitarism if you’ve had a recent head injury or radiation treatment that might have put you at risk of damage to your pituitary gland.
Your doctor will check your hormone levels with blood tests. You may have other tests, such as an MRI of your pituitary gland, to help find the cause of your hypopituitarism.
Tests your doctor may order include:
- Blood tests. They can help detect deficits in hormones as a result of pituitary failure. For example, blood tests can identify low levels of thyroid, adrenal or sex hormones, and can determine if these low levels are associated with inadequate pituitary hormone production.
- ACTH
- Cortisol
- Estradiol (estrogen)
- Follicle-stimulating hormone (FSH)
- Insulin-like growth factor 1 (IGF-1)
- Luteinizing hormone (LH)
- Testosterone level
- Thyroid-stimulating hormone (TSH)
- Thyroid hormone (T4)
- Stimulation or dynamic testing. Your doctor may suggest you go to a specialized endocrine clinic for these tests, which check your body’s secretion of hormones after you’ve taken certain medications that can stimulate hormone production.
- Brain imaging. Magnetic resonance imaging (MRI) of your brain can detect a pituitary tumor or other structural abnormality.
- Vision tests. These tests can determine if growth of a pituitary tumor has impaired your sight or visual fields.
Level of a pituitary hormone may be high in the bloodstream if you have a pituitary tumor that is producing too much of that hormone. The tumor may crush other cells of the pituitary, leading to low levels of other hormones.
Hypopituitarism treatment
Successful treatment of the underlying condition causing hypopituitarism may lead to a complete or partial recovery of your body’s normal production of pituitary hormones.
Treatment with the appropriate hormones is often the first step. These drugs are considered as “replacement,” rather than treatment, because the dosages are set to match the amounts that your body would normally manufacture if it didn’t have a pituitary problem. Treatment sometimes may be lifelong.
Your doctor also will teach you how to take extra cortisone (a hormone) when you are sick or under stress. If a tumor is causing your hypopituitarism, you might need surgery to remove it and/or possibly radiation treatment. If needed, you can take medicine for infertility.
You will need to get regular check-ups. It’s wise to wear medical identification, such as a bracelet or pendant, which provides information about your condition in case of an emergency.
Hypopituitarism medication
Hormone replacement medications may include:
- Corticosteroids. These drugs, such as hydrocortisone or prednisone, replace the adrenal hormones that aren’t being produced because of an adrenocorticotropic hormone (ACTH) deficiency. You take them by mouth.
- Levothyroxine (Levoxyl, Synthroid, others). This medication replaces deficient thyroid hormone levels caused by low or deficient TSH production.
- Sex hormones. These include testosterone in men and estrogen or a combination of estrogen and progesterone in women. Testosterone is administered through the skin with either a patch or a gel or by injection. Female hormone replacement can be administered with pills, gels or patches.
- Growth hormone. Also called somatropin, growth hormone is taken through an injection beneath your skin. It promotes growth, thus producing more normal height in children. Adults with a growth hormone deficiency also may benefit from growth hormone replacement, but they won’t grow taller.
If you’ve become infertile, luteinizing hormone (LH) and follicle-stimulating hormone (FSH) (gonadotropins) can be administered by injection to stimulate ovulation in women and sperm production in men.
Monitoring and adjusting
A doctor who specializes in endocrine disorders (endocrinologist) may monitor the levels of these hormones in your blood to ensure you’re getting adequate — but not excessive — amounts.
Your doctor will advise you to adjust your dosage of corticosteroids if you become seriously ill or experience major physical stress. During these times, your body would ordinarily produce extra cortisol hormone. The same kind of fine-tuning of dosage may be necessary when you have the flu, experience diarrhea or vomiting, or have surgery or dental procedures. Adjustments in dosage may also be necessary during pregnancy or with marked changes in weight. You may need periodic CT or MRI scans as well to monitor a pituitary tumor or other diseases causing the hypopituitarism.
In case of emergency
Wear a medical alert bracelet or pendant, and carry a special card, notifying others — in emergency situations, for example — that you’re taking corticosteroids and other medications.
- Merck Sharp & Dohme Corp. Merck Manual. Overview of the Endocrine System. https://www.merckmanuals.com/professional/endocrine-and-metabolic-disorders/principles-of-endocrinology/overview-of-the-endocrine-system#v27411775[↩]
- Díez JJ, Sangiao-Alvarellos S, Cordido F. Treatment with Growth Hormone for Adults with Growth Hormone Deficiency Syndrome: Benefits and Risks. International Journal of Molecular Sciences. 2018;19(3):893. doi:10.3390/ijms19030893. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5877754/[↩]