Contents
- What is intracranial hemorrhage
- Intracranial hemorrhage prognosis
- Types of intracranial hemorrhage
- Intracranial hemorrhage causes
- Risk factors for intracranial hemorrhage
- Intracranial hemorrhage symptoms
- Intracranial hemorrhage diagnosis
- Intracranial hemorrhage treatment
- Acute management
- Recommended Guidelines from the American Heart Association and American Stroke Association for Treating Elevated BP in Spontaneous intracranial hemorrhage
- Recommendations from European Stroke Initiative for blood pressure management in intracranial hemorrhage
- Intracranial pressure management
- Hyperglycemia management
- Surgical interventions
- Acute management
What is intracranial hemorrhage
Spontaneous intracranial hemorrhage is a serious form of stroke which appears suddenly without warning, unlike ischemic strokes that are often preceded by a transient ischemic attack. Spontaneous intracranial hemorrhage accounts for 10-15% of stroke but it is one of the most disabling forms of stroke 1. Intracranial hemorrhage refers to any bleeding within the intracranial vault, including the brain parenchyma and surrounding meningeal spaces 2. The overall incidence of spontaneous intracranial hemorrhage worldwide is 24.6 per 100,000 person-years with approximately 40,000 to 67,000 cases per year in the United States 3. Intracranial hemorrhage remains the most devastating form of stroke with the 30-day mortality rate ranges from 35% to 52% with only 20% of survivors expected to have full functional recovery at 6 months 4. Approximately half of this mortality occurs within the first 24 hours 5, highlighting the critical importance of early and effective treatment in the emergency department. Death at 1 year for intracranial hemorrhage varies by location of intracranial hemorrhage: 51% for deep hemorrhage, 57% for lobar, 42% for cerebellar, and 65% for brain stem 6. Of the estimated 67,000 patients who had an intracranial hemorrhage in the United States during 2002, only 20% are expected to be functionally independent at 6 months 7.
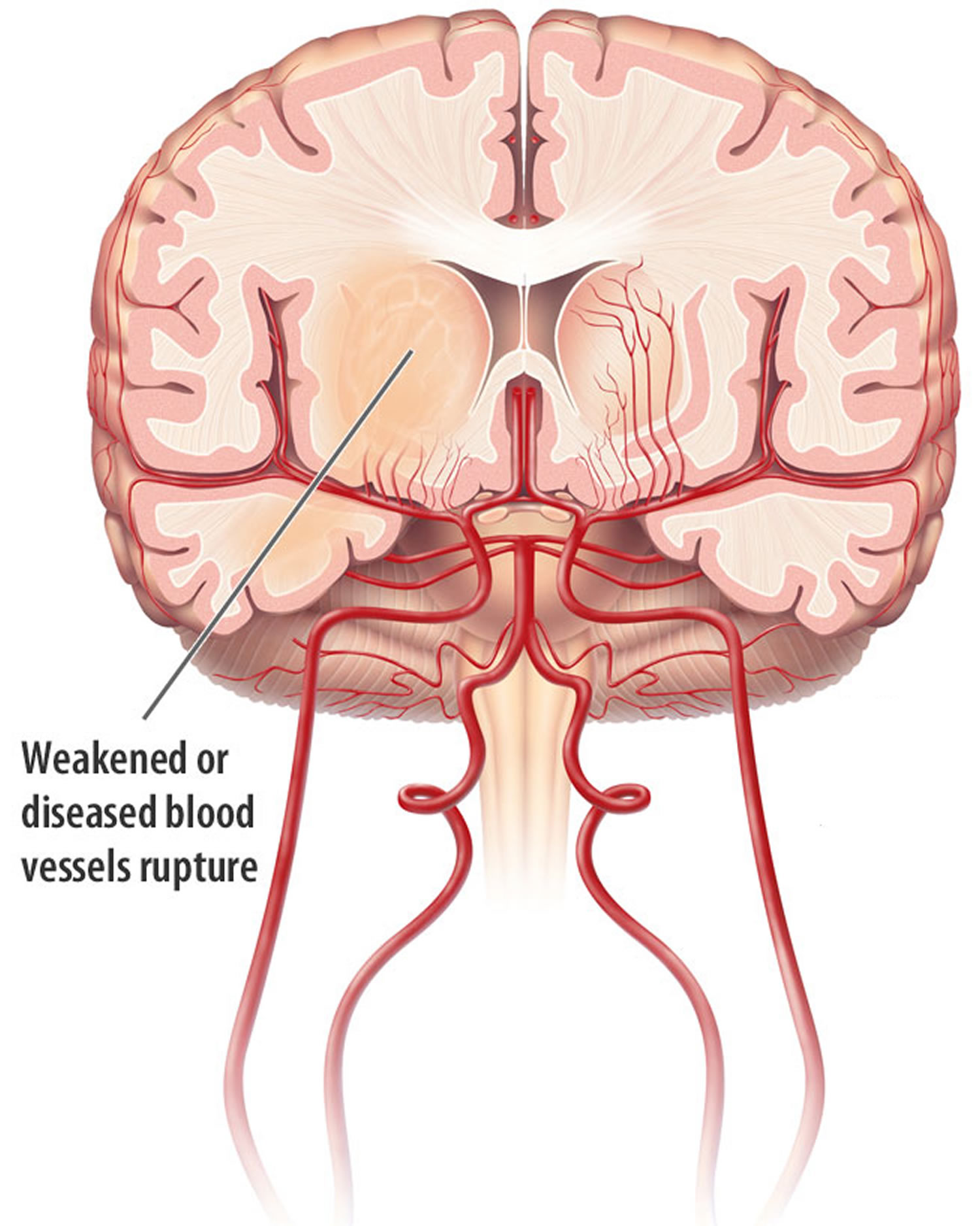
The most common cause of intracranial hemorrhage is high blood pressure (hypertension). Since high blood pressure by itself often causes no symptoms, many people with intracranial hemorrhage are not aware that they have high blood pressure, or that it needs to be treated. Less common causes of intracranial hemorrhage include trauma, infections, tumors, blood clotting deficiencies, and abnormalities in blood vessels (such as arteriovenous malformations).
Intracranial hemorrhage occurs at all ages. The average age is lower than for ischemic stroke. Less common than ischemic strokes, intracranial hemorrhage or hemorrhagic strokes make up about 12 percent of all strokes.
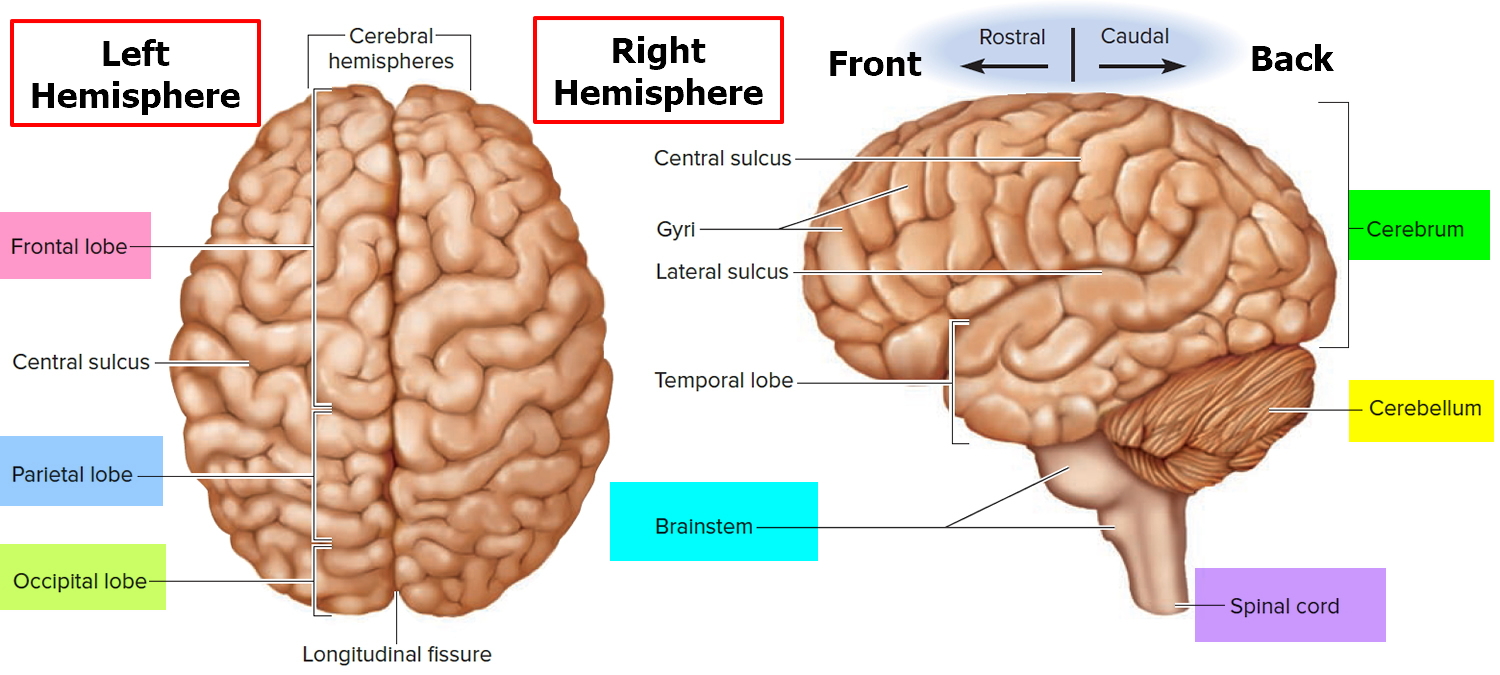
Figure 1. Human brain
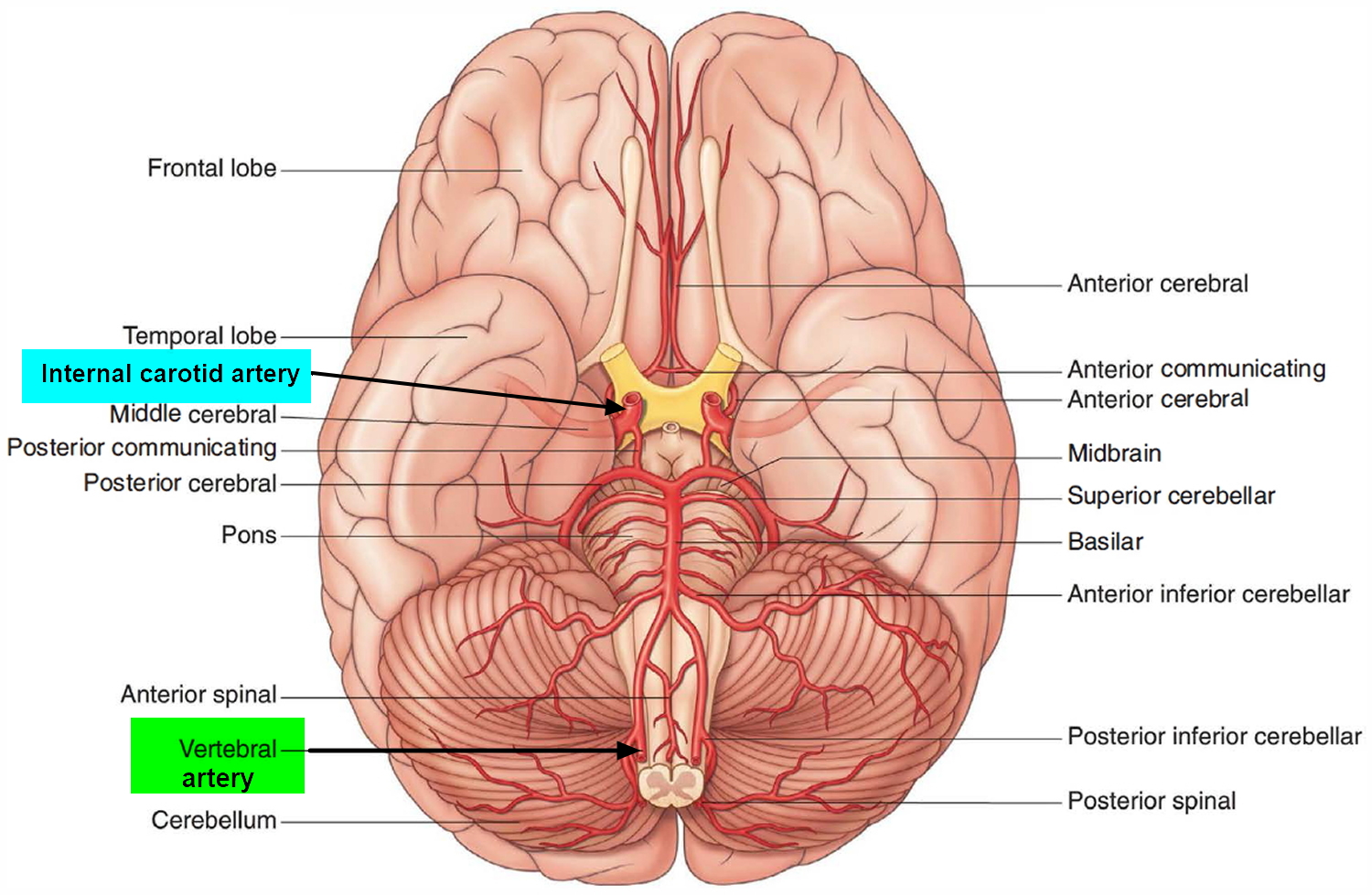
Figure 2. Brain blood supply
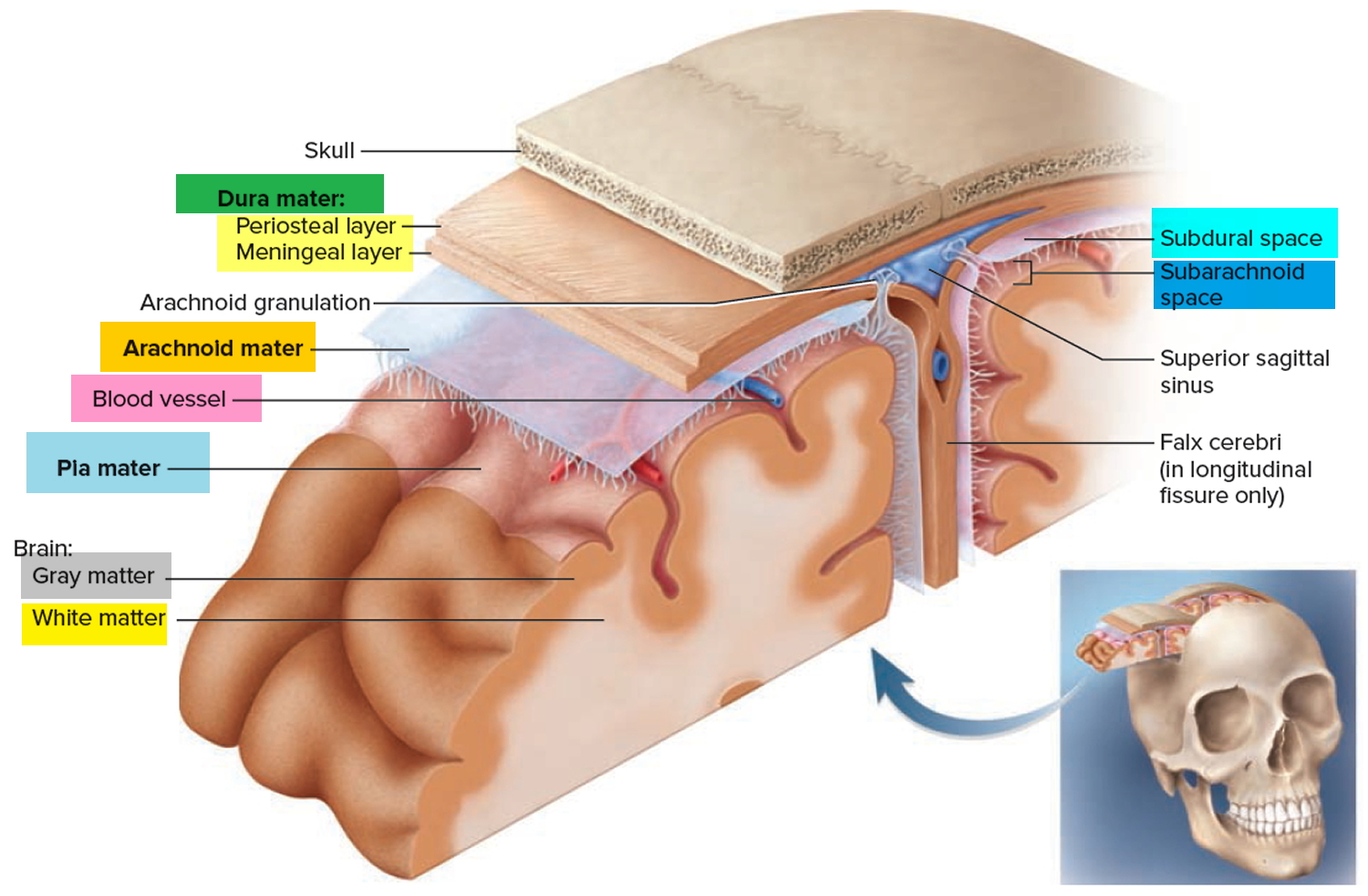
Figure 3. Meninges of the brain
Intracranial hemorrhage prognosis
Multiple grading scores exist that allow for evidence-based risk stratification in the acute phase. First, the intracranial hemorrhage score predicts 30-day mortality using features such as age, intracranial hemorrhage volume and the presence of intraventricular hemorrhage, with higher score associated with worse outcome (Table 1) 8. Second, the FUNC score (FUNCtional outcome risk stratification) predicts functional independence rather than mortality at 90-days (Table 2) 9. The higher the FUNC score, the greater the chance of the patient recovering functional independence.
Table 1. Intracranial hemorrhage score
| Component | Intracranial hemorrhage score points |
|---|---|
| GCS score (Glasgow Coma Score) | |
| 3–4 | 2 |
| 5–12 | 1 |
| 13–15 | 0 |
| Intracranial hemorrhage volume in cm3 | |
| ≥30 | 1 |
| <30 | 0 |
| Intraventricular hemorrhage | |
| Yes | 1 |
| No | 0 |
| Infratentorial origin of Intracranial hemorrhage | |
| Yes | 1 |
| No | 0 |
| Age in years | |
| ≥80 | 1 |
| <80 | 0 |
Note: Intracranial hemorrhage score predicts 30-day mortality with higher score associated with worse outcome
[Source 2]Table 2. FUNC score (FUNCtional outcome risk stratification)
| Component | FUNC Score Points |
|---|---|
| ICH volume, cm3 | |
| <30 | 4 |
| 30–60 | 2 |
| >60 | 0. |
| Age, years | |
| <70 | 2 |
| 70–79 | 1 |
| ≥80 | 0 |
| ICH location | |
| Lobar | 2 |
| Deep | 1 |
| Infratentorial | 0 |
| GCS score | |
| ≥9 | 2 |
| ≤8 | 0 |
| Pre-ICH cognitive impairment | |
| No | 1 |
| Yes | 0 |
| Total FUNC score | 0–11 |
Note: The higher the FUNC score, the greater the chance of the patient recovering functional independence.
[Source 2]Types of intracranial hemorrhage
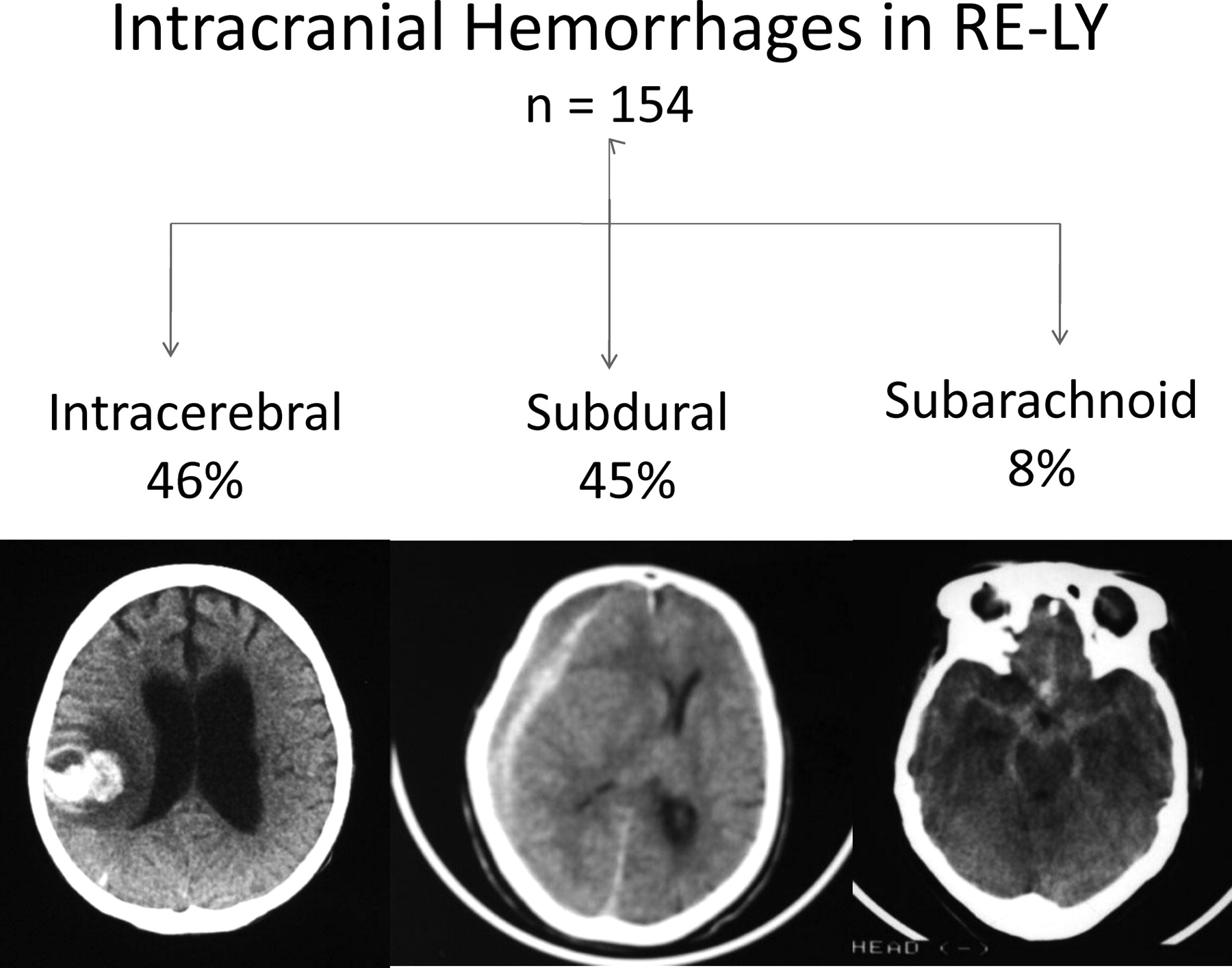
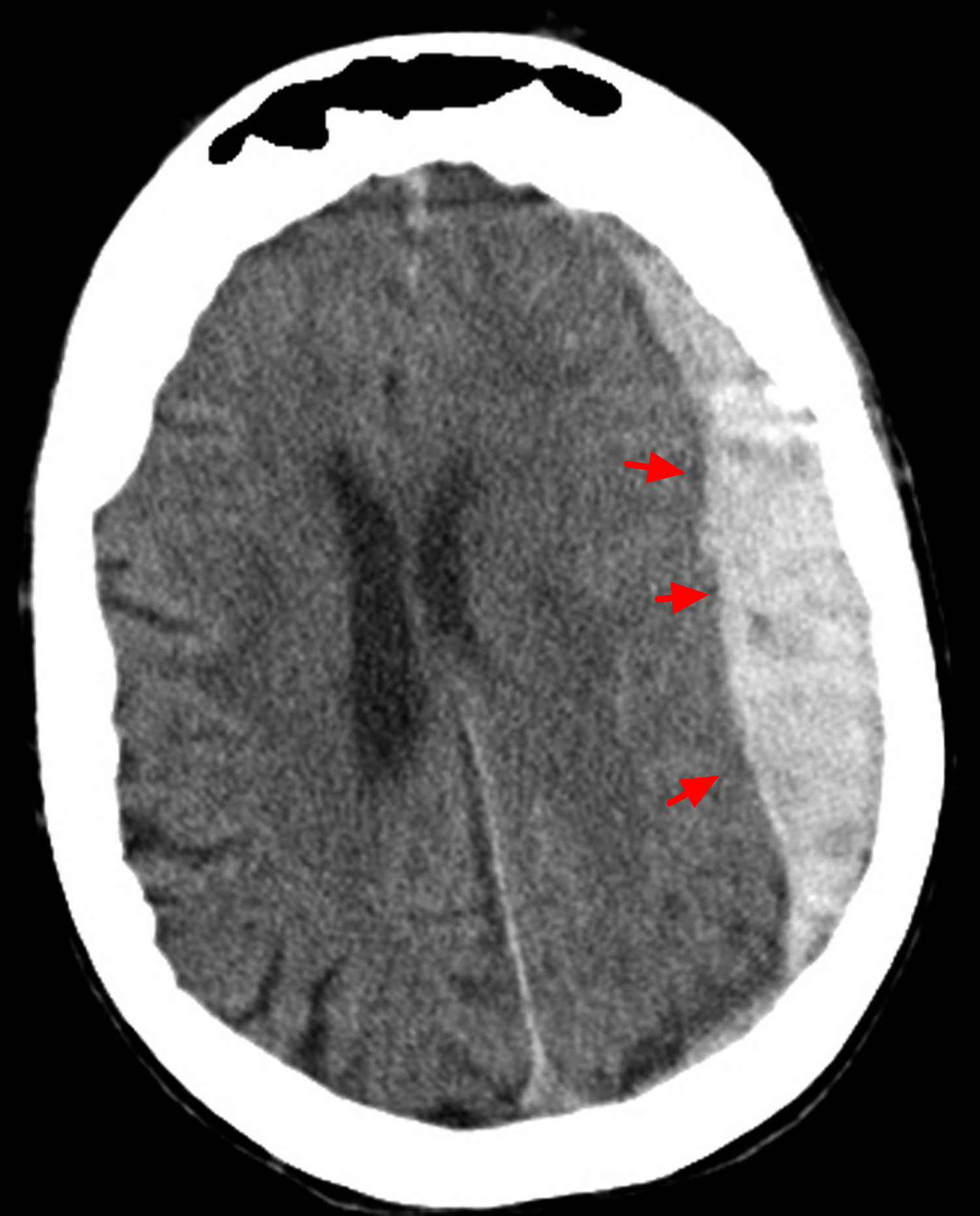
Intracranial hemorrhage is diagnosed by its anatomical location. Intraparenchymal hemorrhage (intracerebral hemorrhage) refers to nontraumatic bleeding into the brain parenchyma. Subarachnoid hemorrhage (SAH) refers to bleeding into the space between the pia and the arachnoid membranes. Nontraumatic causes of rupture include cerebral aneurysms, bleeding from arteriovenous malformations or tumors, cerebral amyloid angiopathy, and vasculopathies (such as vasculitis). A subdural hematoma (Figure 3) is due to bleeding between the dura and the arachnoid, whereas an epidural hematoma involves bleeding between the dura and the bone. Subdural and epidural hematomas are usually traumatic injuries.
Intracranial hemorrhage can be classified by the site of bleeding into intracerebral hemorrhages (classified as hemorrhagic strokes), subdural hematomas, and subarachnoid hemorrhages based on imaging features from review of local interpretation of brain imaging (Figure 5).
Figure 5. Sites of intracranial hemorrhage
Figure 6. Intracerebral hemorrhage (intraparenchymal hemorrhage)
Figure 7. Subarachnoid hemorrhage
Figure 8. Subdural hemorrhage
Primary intracranial hemorrhage
Primary intracranial hemorrhage develops in the absence of any underlying vascular malformation or coagulopathy. Primary intracerebral hemorrhage is more common than secondary intracerebral hemorrhage. Hypertensive arteriosclerosis and cerebral amyloid angiopathy are responsible for 80% of primary hemorrhages 10. At times it may be difficult to identify the underlying cause because poorly controlled hypertension is often identified in most intracranial hemorrhage patients. Patients with cerebral amyloid angiopathy-related intracranial hemorrhage are more likely to be older and the volume of hemorrhage is usually > 30 cc 11. Hypertension related intracranial hemorrhage is frequently seen in younger patients, involving the basal ganglia, and the volume of blood is usually < 30 cc 12. However these characteristics are nonspecific and histopathological studies are needed to confirm a definitive diagnosis of cerebral amyloid angiopathy or hypertension related intracranial hemorrhage. Hypertension causes high pressure within the Circle of Willis resulting in smooth cell proliferation followed by smooth muscle cell death. This may explain why hypertension related intracranial hemorrhage are frequently located deep within the basal ganglia, thalamus (Figure 4 above), cerebellum, pons and rarely the neocortex 10. In contrast, preferential amyloid deposition within leptomeningeal and intraparenchymal cortical vessels may explain the reason for large superficial lobar hemorrhages with amyloid angiopathy 13. It is important to identify those afflicted with cerebral amyloid angiopathy because of the high risk of recurrent lobar hemorrhage and predisposition for symptomatic hemorrhage with anticoagulants and thrombolytics 14.
Secondary intracranial hemorrhage
Secondary intracranial hemorrhage is due to underlying vascular malformation, hemorrhagic conversion of an ischemic stroke, coagulopathy, intracranial tumor, etc. Arteriovenous malformations and cavernous malformations account for majority of underlying vascular malformations 10. An arteriovenous malformation (AVM) is usually a singular lesion composed of an abnormal direct connection between distal arteries and veins. Arteriovenous malformations account for only 2% of all intracranial hemorrhage but are associated with an 18% annual rebleed risk 15. Cavernous malformations are composed of sinusoidal vessels and are typically located in within the supratentorial white matter. The annual risk of recurrent hemorrhage is only 4.5% 16. Intracranial aneurysms usually present with subarachnoid hemorrhage but anterior communicating artery and middle cerebral artery may also have a parenchymal hemorrhagic component near the interhemispheric fissure and perisylvian region respectively 17. Embolic ischemic strokes can often demonstrate hemorrhagic conversion without significant mass effect 18. Sinus thrombosis should be suspected in patients with signs and symptoms suggestive of increased intracranial pressure and radiographic evidence of superficial cortical or bilateral symmetric hemorrhages 19. An underlying cogenial or acquired coagulopathy causing platelet or coagulation cascade dysfunction can result in intracranial hemorrhage. Cogenial disorders account for Hemophilia A, Hemophilia B, and other rare diseases. Acquired coagulopathy may be attributed to longstanding liver disease, renal disease, malignancy, or medication. Particular attention has been directed towards oral anticoagulant associated hemorrhage due to greater risk for hematoma expansion as well as increased 30 day morbidity and mortality rates 20. Metastatic tumors account for less than ten percent of intracranial hemorrhage located near the grey white junction with significant mass effect. The primary malignancy is usually melanoma, choriocarninoma, renal carcinoma, or thyroid carcinoma 16.
Subarachnoid Hemorrhage
Subarachnoid hemorrhage is bleeding in the space between your brain and the surrounding membrane (subarachnoid space).
Bleeding usually results from the rupture of an abnormal bulge in a blood vessel (aneurysm) in your brain – rupture of an intracranial aneurysm is the leading cause, representing up to 85% of cases. Sometimes bleeding is caused by trauma, an abnormal tangle of blood vessels in your brain (arteriovenous malformation), or other blood vessel or health problems.
The overall incidence of subarachnoid hemorrhage is between 9–20 per 100,000 person-years. Subarachnoid hemorrhage is more frequent in women, and the mean age of presentation is 55 years 21. In the U.S. the number of cases of subarachnoid hemorrhage is 30,000 per year 22.
Untreated, a subarachnoid hemorrhage can lead to permanent brain damage or death.
The primary symptom is a sudden, severe headache. This headache is commonly described as being “the worst headache of my life” and with the highest intensity at onset 23. The headache is sometimes associated with nausea, vomiting and a brief loss of consciousness. Although it is frequently accompanied by other symptoms, headache can be the only complaint in up to 40% of patients 24. Recently, a prospective study found that the following clinical characteristics represent the highest risk of belonging to a case of subarachnoid hemorrhage: age>40 years, associated neck pain or stiffness, witnessed loss of consciousness, onset with exertion, vomiting, arrival by ambulance, and blood pressure above 160/100 25.
A subgroup of patients develops “warning signs” before the index subarachnoid hemorrhage. The most common warning sign is again headache, which is of moderate intensity and less severe than those described in subarachnoid hemorrhage. This is commonly referred to as “sentinel headache” or “warning leak” and may be associated with a small leakage of blood into the subarachnoid space or a small bleed into the aneurysmal wall. A thorough evaluation is warranted in these cases, since an subarachnoid hemorrhage can develop up to 110 days later 26.
Risk factors for subarachnoid hemorrhage
Major risk factors associated with subarachnoid hemorrhage are current and former history of smoking, hypertension and excessive alcohol intake 27. Although one third of cases can be attributed to a current smoking status, this risk appears to rapidly disappear after a few years of smoking cessation 28. Cocaine use is also associated with subarachnoid hemorrhage, and these patients tend to be younger and have a worse outcome 29. First degree family history as well as some genetic conditions including autosomal dominant polycystic kidney disease, Marfan’s syndrome and Ehlers-Danlos syndrome are also associated with an increased risk of subarachnoid hemorrhage 30.
A recent meta-analysis reported that in a population without comorbidities, the prevalence of unruptured intracranial aneurysms is 3.2% 31. Only a small percentage of these unruptured intracranial aneurysms will rupture and cause an subarachnoid hemorrhage. The risk of rupture is increased in cases of previous history of subarachnoid hemorrhage, age older than 60, female gender and Japanese or Finnish descent. In addition, the risk is greater for aneurysms >10mm and those located in the posterior circulation 32.
Subarachnoid hemorrhage diagnosis
To diagnose a subarachnoid hemorrhage, your doctor is likely to recommend:
- CT scan. This imaging test can detect bleeding in your brain.Your doctor may inject a contrast dye to view your blood vessels in greater detail (CT angiogram).
- MRI. This imaging test also can detect bleeding in your brain. Your doctor might inject a dye into a blood vessel to view the arteries and veins in greater detail (MR angiogram) and to highlight blood flow.
- Cerebral angiography. You doctor inserts a long, thin tube (catheter) into an artery in your leg and threads it to your brain. Dye is injected into the blood vessels of your brain to make them visible under X-ray imaging. Your doctor might recommend cerebral angiography to obtain more-detailed images or if a subarachnoid hemorrhage is suspected but the cause isn’t clear or doesn’t appear on other imaging.
Up to 22 percent of aneurysmal subarachnoid hemorrhages don’t appear on initial imaging tests. If your initial tests don’t show bleeding, your doctor might recommend:
- Lumbar puncture. Your doctor inserts a needle into your lower back to withdraw a small amount of fluid that surrounds your brain and spinal cord (cerebrospinal fluid). The fluid is examined for the presence of blood, which can indicate a subarachnoid hemorrhage.
- Repeated imaging. The tests might be repeated several days after the initial testing.
Subarachnoid hemorrhage treatment
Treatment initially focuses on stabilizing your condition.
If your bleeding is caused by a ruptured brain aneurysm, your doctor might recommend:
- Surgery. The surgeon makes an incision in your scalp and locates the brain aneurysm. A metal clip is placed across the aneurysm’s neck to stop the blood flow to it.
- Endovascular embolization. The surgeon inserts a catheter into an artery in your groin and threads it to your brain. Detachable platinum coils are guided through the catheter and placed in the aneurysm. The coils fill the aneurysm, reducing blood flow into the aneurysm sac and causing the blood to clot.
- Other endovascular treatments. Certain aneurysms can be treated with endovascular embolization that uses newer technology such as stent-assisted or balloon-assisted coiling or devices that divert blood flow.
Patients with subarachnoid hemorrhage require emergency neurosurgical and/or endovascular consultation. There are currently at least two options for the acute treatment of a ruptured aneurysm: endovascular coiling or surgical clipping. Treatment of a recently ruptured aneurysm reduces the rate of rebleeding, and the benefit is related to the time to treatment initiation 33. Current guidelines recommend that surgical clipping or endovascular coiling should be performed to reduce the rate of rebleeding after aneurysmal subarachnoid hemorrhage, and these procedures should be performed early in the disease course 34.
The selection of the most appropriate intervention depends on a range of characteristics including age, clinical status and medical comorbidities. Aneurysm characteristics, such as location, shape, and size, are taken into consideration as well, which highlights the value of a specialized multidisciplinary group to provide care and decision-making. Some expert consensus groups recommend that subarachnoid hemorrhage be preferentially managed at high volume centers (defined as those centers with greater than 60 cases of subarachnoid hemorrhage per year) 34.
Endovascular procedures sometimes need to be performed again. You’ll have regular follow-up appointments with your doctor to watch for any changes. You may also need physical, occupational and speech therapy.
Intracranial hemorrhage causes
Primary intracranial hemorrhage is typically a manifestation of underlying small vessel disease. First, longstanding hypertension leads to hypertensive vasculopathy causing microscopic degenerative changes in the walls of small-to-medium penetrating vessels, which is known as lipohyalinosis 35. Second, cerebral amyloid angiopathy is characterized by the deposition of amyloid-beta peptide (Aβ) in the walls of small leptomeningeal and cortical vessels 36. Although the underlying mechanism leading to the accumulation of amyloid is still unknown, the final consequences are degenerative changes in the vessel wall characterized by the loss of smooth muscle cells, wall thickening, luminal narrowing, microaneurysm formation and microhemorrhages 37.
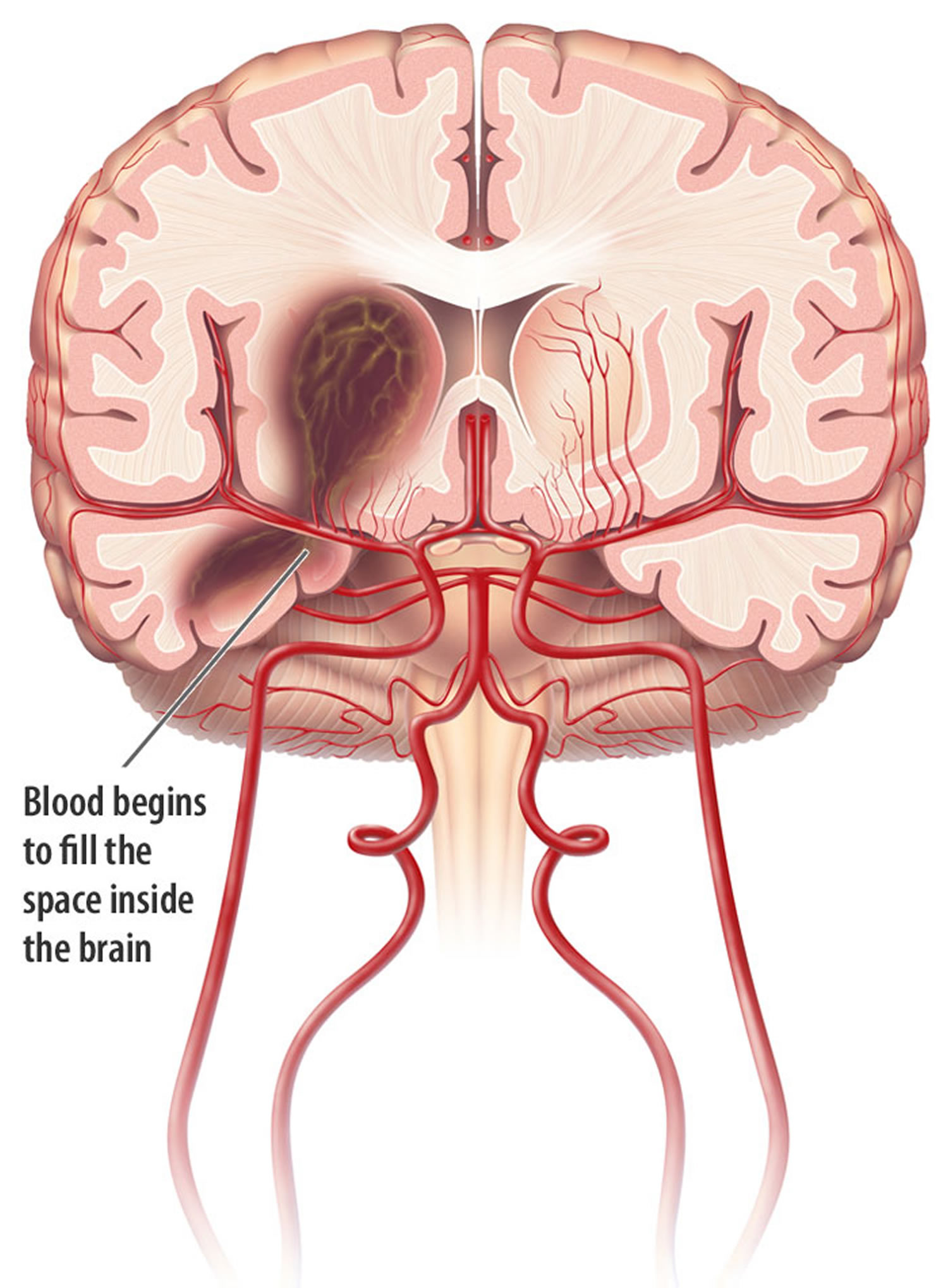
Following initial vessel rupture, the hematoma causes direct mechanical injury to the brain parenchyma. Perihematomal edema develops within the first 3 hours from symptom onset and peaks between 10 to 20 days 38. Next, blood and plasma products mediate secondary injury processes including an inflammatory response, activation of the coagulation cascade, and iron deposition from hemoglobin degradation 38. Finally, the hematoma can continue to expand in up to 38 percent of patients during the first 24 hours 39.
Risk factors for intracranial hemorrhage
A recent population-based meta-analysis showed that risk factors for intracranial hemorrhage include male sex, older age, and Asian ethnicity 3. Intracranial hemorrhage is twice as frequent in low-to-middle income countries compared to high-income countries 40. In the United States, several studies have shown that the incidence of intracranial hemorrhage is greater in African Americans and Hispanics than in whites 41.
The most important risk factors for intracranial hemorrhage include hypertension (high blood pressure) and cerebral amyloid angiopathy. Hypertension-related intracranial hemorrhage is more likely to occur in deep structures 42 and the risk of intracranial hemorrhage increases with increasing blood pressure values 43. Cerebral amyloid angiopathy tends to occur in association with advanced age, and cerebral amyloid angiopathy-related intracranial hemorrhage tends to occur in lobar regions 44.
Other risk factors for intracranial hemorrhage include:
- a) Alcohol intake: This risk appears to be dose-dependent, with a higher risk of intracranial hemorrhage among those with a higher daily alcohol intake 43. Acute changes in blood pressure during ingestion and withdrawal, effects on platelet function and coagulation, and dysfunction of the vascular endothelium may account for this risk 45.
- b) Cholesterol: Low levels of total serum cholesterol are risk factors for intracranial hemorrhage (in contrast to ischemic stroke, for which high cholesterol levels are a risk) 46.
- c) Genetics: The gene most strongly associated with intracranial hemorrhage is the Apolipoprotein E (APOE) gene and its ε2 and ε4 alleles 47. The presence of the ε2 allele was recently also linked to hematoma expansion 48.
- d) Anticoagulation: Oral anticoagulants are widely used as prophylaxis in patients with atrial fibrillation and other cardiovascular and prothrombotic states. The annual risk of intracranial hemorrhage in patients taking warfarin ranges from 0.3 to 1.0% per patient-year with a significantly increased risk when the INR is >3.5 49.
- e) Drug abuse: Sympathomimetic drugs, such as cocaine, are risk factors for intracranial hemorrhage, and patients actively using cocaine at the time of their intracranial hemorrhage have significantly more severe presentations and worse outcomes 50.
Intracranial hemorrhage symptoms
The classic presentation of intracranial hemorrhage is sudden onset of a focal or generalized neurological deficit that progresses over minutes to hours with accompanying headache, nausea, vomiting, decreased consciousness, and elevated blood pressure. The acute presentation of intracranial hemorrhage can be difficult to distinguish from ischemic stroke. Additional symptom may include seizures. Findings such as coma, headache, vomiting, seizures, neck stiffness and raised diastolic blood pressure increase the likelihood of intracranial hemorrhage compared to ischemic stroke, but only neuroimaging can provide a definitive diagnosis 51.
Neurologic deficits are related to the site of parenchymal hemorrhage. Thus, ataxia is the initial deficit noted in cerebellar hemorrhage, whereas weakness may be the initial symptom with a basal ganglia hemorrhage. Early progression of neurologic deficits and decreased level of consciousness can be expected in 50% of patients with intracranial hemorrhage. The progression of neurological deficits in many patients with an intracranial hemorrhage is frequently due to ongoing bleeding and enlargement of the hematoma during the first few hours. Compared with patients with ischemic stroke, headache and vomiting at onset of symptoms is observed three times more often in patients with intracranial hemorrhage 52. Despite the differences in clinical presentation between hemorrhagic and ischemic strokes, brain imaging is required to definitively diagnose intracerebral hemorrhage.
Intracranial hemorrhage diagnosis
Noncontrast computerized tomography
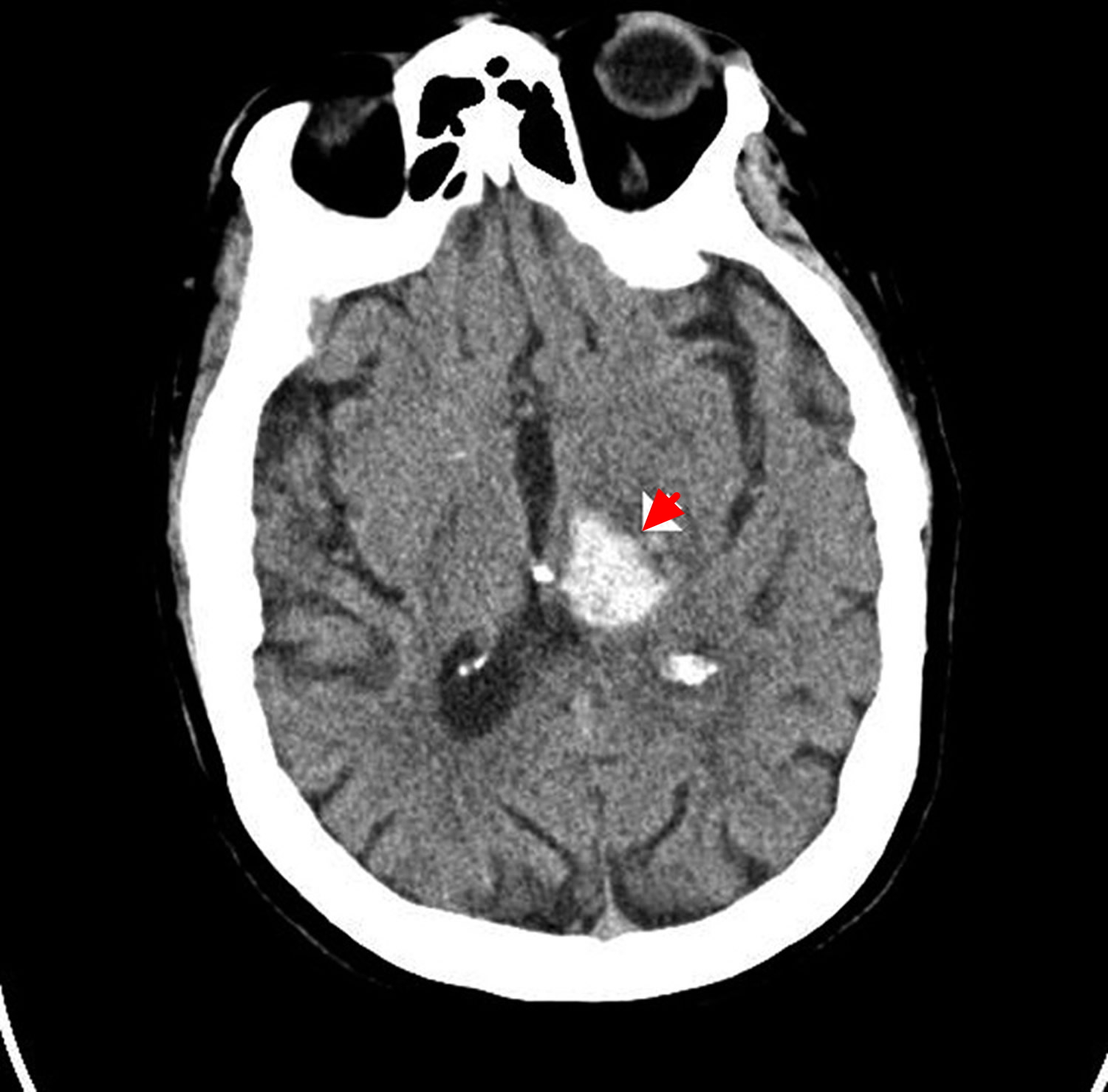
Noncontrast computerized tomography (CT) is the most rapid and readily available tool for the diagnosis of intracranial hemorrhage 53 and remains the most commonly used technique in the emergency department. Besides providing the definitive diagnosis, CT may also show basic characteristics of the hematoma, such as: hematoma location, extension to the ventricular system, presence of surrounding edema, development of mass effect and midline shift.
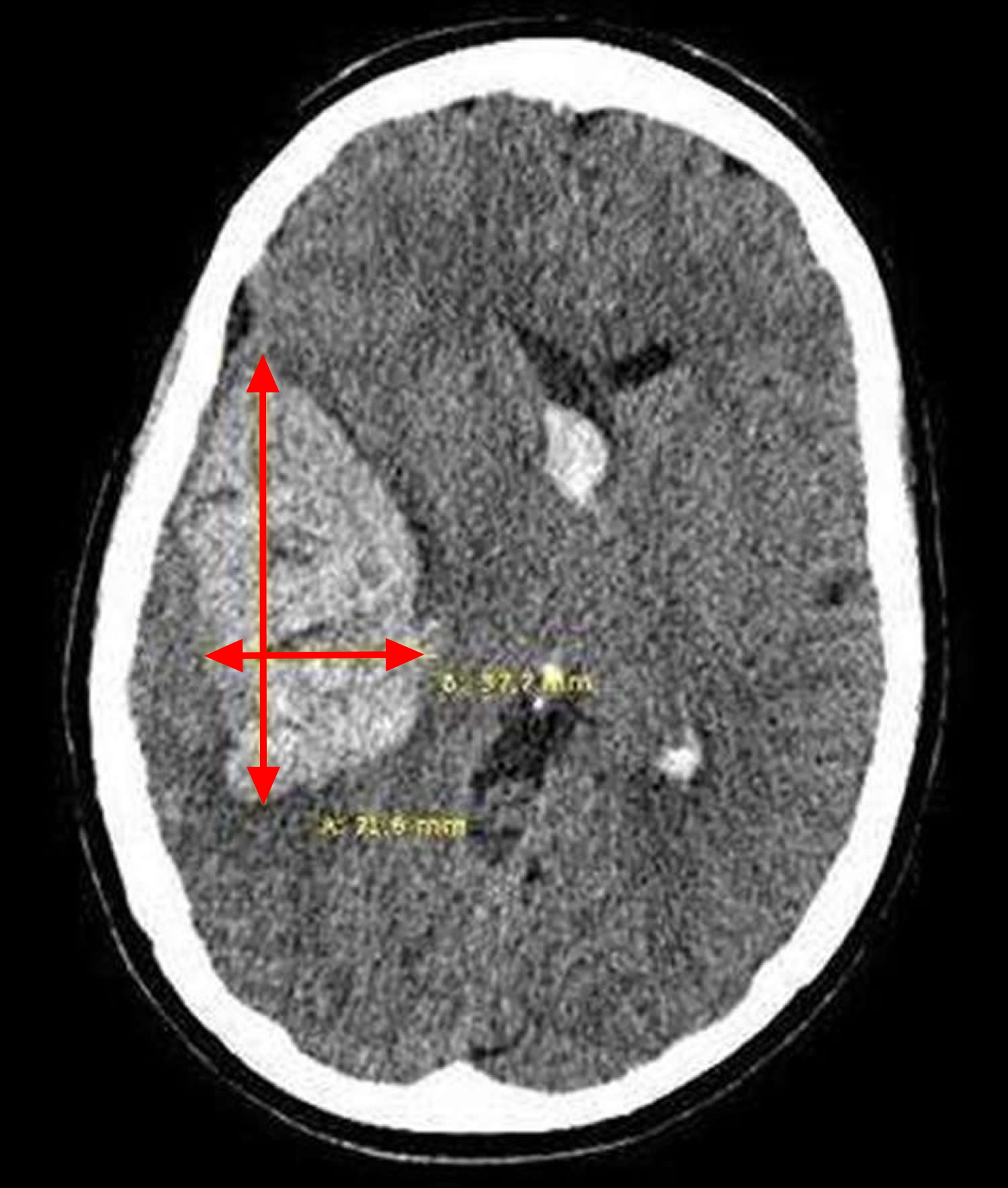
A quick estimation of the hematoma volume can be rapidly performed in the emergency department with the validated ABC/2 technique (Figure 9) 54. The steps to follow using this technique are:
The CT slice with the largest area of hemorrhage is selected.
- A is the largest hemorrhage diameter on the selected slice (in centimeters [cm]).
- B is the largest diameter perpendicular to A on the same slice.
- C is the approximate number of slices in which the hemorrhage is seen multiplied by the slice thickness (often 0.5cm slices).
- A, B, and C are then multiplied and the product divided by 2 [A x B x C/2].
Figure 9. Intracranial hemorrhage
CT Angiography
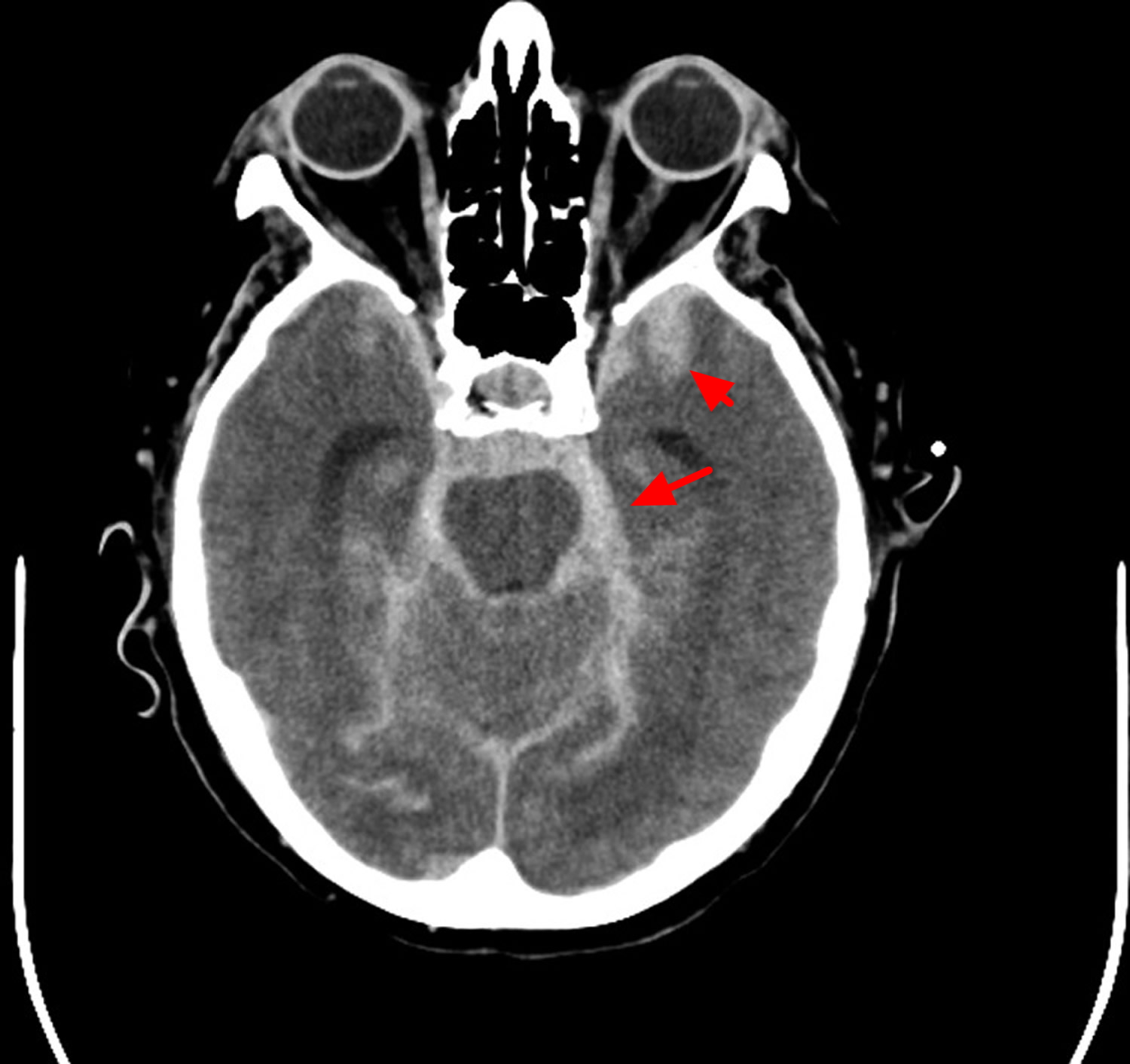
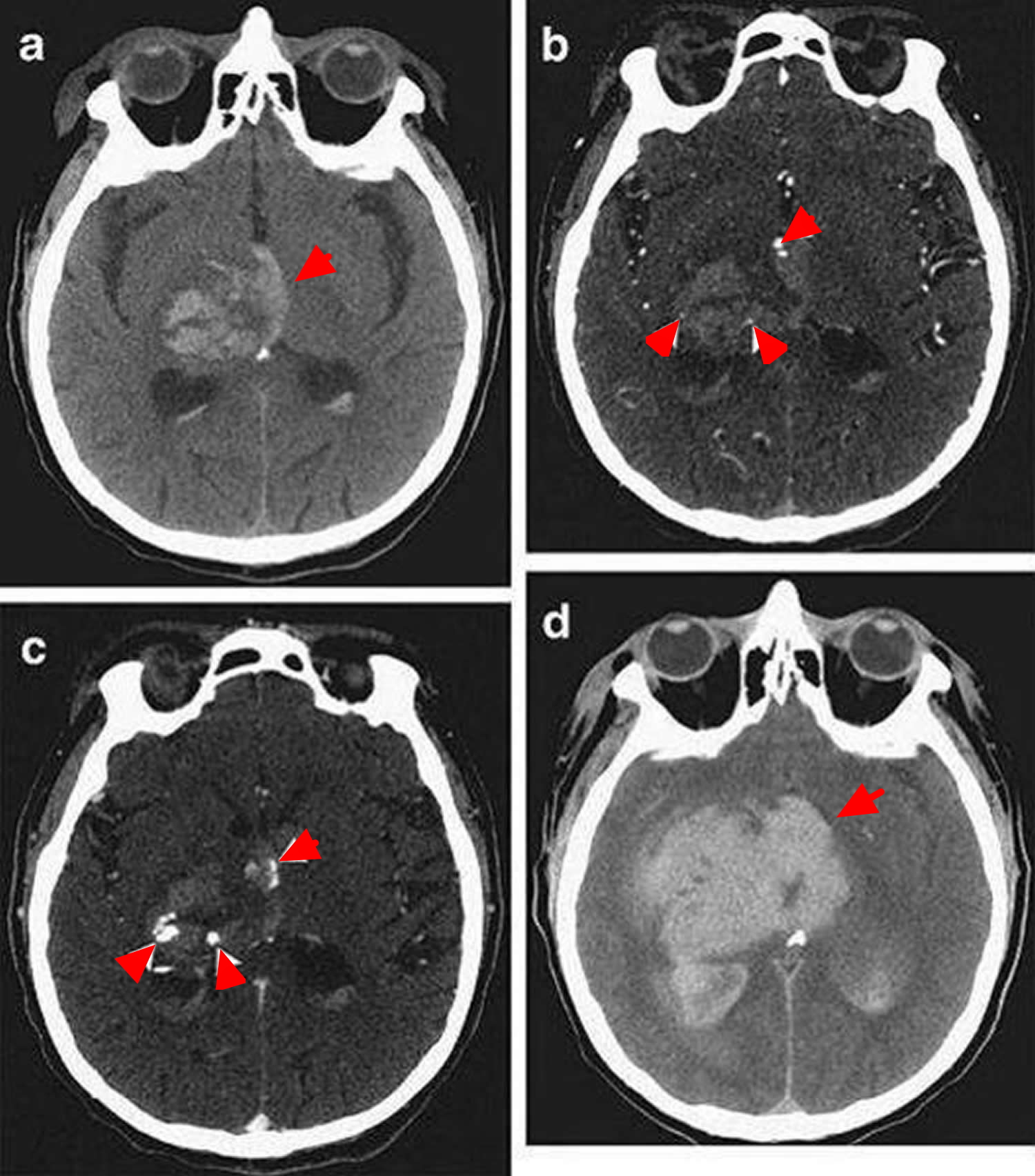
CT Angiography (CTA) is gaining increasing acceptance as a diagnostic tool in the acute setting 55. It is the most widely available, non-invasive technique for ruling out vascular abnormalities as secondary causes of intracranial hemorrhage. The risk of acute nephropathy, if any, is likely quite low 56. Up to 15% of patients with intracranial hemorrhage will show an underlying vascular etiology on CT angiography, potentially changing acute management 57. Finally, contrast extravasation seen on CT angiography images, also known as a “spot sign,” (Figure 5) is thought to represent ongoing bleeding and appears to mark those patients at highest risk of hematoma expansion, poor outcome and mortality 58.
Figure 10. Intracranial hemorrhage (CT angiography)
Note: Computed Tomography (CT) and CT angiography of acute intracerebral hemorrhage. (A) Noncontrast CT shows a right thalamic intracerebral hemorrhage (24mL) with associated intraventricular hemorrhage (6mL). (B) CT angriography demonstrates 3 foci of contrast (spot signs) within the intracerebral hemorrhage (arrowheads) (C). Delayed CT angiography shows increased volume and changed morphology of the spot signs (arrowheads). (D) Noncontrast CT after 8 hours demonstrates expansion of the intracerebral hemorrhage (94ml) and intraventricular hemorrhage (82 mL).
Magnetic resonance imaging (MRI)
magnetic resonance imaging (MRI) is equivalent to CT for the detection of acute intracranial hemorrhage 59. The imaging characteristics of intracranial hemorrhage vary with time as the hemoglobin passes through different stages during the pathological process. In the acute phase, gradient recalled-echo (GRE) imaging techniques with T2*weighting are the best option to detect the presence of intracranial hemorrhage 60. MRI can also detect underlying secondary causes of intracranial hemorrhage such as tumor and hemorrhagic transformation of ischemic stroke. Finally, for patients with poor kidney function or contrast allergies, the cerebral vasculature can be analyzed without contrast using Time-of-Flight MR angiography (MRA) 61.
Intracranial hemorrhage treatment
Acute management
Airway
Patients with intracranial hemorrhage are often unable to protect the airway. Endotracheal intubation may be necessary but this decision should be balanced against the risk of losing the neurologic examination. Rapid sequence intubation is typically the preferred approach in the acute setting. Pretreatment with lidocaine may be considered as it may blunt a rise in intracranial pressure (ICP) associated with intubation. Paralytic agents include succinylcholine, rocuronium or vecuronium, and for postintubation sedation, propofol is a reasonable choice given its short half-life 62.
Blood Pressure management
Elevated blood pressure (BP) is common in the acute setting after an intracranial hemorrhage, and higher blood pressure levels are associated with hematoma expansion and poor prognosis. However, it is not clear that reducing blood pressure improves outcomes 63. While lowering blood pressure may reduce the risk of expansion, it may theoretically also reduce cerebral perfusion. One randomized clinical trial found that lowering systolic blood pressure to 140mmHg compared to 180mmHg reduced the risk of hematoma expansion but had no effect on outcomes 64. A second trial found that rapid blood pressure lowering using intravenous Nicardipine appears safe but again showed no difference in outcomes 65. Multiple clinical trials are currently ongoing to address this issue 66.
Until these trials clarify the role of blood pressure management on hematoma expansion, expert guidelines from the American Heart Association/American Stroke Association recommend blood pressure treatment 67. The European Stroke Initiative guidelines are similar 68.
Recommended Guidelines from the American Heart Association and American Stroke Association for Treating Elevated BP in Spontaneous intracranial hemorrhage
- If systolic blood pressure is >200 mm Hg or mean arterial pressure is >150 mm Hg, then consider aggressive reduction of blood pressure with continuous intravenous infusion, with frequent blood pressure monitoring every 5 min.
- If systolic blood pressure is >180 mm Hg or mean arterial pressure is >130 mm Hg and there is the possibility of elevated intracranial pressure, then consider monitoring intracranial pressure and reducing blood pressure using intermittent or continuous intravenous medications while maintaining a cerebral perfusion pressure ≥60 mm Hg.
- If systolic blood pressure is >180 mm Hg or mean arterial pressure is >130 mm Hg and there is no evidence of elevated intracranial pressure, then consider a modest reduction of blood pressure (e.g., mean arterial pressure of 110 mm Hg or target blood pressure of 160/90 mm Hg) using intermittent or continuous intravenous medications to control blood pressure and clinically reexamine the patient every 15 min.
Recommendations from European Stroke Initiative for blood pressure management in intracranial hemorrhage
- Previous history of hypertension:
- Gradually reduce mean arterial pressure to <120 but >84 mm Hg; avoid a reduction of >20%.
- Blood pressure limit is <180/105 mm Hg, if treatment is necessary target should be <160/100 mm Hg.
- No history of hypertension:
- Reduce mean arterial pressure to 110 mm Hg.
- Blood pressure limit is <160/95 mm Hg, if treatment is necessary target should be <150/90 mm Hg.
- When increased intracranial pressure is present:
- Adapt mean arterial pressure and blood pressure limits to target a cerebral perfusion pressure of 60–70 mm Hg.
In choosing medications to manage hypertension, intravenous antihypertensives with short half-lives should be considered as first-line therapy. The American Heart Association recommends considering IV labetalol, nicardipine, esmolol, enalapril, hydralazine, sodium nitroprusside, or nitroglycerin 63. The European Stroke Initiative recommends IV labetalol, urapidil, sodium nitroprusside, nitroglycerin, or captopril 69.
Hemostatic therapy
It is tempting to consider that in a patient with intracranial hemorrhage, acute hemostatic therapy will provide benefit. One phase III randomized trial in patients with no underlying coagulopathy found no clinical benefit from this approach 70 As a result, current approaches to hemostasis are focused on correcting any underlying coagulopathies.
Oral anticoagulation
The most common class of agent used for oral anticoagulation is warfarin. Many authors believe that early action to rapidly correct the coagulopathy may prevent continued bleeding 71. A number of therapeutic options are available for warfarin reversal.
As warfarin inhibits the vitamin K-dependent carboxylation of factors II, VII, IX, and X, vitamin K is a first line agent to restore these factors. Vitamin K given intravenously lowers the INR as early as 4 hours, but requires over 24 hours for full effect when used as monotherapy 72. Vitamin K infusion at a dose of 5–10 mg should be started promptly and given slowly over 30 minutes 73.
While awaiting the effect of intravenous vitamin K, coagulation factors should be infused emergently. Fresh Frozen Plasma (FFP) contains all coagulation factors and is the most widely available and commonly used agent in the United States 74. Limitations include adverse events such as allergic reactions, potential transmission of infectious agents and transfusion-related acute lung injury 75. There is also significant time needed for its administration in actual practice, including time spent ordering, matching, thawing and delivering to the emergency department 74. The dose of FFP ranges from 10 to 20 ml/kg of body weight. On average, the volume needed to correct the INR varies from 800–3500 ml, which may impose a significant volume load 76. Early administration of coagulation factors maximizes the opportunity for early INR correction 77.
Prothrombin Complex Concentrates (PCCs) provide an alternate source of coagulation factors. Prothrombin Complex Concentrates (PCCs) contain coagulation factors prepared from pooled plasma. All available prothrombin complex concentrates (PCCs) contain factors II, IX, and X, and some contain relevant amounts of factor VII and proteins C and S. In the United States there are currently two commercially available prothrombin complex concentrates (PCCs), Bebulin-VH Factor IX complex (Baxter, Westlake, CA) and Profilnine-SD (GrifolsBiologicals, Los Angeles, CA) 78. Many other products are available in other countries, including Octaplex (Octapharma) and Beriplex (CSL Behring), which include clinically relevant amounts of all 4 vitamin K dependent factors, sometimes termed 4-factor PCCs to differentiate them from the other 3 factor prothrombin complex concentrates 79. Prothrombin complex concentrate offers several advantages over FFP, including smaller infusion volume, faster time to INR correction, and lack of need for blood-type matching 80. Thromboembolic (TE) events are potential complications of the use of prothrombin complex concentrate, although it is not clear that this risk (approximately 1.9%) is any different with FFP 81.
Heparinoids
Heparin-related intracranial hemorrhage is relatively rare, and data is sparse regarding appropriate treatment. One reasonable approach would be to reverse heparin with IV protamine sulfate at a dose of 1 to 1.5 mg per 100 units of heparin with a maximum dose of 50 mg 82.
Platelet function
The two major causes of platelet dysfunction are antiplatelet therapy and thrombocytopenia.
Antiplatelet agents use prior to an intracranial hemorrhage is associated with a small increase in mortality, suggesting an opportunity for intervention 83. The utility and safety of platelet transfusion in such patients is unknown, although some laboratory data suggest that such transfusions may improve platelet activity 84. Platelet transfusion is therefore considered investigational by the AHA and is not recommended by the European Stroke Initiative. The ongoing PATCH clinical trial will investigate whether platelet transfusions can improve outcome 85.
Additionally, it is not clear whether low platelets contribute to ongoing bleeding or worse outcome. Pending further data, current American Heart Association recommendations are that patients with a severe thrombocytopenia should receive platelet transfusion 63. A specific cutoff is not clarified; different groups use thresholds between 10,000 and 50,000 per microliter.
Novel antithrombotics
Recently, a number of new agents, such as factor Xa inhibitors Apixaban and Rivaroxaban and the direct thrombin inhibitor Dabigatran, have become available for stroke prevention 86. There is no currently known antidote for reversal of these agents. Specific hemostatic agents such as rFVIIa and prothrombin complex concentrates may be considered, though there is limited data on their use. For those cases related to Dabigatran use, a recently published expert recommendation states that the drug should be stopped immediately, supportive and symptomatic treatment should be initiated and, due to its renal excretion, aggressive diuresis and potential dialysis could be considered 87.
Intracranial pressure management
An increase in the intracranial pressure (ICP) may arise from the presence of intraventricular hemorrhage (IVH) and subsequent hydrocephalus, or from mass effect from a large hematoma or perihematomal edema. Currently, there are limited data regarding indications for ICP monitoring. Current guidelines from the American Heart Association and American Stroke Association suggest that patients with a GCS (Glasgow Coma Score) score of ≤8, those with clinical evidence of transtentorial herniation, or those with significant IVH or hydrocephalus should be considered for ICP monitoring and treatment 63. Cerebral perfusion pressure (CPP) can then be monitored, and recommendations are to maintain this between 50 to 70 mmHg 57.
The initial management of elevated intracranial pressure (ICP) should comprise simple measures such as elevation of the head of the bed, analgesia, and sedation. Medical options for intracranial pressure (ICP) treatment include mannitol, hypertonic saline (ranging from 3% to 23.4%), and neuromuscular paralysis 4. Barbiturates can be considered in refractory intracranial hypertension 62. Although hyperventilation can produce a rapid decrease in the ICP, its effects are temporary, and its use should be reserved for impending herniation while awaiting surgical decompression.
Hyperglycemia management
Hyperglycemia measured at arrival in the emergency department is associated with worse outcome in both non-diabetic and diabetic patients 88. Declining glucose values after intracranial hemorrhage are associated with a decreased risk of hematoma expansion and poor outcome, suggesting that early glucose control may improve outcomes 71. Early evidence for this intervention comes from the QASC trial, in which patients with intracranial hemorrhage and ischemic stroke were randomized to receive fever, hyperglycemia, and swallow screening, or not 89. The intervention (including glucose management) lowered mortality and improved outcome. This highlights the need for careful glycemic control in the early phase 63.
Temperature
The presence of fever is a common finding in patients with intracranial hemorrhage, especially in those with intraventricular hemorrhage. Again, data from the QASC trial suggests lower mortality and improved outcome in those patients receiving fever control as part of a multidisciplinary approach. Those with fever should undergo a thorough investigation to find a fever source if possible 63.
Anemia
The presence of anemia is common in patients with intracranial hemorrhage. It is present in up to 25% of cases at admission and is associated with larger hematoma volumes 90. It also frequently develops during hospital stay 91. Although current guidelines do not address this issue, a recent study found that packed red blood cell transfusion in these patients was associated with improved survival at 30 days 91. Therefore, transfusion can be considered in such patients, although the ideal target hemoglobin level has not been determined.
Antiepileptics
Patients with intracranial hemorrhage are at an increased risk of developing seizures; however, most of these events are subclinical electroencephalographic findings. Seizures are more common in lobar intracranial hemorrhage and during the first 72 hours after admission 92. The majority of patients develop a single episode of seizure during hospitalization, suggesting that those episodes are related to the pathophysiological processes that occur early after an intracranial hemorrhage 93. The use of prophylactic antiepileptic drugs in patients with intracranial hemorrhage is a common practice, although it is not clear that the presence of seizures and/or the use of prophylactic antiepileptic drugs affect short or long term outcome 94. Some studies have in fact reported an association between antiepileptic drugs and worse outcome, although these patients were disproportionately exposed to phenytoin as the antiepileptic drug of choice 95.
Currently, the American Heart Association and American Stroke Association recommends that antiepileptic drugs should not be used routinely in patients with intracranial hemorrhage. The only clear indications are the presence of clinical seizures or electrographic seizures in patients with a change in mental status. They also suggest that the use of continuous electroencephalography (EEG) monitoring should be considered in those patients with depressed mental status out of proportion to the degree of brain injury 63.
Surgical interventions
External Ventricular Drain Placement
As described previously, some patients may benefit from ICP monitoring. External ventricular drain (EVD) placement not only provides the ability to monitor ICP but has the advantage of allowing therapeutic drainage of the CSF, which is valuable in patients with hydrocephalus 96. The American Heart Association recommends that ICP monitoring and treatment be considered in patients with a GCS score ≤ 8, those with clinical evidence of transtentorial herniation, or those with significant intraventricular hemorrhage or hydrocephalus 97. The European Stroke Initiative recommends considering continuous ICP monitoring in patients who need mechanical ventilation and recommend medical treatment of elevated ICP if clinical deterioration is related to increasing edema.
Intraventricular thrombolysis
Intraventricular hemorrhage occurs when intracranial hemorrhage extends into the ventricles. It occurs in approximately 45% of intracranial hemorrhage, more frequently in relatively large and deeply located (caudate nucleus and thalamus) hemorrhages 98. The presence and the volume of intraventricular hemorrhage are correlated with poor prognosis in patients with intracranial hemorrhage 99. Although evacuation of an intraventricular clot is currently not routinely recommended, a recent study comparing the use of intraventricular rtPA to placebo showed that the use of rtPA was not only feasible and safe, but also showed a significantly greater rate of blood clot resolution 100. In addition, a recent meta-analysis found that adding intraventricular fibrinolysis to EVD placement is associated with better functional outcome 101, although no prospective randomized trial has evaluated this. The CLEAR III study, an ongoing phase III randomized clinical trial was designed to compare the effect on clinical outcome of the intraventricular use of rtPA compared to placebo.
Hematoma evacuation
The role of surgical evacuation is to decrease mass effect related to the presence of blood, as well as to minimize secondary injury. The only clear recommendation for immediate surgical intervention is in patients with cerebellar hemorrhages with neurological deterioration, brainstem compression, and/or hydrocephalus from ventricular obstruction 97. For these patients, emergency neurosurgical consultation should be obtained. However, it is less clear whether patients with supratentorial intracranial hemorrhage will benefit. One large phase III clinical trial, the Surgical Trial intracranial hemorrhage trial, compared early hematoma evacuation with initial conservative treatment for patients with spontaneous supratentorial intracranial hemorrhage 102. This study showed no difference in outcome, suggesting that surgical evacuation provided no benefit. However, a subsequent subgroup analysis raised the possibility that those with hematomas ≤1 cm from the cortical surface (which are more easily accessible) might receive benefit 103. This possibility is being evaluated in the ongoing Surgical Trial intracranial hemorrhage II trial 104. The theoretical idea that hyperacute evacuation of the hematoma would be beneficial was not borne out when a study evaluating the effect of surgery within 4 hours was stopped due to a high rate of rebleeding 105.
Minimally invasive surgery
The development of less invasive surgical techniques may decrease the risk of surgical complications. These techniques are showing promising results, particularly in deep hemorrhages where conventional surgery showed no benefit in the past 106. Minimally invasive stereotactic puncture is reported to be safe and feasible and may lead to better long-term outcome and fewer complications when compared with conventional craniotomy 107 and conventional medical treatment 108.
- A prospective multicenter study to evaluate the feasibility and safety of aggressive antihypertensive treatment in patients with acute intracerebral hemorrhage. Qureshi AI, Mohammad YM, Yahia AM, Suarez JI, Siddiqui AM, Kirmani JF, Suri MF, Kolb J, Zaidat OO. J Intensive Care Med. 2005 Jan-Feb; 20(1):34-42. https://www.ncbi.nlm.nih.gov/pubmed/15665258[↩]
- Caceres JA, Goldstein JN. Intracranial Hemorrhage. Emergency medicine clinics of North America. 2012;30(3):771-794. doi:10.1016/j.emc.2012.06.003. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3443867/[↩][↩][↩]
- van Asch CJJ, et al. Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: a systematic review and meta-analysis. The Lancet Neurology. 2010;9(2):167–176 https://www.ncbi.nlm.nih.gov/pubmed/20056489[↩][↩]
- Broderick J, et al. Guidelines for the Management of Spontaneous Intracerebral Hemorrhage in Adults. Stroke. 2007;38(6):2001–2023 http://stroke.ahajournals.org/content/38/6/2001.long[↩][↩]
- Elliott J, Smith M. The Acute Management of Intracerebral Hemorrhage: A Clinical Review. Anesthesia & Analgesia. 2010;110(5):1419–1427 https://www.ncbi.nlm.nih.gov/pubmed/20332192[↩]
- Flaherty ML, Haverbusch M, Sekar P, Kissela B, Kleindorfer D, Moomaw CJ, Sauerbeck L, Schneider A, Broderick JP, Woo D. Long-term mortality after intracerebral hemorrhage. Neurology. 2006; 66: 1182–1186[↩]
- Counsell C, Boonyakarnkul S, Dennis M, Sandercock P, Bamford J, Burn J, Warlow C. Primary intracerebral haemorrhage in the Oxfordshire Community Stroke Project, 2: prognosis. Cerebrovasc Dis. 1995; 5: 26–34.[↩]
- Hemphill JC, 3rd, et al. The ICH score: a simple, reliable grading scale for intracerebral hemorrhage. Stroke. 2001;32(4):891–7. http://stroke.ahajournals.org/content/32/4/891.long[↩]
- Rost NS, et al. Prediction of functional outcome in patients with primary intracerebral hemorrhage: the FUNC score. Stroke. 2008;39(8):2304–9. http://stroke.ahajournals.org/content/39/8/2304.long[↩]
- Primary intracerebral hemorrhage. Sutherland GR, Auer RN. J Clin Neurosci. 2006 Jun; 13(5):511-7. https://www.ncbi.nlm.nih.gov/pubmed/16769513/[↩][↩][↩]
- Role of cerebral amyloid angiopathy in intracerebral hemorrhage in hypertensive patients. Ritter MA, Droste DW, Hegedüs K, Szepesi R, Nabavi DG, Csiba L, Ringelstein EB. Neurology. 2005 Apr 12; 64(7):1233-7. https://www.ncbi.nlm.nih.gov/pubmed/15824353/[↩]
- Stroke pattern interpretation: the variability of hypertensive versus amyloid angiopathy hemorrhage. Lang EW, Ren Ya Z, Preul C, Hugo HH, Hempelmann RG, Buhl R, Barth H, Klinge H, Mehdorn HM. Cerebrovasc Dis. 2001 Aug; 12(2):121-30. https://www.ncbi.nlm.nih.gov/pubmed/11490106/[↩]
- Primary intracerebral hemorrhage: pathophysiology. Auer RN, Sutherland GR. Can J Neurol Sci. 2005 Dec; 32 Suppl 2():S3-12. https://www.ncbi.nlm.nih.gov/pubmed/16450803/[↩]
- Rosand J, Greenberg SM. Cerebral amyloid angiopathy. Neurologist. 2000;6:315–25[↩]
- A systematic review of the frequency and prognosis of arteriovenous malformations of the brain in adults. Al-Shahi R, Warlow C. Brain. 2001 Oct; 124(Pt 10):1900-26. https://www.ncbi.nlm.nih.gov/pubmed/11571210/[↩]
- The natural history of cerebral cavernous malformations. Kondziolka D, Lunsford LD, Kestle JR. J Neurosurg. 1995 Nov; 83(5):820-4. https://www.ncbi.nlm.nih.gov/pubmed/7472549/[↩][↩]
- Multislice computerized tomography angiography in the evaluation of intracranial aneurysms: a comparison with intraarterial digital subtraction angiography. Wintermark M, Uske A, Chalaron M, Regli L, Maeder P, Meuli R, Schnyder P, Binaghi S. J Neurosurg. 2003 Apr; 98(4):828-36. https://www.ncbi.nlm.nih.gov/pubmed/12691409/[↩]
- The clinical spectrum of hemorrhagic infarction. Ott BR, Zamani A, Kleefield J, Funkenstein HH. Stroke. 1986 Jul-Aug; 17(4):630-7. http://stroke.ahajournals.org/content/17/4/630.long[↩]
- Causes and predictors of death in cerebral venous thrombosis. Canhão P, Ferro JM, Lindgren AG, Bousser MG, Stam J, Barinagarrementeria F, ISCVT Investigators. Stroke. 2005 Aug; 36(8):1720-5. http://stroke.ahajournals.org/content/36/8/1720.long[↩]
- Intracerebral hemorrhage associated with oral anticoagulant therapy: current practices and unresolved questions. Steiner T, Rosand J, Diringer M. Stroke. 2006 Jan; 37(1):256-62. http://stroke.ahajournals.org/content/37/1/256.long[↩]
- Subarachnoid haemorrhage: diagnosis, causes and management. van Gijn J, Rinkel GJ. Brain. 2001 Feb; 124(Pt 2):249-78. https://www.ncbi.nlm.nih.gov/pubmed/11157554/[↩]
- Zacharia BE, et al. Epidemiology of Aneurysmal Subarachnoid Hemorrhage. Neurosurgery clinics of North America. 2010;21(2):221–233 https://www.ncbi.nlm.nih.gov/pubmed/20380965[↩]
- Caceres JA, Goldstein JN. Intracranial Hemorrhage. Emergency medicine clinics of North America. 2012;30(3):771-794. doi:10.1016/j.emc.2012.06.003. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3443867/ [↩]
- Polmear A. Sentinel headaches in aneurysmal subarachnoid haemorrhage: what is the true incidence? A systematic review. Cephalalgia. 2003;23(10):935–41.[↩]
- Perry JJ, et al. High risk clinical characteristics for subarachnoid haemorrhage in patients with acute headache: prospective cohort study. BMJ. 2010;341:c5204. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2966872/[↩]
- Shinohara Y. Hemorrhagic stroke syndromes: clinical manifestations of, intracerebral and subarachnoid hemorrhage. In: Marc F, editor. Handbook of Clinical Neurology. Elsevier; 2008. pp. 577–594[↩]
- Feigin VL, et al. Risk Factors for Subarachnoid Hemorrhage. Stroke. 2005;36(12):2773–2780.[↩]
- Anderson CS, et al. Active and Passive Smoking and the Risk of Subarachnoid Hemorrhage. Stroke. 2004;35(3):633–637[↩]
- Broderick JP, et al. Major Risk Factors for Aneurysmal Subarachnoid Hemorrhage in the Young Are Modifiable. Stroke. 2003;34(6):1375–1381[↩]
- Ferro J, Canhão P, Peralta R. Update on subarachnoid haemorrhage. Journal of Neurology. 2008;255(4):465–479[↩]
- Vlak MHM, et al. Prevalence of unruptured intracranial aneurysms, with emphasis on sex, age, comorbidity, country, and time period: a systematic review and meta-analysis. The Lancet Neurology. 2011;10(7):626–636[↩]
- Wermer MJH, et al. Risk of Rupture of Unruptured Intracranial Aneurysms in Relation to Patient and Aneurysm Characteristics. Stroke. 2007;38(4):1404–1410[↩]
- Investigators* TC. Rates of Delayed Rebleeding From Intracranial Aneurysms Are Low After Surgical and Endovascular Treatment. Stroke. 2006;37(6):1437–1442[↩]
- Diringer M, et al. Critical Care Management of Patients Following Aneurysmal Subarachnoid Hemorrhage: Recommendations from the Neurocritical Care Society’s Multidisciplinary Consensus Conference. Neurocritical Care. 2011;15(2):211–240[↩][↩]
- Fisher CM. Pathological observations in hypertensive cerebral hemorrhage. J Neuropathol Exp Neurol. 1971;30(3):536–50. https://www.ncbi.nlm.nih.gov/pubmed/4105427[↩]
- Vinters H. Cerebral amyloid angiopathy. A critical review. Stroke. 1987;18(2):311–324 http://stroke.ahajournals.org/content/18/2/311.long[↩]
- Viswanathan A, Greenberg SM. Cerebral amyloid angiopathy in the elderly. Annals of Neurology. 2011;70(6):871–880 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4004372[↩]
- Aronowski J, Zhao X. Molecular pathophysiology of cerebral hemorrhage: secondary brain injury. Stroke. 2011;42(6):1781–6 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3123894/[↩][↩]
- Brott T, et al. Early hemorrhage growth in patients with intracerebral hemorrhage. Stroke. 1997;28(1):1–5. http://stroke.ahajournals.org/content/28/1/1.long[↩]
- Feigin VL, et al. Worldwide stroke incidence and early case fatality reported in 56 population-based studies: a systematic review. The Lancet Neurology. 2009;8(4):355–369 https://www.ncbi.nlm.nih.gov/pubmed/19233729[↩]
- Labovitz DL, et al. The incidence of deep and lobar intracerebral hemorrhage in whites, blacks, and Hispanics. Neurology. 2005;65(4):518–522 https://www.ncbi.nlm.nih.gov/pubmed/16116109[↩]
- Matsukawa H, et al. Factors associated with lobar vs. non-lobar intracerebral hemorrhage. Acta Neurologica Scandinavica. 2011 https://www.ncbi.nlm.nih.gov/pubmed/22067041[↩]
- Ariesen MJ, et al. Risk Factors for Intracerebral Hemorrhage in the General Population. Stroke. 2003;34(8):2060–2065 http://stroke.ahajournals.org/content/34/8/2060.long[↩][↩]
- Maia LF, Mackenzie IRA, Feldman HH. Clinical phenotypes of Cerebral Amyloid Angiopathy. Journal of the Neurological Sciences. 2007;257(1–2):23–30 https://www.ncbi.nlm.nih.gov/pubmed/17341423[↩]
- Badjatia N, Rosand J. Intracerebral Hemorrhage. The Neurologist. 2005;11(6):311–324 https://www.ncbi.nlm.nih.gov/pubmed/16286875[↩]
- Wieberdink RG, et al. Serum Lipid Levels and the Risk of Intracerebral Hemorrhage: The Rotterdam Study. Arteriosclerosis, Thrombosis, and Vascular Biology. 2011;31(12):2982–2989 http://atvb.ahajournals.org/content/31/12/2982.long[↩]
- Biffi A, et al. Variants at APOE influence risk of deep and lobar intracerebral hemorrhage. Annals of Neurology. 2010;68(6):934–943 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3058266/[↩]
- Brouwers HB, et al. Apolipoprotein E Genotype Predicts Hematoma Expansion in Lobar Intracerebral Hemorrhage. Stroke. 2012 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3361564/[↩]
- Flaherty ML. Anticoagulant-Associated Intracerebral Hemorrhage. Semin Neurol. 2010;30(05):565, 572. https://www.ncbi.nlm.nih.gov/pubmed/21207349[↩]
- Martin-Schild S, et al. Intracerebral Hemorrhage in Cocaine Users. Stroke. 2010;41(4):680–684. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3412877/[↩]
- Anderson CS, et al. Intensive blood pressure reduction in acute cerebral haemorrhage trial (INTERACT): a randomised pilot trial. The Lancet Neurology. 2008;7(5):391–399 https://www.ncbi.nlm.nih.gov/pubmed/18396107[↩]
- Characterization of incident stroke signs and symptoms: findings from the atherosclerosis risk in communities study. Rathore SS, Hinn AR, Cooper LS, Tyroler HA, Rosamond WD. Stroke. 2002 Nov; 33(11):2718-21. http://stroke.ahajournals.org/content/33/11/2718.long[↩]
- Panagos PD, Jauch EC, Broderick JP. Intracerebral hemorrhage. Emerg Med Clin North Am. 2002;20(3):631–55 https://www.ncbi.nlm.nih.gov/pubmed/12379965[↩]
- Kothari RU, et al. The ABCs of Measuring Intracerebral Hemorrhage Volumes. Stroke. 1996;27(8):1304–1305 http://stroke.ahajournals.org/content/27/8/1304.long[↩]
- Delgado Almandoz JE, Romero JM. Advanced CT Imaging in the Evaluation of Hemorrhagic Stroke. Neuroimaging clinics of North America. 2011;21(2):197–213 https://www.ncbi.nlm.nih.gov/pubmed/21640295[↩]
- Oleinik A, et al. CT Angiography for Intracerebral Hemorrhage Does Not Increase Risk of Acute Nephropathy. Stroke. 2009;40(7):2393–2397 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2726774/[↩]
- Delgado Almandoz JE, et al. Diagnostic Accuracy and Yield of Multidetector CT Angiography in the Evaluation of Spontaneous Intraparenchymal Cerebral Hemorrhage. American Journal of Neuroradiology. 2009;30(6):1213–1221 http://www.ajnr.org/content/30/6/1213.long[↩][↩]
- Li N, et al. Contrast Extravasation on Computed Tomography Angiography Predicts Clinical Outcome in Primary Intracerebral Hemorrhage. Stroke. 2011;42(12):3441–3446 http://stroke.ahajournals.org/content/42/12/3441.long[↩]
- Kidwell CS, et al. Comparison of MRI and CT for Detection of Acute Intracerebral Hemorrhage. JAMA: The Journal of the American Medical Association. 2004;292(15):1823–1830 https://www.ncbi.nlm.nih.gov/pubmed/15494579[↩]
- Smith SD, Eskey CJ. Hemorrhagic Stroke. Radiologic clinics of North America. 2011;49(1):27–45. https://www.ncbi.nlm.nih.gov/pubmed/21111128[↩]
- DeLano MC, DeMarco JK. 3.0 T versus 1.5 T MR Angiography of the Head and Neck. Neuroimaging clinics of North America. 2006;16(2):321–341 https://www.ncbi.nlm.nih.gov/pubmed/16731370[↩]
- Rincon F, Mayer S. Clinical review: Critical care management of spontaneous intracerebral hemorrhage. Critical Care. 2008;12(6):237 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2646334/[↩][↩]
- Morgenstern LB, et al. Guidelines for the Management of Spontaneous Intracerebral Hemorrhage. Stroke. 2010;41(9):2108–2129 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4462131/[↩][↩][↩][↩][↩][↩][↩]
- Anderson CS, et al. Intensive blood pressure reduction in acute cerebral haemorrhage trial (INTERACT): a randomised pilot trial. The Lancet Neurology. 2008;7(5):391–399. https://www.ncbi.nlm.nih.gov/pubmed/18396107[↩]
- Antihypertensive treatment of acute cerebral hemorrhage. Crit Care Med. 2010;38(2):637–48. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5568798/[↩]
- Qureshi A, Palesch Y. Antihypertensive Treatment of Acute Cerebral Hemorrhage (ATACH) II: Design, Methods, and Rationale. Neurocritical Care. 2011;15(3):559–576 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3340125/[↩]
- Morgenstern LB, et al. Guidelines for the Management of Spontaneous Intracerebral Hemorrhage. Stroke. 2010;41(9):2108–2129 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4462131[↩]
- Steiner T, et al. Recommendations for the management of intracranial haemorrhage – part I: spontaneous intracerebral haemorrhage. The European Stroke Initiative Writing Committee and the Writing Committee for the EUSI Executive Committee. Cerebrovasc Dis. 2006;22(4):294–316 https://www.karger.com/Article/Abstract/94831[↩]
- Steiner T, et al. Recommendations for the management of intracranial haemorrhage – part I: spontaneous intracerebral haemorrhage. The European Stroke Initiative Writing Committee and the Writing Committee for the EUSI Executive Committee. Cerebrovasc Dis. 2006;22(4):294–316 https://www.ncbi.nlm.nih.gov/pubmed/16926557[↩]
- Mayer SA, et al. Efficacy and safety of recombinant activated factor VII for acute intracerebral hemorrhage. N Engl J Med. 2008;358(20):2127–37 http://www.nejm.org/doi/10.1056/NEJMoa0707534[↩]
- Aguilar MI, et al. Treatment of Warfarin-Associated Intracerebral Hemorrhage: Literature Review and Expert Opinion. Mayo Clinic proceedings. Mayo Clinic proceedings. Mayo Clinic. 2007;82(1):82–92. https://www.ncbi.nlm.nih.gov/pubmed/17285789[↩][↩]
- Watson HG, et al. A comparison of the efficacy and rate of response to oral and intravenous Vitamin K in reversal of over-anticoagulation with warfarin. Br J Haematol. 2001;115(1):145–9. https://www.ncbi.nlm.nih.gov/pubmed/11722425[↩]
- Goodnough LT, Shander A. How I treat warfarin-associated coagulopathy in patients with intracerebral hemorrhage. Blood. 2011;117(23):6091–6099 http://www.bloodjournal.org/content/117/23/6091.long[↩]
- Goldstein J, Rosand J, Schwamm L. Warfarin Reversal in Anticoagulant- Associated Intracerebral Hemorrhage. Neurocritical Care. 2008;9(2):277–283 https://www.ncbi.nlm.nih.gov/pubmed/18214714[↩][↩]
- O’Shaughnessy DF, et al. Guidelines for the use of fresh-frozen plasma, cryoprecipitate and cryosupernatant. Br J Haematol. 2004;126(1):11–28 https://www.ncbi.nlm.nih.gov/pubmed/15198728[↩]
- Steiner T, Rosand J, Diringer M. Intracerebral Hemorrhage Associated With Oral Anticoagulant Therapy. Stroke. 2006;37(1):256–262 http://stroke.ahajournals.org/content/37/1/256.long[↩]
- Goldstein JN, et al. Timing of Fresh Frozen Plasma Administration and Rapid Correction of Coagulopathy in Warfarin-Related Intracerebral Hemorrhage. Stroke. 2006;37(1):151–155 http://stroke.ahajournals.org/content/37/1/151.long[↩]
- Bershad E, Suarez J. Prothrombin Complex Concentrates for Oral Anticoagulant Therapy-Related Intracranial Hemorrhage: A Review of the Literature. Neurocritical Care. 2010;12(3):403–413 https://www.ncbi.nlm.nih.gov/pubmed/19967567[↩]
- Holland L, et al. Suboptimal effect of a three-factor prothrombin complex concentrate (Profilnine-SD) in correcting supratherapeutic international normalized ratio due to warfarin overdose. Transfusion. 2009;49(6):1171–1177 https://www.ncbi.nlm.nih.gov/pubmed/19210325[↩]
- Bershad E, Suarez J. Prothrombin Complex Concentrates for Oral Anticoagulant Therapy-Related Intracranial Hemorrhage: A Review of the Literature. Neurocritical Care. 2010;12(3):403–413. https://www.ncbi.nlm.nih.gov/pubmed/19967567[↩]
- Dentali F, et al. Safety of prothrombin complex concentrates for rapidanticoagulation reversal of vitamin K antagonists. A meta-analysis. Thromb Haemost. 2011;106(3):429–38 https://www.ncbi.nlm.nih.gov/pubmed/21800002[↩]
- Gurol M, Greenberg S. Management of intracerebral hemorrhage. Current Atherosclerosis Reports. 2008;10(4):324–331 https://www.ncbi.nlm.nih.gov/pubmed/18606103[↩]
- Thompson BB, et al. Prior antiplatelet therapy and outcome following intracerebral hemorrhage: a systematic review. Neurology. 2010;75(15):1333–42 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3013483/[↩]
- Naidech AM, et al. Early platelet transfusion improves platelet activity and may improve outcomes after intracerebral hemorrhage. Neurocrit Care. 2012;16(1):82–7 https://www.ncbi.nlm.nih.gov/pubmed/21837536[↩]
- de Gans K, et al. Patch: platelet transfusion in cerebral haemorrhage: study protocol for a multicentre, randomised, controlled trial. BMC Neurology. 2010;10(1):19 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2851678/[↩]
- Granger CB, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2011;365(11):981–92 http://www.nejm.org/doi/10.1056/NEJMoa1107039[↩]
- Watanabe M, Siddiqui FM, Qureshi AI. Incidence and management of ischemic stroke and intracerebral hemorrhage in patients on dabigatran etexilate treatment. Neurocrit Care. 2012;16(1):203–9 https://www.ncbi.nlm.nih.gov/pubmed/21748501[↩]
- Stead L, et al. Emergency Department Hyperglycemia as a Predictor of Early Mortality and Worse Functional Outcome after Intracerebral Hemorrhage. Neurocritical Care. 2010;13(1):67–74 https://www.ncbi.nlm.nih.gov/pubmed/20390379[↩]
- Middleton S, et al. Implementation of evidence-based treatment protocols to manage fever, hyperglycaemia, and swallowing dysfunction in acute stroke (QASC): a cluster randomised controlled trial. Lancet. 2011;378(9804):1699–706 https://www.ncbi.nlm.nih.gov/pubmed/21996470[↩]
- Kumar MA, et al. Anemia and hematoma volume in acute intracerebral hemorrhage. Critical Care Medicine. 2009;37(4):1442–1447. 10.1097/CCM.0b013e31819ced3a https://www.ncbi.nlm.nih.gov/pubmed/19242340[↩]
- Sheth KN, et al. Packed Red Blood Cell Transfusion and Decreased Mortality in Intracerebral Hemorrhage. Neurosurgery. 2011;68(5):1286–1292. 10.1227/NEU.0b013e31820cccb2 https://www.ncbi.nlm.nih.gov/pubmed/21307801[↩][↩]
- Claassen J, et al. Electrographic seizures and periodic discharges after intracerebral hemorrhage. Neurology. 2007;69(13):1356–1365 https://www.ncbi.nlm.nih.gov/pubmed/17893296[↩]
- De Herdt V, et al. Early seizures in intracerebral hemorrhage. Neurology. 2011;77(20):1794–1800 https://www.ncbi.nlm.nih.gov/pubmed/21975203[↩]
- Reddig RT, Nixdorf KE, Jensen MB. The prophylactic use of an antiepileptic drug in intracerebral hemorrhage. Clinical Neurology and Neurosurgery. 2011;113(10):895–897 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3215779/[↩]
- Naidech AM, et al. Anticonvulsant Use and Outcomes After Intracerebral Hemorrhage. Stroke. 2009;40(12):3810–3815 http://stroke.ahajournals.org/content/40/12/3810.long[↩]
- Smith M. Monitoring Intracranial Pressure in Traumatic Brain Injury. Anesthesia & Analgesia. 2008;106(1):240–248 https://www.ncbi.nlm.nih.gov/pubmed/18165584[↩]
- Morgenstern LB, et al. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2010;41(9):2108–29 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4462131/[↩][↩]
- Hallevi H, et al. Intraventricular hemorrhage. Neurology. 2008;70(11):848–852 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2745649/[↩]
- Tuhrim S, et al. Volume of ventricular blood is an important determinant of outcome in supratentorial intracerebral hemorrhage. Critical Care Medicine. 1999;27(3):617–621 https://www.ncbi.nlm.nih.gov/pubmed/10199544[↩]
- Naff N, et al. Low-Dose Recombinant Tissue-Type Plasminogen Activator Enhances Clot Resolution in Brain Hemorrhage. Stroke. 2011;42(11):3009–3016 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3356690/[↩]
- Gaberel T, et al. Intraventricular Fibrinolysis Versus External Ventricular Drainage Alone in Intraventricular Hemorrhage. Stroke. 2011 http://stroke.ahajournals.org/content/42/10/2776.long[↩]
- Mendelow AD, et al. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial intracerebral haematomas in the International Surgical Trial in Intracerebral Haemorrhage (STICH): a randomised trial. The Lancet. 2005;365(9457):387–397[↩]
- Mendelow AD, et al. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial intracerebral haematomas in the International Surgical Trial in Intracerebral Haemorrhage (STICH): a randomised trial. Lancet. 2005;365(9457):387–97[↩]
- Mendelow AD, et al. Surgical Trial in Lobar Intracerebral Haemorrhage (STICH II) Protocol. Trials. 2011;12(1):124 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3107158/[↩]
- Morgenstern LB, et al. Rebleeding leads to poor outcome in ultra-early craniotomy for intracerebral hemorrhage. Neurology. 2001;56(10):1294–1299 https://www.ncbi.nlm.nih.gov/pubmed/11376176[↩]
- Tan SH, et al. Hypertensive basal ganglia hemorrhage: a prospective study comparing surgical and nonsurgical management. Surgical Neurology. 2001;56(5):287–292[↩]
- Zhou H, et al. A prospective controlled study: Minimally invasive stereotactic puncture therapy versus conventional craniotomy in the treatment of acute intracerebral hemorrhage. BMC Neurology. 2011;11(1):76[↩]
- Wang W-Z, et al. Minimally invasive craniopuncture therapy vs. conservative treatment for spontaneous intracerebral hemorrhage: results from a randomized clinical trial in China. International Journal of Stroke. 2009;4(1):11–16[↩]