Contents
- What is Fish Oil
- What are Omega-3 Fatty Acids
- Recommended Intakes of Omega-3 Fatty Acids
- Fish Oil Omega-3 Polyunsaturated Fatty Acids
- What is fish oil good for ?
- What are the benefits of taking fish-oil supplements ?
- Cardiovascular disease (CVD) and CVD risk factors
- Infant health and neurodevelopment
- Cancer prevention
- Alzheimer’s disease, dementia, and cognitive function
- Age-Related Macular Degeneration (AMD)
- Dry eye disease
- Rheumatoid arthritis
- Depression
- Bipolar disorder
- Inflammatory bowel disease
- Autism Spectrum Disorders
- ADHD
- Childhood allergies
- Cystic fibrosis
- What kinds of omega-3 dietary supplements are available ?
- Fish oil supplement side effects
- Krill oil vs Fish oil
What is Fish Oil
Fish oil is a type of polyunsaturated fatty acids that is derived from fish and contain a rich source of Omega-3 essential fatty acids that are important for a number of functions in your body. Fish oil is commonly found in fatty fish such as salmon, tuna, and trout—and shellfish—such as crab, mussels, and oysters. Omega-3 fatty acids are called essential fatty acids because your body cannot make them and you need it them in your diet.
Although there are other sources you can get the Omega-3 essential fatty acids from, for example Alpha-linolenic acid (ALA) from plant oils such as flaxseed, soybean, and canola oils; only fish oil is rich in Eicosapentaenoic acid (EPA) and Docosahexaenoic acid (DHA) Omega-3 essential fatty acids.
Omega-3 Eicosapentaenoic acid (EPA) and Omega-3 Docosahexaenoic acid (DHA) are found in seafood, including fatty fish (e.g., salmon, tuna, and trout) and shellfish (e.g., crab, mussels, and oysters).
Note the differences between the Omega-3 fatty acids found in flaxseed, soybean, vegetable oils (alpha-linolenic acid (ALA)) from those in fish and seafoods (Eicosapentaenoic acid (EPA) and Docosahexaenoic acid (DHA)).
Our bodies can convert plant omega-3 fatty acids, Alpha-linolenic acid (ALA), to fish omega-3 fatty acids, Eicosapentaenoic acid (EPA) and Docosahexaenoic acid (DHA), but only a small fraction about 5% of Alpha-linolenic acid (ALA) to eicosapentaenoic acid (EPA), the level of eicosapentaenoic acid (EPA) in blood does not rise when we eat α-linolenic acid 1.
What are polyunsaturated fatty acids
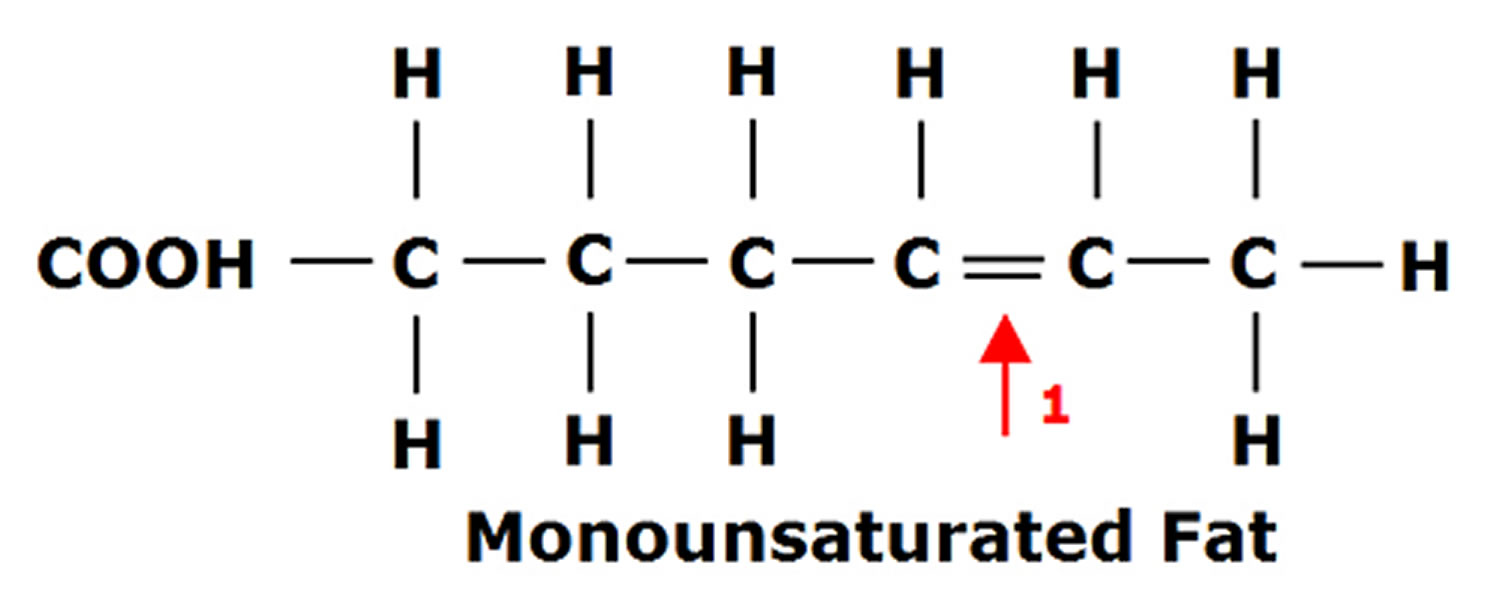
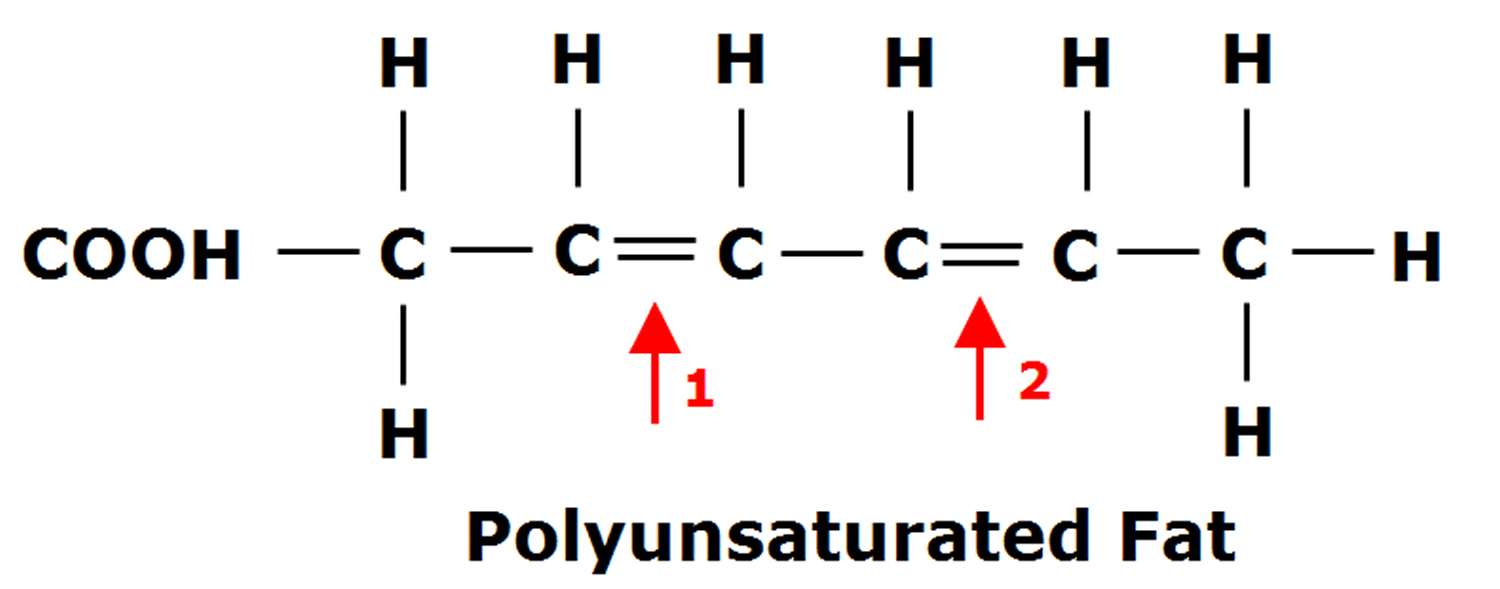
Fats are essential for living organisms. Fatty acid molecules have a variable length carbon chain with a methyl terminus and a carboxylic acid head group 2. They can be categorized based on the degree of saturation of their carbon chains. Saturated fatty acids possess the maximal number of hydrogen atoms, while monounsaturated fatty acids (MUFAs) and polyunsaturated fatty acids (PUFAs) have one, or two or more, double bonds, respectively.
Figure 1. Monounsaturated Fatty Acids Structure
Figure 2. Polyunsaturated Fatty Acids Structure
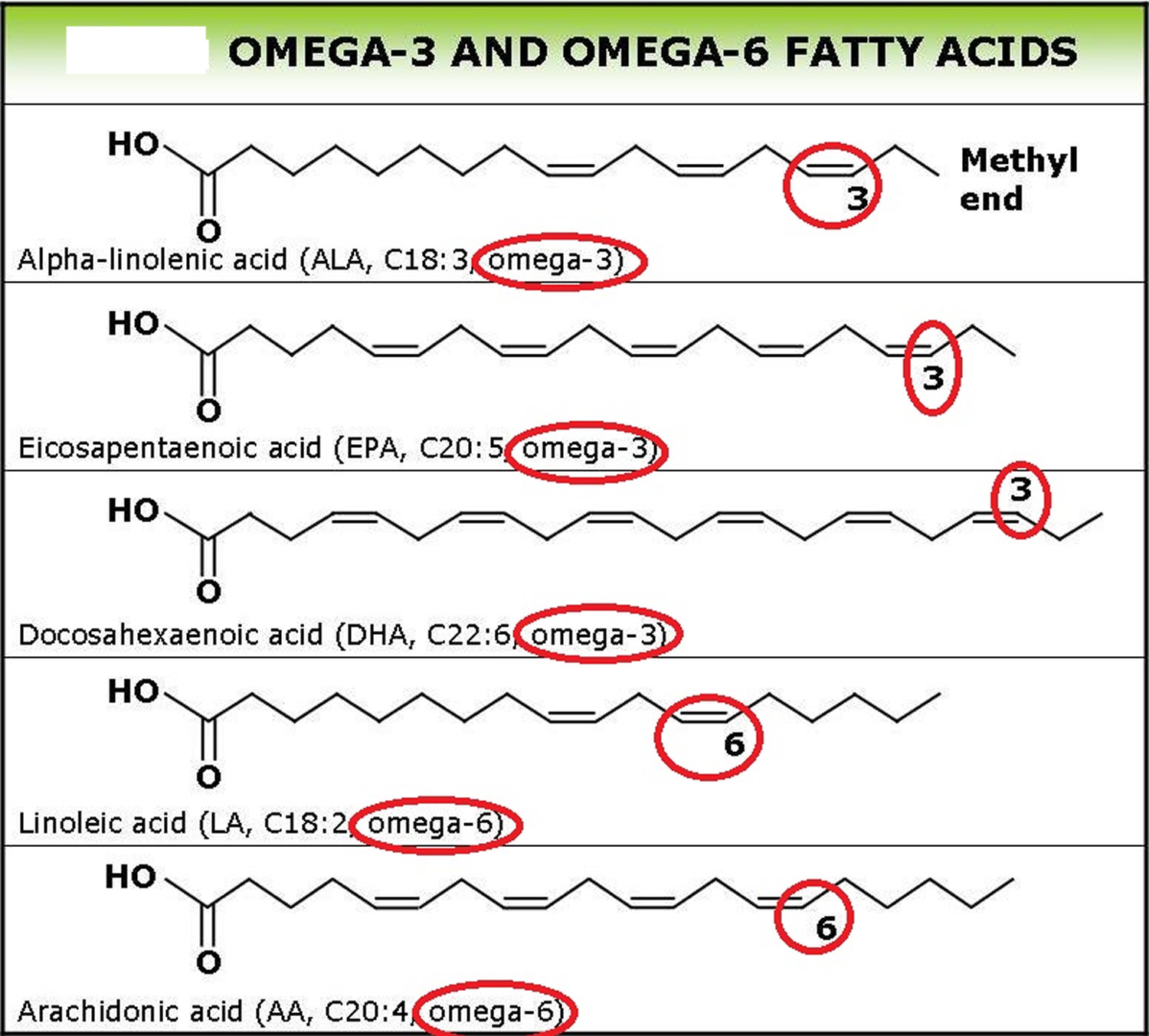
Polyunsaturated fatty acids (PUFAs) can be further subdivided on the basis of the location of the first double bond relative to the methyl terminus of the chain. For example, Omega-3 and Omega-6 fatty acids are two of the most biologically significant polyunsaturated fatty acid classes, and have their first double bond on either the third or sixth carbon from the chain terminus, respectively. The final carbon in the fatty acid chain is also known as the omega carbon, hence the common reference to these fatty acids as Omega-3 or Omega-6 PUFAs. The distinction between omega-6 and omega-3 fatty acids is based on the location of the first double bond, counting from the methyl end of the fatty acid molecule (see Figure 3). Omega-6 fatty acids are represented by Linoleic acid (LA) (18:2ω-6) and Arachidonic acid (AA) (20:4ω-6) and omega-3 fatty acids by Alpha-linolenic acid (ALA) (18:3ω-3), Eicosapentaenoic acid (EPA) (20:5ω-3) and Docosahexaenoic acid (DHA) (22:6ω-3).
Long-chain Omega-3 and Omega-6 PUFAs are synthesized from the essential fatty acids: alpha-linolenic acid (ALA) and linoleic acid, respectively. Basic structures of these two parent PUFAs are shown in Figure 3. An essential fatty acid cannot be made by the body and must be obtained through dietary sources. Animals and humans have the capacity to metabolize essential fatty acids to long-chain derivatives. Because the Omega-3 and Omega-6 pathways compete with one another for enzyme activity, the ratio of Omega-6 to Omega-3 PUFAs is very important to human health. An overabundance of fatty acids from one family will limit the metabolic production of the longer chain products of the other. The typical Western diet provides Omega-6 and Omega-3 PUFAs in a ratio ranging from 8:1 to 25:1 2, values in severe contrast with the recommendations from national health agencies of approximately 4:1 3. Lowering the n-6:n-3 ratio would reduce competition for the enzymes and facilitate the metabolism of more downstream products of ALA.
Mammalian cells cannot convert Omega-6 to Omega-3 fatty acids because they lack the converting enzyme, omega-3 desaturase. Omega-6 and omega-3 fatty acids are not interconvertible, are metabolically and functionally distinct, and often have important opposing physiological effects, therefore their balance in the diet is important 4.
This study showed a balanced omega-6/omega-3 ratio 1–2/1 is one of the most important dietary factors in the prevention of obesity, along with physical activity. A lower omega-6/omega-3 ratio should be considered in the management of obesity 4.
Because most diets are already very rich in Omega-6 PUFAs, greater focus needs to be placed on incorporating Omega-3 PUFAs into the diet. Dietary sources of Omega-3 PUFAs are readily available but in limited quantities. Many foods contain alpha-linolenic acid (ALA), including certain vegetable oils, dairy products, flaxseed, walnuts and vegetables 5. Fatty fish, such as mackerel, herring and salmon, provide an excellent source of the long-chain derivatives of ALA, eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) 3.
What are Omega-3 Fatty Acids
Omega-3 fatty acids are polyunsaturated fatty acids found in certain foods. The term “omega-3” refers to their chemical structure. Omega-3 fatty acids cannot be made in the body and must be obtained from food; thus, they are essential fatty acids. There are many forms of omega-3 fatty acids, each with a different length and chemical structure.
Where are omega-3 fatty acids found ?
- The shortest omega-3 fatty acid, α-linolenic acid or Alpha-linolenic acid (ALA), is found in plants and plant oils, including leafy vegetables, walnuts, mustard seed oil, soy bean oil, canola oil and flaxseed oil (which is made up of 50% Alpha-linolenic acid (ALA)).
- The longer omega-3 fatty acids, eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), are found in algae, which are eaten by fish and baleen whales. We obtain these fatty acids by eating fish (e.g., salmon). Eicosapentaenoic acid and docosahexaenoic acid are often referred to as being from marine or fish sources 1.
Plant omega-3 fatty acids, a different type of omega-3, called Alpha-linolenic acid (ALA), are not the same as fish omega-3 fatty acids. Although their chemical structures are similar, they have important differences, which affect their metabolism in the body.
Plant omega-3 Alpha-linolenic acid (ALA), can be found in some vegetable oils—such as canola and soy oils. Omega-3s are also available as dietary supplements. There is moderate evidence emerging about the health benefits of eating seafood like fish and shellfish. However, the health benefits of taking omega-3 as dietary supplements are unclear.
The main sources of Omega-3 Alpha-linolenic acid (ALA) in the U.S. diet are vegetable oils, particularly canola and soybean oils; flaxseed oil is richer in alpha-linolenic acid (ALA) than soybean and canola oils but is not commonly consumed. The typical North American diet provides approximately 1.4 g of ALA per day, and 0.1 g to 0.2 g of EPA and DHA 6. Alpha-linolenic acid (ALA) can be converted, usually in small amounts, into (Eicosapentaenoic acid) EPA and (Docosahexaenoic acid) DHA in the body.
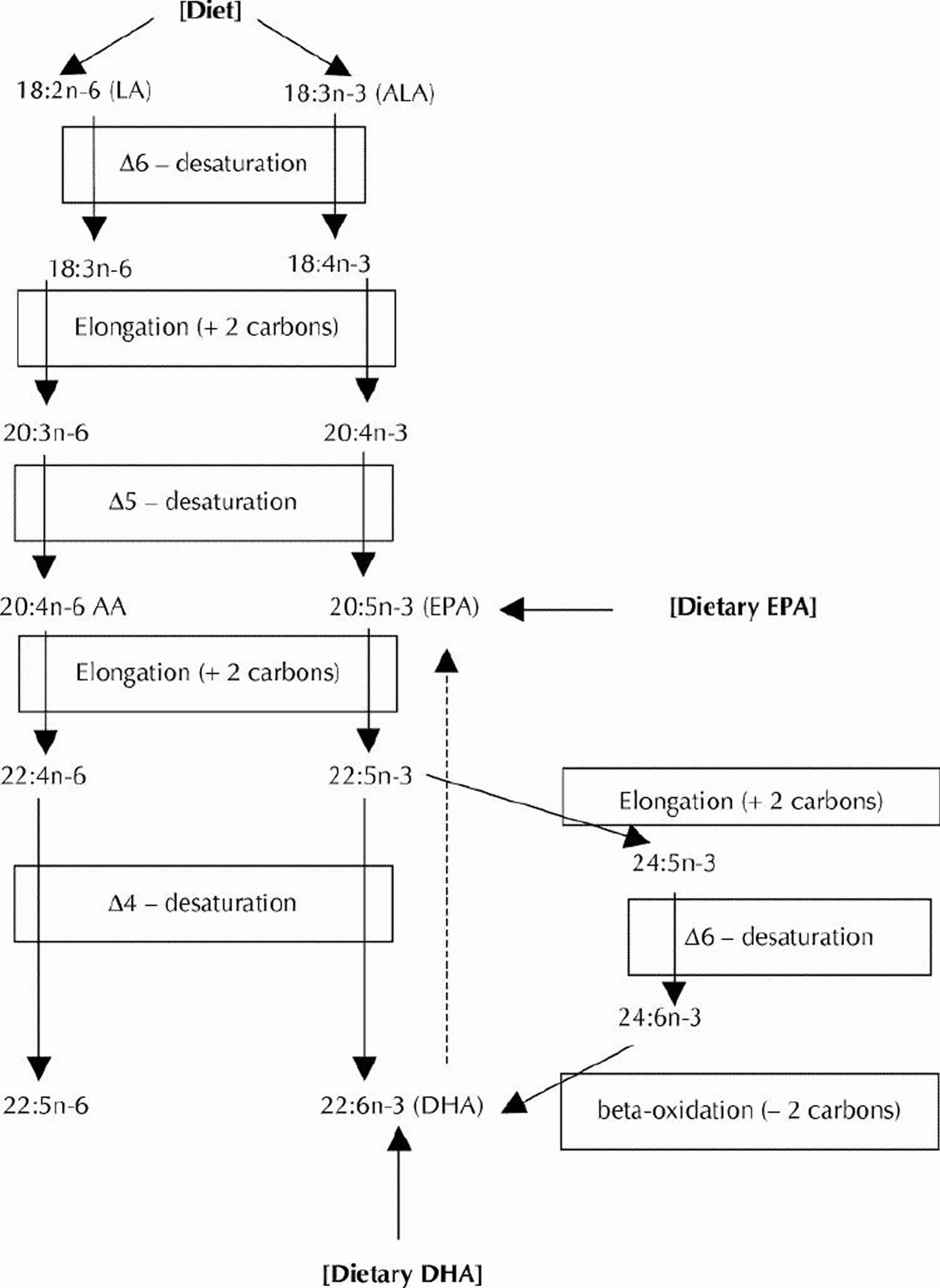
For those who do not consume fish, the omega-3 fatty acid known as alpha-linolenic acid (ALA) can be a dietary source of some metabolically derived Eicosapentaenoic acid (EPA) and Docosahexaenoic acid (DHA). The desaturation plus elongation reactions in the liver and elsewhere in the body that provide for the conversion of dietary ALA to EPA and DHA are depicted in Figure 4. The conversion of ALA to EPA and DHA occurs to a low extent (about 10%–15% efficiency) in the adult human body 7. Nonetheless, there is evidence that the benefits of the Mediterranean-type diet after myocardial infarction may be partly caused by the higher intake of ALA (commonly found in nonhydrogenated canola oil, ground flaxseed and other selected ALA-enriched foods) 8. A prospective cohort study (Nurses Health Study) revealed an inverse relation between ALA intakes and the risk of fatal ischemic heart disease among women 9. However, the Zutphen Elderly Study did not observe a beneficial effect of dietary ALA on the 10-year risk of coronary artery disease 10. The metabolic conversion of ALA to the longer chain omega-3 fatty acids (EPA and DHA) is thought to mediate any possible cardioprotective effects of dietary ALA. In contrast to the well-recognized serum triglyceride-lowering effect of EPA and DHA, most human intervention studies with ALA (e.g., using flaxseed oil) have not exhibited any lipid-lowering effects. Whereas ALA (from flaxseed oil) at a relatively high dose has been found to improve arterial compliance 11, considerably lower supplementation levels of EPA and DHA improved arterial and endothelial functioning in subjects with hypercholesterolemia 12 and subjects with type 2 diabetes mellitus 13.
Recommended Intakes of Omega-3 Fatty Acids
Intake recommendations for fatty acids and other nutrients are provided in the Dietary Reference Intakes (DRIs) developed by the Food and Nutrition Board of the Institute of Medicine. DRI is the general term for a set of reference values used for planning and assessing nutrient intakes of healthy people. These values, which vary by age and sex, include:
- Recommended Dietary Allowance (RDA): average daily level of intake sufficient to meet the nutrient requirements of nearly all (97%–98%) healthy individuals.
- Adequate Intake (AI): established when evidence is insufficient to develop an RDA; intake at this level is assumed to ensure nutritional adequacy.
- Estimated Average Requirement (EAR): average daily level of intake estimated to meet the requirements of 50% of healthy individuals. It is usually used to assess the adequacy of nutrient intakes in populations but not individuals.
- Tolerable Upper Intake Level (UL): maximum daily intake unlikely to cause adverse health effects.
- Acceptable Macronutrient Distribution Range (AMDR): range of intake for a particular energy source (macronutrient) that is associated with reduced risk of chronic disease while providing intakes of essential nutrients.
When the Food and Nutrition Board of the Institute of Medicine last reviewed omega-3s, insufficient data were available to establish an Estimated Average Requirement (EAR), so the Food and Nutrition Board of the Institute of Medicine established AIs for all ages based on omega-3 intakes in healthy populations 14.
Table 1 lists the current Adequate Intake (AIs) for omega-3s in grams per day. Human milk contains omega-3s as ALA, EPA and DHA, so the Institute of Medicine established an AI for infants from birth to 12 months that is equivalent to the mean intake of omega-3s in healthy, breastfed infants.
For infants, the AIs apply to total omega-3s. For ages 1 and older, the AIs apply only to ALA because ALA is the only omega-3 that is essential. The Institute of Medicine did not establish specific intake recommendations for EPA, DHA or other long chain omega-3s.
Table 1: Adequate Intakes (AIs) for Omega-3s
| Age | Male | Female | Pregnancy | Lactation |
|---|---|---|---|---|
| Birth to 6 months* | 0.5 g | 0.5 g | ||
| 7–12 months* | 0.5 g | 0.5 g | ||
| 1–3 years** | 0.7 g | 0.7 g | ||
| 4–8 years** | 0.9 g | 0.9 g | ||
| 9–13 years** | 1.2 g | 1.0 g | ||
| 14–18 years** | 1.6 g | 1.1 g | 1.4 g | 1.3 g |
| 19-50 years** | 1.6 g | 1.1 g | 1.4 g | 1.3 g |
| 51+ years** | 1.6 g | 1.1 g |
*As total omega-3s
**As ALA
Fish Oil Omega-3 Polyunsaturated Fatty Acids
The fish-based and fish-oil–based omega-3 polyunsaturated fatty acids (also referred to as n-3 PUFA) consist of EPA (20 carbon atoms, 5 double bonds) and DHA (22 carbon atoms, 6 double bonds). The general structures for EPA and DHA are shown in Figure 3. Commonly available dietary sources of EPA and DHA are listed in Table 2. Whereas plant foods and vegetable oils lack EPA and DHA, some do contain varying amounts of the n-3 PUFA alpha-linolenic acid (ALA), which has 18 carbon atoms and 3 double bonds (Fig. 3). Many vegetable oils are greatly enriched in omega-6 fatty acids (mainly as linoleic acid in corn, safflower, sunflower and soybean oils), but canola oil (nonhydrogenated), ground flaxseed and walnuts are rich sources of ALA.
The typical North American diet provides about 1–3 g of n-3 PUFA alpha-linolenic acid (ALA) per day but only 0.10–0.15 g of EPA (Eicosapentaenoic acid) plus DHA (Docosahexaenoic acid) per day 15, 16. The very high intake of n-6 PUFA, mostly as linoleic acid (LA) (Fig. 3) in our diet (12–15 g/day) from common vegetable oils (corn, safflower, soybean) and other sources, yields an overall n-6:n-3 dietary ratio (total omega-6 fatty acids in the diet: total omega-3 fatty acids in the diet) of about 8:1. Health Canada has recommended that this ratio be as low as 4:19 to reduce the competitive influence of high LA intakes on ALA metabolism to its longer chain products (such as EPA and DHA). Although high intakes of LA can provide some modest blood cholesterol lowering, experimental studies in animals have raised concerns regarding the enhancing effect of these high intakes on certain cancers 17. This association has not been established in human studies 18.
Figure 3. Omega-3 fatty acids and Omega-6 fatty acids structure
Figure 4. Conversion of dietary ALA to EPA and DHA via Desaturation, elongation and retroconversion of polyunsaturated fatty acids.
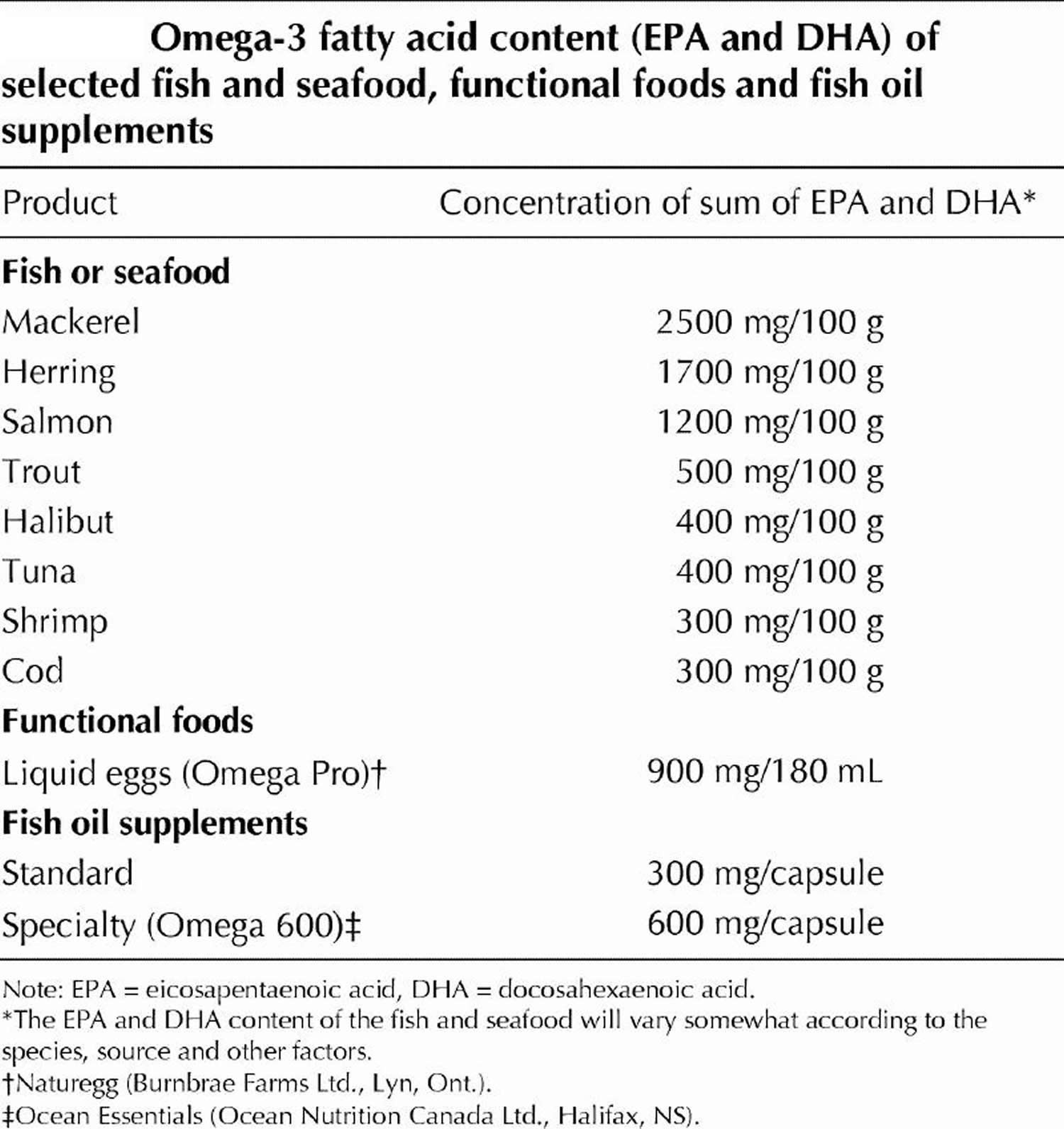
[Source 19]Table 2. Omega-3 Fatty Acid Foods EPA (Eicosapentaenoic acid) and DHA (Docosahexaenoic acid) – Fish and Seafood Sources
[Source 19]Table 3. EPA (Eicosapentaenoic acid) and DHA (Docosahexaenoic acid) Content of Fish Species
| Fish Species and Description | DHA per 100 g | EPA per 100 g | DHA+EPA per 100 g | DHA+EPA per 85 g (3 oz.) |
|---|---|---|---|---|
| Crustaceans, crab, Alaska king, cooked, moist heat | 0.118 | 0.295 | 0.413 | 0.351 |
| Crustaceans, crab, blue, cooked, moist heat | 0.231 | 0.243 | 0.474 | 0.403 |
| Crustaceans, crab, Dungeness, cooked, moist heat | 0.113 | 0.281 | 0.394 | 0.335 |
| Crustaceans, crab, queen, cooked, moist heat | 0.145 | 0.332 | 0.477 | 0.405 |
| Crustaceans, crayfish, mixed species, farmed, cooked, moist heat | 0.038 | 0.124 | 0.162 | 0.138 |
| Crustaceans, crayfish, mixed species, wild, cooked, moist heat | 0.047 | 0.119 | 0.166 | 0.141 |
| Crustaceans, lobster, northern, cooked, moist heat | 0.031 | 0.053 | 0.084 | 0.071 |
| Crustaceans, shrimp, mixed species, cooked, moist heat | 0.144 | 0.171 | 0.315 | 0.268 |
| Crustaceans, spiny lobster, mixed species, cooked, moist heat | 0.139 | 0.341 | 0.480 | 0.408 |
| Fish, anchovy, European, raw | 0.911 | 0.538 | 1.449 | 1.232 |
| Fish, anchovy, European, canned in oil, drained solids | 1.292 | 0.763 | 2.055 | 1.747 |
| Fish, bass, freshwater, mixed species, cooked, dry heat | 0.458 | 0.305 | 0.763 | 0.649 |
| Fish, bass, striped, cooked, dry heat | 0.750 | 0.217 | 0.967 | 0.822 |
| Fish, bluefish, cooked, dry heat | 0.665 | 0.323 | 0.988 | 0.840 |
| Fish, turbot, cooked, dry heat | 0.123 | 0.09 | 0.213 | 0.181 |
| Fish, carp, cooked, dry heat | 0.146 | 0.305 | 0.451 | 0.383 |
| Fish, catfish, channel, farmed, cooked, dry heat | 0.128 | 0.049 | 0.177 | 0.150 |
| Fish, catfish, channel, wild, cooked, dry heat | 0.137 | 0.100 | 0.237 | 0.201 |
| Fish, caviar, black and red, granular | 3.800 | 2.741 | 6.541 | 5.560 |
| Fish, cod, Atlantic, cooked, dry heat | 0.154 | 0.004 | 0.158 | 0.134 |
| Fish, cod, Pacific, cooked, dry heat | 0.173 | 0.103 | 0.276 | 0.235 |
| Fish, croaker, Atlantic, raw | 0.097 | 0.123 | 0.22 | 0.187 |
| Fish, dolphin fish, cooked, dry heat | 0.113 | 0.026 | 0.139 | 0.118 |
| Fish, drum, freshwater, cooked, dry heat | 0.368 | 0.295 | 0.663 | 0.564 |
| Fish, eel, mixed species, cooked, dry heat | 0.081 | 0.108 | 0.189 | 0.161 |
| Fish, fish portions and sticks, frozen, preheated | 0.128 | 0.086 | 0.214 | 0.182 |
| Fish, flatfish (flounder and sole species), cooked, dry heat | 0.258 | 0.243 | 0.501 | 0.426 |
| Fish, grouper, mixed species, cooked, dry heat | 0.213 | 0.035 | 0.248 | 0.211 |
| Fish, haddock, cooked, dry heat | 0.162 | 0.076 | 0.238 | 0.202 |
| Fish, halibut, Atlantic and Pacific, cooked, dry heat | 0.374 | 0.091 | 0.465 | 0.395 |
| Fish, halibut, Greenland, cooked, dry heat | 0.504 | 0.674 | 1.178 | 1.001 |
| Fish, herring, Atlantic, cooked, dry heat | 1.105 | 0.909 | 2.014 | 1.712 |
| Fish, herring, Atlantic, kippered | 1.179 | 0.97 | 2.149 | 1.827 |
| Fish, herring, Pacific, cooked, dry heat | 0.883 | 1.242 | 2.125 | 1.806 |
| Fish, lingcod, cooked, dry heat | 0.130 | 0.133 | 0.263 | 0.224 |
| Fish, mackerel, Atlantic, cooked, dry heat | 0.699 | 0.504 | 1.203 | 1.023 |
| Fish, mackerel, king, cooked, dry heat | 0.227 | 0.174 | 0.401 | 0.341 |
| Fish, mackerel, Pacific and jack, mixed species, cooked, dry heat | 1.195 | 0.653 | 1.848 | 1.571 |
| Fish, mackerel, Spanish, cooked, dry heat | 0.952 | 0.294 | 1.246 | 1.059 |
| Fish, mullet, striped, cooked, dry heat | 0.148 | 0.18 | 0.328 | 0.279 |
| Fish, ocean perch, Atlantic, cooked, dry heat | 0.271 | 0.103 | 0.374 | 0.318 |
| Fish, perch, mixed species, cooked, dry heat | 0.223 | 0.101 | 0.324 | 0.275 |
| Fish, pike, northern, cooked, dry heat | 0.095 | 0.042 | 0.137 | 0.116 |
| Fish, pike, walleye, cooked, dry heat | 0.288 | 0.11 | 0.398 | 0.338 |
| Fish, pollock, Atlantic, cooked, dry heat | 0.451 | 0.091 | 0.542 | 0.461 |
| Fish, pompano, Florida, cooked, dry heat | ?? | ?? | ?? | 0.620 est |
| Fish, rockfish, Pacific, mixed species, cooked, dry heat | 0.262 | 0.181 | 0.443 | 0.377 |
| Fish, roe, mixed species, cooked, dry heat | 1.747 | 1.26 | 3.007 | 2.556 |
| Fish, roe, mixed species, raw | 1.363 | 0.983 | 2.346 | 1.994 |
| Fish, roughy, orange, raw | 0 | 0.001 | 0.001 | 0.001 |
| Fish, sablefish, cooked, dry heat | 0.920 | 0.867 | 1.787 | 1.519 |
| Fish, sablefish, smoked | 0.945 | 0.891 | 1.836 | 1.561 |
| Fish, salmon, Atlantic, farmed, cooked, dry heat | 1.457 | 0.69 | 2.147 | 1.825 |
| Fish, salmon, Atlantic, wild, cooked, dry heat | 1.429 | 0.411 | 1.84 | 1.564 |
| Fish, salmon, Chinook, cooked, dry heat | 0.727 | 1.01 | 1.737 | 1.476 |
| Fish, salmon, chum, cooked, dry heat | 0.505 | 0.299 | 0.804 | 0.683 |
| Fish, salmon, chum, drained solids with bone | 0.702 | 0.473 | 1.175 | 0.999 |
| Fish, salmon, coho, farmed, cooked, dry heat | 0.871 | 0.408 | 1.279 | 1.087 |
| Fish, salmon, coho, wild, cooked, dry heat | 0.658 | 0.401 | 1.059 | 0.900 |
| Fish, salmon, pink, cooked, dry heat | 0.751 | 0.537 | 1.288 | 1.095 |
| Fish, salmon, sockeye, cooked, dry heat | 0.700 | 0.53 | 1.23 | 1.046 |
| Fish, sardine, Atlantic, canned in oil, drained solids with bone | 0.509 | 0.473 | 0.982 | 0.835 |
| Fish, scup, raw (Porgy—assigned to low omega-3 group) | no data | no data | no data | no data |
| Fish, sea bass, mixed species, cooked, dry heat | 0.556 | 0.206 | 0.762 | 0.648 |
| Fish, sea trout, mixed species, cooked, dry heat | 0.265 | 0.211 | 0.476 | 0.405 |
| Fish, shad, American, raw | 1.321 | 1.086 | 2.407 | 2.046 |
| Fish, shark, mixed species, raw | 0.527 | 0.316 | 0.843 | 0.717 |
| Fish, sheepshead, cooked, dry heat | 0.107 | 0.083 | 0.19 | 0.162 |
| Fish, smelt, rainbow, cooked, dry heat | 0.536 | 0.353 | 0.889 | 0.756 |
| Fish, snapper, mixed species, cooked, dry heat | 0.273 | 0.048 | 0.321 | 0.273 |
| Fish, spot, cooked, dry heat | 0.526 | 0.282 | 0.808 | 0.687 |
| Fish, sturgeon, mixed species, cooked, dry heat | 0.119 | 0.249 | 0.368 | 0.313 |
| Fish, sucker, white, cooked, dry heat | 0.371 | 0.244 | 0.615 | 0.523 |
| Fish, sunfish, pumpkin seed, cooked, dry heat | 0.092 | 0.047 | 0.139 | 0.118 |
| Fish, swordfish, cooked, dry heat | 0.681 | 0.138 | 0.819 | 0.696 |
| Fish, tilefish, cooked, dry heat | 0.733 | 0.172 | 0.905 | 0.769 |
| Fish, trout, mixed species, cooked, dry heat | 0.677 | 0.259 | 0.936 | 0.796 |
| Fish, trout, rainbow, farmed, cooked, dry heat | 0.820 | 0.334 | 1.154 | 0.981 |
| Fish, trout, rainbow, wild, cooked, dry heat | 0.520 | 0.468 | 0.988 | 0.840 |
| Fish, tuna, fresh, bluefin, cooked, dry heat | 1.141 | 0.363 | 1.504 | 1.278 |
| Fish, tuna, light, canned in oil, drained solids | 0.101 | 0.027 | 0.128 | 0.109 |
| Fish, tuna, light, canned in water, drained solids | 0.223 | 0.047 | 0.27 | 0.230 |
| Fish, tuna, skipjack, fresh, cooked, dry heat | 0.237 | 0.091 | 0.328 | 0.279 |
| Fish, tuna, white, canned in water, drained solids | 0.629 | 0.233 | 0.862 | 0.733 |
| Fish, tuna, yellowfin, fresh, cooked, dry heat | 0.232 | 0.047 | 0.279 | 0.237 |
| Fish, whitefish, mixed species, cooked, dry heat | 1.206 | 0.406 | 1.612 | 1.370 |
| Fish, whiting, mixed species, cooked, dry heat | 0.235 | 0.283 | 0.518 | 0.440 |
| Fish, wolffish, Atlantic, cooked, dry heat | 0.405 | 0.393 | 0.798 | 0.678 |
| Frog legs, raw | 0.034 | 0.020 | ||
| Mollusks, abalone, mixed species, raw | 0 | 0.049 | 0.049 | 0.042 |
| Mollusks, clam, mixed species, cooked, moist heat | 0.146 | 0.138 | 0.284 | 0.241 |
| Mollusks, conch, baked or broiled | 0.072 | 0.048 | 0.12 | 0.102 |
| Mollusks, cuttlefish, mixed species, cooked, moist heat | 0.132 | 0.078 | 0.21 | 0.179 |
| Mollusks, mussel, blue, cooked, moist heat | 0.506 | 0.276 | 0.782 | 0.665 |
| Mollusks, octopus, common, cooked, moist heat | 0.162 | 0.152 | 0.314 | 0.267 |
| Mollusks, oyster, eastern, farmed, cooked, dry heat | 0.211 | 0.229 | 0.44 | 0.374 |
| Mollusks, oyster, eastern, wild, cooked, dry heat | 0.291 | 0.26 | 0.551 | 0.468 |
| Mollusks, oyster, Pacific, cooked, moist heat | 0.500 | 0.876 | 1.376 | 1.170 |
| Mollusks, scallop, mixed species, cooked, breaded and fried | 0.103 | 0.086 | 0.18 | 0.161 |
| Mollusks, whelk, unspecified, cooked, moist heat | 0.012 | 0.008 | 0.02 | 0.017 |
What is fish oil good for ?
Both omega-3 fatty acids from fish and plants appear to reduce the risk of some types of heart disease. However, the results of all studies have not been consistent. Some suggest that fish oil may reduce mortality among patients with coronary heart disease, possibly by stabilizing the heart’s rhythm and by reducing blood clotting. We don’t know as much about the effects of plant omega-3 fatty acids, but they appear to have similar actions in reducing coronary heart disease events.
Animal and human studies 21, 22, 23, 24 in addition to epidemiological surveys 25, 26 have also shown that Omega-3 PUFAs effectively enhance and improve learning capability as well as memory and cognitive function. Moreover, intake of EPA and DHA is reported to reduce the risk of developing Alzheimer’s disease 27.
Data supporting the use of EPA+DHA for potential cardiovascular benefit were derived from trials involving increased fish consumption or fish oil supplementation. These findings shaped the recommendations for the American Heart Association’s (AHA) Scientific Statement on Fish Consumption, Fish Oil, Omega-3 Fatty Acids, and Cardiovascular Disease 28. Key messages from this document are summarized in Table 4. Essentially, patients must consume a lower 1 g dose of EPA+DHA for cardioprotection or a higher 2 to 4 g dose for triglyceride reduction. Although the lower dose can often be achieved with dietary fish, the high dose generally requires supplementation.
Table 4. American Heart Association guidelines for omega-3 fatty acid consumption
| Goal | Recommendation |
| Cardioprotection | |
| Patients without a history of CVD | Consume oily fish 2 or more times weekly. |
| Patients with a history of CHD | Consume 1 g daily of EPH +DHA, preferably from oily fish. Fish oil supplements may be used with physician consultation. |
| Triglyceride lowering | Consume 2-4 g daily of EPA +DHA as capsules with physician consultation. |
Abbreviations: CHD = coronary heart disease; CVD = cardiovascular disease; EPA+DHA = eicosapentaenoic acid+docosahexaenoic acid.
The data from the secondary- and primary-prevention studies support the idea that consumption of very-long-chain Omega-3 fatty acids from fish and fish-oil supplements (EPA and DHA) reduces all-cause mortality, cardiac and sudden death and stroke 29. This conclusion agrees with 2 recent meta-analyses by He at al. 30, 31, and with the results of a 1999 ecologic study by Zhang et al 32. The latter showed a significant association between fish consumption and total mortality across 36 countries on the basis of data from the Food and Agriculture Organization and the World Health Organization 32. However, this conclusion is not applicable to the population of patients with an coronary heart disease, in whom 3 recent randomized clinical trials found inconsistent antiarrhythmic effects and no significant overall effect on mortality 29.
The evidence appears strong for a beneficial effect of very-long-chain Omega-3 fatty acids intakes on cardiovascular disease risk in secondary, but not in primary, prevention because data from randomized clinical trials are available for the former, but less evidence exists to support a beneficial effect on heart attack. There is no high-quality evidence to support a beneficial effect of alpha-linolenic acid (ALA) 29.
Are fish oils essential for human health ?
Many people believe this to be true, but officials are still divided. Vegetarians, who do not have an obvious source of fish oil in their diet, seem to have a low incidence of coronary heart disease. For certain functions, α-linolenic acid (ALA) from vegetable sources may be able to substitute for longer fatty acids, either directly or after partial conversion to eicosapentaenoic acid.
What are the benefits of taking fish-oil supplements ?
It is usually stated that omega-3 fatty acids from fish may reduce cardiac deaths, inflammatory disease, including rheumatoid arthritis and ulcerative colitis, and promote brain development and mental function.
Some doctors recommend fish-oil supplements to prevent or treat cardiovascular disease. Although fish oil does not reduce LDL “bad” cholesterol, it may be recommended to lower serum triglycerides. In addition, infant formula is supplemented with docosahexaenoic acid in Europe to enhance development of the central nervous system because the rapidly growing brain requires large amounts of this fatty acid 1. Docosahexaenoic acid used in infant formula comes from algae.
Evidence for a benefit for fish oils in preventing coronary artery restenosis after angioplasty and rhythm abnormalities is less clear.
Cardiovascular disease (CVD) and CVD risk factors
Many studies have assessed the effects of omega-3s—primarily EPA and DHA—on cardiovascular disease and cardiovascular disease risk factors, such as high blood pressure and elevated plasma lipids. This interest was spurred by epidemiological research dating back to the 1970s that found low rates of myocardial infarction (heart attack) and other coronary events among Greenland Inuit and other fish-eating populations, such as the Japanese 33. Results from observational studies have been consistent with these findings, with several systematic reviews and meta-analyses showing that higher consumption of fish and higher dietary or plasma levels of omega-3s are associated with a lower risk of heart failure 34, coronary disease, and fatal coronary heart disease 35.
Recommendations from the Dietary Guidelines for Americans: The 2015–2020 Dietary Guidelines for Americans states that strong evidence from mostly prospective cohort studies but also randomized controlled trials has shown that eating patterns that include seafood are associated with reduced risk of cardiovascular disease 36. In addition, consuming about 8 ounces per week of a variety of seafood that provides about 250 mg per day EPA and DHA is associated with fewer cardiac deaths in both healthy individuals and those with preexisting cardiovascular disease.
Conclusions about omega-3s and cardiovascular disease: Overall, research indicates that consuming fish and other types of seafood as part of a balanced diet promotes heart health. Fish oil and other LC omega-3 supplements improve blood lipids and appear to reduce the risk of cardiac death. However, their effects on other cardiovascular endpoints are unclear and might vary based on dietary omega-3 intakes and the use of cardioprotective medications.
The FDA has approved a qualified health claim for conventional foods and dietary supplements that contain EPA and DHA 37. It states, “Supportive but not conclusive research shows that consumption of EPA and DHA omega-3 fatty acids may reduce the risk of coronary heart disease.” The FDA also specifies that the labels of dietary supplements should not recommend a daily intake of EPA and DHA higher than 2 g 37. For patients who need to lower their triglycerides, the American Heart Association recommends 2–4 g/day of EPA plus DHA under the care of a physician. Several prescription omega-3 preparations are also available to treat hypertriglyceridemia.
Scientists hope to gain additional insight on the effects of omega-3s for the prevention of CVD from the VITamin D and OmegA-3 TriaL (VITAL) trial. This clinical trial will examine the effects of EPA (465 mg/day) and DHA (375 mg/day) supplementation with or without 2,000 IU/day vitamin D for 5 years in 25,875 older adults on the primary prevention of cancer and CVD 38. Results from this clinical trial and others will shed more light on possible associations between omega-3s and cardiovascular events as well as blood pressure and atrial fibrillation.
Infant health and neurodevelopment
Numerous studies have examined the effects of maternal seafood and omega-3 intakes on infant birth weight, length of gestation, visual and cognitive development, and other infant health outcomes. High concentrations of DHA are present in the cellular membranes of the brain and retina 14, and DHA is important for fetal growth and development. The accumulation of DHA in the retina is complete by birth, whereas accumulation in the brain continues throughout the first 2 years after birth.
In 2016, Agency for Healthcare Research and Quality (AHRQ) published a review on the effects of omega-3 fatty acids on child and maternal health 39. This comprehensive report evaluated the findings from 95 randomized controlled trials and 48 prospective longitudinal studies and nested case-control studies. Most studies examined the effects of fish oil supplements or other DHA and EPA combinations in pregnant or breastfeeding women or of infant formula fortified with DHA plus arachidonic acid, an omega-6. The authors concluded that, except for small beneficial effects on infant birth weight and length of gestation, omega-3 supplementation or fortification has no consistent effects on infant health outcomes.
Recommendations from the Dietary Guidelines for Americans: The 2015–2020 Dietary Guidelines for Americans states that women who are pregnant or breastfeeding should consume 8–12 ounces of seafood per week, choosing from varieties that are higher in EPA and DHA and lower in methyl mercury, such as salmon, herring, sardines, and trout. These women should not consume certain types of fish, such as king mackerel, shark, swordfish, and tilefish that are high in methyl mercury, and they should limit the amount of white (albacore) tuna they consume to 6 ounces a week. The American Academy of Pediatrics has similar advice for breastfeeding women, recommending intakes of 200–300 mg DHA per day by consuming one to two servings of fish per week to guarantee a sufficient amount of DHA in breast milk 40.
[Source 41]Most currently available infant formulas in the United States contain DHA and arachidonic acid. However, the authors of a paper published by the American Academy of Family Physicians and of two Cochrane reviews (one on full-term infants and one on preterm infants) have concluded that the evidence is insufficient to recommend the use of infant formulas that are supplemented with these fatty acids 42, 43, 44.
Cancer prevention
Researchers have hypothesized that higher intakes of omega-3s from either foods or supplements might reduce the risk of cancer due to their anti-inflammatory effects and potential to inhibit cell growth factors 45. For example, some studies have shown associations between higher intakes and/or blood levels of omega-3s and a decreased risk of certain cancers, including breast and colorectal cancers 46. Other studies have found no associations between omega-3s and cancer risk, and some have even found associations in the opposite direction, suggesting that omega-3s might increase the risk of certain cancers such as prostate cancer 47, 48. To date, no large-scale clinical trials have examined the effects of omega-3s on the primary prevention of cancer in the general population, although a large clinical trial that addresses this question, the VITAL trial, is currently underway 38. The VITAL trial will examine the effects of EPA (465 mg/day) and DHA (375 mg/day) supplementation (with and without 2,000 IU/day vitamin D) for 5 years in 25,875 older adults on the primary prevention of cancer and cardiovascular disease 38. Results from this clinical trial will shed more light on possible associations between these omega-3s and cancer.
Results from observational studies however, have been inconsistent and vary by cancer site and other factors, including gender and genetic risk. Overall, data from observational studies show no consistent relationship between omega-3s and overall cancer risk. Although there are some suggestions of reduced risk for breast and possibly colorectal cancers with higher long chain omega-3 intakes, randomized clinical trials are needed to confirm these findings.
Breast cancer
Evidence from several observational studies suggests that higher intakes of long chain omega-3s are associated with a lower risk of breast cancer, but clinical trials are needed to confirm this finding. In the prospective Singapore Chinese Health Study of 35,298 women aged 45–74 years, those in the top three quartiles of dietary long chain omega-3 intake had a 26% lower risk of breast cancer after an average of 5.3 years of follow-up than those in the lowest quartile 49. Similarly, among 35,016 female participants aged 50–76 years in the Vitamins And Lifestyle cohort, those who reported current use of fish-oil supplements had a 32% lower risk of breast cancer after a mean of 6 years than those who did not take fish oil 50.
According to a systematic review of three case-control studies and five prospective studies published in 2007–2011, evidence is increasing that higher intakes of dietary and supplemental LC omega-3s are associated with a lower risk of breast cancer 51. Similarly, the authors of a meta-analysis of data from 21 prospective cohort studies concluded that women with the highest dietary intakes and/or tissue levels of long chain omega-3s had a 14% lower risk of breast cancer than those with the lowest intakes and tissue levels 46. These authors also found a dose-response relationship between higher intakes of combined long chain omega-3s and reduced breast cancer risk. Intakes of ALA and of fish, however, had no association with differences in breast cancer risk. This finding, which could be due to varying levels of omega-3s in different fish species, warrants further investigation.
Colorectal cancer
Limited evidence from observational studies suggests that greater consumption of fish and long chain omega-3s is associated with a reduced risk of colorectal cancer 51.
The authors of a meta-analysis of 19 prospective cohort studies found no significant association between fish intake and risk of colorectal cancer overall. However, a stratified analysis showed that for participants with the highest fish consumption (those who ate fish at least seven times more often per month than those with the lowest fish consumption), the risk of colorectal cancer was 22% lower than that for the lowest fish consumers 52. Results from a more recent systematic review and meta-analysis of 22 prospective cohort studies and 19 case-control studies indicate that fish consumption is inversely associated with colorectal cancer risk. In this analysis, 21 of the studies distinguished between colon cancer and rectal cancer. The risk of rectal cancer was 21% lower for participants with the highest fish intakes (as much as one serving/day) compared to those with the lowest fish intakes (as little as none), but fish consumption had no significant association with risk of colon cancer alone 53.
Results from the Vitamins And Lifestyle cohort study suggest that associations between fish or LC omega-3 intakes and colorectal cancer risk might vary by such factors as gender and genetic risk. In this study, researchers evaluated associations between colorectal cancer risk and EPA/DHA intakes from fatty fish (salmon and fresh tuna) and fish oil supplements in 68,109 Washington residents aged 50–76 54. The amount of fatty fish consumed ranged from none to 0.8 servings per week or more. Overall, EPA and DHA intakes (from either diet or supplements) and fatty fish consumption were not associated with colorectal cancer risk, but associations varied by genetic characteristics (certain inherited genetic mutations are associated with an increased risk of colorectal cancer). For individuals in the lowest two tertiles of genetic risk, higher fatty fish consumption and higher total EPA and DHA intakes were inversely associated with colorectal cancer risk. For individuals in the highest tertile of genetic risk, higher total EPA and DHA intakes were positively associated with colorectal cancer risk. Risk also varied by gender. Among men, use of fish oil supplements reduced colorectal cancer risk by an average of 34% or more depending on the frequency and duration of use, but this effect did not occur among women. Additional research is needed to clarify possible associations between fish and omega-3 intakes and colorectal cancer risk.
Prostate cancer
Several prospective and case-control studies have investigated associations between either blood levels or intakes of omega-3s and risk of low-grade or high-grade prostate cancer. Results from these studies have been inconsistent.
A few case-control and case-cohort studies have found positive associations between blood levels of LC omega-3s and prostate cancer risk (particularly high-grade disease that is more advanced and more likely to spread than low-grade cancer), suggesting that omega-3s might increase prostate cancer risk. In a nested case-control analysis of men aged 55–84 years participating in the Prostate Cancer Prevention Trial, serum phospholipid levels of DHA were positively associated with risk of high-grade, but not low-grade, prostate cancer 55. Serum EPA levels, however, were not associated with risk of either grade of the disease.
Similarly, results from a case-cohort study within the Selenium and Vitamin E Cancer Prevention (SELECT) trial showed that men in the highest quartile of plasma phospholipid LC omega-3s had a 44% higher risk of low-grade prostate cancer and a 71% higher risk of high-grade prostate cancer than those in the lowest quartile 48. An analysis of data from the European Prospective Investigation into Cancer and Nutrition cohort also found a higher prostate cancer risk in men with higher plasma levels of LC omega-3s 56. Among whites participating in the Multiethnic Cohort Study, higher levels of omega-3s in erythrocyte membranes and higher ratios of omega-3s to omega-6s were both associated with an increased risk of prostate cancer. However, the results showed no associations, even with advanced or high-grade disease, for other ethnic groups or for the population as a whole 57.
Although the findings from the Prostate Cancer Prevention Trial and the SELECT trial suggest that higher LC omega-3 intakes might increase prostate cancer risk, some scientists have questioned the significance of these findings 58. They have noted, for example, that in the SELECT trial 48, the difference in the omega-3 levels in the men with and without prostate cancer was very small and of questionable physiological significance. Other scientists have pointed out that localized (even high-grade) prostate cancers usually progress slowly and are common on autopsy in men who have died from other causes, suggesting that prostate cancer mortality is a more critical endpoint than prostate cancer incidence 59. Finally, desaturation enzymes that convert ALA into EPA and DHA can be upregulated in some cancer cells, suggesting the possibility that it was the disease that raised the omega-3 levels, not the omega-3 levels that raised the disease risk 60.
Results from other observational studies using dietary intake data suggest that higher intakes of fish and/or omega-3s reduce prostate cancer risk. Both fish and omega-3 consumption were associated with a lower risk of fatal prostate cancer in a cohort of 293,464 men participating in the NIH-AARP study 61. In the Health Professionals Follow-up Study, a prospective cohort of over 47,000 men aged 40–75 years, those who consumed fish more than three times per week had a lower risk of metastatic prostate cancer than those who consumed fish less than twice per month 62. However, men who used fish oil supplements did not have a decreased risk of prostate cancer.
A number of systematic reviews and meta-analyses of prospective studies of the effects of fish intakes, omega-3 intakes, and omega-3 blood levels on prostate cancer risk have had inconsistent findings as well. For example, circulating levels of EPA, but not DHA, were positively associated with prostate cancer risk in a meta-analysis of 5,098 men with prostate cancer and 6,649 men without prostate cancer from seven studies 63. Another meta-analysis of 12 studies that included 4,516 men with prostate cancer and 5,728 men without prostate cancer found that high serum levels of these LC omega-3s were positively associated with high-grade disease 64. In other analyses, dietary intakes of LC omega-3s had no effect on prostate cancer risk 65, whereas fish consumption decreased prostate cancer mortality but had no effect on prostate cancer incidence 66. A 2015 meta-analysis found no significant associations between dietary intakes or blood levels of long chain omega-3s and total prostate cancer risk 67. The authors noted that most dietary-intake studies included in their meta-analysis found inverse associations, whereas biomarker studies of blood levels of these fatty acids found positive associations.
Overall, the evidence to date shows no consistent relationships between prostate cancer risk or mortality and omega-3 intakes or blood levels.
Other cancers
Evidence is limited for a role of omega-3s in the prevention of cancers at other sites. For example, evidence is insufficient to determine whether omega-3s affect the risk of skin cancers, including basal-cell carcinoma, squamous-cell carcinoma, and melanoma 68. Findings from the Australian Ovarian Cancer Study suggest that there is no association between total or individual omega-3 intakes from foods and ovarian cancer risk 69.
Associations between omega-3 intakes and endometrial cancer have been mixed. Some evidence indicates that dietary intakes of EPA and DHA may provide protection from the development of endometrial cancer 70. Other evidence indicates that they decrease risk in normal-weight women but have no effect or even increase risk in overweight or obese women 71.
A systematic review and meta-analysis of 9 prospective cohort and 10 case-control studies did not find an association between fish or long chain -omega-3 intakes and risk of pancreatic cancer 72. Similarly, systematic reviews and meta-analyses have not found significant associations between fish consumption and risk of gastric or esophageal cancers 73.
Summary
Overall, data from observational studies show no consistent relationship between omega-3s and overall cancer risk. Although there are some suggestions of reduced risk for breast and possibly colorectal cancers with higher long chain omega-3 intakes, randomized clinical trials are needed to confirm these findings.
The VITAL trial will examine the effects of EPA (465 mg/day) and DHA (375 mg/day) supplementation (with and without 2,000 IU/day vitamin D) for 5 years in 25,875 older adults on the primary prevention of cancer and cardiovascular disease 38. Results from this clinical trial will shed more light on possible associations between these omega-3s and cancer.
Alzheimer’s disease, dementia, and cognitive function
Some, but not all, observational studies suggest that diets high in long chain omega-3s are associated with a reduced risk of cognitive decline, Alzheimer’s disease, and dementia 74, 75. Because DHA is an essential component of cellular membrane phospholipids in the brain, researchers hypothesize that long chain omega-3s might protect cognitive function by helping to maintain neuronal function and cell- membrane integrity within the brain 75. This hypothesis is supported by findings from case-control studies indicating that patients with Alzheimer’s disease have lower serum levels of DHA than cognitively healthy people 76, 77. Lower serum DHA levels are also associated with more cerebral amyloidosis (build-up of protein deposits called amyloids) in healthy older adults, whereas higher DHA is correlated with preservation of brain volume 78.
Several systematic reviews and meta-analyses, including a Cochrane review, have assessed the effects of omega-3 supplementation on cognitive function and dementia in healthy older adults and those with Alzheimer’s disease or cognitive impairment 75, 79, 80, 81. Overall, the findings indicate that LC omega-3 supplementation does not affect cognitive function in healthy older adults or in people with Alzheimer’s disease compared to placebo. For people with mild cognitive impairment, omega-3s may improve certain aspects of cognitive function, including attention, processing speed, and immediate recall 81. However, these findings need to be confirmed in additional clinical trials.
Age-Related Macular Degeneration (AMD)
Age-Related Macular Degeneration (AMD) is a major cause of vision loss among older adults. In most cases, severe vision loss is associated with advanced AMD, which consists of either central geographic atrophy (dry AMD, the most common form) or neovascular AMD (wet AMD). Based on DHA’s presence as a structural lipid in retinal cellular membranes and the beneficial effects of EPA-derived eicosanoids on retinal inflammation, neovascularization, and cell survival, researchers have suggested that these long chain omega-3s have cytoprotective effects in the retina that may help prevent the development or progression of AMD 82.
Results from observational studies suggest that people who consume higher amounts of fatty fish and/or dietary long chain omega-3s have a lower risk of developing AMD. In the cross-sectional EUREYE study of 2,275 participants aged 65 years or older, those who ate fatty fish at least once per week had a 53% lower risk of neovascular AMD than those who consumed fatty fish less often 83. Results were similar in a study in 681 elderly male twins 84 and an analysis of 38,022 healthy female health professionals 85. In the latter study, women in the highest tertiles of dietary DHA plus EPA intake (median of 330 mg/day) had a 38% lower risk of developing AMD during an average of 10 years of follow-up than those in those in the lowest tertile (median intake of 80 mg/day). Higher serum and erythrocyte membrane levels of EPA (but not DHA) have also been associated with a lower risk of neovascular AMD 86.
In the AREDS study, a dietary supplement formulation containing 15 mg beta-carotene, 400 IU vitamin E, 500 mg vitamin C, 80 mg zinc, and 2 mg copper reduced the risk of advanced AMD in people with intermediate AMD or advanced AMD in one eye 87. Data from a nested cohort study within the AREDS population indicated that participants who reported the highest omega-3 intakes were about 30% less likely to develop central geographic atrophy and neovascular AMD than other participants 88.
These findings, combined with other epidemiological evidence, formed the basis for the AREDS2 clinical trial that examined whether adding 350 mg DHA and 650 mg EPA to the AREDS formulation further reduced the risk of progression to advanced AMD 89. The results showed that EPA and DHA did not provide any additional benefits after a median follow-up of 5 years. These findings are in line with those from a Cochrane review 90 that included the results from AREDS2 and the Nutritional AMD Treatment 2 study 91, a 3-year randomized clinical trial of LC omega-3 supplements (840 mg/day DHA and 270 mg/day EPA) in patients with early age-related maculopathy and neovascular AMD. The Cochrane review authors concluded that long chain omega-3 supplementation for up to 5 years in people with AMD does not reduce the risk of progression to advanced AMD or of moderate to severe vision loss.
Dry eye disease
About 14% of adults in the United States have dry eye disease, a chronic condition in which decreased tear volume and quality leads to ocular surface inflammation and damage, causing discomfort and visual impairment 92. Older women, in particular, have a higher risk of dry eye disease than other groups, possibly because of hormonal changes that affect the tear-producing glands 92. Researchers hypothesize that omega 3s—particularly EPA and DHA—might reduce the risk of dry eye disease and relieve its symptoms because of their anti-inflammatory activity, and many patients take them as adjunctive treatments to artificial tears and other medications.
Some, but not all, observational studies show inverse associations between self-reported dietary consumption of omega-3s and risk of dry eye disease. For example, in a cross-sectional study of 32,470 women aged 45–84 participating in the Women’s Health Study, those in the highest quintile of total dietary omega-3 intake (mean of 1,990 mg/day) had a 17% lower risk of dry eye disease than those in the lowest quintile (mean intake of 920 mg/day) 93. The study found a similar association for DHA—women in the highest versus the lowest quintiles of DHA intake had a 12% lower risk of dry eye disease; however, the results showed no significant associations for EPA. But in another cross-sectional study of 322 postmenopausal women, total dietary omega-3 intakes were not correlated with the prevalence of dry eye disease 94.
Results from clinical trials using omega-3 supplementation, primarily EPA and DHA, have had mixed results in reducing the symptoms and signs of dry eye disease. Furthermore, there is no consensus on the optimal dose, composition, or length of omega-3 treatment for this condition 95.
The studies that have found beneficial effects from omega-3 supplementation for symptoms and signs of dry eye disease include one showing that daily supplementation with 1,000 mg omega-3s (650 mg EPA plus 350 mg DHA) for 3 months in 518 men and women (mean age about 40 years) living in northern India reduced symptoms and some signs of dry eye disease compared with placebo 96. In another clinical trial of 105 men and women, daily treatment with supplements containing 2,240 mg omega-3s (1,680 mg EPA and 560 mg DHA as re-esterified triglycerides) for 12 weeks also reduced symptoms of dry eye disease compared with placebo 97. In addition, the supplements increased tear break-up time and decreased tear osmolarity (which would be likely to reduce ocular surface damage).
However, another large, randomized, double-blind clinical trial conducted in the United States found that EPA and DHA from fish oil supplements are no better than placebo at relieving symptoms or signs of dry eye disease 92. This 12-month trial included 535 participants (about 81% female) aged 18 years or older (mean age about 58 years) with at least a 6-month history of moderate to severe dry eye disease. Among them, 349 participants received daily supplements of 3,000 mg omega-3s (2,000 mg EPA plus 1,000 mg DHA), and 186 received a placebo containing 5,000 mg olive oil. Participants could continue taking medications for dry eyes, including artificial tears and prescription anti-inflammatory eye drops, as well as omega-3 supplements as long as the total dose of EPA plus DHA was less than 1,200 mg per day. At the end of the study, symptoms were less severe than at baseline in both groups, but the results showed no significant differences between groups. Groups also showed no significant differences compared with baseline in signs of dry eye disease, including conjunctive and cornea integrity as well as tear volume and quality.
Overall, the evidence to date shows no consistent relationship between omega-3s and dry eye disease. More research is warranted to fully understand whether increased intakes of dietary or supplemental omega-3s help reduce the risk of dry eye disease and whether they are beneficial as an adjunct treatment.
Rheumatoid arthritis
Rheumatoid arthritis (RA) is an autoimmune disease characterized by chronic inflammation of the joints. Its symptoms include pain, swelling, stiffness, and functional impairments. RA is typically treated with nonsteroidal antiinflammatory drugs (NSAIDs), corticosteroids, and disease-modifying antirheumatic drugs. Due to their antiinflammatory effects, some scientists hypothesize that long chain omega-3s reduce some of the symptoms of rheumatoid arthritis and patients’ reliance on NSAIDs and corticosteroids.
A 2012 systematic review concluded that the types of omega-3s found in seafood and fish oil may be modestly helpful in relieving symptoms of rheumatoid arthritis. In the studies included in the review, many of the participants reported that when they were taking fish oil they had briefer morning stiffness, less joint swelling and pain, and less need for anti-inflammatory drugs to control their symptoms.
Reviews and meta-analyses of studies that assessed whether fish oil and long chain omega-3s are beneficial for rheumatoid arthritis have had inconsistent findings. Some suggest that they do not significantly affect the clinical symptoms of RA but do reduce the amounts of NSAIDs and corticosteroids that patients need. Others indicate that LC omega-3s reduce joint swelling and pain, morning stiffness, and number of painful joints in addition to reducing NSAID use. Some researchers suggest that differences in findings could be due in part to whether patient-determined use of NSAIDs is considered a measure of pain.
Findings to date suggest that long chain omega-3s may be helpful as an adjunctive treatment to pharmacotherapy for ameliorating the symptoms of rheumatoid arthritis 98, 99. However, more research is needed to confirm this finding.
Depression
A 2016 meta-analysis of 26 studies found a 17% lower risk of depression with higher fish intake 100. However, a 2015 Cochrane review of 26 studies found insufficient evidence to determine whether omega-3s (1,000 to 6,600 mg/day EPA, DHA, and/or other omega-3s) are beneficial for major depressive disorder in adults 101. The authors did find a small-to-modest beneficial effect on depressive symptoms, but they concluded that this effect was not clinically significant.
Bipolar disorder
This systematic review (source 102) investigated the efficacy of omega-3 fatty acids for bipolar disorder. Five randomised controlled trials met inclusion criteria for the review. Only one trial provided data that could be analysed, investigating ethyl-EPA as an adjunctive treatment in a mixed outpatient population. Some positive benefits were found for depressive symptoms but not for mania, and no adverse events were reported. There is currently insufficient evidence on which to base any clear recommendations concerning omega-3 fatty acids for bipolar disorder. However, given the general health benefits and safety of omega-3, the preliminary evidence from this review provides a strong case for well-powered, high-quality trials in specific index populations.
Results from one study showed positive effects of omega-3 as an adjunctive treatment for depressive but not manic symptoms in bipolar disorder. These findings must be regarded with caution owing to the limited data available. There is an acute need for well-designed and executed randomised controlled trials in this field.
Inflammatory bowel disease
The authors of a systematic review of 19 randomized controlled trials concluded that the available evidence does not support the use of omega-3 supplements to treat active or inactive inflammatory bowel disease 103. Similarly, the authors of a Cochrane review concluded that, based on the evidence from two large, high-quality studies, omega-3 supplements are probably not effective for maintaining remission in people who have Crohn’s disease 104.
Autism Spectrum Disorders
It has been suggested that difficulties associated with autism spectrum disorders may be explained in part by lack of omega-3 fatty acids, and that supplementation of these essential fatty acids may lead to improvement of symptoms. To date there is no high quality evidence that omega-3 fatty acids supplementation is effective for improving core and associated symptoms of autism spectrum disorder. Given the paucity of rigorous studies in this area, there is a need for large well-conducted randomised controlled trials that examine both high and low functioning individuals with autism spectrum disorder and that have longer follow-up periods 105.
ADHD
A systematic review and meta-analysis of 10 studies in children with ADHD or related neurodevelopmental disorders, such as developmental coordination disorder, found no improvements with omega-3 supplementation on measures of emotional lability, oppositional behavior, conduct problems, or aggression 106. However, in subgroup analyses of only the higher-quality studies and those with strict inclusion criteria, omega-3 supplementation (60 to 1,296 mg/day EPA and/or DHA) did significantly improve parent-rated emotional lability and oppositional behavior.
Childhood allergies
A systematic review and meta-analysis of 10 prospective cohort studies and 5 randomized clinical trials on omega-3 intakes during pregnancy and outcomes of childhood allergic disease (eczema, rhino-conjunctivitis, and asthma) found inconsistent results 107. Although the authors could not draw firm conclusions due to the heterogeneity of the studies and their results, they concluded that the overall findings were “suggestive” of a protective association between higher maternal intakes of LC omega-3s or fish and incidence of allergic disease symptoms in the offspring. The authors of a Cochrane review that included eight LC omega-3 supplementation trials concluded that there is limited evidence to support the use of LC omega-3 supplements by women during pregnancy and/or lactation for reducing the risk of allergic disease in their children 108.
Cystic fibrosis
A Cochrane review of four studies of cystic fibrosis found that omega-3 supplements (300 to 5,400 mg/day EPA and/or DHA) might improve lung function and increase blood levels of essential fatty acids in people with cystic fibrosis 109. However, the authors concluded that there is not enough evidence to recommend routine use of omega-3 supplements by people with cystic fibrosis.
What kinds of omega-3 dietary supplements are available ?
Long chain omega-3s are present in several dietary supplement formulations, including fish oil, krill oil, cod liver oil, and vegetarian products that contain algal oil. Commonly used dietary supplements that contain omega-3s include fish oil (which provides EPA and DHA) and flaxseed oil (which provides ALA). Algae oils are a vegetarian source of DHA.
Fish liver oils, such as cod liver oil, are not the same as fish oil. Fish liver oils contain vitamins A and D as well as omega-3 fatty acids 110. Both of these vitamins can be toxic in large doses 110. The amounts of vitamins in fish liver oil supplements vary from one product to another.
A typical fish oil supplement provides about 1,000 mg fish oil, containing 180 mg EPA and 120 mg DHA, but doses vary widely 111. Cod liver oil supplements provide vitamin A and vitamin D in addition to long chain omega-3s. Although seafood contains varying levels of methyl mercury (a toxic heavy metal) 112, omega-3 supplements have not been found to contain this contaminant because it is removed during processing and purification 113.
- Formulations of omega-3 dietary supplements vary widely, so it is important to check product labels to determine the types and amounts of omega-3s in these products. The Dietary Supplement Label Database 111 from the National Institutes of Health contains label information from many dietary supplements on the market that contain omega-3s.
Dietary supplements can contain several different forms of omega-3s, including natural triglycerides, free fatty acids, ethyl esters, re-esterified triglycerides, and phospholipids 113, 114. Natural triglycerides are the form that occur naturally in fish oil, whereas ethyl esters are synthesized from natural triglycerides by replacement of the glycerol molecule of the triglyceride with ethanol. Re-esterified triglycerides are formed by the conversion of ethyl esters back to triglycerides. Omega-3s as re-esterified triglycerides, natural triglycerides, and free fatty acids have somewhat higher bioavailability than ethyl esters, but consumption of all forms significantly increases plasma EPA and DHA levels 115, 116.
Krill oil contains omega-3s primarily as phospholipids, and limited research suggests that these have somewhat higher bioavailability than the omega-3s in fish oil 117, 118, 119.
Plant-based sources of omega-3s from algal oil usually provide around 100–300 mg DHA; some contain EPA as well. These supplements typically contain omega-3s in the triglyceride form 120. According to a small study, the bioavailability of DHA from algal oil is equivalent to that from cooked salmon 121.
Fish oil supplement side effects
In general taking fish oil supplement is quite safe. Commonly reported side effects of omega-3 supplements are usually mild. These include unpleasant taste, bad breath, heartburn, nausea, gastrointestinal discomfort, diarrhea, headache, and odoriferous sweat 81, 104.
Common gastrointestinal symptoms like nausea occurred at rates of ≈4% at dosages <3 g/d and increased to ≈20% at a dosage of 4 g/d.
The Institute of Medicine did not establish a UL for any omega-3s, although it noted that high doses of DHA and/or EPA (900 mg/day of EPA plus 600 mg/day DHA or more for several weeks) might reduce immune function due to suppression of inflammatory responses. Doses of 2–15 g/day EPA and/or DHA might also increase bleeding time by reducing platelet aggregation 14. However, according to the European Food Safety Authority, long-term consumption of EPA and DHA supplements at combined doses of up to about 5 g/day appears to be safe 122. It noted that these doses have not been shown to cause bleeding problems or affect immune function, glucose homeostasis, or lipid peroxidation. The FDA recommends not exceeding 3 g/day EPA and DHA combined, with up to 2 g/day from dietary supplements. Some doses used in clinical trials exceed these levels.
Omega-3 supplements may extend bleeding time (the time it takes for a cut to stop bleeding). People who take drugs that affect bleeding time, such as anticoagulants (“blood thinners”) or nonsteroidal anti-inflammatory drugs (NSAIDs), should discuss the use of omega-3 fatty acid supplements with a health care provider.
Krill oil vs Fish oil
Krill (Euphausia superba) are small red-colored crustaceans, similar to shrimp, that flourish in the extremely cold waters of the Antarctic Ocean. Similar to fish, krill consume a diet rich in omega-3 fatty acids (eg, algae) and are thereby a natural source of EPA and DHA 123.
Although there are similarities, numerous differences exist between krill oil and fish oil. Krill oil is only available as a supplement, primarily in softgel formulation 124. The size of krill oil softgels is markedly smaller and may be more suitable for individuals with swallowing difficulties. On the other hand, fish oil is available in both prescription and supplement formulations including softgels and liquid 124. More important, however, are the differences in omega-3 content and delivery 123. The EPA+DHA content of krill oil is markedly less per softgel (and serving) compared to fish oil. For example, the dose of EPA+DHA of pure krill oil products may range from ~45 to 200 mg/softgel compared to ~300 to 2,250 mg/softgel or teaspoon among the various fish oil formulations 125, 126, 127.
Krills survival in such a frigid environment is attributable to krill having a high content of long-chain polyunsaturated fatty acids, including eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), attached to their cell membranes via phospholipids (primarily phosphatidylcholine). This structure provides optimum membrane fluidity in cold temperatures and an ideal source of omega-3 fatty acids 128. Fish oil contains abundant amounts of Omega-3 PUFAs, most of which is stored as triglycerides. Animal and human studies indicate long-chain polyunsaturated fatty acids bound to phospholipids, like those found in krill oil, have better absorption and delivery to the brain than their methyl ester or triglyceride-formed fish oil counterparts 129, 130, 131.
The fatty acid form is also a key factor differentiating these agents. The EPA+DHA found in krill oil is primarily incorporated in phospholipids, whereas omega-3 fatty acids in fish oil are generally present as triglycerides or ethyl esters 132. Some data have suggested that phospholipids are more efficient at delivering EPA+DHA 133. This difference in chemical form may therefore impact the absorption and bioavailability of the EPA+DHA and the dose required to achieve the various cardiovascular effects.
Although a limited number of studies comparing the effects of krill oil and fish oil on the omega-3 index have suggested greater increases with krill oil 134, 135 newer data do not support these findings 136.
Unlike fish oil, krill oil also contain a potent antioxidant carotenoid, astaxanthin, that helps prevent long-chain polyunsaturated fatty acids oxidation 137. This colorful carotenoid antioxidant, astaxanthin is responsible for the red color observed in salmon meat, cooked shellfish, and certain krill oil supplements. Although this powerful antioxidant has previously demonstrated HDL “good” cholesterol increasing properties, clinical benefits and therapeutic use of astaxanthin remain unclear 138.
Cost may also be an important consideration for patients choosing omega-3 products. Typically, supplemental fish oil is considerably less expensive compared to krill oil. A cost analysis of common products indicated that fish oil supplements range from 1 to 15 cents while krill oil is approximately 30 cents per 100 mg of EPA+DHA 125.
Krill Oil Mechanism of Action
Most of krill oil’s health benefits are attributed to its high EPA and DHA content. Numerous studies have identified the antithrombotic, antiarrhythmic, antiatherosclerotic, and anti-inflammatory mechanisms associated with an increased consumption of these omega-3 fatty acids 139, 140, 141.
More studies are needed to determine whether phosphatidylcholine and astaxanthin provide an additive effect.
Phosphatidylcholine by itself has shown promise as a dietary supplement to improve homocysteine status, liver disorders, and respiratory distress 142.
Astaxanthin may have independent positive effects due to its antioxidant capabilities. When oxygen radical absorptive capacity values of various antioxidants were compared, astaxanthin was found to be 34-times more effective than CoQ10 and 48-times more effective than fish oil 143.
Preliminary human studies indicate krill oil may be superior to fish oil in the reduction of premenstrual syndrome (PMS) complications 144 and biomarkers of dyslipidemia 137.
Krill oil consumption has also shown a positive effect on markers of inflammation 145.
Krill Oil vs Fish Oil on Cholesterol
Researchers in Quebec compared Neptune Krill Oil (NKO®) with high EPA and DHA fish oil (3:2 ratio) or placebo in a 12-week, prospective, randomized, double-blind clinical trial 137. 120 participants ages 25-75, with moderate-to-high cholesterol (193.9-347.9 mg/dL) and triglycerides (203.8-354.4 mg/dL) for a minimum of six months, were divided into four groups.
- Group 1 received 2 or 3 gram Neptune Krill Oil daily, depending on body mass index (BMI).
- Group 2 received 1 or 1.5 g Neptune Krill Oil daily(depending on BMI) during the 12 weeks of the study and an additional 500 mg Neptune Krill Oil as a maintenance dose for a 90 day follow-up period. In bothgroups a BMI >30 justified the higher of the two doses.
- Group 3 received 3 g Fish Oil (180 mg EPA, 120 mg DHA per g) daily, while
- Group 4 received aplacebo.
Parameters examined at baseline, 30, and 90 days (180 days for the follow-up in group 2) included HDL (good cholesterol), LDL (bad cholesterol) and total cholesterol, blood glucose, and triglycerides. At the end of 90 days those taking krill oil at a daily dose of 1 g, 1.5 g, 2 g, or 3 g achieved significant reductions of LDL of 32 %, 36 %, 37% and 39 percent, respectively. Reductions in total cholesterol were 13.4%, 13.7%, 18.1% and 18 percent, respectively, while triglyceride reductions were 11.03%, 11.89%, 27.62%, and 26.51 percent respectively. Blood glucose was reduced 6.3 percent in the 1-g and 1.5-g krill group, and 5.6 percent in those taking 2 or 3 g krill daily.
In comparison, the fish oil group had an average 5.9% reduction in cholesterol and the placebo group had a 9.1-percent increase. Subjects taking the fish oil also had a non-significant 4.6-percent decrease in LDL cholesterol.
Those receiving the placebo had a significant 13% increase in LDL cholesterol. There was a non-significant decrease in glucose and triglycerides for both the 3 g fish oil and placebo groups.
The largest changes in serum lipids occurred for HDL cholesterol. After 90 days, the 1, 1.5, 2, and 3 g krill oil groups had significant HDL “good” increases of 44%, 43%, 55% and 59 percent, while the fish oil group had only a 4.2% increase; the placebo group had a non-significant decrease of HDL.
During the 12-week follow-up, when subjects in the 1- and 1.5-g krill group were maintained on 500 mg krill oil daily, all markers maintained statistical significance except glucose 137.
While the findings for krill oil were impressive, the magnitude of the changes in lipid measures was considerably higher than what one might expect. Additionally, comparison is limited by the fact that the EPA and DHA content of the krill oil supplements was not provided. Thus these findings require further validation.
Another open-label randomized parallel study 135 examined the comparative effects of krill oil and fish oil in 122 participants documented to have no other routine sources of omega-3s. Participants were randomized to 1 of 3 groups: krill oil 3 g daily, fish oil 1.8 g daily, or no supplementation for 7 weeks. A total of 115 participants finished the study. The krill oil used in this study was extracted from Antarctic krill. The daily dose was 6 capsules, each containing 500 mg of oil providing 90.5 mg EPA + DHA and 103.5 mg of omega-3 polyunsaturated fatty acids (PUFAs). The omega-3 fish oil source was a Norway product given in a daily dose of 3 capsules containing 600 mg each of fish oil with 288 mg EPA + DHA and 330 mg of omega-3 PUFAs. In total, the daily amount of krill oil was approximately 63% of the dosage contained in the fish oil 135.
Blood samples were taken after an overnight fast of at least 12 hours at baseline and at the final visit. Plasma levels of EPA, DHA, and docosapentaenoic acid (DPA) increased significantly during the intervention in both the krill oil and fish oil groups compared to control. During the intervention phase, small changes were seen in levels of HDL “good” cholesterol, LDL “bad” cholesterol, and triglycerides. However, the only significant change was the increase in LDL-Cholesterol within the fish oil group. No differences were seen in the HDL-C / triglyceride ratio within the fish oil or control groups. Conversely, the ratio was significantly increased in the krill oil group. Changes in apolipoprotein B (Apo B-100) were minor and nonsignificant for all study groups. Only the within change in Apo B-100 levels for the krill oil group was significant. In addition, no significant increases were seen among the 3 groups in markers for inflammation, hemostasis, or oxidative stress. Withdrawal rates were similar among the 3 groups. The comparable increases in EPA and DHA among the krill oil users despite a lower dose of omega-3 PUFAs compared to the fish oil group are consistent with earlier studies suggesting that krill oil is a more bioavailable source of omega-3 PUFAs 135.
Another study specifically addressed the impact of krill oil on serum triglycerides and the omega-3 index 146. In a double-blind, randomized, placebo-controlled trial, 300 patients with triglyceride levels of 150 to 499 mg/dL were given varying doses of krill oil (0.5, 1, 2, or 4 g per day) or olive oil (placebo) for 12 weeks. These amounts of krill oil corresponded to 100, 200, 400, or 800 mg of EPA+DHA per day. Participants reported low consumption of fatty fish defined as less than twice per month. Fasting triglyceride levels were measured at baseline, 6, and 12 weeks. Compared to placebo, patients on krill oil demonstrated a 10.2% reduction in triglyceride levels 146. No differences in LDL-C levels were observed. Perhaps the most important finding from this study was the significant increase in the omega-3 index in all krill oil groups seen at both 6 and 12 weeks. The corresponding changes were 8%, 18%, 29%, and 73% for the 0.5, 1, 2, and 4 g krill oil dose compared to -3% (minus 3 percent) with placebo. A major limitation of this study is the high intra-individual variability that occurred in the fasting triglyceride measurements. Overall, the average daily dose of krill oil was 1.875 g providing 385 mg of EPA+DHA. This resulted in approximately a 6% reduction in triglyceride levels compared to baseline 146. This daily dose is lower than that used in earlier studies evaluating triglycerides. Additional studies with multiple measurements of triglyceride levels for each individual will be needed to confirm these findings.
The minimal number of clinical studies and substantial heterogeneity of designs preclude any definitive conclusions. Taken as a whole, however, these randomized trials provide preliminary evidence that krill oil is at least equivalent to and perhaps better than fish oil at increasing blood levels of EPA and DHA 147. However, it is not known whether these changes would translate into improved cardiovascular outcomes. Additional studies are needed to further explore the effects of krill oil on the lipid panel and, in particular, the potential impact on triglyceride reduction.
The American Heart Association position statement, published in 2002 28, does not recognize krill oil as an omega-3 supplement. This is understandable considering that the emergence of krill oil began after the American Heart Association recommendations. However, no other major position statements regarding cholesterol or cardiovascular risk prevention that have been published more recently address the appropriate use of krill oil, including the 2013 American College of Cardiology and the American Heart Association cholesterol guidelines 148. As such, krill oil should not be considered first-line therapy for omega-3 intake and cardioprotection. Additionally, krill oil does not appear to be effective for clinically relevant triglyceride lowering, because randomized controlled trials indicate only modest reductions 149. Therefore, krill oil cannot be recommended for hypertriglyceridemia at this time.
Because of these factors, krill oil currently has a limited role in cardiovascular risk prevention, with the potential to expand as more research is completed. Most studies evaluating the impact of krill oil on the omega-3 index indicate efficient absorption of EPA+DHA. The omega-3 index is reflective of the EPA+DHA consumption recommended by the American Heart Association and the potential for cardioprotection 150. Thus, it is reasonable to consider krill oil as an alternative for achieving cardiovascular benefits in patients who are unable to increase fish consumption or utilize fish oil. Specific patient groups that may be candidates for krill oil include those with an aversion to dietary fish and individuals unable to swallow the large fish oil softgels or who have a dislike for the liquid. If the efficient uptake of EPA+DHA from krill oil that has been observed in smaller studies is replicated in larger randomized controlled trials, patients may be able to obtain cardioprotection at a lower EPA+DHA intake than what is currently recommended by the American Heart Association. Additionally, krill is sensitive to degradation and the process for obtaining the key components is more complex than that of fish oil. In view of these issues, we emphasize the importance of getting your krill oil products only from reliable manufacturers.
Krill Oil and Osteoarthritis
A double-blind, placebo-controlled, randomized, prospective study examined the effect of 300 mg Neptune Krill Oil daily on C-reactive protein (CRP) and
functional testing scores for arthritis 145.
Ninety participants had a diagnosis of cardiovascular disease and/or rheumatoid arthritis and/or osteoarthritis, and an elevated CRP (>1.0 mg/dL) for three consecutive weeks. Western Ontario and McMaster Universities (WOMAC) osteoarthritis score and CRP were measured at baseline and at days 7, 14, and 30.
After seven days of krill supplementation, CRP decreased by 19.3 percent in the experimental group, while it increased by 15.7 percent in the control group. By day 14 and day 30, CRP decreased by 29.7 and 30.9 percent, respectively, in the krill oil group, while the placebo group experienced an increase of 32.1 percent by day 14 and a lowering to a total increase of 25.1 percent by day 30. When comparing krill supplementation to placebo, differences at day 7, day 14 and day 30 were all statistically significant. When compared to placebo, WOMAC osteoarthritis score results showed krill significantly reduced pain scores and stiffness scores and functional145.
Krill Oil vs Fish Oil on Premenstrual Syndrome
In a 90-day study, Sampalis et al. 144 compared the effects of Neptune Krill Oil to fish oil on multiple parameters of function in Premenstrual Syndrome (PMS) via an assessment questionnaire and the total consumption of analgesics for pain and discomfort associated with PMS.
Seventy subjects of child-bearing age were assigned to ingest 2 grams krill oil daily (800 mg phospholipids, 600 mg EPA and DHA) (n=36) or 2 grams fish oil daily (600 mg EPA and DHA at an 3:2 ratio) (n=34) for the first 30 days of the trial. In the final 60 days both groups were instructed to take the assigned supplements eight days prior to and two days during menstruation.
Questionnaires were completed and analgesic medication intakes were measured at baseline, 45, and 90 days.
After the first 45 days the krill oil group showed significant for all parameters improvements in breast tenderness, joint pain, weight gain, abdominal pain, swelling, and bloating, as well as feelings of being overwhelmed, stressed, irritable, and depressed. The fish oil group demonstrated significant improvements in weight gain and abdominal pain at day 45, and improvements in weight gain, abdominal pain, and swelling at day 90. When consumption of analgesic medication during menstruation by the krill oil group was compared to the fish oil group, it was found that the average consumption of 1.2 g ibuprofen or 2.5 g
acetaminophen taken at baseline changed in both groups. The krill oil group had reduced consumption to 0.9 g ibuprofen and 1.5 g acetaminophen at day 45, and 0.6 g ibuprofen and 1.0 g acetaminophen at day 9) – a total decrease in analgesics of 50 percent after 90 days. The fish oil group decreased intake to 0.9 g ibuprofen and 1.65 g acetaminophen by day 45, and 0.8 g ibuprofen and 1.48 g acetaminophen by day 90 – a total reduction of 33% and 41 percent, respectively.
Interestingly, the authors noted, “Patients taking NKO did not experience any gastrointestinal difficulties such as regurgitation, while 64 percent of the women in the fish oil group complained of unpleasant reflux” 144.
Krill Oil vs Fish Oil on Human Brain Function in Healthy Elderly Volunteers
Aging reduces regional cerebral blood flow, which causes deterioration of cerebral function 151. Aging has also been shown to reduce the level of dorsolateral prefrontal cortex activity during execution of working memory tasks 152.
In a study involving 45 healthy male subjects in their 60s and 70s who had retired from employment in the Japanese business sector 153. Eligibility for participation was as follows: right-handedness, not requiring prescription medication, no history of mental disorders or cerebrovascular disease, absence of serious liver, kidney, heart, respiratory, endocrine, or metabolic diseases, and absence of food allergies to fish or crustaceans. The participants were clinically confirmed by a psychiatrist to have no cognitive impairment.
Three types of experimental supplement were used: krill oil as a typical oil rich in PUFAs incorporated in phosphatidylcholine, sardine oil as a typical fish oil, and medium-chain triglycerides as placebo. Subjects were asked to take four capsules twice a day after breakfast and dinner for 12 weeks (eight capsules daily), from the day following the initial measurements to the day of the final measurements.
The results of this study suggest that long-term ingestion of krill oil and sardine oil promotes working memory function by activating the dorsolateral prefrontal cortex in elderly people, and thus prevents deterioration in cognitive activity 153. However, sardine oil, which contains the largest amounts of EPA and DHA fatty acids, was not shown to possess any activation effects regarding the calculation task, and only krill oil, containing lesser amounts of these fatty acids, was shown to have an effect, possibly due to its higher percentage of phospholipids, in particular phosphatidylcholine 153. This finding indicates that n-3 PUFAs incorporated in phosphatidylcholine act on brain function more efficiently than those incorporated in triglycerides 154.
Krill Oil Side Effects and Toxicity
There are currently no known toxicity levels for krill oil. Side effects associated with the consumption of krill oil may include an increase in gastrointestinal
complaints such as flatulence, gas, bloating, and/or diarrhea 131.
Krill Oil Dosage
Therapeutic doses range from 1-3 grams krill oil per day, with maintenance dosing of 500 mg daily 128.
- Jenkins DJA, Josse AR. Fish oil and omega-3 fatty acids. CMAJ : Canadian Medical Association Journal. 2008;178(2):150. doi:10.1503/cmaj.071754. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2174990/[↩][↩][↩]
- Salem N., Jr Introduction to polyunsaturated fatty acids. Backgrounder. 1999;3:1–8.[↩][↩]
- Clinical nutrition: 4. Omega-3 fatty acids in cardiovascular care. Holub BJ. CMAJ. 2002 Mar 5; 166(5):608-15. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC99405/[↩][↩]
- Simopoulos AP. An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients. 2016;8(3):128. doi:10.3390/nu8030128. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4808858/[↩][↩]
- Alpha-linolenic acid and cardiovascular diseases. Lanzmann-Petithory D. J Nutr Health Aging. 2001; 5(3):179-83. https://www.ncbi.nlm.nih.gov/pubmed/11458289/[↩]
- Dietary intakes of omega-6 and omega-3 polyunsaturated fatty acids and the risk of breast cancer. Thiébaut AC, Chajès V, Gerber M, Boutron-Ruault MC, Joulin V, Lenoir G, Berrino F, Riboli E, Bénichou J, Clavel-Chapelon F. Int J Cancer. 2009 Feb 15; 124(4):924-31. https://www.ncbi.nlm.nih.gov/pubmed/19035453/[↩]
- Dietary linoleic acid influences desaturation and acylation of deuterium-labeled linoleic and linolenic acids in young adult males. Emken EA, Adlof RO, Gulley RM. Biochim Biophys Acta. 1994 Aug 4; 1213(3):277-88. https://www.ncbi.nlm.nih.gov/pubmed/7914092/[↩]
- Mediterranean diet, traditional risk factors, and the rate of cardiovascular complications after myocardial infarction: final report of the Lyon Diet Heart Study. de Lorgeril M, Salen P, Martin JL, Monjaud I, Delaye J, Mamelle N. Circulation. 1999 Feb 16; 99(6):779-85. https://www.ncbi.nlm.nih.gov/pubmed/9989963/[↩]
- Dietary intake of alpha-linolenic acid and risk of fatal ischemic heart disease among women. Hu FB, Stampfer MJ, Manson JE, Rimm EB, Wolk A, Colditz GA, Hennekens CH, Willett WC. Am J Clin Nutr. 1999 May; 69(5):890-7. http://ajcn.nutrition.org/content/69/5/890.long[↩]
- alpha-Linolenic acid intake is not beneficially associated with 10-y risk of coronary artery disease incidence: the Zutphen Elderly Study. Oomen CM, Ocké MC, Feskens EJ, Kok FJ, Kromhout D. Am J Clin Nutr. 2001 Oct; 74(4):457-63. http://ajcn.nutrition.org/content/74/4/457.long[↩]
- Arterial compliance in obese subjects is improved with dietary plant n-3 fatty acid from flaxseed oil despite increased LDL oxidizability. Nestel PJ, Pomeroy SE, Sasahara T, Yamashita T, Liang YL, Dart AM, Jennings GL, Abbey M, Cameron JD. Arterioscler Thromb Vasc Biol. 1997 Jun; 17(6):1163-70. http://atvb.ahajournals.org/content/17/6/1163.long[↩]
- Dietary supplementation with marine omega-3 fatty acids improve systemic large artery endothelial function in subjects with hypercholesterolemia. Goodfellow J, Bellamy MF, Ramsey MW, Jones CJ, Lewis MJ. J Am Coll Cardiol. 2000 Feb; 35(2):265-70. https://www.ncbi.nlm.nih.gov/pubmed/10676668/[↩]
- Fish oil improves arterial compliance in non-insulin-dependent diabetes mellitus. McVeigh GE, Brennan GM, Cohn JN, Finkelstein SM, Hayes RJ, Johnston GD. Arterioscler Thromb. 1994 Sep; 14(9):1425-9. http://atvb.ahajournals.org/content/14/9/1425.long[↩]
- Institute of Medicine, Food and Nutrition Board. Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids (macronutrients). Washington, DC: National Academy Press; 2005.[↩][↩][↩][↩]
- Omega-3 fatty acid content of the US food supply. Raper NR, Cronin FJ, Exler J. J Am Coll Nutr. 1992 Jun; 11(3):304-8. https://www.ncbi.nlm.nih.gov/pubmed/1619182/[↩]
- Kris-Etherton PM, Taylor DS, Yu-Poth S, Huth P, Moriarty K, Fishell V, et al. Polyunsaturated fatty acids in the food chain in the United States. Am J Clin Nutr 2000;71(Suppl 1):179-88.[↩]
- Breast cancer risk in rats fed a diet high in n-6 polyunsaturated fatty acids during pregnancy. Hilakivi-Clarke L, Onojafe I, Raygada M, Cho E, Clarke R, Lippman ME. J Natl Cancer Inst. 1996 Dec 18; 88(24):1821-7. https://www.ncbi.nlm.nih.gov/pubmed/8961971/[↩]
- Linoleic acid intake and cancer risk: a review and meta-analysis. Zock PL, Katan MB. Am J Clin Nutr. 1998 Jul; 68(1):142-53. https://www.ncbi.nlm.nih.gov/pubmed/9665108/[↩]
- Holub BJ. Clinical nutrition: 4. Omega-3 fatty acids in cardiovascular care. Hoffer LJ, Jones PJ, eds. CMAJ: Canadian Medical Association Journal. 2002;166(5):608-615. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC99405/[↩][↩]
- EPA and DHA Content of Fish Species. https://health.gov/dietaryguidelines/dga2005/report/html/table_g2_adda2.htm[↩]
- Dullemeijer C, Durga J, Brouwer IA, et al. n 3 fatty acid proportions in plasma and cognitive performance in older adults. Am J Clin Nutr. 2007;86:1479–1485. https://www.ncbi.nlm.nih.gov/pubmed/17991662[↩]
- Kannass KN, Colombo J, Carlson SE. Maternal DHA levels and toddler free-play attention. Dev Neuropsychol. 2009;34:159–174. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3752039/[↩]
- Muldoon MF, Ryan CM, Sheu L, Yao JK, Conklin SM, Manuck SB. Serum phospholipid docosahexaenonic acid is associated with cognitive functioning during middle adulthood. J Nutr. 2010;140:848–853. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2838625/[↩]
- Bauer I, Crewther DP, Pipingas A, Rowsell R, Cockerell R, Crewther SG. Omega-3 fatty acids modify human cortical visual processing: a double-blind, crossover study. PLoS One. 2011;6:e28214. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3235106/[↩]
- van Gelder BM, Tijhuis M, Kalmijn S, Kromhout D. Fish consumption, n-3 fatty acids, and subsequent 5-y cognitive decline in elderly men: the Zutphen Elderly Study. Am J Clin Nutr. 2007;85:1142–1147. https://www.ncbi.nlm.nih.gov/pubmed/17413117[↩]
- González S, Huerta JM, Fernández S, Patterson AM, Lasheras C. The relationship between dietary lipids and cognitive performance in an elderly population. Int J Food Sci Nutr. 2010;61:217–225. https://www.ncbi.nlm.nih.gov/pubmed/20001761[↩]
- Jicha GA, Markesbery WR. Omega-3 fatty acids: potential role in the management of early Alzheimer’s disease. Clin Interv Aging. 2010;5:45–61. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2854051/[↩]
- Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Kris-Etherton PM, Harris WS, Appel LJ, American Heart Association. Nutrition Committee. Circulation. 2002 Nov 19; 106(21):2747-57. http://circ.ahajournals.org/content/106/21/2747.long[↩][↩][↩]
- Am J Clin Nutr July 2006, vol. 84 no. 1 5-17. n−3 Fatty acids from fish or fish-oil supplements, but not α-linolenic acid, benefit cardiovascular disease outcomes in primary- and secondary-prevention studies: a systematic review. http://ajcn.nutrition.org/content/84/1/5.long[↩][↩][↩]
- He K, Song Y, Daviglus ML, et al. Accumulated evidence on fish consumption and coronary heart disease mortality: a meta-analysis of cohort studies. Circulation 2004;109:2705–11. http://circ.ahajournals.org/content/109/22/2705[↩]
- He K, Song Y, Daviglus ML, et al. Fish consumption and incidence of stroke. A meta-analysis of cohort studies. Stroke 2004;35:1538–42. http://stroke.ahajournals.org/content/35/7/1538[↩]
- Zhang J, Sasaki S, Amano K, Kesteloot H. Fish consumption and mortality from all causes, ischemic heart disease, and stroke: an ecological study. Prev Med 1999;28:520–9. https://www.ncbi.nlm.nih.gov/pubmed/10329343 [↩][↩]
- Harris WS. Omega-3 fatty acids. In: Coates PM, Betz JM, Blackman MR, et al., eds. Encyclopedia of Dietary Supplements. 2nd ed. London and New York: Informa Healthcare; 2010:577-86.[↩]
- Djousse L, Akinkuolie AO, Wu JH, Ding EL, Gaziano JM. Fish consumption, omega-3 fatty acids and risk of heart failure: a meta-analysis. Clin Nutr 2012;31:846-53. https://www.ncbi.nlm.nih.gov/pubmed/22682084?dopt=Abstract[↩]
- Del Gobbo LC, Imamura F, Aslibekyan S, Marklund M, Virtanen JK, Wennberg M, et al. Omega-3 polyunsaturated fatty acid biomarkers and coronary heart disease: pooling project of 19 cohort studies. JAMA Intern Med 2016;176:1155-66. https://www.ncbi.nlm.nih.gov/pubmed/27357102?dopt=Abstract[↩]
- U.S. Department of Health and Human Services, U.S. Department of Agriculture. https://health.gov/dietaryguidelines/2015/guidelines/[↩]
- U.S. Food and Drug Administration. Summary of qualified health claims subject to enforcement discretion 2014. https://www.fda.gov/Food/FoodborneIllnessContaminants/Metals/ucm393070.htm[↩][↩]
- Pradhan AD, Manson JE. Update on the Vitamin D and OmegA-3 trial (VITAL). J Steroid Biochem Mol Biol 2016;155:252-6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4600007/[↩][↩][↩][↩]
- Newberry SJ, Chung M, Booth M, Maglione M, Tang AM, C.E. OH, et al. Omega-3 fatty acids and maternal and child health: an updated systematic review. Evidence Report/Technology Assessment No. 224. (Prepared by the RAND Southern California Evidence-based Practice Center under Contract No. 290-2012-00006-I.) AHRQ Publication No. 16-E003-EF. Rockville, MD: Agency for Healthcare Research and Quality; 2016.[↩]
- Section on Breastfeeding. Breastfeeding and the use of human milk. Pediatrics 2012;129:2011-3552. https://www.ncbi.nlm.nih.gov/pubmed/22371471?dopt=Abstract[↩]
- U.S. Food and Drug Administration. Eating Fish: What Pregnant Women and Parents Should Know. https://www.fda.gov/downloads/Food/FoodborneIllnessContaminants/Metals/UCM536321.pdf[↩]
- O’Connor NR. Infant formula. Am Fam Physician 2009;79:565-70. https://www.ncbi.nlm.nih.gov/pubmed/19378873?dopt=Abstract[↩]
- Simmer K, Patole SK, Rao SC. Long-chain polyunsaturated fatty acid supplementation in infants born at term. Cochrane Database Syst Rev 2011:CD000376. https://www.ncbi.nlm.nih.gov/pubmed/22161363?dopt=Abstract[↩]
- Schulzke SM, Patole SK, Simmer K. Long-chain polyunsaturated fatty acid supplementation in preterm infants. Cochrane Database Syst Rev 2011:CD000375. https://www.ncbi.nlm.nih.gov/pubmed/21328248?dopt=Abstract[↩]
- Weylandt KH, Serini S, Chen YQ, Su HM, Lim K, Cittadini A, et al. Omega-3 polyunsaturated fatty acids: the way forward in times of mixed evidence. Biomed Res Int 2015;2015:143109.[↩]
- Zheng JS, Hu XJ, Zhao YM, Yang J, Li D. Intake of fish and marine n-3 polyunsaturated fatty acids and risk of breast cancer: meta-analysis of data from 21 independent prospective cohort studies. BMJ 2013;346:f3706.[↩][↩]
- MacLean CH, Newberry SJ, Mojica WA, Khanna P, Issa AM, Suttorp MJ, et al. Effects of omega-3 fatty acids on cancer risk: a systematic review. JAMA 2006;295:403-15. https://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0022772/[↩]
- Brasky TM, Darke AK, Song X, Tangen CM, Goodman PJ, Thompson IM, et al. Plasma phospholipid fatty acids and prostate cancer risk in the SELECT trial. J Natl Cancer Inst 2013;105:1132-41. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3735464/[↩][↩][↩]
- Gago-Dominguez M, Yuan JM, Sun CL, Lee HP, Yu MC. Opposing effects of dietary n-3 and n-6 fatty acids on mammary carcinogenesis: The Singapore Chinese Health Study. Br J Cancer 2003;89:1686-92.[↩]
- Brasky TM, Lampe JW, Potter JD, Patterson RE, White E. Specialty supplements and breast cancer risk in the VITamins And Lifestyle (VITAL) Cohort. Cancer Epidemiol Biomarkers Prev 2010;19:1696-708.[↩]
- Gerber M. Omega-3 fatty acids and cancers: a systematic update review of epidemiological studies. Br J Nutr 2012;107 Suppl 2:S228-39.[↩][↩]
- Geelen A, Schouten JM, Kamphuis C, Stam BE, Burema J, Renkema JM, et al. Fish consumption, n-3 fatty acids, and colorectal cancer: a meta-analysis of prospective cohort studies. Am J Epidemiol 2007;166:1116-25.[↩]
- Wu S, Feng B, Li K, Zhu X, Liang S, Liu X, et al. Fish consumption and colorectal cancer risk in humans: a systematic review and meta-analysis. Am J Med 2012;125:551-9 e5[↩]
- Kantor ED, Lampe JW, Peters U, Vaughan TL, White E. Long-chain omega-3 polyunsaturated fatty acid intake and risk of colorectal cancer. Nutr Cancer 2014;66:716-27.[↩]
- Brasky TM, Till C, White E, Neuhouser ML, Song X, Goodman P, et al. Serum phospholipid fatty acids and prostate cancer risk: results from the prostate cancer prevention trial. Am J Epidemiol 2011;173:1429-39[↩]
- Dahm CC, Gorst-Rasmussen A, Crowe FL, Roswall N, Tjonneland A, Drogan D, et al. Fatty acid patterns and risk of prostate cancer in a case-control study nested within the European Prospective Investigation into Cancer and Nutrition. Am J Clin Nutr 2012;96:1354-61.[↩]
- Park SY, Wilkens LR, Henning SM, Le Marchand L, Gao K, Goodman MT, et al. Circulating fatty acids and prostate cancer risk in a nested case-control study: the Multiethnic Cohort. Cancer Causes Control 2009;20:211-23.[↩]
- Alexander W. Prostate cancer risk and omega-3 Fatty Acid intake from fish oil: a closer look at media messages versus research findings. P T 2013;38:561-4.[↩]
- Torfadottir JE, Stampfer MJ, Mucci LA, Giovannucci EL. RE: Plasma phospholipid fatty acids and prostate cancer risk in the SELECT trial. J Natl Cancer Inst 2014;106:dju018[↩]
- Harris WS, Davidson MH. RE: Plasma phospholipid fatty acids and prostate cancer risk in the SELECT trial. J Natl Cancer Inst 2014;106:dju019[↩]
- Bosire C, Stampfer MJ, Subar AF, Park Y, Kirkpatrick SI, Chiuve SE, et al. Index-based dietary patterns and the risk of prostate cancer in the NIH-AARP diet and health study. Am J Epidemiol 2013;177:504-13.[↩]
- Augustsson K, Michaud DS, Rimm EB, Leitzmann MF, Stampfer MJ, Willett WC, et al. A prospective study of intake of fish and marine fatty acids and prostate cancer. Cancer Epidemiol Biomarkers Prev 2003;12:64-7.[↩]
- Crowe FL, Appleby PN, Travis RC, Barnett M, Brasky TM, Bueno-de-Mesquita HB, et al. Circulating fatty acids and prostate cancer risk: individual participant meta-analysis of prospective studies. J Natl Cancer Inst 2014;106[↩]
- Chua ME, Sio MC, Sorongon MC, Morales ML, Jr. The relevance of serum levels of long chain omega-3 polyunsaturated fatty acids and prostate cancer risk: A meta-analysis. Can Urol Assoc J 2013;7:E333-43.[↩]
- Chua ME, Sio MC, Sorongon MC, Dy JS. Relationship of dietary intake of omega-3 and omega-6 Fatty acids with risk of prostate cancer development: a meta-analysis of prospective studies and review of literature. Prostate Cancer 2012;2012:826254.[↩]
- Szymanski KM, Wheeler DC, Mucci LA. Fish consumption and prostate cancer risk: a review and meta-analysis. Am J Clin Nutr 2010;92:1223-33.[↩]
- Alexander DD, Bassett JK, Weed DL, Barrett EC, Watson H, Harris W. Meta-analysis of long-chain omega-3 polyunsaturated fatty acids (LC omega-3PUFA) and prostate cancer. Nutr Cancer 2015;67:543-54.[↩]
- Noel SE, Stoneham AC, Olsen CM, Rhodes LE, Green AC. Consumption of omega-3 fatty acids and the risk of skin cancers: a systematic review and meta-analysis. Int J Cancer 2014;135:149-56.[↩]
- Ibiebele TI, Nagle CM, Bain CJ, Webb PM. Intake of omega-3 and omega-6 fatty acids and risk of ovarian cancer. Cancer Causes Control 2012;23:1775-83.[↩]
- Arem H, Neuhouser ML, Irwin ML, Cartmel B, Lu L, Risch H, et al. Omega-3 and omega-6 fatty acid intakes and endometrial cancer risk in a population-based case-control study. Eur J Nutr 2013;52:1251-60.[↩]
- Brasky TM, Rodabough RJ, Liu J, Kurta ML, Wise LA, Orchard TS, et al. Long-chain omega-3 fatty acid intake and endometrial cancer risk in the Women’s Health Initiative. Am J Clin Nutr 2015;101:824-34.[↩]
- Qin B, Xun P, He K. Fish or long-chain (n-3) PUFA intake is not associated with pancreatic cancer risk in a meta-analysis and systematic review. J Nutr 2012;142:1067-73.[↩]
- Han YJ, Li J, Huang W, Fang Y, Xiao LN, Liao ZE. Fish consumption and risk of esophageal cancer and its subtypes: a systematic review and meta-analysis of observational studies. Eur J Clin Nutr 2013;67:147-54.[↩]
- Dangour AD, Whitehouse PJ, Rafferty K, Mitchell SA, Smith L, Hawkesworth S, et al. B-vitamins and fatty acids in the prevention and treatment of Alzheimer’s disease and dementia: a systematic review. J Alzheimers Dis 2010;22:205-24. https://www.ncbi.nlm.nih.gov/pubmed/20847412?dopt=Abstract[↩]
- Sydenham E, Dangour AD, Lim WS. Omega 3 fatty acid for the prevention of cognitive decline and dementia. Cochrane Database Syst Rev 2012;6:CD005379. https://www.ncbi.nlm.nih.gov/pubmed/22696350?dopt=Abstract[↩][↩][↩]
- Chew EY, Clemons TE, Agron E, Launer LJ, Grodstein F, Bernstein PS, et al. Effect of omega-3 fatty acids, lutein/zeaxanthin, or other nutrient supplementation on cognitive function: the AREDS2 randomized clinical trial. JAMA 2015;314:791-801. https://www.ncbi.nlm.nih.gov/pubmed/26305649?dopt=Abstract[↩]
- Tully AM, Roche HM, Doyle R, Fallon C, Bruce I, Lawlor B, et al. Low serum cholesteryl ester-docosahexaenoic acid levels in Alzheimer’s disease: a case-control study. Br J Nutr 2003;89:483-9. https://www.ncbi.nlm.nih.gov/pubmed/12654166?dopt=Abstract[↩]
- Yassine HN, Feng Q, Azizkhanian I, Rawat V, Castor K, Fonteh AN, et al. Association of serum docosahexaenoic acid with cerebral amyloidosis. JAMA Neurol 2016. https://www.ncbi.nlm.nih.gov/pubmed/27532692?dopt=Abstract[↩]
- Jiao J, Li Q, Chu J, Zeng W, Yang M, Zhu S. Effect of n-3 PUFA supplementation on cognitive function throughout the life span from infancy to old age: a systematic review and meta-analysis of randomized controlled trials. Am J Clin Nutr 2014;100:1422-36. https://www.ncbi.nlm.nih.gov/pubmed/25411277?dopt=Abstract[↩]
- Yurko-Mauro K, Alexander DD, Van Elswyk ME. Docosahexaenoic acid and adult memory: a systematic review and meta-analysis. PLoS One 2015;10:e0120391. https://www.ncbi.nlm.nih.gov/pubmed/25786262?dopt=Abstract[↩]
- Mazereeuw G, Lanctot KL, Chau SA, Swardfager W, Herrmann N. Effects of omega-3 fatty acids on cognitive performance: a meta-analysis. Neurobiol Aging 2012;33:1482 e17-29. https://www.ncbi.nlm.nih.gov/pubmed/22305186?dopt=Abstract[↩][↩][↩]
- SanGiovanni JP, Chew EY. The role of omega-3 long-chain polyunsaturated fatty acids in health and disease of the retina. Prog Retin Eye Res 2005;24:87-138. https://www.ncbi.nlm.nih.gov/pubmed/15555528?dopt=Abstract[↩]
- Augood C, Chakravarthy U, Young I, Vioque J, de Jong PT, Bentham G, et al. Oily fish consumption, dietary docosahexaenoic acid and eicosapentaenoic acid intakes, and associations with neovascular age-related macular degeneration. Am J Clin Nutr 2008;88:398-406. https://www.ncbi.nlm.nih.gov/pubmed/18689376?dopt=Abstract[↩]
- Seddon JM, George S, Rosner B. Cigarette smoking, fish consumption, omega-3 fatty acid intake, and associations with age-related macular degeneration: the US Twin Study of Age-Related Macular Degeneration. Arch Ophthalmol 2006;124:995-1001. https://www.ncbi.nlm.nih.gov/pubmed/16832023?dopt=Abstract[↩]
- Christen WG, Schaumberg DA, Glynn RJ, Buring JE. Dietary omega-3 fatty acid and fish intake and incident age-related macular degeneration in women. Arch Ophthalmol 2011;129:921-9. https://www.ncbi.nlm.nih.gov/pubmed/21402976?dopt=Abstract[↩]
- Merle BM, Benlian P, Puche N, Bassols A, Delcourt C, Souied EH, et al. Circulating omega-3 Fatty acids and neovascular age-related macular degeneration. Invest Ophthalmol Vis Sci 2014;55:2010-9. https://www.ncbi.nlm.nih.gov/pubmed/24557349?dopt=Abstract[↩]
- A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS report no. 8. Arch Ophthalmol 2001;119:1417-36. https://www.ncbi.nlm.nih.gov/pubmed/11594942?dopt=Abstract[↩]
- Sangiovanni JP, Agron E, Meleth AD, Reed GF, Sperduto RD, Clemons TE, et al. ω-3 Long-chain polyunsaturated fatty acid intake and 12-y incidence of neovascular age-related macular degeneration and central geographic atrophy: AREDS report 30, a prospective cohort study from the Age-Related Eye Disease Study. Am J Clin Nutr 2009;90:1601-7. https://www.ncbi.nlm.nih.gov/pubmed/19812176?dopt=Abstract[↩]
- Age-Related Eye Disease Study 2 Research G. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: the Age-Related Eye Disease Study 2 (AREDS2) randomized clinical trial. JAMA 2013;309:2005-15. https://www.ncbi.nlm.nih.gov/pubmed/23644932?dopt=Abstract[↩]
- Lawrenson JG, Evans JR. Omega 3 fatty acids for preventing or slowing the progression of age-related macular degeneration. Cochrane Database Syst Rev 2015;4:CD010015. www.ncbi.nlm.nih.gov/pubmed/25856365?dopt=Abstract[↩]
- Souied EH, Delcourt C, Querques G, Bassols A, Merle B, Zourdani A, et al. Oral docosahexaenoic acid in the prevention of exudative age-related macular degeneration: the Nutritional AMD Treatment 2 study. Ophthalmology 2013;120:1619-31. https://www.ncbi.nlm.nih.gov/pubmed/23395546?dopt=Abstract[↩]
- The Dry Eye Assessment and Management Study Research Group. Omega-3 fatty acid supplementation for treatment of dry eye disease. N Engl J Med 2018;378:1681-90. https://www.ncbi.nlm.nih.gov/pubmed/29652551[↩][↩][↩]
- Miljanović B, Trivedi KA, Dana MR, Gilbard JP, Buring JE, Schaumberg DA. Relation between dietary n-3 and n-6 fatty acids and clinically diagnosed dry eye syndrome in women. Am J Clin Nutr 2005;82:887-93. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1360504/[↩]
- Ziemanski JF, Wolters LR, Jones-Jordan L, Nichols JJ, Nichols KK. Relation between dietary essential fatty acid intake and dry eye disease and meibomian gland dysfunction in postmenopausal women. Am J Ophthalmol 2018;189:29-40. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5967412/[↩]
- Hom MM, Asbell P, Barry B. Omegas and dry eye: more knowledge, more questions. Optom Vis Sci 2015;92:948-56.[↩]
- Bhargava R, Kumar P, Kumar M, Mehra N, Mishra A. A randomized controlled trial of omega-3 fatty acids in dry eye syndrome. Int J Ophthalmol 2013;18;6:811-6.[↩]
- Epitropoulos AT, Donnenfeld ED, Shah ZA, Holland EJ, Gross M, Faulkner WJ, Matossian C, Lane SS, Toyos M, Bucci FA Jr, Perry HD. Effect of oral re-esterified omega-3 nutritional supplementation on dry eyes. Cornea 2016;35:1185-91.[↩]
- James M, Proudman S, Cleland L. Fish oil and rheumatoid arthritis: past, present and future. Proc Nutr Soc 2010;69:316-23. https://www.ncbi.nlm.nih.gov/pubmed/20509981?dopt=Abstract[↩]
- Goldberg RJ, Katz J. A meta-analysis of the analgesic effects of omega-3 polyunsaturated fatty acid supplementation for inflammatory joint pain. Pain 2007;129:210-23. https://www.ncbi.nlm.nih.gov/pubmed/17335973?dopt=Abstract[↩]
- Li F, Liu X, Zhang D. Fish consumption and risk of depression: a meta-analysis. J Epidemiol Community Health 2016;70:299-304. https://www.ncbi.nlm.nih.gov/pubmed/26359502?dopt=Abstract[↩]
- Appleton KM, Sallis HM, Perry R, Ness AR, Churchill R. Omega-3 fatty acids for depression in adults. Cochrane Database Syst Rev 2015;11:CD004692. https://www.ncbi.nlm.nih.gov/pubmed/26537796?dopt=Abstract[↩]
- Cochcrane Review 16 April 2008 – Omega-3 fatty acids for bipolar disorder – http://www.cochrane.org/CD005169/DEPRESSN_omega-3-fatty-acids-for-bipolar-disorder[↩]
- Cabre E, Manosa M, Gassull MA. Omega-3 fatty acids and inflammatory bowel diseases – a systematic review. Br J Nutr 2012;107 Suppl 2:S240-52. https://www.ncbi.nlm.nih.gov/pubmed/22591898?dopt=Abstract[↩]
- Lev-Tzion R, Griffiths AM, Leder O, Turner D. Omega 3 fatty acids (fish oil) for maintenance of remission in Crohn’s disease. Cochrane Database Syst Rev 2014;2:CD006320. https://www.ncbi.nlm.nih.gov/pubmed/24585498?dopt=Abstract[↩][↩]
- Cochcrane Review 9 November 2011 – Omega-3 fatty acids for autism spectrum disorders – http://www.cochrane.org/CD007992/BEHAV_omega-3-fatty-acids-for-autism-spectrum-disorders-asd[↩]
- Cooper RE, Tye C, Kuntsi J, Vassos E, Asherson P. The effect of omega-3 polyunsaturated fatty acid supplementation on emotional dysregulation, oppositional behaviour and conduct problems in ADHD: A systematic review and meta-analysis. J Affect Disord 2016;190:474-82. https://www.ncbi.nlm.nih.gov/pubmed/26551407?dopt=Abstract[↩]
- Best KP, Gold M, Kennedy D, Martin J, Makrides M. Omega-3 long-chain PUFA intake during pregnancy and allergic disease outcomes in the offspring: a systematic review and meta-analysis of observational studies and randomized controlled trials. Am J Clin Nutr 2016;103:128-43. https://www.ncbi.nlm.nih.gov/pubmed/26675770?dopt=Abstract[↩]
- Gunaratne AW, Makrides M, Collins CT. Maternal prenatal and/or postnatal n-3 long chain polyunsaturated fatty acids (LCPUFA) supplementation for preventing allergies in early childhood. Cochrane Database Syst Rev 2015;7:CD010085. https://www.ncbi.nlm.nih.gov/pubmed/26197477?dopt=Abstract[↩]
- Oliver C, Watson H. Omega-3 fatty acids for cystic fibrosis. Cochrane Database Syst Rev 2016;1:CD002201. https://www.ncbi.nlm.nih.gov/pubmed/26730723?dopt=Abstract[↩]
- National Center for Complementary and Integrative Health. Omega-3 Supplements: In Depth. https://nccih.nih.gov/health/omega3/introduction.htm[↩][↩]
- National Institutes of Health. Dietary Supplement Label Database. https://www.dsld.nlm.nih.gov/dsld-mobile/[↩][↩]
- U.S. Food and Drug Administration. Eating Fish: What Pregnant Women and Parents Should Know. https://www.fda.gov/Food/FoodborneIllnessContaminants/Metals/ucm393070.htm[↩]
- ConsumerLab.com. Product Review: Fish Oil and Omega-3 and -7 Fatty Acid Supplements Review (Including Krill, Algae, Calamari, Green-lipped Mussel Oil, and Sea Buckthorn). https://www.consumerlab.com/reviews/fish_oil_supplements_review/omega3[↩][↩]
- Cunningham E. Are krill oil supplements a better source of n-3 fatty acids than fish oil supplements? J Acad Nutr Diet 2012;112:344. https://www.ncbi.nlm.nih.gov/pubmed/22459227[↩]
- Dyerberg J, Madsen P, Moller JM, Aardestrup I, Schmidt EB. Bioavailability of marine n-3 fatty acid formulations. Prostaglandins Leukot Essent Fatty Acids 2010;83:137-41. https://www.ncbi.nlm.nih.gov/pubmed/20638827[↩]
- Davidson MH, Kling D, Maki KC. Novel developments in omega-3 fatty acid-based strategies. Curr Opin Lipidol 2011;22:437-44. https://www.ncbi.nlm.nih.gov/pubmed/21986642?dopt=Abstract[↩]
- Cunningham E. Are krill oil supplements a better source of n-3 fatty acids than fish oil supplements? J Acad Nutr Diet 2012;112:344. https://www.ncbi.nlm.nih.gov/pubmed/22459227?dopt=Abstract[↩]
- Schuchardt JP, Schneider I, Meyer H, Neubronner J, von Schacky C, Hahn A. Incorporation of EPA and DHA into plasma phospholipids in response to different omega-3 fatty acid formulations–a comparative bioavailability study of fish oil vs. krill oil. Lipids Health Dis 2011;10:145. https://www.ncbi.nlm.nih.gov/pubmed/21854650?dopt=Abstract[↩]
- Ulven SM, Holven KB. Comparison of bioavailability of krill oil versus fish oil and health effect. Vasc Health Risk Manag 2015;11:511-24. https://www.ncbi.nlm.nih.gov/pubmed/26357480?dopt=Abstract[↩]
- ConsumerLab.com.Product Review: Fish Oil and Omega-3 and -7 Fatty Acid Supplements Review (Including Krill, Algae, Calamari, Green-lipped Mussel Oil, and Sea Buckthorn). https://www.consumerlab.com/reviews/fish_oil_supplements_review/omega3[↩]
- Arterburn LM, Oken HA, Bailey Hall E, Hamersley J, Kuratko CN, Hoffman JP. Algal-oil capsules and cooked salmon: nutritionally equivalent sources of docosahexaenoic acid. J Am Diet Assoc 2008;108:1204-9. https://www.ncbi.nlm.nih.gov/pubmed/18589030?dopt=Abstract[↩]
- EFSA Panel on Dietetic Products NaA. Scientific opinion on the tolerable upper intake level of eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA) and docosapentaenoic acid (DPA). EFSA Journal 2012;10:2815.[↩]
- A brief review of krill oil history, research, and the commercial market. Kwantes JM, Grundmann O. J Diet Suppl. 2015 Mar; 12(1):23-35. https://www.ncbi.nlm.nih.gov/pubmed/24689485/[↩][↩]
- ConsumerLab Product Review: Fish oil and omega-3 fatty acid supplements (EPA and DHA from fish A, and krill). 2010. www.consumerlab.com/news/fish_oil_supplements_reviewed/9_28_2010/[↩][↩]
- ConsumerLab Product Review: Fish oil and omega-3 fatty acid supplements (EPA and DHA from fish A, and krill). 2010. https://www.consumerlab.com/news/fish_oil_supplements_reviewed/9_28_2010/[↩][↩]
- Nature Made Krill Oil 300 mg. Nature Made Web site. 2014. http://www.naturemade.com/fish-oil-and-omegas/krill-oil-300-mg[↩]
- MegaRed Ultra Strength Omega-3 Krill Oil. Schiff MegaRed Web site. 2014. http://www.megared.com/?uci=US-MG-O-PS-GA-10002&cid=cov395p6063g-c[↩]
- Altern Med Rev. 2010 Apr;15(1):84-6. Krill oil. Monograph. http://www.altmedrev.com/publications/15/1/84.pdf[↩][↩]
- Wijendran V, Huang MC, Diau GY, et al. Efficacy of dietary arachidonic acid provided as triglyceride or phospholipid as substrates for brain arachidonic acid accretion in baboon neonates. Pediatr Res 2002;51:265-272.[↩]
- Goustard-Langelier B, Guesnet P, Durand G, et al. n-3 and n-6 fatty acid enrichment by dietary fish oil and phospholipid sources in brain cortical areas and nonneural tissues of formula-fed piglets. Lipids 1999;34:5-16.[↩]
- Maki KC, Reeves MS, Farmer M, et al. Krill oil supplementation increases plasma concentrations of eicosapentaenoic and docosahexaenoic acids in overweight and obese men and women. Nutr Res 2009;29:609-615.[↩][↩]
- Krill for human consumption: nutritional value and potential health benefits. Tou JC, Jaczynski J, Chen YC. Nutr Rev. 2007 Feb; 65(2):63-77. https://www.ncbi.nlm.nih.gov/pubmed/17345959/[↩]
- Incorporation of EPA and DHA into plasma phospholipids in response to different omega-3 fatty acid formulations–a comparative bioavailability study of fish oil vs. krill oil. Schuchardt JP, Schneider I, Meyer H, Neubronner J, von Schacky C, Hahn A. Lipids Health Dis. 2011 Aug 22; 10():145. https://www.ncbi.nlm.nih.gov/pubmed/21854650/[↩]
- Maki KC, Reeves MS, Farmer M, et al. Krill oil supplementation increases plasma concentrations of eicosapentaenoic and docosahexaenoic acids in overweight and obese men and women. Nutr Res. 2009;9:609–615. https://www.ncbi.nlm.nih.gov/pubmed/19854375[↩]
- Ulven SM, Kirkhus B, Lamglait A, et al. Metabolic effects of krill oil are essentially similar to those of fish oil but at lower dose of EPA and DHA, in healthy volunteers. Lipids. 2011;46:37–46. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3024511/[↩][↩][↩][↩]
- Laidlaw M, Cockerline CA, Rowe WJ. A randomized clinical trial to determine the efficacy of manufacturers’ recommended doses of omega-3 fatty acids from different sources in facilitating cardiovascular disease risk reduction. Lipids Health Dis. 2014;13:99. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4085663/[↩]
- Bunea R, El Farrah K, Deutsch L. Evaluation of the effects of Neptune Krill Oil on the clinical course of hyperlipidemia. Altern Med Rev 2004:9:420-428.[↩][↩][↩][↩]
- Administration of natural astaxanthin increases serum HDL-cholesterol and adiponectin in subjects with mild hyperlipidemia. Yoshida H, Yanai H, Ito K, Tomono Y, Koikeda T, Tsukahara H, Tada N. Atherosclerosis. 2010 Apr; 209(2):520-3. https://www.ncbi.nlm.nih.gov/pubmed/19892350/[↩]
- Chang JP, Chen YT, Su KP. Omega-3 polyunsaturated fatty acids (n-3 PUFAs) in cardiovascular diseases (CVDs) and depression: the missing link? Cardiovasc Psychiatry Neurol 2009;2009:725310. Epub 2009 Sep 27.[↩]
- Breslow J. n-3 fatty acids and cardiovascular disease. Am J Clin Nutr 2006;83:1477S-1482S.[↩]
- Calzolari I, Fumagalli S, Marchionni N, Di Bari M. Polyunsaturated fatty acids and cardiovascular disease. Curr Pharm Des 2009;15:4094-4102.[↩]
- Phosphatidylcholine. Altern Med Rev 2002;7:150-154.[↩]
- Naguib YM. Antioxidant activities of astaxanthin and related carotenoids. J Agric Food Chem 2000;48:1150-1154.[↩]
- Sampalis F, Bunea R, Pelland MF, et al. Evaluation of the effects of Neptune Krill Oil on the management of premenstrual syndrome and dysmenorrhea. Altern Med Rev 2003;8:171-179.[↩][↩][↩]
- Deutsch L. Evaluation of the effect of Neptune Krill Oil on chronic inflammation and arthritic symptoms. J Am Coll Nutr 2007:26:39-48.[↩][↩][↩]
- Berge K, Musa-Veloso K, Harwood M, Hoem N, Burri L. Krill oil supplementation lowers serum triglycerides without increasing low-density lipoprotein cholesterol in adults with borderline high or high triglyceride levels. Nutr Res. 2014;2:126–133. https://www.ncbi.nlm.nih.gov/pubmed/24461313[↩][↩][↩]
- Backes JM, Howard PA. Krill Oil for Cardiovascular Risk Prevention: Is It for Real? Hospital Pharmacy. 2014;49(10):907-912. doi:10.1310/hpj4910-907. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4252213/[↩]
- Circulation. 2014 Jun 24;129(25 Suppl 2):S1-45. doi: 10.1161/01.cir.0000437738.63853.7a. Epub 2013 Nov 12. 2013 American College of Cardiology and the American Heart Association guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. http://circ.ahajournals.org/content/129/25_suppl_2/S1.long[↩]
- Krill oil supplementation lowers serum triglycerides without increasing low-density lipoprotein cholesterol in adults with borderline high or high triglyceride levels. Berge K, Musa-Veloso K, Harwood M, Hoem N, Burri L. Nutr Res. 2014 Feb; 34(2):126-33. https://www.ncbi.nlm.nih.gov/pubmed/24461313/[↩]
- The omega-3 index as a risk factor for coronary heart disease. Harris WS. Am J Clin Nutr. 2008 Jun; 87(6):1997S-2002S. https://www.ncbi.nlm.nih.gov/pubmed/18541601/[↩]
- Decreases in regional cerebral blood flow with normal aging. Martin AJ, Friston KJ, Colebatch JG, Frackowiak RS. J Cereb Blood Flow Metab. 1991 Jul; 11(4):684-9. https://www.ncbi.nlm.nih.gov/pubmed/2050757/[↩]
- Context-dependent, neural system-specific neurophysiological concomitants of ageing: mapping PET correlates during cognitive activation. Esposito G, Kirkby BS, Van Horn JD, Ellmore TM, Berman KF. Brain. 1999 May; 122 ( Pt 5)():963-79. https://www.ncbi.nlm.nih.gov/pubmed/10355679/[↩]
- Konagai C, Yanagimoto K, Hayamizu K, Han L, Tsuji T, Koga Y. Effects of krill oil containing n-3 polyunsaturated fatty acids in phospholipid form on human brain function: a randomized controlled trial in healthy elderly volunteers. Clinical Interventions in Aging. 2013;8:1247-1257. doi:10.2147/CIA.S50349. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3789637/[↩][↩][↩]
- Correlation between changes in blood fatty acid composition and visual sustained attention performance in children with inattention: effect of dietary n-3 fatty acids containing phospholipids. Vaisman N, Kaysar N, Zaruk-Adasha Y, Pelled D, Brichon G, Zwingelstein G, Bodennec J. Am J Clin Nutr. 2008 May; 87(5):1170-80. https://www.ncbi.nlm.nih.gov/pubmed/18469236/[↩]