Contents
What is kombucha
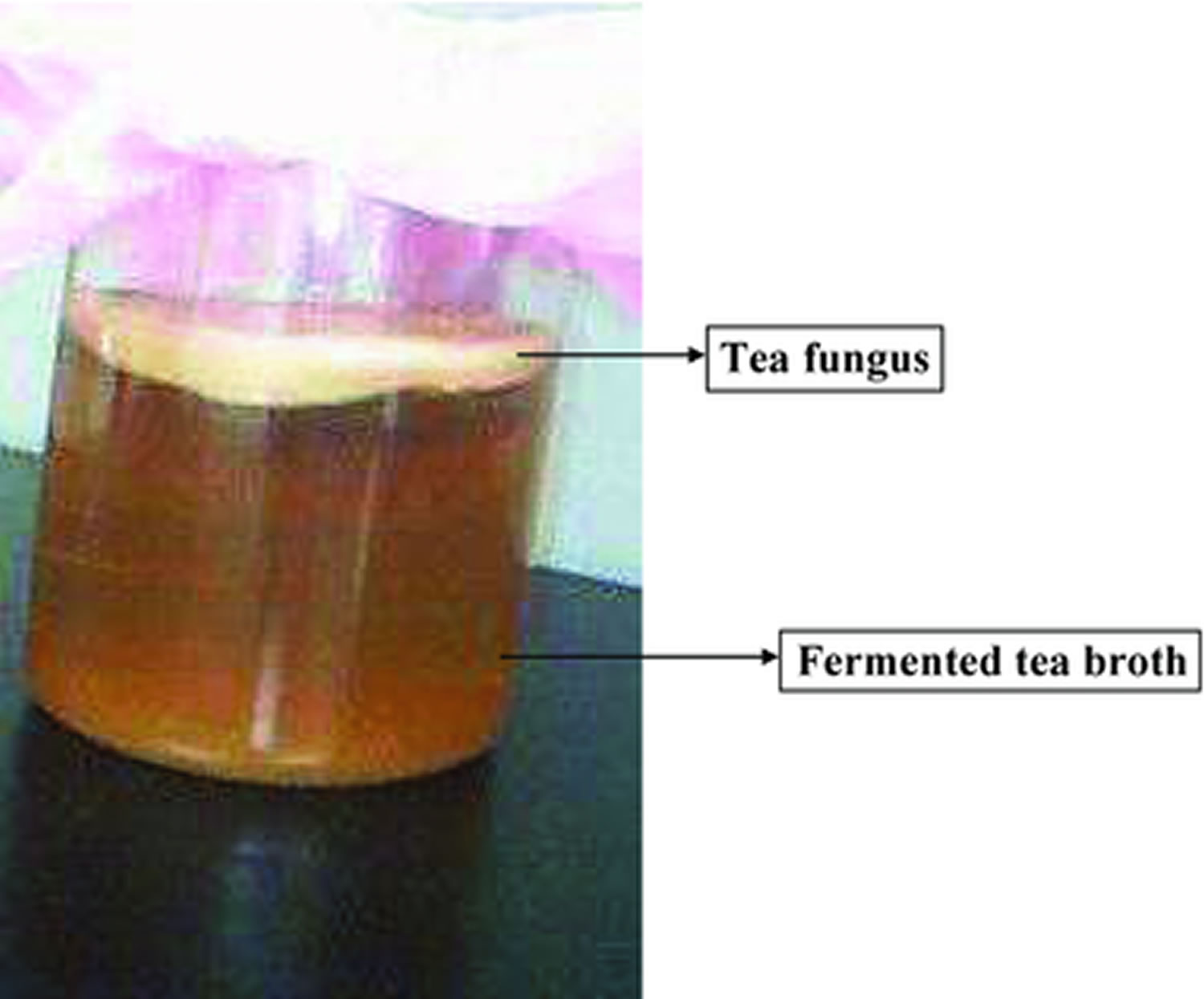
Kombucha tea is a fermented drink made with tea, sugar, bacteria and yeast 1. Although it’s sometimes referred to as kombucha mushroom tea, kombucha is not a mushroom — it’s a colony of bacteria and yeast. Kombucha tea is made by adding the colony (bacteria and yeasts) to sugar and tea, and allowing the mix to ferment 2. The resulting liquid contains vinegar, B vitamins and a number of other chemical compounds. A floating cellulosic pellicle layer and the sour liquid broth are the 2 portions of kombucha tea (Figure 1). It tastes like sparkling apple cider and can be produced in the home by fermentation using mail order or locally available tea fungus. Though green tea can be used for kombucha preparation, black tea and white sugar are considered the finest substrates.
Traditional substrate for the kombucha fermentation is black or green tea extract sweetened with 5% to 8% sucrose. Different tea leaf varieties, amounts of sugar, fermentation time, and composition of tea fungus may account for differences in composition and therefore also the biological activities of kombucha tea.
Figure 1. Kombucha black tea having fermented broth and tea fungus
Proponents claim kombucha tea helps prevent and manage serious health conditions, from blood pressure to cancer. These claims are not backed by scientific evidence 3. Limited evidence suggests kombucha tea may offer benefits similar to probiotic supplements, including promoting a healthy immune system and preventing constipation. All the biological activities have been investigated using animal experimental models. At present, however, valid medical studies of kombucha tea’s role in human health are very limited — and there are risks to consider.
There have been reports of adverse effects, such as stomach upset, infections and allergic reactions in kombucha tea drinkers. Kombucha tea is often brewed in homes under nonsterile conditions, making contamination likely. When improperly manufactured ceramic pots have been used for brewing, lead poisoning has occurred — the acids in the tea can leach lead from the ceramic glaze.
In short, there isn’t enough evidence that kombucha tea delivers on its health claims. At the same time, several cases of harm have been reported. Therefore, the prudent approach is to avoid kombucha tea until more definitive information is available.
Microorganisms of kombucha tea
Tea fungus or kombucha is the common name given to a symbiotic growth of acetic acid bacteria and osmophilic yeast species in a zoogleal mat which has to be cultured in sugared tea. According to Jarrell and others 4, kombucha is a consortium of yeasts and bacteria. The formal botanical name Medusomyces gisevii was given to it by Lindau (Hesseltine 1965). Tea fungus is not a mushroom. That name is wrongly given due to the ability of bacteria to synthesize a floating cellulose network which appears like surface mold on the undisturbed, unshaken medium.
Similarly to milk-derived kefir, the exact microbial composition of kombucha cannot be given because it varies. It depends on the source of the inoculum for the tea fermentation. One of the clearer accounts of the microbes found in kombucha starter is from Hesseltine 5. He isolated an Acetobacter sp. (NRRL B-2357) and 2 yeasts (NRRL YB-4810, NRRL YB-4882) from a kombucha sample received from Switzerland and used these microorganisms to produce kombucha tea.
The most abundant prokaryotes in this culture belong to the bacterial genera Acetobacter and Gluconobacter. The basic bacterium is Acetobacter xylinum 6. It produces a cellulosic floating network on the surface of the fermenting liquid. The network is the secondary metabolite of kombucha fermentation but also one of the unique features of the culture 7. Sievers and others 8 reported that the microflora embedded in the cellulose layer was a mixed culture of A. xylinum and a Zygosaccharomyces sp. The predominant acetic acid bacteria found in the tea fungus are A. xylium, A. pasteurianus, A. aceti, and Gluconobacter oxydans 9. Gluconacetobacter sp. A4 (G. sp. A4), which has strong ability to produce D-saccharic acid-1,4-lactone (DSL), was the key functional bacterial species isolated from a preserved kombucha by Yang and others 10. Strains of a new species in the genus Acetobacter, namely Acetobacter. intermedius sp. nov., were isolated from kombucha beverage and characterized by Boesch and others 11. Dutta and Gachhui 12 isolated the novel nitrogen-fixing Acetobacter nitrogenifigens sp. nov., and the nitrogen-fixing, cellulose-producing Gluconacetobacter kombuchae sp. nov., from kombucha tea. An investigation by Marsh and others 13 indicated that the dominant bacteria in 5 kombucha samples (2 from Canada and one each from Ireland, the United States, and the United Kingdom) belong to Gluconacetobacter (over 85% in most samples) and Lactobacillus (up to 30%) species. Acetobacter was determined in very small number (lower than 2%).
In addition to acetic acid bacteria there are many yeast species in kombucha. A broad spectrum of yeasts has been reported including species of Saccharomyces, Saccharomycodes, Schizosaccharomyces, Zygosaccharomyces, Brettanomyces/Dekkera, Candida, Torulospora, Koleckera, Pichia, Mycotorula, and Mycoderma. The yeasts of Saccharomyces species were identified as Saccharomyces sp. 14 and as Saccharomyces cerevisiae 15, Saccharomyces bisporus 7, Saccharomycoides ludwigii 16, Schizosaccharomyces pombe 17, Zygosaccharomyces sp. 13, Zygosaccharomyces rouxii 18, and Zygosaccharomyces bailii 19. The genus Brettanomyces was isolated by several workers. Herrera and Calderon-Villagomez 18 isolated Brettanomyces intermedius, Liu and others 9 and Teoh and others 17 isolated Brettanomyces bruxellensis, and Jayabalan and others 19 isolated B. claussenii. An examination of 2 commercial kombucha and 32 cultures from private households in Germany 20 showed variable compositions of yeasts. The predominant yeasts were Brettanomyces, Zygosaccharomyces, and Saccharomyces spp. Roussin 6 determined Zygosaccharomyces and S. cerevisiae as the typical yeasts in North American kombucha. Kurtzman and others 21 isolated an ascosporogenous yeast, Zygosaccharomyces kombuchaensis sp. n. (type strain NRRL YB-4811, CBS 8849), from kombucha. An investigation of the physiology of Z. kombuchaensis sp. n., related to the spoilage yeasts Zygosaccharomyces lentus, clearly showed that these 2 species were not same 22.
Candida sp. is included in a great number of kombucha beverages. Kozaki and others 14 isolated Candida famata, Candida guilliermondii, and Candida obutsa. In kombucha samples from Mexico, Herrera and Calderon-Villagomez 18 detected C. famata. Teoh and others 17 identified Candida stellata. From a local kombucha in Saudi Arabia, Ramadani and Abulreesh 16 isolated and identified 4 yeasts: Candida guilliermondi, Candida colleculosa, Candida kefyr, and Candida krusei. C. krusei were identified in kombucha from a district of Ankara 15.
Chemical composition of kombucha tea
Chemical analysis of kombucha showed the presence of various organic acids, such as acetic, gluconic, glucuronic, citric, L-lactic, malic, tartaric, malonic, oxalic, succinic, pyruvic, usnic; also sugars, such as sucrose, glucose, and fructose; the vitamins B1, B2, B6, B12, and C; 14 amino acids, biogenic amines, purines, pigments, lipids, proteins, some hydrolytic enzymes, ethanol, antibiotically active matter, carbon dioxide, phenol, as well as some tea polyphenols, minerals, anions, DSL, as well as insufficiently known products of yeast and bacterial metabolites 23.
Yeasts and bacteria in kombucha are involved in such metabolic activities that utilize substrates by different and in complementary ways. Yeasts hydrolyze sucrose into glucose and fructose by invertase and produce ethanol via glycolysis, with a preference for fructose as a substrate. Acetic acid bacteria make use of glucose to produce gluconic acid and ethanol to produce acetic acid. The pH value of kombucha beverage decreases due to the production of organic acids during fermentation 24.
The results presented in Table 1 indicate the predominant components of traditional kombucha beverage. These data suggest the heterogeneity of investigations performed on kombucha. The main differences in the investigated components are related to the duration of fermentation and the content of black tea. The researchers from different parts of the world used the same initial content of sucrose (10%). Researchers used different amounts of kombucha tea broth for the initial inoculation: 20% 25 and 10% 26. The fermentation process was performed in small volume reactors (glass jar or beaker), up to 1 L. The measured values of components propose that applied parameters (fermentation temperature, fermentation time, and initial content of sucrose and black tea), as well as the composition of kombucha culture have impact on the metabolic activity of kombucha, and therefore, on the end products of the metabolism.
Table 1. Predominant components in kombucha tea at the end of the fermentation on sugared black tea infusion
| Component | Component content (g/L) | Initial sucrose (%) | Black tea | Fermentation temperature (°C) | Fermentation time (d) | Reference |
|---|---|---|---|---|---|---|
| Acetic acid | 8 | 10 | 2 bags | 24 ± 3 | 60 | Chen and Liu 25 |
| 4.69 | 10 | 12 g/L | 24 ± 3 | 18 | Jayabalan and others 26 | |
| Glucuronic acid | 0.0031 | 5 | 1.5 g/L | 28 | 21 | Lončar and others 27 |
| 0.0026 | 7 | 1.5 g/L | 28 | 21 | Lončar and others 28 | |
| 0.0034 | 10 | 1.5 g/L | 28 | 21 | Lončar and others 29 | |
| 1.71 | 10 | 12 g/L | 24 ± 3 | 18 | Jayabalan and others 26 | |
| Gluconic acid | 39 | 10 | 2 bags | 24 ± 3 | 60 | Chen and Liu 25 |
| Glucose | 179.5 | 7 | 1.5 g/L | 28 | 21 | Malbaša and others 30 |
| 24.59 | 7 | 1.5 g/L | 28 | 21 | Lončar and others 27 | |
| 12 | 10 | 2 bags | 24 ± 3 | 60 | Chen and Liu 25 | |
| Fructose | 76.9 | 7 | 1.5 g/L | 28 | 21 | Malbaša and others 30 |
| 5.40 | 7 | 1.5 g/L | 28 | 21 | Lončar and others 27 | |
| 55 | 10 | 2 bags | 24 ± 3 | 60 | Chen and Liu 25 | |
| Remained sucrose | 192.8 | 7 | 1.5 g/L | 28 | 21 | Malbaša and others 30 |
| 11 | 10 | 2 bags | 24 ± 3 | 60 | Chen and Liu 25 | |
| 2.09 | 7 | 1.5 g/L | 28 | 21 | Lončar and others 27 |
Acetic acid bacteria from kombucha produce acetic acid, as one of the main metabolites, when sucrose is used as a carbon source. Many authors determined the content of acetic acid in the beverage obtained after cultivation of kombucha on traditional substrate. Chen and Liu 25 followed extended kombucha fermentation and determined the highest rate of 11 g/L after 30 d. The trend of acetic acid content was slow, increased with time, and then gradually decreased to 8 g/L, at the end of fermentation (60 d; Table 1). The same pattern was established by Jayabalan and others26 who monitored the fermentation until the 18th day on green tea (12 g/L) sweetened with 10% sucrose. The highest content was 9.5 g/L on the 15th day. Molasses was used in place of sucrose by Malbaša and others 30. Kombucha fermentation on molasses produced only 50% of acetic acid in comparison with sucrose at the same stage of fermentation. This might be due to the poor growth of acetic acid bacteria on molasses.
Glucuronic and gluconic acids are also major organic acids that are produced as a result of the kombucha fermentation process on traditional substrate. Lončar and others 27 determined the glucuronic acid after kombucha fermentation on sweetened black tea. The highest amount was measured after 7, and 21 d (0.0034 g/L; Table 1). Jayabalan and others 26 established the maximum value of 2.33 g/L D-glucuronic acid after 12 d of fermentation. Chen and Liu 25 determined that gluconic acid was not produced until the 6th day of fermentation. The ending concentration amounted the about 39 g/L after 60 d (Table 1).
Yavari and others 31 cultivated kombucha on sour cherry juice sweetened with 0.6%, 0.8%, and 1% sucrose. Glucuronic acid was produced in very large amounts of 132.5 g/L which was determined on the 14th day of fermentation, in substrate with 0.8% sucrose. The fermentation process was conducted at 37 °C. Yavari and others 32 used response surface methodology to predict the value of glucuronic acid content in kombucha beverage obtained after fermentation on grape juice sweetened with 0.7% sucrose, and the highest value was achieved after 14 d of fermentation at 37 °C. Franco and others 33 established the presence of glucuronic (0.07 to 9.63 g/L) and gluconic (0.04 to 1.16 g/L) acids in a product obtained after kombucha cultivation on black tea sweetened with glucose (0.062% to 1.51%). Yang and others 10 also determined the presence of gluconic acid and 2-keto gluconic acid, after cultivation of Gluconacetobacter sp. A4 isolated from kombucha and a strain of lactic acid bacteria, on 5 g/L black tea sweetened with 10% glucose.
L-lactic acid is not a characteristic compound for traditional kombucha beverage, but it is detected and determined. Jayabalan and others 26 examined kombucha prepared with green tea to have a higher concentration of lactic acid than kombucha prepared from black tea and tea waste material. The maximum value of 0.54 g/L was established on the 3rd day. Malbaša and others 34 measured the content of L-lactic acid after kombucha fermentation on molasses and established that it is a metabolic product present in large amounts. The presence of L-lactic acid after kombucha fermentation on molasses can be correlated to the L-lactic content of molasses itself which can be produced as a result of degradation of invert sugar in molasses. Molasses also contains amino nitrogen and biotin, which affect the intensity of kombucha fermentation.
The composition of kombucha beverage indicates the presence of numerous compounds and it depends on cultivation substrate, time and temperature of fermentation process, as well as the microorganisms present in the culture, but also on the applied method of analysis.
How to make kombucha
The amounts of tea, sugar, and tea fungus differ in different places. The standard procedure is as follows:
- Tap water (1 L) is boiled and during boiling 50 g sucrose is stirred in.
- Then 5 g tea leaves is added and removed by filtration after 5 min.
- After cooling to room temperature (20 ºC) the tea is inoculated with 24 g tea fungus (the culture) and poured into a beaker (1 L) previously sterilized with boiling water.
- The growth of undesirable microorganisms is inhibited by the addition of 0.2 L previously fermented kombucha, thus lowering the pH.
- The beaker is covered with a paper towel to keep insects, especially Drosophila fruit flies away.
- The incubation is carried out at 20 ºC to 22 ºC. The optimal temperature is in the wide range of 18 ºC and 26 ºC.
- In the next few days, the newly formed daughter culture will start to float and form a clear thin gel-like membrane across the available surface. This is the newly formed tea fungus available as a new layer above the old tea fungus which was inoculated to begin the fermentation. At this time, the tea will start to smell fermented and there will be gas bubbles appearing from the carbonic acid produced during the fermentation. The mother culture will remain at its original volume as it sinks to the bottom of the tea broth where it remains under the newly forming daughter culture.
- After 10 to 14 d, a new tea fungus will have developed on the surface of the tea as a disc of 2-cm thickness covering the whole diameter of the beaker. The newly formed tea fungus is removed with a spoon and kept in a small volume of fermented tea. The remaining beverage is filtered and stored in capped bottles at 4 ºC 35.
- The taste of the kombucha changes during fermentation from a pleasantly fruity sour-like sparkling flavor after a few days to a mild vinegar-like taste after a long incubation period. The 50 g sucrose/L provide the optimal concentrations of ethanol and lactic acid, and this sugar concentration has been used in traditional recipes for the preparation of “teakwass” (another name for kombucha) for a long time 35. An optimum fermentation time is required for the production of kombucha with pleasant flavor and taste. Longer fermentation produces high levels of acids (like mild vinegar) that may pose potential risks when consumed 36.
Kombucha side effects
Although kombucha tea has been reported to have curative effects, there is some evidence of toxicity associated with it. Some individuals have reported dizziness and nausea after consuming certain kombucha products. Two cases of unexplained severe illness have also been reported following kombucha consumption 37. Kombucha tea is contraindicated in pregnant and lactating women. It has been found to cause lead poisoning and gastrointestinal toxicity in 2 individuals. The presence of anthrax Bacillus in kombucha tea fermented in unhygienic condition was reported by Sadjadi 38. Further, Gamundi and Valdivia 39 stated the risks of consuming kombucha beverage by HIV-positive patients. Side effects like allergic reactions, jaundice, nausea, vomiting, and head and neck pain related to consumption of kombucha were reported in 4 patients 40. A married couple who had been drinking kombucha tea for 6 months, which was brewed in a ceramic pot, was reported to have symptomatic lead poisoning requiring chelation therapy 41. It was postulated that acids in the drink eluted lead from the glaze pigment used in the ceramic pot. Sabouraud and others 42 reported cases of lead poisoning in adults identified as anemia due to the lead-glazed earthenware jug which was used to store kombucha. A case of acute renal failure with lactic acidosis and hyperthermia within 15 h of kombucha tea ingestion by a 22-y-old HIV-positive male with a blood lactate level of 12.9 mmol/L and serum creatinine of 2.1 mg/dL was recorded 43. However, all of these cases were very isolated and involved only a small number of individuals. Moreover, there is no substantial evidence to confirm the toxicity of any kombucha tea or the occurrence of illness by earlier studies 44.
- Jayabalan, R., Malbaša, R. V., Lončar, E. S., Vitas, J. S. and Sathishkumar, M. (2014), A Review on Kombucha Tea—Microbiology, Composition, Fermentation, Beneficial Effects, Toxicity, and Tea Fungus. Comprehensive Reviews in Food Science and Food Safety, 13: 538–550. doi:10.1111/1541-4337.12073[↩]
- Chen C, Liu BY. 2000. Changes in major components of tea fungus metabolites during prolonged fermentation. J Appl Microbiol 89:834–9. https://www.ncbi.nlm.nih.gov/pubmed/11119158[↩]
- Ernst E, Kombucha: A Systematic Review of the Clinical Evidence. 2003;10:85-87[↩]
- Jarrell J, Cal T, Bennett JW. 2000. The kombucha consortia of yeasts and bacteria. Mycologist 14:166–70.[↩]
- Hesseltine CW. 1965. A millenium of fungi. Food and fermentation. Mycologia 57:148–67.[↩]
- Roussin MR. 1996. Analyses of kombucha ferments: Report on growers. Information Resources, LC, Salt Lake City, Utah, USA.[↩][↩]
- Markov SL, Malbaša RV, Hauk MJ, Cvetković DD. 2001. Investigation of tea fungus microbe associations. The yeasts. Acta Period Technol 32:133–8.[↩][↩]
- Sievers M, Lanini C, Weber A, Schuler-Schmid U, Teuber M. 1995. Microbiology and fermentation balance in a kombucha beverage obtained from a tea fungus fermentation. Syst Appl Microbiol 18:590–4.[↩]
- Liu CH, Hsu WH, Lee FL, Liao CC. 1996. The isolation and identification of microbes from a fermented tea beverage, Haipao, and their interactions during Haipao fermentation. Food Microbiol 13:407–15.[↩][↩]
- Yang Z, Zhou F, Ji B, Li B, Luo Y, Yang L, Li T. 2010. Symbiosis between microorganisms from kombucha and kefir: potential significance to the enhancement of kombucha function. Appl Biochem Biotechnol 160:446–55.[↩][↩]
- Boesch T, Trček J, Sievers M, Teuber M. 1998. Acetobacter intermedius, sp. nov. Syst Appl Microbiol 21:220–9.[↩]
- Dutta D, Gachhui R. 2007. Nitrogen-fixing and cellulose-producing Gluconacetobacter kombuchae sp. nov., isolated from kombucha tea. Int J Syst Evol Microbiol 57:353–7.[↩]
- Marsh AJ, O’Sullivan O, Hill C, Ross RP, Cotter PD. 2014. Sequence-based analysis of the bacterial and fungal compositions of multiple kombucha (tea fungus) samples. Food Microbiol 38:171–8.[↩][↩]
- Kozaki M, Koizumi A, Kitahara K. 1972. Microorganisms of zoogloeal mats formed in tea decoction. J Food Hyg Soc (Jpn) 13:89–96.[↩][↩]
- Safak S, Mercan N, Aslim B, Beyatli Y. 2002. A study on the production of poly-beta-hydroxybutyrate by some eukaryotic microorganisms. Turk Electron J Biotechnol Special issue 11–7.[↩][↩]
- Ramadani AS, Abulreesh HH. 2010. Isolation and identification of yeast flora in local kombucha sample: AL NABTAH. Umm Al Qura Univ J App Sci 2:42–51.[↩][↩]
- Teoh AL, Heard G, Cox J. 2004. Yeast ecology of kombucha fermentation. Int J Food Microbiol 95:119–26.[↩][↩][↩]
- Herrera T, Calderon-Villagomez A. 1989. Species of yeasts isolated in Mexico from the tea fungus. Rev Mex Micol 5:205–10.[↩][↩][↩]
- Jayabalan R, Marimuthu S, Thangaraj P, Sathishkumar M, Binupriya AR, Swaminathan K, Sei EY. 2008b. Preservation of kombucha teaeffect of temperature on tea components and free radical scavenging properties. J Agri Food Chem 56:9064–71.[↩][↩]
- Mayser P, Fromme S, Leitzmann C, Gründer K. 1995. The yeast spectrum of “tea fungus Kombucha”. Mycodes 38:289–95.[↩]
- Kurtzman CP, Robnett CJ, Basehoar-Powers E. 2001. Zygosaccharomyces kombuchaensis, a new ascosporogenous yeast from “kombucha tea”. FEMS Yeast Res 1:133–8.[↩]
- Steels H, James SA, Bond, CJ, Roberts IN, Straford M. 2002. Zygosaccharomyces kombuchaensis: the physiology of a new species related to the spoilage yeasts Zygosaccharomyces lentus and Zygosaccharomyces bailii. FEMS Yeast Res 2:113–21.[↩]
- Vitas JS, Malbaša RV, Grahovac JA, Lončar ES. 2013. The antioxidant activity of kombucha fermented milk products with stinging nettle and winter savory. CI&CEQ 19:129–39. [↩]
- Dufresne C, Farnworth E. 2000. Tea, kombucha, and health: a review. Food Res Int 33:409–21.[↩]
- Chen C, Liu BY. 2000. Changes in major components of tea fungus metabolites during prolonged fermentation. J Appl Microbiol 89:834–9.[↩][↩][↩][↩][↩][↩][↩][↩]
- Jayabalan R, Marimuthu S, Swaminathan K. 2007. Changes in content of organic acids and tea polyphenols during kombucha tea fermentation. Food Chem 102:392–8.[↩][↩][↩][↩][↩][↩]
- Lončar ES, Petrović SE, Malbaša RV, Verac RM. 2000. Biosynthesis of glucuronic acid by means of tea fungus. Nahrung 44:138–9.[↩][↩][↩][↩][↩]
- Lončar ES, Malbaša RV, Kolarov LjA. 2001. Metabolic activity of tea fungus on molasses as a source of carbon. Acta Period Technol 32:21–6.[↩]
- Lončar ES, Malbaša RV, Kolarov LA. 2007. Kombucha fermentation on raw extracts of different cultivars of Jerusalem artichoke. Acta Period Technol 38:37–44.[↩]
- Malbaša RV, Lončar ES, Kolarov LJA. 2002a. Sucrose and inulin balance during tea fungus fermentation. Roum Biotechnol Lett 7:573–6.[↩][↩][↩][↩]
- Yavari N, Mazaheri Assadi M, Larijani K, Moghadam MB. 2010. Response surface methodology for optimization of glucuronic acid production using kombucha layer on sour cherry juice. Aust J Basic Appl Sci 4(8):3250–6.[↩]
- Yavari N, Assadi MM, Moghadam MB, Larijani K. 2011. Optimizing glucuronic acid production using tea fungus on grape juice by response surface methodology. Aust J Basic Appl Sci 5:1788–94.[↩]
- Franco VG, Perín JC, Mantovani VE, Goicoechea HC. 2006. Monitoring substrate and products in a bioprocess with FTIR spectroscopy coupled to artificial neural networks enhanced with a genetic-algorithm-based method for wavelength selection. Talanta 68:1005–12.[↩]
- Malbaša R, Lončar E, Djurić M, Došenović I. 2008b. Effect of sucrose concentration on the products of kombucha fermentation on molasses. Food Chem 108:926–32.[↩]
- Reiss J. 1994. Influence of different sugars on the metabolism of the tea fungus. Z Lebensm Unters For 198:258–61.[↩][↩]
- Sreeramulu G, Zhu Y, Knol W. 2000. Kombucha fermentation and its antimicrobial activity. J Agri Food Chem 48:2589–94. https://www.ncbi.nlm.nih.gov/pubmed/10888589[↩]
- Centers for Disease Control and Protection. 1995. Unexplained severe illness possibly associated with consumption of kombucha tea—Iowa, 1995. MMWR 44(48):892–3, 899–900. https://www.cdc.gov/mmwr/preview/mmwrhtml/00039742.htm[↩]
- Sadjadi J. 1998.Cutaneous anthrax associated with the kombucha mushroom in Iran. J Am Med Assoc 280:1567–8.[↩]
- Gamundi R, Valdivia M. 1995. El hongo Kombucha: dos opiniones distintas. [The Kombucha mushroom: two different opinions]. SIDAhora : un proyecto del Departamento de Publicaciónes del PWA Coalition, NY (Sidahora) Oct-Nov:35–5.[↩]
- Srinivasan R, Smolinske S, Greenbaum, D. 1997. Probable gastrointestinal toxicity of kombucha tea: is this beverage healthy or harmful?. J Gen Intern Med 12:643–4.[↩]
- Phan TG, Estell J, Duggin G, Beer I, Smith D, Ferson MJ. 1998. Lead poisoning from drinking kombucha tea brewed in a ceramic pot. Med J Aust 169:644–6.[↩]
- Sabouraud S, Coppéré B, Rousseau C, Testud F, Pulce C, Tholly F, Blanc M, Culoma F, Facchin A, Ninet J, Chambon P, Medina B, Descotes J. 2009. Environmental lead poisoning from lead-glazed earthenware used for storing drinks. Rev Med Interne 30:1038–43.[↩]
- Kole AS, Jones HD, Christensen R, Gladstein J. 2009. A case of kombucha tea toxicity. J Intensive Care Med 24:205–7.[↩]
- Vijayaraghavan R, Singh M, Rao PVL, Bhattacharya R, Kumar P, Sugendran K, Kumar O, Pant SC, Singh R. 2000. Subacute (90 days) oral toxicity studies of kombucha tea. Biomed Environ Sci 13:293–9.[↩]





