Contents
- What is leukemia
- How leukemia is classified
- Symptoms of leukemia
- Leukemia causes
- Leukemia Diagnosis
- Treatment for leukemia
- Acute Lymphoblastic Leukemia
- What causes Acute Lymphoblastic Leukemia
- Risk Factors for acute lymphoblastic leukaemia
- Signs and Symptoms of Acute Lymphoblastic Leukemia
- How Is Acute Lymphocytic Leukemia Classified?
- Prognostic factors
- How Is Acute Lymphocytic Leukemia Diagnosed?
- Acute Lymphocytic Leukemia Treatment
- Long-Term and Late Effects of Treatment
- Chronic Lymphocytic Leukemia
- What Causes Chronic Lymphocytic Leukemia?
- Risk Factors for Chronic Lymphocytic Leukemia
- Signs and Symptoms of chronic lymphocytic leukemia
- Chronic Lymphocytic Leukemia Diagnosis
- Chronic Lymphocytic Leukemia Staging
- Prognostic factors for chronic lymphocytic leukemia
- Chronic Lymphocytic Leukemia Treatment
- Complications of Chronic lym:phocytic leukemia
- Acute myeloid leukemia
- How Many People Survive 5 Years Or More after Being Diagnosed with Acute Myeloid Leukemia?
- What Causes Acute Myeloid Leukemia To Develop?
- Risk Factors for acute myeloid leukemia
- How Is Acute Myeloid Leukemia Classified?
- The French-American-British (FAB) classification of Acute Myeloid Leukemia (AML)
- World Health Organization (WHO) classification of Acute Myeloid Leukemia (AML)
- Prognostic factors for acute myeloid leukemia
- Signs and Symptoms of acute myeloid leukemia
- How is acute myeloid leukemia diagnosed
- Acute Myeloid Leukemia Treatment
- What is AML Chemotherapy?
- Stem Cell Transplantation
- Childhood Acute Myeloid Leukemia (AML)
- Acute Myeloid Leukemia Treatment Outcomes
- Refractory Acute Myeloid Leukemia
- Relapsed Acute Myeloid Leukemia
- Chronic myeloid leukemia
- Coping and support
What is leukemia
Leukemia is cancer that starts in the body’s blood-forming tissues, including the bone marrow and the lymphatic system. Most blood cells develop from cells in the bone marrow called stem cells. In a person with leukemia, the bone marrow makes abnormal white blood cells. The abnormal cells are leukemia cells 1. White blood cells help your body fight infection — they normally grow and divide in an orderly way, as your body needs them.
Unlike normal blood cells, leukemia cells don’t die when they should. These cells do not work the way they should and they crowd out normal white blood cells, red blood cells, and platelets in the bone marrow. This makes it hard for normal blood cells to do their work.
Different types of leukemia depend on the type of blood cell that becomes cancer. For example, lymphoblastic leukemia is a cancer of the lymphoblasts (white blood cells, which fight infection). White blood cells are the most common type of blood cell to become cancer. But red blood cells (cells that carry oxygen from the lungs to the rest of the body) and platelets (cells that clot the blood) may also become cancer.
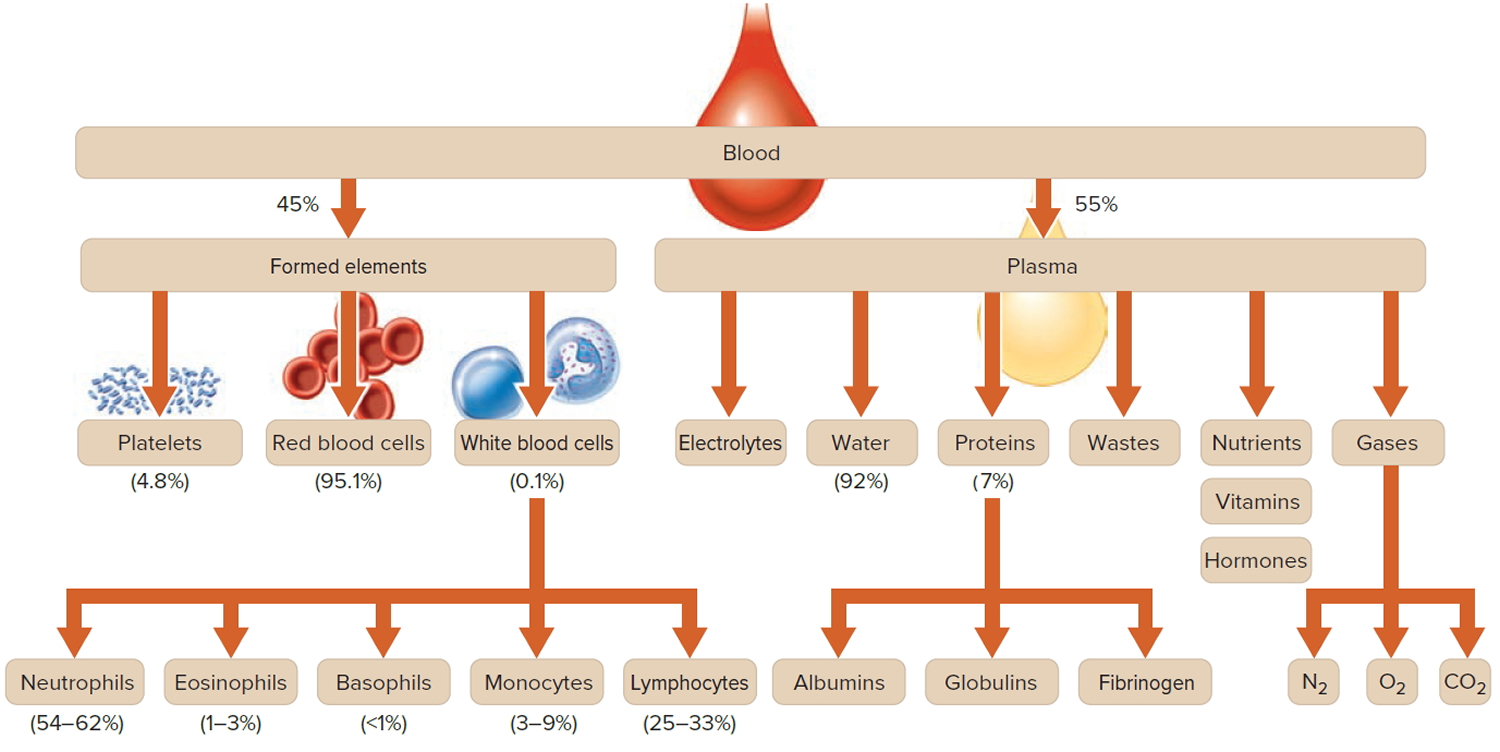
Figure 1. Blood composition
Note: Blood is a complex mixture of formed elements in a liquid extracellular matrix, called blood plasma. Note that water and proteins account for 99% of the blood plasma.
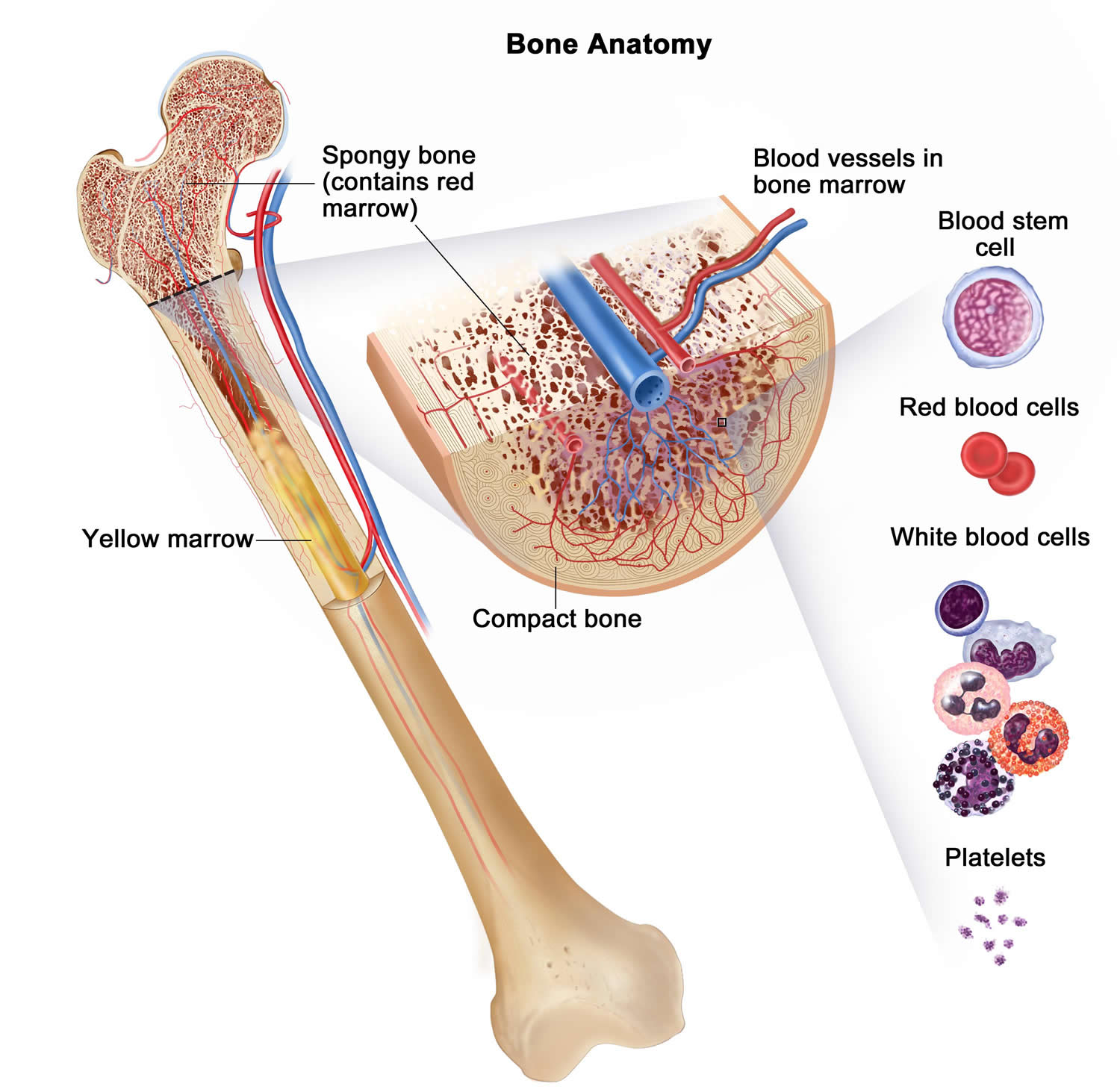
Figure 2. Bone marrow anatomy
Anatomy of the bone. The bone is made up of compact bone, spongy bone, and bone marrow. Compact bone makes up the outer layer of the bone. Spongy bone is found mostly at the ends of bones and contains red marrow. Bone marrow is found in the center of most bones and has many blood vessels. There are two types of bone marrow: red and yellow. Red marrow contains blood stem cells that can become red blood cells, white blood cells, or platelets. Yellow marrow is made mostly of fat.
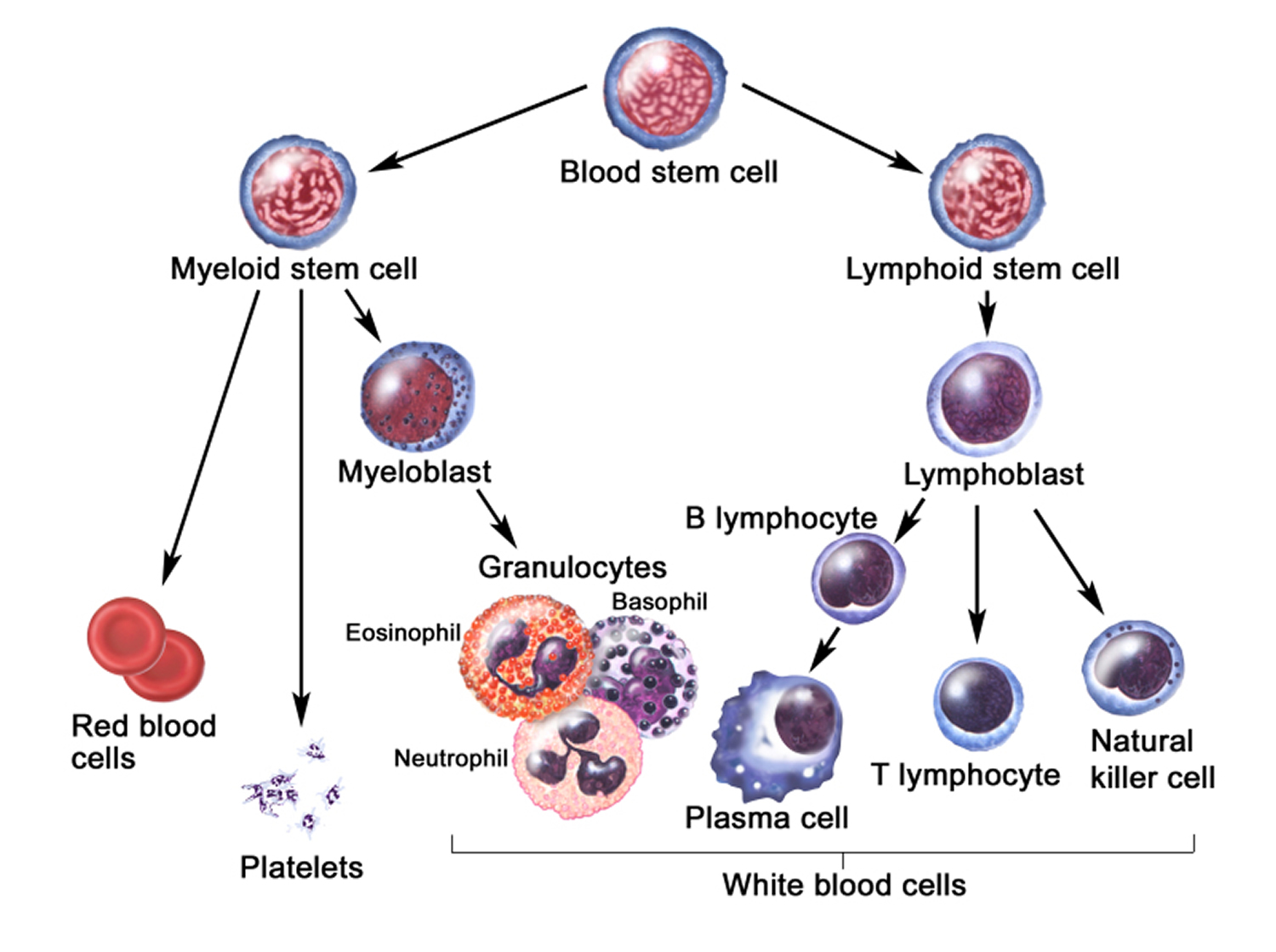
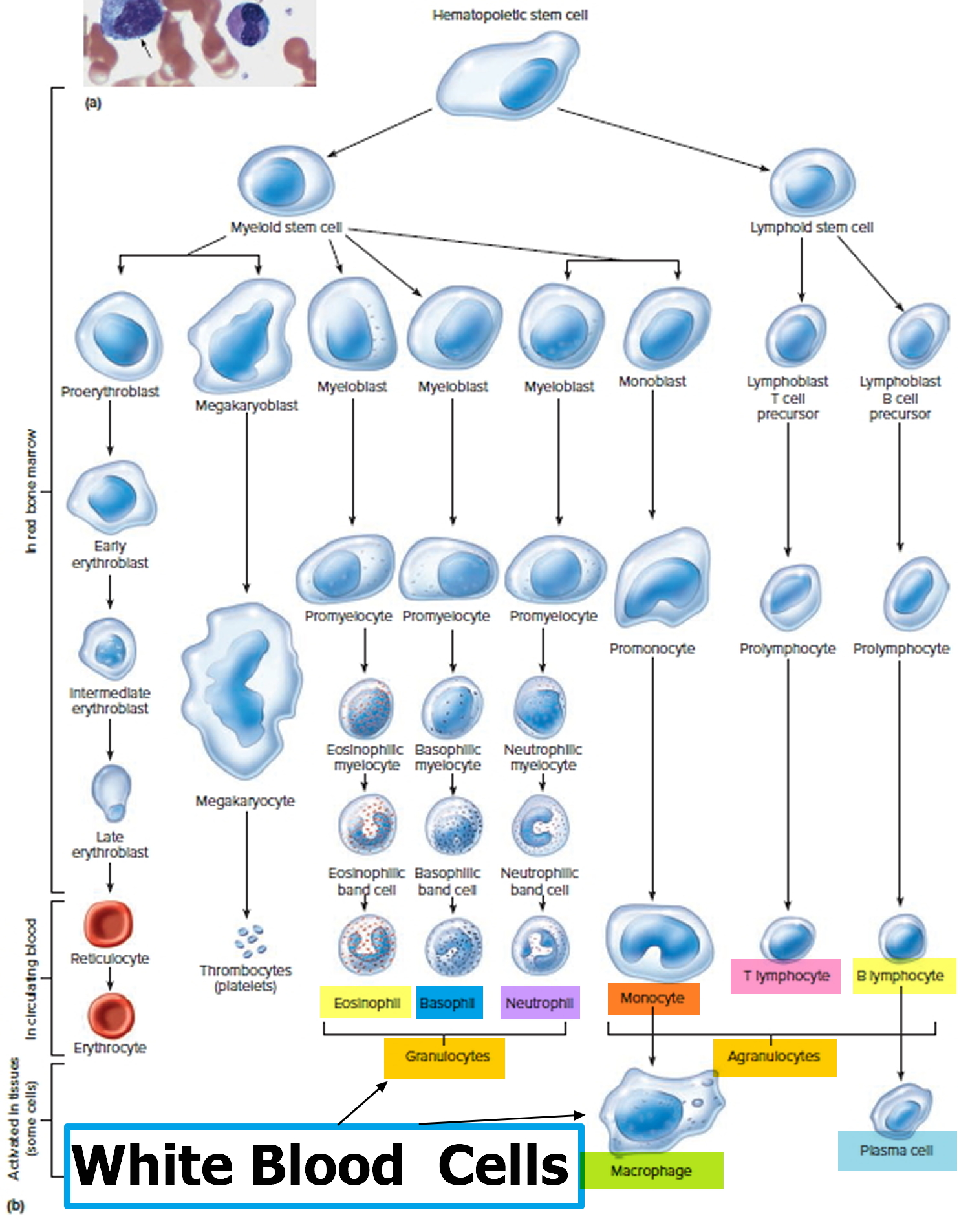
Figure 3. White blood cells development. A blood stem cell goes through several steps to become a red blood cell, platelet, or white blood cell
Figure 4. White blood cells development
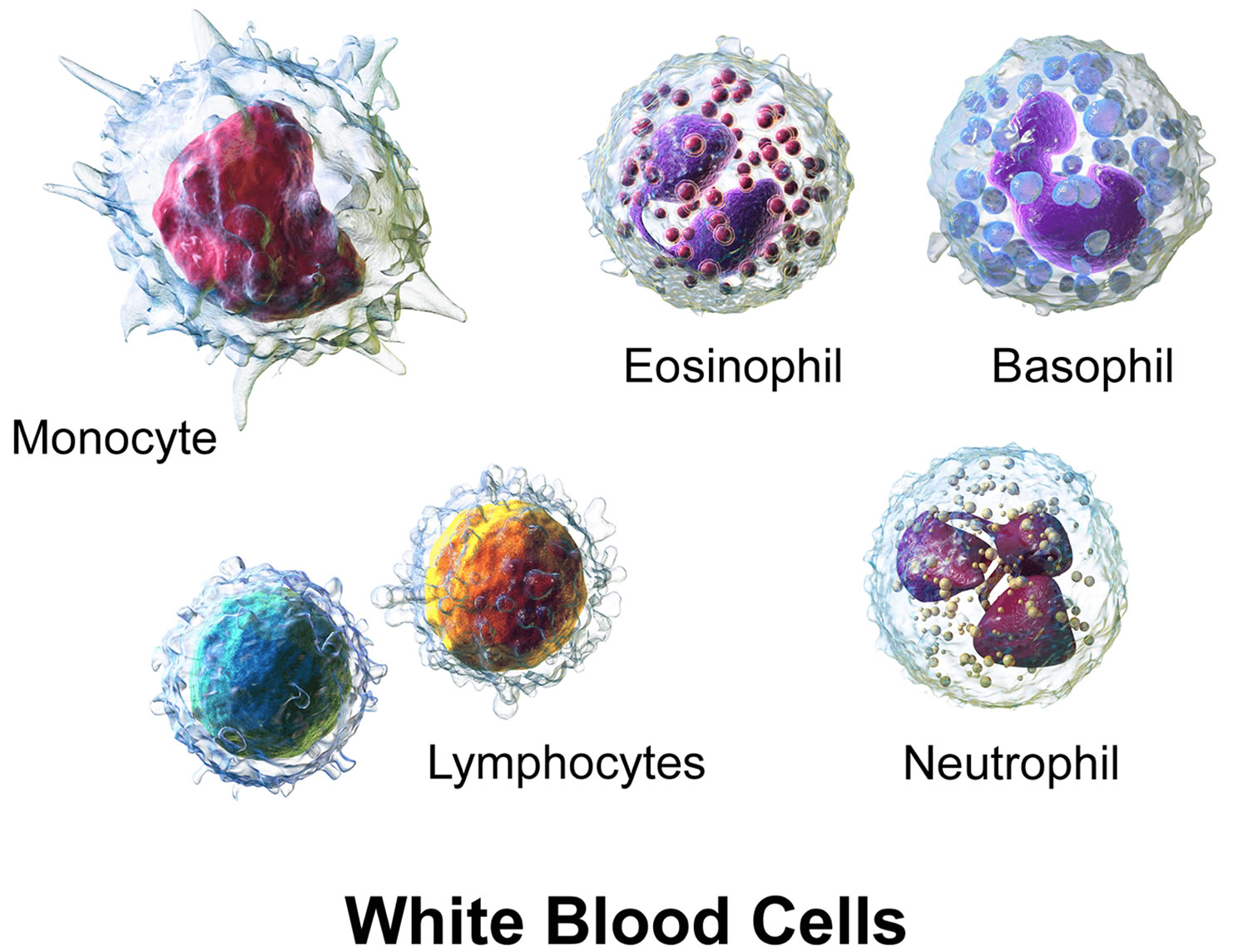
Figure 5. White blood cells
Leukemia occurs most often in adults older than 55 years, but it is also the most common cancer in children younger than 15 years.
Leukemia can be either acute or chronic. Acute leukemia is a fast-growing cancer that usually gets worse quickly. Chronic leukemia is a slower-growing cancer that gets worse slowly over time. In acute leukemia, the cells are very abnormal and their number increases rapidly. Adults can get either type; children with leukemia most often have an acute type. Some leukemias can often be cured. Other types are hard to cure, but you can often control them.
- There is no standard staging system for leukemia. The disease is described as untreated, in remission, or recurrent 2.
Treatment for leukemia can be complex — depending on the type of leukemia and other factors. But there are strategies and resources that can help to make your treatment successful.
Treatments may include chemotherapy, radiation and stem cell transplantation. Even if symptoms disappear, you might need therapy to prevent a relapse.
The treatment and prognosis for leukemia depend on the type of blood cell affected and whether the leukemia is acute or chronic.
The major types of leukemia are:
- Acute lymphoblastic leukaemia (ALL). This is the most common type of leukemia in young children. ALL can also occur in adults.
- Acute myelogenous leukemia (AML). AML is a common type of leukemia. It occurs in children and adults. AML is the most common type of acute leukemia in adults.
- Chronic lymphocytic leukemia (CLL). With CLL, the most common chronic adult leukemia, you may feel well for years without needing treatment.
- Chronic myelogenous leukemia (CML). This type of leukemia mainly affects adults. A person with CML may have few or no symptoms for months or years before entering a phase in which the leukemia cells grow more quickly.
- Other types. Other, rarer types of leukemia exist, including hairy cell leukemia, myelodysplastic syndromes and myeloproliferative disorders.
Rarer forms of lymphocytic leukemia
Prolymphocytic leukemia (PLL): In this type of leukemia the cancer cells are similar to normal cells called prolymphocytes — immature forms of B lymphocytes (B-PLL) or T lymphocytes (T-PLL). Both B-PLL and T-PLL tend to be more aggressive than the usual type of chronic lymphocytic leukemia (CLL). Most people will respond to some form of treatment, but over time they tend to relapse. PLL may develop in someone who already has chronic lymphocytic leukemia (CLL) (in which case it tends to be more aggressive), but it can also occur in people who have never had chronic lymphocytic leukemia (CLL).
Large granular lymphocyte (LGL) leukemia: This is another rare form of chronic leukemia. The cancer cells are large and have features of either T lymphocytes or another type of lymphocyte called natural killer (NK) cells. Most LGL leukemias are slow-growing, but a small number are more aggressive. Drugs that suppress the immune system may be helpful, but aggressive cases are very hard to treat.
Hairy cell leukemia (HCL): This is another cancer of lymphocytes that tends to progress slowly. It accounts for about 2% of all leukemias. The cancer cells are a type of B lymphocyte but are different from those seen in chronic lymphocytic leukemia (CLL). There are also important differences in symptoms and treatment. This type of leukemia gets its name from the way the cells look under the microscope — they have fine projections on their surface that make them look “hairy.”
The overall five-year relative survival rate for leukemia has more than quadrupled since 1960. From 1960 to 1963, the five-year relative survival rate among whites (only data available) with leukemia was 14 percent. From 1975 to 1977, the five-year relative survival rate for the total population with leukemia was 34.2 percent, and from 2006 to 2012, the overall relative survival rate was 62.7 percent.
From 2006-2012, the five-year relative survival rates overall were 3:
- Chronic myeloid leukemia (CML) – 65.9 percent
- Chronic lymphocytic leukemia (CLL) – 85.1 percent
- Acute myeloid leukemia (AML) – 26.8 percent overall and 66.8 percent for children and adolescents younger than 15 years
- Acute lymphoblastic leukaemia (ALL) – 70.7 percent overall, 92.3 percent for children and adolescents younger than 15 years, and 94.1 percent for children younger than 5 years.
How leukemia is classified
Doctors classify leukemia based on its speed of progression and the type of cells involved.
The first type of classification is by How Fast the Leukemia Progresses:
- Acute leukemia. In acute leukemia the bone marrow cells cannot mature properly, the abnormal blood cells are immature blood cells (blasts). Immature leukemia (blasts) cells continue to reproduce and build up. They can’t carry out their normal functions, and they multiply rapidly, so the disease worsens quickly. Acute leukemia requires aggressive, timely treatment. Without treatment, most people with acute leukemia would live only a few months. Some types of acute leukemia respond well to treatment, and many patients can be cured. Other types of acute leukemia have a less favorable outlook.
- Chronic leukemia. There are many types of chronic leukemias. Some produce too many cells and some cause too few cells to be produced. In chronic leukemia, the cells can mature partly but not completely. These cells may look fairly normal, but they generally do not fight infection as well as normal white blood cells do. They also live longer, build up, and crowd out normal cells. These blood cells replicate or accumulate more slowly and can function normally for a period of time. Some forms of chronic leukemia initially produce no early symptoms and can go unnoticed or undiagnosed for years. Chronic leukemias tend to progress over a longer period of time, and most people can live for many years. But chronic leukemias are generally harder to cure than acute leukemias.
The second type of classification is by Type of White Blood Cell affected:
- Lymphocytic leukemia. This type of leukemia affects the lymphoid cells (lymphocytes), which form lymphoid or lymphatic tissue. Lymphatic tissue makes up your immune system. Leukemias that start in immature forms of lymphocytes are called lymphocytic leukemias (also known as lymphoid or lymphoblastic leukemias).
- Myelogenous leukemia. This type of leukemia affects the myeloid cells. Myeloid cells give rise to red blood cells, white blood cells (other than lymphocytes) and platelet-producing cells (megakaryocytes) – are myeloid leukemias (also known as myelocytic, myelogenous, or non-lymphocytic leukemias).
Symptoms of leukemia
Leukemia symptoms vary, depending on the type of leukemia. Common leukemia signs and symptoms include:
- Fever or chills
- Persistent fatigue, weakness
- Frequent or severe infections
- Losing weight without trying
- Swollen lymph nodes, enlarged liver or spleen
- Easy bleeding or bruising
- Recurrent nosebleeds
- Tiny red spots in your skin (petechiae)
- Excessive sweating, especially at night
- Bone pain or tenderness
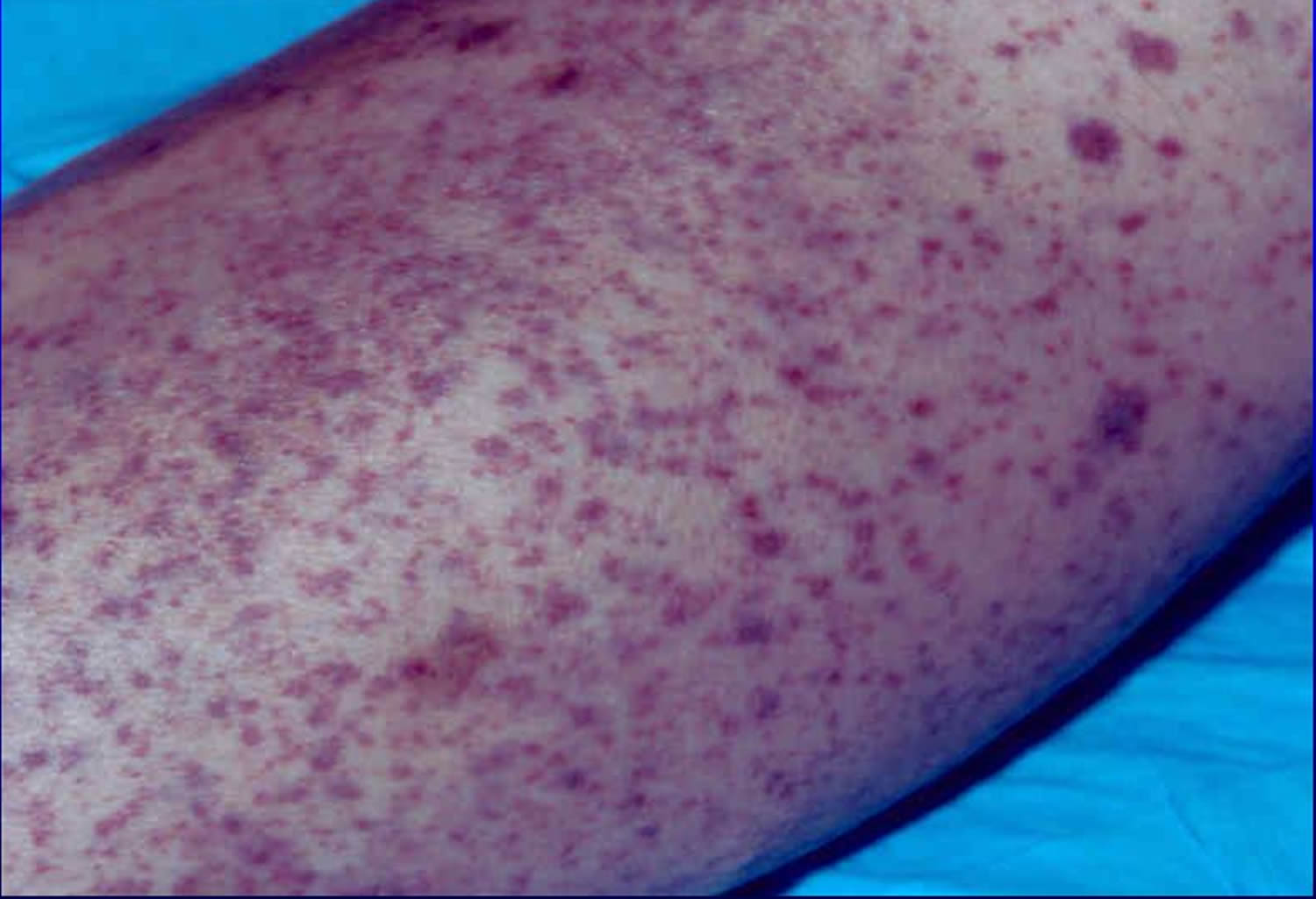
Figure 1. Petechiae
Leukemia causes
How leukemia forms
In general, leukemia is thought to occur when some blood cells acquire mutations in their DNA — the instructions inside each cell that guide its action. There may be other changes in the cells that have yet to be fully understood could contribute to leukemia.
Certain abnormalities cause the cell to grow and divide more rapidly and to continue living when normal cells would die. Over time, these abnormal cells can crowd out healthy blood cells in the bone marrow, leading to fewer healthy white blood cells, red blood cells and platelets, causing the signs and symptoms of leukemia.
Risk factors for leukemia
Factors that may increase your risk of developing some types of leukemia include:
- Previous cancer treatment. People who’ve had certain types of chemotherapy and radiation therapy for other cancers have an increased risk of developing certain types of leukemia.
- Genetic disorders. Genetic abnormalities seem to play a role in the development of leukemia. Certain genetic disorders, such as Down syndrome, are associated with an increased risk of leukemia.
- Exposure to certain chemicals. Exposure to certain chemicals, such as benzene — which is found in gasoline and is used by the chemical industry — also is linked to an increased risk of some kinds of leukemia.
- Smoking. Smoking cigarettes increases the risk of acute myelogenous leukemia.
- Family history of leukemia. If members of your family have been diagnosed with leukemia, your risk for the disease may be increased.
However, most people with known risk factors don’t get leukemia. And many people with leukemia have none of these risk factors.
Leukemia Diagnosis
Doctors may find chronic leukemia in a routine blood test, before symptoms begin. If this happens, or if you have signs or symptoms that suggest leukemia, you may undergo the following diagnostic exams:
- Physical exam. Your doctor will look for physical signs of leukemia, such as pale skin from anemia, swelling of your lymph nodes, and enlargement of your liver and spleen.
- Blood tests. By looking at a sample of your blood, your doctor can determine if you have abnormal levels of white blood cells or platelets — which may suggest leukemia.
- Bone marrow test. Your doctor may recommend a procedure to remove a sample of bone marrow from your hipbone. The bone marrow is removed using a long, thin needle. The sample is sent to a laboratory to look for leukemia cells. Specialized tests of your leukemia cells may reveal certain characteristics that are used to determine your treatment options.
You may undergo additional tests to confirm the diagnosis and to determine the type of leukemia and its extent in your body. Certain types of leukemia are classified into stages, indicating the severity of the disease. Your leukemia’s stage helps your doctor determine a treatment plan.
Treatment for leukemia
Treatment for your leukemia depends on many factors. Your doctor determines your leukemia treatment options based on your age and overall health, the type of leukemia you have, and whether it has spread to other parts of your body.
Common treatments used to fight leukemia include:
- Chemotherapy
Chemotherapy is the major form of treatment for leukemia. This drug treatment uses chemicals to kill leukemia cells.
Depending on the type of leukemia you have, you may receive a single drug or a combination of drugs. These drugs may come in a pill form, or they may be injected directly into a vein.
- Biological therapy
Biological therapy works by using treatments that help your immune system recognize and attack leukemia cells.
- Targeted therapy
Targeted therapy uses drugs that attack specific vulnerabilities within your cancer cells.
For example, the drug imatinib (Gleevec) stops the action of a protein within the leukemia cells of people with chronic myelogenous leukemia. This can help control the disease.
- Radiation therapy
Radiation therapy uses X-rays or other high-energy beams to damage leukemia cells and stop their growth. During radiation therapy, you lie on a table while a large machine moves around you, directing the radiation to precise points on your body.
You may receive radiation in one specific area of your body where there is a collection of leukemia cells, or you may receive radiation over your whole body. Radiation therapy may be used to prepare for a stem cell transplant.
- Stem cell transplant
A stem cell transplant is a procedure to replace your diseased bone marrow with healthy bone marrow.
Before a stem cell transplant, you receive high doses of chemotherapy or radiation therapy to destroy your diseased bone marrow. Then you receive an infusion of blood-forming stem cells that help to rebuild your bone marrow.
You may receive stem cells from a donor, or in some cases you may be able to use your own stem cells. A stem cell transplant is very similar to a bone marrow transplant.
Acute Lymphoblastic Leukemia
Acute lymphoblastic leukemia (ALL), also called acute lymphocytic leukemia, is a cancer that starts from the early version of white blood cells called lymphocytes in the bone marrow (the soft inner part of the bones, where new blood cells are made).
The term “acute” means that the leukemia can progress quickly, and if not treated, would probably be fatal within a few months. Lymphocytic means it develops from early (immature) forms of lymphocytes, a type of white blood cell.
Other types of cancer that start in lymphocytes are known as lymphomas (non-Hodgkin lymphoma or Hodgkin disease). The main difference between these types of cancers is that leukemias like ALL mainly affects the bone marrow and the blood, and may spread to other places, while lymphomas mainly affect the lymph nodes or other organs but may involve the bone marrow. Sometimes cancerous lymphocytes are found in both the bone marrow and lymph nodes when the cancer is first diagnosed, which can make it hard to tell if the cancer is leukemia or lymphoma. If more than 25% of the bone marrow is replaced by cancerous lymphocytes, the disease is usually considered leukemia. The size of lymph nodes is also important. The bigger they are, the more likely the disease will be considered a lymphoma. For more information on lymphomas, see Non-Hodgkin Lymphoma and Hodgkin Disease.
- Acute lymphoblastic leukemia (ALL) a cancer of the bone marrow and blood
- Progresses rapidly without treatment
- Does not have a clear cause
- Both adults and children can be affected.
Acute lymphoblastic leukaemia is very rare, with the number of new cases of acute lymphocytic leukemia was 1.7 per 100,000 men and women per year 4. The number of deaths was 0.4 per 100,000 men and women per year. These rates are age-adjusted and based on 2010-2014 cases and deaths. In 2014, there were an estimated 81,837 people living with acute lymphocytic leukemia in the United States.
The American Cancer Society’s estimates for acute lymphocytic leukemia (ALL) in the United States for 2017 (including both children and adults) are:
- About 5,970 new cases of ALL (3,350 in males and 2,620 in females)
- About 1,440 deaths from ALL (800 in males and 640 in females)
The risk for developing ALL is highest in children younger than 5 years of age. The risk then declines slowly until the mid-20s, and begins to rise again slowly after age 50. Overall, about 4 of every 10 cases of ALL are in adults.
The average person’s lifetime risk of getting ALL is less than 1 in 750. The risk is slightly higher in males than in females, and higher in whites than in African Americans.
Most cases of acute lymphoblastic leukaemia occur in children, but most deaths from acute lymphoblastic leukaemia (about 4 out of 5) occur in adults. Children may do better because of differences in childhood and adult acute lymphoblastic leukaemia in the disease itself, differences in treatment (children’s bodies can often handle aggressive treatment better than adult’s), or some combination of these.
What causes Acute Lymphoblastic Leukemia
The cause of acute lymphoblastic leukaemia remains unknown at this time. Even when a person has one or more risk factors, there is no way to tell whether it actually caused the cancer.
During the past few years, scientists have made great progress in understanding how certain changes in DNA can cause normal bone marrow cells to become leukemia cells. Normal human cells grow and function based mainly on the information contained in each cell’s chromosomes. Chromosomes are like bundles of long molecules of DNA in each cell. DNA is the chemical that makes up your genes – the instructions for how our cells function. You look like your parents because they are the source of your DNA. But your genes affect more than the way you look.
Some genes contain instructions for controlling when your cells grow and divide. Certain genes that help cells grow and divide are called oncogenes. Others that slow down cell growth and division or cause them to die at the right time are called tumor suppressor genes.
Each time a cell prepares to divide into 2 new cells, it must make a new copy of the DNA in its chromosomes. This process is not perfect, and errors can occur that may affect genes within the DNA. Cancers can be caused by DNA mutations (changes) that turn on oncogenes or turn off tumor suppressor genes.
Translocations are the most common type of DNA change that can lead to leukemia. Human DNA is packaged in 23 pairs of chromosomes. A translocation means that DNA from one chromosome breaks off and becomes attached to a different chromosome. The point on the chromosome where the break occurs can affect genes – for example, it can turn on oncogenes or turn off genes that would normally help a cell mature.
The most common translocation in acute lymphoblastic leukaemia in adults is known as the Philadelphia chromosome, which is a swap of DNA between chromosomes 9 and 22, abbreviated as t(9;22). It occurs in about 1 out of 4 adult ALL cases. Other, less common translocations are those between chromosomes 4 and 11, t(4;11), or 8 and 14, t(8;14).
Other chromosome changes such as deletions (the loss of part of a chromosome) and inversions (the rearrangement of the DNA within part of a chromosome) can also affect the development of ALL, although they are less common. In many cases of acute lymphoblastic leukaemia, the gene changes that lead to the leukemia are not known.
Doctors are trying to figure out why these changes occur and how each of them might lead to leukemia. Not all cases of ALL have the same chromosome changes. Some changes are more common than others, and some seem to have more of an effect on a person’s prognosis (outlook) than others.
Some people with certain types of cancer have inherited DNA mutations from a parent. These changes increase their risk for the disease. But ALL is very rarely caused by one of these inherited mutations.
Usually DNA mutations related to ALL occur during the person’s lifetime rather than having been inherited before birth. They may result from exposure to radiation or cancer-causing chemicals, but in most cases the reason they occur is not known.
Therefore, acute lymphoblastic leukaemia may results from either an acquired or a genetic injury to the DNA (genetic material) of a developing stem cell in the bone marrow.
- Stem cells form blood cells (white cells, red cells and platelets).
- Although acute lymphoblastic leukaemia starts in a stem cell in the bone marrow, it can spread to other areas such as the central nervous system, the lymph nodes and, more rarely, the testes.
This damaged cell becomes a leukemic cell and multiplies uncontrollably into billions of cells called leukemic lymphoblasts.
- Leukemic lymphoblasts
- Do not function normally
- Block the production of normal cells
- Grow and survive better than normal cells
As a result, the number of healthy blood cells (red cells, white cells and platelets) is usually lower than normal.
- Anemia is a condition when there is a low number of red cells in the blood which can cause fatigue and shortness of breath.
- Neutropenia is a condition when there is a low number of white cells so that the immune system can’t effectively guard against infection due to a lack of neutrophils (a type of white cell).
- Thrombocytopenia is a condition when there is a low number of platelets which can cause bleeding and easy bruising with no apparent cause.
- Low numbers of all three blood cell counts is called pancytopenia.
Risk Factors for acute lymphoblastic leukaemia
Doctors don’t know why some cells become leukemic cells and others don’t. For most people who have acute lymphoblastic leukemia (ALL), there are no obvious reasons why they developed the disease.
Risk factors associated with the disease include:
- Exposure to high doses of radiation therapy (studied in survivors of atomic bomb detonations in Japan). A child exposed to multiple diagnostic x-rays may be at a slight increased risk of developing ALL, but further research is required to confirm these findings.
- Previous exposure to chemotherapy and radiotherapy may cause ALL in adults.
- Genetic disorders including: Down syndrome, neurofibromatosis, Klinefelter syndrome, Fanconi anemia, Schwachman syndrome, Bloom syndrome and ataxia telangiectasia have been associated with an increased risk of developing ALL.
- Certain chemical exposures: The risk of ALL may be increased by exposure to certain chemotherapy drugs and certain chemicals, including benzene. Benzene is a solvent used in the rubber industry, oil refineries, chemical plants, shoe manufacturing, and gasoline-related industries, and is also present in cigarette smoke, as well as some glues, cleaning products, detergents, art supplies, and paint strippers. Chemical exposure is more strongly linked to an increased risk of acute myeloid leukemia than to acute lymphoblastic leukaemia.
- Certain viral infections: Infection with the human T-cell lymphoma/leukemia virus-1 (HTLV-1) can cause a rare type of T-cell acute lymphocytic leukemia. Most cases occur in Japan and the Caribbean area. This disease is not common in the United States. In Africa, the Epstein-Barr virus (EBV) has been linked to Burkitt lymphoma, as well as to a form of acute lymphocytic leukemia. In the United States, EBV most often causes infectious mononucleosis (“mono”).
- Race/ethnicity: Acute lymphocytic leukemia is more common in whites than in African Americans, but the reasons for this are not clear.
- Gender: Acute lymphocytic leukemia is slightly more common in males than in females. The reason for this is unknown.
- Having an identical twin with ALL: Someone who has an identical twin who develops ALL in the first year of life has an increased risk of getting ALL.
Uncertain, unproven or controversial risk factors
Other factors that have been studied for a possible link to ALL include:
- Exposure to electromagnetic fields (such as living near power lines or using cell phones)
- Workplace exposure to diesel, gasoline, pesticides, and certain other chemicals
- Smoking
- Exposure to hair dyes
So far, none of these factors has been linked conclusively to ALL. Research in these areas continues.
Scientists continue to explore relationships between ALL and lifestyle or environmental factors. More developed countries and higher socioeconomic groups tend to have higher ALL rates.
Research supports the view that a number of complex factors are involved in the risk of developing ALL.
Currently, there’s no way to prevent the disease. You can’t catch ALL from someone else.
Prenatal Development
Some cases of acute lymphoblastic leukaemia relate to a mutation in a lymphocyte that occurs during the prenatal period (in utero). The leukemia is usually diagnosed in infancy or the first few years after birth. However, years may pass before the disease appears. With ALL, it seems that additional genetic abnormalities can occur after birth and allow the unregulated cell growth that triggers the disease, because there are more mutations found in utero than there are cases of childhood acute lymphoblastic leukaemia.
Signs and Symptoms of Acute Lymphoblastic Leukemia
The signs and symptoms of acute lymphoblastic leukemia (ALL) are common to other, less serious illnesses. However, if you’re troubled by any of the following symptoms, see your doctor:
- Aches in the arms, legs or hips
- Black-and-blue marks (bruises) for no clear reason
- Enlarged lymph nodes
- Fever without an obvious cause
- Pale skin
- Pinhead-size red spots under the skin (called petechiae)
- Prolonged bleeding from minor cuts
- Shortness of breath during normal physical activity
- Tiredness or no energy
- Vomiting
- Unexplained weight loss
- Bleeding, such as frequent or severe nosebleeds and bleeding gums
- Infections that don’t go away or keep coming back
- Feeling dizzy or lightheaded
- Feeling weak
Patients with ALL also often have several non-specific symptoms. These can include:
- Weight loss
- Fever
- Night sweats
- Fatigue
- Loss of appetite
Of course, these are not just symptoms of ALL and are more often caused by something other than leukemia.
Swelling in the abdomen
Leukemia cells may build up in the liver and spleen, causing them to enlarge. This might be noticed as a fullness or swelling of the belly or feeling full after eating only a small amount. The lower ribs usually cover these organs, but when they are enlarged the doctor can feel them.
Enlarged lymph nodes
ALL that has spread to lymph nodes close to the surface of the body (such as on the sides of the neck, in the groin, or in underarm areas), might be noticed as lumps under the skin. Lymph nodes inside the chest or abdomen may also swell, but these can be detected only by imaging tests such as CT or MRI scans.
Bone or joint pain
Sometimes leukemia cells build up near the surface of the bone or inside the joint and cause bone or joint pain.
Spread to other organs
Less often, ALL spreads to other organs:
- If ALL spreads to the brain and spinal cord it can cause headaches, weakness, seizures, vomiting, trouble with balance, facial numbness, or blurred vision.
- ALL may spread to the chest cavity, where it can cause fluid buildup and trouble breathing.
- Rarely, ALL may spread to the skin, eyes, testicles, kidneys, or other organs.
Symptoms from an enlarged thymus
The T-cell subtype of ALL often affects the thymus, which is a small organ in the middle of the chest behind the sternum (breastbone) and in front of the trachea (windpipe). An enlarged thymus can press on the trachea, causing coughing or trouble breathing.
The superior vena cava (SVC), a large vein that carries blood from the head and arms back to the heart, passes next to the thymus. If the thymus is enlarged, it may press on the SVC, causing the blood to “back up” in the veins. This is known as SVC syndrome. It can cause swelling in the face, neck, arms, and upper chest (sometimes with a bluish-red color). It can also cause headaches, dizziness, and a change in consciousness if it affects the brain. The SVC syndrome can be life-threatening, and needs to be treated right away.
How Is Acute Lymphocytic Leukemia Classified?
Most types of cancers are assigned numbered stages to describe their extent in the body, based on the size of the tumor and how far the cancer has spread.
Acute lymphocytic leukemia (ALL), on the other hand, does not usually form tumor masses. It generally affects all of the bone marrow in the body and, in many cases, might have spread to other organs, such as the liver, spleen, and lymph nodes. Therefore the outlook for the patient with ALL depends on other information, such as the subtype of ALL (determined by lab tests), the age of the patient, and other lab test results.
Different systems have been used to classify ALL into subtypes.
The French-American-British (FAB) classification
In the 1970s, a group of French, American, and British (FAB) leukemia experts divided ALL into 3 subtypes (L1, L2, and L3), based on the way the leukemia cells looked under the microscope after routine staining. This system has largely been replaced, as newer lab tests now allow doctors to classify ALL more accurately.
Classification based on immunophenotype
Doctors have found that cytogenetic tests, flow cytometry, and oter lab tests provide more detailed information about the subtype of ALL and the patient’s prognosis. These tests help divide ALL into groups based on the immunophenotype of the leukemia, which takes into account:
- The type of lymphocyte (B cell or T cell) the leukemia cells come from
- How mature these leukemia cells are
These groups have largely replaced the FAB classification. The subtypes of ALL are now named as follows:
B-cell ALL
- Early pre-B ALL (also called pro-B ALL) – about 10% of cases
- Common ALL – about 50% of cases
- Pre-B ALL – about 10% of cases
- Mature B-cell ALL (Burkitt leukemia) – about 4% of cases
T-cell ALL
- Pre-T ALL – about 5% to 10% of cases
- Mature T-cell ALL – about 15% to 20% of cases
The subtypes of ALL each carry a slightly different outlook (prognosis), but other factors (like gene changes in the leukemia cells) may also have an impact. Some of these prognostic factors are listed in the next section.
Mixed lineage acute leukemias
In recent years, newer lab tests have shown that a small number of acute leukemias actually have both lymphocytic and myeloid features. Sometimes the leukemia cells have both myeloid and lymphocytic traits in the same cells. In other cases, a person may have some leukemia cells with myeloid features and others with lymphocytic features. These types of leukemias may be called mixed lineage leukemia, ALL with myeloid markers (My+ ALL), AML with lymphoid markers, or biphenotypic acute leukemia (BAL).
Most studies suggest these leukemias tend to have a poorer outlook than standard subtypes of ALL or AML. Not all doctors agree on the best way to treat them.
Intensive treatment (such as a stem cell transplant) is often used when possible, as there is a high risk of recurrence after treatment.
Prognostic factors
As leukemia treatment has improved over the years, research has focused on why some people have a better chance for cure than others. Differences in patients that affect response to treatment are called prognostic factors. They help doctors decide if people with a certain type of leukemia should get more or less treatment.
Age
Younger patients tend to have a better prognosis than older patients. There is no set cutoff for this, but generally those younger than 50 do better than those in their 50s, while people in their 50s do better than those in their 60s or older.
Initial white blood cell count
People with a lower WBC count (less than 30,000 for B-cell ALL and less than 100,000 for T-cell ALL) at the time of diagnosis tend to have a better prognosis.
ALL subtype
In general, T-cell ALL has a better prognosis, while mature B-cell ALL (Burkitt leukemia) has a poorer prognosis. Other subtypes of B-cell ALL fall somewhere in between. It’s important to note that this doesn’t apply to all cases. For instance, some subtypes of T-cell ALL have a better outlook than others.
Chromosome abnormalities
The presence of a translocation between chromosomes 4 and 11 in the leukemia cells predicts a poorer outlook, so does extra chromosome 8 or a missing chromosome 7. The presence of Philadelphia chromosome (a translocation between chromosomes 9 and 22) used to predict a poorer outlook, but not if modern targeted therapy drugs are used.
Response to chemotherapy
Patients who go into a complete remission (no visible leukemia in the bone marrow – see below) within 4 to 5 weeks of starting treatment tend to have a better prognosis than those for whom this takes longer. Patients who don’t achieve a complete remission at all have a poorer outlook. The prognostic value of minimal residual disease (described below) is still being studied.
Status of acute lymphocytic leukemia after treatment
How well leukemia responds to treatment affects the patient’s long-term chance for recovery.
Remission
A remission (complete remission) is usually defined as having no evidence of leukemia after treatment. This means the bone marrow contains fewer than 5% blast cells, the blood cell counts are within normal limits, and there are no signs or symptoms of the disease. A molecular complete remission means no evidence of leukemia cells in the bone marrow is found, even when using very sensitive lab tests, such as polymerase chain reaction (PCR). Even when leukemia is in remission, this does not always mean that it has been cured.
Minimal residual disease
Minimal residual disease (MRD) is a term used after treatment when leukemia cells can’t be found in the bone marrow using standard lab tests (such as looking at cells under a microscope), but they can still be detected with more sensitive tests (such as flow cytometry or PCR). Patients with minimal residual disease after treatment are more likely to have the leukemia relapse (come back after treatment) and overall have a poorer outlook than those who achieve a complete remission. Doctors are looking to see if these patients could benefit from further or more intensive treatment.
Active disease
Active disease means that either there is evidence that the leukemia is still present during treatment or that the disease has relapsed (come back) after treatment. For a patient to be in relapse, more than 5% of the bone marrow must be made up of blast cells.
How Is Acute Lymphocytic Leukemia Diagnosed?
- Diagnosing acute lymphoblastic leukemia (ALL) and your ALL subtype usually involves a series of tests. An accurate diagnosis of the subtype is important. The exact diagnosis helps the doctor
- Estimate how the disease will progress
- Determine the appropriate treatment.
Tests your doctor may use to diagnose ALL:
Blood Tests
Your doctor needs to test your blood to make a diagnosis. Your blood is sent to a lab for:
- A complete blood count (CBC), which shows the number of red cells, white cells and platelets in your blood. Usually, patients with ALL have lower-than-expected red blood cells and platelets.
- A peripheral blood smear, which examines the cells within the blood and shows whether there are too many immature white cells (leukemic blast cells). The blood smear sample can also be used for
Cytogenetic analysis, which identifies certain changes in the number and size of chromosomes within cells.
Immunophenotyping, which identifies cells based on the types of proteins (antigens) on the cell surface to find out if the ALL cells are B cells or T cells. “Flow cytometry” is one type of test used for immunophenotyping.
Bone Marrow Tests
Your doctor or oncologist (cancer specialist) tests your bone marrow. Bone marrow testing involves two steps usually performed at the same time in a doctor’s office or a hospital:
- A bone marrow aspiration to remove a liquid marrow sample
- A bone marrow biopsy to remove a small amount of bone filled with marrow
For both procedures, the patient is given medication to numb the area, or given a general anesthesia, and the sample is taken from the hip bone.
The tests’ purpose is to confirm an ALL diagnosis and
- Find out the percentage of ALL cells are in your bone marrow
- Examine the ALL cells to find abnormalities.
Chromosome testing
Normal human cells contain 23 pairs of chromosomes (bundles of DNA). In some cases of leukemia, the cells have chromosome changes. Sometimes a piece of a chromosome is missing – called a deletion.
More often in ALL, 2 chromosomes swap some of their DNA, so that part of one chromosome becomes attached to part of a different chromosome. This is called a translocation. The most common chromosome change in adult ALL is a translocation between chromosomes 9 and 22 [often written t(9;22)], which results in a shortened chromosome 22 (called the Philadelphia chromosome). About 1 out of 4 adults with ALL have this abnormality in their leukemia cells. This change is especially important because it can be targeted with certain drugs.
Information about chromosome changes can be useful in predicting a person’s outlook and response to treatment. For this reason, chromosome testing is a standard part of the work-up of ALL patients.
Cytogenetics: For this test, the cells are grown in lab dishes until they start dividing and the chromosomes can be seen under a microscope. Then the chromosomes are looked at under a microscope to detect any changes.
Because it takes time for the cells to start dividing, cytogenetic testing often takes about 2 to 3 weeks. It is often used to look at cells in the bone marrow, but it can also be used to look at cells from the blood. An advantage of cytogenetic testing is that it looks at all of the chromosomes, and the doctor doesn’t have to know in advance what changes to test for.
Not all chromosome changes can be seen under a microscope. Other lab tests can often help find these changes.
Fluorescent in situ hybridization (FISH): This is another way to look at chromosomes and genes. It uses special fluorescent dyes that only attach to specific genes or parts of particular chromosomes. FISH can find most chromosome changes (such as translocations) that are visible under a microscope in standard cytogenetic tests, as well as some changes too small to be seen with usual cytogenetic testing.
FISH can be used on regular blood or bone marrow samples. Because the cells don’t have to be able to divide for this test, it can also be used to look at cells from other tissues, like lymph node samples. It is very accurate and can usually provide results within a couple of days. But because FISH only tests for certain gene changes (and doesn’t look at the chromosomes overall), it is best for looking for the changes that are important based on the kind of leukemia a person has.
Polymerase chain reaction (PCR): This is a very sensitive DNA test that can also find certain gene changes too small to be seen with a microscope, even if very few leukemia cells are present in a sample. Like FISH, it is used to find particular gene changes and not to look at the chromosomes overall. For ALL, it is often used to look for the gene made by the Philadelphia chromosome.
If the leukemia cells have a particular gene (or chromosome) change, PCR can be used after treatment to try to find small numbers of leukemia cells that may not be visible with a microscope.
Lumbar puncture (spinal tap)
ALL can spread to the area around the brain and spinal cord. To check for this spread, doctors remove a sample of the fluid from that area (cerebrospinal fluid or CSF) for testing.
You may lay on your side or sit up for this test. The doctor first numbs an area in the lower part of the back over the spine. A small, hollow needle is then placed between the bones of the spine and into the area around the spinal cord to collect some fluid.
A lumbar puncture can also be used to put chemotherapy drugs into the CSF to try to prevent or treat the spread of leukemia to the spinal cord and brain.
Lymph node biopsy
Removing a lymph node or part of a lymph node is often done to help diagnose lymphomas, but is only rarely needed with leukemia because the diagnosis is usually made looking at blood and bone marrow.
In this procedure, a surgeon cuts through the skin to remove all or part of a lymph node. If the node is near the skin surface, this is a simple operation that can often be done with local anesthesia, but if the node is inside the chest or abdomen, general anesthesia is used to keep you asleep during the biopsy.
When the entire lymph node is removed, it is called an excisional lymph node biopsy. If only part of the lymph node is removed, it is called an incisional lymph node biopsy.
Imaging tests
Imaging tests use x-rays, sound waves, magnetic fields, or radioactive particles to produce pictures of the inside of the body. Because leukemia does not usually form tumors, imaging tests aren’t as useful as they are for other types of cancer.
Imaging tests might be done in people with ALL, but they are done more often to look for infections or other problems, rather than for the leukemia itself. In some cases they may be done to help determine the extent of the disease, if it is thought it may have spread beyond the bone marrow and blood.
X-rays
Chest x-rays may be done if the doctor suspects a lung infection. They may also be done to look for enlarged lymph nodes in the chest.
Computed tomography (CT) scan
The CT scan is a type of x-ray test that produces detailed, cross-sectional images of your body. Unlike a regular x-ray, CT scans can show the detail in soft tissues (such as internal organs).
This test can help tell if any lymph nodes or organs in your body are enlarged. It isn’t usually needed to diagnose ALL, but it may be done if your doctor suspects leukemia cells are growing in an organ, like your spleen.
Sometimes a test that combines the CT scan with a PET (positron emission tomography) scan (PET/CT scan) is done. This is not often needed for patients with ALL.
Magnetic resonance imaging (MRI) scan
MRI scans are very helpful in looking at the brain and spinal cord.
MRI scans take longer than CT scans − often up to an hour. You may have to lie inside a narrow tube, which is confining and can be distressing to some people. Newer, more open MRI machines may be another option. The MRI machine makes loud buzzing and clicking noises that you may find disturbing. Some places provide headphones or earplugs to help block this noise out.
Ultrasound
Ultrasound can be used to look at lymph nodes near the surface of the body or to look for enlarged organs inside your abdomen such as the kidneys, liver, and spleen.
This is an easy test to have, and it uses no radiation. For most ultrasounds, you simply lie on a table, and a technician moves the transducer over the part of your body being looked at.
Gallium scan and bone scan
Gallium and bone scans are not often done for ALL, but they may be useful if you have bone pain that might be caused by either an infection or cancer in the bones.
Diagnosing ALL
After your doctor takes samples of your blood and bone marrow, a hematopathologist confirms a diagnosis and identifies the ALL subtype. A hematopathologist is a specialist who studies blood cell diseases by looking at samples of blood and marrow cells and other tissues.
The diagnosis of ALL is confirmed by identifying:
- Leukemic blast cells in the bone marrow samples
- The percentage of blast cells in the bone marrow
- About 1 to 5 percent of normal marrow cells are blast cells. In ALL, at least 20 percent of marrow cells are blast cells.
If you’re diagnosed with ALL, blood and bone marrow tests during or after treatment to see how your ALL cells are responding to therapy.
Acute Lymphocytic Leukemia Treatment
Adult acute lymphocytic leukemia (ALL) is not a single disease. It is really a group of related diseases, and patients with different subtypes of ALL may have different outlooks and responses to treatment.
After your cancer is diagnosed and staged, your cancer care team will discuss your treatment options with you. Choosing a treatment plan is an important decision, so it is important to take time and think about your choices. Treatment options for each patient are based on the leukemia subtype as well as certain prognostic features.
The number of patients with acute lymphoblastic leukemia who enter remission, stay in remission for years, or are cured, has increased significantly over the past 30 years. Most children with ALL are cured of their disease after treatment. Several areas of research have contributed to this progress.
The main types of treatment used for ALL are:
- Chemotherapy
- Targeted therapy
- Stem cell transplant
Other treatments such as surgery, radiation therapy, or monoclonal antibodies, may be used in special circumstances.
Treatment of ALL typically lasts for about 2 years. It is often intense, especially in the first few months of treatment, so it is important that you are treated in a center that has experience with this disease. See Typical Treatment of Acute Lymphocytic Leukemia for information about common treatment plans.
You may have different types of doctors on your treatment team. The doctor in charge or your team will most likely be a hematologist, a doctor who specializes in treating blood diseases, including leukemia. Many other specialists may be involved in your care as well, including nurse practitioners, nurses, nutrition specialists, social workers, and other health professionals.
It is important to discuss all of your treatment options, including their goals and possible side effects, with your doctors to help make the decision that best fits your needs. It’s also very important to ask questions if there is anything you’re not sure about.
Treatment for ALL usually needs to start very soon after it is diagnosed, but if time permits, it is often a good idea to seek a second opinion. A second opinion might give you more information and help you feel confident about your chosen treatment plan.
Taking part in a clinical trial
Clinical trials are carefully controlled research studies that are done to get a closer look at promising new treatments or procedures. Clinical trials are one way to get state-of-the art cancer treatment. In some cases they may be the only way to get access to newer treatments. They are also the best way for doctors to learn better methods to treat cancer. Still, they are not right for everyone.
If you would like to learn more about clinical trials that might be right for you, start by asking your doctor if your clinic or hospital conducts clinical trials.
Considering complementary and alternative methods
You may hear about alternative or complementary methods that your doctor hasn’t mentioned to treat your cancer or relieve symptoms. These methods can include vitamins, herbs, and special diets, or other methods such as acupuncture or massage, to name a few.
Complementary methods refer to treatments that are used along with your regular medical care. Alternative treatments are used instead of a doctor’s medical treatment. Although some of these methods might be helpful in relieving symptoms or helping you feel better, many have not been proven to work. Some might even be dangerous.
Be sure to talk to your cancer care team about any method you are thinking about using. They can help you learn what is known (or not known) about the method, which can help you make an informed decision.
Long-Term and Late Effects of Treatment
Some side effects of cancer treatment, such as fatigue, can linger for months or years after therapy. Some medical conditions like heart disease and other cancers may not appear until years after treatment ends.
Most childhood survivors of leukemia don’t develop significant long-term or late effects of treatment. However, for some patients the effects can range from mild to severe.
Talk to your child’s treatment team about possible long-term and late effects. Your child’s risk for developing long-term or late effects can be influenced by:
- Treatment type and duration
- Age at the time of treatment
- Gender
- Overall health
Some long-term and late effects become evident with maturation (puberty), growth and the normal aging process. Have your child evaluated with a physical exam yearly or more often as needed. Early intervention and healthy lifestyle practices (not smoking, good nutrition, exercise, regular screenings and follow-up) help.
Long-term and late effects can impact your child’s physical, mental and cognitive (brain function) health in several ways.
Physical Effects. Children treated for leukemia may be at increased risk for:
- Fatigue
- Growth delays
- Thyroid dysfunction
- Hearing loss
- A secondary cancer
Mental Effects. Most childhood survivors of cancer are psychologically healthy. However, some studies have indicated that a small number of childhood leukemia survivors were more likely than healthy peers to report changes in mood, feelings and behavior, including depression and posttraumatic stress disorder.
Cognitive Effects. Learning disabilities can begin during treatment or appear months or years afterward. Areas that can be affected include:
- Mathematics
- Spatial relationships
- Problem solving
- Attention span
- Reading
- Spelling
- Information processing
- Planning and organizing
- Concentration skills
- Fine motor coordination
Returning to School
Once your child is in remission, he or she will likely be going back to school. This reentry to the classroom can be daunting for a child of any age. Educate family members, friends, school personnel and healthcare providers about your child’s possible long-term and late effects of treatment. Talk with teachers about your child’s needs before he or she returns to school. Work with your child’s teachers and medical providers to develop a program tailored to his or her needs that features baseline testing, special accommodations and long-term planning.
Chronic Lymphocytic Leukemia
Chronic lymphocytic leukemia (CLL) is a type of cancer that starts from cells that become certain white blood cells (called lymphocytes) in the bone marrow. The cancer (leukemia) cells start in the bone marrow but then go into the blood.
In chronic lymphocytic leukemia, the leukemia cells often build up slowly over time, and many people don’t have any symptoms for at least a few years. In time, the cells can spread to other parts of the body, including the lymph nodes, liver, and spleen.
- CLL is the most common type of leukemia in adults.
- Some people have CLL that grows slowly while other people have CLL that grows faster.
- CLL patients have a number of effective treatment options available to them.
- For slow-growing CLL, watchful waiting may be an appropriate treatment approach.
- Many people with CLL live good-quality lives for years with medical care.
Doctors have found that there seem to be 2 different kinds of chronic lymphocytic leukemia (CLL):
- The slower-growing form has an increased number of lymphocytes but a normal or slightly below normal level of red cells, platelets and neutrophils (another type of white cell) in the blood. This form can remain stable for years. So it may take a long time before the patient needs treatment.
- The faster-growing form has too many CLL cells in the blood that block normal cell production. As a result, the number of fully functioning red cells and platelet levels drop lower than normal. The faster growing chronic lymphocytic leukemia is a more serious disease.
People with the faster-growing variety may have:
- Enlarged lymph nodes. The nodes can compress nearby organs, causing them to function improperly. For example, an enlarged node pressing on the stomach can interfere with gastrointestinal or urinary tract functions.
- A severe immunoglobulin deficiency. Immunoglobulins are proteins in the blood that fight infection. Low levels of immunoglobulins, sometimes combined with low neutrophil levels, can lead to recurrent infections.
- An enlarged spleen. The spleen can press on the stomach causing early fullness (satiety) while eating food and also discomfort in the left upper part of the abdomen.
If not treated, the faster-growing form of CLL can eventually lead to anemia, neutropenia or thrombocytopenia.
- The leukemia cells from these 2 types look alike, but lab tests can tell the difference between them. The tests look for proteins called ZAP-70 and CD38. If the chronic lymphocytic leukemia (CLL) cells contain low amounts of these proteins, the leukemia tends to grow more slowly.
The American Cancer Society’s estimates for leukemia in the United States for 2017 are 5:
- About 20,110 new cases of chronic lymphocytic leukemia (CLL)
- About 4,660 deaths from CLL
CLL accounts for about one-quarter of the new cases of leukemia. The average person’s lifetime risk of getting chronic lymphocytic leukemia (CLL) is about ½ of 1% (about 1 in 200). The risk is slightly higher in men than in women.
Chronic lymphocytic leukemia (CLL) mainly affects older adults. The average age at the time of diagnosis is around 71 years. It is rarely seen in people under age 40, and is extremely rare in children.
What Causes Chronic Lymphocytic Leukemia?
The exact cause of most cases of chronic lymphocytic leukemia (CLL) is not known. But scientists have learned a great deal about the differences between normal lymphocytes and CLL cells in recent years.
Normal human cells grow and function based mainly on the information contained in each cell’s chromosomes. Chromosomes are long molecules of DNA. DNA is the chemical that carries our genes — the instructions for how our cells function. We look like our parents because they are the source of our DNA. But our genes affect more than the way we look.
Each time a cell prepares to divide into 2 new cells, it must make a new copy of the DNA in its chromosomes. This process is not perfect, and errors can occur that may affect genes within the DNA.
Some genes contain instructions for controlling when our cells grow and divide. Certain genes that promote cell growth and division are called oncogenes. Others that slow down cell division or cause cells to die at the right time are called tumor suppressor genes. Cancers can be caused by DNA mutations (changes) that turn on oncogenes or turn off tumor suppressor genes.
Each human cell contains 23 pairs of chromosomes. In most cases of CLL, a change can be found in at least one of these chromosomes. Most often this change is a deletion − that is, loss of part of a chromosome. The loss of part of chromosome 13 is the most common deletion, but other chromosomes such as 11 and 17 can also be affected. Sometimes there is an extra chromosome 12 (trisomy 12). Other, less common abnormalities may also be found. Scientists know these chromosome changes are important in CLL, but it’s not yet clear which genes they involve or exactly how they lead to leukemia.
We do know that normal B lymphocytes are part of the immune system. They are programmed to grow and divide when they come into contact with a foreign substance called an antigen. (Scientists call substances foreign if they don’t normally occur in a person’s body and can be recognized by their immune system. Germs contain foreign antigens. So do blood cells from someone else with a different blood type.) Scientists think that CLL begins when B lymphocytes continue to divide without restraint after they have reacted to an antigen. But why this happens is not yet known.
Sometimes people inherit DNA mutations from a parent that greatly increase their risk of getting certain types of cancer. But inherited mutations rarely cause CLL. DNA changes related to CLL usually occur during the person’s lifetime, rather than having been inherited before birth.
Risk Factors for Chronic Lymphocytic Leukemia
A risk factor is something that affects a person’s chance of getting a disease like cancer. For example, exposing skin to strong sunlight is a risk factor for skin cancer. Smoking is a risk factor for a number of cancers.
But risk factors don’t tell us everything. Having a risk factor, or even several risk factors, doesn’t mean that you will get the disease. And many people who get the disease may not have had any known risk factors. Even if a person has a risk factor and develops cancer, it is often very hard to know how much that risk factor may have contributed to the cancer.
There are very few known risk factors for chronic lymphocytic leukemia (CLL). These include:
- Exposure to certain chemicals
- Family history
- Gender
- Race/ethnicity
The risk of getting CLL does not seem to be affected by smoking, diet, or infections.
Certain chemical exposures
Some studies have linked exposure to Agent Orange, an herbicide used during the Vietnam War, to an increased risk of CLL. Some other studies have suggested that farming and long-term exposure to some pesticides may be linked to an increased risk of CLL, but more research in this area is needed.
Family history
First-degree relatives (parents, siblings, or children) of CLL patients have more than twice the risk for this cancer.
Gender
CLL is slightly more common in males than females, but the reasons for this are not known.
Race/ethnicity
CLL is more common in North America and Europe than in Asia. Asian people who live in the United States do not have a higher risk than those living in Asia. This is why experts think the differences in risk are related to genetics rather than environmental factors.
Signs and Symptoms of chronic lymphocytic leukemia
Chronic lymphocytic leukemia (CLL) symptoms usually develop over time. Early in the course of the disease, CLL often has little effect on a person’s well-being. Some people with CLL do not have any symptoms. The disease may be suspected because of abnormal results from blood tests that were ordered either as part of an annual physical or a medical examination for an unrelated condition. An unexplained elevated white blood cell (lymphocyte) count is the most common finding that leads a doctor to consider a CLL diagnosis.
People with CLL who do have symptoms may:
- Tire more easily, and/or feel short of breath during day-to-day physical activities—as a result of anemia (low red blood cell count)
- Lose weight because of decreased appetite and/or increased use of energy
- Have lymph nodes and a spleen that may become enlarged as a result of an accumulation of CLL cells (leukemic lymphocytes)
- Have infections of the skin, lungs, kidneys or other sites that may occur as result of low immunoglobulin levels and decreased neutrophil counts.
Chronic Lymphocytic Leukemia Diagnosis
Certain signs and symptoms might suggest that a person has chronic lymphocytic leukemia (CLL), but tests are needed to confirm the diagnosis.
Medical history and physical exam
If you have any signs or symptoms that suggest you might have leukemia, your doctor will want to take a complete medical history to check for symptoms and possible risk factors. You will also be asked about your general health.
A physical exam provides information about your general health, possible signs of leukemia, and other health problems. During the physical exam, your doctor will pay close attention to your lymph nodes and other areas that might be affected.
Tests used to diagnose and classify leukemia
If symptoms and/or the results of the physical exam suggest you could have leukemia, the doctor will need to check samples of blood and bone marrow to be certain of this diagnosis. Other tissue and cell samples may also be taken to help guide treatment.
Blood tests
Blood samples for tests for CLL generally are taken from a vein in the arm.
Complete blood count and blood cell exam (peripheral blood smear)
The complete blood count (CBC) is a test that measures the different cells in the blood, such as the red blood cells, the white blood cells, and the platelets. This test is often done along with a differential (or diff) which looks at the numbers of the different types of white blood cells. These tests are often the first ones done on patients with a suspected blood problem.
People with CLL have too many lymphocytes (called lymphocytosis). Having more than 10,000 lymphocytes/mm³ (per cubic millimeter) of blood strongly suggests that CLL is present, but other tests are needed to know for certain. You might also have too few red blood cells and blood platelets as well.
For the peripheral blood smear, a sample of blood is looked at under the microscope. If you have CLL, the blood smear could show many abnormal looking lymphocytes called smudge cells.
Flow cytometry
This test is important in diagnosing CLL. It uses a machine that looks for certain substances on or in cells that help identify what types of cells they are (markers).
This test can be used to see if the lymphocytes in a sample of blood contain CLL cells. It can also be used to look for CLL cells in bone marrow or other fluids. CLL cells can have many of the same markers as normal B-cells. However, they also have a marker called CD5 that is normally found on T-cells, but not on normal B cells. For someone to have CLL, there must be at least 5,000 of these cells (per mm3) in the blood.
Flow cytometry can also be used to test for substances called ZAP-70 and CD38 on the cells. Studies suggest that CLL with fewer cells that have these substances seem to have a better outlook.
Other blood tests
Other tests may be done to measure the amount of certain chemicals in the blood, but they are not used to diagnose leukemia. In patients already known to have CLL, these tests help detect liver or kidney problems caused by the spread of leukemia cells or due to the side effects of certain chemotherapy (chemo) drugs. These tests also help determine if treatment is needed to correct low or high blood levels of certain minerals. If treatment with the drug rituximab (Rituxan®) is planned, the doctor may order blood tests to check for previous hepatitis infection.
Blood immunoglobulin (antibody) levels may be tested to check if you enough antibodies to fight infections, especially if you have recently had many infections. Another blood protein called beta-2-microglobulin may be measured. High levels of this protein generally indicate a more advanced CLL.
Bone marrow tests
Blood tests are often enough to diagnose CLL, but testing the bone marrow is helpful to tell how advanced it is. Bone marrow tests are often done before starting treatment for that reason. They might also be repeated during or after treatment to see if the treatment is effective.
Bone marrow aspiration and biopsy
Bone marrow aspiration and biopsy are done to get bone marrow samples for testing. They are usually done together, as part of the same procedure. The samples are usually taken from the back of the pelvic (hip) bone, but sometimes they may be taken from other bones.
For a bone marrow aspiration, you lie on a table (either on your side or on your belly). After cleaning the skin over the hip, the doctor numbs the area and the surface of the bone with local anesthetic, which may cause a brief stinging or burning sensation. A thin, hollow needle is then inserted into the bone and a syringe is used to suck out a small amount (about 1 teaspoon) of liquid bone marrow. Even with the anesthetic, most people still have some brief pain when the marrow is removed.
A bone marrow biopsy is usually done just after the aspiration. A small piece of bone and marrow (about 1/16 inch in diameter and 1/2 inch long) is removed with a slightly larger needle that is twisted as it is pushed down into the bone. With the local anesthetic, this most often causes a feeling of pressure or tugging, but is not often painful. Once the biopsy is done, pressure will be applied to the site to help prevent bleeding.
Routine microscopic exams
The bone marrow samples are looked at under a microscope by a pathologist (a doctor specializing in lab tests) and may be reviewed by the patient’s hematologist/oncologist (a doctor specializing in blood diseases and cancer).
The doctors will look at the size, shape, and other traits of the white blood cells in the samples to classify them into specific types.
An important factor is if the cells look mature (like normal blood cells that can fight infections). Chronic lymphocytic leukemia cells usually appear mature, while cells of acute leukemias look immature.
A key feature of a bone marrow sample is its cellularity. Normal bone marrow has a certain number of blood-forming cells and fat cells. Marrow with too many blood-forming cells is said to be hypercellular. This is often seen in bone marrow of CLL patients. Doctors also look to see how much of the normal cells in the bone marrow have been replaced by CLL cells.
The pattern of spread of CLL cells in the bone marrow is also important. A pattern where the cells are in small groups (nodular or interstitial pattern) often indicates a better outlook than if the cells are scattered throughout the marrow (a diffuse pattern).
Stains and/or antibody tests such as cytochemistry, immunocytochemistry, immunohistochemistry, and flow cytometry may be used on the bone marrow samples to diagnose CLL.
Gene tests
Cytogenetics: For this test, bone marrow cells (or sometimes cells from the blood or other tissues) are grown in the lab, and the chromosomes are examined under a microscope. Because it takes time for the cells to start dividing, this test usually takes weeks to complete. Normal human cells contain 23 pairs of chromosomes, but some cases of CLL have chromosome changes that can be seen under the microscope.
In some cases of CLL, part of a chromosome may be missing. This is called a deletion. The most common deletions occur in parts of chromosomes 13, 11, or 17. Deletion of part of chromosome 17 (often written as del[17p]) is linked to a poor outlook. Other, less common chromosome changes include an extra copy of chromosome 12 (trisomy 12) or a translocation (swapping of DNA) between chromosomes 11 and 14 (written as t[11;14]).
This information may be helpful to determine a patient’s prognosis (outlook), but it needs to be looked at along with other factors, such as the stage of CLL. The loss of part of chromosome 13 is usually linked with a slower-growing disease and a better outlook, while defects in chromosomes 11 or 17 often indicate a poorer outlook. Trisomy 12 does not seem to have much of an effect on prognosis.
Fluorescent in situ hybridization (FISH): This is a type of chromosome test that can be used to look at the cells’ chromosomes and DNA without having to grow the cells in the lab. It uses special fluorescent dyes that only attach to specific parts of particular chromosomes. FISH is used to look for certain genes or chromosome changes (not just any change). It can be used on regular blood or bone marrow samples. Because the cells don’t have to grow in the lab first, it can usually provide results more quickly than cytogenetics, often within a couple of days.
Molecular tests: Immunoglobulins, the antibodies that help your body fight infections, are made up of light chains and heavy chains. Whether the gene for the immunoglobulin heavy chain variable region (IGHV or IgVH) has changed (mutated) can help your doctor know how aggressive your CLL is. That gene is looked at in a test called cDNA sequencing.
Lymph node biopsy
In a lymph node biopsy, all or part of a lymph node is removed so that it can be examined under the microscope to see if it contains cancer cells. Although this is often done to diagnose lymphomas, it is only rarely needed in CLL. It may be used if a lymph node has grown very large and the doctor wants to know if the leukemia has changed (transformed) into a more aggressive lymphoma.
In an excisional lymph node biopsy, an entire lymph node is removed through a cut in the skin. If the node is near the skin surface, this is a simple operation that can be done with local anesthesia, but if the node is inside the chest or abdomen, general anesthesia (where the patient is asleep) is used. If the lymph node is very large, only part of it may be removed. This is called an incisional biopsy.
Lumbar puncture (or spinal tap)
This procedure is used to take samples of the fluid that surrounds the brain and spinal cord (the cerebrospinal fluid or CSF) for testing. This is not a routine test for people with CLL. It is only done if the doctor suspects leukemia cells may have spread to the area around the brain or spinal cord (which is rare), or if there might be an infection in those areas.
For this test, the doctor first numbs an area in the lower part of the back over the spine. A small, hollow needle is then placed between the bones of the spine and into the space around the spinal cord to collect some of the fluid.
Imaging tests
Imaging tests use x-rays, sound waves, or magnetic fields to create pictures of the inside of the body. Imaging tests are not done to diagnose the leukemia, but they may be done for other reasons, including to help find a suspicious area that might be cancerous, to learn how far a cancer may have spread, or to help determine if treatment has been effective.
Computed tomography (CT) scan
The CT scan can help tell if any lymph nodes or organs in your body are enlarged. It isn’t usually needed to diagnose CLL, but it may be done if your doctor suspects the leukemia is growing in an organ, like your spleen.
Sometimes a CT scan is combined with a PET scan in a test known as a PET/CT scan. For a PET scan, glucose (a form of sugar) containing a radioactive atom is injected into the blood. Because cancer cells grow rapidly, they absorb large amounts of the radioactive sugar. A special camera can then create a picture of the areas of radioactivity in the body. The PET/CT scan combines both tests in one machine. This test allows the doctor to compare areas of higher radioactivity on the PET scan with the more detailed appearance of that area on the CT.
Magnetic resonance imaging (MRI) scan
MRI scans are most useful in looking the brain and spinal cord, but they are not often needed in people with CLL.
MRI scans take longer than CT scans − often up to an hour. You might have to lie inside a narrow tube, which is confining and can be distressing to some people. Newer, more open MRI machines may be another option. The MRI machine makes loud buzzing and clicking noises that you may find disturbing. Some places provide headphones or earplugs to help block this noise out.
Ultrasound
Ultrasound can be used to look at lymph nodes near the surface of the body or to look for enlarged organs (like the liver and spleen) inside your abdomen.
This is an easy test to have, and it uses no radiation. For most ultrasound exams, you simply lie on a table, and a technician moves the transducer over the part of your body being looked at.
Chronic Lymphocytic Leukemia Staging
For most cancers, staging is the process of finding out how far the cancer has spread. Stages are often useful because they can help guide treatment and determine a person’s prognosis (outlook). Most types of cancer are staged based on the size of the tumor and how far the cancer has spread.
Chronic lymphocytic leukemia (CLL), on the other hand, does not usually form tumor masses. It generally is present in the bone marrow and blood, and, in many cases, it has spread to other organs such as the spleen, liver, and lymph nodes by the time it is found. Therefore, the outlook for a person with CLL depends on other information, such as the lab test results and the results of imaging tests.
Staging for chronic lymphocytic leukemia
A staging system is a standardized way for the cancer care team to summarize information about how far a cancer has spread. There are 2 different systems for staging CLL:
- Rai system: This is used more often in the United States.
- Binet system: This is used more widely in Europe.
Rai staging system
The Rai system was originally devised in 1968. At that time, all that was needed to diagnose CLL was lymphocytosis – a high number of lymphocytes in the blood and bone marrow that didn’t have any other cause (like infection). This was originally defined as over 15,000 lymphocytes/mm3 of blood and at least 40% of the bone marrow being made up of lymphocytes.
Now, for a diagnosis of CLL, the patient must have at least 5,000/mm3 of monoclonal lymphocytes (sometimes called a monoclonal lymphocytosis), but the overall lymphocyte count does not have to be high. Monoclonal means that the cells all came from the same cell, which can lead to them having the same chemical pattern on special testing.
For the purposes of this staging, you can substitute a diagnosis of CLL (such as with a monoclonal lymphocytosis) for lymphocytosis.
This system divides CLL into 5 stages:
- Rai stage 0: Lymphocytosis and no enlargement of the lymph nodes, spleen, or liver, and with near normal red blood cell and platelet counts.
- Rai stage I: Lymphocytosis plus enlarged lymph nodes. The spleen and liver are not enlarged and the red blood cell and platelet counts are near normal.
- Rai stage II: Lymphocytosis plus an enlarged spleen (and possibly an enlarged liver), with or without enlarged lymph nodes. The red blood cell and platelet counts are near normal.
- Rai stage III: Lymphocytosis plus anemia (too few red blood cells), with or without enlarged lymph nodes, spleen, or liver. Platelet counts are near normal.
- Rai stage IV: Lymphocytosis plus thrombocytopenia (too few blood platelets), with or without anemia, enlarged lymph nodes, spleen, or liver.
Doctors separate the Rai stages into low-, intermediate-, and high-risk groups when determining treatment options.
- Stage 0 is considered low risk.
- Stages I and II are considered intermediate risk.
- Stages III and IV are considered high risk.
These risk groups are used later chronic lymphocytic leukemia treatment.
Binet staging system
In the Binet staging system, CLL is classified by the number of affected lymphoid tissue groups (neck lymph nodes, groin lymph nodes, underarm lymph nodes, spleen, and liver) and by whether or not the patient has anemia (too few red blood cells) or thrombocytopenia (too few blood platelets).
- Binet stage A: Fewer than 3 areas of lymphoid tissue are enlarged, with no anemia or thrombocytopenia.
- Binet stage B: 3 or more areas of lymphoid tissue are enlarged, with no anemia or thrombocytopenia.
- Binet stage C: Anemia and/or thrombocytopenia are present.
Both of these staging systems are helpful and have been in use for many years.
Other factors can also help predict a person’s outlook. The factors described below are not part of formal staging systems (at least at this time), but they can also provide helpful information.
Prognostic factors for chronic lymphocytic leukemia
Along with the stage, there are other factors that help predict a person’s outlook. These factors are sometimes taken into account when looking at possible treatment options. Factors that tend to be linked with shorter survival time are called adverse prognostic factors. Those that predict longer survival are favorable prognostic factors.
Adverse prognostic factors
- Diffuse pattern of bone marrow involvement (more widespread replacement of normal marrow by leukemia)
- Advanced age
- Male gender
- Deletions of parts of chromosomes 17 or 11
- High blood levels of certain substances, such as beta-2-microglobulin
- Lymphocyte doubling time (the time it takes for the lymphocyte count to double) of less than 6 months
- Increased fraction of prolymphocytes (an early form of the lymphocyte) in the blood
- High proportion of CLL cells containing ZAP-70 (more than 20%) or CD38 (more than 30%)
- CLL cells with unchanged (not mutated) gene for the immunoglobulin heavy chain variable region (IGHV)
Favorable prognostic factors
- Non-diffuse (nodular or interstitial) pattern of bone marrow involvement
- Deletion of part of chromosome 13 (with no other chromosome abnormalities)
- Low proportion of CLL cells containing ZAP-70 (20% or less) or CD38 (30% or less)
- CLL cells with a mutated gene for IGHV
Certain prognostic factors such as the presence or absence of ZAP-70, CD38, and a mutated gene for IGHV help divide cases of CLL into 2 groups, slow growing and fast growing. People with the slower growing kind of CLL tend to live longer and may be able to delay treatment longer as well.
Staging for hairy cell leukemia
There is no generally accepted staging system for hairy cell leukemia.
Monoclonal B-lymphocytosis
Some people have monoclonal lymphocytes in their blood, but not enough to make the diagnosis of CLL. If someone has less than 5,000 monoclonal lymphocytes (per mm3), normal counts of red blood cells and platelets, and no enlarged lymph nodes (or enlarged spleen), they have a condition called monoclonal B-lymphocytosis (MBL). MBL doesn’t need to be treated, but about one patient of every 100 with this condition will go on to need treatment for CLL.
Small lymphocytic lymphoma
The cancer cells of small lymphocytic lymphoma (SLL) and CLL look the same under the microscope and have the same marker proteins on the surface of the cells. Whether someone is diagnosed with SLL or CLL depends largely on the number of lymphocytes in the blood. To be diagnosed with CLL, there must at least 5,000 monoclonal lymphocytes (per mm3) in the blood. For it to be called SLL, the patient must have enlarged lymph nodes or an enlarged spleen with fewer than 5,000 lymphocytes (per mm3) in the blood. Still, since SLL and CLL can be treated the same, the difference between them is not really important.
Chronic Lymphocytic Leukemia Treatment
After the leukemia is found and staged, your cancer care team will discuss your treatment options with you. Because chronic lymphocytic leukemia often grows slowly, not everyone needs to be treated right away. When treatment is needed, the main treatments used are:
- Chemotherapy
- Monoclonal antibodies
- Targeted therapy
- Supportive care
- Splenectomy
- Stem cell transplant
Less often, leukapheresis, surgery, or radiation therapy may also be used.
It is important to take time and think about your possible choices. In choosing a treatment plan, the stage of the leukemia and other prognostic factors are important. Other factors to consider include whether or not you are having symptoms, your age and overall health, and the likely benefits and side effects of treatment.
In considering your treatment options it is often a good idea to seek a second opinion, if possible. This could give you more information and help you feel more confident about the treatment plan you have chosen.
Complications of Chronic lym:phocytic leukemia
Chronic lymphocytic leukemia (CLL) may cause complications such as those outlined below.
Infection
Infection is a common complication for people with CLL. Because of the high risk of infections, immediate vaccination for pneumococcal pneumonia with Prevnar 13® and a yearly flu vaccine is recommended. CLL patients do not respond well to vaccines due to their immune system depression. CLL patients should never receive live vaccines (such as the shingles vaccine).
A higher risk of infection is caused by:
- The inability of the person’s CLL cells to make antibodies needed to fight infections
- The effect of chemotherapy, which causes reduced cell counts for certain infection-fighting white cells in the blood, specifically neutrophils and monocytes.
Antibiotic therapy is usually required to treat bacterial or fungal infections during the course of the disease. People who get recurrent infections may also receive injections of immunoglobulin (gamma globulin) on a regular basis to correct the immune deficiency. While immunoglobulin is expensive, it does have benefit in decreasing the frequency of infections in CLL patients with low levels of this in the blood.
CLL-related low blood counts are often efficiently corrected by CLL therapy. However, the use of white cell growth factors may benefit patients who experience prolonged low white cell counts after treatment. Examples of white cell growth factors are
- Granulocyte-colony stimulating factor (G-CSF) (filgrastim [Neupogen®] or pegfilgrastim [Neulasta®]) that can increase the number of neutrophils
- Granulocyte-macrophage colony-stimulating growth factor (GM-CSF) (sargramostim [Leukine®]) that can increase the number of neutrophils and monocytes.
Anemia
Anemia (low numbers of red cells) is a common side effect of chemotherapy. Some people with CLL may need blood transfusions.
Richter Transformation
In about 3 to 5 percent of people with CLL, the disease transforms into an aggressive lymphoma because of a change in the characteristics of the CLL cells. This is much more common in IgHv-unmutated CLL. This pattern is referred to as a “Richter transformation” or “large cell transformation.” People with this type of CLL may have significantly enlarged lymph nodes, and may have fevers and weight loss. Tumors of lymphocytes may also develop in parts of the body other than the lymph nodes. Richter transformation is treated with aggressive chemotherapy and reduced-intensity allogeneic transplantation if feasible. Outcome for patients with Richter transformation is generally poor unless it is diagnosed prior to receiving a lot of treatment for CLL.
Prolymphocytes
About 15 percent of people with CLL have leukemia cells that are a mix of lymphocytes and another type of white cell, called a “prolymphocyte.” Most people with this type of CLL follow a similar course to that of other people with CLL. However, for a relatively small subset of patients with this type of CLL, the blood cells may become mainly composed of prolymphocytes; the spleen may enlarge further, and the disease may become less responsive to treatment. In these cases, individuals are encouraged to talk to their doctors about the potential benefits of treatment in a clinical trial.
Autoimmune Hemolytic Anemia
Some people with CLL produce a type of antibody that works against their own cells. These “autoantibodies” are usually directed against the patient’s red cells and causes them to be removed rapidly from the blood. This condition, called “autoimmune hemolytic anemia,” can worsen the effects of already low red cell counts. The “antiglobulin test” or “Coombs’ test” is used to identify the autoantibodies. Less often, the antibody works against the platelets. This condition, called “immune thrombocytopenia” results in significantly decreased platelet counts. The drugs prednisone, Rituxan and cyclosporine are sometimes used to treat autoimmune hemolytic anemia and immune thrombocytopenia.
Second Cancers
People with CLL have a higher risk than the general population of developing a second cancer. The cancers that tend to occur most often include:
- Soft tissue sarcoma
- Melanoma
- Colorectal cancer
- Lung cancer
- Squamous cell skin cancer
- Basal cell carcinoma, with a high recurrence rate
CLL patients are also at higher risk for other blood cancers and disorders such as:
- Acute myeloid leukemia
- Myelodysplastic syndromes
This complication is more common after treatment with fludarabine and cyclophosphamide (FC or FCR). Further evaluation is needed to determine whether treatment with fludarabine may increase the risk of second solid tumor cancers. It is important to follow up with your oncologist on a regular basis.
Acute myeloid leukemia
Acute myeloid leukemia (a cancer of the bone marrow and the blood) is also called acute myelocytic leukemia, acute myelogenous leukemia, acute granulocytic leukemia, acute non-lymphocytic leukemia, or sometimes just AML 6. “Acute” means that this leukemia can progress quickly if not treated, and would probably be fatal in a few months. “Myeloid” refers to the type of cell this leukemia starts from.
Acute myeloid leukemia starts in the bone marrow (the soft inner part of certain bones, where new blood cells are made), but in most cases it quickly moves into the blood and progresses rapidly without treatment and can be difficult to treat. It can sometimes spread to other parts of the body including the lymph nodes, liver, spleen, central nervous system (brain and spinal cord), and testicles.
Acute myeloid leukemia is characterized by a clonal (starting from a single malignant cell) proliferation of myeloid precursors with a reduced capacity to differentiate into the different blood cells. As a result, there is an accumulation of immature leukemic cells, called “blasts.” These are found in the bone marrow, peripheral blood, and occasionally in other tissues, with a variable reduction in the numbers of normal red blood cells, platelets, and mature granulocytes. Acute myeloid leukemia that transforms from a myelodysplastic syndrome (MDS) is still considered to be acute myeloid leukemia. In disease that transforms from other myeloproliferative or myelodysplastic/myeloproliferative neoplasms, it helps to know if the acute process arose from an underlying chronic disease. Leukemia that arises from an underlying chronic disease or from previous therapy is less likely to respond to current anticancer treatments and, therefore, it is less likely to be cured 7.
Acute myeloid leukemia (AML) is the most common acute leukemia in the United States and worldwide 7. There are 40,255 people living with Acute Myeloid Leukemia (AML) in the United States, with an estimated 19,950 new cases that will be diagnosed in 2016. According to the Surveillance, Epidemiology and End Results Program 8, the number of new cases of acute myeloid leukemia was 4.1 per 100,000 men and women per year. The number of deaths was 2.8 per 100,000 men and women per year. These rates are age-adjusted and based on 2009-2013 cases and deaths.
Acute myeloid leukemia is most common in older people, but despite advances in the treatment of other blood cancers, the standard of care for most acute myeloid leukemia patients has changed very little in 40 years.
How Many People Survive 5 Years Or More after Being Diagnosed with Acute Myeloid Leukemia?
Relative survival statistics compare the survival of patients diagnosed with cancer with the survival of people in the general population who are the same age, race, and sex and who have not been diagnosed with cancer. Because survival statistics are based on large groups of people, they cannot be used to predict exactly what will happen to an individual patient. No two patients are entirely alike, and treatment and responses to treatment can vary greatly. However, based on data from SEER 18 2007-2013 2, 26.9 percent of those with acute myeloid leukemia (AML) survived 5 years or more.
What Causes Acute Myeloid Leukemia To Develop?
The DNA (genetic material) of a developing stem cell in the bone marrow is damaged 9. This is called an “acquired mutation.”
Stem cells form blood cells (red cells, white cells and platelets).
This damaged cell becomes a leukemic cell and multiplies into billions of cells called leukemic blasts.
- Leukemic blasts
- Do not function normally
- Block the production of normal cells
- Grow and survive better than normal cells
As a result, the number of healthy blood cells (red cells, white cells and platelets) is usually lower than normal.
- Anemia is a condition when there is a low number of red cells in the blood which can cause fatigue and shortness of breath.
- Neutropenia is a condition when there is a low number of white cells so that the immune system can’t effectively guard against infection due to a lack of neutrophils (a type of white cell).
- Thrombocytopenia is a condition when there is a low number of platelets which can cause bleeding and easy bruising with no apparent cause.
- Low numbers of all three blood cell counts is called pancytopenia.
Risk Factors for acute myeloid leukemia
For most people who have acute myeloid leukemia, there are no obvious reasons (risk factors) why they developed the disease. You cannot catch acute myeloid leukemia from someone else.
Researchers have identified potential risk factors, including:
- Repeated exposure to the chemical benzene, which damages the DNA of normal marrow cells. According to the Agency for Toxic Substances and Disease Registry, half of the total national personal exposure to benzene comes from cigarette smoke even though petroleum products contribute to the majority of benzene in the atmosphere. Benzene is also found in certain industrial settings; however, the strict regulation of its use has decreased benzene exposure in the workplace.
- Certain genetic disorders such as Down syndrome, Fanconi’s anemia, Shwachman syndrome and Diamond-Blackfan syndrome
- Past chemotherapy or radiation treatments for other cancers.
- Progression of other blood cancers or disorders, polycythemia vera, primary myelofibrosis, essential thrombocythemia and myelodysplastic syndromes (MDS).
How Is Acute Myeloid Leukemia Classified?
Most cases of acute myeloid leukemia develop from cells that would turn into white blood cells (other than lymphocytes), but some cases of acute myeloid leukemia develop in other types of blood-forming cells.
For most types of cancer, determining the stage (extent) of the cancer is very important. The stage is based on the size of the tumor and how far the cancer has spread. This can be helpful in predicting a person’s outlook and deciding on treatment.
Acute myeloid leukemia (AML), on the other hand, does not usually form tumors. It generally is in all of the bone marrow in the body and in some cases, has spread to other organs, such as the liver and spleen. Therefore acute myeloid leukemia is not staged like most other cancers. The outlook for a person with acute myeloid leukemia depends instead on other information, such as the subtype of acute myeloid leukemia (determined by lab tests), the patient’s age, and other lab test results.
Knowing the subtype of acute myeloid leukemia can be very important, as it sometimes affects both a patient’s outlook and the best treatment. For example, the acute promyelocytic leukemia (APL) subtype is often treated using drugs that are different from those used for other subtypes of acute myeloid leukemia.
Two of the main systems that have been used to classify acute myeloid leukemia into subtypes are the French-American-British (FAB) classification and the newer World Health Organization (WHO) classification.
The French-American-British (FAB) classification of Acute Myeloid Leukemia (AML)
In the 1970s, a group of French, American, and British leukemia experts divided acute myeloid leukemia into subtypes, M0 through M7, based on the type of cell from which the leukemia develops and how mature the cells are. This was based largely on how the leukemia cells looked under the microscope after routine staining.
| FAB subtype | Name |
| M0 | Undifferentiated acute myeloblastic leukemia |
| M1 | Acute myeloblastic leukemia with minimal maturation |
| M2 | Acute myeloblastic leukemia with maturation |
| M3 | Acute promyelocytic leukemia (APL) |
| M4 | Acute myelomonocytic leukemia |
| M4 eos | Acute myelomonocytic leukemia with eosinophilia |
| M5 | Acute monocytic leukemia |
| M6 | Acute erythroid leukemia |
| M7 | Acute megakaryoblastic leukemia |
Note: Subtypes M0 through M5 all start in immature forms of white blood cells. M6 AML starts in very immature forms of red blood cells, while M7 AML starts in immature forms of cells that make platelets.
[Source 10]World Health Organization (WHO) classification of Acute Myeloid Leukemia (AML)
The French-American-British (FAB) classification system is useful and is still commonly used to group acute myeloid leukemia into subtypes. But it doesn’t take into account many of the factors that are now known to affect prognosis (outlook). The World Health Organization (WHO) has developed a newer system that includes some of these factors to try to better classify acute myeloid leukemia.
The WHO system divides acute myeloid leukemia into several groups:
- Acute myeloid leukemia with certain genetic abnormalities
- AML with a translocation between chromosomes 8 and 21
- AML with a translocation or inversion in chromosome 16
- AML with a translocation between chromosomes 9 and 11
- Acute promyelocytic leukemia [APL] (M3) with a translocation between chromosomes 15 and 17
- AML with a translocation between chromosomes 6 and 9
- AML with a translocation or inversion in chromosome 3
- AML (megakaryoblastic) with a translocation between chromosomes 1 and 22
- Acute myeloid leukemia with myelodysplasia-related changes
- Acute myeloid leukemia related to previous chemotherapy or radiation
- Acute myeloid leukemia not otherwise specified (This includes cases of AML that don’t fall into one of the above groups, and is similar to the FAB classification.)
- AML with minimal differentiation (M0)
- AML without maturation (M1)
- AML with maturation (M2)
- Acute myelomonocytic leukemia (M4)
- Acute monocytic leukemia (M5)
- Acute erythroid leukemia (M6)
- Acute megakaryoblastic leukemia (M7)
- Acute basophilic leukemia
- Acute panmyelosis with fibrosis
- Myeloid sarcoma (also known as granulocytic sarcoma or chloroma)
- Myeloid proliferations related to Down syndrome
- Undifferentiated and biphenotypic acute leukemias (leukemias that have both lymphocytic and myeloid features). Sometimes called acute lymphocytic leukemia (ALL) with myeloid markers, acute myeloid leukemia with lymphoid markers, or mixed phenotype acute leukemias.
Prognostic factors for acute myeloid leukemia
In recent years, research has focused on why some patients have a better chance to be cured than others. Differences among patients (or their leukemias) that affect response to treatment are called prognostic factors. Prognostic factors help doctors decide if people with a certain type of acute myeloid leukemia should get more or less treatment. Some of these include:
Chromosome abnormalities
Acute myeloid leukemia cells can have many kinds of chromosome changes, some of which can affect a person’s prognosis. Those listed below are some of the most common, but there are many others. Not all patients have these abnormalities. Patients without any of these usually have an outlook that is between favorable and unfavorable.
Favorable abnormalities:
- Translocation between chromosomes 8 and 21 (seen most often in patients with M2)
- Inversion of chromosome 16 (seen most often in patients with M4 eos) or a translocation between chromosome 16 and itself
- Translocation between chromosomes 15 and 17 (seen most often in patients with M3)
Unfavorable abnormalities:
- Deletion (loss) of part of chromosome 5 or 7 (no specific AML type)
- Translocation or inversion of chromosome 3
- Translocation between chromosomes 6 and 9
- Translocation between chromosomes 9 and 22
- Abnormalities of chromosome 11 (at the spot q23)
- Complex changes – those involving several chromosomes (no specific AML type)
Gene mutations
People whose leukemia cells have certain gene mutations may have a better or worse outlook.
For instance, about 1 patient out of 3 with acute myeloid leukemia has a mutation in the FLT3 gene. These people tend to have a poorer outcome, but new drugs that target this abnormal gene are now being studied, which may lead to better outcomes.
On the other hand, people with changes in the NPM1 gene (and no other abnormalities) seem to have a better prognosis than people without this change. Changes in the CEBPA gene are also linked to a better outcome.
In the coming years, doctors will use newer lab tests to learn more about the underlying genetic defects that cause acute myeloid leukemia and how they can be used to predict a patient’s prognosis. These genetic defects might also form the basis for treating these leukemias.
Markers on the leukemia cells
If the leukemia cells have the CD34 protein and/or the P-glycoprotein (MDR1 gene product) on their surface, it is linked to a worse outcome.
Age
Older patients (over 60) generally don’t do as well as younger patients. Some of this may be because they are more likely to have unfavorable chromosome abnormalities. Older patients sometimes also have other medical conditions that can make it harder to treat them with more intense chemotherapy regimens.
White blood cell count
A high white blood cell count (>100,000) at the time of diagnosis is linked to a worse outlook.
Prior blood disorder leading to acute myeloid leukemia
Having a prior blood disorder such as a myelodysplastic syndrome is linked to a worse outcome.
Acute myeloid leukemia that develops after treatment for another cancer tends is linked to a worse outcome.
Infection
Having an active systemic (blood) infection at the time of diagnosis makes a poor outcome more likely.
Leukemia cells in the central nervous system
Leukemia that has spread to the area around the brain and spinal cord can be hard to treat, since most chemotherapy drugs can’t reach that area.
Status of acute myeloid leukemia after treatment
Not surprisingly, how well (and how quickly) the leukemia responds to treatment also affects long-term prognosis. Better responses have been linked with better long-term outcomes.
A remission (complete remission) is usually defined as having no evidence of disease after treatment. This means the bone marrow contains fewer than 5% blast cells, the blood cell counts are within normal limits, and there are no signs or symptoms of the disease. A molecular complete remission means there is no evidence of leukemia cells in the bone marrow, even when using very sensitive tests, such as PCR (polymerase chain reaction).
Minimal residual disease is a term used after treatment when leukemia cells can’t be found in the bone marrow using standard tests (such as looking at cells under a microscope), but more sensitive tests (such as flow cytometry or PCR) find evidence that there are still leukemia cells in the bone marrow.
Active disease means that either there is evidence that the leukemia is still present during treatment or that the disease has come back after treatment (relapsed). For a patient to have relapsed, they must have more than 5% blast cells in their bone marrow.
Signs and Symptoms of acute myeloid leukemia
General signs and symptoms of the early stages of acute myeloid leukemia may mimic those of the flu or other common diseases. Furthermore, signs and symptoms may vary.
It is common for people with acute myeloid leukemia to feel a loss of well-being – lethargy and fatigue, because of the underproduction of normal bone marrow cells. The person may tire more easily and have shortness of breath during normal physical activities.
People with acute myeloid leukemia may also have 11:
- A pale complexion from anemia
- Signs of bleeding caused by a very low platelet count, including:
- Black-and-blue marks or bruises occurring for no reason or because of a minor injury
- The appearance of pinhead-sized red spots on the skin, called “petechiae”
- Prolonged bleeding from minor cuts
- Mild fever
- Swollen gums
- Frequent minor infections
- Loss of appetite and weight loss
- Discomfort in bones or joints
- Enlarged spleen
- Enlarged liver.
- Shortness of breath
- Easy bruising
- Unusual bleeding, such as frequent nosebleeds and bleeding from the gums.
In addition to the signs and symptoms above, you may experience these conditions, which can be dangerous without treatment:
- Bleeding in the brain or a lung
- Infection, especially if your body produces too few white cells known as neutrophils
- Myeloid sarcoma, when a mass of acute myeloid leukemia cells can form a tumor elsewhere in the body
How is acute myeloid leukemia diagnosed
An accurate diagnosis of the type of leukemia is important. The exact diagnosis helps the doctor to:
- Estimate how the disease will progress
- Determine the appropriate treatment.
Diagnosing acute myeloid leukemia and your AML subtype usually involves a series of tests. Some of these tests may be repeated during and after therapy to measure the effects of treatment.
Tests Your Doctor May Use to Diagnose AML
Blood and Bone Marrow Tests
Blood and bone marrow tests are used to diagnose acute myeloid leukemia and the AML subtype. A change in the number and appearance of blood cells helps to make the diagnosis. Acute myeloid leukemia cells look similar to normal immature white cells. However, their development is incomplete.
Blood Tests
Blood samples are generally taken from a vein in the patient’s arm. Your blood is sent to a lab for:
- A complete blood count (CBC), which counts the number of red cells, white cells and platelets in your blood. Usually, patients with AML have lower-than-expected numbers of red blood cells and platelets.
- A peripheral blood smear, which shows the presence of leukemic blast cells (myeloblasts). A person with AML usually has too many leukemic blast cells in the blood. These cells do not function like normal cells.
Bone Marrow Tests
Samples of marrow cells are obtained by bone marrow aspiration and biopsy. Bone marrow testing involves two steps usually performed at the same time in a doctor’s office or a hospital:
- A bone marrow aspiration to remove a liquid marrow sample
- A bone marrow biopsy to remove a small amount of bone filled with marrow
Both samples are examined under a microscope to look for chromosomal and other cell changes.
Other Tests
“Karyotyping” and “cytogenetic analysis” are processes used to identify certain changes in chromosomes and genes. A laboratory test called “polymerase chain reaction (PCR)” may be done, in which cells in a sample of blood or marrow are studied to look for certain changes in the structure or function of genes, such as FLT3 and NPM1.
Diagnosing acute myeloid leukemia
In addition to looking at the number and appearance of the cells in the blood samples, your doctor will also order other tests to:
- Confirm the diagnosis
- Identify the AML subtype
- Develop a treatment plan.
Your doctor will work with a hematopathologist to confirm the diagnosis. A hematopathologist is a specialist who studies blood cell diseases by looking at samples of blood and marrow cells and other tissues.
The diagnosis of AML is confirmed by identifying:
- Leukemic blast cells in bone marrow samples
- The percentage of blast cells. Blasts are normally 1 to 5 percent of marrow cells. Having at least 20 percent blasts is generally required for a diagnosis of AML. But AML can also be diagnosed if the blasts have a chromosome change that occurs in a specific type of AML, even if the blast percentage is less than 20 percent.
- Characteristic markers (antigens) on the surface of blast cells, such as CD13 or CD33 (CD is an abbreviation for “cluster designation”).
- Cells based on the types of markers (antigens) on the cell surface, using a process called “immunophenotyping.” “Flow cytometry” is the name of one test that may be used to do immunophenotyping.
Chromosome and Gene Abnormalities
About 60 percent of people with AML have abnormal chromosomes (number and/or structure). These may affect a patient’s response to treatment. Certain changes to chromosomes and genes can provide important information for risk assessment and treatment planning.
Acute myeloid leukemia patients with normal chromosomes may have genetic mutations. Your doctor will perform a molecular analysis on your cells to identify genetic changes.
Acute Myeloid Leukemia Treatment
It’s important that your doctor is experienced in treating patients with acute leukemia or has access to an acute myeloid leukemia specialist.
Acute myeloid leukemia (AML) is often treated in two phases, induction therapy and postremission (consolidation) therapy.
Types of acute myeloid leukemia Treatment
Doctors use several types of treatment for adults with AML, some at different stages.
- Chemotherapy or other drug therapies. Chemotherapy is the use of potent drugs or chemicals, often in combinations or intervals, to kill or damage cancer cells. Some different anticancer drugs are used to treat acute promyelocytic leukemia (subtype M3).
- Stem cell transplantation. This may be used with a second phase of chemotherapy.
- Clinical trials. Clinical trials can involve therapy with new drugs and new drug combinations or new approaches to stem cell transplantation.
Finding the Best Treatment Approach
Most acute myeloid leukemia patients, particularly patients with high white cell counts, need treatment soon after diagnosis because of the disease’s rapid progression. The initial goal of treatment usually is to get the patient into remission. The long-term goal is to cure the disease.
A number of factors affect the choice and outcome of treatment, including:
- Your AML subtype
- The results of cytogenetic analysis
- Whether you have received chemotherapy in the past to treat another type of cancer
- Whether you have had myelodysplastic syndrome (MDS) or another blood cancer
- Whether the AML is in your central nervous system
- Whether your AML has not responded to treatment or has relapsed
- The presence of systemic infection at diagnosis
- Your age and general health.
As you develop a treatment plan with your doctor, be sure to discuss:
- The results you can expect from treatment
- The possibility of participating in a clinical trial, where you’ll have access to advanced medical treatment that may be more beneficial to you than standard treatment
- Potential side effects, including long-term and late effects
You may find it helpful to bring a loved one with you to your doctor’s visits for support, to take notes and ask follow-up questions. It’s a good idea to prepare questions you’d like to ask when you visit your doctor. You can also record your conversations with your doctor and listen more closely when you get home.
Age 60 or older with acute myeloid leukemia
Age 60 or older, patient performance status, other health issues and AML risk factors are all considered in developing your treatment plan, which may vary from standard approaches. For instance, your body may not be able to tolerate toxic chemotherapy drugs or you may have other ailments that are more common as you age. These factors, among others, may make deciding on a treatment more complicated.
What is AML Chemotherapy?
- Chemotherapy is the use of potent drugs or chemicals, often in combinations or intervals, to kill or damage cancer cells.
- Chemotherapy can be hard on the body: The drugs’ toxicity can also damage or kill healthy cells and cause side effects.
- Everyone experiences side effects differently.
Acute myeloid leukemia treatment is generally done in two phases (cycles):
- Induction therapy
- Postremission (consolidation) therapy
- Central nervous system prophylaxis
Treatment for patients with acute promyelocytic leukemia (APL), the M3 subtype of AML, differs from other AML treatments.
Treatment for patients with the FLT3 mutation may receive a different treatment combination.
Induction Therapy
The first phase of your treatment is induction therapy. Its goal is to “induce” (bring on) remission (when no evidence of the disease is left). Specifically, induction therapy for AML attempts to:
- Kill as many AML cells as possible with chemotherapy
- Get healthy blood cell counts back to normal
- Get rid of all signs of the disease for an extended time
What type of treatment is used for AML induction therapy?
- Doctors commonly combine two or more chemotherapy drugs to treat AML. Each type of drug works in a different way to kill the cancerous cells.
- Combining drug types can strengthen their effectiveness.
- Most AML patients are treated with a combination of an anthracycline (such as daunorubicin [Cerubidine®], doxorubicin [Adriamycin® PFS, Adriamycin®] or idarubicin [Idamycin®]) and cytarabine (also called cytosine arabinoside or ara-C [Cytosar-U®]).
- Gemtuzumab ozogamicin (MylotargTM) is FDA approved for the treatment of adults with newly diagnosed AML whose tumors express the CD33 antigen (CD33-positive AML).
- Other drugs may be added or substituted for higher-risk, refractory or relapsed patients.
How are these drugs administered?
These drugs are administered through a catheter (a thin, flexible tube or intravenous line, which is surgically placed in a vein, normally in the upper chest). The anthracycline is usually given in the first 3 days of treatment. The cytarabine is started at the same time but is given for 7 to 10 days. This treatment is called “7 plus 3”.
Where is the treatment done?
- Induction therapy happens in the hospital, usually over 4-6 weeks. You may have to go through several rounds of induction therapy before you go into remission. Usually the same drugs are used for more rounds of treatment.
Post-remission Therapy
After induction therapy is complete and the patient is in remission, there will be another phase of treatment is needed called “postremission therapy,” or “consolidation therapy.” This second phase of treatment is used to destroy any stray AML cells not found by blood or marrow tests. Without postremission therapy, the AML will likely return.
What type of treatment is used for AML postremission therapy?
Postremission therapy consists of additional intensive chemotherapy after remission has been achieved, with or without autologous or allogeneic stem cell transplantation. If stem cell transplantation is not used, the treatment will generally consist of four cycles of chemotherapy. For best results, intensive chemotherapy is given with high doses of cytarabine or other drugs.
Where is the treatment done?
Postremission therapy happens in the hospital and the length of stay depends on the treatment and other factors.
Central Nervous System (CNS) Prophylaxis
CNS prophylaxis is a postremission treatment used to prevent central nervous system (CNS) AML.
What is central nervous system (CNS) AML?
- CNS AML, or meningeal leukemia, is when AML spreads to the meninges (the covering of the spinal cord and brain).
- CNS AML has a tendency to occur in patients with acute lymphoblastic leukemia and acute monocytic leukemia
- It is not common for CNS AML to be present at the time of initial AML diagnosis, and it more commonly occurs in the form of relapse (the cancer returns after the patient is in remission).
Who is treated with central nervous system (CNS) prophylaxis?
Preventative therapy is usually not indicated for CNS AML but examination of the spinal fluid after remission should be considered for some patients.
What type of treatment is used for CNS prophylaxis?
Chemotherapy drugs are injected into the spinal fluid (fluid that surrounds the spinal cord and brain), using a treatment called intrathecal therapy.
Stem Cell Transplantation
Your doctor may recommend an allogeneic stem cell transplantation or an autologous stem cell transplantation to treat your acute myeloid leukemia (AML). Stem cell transplantation isn’t an option for everyone, especially because of the high, sometimes life-threatening risks associated with it. The question of which patients are likely to benefit from transplantation after their first complete remission is under study in clinical trials.
Studies show that allogeneic stem cell transplantation may benefit high-risk and intermediate-risk patients who are younger than 60 and have a sibling match. Treating with a reduced-intensity transplant has shown some benefit for healthier older patients, up to age 75. There does not seem to be any clear advantage for patients considered favorable or chemo-sensitive. Autologous transplant is being used in some centers as an alternative to multiple cycles of chemotherapy.
Allogeneic Stem Cell Transplantation
Allogeneic stem cell transplantation involves transferring stem cells from a healthy person (the donor) to the patient. The procedure follows high-intensity chemotherapy, potent drugs that must be toxic enough to kill leukemic cells. Unfortunately, the drugs also take aim at normal stem cells in the bone marrow.
The main reasons for doing an allogeneic stem cell transplant are:
- To start a new supply of red cells, white cells and platelets with help from the transplanted donor stem cells
- To give strong doses of chemotherapy to kill AML cells
The decision to do a stem cell transplant depends on:
- The availability of a matched donor
- Your response to oral drug therapy
- Your understanding of the transplant’s benefits and risks
Allogeneic stem cell transplantation is used to treat certain AML patients. It is a curative treatment option for some AML patients in first remission. Allogeneic transplantation is associated with a higher rate of side effects and mortality than autologous transplant. However, it may be considered for patients with higher-risk AML, based on cytogenetic and molecular test results. The decision to perform an allogeneic transplant also depends on the age of the patient and the patient’s (or his or her family’s) understanding of the potential benefits and risks. The upper age limit for transplantation varies by treatment center; many centers use age 60 or 65 years for allogeneic transplantation and 70 or 75 years for reduced-intensity allogeneic transplantation.
Reduced-Intensity Allogeneic Stem Cell Transplantation
Reduced-intensity allogeneic stem cell transplantation may be a treatment option for patients who are too old or who may have other medical conditions that prevent them from having a standard allogeneic stem cell transplant. The conditioning therapy used for a reduced-intensity transplant is of lower intensity than that for a standard stem cell transplant; it does not completely inactivate the patient’s immune system or treat the AML as intensively. Thus, if a suitable donor is available, patients up to age 75 may benefit from this form of treatment.
Graft Versus Host Disease
A serious risk of allogeneic and reduced-intensity allogenic stem cell transplantation is graft versus host disease (GVHD), which develops if the donor’s immune cells attack your normal tissue. Graft versus host disease effects can range from minor to life threatening.
Autologous Stem Cell Transplantation
Autologous stem cell transplantation involves “harvesting,” or retrieving, noncancerous stem cells from the patient’s own body and freezing them. The cells are returned to the patient’s body after receiving intensive chemotherapy. The procedure is only appropriate for certain patients.
The question of which patients are likely to benefit from transplantation after their first complete remission is under study in clinical trials. The decision to do a stem cell transplant depends on whether the patient’s AML is favorable risk, intermediate risk or high risk. The doctor also considers:
- The patient’s overall health
- The chances that chemotherapy alone will cure the AML
- The type of abnormal changes to the chromosomes and cells
- The availability of a matched donor, if necessary
- The patient’s understanding of the benefits and risks of transplant
Autologous transplantation is relatively safe for many patients, including older patients. For some AML patients who do not have an HLA-matched stem cell donor, therapy can be further intensified with very-high-dose chemotherapy followed by an autologous transplant.
Childhood Acute Myeloid Leukemia (AML)
Acute myeloid leukemia is one of the most challenging childhood cancers to treat. Because of the intensity of therapy utilized to treat AML, children with this disease should have their care coordinated by pediatric hematology-oncology specialists and be treated in cancer centers or hospitals with the appropriate supportive care facilities and services.
Chemotherapy. Children who have AML are treated with an induction therapy similar to that for adults with AML: cytarabine and drugs such as doxorubicin or daunorubicin, or a third drug, such as mitoxantrone. More treatment is needed after a child with AML is in remission (postremission therapy). This is because some AML cells may remain after induction therapy and these AML cells do not show up in standard blood or marrow tests. Postremission therapy in children includes a number of chemotherapy drugs. This treatment results in about an 80 percent remission rate. Slightly more than half of the children in relapse-free remission are considered cured.
Generally, AML treatment is less effective for children who 12:
- Are younger than 2 years old
- Have the AML subtype acute monocytic leukemia
- Have a type of leukemia called hyperleukocytic leukemia, which has a high number of blast cells
For children who receive intensive chemotherapy, including anthracyclines, ongoing monitoring of cardiac function is critical since these drugs may affect the heart. Periodic examination of kidney function and auditory exams are also recommended.
Clinical Trials. When it comes to finding the right treatment for a child’s AML, a clinical trial may be the best treatment option, as clinical trials provide access to new or improved therapies under study and not yet on the market. Treatment in clinical trials is administered in a safe, closely monitored environment. The possibility of participating in a clinical trial should be discussed with the doctor.
Researchers are studying treatment in clinical trials for children and young adults with AML with the aim to:
- Improve cure rates
- Decrease side effects and long-term and late effects of chemotherapy
- Make AML therapy safer and reduce chemotherapy side effects and complications, such as infections
- Determine the best treatments for children who have a poor chance of recovering from AML
Researchers have identified cell targets that appear to be the key to treatment with the new generation of chemotherapy agents. These new targeted agents are being studied in conjunction with chemotherapy to examine their impact upon cure rates and their effect on toxic complications associated with traditional chemotherapy.
Stem Cell Transplantation. An allogeneic stem cell transplantation may be used to treat children who have:
- High-risk AML, based on cytogenetic and molecular test results
- Primary induction failure
- Relapse after intensive multidrug therapy.
Survivorship and Special Healthcare Needs
After treatment, most children can expect to have full and productive lives. Many survivors return to school, attend college, enter the workforce, marry and become parents.
Childhood cancer survivors have special long-term healthcare needs. Survivorship programs focus on life after cancer and can be very helpful for children with cancer. Several major hospitals around the country offer these programs.
Follow-Up Care
A child should visit his or her pediatrician or doctor at least once a year for a complete physical exam and any additional needed tests. The oncologist should also regularly examine the child.
Regular doctor visits are encouraged to:
- Enable doctors to assess the full effect of therapy
- Detect and treat disease recurrence
- Identify and manage long-term and late effects of treatment
The pediatrician should recommend a schedule for having the child’s learning skills assessed. If the child appears to be experiencing learning disabilities, special education methods can help.
Coordination between the child’s pediatrician and oncologist is important for the best care possible. Some treatment centers offer comprehensive follow-up care clinics for childhood cancer survivors.
Long-Term and Late Effects of Treatment
Some side effects of cancer treatment, such as fatigue, can linger for months or years after therapy. Some medical conditions like heart disease and other cancers may not appear until years after treatment ends.
Most childhood survivors of leukemia don’t develop significant long-term or late effects of treatment. However, for some patients the effects can range from mild to severe. For children who receive intensive chemotherapy, including anthracyclines, ongoing monitoring of cardiac function is critical since these drugs may affect the heart. Periodic examination of kidney function and auditory exams are also recommended.
Talk to the child’s treatment team about possible long-term and late effects. His or her risk for developing long-term or late effects can be influenced by:
- Treatment type and duration
- Age at the time of treatment
- Gender
- Overall health
Some long-term and late effects become evident with maturation (puberty), growth and the normal aging process. Have the child evaluated with a physical exam yearly or more often as needed. Early intervention and healthy lifestyle practices (not smoking, good nutrition, exercise, regular screenings and follow-up) help.
Long-term and late effects can impact a child’s physical, mental and cognitive health in a number of ways.
Physical Effects. Children treated for acute myeloid leukemia may be at increased risk for:
- Fatigue
- Growth delays
- Thyroid dysfunction
- Hearing loss
- A secondary cancer
Mental Effects. Most childhood survivors of cancer are psychologically healthy. However, some studies have indicated that a small number of childhood leukemia survivors were more likely than healthy peers to report changes in mood, feelings and behavior, including depression and posttraumatic stress disorder.
Cognitive Effects. Learning disabilities can begin during treatment or appear months or years afterward. Areas that can be affected include:
- Mathematics
- Spatial relationships
- Problem solving
- Attention span
- Reading
- Spelling
- Information processing
- Planning and organizing
- Concentration skills
- Fine motor coordination
Returning to School
Once your child is in remission, he or she will likely be going back to school. This reentry to the classroom can be daunting for a child of any age. Educate family members, friends, school personnel and healthcare providers about your child’s possible long-term and late effects of treatment. Talk with teachers about your child’s needs before he or she returns to school. Work with your child’s teachers and medical providers to develop a program tailored to his or her needs that features baseline testing, special accommodations and long-term planning.
Acute Myeloid Leukemia Treatment Outcomes
Acute myeloid leukemia can be a difficult disease to cure. However, advances in AML treatment have resulted in improved remission (an absence of signs and symptoms) and cure rates.
Treatment outcomes can be broken down into four categories. The table below describes the difference between each category.
| Active disease |
|
| Minimal residual disease |
|
| Remission |
|
| Complete molecular remission |
|
Refractory Acute Myeloid Leukemia
Most patients achieve a remission (an absence of signs and symptoms) after initial treatment for acute myeloid leukemia. However, some patients have residual leukemic cells in their marrow even after intensive treatment. This is referred to as “refractory leukemia.”
Gemtuzumab ozogamicin (MylotargTM) is FDA approved for the treatment of patients aged 2 years and older with CD33-positive AML who have experienced a relapse or who have not responded to initial treatment (refractory).
For refractory AML, treatment options may include drugs not already used during the first course of treatment. Stem cell transplantation may be used when remission is achieved, which may result in a more durable remission.
Relapsed Acute Myeloid Leukemia
Some patients reach remission and then have a return of leukemia cells in the marrow and a decrease in normal blood cells. This is called “relapsed leukemia.”
In patients who relapse, the duration of the remission, the patient’s age and the cytogenetic findings in the leukemia cells influence the approach to therapy. Drugs similar to those administered initially, different drugs or stem cell transplantation may be used to treat the leukemia.
Gemtuzumab ozogamicin (MylotargTM) is FDA approved for the treatment of patients aged 2 years and older with CD33-positive AML who have experienced a relapse or who have not responded to initial treatment (refractory).
Stem Cell Transplantation in Relapsed Patients
Allogeneic stem cell transplantation may be a treatment option for patients in early first relapse or second remission, although this is a high-risk procedure. For patients who lack a sibling donor, matched-unrelated donor transplants can be effective. Patients with AML who relapse after allogeneic stem cell transplantation may have a long-term remission if they have a second transplant. Donor leukocyte infusion is sometimes used to treat relapsed AML post transplant. This therapy is most effective in early relapses and in the absence of extensive chronic graft-versus-host disease (GVHD).
Clinical Trials
For some patients with relapsed or refractory AML, the best treatment route may be one that is being studied in a clinical trial. Several drugs and drug combinations used to treat AML are currently being studied.
Chronic myeloid leukemia
Chronic myeloid leukemia (CML), also known as chronic myelogenous leukemia, chronic granulocytic leukemia and chronic myelocytic leukemia, is a type of cancer that starts in certain blood-forming cells of the bone marrow. In CML, a genetic change takes place in an early (immature) version of myeloid cells – the cells that make red blood cells, platelets, and most types of white blood cells (except lymphocytes). This change forms an abnormal gene called BCR-ABL, which turns the cell into a CML cell. The leukemia cells grow and divide, building up in the bone marrow and spilling over into the blood. In time, the cells can also settle in other parts of the body, including the spleen. CML is a fairly slow growing leukemia, but it can also change into a fast-growing acute leukemia that is hard to treat.
Most cases of CML occur in adults, but very rarely it occurs in children, too. In general, their treatment is the same as for adults.
The American Cancer Society’s estimates for chronic myeloid leukemia (CML) in the United States for 2017 are:
- About 8,950 new cases will be diagnosed with CML (5,230 in men and 3,720 in women).
- About 1,080 people will die of CML (610 men and 470 women).
A little over 10% of all new cases of leukemia are chronic myeloid leukemia. About 1 person in 555 will get CML in their lifetime in the United States.
The average age at diagnosis of CML is around 64 years. Almost half of cases are diagnosed in people 65 and older. This type of leukemia mainly affects adults, and is only rarely seen in children.
What Causes Chronic Myeloid Leukemia?
Most cases of chronic myeloid leukemia (CML) start when a “swapping” of chromosomal material (DNA) occurs between chromosomes 9 and 22 during cell division. Part of chromosome 9 goes to 22 and part of 22 goes to 9. This is known as a translocation and gives rise to a chromosome 22 that is shorter than normal. This new abnormal chromosome is known as the Philadelphia chromosome. The Philadelphia chromosome is found in the leukemia cells of almost all patients with chronic myeloid leukemia (CML).
The swapping of DNA between the chromosomes leads to the formation of a new gene (an oncogene) called BCR-ABL. This gene then produces the BCR-ABL protein, which is the type of protein called a tyrosine kinase. This protein causes chronic myeloid leukemia (CML) cells to grow and reproduce out of control.
In a very small number of chronic myeloid leukemia (CML) patients, the leukemia cells have the BCR-ABL oncogene but not the Philadelphia chromosome. It is thought that the BCR-ABL gene must form in a different way in these people. In a very small number of people who seem to have CML, neither the Philadelphia chromosome nor the BCR-ABL oncogene can be found. They might have other, unknown oncogenes causing their disease and are not considered to truly have chronic myeloid leukemia (CML).
Sometimes people inherit DNA mutations from a parent that greatly increase their risk of getting certain types of cancer. But inherited mutations do not cause chronic myeloid leukemia (CML). DNA changes related to chronic myeloid leukemia (CML) occur during the person’s lifetime, rather than having been inherited before birth.
Risk Factors for Chronic Myeloid Leukemia
For most people who have chronic myeloid leukemia (CML), there are no obvious reasons why they develop the disease. No one is born with CML. It happens when there is an injury to the DNA of a single bone marrow cell. Risk factors for CML are:
- Gender – CML is slightly more common in males than females.
- Age – The risk of getting CML increases with age.
- Radiation exposure – In a small number of patients, CML is caused by exposure to very high doses of radiation (such as being a survivor of an atomic bomb blast or a nuclear reactor accident). A slight increase in risk also occurs in some individuals treated with high-dose radiation therapy for other cancers, such as lymphoma. Most people treated for cancer with radiation do not develop CML, and most people who have CML have not been exposed to high doses of radiation. Exposures to diagnostic dental or medical x-rays have not been associated with an increased risk of CML.
Signs and Symptoms of chronic myeloid leukemia
People who have chronic myeloid leukemia (CML) may not have any symptoms at first. Often, patients learn they have CML after a routine physical exam or a blood test. CML signs and symptoms tend to develop gradually. Those with symptoms often report
- Weakness
- Fatigue
- Shortness of breath during basic everyday activities
- Fever
- Bone pain
- Unexplained weight loss
- Pain or a feeling of fullness below the ribs on the left side due to an enlarged spleen
- Night sweats.
Many signs and symptoms of CML occur because the CML cells crowd out the bone marrow’s healthy red blood cells, white blood cells and platelets. Anemia is a shortage of red blood cells that can cause weakness, fatigue and shortness of breath. A shortage of normal white blood cells can increase a CML patient’s risk of infection, and a shortage of platelets can lead to excessive bruising or bleeding. Symptoms may also occur because CML cells collect in organs such as the spleen.
Diagnosis of chronic myeloid leukemia
Many people with CML do not have symptoms when diagnosed. The most common sign of CML is an abnormal white blood cell count often found during blood tests for an unrelated health problem or during a routine checkup. To diagnose CML, doctors use a variety of tests to analyze blood and bone marrow cells. A pathologist, a doctor who specializes in identifying diseases by studying cells under a microscope, will examine the blood cells and the bone marrow cells. The samples should also be examined by a hematopathologist, a specialist who diagnoses diseases of the blood and marrow.
Tests that are used to diagnose CML
Complete Blood Count (CBC) with Differential. This test is used to measure the number red blood cells, white blood cells and platelets in a sample of blood. It also measures the amount of hemoglobin in the red blood cells and the percentage of red blood cells in the sample. The CBC should include a differential. The differential measures the different types of white blood cells in the sample.
People with CML often have
- An increased white blood cell count, often to very high levels
- A decreased red blood cell count
- A possible increase or decrease in the number of platelets, depending on the severity of the person’s CML.
Peripheral Blood Smear. Blood cells are stained (dyed) and examined with a light microscope. These samples show
- The number, size, shape and type of blood cells
- Specific pattern of white blood cells
- The proportion of immature cells (blast cells) compared to the proportion of maturing and fully matured white blood cells. Blast cells are normally not present in the blood of healthy individuals.
Bone Marrow Aspiration and Biopsy. These tests are used to examine bone marrow cells to find abnormalities, and are generally done at the same time. The sample is usually taken from the patient’s hip bone after medicine has been given to numb the skin. For a bone marrow aspiration, a hollow needle is inserted through the hip bone and into the bone marrow to remove a liquid sample of cells. For a bone marrow biopsy, a wider needle is used to remove a small piece of bone that contains marrow. Both samples are examined under a microscope to look for chromosomal and other cell changes.
Cytogenetic Analysis. Cytogenetics is the study of chromosomes and chromosomal abnormalities. Samples from the bone marrow are examined under a microscope for chromosomal changes or abnormalities such as the Philadelphia (Ph) chromosome. The presence of the Ph chromosome in the bone marrow cells, along with a high white blood cell count and other characteristic blood and bone marrow test findings, confirm the diagnosis of CML. The bone marrow cells of about 95 percent of people with CML have a Ph chromosome that is detectable by cytogenetic analysis. A small percentage of people with clinical signs of CML do not have cytogenetically detectable Ph chromosome, but they almost always test positive for the BCR-ABL fusion gene on chromosome 22 with other types of tests.
FISH (Fluorescence In Situ Hybridization). FISH is a laboratory test used to examine genes and chromosomes in cells. FISH is a more sensitive method for detecting CML than the standard cytogenetic tests that identify the Ph chromosome. FISH can identify the presence of the BCR-ABL gene. Genes are made up of DNA segments. FISH uses color probes that bind to DNA to locate the BCR and ABL genes in chromosomes. Both BCR and ABL genes are labeled with different chemicals, each of which releases a different color. The color shows up on the chromosome that contains the gene—normally chromosome 9 for ABL and chromosome 22 for BCR—so FISH can detect the piece of chromosome 9 that has moved to chromosome 22 in CML cells. The BCR-ABL fusion gene is shown by the overlapping colors of the two probes.
Quantitative Polymerase Chain Reaction (qPCR). qPCR is the most sensitive test that detects and measures the quantity of the BCR-ABL gene in blood or bone marrow samples. It can detect very small amounts of the BCR-ABL gene (even when the Ph chromosome cannot be detected in blood or bone marrow cells with cytogenetic testing) to a level of one CML cell in a background of 100,000 to 1,000,000 normal cells.
Blood cell counts, bone marrow examinations, FISH and qPCR may also be used to track a person’s response to therapy once treatment has begun. qPCR testing is recommended every 3 months for 2 years as long as the patient’s CML is responding to treatment. After 2 years, the test should be done every 3 to 6 months.
Chronic Myeloid Leukemia Staging
Most types of cancer are assigned a stage based on the size of the tumor and the extent of cancer spread. Stages can be helpful in predicting prognosis (outlook).
But because chronic myeloid leukemia (CML) is a disease of the bone marrow, it isn’t staged like most cancers. The outlook for someone with CML depends on other information, such as the phase of the disease, as well as factors like the age of the patient, blood counts, and if the spleen is enlarged.
Phases of chronic myeloid leukemia
CML is classified into 3 groups that help predict outlook. Doctors call these groups phases instead of stages. The phases are based mainly on the number of immature white blood cells — myeloblasts (blasts) — that are seen in the blood or bone marrow. Different groups of experts have suggested slightly different cutoffs to define the phases, but a common system (proposed by the World Health Organization) is described below. Not all doctors may agree with or follow these cutoff points for the different phases. If you have questions about what phase your CML is in, be sure to have your doctor explain it to you.
Chronic phase
Patients in this phase typically have less than 10% blasts in their blood or bone marrow samples. These patients usually have fairly mild symptoms (if any) and usually respond to standard treatments. Most patients are diagnosed in the chronic phase.
Accelerated phase
Patients are considered to be in accelerated phase if any of the following are true:
- The bone marrow or blood samples have more than 10% but fewer than 20% blasts
- High blood basophil count (basophils making up at least 20% of the white blood cells)
- High white blood cell counts that do not go down with treatment
- Very high or very low platelet counts that are not caused by treatment
- New chromosome changes in the leukemia cells
Patients whose CML is in accelerated phase may have symptoms such as fever, poor appetite, and weight loss. CML in the accelerated phase does not respond as well to treatment as CML in the chronic phase.
Blast phase (also called acute phase or blast crisis)
Bone marrow and/or blood samples from a patient in this phase have more than 20% blasts. The blast cells often spread to tissues and organs beyond the bone marrow. These patients often have fever, poor appetite, and weight loss. In this phase, the CML acts much like an aggressive acute leukemia.
Prognostic factors for chronic myeloid leukemia
Along with the phase of CML, there are other factors that may help predict the outlook for survival. These factors are sometimes helpful when choosing treatment. Factors that tend to be linked with shorter survival time are called adverse prognostic factors.
Adverse prognostic factors:
- Accelerated phase or blast phase
- Enlarged spleen
- Areas of bone damage from growth of leukemia
- Increased number of basophils and eosinophils (certain types of granulocytes) in blood samples
- Very high or very low platelet counts
- Age 60 years or older
- Multiple chromosome changes in the CML cells
Many of these factors are taken into account in the Sokal system, which develops a score used to help predict prognosis. This system considers the person’s age, the percentage of blasts in the blood, the size of the spleen, and the number of platelets. These factors are used to divide patients into low-, intermediate-, or high-risk groups. Another system, called the Euro score, includes the above factors, as well as the number of blood basophils and eosinophils. Having more of these cells indicates a poorer outlook.
The Sokal and Euro models were helpful in the past, before the newer, more effective drugs for CML were developed. It’s not clear how helpful they are at this time in predicting a person’s outlook. Targeted therapy drugs like imatinib (Gleevec®) have changed the treatment of CML dramatically over the last several years. These models haven’t been tested in people who are being treated with these drugs.
Chronic Myeloid Leukemia Treatment
Targeted therapy drugs are the main treatment for CML. Some patients might also need other treatments, such as:
- Interferon
- Chemotherapy
- Radiation therapy
- Surgery
- Stem cell transplant
Targeted Therapies for Chronic Myeloid Leukemia
Chronic myeloid leukemia (CML) cells contain an oncogene, BCR-ABL, that isn’t found in normal cells. This gene makes a protein, BCR-ABL, which causes CML cells to grow and reproduce out of control. BCR-ABL is a type of protein known as a tyrosine kinase. Drugs known as tyrosine kinase inhibitors (TKIs) that target BCR-ABL are the standard treatment for CML. These include:
- Imatinib (Gleevec)
- Dasatinib (Sprycel®)
- Nilotinib (Tasigna®)
- Bosutinib (Bosulif®)
- Ponatinib (Iclusig®)
All of these drugs can have serious or even deadly interactions with other drugs, over the counter supplements, and even certain foods (such as grapefruit and pomegranate). Be sure that your doctor always has an up-to-date list of any medicines you are taking, including over-the-counter medicines, vitamins, and herbal supplements. You also need to check with your doctor before starting any new medicine, to be sure it is safe.
It is also important to understand that all of the TKIs can harm the fetus if taken during pregnancy. These drugs seem to work best on CML that is still in the chronic phase, but they also can help patients with more advanced disease for some time.
Imatinib
Imatinib (Gleevec) was the first drug to specifically target the BCR-ABL tyrosine kinase protein, and it quickly became the standard treatment for CML patients. Because it was the first TKI, imatinib is known as a first generation tyrosine kinase inhibitor.
Almost all CML patients respond to treatment with imatinib, and most of these responses seem to last for many years. This drug doesn’t seem to make the leukemia go away and stay away, so patients need to take it indefinitely (or until it stops working). Imatinib is taken by mouth as a pill with food, usually once a day.
Common side effects can include diarrhea, nausea, muscle pain, and fatigue. These are generally mild. About 30% of people taking the drug have itchy skin rashes. Most of these symptoms can be treated effectively, if needed.
Another common side effect is fluid buildup around the eyes, feet, or abdomen. In rare cases the fluid may collect in the lungs or around the heart, which can cause trouble breathing. Some studies have suggested that some of this fluid buildup may be caused by effects of the drug on the heart, though this is rare. It’s not yet clear how serious this is or if it might go away if treatment is stopped. If you are taking this drug, tell your doctor right away if you notice sudden weight gain or fluid buildup anywhere in the body or have trouble breathing.
Another possible side effect is a drop in a person’s white blood cell and platelet counts. When this happens at the beginning of treatment, it might be because the blood-forming cells that are making these are part of the malignant process. If this is the case, normal blood-forming cells take over and the blood counts will begin to rise to normal over time.
Your doctor might tell you to stop taking the drug for a short period if your blood counts get too low. This can also happen later on in treatment. In the past, low red blood cell counts were treated with a red cell growth factor, such as erythropoietin (Procrit®) or darbepoietin (Aranesp®), but these drugs are used less often now. Instead, your doctor may lower the dose of imatinib to see if your blood counts improve.
In some patients, imatinib eventually seems to stop working. This is known as imatinib resistance. Resistance to imatinib seems to be caused by changes in the genes of the CML cells. Sometimes this resistance can be overcome by increasing the dose of imatinib, but some patients need to change to a different drug, such as one of the other TKIs described further on.
Dasatinib
Dasatinib (Sprycel) is another tyrosine kinase inhibitor that targets the BCR-ABL protein. Because it was developed after imatinib, it is called a second generation TKI. Like imatinib, this drug is a pill taken by mouth.
Dasatinib can be used as the first treatment for CML, but it can also be helpful for patients who can’t take imatinib because of side effects or because imatinib isn’t working.
When it was first approved, dasatinib was a pill taken twice a day, but more often now a larger dose is taken once a day.
The possible side effects of dasatinib seem to be similar to those of imatinib, including fluid buildup, lowered blood cell counts, nausea, diarrhea, and skin rashes. A serious side effect that can occur with this drug is fluid buildup around the lung (called a pleural effusion). This side effect is more common in patients taking this drug twice a day. The fluid can be drained off with a needle, but it can build up again, and may require the dose of dasatinib to be decreased.
Nilotinib
Nilotinib (Tasigna) is another second generation TKI that targets the BCR-ABL protein. Like dasatinib, this drug can be used as a first treatment for CML, as well as for use in people who can’t take imatinib or whose CML no longer responds to it.
Side effects of nilotinib seem to be mild, but can include fluid buildup, lowered blood cell counts, nausea, diarrhea, and some lab test abnormalities. It can cause high blood sugar and pancreatitis, although this is rare.
This drug can also affect the rhythm of the heart, causing a condition called prolonged QT syndrome. This usually doesn’t cause any symptoms, but can be serious or even fatal. This is why patients should have an electrocardiogram (EKG) before starting nilotinib and then again while being treated. This heart rhythm problem can sometimes be caused by nilotinib interacting with other drugs or supplements, so it’s especially important to be sure that your cancer doctor knows about any medicines you take, including over-the-counter medicines and supplements. You also need to check with your doctor before starting any new medicine, to be sure it is safe.
Bosutinib
Bosutinib (Bosulif®) is another TKI that targets the BCR-ABL protein. At this time, this drug is only approved by the US Food and Drug Administration to treat patients after they have been treated with another TKI.
Common side effects are usually mild and include diarrhea, nausea, vomiting, abdominal pain, rash, fever, fatigue, and low blood cell counts (including low platelet counts, low red blood cell counts, and low white blood cell counts). Less often, this drug can also cause problems with fluid retention, liver damage, and severe allergic reaction. Your doctor will check your blood test results regularly to watch for problems with your liver and low blood counts.
Ponatinib
Ponatinib (Iclusig®) is a new TKI targeting the BCR-ABL protein. Because of risks of some serious side effects, this drug is only used to treat patients with CML if all of the other TKIs don’t work or if their leukemia cells have a certain gene change called the T315I mutation. This mutation (gene change) occurs in the leukemia cells of some CML patients who are treated with a TKI, and it prevents other TKIs from working. Ponatinib is the first TKI to work against CML cells that have this mutation.
This drug is a pill taken once a day.
Most side effects are mild and can include abdominal (belly) pain, headache, rash or other skin problems, and fatigue.
High blood pressure is also fairly common, and it may need to be treated with a blood pressure drug.
There is also a risk of serious blood clots that can lead to heart attacks and strokes, or block arteries and veins in the arms and legs. Rarely, blood clots in patients taking this drug have cut off circulation, and lead to an arm or leg needing to amputated (cut off). Surgery or some other procedure may be needed to treat these blood clots. The risk of serious blood clots is higher in older patients, those with certain risk factors, such as high blood pressure, high cholesterol, or diabetes, and those who have already had a heart attack, stroke, or poor circulation.
More rarely, this drug can also weaken the heart muscle, leading to a condition known as congestive heart failure. It can also cause liver problems, including liver failure, as well as pancreatitis (inflammation of the pancreas, which can lead to severe belly pain, nausea, and vomiting).
Coping and support
A diagnosis of leukemia may be devastating — especially for the family of a newly diagnosed child. With time you’ll find ways to cope with the distress and uncertainty of cancer. Until then, you may find it helps to:
- Learn enough about leukemia to make decisions about your care. Ask your doctor about your leukemia, including your treatment options and, if you like, your prognosis. As you learn more about leukemia, you may become more confident in making treatment decisions.
- Keep friends and family close. Keeping your close relationships strong will help you deal with your leukemia. Friends and family can provide the practical support you’ll need, such as helping take care of your house if you’re in the hospital. And they can serve as emotional support when you feel overwhelmed by cancer.
- Find someone to talk with. Find a good listener who is willing to listen to you talk about your hopes and fears. This may be a friend or family member. The concern and understanding of a counselor, medical social worker, clergy member or cancer support group also may be helpful.
- Ask your doctor about support groups in your area. Or check your phone book, library or a cancer organization, such as:
- Leukemia. https://www.cancer.gov/types/leukemia[↩]
- Cancer Stat Facts: Leukemia – Acute Myeloid Leukemia (AML). https://seer.cancer.gov/statfacts/html/amyl.html[↩][↩]
- Facts and Statistics. http://www.lls.org/facts-and-statistics/facts-and-statistics-overview[↩]
- Cancer Stat Facts: Leukemia – Acute Lymphocytic Leukemia (ALL). https://seer.cancer.gov/statfacts/html/alyl.html[↩]
- https://cancerstatisticscenter.cancer.org/#!/cancer-site/Leukemia[↩]
- Acute Myeloid Leukemia (AML). https://www.cancer.org/cancer/acute-myeloid-leukemia.html[↩]
- Treating AML: Current and Novel Approaches (ASH/LLS). https://ashondemand.org/Webinar/074/June-16-2017[↩][↩]
- Surveillance, Epidemiology and End Results Program. https://seer.cancer.gov/statfacts/html/amyl.html[↩]
- Acute myeloid leukemia. http://www.lls.org/leukemia/acute-myeloid-leukemia?src1=20032&src2=[↩]
- How Is Acute Myeloid Leukemia Classified? https://www.cancer.org/cancer/acute-myeloid-leukemia/detection-diagnosis-staging/how-classified.html[↩]
- Acute myeloid leukemia (AML). http://www.lls.org/leukemia/acute-myeloid-leukemia?src1=20032&src2=[↩]
- Childhood AML. http://www.lls.org/leukemia/acute-myeloid-leukemia/childhood-aml[↩]
- National Cancer Institute. https://www.cancer.gov/[↩]
- Leukemia and Lymphoma Society. http://www.lls.org/[↩]