Contents
What is macrocytic anemia
The term macrocytosis refers to a blood condition in which red blood cells (RBCs) are larger than normal. Macrocytosis is reported in terms of mean corpuscular volume (MCV). Normal MCV values range from 80 to 100 femtoliters (fl) and vary by age and reference laboratory 1.
Macrocytosis can be identified by reviewing peripheral blood smears and/or by automated red blood cell indices. The peripheral blood smear is more sensitive than red blood cell indices for identifying early macrocytic changes because the mean corpuscular volume (MCV) represents the mean of the distribution curve and is insensitive to the presence of small numbers of macrocytes 2. However, compared to the peripheral blood smear, mean corpuscular volume (MCV) may underestimate macrocytosis in over 30% of cases 3.
Macrocytosis is a relatively common finding in the era of automated blood cell counters, with prevalence estimates ranging from 1.7% to 3.6% 4. Its significance tends to be underestimated by physicians, since about 60% of patients present without associated anemia 4, unless there are other accompanying abnormalities noted.
Macrocytic anemia describes an anemic state characterized by the presence of abnormally large red blood cells in the peripheral blood. This abnormality is usually recognized by the automated blood cell counter and confirmed on review of the peripheral blood smear. The cause of macrocytic anemia may be due to a variety of illnesses and demands further clinical and laboratory assessment.
Macrocytosis Without Anemia
Large circulating erythrocytes (red blood cells) are not always associated with a pathologic process or condition. In fact, red blood cells of newborns and infants tend to be larger (mean MCV = 108 fl) than normal adult red blood cells 5 and large red blood cells can be seen during pregnancy in the absence of an obvious etiology. Macrocytosis without anemia may be a normal variant and is only noted as a result of repeated peripheral red blood cell indices in the absence of any known or existing clinical problems. In some instances this variation from normal can be found in other family members, which suggests a genetic predisposition, and requires no therapeutic intervention or further investigation 5, 6.
Macrocytic anemia causes
Macrocytic anemia can usually be divided into two categories:
- Megaloblastic: Megaloblastic anemia is caused by deficiency or impairment of utilization of vitamin B12 or folate.
- Nonmegaloblastic: Nonmegaloblastic anemia may be the result of liver dysfunction, alcoholism, myelodysplastic syndrome (MDS), or hypothyroidism.
This categorization is important and frequently aids in determining the etiology of the anemia. Additionally, a careful review of the peripheral blood smear noting the morphology of the red blood cells, as well as the other cellular elements and features on the smear, can provide important clues as to the etiology of the anemia.
Common causes of macrocytosis are different by region and setting. For example, in New York, 37% of cases diagnosed in hospitalized patients were medication related 7. Antiretroviral therapy (ART) for human immunodeficiency virus (HIV) infections accounted for 13% 7. In Finland, the common causes of macrocytic anemias were alcoholism (65%) 8 and vitamin B12 or folate deficiency (28%) 9 in outpatients over 75 years of age.
Vitamin B12 deficiency is the most common cause of megaloblastic anemia. Vitamin B12 deficiency is caused by insufficient dietary intake, as in the cases of vegetarians or malnutrition, malabsorption due to the absence of intrinsic factor caused by pernicious anemia or following gastric surgery, congenital disorders, such as transcobalamin II deficiency, or exposure to nitrous oxide.
The result of one study, conducted in Japan, indicated that the most common cause of megaloblastic anemia is pernicious anemia (61%), followed by vitamin B12 deficiency due to gastrectomy (34%), vitamin B12 deficiency due to other causes (2%), and folate deficiency (2%) 10. Vitamin B12 is contained in animal foods, and the daily intake is approximately 3‐30 μg. The daily required amount is approximately 1‐3 μg, and except for stomach or intestinal obstruction, or being a strict vegetarian, vitamin B12 deficiency is rare.
Vitamin B12 binds to intrinsic factor secreted by the gastric parietal cells, and it is absorbed in the terminal ileum. Once absorbed, vitamin B12 acts as a coenzyme in the enzymatic reaction that produces methionine from homocysteine. As a result, folic acid is converted into its active form. When vitamin B12 is deficient, active folic acid is also deficient. As a result, the intracellular reaction involving the coenzyme form of folic acid is affected. Thus, not only vitamin B12 but also folate deficiencies impair DNA synthesis. Because a large amount of vitamin B12 is stored in the liver, it takes 5‐10 years for clinical problems to manifest following decreased intake or absorption of vitamin B12 11.
Folic acid is contained in green vegetables and animal products, such as liver. The recommended dietary allowance of folic acid for adults is 240 μg a day, and an intake of around 400 μg each day is necessary for pregnant or lactating women. Folate deficiency may increase the risk of a congenital neural tube stenosis during pregnancy. Folic acid is absorbed in the upper jejunum by both passive diffusion and active uptake. Folate deficiency is caused by nutritional deficiency (eg, poor diet, alcoholism), malabsorption (eg, celiac disease, inflammatory bowel disease), increased requirements (eg, pregnancy, lactation, chronic hemolysis), or medication (eg, methotrexate, trimethoprim, phenytoin). Because serum folate levels fluctuate with dietary intake, measurement of red blood cell folate levels, which reflect folate stores in tissue, has been considered more reliable 12. Patients are usually treated with oral folic acid if the cause of folate deficiency is nutritional deficiency or increased nutritional requirements.
The spectrum of causes associated with macrocytic anemia includes:
- Nutritional deficiencies (e.g., vitamin B12 and folate),
- Primary bone marrow disorders (e.g., myelodysplasia and leukemia) and other chronic illnesses (Table 1) and
- Drugs (Table 2).
Macrocytosis due to vitamin B12 or folate deficiency is a direct result of ineffective or dysplastic erythropoiesis. These important vitamins and cofactors are required for normal maturation of all cells. Marrow erythroblasts are no exception. When either of these two factors is deficient, red blood cell proliferation and maturation result in large erythroblasts with nuclear/cytoplasmic asynchrony. These abnormalities are caused by a defect in DNA synthesis that interferes with cellular proliferation and maturation. RNA synthesis and cytoplasmic components remain relatively unaffected. The marrow is hypercellular with all forms of the myeloid cell line being increased and erythroid elements being dominant on the marrow aspirate smear preparations. The erythroblasts become large, oval shaped and contain a characteristic immature, lacy nucleus. These bone marrow features are called “megaloblastic” and are highly suspicious of a vitamin B12 or folate deficiency. Megaloblastoid (megaloblastic-like) abnormalities of the marrow are frequently seen in other hematologic disorders not associated with vitamin B12 or folate deficiency, (e.g., myelodysplasia and leukemia) and a careful examination of the bone marrow is necessary to make this distinction.
Macrocytosis is frequently linked to alcoholism, with or without liver disease. In fact, it is purported to be one of the most common causes of nonmegaloblastic macrocytosis 13.
Table 1. Common causes of macrocytosis
| Drugs |
| Alcoholism |
| Reticulocytosis |
| Nonalcoholic and alcoholic liver disease |
| Hypothyroidism |
| Vitamin B12 deficiency |
| Folate deficiency |
| Multiple myeloma |
| Myelodysplastic syndromes |
| Aplastic anemia |
| Acute leukemia |
Table 2. Drugs that may induce macrocytosis
| Chemotherapeutic agents | Antimicrobials |
| Cyclophosphamide | Pyrimethamine |
| Hydroxyurea | Sulfamethoxazole |
| Methotrexate | Trimethoprim |
| Azathioprine | Valacyclovir |
| Mercaptopurine | |
| Cladribine | Diuretics |
| Cytosine arabinoside | Triamterene |
| 5-Fluouracil | |
| Antiretroviral | Anticonvulsant agents |
| Zidovudine | Phenytoin |
| Stavudine | Primidone |
| Valproic acid | |
| Hypoglycemic | Anti-inflammatory |
| Metformin | Sulfasalazine |
| Other | |
| Nitrous oxide |
Figure 1. Flowchart for the differential diagnosis of macrocytic anemias (Note: MDS = myelodysplastic syndrome)
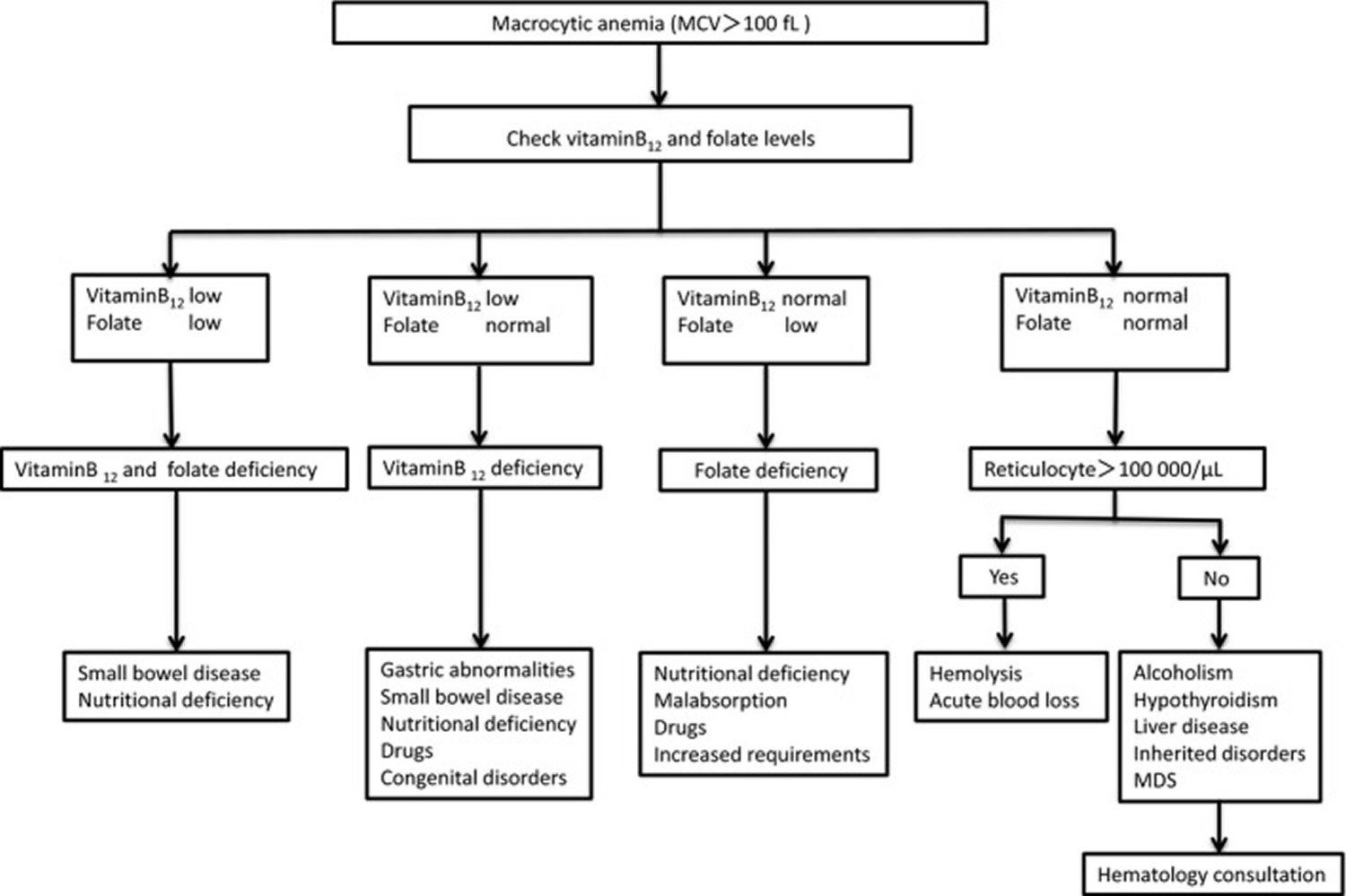 [Source 15]
[Source 15]Macrocytic anemia symptoms
If you have anemia, you will feel tired and short of breath, even when doing things you could usually do easily.
You may also have a fast or irregular heartbeat, look pale, have cold feet or hands, feel dizzy or have problems thinking.
It’s important to see your doctor if you have any of these symptoms.
Signs and symptoms of anemia
- Fatigue
- Weakness
- Pale or yellowish skin
- Irregular heartbeats
- Shortness of breath
- Dizziness or lightheadedness
- Chest pain
- Cold hands and feet
- Headache
At first anemia can be so mild that it goes unnoticed. But symptoms worsen as anemia worsens.
Pernicious anemia signs and symptoms
Some people do not have symptoms. Symptoms may be mild.
Without enough vitamin B12, your body can’t make enough healthy red blood cells, which causes anemia.
Some of the signs and symptoms of pernicious anemia apply to all types of anemia. Other signs and symptoms are specific to a lack of vitamin B12.
- Desire to eat ice or other non-food things (pica)
- Diarrhea or constipation
- Fatigue, lack of energy, or lightheadedness when standing up or with exertion
- Loss of appetite
- Pale skin
- Problems concentrating
- Shortness of breath, mostly during exercise
- Swollen, red tongue or bleeding gums.
Macrocytic anemia diagnosis
The evaluation of the patient with macrocytosis requires a systemic approach. It should begin with a comprehensive history and physical examination followed by appropriate laboratory studies that include a complete blood count, a peripheral blood smear and reticulocyte count. In some cases, a bone marrow examination may be necessary.
Determining the underlying cause of the macrocytosis can be particularly challenging when thalassemia trait or iron deficiency or other nutritional deficiencies coexist with a vitamin B12 or folate deficiency. In these instances the peripheral blood smear may show a mixed population of microcytic and macrocytic red blood cells with an elevated distribution width. In cases of macrocytosis related to alcoholism the elevated MCV may be due to the direct effect of the alcohol, liver disease and/or folate deficiency.
History and Physical Examination
Evaluation of macrocytosis begins with a complete history and physical examination to search for signs and symptoms related to an acute or chronic underlying illness that may be obvious or occult in nature. Medications such as antimicrobial, chemotherapeutic and anticonvulsant agents can account for a significant number of cases of macrocytosis, with or without anemia (see Table 2 above), emphasizing the importance of taking a careful inventory of the patient’s medications. In some instances, macrocytosis may serve as a surrogate marker indicating the patient’s compliance in taking his/her medications 16. A similar degree of importance applies to the patient’s dietary history and his/her use of alcohol.
Peripheral Blood Smear
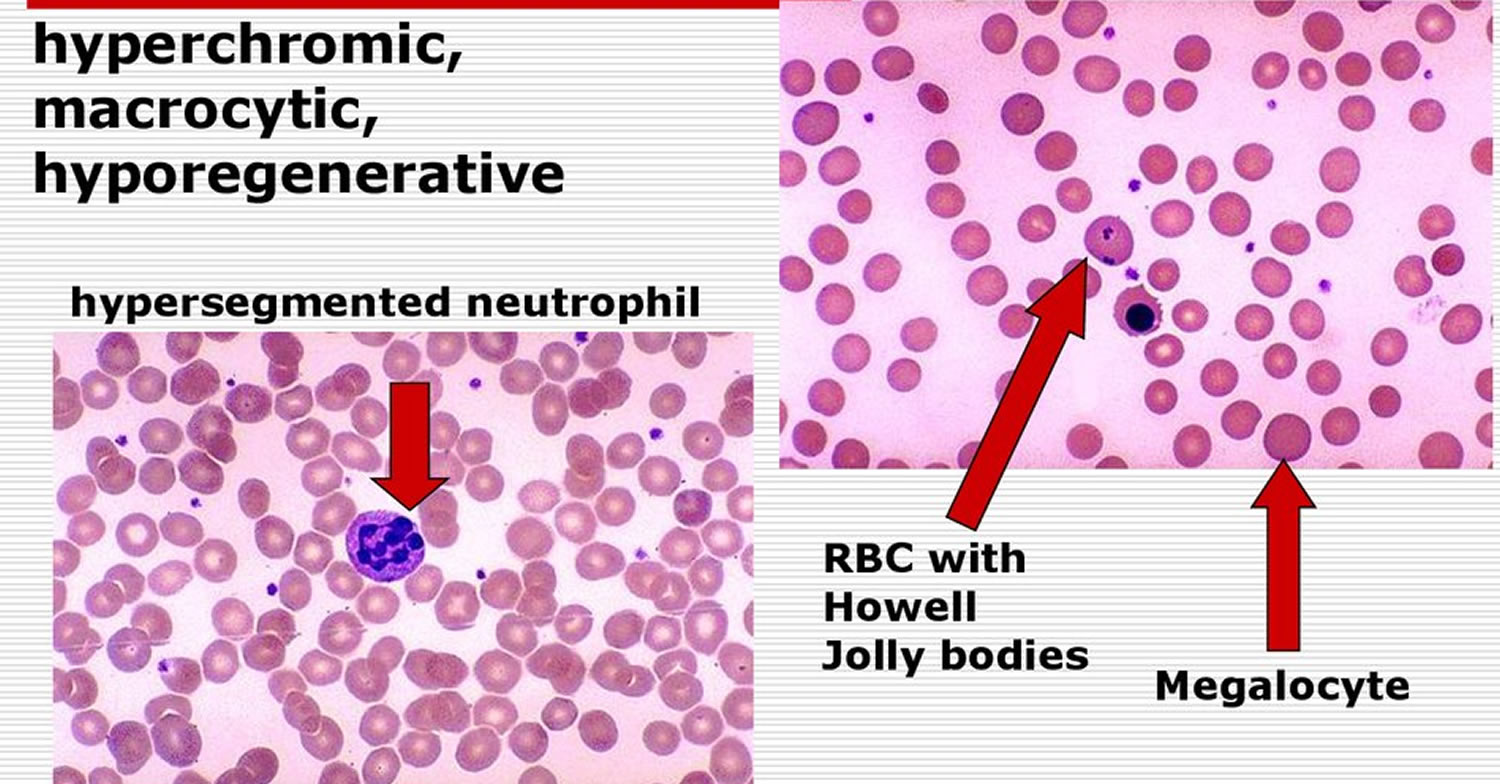
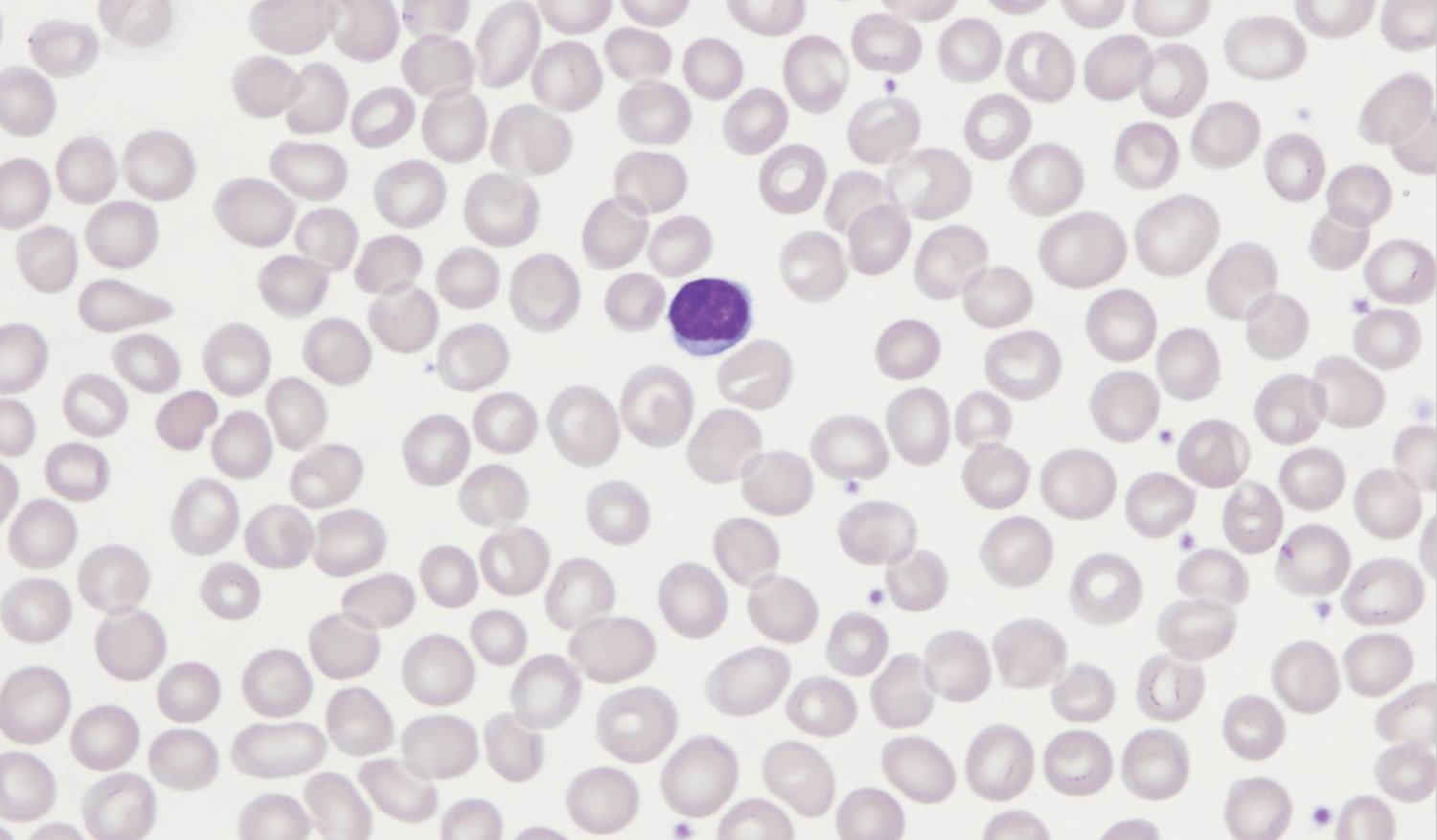
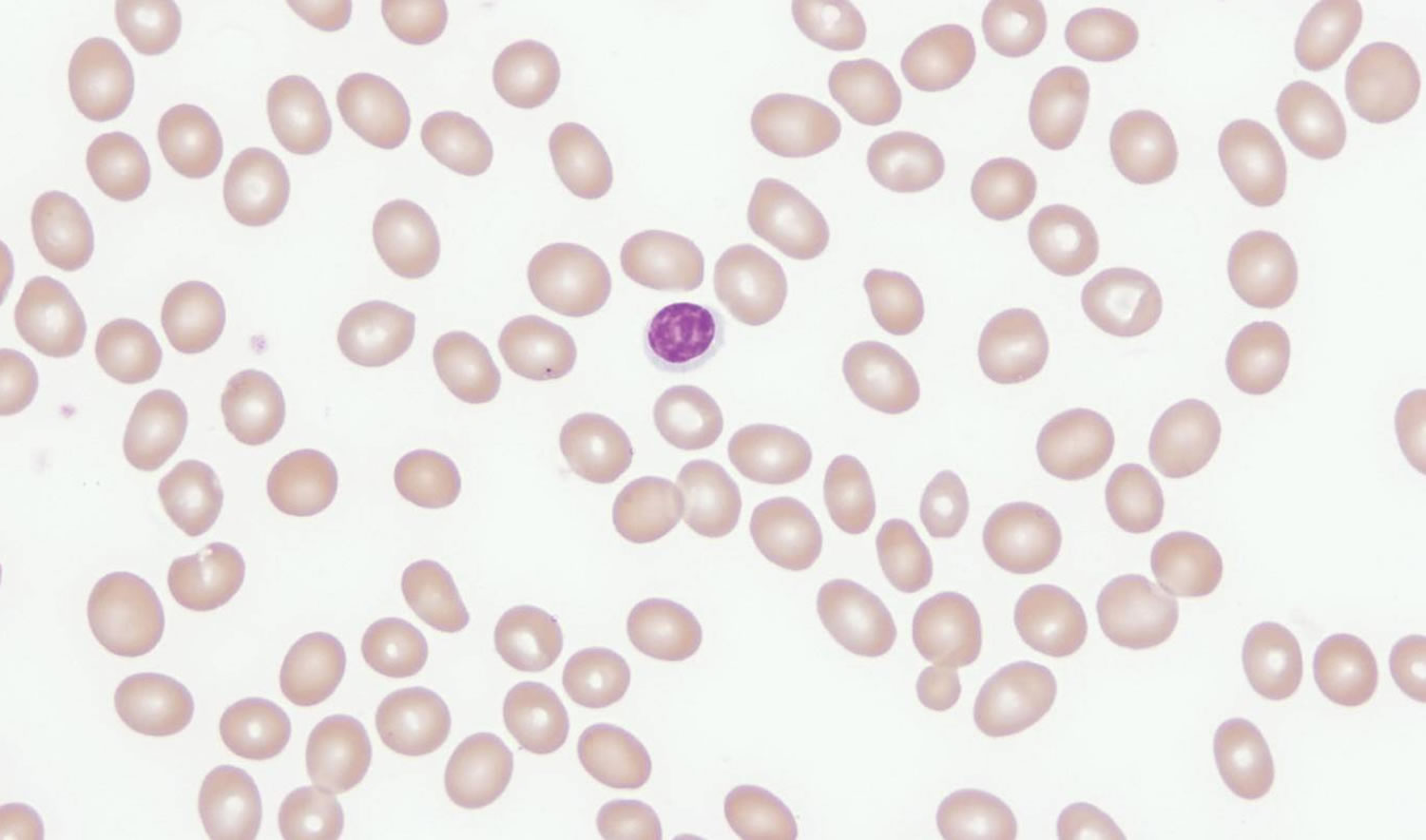
A review of the peripheral smear is imperative in determining the etiology of macrocytosis. The presence of macro-ovalocytes having an MCV >115 fl, anisocytosis, poikilocytosis and hypersegmented neutrophils suggests a megaloblastic disorder associated with a nutritional deficiency, i.e., vitamin B12 or folate deficiency (Figure 2). Round macrocytes are commonly seen in a variety of chronic illnesses, and round target-appearing macrocytes are characteristic of liver disease such as hepatitis, obstructive jaundice, and acute and chronic alcoholism with liver disease (Figure 3). For patients who present with disordered immaturity, hypogranulated or hyposegmented neutrophils, and cytopenias, a bone marrow examination is necessary to rule out or confirm a primary bone marrow disorder such as a myelodysplastic syndrome or leukemia.
Figure 2. Macrocytic anemia peripheral blood smear of a patient with vitamin B12 deficiency showing marked macro-ovalocytosis (MCV 134 fl)
Figure 3. Macrocytic anemia peripheral blood smear of a patient with liver disease showing predominantly round macrocytes (MCV = 114 fl), some targeted
[Source 14]Reticulocyte Count
A reticulocyte count should be obtained if there is evidence of hemolysis on the peripheral smear, i.e., increased polychromasia, nucleated red blood cells, spherocytes or schistocytes 17. The presence of increased polychromasia of the macrocytes on the peripheral smear and a reticulocyte count of >10% should raise suspicion of hemolysis or an acute bleed. These large polychromatophilic erythrocytes noted on the peripheral smear represent reticulocytes, immature red blood cells that are larger than mature red blood cells, and are indicative of increased erythropoiesis or red blood cell production and, if present in increased number, can raise the MCV. Additionally, the reticulocyte maturation parameters performed on the peripheral blood may also be helpful to differentiate megaloblastic from nonmegaloblastic causes of the macrocytosis 18. An elevated reticulocyte maturation value is more suggestive of a megaloblastic rather than a non-megaloblastic anemia.
Bone Marrow Examination
Macrocytosis associated with a megaloblastic marrow is usually accompanied by anemia due to ineffective erythropoiesis. The bone marrow is hypercellular, showing evidence of abnormal proliferation and maturation of multiple myeloid cell lines. These abnormalities are most evident in the erythroid precursors with large megaloblastic erythroblasts present in increased numbers throughout the marrow. Similar morphologic abnormalities can be seen in the other myeloid elements, e.g., large or giant metamyelocytes and other granulocytic precursors. This ineffective erythropoiesis is accompanied by intramedullary hemolysis causing an elevated lactate dehydrogenase and indirect bilirubin in the serum 19. However, the reticulocyte count is low due to the abnormal maturation process. More severe degrees of abnormal proliferation and maturation are seen with myelodysplasia and myeloid leukemias. It is imperative that a hematologist or hematopathologist examine the marrow in order to appreciate these important, subtle, hematopoietic abnormalities. Patients with macrocytosis who are not anemic and have no other abnormalities noted on the peripheral blood smear do not usually need a bone marrow examination.
Investigation of Vitamin B12 and Folate Deficiencies
Macrocytosis is the earliest abnormality seen in complete blood counts of patients with folate or vitamin B12 deficiency. In patients with elevated MCV values, laboratory tests for vitamin B12 and folate deficiencies are routinely ordered by physicians, although these tests are limited by their low sensitivity and specificity. Like the red blood cell MCV, the lower limits of normal for vitamin B12 levels are not well defined 20.
Serum B12 Levels
Vitamin B12 levels may be reported as normal or elevated in myeloproliferative disorders, liver disease, congenital transcobalamin II deficiency, intestinal bacterial overgrowth and antecedent administration of vitamin B12 21. Moreover, there are reports of falsely low vitamin B12 levels with folate deficiency, pregnancy, use of oral contraceptives, congenital deficiency of serum haptocorrins and multiple myeloma 21. The prevalence of vitamin B12 deficiency among the elderly ranges from 1.5% to 4.6% 22 and was reported to be as high as 15% in the population over the age of 60 years 23. The deficiency in many cases is associated with gastric achlorhydria, resulting in decreased synthesis and availability of intrinsic factor, a necessary binding protein that facilitates vitamin B12 absorption in the ileum. This constellation of events eventually leads to pernicious anemia and requires prompt intervention with exogenous vitamin B12 preparations. The diagnosis of pernicious anemia can be confirmed by identifying and measuring intrinsic antibody levels in the serum. Parietal cell antibodies, although not specific, are also commonly present. However, these tests are expensive and not always available to the practicing clinician.
Serum Folate Levels
Folic acid deficiency in the United States is extremely rare because of the fortification of foods 24. Although tissue stores may be normal, serum folate levels can decrease within a few days of dietary folate restriction 21. Thus, patients should fast prior to testing for serum folate levels, as serum folate levels increase with feeding. Because of the high concentration of folate within the red blood cell, mild degrees of hemolysis can falsely elevate serum folate levels 21. Pregnancy, certain anticonvulsant drugs, and alcohol intake may also cause a decrease in folate serum levels despite adequate tissue stores. Serum folate levels tend to be increased in patients with vitamin B12 deficiency, presumably because of impairment of the methionine synthase pathway and accumulation of methyltetrahydrofolate, the principal form of folate in the serum 25.
Red blood cell Folate
Because of the limitations of measuring serum folate, RBC folate levels have been advocated as a more reliable source of measuring tissue stores of folate. RBC folate levels remain constant throughout the lifespan of the cell and are not affected by short-term dietary changes that can alter serum levels. However, assays for measuring RBC folate levels have also been fraught with unreliability.3,31–33 It should be noted that low RBC folate levels have been reported with alcohol use, pregnancy and anticonvulsant medications.34 Another important cause of low RBC folate levels is vitamin B12 deficiency.29,30,35 It is estimated that approximately 60% of patients with pernicious anemia have low RBC folate levels, presumably because vitamin B12 is necessary for normal transfer of methyltetrahydrofolate from plasma to RBCs.28,36
Methylmalonic Acid (MMA) and Homocysteine Serum Concentrations
Cobalamin and folate are cofactors in several important metabolic pathways in the cell. The hydroxylated form of cobalamin plays an important role in the metabolism of homocysteine and methylmalonic acid. The conversion of homocysteine to methionine requires both vitamin B12 and folate as cofactors. However, the metabolism of L-methylmalonyl CoA to succinyl CoA, an enzymatic pathway involved in oxidative phosphorylation reactions within the cell, only requires vitamin B12. These metabolites provide early information regarding the cellular state of vitamin B12 and folate and can be used to distinguish folate from vitamin B12 deficiency, since most patients with folate deficiency have normal methylmalonic acid (MMA) or mildly elevated levels 26. It should be kept in mind that nearly 50% of those with elevation of these metabolites will have normal serum vitamin B12 levels 27. This emphasizes the low sensitivity of using vitamin B12 levels, particularly in the presence of other signs or symptoms.
Previous studies on large groups of cobalamine and/or folate deficient patients have shown the ability of differentiating cobalamin deficiency from folate deficiency by measuring serum methylmalonic acid (MMA) and homocysteine levels. Both of these metabolites are elevated in cobalamin deficiency, with anemic cobalamin deficient patients showing marked elevations. However, methylmalonic acid (MMA) is more sensitive for identifying non-anemic cobalamin deficiency patients than homocysteine. In folate deficiency patients, serum homocysteine levels are markedly increased, while serum methylmalonic acid (MMA) levels are not elevated 28. Therefore, measuring the serum levels of these two metabolites not only helps in differentiating cobalamine (vitamin B12) deficiency from folate deficiency, but also provides a reliable degree of both sensitivity and specificity in diagnosing these important deficiency states 28. An important limitation that must be considered when measuring methylmalonic acid (MMA) is the presence of renal insufficiency, i.e., an elevated serum creatinine, and hypovolemia. Serum methylmalonic acid (MMA) will be elevated in patients with underlying renal dysfunction, decreasing its specificity and sensitivity in identifying patients with cobalamin deficiency 29. Similarly, hereditary homocysteinemia is a condition in which the serum homocysteine levels are elevated. Measurements of methylmalonic acid (MMA) levels are recommended when initial vitamin B12 and/or homocysteine levels are abnormal.
Macrocytic anemia treatment
Traditionally, patients with vitamin B12 deficiency from any cause have received cyanocobalamin intramuscularly or subcutaneously 1000 μg/week for 1 month and monthly, thereafter. This time-honored method remains an acceptable form of treatment for all causes of vitamin B12 deficiency, particularly when cognitive impairment or neurologic disease is present. Alternatively, hydroxocobalamin given in the same dose every 1–3 months intramuscularly is also effective therapy. This form of cobalamin remains in the tissues longer than the cyanocobalamin forms and can, therefore, be given less frequently 30. In cases of deficiency due to inadequate intake, food-cobalamin malabsorption and pernicious anemia, oral cyanocobalamin administered at 1000–2000 μg/day for 1 month, followed by 125–500 μg/day is recommended and considered a safe and effective method of treatment 31. Other oral administration regimens have demonstrated efficacy and have proven to be equally as effective as intramuscular administration 32. In some cases, nasal or sublingual cyanocobalamin may also be useful in replenishing vitamin B12 stores 33. Because of the inherent unreliability of measuring either serum or red blood cell folate, it is recommended that patients receiving treatment for vitamin B12 deficiency receive empiric folate supplementation of 400 μg/day to 1 mg/day 34. Maintenance therapy should continue until the underlying cause of the deficiency is corrected.
In folate deficiency, the serum folate level is very sensitive to dietary folate intake and responds well to short-term treatment. Long-term treatment is not warranted except with chronic conditions such as malnutrition, exfoliative dermatitis or hemolysis 35. A complete blood cell count 10–14 days after starting the treatment for vitamin B12 or folate deficiency should reveal a rise in hemoglobin and a decrease in MCV. A full hematologic response should occur within 8 weeks. During treatment, further monitoring of the complete blood cell count or measuring vitamin B12 and folate levels or their metabolites is not necessary 35. In patients taking long-term treatment for vitamin B12 deficiency, an annual complete blood cell count may be a reasonable consideration to monitor therapy.
- Diagnosis of cobalamin deficiency: the old and the new. Chanarin I, Metz J. Br J Haematol. 1997 Jun; 97(4):695-700. https://www.ncbi.nlm.nih.gov/pubmed/9217166/[↩]
- Kasper DL, Braunwald E, Fauci A, Hauser S, Longo D, Jameson JL. Harrison’s principles of internal medicine. 16th ed. New York: McGraw-Hill Medical Publishing Division; 2005.[↩]
- Davidson RJ, Hamilton PJ. High mean red cell volume: its incidence and significance in routine haematology. J Clin Pathol 1978;31:493–498. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1145310/pdf/jclinpath00441-0099.pdf[↩]
- A practical approach to the differential diagnosis and evaluation of the adult patient with macrocytic anemia. Colon-Otero G, Menke D, Hook CC. Med Clin North Am. 1992 May; 76(3):581-97. https://www.ncbi.nlm.nih.gov/pubmed/1578958/[↩][↩]
- Hoffbrand V, Provan D. ABC of clinical haematology. Macrocytic anaemias. BMJ 1997;314:430–433. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2125890/pdf/9040391.pdf[↩][↩]
- Sechi LA, De Carli S, Catena C, Zingaro L, Bartoli E. Benign familial macrocytosis. Clin Lab Haematol 1996;18:41–43. https://www.ncbi.nlm.nih.gov/pubmed/9118603[↩]
- Savage DG, Ogundipe A, Allen RH, Stabler SP, Lindenbaum J. Etiology and diagnostic evaluation of macrocytosis. Am J Med Sci 2000;319:343–52. https://www.ncbi.nlm.nih.gov/pubmed/10875288[↩][↩]
- Seppä K, Heinilä K, Sillanaukee P, Saarni M. Evaluation of macrocytosis by general practitioners. J Stud Alcohol 1996;57:97–100. https://www.ncbi.nlm.nih.gov/pubmed/8747507[↩]
- Mahmoud MY, Lugon M, Anderson CC. Unexplained macrocytosis in elderly patient. Age Ageing 1996;25:310–2. https://www.ncbi.nlm.nih.gov/pubmed/8831877[↩]
- Komine M. Int J Hematol 2000;71(suppl 1):8.[↩]
- Shipton MJ, Thachil J. Vitamin B12 deficiency‐A21st century perspective. Clin Med (Lond). 2015;15:145–50. https://www.ncbi.nlm.nih.gov/pubmed/25824066[↩]
- Kaferle J, Strzoda CE. Evaluation of macrocytosis. Am Fam Physician 2009;79:203–8. https://www.aafp.org/afp/2009/0201/p203.html[↩]
- Savage DG, Ogundipe A, Allen RH, Stabler SP, Lindenbaum J. Etiology and diagnostic evaluation of macrocytosis. Am J Med Sci 2000;319:343–352. https://www.ncbi.nlm.nih.gov/pubmed/10875288[↩]
- Aslinia F, Mazza JJ, Yale SH. Megaloblastic Anemia and Other Causes of Macrocytosis. Clinical Medicine and Research. 2006;4(3):236-241. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1570488/[↩][↩][↩][↩]
- Nagao T, Hirokawa M. Diagnosis and treatment of macrocytic anemias in adults. Journal of General and Family Medicine. 2017;18(5):200-204. doi:10.1002/jgf2.31. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5689413/[↩]
- Thomas CW Jr, Lowry PW, Franklin CL, Weaver AL, Myhre GM, Mays DC, Tremaine WJ, Lipsky JJ, Sandborn WJ. Erythrocyte mean corpuscular volume as a surrogate marker for 6-thioguanine nucleotide concentration monitoring in patients with inflammatory bowel disease treated with azathioprine or 6-mercaptopurine. Inflamm Bowel Dis 2003;9:237–245. https://www.ncbi.nlm.nih.gov/pubmed/12902847[↩]
- Bain BJ. Diagnosis from the blood smear. N Engl J Med 2005;353:498–507. http://www.nejm.org/doi/full/10.1056/NEJMra043442[↩]
- Torres Gomez A, Casano J, Sanchez J, Madrigal E, Blanco F, Alvarez MA. Utility of reticulocyte maturation parameters in the differential diagnosis of macrocytic anemias. Clin Lab Haematol 2003;25:283–288. https://www.ncbi.nlm.nih.gov/pubmed/12974717[↩]
- Hoffman R, Benz EJ Jr, Shattil SJ, Furie B, Cohen HJ, Silberstein LE, McGlave P, eds. Hematology: basic principles and practice. 4th ed. New York, NY: Churchill Livingston; 2005.[↩]
- Ward PC. Modern approaches to the investigation of vitamin B12 deficiency. Clin Lab Med 2002;22:435–445.[↩]
- Snow CF. Laboratory diagnosis of vitamin B12 and folate deficiency: a guide for the primary care physician. Arch Intern Med 1999;159:1289–1298. https://jamanetwork.com/journals/jamainternalmedicine/fullarticle/485067[↩][↩][↩][↩]
- Andres E, Loukili NH, Noel E, Kaltenbach G, Abdelgheni MB, Perrin AE, Noblet-Dick M, Maloisel F, Schlienger JL, Blickle JF. Vitamin B12 (cobalamin) deficiency in elderly patients. CMAJ 2004;3:251–259. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC490077/[↩]
- Oh RC, Brown DL. Vitamin B12 deficiency. Am Fam Physician 2003;67:979–986. https://www.ncbi.nlm.nih.gov/pubmed/12643357[↩]
- Ashraf MJ, Goyal M, Hinchey K, Cook JR. Clinical utility of folic acid testing for anemia and dementia screen. J Gen Intern Med 2004;19(s1):130.[↩]
- Handin RI, Lux SE, Stossel TP. Blood: principles and practice of hematology. 1st ed. Philadelphia, PA: Lippincott Williams & Wilkins; 1995; 1421.[↩]
- Lindenbaum J, Savage DG, Stabler SP, Allen RH. Diagnosis of cobalamine deficiency: II. Relative sensitivities of serum cobalamine, methylmalonic acid, and total homocysteine concentrations. Am J Hematol 1990;34:99–107. https://www.ncbi.nlm.nih.gov/pubmed/2339684[↩]
- Chanarin I, Metz J. Diagnosis of cobalamin deficiency: the old and new. Br J Haematol 1997;97:695–700. https://www.ncbi.nlm.nih.gov/pubmed/9217166[↩]
- Savage DG, Lindenbaum J, Stabler SP, Allen RH. Sensitivity of serum methylmalonic acid and total homocysteine determinations for diagnosing cobalamin and folate deficiencies. Am J Med 1994;96:239–246. https://www.ncbi.nlm.nih.gov/pubmed/8154512[↩][↩]
- Ward PC. Modern approaches to the investigation of vitamin B12 deficiency. Clin Lab Med 2002;22:435–445. https://www.ncbi.nlm.nih.gov/pubmed/12134470[↩]
- Shojania AM. Protein synthesis-megaloblastic disorders. In: Gross S, Roath S, eds. Hematology. A problem-oriented approach. Baltimore: Williams & Wilkins. 1996. 25–54.[↩]
- Nyholm E, Turpin P, Swain D, Cunningham B, Daly S, Nightingale P, Fegan C. Oral vitamin B12 can change our practice. Postgrad Med J 2003;79:218–220. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1742688/pdf/v079p00218.pdf[↩]
- Bolaman Z, Kadikoylu G, Yukselen V, Yavasoglu I, Barutca S, Senturk T. Oral versus intramuscular cobalamin treatment in megaloblastic anemia: a single-center, prospective, randomized, open-label study. Clin Ther 2003;25:3124–3134. https://www.ncbi.nlm.nih.gov/pubmed/14749150[↩]
- Sharabi A, Cohen E, Sulkes J, Garty M. Replacement therapy for vitamin B12 deficiency: comparison between the sublingual and oral route. Br J Clin Pharmacol 2003;56:635–638. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1884303/[↩]
- Latif T, Hsi ED, Rybicki LA, Adelstein DJ. Is there a role for folate determinations in current clinical practice in the USA? Clin Lab Haematol 2004;26:379–383. https://www.ncbi.nlm.nih.gov/pubmed/15595994[↩]
- Smellie WS, Wilson D, McNulty CA, Galloway MJ, Spickett GA, Finnigan DI, Bareford DA, Greig MA, Richards J. Best practice in primary care pathology: review 1. J Clin Pathol 2005;58:1016–1024. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1770731/[↩][↩]