Contents
What is melatonin
Melatonin is a natural hormone produced by the pineal gland that plays a role in sleep. Melatonin production and release in the brain by the pineal gland is related to time of day. Melatonin secretion increases in darkness – rising in the evening and decreases during exposure to light – falling in the morning. Light at night blocks melatonin production. Melatonin has multiple effects and is implicated in the regulation of sleep-wake cycle, mood, and reproduction. Melatonin acts by engagement of melatonin receptors (MT-1 and MT-2) causing a reduction of body temperature and alternation of brain monoamine levels, which can induce somnolence (sleepiness or drowsiness) that helps promote sleep. Melatonin is also an effective antioxidant and may have reproductive effects and modulate immune reactions. Melatonin has been proposed as therapy of sleep disturbances including insomnia and jet lag, but it has not been approved for this indication in the United States.
Melatonin is a small molecular weight amine (N-actyl-5-methoxytryptamine) synthesized in the pineal gland from serotonin (5-HT). Melatonin is released into the circulation in a circadian pattern, the plasma levels being low during the day and high at night. In persons with normal sleep-wake patterns, peak melatonin values are present between 2:00 am and 4:00 am and drop before light onset in the morning.
Melatonin is available over-the-counter as melatonin supplement in multiple generic forms and formulations. Melatonin is also found in many herbal and nutrition supplement mixtures. Most melatonin supplements are made in a lab. A common dose is 10 mg once daily, but less is better, take 1 to 3 milligrams two hours before bedtime or taken orally one to three hours before sleep time.
Melatonin dietary supplements have been studied for sleep disorders, such as jet lag, disruptions of the body’s internal “clock,” insomnia, and problems with sleep among people who work night shifts. However, important questions remain about its usefulness, how much to take, when to take it, and its long-term safety. Melatonin has also been studied for dementia symptoms.
In several clinical trials, melatonin was found to be well tolerated and not associated with serum enzyme elevations or evidence of liver injury. Despite wide scale use, melatonin has not been convincingly linked to instances of clinically apparent liver injury 1. Melatonin side effects are few but may include daytime somnolence and headache.
Melatonin synthesis and secretion
Melatonin is the main hormone secreted by the pineal gland. Extrapineal sources of melatonin were reported in the retina, bone marrow cells, platelets, skin, lymphocytes, Harderian gland, cerebellum, and especially in the gastrointestinal tract of vertebrate species 2. Indeed, melatonin is present but can also be synthesized in the enterochromaffin cells; the release of gastrointestinal melatonin into the circulation seems to follow the periodicity of food intake, particularly tryptophan intake 3. It is noteworthy that the concentration of melatonin in the gastrointestinal tract surpasses blood levels by 10-100 times and there is at least 400 times more melatonin in the gastrointestinal tract than in the pineal gland 3. Melatonin in the gastrointestinal tract of newborn and infant mammals is of maternal origin given that melatonin penetrates easily the placenta and is after secreted into the mother’s milk 4. It has even been suggested that melatonin is involved in the production of mekonium 3. Melatonin in human breast milk follows a circadian rhythm in both preterm and term milk, with high levels during the night and undetectable levels during the day 5. No correlation was found between gestational age and concentration of melatonin. It is noteworthy that bottle milk composition does not contain melatonin in powder formula. Also, human colostrum, during the first 4 or 5 days after birth, contains immune – competent cells (colostral mononuclear cells) which are able to synthesize melatonin in an autocrine manner 6.
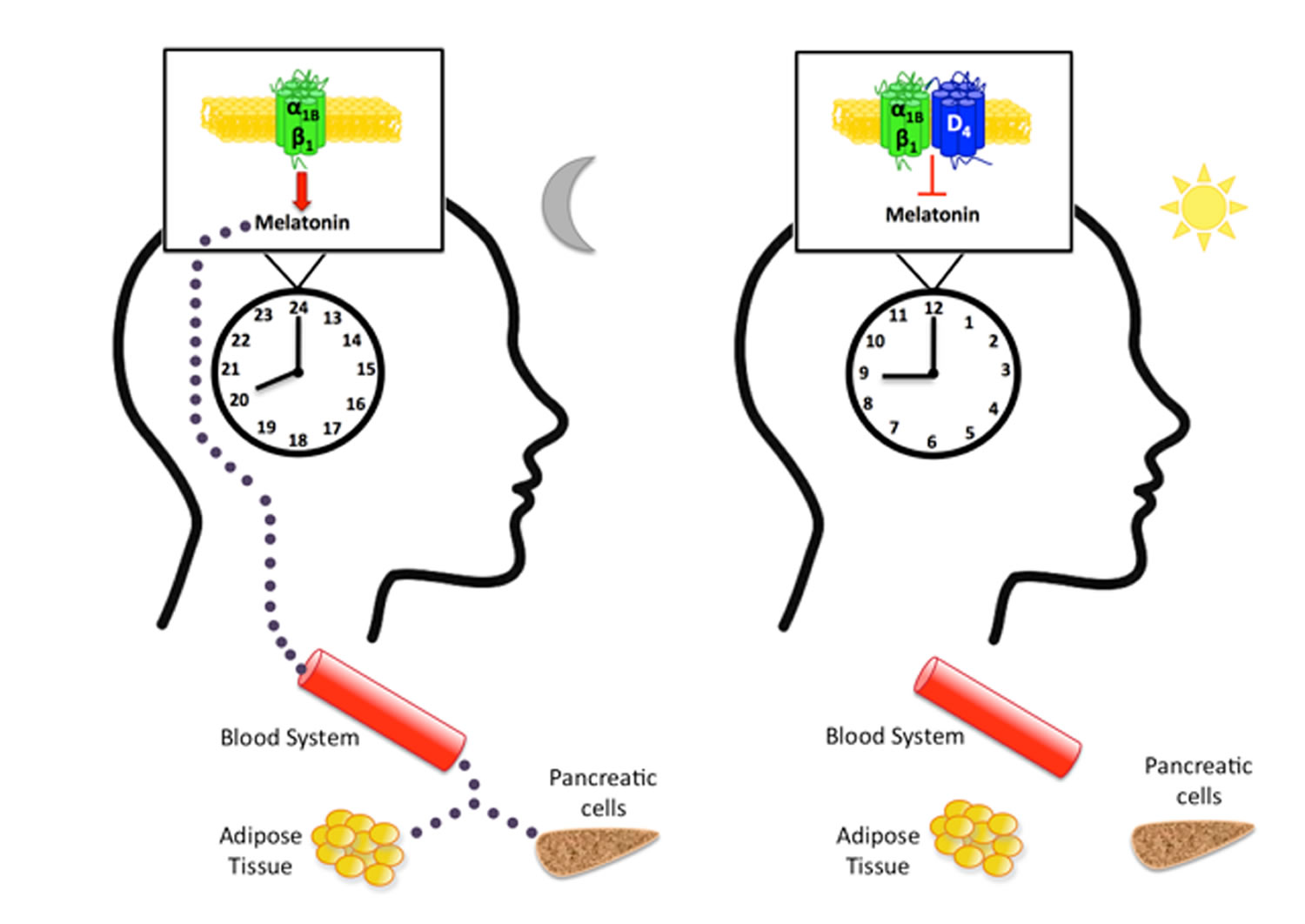
Melatonin is mainly synthesized by the pinealocytes from the amino acid tryptophan, which is hydroxylated (by the tryptophan-5-hydroxylase) in 5-hydroxytryptophan, then decarboxylated (by the 5-hydroxytryptophan decarboxylase) in serotonin (see Figure 3). Two enzymes, found mainly in the pineal gland, transform serotonin to melatonin 7: serotonin is first acetylated to form N-acetylserotonin by arylalkylamine-N-acetyltransferase (AA-NAT, also called “Timezyme”, is the rate-limiting enzyme for melatonin synthesis), and then N-acetylserotonin is methylated by acetylserotonin-O-methyltransferase (ASMT, also called hydroxyindole-O- methyltransferase or HIOMT) to form melatonin (Figure 2). Both AA-NAT and ASMT activities are controlled by noradrenergic and neuropeptidergic projections to the pineal gland 8. Norepinephrine, also called noradrenaline, activates adenylate cyclase which in turn promotes the melatonin biosynthesis enzymes, especially AA-NAT 9. Once synthesized, melatonin is quickly released into the systemic circulation to reach central and peripheral target tissues.
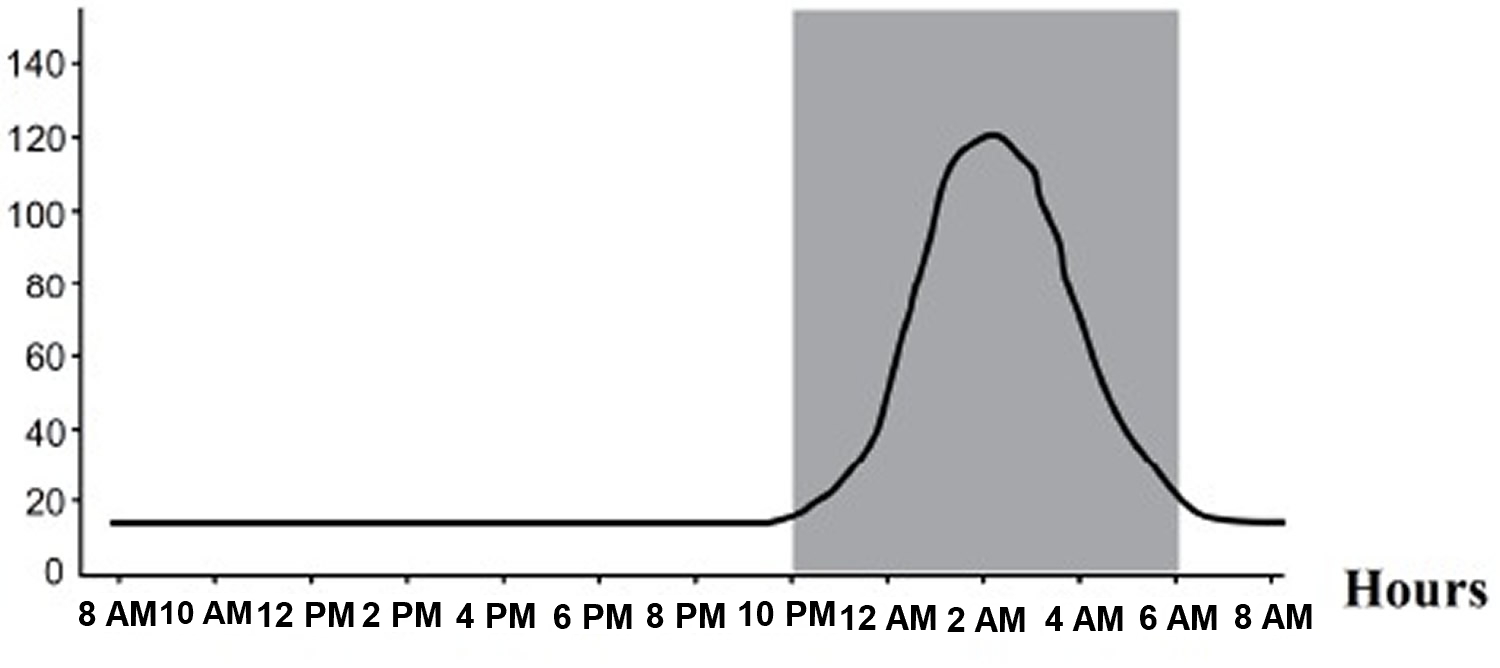
Melatonin synthesis and secretion is enhanced by darkness and inhibited by light (Figure 1) 10. Luminous information is transmitted from the retina to pineal gland through the suprachiasmatic nucleus (SCN) of the hypothalamus. In humans, its secretion starts soon after sundown, reaches a peak in the middle of the night (between 2 and 4 in the morning) and decreases gradually during the second half of the night 11. Nearly 80% of the melatonin is synthesized at night, with serum concentrations varying between 80 and 120 pg/ml. During daylight hours, serum concentrations are low (10-20 pg/ml) 12.
Serum concentrations of melatonin vary considerably with age, and infants secrete very low levels of melatonin before 3 months of age. Melatonin secretion increases and becomes circadian along with child development: Sadeh 13 reported an association between melatonin secretion and organization of sleep-wake rhythm from 6 months of age. However, more recent studies suggest that melatonin rhythm is set around 3 months of age in typical development, at the same time that infants begin to have more regular sleep–wake cycles associated with nighttime sleep lasting 6-8 h 14. In 3-years-old children, a stabilization of the sleep-wake rhythm is observed, which corresponds to a regular melatonin secretion rhythm [26]. Nocturnal concentration peaks are the highest between the 4th and 7th years of age 15 and then decline progressively 16.
After intravenous or oral administration, melatonin is quickly metabolized, mainly in the liver and secondarily in the kidney. However, after intravenous administration, the hepatic bio-degradation is less important due to the absence of hepatic first pass. It undergoes hydroxylation to 6-hydroxymelatonin by the action of the cytochrome P450 enzyme CYP1A2, followed by conjugation with sulfuric acid (90%) or glucuronic acid (10%) and is excreted in the urine. About 5% of serum melatonin is excreted unmetabolized also in urine. The principal metabolite, the 6-sulfatoxy-melatonin, is inactive, and its urinary excretion reflects melatonin plasma concentrations 17. Plasma levels can be also measured directly or indirectly assessed through salivary measures. A reverse relation between bioavailability of melatonin and the 6-sulfatoxy-melatonin concentrations area under the curve has been shown, the low bioavailability being explained by an important hepatic first pass 18.
Figure 1. Melatonin plasma concentrations – Circadian profile (in grey is represented the period of darkness)
Figure 2. Melatonin chemical structure
Figure 3. Melatonin synthesis
Note: AA-NAT = arylalkylamine-N-acetyl-transferase; ASMT = acetylserotonin-O-methyltransferase
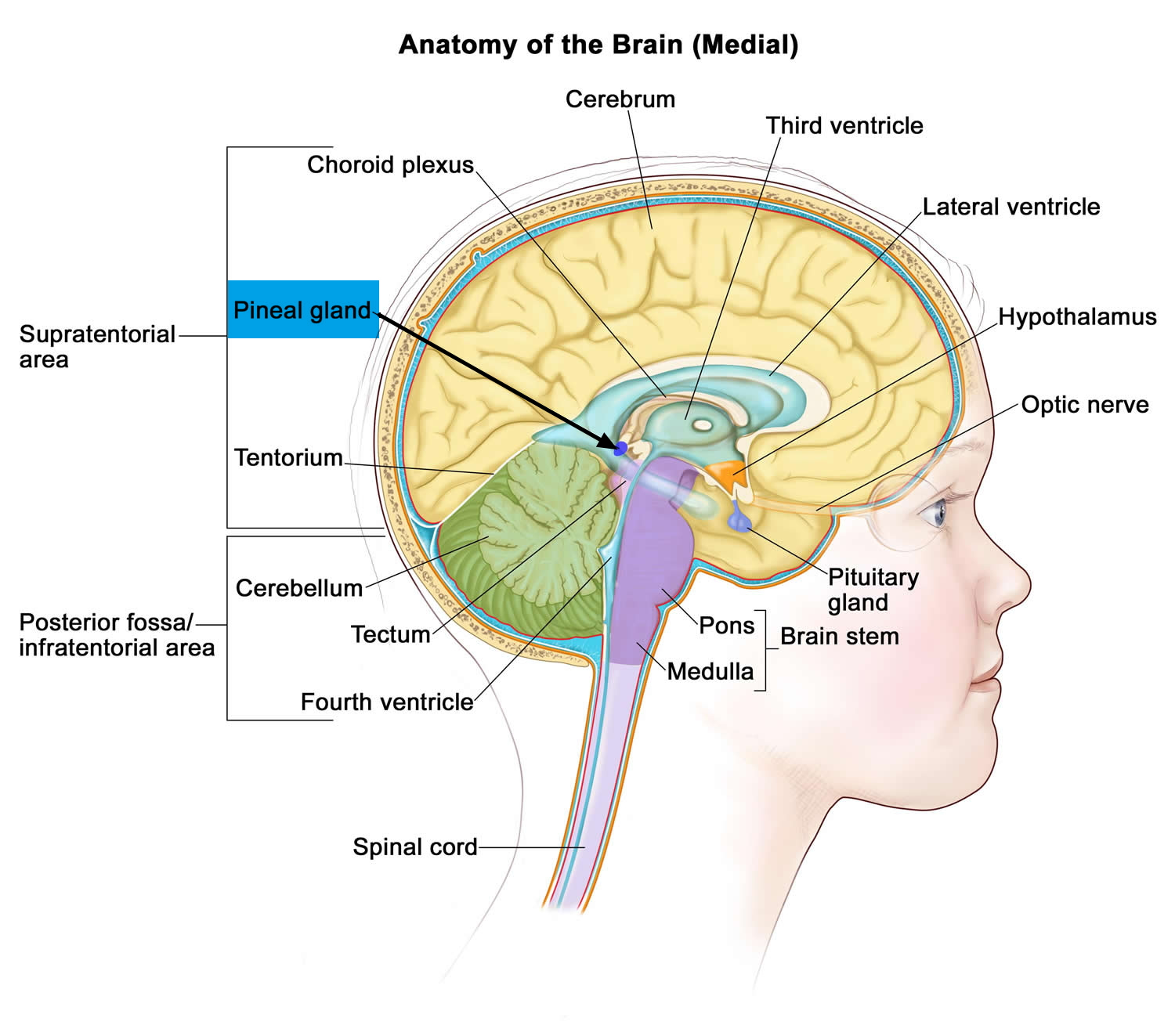
[Source 19]Figure 4. Pineal gland
Note: The pineal gland is commonly located along the midline above the superior colliculi and inferior to the splenium of the corpus callosum. It is attached to the superior aspect of the posterior border of the third ventricle.
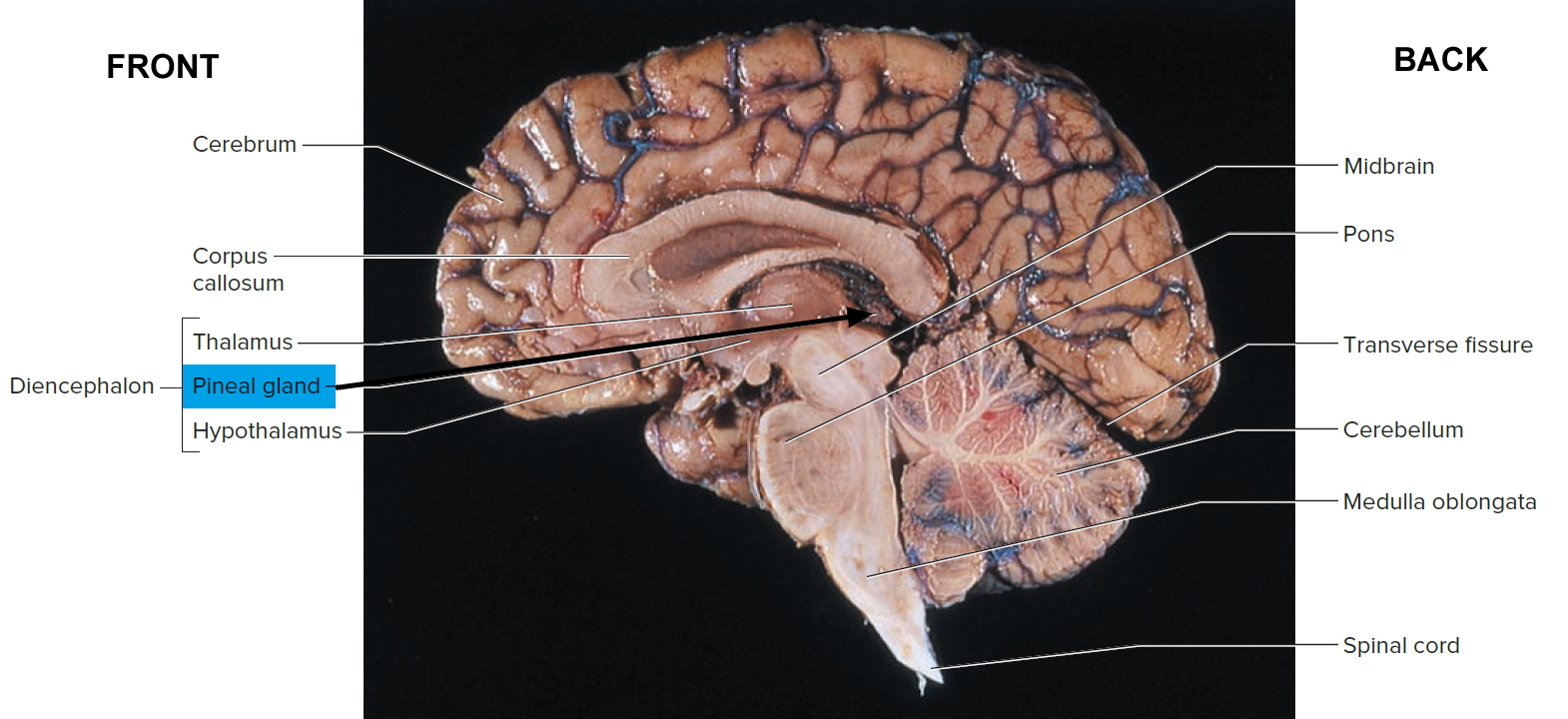
Figure 5. Pineal gland
Physiological effects of melatonin
Melatonin regulates circadian rhythms such as the sleep-wake rhythm, neuroendocrine rhythms or body temperature cycles through its action on melatonin receptors (MT-1 and MT-2) 20. Ingestion of melatonin induces fatigue, sleepiness and a diminution of sleep latency 21. Disturbed circadian rhythms are associated with sleep disorders and impaired health 22. For example, children with multiple developmental, neuro-psychiatric and health difficulties often show melatonin deficiency 23. When circadian rhythms are restored, behavior, mood, development, intellectual function, health, and even seizure control may improve 22. It should be noted that according to several studies, circadian rhythms are important for typical (normal) neurodevelopment and their absence suppresses neurogenesis in animal models 24.
Finally, melatonin may be involved in early fetal development, with direct effects on placenta, glial and neuronal development, and could play an ontogenic role in the establishment of diurnal rhythms and synchronization of the fetal biological clock 25. Iwasaki et al. 25 investigated the expression of the two enzymes involved in the conversion of serotonin to melatonin (AA-NAT and ASMT) (see Figure 3 above) and found that transcripts of these enzymes were present in the first-trimester human placenta. Moreover, they found also that melatonin significantly potentiated hCG (human chorionic gonadotropin) secretion at optimal concentrations on cultured human trophoblast cells. These results suggest that melatonin regulates in a paracrine/autocrine way human placental function with a potential role in human reproduction. Test tube studies have shown that neural stem/progenitor cells express melatonin MT1 receptors and melatonin induces glial cell-line derived neurotrophic factor (GDNF) expression in neural stem cells, suggesting an early role for melatonin in central nervous system development. Indeed, as indicated previously, melatonin of maternal origin crosses the placenta and can therefore influence fetal development. Studies in humans have repeatedly confirmed that the cycle of melatonin in maternal blood occurs also in fetal circulation 26. The maturation and synchronization of the fetal circadian system have not been thoroughly studied. However, studies in nonhuman primate fetus have shown that maternal melatonin stimulates growth of the primate fetal adrenal gland and entrains fetal circadian rhythms, including suprachiasmatic nucleus (SCN) rhythms 27. Furthermore, in mice, the suppression of melatonin rhythm by maternal exposure to constant light changes the rhythmic expression in fetal clock genes; these changes are reversed when melatonin is injected daily to the mother 28. These results document that the fetal clock is imprinted by melatonin, which under normal circumstances is of maternal origin. In addition, some studies in humans and nonhuman primates show 24h rhythms in fetal heart rate and respiratory movements during the latter half of pregnancy. Whether the circadian system of the human fetus, particularly in late pregnancy, is under the influence of maternal suprachiasmatic nucleus (SCN) remains to be better ascertained 29.
Besides the well-known effects of melatonin on the regulation of sleep-wake rhythms, melatonin is considered as an endogenous synchronizer and a chronobiotic molecule, i.e. a substance that reinforces oscillations or adjusts the timing of the central biological clock located in the suprachiasmatic nuclei of the hypothalamus to stabilize bodily rhythms 30. Furthermore, Pevet and Challet 31 view melatonin as both the master clock output and internal time-giver in the complex circadian clocks network: as a major hormonal output, melatonin distributes, through its daily rhythm of secretion, temporal cues to the numerous tissue targets with melatonin receptors, driving circadian rhythms in some tissue structures such as the adenohypophysis or synchronizing peripheral oscillators such as the fetal adrenal gland but also many other peripheral tissues (pancreas, liver, kidney, heart, lung, fat, gut, etc.). Circadian rhythms, and more precisely the circadian clocks network, allow temporal organization of biological functions in relation to periodic environmental changes and therefore reflect adaptation to the environment. Thus, the sleep–wake rhythm associated with biological circadian rhythms can be seen as an adaptation to the day–night cycle. Moreover, the synchronization by melatonin of peripheral oscillators reflects adaptation of the individual to his/her internal and external environment (for example, the synchronized effects of melatonin on cortisol and insulin secretion allow the individual to be fully awake at 8am and able to start the day by eating and getting some energy from food intake). Given the major synchronizing effects of melatonin on central and peripheral oscillators, measures of melatonin are considered the best peripheral indices of human circadian timing 32.
Futhermore, melatonin is involved in blood pressure and autonomic cardiovascular regulation, immune system regulation but also various physiological functions such as retinal functions, detoxification of free radicals and antioxidant actions through its action on melatonin MT3 receptors protecting the brain from oxidative stress 33. A through its action on MT3 receptors specific section is developed below on melatonin and brain protection. The antioxidant actions of melatonin protect also the gastrointestinal tract from ulcerations by reducing secretion of hydrochloric acid and the oxidative effects of bile acids on the intestinal epithelium, and by increasing duodenal mucosal secretion of bicarbonate through its action on MT2 receptors (this alkaline secretion is an important mechanism for duodenal protection against gastric acid); melatonin prevents also ulcerations of gastrointestinal tract by increasing microcirculation and fostering epithelial regeneration 34. Concerning the role of melatonin in immune regulation, melatonin has direct immuno-enhancement effects in animals and humans 35. Indeed, melatonin stimulates the production of cytokines and more specifically interleukins (IL-2, IL-6, IL-12) 36. In addition, melatonin enhances T helper immune responses 37. Furthermore, the melatonin antioxidant actions contribute to its immuno-enhancing effects 36 and have also an indirect effect by reducing nitric oxide formation which facilitates the decrease of the inflammatory response 38. As suggested by Esquifino et al. 39, melatonin might provide a time-related signal to the immune network.
In addition, effects of melatonin on body mass and bone mass regulation have been reported. Melatonin is known for its role in energy expenditure and body mass regulation in mammals by preventing the increase in body fat with age 40. These effects are mediated by MT2 receptors in adipose tissue 41. Moreover, melatonin increases bone mass by promoting osteoblast cell differentiation and bone formation 42. In humans, melatonin stimulates bone cell proliferation and Type I collagen synthesis in these cells, and inhibits bone resorption through down-regulation of the RANKL-mediated osteoclast formation and activation 43. Also, a deficit of melatonin has been found to be associated with animal scoliosis following pinealectomy and human idiopathic scoliosis 44.
Finally, melatonin has physiological effects on reproduction and sexual maturation in mammals through down-regulation of gonadotropin-releasing hormone (GnRH) gene expression in a cyclical pattern over a 24-hour period 45. The rhythmic release of GnRH controls luteinizing hormone (LH) and follicule-stimulating hormone (FSH) secretion. The daily profile of melatonin secretion conveys internal information used for both circadian and seasonal temporal organization 31. The melatonin rhythmic pattern entrains the reproductive rhythm via the influence of photoperiod on LH pulsatile secretion and therefore mediates the seasonal fluctuations of reproduction clearly observed in animals (seasonal breading as species-specific seasons for reproduction) and moderately observed in humans 46.
Is melatonin safe
Melatonin supplements appear to be safe when used short-term; less is known about its long-term safety. Further study is needed to find out more about melatonin’s side effects, especially the delayed or long-term effects. It is unknown if melatonin causes problems when taken with other medicines. It also is unknown if melatonin affects people who have certain diseases and conditions.
- In one study, researchers noted that melatonin supplements may worsen mood in people with dementia.
- The U.S. Food and Drug Administration (FDA) regulates dietary supplements such as melatonin, but the regulations for dietary supplements are different and less strict than those for prescription or over-the-counter drugs. In 2011, the U.S. Food and Drug Administration (FDA) issued a warning to a company that makes and sells “relaxation brownies,” stating that the melatonin in them hasn’t been deemed a safe food additive.
- Most dietary supplements haven’t been tested in pregnant women, nursing mothers, or children. If you’re pregnant or nursing a child, it’s especially important to see your health care provider before taking any medication or supplement, including melatonin.
Melatonin benefits
Melatonin supplements may help some people with certain sleep disorders, including jet lag, sleep problems related to shift work, and delayed sleep phase disorder (one in which people go to bed but can’t fall asleep until hours later), and insomnia. Unlike many other sleep medications, with melatonin you are unlikely to become dependent, have a diminished response after repeated use (habituation), or experience a hangover effect.
If melatonin for sleep isn’t helping after a week or two, stop using it. And if your sleep problems continue, talk with your health care provider.
If melatonin does seem to help, it’s safe for most people to take nightly for one to two months. After that, stop and see how your sleep is. Be sure you’re also relaxing before bed, keeping the lights low and sleeping in a cool, dark, comfortable bedroom for optimal results.
Sleep Disorders
Studies suggest that melatonin may help with certain sleep disorders, such as jet lag, delayed sleep phase disorder (a disruption of the body’s biological clock in which a person’s sleep-wake timing cycle is delayed by 3 to 6 hours), sleep problems related to shift work, and some sleep disorders in children. It’s also been shown to be helpful for a circadian rhythm sleep disorders in the blind that causes changes in blind peoples’ sleep and wake times. Melatonin can help improve these disorders in adults and children.
However, study results are mixed on whether melatonin is effective for insomnia in adults, but some studies suggest it may slightly reduce the time it takes to fall asleep.
Jet lag
Jet lag is caused by rapid travel across several time zones; its symptoms include disturbed sleep, daytime fatigue, indigestion, and a general feeling of discomfort. To ease jet lag, try taking melatonin two hours before your bedtime at your destination, starting a few days before your trip.
- In a 2009 research review, results from six small studies and two large studies suggested that melatonin may ease jet lag symptoms, such as alertness.
- In a 2007 clinical practice guideline, the American Academy of Sleep Medicine supported using melatonin to reduce jet lag symptoms and improve sleep after traveling across more than one time zone.
You can also adjust your sleep-wake schedule to be in sync with your new time zone by simply staying awake when you reach your destination—delaying sleep until your usual bedtime in the new time zone. Also, get outside for natural light exposure.
Melatonin for Jet Lag:
- Eastbound: If you are traveling east, say from the US to Europe, take melatonin after dark, 30 minutes before bedtime in the new time zone or if you are on the plane. Then take it for the next 4 nights in the new time zone, after dark, 30 minutes before bedtime. If drowsy the day after melatonin use, try a lower dose.
- Westbound: If you are heading west, for example, from the US to Australia, a dose is not needed for your first travel night, but you then may take it for the next 4 nights in the new time zone, after dark, 30 minutes before bedtime. Melatonin may not always be needed for westbound travel.
Given enough time (usually 3 to 5 days), jet lag will usually resolve on its own, but this is not always optimal when traveling.
Delayed Sleep Phase Disorder
In this disorder your sleep pattern is delayed two hours or more from a conventional sleep pattern, causing you to go to sleep later and wake up later. Adults and teens with delayed sleep-wake phase sleep disorder have trouble falling asleep before 2 a.m. and have trouble waking up in the morning. Research shows that melatonin reduces the length of time needed to fall asleep and advances the start of sleep in young adults and children with this condition. Talk to your child’s doctor before giving melatonin to a child.
- In a 2007 review of the literature, researchers suggested that a combination of melatonin supplements, a behavioral approach to delay sleep and wake times until the desired sleep time is achieved, and reduced evening light may even out sleep cycles in people with this sleep disorder.
- In a 2007 clinical practice guideline, the American Academy of Sleep Medicine recommended timed melatonin supplementation for this sleep disorder.
Shift Work Disorder
Shift work refers to job-related duties conducted outside of morning to evening working hours. About 2 million Americans who work afternoon to nighttime or nighttime to early morning hours are affected by shift work disorder.
- A 2007 clinical practice guideline and 2010 review of the evidence concluded that melatonin may improve daytime sleep quality and duration, but not nighttime alertness, in people whose jobs require them to work outside the traditional morning to evening schedule.
- The American Academy of Sleep Medicine recommended taking melatonin prior to daytime sleep for night shift workers with shift work disorder to enhance daytime sleep.
Insomnia
Insomnia is a general term for a group of problems characterized by an inability to fall asleep and stay asleep. Research suggests that melatonin might provide relief from the inability to fall asleep and stay asleep (insomnia) by slightly improving your total sleep time, sleep quality and how long it takes you to fall asleep.
- In adults. A 2013 analysis of 19 studies of people with primary sleep disorders found that melatonin slightly improved time to fall asleep, total sleep time, and overall sleep quality. In a 2007 study of people with insomnia, aged 55 years or older, researchers found that prolonged-release melatonin significantly improved quality of sleep and morning alertness.
- In children. There’s limited evidence from rigorous studies of melatonin for sleep disorders among young people. A 2011 literature review suggested a benefit with minimal side effects in healthy children as well as youth with attention-deficit hyperactivity disorder, autism, and several other populations. There’s insufficient information to make conclusions about the safety and effectiveness of long-term melatonin use.
Sleep-wake cycle disturbances in children
Sleep problems are one of the most common problems parents encounter with their children. There are some simple steps parents can take to improve their children’s sleep, such as having a set bedtime and bedtime routine, avoiding foods or drinks with caffeine, and limiting the amount of screen time. Melatonin can help treat these sleep-wake cycle disturbances in children with a number of disabilities. For example, children with multiple developmental, neuro-psychiatric and health difficulties often show melatonin deficiency 23. When circadian rhythms are restored, behavior, mood, development, intellectual function, health, and even seizure control may improve 22.
Healthy sleep tips for children
Good sleep is important for your child’s physical and mental wellbeing.
A relaxing bedtime routine is one important way to help your child get a good night’s sleep.
Relaxation tips to help sleep
Doing the same relaxing things in the same order and at the same time each night helps promote good sleep:
- A warm (not hot) bath will help your child relax and get ready for sleep.
- Keeping lights dim encourages your child’s body to produce the sleep hormone, melatonin.
- Once they’re in bed, encourage your child to read quietly or listen to some relaxing music, or read a story together.
- You could also suggest your child tries this relaxing breathing exercise before bed.
Avoid screens in the bedroom
Tablets, smartphones, TVs and other electronic gadgets can affect how easily children get to sleep.
Older children may also stay up late or even wake in the middle of the night to use social media.
Try to keep your child’s bedroom a screen-free zone, and get them to charge their phones in another room.
Encourage your child to stop using screens an hour before bedtime.
Your child’s bedroom
Your child’s bedroom should ideally be dark, quiet and tidy. It should be well ventilated and kept at a temperature of about 18 to 24C.
Fit some thick curtains to block out any daylight. If there’s noise outside, consider investing in double glazing or, for a cheaper option, offer your child earplugs.
Get help with sleep problems
If you’ve tried these tips but your child keeps having problems getting to sleep or sleeping through the night, you may feel you want more support.
You can speak to your doctor to begin with. They may refer you to a child psychologist or sleep expert.
Know how much sleep your child needs
The amount of sleep your child needs changes as they get older.
A 5-year-old needs about 11 hours a night, for example, while a 9-year-old needs roughly 10 hours.
How much sleep do children need?
Below are the approximate hours of sleep needed by children of different ages, as recommended by the Children’s Sleep Clinic.
1 week old
- daytime: 8 hours
- night time: 8 hours 30 minutes
4 weeks old
- daytime: 6 to 7 hours
- night time: 8 to 9 hours
3 months old
- daytime: 4 to 5 hours
- night time: 10 to 11 hours
6 months old
- daytime: 3 hours
- night time: 11 hours
9 months old
- daytime: 2 hours 30 minutes
- night time: 11 hours
12 months of age
- daytime: 2 hours 30 minutes
- night time: 11 hours
2 years of age
- daytime: 1 hour 30 minutes
- night time: 11 hours 30 minutes
3 years of age
- daytime: 0 to 45 minutes
- night time: 11 hours 30 minutes to 12 hours
4 years of age
- night time: 11 hours 30 minutes
5 years of age
- night time: 11 hours
6 years of age
- night time: 10 hours 45 minutes
7 years of age
- night time: 10 hours 30 minutes
8 years of age
- night time: 10 hours 15 minutes
9 years of age
- night time: 10 hours
10 years of age
- night time: 9 hours 45 minutes
11 years of age
- night time: 9 hours 30 minutes
12 to 13 years of age
- night time: 9 hours 15 minutes
14 to 16 years of age
- night time: 9 hours
Why are teens always tired?
Trouble getting up on school days, dozing off in class, marathon lie-ins at weekends. It may feel like your teenager is sleeping their life away.
In fact, the opposite is probably true. Sleep experts say teens today are sleeping less than they ever have. This is a worry, as there’s a link between sleep deprivation and accidents, obesity and cardiovascular disease in later life.
Lack of sleep also affects teenagers’ education, as it can leave them too tired to concentrate in class and perform well in exams.
Teenagers’ sleep patterns
Your sleep patterns are dictated by light and hormones. When light dims in the evening, your brain produce a chemical called melatonin, which tells you it’s time to sleep.
The problem is that modern life has disrupted this pattern. Bright room lighting, TVs, games consoles, mobiles, tablets and PCs can all emit enough light to stop your bodies producing melatonin.
On top of this, research suggests that teenagers’ body clocks are set later than adults’ and younger children’s. In other words, they are programmed to stay up later and also get up later than the rest of us.
This wouldn’t be a problem if your teenager didn’t have to get up early for school. These early-morning wake-up times mean they’re not getting the 8 to 9 hours of sleep they need. The result is a tired, cranky teenager.
Sleep tips for teenagers
A minimum of 8 to 9 hours’ good sleep on school nights is recommended for teens.
Catching up on sleep at weekends isn’t ideal. Late nights and long lie-ins will just disrupt your teenager’s body clock even more.
However tired they feel, teenagers should avoid lie-ins at the weekend. They should also get out into the daylight during the day.
Both these things will help to keep their body clock regular, and make it easier to go to sleep and get up at a reasonable time.
Here’s how to make sure your teen is getting enough sleep to stay healthy and do well at school.
Limit screens in the bedroom
If possible, don’t have a mobile, tablet, TV or computer in the bedroom at night, as the light from the screen interferes with sleep.
Having screens in the bedroom also means your teen is more likely to stay up late interacting with friends on social media.
Encourage your teenager to have at least 30 minutes of screen-free time before going to sleep.
Cut out the caffeine
Suggest that your teenager drinks less caffeine – found in drinks such as cola, tea and coffee – particularly in the 4 hours before bed.
Too much caffeine can stop them falling asleep and reduce the amount of deep sleep they have.
Don’t binge before bedtime
Let teenagers know that eating too much, or too little, close to bedtime can lead to an overfull or empty stomach. This can be a cause of discomfort during the night and may prevent sleep.
Have a good routine
Encourage your teenager to get into a regular bedtime routine. Doing the same things in the same order an hour or so before bed can help them drift off to sleep.
Exercise for better sleep
It’s official: regular exercise helps you sleep more soundly, as well as improving your general health.
Teenagers should be aiming for at least 60 minutes’ exercise every day, including aerobic activities such as fast walking and running.
Exercising out in daylight will help to encourage healthy sleep patterns, too.
Create a sleep-friendly bedroom
Ensure your teenager has a good sleeping environment – ideally a room that is dark, cool, quiet and comfortable.
It might be worth investing in thicker curtains or a blackout blind to help block out early summer mornings and light evenings.
Talk through any problems
Talk to your teenager about anything they’re worried about. This will help them to put their problems into perspective and sleep better.
You could also encourage them to jot down their worries or make a to-do list before they go to bed. This should mean they’re less likely to lie awake worrying during the night.
Other therapeutic effects of melatonin
Therapeutic effects of melatonin have been reported in several disorders such as certain tumors, cardiovascular diseases or psychiatric disorders. The part concerning melatonin and psychiatric disorders is in particular developed given our past and current work on this topic.
Indeed, oncostatic effects of melatonin have been reported in several tumors (breast cancer, ovarian and endometrial carcinoma, human uveal melanoma, prostate cancer, hepatomas, and intestinal tumors) 47. These oncostatic effects have been attributed to the anti-oxidative role of melatonin given that oxidative stress is involved in the initiation, promotion and progression of carcinogenesis 48. Also, decreased melatonin levels (measures of blood melatonin or urinary excretion of 6-SM) were reported in patients with cardiovascular diseases 49. Inversely, melatonin treatment reduces blood pressure in patients with hypertension 50.
Concerning psychiatric disorders, secretion disturbances of the pineal gland have been described in child and adult psychiatry, with notably in most studies a decreased nocturnal melatonin secretion observed in major depressive disorder, bipolar disorder, schizophrenia or autism spectrum disorder 51.
Also, a phase-shift of melatonin has been reported in major depressive disorder and bipolar disorder, including in particular a delayed melatonin peak secretion 52. It is noteworthy that increased melatonin levels (measures of blood melatonin and urinary excretion of 6-SM) were found when clinical therapeutic benefits were observed following the use of antidepressants 53. Furthermore, significant improvement of major depressive disorder and anxiety was described following administration of 25mg per day of agomelatine, a MT1/MT2 melatonin agonist and selective antagonist of 5-HT2C receptors 54.
Autism spectrum disorder
Concerning autism spectrum disorder (ASD), abnormalities in the serotoninergic system and sleep-wake rhythm disturbances observed in children with autism spectrum disorder suggest altered melatonin secretion in autism 55. Sleep disorders (mostly increased sleep latency, reduced total sleep and nocturnal awakenings with insomnia) are observed in 50-80% of individuals with autism 56. It is noteworthy that sleep problems are not specific of autism and are also observed in children with intellectual disability associated or not with autism 57. However, melatonin measures in children with intellectual disability not associated with autism, such as some children with Down syndrome and Fragile X syndrome, showed respectively normal melatonin production despite delayed nocturnal melatonin peak secretion and increased levels of melatonin 58, whereas decreased nocturnal melatonin secretion was mostly observed in children with autism 57. Scientists reported in two different large samples of children with autism significant relationships between decreased nocturnal urinary excretion and severity of autistic impairments in social communication 59, 60. These results suggest that abnormalities in melatonin physiology might contribute not only to sleep problems in autism, but also to biological and psychopathological mechanisms involved in the development of autism spectrum disorder (for example, certain immunological abnormalities found in autism, such as a decrease number of T lymphocytes, might be explained by the hypo-functioning of the melatonin system).
Schizophrenia
Concerning schizophrenia, as suggested by Morera-Fumero and Abreu-Gonzalez 51, a possible explanation for the “low melatonin syndrome” present in some individuals with schizophrenia may stem from the study of the melatonin-synthesizing enzymes, the AA-NAT and ASMT. Furthermore, according to some authors, MT3 might be involved in the melatonin disturbances observed in schiozophrenia 61. Finally, melatonin secretion was also studied in obsessive compulsive disorder but no abnormalities in melatonin levels were reported.
Brain Protection
Neurological and neuropsychological disabilities caused by brain injuries are a major public health concern. Thus, reducing deficits after a stroke is a major issue. In this line, a number of recent studies have reported the important role of melatonin in neuroprotection in animal models of stroke 62. Indeed, melatonin administration after an experimental stroke in animals reduces infarction volume 63. Such a protective effect can be seen in both gray and white matter 64 and melatonin reduces also inflammatory response 65, cerebral oedema formation 66, and blood-brain barrier permeability 67. In addition, Kilic et al. 68 investigated how sub-acute delivery of melatonin, starting at 24 hours after stroke onset, and continuing for 29 days can influences neuronal survival, endogenous neurogenesis, motor recovery and locomotor activity in mice submitted to an occlusion of the middle cerebral artery during 30 minutes. Furthermore, melatonin improved neuronal survival and enhanced neurogenesis, even when applied one day after stroke. In addition, the authors showed both motor as well as behavioral improvements after melatonin administration. Indeed, the results indicate that cell survival was associated with a long-lasting improvement of motor and coordination deficits as well as with attenuation of hyperactivity and anxiety of the animals as revealed in open field tests. Its neuroprotective activity in animal models of ischemic stroke, as well as its lack of serious toxicity suggests that melatonin could be used for human stroke treatment in the future.
In addition to its protective effect after stroke, experimental data obtained in various independent animal models of brain lesions in neonates support the notion of a neuroprotective effect of melatonin in preterm neonates {see for a review, Biran et al., 69]. In infants, a major source of brain injury is preterm birth, often associated to long-term neurological, cognitive, educational, and social problems. Neurodevelopmental disorders are not only seen in extremely preterm birth 70 but also in late prematurity 71. A large number of infants who survive very preterm birth develop cerebral palsy 72 with a high occurrence of associated motor, perceptual and cognitive deficiencies in childhood 73. Nowadays, the most common brain damage observed in preterm children is diffuse white matter damage as well as reduced neural connectivity 74 in the context of infection, inflammation, and hypoxia-ischemia 75. Although a number of treatments have been tested in preclinical animal models of perinatal brain injury, none of them had been proved to be efficient as a neuroprotector nor translated in clinical practice. Among the molecules proposed, melatonin is a very good candidate, given its effect on brain development, neuroprotection as well as regarding its absence of adverse effects 76. As discussed by Biran et al., 69, in addition to its good safety profile, melatonin easily crosses the placenta as the blood-brain barriers and blocks oxidative, excitotoxic and inflammatory pathways, all involved in the pathogenesis of perinatal brain damage caused by preterm birth. However, only a few studies have looked at the synthesis of melatonin in preterm and term neonates. These studies point to a reduced urinary concentration of melatonin during the first 3 months after birth in preterm infants 69. As these authors discussed, compared with term neonates, preterm neonates show a delayed secretion of melatonin which persists after correction for gestational age up to 8 to 9 months of age. In the absence of maternal melatonin, the appearance of circadian rhythms depends principally on neurological maturation, and very little on the environment 77.
Since, melatonin easily crosses the blood–brain and placental barriers, it can be administered antenatally in order to reduce or prevent the impact of
brain lesions in preterm neonates. Currently, two therapeutic trials testing the neuroprotective properties of melatonin administration in the immediate prepartum period in very preterm infants are under way in France and in the United Kingdom69. The French trial aims to determine the dose of melatonin to be administered in prepartum by parenteral route to mothers at risk of preterm delivery, to decrease the extent of white matter damage detected by diffusion tensor imaging in infants born preterm. The objective of the English trial is to prove that melatonin is capable of reducing brain injury and white matter disease as defined by magnetic resonance imaging at term. These trials will probably lead to a clinical use of melatonin before preterm birth (in case of at risk mother) of just after birth in preterm neonates in order to prevent neurodevelopmental deficits in these children.
Interestingly, from a functional point of view, abnormalities in melatonin physiology associated with sleep disorders, and in particular sleep deprivation, are seen to endanger cerebral and more specifically hippocampal integrity, leading to cognitive dysfunction and contributing to the development of mood disorders 78. The involvement of melatonin in the development of mood disorders was discussed in the previous section.
Finally, based on the brain protective role of melatonin against oxidative stress previously described in this article, there is also increased experimental evidence showing the therapeutic potential of melatonin in neurodegenerative conditions such as Alzheimer disease, Parkinson disease, Huntington’s disease and amyotrophic lateral sclerosis 79. Additional studies and clinical trials are now required both in preterm neonates and aging adults to test the clinical efficacy of melatonin supplementation in such disorders, and to identify the specific therapeutic concentrations needed regarding the subject’s age, disease and brain lesion as well as the short and long-term effects of melatonin both on physiological, functional and cognitive outcomes.
Melatonin supplement
Evidence suggests that melatonin promotes sleep and is safe for short-term use. Melatonin can be used to treat delayed sleep phase and circadian rhythm sleep disorders in the blind and provide some insomnia relief. Treat melatonin as you would any sleeping pill and use it under your doctor’s supervision.
There is little scientific evidence that melatonin has a role in promoting health or treating disease. Melatonin also is not proven to slow down the aging process or prolong your life.
How much melatonin should I take?
The typical adult dose ranges from 0.3 mg to 5 mg at bedtime. Lower doses often work as well as higher doses. Intake of an usual dose (i.e., 1 to 5 mg), allows within the hour after ingestion, melatonin concentrations 10 to 100 times higher than the physiological nocturnal peak to be obtained, with a return to basal concentrations in 4 to 8 hours 19. A bioavailability study in four male healthy volunteers 80 showed a plasma melatonin peak varying between 2 and 395 nmol/L and an elimination half-life of 47± 3 min (mean ± SD) after oral administration of a 0.5 mg dose. Bioavailability varied from 10 to 56% (mean 33%).
Read the directions on the label of the pill bottle. These will tell you how much melatonin to take and how often to take it. If you have questions about how to take melatonin, call your doctor or pharmacist. Do not take more than the recommended amount. Taking more melatonin does not make it work quicker or better. Overdosing on any medicine can be dangerous.
Keep a record of all medicines and supplements you take and when you take them. Take this list with you when you go to the doctor. Ask your doctor if it’s okay to take melatonin if:
- You take other prescription or over the counter (OTC) medicines.
- You have ongoing health problems.
- You are pregnant or nursing (it is unclear what effect melatonin can have on an unborn baby or nursing infant).
Melatonin side effects
Melatonin taken orally in appropriate amounts is generally safe. Side effects of melatonin are uncommon but can include:
- drowsiness,
- a “heavy-head” feeling,
- headache,
- dizziness,
- feeling hungover,
- nausea.
There have been no reports of significant side effects of melatonin in children.
Other, less common melatonin side effects might include short-lasting feelings of depression, mild tremor, mild anxiety, abdominal cramps, irritability, reduced alertness, confusion or disorientation, and abnormally low blood pressure (hypotension). Because melatonin can cause daytime drowsiness, don’t drive or use machinery within five hours of taking the supplement.
- Do not use melatonin if you are pregnant or breastfeeding or have an autoimmune disorder, a seizure disorder or depression. Talk to your health care provider if you have diabetes or high blood pressure. Melatonin supplements may also raise blood-sugar levels and increase blood pressure levels in people taking some hypertension medications.
In addition, melatonin supplements can interact with various medications, including:
- Anticoagulants and anti-platelet drugs. These types of drugs, herbs and supplements reduce blood clotting. Combining use of melatonin with them might increase the risk of bleeding.
- Anticonvulsants. Melatonin might inhibit the effects of anticonvulsants in neurologically disabled children.
- Blood pressure drugs. Melatonin might worsen blood pressure in people taking blood pressure medications.
- CNS depressants. Melatonin use with use of these medications might cause an additive sedative effect.
- Diabetes medications. Melatonin might affect sugar levels. If you use diabetes medications, use melatonin cautiously.
- Contraceptive drugs. Use of contraceptive drugs with melatonin might increase the effects and possible side effects of melatonin.
- Cytochrome P450 1A2 (CYP1A2) and cytochrome P450 2C19 (CPY2C19) substrates. Use melatonin cautiously if you take drugs such as diazepam (Valium) and others that are affected by these enzymes.
- Fluvoxamine (Luvox). This selective serotonin reuptake inhibitor can increase melatonin levels, causing unwanted excessive drowsiness.
- Medications that suppress the immune system (immunosuppressants). Melatonin can stimulate immune function and interfere with immunosuppressive therapy.
- Seizure threshold lowering drugs. Taking melatonin with these drugs might increase the risk of seizures.
If you’re considering taking melatonin supplements, check with your doctor first — especially if you have any health conditions. He or she can help you determine if melatonin is right for you.
Melatonin Dosage
Melatonin is considered an effective treatment for jet lag and can aid sleep during times when you would not normally be awake. However, melatonin is not approved by the FDA for any use. Melatonin falls under the FDA’s Dietary Health and Education Act as a dietary supplement. The FDA is not authorized to review the safety or effectiveness of dietary supplements like melatonin before they are marketed.
Effective starting doses for jet lag range from 0.3 to 0.5 mg. One milligram tablets can be cut in half to achieve a 0.5 mg dose if smaller doses are not available for purchase. Lower doses may work for some people, while others may need a higher dose, up to 3 to 5 mg. However, higher doses may be associated with more side effects such as headache, next day grogginess, or vivid dreams.
Always start with the lowest melatonin dose. According to a Cochrane review 81, doses over 5 mg appear to be no more effective than lower doses. It is important to note that much higher doses are available for sale in the U.S., but these doses may result in excessively high levels of physiologic melatonin.
Melatonin for sleep
How to Take Melatonin for Sleep (Insomnia):
Dosage: Take melatonin 0.1 mg to 0.5 mg thirty minutes before bedtime. Studies suggest melatonin for sleep may be effective in promoting but not maintaining sleep (early morning awakening).
How to Take Melatonin for Shift-Work Sleep Disorders
Dosage: Take melatonin 1.8 mg to 3 mg thirty minutes prior to the desired onset of daytime sleep; melatonin may NOT lead to improved alertness during the nighttime work shift and may only improve daytime sleep time by about 30 minutes.
How to Take Melatonin for Delayed Sleep Phase Disorder
Delayed sleep phase disorder most often occurs in adolescents, possibly due to reduced melatonin production and melatonin deficiency at this age. Sleep onset is delayed by 3 to 6 hours compared with conventional bedtimes (10 to 11 pm). Delayed sleep phase disorder can negatively affect school performance, daily activities, and lead to morning drowsiness which can be dangerous for teen drivers. Any sleep disorder in an adolescent should be evaluated by a physician.
Dosage: Take melatonin 1 mg four to six hours before set bedtime. Once a set bedtime is achieved, use maintenance doses of 0.5 mg melatonin 2 hours before expected sleep onset. Bright light therapy and behavioral management may enhance results. Be aware drowsiness may occur after melatonin dose, so avoid hazardous activities such as driving.
How to Take Melatonin for Non-24-Hour Sleep Wake Disorder (Non-24)
More than 70% of people who are totally blind have Non-24, a circadian rhythm disorder. For people who are totally blind, there are no light cues to help reset the biological clock. The sleep time and wake up time of people who have Non-24-Hour Sleep Wake Disorder shifts a little later every day. Sleep times go in and out of alignment compared to a normal sleep-wake phase. Extra minutes add up each day by day and disrupt the normal wake-sleep pattern.
Use of melatonin in Non-24 is to aid in stimulation to reset the biological clock with one long sleep time at night and one long awake time during the day. However, no large-scale clinical trials of melatonin therapy for Non-24 have been conducted to date.
Dosage: Studies on the blind suggest that 0.5 mg/day melatonin is an effective dose.
Hetlioz, a prescription-only melatonin agonist is also approved for use in Non-24-Hour Sleep Wake Disorder in blind individuals.
Hetlioz (tasimelteon)
Fast-dissolving Melatonin
Some melatonin tablets are available in fast-dissolving formulations in the U.S. To take the orally disintegrating tablet:
- Use dry hands to remove the tablet and place it in your mouth.
- Do not swallow the tablet whole. Allow it to dissolve in your mouth without chewing. If desired, you may drink liquid to help swallow the dissolved tablet.
Call your doctor if the condition you are treating with melatonin does not improve, or if it gets worse while using this product.
Store at room temperature away from moisture and heat.
Melatonin for children
Parents may consider using melatonin to help their child who has a trouble falling asleep. Only use melatonin for your child under the care of a pediatrician or other medical sleep specialist. Insomnia or other sleeping disorders in children should always be evaluated by a medical professional. Children 6 months to 14 years of age with sleep disorders : Melatonin 2 to 5 mg has been used.
Melatonin should not be used as a substitute for good sleep hygiene and consistent bedtime routines in children.
Products containing lower-dose melatonin for kids do exist on the U.S. market. However, long-term use of melatonin has not been studied in children and possible side effects with prolonged use are not known. Use for children with autism spectrum disorder or attention-deficit hyperactivity disorder should involve behavioral interventions and should be directed by a physician.
Delayed sleep phase disorder often occurs in teenagers and young adults, possibly due to alterations in endogenous melatonin production. Sleep onset is delayed by 3 to 6 hours compared with normal bedtime hours of 10 to 11 PM. Maintaining a consistent bedtime free of electronics for at least one hour prior to bedtime is especially important for children and adolescents.
Melatonin Side Effects in Children
The most common melatonin side effect in children is morning drowsiness. Other common side effects in children include:
- Bedwetting
- Headache
- Dizziness
- Nausea
- Diarrhea
- Possible increased risk for seizures in children with severe neurological disorders.
Dietary melatonin supplements can still have drug interactions or health risks if you have certain medical conditions, upcoming surgery, or other health concerns.
Melatonin overdose
Melatonin is thought to be very safe in the short-term with a low risk for overdose. Melatonin is not known to be a potential cause of death, but you need to be aware that it could lead to certain complications if you overdose. Taking too much melatonin can disrupt your circadian rhythms (sleep-wake cycle). It may also cause other unwanted side effects. So, yes, you can technically overdose on melatonin. However, a melatonin overdose can be hard to define since there isn’t an official standard safe dose for everyone.
The acute toxicity of melatonin as seen in both animal and human studies is extremely low. Melatonin may cause minor adverse effects, such as headache, insomnia, rash, upset stomach, and nightmares 82. In animals, an LD50 (the lethal dose which suggests that this is an amount of which at least 50% of the experimental animals (rat or mouse) would die of exposure) could not be established 82. Even 800 mg/kg bodyweight (high dose) was not lethal 83. Studies of human subjects given varying doses of melatonin (1–6.6 g/day) for 30–45 days, and followed with an elaborate battery of biochemical tests to detect potential toxicity, have concluded that, aside from drowsiness, all findings were normal at the end of the test period 84.
Melatonin is widely available as an over-the-counter supplement marketed by different companies. These supplements may not be similar in dosage and/or composition, and some of them may contain additional vitamins. Moreover, melatonin may interact with other over-the-counter drugs, although such interactions have not been systematically evaluated and, therefore, remain unreported. Some people are more sensitive than others to the effects of melatonin. A dose that might trigger side effects in one person may have little effect on someone else.
Young children should avoid melatonin unless otherwise directed by a doctor. Doses between 1 and 5 milligrams (mg) may cause seizures or other complications for young children. In adults, doses in the 30-mg range may be harmful. In general, it’s better to start low and move up slowly and carefully if you see encouraging results.
Some people can have side effects from melatonin that may include:
- daytime drowsiness, dizziness, weakness, or confusion
- vivid dreams, nightmares
- feeling depressed, anxious, irritable
- headache
- loss of appetite, diarrhea, nausea, stomach pain
- blood pressure changes
- joint or back pain
- elevated risk for seizures
Animal studies suggest that melatonin can downregulate the pituitary/gonadal axis resulting in hypogonadism and/or delayed puberty. However chronic administration of low-dose melatonin in men did not alter blood levels of testosterone or luteinizing hormone 85. One case of extremely high melatonin levels associated with delayed puberty and hypogonadism has been reported 86. Pubertal development and resolution of the hypogonadism occurred spontaneously as melatonin levels declined over several years. Recent experimental evidence demonstrates that melatonin reduces sperm motility 87 and that long-term administration inhibits testicular aromatase levels 88.
Melatonin has also been suggested for use as a contraceptive for women 89, which might raise the question of whether melatonin damages the female reproductive system. Notably, no side effects were reported in a report of a phase 2 clinical trial in which 1400 women were treated with 75 mg of melatonin nightly for 4 years 89.
Preliminary animal studies suggest that melatonin may accelerate the development of autoimmune conditions 90. Melatonin transiently exacerbated neurologic symptoms in 1 patient with multiple sclerosis 91.
Although melatonin is a potential adjunctive agent in the treatment of cancer and immune deficiency, poorly timed administration can produce opposite effects. Melatonin injections given in the morning stimulate tumor growth 92, whereas the same doses in mid-afternoon have no effect but in the evening have a retarding effect. And although some people with depression may suffer from a “low melatonin syndrome” 93, melatonin administration that unduly prolongs the nocturnal melatonin rise, or that is given throughout the day, may exacerbate seasonal affective disorder 94 and bipolar and classic depression 95. Finally, animal studies have shown that moderately large doses of melatonin (equivalent in one study to about 30 mg in adult humans) increased light-induced damage to retinal photoreceptors 96.
There is also some concern regarding increased atherosclerosis in the aorta in hypercholesterolemic rats caused by melatonin 97. Moreover, in these animals LDL “bad” cholesterol were less well recognized by LDL-receptor metabolic pathways when melatonin was administered.
- https://livertox.nlm.nih.gov//Melatonin.htm[↩]
- Carrillo-Vico A., Calvo J.R., Abreu P., Lardone P.J., Garcia-Maurino S., Reiter R.J., Guerrero J.M. Evidence of melatonin synthesis by human lymphocytes and its physiological significance: possible role as intracrine, autocrine, and/or paracrine substance. FASEB J. 2004;18:537–539.[↩]
- Bubenik G.A. Gastrointestinal melatonin: localization, function, and clinical relevance. Dig. Dis. Sci. 2002;47:2336–2348.[↩][↩][↩]
- Reiter R.J., Tan D.X., Maldonado M.D. Melatonin as an antioxidant: physiology versus pharmacology. J. Pineal Res. 2005;39:215–216.[↩]
- Katzer D., Pauli L., Mueller A., Reutter H., Reinsberg J., Fimmers R., Bartmann P., Bagci S. Melatonin concentrations and antioxidative capacity of human breast milk according to gestational age and the time of the day. 2016.[↩]
- Pires-Lapa M.A., Tamura E.K., Salustiano E.M., Markus R.P. Melatonin synthesis in human colostrum mononuclear cells enhances dectin-1-mediated phagocytosis by mononuclear cells. 2013.[↩]
- Coon S.L., Roseboom P.H., Baler R., Weller J.L., Namboodiri M.A., Koonin E.V., Klein D.C. Pineal serotonin N-acetyltrans- ferase: expression cloning and molecular analysis. Science. 1995;270(5242):1681–1683[↩]
- Simonneaux V., Ribelayga C. Generation of the melatonin endocrine message in mammals: A review of the complex regulation of melatonin synthesis by norepinephrine, peptides, and other pineal transmitters. Pharmacol. Rev. 2003;55:325–395.[↩]
- Klein D.C. 2004.[↩]
- Touitou Y. La mélatonine: hormone et médicament. C. R. Soc. Biol. 1998;192:643–657[↩]
- Brzezinski A. Melatonin in humans. N. Engl. J. Med. 1997;336(3):186–195[↩]
- Karasek K., Winczyk K. Melatonin in humans. J. Physiol. Pharmacol. 2006;57(5):19–39[↩]
- Sadeh A. Sleep and melatonin in infants: a preliminary study. Sleep. 1997;20(3):185–191[↩]
- Joseph D., Chong N.W., Shanks M.E., Rosato E., Taub N.A., Petersen S.A. Get- ting rhythm: how do babies do it? Arch. Dis. Child. Fetal Neonatal Ed. 2014;100(1):F50–F54. doi: 10.1136/archdischild-204-306104.[↩]
- Karasek K., Winczyk K. Melatonin in humans. J. Physiol. Pharmacol. 2006;57(5):19–39.[↩]
- Waldhauser F., Weiszenbacher G., Frisch H., Zeitlhuber U., Waldhauser M., Wurtman R.J. Fall in nocturnal serum melatonin during prepuberty and pubescence. Lancet. 1984;1(8373):362–365.[↩]
- Lynch H.J., Wurtman R.J., Moskowitz M.A., Archer M.C., Ho M.H. Daily rhythm in human urinary melatonin. Science. 1975;187(4172):169–171[↩]
- Di W.L., Kadva A., Johnston A., Silman R. Variable bioavailability of oral melatonin. N. Engl. J. Med. 1997;336(14):1028–1029[↩]
- Tordjman S, Chokron S, Delorme R, et al. Melatonin: Pharmacology, Functions and Therapeutic Benefits. Current Neuropharmacology. 2017;15(3):434-443. doi:10.2174/1570159X14666161228122115. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5405617/[↩][↩]
- Karasek K., Winczyk K. Melatonin in humans. J. Physiol. Pharmacol. 2006;57(5):19–39. https://www.ncbi.nlm.nih.gov/pubmed/17218758[↩]
- Zhdanova I., Lynch H., Wurtman R. Melatonin: a sleep- promoting hormone. Sleep. 1997;20:899–907 https://www.ncbi.nlm.nih.gov/pubmed/9415953[↩]
- Arendt J. Melatonin: characteristics, concerns, and prospects. 2005. https://www.ncbi.nlm.nih.gov/pubmed/16077149[↩][↩][↩]
- Jan J.E., Bax M.C., Owens J.A., Ipsiroglu O.S., Wasdell M.B. Neurophysiology of circadian rhythm sleep disorders of children with neurodevelopmental disabilities. Eur. J. Paediatr. Neurol. 2012;16(5):403–412. https://www.ncbi.nlm.nih.gov/pubmed/22264650[↩][↩]
- Guzman-Marin R., Suntsova N., Methippara M., Greiffenstein R., Szymusiak R., McGinty D. Sleep deprivation suppresses neurogenesis in the adult hippocampus of rats. 2005 https://www.ncbi.nlm.nih.gov/pubmed/16262649[↩]
- Iwasaki S., Nakazawa K., Sakai J., Kometani K., Iwashita M., Yoshimura Y., Maruyama T. Melatonin as a local regulator of human placental function. J. Pineal Res. 2005;39:261–265. https://www.ncbi.nlm.nih.gov/pubmed/16150106[↩][↩]
- Okatani Y., Morioka N., Hayashi K. Changes in nocturnal pineal melatonin synthesis during the perimenopausal period: relation to estrogen levels in female rats. J. Pineal Res. 1999;27(2):65–72.[↩]
- Torres-Farfan C., Valenzuela F.J., Germain A.M., Viale M.L., Campino C., Torrealba F., Valenzuela G.J., Richter H.G., Serón-Ferré M. Maternal melatonin stimulates growth and prevents maturation of the capuchin monkey fetal adrenal gland. 2006[↩]
- Torres-Farfan C., Serón-Ferré M., Dinet V., Korf H.W. Immunocytochemical demonstration of day/night changes of clock gene protein levels in the murine adrenal gland: differences between melatonin-proficient (C3H) and melatonin-deficient (C57BL) mice. J. Pineal Res. 2006;40(1):64–70.[↩]
- Reiter R.J., Tan D.X., Korkmaz A., Rosales-Corral S.A. Melatonin and stable circadian rhythms optimize maternal, placental and fetal physiology. Hum. Reprod. Update. 2014;20(2):293–307 [↩]
- Pandi-Perumal S.R., Srinivasan V., Maestroni G.J., Cardinali D.P., Poeggeler B., Hardeland R. Melatonin: Nature’s most versatile biological signal? FEBS J. 2006;273:2813–2838.[↩]
- Pevet P., Challet E. 2011.[↩][↩]
- Arendt J. Melatonin and human rhythms. Chronobiol. Int. 2006;29:21–37.[↩]
- Reiter R.J., Tan D.X., Maldonado M.D. Melatonin as an antioxidant: physiology versus pharmacology. J. Pineal Res. 2005;39:215–216[↩]
- Bubenik G.A. Gastrointestinal melatonin: localization, function, and clinical relevance. Dig. Dis. Sci. 2002;47:2336–2348[↩]
- Pandi-Perumal S.R., Esquifino A.I., Cardinali D.P. The role of melatonin in immunoenhancement: Potential application in cancer. Int. J. Exp. Pathol. 2006;87:81–87.[↩]
- Srinivasan V., Pandi-Perumal S.R., Maestroni G.J., Esquifino A.I., Hardeland R., Cardinali D.P. Role of melatonin in neurodegenerative diseases. Neurotox. Res. 2005;7:293–318.[↩][↩]
- Carrillo-Vico A., Guerrero J.M., Lardone P.J., Reiter R.J. A review of the multiple actions of melatonin on the immune system. 2005.[↩]
- Hardeland R., Pandi-Perumal S.R. Melatonin, a potent agent in antioxidative defense: Actions as a natural food constituent, gastrointestinal factor, drug and prodrug. Nutr. Metab. 2005;2:22.[↩]
- Esquifino A.I., Pandi-Perumal S.R., Cardinali D.P. Circadian organization of the immune response: a role for melatonin. 2004.[↩]
- Bartness T.J., Demas G.E., Song C.K. Seasonal changes in adiposity: the roles of the photoperiod, melatonin and other hormones, and sympathetic nervous system. Exp. Biol. Med. (Maywood) 2002;227:363–376.[↩]
- Brydon L., Petit L., Delagrange P., Strosberg A.D., Jockers R. Functional expression of mt2 (mel1b) melatonin receptors in human paz6 adipocytes. Endocrinology. 2001;142:4264–4271.[↩]
- Ladizesky M.G., Cutrera R.A., Boggio V., Somoza J., Centrella J.M., Mautalen C., Cardinali D.P. Effect of melatonin on bone metabolism in ovariectomized rats. Life Sci. 2001;70:557–565.[↩]
- Koyama H., Nakade O., Takada Y., Kaku T., Lau K.H. Melatonin at pharmacologic doses increases bone mass by suppressing resorption throught downregulation of the RANKL-mediated osteoclast formation and activation. J. Bone Miner. Res. 2002;17:1219–1229.[↩]
- Fjelldal P.G., Grotmol S., Kryvi H., Gjerdet N.R., Taranger G.L., Hansen T., Porter M.J., Totland G.K. Pinealectomy induces malformation of the spine and reduces the mechanical strength of the vertebrae in Atlantic salmon, Salmo salar. J. Pineal Res. 2004;36:132–139.[↩]
- Balik A., Kretschmannova K., Mazna P., Svobodova I., Zemkova H. Melatonin action in neonatal gonadotrophs. Physiol. Res. 2004;53(1):S153–S166.[↩]
- Barrell G.K., Thrun L.A., Brown M.E., Viguie C., Karsch F.J. Importance of photoperiodic signal quality to entrainment of the circannual reproductive rhythm of the Ewe. Biol. Reprod. 2000;63:769–774.[↩]
- Blask D.E., Dauchy R.T., Sauer L.A. Putting cancer to sleep at night: the neuroendocrine/circadian melatonin signal. Endocrine. 2005;27:179–188.[↩]
- Karbownik M., Lewinski A., Reiter R.J. Anticarcinogenic actions of melatonin which involve antioxidative processes: comparison with other antioxidants. Int. J. Biochem. Cell Biol. 2001;33:735–753.[↩]
- Yaprak M., Altun A., Vardar A., Aktoz M., Ciftci S., Ozbay G. Decreased nocturnal synthesis of melatonin in patients with coronary artery disease. Int. J. Cardiol. 2003;89:103–107.[↩]
- Scheer F.A. Potential use of melatonin as adjunct antihypertensive therapy. 2005.[↩]
- Morera-Fumero A.L., Abreu-Gonzalez P. Role of melatonin in schizophrenia. Int. J. Mol. Sci. 2013;14:9037–9050. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3676771/[↩][↩]
- Milhiet V., Etain B., Boudebesse C., Bellivier F. Circadian biomakers, circadian genes and bipolar disorders. J. Physiol. 2011;105:183–189.[↩]
- Thompson C., Mezey G., Corn T., Franey C., English J., Arendt J., Checkley S.A. The effect of desipramine upon melatonin and cortisol secretion in depressed and normal subjects. 1985.[↩]
- Den Boer J.A., Bosker F.J., Meesters Y. Clinical efficacy of agomelatine in depression: the evidence. 2006.[↩]
- Kotagal S., Broomall E. Sleep in children with autisms pectrum disorder. J. Pediatr. Neurol. 2012;47(4):242–251. doi: 10.1016/j.pediatrneurol.2012.05.007. [↩]
- Lai M.C., Lombardo M.V., Baron-Cohen S. Autism. Lancet. 2014;383:896–910.[↩]
- Tordjman S., Najjar I., Bellissant E., Anderson G.M., Barburoth M., Cohen D., Nemat Jaafari N., Schischmanoff O., Fagard R., Lagdas E., Kermarrec S., Ribardiere R., Botbol M., Fougerou C., Bronsard G., Vernay-Leconte J. Advances in the research of melatonin in autism spectrum disorders: literature review and new perspectives. Int. J. Mol. Sci. 2013;14(10):20508–20542. doi: 10.3390/ijms141020508. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3821628/[↩][↩]
- Gould E.L., Loesch D.Z., Martin M.J., Hagerman R.J., Amstrong S.M., Huggins R.M. Melatonin profiles and sleep characteristics in boys with fragile X syndrome: a preliminary study. 2000.[↩]
- Tordjman S., Anderson G.M., Pichard N., Charbuy H., Touitou Y. Nocturnal excretion of 6-sulphatoxymelatonin in children and adolescents with autistic disorder. Biol. Psychiatry. 2005;57:134–138. https://www.ncbi.nlm.nih.gov/pubmed/15652871[↩]
- Tordjman S., Anderson G.M., Bellissant E., Botbol M., Charbuy H., Camus F., Graignic R., Kermarrec S., Fougerou C., Cohen D., Touitou Y. Day and nighttime excretion of 6-sulphatoxymelatonin in adolescents and young adults with autistic disorder. Psychoneuroendocrinology. 2012;37:1996–1997. https://www.ncbi.nlm.nih.gov/pubmed/22613035[↩]
- Jockers R., Maurice P., Boutin J.A., Delagrange P. Melatonin receptors, heterodimerization, signal transduction and binding sites: what’s new? Br. J. Pharmacol. 2008;154(6):1182–1195 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2483381/[↩]
- Kilic E., Ozdemir Y.G., Bolay H., Kelestimur H., Dalkara T. Pinealectomy aggravates and melatonin administration attenuates brain damage in focal ischemia. J. Cereb. Blood Flow Metab. 1999;19:511–516. https://www.ncbi.nlm.nih.gov/pubmed/10326718[↩]
- Pei Z., Pang S.F., Cheung R.T. Administration of melatonin after onset of ischemia reduces the volume of cerebral infarction in a rat middle cerebral artery occlusion stroke model. Stroke. 2003;34:770–775. http://stroke.ahajournals.org/content/34/3/770.long[↩]
- Lee E.J., Lee M.Y., Chen H.Y., Hsu Y.S., Wu T.S., Chen S.T., Chang G.L. Melatonin attenuates gray and white matter damage in a mouse model of transient focal cerebral ischemia. J. Pineal Res. 2005;38:42–52 https://www.ncbi.nlm.nih.gov/pubmed/15617536[↩]
- Lee M.Y., Kuan Y.H., Chen H.Y., Chen T.Y., Chen S.T., Huang C.C., Yang I.P., Hsu Y.S., Wu T.S., Lee E.J. Intravenous administration of melatonin reduces the intracerebral cellular inflammatory response following transient focal cerebral ischemia in rats. 2007 https://www.ncbi.nlm.nih.gov/pubmed/17349029[↩]
- Kondoh T., Uneyama H., Nishino H., Torii K. Melatonin reduces cerebral edema formation caused by transient forebrain ischemia in rats. Life Sci. 2002;72:583–590 https://www.ncbi.nlm.nih.gov/pubmed/12467899[↩]
- Chen T.Y., Lee M.Y., Chen H.Y., Kuo Y.L., Lin S.C., Wu T.S., Lee E.J. Melatonin attenuates the postischemic increase in blood-brain barrier permeability and decreases hemorrhagic transformation of tissue-plasminogen activator therapy following ischemic stroke in mice. J. Pineal Res. 2006;40:242–250. https://www.ncbi.nlm.nih.gov/pubmed/16499561[↩]
- Kilic E., Kilic U., Bacigaluppi M., Guo Z., Ben Abdallah N., Wolfer D.P., Reiter R.J., Hermann D.M., Bassetti C.L. Delayed melatonin administration promotes neuronal survival, neurogenesis and motor recovery, and attenuates hyperactivity and anxiety after mild focal cerebral ischemia in mice. J. Pineal Res. 2008;45:142–148. https://www.ncbi.nlm.nih.gov/pubmed/18284547[↩]
- Biran V., Phan Duy A., Decobert F., Bednarek N., Alberti C., Baud O. Is melatonin ready to be used in preterm infants as a neuroprotectant? Dev. Med. Child Neurol. 2014;56(8):717–723. https://www.ncbi.nlm.nih.gov/pubmed/24575840[↩][↩][↩][↩]
- Arpino C., Compagnone E., Montanaro M.L., Cacciatore D., De Luca A., Cerulli A., Di Girolamo S., Curatolo P. 2010.[↩]
- Machado J. L.C.; Passini, Júnior, R.; Rodrigues, M. R. I. Late prematurity: a systematic review. J. Pediatr. (Rio J.) 2014;90(3):221–231 https://www.ncbi.nlm.nih.gov/pubmed/24508009[↩]
- Allen M.C. Neurodevelopmental outcomes of preterm infants. 2008. https://www.ncbi.nlm.nih.gov/pubmed/18317268[↩]
- Vincer M.J., Allen A.C., Allen V.M., Baskett T.F., O’Connell C.M. Trends in the prevalence of cerebral palsy among very preterm infants. Paediatr. Child Health. 2014;19(4):185–189. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4028643/[↩]
- Pineda R.G., Neil J., Dierker D., Smyser C.D., Wallendorf M., Kidokoro H., Reynolds L.C., Walker S., Rogers C., Mathur A.M., Van Essen D.C., Inder T. Alterations in brain structure and neurodevelopmental outcome in preterm infants hospitalized in different neonatal intensive care unit environments. J. Pediatr. 2014;164(1):52–60.e2. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3872171/[↩]
- Benders M.J., Kersbergen K.J., de Vries L.S. Neuroimaging of white matter injury, intraventricular and cerebellar hemorrhage. 2014. https://www.ncbi.nlm.nih.gov/pubmed/24524447[↩]
- Reiter R.J., Tan D.X., Galano A. Melatonin: exceeding expectations. Physiology (Bethesda) 2014;29(5):325–333. https://www.ncbi.nlm.nih.gov/pubmed/25180262[↩]
- Gertner S., Greenbaum C.W., Sadeh A., Dolfin Z., Sirota L., Ben-Nun Y. Sleep-wake patterns in preterm infants and 6 month’s home environment: implications for early cognitive development. 2002. https://www.ncbi.nlm.nih.gov/pubmed/12113995[↩]
- Meerlo P., Mistlberger R.E., Jacobs B.L., Heller H.C., McGinty D. New neurons in the adult brain: the role of sleep and consequences of sleep loss. Sleep Med. Rev. 2009;13(3):187–194 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2771197/[↩]
- Polimeni G., Esposito E., Bevelacqua V., Guarneri C., Cuzzocrea S. Role of melatonin supplementation in neurodegenerative disorders. Front. Biosci. (Landmark Ed.) 2014;19:429–446. https://www.ncbi.nlm.nih.gov/pubmed/24389194[↩]
- Di W.L., Kadva A., Johnston A., Silman R. Variable bioavailability of oral melatonin. N. Engl. J. Med. 1997;336(14):1028–1029. http://www.nejm.org/doi/10.1056/NEJM199704033361418[↩]
- Herxheimer A, Petrie KJ. Melatonin for the prevention and treatment of jet lag. Cochrane Database of Systematic Reviews 2002, Issue 2. Art. No.: CD001520. DOI: 10.1002/14651858.CD001520.[↩]
- Malhotra S, Sawhney G, Pandhi P. The Therapeutic Potential of Melatonin: A Review of the Science. Medscape General Medicine. 2004;6(2):46. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1395802/[↩][↩]
- Barchas J, DaCosta F, Spector S. Acute pharmacology of melatonin. Nature. 1967; 214: 919-920. https://www.ncbi.nlm.nih.gov/pubmed/6054984[↩]
- Nordlund JJ, Lerner AB. The effects of oral melatonin on skin color and on the release of pituitary hormones. J Clin Endocrinol Metab. 1977; 45: 768-774 https://www.ncbi.nlm.nih.gov/pubmed/914981[↩]
- Wright J, Aldhous M, Franey C, English J, Arendt J. The effect of exogenous melatonin in endocrine function in man. Clin Endocrinol. 1986; 24: 375-382. https://www.ncbi.nlm.nih.gov/pubmed/3742833[↩]
- Puig-Domingo M, Webb SM, Serrano J, et al. Brief report: melatonin-related hypogonadotropic hypogonadism. N Engl J Med. 1992; 327: 1356-1359. http://www.nejm.org/doi/10.1056/NEJM199211053271905[↩]
- Gwayi N, Bernard RT. The effects of melatonin on sperm motility in vitro in Wistar rats. Andrologia. 2002; 34: 391-396 https://www.ncbi.nlm.nih.gov/pubmed/12472624[↩]
- Luboshitzky R, Shen-Orr Z, Nave R, Lavi S, Lavie P. Melatonin administration alters semen quality in healthy men. J Androl. 2002; 23: 572-578. https://www.ncbi.nlm.nih.gov/pubmed/12065466[↩]
- Silman RE. Melatonin: a contraceptive for the nineties. Eur J Obstet Gynecol Reprod Biol. 1993; 49: 3-9. https://www.ncbi.nlm.nih.gov/pubmed/8365512[↩][↩]
- Mattsson R, Hannsson I, Holmdahl R. Pineal gland in autoimmunity: melatonin-dependent exaggeration of collagen-induced arthritis in mice. Autoimmunity. 1994; 17: 83-86. https://www.ncbi.nlm.nih.gov/pubmed/8025216[↩]
- Sandyk R. Successful treatment of multiple sclerosis with magnetic fields. Int J Neurosci. 1992; 66: 237-250 https://www.ncbi.nlm.nih.gov/pubmed/1305621[↩]
- Blask DE, Cos S, Hill SM, Burns DM, Lemus-Wilson A, Grosso DS. Melatonin action on oncogenesis. In: Fraschini, F, Reiter, RJ, eds. Role of Melatonin and Pineal Peptides in Neuroimmunomodulation. New York, NY: Plenum; 1991:233-240.[↩]
- Beck-Friis J, Kjellman BF, Aperia B, et al. Serum melatonin in relation to clinical variables in patients with major depressive disorder and a hypothesis of a low melatonin syndrome. Acta Psychiatr Scand. 1985; 71: 319-330 https://www.ncbi.nlm.nih.gov/pubmed/4039876[↩]
- Rosenthal NE, James SP, Sack DA, et al. Seasonal affective disorder and phototherapy. N Y Acad Sci Ann. 1985; 453: 260-269. https://www.ncbi.nlm.nih.gov/pubmed/3865586[↩]
- Carman JS, Post RM, Buswell R, Goodwin FK. Negative effects of melatonin on depression. Am J Psychiatry. 1976; 133: 1181-1186 https://www.ncbi.nlm.nih.gov/pubmed/788529[↩]
- Wiechmann AF, O’Steen WK. Melatonin increases photoreceptor susceptibility to light-induced damage. Invest Ophthalmol Visual Sci. 1992; 33: 1894-1902. https://www.ncbi.nlm.nih.gov/pubmed/1582795[↩]
- Tailleux A, Torpier G, Bonnefont-Rousselot D, et al. Daily melatonin supplementation in mice increases atherosclerosis in proximal aorta. Biochem Biophys Res Commun. 2002; 293: 1114-1123. https://www.ncbi.nlm.nih.gov/pubmed/12051775[↩]