Contents
What is mild cognitive impairment
Mild cognitive impairment is a condition in which an individual experiences a mild but noticeable and measurable decline in cognitive abilities, including memory, judgment, language and thinking skills that are greater than normal age-related changes, but the loss doesn’t significantly interfere with your ability to handle everyday activities. Mild cognitive impairment is an intermediate stage between normal cognitive changes that may occur with age and more serious symptoms that indicate dementia. If you have mild cognitive impairment, you may be aware that your memory or mental function has “slipped.” Your family and close friends also may notice a change. But generally these changes aren’t severe enough to significantly interfere with your day-to-day life and usual activities.
Approximately 15 to 20 percent of people age 65 or older have mild cognitive impairment.
Symptoms of mild cognitive impairment include mild memory loss; difficulty with planning or organization; trouble finding words; frequently losing or misplacing things; and forgetting names, conversations, and events. Someone who has mild cognitive impairment may be at greater risk of eventually developing Alzheimer’s disease or another type of dementia, particularly if the degree of memory impairment is significant, but mild cognitive impairment does not always progress to dementia. Symptoms of mild cognitive impairment may remain stable for several years, and even improve over time in some people. In other cases, such as when a medication causes cognitive impairment, mild cognitive impairment is mistakenly diagnosed. Therefore, it’s important that people experiencing cognitive impairment seek help as soon as possible for diagnosis and possible treatment.
Experts classify mild cognitive impairment based on the thinking skills affected:
- Amnestic mild cognitive impairment: mild cognitive impairment that primarily affects memory. A person may start to forget important information that he or she would previously have recalled easily, such as appointments, conversations or recent events.
- Nonamnestic mild cognitive impairment: mild cognitive impairment that affects thinking skills other than memory, including the ability to make sound decisions, judge the time or sequence of steps needed to complete a complex task, or visual perception.
In 2011, the National Institute on Aging and the Alzheimer’s Association published revised guidelines for the diagnosis of Alzheimer’s disease, suggesting that in some cases mild cognitive impairment is an early stage of Alzheimer’s disease or another dementia. The 2011 guidelines recommend finding a biomarker (a measurable biological factor, such as levels of a protein, that indicates the presence or absence of a disease) for people with mild cognitive impairment to learn whether they have brain changes that put them at high risk of developing Alzheimer’s disease and other dementias.
More research is needed on the biological changes associated with normal aging, mild cognitive impairment and Alzheimer’s disease and other dementias to better understand the causes of and risk factors for mild cognitive impairment and the prognosis for those with the condition. If it can be shown that changes in the brain, cerebrospinal fluid and/or blood are caused by physiologic processes associated with Alzheimer’s disease, the revised guidelines recommend a diagnosis of mild cognitive impairment due to Alzheimer’s disease.
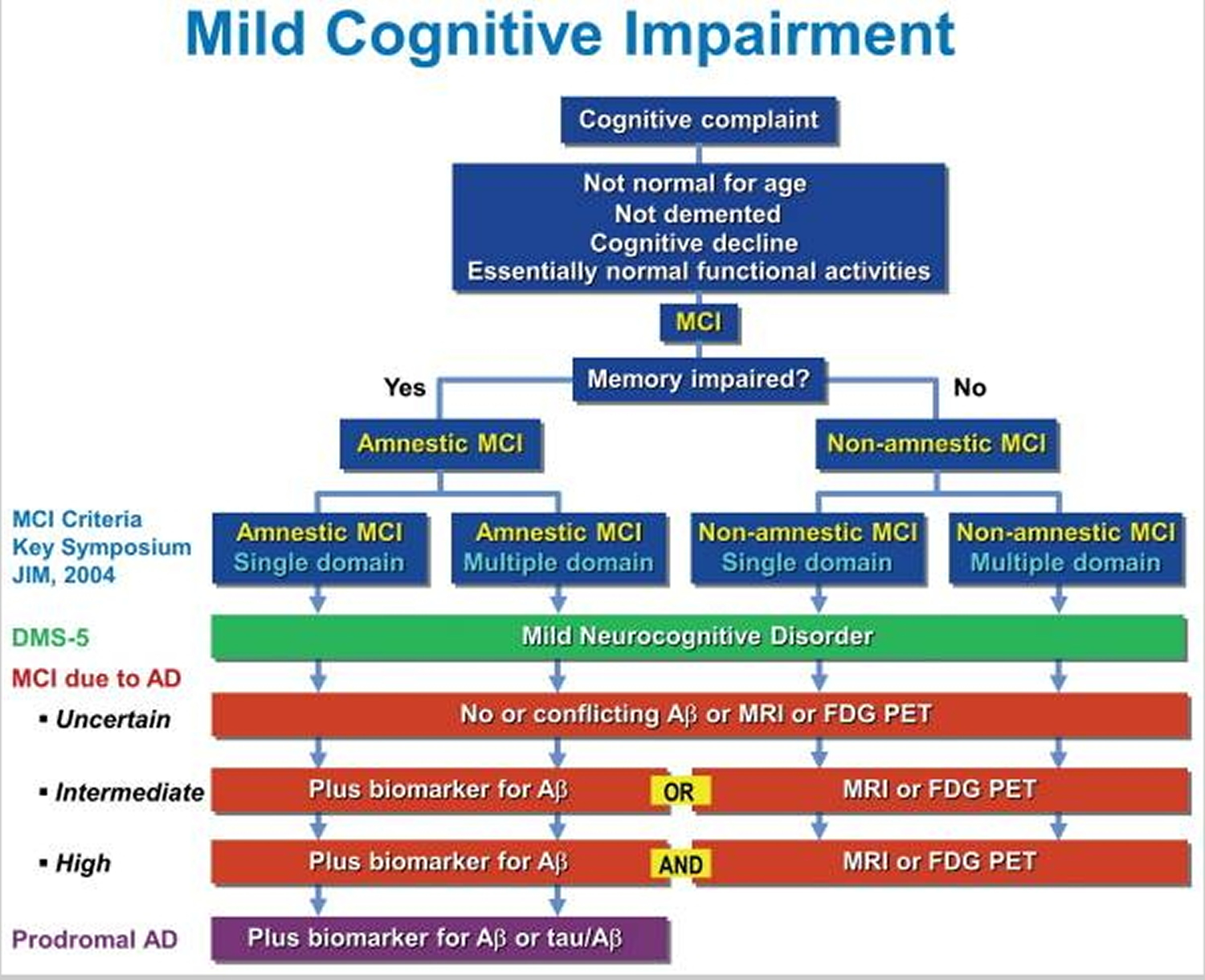
Figure 1 (below) shows a comparison of the current criteria for mild cognitive impairment. As you can see, scientists have not completely characterized and defined mild cognitive impairment and the causes of mild cognitive impairment are not yet completely understood. Experts believe that many mild cognitive impairment cases — but not all — result from brain changes occurring in the very early stages of Alzheimer’s disease or other dementias. Amnestic mild cognitive impairment may be a forerunner of Alzheimer’s disease. Yet mild memory impairments could also be associated with preclinical signs of any dementing illness. Studies have found that individuals diagnosed with amnestic mild cognitive impairment had some type of abnormality affecting key memory regions, and most seemed to be on their way toward meeting standards for Alzheimer’s. Most also had evidence of other abnormalities in addition to the characteristic plaques and tangles of Alzheimer’s disease. The risk factors most strongly linked to mild cognitive impairment are the same as those for dementia: advancing age, family history of Alzheimer’s disease or another dementia, and conditions that raise risk for cardiovascular disease.
It’s not yet possible to tell for certain what the outcome of mild cognitive impairment will be for a specific person or to determine the underlying cause of mild cognitive impairment from a person’s symptoms.
Researchers hope to increase the power to predict mild cognitive impairment outcomes by developing new diagnostic tools to identify and measure underlying brain changes linked to specific types of dementia.
Figure 1. Mild cognitive impairment current diagnostic criteria
Note: Comparison of current criteria for mild cognitive impairment (MCI). The criteria outlined in blue were proposed at the 2003 Working Group on Mild Cognitive Impairment Symposium 1. Other criteria include those of the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) 2 and mild cognitive impairment due to Alzheimer’s disease (AD) 3. Aβ amyloid-beta; MRI,magnetic resonance imaging; FDG PET, fluorodeoxyglucose positron emission tomography; tau, tau protein.
[Source 4]Mild cognitive impairment complications
People with mild cognitive impairment have a significantly increased risk — but not a certainty — of developing dementia. Overall, about 1 to 2 percent of older adults develop dementia every year. Among older adults with mild cognitive impairment, studies suggest that around 10 to 15 percent develop dementia every year.
Mild cognitive impairment causes
There’s no single cause of mild cognitive impairment (MCI), just as there’s no single outcome for the disorder. Symptoms of mild cognitive impairment (MCI) may remain stable for years, progress to Alzheimer’s disease or another type of dementia, or improve over time.
Current evidence indicates that mild cognitive impairment often, but not always, arises from a lesser degree of the same types of brain changes seen in Alzheimer’s disease or other forms of dementia. Some of these changes have been identified in autopsy studies of people with mild cognitive impairment. These changes include:
- Abnormal clumps of beta-amyloid protein (plaques) and microscopic protein clumps of tau characteristic of Alzheimer’s disease (tangles)
- Lewy bodies, which are microscopic clumps of another protein associated with Parkinson’s disease, dementia with Lewy bodies and some cases of Alzheimer’s disease
- Small strokes or reduced blood flow through brain blood vessels
Brain-imaging studies show that the following changes may be associated with mild cognitive impairment:
- Shrinkage of the hippocampus, a brain region important for memory
- Enlargement of the brain’s fluid-filled spaces (ventricles)
- Reduced use of glucose, the sugar that’s the primary source of energy for cells, in key brain regions
Risk factors for mild cognitive impairment
The strongest risk factors for mild cognitive impairment (MCI) are:
- Increasing age
- Having a specific form of a gene known as APOE-e4, also linked to Alzheimer’s disease — though having the gene doesn’t guarantee that you’ll experience cognitive decline
Other medical conditions and lifestyle factors have been linked to an increased risk of cognitive change, including:
- Diabetes
- Smoking
- High blood pressure
- Elevated cholesterol
- Depression
- Lack of physical exercise
- Infrequent participation in mentally or socially stimulating activities
The study of the possible factors that can promote or predict the development of mild cognitive impairment and the different subtypes is relatively new to dementia research and most of the available information has been collected during the last 10 years 4. Also, it is often unclear whether identified factors are true hazards for mild cognitive impairment or merely predictors of its occurrence, including reverse causation.
As expected, the major risk factors associated with cognitive decline and dementia, i.e. older age and lower level of education, have also been repeatedly associated with mild cognitive impairment 5. However, there is no strong agreement on the extent of the association. This is probably due to the fact that neuropsychological test scores used in the mild cognitive impairment classification are generally corrected for age and education. Moreover, mild cognitive impairment is by definition often a transitory state and the setting in which the case series are collected can also impact on risk profiles.
There is also no strong agreement regarding the effect of gender on mild cognitive impairment occurrence; however, some recent findings point towards a possible association with male gender 6. The association between mild cognitive impairment and the well-known ε4 allele of the Alzheimer’s disease susceptibility gene APOE has been widely investigated and findings have been generally consistent with results for dementia and Alzheimer’s disease 7. However, a strong genetic base of mild cognitive impairment is not supported by concordance studies among twins, which showed no difference in level of concordance between monozygotic and dizygotic twins 8. The presence of comorbidities is another widely studied variable in relation to mild cognitive impairment. In particular, the association between mild cognitive impairment and vascular diseases was assessed in several studies, with mixed results. Indeed, while cross-sectional studies have often demonstrated increased odds of mild cognitive impairment in individuals with vascular diseases such as stroke 9 or heart disease 10, the majority of prospective studies showed no association 11. A similar pattern can be observed for diabetes, with the findings of cross-sectional studies suggesting an association 12 and the majority of prospective studies showing no association with incident mild cognitive impairment 13. A possible explanation for these discrepant findings can be identified in competing outcomes, i.e. the elevated risk among patients with vascular disease of death or progression to dementia during the follow-up interval and before mild cognitive impairment can be detected. Neuropsychiatric symptoms are common in individuals with mild cognitive impairment and in particular depression has been extensively studied as a possible risk factor/predictor of mild cognitive impairment 14. Regarding environmental exposures/lifestyle, physical activity is currently considered a key factor in the prevention of mild cognitive impairment 15, as for dementia and cognitive decline, although the degree of preventive potential is unknown and as yet few prospective studies of physical activity and cognitive functioning have been conducted. Moreover, a careful review of the current evidence suggests that sustained physical, social and cognitive activities can all contribute to postponing or preventing mild cognitive impairment 16, and therefore prospective studies to investigate the combined effect, as well as the individual contribution of each of these factors, are warranted.
Dementia prevention
Some risk factors for dementia, such as age and genetics, cannot be changed. But researchers continue to explore the impact of other risk factors on brain health and prevention of dementia. Some of the most active areas of research in risk reduction and prevention include cardiovascular factors, physical fitness, and diet.
- Cardiovascular risk factors: Your brain is nourished by one of your body’s richest networks of blood vessels. Anything that damages blood vessels anywhere in your body can damage blood vessels in your brain, depriving brain cells of vital food and oxygen. Blood vessel changes in the brain are linked to vascular dementia. They often are present along with changes caused by other types of dementia, including Alzheimer’s disease and dementia with Lewy bodies. These changes may interact to cause faster decline or make impairments more severe. You can help protect your brain with some of the same strategies that protect your heart – don’t smoke; take steps to keep your blood pressure, cholesterol and blood sugar within recommended limits; and maintain a healthy weight.
- Physical exercise: Regular physical exercise may help lower the risk of some types of dementia. Evidence suggests exercise may directly benefit brain cells by increasing blood and oxygen flow to the brain.
- Diet: What you eat may have its greatest impact on brain health through its effect on heart health. The best current evidence suggests that heart-healthy eating patterns, such as the Mediterranean diet, also may help protect the brain. A Mediterranean diet includes relatively little red meat and emphasizes whole grains, fruits and vegetables, fish and shellfish, and nuts, olive oil and other healthy fats.
Mild cognitive impairment symptoms
Your brain, like the rest of your body, changes as you grow older. Many people notice gradually increasing forgetfulness as they age. It may take longer to think of a word or to recall a person’s name.
But consistent or increasing concern about your mental performance may suggest mild cognitive impairment (mild cognitive impairment). Cognitive issues may go beyond what’s expected and indicate possible mild cognitive impairment if you experience any or all of the following:
- You forget things more often.
- You forget important events such as appointments or social engagements.
- You lose your train of thought or the thread of conversations, books or movies.
- You feel increasingly overwhelmed by making decisions, planning steps to accomplish a task or interpreting instructions.
- You start to have trouble finding your way around familiar environments.
- You become more impulsive or show increasingly poor judgment.
- Your family and friends notice any of these changes.
If you have mild cognitive impairment, you may also experience:
- Depression
- Irritability and aggression
- Anxiety
- Apathy
Mild cognitive impairment diagnosis
There is no specific test to confirm a diagnosis of mild cognitive impairment (MCI). Mild cognitive impairment is a “clinical” diagnosis representing a doctor’s best professional judgment about the reason for a person’s symptoms. Your doctor will decide whether mild cognitive impairment is the most likely cause of your symptoms based on the information you provide and results of various tests that can help clarify the diagnosis.
If a physician has difficulty confirming a diagnosis of mild cognitive impairment or the cause of mild cognitive impairment, biomarker tests such as brain imaging and cerebrospinal fluid tests may be performed to determine if the individual has mild cognitive impairment due to Alzheimer’s disease.
A medical workup for mild cognitive impairment includes the following core elements:
- Thorough medical history, where the physician documents current symptoms, previous illnesses and medical conditions, and any family history of significant memory problems or dementia.
- Assessment of independent function and daily activities, which focuses on any changes from a person’s usual level of function.
- Input from a family member or trusted friend to provide additional perspective on how function may have changed.
- Assessment of mental status using brief tests designed to evaluate memory, planning, judgment, ability to understand visual information and other key thinking skills.
- In-office neurological examination to assess the function of nerves and reflexes, movement, coordination, balance and senses.
- Evaluation of mood to detect depression; symptoms may include problems with memory or feeling “foggy.” Depression is widespread and may be especially common in older adults.
- Laboratory tests including blood tests and imaging of the brain’s structure.
If the workup doesn’t create a clear clinical picture, the doctor may recommend neuropsychological testing, which involves a series of written or computerized tests to evaluate specific thinking skills.
Many doctors diagnose mild cognitive impairment based on the following criteria developed by a panel of international experts:
- You have problems with memory or another mental function. You may have problems with your memory, planning, following instructions or making decisions. Your own impressions should be corroborated by someone close to you.
- You’ve declined over time. A careful medical history reveals that your ability has declined from a higher level. This change ideally is confirmed by a family member or a close friend.
- Your overall mental function and daily activities aren’t affected. Your medical history shows that your overall abilities and daily activities generally aren’t impaired, although specific symptoms may cause worry and inconvenience.
- Mental status testing shows a mild level of impairment for your age and education level. Doctors often assess mental performance with a brief test such as the Mini-Mental State Examination (MMSE). More-detailed neuropsychological testing may shed additional light on the degree of memory impairment, which types of memory are most affected and whether other mental skills also are impaired.
- Your diagnosis isn’t dementia. The problems that you describe and that your doctor documents through corroborating reports, your medical history or mental status testing aren’t severe enough to be diagnosed as Alzheimer’s disease or another type of dementia.
Neurological exam
As part of your physical exam, your doctor may perform some basic tests that indicate how well your brain and nervous system are working. These tests can help detect neurological signs of Parkinson’s disease, strokes, tumors or other medical conditions that can impair your memory as well as your physical function. The neurological exam may test:
- Reflexes
- Eye movements
- Walking and balance
Lab tests
Blood tests can help rule out physical problems that can affect memory, such as a vitamin B-12 deficiency or an underactive thyroid gland.
Brain imaging
Your doctor may order an MRI or CT scan to check for evidence of a brain tumor, stroke or bleeding.
Mental status testing
Short forms of mental status testing can be done in about 10 minutes. In testing, doctors ask people to conduct several specific tasks and answer several questions, such as naming today’s date or following a written instruction [see The Mini Mental State Examination (MMSE)].
Longer forms of neuropsychological testing can provide additional details about your mental function compared with others’ of a similar age and education level. These tests may also help identify patterns of change that offer clues about the underlying cause of your symptoms.
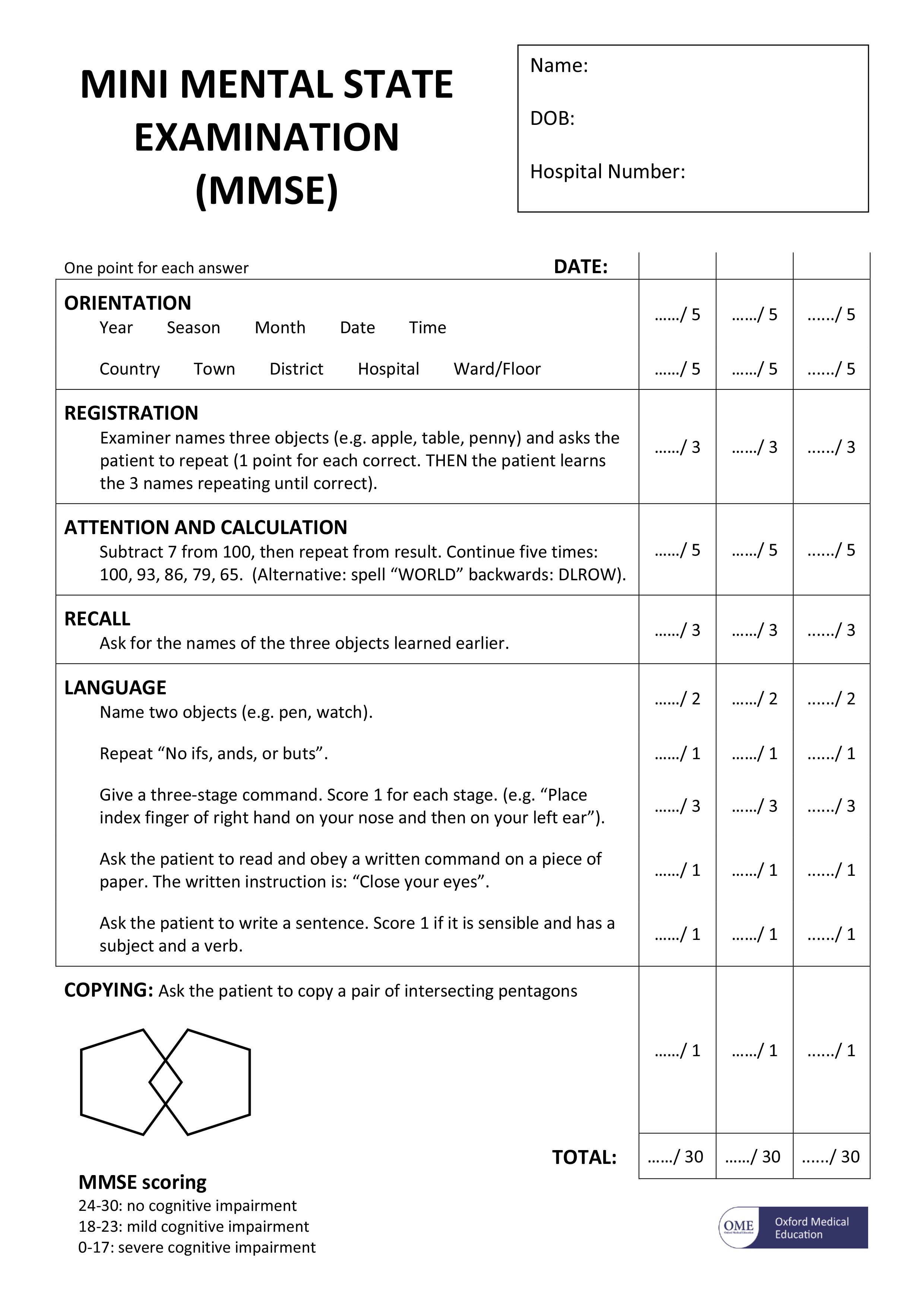
The Mini Mental State Examination (MMSE)
The mini mental state examination (MMSE) assesses a number of different mental abilities, including:
- short- and long-term memory
- attention span
- concentration
- language and communication skills
- ability to plan
- ability to understand instructions
The mini mental state examination (MMSE) is a series of exercises, each carrying a score with a maximum of 30 points. These exercises include:
- memorising a short list of objects and then repeating the list
- writing a short sentence that is grammatically correct, such as “the dog sat on the floor”
- correctly answering time-orientation questions, such as identifying the day of the week, the date or the year
Figure 2. The Mini Mental State Examination (MMSE)
[Source 17]Note: The mini mental state examination (MMSE) is not a test to diagnose dementia. However, it is useful for assessing the level of mental impairment that a person with dementia may have. Test scores may be influenced by a person’s level of education. For example, someone who cannot read or write very well may have a lower score, but they may not have dementia. Similarly, someone with a higher level of education may achieve a higher score, but still have dementia.
The mini mental state examination (MMSE) is a 30-point test
Advantages
- Relatively quick and easy to perform
- Requires no additional equipment
- Can provide a method of monitoring deterioration over time
Disadvantages
- Biased against people with poor education due to elements of language and mathematical testing
- Bias against visually impaired
- Limited examination of visuospatial cognitive ability
- Poor sensitivity at detected mild/early dementia
Mild cognitive impairment treatment
Currently, no mild cognitive impairment (mild cognitive impairment) drugs or other treatments are specifically approved by the Food and Drug Administration (FDA). However, mild cognitive impairment is an active area of research. Clinical studies are underway to shed more light on the disorder and find treatments that may improve symptoms or prevent or delay progression to dementia.
Experts recommend that a person diagnosed with mild cognitive impairment be re-evaluated every six months to determine if symptoms are staying the same, improving or growing worse.
Mild cognitive impairment increases the risk of later developing dementia, but some people with mild cognitive impairment never get worse. Others with mild cognitive impairment later have test results that return to normal for their age and education.
Alzheimer’s drugs
Doctors sometimes prescribe cholinesterase inhibitors – donepezil (Aricept), a type of drug approved for Alzheimer’s disease, for people with mild cognitive impairment whose main symptom is memory loss. However, cholinesterase inhibitors aren’t recommended for routine treatment of mild cognitive impairment. Results of a large, federally funded trial showed that 10 milligrams of donepezil (Aricept) daily reduced the risk of progressing from amnestic mild cognitive impairment to Alzheimer’s disease for about a year, but the benefit disappeared within three years.
The study’s principal investigators said the results were not strong enough to clearly recommend donepezil as a treatment for mild cognitive impairment. However, it might be reasonable for patients and their physicians to talk about the possible benefits and risks of such treatment on an individual basis.
Treating other conditions that can affect mental function
Other common conditions besides mild cognitive impairment can make you feel forgetful or less mentally sharp than usual. Treating these conditions can help improve your memory and overall mental function. Conditions that can affect memory include:
- High blood pressure. People with mild cognitive impairment tend to be more likely to have problems with the blood vessels inside their brains. High blood pressure can worsen these problems and cause memory difficulties. Your doctor will monitor your blood pressure and recommend steps to lower it if it’s too high.
- Depression. When you’re depressed, you often feel forgetful and mentally “foggy.” Depression is common in people with mild cognitive impairment. Treating depression may help improve memory, while making it easier to cope with the changes in your life.
- Sleep apnea. In this condition, your breathing repeatedly stops and starts while you’re asleep, making it difficult to get a good night’s rest. Sleep apnea can make you feel excessively tired during the day, forgetful and unable to concentrate. Treatment can improve these symptoms and restore alertness.
Home remedies
Study results have been mixed about whether diet, exercise or other healthy lifestyle choices can prevent or reverse cognitive decline. Regardless, these healthy choices promote good overall health and may play a role in good cognitive health.
- Regular physical exercise has known benefits for heart health and may also help prevent or slow cognitive decline.
- A diet low in fat and rich in fruits and vegetables is another heart-healthy choice that also may help protect cognitive health.
- Omega-3 fatty acids also are good for the heart. Most research showing a possible benefit for cognitive health uses fish consumption as a yardstick for the amount of omega-3 fatty acids eaten.
- Intellectual stimulation may prevent cognitive decline. Studies have shown computer use, playing games, reading books and other intellectual activities may help preserve function and prevent cognitive decline.
- Social engagement may make life more satisfying, and help preserve mental function and slow mental decline.
- Memory training and other thinking (cognitive) training may help improve your function.
There is some evidence of a possible benefit on mild cognitive impairment from non-pharmacological interventions, such as cognitive training and physical exercise, activities that may be neuroprotective or compensatory. A recent review showed how several studies demonstrated the efficacy of cognitive training in mild cognitive impairment measured as improved performances in tests of global cognitive functioning, memory and meta-memory 18. A limitation of these findings is the small sample sizes of the individual studies. Only seven randomized control trials were identified by a systematic review 19, with a total of 296 mild cognitive impairment subjects who were cognitively treated. Most of these studies in fact included samples of fewer than 50 individuals; therefore, replication of the findings in larger randomized control trials is warranted.
A rapidly growing body of evidence suggests that exercise, specifically aerobic exercise, may attenuate cognitive impairment 20. A systematic review of the effect of aerobic exercise on cognitive performance in individuals with neurological disorders found modest improvements in attention and processing speed, executive function and memory 21. Therefore, as for the case of cognitive training, larger randomized control trials specifically in subjects with mild cognitive impairment, are warranted to confirm or refute these preliminary results. In particular, there is a strong need for evidence regarding the combined effect of multiple non-pharmacological interventions on mild cognitive impairment evolution and ongoing multidomain randomized control trials of mild cognitive impairment are particularly relevant 22. A further possibility is to combine pharmacological and non-pharmacological interventions and evaluate whether their joint effect has more therapeutic value than the individual treatments alone. Such studies could also be combined with therapeutic trials.
Alternative medicine
Some supplements — including vitamin E, ginkgo and others — have been purported to help prevent or delay the progression of mild cognitive impairment. However, no supplement has shown any benefit in a clinical trial.
- Winblad B, Palmer K, Kivipelto M, et al. Mild cognitive impairment—beyond controversies, towards a consensus: report of the International Working Group on Mild Cognitive Impairment. J Intern Med. 2004;256:240–6. https://www.ncbi.nlm.nih.gov/pubmed/15324367[↩]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5. Washington, D.C: American Psychiatric Association; 2013.[↩]
- Albert MS, DeKosky ST, Dickson D, et al. The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7:270–9. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3312027/[↩]
- Petersen RC, Caracciolo B, Brayne C, Gauthier S, Jelic V, Fratiglioni L. Mild cognitive impairment: a concept in evolution. Journal of internal medicine. 2014;275(3):214-228. doi:10.1111/joim.12190. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3967548/[↩][↩]
- Luck T, Luppa M, Briel S, Riedel-Heller SG. Incidence of mild cognitive impairment: a systematic review. Dement Geriatr Cogn Disord. 2010;29:164–75. https://www.karger.com/Article/Pdf/272424[↩]
- Roberts RO, Geda YE, Knopman DS, et al. The incidence of MCI differs by subtype and is higher in men: the Mayo Clinic Study of Aging. Neurology. 2012;78:342–51. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3280046[↩]
- Boyle PA, Buchman AS, Wilson RS, Leurgans SE, Bennett DA. Physical frailty is associated with incident mild cognitive impairment in community-based older persons. J Am Geriatr Soc. 2010;58:248–55. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3150526/[↩]
- Caracciolo B, Gatz M, Xu W, Pedersen NL, Fratiglioni L. Differential distribution of subjective and objective cognitive impairment in the population: a nation-wide twin-study. J Alzheimers Dis. 2012;29:393–403. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4063353/[↩]
- Atti AR, Forlani C, De Ronchi D, Palmer K, Casadio P, Dalmonte E, Fratiglioni L. Cognitive Impairment after Age 60: Clinical and Social Correlates in the “Faenza Project” J Alzheimers Dis. 2010;21:1325–34. https://www.ncbi.nlm.nih.gov/pubmed/21504137[↩]
- Roberts RO, Knopman DS, Geda YE, Cha RH, Roger VL, Petersen RC. Coronary heart disease is associated with non-amnestic mild cognitive impairment. Neurobiol Aging. 2010;31:1894–1902. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2888961/[↩]
- Luck T, Luppa M, Briel S, et al. Mild cognitive impairment: incidence and risk factors: results of the Leipzig longitudinal study of the aged. J Am Geriatr Soc. 2010;58:1903–10. https://www.ncbi.nlm.nih.gov/pubmed/20840461[↩]
- Roberts RO, Geda YE, Knopman DS, et al. Association of duration and severity of diabetes mellitus with mild cognitive impairment. Arch Neurol. 2008;65:1066–73. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2630223/[↩]
- Xu W, Caracciolo B, Wang HX, Winblad B, Backman L, Qiu C, Fratiglioni L. Accelerated progression from mild cognitive impairment to dementia in people with diabetes. Diabetes. 2010;59:2928–35. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2963552/[↩]
- van der Linde RM, Stephan BC, Matthews FE, Brayne C, Savva GM Medical Research Council Cognitive Function and Ageing Study. The presence of behavioral and psychological symptoms and progression to dementia in the cognitively impaired older population. Int J Geriatr Psychiatry. 2013;28:700–9. https://www.ncbi.nlm.nih.gov/pubmed/22887592[↩]
- Ahlskog JE, Geda YE, Graff-Radford NR, Petersen RC. Physical exercise as a preventive or disease-modifying treatment of dementia and brain aging. Mayo Clin Proc. 2011;86:876–84. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3258000/[↩]
- Marioni RE, Valenzuela MJ, van den Hout A, Brayne C, Matthews FE MRC Cognitive Function and Ageing Study. Active cognitive lifestyle is associated with positive cognitive health transitions and compression of morbidity from age sixty-five. PLoS One. 2012;7:e50940. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3521012/[↩]
- Oxford Medical Education. Mini-mental state examination (MMSE). http://www.oxfordmedicaleducation.com/geriatrics/mini-mental-state-examination-mmse/[↩]
- Simon SS, Yokomizo JE, Bottino CM. Cognitive intervention in amnestic Mild Cognitive Impairment: a systematic review. Neurosci Biobehav Rev. 2012;36:1163–78. https://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0048106/[↩]
- Gates NJ, Sachdev PS, Fiatarone Singh MA, Valenzuela M. Cognitive and memory training in adults at risk of dementia: a systematic review. BMC Geriatr. 2011;25;11:55. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3191477/[↩]
- Miller DI, Taler V, Davidson PS, Messier C. Measuring the impact of exercise on cognitive aging: methodological issues. Neurobiol Aging. 2012;33:622.e29–43. https://www.ncbi.nlm.nih.gov/pubmed/21514694[↩]
- McDonnell MN, Smith AE, Mackintosh SF. Aerobic exercise to improve cognitive function in adults with neurological disorders: a systematic review. Arch Phys Med Rehabil. 2011;92:1044–52. https://www.ncbi.nlm.nih.gov/pubmed/21704783[↩]
- Lautenschlager NT, Cox K, Kurz AF. Physical activity and mild cognitive impairment and Alzheimer’s disease. Curr Neurol Neurosci Rep. 2010;10:352–58. https://www.ncbi.nlm.nih.gov/pubmed/20556547[↩]