Contents
What is myocarditis
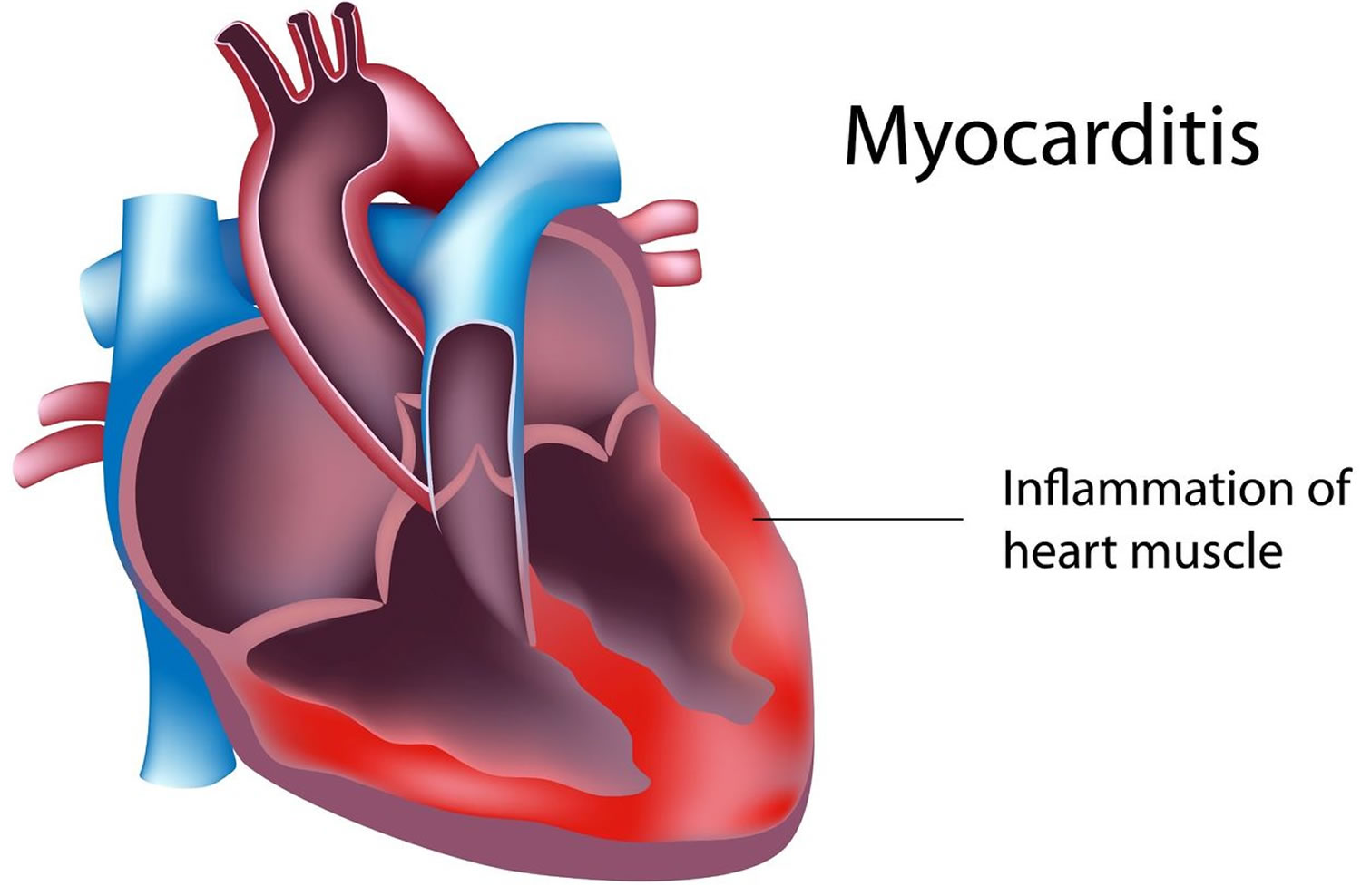
Myocarditis is inflammation of the myocardium – the heart muscle. Myocarditis can affect your heart muscle and your heart’s electrical system, reducing your heart’s ability to pump and causing rapid or abnormal heart rhythms (arrhythmias). Myocarditis is not related to lifestyle and there are no medical treatments to prevent it.
Myocarditis is usually caused by a viral infection, particularly adenovirus (a virus that contains DNA) and enterovirus (a group of viruses that infect the gastrointestinal tract and can spread to other areas) infections, although many infectious organisms commonly seen in infants and children have been implicated. Myocarditis can also result from a reaction to a drug or be part of a more general inflammatory condition. Signs and symptoms of myocarditis include chest pain, fatigue, shortness of breath, and arrhythmias.
Lieberman further classified myocarditis as follows 1:
- Fulminant myocarditis – Follows a viral prodrome; distinct onset of illness consisting of severe cardiovascular compromise with ventricular dysfunction and multiple foci of active myocarditis; either resolves spontaneously or results in death
- Acute myocarditis – Less distinct onset of illness, with established ventricular dysfunction; may progress to dilated cardiomyopathy
- Chronic active myocarditis – Less distinct onset of illness, with clinical and histologic relapses; development of ventricular dysfunction associated with chronic inflammatory changes (including giant cells)
- Chronic persistent myocarditis – Less distinct onset of illness; persistent histologic infiltrate with foci of myocyte necrosis but without ventricular dysfunction (despite symptoms, e.g, chest pain, palpitations)
These terms are still used to describe the clinical presentation and progression of myocarditis, particularly in the absence of ongoing histologic evaluation.
Myocarditis is a rare disease. The frequency of myocarditis is difficult to ascertain, owing to the wide variation of clinical presentation. Incidence varies greatly among countries and is related to hygiene and socioeconomic conditions. Incidence is usually estimated at 1-10 cases per 100,000 persons. Availability of medical services and immunizations also affect incidence. Occasional epidemics of viral infections have been reported with an associated higher incidence of myocarditis. Incidence of positive right ventricular biopsy findings in patients with suspected myocarditis is highly variable (ranging from 0-80%). According to estimates, as many as 1-5% of patients with acute viral infections may have involvement of the myocardium.
A population study in Finland found that, in a study of more than 670,000 healthy young male military recruits, 98 cases had myocarditis mimicking myocardial ischemia, 1 case presented as sudden death, and 9 cases presented as recent-onset dilated cardiomyopathy 2.
A Japanese 20-year series of 377,841 autopsies found idiopathic, nonspecific, interstitial, or viral myocarditis in only 0.11% of individuals 3.
Studies give a wide spectrum of mortality and morbidity statistics. With suspected coxsackievirus B, the mortality rate is higher in newborns (75%) than in older infants and children (10-25%). Complete recovery of ventricular function has been reported in as many as 50% of patients. Some patients develop chronic myocarditis (ongoing or resolving) and/or dilated cardiomyopathy and may eventually require cardiac transplantation.
No particular race predilection is noted for myocarditis except for peripartum cardiomyopathy (a specific form of myocarditis that appears to have a higher incidence in patients of African descent).
The incidence of myocarditis is similar between males and females, although young males are particularly susceptible.
Patients are usually fairly young. The median age of patients affected with lymphocytic myocarditis is 42 years. Patients with giant cell myocarditis may be older (mean age 58 years), but this condition usually does not discriminate with respect to age, sex, or presenting symptoms.
Other susceptible groups include immunocompromised individuals, pregnant women, and children (particularly neonates).
There is no age predilection exists for myocarditis. However, younger patients, especially newborns and infants, and immunocompromised patients may have increased susceptibility to myocarditis.
Some research in laboratory animals suggests that myocarditis may be more aggressive in males than in females. Certain strains of female mice had a reduced inflammatory process when treated with estradiol. In other studies, testosterone appeared to increase cytolytic activity of T lymphocytes in male mice.
It is difficult to diagnose myocarditis as the test involves doing an invasive biopsy of the heart muscle, and it is not very sensitive and can miss the diagnosis. There is also no specific treatment for myocarditis. Treatment for myocarditis depends on the cause. Although scientists know that immune cells called T cells cause inflammation of the heart muscle in myocarditis, they don’t know exactly which T cells are involved and how they are encouraged to travel to the heart.
Most people suffering with myocarditis recover without complications, but in rare cases when inflammation is severe, there can be damage to the heart. Severe myocarditis weakens your heart so that the rest of your body doesn’t get enough blood. Clots can form in your heart, leading to a stroke or heart attack.
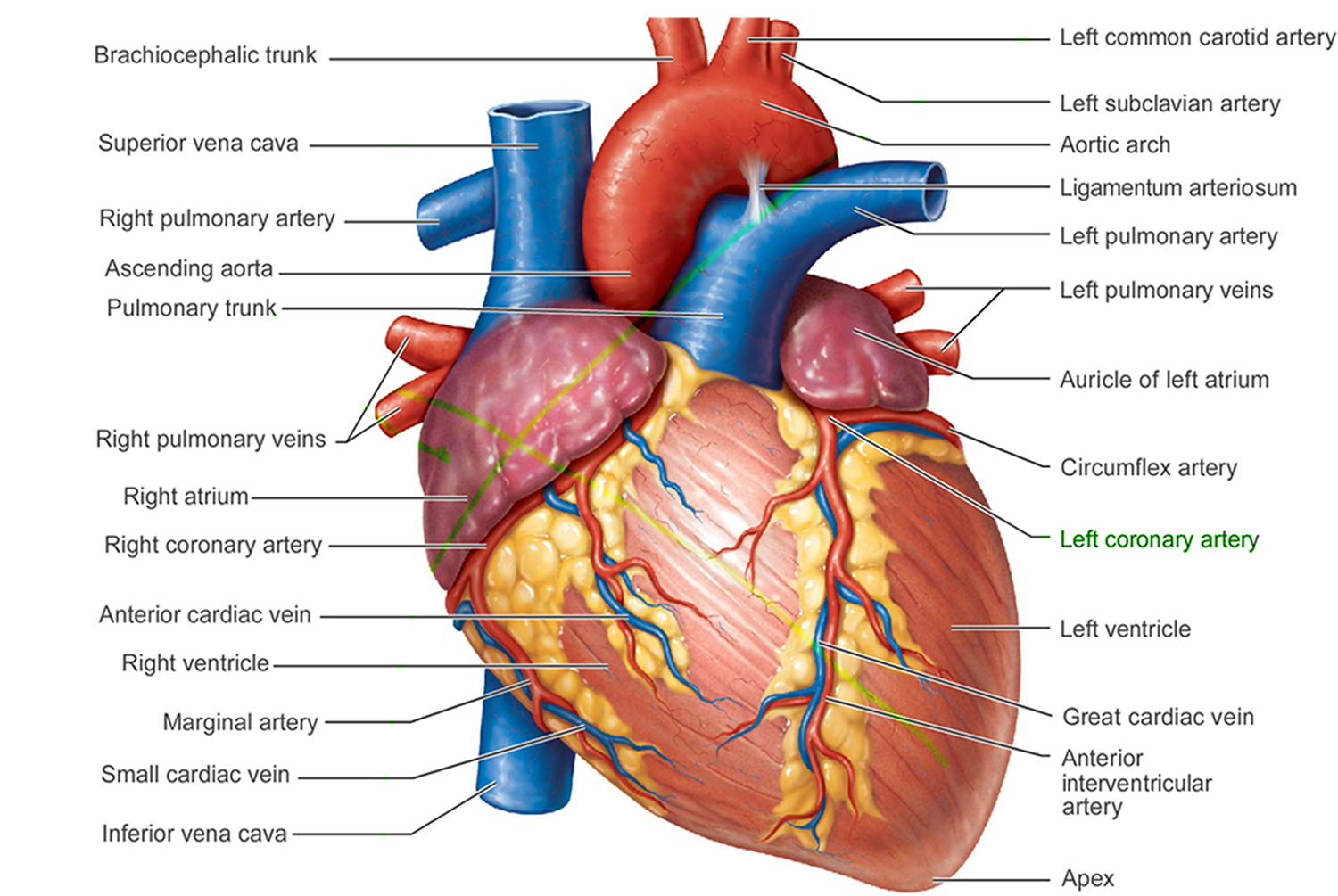
Figure 1. The anatomy of the heart
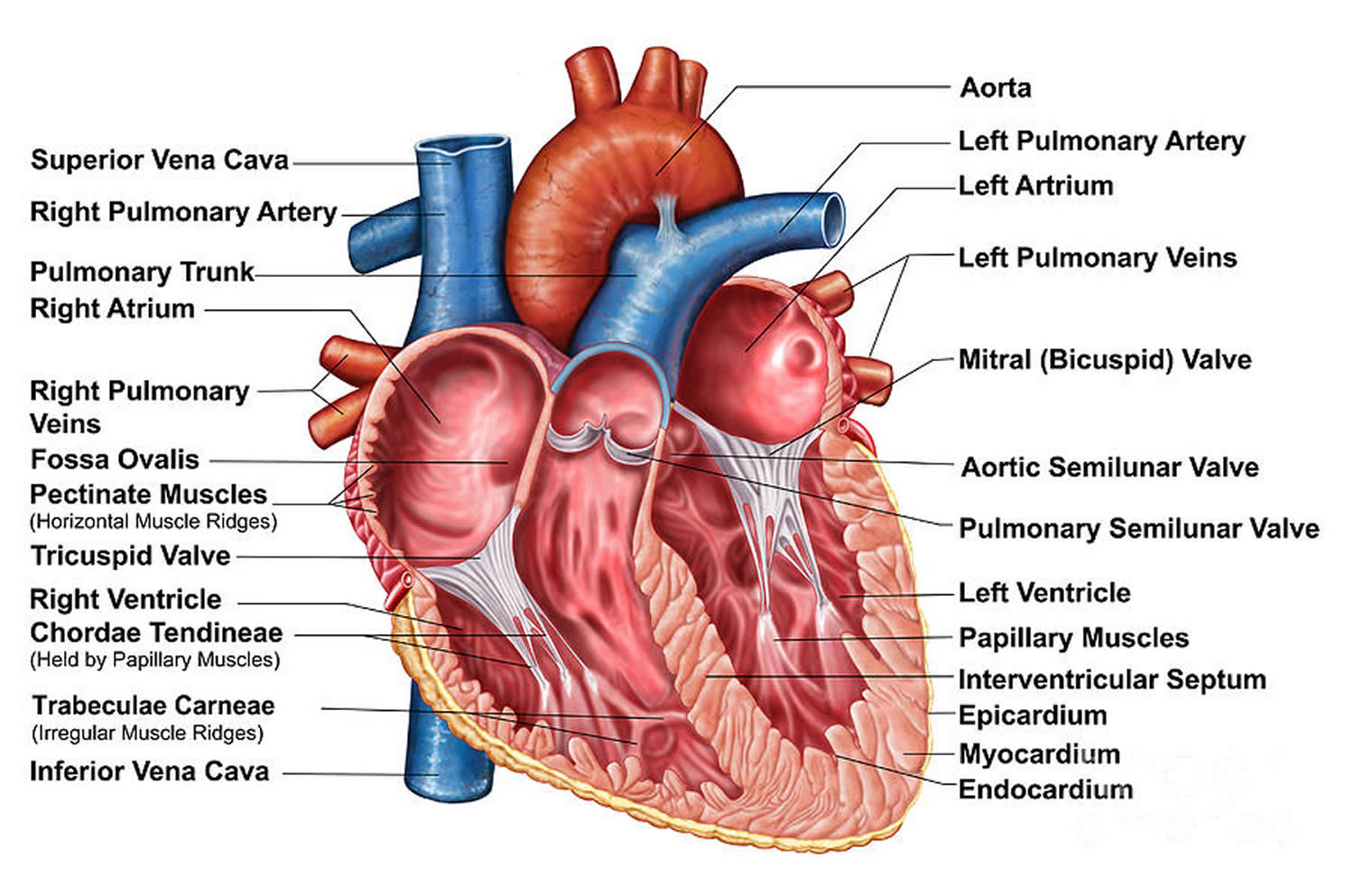
Figure 2. The anatomy of the heart chambers
 Progression of Myocarditis
Progression of Myocarditis
The disease usually develops acutely and may be linked to a causative agent (rarely is a single agent identified). A generalised inflammatory illness ensues and heart failure may develop. Recovery may be spontaneous or intractable heart failure may develop.
Heart blocks and ventricular tachyarrhythmias are associated with some of the more aggressive types of myocarditis.
Eosinophilic myocarditis
Eosinophilic myocarditis represents a rare form of myocardial inflammation with very heterogeneous aetiology 4. Eosinophilic myocarditis is a condition resulting from various eosinophilic diseases, including helminth infection, drug hypersensitivity, systemic vasculitis or idiopathic hypereosinophilic syndromes 5. In developed countries, the most prevalent causes of eosinophilic myocarditis are hypersensitivity or allergic reactions, as well as hematological diseases leading to eosinophilia. Eosinophilic myocarditis may have a variable clinical presentation, ranging from asymptomatic forms to life-threatening conditions. Most patients with eosinophilic myocarditis have marked eosinophilia in peripheral blood. Endomyocardial biopsy needs to be performed in most cases in order to establish a definitive diagnosis of eosinophilic myocarditis. The therapy depends on the underlying aetiology. Immunosuppressive therapy represents the treatment mainstay in the majority of eosinophilic myocarditis forms.
The principal aetiologic factors associated with eosinophilic myocarditis are hypersensitivity or allergic reactions, infections, malignancies, vasculitis, and hypereosinophilic syndromes. In developed countries, eosinophilic myocarditis seems to be predominantly connected with hypersensitivity or allergic reactions due to various stimuli including drug reactions. Drugs that are most frequently associated with eosinophilic myocarditis are listed as follows 6.
Principal Drugs Associated with Eosinophilic Myocarditis 6:
- Antimicrobial drugs (amphotericin B, ampicillin, chloramphenicol, penicillin, tetracycline, streptomycin, cephalosporin, sulfonamides, and antituberculous drugs).
- Antipsychotics (clozapine).
- Anti-inflammatory drugs (indomethacin, oxyphenbutazone, and phenylbutazone).
- Diuretics (acetazolamide, chlorthalidone, hydrochlorothiazide, and spironolactone).
- Angiotensin converting enzyme inhibitors (captopril, enalapril).
- Inotropes (dobutamine, digoxin). There may be an association between eosinophilic myocarditis and dobutamine use, particularly prolonged intravenous administration 7.
- Others (tetanus toxoid, methyldopa, amitriptyline, lenalidomide, and sulfonylurea).
This form of myocarditis has been identified in 0.5% of cases in an unselected autopsy series 8 and in 0.1% of cases among a cohort of patients biopsied for suspected myocarditis 9. The prevalence of eosinophilic myocarditis in patients undergoing heart transplantation differs among published studies; usually it is reported between 3 and 7% 10.
Eosinophilic heart disease includes several types of cardiac damage from acute myocarditis to endomyocardial fibrosis. The degree of heart involvement associated with eosinophilic infiltration of cardiac tissue depends on the stimulus attracting the eosinophils, the duration of eosinophilia, and the degree of eosinophil activation. Deleterious effects are more common in subjects with profound blood eosinophilia (>5000/mm3) 11. Three phases of eosinophilic heart disease are classically described. The first stage represented by EM is due to initial eosinophilic infiltration of the heart and subsequent myocardial necrosis associated with the degranulation of eosinophils. When endomyocardial biopsies are performed, deposits of ECP, MBP, and eosinophil peroxidase have been consistently detected 11. The second phase, known as the thrombotic stage, is mainly associated with a hypercoagulable state associated with increased levels of circulating thrombin. Because eosinophil cationic proteins normally bind to an anionic exosite on thrombomodulin, higher numbers of circulating eosinophils bind available thrombomodulin, causing impaired formation of the thrombomodulin-thrombin complex. Moreover, eosinophils store tissue factor, the main initiator of blood coagulation, in their specific granules. Furthermore, it has been recently shown that tissue factor expression is higher in subjects with hypereosinophilia 12. The third and final phase of eosinophilic myocarditisis represented by fibrotic scarring. Eosinophil-associated fibrosis is observed specifically in the endocardium because endothelial cells are very sensitive to eosinophil granule constituents, especially to eosinophil cationic protein and major basic protein. Eosinophils have the potential to promote fibroblast activation, proliferation, and extracellular matrix production, likely through secretion of transforming growth factor- (TGF-) β and IL-1 13.
Eosinophilia may be associated with a number of neoplastic disorders. It is considered to be reactive in some solid lung, gastrointestinal tract, and urogenital tumors as well as in certain types of hematologic disorders such as T-cell and Hodgkin lymphomas, acute lymphoblastic leukemia, or mastocytosis. Eosinophilia can also be part of the neoplastic clone in hematologic disorders, such as in acute and chronic myeloid leukemia, myelodysplastic syndrome, or other myeloproliferative diseases including polycythemia vera or essential thrombocythemia 14. Reactive eosinophilia can be associated with various microbial agents but it usually represents a sequela of parasitic infections. Protozoal infections caused by Trypanosoma, Toxoplasma, Trichinella, Entamoeba, or Echinococcus are usually among the reported infectious causes of eosinophilic myocarditis 6.
Eosinophilic myocarditis may develop in individuals suffering from certain types of vasculitis, namely, Churg-Strauss syndrome (eosinophilic granulomatosis with polyangiitis). This rare entity is also known as eosinophilic granulomatosis with polyangiitis. The syndrome was first described by Churg and Strauss as a disease characterized by disseminated necrotizing vasculitis with extravascular granulomas occurring among patients with bronchial asthma and tissue eosinophilia. Currently, diagnosis of Churg-Strauss syndrome (eosinophilic granulomatosis with polyangiitis) is based on criteria described by the American College of Rheumatology. According to this classification at least four of the following criteria must be met for diagnosis of Churg-Strauss syndrome (eosinophilic granulomatosis with polyangiitis): (1) marked peripheral eosinophilia >10%, (2) paranasal sinus abnormality, (3) bronchial asthma, (4) nonfixed pulmonary infiltrates, (5) mononeuropathy or polyneuropathy, and (6) extravascular eosinophil infiltration on biopsy findings. Heart involvement is more common in the subgroup of Churg-Strauss syndrome (eosinophilic granulomatosis with polyangiitis) patients with absence of ANCA (anti-neutrophil cytoplasmic antibody). Symptomatic cardiac manifestations occur in 27–47% of Churg-Strauss syndrome (eosinophilic granulomatosis with polyangiitis) cases and represent the major cause of death and poor long-term prognosis in these individuals 15.
Eosinophilic myocarditis can be also caused by a heterogeneous group of hematologic disorders called idiopathic hypereosinophilic syndrome. This rare condition is defined as unexplained peripheral blood eosinophilia (>1500/mm3) persisting for at least six months associated with tissue damage 16. If tissue damage is absent, idiopathic hypereosinophilia is the preferred diagnosis. Dermatologic involvement followed by pulmonary and gastrointestinal manifestations is most common in idiopathic hypereosinophilic syndrome. Heart involvement occurs in approximately 20% of patients with idiopathic hypereosinophilic syndrome and only a minority has cardiac manifestations at the time of initial presentation 17.
Generally, strict restriction of physical activity is recommended in all patients during the acute phase of eosinophilic myocarditis with subsequent exclusion of sporting activities in next 6 months. Pharmacological and nonpharmacological treatment of patients with eosinophilic myocarditis manifesting with heart failure or arrhythmias are managed according to current guidelines.
Specific treatment of eosinophilic myocarditis differs significantly based on its underlying aetiology. In patients with suspicion for hypersensitivity or allergic aetiology of eosinophilic myocarditis, it is of utmost importance to eliminate possible causative factors. If eosinophilic myocarditis is related to infectious agents such as parasites, targeted antimicrobial treatment is obviously essential. In myeloproliferative disorders associated with fusion genes FIP1L1 (FIP 1 like 1), PDGFRA (platelet derived growth factor receptor alpha), and PDGFRB (platelet derived growth factor receptor beta), respectively, tyrosine kinase activity is constitutively present. The administration of the tyrosine kinase inhibitor imatinib is clearly indicated in this situation. Oral treatment with imatinib can effectively suppress but not eliminate the FIP1L1-PDGFRA clone in most patients, although some may experience remissions after imatinib discontinuation. In patients with eosinophilic myocarditis, prophylactic use of steroids during the first days of imatinib treatment is recommended 16. The majority of individuals with eosinophilic myocarditis are treated with immunosuppressive treatment, namely, corticosteroids. Nevertheless, the evidence supporting this widely used therapy in non-Churg-Strauss syndrome patients is modest and is based only on case reports, case series, and small nonrandomized studies 17. Moreover, the initial dosage of corticosteroids and the treatment duration vary among the published studies and thus no clear evidence-based recommendations can be given at this time. It seems reasonable to adjust the dosage of corticosteroids and the treatment duration with respect to the severity of eosinophilic myocarditis manifestation as well as the primary underlying disorder. In patients with Churg-Strauss syndrome, corticosteroids are the mainstay of treatment. Patients with Churg-Strauss syndrome are most frequently treated with 1 mg/kg per day of prednisone or its equivalent administered orally. When a clinical response is reached, usually in several weeks, steroids are tapered down slowly. If a more advanced stage of the disease is present, combined immunosuppressive therapy comprising corticosteroids and cyclophosphamide or azathioprine is usually administered. In a study conducted by Miszalski-Jamka et al. 18, patients suffering from Churg-Strauss syndrome in whom noncorticosteroid immunosuppressive treatment was initiated at the time of diagnosis less frequently had new onset or progression of heart failure in comparison with subjects in whom this therapy was started later on. In patients with Churg-Strauss syndrome or idiopathic hypereosinophilic syndrome there is also evidence showing the usefulness of mepolizumab administration. Mepolizumab is a humanized monoclonal antibody that inhibits binding of IL-5 to its receptor expressed on eosinophils. Initial experience with mepolizumab demonstrates its safety and tolerability; a main advantage is its corticoid-sparing effect 19.
Interestingly, there is also evidence that certain patients with eosinophilic myocarditis do not need to be treated with corticosteroids. In a retrospective study by Yanagisawa et al. 20, which included 22 patients with idiopathic eosinophilia and histologically proven eosinophilic myocarditis and 7 subjects with lymphocytic myocarditis, a similar outcome in terms of LV ejection fraction improvement as well as mortality was observed at 1-year follow-up in both study groups with only conventional heart failure therapy.
Recently, new therapeutic strategies for eosinophil-associated disorders have been suggested. Among the plethora of eosinophils receptors described so far, only several receptors such as IL-5 receptor alpha, chemokine receptor CCR3, and sialic acid-binding immunoglobulin-like lectin 8 are considered to be relatively specific for eosinophil lineage and are thus potentially suitable for antibody targeting 21. Among the drugs affecting these receptors benralizumab, representing humanized antibody to IL-5 receptor alpha, seems to be very promising and has evidence on blood and tissue eosinophilia reduction from several randomized studies 21. Unfortunately, benralizumab similar to other new drugs targeting specifically eosinophil receptors lacks evidence from randomized trial for the treatment of eosinophilic myocarditis.
Giant cell myocarditis
Giant cell myocarditis is a rare and commonly fatal form of fulminant myocarditis of unknown origin 22. The disease usually occurs in previously healthy young people and is typified by rapidly progressive cardiac dysfunction, often requiring cardiac transplantation because of its poor prognosis. Giant cell myocarditis is a rare clinical condition characterized by rapid compromise of cardiac systolic function, ultimately leading to severe cardiogenic shock. It has a grave prognosis with a rate of death or heart transplantation of 70% at 1 year 23. Recently, venoarterial extracorporeal membrane oxygenation (VA-ECMO) has been used as a bridge to cardiac transplantation or recovery 24. Although no current guidelines are available for an optimal monitoring device for patients under extracorporeal membrane oxygenation (ECMO) support, conventional transesophageal echocardiography (TEE) or transthoracic echocardiography (TTE) is commonly used for this purpose 25. Nevertheless, both techniques have limitations 26.
Giant cell myocarditis was first described by Saltykow in 1905 27 and is histologically characterized by diffuse intramyocardial inflammatory infiltrate consisting of lymphocytes, eosinophils and plasma cells, diffuse myocardial necrosis with multinucleated giant cells. Giant cell myocarditis can be idiopathic when any association with another illness is found, or can be secondary to other diseases with a myocardial granulomatous component such as sarcoidosis, mycobacteria or fungal infections and autoimmune diseases. Differentiation of idiopathic giant cell myocarditis from cardiac sarcoidosis might be challenging and perhaps these two entities represent two ends of a disease spectrum as suggested by Stoica et al. 28.
Several experiments suggest that Giant cell myocarditis is an autoimmune disorder dependant on CD4-positive T lymphocytes 29. Until 1996, the multicenter Giant Cell Myocarditis Registry recorded 63 cases from 36 centers. Median survival of such patients was 5.5 months from the onset of symptoms until death or transplantation 30.
Giant cell myocarditis is an uncommon disorder which typically affects young to middle-aged adults, with a slight male preponderance 31). Congestive heart failure is the most common cardiac presentation. On occasion, the initial manifestations may be quite dramatic, including rapidly progressive hemodynamic deterioration, intractable arrythmias and sudden death. Although the early symptoms of Giant cell myocarditis may not be different from other types of acute myocarditis, the condition progresses rapidly to death, often within days to months. Because of this fatal nature of the disease, early right ventricular endomyocardial biopsy in previously healthy patients presenting unexplained acute heart failure should be performed to allow accurate diagnosis and adapted treatment with immunosuppression or heart transplantation 32). They must be repeated because Giant cell myocarditis which may be focal can be undiagnosed if endomyocardial biopsy are performed just once.
Several treatment options have been described in the literature. Immunosuppressive therapy with regimens including cyclosporine, azathioprine or both, but not corticosteroids alone, may be considered but only prolongs the time to transplantation. Indeed, survival of patients presenting Giant cell myocarditis and receiving a double immunosuppressive therapy is less than one year 30. The rapid and precipitous clinical deterioration into congestive heart failure can be bridged to transplantation with mechanical circulatory support, as demonstrated by our case using a peripheral extracorporeal membrane oxygenation (ECMO). Extracorporeal membrane oxygenation (ECMO) permits to stabilize the patient regarding hemodynamic conditions and peripheral organ perfusion during a short period of time. It is also less expensive than mechanical assist devices such as Thoratec Paracorporeal Ventricular Assist Device or Cardiowest Total Artificial Heart used by Davies et al. in patients presenting Giant cell myocarditis myocarditis who were further transplanted within 2 to 53 days after implantation 33. In case of no myocarditis cause being found by endomyocardial biopsy and if no heart recovery is observed within the first 7–10 days after ECMO implantation, the patient can be either bridged to another mechanical assist device or transplanted in emergency.
Heart transplantation may remain the only possibility for prolonged survival, which is almost similar to the overall survival rates for ischemic or dilated cardiomyopathy patients 34. The recurrence of Giant cell myocarditis in cardiac transplants has been described in only 5 cases with good response to increased immunosuppression, which differs dramatically from that in the native heart 35. The post-transplantation care of these patients should probably be different from other heart transplant patients with a more frequent follow-up maintained indefinitely to assess recurrence of Giant cell myocarditis.
Myocarditis prognosis (outlook)
This is usually good and depends somewhat on the underlying causes and origins of the disease. Chronic heart failure or cardiomyopathy may result with poorer outlook.
Patients who survive fulminant myocarditis have a good prognosis. In a study of 147 cases of myocarditis monitored for an average of 5.6 years, 93% of the 15 patients with fulminant disease were alive without transplant 11 years after biopsy, compared with 45% of the 132 patients with less severe disease. Left ventricular dilation was not as severe in the fulminant cases as in the nonfulminant ones 36.
Expression of soluble Fas and Fas ligands at initial presentation appears to be a good serologic marker to predict the prognosis of acute myocarditis, while antimyosin autoantibodies are associated with development of worse cardiac dysfunction in chronic myocarditis 37.
Predictors of death or need for heart transplantation after acute myocarditis in multivariate analyses include syncope, low ejection fraction, and left bundle-branch block, all indicators of advanced cardiomyopathy 38.
Morbidity and mortality
Most patients with mild symptoms recover completely without any residual cardiac dysfunction, although a third subsequently developing dilated cardiomyopathy 2. Cardiogenic shock may occur in fulminant cases of myocarditis.
In the Myocarditis Treatment Trial, the 1-year mortality rate was 20% and the 4-year mortality rate was 56% in a population with symptomatic heart failure presentation and left ventricular ejection fraction lower than 45% at baseline 39. Severe heart block requiring permanent pacemaker placement occurred in 1% of patients in the trial.
In a study of patients with giant cell myocarditis, 89% of patients either died or underwent transplantation, with median survival from symptom onset to death or transplantation being only 5.5 months 40.
A study by Klugman et al 41 reported a 92% survival rate among 216 pediatric patients with myocarditis. According to the investigators, nonsurviving patients were characterized by a greater severity of illness at presentation and a frequent need for extracorporeal membrane oxygenation and other intensive care unit therapies. With regard to postpartum cardiomyopathy, the mortality rate at 1 year can be as high as 50%.
Myocarditis complications
Severe myocarditis can permanently damage your heart muscle, possibly causing:
- Heart failure. Untreated, myocarditis can damage your heart’s muscle so that it can’t pump blood effectively. In severe cases, myocarditis-related heart failure may require a ventricular assist device or a heart transplant.
- Heart attack or stroke. If your heart’s muscle is injured and can’t pump blood, the blood that pools in your heart can form clots. If a clot blocks one of your heart’s arteries, you can have a heart attack. If a blood clot in your heart travels to an artery leading to your brain before becoming lodged, you can have a stroke.
- Rapid or abnormal heart rhythms (arrhythmias). Damage to your heart muscle can cause arrhythmias.
- Sudden cardiac death. Certain serious arrhythmias can cause your heart to stop beating (sudden cardiac arrest). It’s fatal if not treated immediately.
Myocarditis causes
Often, the cause of myocarditis isn’t identified.
Myocardial damage has 2 main phases, as follows:
- Acute phase (first 2 weeks) – Myocyte destruction is a direct consequence of the offending agent, which causes cell-mediated cytotoxicity and cytokine release, contributing to myocardial damage and dysfunction; detection of the causal agent is uncommon during this stage
- Chronic phase (>2 weeks) – Continuing myocyte destruction is autoimmune in nature, with associated abnormal expression of human leukocyte antigen (HLA) in myocytes (and in the case of viral myocarditis, persistence of the viral genome in myocardium)
Potential causes are many, but the likelihood of developing myocarditis is rare. Potential causes include:
- Viruses. Many viruses are commonly associated with myocarditis, including the viruses that cause the common cold (adenovirus); cytomegalovirus ; hepatitis B and C; parvovirus, which causes a mild rash, usually in children (fifth disease); rubeola, varicella, variola/vaccinia, arbovirus, respiratory syncytial virus, yellow fever virus, rabies, parvovirus and herpes simplex virus.Gastrointestinal infections (echoviruses), mononucleosis (Epstein-Barr virus), poliomyelitis, mumps, and German measles (rubella) also can cause myocarditis. It’s also common in people with HIV, the virus that causes AIDS.
- Bacteria. Numerous bacteria can cause myocarditis, including staphylococcus, streptococcus, mycoplasma, tuberculosis, chlamydia, meningococci, brucellosis, clostridia, the bacterium that causes diphtheria, melioidosis, Mycoplasma pneumoniae, psittacosis and the tick-borne bacterium responsible for Lyme disease.
- A chest infection
- Fungal infection
- Parasites. Among these are such parasites as Trypanosoma cruzi, trypanosomiasis, malaria, leishmaniasis, balantidiasis, sarcosporidiosis and toxoplasma, including some that are transmitted by insects and can cause a condition called Chagas disease. This disease is much more prevalent in Central and South America than in the United States, but it can occur in travelers and in immigrants from that part of the world.
- Rickettsial – Scrub typhus, Rocky Mountain spotted fever, Q fever
- Spirochetal – Syphilis, leptospirosis/Weil disease, relapsing fever/Borrelia, Lyme disease
- Fungi. Yeast infections, such as candida; molds, such as aspergillus; cryptococcosis, actinomycosis, blastomycosis, coccidioidomycosis, mucormycosis and other fungi, such as histoplasma, often found in bird droppings, can sometimes cause myocarditis, particularly in people with weakened immune systems.
- Helminthic – Trichinosis, echinococcosis, schistosomiasis, heterophyiasis, cysticercosis, visceral larva migrans, filariasis
- Bites/stings – Scorpion venom, snake venom, black widow spider venom, wasp venom, tick paralysis
- Drugs (usually causing hypersensitivity myocarditis)
- Chemotherapeutic drugs – Doxorubicin and anthracyclines, streptomycin, cyclophosphamide, interleukin-2, anti-HER-2 receptor antibody/Herceptin
- Antibiotics – Penicillin, chloramphenicol, sulfonamides
- Antihypertensive drugs – Methyldopa, spironolactone
- Antiseizure drugs – Phenytoin, carbamazepine
- Amphetamines, cocaine, catecholamines
- Chemicals – Hydrocarbons, carbon monoxide, arsenic, lead, phosphorus, mercury, cobalt
- Physical agents (radiation, heatstroke, hypothermia)
- Acute rheumatic fever
- Systemic inflammatory disease – Giant cell myocarditis, sarcoidosis, Kawasaki disease, Crohn disease, systemic lupus erythematosus, ulcerative colitis,
- Wegener granulomatosis, thyrotoxicosis, scleroderma, rheumatoid arthritis
- Peripartum cardiomyopathy
- Posttransplant cellular rejection
Myocarditis also sometimes occurs if you’re exposed to:
- Medications or illegal drugs that might cause an allergic or toxic reaction. These include drugs used to treat cancer; antibiotics, such as penicillin and sulfonamide drugs; some anti-seizure medications; and some illegal substances, such as cocaine.
- Chemicals or radiation. Exposure to certain chemicals, such as carbon monoxide, and radiation can sometimes cause myocarditis.
- Other diseases. An auto immune disease (when a person’s own immune system attacks their body). These include disorders such as lupus, Wegener’s granulomatosis, giant cell arteritis and Takayasu’s arteritis.
Viral myocarditis
In viral myocarditis, viral isolates differ in tissue tropism and virulence. For example, coxsackievirus A9 is a self-limiting myocarditis, whereas coxsackievirus B3 causes severe myocarditis resulting in a high mortality rate. The induction of the coxsackie-adenovirus receptor (CAR) and the complement deflecting protein decay accelerating factor (DAF, CD55) may allow efficient internationalization of the viral genome.
Viral replication may lead to further disruption of metabolism and to perturbation of inflammation and its response. Vasospasm induced by endothelial cell viral infection may also contribute to further damage 42.
New evidence of dystrophin disruption by expression of enteroviral protease 2A points to yet another unique pathogenic mechanism. 43 In contrast, some viruses (such as parvovirus B19) may focus on pericapillary depositions, contributing to diastolic dysfunction rather than to direct myocyte destruction. Regardless, viral persistence provides the necessary stimuli for autoimmune or other inflammatory responses.
Idiopathic myocarditis
Approximately 50% of the time, myocarditis is classified as idiopathic, although a report by Klugman et al 41 found that 82% of the pediatric cases studied were considered idiopathic. The investigators also determined that 3% of cases in the study had a known bacterial or viral etiology, and that 6% of cases were related to other diseases.
In idiopathic cases, a viral etiology is often suspected but unproved, even with sophisticated immunohistochemical and genomic studies. Studies on patients with idiopathic dilated cardiomyopathy found evidence of viral particles in endomyocardial biopsy specimens in up to two thirds of the patients 44.
Risk Factors for Myocarditis
These include:
- 1) Viral infections: Coxsackie, Influenza, Rubella, Polio, Adeno and HIV have all been implicated.
- 2) Bacterial infections with agents such as Corynebacterium, Rickettsia, Chlamydia and Coxiella.
- 3) Protozoal infections: Trypanosoma Cruzi which causes Chagas’ disease and is common in South America and Toxoplasma Gondii are both known to cause myocarditis.
- 4) Radiation and poisoning can cause myocarditis (agents such as lead).
- 5) Rarely, autoimmune processes may cause myocarditis.
Myocarditis prevention
There’s no specific prevention for myocarditis. However, taking these steps to prevent infections might help:
- Avoid people who have a viral or flu-like illness until they’ve recovered. If you’re sick with viral symptoms, try to avoid exposing others.
- Follow good hygiene. Regular hand-washing can help prevent spreading illness.
- Avoid risky behaviors. To reduce your chances of getting an HIV-related myocardial infection, practice safe sex and don’t use illegal drugs.
- Minimize exposure to ticks. If you spend time in tick-infested areas, wear long-sleeved shirts and long pants to cover as much of your skin as possible. Apply tick or insect repellents that contain DEET.
- Get your vaccines. Stay up to date on the recommended vaccines, including those that protect against rubella and influenza — diseases that can cause myocarditis.
Myocarditis symptoms
People who have myocarditis usually develop symptoms one to two weeks after the initial viral illness.
If you have a mild case of myocarditis or are in the early stages, you might have no symptoms or mild ones, such as chest pain or shortness of breath.
In serious cases, the signs and symptoms of myocarditis vary, depending on the cause of the disease.
Common myocarditis signs and symptoms include:
- A stabbing pain and/or tightness in the chest which may spread across the body
- Rapid or abnormal heart rhythms (arrhythmias)
- Shortness of breath when lightly exercising or walking
- Difficulty breathing when resting
- Flu like symptoms such as a high temperature, tiredness and fatigue
- Fluid retention with swelling of your legs, ankles and feet
- Fatigue
- Other signs and symptoms of a viral infection, such as a headache, body aches, joint pain, fever, a sore throat or diarrhea.
Myocarditis in children
When children develop myocarditis, they might have signs and symptoms including:
- Fever
- Fainting
- Breathing difficulties
- Rapid breathing
- Rapid or abnormal heart rhythms (arrhythmias).
Myocarditis diagnosis
The diagnosis of acute myocarditis is usually presumptive, based on patient demographics and the clinical course. Because many cases of myocarditis are not clinically obvious, a high degree of suspicion is required.
Patients with myocarditis usually present with signs and symptoms of acute decompensation of heart failure (e.g, tachycardia, gallop, mitral regurgitation, edema) and, in those with concomitant pericarditis, with pericardial friction rub.
Specific findings in special cases are as follows:
- Sarcoid myocarditis: Lymphadenopathy, also with arrhythmias, sarcoid involvement in other organs (up to 70%)
- Acute rheumatic fever: Usually affects heart in 50-90%; associated signs, such as erythema marginatum, polyarthralgia, chorea, subcutaneous nodules (Jones criteria)
- Hypersensitive/eosinophilic myocarditis: Pruritic maculopapular rash and history of using offending drug
- Giant cell myocarditis: Sustained ventricular tachycardia in rapidly progressive heart failure
- Peripartum cardiomyopathy – Heart failure developing in the last month of pregnancy or within 5 months following delivery
Early diagnosis is key to preventing long-term heart damage. After a physical examination, your doctor might order one or more tests to confirm that you have myocarditis and determine its severity. Tests might include:
- Chest x-ray: there may be some cardiac enlargement depending on the cause and stage.
- An electrocardiogram (ECG) to check for an abnormal heart rhythm, if you have experienced palpitations. This noninvasive test shows your heart’s electrical patterns and can detect abnormal rhythms.
- An echocardiogram (echo) if your doctor is concerned there may be heart muscle damage. Sound waves create moving images of the beating heart. An echocardiogram might detect enlargement of your heart, poor pumping function, valve problems, a clot within the heart or fluid around your heart.
- MRI. Cardiac MRI will show your heart’s size, shape and structure. This test can show signs of inflammation of the heart muscle.
- Blood tests to check for infection. These measure white and red blood cell counts, as well as levels of certain enzymes that indicate damage to your heart muscle. Blood tests can also detect antibodies against viruses and other organisms that might indicate a myocarditis-related infection.
- Cardiac catheterization and endomyocardial biopsy. A small tube (catheter) is inserted into a vein in your leg or neck and threaded into your heart. In some cases, doctors use a special instrument to remove a tiny sample of heart muscle tissue (biopsy) for analysis in the lab to check for inflammation or infection.
- Endomyocardial biopsy is the standard tool for diagnosing myocarditis. However, the use of routine endomyocardial biopsy in establishing the diagnosis of myocarditis rarely is helpful clinically, since histologic diagnosis seldom has an impact on therapeutic strategies, unless giant cell myocarditis is suspected.
Myocarditis treatment
In many cases, myocarditis improves on its own or with treatment, leading to a complete recovery. Myocarditis treatment focuses on the cause and the symptoms, such as heart failure.
In mild cases, persons should avoid competitive sports for at least three to six months. Rest and medication to help your body fight off the infection causing myocarditis might be all you need. Although antiviral medications are available, they haven’t proved effective in the treatment of most cases of myocarditis.
Certain rare types of viral myocarditis, such as giant cell and eosinophilic myocarditis, respond to corticosteroids or other medications to suppress your immune system. In some cases caused by chronic illnesses, such as lupus, treatment is directed at the underlying disease.
In prolonged cases, myocarditis might affect your heart muscle and tissue, meaning you could develop heart failure. Treatment involves close monitoring and medication, including anti-inflammatory medicines and, depending on your condition, antibiotics.
Rarely, a heart transplant may be needed if the heart muscle has become very weak or you have other health problems.
Treatment is aimed at the cause of the problem, and may involve:
- Antibiotics to fight bacterial infection
- Medicines called steroids to reduce swelling
- Intravenous immunoglobulin (IVIG), a medicine made of substances (called antibodies) that the body produces to fight infection, to control the inflammatory process
- Diuretics to remove excess water from the body
- Low-salt diet
- Reduced activity
Drugs to help your heart
If myocarditis is causing heart failure or arrhythmias, your doctor might hospitalize you and prescribe drugs or other treatments. For certain abnormal heart rhythms or severe heart failure, you may be given medications to reduce the risk of blood clots forming in your heart.
If your heart is weak, your doctor might prescribe medications to reduce your heart’s workload or help you eliminate excess fluid, including:
- Angiotensin-converting enzyme (ACE) inhibitors. These medications, such as enalapril (Vasotec), captopril (Capoten), lisinopril (Zestril, Prinivil) and ramipril (Altace), relax the blood vessels in your heart and help blood flow more easily.
- Angiotensin II receptor blockers (ARBs). These medications, such as losartan (Cozaar) and valsartan (Diovan), relax the blood vessels in your heart and help blood flow more easily.
- Beta blockers. Beta blockers, such as metoprolol (Lopressor, Toprol-XL), bisoprolol and carvedilol (Coreg), work in multiple ways to treat heart failure and help control arrhythmias.
- Diuretics. These medications, such as furosemide (Lasix), relieve sodium and fluid retention.
Treating severe cases
In some severe cases of myocarditis, aggressive treatment might include:
- Intravenous (IV) medications. These might improve the heart-pumping function more quickly.
- Ventricular assist devices. Ventricular assist devices (VADs) are mechanical pumps that help pump blood from the lower chambers of your heart (the ventricles) to the rest of your body. VADs are used in people who have weakened hearts or heart failure. This treatment may be used to allow the heart to recover or while waiting for other treatments, such as a heart transplant.
- Intra-aortic balloon pump. Doctors insert a thin tube (catheter) in a blood vessel in your leg and guide it to your heart using X-ray imaging. Doctors place a balloon attached to the end of the catheter in the main artery leading out to the body from the heart (aorta). As the balloon inflates and deflates, it helps to increase blood flow and decrease the workload on the heart.
- Extracorporeal membrane oxygenation (ECMO). With severe heart failure, this device can provide oxygen to the body. When blood is removed from the body, it passes through a special membrane in the extracorporeal membrane oxygenation (ECMO) machine that removes carbon dioxide and adds oxygen to the blood. The newly oxygenated blood is then returned to the body.
The extracorporeal membrane oxygenation (ECMO) machine takes over the work of the heart. This treatment may be used to allow the heart to recover or while waiting for other treatments, such as a heart transplant.
In the most severe cases, doctors might consider urgent heart transplantation.
Some people might have chronic and irreversible damage to the heart muscle requiring lifelong medications, while other people need medications for just a few months and then recover completely. Either way, your doctor is likely to recommend regular follow-up appointments, including tests to evaluate your condition.
Lifestyle and home remedies
Rest and reducing the workload on your heart is an important part of recovery. Your doctor will likely tell you what type of physical activity you can do during the months your heart is healing and when you can resume normal activities.
Patients should consume a low-sodium diet similar to that for heart failure management. Bedrest and avoidance of athletic activities are recommended from anecdotal experiences (with lower incidence of arrhythmia).
If you have lingering heart damage, it’s important to keep salt to a minimum, restrict the amount of fluids you drink, drink a minimum amount of alcohol, if any, and avoid smoking. Your doctor will let you know what your fluid intake should be, as well as the amount of salt that’s okay for you to include in your diet.
- Feldman AM, McNamara D. Myocarditis. N Engl J Med. 2000 Nov 9. 343(19):1388-98.[↩]
- Durani Y, Egan M, Baffa J, et al. Pediatric myocarditis: presenting clinical characteristics. Am J Emerg Med. 2009 Oct. 27(8):942-7.[↩][↩]
- Wakafuji S, Okada R. Twenty year autopsy statistics of myocarditis incidence in Japan. Jpn Circ J. 1986 Dec. 50(12):1288-93.[↩]
- Kuchynka P, Palecek T, Masek M, et al. Current Diagnostic and Therapeutic Aspects of Eosinophilic Myocarditis. BioMed Research International. 2016;2016:2829583. doi:10.1155/2016/2829583. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4738989/[↩]
- Sohn K-H, Song W-J, Kim B-K, et al. Eosinophilic myocarditis: case series and literature review. Asia Pacific Allergy. 2015;5(2):123-127. doi:10.5415/apallergy.2015.5.2.123. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4415178/[↩]
- Baandrup U. Eosinophilic myocarditis. Herz. 2012;37(8):849–853. doi: 10.1007/s00059-012-3701-2. https://www.ncbi.nlm.nih.gov/pubmed/23179050[↩][↩][↩]
- Spear G. S. Eosinophilic explant carditis with eosinophilia:? Hypersensitivity to dobutamine infusion. Journal of Heart and Lung Transplantation. 1995;14(4):755–760. https://www.ncbi.nlm.nih.gov/pubmed/7578186[↩]
- Al Ali A. M., Straatman L. P., Allard M. F., Ignaszewski A. P. Eosinophilic myocarditis: case series and review of literature. Canadian Journal of Cardiology. 2006;22(14):1233–1237. doi: 10.1016/s0828-282x(06)70965-5. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2569073/[↩]
- Maisch B., Pankuweit S. Current treatment options in (peri)myocarditis and inflammatory cardiomyopathy. Herz. 2012;37(6):644–656. doi: 10.1007/s00059-012-3679-9. https://www.ncbi.nlm.nih.gov/pubmed/22996288[↩]
- Yoshizawa S., Kato T. S., Mancini D., Marboe C. C. Characteristics of patients with advanced heart failure having eosinophilic infiltration of the myocardium in the recent era: investigation of explanted hearts for transplantation. International Heart Journal. 2013;54(3):146–148. doi: 10.1536/ihj.54.146. https://www.jstage.jst.go.jp/article/ihj/54/3/54_146/_pdf/-char/en[↩]
- Oakley C. M., Olsen E. G. J. Eosinophilia and heart disease. British Heart Journal. 1977;39(3):233–237. doi: 10.1136/hrt.39.3.233. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC483226/pdf/brheartj00229-0001.pdf[↩][↩]
- Cugno M., Marzano A. V., Lorini M., Carbonelli V., Tedeschi A. Enhanced tissue factor expression by blood eosinophils from patients with hypereosinophilia: a possible link with thrombosis. PLoS ONE. 2014;9(11) doi: 10.1371/journal.pone.0111862.e111862 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4222944[↩]
- Séguéla P. E., Iriart X., Acar P., Montaudon M., Roudaut R., Thambo J. B. Eosinophilic cardiac disease: molecular, clinical and imaging aspects. Archives of Cardiovascular Diseases. 2015;108(4):258–268. doi: 10.1016/j.acvd.2015.01.006. https://www.sciencedirect.com/science/article/pii/S1875213615000182[↩]
- Gotlib J. Molecular classification and pathogenesis of eosinophilic disorders: 2005 update. Acta Haematologica. 2005;114(1):7–25. doi: 10.1159/000085559. https://www.ncbi.nlm.nih.gov/pubmed/15995322[↩]
- Greco A., Rizzo M. I., De Virgilio A., et al. Churg-Strauss syndrome. Autoimmunity Reviews. 2015;14(4):341–348. doi: 10.1016/j.autrev.2014.12.004. https://www.ncbi.nlm.nih.gov/pubmed/25500434[↩]
- Akuthota P., Weller P. F. Spectrum of eosinophilic end-organ manifestations. Immunology and Allergy Clinics of North America. 2015;35(3):403–411. doi: 10.1016/j.iac.2015.04.002. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4515759/[↩][↩]
- Ogbogu P. U., Bochner B. S., Butterfield J. H., et al. Hypereosinophilic syndrome: a multicenter, retrospective analysis of clinical characteristics and response to therapy. Journal of Allergy and Clinical Immunology. 2009;124(6):1319.e3–1325.e3. doi: 10.1016/j.jaci.2009.09.022. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2829669/[↩][↩]
- Miszalski-Jamka T., Szczeklik W., Sokołowska B., et al. Noncorticosteroid immunosuppression limits myocardial damage and contractile dysfunction in eosinophilic granulomatosis with polyangiitis (Churg-Strauss syndrome) Journal of the American College of Cardiology. 2015;65(1):103–105. doi: 10.1016/j.jacc.2014.08.055. https://www.sciencedirect.com/science/article/pii/S0735109714068405[↩]
- Rothenberg M. E., Klion A. D., Roufosse F. E., et al. Treatment of patients with the hypereosinophilic syndrome with mepolizumab. The New England Journal of Medicine. 2008;358(12):1215–1228. doi: 10.1056/nejmoa070812 http://www.nejm.org/doi/full/10.1056/NEJMoa070812[↩]
- Yanagisawa T., Inomata T., Watanabe I., et al. Clinical significance of corticosteroid therapy for eosinophilic myocarditis. International Heart Journal. 2011;52(2):110–113. doi: 10.1536/ihj.52.110. https://www.jstage.jst.go.jp/article/ihj/52/2/52_2_110/_pdf/-char/en[↩]
- Legrand F., Klion A. D. Biologic therapies targeting eosinophils: current status and future prospects. The Journal of Allergy and Clinical Immunology: In Practice. 2015;3(2):167–174. doi: 10.1016/j.jaip.2015.01.013. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4418449/[↩][↩]
- Cooper L, Berry GJ, Shabetai R for the Multicenter Giant Cell Myocarditis Study Group Investigators . Idiopathic giant‐cell myocarditis—natural history and treatment. N Engl J Med 1997; 336: 1860–1866. http://www.nejm.org/doi/full/10.1056/NEJM199706263362603[↩]
- Ripoll JG, Ratzlaff RA, Menke DM, Olave MC, Maleszewski JJ, Díaz-Gómez JL. Hemodynamic Transesophageal Echocardiography-Guided Venous-Arterial Extracorporeal Membrane Oxygenation Support in a Case of Giant Cell Myocarditis. Case Reports in Critical Care. 2016;2016:5407597. doi:10.1155/2016/5407597. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5014940/[↩]
- Le Guyader A., Rollé F., Karoutsos S., Cornu E. Acute myocarditis supported by extracorporeal membrane oxygenation successfully bridged to transplantation: a giant cell myocarditis. Interactive Cardiovascular and Thoracic Surgery. 2006;5(6):782–784. doi: 10.1510/icvts.2006.136697. https://academic.oup.com/icvts/article/5/6/782/719735[↩]
- Doufle G., Roscoe A., Billia F., Fan E. Echocardiography for adult patients supported with extracorporeal membrane oxygenation. Critical Care. 2015;19, article 326 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4591622/[↩]
- Vignon P. Hemodynamic assessment of critically ill patients using echocardiography Doppler. Current Opinion in Critical Care. 2005;11(3):227–234. doi: 10.1097/01.ccx.0000159946.89658.51. https://www.ncbi.nlm.nih.gov/pubmed/15928471[↩]
- Saltykow S. Uber diffuse MyoKarditis, Virchows Arch Pathol Anat , 1905, vol. 182,pg. 1-39[↩]
- Stoica SC, Goddard M, Tsui S, Dunning J, McNeil K, Parameshwar J, Large SR. Ventricular assist surprise: giant cell myocarditis or sarcoidosis, J Thorac Cardiovasc Surg , 2003, vol. 126 (pg. 2072-2074).[↩]
- Neu N, Pummerer C, Rieker T, Berger P. T cells in cardiac myosin-induced myocarditis, Clin Immunol Immunopathol , 1993, vol. 68, pg. 107-110[↩]
- Cooper LT, Berry GJ, Shabetai R. Idiopathic giant-cell myocarditis – natural history and treatment, N Eng J Med , 1997, vol. 336, pg. 1860-1866[↩][↩]
- Cooper LT, Berry GJ, Shabetai R. Idiopathic giant-cell myocarditis – natural history and treatment, N Eng J Med , 1997, vol. 336 (pg. 1860-1866[↩]
- Moloney ED, Egan JJ, Kelly P, Wood AE, Cooper LTJr. Transplantation for myocarditis: a controversy revisited, J Heart Lung Transplant , 2005, vol. 24 (pg. 1103-1110[↩]
- Davies RA, Veinot JP, Smith S, Struthers C, Hendry P, Masters R. Giant cell myocarditis: clinical presentation, bridge to transplantation with mechanical circulatory support, and long-term outcome, J Heart Lung Transplant , 2002, vol. 21, pg. 674-679[↩]
- Moloney ED, Egan JJ, Kelly P, Wood AE, Cooper LTJr. Transplantation for myocarditis: a controversy revisited, J Heart Lung Transplant , 2005, vol. 24, pg. 1103-1110[↩]
- Scott RL, Ratliff NB, Starling RC, Young JB. Recurrence of giant cell myocarditis in cardiac allograft, J Heart Lung Transplant , 2001, vol. 20 , pg. 375-380[↩]
- Pulerwitz TC, Cappola TP, Felker GM, et al. Mortality in primary and secondary myocarditis. Am Heart J. 2004 Apr. 147(4):746-50.[↩]
- Lauer B, Schannwell M, Kuhl U, et al. Antimyosin autoantibodies are associated with deterioration of systolic and diastolic left ventricular function in patients with chronic myocarditis. J Am Coll Cardiol. 2000 Jan. 35(1):11-8.[↩]
- Fuse K, Kodama M, Okura Y, et al. Predictors of disease course in patients with acute myocarditis. Circulation. 2000 Dec 5. 102(23):2829-35.[↩]
- Mason JW, O’Connell JB, Herskowitz A, et al. A clinical trial of immunosuppressive therapy for myocarditis. The Myocarditis Treatment Trial Investigators. N Engl J Med. 1995 Aug 3. 333(5):269-75.[↩]
- Cooper LT Jr, Berry GJ, Shabetai R. Idiopathic giant-cell myocarditis–natural history and treatment. Multicenter Giant Cell Myocarditis Study Group Investigators. N Engl J Med. 1997 Jun 26. 336(26):1860-6.[↩]
- Klugman D, Berger JT, Sable CA, He J, Khandelwal SG, Slonim AD. Pediatric patients hospitalized with myocarditis: a multi-institutional analysis. Pediatr Cardiol. 2010 Feb. 31(2):222-8.[↩][↩]
- Bowles NE, Towbin JA. Molecular aspects of myocarditis. Curr Opin Cardiol. 1998 May. 13(3):179-84.[↩]
- Badorff C, Knowlton KU. Dystrophin disruption in enterovirus-induced myocarditis and dilated cardiomyopathy: from bench to bedside. Med Microbiol Immunol (Berl). 2004 May. 193(2-3):121-6.[↩]
- Kuhl U, Pauschinger M, Noutsias M, et al. High prevalence of viral genomes and multiple viral infections in the myocardium of adults with “idiopathic” left ventricular dysfunction. Circulation. 2005 Feb 22. 111(7):887-93.[↩]