What are causes of nausea
Nausea is a feeling of sickness or unpleasant painless subjective feeling or discomfort characterized by a loss of appetite and the sensation of impending vomiting 1. Nausea is considered to function as a protective mechanism, warning the organism to avoid potential toxic ingestion 2. Less adaptive circumstances are also associated with nausea, including post-operative nausea, chemotherapy-induced nausea and motion sickness.
Nausea is a commonly encountered symptom with a broad list of possible causes (see Table 1). While nausea and vomiting are often thought to exist on a temporal continuum, this is not always the case. There are situations when severe nausea may be present without vomiting and less frequently, when vomiting may be present without preceding nausea. Most individuals report that nausea is more common, more disabling, feels worse and lasts longer than vomiting 3.
In the Diagnosis and Management of Nausea – Key Clinical Questions are 4:
- Is the nausea symptom acute (less than 4 weeks) or chronic (longer than 4 weeks) ?
- Are warning signs present ?
- Is this related to the GI (gastrointestinal) tract or to another organ system ?
- Are special conditions present ?
- What tests have been performed ?
- What treatment options are available ?
Is the Nause Acute or Chronic ?
- Acute less than 4 weeks in duration
- Infectious, toxins, medications, recent surgery, obstruction, inner ear disorders.
- Chronic longer than 4 weeks in duration
- Gastroparesis
- Chronic intestinal pseudo-obstruction
- Dyspepsia
- Hepatobiliary
- Medications
- Functional abdominal pain
- Narcotic bowel syndrome
- Psychogenic/psychological (bulimia)
- Other (renal, cardiac, urinary, CNS, endocrine).
Are warning signs present ?
- Persistent vomiting/hematemesis
- Odynophagia: Pain on swallowing food and fluids
- Dysphagia: Difficulty or discomfort in swallowing food and fluids
- Unintentional weight loss
- Significant abdominal pain (out of proportion)
- Evidence of obstruction (distention)
- Associated headaches/CNS findings
- Change in mental status/vision
- Adverse events of chronic nausea & vomiting
- Dehydration, hypokalemia, metabolic alkalosis
- Fever
- Reduce consciousness
- Severe headache with neck stiffness
Table 1. Common causes of nausea
| Medications and toxic etiologies | Disorders of the gut and peritoneum |
|---|---|
| Cancer chemotherapy | Mechanical obstruction |
| Analgesics | Gastric outlet obstruction |
| Cardiovascular medications | Small bowel obstruction |
| Digoxin | Functional gastrointestinal disorders |
| Antiarrhythmics | Functional dyspepsia |
| Antihypertensives | Chronic idiopathic nausea |
| β-Blockers | Cyclic Vomiting Syndrome |
| Calcium-channel antagonists | Idiopathic vomiting |
| Hormonal preparations/therapies | |
| Oral antidiabetics | Non-ulcer dyspepsia |
| Oral contraceptives | Irritable bowel syndrome |
| Antibiotics/antivirals | Organic gastrointestinal disorders |
| Erythromycin | Pancreatic adenocarcinoma |
| Tetracycline | Peptic ulcer disease |
| Sulfonamides | Cholecystitis |
| Antituberculous drugs | Pancreatitis |
| Acyclovir | Hepatitis |
| Gastrointestinal medications | Crohn’s disease |
| Sulfasalazine | Neuromuscular disorders of the gastrointestinal tract |
| Azathioprine | Gastroperesis |
| Nicotine | Post-operative nausea and vomiting |
| CNS active | Chronic intestinal pseudo-obstruction |
| Narcotics | CNS causes |
| Antiparkinsonian drugs | Migraine |
| Anticonvulsants | Increased intracranial pressure |
| Malignancy | |
| Radiation therapy | Hemorrhage |
| Infarction | |
| Ethanol abuse | Abscess |
| Meningitis | |
| Infectious causes | Congenital malformation |
| Gastroenteritis | Hydrocephalus |
| Otitis media | Pseudotumor cerebri |
| Acute intermittent porphyria | Seizure disorders |
| Demyelinating disorders | |
| Miscellaneous causes | Psychiatric disease |
| Cardiac disease | Psychogenic vomiting |
| Myocardial infarction | Anxiety disorders |
| Congestive heart failure | Depression |
| Radiofrequency ablation | Pain |
| Starvation | Eating disorders |
| Labyrinthine disorders | |
| Motion sickness | |
| Labyrinthitis | |
| Tumors | |
| Meniere’s disease | |
| Iatrogenic | |
| Endocrinological and metabolic causes | |
| Pregnancy | |
| Other endocrine and metabolic | |
| Uremia | |
| Diabetic ketoacidosis | |
| Hyperparathyroidism | |
| Hypoparathyroidism | |
| Hyperthyroidism | |
| Addison’s disease | |
Table 2. Presentations of Nausea and Vomiting That Suggest Specific Diagnoses
| Clinical presentation | Suggested diagnoses | Suggested tests |
|---|---|---|
| Acute onset |
| Cholecystitis: right upper-quadrant ultrasonography |
| Pancreatitis: amylase and lipase levels, ultrasonography to assess for gallstones, contrast-enhanced abdominal computed tomography in patients with severe illness | ||
| Associated with diarrhea, headache, and myalgias | Viral gastroenteritis | None |
| Bilious vomiting | Small bowel obstruction | Abdominal radiography or computed tomography |
| Continuous vomiting | Conversion disorders | Electrolyte levels |
| Delayed vomiting (more than one hour after meals) |
| Obstruction: abdominal radiography |
| Gastroparesis: gastric emptying study | ||
| Feculent or foul odor to vomitus | Intestinal obstruction | Abdominal radiography |
| Habitual postprandial, irregular vomiting | Major depression | Patient Health Questionnaire-9, Beck Depression Inventory |
| Insidious onset |
| Gastroesophageal reflux: esophagogastroduodenoscopy if patient has warning signs or does not improve with empiric therapy |
| Gastroparesis: gastric emptying study | ||
| Metabolic disorders: pulse oximetry, arterial blood gases, serum chemistries, chest radiography | ||
| Pregnancy: pregnancy test in women of childbearing age, with pelvic ultrasonography if ectopic pregnancy is suspected | ||
| Patient report of previous organic or functional gastrointestinal illness | Chronic psychogenic vomiting | Electrolyte levels, further evaluation if organic cause is suspected |
| Projectile vomiting, may not be preceded by nausea | Intracranial disorders, increased intracranial pressure (also associated with normal emesis) | Brain computed tomography |
| Regurgitation of undigested food |
| Esophagogastroduodenoscopy, upper gastrointestinal barium study |
| Vomiting before breakfast |
| Increased intracranial pressure: brain computed tomography |
| Pregnancy: pregnancy test in women of childbearing age, with pelvic ultrasonography if ectopic pregnancy is suspected | ||
| Uremia: renal function testing, electrolyte levels | ||
| Vomiting during or soon after meals |
| Electrolyte levels |
| Vomiting partly digested food or chyme several hours after meals |
| Obstruction: abdominal radiography |
In population studies, more than 50% of adults reported at least one episode of nausea, and more than 30% of adults reported one episode of vomiting within the preceding 12 months, with women reporting more episodes of nausea than men 6. In a large population based study of 62,651 individuals, 12.5% of the individuals reported nausea as ‘minor or major complaint’ in the last 12 months with the prevalence of nausea being three times higher in women than in men 7. Similar results have been reported in other epidemiological studies 8. In addition, race has also been shown to be associated with differential rates of the experience of nausea with White/African-Americans experiencing less nausea than Asian/Asian-American subjects 9.
Chronic, unexplained nausea alone or with vomiting occurs less commonly but is associated with significant comorbidity and poses a therapeutic challenge to health care providers 10. The exact prevalence of chronic idiopathic nausea or functional vomiting is not well studied and population-based studies are needed to estimate the disease burden.
Pathophysiology of nausea
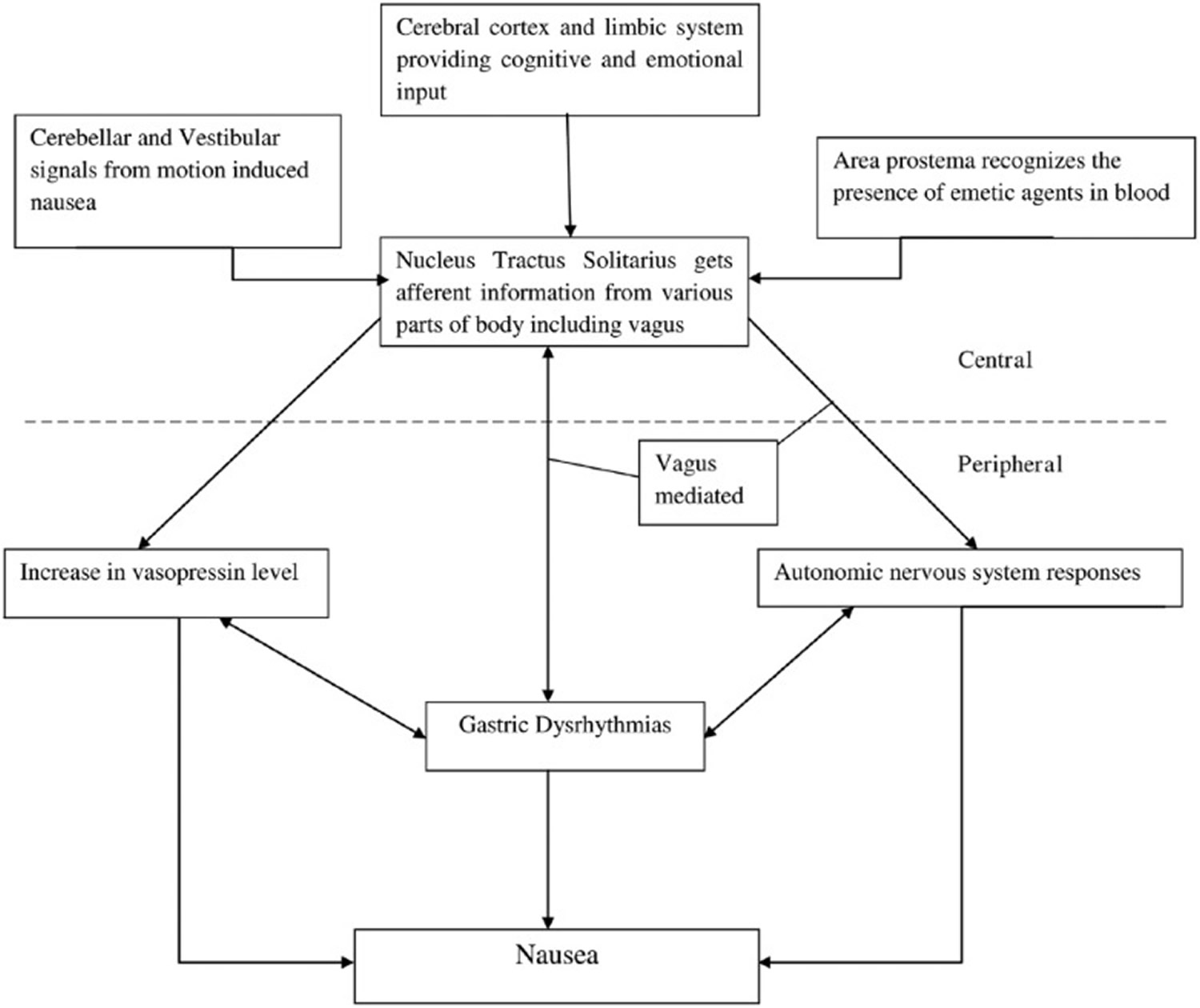
The underlying mechanisms involved in nausea are complex and encompass psychological states, the central nervous system, autonomic nervous system, gastric dysrhythmias, and the endocrine system (see Figure 1).
Central and peripheral pathways involved in pathogenesis of nausea. Afferent information from various stimuli are relayed to nucleus tractus solitarius via four pathways: vestibular and cerebellar, cerebral cortex and limbic system, area postrema and gastrointestinal tract via vagus nerve.*
*Once any of these neural pathways are activated, it culminates into sensation of nausea with or without vomiting. The efferent information from nucleus tractus solitarius is also responsible for activation of autonomic nervous response via vagal pathways. Nausea is also associated with gastric dysrhythmia and release of vasopressin. However, the cause–effect relationship of this triad is not very well understood and warrants further studying.
In order to understand the pathophysiology underlying nausea, it is important to introduce the concept of the dynamic threshold 11. It is proposed that each individual has a threshold for nausea that changes minute by minute. At any given moment, the threshold depends on the interaction of certain inherent factors of the individual with the more changeable psychological states of anxiety, anticipation, expectation and adaptation 11. This dynamic interaction likely explains the inter- and intra-individual variability that is typically encountered in the face of a nauseogenic stimulus 11.
Stimuli giving rise to nausea and vomiting originate from visceral, vestibular, and chemoreceptor trigger zone inputs which are mediated by serotonin/dopamine, histamine/acetylcholine and serotonin/dopamine, respectively. These relationships serve as the basis on which current pharmacological therapy for nausea and vomiting is recommended 12.
Figure 1. Pathogenesis of nausea
Central nervous system (brain & spinal cord)
Despite the prevalence and importance of nausea, surprisingly little is known regarding the central mechanisms underlying this sensation 13. The neurocircuitry involved in vomiting is better characterized. Associated autonomic changes which occur during nausea and emesis (e.g. salivation, sweating) are coordinated at the level of the medulla oblongata 14. Chemosensitive receptors detect the presence of emetic agents in the blood and this information is relayed via the area postrema to the nucleus tractus solitarius (NTS) 14. Abdominal vagal afferents which detect gastric tone and contents also project to the NTS 14. Neurons from the nucleus tractus solitarius (NTS) then project to a central-pattern generator which coordinates the various actions involved in the act of emesis in addition to directly projecting to neurons in the ventral medulla and hypothalamus, from which higher brain areas can be reached 14.
Many studies have suggested that the cerberal cortex is also involved in pathways of nausea 13. Recent investigations using functional magnetic resonance imaging (fMRI) techniques in healthy adults have shown that the medial prefrontal cortex and the pregenual anterior cingulate cortex, areas of the brain involved in higher cognitive function and emotion, are positively correlated with an increase in heart rate during nausea, suggesting the importance of cognitive and emotional centers in modulating the parasympathetic to sympathetic shift associated with nausea 13. Napadow and colleagues 13 studied humans predisposed to motion sickness and suggested that nauseogenic stimulus causes activation of amygdala, putamen and locus ceruleus which translates into fear conditioning and emotional triggering. This ultimately leads to the sensation of strong nausea 13. This is followed by continued, sustained activation in cortical areas such as insula, anterior cingulated cortex, nucleus accumbens, orbitofrontal, somatosensory and prefrontal cortex. These areas are involved in the interoceptive, limbic, somatosensory and cognitive network which alerts the suffering individual of the changes in interoceptive signaling so that appropriate autonomic and motor responses are initiated in a timely manner 13. Many of these areas involved in the nausea circuit specifically anterior cingulate cortex, insular cortex, nucleus accumbens and amygdala are known to be involved in processing of acute as well as chronic painful stimulus 15. Furthermore, the medial prefrontal cortex, which appears to be much more involved in chronic pain than acute pain perception, was also found to be a part of the nausea circuit 16. It is plausible that the brain perceives peripheral noxious stimuli through similar pathways, which in some cases lead to chronic pain and in others to chronic nausea. Understanding the central mechanisms of nausea, especially chronic unexplained nausea will be important for the development of therapies in the treatment of chronic nausea.
Autonomic nervous system
Characteristic physiological changes (sweating, pallor, salivation, increase in blood pressure, tachycardia, cutaneous vasoconstriction, decreased gastrointestinal motility) occurring before vomiting are mediated by the autonomic nervous system (ANS) and are well described 17. Afferent signaling arises from vagal inputs, reflecting mechanical or chemical stimuli 18. Several studies have now shown that increasing nausea perception is associated with decreased parasympathetic and increased sympathetic modulation which accounts for majority of above mentioned symptoms 19. In addition, LaCount and colleagues 19 have shown that bursts of cardiovagal modulation precedes transition to a higher level of nausea, perhaps by prompting interoceptive re-evaluation by the subject culminating in rating nausea at a higher level. This autonomic outflow during nausea is likely modulated by the central nervous system (CNS). While some areas of the brain, such as insula appear to be modulating both sympathetic as well as parasympathetic response, there also appears to be a divergent central control for autonomic response of nausea 20. Thus, ANS outflow and the CNS network controlling it could be determinant of overall nausea intensity, and understanding them in more detail could be of therapeutic importance.
Endocrine
Several hormones have been studied in the pathogenesis of nausea with the most important being vasopressin [also called antidiuretic hormone (ADH)]. Increase in antidiuretic hormone (ADH) secretion in various emetogenic situations is clear evidence that the rise in antidiuretic hormone (ADH) level precedes emesis, indicating that this rise does not appear in response to volume depletion or hyperosmolarity 21. Studies have also reported a positive correlation between serum antidiuretic hormone (ADH) level and nausea intensity 22. However, the cause–effect relationship between nausea and antidiuretic hormone (ADH) is still not clear. Although some studies have shown that supra-physiological levels of antidiuretic hormone (ADH) can induce gastric dysrhythmias and nausea whether this happens under normal physiological circumstances is not clear 23. The precise temporal relationship between vasopressin, gastric dysrhythmias and nausea should be studied in order to give us insight into pathogenesis of acute and chronic nausea syndromes. In addition to vasopressin, corticotropin-releasing factor has been established as brain–gut mediator in foregut function and can stimulate inhibitory motor nerves in the dorsal motor nucleus of the vagus, causing delayed gastric emptying and nausea 24.
Gastric dysrhythmias
The stomach is a neuromuscular organ, the myoelectrical activity of which can be measured by a number of techniques including electrogastrography. Normal gastric myoelectrical activity reflects the balance of the intrinsic pacemaker activity of the stomach, smooth muscle, the enteric nervous system, the autonomic nervous system and hormone levels 21. Activity frequency slower than the intrinsic rate is termed bradygastria; a faster frequency is termed tachygastria. There are numerous studies demonstrating the relationship of nausea with the onset of dysrhythmias in individuals with motion sickness, pregnant females, drug-induced nausea and gastroparesis 25. Xu and colleagues 26 have shown that in individuals experiencing vection-induced nausea, experienced tachygastria a few minutes beforehand, suggesting a relationship between gastric dysrhythmias and nausea. Interestingly, medications and interventions that promote normalization of myoelectrical activity from tachygastria decrease nausea, and conversely, stimuli that decrease normal myoelectrical activity and increase dysrhythmias promote the sensation of nausea 25. Whether activation of sympathetic nervous system precedes onset of gastric dysrhythmias or vice versa is not clear and needs to be studied in future 25. High amplitude, retrograde persistaltic contractions of small intestine from the distal to the proximal part on antroduodenal manometry has been shown to precede vomiting episodes 27. However, the causal relationship between gastric dysrhythmias or small intestinal dysmotility and nausea is not well established.
Diagnosis of Nausea
Detailed history and physical exam forms the cornerstone of evaluating patients with the chief complaint of nausea. History and physical exam help to rule out the non-gastrointestinal causes of nausea such as CNS, endocrinological and psychiatric diagnoses, which might be the primary cause or could be a contributing factor (see Table 1 above). The mainstay of diagnostic evaluation of the patient with nausea and vomiting is correcting any symptom consequences (electrolyte abnormalities, dehydration, malnutrition), identifying the cause of the symptoms and initiating targeted therapy, and lastly, if no cause is found, initiating therapy directed at suppressing the symptoms 1. There are no well-designed, controlled trials to guide diagnostic evaluation in patients with acute or chronic nausea. Basic blood work, including electrolytes, liver function tests, pancreatic enzymes and pregnancy test (wherever applicable) should be performed to help establish etiology. If mechanical obstruction is suggested by clinical presentation, radiological investigations such as abdominal X-ray and abdominal CT scan are typically the first-line investigations 1. If mucosal diseases such as ulcer or mass are suspected, esophagogastroduodenoscopy remains the most sensitive and specific investigation to study the esophageal, gastric and duodenal mucosa. If needed, small bowel mucosa can be studied by small bowel follow through and enteroclysis.
Scintigraphic measures of solid phase gastric emptying (such as 99mTc-sulfur colloid in egg) are commonly used to evaluate gastric motion function in suspected gastroparesis 28. Following the test for four instead of two hours has been shown to improve accuracy in diagnosing gastroparesis 29. One should remember that these studies do not establish cause–effect relationship in patients with nausea. Their utility is further limited by the finding that symptoms are not well correlated with abnormalities of gastric emptying and medications to improve motility have been shown to improve symptoms without changing emptying delay 1. In general, most health providers would advocate an empirical trial of antiemetic or prokinetic medications prior to specialized testing 1. Moreover, many of these patients need to be on antiemetics to be able to tolerate these tests. However, many of these agents, specifically prokinetics, can alter gut motility. Thus, it is important to interpret the results in the context of being on or off medication. Investigations such as cutaneous electrogastrography and antroduodenal manometry to study gastric motor functions are not widely available, are expensive and their role in the diagnostic algorithm of patients with chronic nausea is not well established 30. A reduction or decrease of expected postprandial electrogastric amplitude has been shown to correlate with delayed gastric emptying 30. Similarly, postprandial antral hypomotility on atroduodenal manometry is a common finding in patients with unexplained nausea, as well as gastroparesis, and could be used if other tests for gastric motility are normal in a patient with persistent symptoms despite empirical treatment 31. It aids in the diagnosis of motor disorders in such cases but when normal, helps to rule out dysmotility as a cause of nausea.
Patients with unexplained chronic nausea (even after thorough investigation including normal gastric emptying studies) pose a diagnostic dilemma and they remain indistinguishable from those with documented gastric delay with respect to demographics, stability of symptoms over time, healthcare utilization and health-related quality of life 32. In the absence of an identifiable cause, Rome III criteria for functional gastroduodenal disorders can be utilized to diagnose functional disorders related to nausea and vomiting, including chronic idiopathic nausea, functional vomiting, cyclical vomiting syndrome and rumination syndrome. Pasricha and colleagues showed that current Rome III diagnostic criteria may be inadequate for differentiating this group of patients, with significant overlap of diagnoses; many patients met diagnostic criteria for functional dyspepsia, irritable bowel syndrome and chronic idiopathic nausea/functional vomiting 32. Further refinement will be guided by evolving experience and knowledge of this specific group of patients.
What helps nausea
A distinction should be made with respect to management of acute (less than 4 weeks) versus chronic (greater than 4 weeks) symptoms as they are likely entirely different entities and response to therapeutics differ between the two. There is a paucity of literature evaluating the pharmacological therapy of chronic, unexplained nausea 31. This is likely related to the fact that most clinically encountered episodes of nausea and vomiting are typically short lived and self-limited 31. Most of the literature is focused on those clinical situations where the risk of nausea and vomiting is high, such as in pregnancy, the post-operative time period, post chemotherapy, and post radiation 31. It is important to realize the inconsistent effect on the relief of nausea compared with vomiting, with nausea being more resistant to interventions. This finding likely reflects the different physiology of these two distinct symptoms 31. Effect of various antiemetics on different types of nausea, e.g. benzodiazepines for anticipatory nausea and serotonin antagonists for chemotherapy-induced nausea again highlights the complex pathophysiology of nausea.
Current medical therapy generally falls into two categories:
- Agents directed at suppressing nausea and preventing vomiting (antiemetic) which typically act centrally in the brain, and
- Agents directed at modulating gastrointestinal motility (prokinetic).
Commonly utilized antiemetics are listed (see Table 3).
Table 3. Common anti nausea medications
| Medications | Typical dosage | Side effects | Route of administration |
|---|---|---|---|
| Anticholinergics | |||
| Scopolamine | 0.3–0.6 mg q24 hours | Tachycardia, confusion, dry mouth, constipation, urinary retention, blurred vision | SL, IV, IM, transdermal |
| Antihistamines | |||
| Meclizine Diphenhydramine Cinnarizine Cyclizine Hydroxyzine | 25–50 mg q24 hours 25–50 mg q6–8 hours 25–75 mg q8 hours 25–50 mg q4–6 hours 25–100 mg q6–8 hours | Drowsiness, confusion, blurred vision, constipation, urinary retention | Oral Oral, IM, IV Oral Oral Oral, IM |
| Phenothiazines | |||
| Prochlorperazine Promethazine Chlorpromazine Perphenazine | 5–10 mg q6–8 hours 12.5 –25 mg q4–6 hours 10–25 mg q4–6 hours 4–8 mg q8–12 hours | Extrapyramidal side effects, tardive dyskinesias, neuroleptic malignant syndrome, hyperprolactinemia, QT prolongation | Oral, IM, IV, rectal Oral, IM, IV, rectal Oral, IM, IV Oral |
| Benzamides | |||
| Metoclopramide Domperidone | 10–20 mg q6–8 hours 10 mg q8–24 hours | Sedation Anxiety Mood disturbances Sleep disruption Dystonic reactions Tardive dyskinesia Galactorrhea Sexual dysfunction | Oral, IM, IV Oral |
| 5-HT3 antagonists | |||
| Ondansetron Granisetron Palonosetron | 4–8 mg q4–8 hours 1–2 mg q24 hours 0.075–0.25 mg q24 hours | Headache, fatigue, malaise, constipation | Oral, IV Oral, IV, transdermal IV |
| Cannabinoids | |||
| Dronabinol Nabilone | 2.5–10 mg q6–8 hours 1–2 mg q8–12 hours | Palpitations, tachycardia, vasodilation/facial flushing, euphoria, abnormal thinking, dizziness, paranoia, depersonalization, hallucinations, visual changes | Oral Oral |
| Benzodiazepines | |||
| Lorazepam Alprazolam | 0.5–2 mg 0.25–1 mg | Ataxia, cognitive dysfunction, depression, dizziness, drowsiness, dysarthria, fatigue, irritability, memory impairment, sedation | Oral, SL, IM, IV Oral |
| Corticosteroids | |||
| Dexamethasone | 4–8 mg q4–6 hours | Emotional instability, acne, bruising, hyperglycemia, adrenal suppression, Cushing’s syndrome | Oral, IM, IV |
| Droperidol | 0.625–1.25 mg q24hours | QTc prolongation, orthostatic hypotension, extrapyramidal symptoms, CNS effects | IM, IV |
| NK-1 Receptor Antagonists | |||
| Aprepitant | 80–125 mg q24 hours | Fatigue, constipation, hiccups | Oral |
SL, sublingual; IV, intravenous; IM, intramuscular; CNS, central nervous system.
Table 4. Treatment of Nausea and Vomiting in Adults according to disease condition
| Cause | Treatment |
|---|---|
| Benign positional vertigo | Meclizine (Antivert) |
| Benign, self-limited, no alarm signs | Cola, ginger ale, mints, oral rehydration, supportive therapy, treatment of underlying cause |
| Gastroenteritis | Ondansetron (Zofran), promethazine |
| Gastroesophageal reflux disease, gastritis | Histamine H2 antagonist, proton pump inhibitor |
| Gastroparesis | Erythromycin, metoclopramide (Reglan) |
| Migraine | Metoclopramide, nonsteroidal anti-inflammatory drugs, prochlorperazine plus antihistamines (NNT = 5) |
| Motion sickness | Antihistamines, scopolamine |
| Peptic ulcer disease | Proton pump inhibitor (NNT = 3) |
| Pregnancy | Doxylamine (Unisom), ginger, vitamin B6 |
| Renal colic | Intravenous or intramuscular nonsteroidal anti-inflammatory drugs, with or without opioids (NNT = 16) |
NNT = number needed to treat.
Anti nausea medicines
Serotonin 5-HT3 antagonists
Serotonin 5-HT3 antagonists such as granisetron and ondansetron have utility in post-operative vomiting, post-radiation therapy, and in preventing chemotherapy-related emesis 1. Their mechanism of action is mediated primarily though central 5-HT3 receptor blockade (mainly in the chemoreceptor trigger zone) and peripheral blockade of 5-HT3 receptors on intestinal vagal and spinal afferent nerves 33. Their effect in relieving nausea is less robust and prevalence of chemotherapy-induced nausea has increased despite the introduction of potent 5-HT3 antagonists 34. Its utility in other forms of nausea, such as gastroparesis and functional vomiting, is not well studied and it has not been shown to be superior to metaclopramide or promethazine in a double-blinded randomized controlled trial in controlling nausea symptoms in adults visiting the emergency department 28.
Antihistamines
Antihistamines have a therapeutic role in motion sickness and labrynthitis and exert their antiemetic action through central anticholinergic (M1 receptor) and antihistamine (H1 receptor) effects 33. These drugs suppress labyrinthine and vestibular stimulation and that of the chemoreceptor zone in the brainstem 35.
Anticholinergic agents
Anticholinergic agents act centrally via the muscarinic receptors and block the pathway from the inner ear to the brainstem and the ‘vomiting center’ 36. Scopolamine is the most widely used anticholinergic and is administered as a transdermal patch for both prophylaxis and treatment of motion sickness, but its use in other forms of nausea are not well established 31. Selective M3 and M5 antagonist (Zamifenacin) appears to be equally effective implicating these two receptor subtypes 36.
Phenothiazines
Phenothiazines are antidopaminergic agents which act via nonselective inhibition of mainly D2 and D3 receptors in the region of the area postrema, but also muscarinic and H1 receptors 37. They have demonstrated efficacy in treating nausea related to migraine, motion sickness and vertigo, as well as post-operative and post-chemotherapy nausea and vomiting 31. The butyrophenone, droperidol, is only available as a restricted use drug by the FDA, primarily due to its effects on QT prolongation. Its efficacy is well documented in post-operative and chemotherapy-associated nausea and vomiting and like phenothiazines, the mechanism of action is primarily via antidopaminergic activity in the chemoreceptor zone 31.
Cannabinoids
Cannabinoids have been investigated in chemotherapy-related nausea and vomiting 38. They are thought to act primarily via the cannabinoid receptor (CB1) in the ‘vomiting center’ of the medulla and the area subpostrema of the NTS, although potential to modulate 5-HT3 activation in nodose ganglions and substance P release in the spinal cord could also contribute to their antiemesic activity 12. Like many of the other antiemetic agents, the antinausea effect of cannabinoids is not as well established as their antiemetic effect 39.
Benzodiazepines
Benzodiazepines have been investigated as adjunctive therapy in post-operative nausea and small reports have shown that its use reduces anticipatory nausea associated with chemotherapy 40. It is postulated that the antiemetic mechanism of action involves dopamine in the chemoreceptor trigger zone 40. Its primary mode of action is via its sedative, anxiolytic, and amnestic properties in reducing the anticipatory component of nausea 12.
Corticosteroids
Corticosteroids are often used concomitantly with 5-HT3 antagonists and other agents for acute, as well as delayed chemotherapy-induced nausea and vomiting. They have also been shown to have efficacy equivalent to ondansetron and droperidol in post-operative nausea 41. The mechanism of action is unclear but most likely involves its effect on prostaglandin formation and inflammation 12.
Tachykinin peptides
The role of tachykinin peptides such as substance P in the vomiting reflex has been exploited therapeutically, with aprepitant, an antagonist of the tachykinin receptor NK1. Aprepitant is FDA approved for the prevention of both acute, as well as delayed chemotherapy-induced nausea and vomiting and has been showed to potentiate the effects of 5-HT3 receptor antagonists and corticosteroids 42. Case reports have demonstrated the use of aprepitant in the treatment of gastroparesis-associated nausea and vomiting with no demonstrated change in gastric emptying 43. Formal investigation into the efficacy of aprepitant in the treatment of chronic, refractory nausea and vomiting is needed.
Prokinetics
Prokinetic medications are listed (Table 5) and include agents that act primarily as a prokinetic (e.g. erythromycin) versus those that have both prokinetic and antiemetic properties (e.g. metoclopramide). Erythromycin exerts its effects via activation of motilin receptors present on gut smooth muscle, possibly leading to modulation of vagal nerve pathways involved in emesis 44. At low doses of 50–100 mg before meals, it has been shown to be efficacious in controlling nausea and vomiting in patients with delayed gastric emptying. However, when used as antibiotic at higher doses (250–500 mg, 2–4 times a day), it induces nausea, possibly with vomiting 44. At higher concentrations it probably promotes nausea by contracting the gastric fundus and thus inducing gastric dysrythmias and prolonged, nonpropulsive hypermotility of the antrum 44. Recently, ghrelin has been shown to increase gastric emptying in patients with diabetic gastroparesis 45. It has also been shown to control nausea in ferrets exposed to cisplatin; but its effect on nausea in humans is not yet studied 46. However, it is important to realize that nausea likely has both peripheral and central components, and the prokinetic mechanism of action is likely restricted to the periphery. Metoclopramide and domperidone are benzamides with potent antiemetic and prokinetic properties. Their mechanism of action is complex and involves vagal and central 5-HT3 and D2 receptor antagonism with prokinetic properties via gut dopamine receptor antagonism and 5-HT4-receptor agonist activity. They are differentiated by the fact that domperidone does not cross blood–brain barrier and is thus free of extrapyramidal side effects which are common with metaclopramide. Domperidone is not available in the US but metaclopramide is manufactured for both oral and parenteral use. These drugs have been shown to be efficacious in post-chemotherapy vomiting and gastroparesis. Clinically, the use of prokinetic agents is unfortunately limited by numerous side effects, some of which may be irreversible.
Table 5. Prokinetic medications
| Agent | Typical Dosage | Side Effects | Route of administration |
|---|---|---|---|
| Metoclopramide | 10–20 mg q6–8 hours | Sedation Anxiety Mood disturbances Sleep disruption Dystonic reactions Tardive dyskinesia Galactorrhea Sexual dysfunction | Oral, IM, IV |
| Domperidone | 10mg q8–24 hours | Galactorrhea/gynecomastia Sexual dysfunction | Oral |
| Erythromycin | 250–500mg q8 hours | Nausea and vomiting Diarrhea | Oral, IV |
IV, intravenous; IM, intramuscular.
[Source 2]
Novel therapies
Novel and nontraditional medical therapies for nausea and vomiting have been studied (Table 6), and include low-dose antidepressants such as tricyclic antidepressants (TCAs). Retrospective studies have showed moderate symptom reduction in patients with chronic nausea and vomiting including cyclical vomiting syndrome in both diabetic and non-diabetic population; however, given lack of good prospective studies, their use is typically reserved for those with moderate to severe or refractory symptoms 47. In a retrospective study of 37 patients with chronic functional nausea, 51% patients had a complete response and an additional 33% had at least moderate symptoms reduction with low dose TCAs 48. TCAs such as amitriptyline have been shown to have some benefit in functional upper gastrointestinal symptoms such as painful dyspepsia in patients who do not have prolonged gastric emptying 49. This is likely true for other symptoms such as chronic nausea where TCAs are likely to benefit only those patients without delayed gastric emptying.
Table 6. Novel and non-traditional therapies for nausea
| Agent | Mechanism of action | Typical dosage | Side effects | Route of administration |
|---|---|---|---|---|
| Tricyclic antidepressants Amitriptyline Nortriptyline Doxepin Desipramine Imipramine | Antihistaminic and muscarinic activity | 10–100 mg/day is the common range for most of these | Constipation, gastric emptying delay, agitation, sedation | Oral |
| Gabapentin | Mitigation of calcium currents and tachykinin in areas such as the area postrema | 300–900 mg thrice daily For sensitive patients, consider starting at between 12.5 to 25 mg daily | Clumsiness, somnolence | Oral |
| Olanzapine | Targets dopaminergic (D1, D2, D3 and D4), serotonergic (5-HT2A, 5-HT2C, 5-HT3, 5-HT6), histaminic and muscarinic receptor | 5–10 mg/day | Somnolence, postural hypotension, dizziness, dyspepsia, restlessness, weight gain and rarely extrapyramidal symptoms | Oral |
Gabapentin
Gabapentin, a γ-aminobutyric acid analog has been shown to be efficacious in preventing post-operative nausea and vomiting in multiple randomized controlled trials 50. There is also some use of gabapentin in preventing acute and delayed chemotherapy-associated nausea and vomiting, hyperemesis gravidarum, and life-threatening refractory emesis following posterior fossa surgery 50. It is suggested that the mechanism of action involves mitigation of calcium currents in areas such as area postrema 50. Future studies should explore its use in patients with chronic functional nausea vomiting.
Olanzapine
Olanzapine is a known antagonist for dopamine and serotonin receptors. Clinical trials have demonstrated the efficacy of olanzapine in the control of both acute and delayed chemotherapy induced nausea and vomiting 51. In animal studies, olanzapine has been shown to be effective for the control of narcotic-induced nausea and sleep disturbance associated with chronic pain 52.
Gastric electrical stimulation (GES)
Gastric electrical stimulation (GES) is a technique used for refractory gastroparesis. A recent analysis of a large series at one institution demonstrated symptomatic improvement after GES but a need for additional surgical procedures and complications after GES implant 53. Symptomatic improvement achieved by GES in patients with gastroparesis is best explained by activation of vagal afferent pathways to influence CNS control mechanisms for nausea and vomiting, accompanied by enhanced vagal efferent autonomic function and decreased gastric sensitivity to volume distention which enhances postprandial gastric accommodation 54. GES with short pulses can improve gastroparesis symptoms such as nausea and vomiting and GES with long pulses has been shown to improve gastric motility 55. Evidence for the efficacy of GES in patients without demonstrated gastric-emptying delay (such as patients with functional dyspepsia and chronic, refractory, idiopathic nausea and vomiting) is evolving 56.
Home remedies for nausea
With respect to dietary recommendations in the management of nausea and vomiting, a low-fat, low-fiber diet with small frequent meals is typically recommended if patients are able to tolerate oral intake. A short-term liquid diet in extreme cases of patients not tolerating a solid diet is also routinely recommended 57. For patients with chronic, unexplained symptoms, dietary recommendations typically follow those routinely recommended for patients with documented gastric emptying delay, although there are no large, well-designed trials evaluating this strategy.
Dietary manipulation and supplements have also been investigated for the management of nausea and vomiting. Use over-the-counter (OTC) motion sickness medicines. If you’re planning a trip, OTC motion sickness drugs, such as dimenhydrinate (Dramamine) or meclizine (Rugby Travel Sickness) may help calm your queasy stomach. For longer journeys, such as a cruise, ask your doctor about prescription motion sickness adhesive patches, such as scopolamine (Transderm Scop). Ginger is a herbal supplement that has been shown to have some efficacy in small studies to reduce severity of post-operative nausea and vomiting, morning sickness and motion sickness 58. Studies involving the use of ginger in prevention of chemotherapy-induced nausea and vomiting are conflicting and do not support its use 59. Its mechanism of action is largely unknown but improvements in gastric motility, anti-5-hydroxytryptamine activity and central antiemetic effects have been postulated 59. Rieber and colleagues investigated human subjects, looking at the effect of acute tryptophan depletion when subjected to a rotational procedure intended to induce nausea and found that it increased 60. Interestingly, acute tryptophan depletion was also shown to slow gastric emptying time in females 61. These findings suggest a potential role of diet and supplements in modulating the sensation of pain and nausea, which would be important to pursue with further research.
Alternative and complementary approaches to the management of nausea and vomiting include hypnosis, acupressure and acupuncture. In a systematic review, P6 acupoint stimulation significantly reduced nausea, vomiting and the need for rescue medications in the post-operative setting 62. Another randomized controlled trial involving 63 subjects with post-surgical gastroparesis showed that P6-acupoint stimulation was superior to metaclopramide in achieving complete recovery rate 63. Marchioro and colleagues 64 have also shown efficacy of hypnosis in preventing chemotherapy-induced anticipatory nausea and vomiting in 16 patients.
Summary
Acute nausea (e.g. related to chemotherapy) is easier to control than chronic unexplained nausea. Clinically, patients with chronic and refractory symptoms in the absence of an identifiable cause pose a significant challenge and should be the focus of clinical investigation. Antiemetics such as antihistaminics, antidopaminergics and 5-HT3 antagonists are often the first-line agents used for common causes of acute nausea. Other agents such as steroids and aprepitant are most commonly used for acute and delayed phase of chemotherapy-induced nausea. Benzodiazepines are best suited for the anticipatory component of post-operative or chemotherapy-induced nausea.
Chronic nausea is much more difficult to control and poses a therapeutic challenge for most health care providers. As discussed above, the central pathways of chronic nausea are very close to chronic neuropathic pain and thus therapeutic options are also directed towards the same. The first line therapeutic options for these patients are neuromodulators such as TCAs, olanzapine, gabapentin and possibly cannabinoids and benzodiazepines. However, if a patient is found to have delayed gastric emptying, prokinetics are the agents of choice. For patients with chronic nausea, we recommend starting with the lowest possible dose of antinausea agents and titrating slowly, as most of these types of patient are sensitive to triggers such as food and medications, which could worsen their symptoms.
- Nausea and vomiting. Hasler WL, Chey WD. Gastroenterology. 2003 Dec; 125(6):1860-7.[↩][↩][↩][↩][↩][↩]
- Singh P, Yoon SS, Kuo B. Nausea: a review of pathophysiology and therapeutics. Therapeutic Advances in Gastroenterology. 2016;9(1):98-112. doi:10.1177/1756283X15618131. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4699282/[↩][↩][↩][↩][↩][↩]
- Stern R., Koch K., Andrews P. (2011) Nausea: mechanisms and management. New York: Oxford University Press.[↩]
- Functional Dyspepsia & Nausea: Where Do We Stand in 2015? http://s3.gi.org/meetings/na2015/15ACG_Southern_Regional_0004.pdf[↩]
- Evaluation of Nausea and Vomiting in Adults: A Case-Based Approach. Am Fam Physician. 2013 Sep 15;88(6):371-379. http://www.aafp.org/afp/2013/0915/p371.html[↩][↩]
- Rub R., Andrews P., Whitehead S. (1992) Vomiting—incidence, causes, ageing and sex. In Bianchi A, Grelot L., Miller A., King G., editors. (eds), Mechanisms and Control of Emesis, Montrouge: John Libbey Eurotext.[↩]
- The prevalence of nausea in the community: psychological, social and somatic factors. Haug TT, Mykletun A, Dahl AA. Gen Hosp Psychiatry. 2002 Mar-Apr; 24(2):81-6.[↩]
- Comorbidity of gastrointestinal complaints, depression, and anxiety in the Epidemiologic Catchment Area (ECA) Study. Walker EA, Katon WJ, Jemelka RP, Roy-Bryne PP. Am J Med. 1992 Jan 24; 92(1A):26S-30S.[↩]
- Asian hypersusceptibility to motion sickness. Stern RM, Hu S, Uijtdehaage SH, Muth ER, Xu LH, Koch KL. Hum Hered. 1996 Jan-Feb; 46(1):7-14.[↩]
- Characteristics of patients with chronic unexplained nausea and vomiting and normal gastric emptying. Pasricha PJ, Colvin R, Yates K, Hasler WL, Abell TL, Unalp-Arida A, Nguyen L, Farrugia G, Koch KL, Parkman HP, Snape WJ, Lee L, Tonascia J, Hamilton F. Clin Gastroenterol Hepatol. 2011 Jul; 9(7):567-76.e1-4. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3123425/[↩]
- The psychophysiology of nausea. Stern RM. Acta Biol Hung. 2002; 53(4):589-99.[↩][↩][↩]
- Nausea and vomiting. Chepyala P, Olden KW. Curr Treat Options Gastroenterol. 2008 Apr; 11(2):135-44. https://www.ncbi.nlm.nih.gov/pubmed/18321441/[↩][↩][↩][↩]
- The brain circuitry underlying the temporal evolution of nausea in humans. Napadow V, Sheehan JD, Kim J, Lacount LT, Park K, Kaptchuk TJ, Rosen BR, Kuo B. Cereb Cortex. 2013 Apr; 23(4):806-13.[↩][↩][↩][↩][↩][↩]
- Central neurocircuitry associated with emesis. Hornby PJ. Am J Med. 2001 Dec 3; 111 Suppl 8A():106S-112S. https://www.ncbi.nlm.nih.gov/pubmed/11749934/[↩][↩][↩][↩]
- Neuroplasticity underlying the comorbidity of pain and depression. Doan L, Manders T, Wang J. Neural Plast. 2015; 2015():504691. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4355564/[↩]
- Shape shifting pain: chronification of back pain shifts brain representation from nociceptive to emotional circuits. Hashmi JA, Baliki MN, Huang L, Baria AT, Torbey S, Hermann KM, Schnitzer TJ, Apkarian AV. Brain. 2013 Sep; 136(Pt 9):2751-68. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3754458/[↩]
- Why is the neurobiology of nausea and vomiting so important? Horn CC. Appetite. 2008 Mar-May; 50(2-3):430-4.[↩]
- The medical implications of gastrointestinal vagal afferent pathways in nausea and vomiting. Horn CC. Curr Pharm Des. 2014; 20(16):2703-12.[↩]
- Static and dynamic autonomic response with increasing nausea perception. LaCount LT, Barbieri R, Park K, Kim J, Brown EN, Kuo B, Napadow V. Aviat Space Environ Med. 2011 Apr; 82(4):424-33. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3137518/[↩][↩]
- Sclocco R., Kim J., Garcia R., Sheehan J., Beissner F., Bianchi A., et al. (2014) Brain circuitry supporting multi-organ autonomic outflow in response to nausea. Cereb Cortex August 12. pii: bhu172. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4712791/[↩]
- A noxious trio: nausea, gastric dysrhythmias and vasopressin. Koch KL. Neurogastroenterol Motil. 1997 Sep; 9(3):141-2.[↩][↩]
- The effect of intravenous vasopressin on gastric myoelectrical activity in human subjects. Caras SD, Soykan I, Beverly V, Lin Z, McCallum RW. Neurogastroenterol Motil. 1997 Sep; 9(3):151-6.[↩]
- Role of plasma vasopressin as a mediator of nausea and gastric slow wave dysrhythmias in motion sickness. Kim MS, Chey WD, Owyang C, Hasler WL. Am J Physiol. 1997 Apr; 272(4 Pt 1):G853-62.[↩]
- Cyclic vomiting syndrome: the corticotropin-releasing-factor hypothesis. Taché Y. Dig Dis Sci. 1999 Aug; 44(8 Suppl):79S-86S.[↩]
- Gastric dysrhythmias: a potential objective measure of nausea. Koch KL. Exp Brain Res. 2014 Aug; 232(8):2553-61.[↩][↩][↩]
- Hypothalamic and gastric myoelectrical responses during vection-induced nausea in healthy Chinese subjects. Xu LH, Koch KL, Summy-Long J, Stern RM, Seaton JF, Harrison TS, Demers LM, Bingaman S. Am J Physiol. 1993 Oct; 265(4 Pt 1):E578-84.[↩]
- Vomiting and the small intestine. Thompson DG, Malagelada JR. Dig Dis Sci. 1982 Dec; 27(12):1121-5.[↩]
- Clinical guideline: management of gastroparesis. Camilleri M, Parkman HP, Shafi MA, Abell TL, Gerson L, American College of Gastroenterology. Am J Gastroenterol. 2013 Jan; 108(1):18-37; quiz 38.[↩][↩]
- Assessment of gastric emptying using a low fat meal: establishment of international control values. Tougas G, Eaker EY, Abell TL, Abrahamsson H, Boivin M, Chen J, Hocking MP, Quigley EM, Koch KL, Tokayer AZ, Stanghellini V, Chen Y, Huizinga JD, Rydén J, Bourgeois I, McCallum RW. Am J Gastroenterol. 2000 Jun; 95(6):1456-62.[↩]
- Electrogastrography: a document prepared by the gastric section of the American Motility Society Clinical GI Motility Testing Task Force. Parkman HP, Hasler WL, Barnett JL, Eaker EY, American Motility Society Clinical GI Motility Testing Task Force. Neurogastroenterol Motil. 2003 Apr; 15(2):89-102.[↩][↩]
- AGA technical review on nausea and vomiting. Quigley EM, Hasler WL, Parkman HP. Gastroenterology. 2001 Jan; 120(1):263-86.[↩][↩][↩][↩][↩][↩][↩][↩]
- Characteristics of patients with chronic unexplained nausea and vomiting and normal gastric emptying. Pasricha PJ, Colvin R, Yates K, Hasler WL, Abell TL, Unalp-Arida A, Nguyen L, Farrugia G, Koch KL, Parkman HP, Snape WJ, Lee L, Tonascia J, Hamilton F. Clin Gastroenterol Hepatol. 2011 Jul; 9(7):567-76.e1-4.[↩][↩]
- Nausea and vomiting. Chepyala P, Olden KW. Curr Treat Options Gastroenterol. 2008 Apr; 11(2):135-44.[↩][↩]
- Nausea and vomiting remain a significant clinical problem: trends over time in controlling chemotherapy-induced nausea and vomiting in 1413 patients treated in community clinical practices. Roscoe JA, Morrow GR, Hickok JT, Stern RM. J Pain Symptom Manage. 2000 Aug; 20(2):113-21.[↩]
- Practical selection of antiemetics. Flake ZA, Scalley RD, Bailey AG. Am Fam Physician. 2004 Mar 1; 69(5):1169-74.[↩]
- Comparison of the effects of a selective muscarinic receptor antagonist and hyoscine (scopolamine) on motion sickness, skin conductance and heart rate. Golding JF, Stott JR. Br J Clin Pharmacol. 1997 Jun; 43(6):633-7.[↩][↩]
- Treatment of nausea and vomiting: gaps in our knowledge. Sanger GJ, Andrews PL. Auton Neurosci. 2006 Oct 30; 129(1-2):3-16.[↩]
- Superiority of nabilone over prochlorperazine as an antiemetic in patients receiving cancer chemotherapy. Herman TS, Einhorn LH, Jones SE, Nagy C, Chester AB, Dean JC, Furnas B, Williams SD, Leigh SA, Dorr RT, Moon TE. N Engl J Med. 1979 Jun 7; 300(23):1295-7. http://www.nejm.org/doi/full/10.1056/NEJM197906073002302[↩]
- Treatment of nausea and vomiting: gaps in our knowledge. Sanger GJ, Andrews PL. Auton Neurosci. 2006 Oct 30; 129(1-2):3-16. https://www.ncbi.nlm.nih.gov/pubmed/16934536/[↩]
- Midazolam as an anti-emetic. Rodolà F. Eur Rev Med Pharmacol Sci. 2006 May-Jun; 10(3):121-6.[↩][↩]
- A factorial trial of six interventions for the prevention of postoperative nausea and vomiting. Apfel CC, Korttila K, Abdalla M, Kerger H, Turan A, Vedder I, Zernak C, Danner K, Jokela R, Pocock SJ, Trenkler S, Kredel M, Biedler A, Sessler DI, Roewer N, IMPACT Investigators. N Engl J Med. 2004 Jun 10; 350(24):2441-51.[↩]
- Guideline update for MASCC and ESMO in the prevention of chemotherapy- and radiotherapy-induced nausea and vomiting: results of the Perugia consensus conference. Roila F, Herrstedt J, Aapro M, Gralla RJ, Einhorn LH, Ballatori E, Bria E, Clark-Snow RA, Espersen BT, Feyer P, Grunberg SM, Hesketh PJ, Jordan K, Kris MG, Maranzano E, Molassiotis A, Morrow G, Olver I, Rapoport BL, Rittenberg C, Saito M, Tonato M, Warr D, ESMO/MASCC Guidelines Working Group. Ann Oncol. 2010 May; 21 Suppl 5():v232-43.[↩]
- Gastroparesis-associated refractory nausea treated with aprepitant. Fahler J, Wall GC, Leman BI. Ann Pharmacother. 2012 Dec; 46(12):e38.[↩]
- Anti-emetic and emetic effects of erythromycin in Suncus murinus: role of vagal nerve activation, gastric motility stimulation and motilin receptors. Javid FA, Bulmer DC, Broad J, Aziz Q, Dukes GE, Sanger GJ. Eur J Pharmacol. 2013 Jan 15; 699(1-3):48-54.[↩][↩][↩]
- Ghrelin enhances gastric emptying in diabetic gastroparesis: a double blind, placebo controlled, crossover study. Murray CD, Martin NM, Patterson M, Taylor SA, Ghatei MA, Kamm MA, Johnston C, Bloom SR, Emmanuel AV. Gut. 2005 Dec; 54(12):1693-8.[↩]
- Anti-emetic activity of ghrelin in ferrets exposed to the cytotoxic anti-cancer agent cisplatin. Rudd JA, Ngan MP, Wai MK, King AG, Witherington J, Andrews PL, Sanger GJ. Neurosci Lett. 2006 Jan 9; 392(1-2):79-83.[↩]
- Tricyclic antidepressants for chronic vomiting in diabetic patients. Sawhney MS, Prakash C, Lustman PJ, Clouse RE. Dig Dis Sci. 2007 Feb; 52(2):418-24.[↩]
- Tricyclic antidepressants for functional nausea and vomiting: clinical outcome in 37 patients. Prakash C, Lustman PJ, Freedland KE, Clouse RE. Dig Dis Sci. 1998 Sep; 43(9):1951-6.[↩]
- Effect of Amitriptyline and Escitalopram on Functional Dyspepsia: A Multicenter, Randomized Controlled Study. Talley NJ, Locke GR, Saito YA, Almazar AE, Bouras EP, Howden CW, Lacy BE, DiBaise JK, Prather CM, Abraham BP, El-Serag HB, Moayyedi P, Herrick LM, Szarka LA, Camilleri M, Hamilton FA, Schleck CD, Tilkes KE, Zinsmeister AR. Gastroenterology. 2015 Aug; 149(2):340-9.e2.[↩]
- Gabapentin’s anti-nausea and anti-emetic effects: a review. Guttuso T Jr. Exp Brain Res. 2014 Aug; 232(8):2535-9.[↩][↩][↩]
- Navari R. (2013) Management of chemotherapy-induced nausea and vomiting: focus on newer agents and new uses for older agents. Drugs 73: 249–262.[↩]
- Usefulness of olanzapine as an adjunct to opioid treatment and for the treatment of neuropathic pain. Torigoe K, Nakahara K, Rahmadi M, Yoshizawa K, Horiuchi H, Hirayama S, Imai S, Kuzumaki N, Itoh T, Yamashita A, Shakunaga K, Yamasaki M, Nagase H, Matoba M, Suzuki T, Narita M. Anesthesiology. 2012 Jan; 116(1):159-69.[↩]
- Keller D., Parkman H., Boucek D., Sankineni A., Meilahn J., Gaughan J., et al. (2013) Surgical Outcomes After Gastric Electric Stimulator Placement for Refractory Gastroparesis. J Gastrointest Surg 17: 620–626[↩]
- McCallum R., Dusing R., Sarosiek I., Cocjin J., Forster J., Lin Z. (2010) Mechanisms of symptomatic improvement after gastric electrical stimulation in gastroparetic patients. Neurogastroenterol Motil 22: 161–167.[↩]
- Lei Y., Chen J. (2009) Effects of dual-pulse gastric electrical stimulation on gastric tone and compliance in dogs. Dig Liver Dis 41: 277–282.[↩]
- Lu P., Teich S., Di Lorenzo C., Skaggs B., Alhajj M., Mousa H. (2013) Improvement of quality of life and symptoms after gastric electrical stimulation in children with functional dyspepsia. Neurogastroenterol Motil 25: 567–e456.[↩]
- Hasler W., Chey W. (2003) Nausea and vomiting. Gastroenterology 125: 1860–1867. https://www.ncbi.nlm.nih.gov/pubmed/14724837[↩]
- Keating A., Chez R. (2002) Ginger syrup as an antiemetic in early pregnancy. Altern Ther Health Med.8: 89–91.[↩]
- Navari R. (2013) Management of chemotherapy-induced nausea and vomiting: focus on newer agents and new uses for older agents. Drugs 73: 249–262. https://www.ncbi.nlm.nih.gov/pubmed/23404093[↩][↩]
- Rieber N., Mischler D., Schumacher V., Muth E., Bischoff S., Klosterhalfen S., et al. (2010) Acute tryptophan depletion increases experimental nausea but also induces hunger in healthy female subjects. Neurogastroenterol Motil 22: 752–757.[↩]
- Van Nieuwenhoven M., Valks S., Sobczak S., Riedel W., Brummer R. (2004) Acute tryptophan depletion slows gastric emptying in females. Br J Nutr 91: 351–355.[↩]
- Lee A., Fan L. (2009) Stimulation of the wrist acupuncture point P6 for preventing postoperative nausea and vomiting. Cochrane Database Syst Rev CD003281.[↩]
- Sun B., Luo M., Wu S., Chen X., Wu M. (2010) Acupuncture versus metoclopramide in treatment of postoperative gastroparesis syndrome in abdominal surgical patients: a randomized-controlled trial. Zhong Xi Yi Jie He Xue Bao 8: 641–644.[↩]
- Marchioro G., Azzarello G., Viviani F., Barbato F., Pavanetto M., Rosetti F., et al. (2000) Hypnosis in the treatment of anticipatory nausea and vomiting in patients receiving cancer chemotherapy. Oncology 59: 100–104.[↩]





