Contents
What is neurogenic bladder
Neurogenic bladder is a problem in which a person lacks bladder control due to a brain, spinal cord, or nerve condition 1. A spinal cord injury may interrupt communication between the nerves in the spinal cord that control bladder and bowel function and the brain, causing bladder- and bowel-function problems. This results in bladder or bowel dysfunction that is termed “neurogenic bladder” or “neurogenic bowel.” People with multiple sclerosis or spina bifida may have similar problems 2.
If you have neurogenic bladder, signs may include loss of bladder control (urinary incontinence), inability to empty the bladder, urinary frequency and urinary tract infections. Signs of neurogenic bowel include loss of bowel control (bowel incontinence), constipation, bowel frequency and lack of bowel movements.
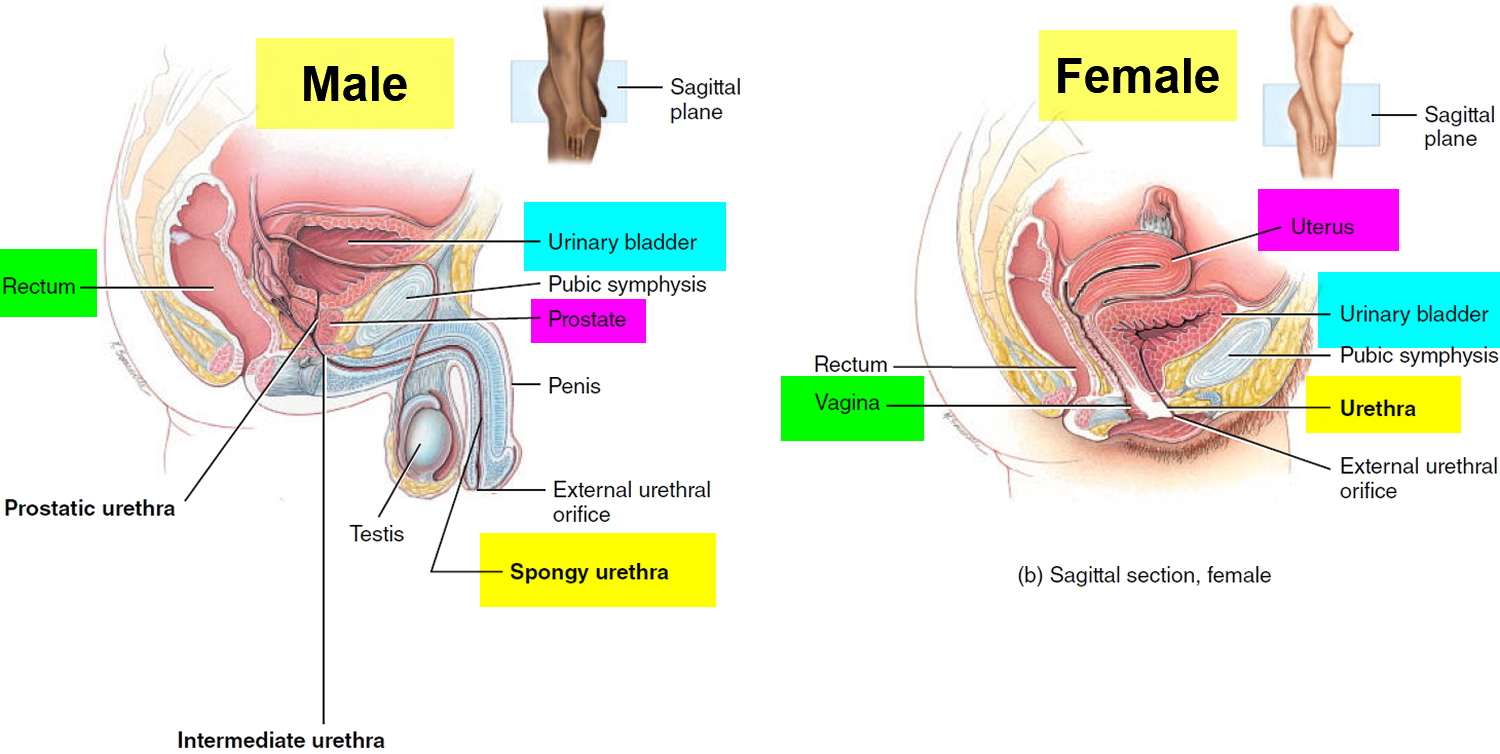
Figure 1. Urinary bladder location
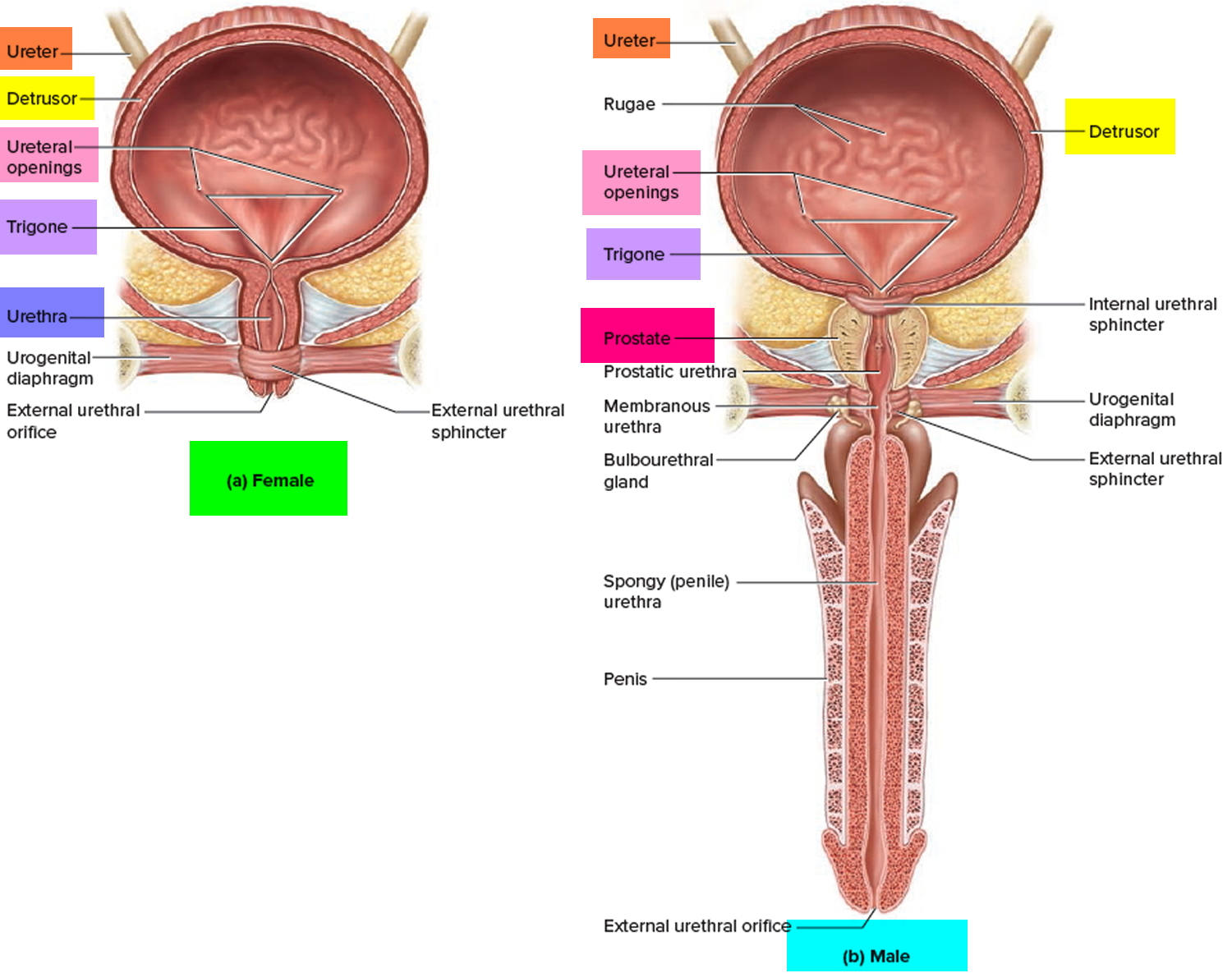
Figure 2. Urinary bladder anatomy
Normal bladder function
The urinary bladder is a hollow, collapsible muscular sac that stores and expels urine situated in the pelvic cavity posterior to the pubic symphysis. When slightly distended due to the accumulation of urine, the urinary bladder is spherical. When it is empty, it collapses. As urine volume increases, it becomes pear shaped and rises into the abdominal cavity.
Urinary bladder capacity averages 700–800 mL. It is smaller in females because the uterus occupies the space just superior to the urinary bladder.
For the urinary system to do its job, muscles and nerves must work together to hold urine in the bladder and then release it at the right time. Nerves carry messages from the bladder to the brain to let it know when the bladder is full. They also carry messages from the brain to the bladder, telling muscles either to tighten or release. A nerve problem might affect your bladder control if the nerves that are supposed to carry messages between the brain and the bladder do not work properly.
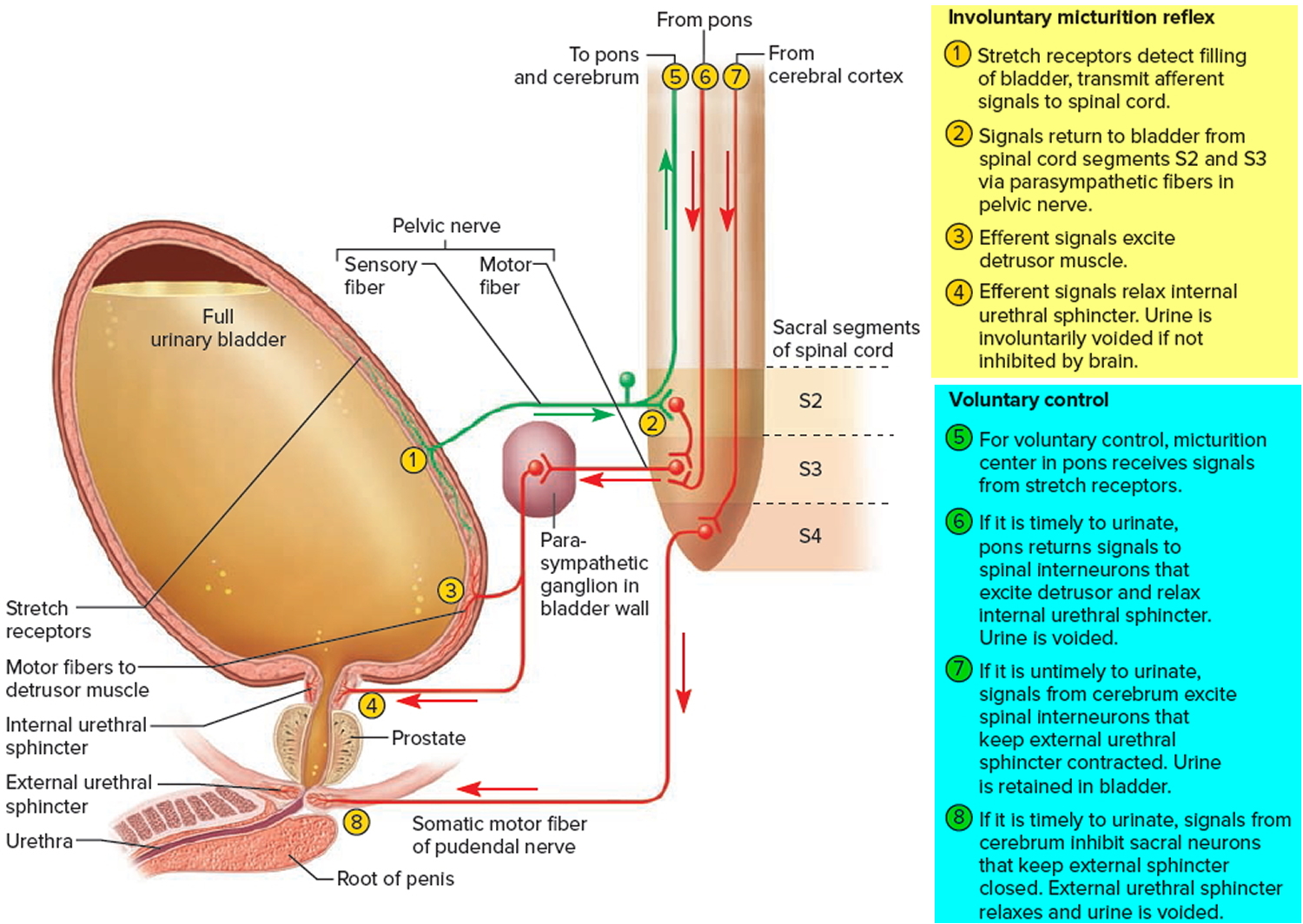
Discharge of urine from the urinary bladder, called micturition, is also known as urination or voiding. Micturition occurs via a combination of involuntary and voluntary muscle contractions. When the volume of urine in the urinary bladder exceeds 200–400 mL, pressure within the bladder increases considerably, and stretch receptors in its wall transmit nerve impulses into the spinal cord. These impulses propagate to the micturition center in sacral spinal cord segments S2 and S3 and trigger a spinal reflex called the micturition reflex. In this reflex arc, parasympathetic impulses from the micturition center propagate to the urinary bladder wall and internal urethral sphincter.
The nerve impulses cause contraction of the detrusor muscle and relaxation of the internal urethral sphincter muscle. Simultaneously, the micturition center inhibits somatic motor neurons that innervate skeletal muscle in the external urethral sphincter. On contraction of the urinary bladder wall and relaxation of the sphincters, urination takes place. Urinary bladder filling causes a sensation of fullness that initiates a conscious desire to urinate before the micturition reflex actually occurs. Although emptying of the urinary bladder is a reflex, in early childhood we learn to initiate it and stop it voluntarily.
Human bladder contraction is mediated mainly through stimulation of muscarinic receptors in the detrusor muscle by acetylcholine 3. Antimuscarinic agents act during the filling/storage phase of the micturition cycle by inhibiting afferent (sensory) input from the bladder, and directly inhibiting smooth muscle contractility 4. They are competitive antagonists, so with massive release of acetylcholine during micturition the drug effect is decreased and the muscle can contract 5.
Through learned control of the external urethral sphincter muscle and certain muscles of the pelvic floor, the cerebral cortex can initiate micturition or delay its occurrence for a limited period.
Figure 3. Bladder control
Causes of neurogenic bladder
Several muscles and nerves must work together for your bladder to hold urine until you’re ready to empty it. Nerve messages go back and forth between the brain and the muscles that control bladder emptying. If these nerves are damaged by illness or injury, the muscles may not be able to tighten or relax at the right time.
Disorders of the central nervous system commonly cause neurogenic bladder. These can include:
- Alzheimer’s disease
- Birth defects of the spinal cord
- Brain or spinal cord tumors
- Cerebral palsy
- Encephalitis
- Learning disabilities such as attention deficit hyperactivity disorder (ADHD)
- Multiple sclerosis
- Parkinson’s disease
- Spinal cord injury
- Stroke recovery
Damage or disorders of the nerves that supply the bladder can also cause this condition. This can include:
- Alcoholic neuropathy (nerve damage due to long-term, heavy alcohol use)
- Diabetic neuropathy (nerve damage due to long-term diabetes)
- Nerve damage due to pelvic surgery
- Nerve damage from a herniated disc.
- Vitamin B12 deficiency
- Nerve damage from syphilis
- Nerve damage due to pelvic surgery
- Nerve damage from a herniated disk or spinal canal stenosis
Possible Complications of neurogenic bladder
- Constant urine leakage can cause skin to break down and lead to pressure sores
- Kidney damage may occur if the bladder becomes too full, causing pressure to build up in the tubes leading to the kidneys and in the kidneys themselves
- Urinary tract infections
Complications of neurogenic bladder following Spinal Cord Injury
The most common complications of neurogenic bladder due to spinal cord injury are urinary tract infection (UTI), urinary stones, and renal impairment. These complications are associated with the pathology of bladder dysfunction itself or occur as a consequence of the use of urinary catheters for drainage.
Urinary tract infection
Patients with spinal cord injury bladder and stable bladder management are prone to repeated urinary tract infections. Symptomatic urinary tract infections continue to pose a significant problem for these patients. Symptomatic urinary tract infections might include fever, foul smelling urine, and/or hematuria. A comparative study followed patients for 1 year and showed that episodes of bacteriuria with fever were significantly more common in patients who were catheterized by someone else than in patients on self-clean intermittent catheterization or with indwelling catheters. Patients who exhibited the least frequent episodes of symptomatic urinary tract infection were those with external sphincterotomies 6.
The type of catheter used for clean intermittent catheterization in patients with spinal cord injury-neurogenic bladder dysfunction might affect the rate of infection. In a prospective randomized study that compared hydrophilic-coated catheters with uncoated polyvinyl chloride catheters, significantly fewer patients using the hydrophilic-coated catheters (64%) experienced one or more urinary tract infections compared with the control group (82%) over a period of 1 year 7.
Several studies have examined the value of prophylactic antibiotics for prevention of urinary tract infections in spinal cord injury patients on clean intermittent catheterization. A prospective randomized trial evaluated the possible role of trimethoprim-sulfamethoxazole prophylaxis in patients with recent spinal cord injury during the first 4 months of clean intermittent catheterization during the bladder training phase. A significant reduction in bacteriuria and symptomatic urinary tract infection was observed in men in the antibiotic group compared with those on placebo. This difference was not observed in female patients. However, adverse events related to antibiotic treatment and emergence of drug resistance were common and limit the usefulness of this treatment regimen 8.
Another prospective study in patients with spinal cord injury-neurogenic bladder dysfunction conducted during initial management with clean intermittent catheterization found that prophylactic antibiotics significantly reduced bacteriuria but not clinical urinary tract infections 9. A meta-analysis examined the benefits and harms of antimicrobial prophylaxis for UTI in patients with spinal cord injury 10. The authors pooled the results from 15 clinical trials and reached the conclusion that there is no evidence to support the use of prophylactic antibiotics for most patients. Prophylaxis did not significantly decrease symptomatic infections but was associated with a reduction in asymptomatic bacteriuria. Importantly, prophylaxis resulted in a two-fold increase in antimicrobial-resistant bacteria.
Modification of antibiotic treatment regimens might reduce drug resistance. Spinal cord injury-neurogenic bladder dysfunction on intermittent catheterization and recurrent urinary tract infections require multiple courses of antibiotic therapy that increase the incidence of multidrug-resistant bacteria. A prospective study determined the efficacy of alternate administration of an oral antibiotic once per week to prevent urinary tract infections. Two years of follow-up revealed a significant decrease in symptomatic urinary tract infections from 9.4 to 1.8 per patient-year with no development of new drug-resistant bacteria 11.
Alternative treatment options for reducing infection include daily administration of methenamine hippurate or cranberry tablets. However, in patients with stable bladder management following spinal cord injury, a prospective double-blind study failed to identify any benefit of such regimens in terms of reducing symptomatic UTIs over a follow-up period of 6 months 12. An interesting technique to prevent urinary tract infection in spinal cord injury patients is inoculation of the bladder with the innocuous Escherichia coli strain 83972. With a mean duration of colonization of 12 months, the patients exhibited no symptoms of urinary tract infection compared with a mean of 3.1 symptomatic urinary tract infections per year before colonization. Symptomatic urinary tract infections occurred in the patients who were not successfully colonized and after spontaneous loss of colonization 13.
Urethritis and prostatitis
To decrease the possibility of urethritis, patients should use clean intermittent catheterizations and avoid indwelling catheters. Occasionally, blockage of the periurethral glands can cause periurethral abscesses. E. coli is the most frequent bacteria in prostatitis. Most antibiotics diffuse poorly into prostatic tissue, and trimethoprim is one of the recommended antibiotics because of its ability to cross the blood-prostatic barrier 14.
Epididymitis and epididymoorchitis
These conditions are catheter-related complications. Spinal cord injury patients usually will not experience pain, but they might exhibit swelling and occasionally skin redness. Treatment consists of antibiotic therapy. Occasionally, testicular abscesses that require drainage might form 14.
Bladder stones
Infections of the bladder with urea-splitting organisms, indwelling catheters, and high residual urine levels are the primary reasons for bladder stone formation. Stone formation might cause severe irritative symptoms and hematuria. Weekly changing of the catheters might decrease the potential for bladder stone formation 15.
Kidney stones
The overall incidence of renal stones is 3.5% in patients with neurogenic bladder. The most common are struvite stones. Successful treatment depends on complete elimination of the calculus and eradication of the infection 16.
Reflux and renal insufficiency
Vesicoureteral reflux occurs in more than 20% of patients with neurogenic bladder. This condition is more common when the lesion is suprasacral. High detrusor pressure and reflux are responsible for renal damage and renal failure. The best treatment for reflux is to normalize the detrusor pressure 17.
Bladder cancer
The risk of bladder cancer is 20 times higher in spinal cord injury patients compared to the general population, with squamous cell cancer occurring more commonly than transitional cell cancer. The risk factors are UTI, bladder stones, and indwelling catheters. Unfortunately, the diagnosis is usually made in the more advanced stages 18.
Neurogenic bladder symptoms
The symptoms depend on the cause. They often include symptoms of urinary incontinence.
Symptoms of overactive bladder:
- Having to urinate too often in small amounts
- Problems emptying all the urine from the bladder
- Loss of bladder control
Symptoms of underactive bladder:
- Bladder becomes too full and you may leak urine
- Inability to tell when the bladder is full
- Problems starting to urinate or emptying all the urine from the bladder
- Urinary retention.
When to Contact a Medical Professional
See your health care provider if you:
- Are unable to empty your bladder at all
- Have signs of a bladder infection (fever, burning when you urinate, frequent urination)
- Urinate small amounts, frequently.
How is neurogenic bladder diagnosed
The initial evaluation includes history-taking and a voiding diary. A focused physical examination is essential to evaluate the pelvic anatomy and the nervous system. The examiner should determine the level of the spinal motor lesion, the extent of injury, ie, whether it is complete or incomplete, limb tone, rectal tone, and the bulbocavernosus reflex. To properly assess the patient’s symptoms, a validated neurogenic bladder symptom score was recently developed as a tool to measure urinary symptoms and their consequences in patients with acquired or congenital neurogenic bladder 19. Urinalysis, urine culture and sensitivity, serum blood urea nitrogen/creatinine, creatinine clearance, urodynamics, and urinary tract imaging should be performed during the initial evaluation of spinal cord injury patients. Urologic follow-up evaluations of patients with neurogenic bladder are typically performed yearly. Doctors recommend urinalysis every 6 months, urine culture whenever the patient is symptomatic, ultrasound of the upper tract every 6 months, and urodynamic evaluation every 1–2 years. Patients with high detrusor pressure or decreased compliance require urodynamic evaluation within 12 months of treatment 20. Detection of hydronephrosis can be used as a marker of badly managed neurogenic bladder.
Urodynamics
Urodynamics are the mainstay for evaluation of neurogenic lower urinary tract dysfunction. These assessments include urinary flowmetry, bladder cystometry, electromyogram, Valsalva leak point pressure, and detrusor leak point pressure. Urodynamic evaluations provide the most definitive diagnoses of abnormalities in the bladder and urethra during the filling/storage phase. The initial urodynamic studies should be delayed until after the spinal shock phase has passed. Because the anatomic site of the spinal cord injury does not predict bladder and urethral function, management should be based on urodynamic findings rather than on findings from the neurologic evaluation 21.
Initial studies to identify patients who are at risk for upper tract disease should be conducted early because the effects of the disease on the urinary tract can be silent. The goals of assessment are to observe the maintenance of low bladder pressures, decrease the risk of infection, maintain continence, and prevent upper tract deterioration.36 Lower urinary tract function is subject to change in most cases of neurogenic bladder; hence, regular urodynamic follow-ups are warranted. In a long-term study, the treatment strategy for neurogenic bladder dysfunction in spinal cord injury patients had to be modified in all patients 22. Videourodynamic studies combine fluoroscopic voiding cystourethrography with multichannel urodynamics to provide the most comprehensive assessment of the lower urinary tract in spinal cord injury patients. When vesicoureteral reflux occurs, the volume and pressure at which the reflux is initiated can be documented. Videourodynamic studies are the definitive tests for diagnosis of detrusor striated sphincter dyssynergia during detrusor contraction 23.
Cystoscopy
Cystoscopy is not useful in the initial evaluation of neurogenic bladder but becomes very important for assessing lower urinary tract complications.
Treatment of neurogenic bladder
Conservative management
Conservative management is the most frequently applied treatment option in neurogenic bladder. Conservative management requires patient education and might include timed voiding, the Valsalva and Credé maneuvers, medications, intermittent catheterization, or an indwelling urinary catheter. Regular bladder emptying with or without anticholinergic medications is important to prevent UTIs, upper tract damage, and incontinence. However, the Credé and Valsalva maneuvers are generally not recommended as primary methods of bladder emptying. Potential complications of the Credé and Valsalva maneuvers include high bladder pressure, abdominal bruising with the Credé method, hernia, and hemorrhoids. Long-term use of the Credé maneuver to evacuate the bladder is not safe. This conclusion was reached in a cross-sectional study of spinal cord injury patients with flaccid bladders 20 years after an earthquake in the People’s Republic of China 24. Most of the patients exhibited residuals exceeding 100 mL, and half exhibited more than 300 mL of residual urine following the Credé maneuver. Urologic complications were common and included pyuria (82%), urinary lithiasis (31%), ureteral dilatation (60%), hydronephrosis (35%), and renal damage (16%). Upper urinary tract deterioration was more common in men. Women who perform the Credé maneuver might have an anatomic advantage over men. However, in a prospective, long-term follow-up of men and women with spinal cord injury-induced neurogenic bladder dysfunction, no specific method of bladder management was observed to cause renal impairment in women 25. The only subset of patients in which significant deterioration was observed was men with quadriplegia and ileal conduits when compared with the women.
The type of bladder management used and frequency of urinary incontinence strongly influence health-related quality of life. Several factors influence patient choice for bladder management. Patients are likely to choose treatments that they perceive as convenient to use and will maintain continence. In a cross-sectional study of patients with spinal cord injury, only 21% reported normal voiding 26. In addition to frequency of incontinence, health-related quality of life is affected by the type of bladder management. The worst mental status scores are associated with attendants performing clean intermittent catheterization and use of indwelling urethral catheters or indwelling suprapubic catheters 26. A prospective study evaluated the psychologic and social status of patients using clean intermittent catheterization for spinal cord injury-associated neurogenic bladder dysfunction. The patients generally reported reduced health-related quality of life scores in all health domains 27.
Your doctor may refer you someone who has been trained to help people manage bladder problems. Skills or techniques you may learn include:
- Exercises to strengthen your pelvic floor muscles (Kegel exercises)
- Keeping a diary of when you urinate, the amount you urinated, and if you leaked urine. This may help you learn when you should empty your bladder and when it may be best to be near a bathroom.
Learn to recognize the symptoms of urinary infections (UTIs), such as burning when you urinate, fever, low back pain on one side, and a more frequent need to urinate. Cranberry tablets are used to prevent UTIs.
Some people may need to use a urinary catheter. This is a thin tube that is inserted into your bladder:
- You may need a catheter to be in place all the time (indwelling catheter)
- You may need a catheter to be placed in your bladder 4 to 6 times a day to keep your bladder from becoming too full (intermittent catheterization)
What are Kegel exercises ?
To do Kegel exercises, you just squeeze your pelvic floor muscles. The part of your body including your hip bones is the pelvic area. At the bottom of the pelvis, several layers of muscle stretch between your legs. The muscles attach to the front, back, and sides of the pelvic bone.
Kegel exercises are designed to make your pelvic floor muscles stronger. These are the muscles that hold up your bladder and help keep it from leaking.
Building up your pelvic muscles with Kegel exercises can help with your bladder control.
How do you exercise your pelvic muscles ?
Find the right muscles. Try one of the following ways to find the right muscles to squeeze.
- Imagine that you are trying to stop passing gas. Squeeze the muscles you would use. If you sense a “pulling” feeling, you are squeezing the right muscles for pelvic exercises.
- Imagine that you are sitting on a marble and want to pick up the marble with your vagina. Imagine “sucking” the marble into your vagina.
- For Women: If you still are not sure you are tightening the right muscles – lie down and put your finger inside your vagina. Squeeze as if you were trying to stop urine from coming out. If you feel tightness on your finger, you are squeezing the right pelvic muscles.
- For Men: Insert a finger into your rectum. Tighten the muscles as if you are holding in your urine, then let go. You should feel the muscles tighten and move up and down.
Once you know what the movement feels like, do Kegel exercises three times a day:
- Make sure your bladder is empty, then sit or lie down.
- Tighten the pelvic floor muscles. Hold tight and count to 6 to 8.
- Relax the muscles and count to 10.
- Repeat 10 times, three times a day (morning, afternoon, and night).
At first, find a quiet spot to practice—your bathroom or bedroom—so you can concentrate. Lie on the floor. Pull in the pelvic muscles and hold for a count of 6 to 8. Then relax for a count of 10. Work up to 10 to 15 repeats each time you exercise.
Do your pelvic exercises at least three times a day. Every day, use three positions: lying down, sitting, and standing. You can exercise while lying on the floor, sitting at a desk, or standing in the kitchen. Using all three positions makes the muscles strongest.
Breathe deeply, and relax your body when you are doing these exercises. Make sure you are not tightening your stomach, thigh, buttock, or chest muscles.
After 4 to 6 weeks, you should feel better and have fewer symptoms. Keep doing the exercises, but do not increase how many you do. Overdoing it can lead to straining when you urinate or move your bowels.
Some notes of caution:
- Once you learn how to do them, do not practice Kegel exercises at the same time you are urinating more than twice a month. Doing the exercises while you are urinating can weaken your pelvic floor muscles over time.
- In women, doing Kegel exercises incorrectly or with too much force may cause vaginal muscles to tighten too much. This can cause pain during sexual intercourse.
- Incontinence will return if you stop doing these exercises. Once you start doing them, you may need to do them for the rest of your life.
It may take several months for your incontinence to lessen once you start doing these exercises.
Let your doctor, nurse, or therapist help you. Many people have trouble finding the right muscles. Your doctor, nurse, or therapist can check to make sure you are doing the exercises correctly. You can also exercise by using special weights or biofeedback. Ask your health care team about these exercise aids.
Don’t squeeze other muscles at the same time. Be careful not to tighten your stomach, legs, or other muscles. Squeezing the wrong muscles can put more pressure on your bladder control muscles. Just squeeze the pelvic muscle. Don’t hold your breath.
Be patient. Don’t give up. It’s just 5 minutes, three times a day. You may not feel your bladder control improve until after 3 to 6 weeks. Still, most women do notice an improvement after a few weeks.
Medications
Anticholinergic medications
Anticholinergic treatment is the first-line therapy for neurogenic detrusor overactivity. This treatment works by blocking cholinergic transmission at muscarinic receptors. Anticholinergic agents have similar efficacy; however, they have different side effect and tolerability profiles that depend on their muscarinic receptor selectivity and the rate of drug distribution. Several studies have shown that anticholinergic treatment increases bladder capacity, reduces bladder pressure, and improves compliance and quality of life. The majority of the studies have examined oxybutynin immediate-release, trospium chloride immediate-release, propiverine immediate-release, and propiverine extended-release 28.
Anticholinergic drugs that bind M1 receptors might produce impairments of memory and cognition. Agents that bind M2 receptors can produce QT interval prolongation that causes tachycardia and arrhythmias. Anticholinergic drugs that bind M3 receptors might produce visual blurring, xerostomia, and constipation. Different routes of application have been used to reduce the side effects of oral anticholinergics. A transdermal oxybutynin system was found to be efficacious in spinal cord injury patients with neurogenic detrusor overactivity and was well tolerated at up to three times the standard dose; however, skin irritation was a common side effect 29. Intravesical therapy with oxybutynin has been shown to be effective and helps to reduce the systemic side effects associated with oral administration of this drug 30.
A combined antimuscarinic treatment has been used in refractory neurogenic bladder; despite the increased side effects, the combined treatment delays the need for other more invasive treatments.68 A higher dose of oxybutynin (30 mg) has been used to improve patient symptoms; this dose was found to be effective, safe, and well tolerated 31.
Medications may help manage your symptoms. Your doctor may recommend:
- Medicines that relax the bladder (oxybutynin, tolterodine, or propantheline)
- Medicines that make certain nerves more active (bethanechol)
- Botulinum toxin (Botox)
- GABA supplements
- Antiepileptic drugs
Newer drugs are also being studied.
Sometimes surgery is needed. Surgeries for neurogenic bladder include:
- Artificial sphincter
- Electrical device implanted near the bladder nerves, to stimulate the bladder muscles
- Sling surgery
- Creation of an opening (stoma) in which urine flows into a special pouch (this is called urinary diversion).
- Neurogenic bladder. U.S. National Library of Medicine. https://wwwqa.nlm.nih.gov/medlineplus/275/ency/article/000754.htm[↩]
- Neurogenic bladder and bowel management. Mayo Clinic. https://www.mayoclinic.org/tests-procedures/neurogenic-bladder-bowel-management/basics/definition/prc-20013781[↩]
- Ballert K, Biggs G, Nitti V. Antimuscarinic agents. AUA update series 2008;27:137–47.[↩]
- Saks EK, Arya LA. Pharmacologic management of urinary incontinence, voiding dysfunction, and overactive bladder. Obstet Gynecol Clin North Am 2009;36:493–507.[↩]
- Andersson K-E. Antimuscarinics for treatment of overactive bladder. Lancet Neurol 2004;3:46–53.[↩]
- Bacteriuria with fever after spinal cord injury. Cardenas DD, Mayo ME. Arch Phys Med Rehabil. 1987 May; 68(5 Pt 1):291-3. https://www.ncbi.nlm.nih.gov/pubmed/3579535/[↩]
- Cardenas DD, Mayo ME. Bacteriuria with fever after spinal cord injury. Arch Phys Med Rehabil. 1987;68(5 Pt 1):291–293. https://www.ncbi.nlm.nih.gov/pubmed/3579535[↩]
- Gribble MJ, Puterman ML. Prophylaxis of urinary tract infection in persons with recent spinal cord injury: a prospective, randomized, double-blind, placebo-controlled study of trimethoprim-sulfamethoxazole. Am J Med. 1993;95(2):141–152. https://www.ncbi.nlm.nih.gov/pubmed/8356980[↩]
- Maynard FM, Diokno AC. Urinary infection and complications during clean intermittent catheterization following spinal cord injury. J Urol. 1984;132(5):943–946. https://www.ncbi.nlm.nih.gov/pubmed/6333518[↩]
- Morton SC, Shekelle PG, Adams JL, et al. Antimicrobial prophylaxis for urinary tract infection in persons with spinal cord dysfunction. Arch Phys Med Rehabil. 2002;83(1):129–138. https://www.ncbi.nlm.nih.gov/pubmed/11782843[↩]
- Salomon J, Denys P, Merle C, et al. Prevention of urinary tract infection in spinal cord-injured patients: safety and efficacy of a weekly oral cyclic antibiotic (WOCA) programme with a 2 year follow-up – an observational prospective study. J Antimicrob Chemother. 2006;57(4):784–788. https://www.ncbi.nlm.nih.gov/pubmed/16473921[↩]
- Lee BB, Haran MJ, Hunt LM, et al. Spinal-injured neuropathic bladder antisepsis (SINBA) trial. Spinal Cord. 2007;45(8):542–550. https://www.ncbi.nlm.nih.gov/pubmed/17043681[↩]
- Hull R, Rudy D, Donovan W, et al. Urinary tract infection prophylaxis using Escherichia coli 83972 in spinal cord injured patients. J Urol. 2000;163(3):872–877. https://www.ncbi.nlm.nih.gov/pubmed/10687996[↩]
- Buczyński AZ. Principles for urological management of SCI patients. Ortop Traumatol Rehabil. 2000;2(1):57–60. https://www.ncbi.nlm.nih.gov/pubmed/18033215[↩][↩]
- Park YI, Linsenmeyer TA. A method to minimize indwelling catheter calcification and bladder stones in individuals with spinal cord injury. J Spinal Cord Med. 2001;24(2):105–108. https://www.ncbi.nlm.nih.gov/pubmed/11587416[↩]
- Chen Y, DeVivo MJ, Stover SL, Lloyd LK. Recurrent kidney stone: a 25-year follow-up study in persons with spinal cord injury. Urology. 2002;60(2):228–232. https://www.ncbi.nlm.nih.gov/pubmed/12137815[↩]
- Hackler RH, Dalton JJ, Jr, Bunts RC. Changing concepts in the preservation of renal function in the paraplegic. J Urol. 1965;94:107–111. https://www.ncbi.nlm.nih.gov/pubmed/14334300[↩]
- Hess MJ, Zhan EH, Foo DK, Yalla SV. Bladder cancer in patients with spinal cord injury. J Spinal Cord Med. 2003;26(4):335–338. https://www.ncbi.nlm.nih.gov/pubmed/14992333[↩]
- The validity and reliability of the neurogenic bladder symptom score. Welk B, Morrow S, Madarasz W, Baverstock R, Macnab J, Sequeira K. J Urol. 2014 Aug; 192(2):452-7. https://www.ncbi.nlm.nih.gov/pubmed/24518764/[↩]
- EAU guidelines on neurogenic lower urinary tract dysfunction. Stöhrer M, Blok B, Castro-Diaz D, Chartier-Kastler E, Del Popolo G, Kramer G, Pannek J, Radziszewski P, Wyndaele JJ. Eur Urol. 2009 Jul; 56(1):81-8. https://www.ncbi.nlm.nih.gov/pubmed/19403235/[↩]
- Urodynamics of spinal cord injury. Watanabe T, Rivas DA, Chancellor MB. Urol Clin North Am. 1996 Aug; 23(3):459-73. https://www.ncbi.nlm.nih.gov/pubmed/8701559/[↩]
- Clinical usefulness of urodynamic assessment for maintenance of bladder function in patients with spinal cord injury. Nosseir M, Hinkel A, Pannek J. Neurourol Urodyn. 2007; 26(2):228-33. https://www.ncbi.nlm.nih.gov/pubmed/16998859/[↩]
- Videourodynamics: indications and technique. Marks BK, Goldman HB. Urol Clin North Am. 2014 Aug; 41(3):383-91, vii-viii. https://www.ncbi.nlm.nih.gov/pubmed/25063594/[↩]
- Chang SM, Hou CL, Dong DQ, Zhang H. Urologic status of 74 spinal cord injury patients from the 1976 Tangshan earthquake, and managed for over 20 years using the Credé maneuver. Spinal Cord. 2000;38(9):552–554. https://www.ncbi.nlm.nih.gov/pubmed/11035477[↩]
- Jackson AB, DeVivo M. Urological long-term follow-up in women with spinal cord injuries. Arch Phys Med Rehabil. 1992;73(11):1029–1035. https://www.ncbi.nlm.nih.gov/pubmed/1444767[↩]
- Liu CW, Attar KH, Gall A, Shah J, Craggs M. The relationship between bladder management and health-related quality of life in patients with spinal cord injury in the UK. Spinal Cord. 2010;48(4):319–324. https://www.ncbi.nlm.nih.gov/pubmed/19841636[↩][↩]
- Oh SJ, Ku JH, Jeon HG, Shin HI, Paik NJ, Yoo T. Health-related quality of life of patients using clean intermittent catheterization for neurogenic bladder secondary to spinal cord injury. Urology. 2005;65(2):306–310. https://www.ncbi.nlm.nih.gov/pubmed/15708043[↩]
- Madersbacher H, Mürtz G, Stöhrer M. Neurogenic detrusor overactivity in adults: a review on efficacy, tolerability and safety of oral antimuscarinics. Spinal Cord. 2013;51(6):432–441. https://www.ncbi.nlm.nih.gov/pubmed/23743498[↩]
- Kennelly MJ, Lemack GE, Foote JE, Trop CS. Efficacy and safety of oxybutynin transdermal system in spinal cord injury patients with neurogenic detrusor overactivity and incontinence: an open-label, dose-titration study. Urology. 2009;74(4):741–745. https://www.ncbi.nlm.nih.gov/pubmed/19628264[↩]
- George J, Tharion G, Richar J, Macaden AS, Thomas R, Bhattacharji S. The effectiveness of intravesical oxybutynin, propantheline, and capsaicin in the management of neuropathic bladder following spinal cord injury. Scientific World Journal. 2007;7:1683–1690. https://www.ncbi.nlm.nih.gov/pubmed/17982563[↩]
- Bennett N, O’Leary M, Patel AS, Xavier M, Erickson JR, Chancellor MB. Can higher doses of oxybutynin improve efficacy in neurogenic bladder? J Urol. 2004;171(2 Pt 1):749–751. https://www.ncbi.nlm.nih.gov/pubmed/14713802[↩]