Contents
Nutmeg essential oil
Nutmeg oil is a volatile essential oil from nutmeg (the seed of Myristica fragrans Houtt., family Myristicaceae) 1. At present, nutmeg still maintains its status as a unique kitchen spice with growing evidence for many of its traditional uses as a natural remedy. The evergreen nutmeg tree can reach 20 m in height and continues to be cultivated in its original location (Indonesian East Indies islands and Srilanka) as well as the West Indies (Caribbean Grenada islands) where the tree was introduced in the 19th century 2. The lemon-like yellowish fruit contains the seed (nutmeg kernel, Figure 1) which is enveloped in a reddish net-like spongy tissue known as the aril or arillus (nutmeg mace, Figure 1). Both nutmeg kernel and nutmeg arillus are rich in essential oil that imparts the characteristic aroma and taste to nutmeg as a unique culinary ingredient. Primary metabolites (carbohydrates, lipids/fatty acids and proteins) constitute up to 80% of the weight of dry nutmeg kernel while the remaining weight comprises secondary metabolites of diverse chemical nature. They include essential oils (terpenes and phenylpropanoids) and phenolic compounds (caffeic, ferulic and protocatechuic acids, lignans/neolignans, and diarylalaknes) as the major constituents 1. Polyphenols and pigments (catechins, epicatechins, falvonoids, and cyanidins) are also present (Table 1). Nutmeg mace is separately processed from nutmeg and has relatively less fats and carbohydrates.
Table 1. Relative chemical composition of nutmeg kernel and methods of preparation of major constituents
| Chemical class | Concentration (w/w, dry) | Main Constituents (abundance within class) | Preparation Method |
|---|---|---|---|
| I) Primary metabolites: | |||
| Fixed oil (nutmeg butter) | up to 40% | Myristic acid (~8%) & trimyristin triglyceride (~73%) | Expression |
| Carbohydrates | up to 30% | Starch | Insoluble residue |
| Protein | up to 6% | Insoluble residue | |
| II) Secondary metabolites: | |||
| Nutmeg Essential oil | 2–16% | Terpenes (~88%), Phenylpropanoids (~12%) | Steam distillation |
| Small phenolic compounds | varies | Phenolic acids, lignans, diarylalkanes, flavonoids | Organic solvent extraction |
| Resins & pigments | varies | Polyphenolics, polycatechins, tannins, anthocyanins | Variable |
Figure 1. Nutmeg fruit anatomy
Figure 2. Nutmeg kernel
Figure 3. Nutmeg oil
Chemistry of major secondary metabolites of nutmeg essential oil
The essential oil is obtained by the steam distillation of ground nutmeg and is used heavily in the perfumery and pharmaceutical industries. The nutmeg essential oil is used as a natural food flavoring in baked goods, syrups, beverage and sweets etc. Nutmeg essential oil replaces ground nutmeg as it leaves no particles in the food. The essential oil is also used in the cosmetic and pharmaceutical industries for instance in tooth paste and as a major ingredient in some cough syrups. In traditional medicine nutmeg and nutmeg oil were used for illnesses related to the nervous and digestive systems.
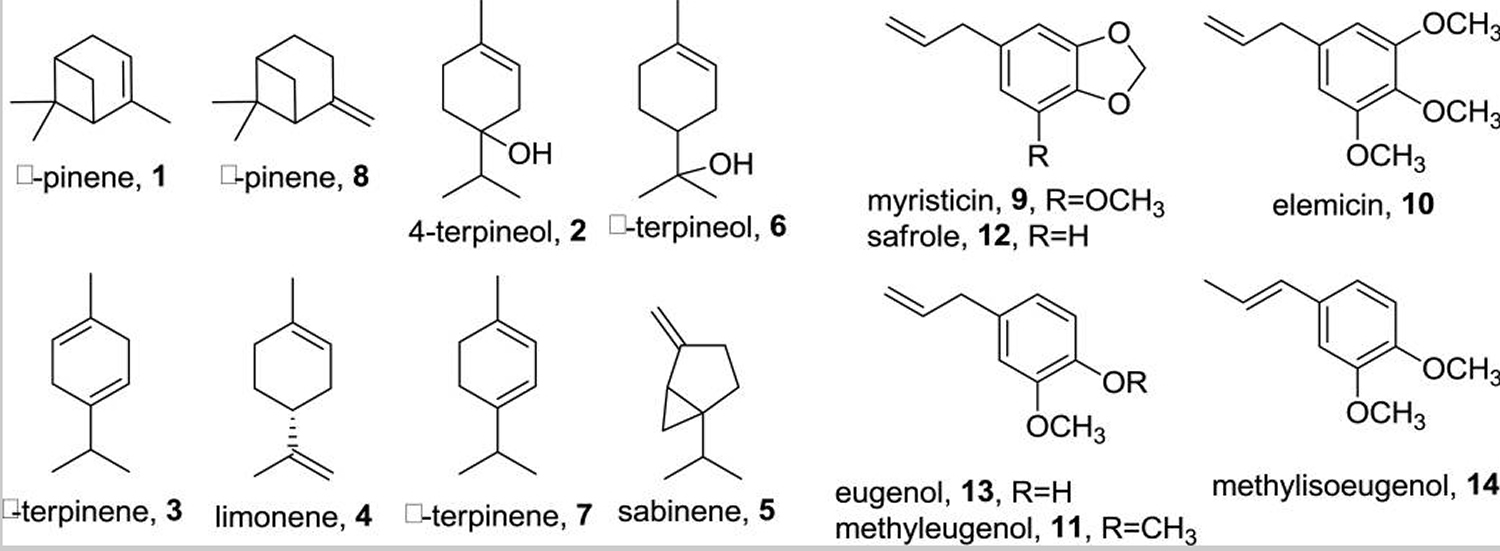
The essential oil constitutes up to 16% of nutmeg (w/w) and is rich in monoterpenes (approximately 90%) and phenylpropanoids 1. A number of recent reports listed between 27–37 components present at various concentrations as determined by gas chromatography–mass spectrometry. In two independent studies by Wahab et al. 5 and Piaru et al. 6, the total number of compounds identified in the nutmeg essential oil was 37. In other studies, Du et al. 7 reported 27 compounds, Muchtaridi and co-workers 8 listed 32 compounds while Piras et al. 9 identified 30 compounds. Based on these most recent reports, the average number of compounds identified in nutmeg essential oil is 34. The monoterpenes -pinene (1, 7.4±3.5 %), 4-terpineol (2, 16.0±10.6 %), -terpinene (3, 5.3±3.3 %), limonene (4, 5.9±2.8 %), sabinene (5, 16.4±4.8 %), -terpineol (6, 2.4±2.2 %), -terpinene (7, 4.4±3.7 %), and -pinene (8, 5.2±3.0 %); and the phenylpropanoids myristicin (9, 12.4±11.7 %), elemicin (10, 1.9±1.7 %), methyleugenol (11, 3.8±7.2 %), safrole (12, 2.6±1.9 %), eugenol (13, 6.8±11.4 %), and methylisoeugenol (14, 5.7±9.6 %) were the most detected constituents in the five reported samples. Interestingly, isoeugenol the propenyl isomer of 13 or the O-demethyl isomer of 14, was present in trace amounts in only one of the five recent reports on the essential oil of nutmeg (Muchtaridi et al. 2010). Mean concentrations shown between parentheses reflects a wide range of variability (chemical structures are shown in Figure 2).
Table 1. Chemical composition of nutmeg essential oil from nutmeg seeds
| No. | Compounds | Percentage % |
| 1 | α-thujene | 0.78 |
| 2 | α-pinene | 10.23 |
| 3 | Camphene | 0.16 |
| 4 | Sabinene | 21.38 |
| 5 | α-myrcena | 2.38 |
| 6 | α-terpinene | 2.72 |
| 7 | Limonene | 5.57 |
| 8 | β-ocimene | 0.03 |
| 9 | γ-terpinene | 3.98 |
| 10 | trans-sabinene hydrate | 0.03 |
| 11 | Terpinolene | 1.62 |
| 12 | Linalool | 0.75 |
| 13 | Fenchyl alcohol | 0.05 |
| 14 | cis-sabinene hydrate | 0.06 |
| 15 | 4-terpineol | 13.92 |
| 16 | α-terpineol | 3.11 |
| 17 | Citronellol | 0.77 |
| 18 | Linalyl acetate | 0.06 |
| 19 | Bornyl acetate | 0.24 |
| 20 | Safrole | 4.28 |
| 21 | Methyl eugeunol | 0.77 |
| 22 | Isoeugeunol | 1.74 |
| 23 | Myristicin | 13.57 |
| 24 | Elimicin | 1.42 |
| 25 | Metoxyeugeunol | 0.1 |
| 26 | β-asaron | 0.03 |
| 27 | Myristic acid | 0.11 |
| 28 | Ethyl miristate | 0.04 |
| 29 | Palmitic acid | 0.03 |
| 30 | Ethyl palmitate | 0.07 |
| 31 | Stearic acid | 0.01 |
| 32 | Ethyl oleate | 0.01 |
Figure 4. Nutmeg essential oil major secondary metabolites – monoterpenes (90% approx.) and phenylpropanoids
[Source 1]Lignans and neolignans
Based on the number of identified compounds, lignans and neolignans constitute the most abundant class of secondary metabolites present in nutmeg kernel and mace 1. A lignan can be loosely identified as a product of tail-to-tail dimerization of two phenylpropanoids while neolignans are formed mainly via head-to-tail coupling, with other possible non-C-2/C-2 couplings. Thus, the basic skeleton of a lignan usually comprises a dibenzyl-substituted tetrahydrofuran, a hexahydrofurofuran or a butane moiety while that of a neolignan may be an ether, a benzofuran, a benzodioxane, or a biphenyl (head-to-head coupling). Due to the abundance of phenylpropanoids (see Figure 4 above number 9-14) in nutmeg, all identified lignans and neolignans represent various dimerization patterns between these five phenylpropanoids.
Diphenylalkanes
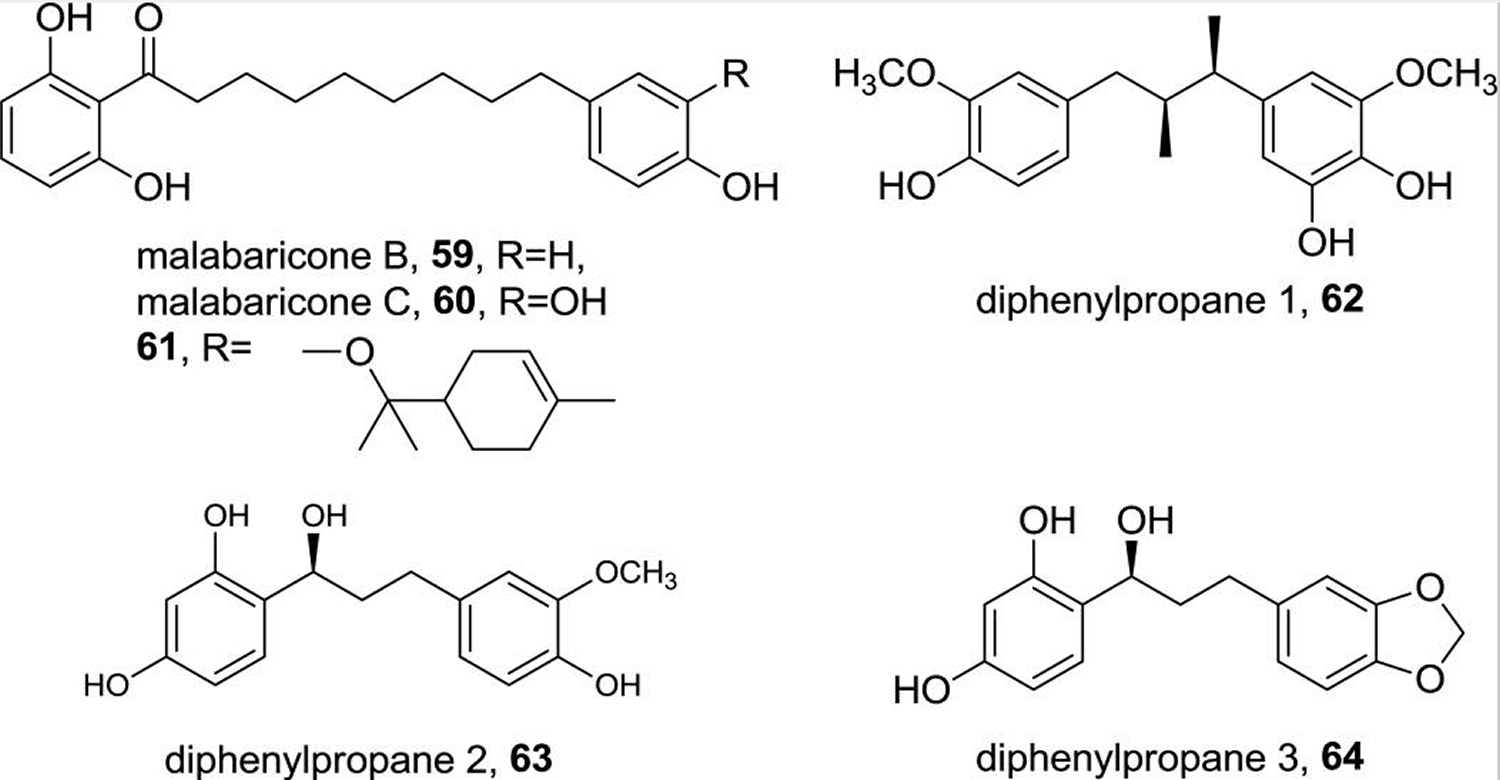
In addition to lignans, which can be chemically classified as diphenylbutanes, two types of diphenylalkanes have been identified in nutmeg. The diphenylnonanoid malabaricones A-D were originally isolated Myristica malabarica (wild nutmeg), and, of these malabaricone B and C (59 & 60, respectively, Figure 5) are among common constituents later isolated from the kernel and mace of nutmeg as well as the bark of another Myristica species, Myristica cinnamomea (compound 60) 10. Another -terpineol ether of malabaricone C (61) was isolated and reported as a new compound by Duan et al 11. 1,3-Diphenylpropanes constitute a number of less common diphenylalkanes that were recently reported by Cuong et al. in the course of a bioassay-directed isolation project. Three compounds were isolated and identified as 5-12. It is noteworthy that diarylpropanoids reported by Forrest et al. in 1974 13 are more appropriately classified as neolignans based on their benzofuranoid and 8,4’-oxyneolignan core structures.
Figure 5. Nutmeg essential oil major secondary metabolites – Diphenylalkanes
It seems that most promising biologically active secondary metabolites of nutmeg are either lignans/neolignans or diarylnonanoids.
The chemical groups, lignans and neolignans seem to be among the most studied. Nguyen et al. 14, in search of novel AMP-protein kinase (5’-adenosine monophosphate-activated protein kinase, AMPK) activators, isolated 2,5-bis-aryl-3,4-dimethyltetarhydrofuran lignans 15-20 & 22 from nutmeg. AMPK enzyme system plays a crucial role in regulating lipid and glucose homeostasis in a various tissue types. Studies have emphasized its role in obesity, diabetes, and cardiovascular diseases. Activation of AMPK has recently emerged as a therapeutic target for the aforementioned disease states 15. Because of antihyperlipidemic and anti-atheroscelorotic activities reported for nutmeg extract 16, Nguyen et al. 14 screened the seed extract of AMPK activator activity, followed by isolation of pure lignan compounds. Out of the seven isolated compounds, tetrahydrofuroguaiacin, nectandrin A, and nectandrin B exerted strong activation of AMPK in differentiated C2C12 muscle cells, at 5 M concentration 14. The study also examined the effect of administration of a nectandrin B-rich active fraction in a high fat-induced animal model of obesity. The results showed a protective effect of the fraction against weight gain and blood glucose elevation caused by the high fat diet, suggesting potential application for nutmeg and its compounds in obesity, type 2 diabetes mellitus, and metabolic syndrome. A follow up study reported that nectandrin B activated AMPK in vascular smooth muscle cells and inhibited vascular smooth muscle cells proliferation and neointima formation, events that are critical in the development of vascular occlusive diseases. An elaborate study of the mechanism of vascular smooth muscle cells anti-proliferative effect revealed that AMPK activation resulted in P53 and P21 induction, that in turn downregulated retinoblastoma (Rb) phosphorylation, E2 transcription factor 1 (E2F1) resulting in inhibition of pin1 gene expression. The cascade of events results in inhibition of intimal hyperplasia 17. These results support a therapeutic potential for nectandrin B in the treatment or prevention of various occlusive vascular diseases, however further studies, particularly clinical studies are still needed.
Reviewing the literature reveals that lignans have received wide attention for their potential role in prevention of osteoporosis. Lignans are classified as phytoestrogens that exert estrogenic activity through binding to the estrogen receptor. Various phytoestrogens, especially isoflavones, have demonstrated beneficial clinical effects against bone loss in both preclinical and clinical studies 18. Machilin A, one of the lignan components of nutmeg, was reported to stimulate osteoblast differentiation through activation of the p38 mitogen activated protein kinase pathway 19. In early stage osteoblast differentiation, machilin A significantly increase alkaline phosphatase (ALP) activity, a commonly used marker for stimulating differentiation. Similarly, machilin A activated late stage differentiation and significant bone mineralization. The observed bone anabolic activity occurred in a dose-dependent manner. Other activities for machilin A include inhibition of proliferation of blood lymphocytes, human leukemia HL-60 cells, and topoisomerase I and II inhibition, suggesting potential anticancer properties 20. Follow-up studies and proper clinical evaluation of these properties remain uninvestigated.
Several pharmacological activities have been attributed to macelignan the main bioactive component identified in nutmeg mace. The activities range from anti-microbial, anti-inflammatory, anti-cancer, to antidiabetic, hepato- and neuro-protective 21. The anti-inflammatory effects of macelignan have been extensively studied. Shin et al. 22 reported that treatment with macelignan prevents the development of allergen-induced asthma in experimental animal models. The protective effect was coupled to a reduction in CD4+ T cells production of interleukin-4 (IL-4), but with no apparent effect on IL-17 or interferon-cells. Animals administered macelignan showed lower expression of the type-2 T helper cell (Th2) transcription factor, GATA3, an effect that needs might contribute to the protective anti-asthma activity, but requires further mechanistic studies. An earlier study demonstrated that macelignan inhibits the activation of mast cells in response to allergen exposure. Macelignan inhibited the release of histamine, calcium influx, degranulation, as well as various inflammatory mediators’ release 23. The protective effect of macelignan has been tested in a variety of models of neurological dysfunction. Cui et al. 24 reported a protective effect of oral administration of macelignan against lipopolysaccharide-induced hippocampus microglial cells in rats. It also protected against impaired spatial learning induced by chronic lipopolysaccharide administration, implying potential therapeutic benefit for Alzheimer’s Disease patients. The mechanism of anti-inflammatory effect was investigated by Ma et al. 25. The study reported that macelignan suppressed lipopolysaccharide-induced activation of the Toll-like receptor 4 pathway, as evidenced by suppression of the nuclear factor NF-κB, reduction in cyclooxygenase type-2 (COX-2) expression, and inhibition of reactive oxygen species (ROS) generation. The results were in agreement with anti-inflammatory and protective effects of macelignan observed in animal models of diabetes and hepatotoxicity 21. A recent study corroborated the neuroprotective effect of macelignan. Using midbrain slice cultures, macelignan treatment protected dopaminergic neurons against the interferon (IFN)-γ and lipopolysaccharide-induced degeneration 26. Mechanistic studies revealed the protective neuroprotective effect observed was mediated by macelignan activation of the peroxisome proliferator activated receptor (PPAR-γ), which in turns activated arginase 1 enzyme expression. The result of the study implicates probable protective role of maceliganan against Parkinson’s Disease and other neurodegenerative disorders. The use of macelignan as an antiphotoaging agent stems from its antioxidant, antionflammtory, as well as its documented ability to protect human skin fibroblasts against damaging effects of UVB irradiation. The observed protective effect is mediated by suppression of two cellular responses involved in premature skin aging: the upregulation of matrix metalloproteinases and reduced collagen synthesis 27. Another potential skin application for macelignan was reported by Choi et al. 28 whereby the authors suggested the use of macelignan as a natural depigmenting agent based on its ability, at 10 M concentration, to inhibit melanosome transfer and dendrite formation in B16F10 melanoma cells. In addition to its earlier documented anti-diabetic effect, macelignan, isolated from Schisandra grandiflora, was recently reported to possess an inhibitory action against advanced glycation end products, an effect that adds to its potential role in the management of diabetes and metabolic syndrome 29.
Nutmeg essential oil uses and potential benefits
In folkloric medicine, nutmeg has long been used as a remedy for gastrointestinal problems, such as flatulence, colic, indigestion and diarrhea 1. The traditional use of nutmeg to treat tumors and infectious diseases, such as parasites and plague, has also been reported 1. Nutmeg has been used externally to treat skin infections, rheumatism and paralysis 2. Other interesting uses for nutmeg include the treatment of psychological disorders 30, and as a cheap substitute for marijuana especially among teenagers, sailors and prison inmates 31.
Despite the myriad of folkloric uses of nutmeg, preclinical and clinical studies supporting such uses are relatively limited. Pharmacological studies have confirmed a few activities of nutmeg extracts, including antidiarrheal, antimicrobial, antioxidant, and different central nervous system (CNS) activities. Gorver et al. 32 examined the effects of crude nutmeg suspension, aqueous, as well as petroleum ether extracts in a variety of activities. The study 32 revealed a significant antidiarrheal effect exerted by both the crude suspension and petroleum ether nutmeg extract. However, only the petroleum ether extract had a significant sedative effect. None of the extracts showed significant cardiovascular effects as measured by blood pressure and ECG changes. On the other hand, a cardioprotective effect against myocardial infarction (heart attack) has been reported. Abdul Kareem et al. 1 examined the effect of daily administration of aqueous nutmeg extract (100 mg/kg, orally) for 30 days on isoproterenol-induced heart attack in adult male rats. Data collected show that pretreatment with nutmeg extract offered protection against isoproterenol effects on blood glucose, plasma lipids, as well as histological myocardiac changes, suggesting a potential cardiovascular protective role of nutmeg consumption.
Very few studies examined the antimicrobial activity of nutmeg extracts. Methanolic extracts possess potent antifungal activity against various plant pathogenic fungi, with three lignans (erythro-austrobailignan-6 [macelignan, 23], meso-dihydroguaiaretic acid [30] and nectandrin B [19]) identified as the primary constituents responsible for the reported antifungal activity 33. Similar antimicrobial activity was reported for nutmeg against the pathogenic Escherichia coli O157 and O111. The activity proved to be selective against the pathogenic versus the non-pathogenic E. coli strains 34. On the other hand, evaluation of nutmeg ethanolic extract against clinical isolates of Staphylococcus aureus, E. coli, and Streptococcus pyogenes resulted in lack of antimicrobial activity against all tested bacteria 35. Ethyl acetate and ethanolic extracts of the seed, mace, and flesh of nutmeg demonstrated high bactericidal activity against several Gram positive and Gram negative oral pathogens 36. In vitro evaluation of antimalarial activity of 27 herbal extracts and 5 formulations proved nutmeg to be among the identified eight extracts that exhibited potent antimalarial activity with IC50 value less than 10 mg/mL. In addition, the activity showed a selectivity index of >10 against multidrug resistant Plasmodium falciparum versus human renal epithelial cells 37. Chemical investigation of nutmeg resulted in the isolation of a wide range of phenolic compounds belonging to the lignan group. With such compounds identified, research efforts intensified at evaluating the antioxidant potential of nutmeg. A study conducted by Assa et al. 38 examined the antioxidant capacity of methanolic extracts of nutmeg mace, seed, and flesh. Results attributed the highest free radical scavenging antioxidant activity to the seed extract based on the 1,1-diphneyl-2-picrylhydrazyl (DPPH) and ferric-reducing antioxidant power assays. Phytochemical evaluation correlated the high antioxidant capacity of the seed extract to its tannin, terpenoid, and flavonoid components. Lack of antioxidant activity of mace was also reported by Yadav and Bhatnagar 39 and was attributed to the relatively low polyphenolic content. The antioxidant properties of the aqueous extract of nutmeg seem to be responsible for its observed antimuatgenic and antimitotic actions against the cyclophosphamide-induced carcinogenic effects in the Allium cepa test 40.
Unlike the plethora of preclinical studies evaluating the nutmeg extracts, very few studies examined the potential activities of nutmeg oil. Wahab et al. 5 reported a dose-dependent activity of nutmeg oil in various animal seizure models. The oil exerted significant anticonvulsant effect against seizures induced by maximal electroshock, pentylenetetrazole, and strychnine, at doses that did not impair locomotor activity. Higher doses of the nutmeg essential oil seem to possess a weak proconvulsant effect, potentiating the clonic seizures induced by both pentylenetetrazole and bicuculline. Administration of nutmeg essential oil to experimental animals via the inhalation route showed a dose-dependent depressant effect on locomotor activity, with potential sedative effect attributed to myristicin, safrole, and 4-terpineol oil constituents 8. Using the DPPH (2,2-diphenyl-1-picrylhydrazyl) free radical scavenging assay, Piaru et al. 6 reported a significant antioxidant activity of nutmeg oil. The oil also showed strong cytotoxic action against colorectal cancer carcinoma cell line and breast carcinoma cell line.
Limited studies focused on preclinical pharmacological evaluation of myristicin. Leiter et al. 41 examined the anxiolytic effect of myristicin (see Figure 4 above – phenylpropanoids of nutmeg essential oil – number 9) in the experimental elevated plus maze animal model. In line with a previous report by Sonvane and co-workers 42, myristicin failed to show anxiolytic action in the Leiter et al. study. The results demonstrated that myristicin may actually have some anxiogenic effect and may antagonize the actions of benzodiazepine, the GABAA receptors allosteric potentiators. In addition the CNS activity, antihelmintic 43, insecticidal 7, apoptotic, and protection against DNA damage 44 effects were reported to myristicin 1. However, most of these activities are scattered in the literature, with no systematic follow up studies that further corroborate the findings or carry them further to application.
The most studied pharmacological activity attributed to nutmeg is its effects on the central nervous system (CNS). As early as the 12th century, nutmeg has been used and known for its central nervous system activity. Available literature has recognized a variety of nervous system effects of nutmeg and its major constituents. Earlier anecdotes report psychoactive and hallucinogenic properties of nutmeg 45. These reports were the basis of Shulgin’s hypothesis that attributed nutmeg’s psychoactivity to metabolic conversion of its main phenylpropanoid constituents to amphetamine-like metabolites 46. So far, the hypothesis has not been experimentally supported. Inconsistent animal findings and lack of detection of the amphetamine-like metabolites in biological fluids of nutmeg abusers led to reevaluation of the validity of the hypothesis 47. Further experimental data have ascribed several additional nervous system effects to nutmeg. Hayfaa et al. 48 reported analgesic activity of acidulated ethanolic extract of nutmeg (1 g/kg dose) in acetic acid-induced writhing animal model in support of earlier reports of the analgesic activity of the n-hexane nutmeg extract 32. Neurobehavioral effects exerted by nutmeg have been documented in various animal models, with numerous activities reported. Sonavane et al. 49 reported an anxiogenic activity exerted by the n-hexane extract of nutmeg, at doses of 10 and 30 mg/kg, i.p., as well by trimyristin (10, 30, and 100 mg/kg, intra-peritoneal). On the other hand, Ayurvedic literature reports the use of aqueous nutmeg extract as an anxiolytic agent 16. Such claim has not been substantiated by experimental dependent anxiolytic activity of aqueous nutmeg extract in the open field test experimental model. Similar to reported results for its effect on anxiety, conflicting data have been documented for nutmeg’s (and its components) effect on depression. Dhingra et al. 50 and Moinuddin et al. 51 reported antidepressant activity of nutmeg extracts in both models of behavioral despair as well in reserpine reversal test paradigms, respectively. The studies also suggested the involvement of adrenergic, serotonergic, and dopaminergic systems in the observed antidepressant effect, since it was inhibited by 1 and dopaminergic receptor antagonists as well as a serotonin synthesis inhibitor. On the other hand, tryimyristin exerted a depressant effect when tested in behavioral despair animal models and potentiated hypothermia induced by reserpine. The observed effects were blocked by pre-administration of a serotonin 5-HT2A receptor antagonist 52. A previous study in a laboratory 53 evaluated the neurobehavioral effects of nutmeg in the four-point tetrad assay as compared to common drugs of abuse, Δ9-tetrahydrocannabinol (Δ9-THC), morphine, and amphetamine. The results of the study showed that nutmeg extracts have various activities in the assay, depending on the nature of the extract, as well as the route of administration. The study demonstrated that the dichloromethane nutmeg extract, when injected at 100 and 300 mg/kg doses, intra-peritoneal, exerted some cannabimimetic activity in the tetrad assay at 53.
Maity and co-workers reported a significant healing effect of malabaricone B against indomethacin-induced stomach ulcer 54. Administration of malabaricone B attenuated the increased nitric oxide synthesis induced by indomethacin, while enhancing the arginase pathway, thus favoring an anti / pro inflammatory cytokine ratio. Malabaricone C has potential pharmacological benefits in vascular disease 55, promotion of healing, anti-inflammation and angiogenesis caused by stomach ulcers 54, antioxidant activity 56 and cytotoxic activity against certain cancers 57. Malabaricone C also has anti-anaerobic, antifungal, and antibacterial properties 58 as well as anti-parasitic, leishmanicidal and nematocidal activity 59.
For a long time studies have focused on lignans as the main active components of nutmeg. Recent research efforts explored the activities of neolignan-type compounds. Kang et al. reported an antiplatelet activity to erythro-(7S,8R)-7-acetoxy-3,4,3’,5’-tetramethoxy-8-O-4’-neolignan (EATN) 60. Results of the study showed that erythro-(7S,8R)-7-acetoxy-3,4,3’,5’-tetramethoxy-8-O-4’-neolignan (EATN), exerted a concentration dependent inhibition of platelet aggregation induced by platelet activating-factor and thrombin, and arachidonic acid. EATN had IC50 values of 3.2 ± 0.4 and 3.4 ± 0.4 M against platelet activating factor and thrombin-induced platelet aggregation, respectively. Further mechanistic investigation delineated the mechanism of anti-platelet activity. EATN regulates the level of cAMP, a crucial second messenger in the activation of platelet aggregation. EATN elevates intracellular cAMP levels inhibiting the Ca2+-induced mobilization of platelets activated by thrombin. Though these results are promising and elude to potential therapeutic application of EATN in atherothrombotic diseases, in vivo and clinical studies are still in demand. Licarin E, another neolignan, proved to protect against UVB irradiation damage to human skin fibroblasts 61. It reversed the two events induced by UVB: elevation of matrix metalloproteinase-1 and reduction of procollagen expression. The molecular mechanism of these effects proved to be via stimulation of transforming growth factor (TGF )/Smad signaling pathway. Similar to macelignan, licarin E could offer a novel therapeutic agent for photoaging. Using rat basophilic leukemia cells, stimulated by dinitrophenyl-human serum albumin, the effect of the neolignan licarin A on histamine release and mast cell activation was examined 62. The results demonstrated that licarin A inhibited mast cell activation, as evidenced by inhibiting tumor necrosis factor (TNF ), COX-2, and prostaglandin production. Further studies are needed to corroborate the role of licarin A in the treatment of immediate hypersensitivity cases. In vitro studies have elucidated that licarin A, isolated from Machilus thunbergii, may possess neuroprotective value against glutamate-induced toxicity of rat cortical cells 63. The protective effect was also evident against kainic acid-induced neurotoxicity, though more selective protection was observed for glutamate toxicity. The neuroprotective effect was attributed to the potent antioxidant properties of licarin A, evidenced by reduction of NO (nitric oxide), peroxide, free radical production as well as enhancing the activity of antioxidant enzyme systems. In addition, licarin A effectively suppressed Ca2+ influx that is typically induced by glutamate. Synthetic (−)-licarin A was reported to possess concentration-dependent anti-parasitic activity 64. In vitro studies reported potent inhibition of growth Leishmania promastigotes, suggesting promising application as a leishmanicidal agent. Using a tuberculosis murine animal model, Leon-Diaz et al. 65 demonstrated that licarin A possesses a significant suppressant action of the pulmonary burden and pneumonia in animals infected with both drug sensitive as well as drug-resistant tuberculosis strains. Animals administered licarin A (5mg/kg for 30 days) showed significant reduction in lung bacilli and pneumonia incidence. The anti-parasitic effect of licarin A was further corroborated against both Schistosoma mansoni and Trypanosoma cruzi 66. It is evident that the potential anti-parasitic activity of licarin A is worth further investigation in the hope of future development of effective medications.
Nutmeg essential oil side effects
Myristicin (see Figure 4 above – phenylpropanoids of nutmeg essential oil – number 9) is the major phenylpropanoid constituent of nutmeg essential oil. Various toxicological reports have attributed adverse effects associated with nutmeg ingestion to myristicin 1. These adverse effects include gastrointestinal; vomiting, and paralytic ileus, nervous system; drowsiness, paresthesia, numbness, reality detachment, and cardiovascular; hypotension, tachycardia, symptoms 32, 67.
The central nervous system depressant effect of trimyristin, the main triglyceride constituent of nutmeg butter, was documented in mice 52. Trimyristin significantly increased immobility time in both forced swim and tail suspension murine tests, indicating a depressant-like action, when administered intraperitoneal injection (abdominal cavity injection) at 10 and 30 mg/kg doses. Furthermore the activity was inhibited by various antidepressant medications including the selective serotonin reuptake inhibitor fluoxetine, the tricyclic antidepressants imipramine, and the atypical antidepressant mianserin. However, the mechanism of such depressant action remains unclear.
Summary of nutmeg essential oil uses and benefits
Nutmeg kernel and mace have a long history of use as a spice and traditional remedy that goes back to the 12th century. Traditional uses of nutmeg in alleviating gastrointestinal disorders, managing rheumatic pain, healing skin wounds and infections as well as its use as a calming agent resulted in massive contemporary efforts to evaluate its different extracts, fractions. Despite the traditional uses and activities reported for nutmeg, the mechanisms underlying its effects remain unclear and further pharmacological studies are certainly needed to properly assess the therapeutic potential of this natural product 68, 2.
One of the most commonly evaluated activities of nutmeg essential oil and extracts is their effect on the CNS. In addition to the essential oil which is rich in terpenes and phenylpropanoids, nutmeg has significant levels of non-volatile secondary metabolites of the lignan/neolignan-type as well as diarylalkanes that have been isolated and identified. Preclinical studies to evaluate such compounds as myristicin, macelignan, nectandrin A & B, licarin A-E, and malabaricone B & C are abundant in recent literature with focus on anxiolytic, antioxidant/chemopreventive, anti-inflammatory, anti-infective effects of individual compounds. On the other hand, clinical trials are not as abundant in the current literature. They are limited in size, design and focus (topical application of nutmeg or use of a mixed herbal product containing nutmeg among other extracts) with no individual pure compounds included.
While phytochemical studies have supported some of the folkloric uses of nutmeg, very few clinical studies systematically investigated the clinical efficacy of nutmeg uses. A thorough literature search revealed merely two small clinical studies. A randomized, placebo-controlled, double blind trial examined the clinical effects of using topical nutmeg extract in patients with painful diabetic neuropathy 69. The study included 74 diabetic neuropathy patients (males and females, ages 30–85 years) who were randomized to receive the different topical treatment: nutmeg extract, mace oil, nutmeg oil, coconut oil, methyl salicylate, menthol, or placebo. The study used a validated Brief Pain Inventory that has been modified for painful diabetic neuropathy in addition to the Neuropathic Pain Symptom Inventory. Following four weeks of treatment, patients showed within group significant improvement of pain, mood scores, and daily functions. However no statistically significant effect was reported between the nutmeg treated and placebo groups. Being the only study that assessed the clinical analgesic effect of nutmeg, it is hard to draw conclusions because of the study limitations. The small sample size, short duration of the study, lack of inert placebo, poor patient compliance, and use of non-standardized nutmeg preparations are all limitations that hinder proper evaluation of the therapeutic role of nutmeg in pain disorders. The other study retrieved describes an open, uncontrolled trial that examined the effect of administration of a nutmeg-containing herbal product in a total of 251 patients 70. The product used, Revivin, contained a mixture of various plant extracts including nutmeg in addition to carbohydrate molecules, and is commonly used to enhance performance, improve appetite, and reduce weakness and fatigue. Patients (average age 44 years, males and females) received the capsule daily for 4 weeks. Outcomes were assessed by self-filled patients’ questionnaire. Patients reported improvement in mood, insomnia, and overall weakness, and no adverse effects were reported by the patient. The study suffers from several limitations: subjectivity of outcome evaluation, short duration, and lack of placebo control. In addition, since the study used a product where nutmeg constituted only one component of the mixture, the clinical nutmeg effect cannot be isolated. As evident, there is a lack of well-designed controlled clinical trials that evaluate the potential therapeutic place of nutmeg and its components.
- Abourashed EA, El-Alfy AT. Chemical diversity and pharmacological significance of the secondary metabolites of nutmeg (Myristica fragrans Houtt.). Phytochemistry reviews : proceedings of the Phytochemical Society of Europe. 2016;15(6):1035-1056. doi:10.1007/s11101-016-9469-x. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5222521/[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Abourashed EA, Khan IA. Nutmeg. In: Khan IA, Abourashed EA, editors. Leung’s Encyclopedia of Common Natural Ingredients Used in Food, Drugs and Cosmetics. 3rd. Hoboken, NJ: Wiley; 2010. pp. 467–470.[↩][↩][↩][↩]
- Olaleye MT, Akinmoladun AC, Akindahunsi AA. Antioxidant properties of Myristica fragrans (Houtt) and its effect on selected organs of albino rats. Afr J Biotechnol. 2006;5:1274–1278.[↩]
- Daniel D. Nutmeg processing and marketing in Grenada. Food And Agriculture Organization of the United Nations Report. 1994:6–10.[↩]
- Anticonvulsant activities of nutmeg oil of Myristica fragrans. Wahab A, Ul Haq R, Ahmed A, Khan RA, Raza M. Phytother Res. 2009 Feb; 23(2):153-8. https://www.ncbi.nlm.nih.gov/pubmed/19067329/[↩][↩]
- Chemical composition, antioxidant and cytotoxicity activities of the essential oils of Myristica fragrans and Morinda citrifolia. Piaru SP, Mahmud R, Abdul Majid AM, Ismail S, Man CN. J Sci Food Agric. 2012 Feb; 92(3):593-7. https://www.ncbi.nlm.nih.gov/pubmed/25520982/[↩][↩]
- Chemical constituents and activities of the essential oil from Myristica fragrans against cigarette beetle Lasioderma serricorne. Du SS, Yang K, Wang CF, You CX, Geng ZF, Guo SS, Deng ZW, Liu ZL. Chem Biodivers. 2014 Sep; 11(9):1449-56. https://www.ncbi.nlm.nih.gov/pubmed/25238085/[↩][↩]
- Identification of compounds in the essential oil of nutmeg seeds (Myristica fragrans Houtt.) that inhibit locomotor activity in mice. Muchtaridi, Subarnas A, Apriyantono A, Mustarichie R. Int J Mol Sci. 2010 Nov 23; 11(11):4771-81. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3000115/[↩][↩][↩]
- Extraction and separation of volatile and fixed oils from seeds of Myristica fragrans by supercritical CO₂: chemical composition and cytotoxic activity on Caco-2 cancer cells. Piras A, Rosa A, Marongiu B, Atzeri A, Dessì MA, Falconieri D, Porcedda S. J Food Sci. 2012 Apr; 77(4):C448-53. https://www.ncbi.nlm.nih.gov/pubmed/22429024/[↩]
- Malabaricone C from Myristica cinnamomea exhibits anti-quorum sensing activity. Chong YM, Yin WF, Ho CY, Mustafa MR, Hadi AH, Awang K, Narrima P, Koh CL, Appleton DR, Chan KG. J Nat Prod. 2011 Oct 28; 74(10):2261-4. https://www.ncbi.nlm.nih.gov/pubmed/21910441/[↩]
- Cytotoxic and antioxidative phenolic compounds from the traditional Chinese medicinal plant, Myristica fragrans. Duan L, Tao HW, Hao XJ, Gu QQ, Zhu WM. Planta Med. 2009 Sep; 75(11):1241-5. https://www.ncbi.nlm.nih.gov/pubmed/19326328/[↩]
- 2R,3S)-4-(4-hydroxy-3-methoxyphenyl)-3-methylbutan-2-yl)-3-methoxybenzene-1,2-diol (62), (S)-4-(1-hydroxy-3-(4-hydroxy-3-methoxyphenyl)propyl)benzene-1,3-diol (63) and (S)-4-(3-(benzo[d][1,3]dioxol-5-yl)-1-hydroxypropyl)benzene-1,3-diol (64) ((Inhibitory effect on NO production of phenolic compounds from Myristica fragrans. Cuong TD, Hung TM, Na M, Ha do T, Kim JC, Lee D, Ryoo S, Lee JH, Choi JS, Min BS. Bioorg Med Chem Lett. 2011 Nov 15; 21(22):6884-7. https://www.ncbi.nlm.nih.gov/pubmed/21978676/[↩]
- Diarylpropanoids from nutmeg and mace (Myristica fragrans Houtt.). Forrest JE, Heacock RA, Forrest TP. J Chem Soc Perkin 1. 1974; 2():205-9. https://www.ncbi.nlm.nih.gov/pubmed/4856212/[↩]
- AMP-activated protein kinase (AMPK) activators from Myristica fragrans (nutmeg) and their anti-obesity effect. Nguyen PH, Le TV, Kang HW, Chae J, Kim SK, Kwon KI, Seo DB, Lee SJ, Oh WK. Bioorg Med Chem Lett. 2010 Jul 15; 20(14):4128-31. https://www.ncbi.nlm.nih.gov/pubmed/20541406/[↩][↩][↩]
- AMP-Activated Protein Kinase Signalling in Cancer and Cardiac Hypertrophy. Lipovka Y, Konhilas JP. Cardiovasc Pharm Open Access. 2015 Aug; 4(3). https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4718578/[↩]
- Sharma PV. Dravyaguna Vijnana (Vegetable Drugs), Vol. 1 edn. India: Varanasi, Chaukambha Bharti Academy; 2001. pp. 151–160.[↩][↩]
- Protective effect of nectandrin B, a potent AMPK activator on neointima formation: inhibition of Pin1 expression through AMPK activation. Ki SH, Lee JW, Lim SC, Hien TT, Im JH, Oh WK, Lee MY, Ji YH, Kim YG, Kang KW. Br J Pharmacol. 2013 Feb; 168(4):932-45. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3631381/[↩]
- Phytoestrogens in postmenopause: the state of the art from a chemical, pharmacological and regulatory perspective. Poluzzi E, Piccinni C, Raschi E, Rampa A, Recanatini M, De Ponti F. Curr Med Chem. 2014; 21(4):417-36. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3963458/[↩]
- Machilin A isolated from Myristica fragrans stimulates osteoblast differentiation. Lee SU, Shim KS, Ryu SY, Min YK, Kim SH. Planta Med. 2009 Feb; 75(2):152-7. https://www.ncbi.nlm.nih.gov/pubmed/19096999/[↩]
- Lignans from the bark of Machilus thunbergii and their DNA topoisomerases I and II inhibition and cytotoxicity. Li G, Lee CS, Woo MH, Lee SH, Chang HW, Son JK. Biol Pharm Bull. 2004 Jul; 27(7):1147-50. https://www.ncbi.nlm.nih.gov/pubmed/15256759/[↩]
- Multiple biological properties of macelignan and its pharmacological implications. Paul S, Hwang JK, Kim HY, Jeon WK, Chung C, Han JS. Arch Pharm Res. 2013 Mar; 36(3):264-72. https://www.ncbi.nlm.nih.gov/pubmed/23435944/[↩][↩]
- Macelignan attenuated allergic lung inflammation and airway hyper-responsiveness in murine experimental asthma. Shin K, Chung HC, Kim DU, Hwang JK, Lee SH. Life Sci. 2013 Jun 13; 92(22):1093-9. https://www.ncbi.nlm.nih.gov/pubmed/23643632/[↩]
- Macelignan inhibits histamine release and inflammatory mediator production in activated rat basophilic leukemia mast cells. Han YS, Kim MS, Hwang JK. Inflammation. 2012 Oct; 35(5):1723-31. https://www.ncbi.nlm.nih.gov/pubmed/22729280/[↩]
- Macelignan attenuates LPS-induced inflammation and reduces LPS-induced spatial learning impairments in rats. Cui CA, Jin DQ, Hwang YK, Lee IS, Hwang JK, Ha I, Han JS. Neurosci Lett. 2008 Dec 19; 448(1):110-4. https://www.ncbi.nlm.nih.gov/pubmed/18940231/[↩]
- Macelignan attenuates activations of mitogen-activated protein kinases and nuclear factor kappa B induced by lipopolysaccharide in microglial cells. Ma J, Hwang YK, Cho WH, Han SH, Hwang JK, Han JS. Biol Pharm Bull. 2009 Jun; 32(6):1085-90. https://www.ncbi.nlm.nih.gov/pubmed/19483320/[↩]
- A natural compound macelignan protects midbrain dopaminergic neurons from inflammatory degeneration via microglial arginase-1 expression. Kiyofuji K, Kurauchi Y, Hisatsune A, Seki T, Mishima S, Katsuki H. Eur J Pharmacol. 2015 Aug 5; 760():129-35. https://www.ncbi.nlm.nih.gov/pubmed/25917324[↩]
- Effects of macelignan isolated from Myristica fragrans (Nutmeg) on expression of matrix metalloproteinase-1 and type I procollagen in UVB-irradiated human skin fibroblasts. Lee KE, Mun S, Pyun HB, Kim MS, Hwang JK. Biol Pharm Bull. 2012; 35(10):1669-75. https://www.ncbi.nlm.nih.gov/pubmed/23037157/[↩]
- Macelignan inhibits melanosome transfer mediated by protease-activated receptor-2 in keratinocytes. Choi EJ, Kang YG, Kim J, Hwang JK. Biol Pharm Bull. 2011; 34(5):748-54. https://www.ncbi.nlm.nih.gov/pubmed/21532167/[↩]
- Advanced glycation end-products inhibitors isolated from Schisandra grandiflora. Poornima B, Kumar DA, Siva B, Venkanna A, Vadaparthi PR, Kumar K, Tiwari AK, Babu KS. Nat Prod Res. 2016; 30(4):493-6. https://www.ncbi.nlm.nih.gov/pubmed/25812677/[↩]
- Formulas used by Tibetan doctors at Men-Tsee-Khang in India for the treatment of neuropsychiatric disorders and their correlation with pharmacological data. Antonio RL, Kozasa EH, Galduróz JC, Dawa, Dorjee Y, Kalsang T, Norbu T, Tenzin T, Rodrigues E. Phytother Res. 2013 Apr; 27(4):552-63. https://www.ncbi.nlm.nih.gov/pubmed/22674653/[↩]
- Weil AT. The use of nutmeg as a psychotropic agent. B Narcotics. 1966;18:15–23.[↩]
- Pharmacological studies on Myristica fragrans–antidiarrheal, hypnotic, analgesic and hemodynamic (blood pressure) parameters. Grover JK, Khandkar S, Vats V, Dhunnoo Y, Das D. Methods Find Exp Clin Pharmacol. 2002 Dec; 24(10):675-80. https://www.ncbi.nlm.nih.gov/pubmed/12616960/[↩][↩][↩][↩]
- Isolation and antifungal activity of lignans from Myristica fragrans against various plant pathogenic fungi. Cho JY, Choi GJ, Son SW, Jang KS, Lim HK, Lee SO, Sung ND, Cho KY, Kim JC. Pest Manag Sci. 2007 Sep; 63(9):935-40. https://www.ncbi.nlm.nih.gov/pubmed/17659535/[↩]
- Antimicrobial activity of nutmeg against Escherichia coli O157. Takikawa A, Abe K, Yamamoto M, Ishimaru S, Yasui M, Okubo Y, Yokoigawa K. J Biosci Bioeng. 2002; 94(4):315-20. https://www.ncbi.nlm.nih.gov/pubmed/16233309/[↩]
- Antibacterial activity of crude extracts of prasaprohyai formula and its components against pathogenic bacteria. Sattaponpan C, Kondo S. J Med Assoc Thai. 2011 Dec; 94 Suppl 7():S153-61. https://www.ncbi.nlm.nih.gov/pubmed/22619922/[↩]
- Antibacterial Activity of Myristica fragrans against Oral Pathogens. Shafiei Z, Shuhairi NN, Md Fazly Shah Yap N, Harry Sibungkil CA, Latip J. Evid Based Complement Alternat Med. 2012; 2012():825362. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3434417/[↩]
- Antimalarial activities of medicinal plants and herbal formulations used in Thai traditional medicine. Thiengsusuk A, Chaijaroenkul W, Na-Bangchang K. Parasitol Res. 2013 Apr; 112(4):1475-81. https://www.ncbi.nlm.nih.gov/pubmed/23340720[↩]
- Assa JR, Widjanarko SB, Kusnadi J, Berhimpon S. Antioxidant potential of flesh, seed, and mace of nutmeg (Myristica fragrans Houtt) Int J Chem Tech Res. 2014;6:2460–2468.[↩]
- Free radical scavenging activity, metal chelation and antioxidant power of some of the Indian spices. Yadav AS, Bhatnagar D. Biofactors. 2007; 31(3-4):219-27. https://www.ncbi.nlm.nih.gov/pubmed/18997285/[↩]
- Antioxidants in aqueous extract of Myristica fragrans (Houtt.) suppress mitosis and cyclophosphamide-induced chromosomal aberrations in Allium cepa L. cells. Akinboro A, Mohamed KB, Asmawi MZ, Sulaiman SF, Sofiman OA. J Zhejiang Univ Sci B. 2011 Nov; 12(11):915-22. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3208171/[↩]
- Evaluation of the anxiolytic properties of myristicin, a component of nutmeg, in the male Sprague-Dawley rat. Leiter E, Hitchcock G, Godwin S, Johnson M, Sedgwick W, Jones W, McCall S, Ceremuga TE. AANA J. 2011 Apr; 79(2):109-14. https://www.ncbi.nlm.nih.gov/pubmed/21560973/[↩]
- Anxiogenic activity of Myristica fragrans seeds. Sonavane GS, Sarveiya VP, Kasture VS, Kasture SB. Pharmacol Biochem Behav. 2002 Jan-Feb; 71(1-2):239-44. https://www.ncbi.nlm.nih.gov/pubmed/11812528/[↩]
- Antihelmintic effects of nutmeg (Myristica fragans) on Anisakis simplex L3 larvae obtained from Micromesistius potassou. López V, Gerique J, Langa E, Berzosa C, Valero MS, Gómez-Rincón C. Res Vet Sci. 2015 Jun; 100():148-52. https://www.ncbi.nlm.nih.gov/pubmed/25890576/[↩]
- Myristicin from nutmeg induces apoptosis via the mitochondrial pathway and down regulates genes of the DNA damage response pathways in human leukaemia K562 cells. Martins C, Doran C, Silva IC, Miranda C, Rueff J, Rodrigues AS. Chem Biol Interact. 2014 Jul 25; 218():1-9. https://www.ncbi.nlm.nih.gov/pubmed/24792648/[↩]
- The pharmacology of myristicin. A contribution to the psychopharmacology of nutmeg. TRUITT EB Jr, CALLAWAY E 3rd, BRAUDE MC, KRANTZ JC Jr. J Neuropsychiatr. 1961 Apr; 2():205-10. https://www.ncbi.nlm.nih.gov/pubmed/13778274/[↩]
- Possible implication of myristicin as a psychotropic substance. Shulgin AT. Nature. 1966 Apr 23; 210(5034):380-4. https://www.ncbi.nlm.nih.gov/pubmed/5336379/[↩]
- Abuse of nutmeg (Myristica fragrans Houtt.): studies on the metabolism and the toxicologic detection of its ingredients elemicin, myristicin, and safrole in rat and human urine using gas chromatography/mass spectrometry. Beyer J, Ehlers D, Maurer HH. Ther Drug Monit. 2006 Aug; 28(4):568-75. https://www.ncbi.nlm.nih.gov/pubmed/16885726/[↩]
- Evaluation of analgesic activity and toxicity of alkaloids in Myristica fragrans seeds in mice. Hayfaa AA, Sahar AM, Awatif MA. J Pain Res. 2013; 6():611-5. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3738253/[↩]
- Sonavane GS, Sarveiya V, Kasture V, Kastre SB. Behavioral actions of Myristica fragrans seeds. Indian J Pharmacol. 2001;33:417–424.[↩]
- Comparative brain cholinesterase-inhibiting activity of Glycyrrhiza glabra, Myristica fragrans, ascorbic acid, and metrifonate in mice. Dhingra D, Parle M, Kulkarni SK. J Med Food. 2006 Summer; 9(2):281-3. https://www.ncbi.nlm.nih.gov/pubmed/16822217/[↩]
- Evaluation of the anti-depressant activity of Myristica fragrans (Nutmeg) in male rats. Moinuddin G, Devi K, Kumar Khajuria D. Avicenna J Phytomed. 2012 Spring; 2(2):72-8. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4075663/[↩]
- Kasture SB, Gujar KN. Depressant effect of trimyristin and its inhibition by some antidepressants in mice. In: Bernath J, Nemeth E, Cracker LE, Gardner ZE, editors. Proceedings WOCMAO III 2005, Vol. 1: Bioprospecting & Ethnopharmacology. Acta Hort. Vol. 675. ISHS; 2005. pp. 147–152.[↩][↩]
- Towards a better understanding of the psychopharmacology of nutmeg: Activities in the mouse tetrad assay. El-Alfy AT, Wilson L, ElSohly MA, Abourashed EA. J Ethnopharmacol. 2009 Nov 12; 126(2):280-6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2783227/[↩][↩]
- Regulation of arginase/nitric oxide synthesis axis via cytokine balance contributes to the healing action of malabaricone B against indomethacin-induced gastric ulceration in mice. Maity B, Banerjee D, Bandyopadhyay SK, Chattopadhyay S. Int Immunopharmacol. 2009 Apr; 9(4):491-8. https://www.ncbi.nlm.nih.gov/pubmed/19291837[↩][↩]
- Malabaricone C inhibits PDGF-induced proliferation and migration of aortic smooth muscle cells through induction of heme oxygenase-1. Lee S, Seo J, Ryoo S, Cuong TD, Min BS, Lee JH. J Cell Biochem. 2012 Sep; 113(9):2866-76. https://www.ncbi.nlm.nih.gov/pubmed/22511233[↩]
- Antioxidant activity of Myristica malabarica extracts and their constituents. Patro BS, Bauri AK, Mishra S, Chattopadhyay S. J Agric Food Chem. 2005 Aug 24; 53(17):6912-8. https://www.ncbi.nlm.nih.gov/pubmed/16104820/[↩]
- Mechanism of the malabaricone C-induced toxicity to the MCF-7 cell line. Tyagi M, Patro BS, Chattopadhyay S. Free Radic Res. 2014 Apr; 48(4):466-77. https://www.ncbi.nlm.nih.gov/pubmed/24456233/[↩]
- Malabaricone C from Myristica cinnamomea exhibits anti-quorum sensing activity. Chong YM, Yin WF, Ho CY, Mustafa MR, Hadi AH, Awang K, Narrima P, Koh CL, Appleton DR, Chan KG. J Nat Prod. 2011 Oct 28; 74(10):2261-4. https://www.ncbi.nlm.nih.gov/pubmed/21910441[↩]
- Antipromastigote activity of the malabaricones of Myristica malabarica (rampatri). Sen R, Bauri AK, Chattopadhyay S, Chatterjee M. Phytother Res. 2007 Jun; 21(6):592-5. https://www.ncbi.nlm.nih.gov/pubmed/17335115/[↩]
- Anti-platelet activity of erythro-(7S,8R)-7-acetoxy-3,4,3′,5′-tetramethoxy-8-O-4′-neolignan from Myristica fragrans. Kang JW, Min BS, Lee JH. Phytother Res. 2013 Nov; 27(11):1694-9. https://www.ncbi.nlm.nih.gov/pubmed/23296979/[↩]
- Effects of licarin E on expression of matrix metalloproteinase-1 and type-1 procollagen in UVB-irradiated human skin fibroblasts. Kwon YY, Kim D, Kim J, Hwang JK. Phytother Res. 2011 Dec; 25(12):1891-4. https://www.ncbi.nlm.nih.gov/pubmed/21544886/[↩]
- Licarin A is a candidate compound for the treatment of immediate hypersensitivity via inhibition of rat mast cell line RBL-2H3 cells. Matsui T, Ito C, Masubuchi S, Itoigawa M. J Pharm Pharmacol. 2015 Dec; 67(12):1723-32. https://www.ncbi.nlm.nih.gov/pubmed/26376734/[↩]
- Meso-dihydroguaiaretic acid and licarin A of Machilus thunbergii protect against glutamate-induced toxicity in primary cultures of a rat cortical cells. Ma CJ, Kim SR, Kim J, Kim YC. Br J Pharmacol. 2005 Nov; 146(5):752-9. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1751201/[↩]
- Neolignan Licarin A presents effect against Leishmania (Leishmania) major associated with immunomodulation in vitro. Néris PL, Caldas JP, Rodrigues YK, Amorim FM, Leite JA, Rodrigues-Mascarenhas S, Barbosa-Filho JM, Rodrigues LC, Oliveira MR. Exp Parasitol. 2013 Oct; 135(2):307-13. https://www.ncbi.nlm.nih.gov/pubmed/23891943/[↩]
- Antitubercular activity and the subacute toxicity of (-)-Licarin A in BALB/c mice: a neolignan isolated from Aristolochia taliscana. León-Díaz R, Meckes-Fischer M, Valdovinos-Martínez L, Campos MG, Hernández-Pando R, Jiménez-Arellanes MA. Arch Med Res. 2013 Feb; 44(2):99-104. https://www.ncbi.nlm.nih.gov/pubmed/23291382/[↩]
- Schistosomicidal and trypanocidal structure-activity relationships for (±)-licarin A and its (-)- and (+)-enantiomers. Pereira AC, Magalhães LG, Gonçalves UO, Luz PP, Moraes AC, Rodrigues V, da Matta Guedes PM, da Silva Filho AA, Cunha WR, Bastos JK, Nanayakkara NP, e Silva ML. Phytochemistry. 2011 Aug; 72(11-12):1424-30. https://www.ncbi.nlm.nih.gov/pubmed/21570099/[↩]
- Toxicology of nutmeg abuse. Sangalli BC, Chiang W. J Toxicol Clin Toxicol. 2000; 38(6):671-8. https://www.ncbi.nlm.nih.gov/pubmed/11185977/[↩]
- Asgarpanah J, Kazemivash N. Phytochemistry and pharmacologic properties of Myristica fragrans Houtt.: A review. Afr J Biotechnol. 2012;11:12787–12793.[↩]
- Nutmeg extracts for painful diabetic neuropathy: a randomized, double-blind, controlled study. Motilal S, Maharaj RG. J Altern Complement Med. 2013 Apr; 19(4):347-52. https://www.ncbi.nlm.nih.gov/pubmed/23098698/[↩]
- Evaluation of the efficacy and safety of a new herbal revitalizer revivin. Rani PU, Naidu MU, Kumar TR, Shobha JC. Anc Sci Life. 1997 Jan; 16(3):190-5. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3331163/pdf/ASL-16-190.pdf[↩]