Contents
What is orchitis
Orchitis is swelling (inflammation) of one or both of the testicles, which is rarely isolated. Usually, bacteria retrogradely seed into the testis from the bladder or prostate. Can also be secondary to viral infection (e.g. mumps, Coxsackie virus). Isolated orchitis can be seen in mumps or bacterial infection.
Bacterial orchitis can be caused by sexually transmitted infections (STIs), particularly gonorrhea or chlamydia. Bacterial orchitis often results from epididymitis, an inflammation of the coiled tube (epididymis) at the back of the testicle that stores and carries sperm. In that case, it’s called epididymo-orchitis.
Orchitis causes pain and can affect fertility. Medication can treat the causes of bacterial orchitis and can ease some signs and symptoms of viral orchitis. But it may take several weeks for scrotal tenderness to disappear.
If you experience pain or swelling in your scrotum, especially if the pain occurs suddenly, see your doctor right away.
A number of conditions can cause testicle pain, and some of the conditions require immediate treatment. One such condition involves twisting of the spermatic cord (testicular torsion), which may cause pain similar to that caused by orchitis. Your doctor can perform tests to determine which condition is causing your pain.
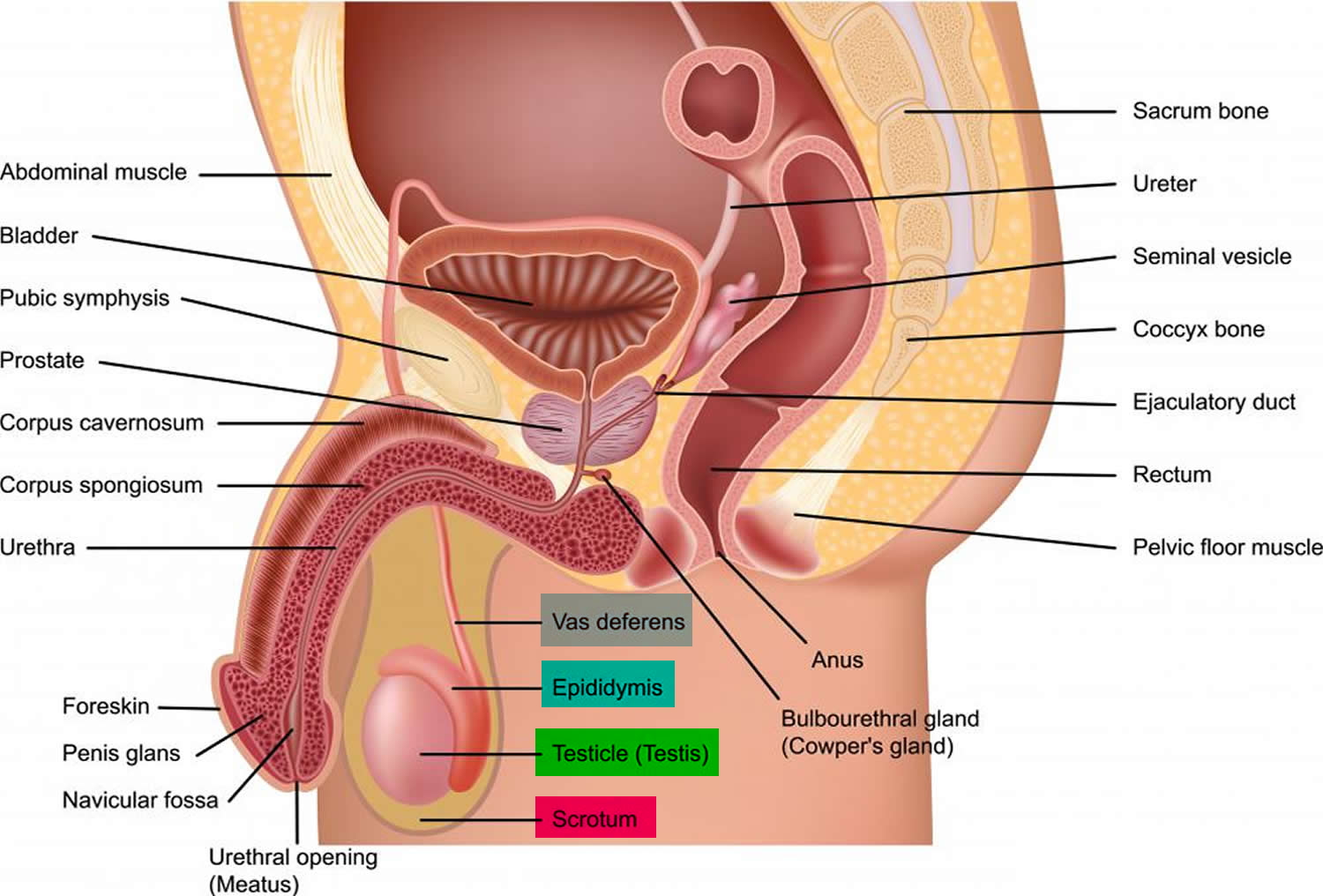
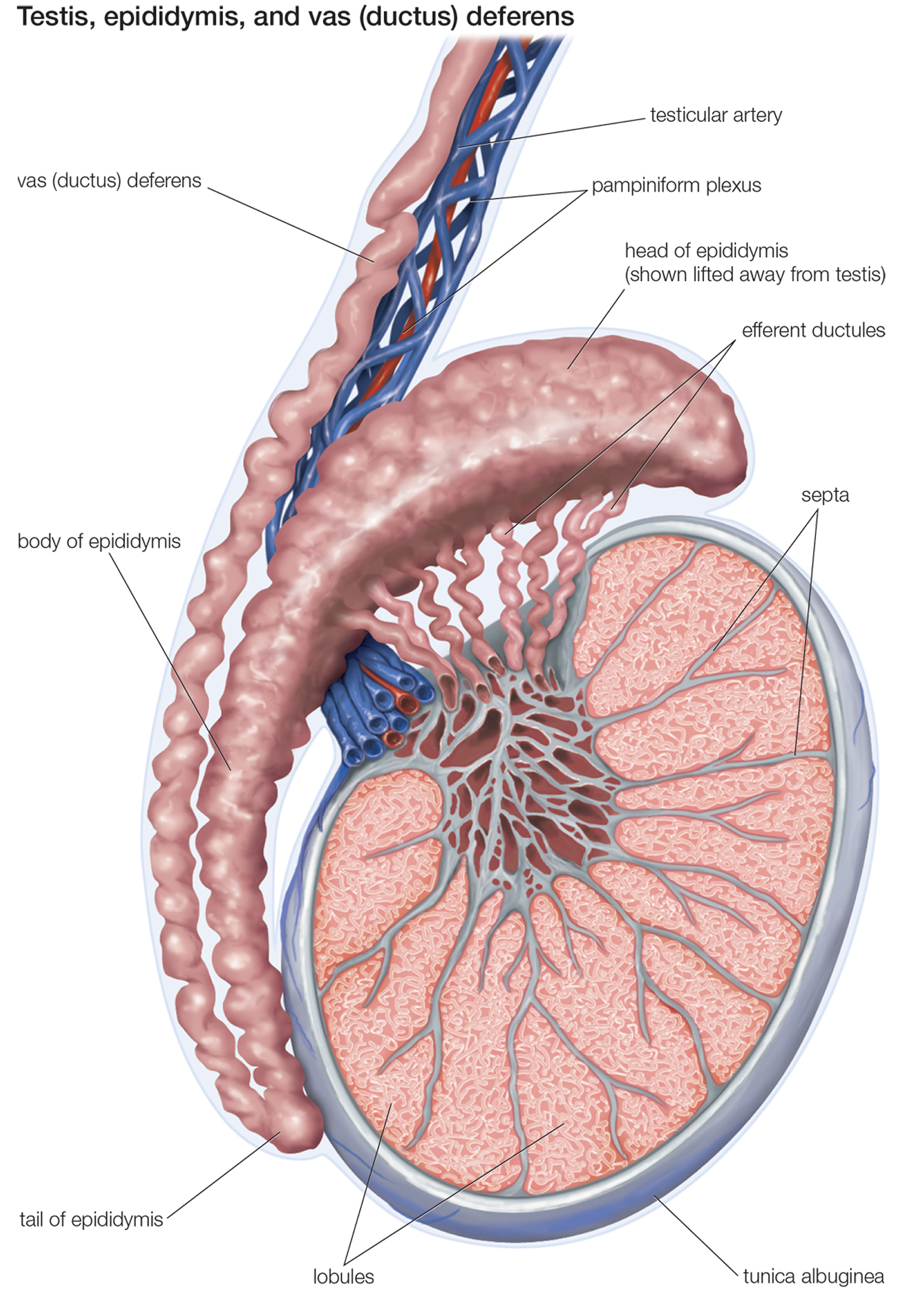
Figure 1. Testis
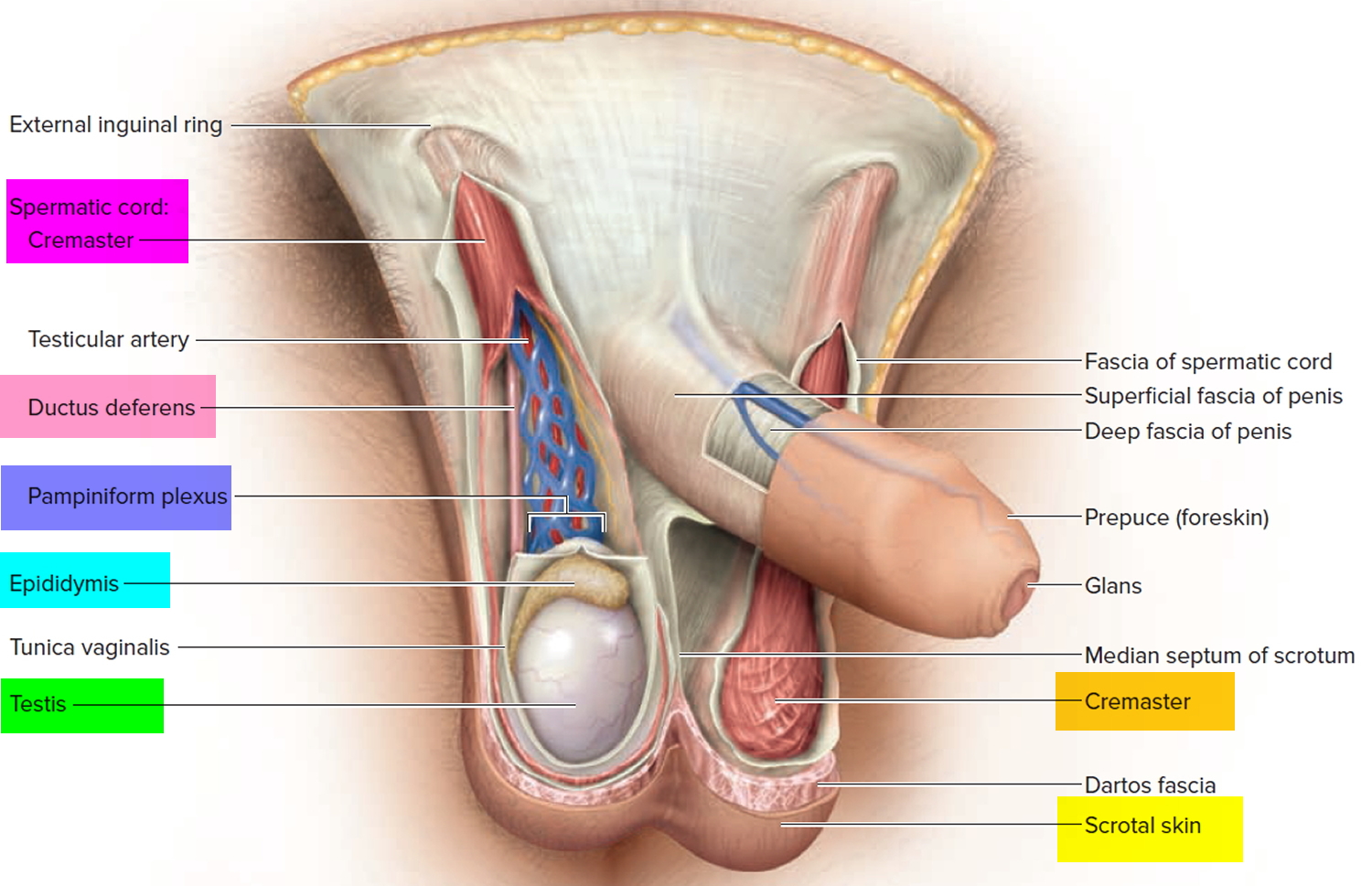
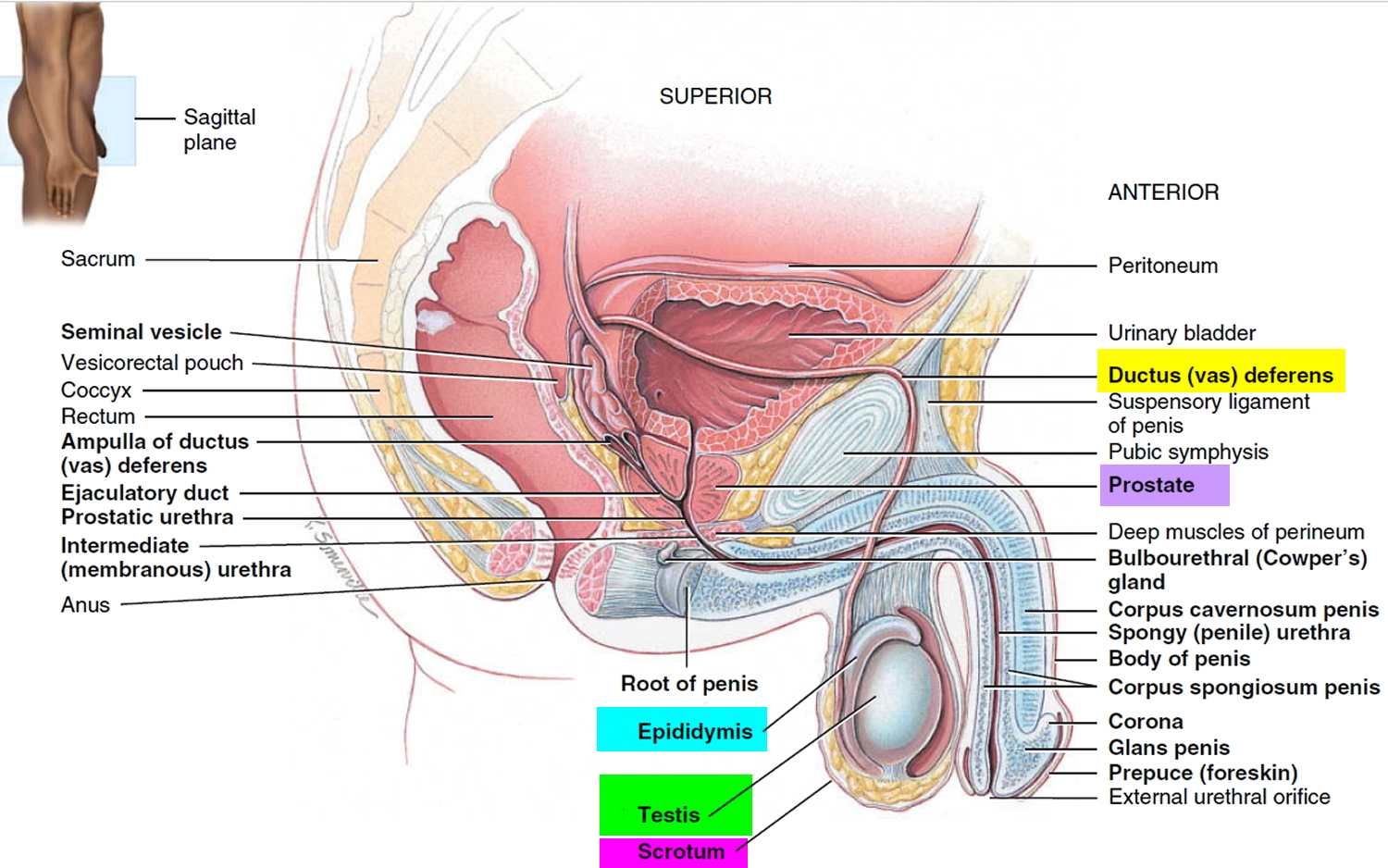
Figure 3. Testis and scrotum (cross section view)
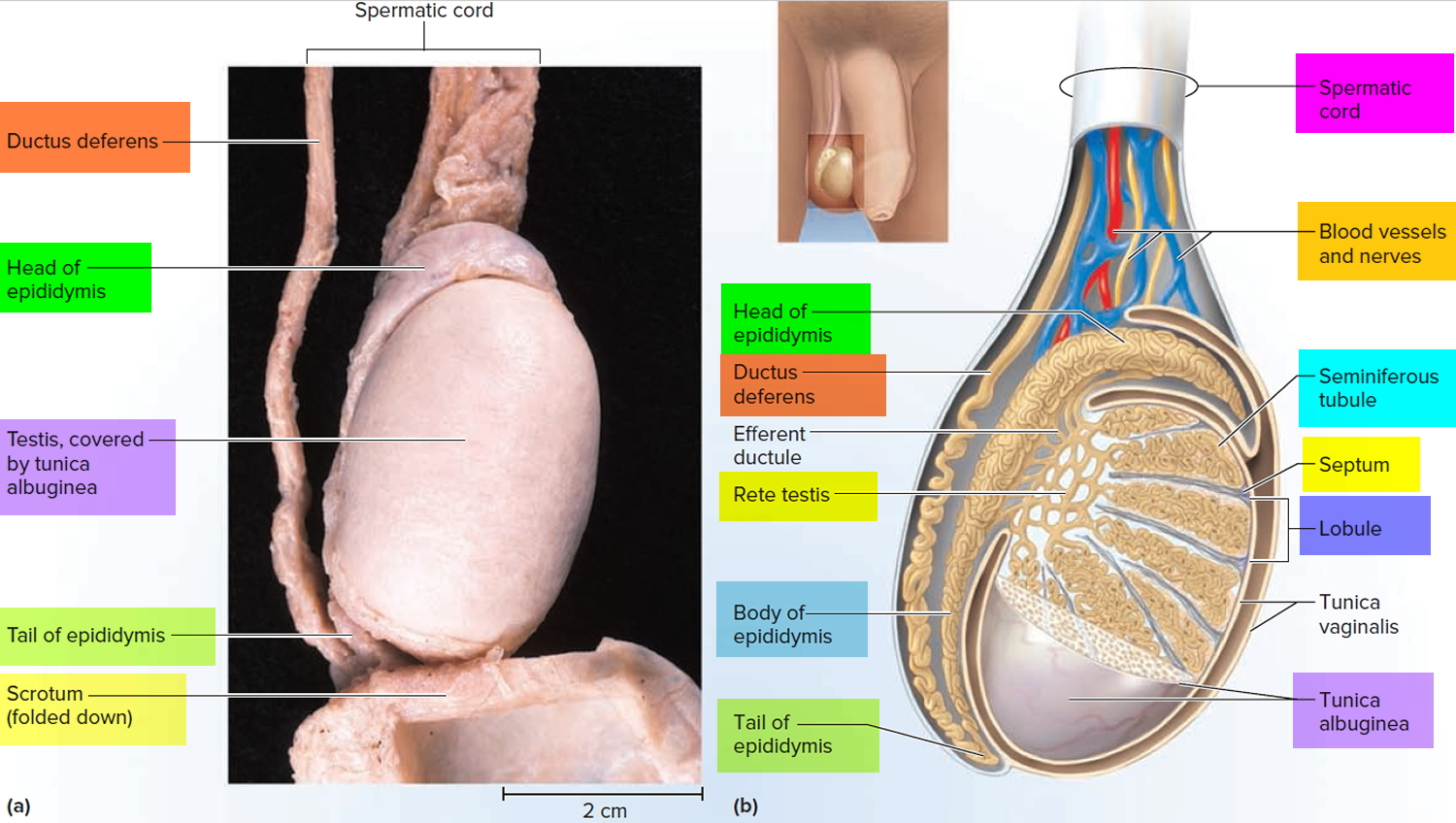
Figure 4. Testis
Mumps orchitis
Mumps is a contagious viral disease that often results in painful swelling of the parotid gland. Mumps virus is an RNA virus of the genus paramyxovirus which is spread from human reservoir by direct contact, airborne droplets, fomites contaminated by saliva and possibly by urine. Mumps typically starts with a few days of fever, headache, muscle aches, tiredness, and loss of appetite, followed by swollen salivary glands. This is what causes the puffy cheeks and a tender, swollen jaw. You can protect yourself and your family against mumps with with MMR (measles, mumps and rubella) vaccine. Centers for Disease Control and Prevention (CDC) recommends children get two doses of MMR vaccine, starting with the first dose at 12 through 15 months of age, and the second dose at 4 through 6 years of age. Teens and adults also should also be up to date on their MMR vaccination.
The incidence of mumps orchitis has declined dramatically since the introduction of the childhood MMR (measles, mumps and rubella) vaccination programme. Mumps is a contagious viral disease that often results in painful swelling of the parotid gland. About 30%-40% of patients with mumps do not develop parotitis.
Mumps orchitis is now rarely seen in children under 10.4 Orchitis is the most common complication of mumps in post-pubertal men, affecting about 20%-30% of cases:5 10%-30% are bilateral.2 Orchitis usually occurs 1-2 weeks after parotitis.
Of affected testicles, 30%-50 % show a degree of testicular atrophy.2 Within the first few days of infection the virus attacks the testicular glands, leading to parenchymal inflammation, separation of seminiferous tubules and perivascular interstitial lymphocyte infiltration. The tunica albugenia forms a barrier against oedema, and the subsequent rise in intratesticular pressure leads to pressure-induced testicular atrophy.5
Adamopoulos et al.6 studied the effects of mumps orchitis on Leydig cell function and found low testosterone levels, elevated leuteinizing hormone levels and an exaggerated pituitary response to leuteinizing hormone-releasing hormone (LHRH) stimulation in the acute phase. Whilst basal testosterone concentrations returned to normal after several months, mean basal follicle stimulating hormone and leuteinizing hormone concentrations remained significantly increased at 10 and 12 months after the acute phase.
The causal link between mumps orchitis and anti-sperm antibodies has been unclear. Although the antibodies were suspected to impair fertility, Kalaydjiev et al.7 demonstrated that both the incidence and the level of serum anti-sperm antibodies among mumps orchitis patients were low, and did not support the hypothesis of an enhanced humoral immunity against spermatozoa.
Mumps orchitis rarely leads to sterility but it may contribute to subfertility. It can also can lead to oligospermia, azoospermia, and asthenospermia (defects in sperm movement). Unilateral disease can significantly, but only transiently, diminish the sperm count, mobility, and morphology. Impairment of fertility is estimated to occur in about 13% of patients,2 while 30%-87% of patients with bilateral mumps orchitis experience infertility.8
Mumps orchitis treatment
Treatment is supportive (bed rest, scrotal support, and the use of nonsteroidal anti-inflammatory agents). Two studies 1, 2 supported prescribing broadspectrum antibiotics. They claimed that bacterial infection of the oedematous testicular tissues cannot always be ruled out.
Steroid administration helps in diminishing pain and edema, but it does not alter the clinical course of the disease or prevent future complications 3. The benefits must be weighed against the self-limiting nature of mumps orchitis and the potential side effects of steroid treatment. Adamopoulos et al. 4 found that corticosteroids may reduce pain and edema, but it caused the testosterone levels to decrease and follicle stimulating hormone (FSH) and leuteinizing hormone (LH) levels to increase. Smith and Bishir 5 described the use of high doses of steroids in reducing pain, but they had little effect on local findings and subsequent clinical course of the disease. Bertschat et al. 6 in his small series found that patients on corticosteroids showed better semen analysis parameters at follow up examinations, although it was not significant.
As interferon inhibits transcriptase-induced viral replication, it would be expected that systemic interferon treatment could prevent the development of testicular atrophy and infertility. Erpenbach et al. 7 claimed in 1991 to have prevented testicular damage and infertility in four patients who had bilateral mumps orchitis by using systemic interferon-α 2B for seven days. Those cases with severe subfertility before treatment improved to normospermia and remained fertile during the 12-20 month follow-up period. Ku et al. 8 studied the effect of interferon-α 2B on 13 patients with mumps orchitis in comparison to eight infected patients who did not receive therapy. No patient in the treatment group developed testicular atrophy as opposed to three in the control group. The sperm count was improved in all patients treated with interferon, but low sperm count persisted in four from the control group. Abnormal sperm morphology persisted in both patient groups. Yeniyol et al. 9 studied the effects of interferon-α 2B on 18 patients with mumps orchitis. Patients were evaluated by testicular biopsy after a year of treatment. The biopsies showed total atrophy of the seminiferous tubules in 39%, 10% atrophy in 16%, and no apparent histological alterations, except an arrest in spermatogenesis, in 45%. They concluded that systemic treatment with interferon is not completely effective in preventing atrophy after mumps orchitis.
When azoospermia occurs as a consequence of mumps orchitis, it does not necessarily cause complete absence of spermatozoa in the testes. Testicular biopsy often yields occasional spermatozoa. The development of intracytoplasmic sperm injection has significantly changed the treatment of male subfertility. Fertilization and pregnancy can be achieved after retrieving a few spermatozoa.
Orchitis complications
Complications of orchitis may include:
- Testicular atrophy. Orchitis may eventually cause the affected testicle to shrink.
- Scrotal abscess. The infected tissue fills with pus.
- Repeated epididymitis. Orchitis can lead to recurrent episodes of epididymitis.
- Infertility. Occasionally, orchitis may cause infertility or inadequate testosterone production (hypogonadism). But infertility and hypogonadism are less likely if orchitis affects only one testicle.
Orchitis causes
Orchitis may be caused by an infection. Many types of bacteria and viruses can cause orchitis. Sometimes a cause of orchitis can’t be determined (idiopathic orchitis).
The most common virus that causes orchitis is mumps. It most often occurs in boys after puberty. Orchitis most often develops 4 to 6 days after the mumps begins.
Orchitis may also occur along with infections of the prostate or epididymis.
Orchitis may be caused by an sexually transmitted infection (STI), such as gonorrhea or chlamydia. The rate of sexually transmitted orchitis or epididymitis is higher in men ages 19 to 35.
Bacterial orchitis
Most often, bacterial orchitis is the result of epididymitis. Epididymitis usually is caused by an infection of the urethra or bladder that spreads to the epididymis.
Often, the cause of the infection is an sexually transmitted infection (STI). Other causes of infection may be related to having been born with abnormalities in your urinary tract or having had a catheter or medical instruments inserted into your penis.
Viral orchitis
Viral orchitis is usually caused by the mumps virus. About one-third of males who contract the mumps after puberty develop orchitis, usually four to seven days after onset of the mumps.
Risk factors for orchitis
Risk factors for sexually transmitted orchitis include:
- High-risk sexual behaviors
- Sex without a condom
- Multiple sexual partners
- Personal history of gonorrhea or another sexually transmitted infection (STI)
- Sexual partner with a diagnosed sexually transmitted infection (STI)
Risk factors for orchitis not due to an STI include:
- Being older than age 45
- Long-term use of a Foley catheter
- Not being vaccinated against the mumps
- Problems of the urinary tract that were present at birth (congenital)
- Repeated urinary tract infections
- Surgery of the urinary tract (genitourinary surgery)
Orchitis prevention
To prevent orchitis:
- Get immunized against mumps, the most common cause of viral orchitis
- Practice safe sex, to help protect against sexually transmitted infections (STIs) that can cause bacterial orchitis.
Orchitis symptoms
Orchitis signs and symptoms usually develop suddenly and may include:
- Pain in the testicle
- Blood in the semen
- Discharge from penis
- Fever
- Groin pain
- Pain with intercourse or ejaculation
- Pain with urination (dysuria)
- Scrotal swelling
- Tender, swollen groin area on affected side
- Tender, swollen, heavy feeling in the testicle, which may last for weeks
- Fever
- Nausea and vomiting
The terms “testicle pain” and “groin pain” are sometimes used interchangeably. But groin pain occurs in the fold of skin between the thigh and abdomen — not in the testicle. The causes of groin pain are different from the causes of testicle pain.
Orchitis diagnosis
Your doctor is likely to start with a physical exam to check for enlarged lymph nodes in your groin and an enlarged testicle on the affected side. Your doctor may also do a rectal examination to check for prostate enlargement or tenderness.
A physical exam may show:
- Enlarged or tender prostate gland
- Tender and enlarged lymph nodes in the groin (inguinal) area on the affected side
- Tender and enlarged testicle on the affected side
- Redness or tenderness of scrotum
After that, your doctor may recommend:
- Complete blood count (CBC)
- Sexually transmitted infection (STI) screening. A narrow swab is inserted into the end of your penis (urethral smear) to obtain a sample of discharge from your urethra. The sample is checked in the laboratory for gonorrhea and chlamydia.
- Testicular ultrasound
- Nuclear scan of the testicles. A radioactive tracer is inserted into your bloodstream. The scanner then maps blood flow to your testicles, which can indicate torsion or orchitis.
- Urinalysis
- Urine culture (clean catch) — may need several samples, including initial stream, midstream, and after prostate massage
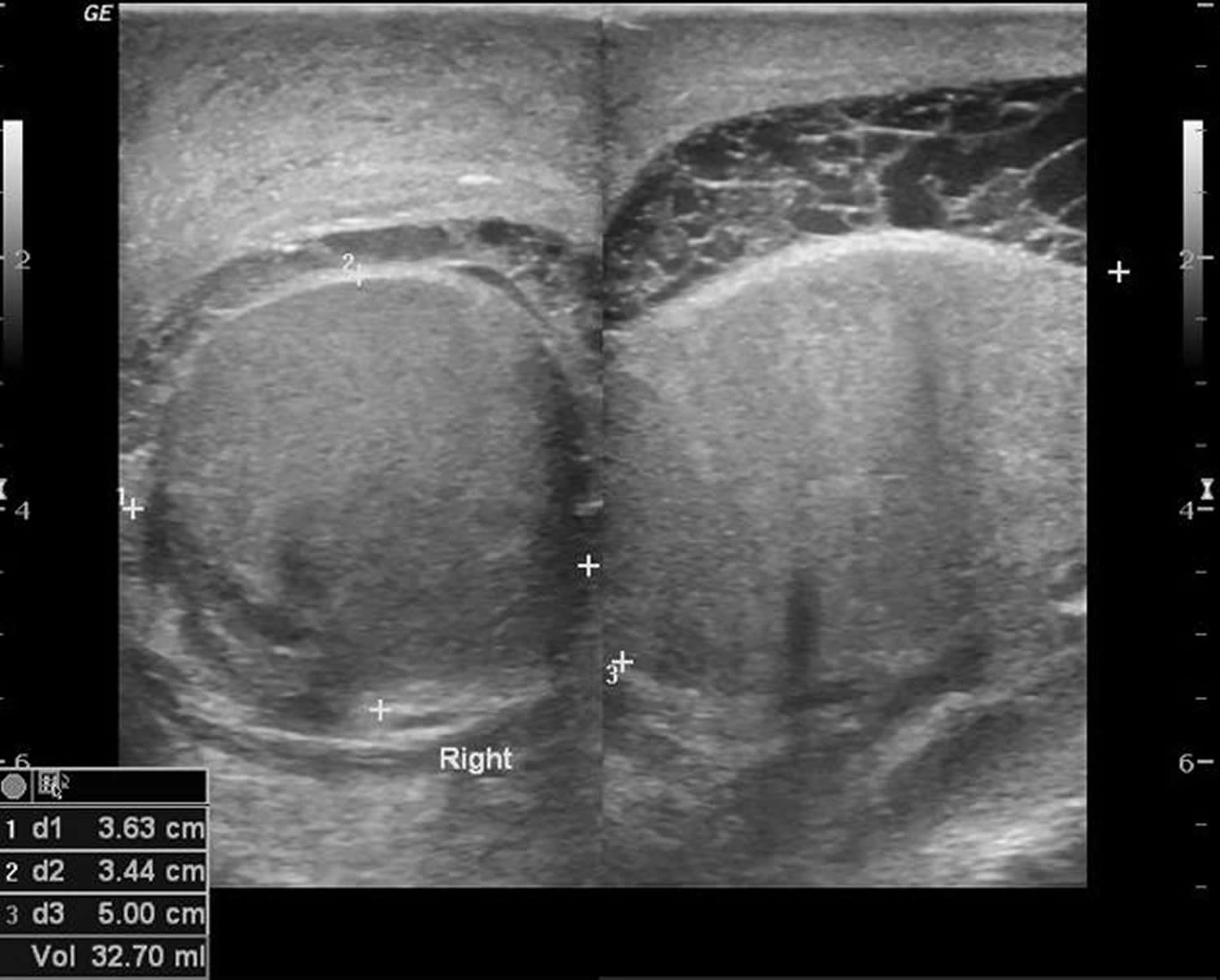
Figure 5. Orchitis ultrasound (enlarged right testis with extra-testicular tiny floating echoes with thin septa favor pyocele [pus])
Orchitis treatment
Treatment depends on the cause of orchitis.
Treating bacterial orchitis
Antibiotics are needed to treat bacterial orchitis and epididymo-orchitis. If the cause of the bacterial infection is an sexually transmitted infection (STI), your sexual partner also needs treatment.
Be sure to take the entire course of antibiotics prescribed by your doctor, even if your symptoms ease sooner, to ensure that the infection is gone.
It may take several weeks for the tenderness to disappear. Resting, supporting the scrotum with an athletic strap, applying ice packs and taking pain medication can help relieve discomfort.
Treatment is aimed at relieving symptoms. Your doctor may recommend:
- Nonsteroidal anti-inflammatory drugs, such as ibuprofen (Advil, Motrin IB, others) or naproxen (Aleve, others)
- Bed rest and elevating your scrotum
- Cold packs
Most people with viral orchitis start to feel better in three to 10 days, although it may take several weeks for the scrotal tenderness to disappear.
Treating idiopathic orchitis
Your doctor may recommend antibiotics and anti-inflammatory medications.
- Casella R, Leibundgut B, Lehmann K, Gasser TC. Mumps orchitis: report of a mini-epidemic. J Urol 1997;158: 2158-61 https://www.ncbi.nlm.nih.gov/pubmed/9366335[↩]
- Scholz M, Graf N, Steffens J, et al. mumps-orchitis im jugend-und erwachsenenalter: ein vergessenes krankheitsbild. Dt Arztbl 1996;93: 2087[↩]
- Masarani M, Wazait H, Dinneen M. Mumps orchitis. Journal of the Royal Society of Medicine. 2006;99(11):573-575. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1633545/[↩]
- Adamopoulos DA, Lawrence DM, Vassilopoulos P, Contoyiannis PA, Swyer GI. Pituitary-testicular interrelationships in mumps orchitis and other infections. BMJ 1978;i: 1177-80 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1604202/pdf/brmedj00124-0019.pdf[↩]
- Smith IM, Bishir JE. Treatment of mumps orchitis with ACTH and cortisone. N Engl J Med 1958;258: 120-4 https://www.ncbi.nlm.nih.gov/pubmed/13493751[↩]
- Steinberger E. The etiology and pathophysiology of testicular dysfunction in man. Fertil Steril 1978;29: 481-91 https://www.ncbi.nlm.nih.gov/pubmed/352731[↩]
- Erpenbach K. Systemic treatment with interferon-alpha 2B: an effective method to prevent sterility after bilateral mumps orchitis. J Urol 1991;146: 54-6 https://www.ncbi.nlm.nih.gov/pubmed/2056606[↩]
- Ku JH, Kim YS, Lee NK. The preventive effect of systemic treatment with interferon-alpha 2B for infertility from mumps orchitis. BJU Int 1999;84: 839-42 https://www.ncbi.nlm.nih.gov/pubmed/10532982[↩]
- Yeniyol CO, Sorguc S, Minareci S, Ayder AR. Role of interferon-alpha 2B in prevention of testicular atrophy with unilateral mumps orchitis. Adult Urol 2000;55: 931-3 https://www.ncbi.nlm.nih.gov/pubmed/10840112[↩]