Contents
What is osteomyelitis
Osteomyelitis is the medical term for an infection in a bone, often caused by bacteria called Staphylococcus aureus. Depending on how the bone becomes infected and the age of the person, other types of bacteria can cause it, too. Osteomyelitis is usually caused by bacteria that enters the body through a wound or spreads from an infection elsewhere in the body.
Although any bone in the body can be affected, the long bones of the arms and legs are most commonly infected in children, while the feet, spine bones, and hips are primarily affected in adults.
Bacteria can infect bones in a number of ways. Bacteria can travel into the bone through the bloodstream from other infected areas in the body. This is called hematogenous (hema refers to the blood) osteomyelitis and is the most common way that people get bone infections.
Another way is by direct infection, when bacteria enter the body’s tissues through a wound and travel to the bone (like after an injury). Open fractures — breaks in the bone with the skin also open — are the injuries that most often develop osteomyelitis.
A bone also can become infected when the blood supply to that area of the bone is disrupted. This can happen in older people with atherosclerosis, which is a narrowing of the blood vessels, or in association with diabetes. Most infections of this kind occur in the toes or feet.
Osteomyelitis is generally categorized as acute or chronic based on histopathologic findings, rather than duration of the infection. Acute osteomyelitis is associated with inflammatory bone changes caused by pathogenic bacteria, and symptoms typically present within two weeks after infection. Necrotic bone is present in chronic osteomyelitis, and symptoms may not occur until six weeks after the onset of infection 1. Further classification of osteomyelitis is based on the presumed mechanism of infection (e.g., hematogenous or direct inoculation of bacteria into bone from contiguous soft tissue infection or a chronic overlying open wound) 2. The more complex Cierny-Mader classification system was developed to help guide surgical management, but is generally not used in primary care 3.
The incidence of chronic osteomyelitis is increasing because of the prevalence of predisposing conditions such as diabetes mellitus and peripheral vascular disease.
Acute hematogenous osteomyelitis results from bacteremic seeding of bone. Children are most often affected because the metaphyseal (growing) regions of the long bones are highly vascular and susceptible to even minor trauma. More than one-half of cases of acute hematogenous osteomyelitis in children occur in patients younger than five years 4. Children typically present within two weeks of disease onset with systemic symptoms, including fever and irritability, as well as local erythema, swelling, and tenderness over the involved bone 5. Chronic osteomyelitis in children is uncommon 6.
Chronic osteomyelitis is generally secondary to open fractures, bacteremia, or contiguous soft issue infection. The incidence of significant infection within three months after an open fracture has been reported to be as high as 27 percent 7. The incidence appears to be independent of the length of time from the injury to surgery 7. Only 1 to 2 percent of prosthetic joints become infected 8.
Hematogenous osteomyelitis is much less common in adults than in children. It typically involves the vertebrae, but can occur in the long bones, pelvis, or clavicle. Patients with vertebral osteomyelitis often have underlying medical conditions (e.g., diabetes mellitus, cancer, chronic renal disease) or a history of intravenous drug use 9. Back pain is the primary presenting symptom.
Chronic osteomyelitis from contiguous soft tissue infection is becoming more common because of the increasing prevalence of diabetic foot infections and peripheral vascular disease. Up to one-half of patients with diabetes develop peripheral neuropathy, which may reduce their awareness of wounds and increase the risk of unrecognized infections 10. Peripheral vascular disease, which is also common in patients with diabetes, reduces the body’s healing response and contributes to chronically open wounds and subsequent soft tissue infection. These conditions may act synergistically to significantly increase the risk of osteomyelitis in these patients 11.
Clinical symptoms of osteomyelitis can be nonspecific and difficult to recognize. They include chronic pain, persistent sinus tract or wound drainage, poor wound healing, malaise, and sometimes fever.
Osteomyelitis needs to be treated early to get rid of the infection and prevent damage to the bone.
Outlook (Prognosis) of osteomyelitis
With treatment, the outcome for acute osteomyelitis is often good.
The outlook is worse for those with long-term (chronic) osteomyelitis. Symptoms may come and go for years, even with surgery. Amputation may be needed, especially in people with diabetes or poor blood circulation.
The outlook for people with an infection of a prosthesis depends partly on:
- The person’s health
- The type of infection
- Whether the infected prosthesis can be safely removed
Complications of Osteomyelitis
Osteomyelitis complications may include:
- Bone death (osteonecrosis). An infection in your bone can impede blood circulation within the bone, leading to bone death. Your bone can heal after surgery to remove small sections of dead bone. If a large section of your bone has died, however, you may need to have that limb surgically removed (amputated) to prevent spread of the infection.
- Septic arthritis. In some cases, infection within bones can spread into a nearby joint.
- Impaired growth. In children, the most common location for osteomyelitis is in the softer areas, called growth plates, at either end of the long bones of the arms and legs. Normal growth may be interrupted in infected bones.
- Skin cancer. If your osteomyelitis has resulted in an open sore that is draining pus, the surrounding skin is at higher risk of developing squamous cell cancer.
Jaw osteomyelitis
An overview of the literature on osteomyelitis of the jaw reveals a wide variety of proposed classifications based on different aspects such as clinical course, radiological features, pathogenesis, and etiology. However, most agree that osteomyelitis can be classified as acute or chronic. Acute osteomyelitis differs from chronic osteomyelitis, which has duration of four weeks after the onset of clinical symptoms 12, 13. Many authors advocate that chronic osteomyelitis involving the jawbone can be further divided into two major categories: suppurative and nonsuppurative forms 14. The main cause of chronic suppurative osteomyelitis of the jaws is odontogenic infections, which might occur as a complication of dental extractions, maxillofacial trauma, or irradiation of the facial skeleton 15. The clinical findings of this disease were characterized by the presence of pus, fistula, and sequestration 16. Odontogenic microorganisms such as Staphylococcus aureus, Staphylococcus epidermidis, and Actinomyces usually contribute to the pathogenesis of osteomyelitis of the jaws 17.
The term nonsuppurative osteomyelitis is characterized by the absence of the formation of pus, fistula, and sequestration 17. These forms of osteomyelitis of the jaw include osteoradionecrosis, bisphosphonate-related osteonecrosis of the jaws, chronic recurrent multifocal osteomyelitis of children, and chronic sclerosing osteomyelitis 17. Diffuse Sclerosing Osteomyelitis (DSO) is a radiographic term that has been used to describe the radiographic pattern associated with both primary chronic osteomyelitis and chronic suppurative osteomyelitis 18. Chronic recurrent multifocal osteomyelitis (CRMO) is a nonautoimmune disorder that mostly affects children and is characterized by periods of exacerbations and remissions over many years 19. It causes periodic bone pain, fever, and the appearance of multiple bone lesions that can occur in any skeletal site. SAPHO [synovitis, acne, pustulosis, hyperostosis, osteitis] syndrome is the adult version of chronic recurrent multifocal osteomyelitis, which is associated with synovitis, acne, pustulosis, hyperostosis, and osteitis 20.
Primary Chronic Osteomyelitis is a nonodontogenic and nonsuppurative chronic inflammatory condition of unknown origin 16. Several authors have pointed out a possible association between primary chronic osteomyelitis of the jaw and other syndromes, such as CRMO [chronic recurrent multifocal osteomyelitis], DSO [Diffuse Sclerosing Osteomyelitis], and SAPHO [synovitis, acne, pustulosis, hyperostosis, osteitis] 21, 22.
Primary chronic osteomyelitis is a nonbacterial chronic inflammatory disease of unknown cause, which can also be associated with other conditions, including autoimmune diseases and syndromes such as “SAPHO (Synovitis, Acne, Pustulosis, Hyperostosis, and Osteitis) syndrome,” Majeed syndrome, and cherubism (a disorder characterized by abnormal bone tissue in the lower part of the face) 23. Hematogenous spread of inflammation has also been mentioned in the literature, especially in osteomyelitis of the long bones of infants and children 24.
Primary chronic osteomyelitis of the jaw has been reported in children of both sexes with a peak onset between 10 and 20 years 25. In the maxillofacial region, the most affected site is the mandible 26.
The bacterial contamination of bone tissue is best determined by bone biopsy under radiographic guide. The most frequently bacteria associated with osteomyelitis are Staphylococcus aureus, Gram negatives (Pseudomonas aeruginosa), and anaerobe bacteria (Bacteroides fragilis) 27.
Secondary chronic osteomyelitis of the jaw is usually caused by bacterial infection of dental origin (pulpal disease, posttooth extraction, or foreign bodies) and is much more common than primary chronic osteomyelitis 28.
Treatment of primary chronic osteomyelitis of the jaw includes anti-inflammatory drug therapy such as corticosteroids in combination with surgical removal of inflammatory or necrotic bone tissue 29. Nonsteroidal anti-inflammatory drugs (NSAID) are beneficial and considerably improve the patient’s quality of life 30. Antibiotics have been given most of preventive reasons and hyperbaric oxygen therapy has been used with varying results 31.
Osteomyelitis causes
In most cases, osteomyelitis is caused by a type of bacteria found on the skin, the staphylococcus bacteria. But it can also be caused by fungi or other germs. The bone may become infected after an injury, such as a bone fracture, or surgery.
The most common pathogens in osteomyelitis depend on the patient’s age. Staphylococcus aureus is the most common cause of acute and chronic hematogenous osteomyelitis in adults and children. Group A streptococcus, Streptococcus pneumoniae, and Kingella kingae are the next most common pathogens in children. Group B streptococcal infection occurs primarily in newborns 32. In adults, S. aureus is the most common pathogen in bone and prosthetic joint infections. Increasingly, methicillin-resistant S. aureus (MRSA) is isolated from patients with osteomyelitis. In some studies, MRSA accounted for more than one-third of staphylococcal isolates 33. In more chronic cases that may be caused by contiguous infection, Staphylococcus epidermidis, Pseudomonas aeruginosa, Serratia marcescens, and Escherichia coli may be isolated. Fungal and mycobacterial infections have been reported in patients with osteomyelitis, but these are uncommon and are generally found in patients with impaired immune function 34.
Germs can enter a bone in a variety of ways, including:
- Bacteria or other germs may spread to a bone from infected skin, muscles, or tendons next to the bone. This may occur under a skin sore.
- The infection can start in another part of the body and spread to the bone through the blood. Infection such as a urinary tract infection or pneumonia, spreads through the blood to the bone.
- The infection can also start after bone surgery. This is more likely if the surgery is done after an injury or if metal rods or plates are placed in the bone.
- Infected tissue or an infected prosthetic joint. Severe puncture wounds can carry germs deep inside your body. If such an injury becomes infected, the germs can spread into a nearby bone.
- Open wounds. Germs can enter the body if you have broken a bone so severely that part of it is sticking out through your skin. Direct contamination can also occur during surgeries to replace joints or repair fractures.
Risk factors for osteomyelitis
Your bones are normally resistant to infection. For osteomyelitis to occur, a situation that makes your bones vulnerable must be present.
Risk factors for osteomyelitis include:
- diabetes,
- hemodialysis,
- poor blood supply,
- recent injury,
- intravenous drug abuse,
- surgery involving the bones, and
- a weakened immune system.
Recent injury or orthopedic surgery
A severe bone fracture or a deep puncture wound gives infections a route to enter your bone or nearby tissue. Surgery to repair broken bones or replace worn joints also can accidentally open a path for germs to enter a bone.
Implanted orthopedic hardware is a risk factor for infection. Deep animal bites also can provide a pathway for infection.
Circulation disorders
When blood vessels are damaged or blocked, your body has trouble distributing the infection-fighting cells needed to keep a small infection from growing larger. What begins as a small cut can progress to a deep ulcer that may expose deep tissue and bone to infection.
Diseases that impair blood circulation include:
- Poorly controlled diabetes
- Peripheral arterial disease, often related to smoking
- Sickle cell disease
Problems requiring intravenous lines or catheters
There are a number of conditions that require the use of medical tubing to connect the outside world with your internal organs. However, this tubing can also serve as a way for germs to get into your body, increasing your risk of an infection in general, which can lead to osteomyelitis.
Examples of when this type of tubing might be used include:
- Dialysis machine tubing
- Urinary catheters
- Long-term intravenous tubing, sometimes called central lines
Conditions that impair the immune system
If your immune system is affected by a medical condition or medication, you have a greater risk of osteomyelitis. Factors that may suppress your immune system include:
- Chemotherapy
- Poorly controlled diabetes
- Needing to take corticosteroids or drugs called tumor necrosis factor (TNF) inhibitors
Illicit drugs
People who inject illicit drugs are more likely to develop osteomyelitis because they typically use nonsterile needles and don’t sterilize their skin before injections.
Osteomyelitis Prevention
If you’ve been told that you have an increased risk of infection, talk to your doctor about ways to prevent infections from occurring. Reducing your risk of infection will also reduce your risk of developing osteomyelitis.
In general, take precautions to avoid cuts and scrapes, which give germs easy access to your body. If you do get any cuts and scrapes, clean the area immediately and apply a clean bandage. Check wounds frequently for signs of infection.
Osteomyelitis symptoms
If you have osteomyelitis, you may experience:
- high fever
- ill feeling (malaise)
- pain in the affected bones
- swelling, redness and warmth at the site of the bone infection
- general discomfort
- an open wound that may show pus
People with bone infection (osteomyelitis) often feel severe pain in the infected bone. They might have fever and chills, feel tired or nauseated, or have a general feeling of not being well. The skin above the infected bone may be sore, red, and swollen. It’s sometimes difficult to diagnose osteomyelitis in infants and young children because they don’t always show pain or specific symptoms in the area of the infection. Also, older people with diabetes or another problem with their blood vessels don’t always show signs of fever or pain. For teenagers, it’s frequently a preceding accident or injury that leads to the infection.
If the osteomyelitis developed after an injury, the injured area may begin to hurt again after initially seeming to get better.
Osteomyelitis diagnosis
Your doctor may diagnose osteomyelitis by examining you, doing a blood test and a bone scan or other scans. Although a blood test cannot tell whether you have osteomyelitis, it may be used to detect signs of infection, including the bacteria causing the infection. Sometimes your doctor will take a bone biopsy, where a small piece of bone is removed for testing.
Blood tests
Blood tests may reveal elevated levels of white blood cells and other factors that may indicate that your body is fighting an infection. If your osteomyelitis was caused by an infection in the blood, tests may reveal what germs are to blame.
No blood test exists that tells your doctor whether you do or do not have osteomyelitis. However, blood tests do give clues that your doctor uses to decide what further tests and procedures you may need.
Imaging tests
- X-rays. X-rays can reveal damage to your bone. However, damage may not be visible until osteomyelitis has been present for several weeks. More-detailed imaging tests may be necessary if your osteomyelitis has developed more recently.
- Computerized tomography (CT) scan. A CT scan combines X-ray images taken from many different angles, creating detailed cross-sectional views of a person’s internal structures.
- Magnetic resonance imaging (MRI). Using radio waves and a strong magnetic field, MRI scans can produce exceptionally detailed images of bones and the soft tissues that surround them.
Imaging is useful to characterize the infection and to rule out other potential causes of symptoms. Plain radiography, technetium-99 bone scintigraphy, and magnetic resonance imaging (MRI) are the most useful modalities (Table 1) 35. Plain radiography usually does not show abnormalities caused by osteomyelitis until about two weeks after the initial infection, when nearly 50 percent of the bone mineral content has been lost 35. Typical findings include non-specific periosteal reaction and osteolysis (Figure 1). Plain radiography is a useful first step that may reveal other diagnoses, such as metastases or osteoporotic fractures. It generally complements information provided by other modalities and should not be omitted, even if more advanced imaging is planned.
The role of computed tomography in the diagnosis of osteomyelitis is limited. Although computed tomography is superior to MRI in detecting necrotic fragments of bone, its overall value is generally less than that of other imaging modalities. Computed tomography should be used only to determine the extent of bony destruction (especially in the spine), to guide biopsies, or in patients with contraindications to MRI 36.
MRI provides better information for early detection of osteomyelitis than do other imaging modalities (Figure 1). MRI can detect osteomyelitis within three to five days of disease onset 36. Most studies of the diagnostic accuracy of MRI in detecting osteomyelitis included patients with diabetic foot ulcers 37. The sensitivity and specificity of MRI in the diagnosis of osteomyelitis may be as high as 90 percent 38. Because MRI can also detect necrotic bone, sinus tracts, or abscesses, it is superior to bone scintigraphy in diagnosing and characterizing osteomyelitis 38. Its use can be limited, however, if surgical hardware is present.
Nuclear imaging can be helpful in diagnosing osteomyelitis (Figure 2). Three-phase technetium-99 bone scintigraphy and leukocyte scintigraphy are usually positive within a few days of the onset of symptoms 36. The sensitivity of bone scintigraphy is comparable to MRI, but the specificity is poor. Leukocyte scintigraphy also has poor specificity, but when combined with three-phase bone scintigraphy, sensitivity and specificity are improved 39. Bone and leukocyte scintigraphy can provide valuable information if MRI is contraindicated or unavailable 40.
Other imaging modalities seem promising for the diagnosis of osteomyelitis, but they are not routinely used. Positron emission tomography has the highest sensitivity and specificity—more than 90 percent—but it is expensive and not as widely available as other modalities 39. The role of musculoskeletal ultrasonography in the diagnosis of osteomyelitis is evolving. Some studies suggest that in some patients, such as those with sickle cell disease, detection of subperiosteal fluid collections can be useful or even diagnostic; however, reliable estimates of sensitivity and specificity are lacking 36.
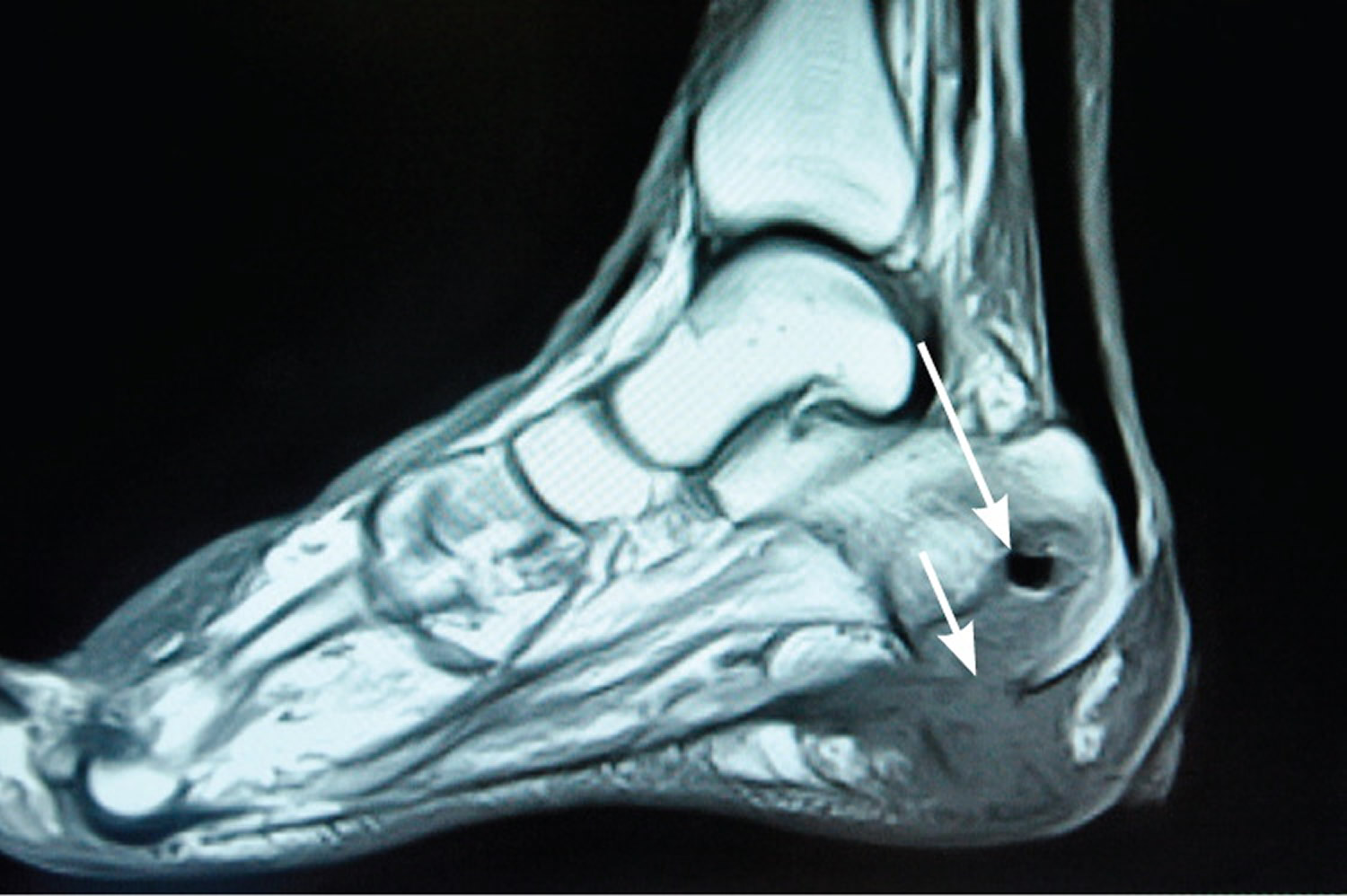
Figure 1. Magnetic resonance image (MRI) of foot osteomyelitis
Note: Magnetic resonance image demonstrating abnormal T1-weighted signal within the calcaneus (long arrow), consistent with osteomyelitis. Inferior cortical disruption and contiguous soft tissue fluid and edema are also present (short arrow).
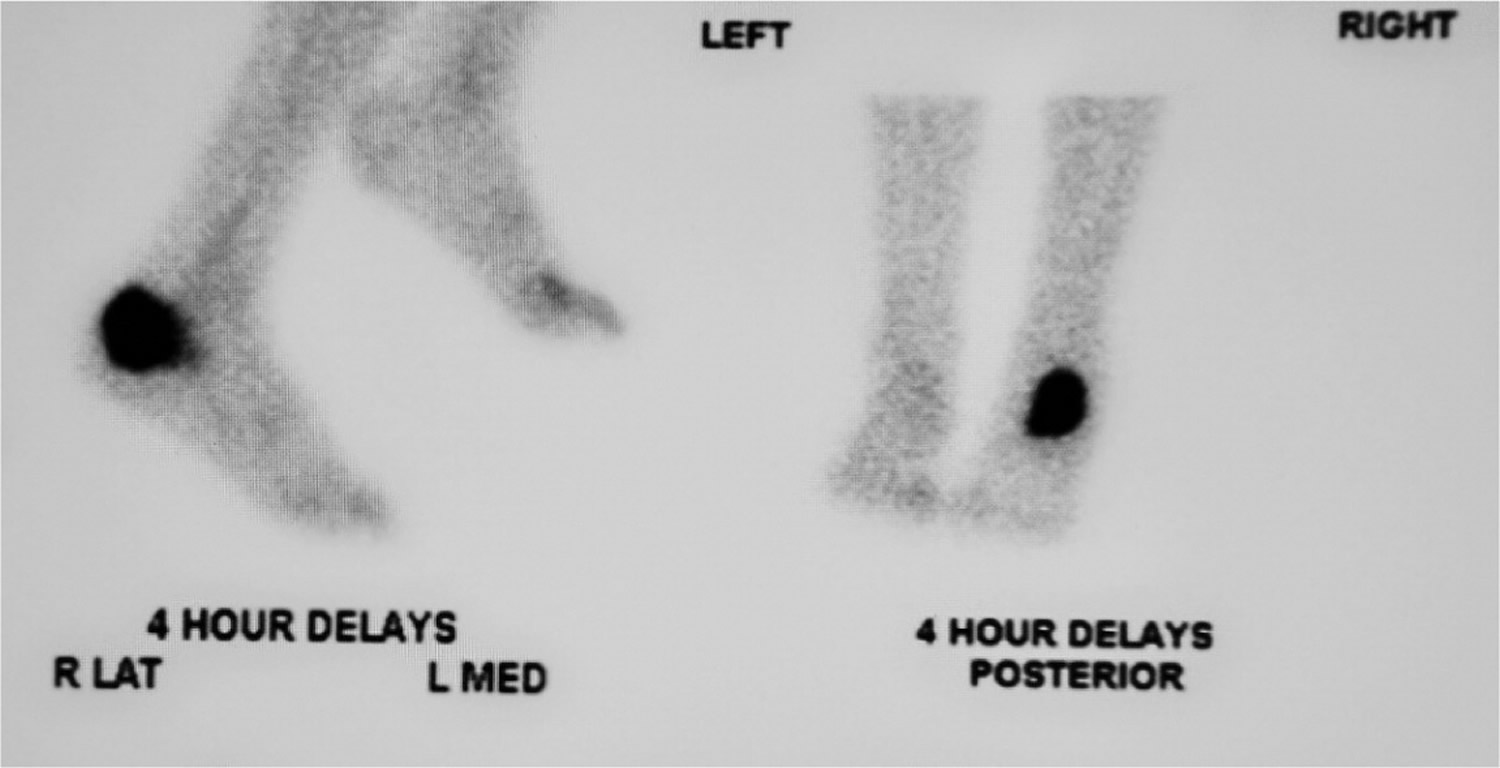
Figure 2. Bone scintigraphy images demonstrating localized increased radioactive tracer uptake within the left calcaneus, consistent with osteomyelitis.
Table 1. Diagnostic Imaging Studies for Osteomyelitis
| Imaging modality | Sensitivity (%) | Specificity (%) | Comments |
|---|---|---|---|
Computed tomography | 67 | 50 | Generally should not be used in osteomyelitis evaluation |
Leukocyte scintigraphy | 61 to 84 | 60 to 68 | Combining with technetium-99 bone scintigraphy can increase specificity |
Magnetic resonance imaging | 78 to 90 | 60 to 90 | Useful to distinguish between soft tissue and bone infection, and to determine extent of infection; less useful in locations of surgical hardware because of image distortion |
Plain radiography(anteroposterior, lateral, and oblique views) | 14 to 54 | 68 to 70 | Preferred imaging modality; useful to rule out other pathology |
Positron emission tomography | 96 | 91 | Expensive; limited availability |
Technetium-99 bone scintigraphy | 82 | 25 | Low specificity, especially if patient has had recent trauma or surgery; useful to differentiate osteomyelitis from cellulitis, and in patients in whom magnetic resonance imaging is contraindicated |
Bone biopsy
A bone biopsy is the gold standard for diagnosing osteomyelitis, because it can also reveal what particular type of germ has infected your bone. Knowing the type of germ allows your doctor to choose an antibiotic that works particularly well for that type of infection.
An open biopsy requires anesthesia and surgery to access the bone. In some situations, a surgeon inserts a long needle through your skin and into your bone to take a biopsy. This procedure requires local anesthetics to numb the area where the needle is inserted. X-ray or other imaging scans may be used for guidance.
Osteomyelitis treatment
Treatment of osteomyelitis depends on appropriate antibiotic therapy and often requires surgical removal of infected and necrotic tissue. Choice of antibiotic therapy should be determined by culture and susceptibility results, if possible (Table 1) 42. In the absence of such information, broad-spectrum, empiric antibiotics should be administered. False-negative blood or biopsy cultures are common in patients who have begun antibiotic therapy. If clinically possible, delaying antibiotics is recommended until microbial culture and sensitivity results are available. Indications for surgery include antibiotic failure, infected surgical hardware, and chronic osteomyelitis with necrotic bone and soft tissue 43.
Acute osteomyelitis is usually treated with antibiotics for at least four to six weeks. The antibiotics are first given intravenously (through a vein), then as tablets once symptoms improve.
Acute hematogenous osteomyelitis in children typically requires a much shorter course of antibiotic therapy than does chronic osteomyelitis in adults. Although randomized controlled trials are lacking, therapy with four days of parenteral antibiotics followed by oral antibiotics for a total of four weeks seems to prevent recurrence in children who have no serious underlying pathology 44. In immunocompromised children, the transition to oral antibiotics should be delayed, and treatment should continue for at least six weeks based on clinical response 4. Recurrence rates are typically higher in this population. Surgical treatment in immunocompetent children is rare.
Despite the use of surgical debridement and long-term antibiotic therapy, the recurrence rate of chronic osteomyelitis in adults is about 30 percent at 12 months 45. Recurrence rates in cases involving P. aeruginosa are even higher, nearing 50 percent. The optimal duration of antibiotic treatment and route of delivery are unclear 46. For chronic osteomyelitis, parenteral antibiotic therapy for two to six weeks is generally recommended, with a transition to oral antibiotics for a total treatment period of four to eight weeks 47. Long-term parenteral therapy is likely as effective as transitioning to oral medications, but has similar recurrence rates with increased adverse effects 46. In some cases, surgery is necessary to preserve viable tissue and prevent recurrent systemic infection.
Antibiotic regimens for the empiric treatment of acute osteomyelitis, particularly in children, should include an agent directed against Staphylococcus aureus. Betalactam antibiotics are first-line options unless MRSA (methicillin-resistant Staphylococcus aureus) is suspected. If methicillin resistance among community isolates of Staphylococcus is greater than 10 percent, MRSA (methicillin-resistant Staphylococcus aureus) should be considered in initial antibiotic coverage 44. Intravenous vancomycin is the first-line choice. In patients with diabetic foot infections or penicillin allergies, fluoroquinolones are an alternate option for staphylococcal infections; these agents seem to be as effective as beta-lactams 42. Fluoroquinolones also cover quinolone-sensitive enterobacteria and other gram-negative rods.
Table 2. Initial Antibiotic Therapy for Treatment of Osteomyelitis in Adults
| Organism | Preferred regimens | Alternative regimens |
|---|---|---|
Anaerobes |
|
|
Enterobacteriaceae (e.g., Escherichia coli), quinolone-resistant |
|
|
Enterobacteriaceae, quinolone-sensitive |
|
|
Pseudomonas aeruginosa |
|
|
Staphylococcus aureus, methicillin-resistant |
|
|
S. aureus, methicillin sensitive |
|
|
Streptococcus species |
|
|
IV = intravenously.
In more severe cases, and for chronic osteomyelitis, you may also need surgery to remove damaged bone or tissue.
Surgery
Depending on the severity of the infection, osteomyelitis surgery may include one or more of the following procedures:
- Drain the infected area. Opening up the area around your infected bone allows your surgeon to drain any pus or fluid that has accumulated in response to the infection.
- Remove diseased bone and tissue. In a procedure called debridement, the surgeon removes as much of the diseased bone as possible, and takes a small margin of healthy bone to ensure that all the infected areas have been removed. Surrounding tissue that shows signs of infection also may be removed.
- Restore blood flow to the bone. Your surgeon may fill any empty space left by the debridement procedure with a piece of bone or other tissue, such as skin or muscle, from another part of your body. Sometimes temporary fillers are placed in the pocket until you’re healthy enough to undergo a bone graft or tissue graft. The graft helps your body repair damaged blood vessels and form new bone.
- Remove any foreign objects. In some cases, foreign objects, such as surgical plates or screws placed during a previous surgery, may have to be removed.
- Amputate the limb. As a last resort, surgeons may amputate the affected limb to stop the infection from spreading further.
Osteomyelitis can be successfully treated. However, it is important to prevent it from happening again. Your doctor will advise you on the steps you should take.
- Mylona E, Samarkos M, Kakalou E, Fanourgiakis P, Skoutelis A. Pyogenic vertebral osteomyelitis: a systematic review of clinical characteristics. Semin Arthritis Rheum. 2009;39(1):10–17.[↩]
- Waldvogel FA, Medoff G, Swartz MN. Osteomyelitis: a review of clinical features, therapeutic considerations and unusual aspects. N Engl J Med. 1970;282(4):198–206.[↩]
- Cierny G III, Mader JT, Penninck JJ. A clinical staging system for adult osteomyelitis. Clin Orthop Relat Res. 2003;(414):7–24.[↩]
- Gutierrez K. Bone and joint infections in children. Pediatr Clin North Am. 2005;52(3):779–794.[↩][↩]
- Saavedra-Lozano J, Mejías A, Ahmad N, et al. Changing trends in acute osteomyelitis in children: impact of methicillin-resistant Staphylococcus aureus infections. J Pediatr Orthop. 2008;28(5):569–575.[↩]
- Auh JS, Binns HJ, Katz BZ. Retrospective assessment of subacute or chronic osteomyelitis in children and young adults. Clin Pediatr (Phila). 2004;43(6):549–555.[↩]
- Pollak AN, Jones AL, Castillo RC, Bosse MJ, MacKenzie EJ; LEAP Study Group. The relationship between time to surgical debridement and incidence of infection after open high-energy lower extremity trauma J Bone Joint Surg Am. 2010;92(1):7–15.[↩][↩]
- Kurtz SM, Lau E, Schmier J, Ong KL, Zhao K, Parvizi J. Infection burden for hip and knee arthroplasty in the United States. J Arthroplasty. 2008;23(7):984–991.[↩]
- Zimmerli W. Clinical practice. Vertebral osteomyelitis. N Engl J Med. 2010;362(11):1022–1029.[↩]
- Barrett AM, Lucero MA, Le T, Robinson RL, Dworkin RH, Chappell AS. Epidemiology, public health burden, and treatment of diabetic peripheral neuropathic pain: a review. Pain Med. 2007;8(suppl 2):S50–S62.[↩]
- Abdulrazak A, Bitar ZI, Al-Shamali AA, Mobasher LA. Bacteriological study of diabetic foot infections. J Diabetes Complications. 2005;19(3):138–141.[↩]
- Mercuri L. G. Acute osteomyelitis of the jaws. Oral & Maxillofacial Surgery Clinics of North America. 1991;3(2):355–365.[↩]
- Marx R. E. Chronic osteomyelitis of the jaws. Oral & Maxillofacial Surgery Clinics of North America. 1991;3(2):367–381.[↩]
- Bernier S., Clermont S., Maranda G., Turcotte J. Y. Osteomyelitis of the jaws. Journal of the Canadian Dental Association. 1995;61(5):441-2–445-8. https://www.ncbi.nlm.nih.gov/pubmed/7773870[↩]
- Koorbusch G. F., Fotos P., Goll K. T. Retrospective assessment of osteomyelitis: etiology, demographics, risk factors, and management in 35 cases. Oral Surgery, Oral Medicine, Oral Pathology. 1992;74(2):149–154. doi: 10.1016/0030-4220(92)90373-x. https://www.ncbi.nlm.nih.gov/pubmed/1508521[↩]
- Eyrich G. K. H., Harder C., Sailer H. F., Langenegger T., Bruder E., Michel B. A. Primary chronic osteomyelitis associated with synovitis, acne, pustulosis, hyperostosis and osteitis (SAPHO syndrome) Journal of Oral Pathology and Medicine. 1999;28(10):456–464. https://www.ncbi.nlm.nih.gov/pubmed/10551743[↩][↩]
- Frid P., Tornes K., Nielsen Ø., Skaug N. Primary chronic osteomyelitis of the jaw-a microbial investigation using cultivation and DNA analysis: a pilot study. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology and Endodontology. 2009;107(5):641–647. doi: 10.1016/j.tripleo.2009.01.020. https://www.ncbi.nlm.nih.gov/pubmed/19426919[↩][↩][↩]
- Bevin C. R., Inwards C. Y., Keller E. E. Surgical management of primary chronic osteomyelitis: a long-term retrospective analysis. Journal of Oral and Maxillofacial Surgery. 2008;66(10):2073–2085. doi: 10.1016/j.joms.2008.06.029. https://www.ncbi.nlm.nih.gov/pubmed/18848105[↩]
- Monsour P. A. J., Dalton J. B. Chronic recurrent multifocal osteomyelitis involving the mandible: case reports and review of the literature. Dentomaxillofacial Radiology. 2010;39(3):184–190. doi: 10.1259/dmfr/23060413 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3520218/[↩]
- Magrey M., Khan M. A. New insights into synovitis, acne, pustulosis, hyperostosis, and osteitis (SAPHO) syndrome. Current Rheumatology Reports. 2009;11(5):329–333. doi: 10.1007/s11926-009-0047-8. https://www.ncbi.nlm.nih.gov/pubmed/19772827[↩]
- Kahn M. F., Hayem F., Hayem G., Grossin M. Is diffuse sclerosing osteomyelitis of the mandible part of the synovitis, acne, pustulosis, hyperostosis, osteitis (SAPHO) syndrome?. Analysis of seven cases. Oral Surgery, Oral Medicine, Oral Pathology. 1994;78(5):594–598. doi: 10.1016/0030-4220(94)90170-8. https://www.ncbi.nlm.nih.gov/pubmed/7838465[↩]
- Flygare L., Norderyd J., Kubista J., Ohlsson J., Vallö-Christiansen J., Magnusson B. Chronic recurrent multifocal osteomyelitis involving both jaws: report of a case including magnetic resonance correlation. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontics. 1997;83(2):300–305. doi: 10.1016/s1079-2104(97)90020-2. https://www.ncbi.nlm.nih.gov/pubmed/9117765[↩]
- Stern S., Ferguson P. Auto inflammatory bone diseases. Rheumatic Disease Clinics of North America. 2013;39(4):735–749. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3823499/[↩]
- Mader J. T., Shirtliff M. E., Bergquist S., Calhoun J. H. Bone and joint infections in the elderly: practical treatment guidelines. Drugs and Aging. 2000;16(1):67–80. doi: 10.2165/00002512-200016010-00006. https://www.ncbi.nlm.nih.gov/pubmed/10733265[↩]
- Gentry L. O. Osteomyelitis: options for diagnosis and management. Journal of Antimicrobial Chemotherapy. 1988;21:115–131. https://www.ncbi.nlm.nih.gov/pubmed/3290180[↩]
- Idahosa C. H., Boggess W., Levin L. M., Alawi F. Primary chronic osteomyelitis of the mandible in a child. Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology. 2014;117(5, article e342) doi: 10.1016/j.oooo.2014.01.055.[↩]
- van Merkesteyn J. P. R., Groot R. H., Bras J., McCarroll R. S., Bakker D. J. Diffuse sclerosing osteomyelitis of the mandible: a new concept of its etiology. Oral Surgery, Oral Medicine, Oral Pathology. 1990;70(4):414–419. doi: 10.1016/0030-4220(90)90200-c. https://www.ncbi.nlm.nih.gov/pubmed/2216378[↩]
- Eyrich G. K. H., Baltensperger M. M., Bruder E., Grätz K. W. Primary chronic osteomyelitis in childhood and adolescence: a retrospective analysis of 11 cases and review of the literature. Journal of Oral and Maxillofacial Surgery. 2003;61(5):561–573. doi: 10.1053/joms.2003.50110. https://www.ncbi.nlm.nih.gov/pubmed/12730835[↩]
- Hjørting-Hansen E. Decortication in treatment of osteomyelitis of the mandible. Oral Surgery, Oral Medicine, Oral Pathology. 1970;29(5):641–655. doi: 10.1016/0030-4220(70)90259-8. https://www.ncbi.nlm.nih.gov/pubmed/5265872[↩]
- Theologie-Lygidakis N., Schoinohoriti O., Iatrou I. Surgical management of primary chronic osteomyelitis of the jaws in children: a prospective analysis of five cases and review of the literature. Oral & Maxillofacial Surgery. 2011;15(1):41–50. doi: 10.1007/s10006-010-0248-3. https://www.ncbi.nlm.nih.gov/pubmed/20978813[↩]
- Lentrodt S., Lentrodt J., Kübler N., Mödder U. Hyperbaric oxygen for adjuvant therapy for chronically recurrent mandibular osteomyelitis in childhood and adolescence. Journal of Oral and Maxillofacial Surgery. 2007;65(2):186–191. doi: 10.1016/j.joms.2005.11.106. https://www.ncbi.nlm.nih.gov/pubmed/17236919[↩]
- Kaplan SL. Osteomyelitis in children. Infect Dis Clin North Am. 2005;19(4):787–797.[↩]
- Aragón-Sánchez J, Lázaro-Martínez JL, Quintana-Mar-rero Y, et al. Are diabetic foot ulcers complicated by MRSA osteomyelitis associated with worse prognosis? Outcomes of a surgical series. Diabet Med. 2009;26(5):552–555.[↩]
- Kohli R, Hadley S. Fungal arthritis and osteomyelitis. Infect Dis Clin North Am. 2005;19(4):831–851.[↩]
- Pineda C, Espinosa R, Pena A. Radiographic imaging in osteomyelitis: the role of plain radiography, computed tomography, ultrasonography, magnetic resonance imaging, and scintigraphy. Semin Plast Surg. 2009;23(2):80–89.[↩][↩]
- Pineda C, Vargas A, Rodríguez AV. Imaging of osteomyelitis: current concepts. Infect Dis Clin North Am. 2006;20(4):789–825.[↩][↩][↩][↩]
- Dinh MT, Abad CL, Safdar N. Diagnostic accuracy of the physical examination and imaging tests for osteomyelitis underlying diabetic foot ulcers: meta-analysis. Clin Infect Dis. 2008;47(4):519–527.[↩]
- Kapoor A, Page S, Lavalley M, Gale DR, Felson DT. Magnetic resonance imaging for diagnosing foot osteomyelitis: a meta-analysis. Arch Intern Med. 2007;167(2):125–132.[↩][↩]
- Termaat MF, Raijmakers PG, Scholten HJ, Bakker FC, Patka P, Haarman HJ. The accuracy of diagnostic imaging for the assessment of chronic osteomyelitis: a systematic review and meta-analysis. J Bone Joint Surg Am. 2005;87(11):2464–2471.[↩][↩]
- Love C, Patel M, Lonner BS, Tomas MB, Palestro CJ. Diagnosing spinal osteomyelitis: a comparison of bone and Ga-67 scintigraphy and magnetic resonance imaging. Clin Nucl Med. 2000;25(12):963–977.[↩]
- Diagnosis and Management of Osteomyelitis. https://www.aafp.org/afp/2011/1101/p1027.html[↩][↩]
- Karamanis EM, Matthaiou DK, Moraitis LI, Falagas ME. Fluoroquinolones versus betalactam based regimens for the treatment of osteomyelitis: a meta-analysis of randomized controlled trials. Spine (Phila Pa 1976). 2008;33(10):E297–E304.[↩][↩]
- Davis JS. Management of bone and joint infections due to Staphylococcus aureus. Intern Med J. 2005;35(suppl 2):S79–S96.[↩]
- Bachur R, Pagon Z. Success of short-course parenteral antibiotic therapy for acute osteomyelitis of childhood. Clin Pediatr (Phila). 2007;46(1):30–35.[↩][↩]
- Tice AD, Hoaglund PA, Shoultz DA. Outcomes of osteomyelitis among patients treated with outpatient parenteral antimicrobial therapy. Am J Med. 2003;114(9):723–728.[↩]
- Conterno LO, da Silva Filho CR. Antibiotics for treating chronic osteomyelitis in adults. Cochrane Database Syst Rev. 2009;(3):CD004439.[↩][↩]
- Roblot F, Besnier JM, Juhel L, et al. Optimal duration of antibiotic therapy in vertebral osteomyelitis. Semin Arthritis Rheum. 2007;36(5):269–277.[↩]