Contents
What is phosphatidylcholine
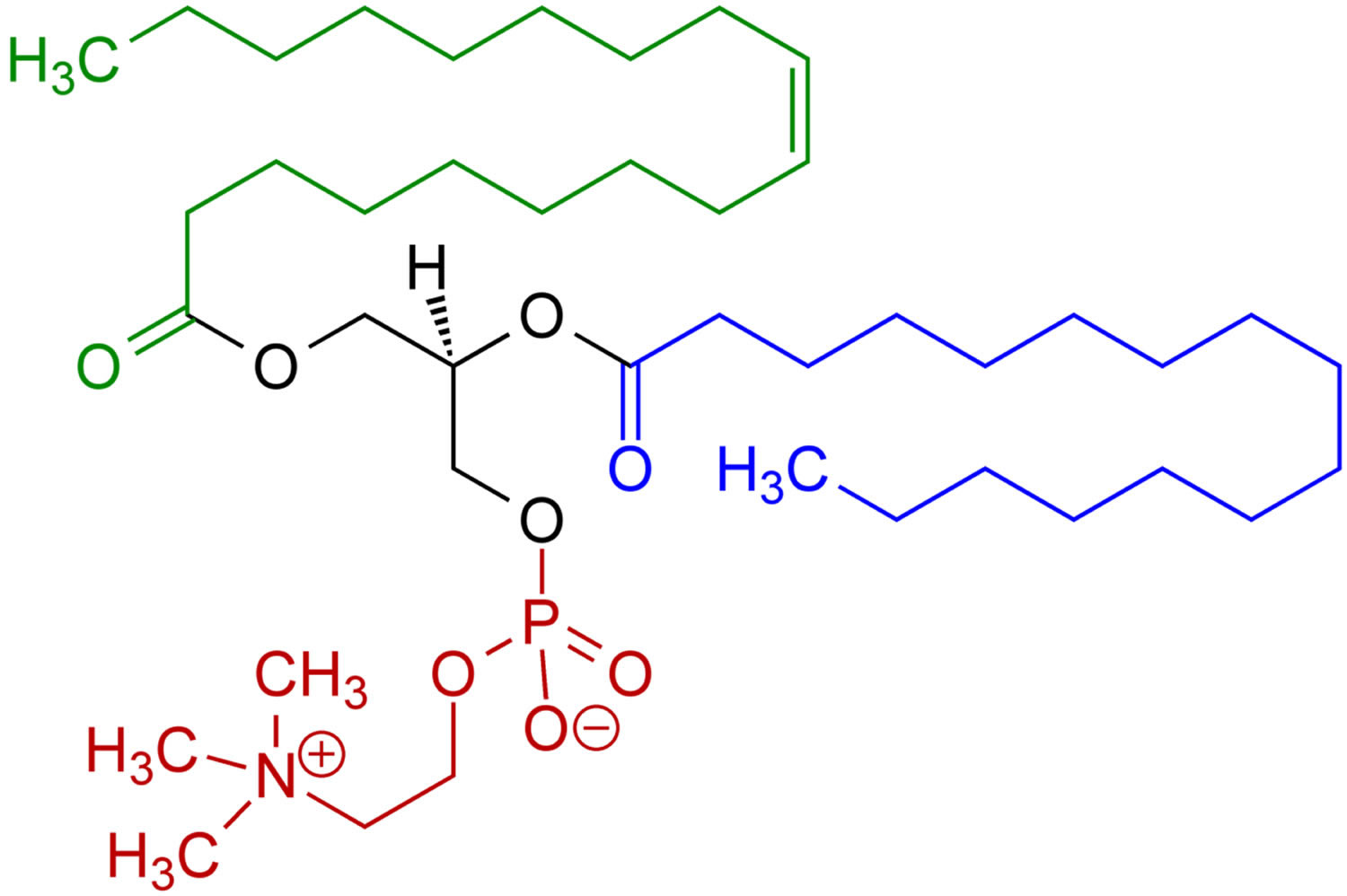
Phosphatidylcholine also called lecithin is a major constituent of the cell membranes which is more commonly found in the exoplasmic or outer leaflet of the plasma membrane 1. Phosphatidylcholine, the main component of lecithin, is a glycerophospholipid that incorporate choline as the head group. The fatty acids bound to the glycerophosphatidic acid can vary but generally one of them is unsaturated and the other one is saturated. The phospholipid phosphatidylcholine (lecithin) is the major dietary source of choline, a semiessential nutrient that is part of the vitamin B-complex family 2. Virtually all eukaryotic cells have an asymmetric distribution of phospholipids across their bilayer membrane, where the choline-containing phospholipids, phosphatidylcholine and sphingomyelin are predominately maintained on the outer membrane leaflet, and the amino-phospholipids (phosphatidylserine and phosphatidylethanolamine) are predominately localized in the inner membrane leaflet 3. This asymmetry is actively maintained by the regulated activity of ATP-dependent lipid transporters. However, membrane asymmetry collapses under a variety of physiological and pathological conditions resulting in dramatic changes in the biochemical properties of the membrane. For example, the redistribution of phosphatidylserine to the external face of the plasma membrane flags cells for their recognition, phagocytosis 4, and ultimate degradation by phagocytes (efferocytosis). Moreover, the interaction between phosphatidylserine-expressing cells and immune cells elicits profound immunological consequences by triggering immunosuppressive pathways that prevent both local and systemic immune activation. Although these pathways are used by apoptotic cells to quell potential immune sequelae against ‘self’, these same pathways are hijacked by pathogens and tumors to promote their sinister life-threatening expansion. Taken together, these observations suggest that phosphatidylserine functions as an upstream immune checkpoint that suppresses the development of immunity. This raises the possibility that phosphatidylserine blockade by the therapeutic administration of phosphatidylserine-targeting agents can restore pathogen and tumor immunity.
In mice, higher dietary phosphatidylcholine led to increases in plasma concentrations of trimethylamine-N-oxide (TMAO) and eventually promoted atherosclerosis 5. In humans, a dietary phosphatidylcholine challenge induced increased postprandial concentrations of trimethylamine-N-oxide (TMAO) in both blood and urine samples 6. However, the evidence for an association of dietary phosphatidylcholine with cardiovascular disease and mortality in humans is limited.
Trimethylamine-N-oxide (TMAO), an oxidation product of trimethylamine (TMA), is a relatively common metabolite of choline in animals 7. Foods rich in the lipid phosphatidylcholine, which predominantly includes eggs, milk, liver, red meat, poultry, shell fish and fish, are believed to be the major dietary sources for choline, and hence trimethylamine-N-oxide (TMAO) production 8. Briefly, initial catabolism of choline and other trimethylamine containing species (e.g. betaine) by intestinal microbes forms the gas trimethylamine (TMA) 9, which is efficiently absorbed and rapidly metabolized by at least one member of the hepatic flavin monooxygenase family of enzymes, FMO3, to form trimethylamine-N-oxide (TMAO) 10. Increased trimethylamine-N-oxide (TMAO) levels are associated with an increased risk of incident major adverse cardiovascular events 11.
Studies in germ-free mice and cross-sectional clinical studies in humans have suggested a role for the intestinal microbiota in the pathogenesis of atherosclerosis in patients with a diet rich in phosphatidylcholine (with major sources including eggs, liver, beef, and pork) through the formation of the metabolite trimethylamine and conversion to trimethylamine-N-oxide (TMAO) 12.
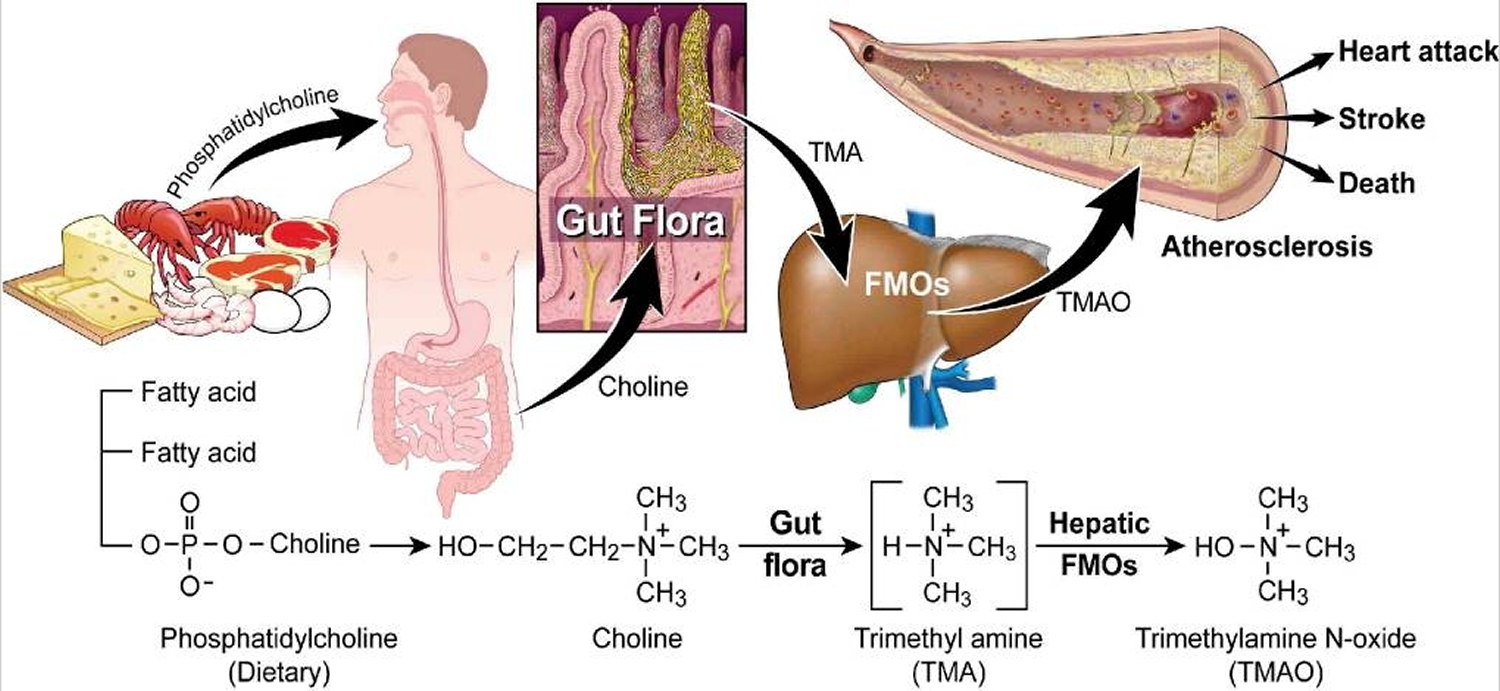
Ingested phosphatidylcholine (lecithin), the major dietary source of total choline, is acted on by intestinal lipases to form a variety of metabolic products, including the choline-containing nutrients glycerophosphocholine, phosphocholine, and choline. Choline-containing nutrients that reach the cecum and large intestine may serve as fuel for intestinal microbiota (gut flora), producing trimethylamine (TMA). TMA is rapidly further oxidized to trimethylamine-N-oxide (TMAO) by hepatic flavin-containing monooxygenases (FMOs). TMAO enhances the accumulation of cholesterol in macro-phages, the accumulation of foam cells in artery walls, and atherosclerosis 12, all factors that are associated with an increased risk of heart attack, stroke, and death. Choline can also be oxidized to betaine in both the liver and kidneys. Dietary betaine can serve as a substrate for bacteria to form TMA 13 and presumably TMAO 14. Nevertheless, the high correlation between urine and plasma levels of trimethylamine-N-oxide (TMAO) argues for effective urinary clearance of TMAO. Hence, an efficient excretion mechanism may provide protection by preventing the accumulation of TMAO and does not undermine the mechanistic link between TMAO and cardiovascular risk 11.
In another study, dietary phosphatidylcholine intake was consistently associated with an increased risk of type 2 diabetes in 3 US populations 15. Furthermore, patients with type 2 diabetes had a 2- to 3-fold higher risk of cardiovascular disease, cardiovascular-specific mortality, or premature mortality than did the general population 16. Thus far, the exact pathogenic mechanisms underlying this increased risk are not fully understood. A previous study showed that the risk of incident major adverse cardiovascular events, including mortality attributable to increased blood TMAO concentrations, appeared to be greater in diabetic patients than in those without diabetes (hazard ratios comparing the top and bottom quintiles of TMAO concentrations were 2.5 and 1.6 in diabetic and nondiabetic subjects, respectively) 11.
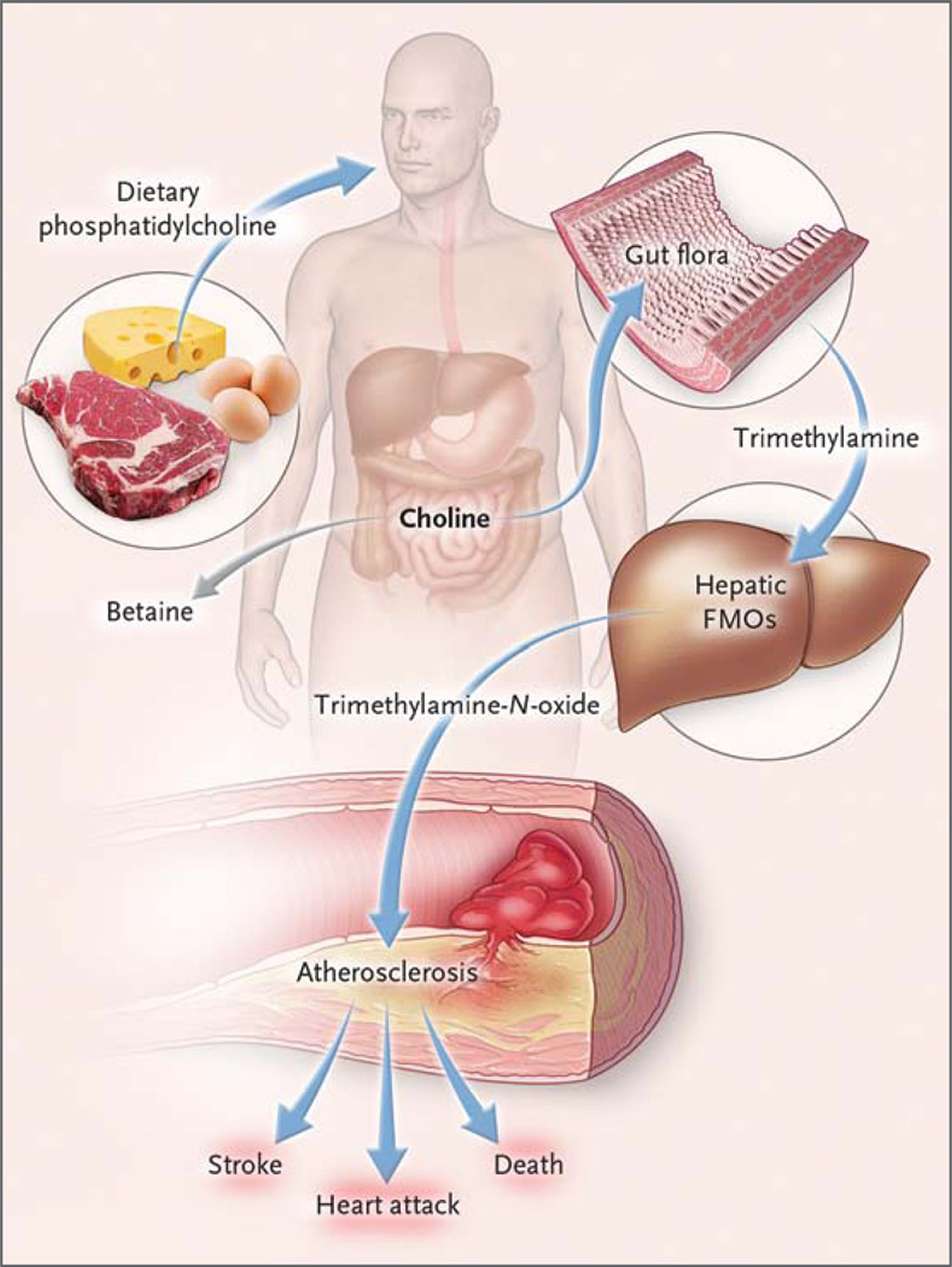
In this 2016 prospective study 17 involving 80,978 women and 39,434 men who were followed for up to 32 years, the researchers observed that higher habitual dietary intakes of phosphatidylcholine were associated with an increased risk of all-cause mortality, especially cardiovascular disease-specific mortality. Such associations were independent of traditional cardiovascular disease risk factors. Compelling evidence suggests that circulating TMAO might advance atherosclerosis by disturbing the clearance of cholesterol in the liver 18, and higher blood TMAO concentrations have been related to an increased risk of cardiovascular disease and mortality 19. Other putative mechanisms may involve upregulation of macrophage scavenger receptors, augmented macrophage cholesterol accumulation, and foam cell formation, which result in increased inflammation and oxidation of LDL “bad” cholesterol 12.
The associations of dietary phosphatidylcholine with all-cause and cardiovascular disease-specific mortality were stronger among diabetic than in nondiabetic participants, although in both populations such associations were all significant. The associations of dietary phosphatidylcholine with all-cause and cardiovascular disease-specific mortality appeared to be consistent in both men and women. This observation is in line with a previous report that the associations of TMAO concentrations with composite risk of incident cardiovascular disease and mortality were comparable, in general, between sexes 6.
In conclusion, research have found that a high intake of phosphatidylcholine, which could lead to a higher production of TMAO, was significantly associated with an increased risk of all-cause and cardiovascular disease-specific mortality, in particular among diabetic patients.
Figure 1. Phosphatidylcholine
Figure 2. Gut flora dependent metabolism of dietary phosphatidylcholine and atherosclerosis. Schematic summary illustrating newly discovered pathway for gut flora mediated generation of pro-atherosclerotic metabolite from dietary phosphatidylcholine
[Source 20]Figure 3. Pathways Linking Dietary Phosphatidylcholine, Intestinal Microbiota, and Incident Adverse Cardiovascular Events
Phosphatidylcholine also plays a role in membrane-mediated cell signaling. The phospholipase D-mediated catabolism of phosphatidylcholine yields phosphatidic acid and choline, which are important lipid second messengers involved in several signaling pathways 21. Phosphatidic acid binds to Raf-1 and promotes its recruitment to the plasma membrane where it is activated by direct interaction with Ras 22. Ras-mediated Raf-1 activation leads to mitogen-activated protein kinase (MAPK) and PI3K/Akt activation 23. Therefore, PA would have a pivotal role in the amplification of signaling cascades required for survival and growth 24. Phosphatidic acid also binds the mammalian target of rapamycin (mTOR), a protein kinase that regulates cell cycle progression and cell growth regulating several cellular events like translation, transcription, membrane trafficking, and protein degradation 25.
Phosphatidylcholine vs Choline
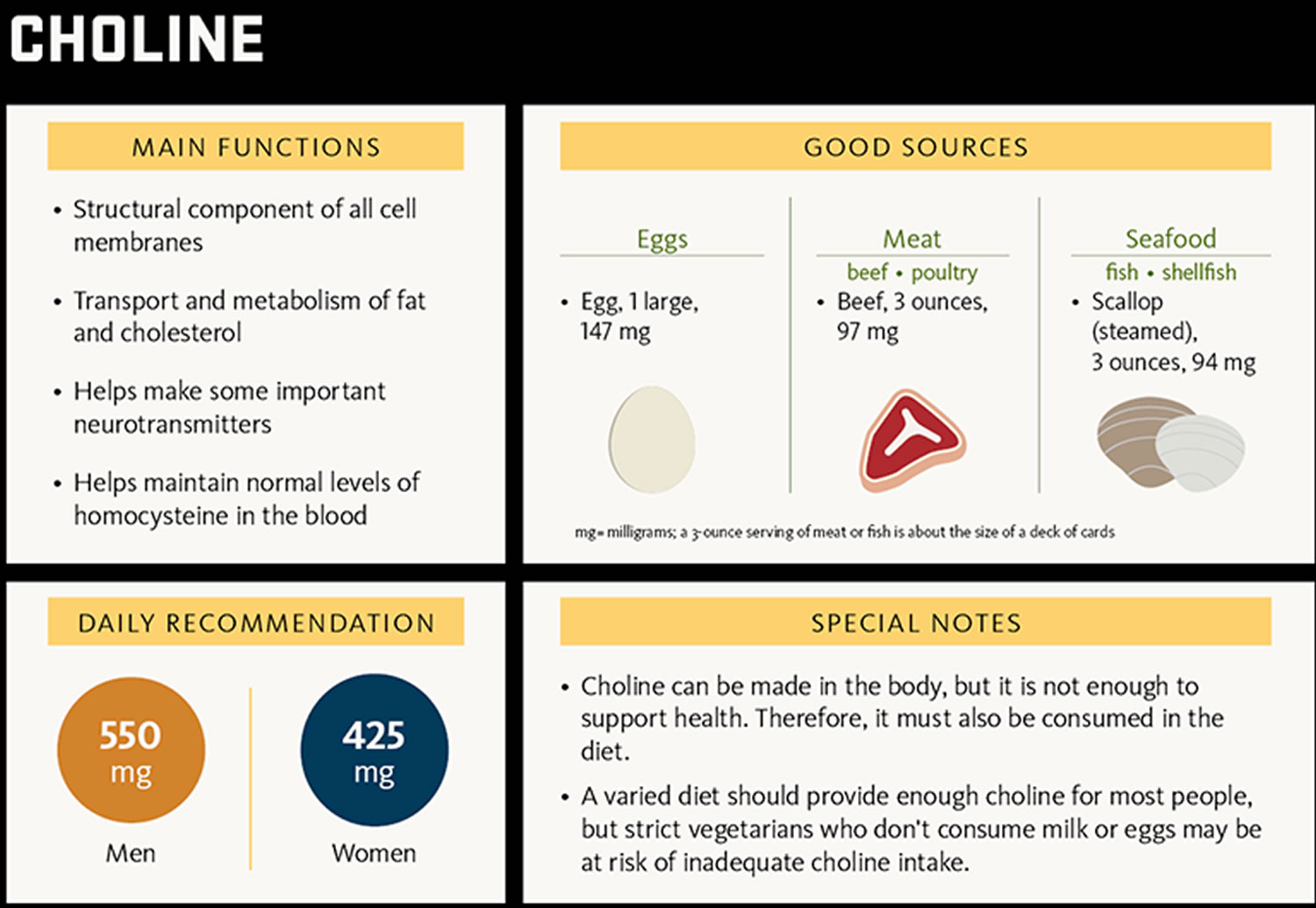
Choline (2-hydroxy-N,N,N-trimethylethanaminium) is an essential nutrient; that is, together with essential amino acids, fatty acids, vitamins and minerals, choline must be obtained from the diet to maintain health 26. Humans can synthesize small amounts of choline but not enough to support health. Therefore, choline is considered an essential nutrient and must be consumed in the diet. Choline is naturally present in some foods and available as a dietary supplement. Choline is a source of methyl groups needed for many steps in metabolism. The body needs choline to synthesize phosphatidylcholine and sphingomyelin, two major phospholipids vital for cell membranes 27. Therefore, all plant and animal cells need choline to preserve their structural integrity 28. In addition, choline is needed to produce acetylcholine, an important neurotransmitter for memory, mood, muscle control, and other brain and nervous system functions 28. Choline also plays important roles in modulating gene expression, cell membrane signaling, lipid transport and metabolism, and early brain development 28.
Humans can produce choline endogenously in the liver, mostly as phosphatidylcholine, but the amount that the body naturally synthesizes is not sufficient to meet human needs 29. As a result, humans must obtain some choline from the diet. Premenopausal women might need less choline from the diet than children or other adults because estrogen induces the gene that catalyzes the biosynthesis of choline 29. When a diet is deficient in folate, a B-vitamin that is also a methyl donor, the need for dietary choline rises because choline becomes the primary methyl donor 28.
The most common sources of choline in foods are the fat-soluble phospholipids phosphatidylcholine and sphingomyelin as well as the water-soluble compounds phosphocholine, glycerolphosphocholine, and free choline 28. When these choline-containing compounds are ingested, pancreatic and mucosal enzymes liberate free choline from about half of the fat-soluble forms and some water-soluble forms 30. Free choline, phosphocholine, and glycerophosphocholine are absorbed in the small intestine, enter the portal circulation, and are stored in the liver, where they are subsequently phosphorylated and distributed throughout the body to make cell membranes 28. The remaining fat-soluble phospholipids (phosphatidylcholine and sphingomyelin) are absorbed intact, incorporated into chylomicrons, and secreted into the lymphatic circulation, where they are distributed to tissues and other organs, including the brain and placenta 28.
Choline status is not routinely measured in healthy people. In healthy adults, the concentration of choline in plasma ranges from 7 to 20 mcmol/L 31. According to one study, the range is 7–9.3 mcmol/L in fasting adults 32. Plasma choline levels do not decline below 50% of normal, even in individuals who have not eaten for more than a week 33. This may be due to the hydrolysis of membrane phospholipids, a source of choline, to maintain plasma choline concentrations above this minimal level, or to endogenous synthesis 31.
Intake recommendations for choline and other nutrients are provided in the Dietary Reference Intakes (DRIs) developed by the Food and Nutrition Board of the Institute of Medicine (IOM) 31. Dietary Reference Intakes (DRIs) is the general term for a set of reference values used for planning and assessing nutrient intakes of healthy people. These values, which vary by age and sex, include:
- Recommended Dietary Allowance (RDA): Average daily level of intake sufficient to meet the nutrient requirements of nearly all (97%–98%) healthy individuals; often used to plan nutritionally adequate diets for individuals.
- Adequate Intake (AI): Intake at this level is assumed to ensure nutritional adequacy; established when evidence is insufficient to develop an RDA.
- Estimated Average Requirement (EAR): Average daily level of intake estimated to meet the requirements of 50% of healthy individuals; usually used to assess the nutrient intakes of groups of people and to plan nutritionally adequate diets for them; can also be used to assess the nutrient intakes of individuals.
- Tolerable Upper Intake Level (UL): Maximum daily intake unlikely to cause adverse health effects.
Insufficient data were available to establish an estimated Average Requirement (EAR) for choline, so the Food and Nutrition Board established adequate intakes (AIs) for all ages that are based on the prevention of liver damage as measured by serum alanine aminostransferase levels 31. The amount of choline that individuals need is influenced by the amount of methionine, betaine, and folate in the diet; gender; pregnancy; lactation; stage of development; ability to produce choline endogenously; and genetic mutations that affect choline needs 31. Table 1 lists the current adequate intakes (AIs) for choline.
Table 1. Adequate Intakes (AIs) for Choline
| Age | Male | Female | Pregnancy | Lactation |
|---|---|---|---|---|
| Birth to 6 months | 125 mg/day | 125 mg/day | ||
| 7–12 months | 150 mg/day | 150 mg/day | ||
| 1–3 years | 200 mg/day | 200 mg/day | ||
| 4–8 years | 250 mg/day | 250 mg/day | ||
| 9–13 years | 375 mg/day | 375 mg/day | ||
| 14–18 years | 550 mg/day | 400 mg/day | 450 mg/day | 550 mg/day |
| 19+ years | 550 mg/day | 425 mg/day | 450 mg/day | 550 mg/day |
Most people in the United States consume less than the adequate intakes (AIs) for choline. An analysis of data from the 2013–2014 National Health and Nutrition Examination Survey (NHANES) found that the average daily choline intake from foods and beverages among children and teens is 256 mg for ages 2–19 34. In adults, the average daily choline intake from foods and beverages is 402 mg in men and 278 mg in women. Intakes from supplements contribute a very small amount to total choline intakes.
According to an analysis of 2007–2008 NHANES data, black males of all ages had lower mean choline intakes than their white and Hispanic counterparts, but choline intakes did not differ substantially among females of different races/ethnicities 35.
Table 2. Foods high in choline
| Food | Milligrams (mg) per serving | Percent DV* |
|---|---|---|
| Beef liver, pan fried, 3 ounces | 356 | 65 |
| Egg, hard boiled, 1 large egg | 147 | 27 |
| Beef top round, separable lean only, braised, 3 ounces | 117 | 21 |
| Soybeans, roasted, ½ cup | 107 | 19 |
| Chicken breast, roasted, 3 ounces | 72 | 13 |
| Beef, ground, 93% lean meat, broiled, 3 ounces | 72 | 13 |
| Fish, cod, Atlantic, cooked, dry heat, 3 ounces | 71 | 13 |
| Mushrooms, shiitake, cooked, ½ cup pieces | 58 | 11 |
| Potatoes, red, baked, flesh and skin, 1 large potato | 57 | 10 |
| Wheat germ, toasted, 1 ounce | 51 | 9 |
| Beans, kidney, canned, ½ cup | 45 | 8 |
| Quinoa, cooked, 1 cup | 43 | 8 |
| Milk, 1% fat, 1 cup | 43 | 8 |
| Yogurt, vanilla, nonfat, 1 cup | 38 | 7 |
| Brussels sprouts, boiled, ½ cup | 32 | 6 |
| Broccoli, chopped, boiled, drained, ½ cup | 31 | 6 |
| Cottage cheese, nonfat, 1 cup | 26 | 5 |
| Fish, tuna, white, canned in water, drained in solids, 3 ounces | 25 | 5 |
| Peanuts, dry roasted, ¼ cup | 24 | 4 |
| Cauliflower, 1” pieces, boiled, drained, ½ cup | 24 | 4 |
| Peas, green, boiled, ½ cup | 24 | 4 |
| Sunflower seeds, oil roasted, ¼ cup | 19 | 3 |
| Rice, brown, long-grain, cooked, 1 cup | 19 | 3 |
| Bread, pita, whole wheat, 1 large (6½ inch diameter) | 17 | 3 |
| Cabbage, boiled, ½ cup | 15 | 3 |
| Tangerine (mandarin orange), sections, ½ cup | 10 | 2 |
| Beans, snap, raw, ½ cup | 8 | 1 |
| Kiwifruit, raw, ½ cup sliced | 7 | 1 |
| Carrots, raw, chopped, ½ cup | 6 | 1 |
| Apples, raw, with skin, quartered or chopped, ½ cup | 2 | 0 |
*DV = Daily Value. DVs were developed by the U.S. Food and Drug Administration (FDA) to help consumers compare the nutrient contents of products within the context of a total diet. The DV for choline is 550 mg for adults and children age 4 and older 36. However, the FDA does not require food labels to list choline content unless a food has been fortified with this nutrient. Foods providing 20% or more of the DV are considered to be high sources of a nutrient.
The U.S. Department of Agriculture’s (USDA’s) National Nutrient Database for Standard Reference 37 lists the nutrient content of many foods and provides a comprehensive list of foods containing choline arranged by choline content 38 and by food name 39.
Phosphatidylserine vs phosphatidylcholine
Phosphatidylserine is the major anionic phospholipid class particularly enriched in the inner leaflet of the plasma membrane in nerve tissues 40. The phosphatidylserine biosynthesis preferentially utilizes docosahexaenoic acid (DHA)-containing phospholipids as substrates. Although membrane phospholipids are under a tight homeostatic regulation, the phosphatidylserine level can be altered according to the docosahexaenoic acid (DHA) status, specifically in the brain. Therefore, diet- or ethanol-induced alteration of the brain docosahexaenoic acid (DHA) level and membrane phosphatidylserine can influence the signaling platform in the membrane and the transmission of the signaling cues. Detailed molecular mechanisms, particularly membrane phosphatidylserine-protein interactions, warrant further investigation in order to obtain more insight into the functional significance of neuronal phosphatidylserine.
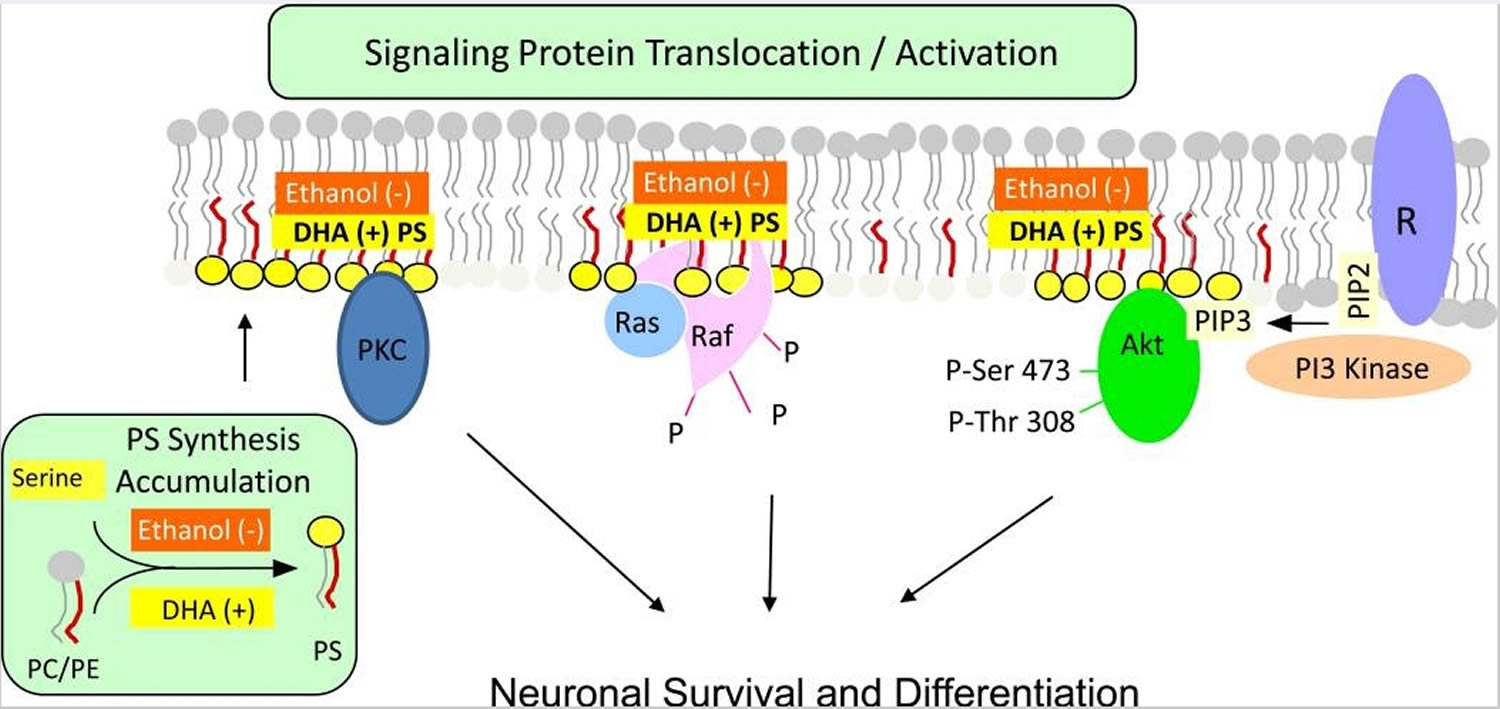
Phosphatidylserine is the major acidic phospholipid class that accounts for 13–15 % of the phospholipids in the human cerebral cortex 41. In the plasma membrane, phosphatidylserine is localized exclusively in the cytoplasmic leaflet where it forms part of protein docking sites necessary for the activation of several key signaling pathways. These include the Akt, protein kinase C (PKC) and Raf-1 signaling that is known to stimulate neuronal survival, neurite growth and synaptogenesis 42. Modulation of the phosphatidylserine level in the plasma membrane of neurons has significant impact on these signaling processes. The mechanism of phosphatidylserine-mediated activation of these neuronal signaling pathways is illustrated in Figure 4.
Figure 4. Phosphatidylserine facilitating activation of neuronal signaling. Phosphatidylserine facilitates the activation of signaling proteins and receptors that are critical for neuronal survival, differentiation and synaptic neurotransmission.
Footnotes: Activation of Akt, protein kinase C and Raf-1 requires translocation from the cytosol to the cytoplasmic surface of the plasma membrane. Translocation is initiated by specific stimuli, for example, growth factor-dependent PIP3 generation from PIP2 by PI3 kinase in the case of Akt. Binding to the membrane occurs in part through an interaction of these proteins with phosphatidylserine present in anionic domains of the lipid bilayer, activating the signaling pathways leading to neuronal differentiation and survival. DHA facilitates this mechanism by increasing phosphatidylserine production in neurons, while ethanol has the opposite effect because it inhibits the DHA-induced increase in phosphatidylserine production. R: receptor
Abbreviations: PS= phosphatidylserine
[Source 40]Phosphatidylserine is synthesized from phosphatidylcholine or phosphatidylethanolamine by exchanging the base head group with serine in reactions are catalyzed by phosphatidylserine synthase 1 and phosphatidylserine synthase 2 located in the endoplasmic reticulum. Activation of Akt, Raf-1 and protein kinase C signaling, which supports neuronal survival and differentiation, requires interaction of these proteins with phosphatidylserine localized in the cytoplasmic leaflet of the plasma membrane. Furthermore, neurotransmitter release by exocytosis and a number of synaptic receptors and proteins are modulated by phosphatidylserine present in the neuronal membranes. Brain is highly enriched with docosahexaenoic acid (DHA), and brain phosphatidylserine has a high docosahexaenoic acid (DHA) content. By promoting phosphatidylserine synthesis, docosahexaenoic acid (DHA) can uniquely expand the phosphatidylserine pool in neuronal membranes and thereby influence phosphatidylserine-dependent signaling and protein function. Ethanol decreases docosahexaenoic acid (DHA)-promoted phosphatidylserine synthesis and accumulation in neurons, which may contribute to the deleterious effects of ethanol intake. Improvement of some memory functions has been observed in cognitively impaired subjects as a result of phosphatidylserine supplementation, but the mechanism is unclear.
A decrease of the DHA content in phosphatidylserine has been reported in cognitive impairment. A small reduction in the docosahexaenoic acid (DHA) content of hippocampal phosphatidylserine was observed in 12 month-old senescence-accelerated prone mice that have a shorter life span, learning and memory deficit, and an increase in hippocampal Aβ-peptide content 43. The decrease in DHA was associated with a corresponding increase in the arachidonic acid content of hippocampal phosphatidylserine. Likewise, the DHA content of phosphatidylserine in the superior temporal and mid-frontal cortex was reduced by 12 and 14 %, respectively, in brain tissues obtained from patients with Alzheimer’s disease 44. However, there is no information as to how a decrease in the DHA content of phosphatidylserine might contribute to the pathogenesis of cognitive impairment, and a decrease in DHA content is not a uniform finding in animal models of cognitive impairment. For example, substantial fatty acyl compositional changes, including reductions in arachidonic acid, have been observed in brain phosphatidylserine of aged Wistar rats with cognitive deficits, but there is no difference in the DHA content of the phosphatidylserine 45. Therefore, the putative linkage between DHA reductions in phosphatidylserine and cognitive impairment remains open to question.
Dietary phosphatidylserine supplements are reported to improve cognitive function in experimental animals 46, and a similar result has been obtained recently with krill phosphatidylserine which has a high content of omega-3 fatty acid. Aged rats given daily doses of krill phosphatidylserine orally for 7 days showed improvement in the Morris water maze test. There was less loss of choline acetyltransferase and acetylcholine esterase transporter mRNA in the hippocampus. The neuroprotective activity of 20 mg/kg krill phosphatidylserine was equivalent to that of 50 mg/kg soy phosphatidylserine in these aged rats 47. Normal young rats given 100 mg/kg krill phosphatidylserine orally for 30 days also showed improvement in the Morris water maze test 48.
Likewise, cognitive improvement was reported in humans given oral phosphatidylserine supplements 49, and these findings subsequently were confirmed and extended. Human subjects treated for 42 days with 200 mg of soy-based phosphatidylserine showed a more relaxed state before and after mental stress as measured by electro-encephalography 50, and no adverse effects were evident at this dose given three-times a day for 6 to 12 weeks 51. The ability to recall words increased by 42% in male and female subjects who were older than 60 years and complained of subjective memory loss when they were treated with 300 mg/day of phosphatidylserine containing 37.5 mg of eicosapentaenoic acid and DHA 52. Improved verbal immediate recall was also observed in a double-blind, placebo controlled clinical trial in a large group of elderly subjects with memory complaints when treated with a daily dose of 300 mg phosphatidylserine containing DHA and eicosapentaenoic acid in a 3:1 ratio. A subset with relatively good cognitive performance at baseline showed the greatest improvement 53. In a similar double blind study in Japanese subjects between the ages of 50 and 69 years with memory complaints, subjects having low scores at baseline showed greater improvement in delayed verbal recall after treatment for 6 months with soybean phosphatidylserine 54.
Several biochemical responses to phosphatidylserine administration that have been reported in experimental animals could be involved in the mechanism of phosphatidylserine-mediated improvement in cognition. Stimulation of dopamine-dependent adenylate cyclase activity was observed in mouse brain following an intravenous infusion of a sonicated preparation of bovine brain phosphatidylserine 55. Sonicated suspensions of phosphatidylserine injected intravenously also increased calcium-dependent acetylcholine output from the cerebral cortex in urethane anesthetized rats 56. Likewise, intravenous injection of purified bovine brain phosphatidylserine for 8 days attenuated the decrease in acetylcholine release from the parietal cortex in aged rats, possibly by providing more choline for acetylcholine synthesis 57. Furthermore, orally administered krill phosphatidylserine in normal young rats for 30 days produced an increase in neurons positive for brain-derived neurotrophic factor and insulin-like growth factor in the hippocampal CA1 region 48.
A current hypothesis is that these biochemical responses and the resulting cognitive improvements are due to phosphatidylserine mediated effects on neuronal membrane properties 58. However, experimental evidence indicating that orally or intravenously administered phosphatidylserine actually alters neuronal membrane properties is lacking. How the administered phosphatidylserine is transported in the plasma, how much enters the brain, whether it is taken up intact, and whether it is incorporated into neurons or glia are not known. Dietary phospholipids are hydrolyzed during digestion, so orally administered phosphatidylserine most likely is not absorbed intact. phosphatidylserine preparations are rich in DHA, and DHA supplementation is known to improve hippocampal function 59. Because the administered phosphatidylserine probably undergoes partial or complete hydrolysis, the beneficial effects of phosphatidylserine on cognition, particularly from krill or bovine sources, possibly are produced by DHA released from the phosphatidylserine rather than the intact phosphatidylserine itself. These issues will have to be investigated in order to obtain some mechanistic insight into how dietary or intravenously administered phosphatidylserine supplements function to produce cognitive improvement.
- Gándola YB, Pérez SE, Irene PE, et al. Mitogenic Effects of Phosphatidylcholine Nanoparticles on MCF-7 Breast Cancer Cells. BioMed Research International. 2014;2014:687037. doi:10.1155/2014/687037. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3977480/[↩]
- Patterson KY, Bhagwat SA, Williams JR, Howe JC, Holden JM. USDA database for the choline content of common foods: release two. 2008 https://www.ars.usda.gov/ARSUserFiles/80400525/Data/Choline/Choln02.pdf[↩]
- The distribution and function of phosphatidylserine in cellular membranes. Leventis PA, Grinstein S. Annu Rev Biophys. 2010; 39():407-27. https://www.annualreviews.org/doi/full/10.1146/annurev.biophys.093008.131234[↩]
- In vivo recognition and clearance of red blood cells containing phosphatidylserine in their plasma membranes. Schroit AJ, Madsen JW, Tanaka Y. J Biol Chem. 1985 Apr 25; 260(8):5131-8. http://www.jbc.org/content/260/8/5131.long[↩]
- Wang Z, Klipfell E, Bennett BJ, Koeth R, Levison BS, Dugar B, Feldstein AE, Britt EB, Fu X, Chung YM, et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011;472:57–63 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3086762/[↩]
- Tang WH, Wang Z, Levison BS, Koeth RA, Britt EB, Fu X, Wu Y, Hazen SL. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N Engl J Med 2013;368:1575–84. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3701945/[↩][↩]
- Biochemical and clinical aspects of the human flavin-containing monooxygenase form 3 (FMO3) related to trimethylaminuria. Cashman JR, Camp K, Fakharzadeh SS, Fennessey PV, Hines RN, Mamer OA, Mitchell SC, Nguyen GP, Schlenk D, Smith RL, Tjoa SS, Williams DE, Yannicelli S. Curr Drug Metab. 2003 Apr; 4(2):151-70. https://www.ncbi.nlm.nih.gov/pubmed/12678693/[↩]
- Zeisel SH, Mar MH, Howe JC, Holden JM. Concentrations of choline-containing compounds and betaine in common foods. J Nutr. 2003;133:1302–1307[↩]
- al-Waiz M, Mikov M, Mitchell SC, Smith RL. The exogenous origin of trimethylamine in the mouse. Metabolism. 1992;41:135–136. https://www.ncbi.nlm.nih.gov/pubmed/1736035[↩]
- Zhang AQ, Mitchell SC, Smith RL. Dietary precursors of trimethylamine in man: a pilot study. Food Chem Toxicol. 1999;37:515–520.[↩]
- Tang WHW, Wang Z, Levison BS, et al. Intestinal Microbial Metabolism of Phosphatidylcholine and Cardiovascular Risk. The New England journal of medicine. 2013;368(17):1575-1584. doi:10.1056/NEJMoa1109400. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3701945/[↩][↩][↩]
- Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Wang Z, Klipfell E, Bennett BJ, Koeth R, Levison BS, Dugar B, Feldstein AE, Britt EB, Fu X, Chung YM, Wu Y, Schauer P, Smith JD, Allayee H, Tang WH, DiDonato JA, Lusis AJ, Hazen SL. Nature. 2011 Apr 7; 472(7341):57-63. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3086762/[↩][↩][↩]
- Bennett BJ, de Aguiar Vallim TQ, Wang Z, et al. Trimethylamine-N-oxide, a metabolite associated with atherosclerosis, exhibits complex genetic and dietary regulation. Cell Metab. 2013;17:49–60. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3771112/[↩]
- Möller B, Hippe H, Gottschalk G. Degradation of various amine compounds by mesophilic clostridia. Arch Microbiol. 1986;145:85–90. https://www.ncbi.nlm.nih.gov/pubmed/3753143[↩]
- Dietary phosphatidylcholine intake and type 2 diabetes in men and women. Li Y, Wang DD, Chiuve SE, Manson JE, Willett WC, Hu FB, Qi L. Diabetes Care. 2015 Feb; 38(2):e13-4. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4302257/[↩]
- The independent effect of type 2 diabetes mellitus on ischemic heart disease, stroke, and death: a population-based study of 13,000 men and women with 20 years of follow-up. Almdal T, Scharling H, Jensen JS, Vestergaard H. Arch Intern Med. 2004 Jul 12; 164(13):1422-6. https://jamanetwork.com/journals/jamainternalmedicine/fullarticle/217184[↩]
- Zheng Y, Li Y, Rimm EB, et al. Dietary phosphatidylcholine and risk of all-cause and cardiovascular-specific mortality among US women and men. The American Journal of Clinical Nutrition. 2016;104(1):173-180. doi:10.3945/ajcn.116.131771. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4919531/[↩]
- Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. Tang WH, Wang Z, Levison BS, Koeth RA, Britt EB, Fu X, Wu Y, Hazen SL. N Engl J Med. 2013 Apr 25; 368(17):1575-84. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3701945/[↩]
- Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Koeth RA, Wang Z, Levison BS, Buffa JA, Org E, Sheehy BT, Britt EB, Fu X, Wu Y, Li L, Smith JD, DiDonato JA, Chen J, Li H, Wu GD, Lewis JD, Warrier M, Brown JM, Krauss RM, Tang WH, Bushman FD, Lusis AJ, Hazen SL. Nat Med. 2013 May; 19(5):576-85. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3650111/[↩]
- Wang Z, Klipfell E, Bennett BJ, et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature. 2011;472(7341):57-63. doi:10.1038/nature09922. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3086762/[↩]
- Rizzo M, Romero G. Pharmacological importance of phospholipase D and phosphatidic acid in the regulation of the mitogen-activated protein kinase cascade. Pharmacology & Therapeutics. 2002;94(1-2):35–50[↩]
- Rizzo MA, Shome K, Watkins SC, Romero G. The recruitment of Raf-1 to membranes is mediated by direct interaction with phosphatidic acid and is independent of association with Ras. The Journal of Biological Chemistry. 2000;275(31):23911–23918[↩]
- Clem BF, Clem AL, Yalcin A, et al. A novel small molecule antagonist of choline kinase-α that simultaneously suppresses MAPK and PI3K/AKT signaling. Oncogene. 2011;30(30):3370–3380.[↩]
- Gomez-Cambronero J. New concepts in phospholipase D signaling in inflammation and cancer. TheScientificWorldJournal. 2010;10:1356–1369.[↩]
- Laplante M, Sabatini DM. mTOR signaling in growth control and disease. Cell. 2012;149(2):274–293.[↩]
- Food and Nutrition Board . Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Panthotenic Acid, Biotin, and Cholin. National Academy Press; Washington, DC, USA: 1998[↩]
- Blusztajn JK, Slack BE, Mellott TJ. Neuroprotective Actions of Dietary Choline. Nutrients. 2017;9(8):815. doi:10.3390/nu9080815. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5579609/[↩]
- Zeisel SH, Corbin KD. Choline. In: Erdman JW, Macdonald IA, Zeisel SH, eds. Present Knowledge in Nutrition. 10th ed. Washington, DC: Wiley-Blackwell; 2012:405-18.[↩][↩][↩][↩][↩][↩][↩]
- Zeisel SH. Choline. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2014:416-26.[↩][↩]
- Hollenbeck CB. An introduction to the nutrition and metabolism of choline. Cent Nerv Syst Agents Med Chem 2012;12:100-13.[↩]
- Institute of Medicine. Food and Nutrition Board. Dietary Reference Intakes: Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: National Academy Press; 1998.[↩][↩][↩][↩][↩][↩]
- Holm PI, Ueland PM, Kvalheim G, Lien EA. Determination of choline, betaine, and dimethylglycine in plasma by a high-throughput method based on normal-phase chromatography-tandem mass spectrometry. Clin Chem 2003;49:286-94[↩]
- Zeisel SH. Choline. In: Coates PM, Betz JM, Blackman MR, et al., eds. Encyclopedia of Dietary Supplements. 2nd ed. London and New York: Informa Healthcare; 2010:136-43.[↩]
- U.S. Department of Agriculture, Agricultural Research Service. Nutrient Intakes from Food and Beverages: Mean Amounts Consumed per Individual, by Gender and Age, What We Eat in America, NHANES 2013-2014. 2016 https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/food-surveys-research-group/docs/wweia-data-tables/[↩]
- Chester DN, Goldman JD, Ahuja JK, Moshfegh AJ. Dietary Intakes of Choline: What We Eat in America, NHANES 2007-2008. Dietary Data Brief 2011;9:1-4.[↩]
- U.S. Food and Drug Administration. https://www.federalregister.gov/documents/2016/05/27/2016-11867/food-labeling-revision-of-the-nutrition-and-supplement-facts-labels[↩]
- U.S. Department of Agriculture, Agricultural Research Service. USDA National Nutrient Database for Standard Reference. https://ndb.nal.usda.gov/ndb/[↩]
- https://ndb.nal.usda.gov/ndb/nutrients/report/nutrientsfrm?max=25&offset=0&totCount=0&nutrient1=421&nutrient2=&nutrient3=&subset=0&fg=&sort=c&measureby=m[↩]
- https://ndb.nal.usda.gov/ndb/nutrients/report/nutrientsfrm?max=25&offset=0&totCount=0&nutrient1=421&nutrient2=&nutrient3=&subset=0&fg=&sort=f&measureby=m[↩]
- Kim H-Y, Huang BX, Spector AA. Phosphatidylserine in the Brain: Metabolism and Function. Progress in lipid research. 2014;0:1-18. doi:10.1016/j.plipres.2014.06.002. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4258547/[↩][↩]
- Svennerholm L. Distribution and fatty acid composition of normal human brain. J Lipid Res. 1968;9:570–579.[↩]
- Akbar M, Calderon F, Wen Z, Kim HY. Docosahexaenoic acid: a positive modulator of Akt signaling in neuronal survival. Proc Natl Acad Sci USA. 2005;102:10858–10863 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1182431/[↩]
- Petursdottir AL, Farr SA, Morley JE, Banks WA, Skuladottir GV. Lipid peroxidation in brain during aging in the senescence-accelerated mouse (SAM) Neurobiol Aging. 2007;28:1170–1178. https://www.ncbi.nlm.nih.gov/pubmed/16846666[↩]
- Cunnane SC, Schneider JA, Tangney C, Tremblay-Mercier J, Fortier M, Bennett DA, et al. Plasma and brain fatty acid profiles in mild cognitive impairment and Alzheimer’s disease. J Alzheimer’s Dis. 2012;29:691–697 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3409580/[↩]
- Ullman L, Mimouni V, Roxx S, Porsolt R, Poisson JP. Brain and hippocampus fatty acid composition in the phospholipid classes of age-related cognitive deficit rats. Prostagl Leukot Essent Fatty Acid. 2001;64:189–195. https://www.ncbi.nlm.nih.gov/pubmed/11334555[↩]
- Corwin J, Dean RL, III, Bartus RT, Rotrosen J, Watkins DL. Behavioral effects of phosphatidylserine in the aged Fischer 344 rats: Amelioration of passive avoidance deficits without changes in psychomotor task performance. Neurobiol Aging. 1985;6:11–15. https://www.ncbi.nlm.nih.gov/pubmed/4000381[↩]
- Lee B, Sur BJ, Han JJ, Shim I, Her S, Lee HJ, et al. Krill phosphatidylserine improves learning and memory in Morris water maze in aged rats. Prog Neuropsychopharmacol Biol Psych. 2010;34:1085–1093.[↩]
- Park HJ, Shim HS, Kim KS, Han JJ, Kim JS, Yu AR, et al. Enhanced learning and memory of normal young rats by repeated oral administration of Krill phosphatidylserine. Nutr Neurosci. 2012;16:47–53.[↩][↩]
- Delwaide PJ, Gyselynck-Mambourg AM, Hurlet A, Ylieff M. Double blind randomized controlled study of phosphatidylserine in senile demented patients. Acta Neurol Scand. 1986;73:136–140. https://www.ncbi.nlm.nih.gov/pubmed/3518329[↩]
- Baumeister J, Barthel T, Geiss KR, Weiss M. Influence of phosphatidylserine on cognitive performance and cortical activity after induced stress. Nutr Neurosci. 2008;11:103–110. https://www.ncbi.nlm.nih.gov/pubmed/18616866[↩]
- Jorissen BL, Brorens F, Van Boxtel MPJ, Riedel WJ. Safety of soy-derived phosphatidylserine in elderly people. Nutr Neurosci. 2002;5:337–343. https://www.ncbi.nlm.nih.gov/pubmed/12385596[↩]
- Richter Y, Herzog Y, Cohen T, Steinhart Y. The effect of phosphatidylserine-containin omega-3 fatty acids on memory abilities in subjects with subjective memory complaints; a pilot study. Clin Intervent Aging. 2010;5:313–316 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2981104/[↩]
- Vakhapova V, Cohen T, Richter Y, Herzog Y, Korczyn AD. Phosphatidylserine containing ω-3 fatty acids may improve memory abilities in non-demented elderly with memory complaints: A double-blind placebo-controlled trial. Dement Geriatr Cogn Disord. 2010;29:467–474. https://www.karger.com/Article/Abstract/310330[↩]
- Kato-Kataoka A, Sakai M, Ebina R, Nonaka C. Soybean derived phosphatidylserine improves memory function of the elderly Japanese subjects with memory complaints. J Clin Biochem Nutr. 2010;47:246–255. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2966935/[↩]
- Leon A, Benvegnú D, Toffano G, Orlando P, Massari P. Effect of brain cortex phospholipids on adenylate-cyclase activity in mouse brain. J Neurochem. 1978;30:23–26.[↩]
- Casamenti F, Mantovani P, Amaducci L, Papeu G. Effect of phosphatidylserine on acetylcholine output from the cerebral cortex of rats. J Neurochem. 1979;32:529–533[↩]
- Casamenti F, Scali C, Papeu G. Phosphatidylserine reverses the age-dependent decrease in cortical acetylcholine release: a microdialysis study. Eur J Pharmacol. 1991;194:11–16.[↩]
- Delwaide PJ, Gyselynck-Mambourg AM, Hurlet A, Ylieff M. Double blind randomized controlled study of phosphatidylserine in senile demented patients. Acta Neurol Scand. 1986;73:136–140.[↩]
- Kim HY. Novel metabolism of docosahexaenoic acid in neural cells. J Biol Chem. 2007;282:18661–18665. http://www.jbc.org/content/282/26/18661.long[↩]