Contents
- What is retinol
- What does retinol do
- Serum retinol concentrations
- Retinol for acne
- Topical retinoids
- What is isotretinoin ?
- What is isotretinoin used for ?
- Contraindications to isotretinoin
- How does isotretinoin work ?
- What is the usual dose of isotretinoin ?
- For how long is isotretinoin taken ?
- Drug interactions with isotretinoin
- What are the side effects and risks of isotretinoin ?
- Cutaneous and mucocutaneous side effects
- Monitoring isotretinoin
- Contraception in females considering isotretinoin
- What happens if a pregnant woman takes isotretinoin ?
- Does acne ever fail to clear on isotretinoin ?
- Can isotretinoin be used again if acne recurs ?
- Special precautions for pilots considering isotretinoin
- Retinoids as Antiaging
- Retinol side effects
What is retinol
Retinol is the alcohol form of vitamin A 1. Vitamin A cannot be synthesized by the body; hence it needs to be supplied to the body. Naturally, vitamin A is present as retinyl esters and beta-carotene. The retinyl esters are converted to retinol before absorption from food in the small intestine and back to retinyl esters for storage in the liver.
Vitamin A is a fat soluble vitamin necessary for health, deficiency of which can cause disorders of vision, skin, bone and immunity. The recommended daily allowance for vitamin A is 300 to 700 μg for children and approximately 700 to 900 μg for adults, amounts which can be provided by a normal diet. Higher doses of vitamin A can be toxic, leading to a constellation of signs and symptoms as well as liver injury, jaundice, enlargement of the liver and spleen, portal hypertension and cirrhosis.
Retinol/vitamin A is generally stored in the liver from where it is mobilized into blood circulation bound to retinol binding protein (RBP) also known as RBP4. Liver exhibits high affinity binding sites for RBP4 2. The stored vitamin A/retinol is mobilized into blood plasma by the enzymes retinyl ester hydrolases 3. The normal concentration of retinol in blood plasma varies between 1.0 and 2.0 μM 4.
The mechanism/s by which retinol executes its function however, remain poorly understood.
Retinol is metabolized into four important products:
- Retinyl esters,
- All-trans retinoic acid,
- 14-hydroxy-4, 14-retro retinol, and
- All-trans 3, 4-didehydroretinol, and its esters.
Retinoids are required for a vast number of biological processes. In particular, they are involved in embryogenesis, reproduction, vision, growth, inflammation, differentiation, proliferation, and apoptosis. Retinal is an essential part of the rhodopsin pigment, necessary for vision 5. Retinoids are found in the keratinocytes in two forms: retinol and retinyl esters – probably the storage form.
Table 1. Recommendations for vitamin A intake by age or population group by the Institute of Medicine 6 and the FAO 7
| US Institute of Medicine | Food and Agriculture Organization of the United Nations (FAO) | ||||
| Life-stage group | EAR, μg RAEs/d | AI or RDA, μg RAEs/d | UL,2 μg REs/d | Mean requirement, μg REs/d | Recommended safe intake, μg REs/d |
| Infants | |||||
| 0–6 mo | — | 400 | 600 | 180 | 375 |
| 7–12 mo | — | 500 | 600 | 190 | 400 |
| Children | |||||
| 1–3 y | 210 | 300 | 600 | 200 | 400 |
| 4–6 y | — | — | — | 200 | 450 |
| 4–8 y | 275 | 400 | 900 | ||
| 7–9 y | — | — | — | 250 | 500 |
| 9–13 y | — | — | — | — | — |
| Male | 445 | 600 | 1700 | — | — |
| Female | 420 | 600 | 1700 | — | — |
| Adolescents aged 10–18 y | 330–400 | 600 | |||
| Adults | |||||
| Females | |||||
| 14–18 y | 485 | 700 | 2800 | — | — |
| ≥19 y | 500 | 700 | 3000 | 270–300 | 500–600 |
| Males | |||||
| 14–18 y | 630 | 900 | 2800 | 300 | 600 |
| ≥19 y | 625 | 900 | 3000 | 300 | 600 |
| Pregnancy | |||||
| 14–18 y | 530 | 750 | 2800 | 370 | 800 |
| 19–50 y | 550 | 770 | 3000 | 370 | 800 |
| Lactation | |||||
| 14–18 y | 885 | 1200 | 2800 | 450 | 850 |
| 19–50 y | 900 | 1300 | 3000 | 450 | 850 |
Studies of the past two decades have shown that retinol is associated with cell differentiation via its most potent metabolite retinoic acid 1. Retinol (vitamin A) executes its function via retinoic acid and regulates the function of >500 genes involved in development and cell differentiation 9. The circulating retinol in blood plasma binds to a 21 kDa retinol binding protein (RBP) also known as RBP4 and thyroxine binding-protein transthyretin (TTR) to form a ternary retinol-RBP-TTR complex in 1:1:1 molar proportion that binds to the target cell via cell surface receptor STRA6 (stimulated by retinoic acid 6) for transport into the target cell 10.
Inside the cytoplasm, retinol/vitamin A binds to a 15 kDa cellular retinol binding protein (CRBP) and is converted into retinoic acid by two sequential oxidation steps that convert first retinol into retinaldehyde and then to retinoic acid. Though the conversion of retinol into retinaldehyde is reversible, the retinoic acid cannot be reduced back to retinol. Retinol is converted into retinaldehyde by retinol dehydrogenases (Rdh10) whereas the enzymes that metabolize retinaldehyde into retinoic acid include retinaldehyde dehydrogenases Ralhd1 (Aldhd1), Ralhd2 (Alhd1A2) and Ralhd3 (Alhd1A3) 11.
Retinoic acid is then transported to the nucleus where it binds to heterodimer receptors, retinoic acid receptor (RAR) and retinoid X receptor (RXR) which belong to the superfamily of ligand-inducible transcriptional regulators that include steroid hormone receptors, thyroid hormone receptors, and vitamin D3 receptors. This complex then binds to the retinoic acid responsive elements (RARE) in the promoter region of the retinoic acid-responsive genes to activate their expression 12.
Figure 1. Retinol
What does retinol do
The importance of retinol (vitamin A) was discovered during World War I and subsequent research showed that its deficiency gives rise to xerosis (dry skin) and follicular hyperkeratosis (skin condition characterized by excessive development of keratin in hair follicles, resulting in rough, cone-shaped, elevated papules) 13. These observations were followed by numerous studies focused on the metabolism and pharmacological action of retinoids in the skin leading to the establishment of retinoic acid treatment for various skin diseases 14.
Vitamin A is required for the regulation of numerous key biological processes including roles in the following:
- vision (proper functioning of the retina),
- growth and differentiation of target tissues,
- maintenance of epithelial surfaces,
- modulation of immune function,
- reproduction and proper functioning of the reproductive organs,
- embryonic growth and development.
Retinol is the precursor for ≥2 essential biologically active molecules: all-trans retinoic acid as the ligand of nuclear receptors, such as RARs 15, and 11-cis-retinal required in the visual cycle 16.
Critical Steps in the Functions of Vitamin A
Vision 16
- 11-cis Retinal binds to opsin to form rhodopsin, which can absorb light within the visible spectrum.
- When 11-cis retinal absorbs a photon, it isomerizes to all-trans retinal through several intermediate species and is rapidly released from opsin.
- The isomerization of 11-cis retinal is the initial step in vision. An excited intermediate of rhodopsin greatly amplifies light-induced hyperpolarization of the rod membrane, resulting in generation of the nerve impulses for vision.
Nuclear receptors/gene regulation
- All-trans retinoic acid is the most biologically relevant metabolite of vitamin A.
- Retinoic acid binds and activates several nuclear receptors, i.e., RARs, retinoid X receptor, and PPARs 17.
- Upon ligand binding, RAR dimerizes with a retinoid X receptor to form a heterodimer, which then initiates gene transcription by binding to the retinoic acid response element in the promoter region of >500 target genes 18.
Anemia
Vitamin A deficiency negatively affects hemoglobin concentrations. Supplementing women with vitamin A and iron enhanced hemoglobin concentrations more than either nutrient alone 19.
Immune Function
- The regulatory roles of vitamin A in mammals include the maintenance of epithelial cell differentiation and immune competence 20.
- Vitamin A supports innate immune function by supporting regeneration of mucosal barriers damaged by infection in children and by enhancing the function of neutrophils, macrophages, and NK cells, as described primarily in rodents 20.
- Vitamin A is required for adaptive immunity and plays a role in the development of T-helper (Th), T-regulatory, and B cells, but more in vivo work is needed 20.
- All-trans retinoic acid is a potent regulator of gene expression, and it controls leukocyte homing and T-regulatory function 21. Its production by cells of the immune system is regulated during an immune response in a manner that is still being elucidated 22.
- In human T lymphocytes, all-trans retinoic acid inhibited the production of cytokines that favor the generation of Th1-type T cells and enhanced the production of cytokines favoring the Th2-type T cells 23.
Cancer
- Meta-analyses of human studies have shown an inverse relation between dietary amounts of vitamin A and various cancers, for example, bladder 24, breast 25, cervical 26, and gastric 27.
- Synthetic retinoids and some naturally occurring retinoids (e.g., all-trans retinoic acid, 9-cis retinoic acid, 13-cis retinoic acid) have been used in clinical studies 28.
- Certain retinoids inhibit the growth of various tumors (e.g., lung, gastrointestinal, breast) and may have chemopreventive and/or chemotherapeutic properties 29.
- All-trans retinoic acid is used as a chemotherapeutic agent to treat acute promyelocytic leukemia, and in the vast majority of these patients this treatment leads to a complete remission 30.
Retinoids, the natural and synthetic analogues of retinol, have been of significant interest as cancer preventive agents since late 1970s 31. They have been used in many clinical trials to assess the role of β-carotene in cancer prevention. Almost 27 years ago, the first phase of β-Carotene and Retinol Efficacy Trial (CARET) among men and women at high risk of lung cancer was stopped early primarily due to an increased risk of lung cancer 32. Similarly α-tocopherol and β-carotene (ATBC) and CARET study found an increased risk of lung cancer among those assigned to active β-carotene treatment 33 or no significant benefit of β-carotene 34. On the other hand, β-carotene alone was found to be associated with an increased risk of aggressive prostate cancer in a nested case-control study 35. The outcome of these trials therefore, led some to question the safety of β-carotene supplementation for cancer prevention.
Diabetes
- Type 1 diabetes has been associated with lower concentrations of serum retinol and its carrier proteins (RBP and transthyretin) in patients 36.
- Type 2 diabetes has a less-clear relation with serum retinol and carrier proteins, with some studies showing no change and others showing reductions in type 2 diabetes 37.
- Vitamin A deficiency and excess have varying and discordant effects on macronutrient metabolism in various tissues and cell types 38.
- Vitamin A is involved in pancreatic development and function: deficiency caused reduced β cell mass in fetal islets 39 and reduced glucagon and insulin secretion from pancreatic α and β cells, respectively 40.
- Retinol Binding Protein secreted by adipose tissue has been implicated as a link between obesity and insulin resistance by interrupting insulin signaling in muscle and increasing hepatic glucose output 41.
- In human macrophages, Retinol Binding Protein may cause insulin resistance by contributing to adipocyte inflammation through proinflammatory cytokine activation, and the mechanism is retinol- and STRA6-independent 42.
- The public health link of vitamin A status to diabetes needs further investigation.
Energy metabolism and obesity
- Evidence in animals exists for a role of vitamin A in maintaining energy metabolism; however, more research is needed.
- Retinoic acid exerts its broad range of biological effects in large part by controlling gene expression. Early in adipogenesis, retinoic acid blocks differentiation, whereas after 48 h of differentiation, it promotes fat cell formation 43.
- Mice lacking retinaldehyde dehydrogenase 1 (Raldh1) resisted diet-induced obesity and insulin resistance. Administration of retinal or an Raldh1 inhibitor to obesity-susceptible mice reduced fat accumulation and increased insulin sensitivity 43.
- In mice with active protein kinase C, retinol supplementation showed that retinol is a metabolic cofactor involved in the regulation of mitochondrial fuel utilization 44.
HIV and pregnancy
- Currently, no conclusive evidence of vitamin A supplementation on vertical HIV transmission exists; therefore, the WHO does not recommend supplementation in HIV-positive pregnant women to reduce the risk of mother-to-child transmission 45, 46.
- Cochrane reviews indicated that vitamin A alone 47 and micronutrient supplementation 48 should not be used as a substitute for recommended antiretroviral medication.
- In infants with mannose-binding lectin-2 variants, vitamin A supplementation to mothers at delivery was associated with a decreased risk of HIV transmission 49.
- Future work is needed on the effect of vitamin A supplementation on HIV transmission from mother to child that accounts for the potential effect of an innate immune deficiency.
Measles
- On the basis of a randomized, placebo-controlled clinical trial in children with measles, along with other clinical research, the WHO recommends that age-appropriate doses of vitamin A be given twice 24 h apart to infants and children with measles in populations in whom vitamin A deficiency may be present to decrease the risk of death from measles 50.
- Prevention of vitamin A deficiency by using periodic, high-dose supplements in communities in which vitamin A deficiency is a public health problem decreases the risk of developing measles in children 6–59 mo of age 51.
Figure 2. Retinoids in dermatological therapy
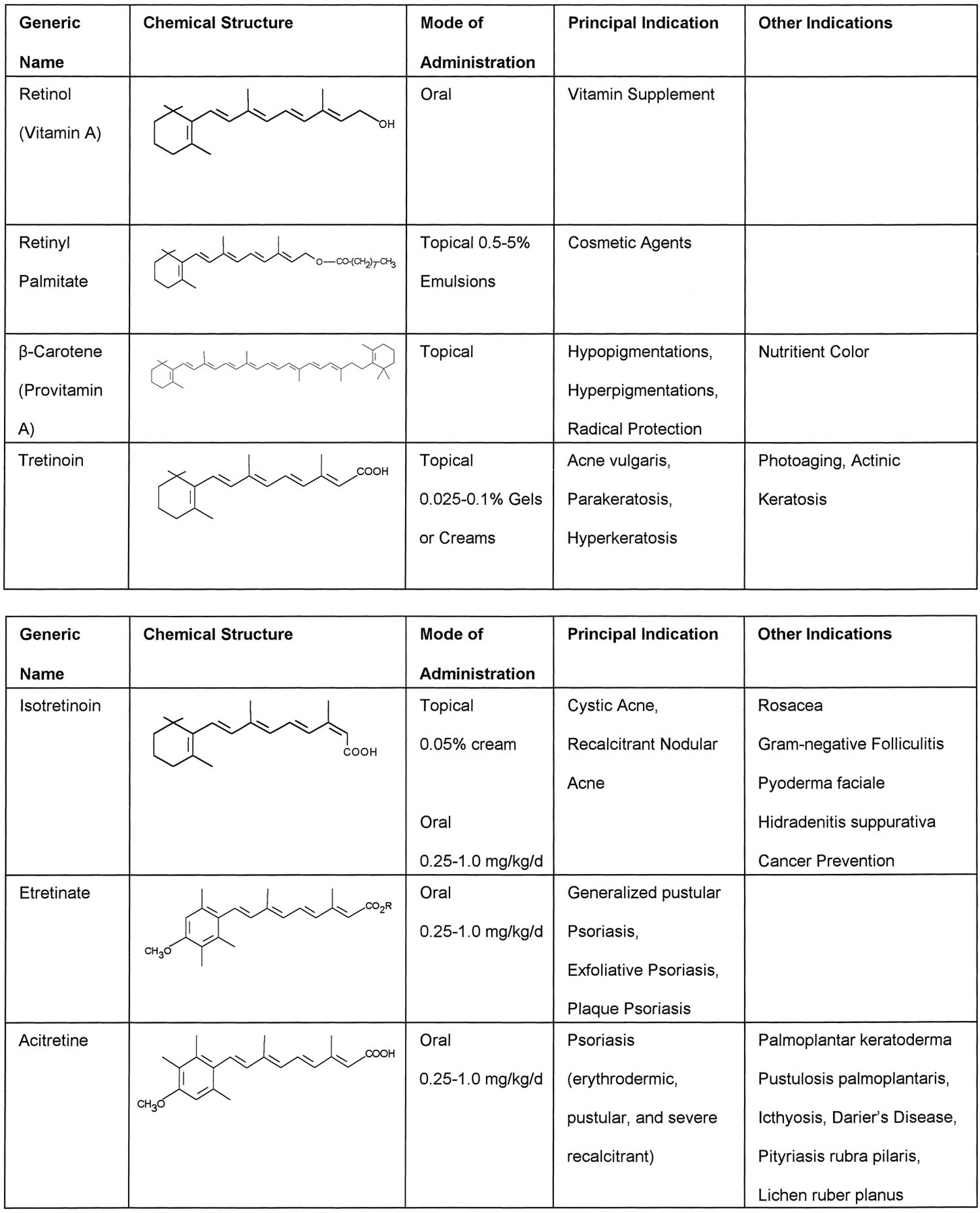
Serum retinol concentrations
Serum retinol concentration measurements by high-performance liquid chromatography are a common method used to assess vitamin A status of populations 52. The current cutoff used to define a severe public health problem for vitamin A deficiency is when 20% of children aged 6–71 months have a serum retinol concentration <0.7 μmol/L 53. Although currently recommended for use by the WHO, serum retinol concentrations should not be used alone to define the degree of public health significance but should be used in conjunction with another biological indicator or when ≥4 of the following risk factors are found in the population being evaluated 53:
- infant mortality rate >75 of 1000 live births and under-5-y mortality rate of >100 of 1000 live births;
- full immunization coverage in <50% of children at 12–23 mo of age;
- <50% prevalence of breastfeeding in 6-mo-old infants;
- median dietary intakes <50% of recommended safe levels of intake among 75% of children 1–6 y of age;
- 2-wk period prevalence of diarrhea of ≥20%;
- measles case fatality rate of ≥1%;
- no formal schooling for ≥50% of women 15–44 y of age; and
- <50% of households with a safe water source (e.g., boiled, treated, filtered, properly stored).
Serum retinol and retinol-binding protein tend to be lower in infants and young children than in adults, even in vitamin A–adequate populations. Therefore, the same cutoff cannot be used for infants <6 mo of age 54. However, the relative concentration to define vitamin A deficiency in infants has not been determined and is a research question that needs to be addressed. Serum retinol concentration tends to increase through middle-age in the US population (particularly in men but also in women >60 y of age) and may represent a continuing, although less pronounced, association of liver vitamin A stores 55.
Retinol-binding protein concentrations do not vary by sex, generally speaking, and the lower serum retinol concentrations in women than in men in middle-age may be due to differences in liver stores or to the higher prevalence of inflammation in women 55. The cutoff used for deficiency in adults varies, and some researchers choose to use 0.7 μmol/L whereas others suggest 1.05 μmol/L 54. The hepatic synthesis of retinol-binding protein is depressed during zinc deficiency 56, which results in lower plasma retinol concentrations that confound measurements in zinc-deficient individuals. Pregnancy lowers serum retinol concentrations through hemodilution.
Serum Retinol Concentration Use in Clinical Application
Serum retinol concentrations are affected by infection status. Retinol-binding protein is a negative acute-phase protein, and thus retinol and retinol-binding protein concentrations decrease during the acute-phase response 57. Therefore, serum retinol has very little utility to diagnose vitamin A deficiency when individuals have an infection. In addition, serum retinol concentrations are homeostatically controlled over a broad range of liver reserves and thus are best used at the population level as detailed above.
Use of serum retinol concentrations in epidemiologic studies
Serum retinol concentrations are determined and have been evaluated in epidemiologic surveys. A recent evaluation of NHANES III data in adults ≥50 y of age determined that serum retinol concentrations that were either <1 μmol/L or >2.8 μmol/L were associated with increased risk of all-cause and cardiovascular or coronary artery disease–related mortality 57. The significance of these findings is not entirely known. In otherwise well-nourished individuals who are not experiencing inflammation, serum retinol concentrations <1 μmol/L may reflect vitamin A deficiency 54. It appears that serum retinol concentrations are increasing over time in well-nourished Americans, which may be associated with increased catabolism 58 due to the fortification of a variety of foods and multivitamin supplement use that increase total body stores over time 59.
Effect of infection or inflammation on serum retinol concentrations
The acute-phase response to either infection or inflammation affects retinol homeostasis. The acute-phase response on micronutrient status markers has been reviewed 60. Currently, concentrations of C-reactive protein (CRP) and α1-acid glycoprotein (AGP) have been used to identify infection stage according to a proposed published paradigm: elevated CRP alone indicates the incubation or early phase of an acute infection, elevated CRP and AGP indicate early convalescence, and elevated AGP only indicates late convalescence. These inflammation stages have been used to correct serum retinol concentrations or to choose lower cutoffs for serum retinol as a marker of deficiency 61. During early convalescence, serum retinol concentrations are shifted further to the left than during late convalescence or incubation 57.
Retinol for acne
Topical retinoids
Topical retinoids are creams, lotions and gels containing one or other of group of medicines derived from Vitamin A. These compounds result in proliferation and reduced keratinisation of skin cells independent of their functions as a vitamin.
Many brand-name creams containing the retinoids retinol and retinaldehyde can be obtained over the counter at pharmacies and supermarkets.
Approved Topical Retinol creams
Naturally occurring retinoids
- All-trans retinol (used in over-the-counter cosmeceuticals)
- All-trans retinoic acid (tretinoin)
- Alitretinoin (US FDA approved for AIDS-related Kaposi’s sarcoma)
Synthesized retinoids
- Adapalene
- Tazarotene
- Bexarotene (US FDA approved for cutaneous lymphoma)
Adapalene gel has received approval from the FDA in the USA for over-the-counter use of acne treatment in patients 12 or older (July 2016).
Adapalene is also available to treat acne in combination with benzoyl peroxide, as Epiduo® gel.
The more potent topical retinoids available on prescription are:
- ReTrieve™ cream (tretinoin)
- Retin-A™ Cream (tretinoin or retinoic acid)
- Retinova™ Cream (tretinoin emollient)
- Isotrex™ Gel (isotretinoin)
- Differin™ Gel, Cream (adapalene)
What are they used for ?
Topical retinoids are effective treatments for mild to moderately severe acne. The effect is often noticeable within a few weeks, but it may take 6 weeks or longer before improvement occurs.
Tretinoin has also been shown to reverse some of the changes due to photo-aging, i.e. sun damage. If used long term, it may reduce some fine wrinkles, freckles, solar comedones (whiteheads and blackheads), and actinic keratoses (tender, dry sun-spots).
They may also be used in bleaching creams to reduce pigmentation in melasma.
Topical retinoids can be applied to any area but are most often used on the face, the neck and the back of hands.
Do topical retinoids have any side effects ?
Topical retinoids can irritate the skin, especially when they are first used. This is more likely in those with sensitive skin, resulting in stinging. Excessive use results in redness, swelling, peeling and blistering in treated areas. It may cause or aggravate eczema, particularly atopic dermatitis.
By peeling off the top layer of skin, they may increase the chance of sunburn. Irritation may also be aggravated by exposure to wind or cold, use of soaps and cleansers, astringents, peeling agents and certain cosmetics.
Some people have reported a flare of acne in the first few weeks of treatment. This usually settles with continued use.
Retinoids taken by mouth may cause birth deformities. Manufacturers recommend that topical retinoids are not used in pregnancy or breastfeeding as negative animal studies are not always predictive of human response.
How to use topical retinoids
Follow these instructions carefully:
- Be cautious if you are using other topical acne treatments – ask your doctor if you should stop these.
- In general, a cream is less iritating than a gel. If there is a choice, start with a lower concentration product.
- Use your topical retinoid on alternate nights at first. If you have sensitive skin, wash it off after an hour or so. If it irritates, apply it less often. If it doesn’t, try every night, and if possible twice daily. In most people, the skin gradually gets used to it.
- To reduce stinging, apply it to dry skin, that is, 30 minutes or longer after washing.
- Apply a tiny amount to all the areas affected, and spread it as far as it will go.
- Don’t get it in your eyes or mouth.
- Apply a sunscreen to exposed skin in the morning.
- Wear your usual make-up if you wish, and use gentle cleansers (avoid soap) and apply non-greasy moisturisers as often as required.
- If you have acne, choose oil-free cosmetics.
- If your skin goes scarlet and peels dramatically even with cautious use, the retinoid may be unsuitable for your sensitive skin.
- Tolerance to topical retinoids often develops over time.
What is isotretinoin ?
Isotretinoin (13-cis retinoic acid) is a vitamin-A derivative (retinoid). The liver naturally makes small quantities of isotretinoin from vitamin-A.
Isotretinoin was developed in the 1950s, but only started being used in the mid 1970s. The original brand names were Accutane® and Roaccutane®, but there are now many generic versions on the market, of varying potency.
What is isotretinoin used for ?
Isotretinoin is a very effective medication for the treatment of acne. Originally licensed for use in severe disease, it is increasingly prescribed for all grades of acne.
Isotretinoin is also useful for other follicular conditions, such as:
- Rosacea
- Seborrhoea
- Hidradenitis suppurativa
- Scalp folliculitis
It is also prescribed for many other skin diseases. Examples include:
- Discoid lupus erythematosus
- Granuloma annulare
- Grover disease
- Sarcoidosis
- Extensive actinic keratoses
- Prevention of squamous cell carcinoma
- As an adjuvant in neuroblastoma
Contraindications to isotretinoin
- Isotretinoin must not be taken in pregnancy, or if there is a significant risk of pregnancy.
- Blood donation by males and females on isotretinoin is not allowed in case the blood is used for a pregnant woman.
Precautions when taking isotretinoin
- Isotretinoin should be used with caution during breastfeeding.
- Commercial pilots may be subject to flying restrictions if they take isotretinoin.
- High dose isotretinoin in very young children has been associated with premature epiphyseal closure, leading to shorter stature (not seen in low dose for the treatment of acne).
How does isotretinoin work ?
In acne, isotretinoin:
- Reduces sebum production
- Shrinks the sebaceous glands
- Reduces follicular occlusion
- Inhibits growth of bacteria
- Has anti-inflammatory properties
What is the usual dose of isotretinoin ?
The range of doses used each day for acne is less than 0.1 to over 1 mg/kg body weight. Some patients may only need a small dose once or twice a week. A course of treatment may be completed in a few months or continue for several years. For acne, some prescribers have targeted a total cumulative dose of 120–140 mg/kg, in the hope of reducing relapse, but the evidence for this remains controversial. The general trend has been to use lower dosages, unrelated to body weight (eg 10 mg/day).
The individual dose prescribed by the dermatologist depends on:
- Prescriber preference
- Patient body weight
- The specific condition being treated
- Severity of the skin condition
- Response to treatment
- Other treatment used at the same time
- Side effects experienced
Isotretinoin is better taken with water or milk after food to help with its absorption. It may be taken on an empty stomach, but absorption may be halved. There is no particular advantage in splitting the dose over the day. A newer formulation (isotretinoin-lidose) can be taken without food.
For how long is isotretinoin taken ?
Most patients should be treated until their skin condition clears and then for a further few months. However, courses have often been restricted to 16–30 weeks (4–7 months) to minimise risk of teratogenicity (risk of congenital abnormalities), and to comply with local regulatory authorities. Isotretinoin may be prescribed for years, usually in low dose or intermittently.
Drug interactions with isotretinoin
Care should be taken with the following medications:
- Vitamin-A (retinoic acid): side effects are cumulative and could be severe. Beta-carotene (provitamin-A) is permitted.
- Tetracyclines (including doxycycline, minocycline): these could increase the risk of headaches and blurred vision due to raised intracranial pressure.
- Warfarin: monitor INR carefully.
What are the side effects and risks of isotretinoin ?
The side effects of isotretinoin are dose dependent; at 1 mg/kg/day, nearly all patients will have some side effects, whereas at 0.1 mg/kg/day, most patients will not. The range and severity of the side effects also depends on personal factors and the disease being treated.
Patients with significant liver or kidney disease, high blood fats, diabetes and depression may be advised not to take isotretinoin or to be on a lower dose than usual and to have regular follow-up visits.
Cutaneous and mucocutaneous side effects
Most of the side effects due to isotretinoin are cutaneous or mucocutaneous and relate to the mode of action of the drug. The most common are listed here. When side effects are troublesome, isotretinoin may need to be withheld or the dose reduced.
- Acne flare-up (particularly if starting dose is >0.5 mg/kg/day)
- Dry lips, cheilitis (sore, cracked or scaly lips) (100% of patients on 1 mg/kg/day)
- Dry skin, fragile skin, eczema/dermatitis (itchy, red patches of skin). Note: atopic eczema may improve.
- Increased sweating
- Dry nostrils, epistaxis (nose bleeds)
- Dry, watery or irritable eyes (especially in contact lens wearers), conjunctivitis, keratitis
- Dry anal mucosa, bleeding at the time of a bowel motion
- Dry genitals, dyspareunia (discomfort during intercourse)
- Facial erythema
- Sunburn on exposure to the sun
- Temporary hair loss
- Brittle nails
- Skin infections: impetigo, acute paronychia, pyogenic granuloma
Treatment of mucocutaneous side effects
- Reduce the dosage (eg to 5–10 mg/day)
- Emollients, lip balm, petroleum jelly, sunscreen, eye drops and lubricants should be applied frequently and liberally when needed
- Dermatitis can be treated with topical steroids
- Take short, cool showers without using soap
- Use mild or diluted shampoo
- Do not start wearing contact lenses for the first time
- Do not have elective eye surgery while on isotretinoin or for 6 months afterwards.
- Do not have ablative laser treatments (eg CO2 resurfacing) while on isotretinoin or for 6 months afterwards. Other laser and light treatments may be performed with care
- Shave rather than wax
- Topical and/or oral antibiotics may be prescribed for impetigo
Other common dose-related side effects of isotretinoin
- Headache
- Myalgia (muscle aches) and arthralgia (joint aches), especially after exercise
- Tiredness (lethargy and drowsiness)
- Disturbed night vision and slow adaptation to the dark. Drivers may experience increased glare from car headlights at night
- Hypertriglyceridaemia (high levels of triglyceride in the blood), usually of no clinical relevance
- Irregular or heavy menstrual periods
Rare side effects of isotretinoin
- Causality of the listed side effects may not have been confirmed
- Severe headache with blurred vision due to raised intracranial pressure
- Mood changes and depression. Note: depression is more often related to the skin condition being treated or other health or psychosocial problems.
- Antidepressant medications may be helpful
- Corneal opacities and cataracts
- High-tone deafness
- Accelerated diffuse interstitial skeletal hyperostosis (bony change)
- Abnormal liver function tests or symptomatic hepatitis
- Diarrhea or bleeding from the bowel
- Pancreatitis
- Allergy to isotretinoin causing liver disease and a febrile illness
Treatment of systemic side effects
- Drink minimal alcohol
- Take paracetamol for headache and for mild aches and pains
- Seek medical attention early, if unwell
Monitoring isotretinoin
- Pregnancy must be excluded before and during treatment with isotretinoin.
In an otherwise healthy individual, blood tests are generally not needed. However, consider the following if using high dose (1 mg/kg/day), prolonged courses (>12 months), or if patients have specific risk factors (eg family history of dyslipidaemia, higher risk of viral hepatitis, etc):
- Cholesterol and triglyceride levels
- Liver function tests
- Blood count
Contraception in females considering isotretinoin
- Isotretinoin must not be taken in pregnancy because of a very high risk of serious congenital abnormalities in the baby. Caution needs to be used during breast-feeding as it enters the breast milk and might affect the baby.
All females who could biologically have a child should take the following precautions during treatment with isotretinoin and for four weeks after the medication has been discontinued:
- Abstinence. The most reliable method of avoiding pregnancy is not to have sex. No method of contraception is completely reliable. “Natural” family planning is particularly risky.
- If sexually active, two reliable methods of contraception should be used. Discuss contraception with your doctor (general practitioner, family planning specialist, gynaecologist or dermatologist). The combined oral contraceptive, IUD (intrauterine device), progesterone implant, or medroxyprogesterone injections may be suitable.
- The low-dose progesterone mini-pill on its own is not recommended.
A prescription for emergency contraception may be available from a medical practitioner (your doctor or family planning clinic) or accredited pharmacy. It prevents 85% of pregnancies if taken within 72 hours of unprotected sexual intercourse.
If contraception fails, termination of pregnancy (an abortion) may be advised if pregnancy arises during treatment with isotretinoin or within a month of discontinuing it.
What happens if a pregnant woman takes isotretinoin ?
Isotretinoin has a very high chance of resulting in a spontaneous miscarriage or a severe birth deformity if a fetus is exposed to it during the first half of pregnancy. The deformities affect the growth of tissues developing at the time of exposure to the drug:
- Cranium (skull and brain)
- Cardiac (heart)
- Eye, ear
- Limbs
No contraceptive precautions are necessary for men
Isotretinoin has no effect on sperm or male fertility and has not been shown to cause birth defects in children fathered by men taking it.
Does acne ever fail to clear on isotretinoin ?
Although isotretinoin is usually very effective for acne, occasionally it responds unexpectedly slowly and incompletely. Poor response is associated with:
- Macrocomedones (large whiteheads)
- Nodules (large, deep inflammatory lesions)
- Secondary infection
- Smoking
- Polycystic ovarian syndrome
- Younger age (<14 years)
Options available to slow responders include:
- Electrocautery of comedones
- Prolonged course of isotretinoin
- Additional treatment with oral antibiotics and oral steroids
Can isotretinoin be used again if acne recurs ?
At least fifty per cent of patients with acne have a long lasting response after a single adequate course of isotretinoin. In others, acne may recur a few months to a few years after the medication has been discontinued. Relapse is more common in females than in males, and in patients >25 years of age. These patients may receive one or more further courses of isotretinoin.
Long-term treatment (>1 year) is often used for patients with:
- Persistent acne
- Seborrhoea
- Rosacea
- Scalp folliculitis
- Skin cancer
Special precautions for pilots considering isotretinoin
Good night vision is important for airline pilots and those flying after dark. Night vision may be affected by isotretinoin. Pilots taking isotretinoin or considering a course of isotretinoin must report to their national aviation authority to discuss how this treatment affects their flying privileges.
Retinoids as Antiaging
Retinoids are among the most common ingredients found in cosmeceuticals. In fact, they are the most studied and have the most data behind them. They consist of natural and synthetic derivatives of vitamin A that reduce hyperpigmentation and inhibit enzymes from breaking down collagen. Many of their cosmeceutical claims are based on data derived from studies on tretinoin and other classes of retinoid drugs. Some key retinoids include retinoic acid (tretinoin), retinol, retinaldehyde.
Over-The-Counter (OTC) Retinoids
- They reduce wrinkles and lentigines.
- Common side-effects include redness, irritation, and an increase in photosensitivity.
- Certain retinoid analogues within the same class of molecules have been shown to provide less irritation, but maintain comparable levels of efficacy 62.
- 3 classes of retinoids exhibit distinct properties:
- Vitamin A metabolites – Trans-retinoic acid, Retinaldehyde, Adapalene, and Tazarotene
- Vitamin A – Retinol
- vitamin A esters – retinyl acetate, retinyl propionate, and retinyl palmitate.
- In randomized, double-blind, placebo-controlled, human studies comparing retinol, retinyl acetate, retinyl propionate and trans-retinoic acid
- 62
- propionate exhibited the highest rating when evaluated for efficacy and non-irritation.
- 0.30% retinyl propionate demonstrated superior reductions in wrinkles, redness and hyper-pigmentation vs. 0.15% retinol.
Retinol (Vitamin A)
Retinol is oxidized into retinaldehyde and then into retinoic acid, the biologically active form of vitamin A. In vivo studies showed that topical retinol had only a modest retinoid-like biologic activity compared with topical retinaldehyde and retinoic acid 63. Two randomized, controlled trials reported significant improvement in fine wrinkles after 12 and 24 weeks of treatment, respectively 64, 65.
- Cosmeceutical retinol, retinaldehyde, retinyl propionate, retinyl palmitate – in many cases, bioavailability and activity are unproven when formulated.
Retinaldehyde
Retinaldehyde is viewed in a large part as an intermediate form during the conversion of retinol to retinoic acid. Studies have shown that it does have activity in human skin 66. Moreover, some studies have reported that this retinoid can produce significant clinical improvement in the appearance of fine and deep wrinkles 67.
Retinoic Acid (Tretinoin)
Retinoic acid is considered by dermatologists to be the anti-aging gold standard.
- Available only through a doctor’s prescription.
There is extensive literature on the use of tretinoin, which is considered to be one of the most potent compounds for treating the signs of aging and/or photodamaged skin, including fine lines, hyperpigmented spots, and wrinkles 63. However, side-effects such as burning and scaling have limited its acceptance. In order to minimize these side-effects, various novel drug delivery systems are being developed 68.
Precautions
- Exercise care when using with other photosensitizing drugs, e.g., tetracyclines and thiazides.
- Sun avoidance or sun protection must be encouraged as tretinoin thins the stratum corneum, and allows greater entry of UV light into the skin. People using tretinoin should avoid unnecessary or prolonged exposure to sunlight, and wear sunscreen and protective clothing.
- Tretinoin can also cause skin irritation and hypo- or hyperpigmentation.
Retinol side effects
Retinol (vitamin A) is a fat soluble retinoid. Retinol (vitamin A) is needed for immunity, visual and dermatological health as well as cell communication and growth. Normal doses of vitamin A are not associated with liver injury or liver test abnormalities, but higher doses (generally more than 40,000 IU daily, ~12,000 μg) can be toxic. In excessive amounts, retinol (vitamin A) can accumulate in the liver and cause a wide array of symptoms. Toxicity is classified as either acute or chronic.
Vitamin A toxicity is also known as hypervitaminosis A.
What causes vitamin A toxicity ?
Acute toxicity
Acute toxicity is caused by a single or a few repeated very high doses – the most common cause of acute vitamin A toxicity is the ingestion (generally accidental) of over 300,000 IU of vitamin A (generally >100 times the Recommended Dietary Allowance [RDA]), arising within days to weeks with a typical symptom complex of severe headache, nausea, vertigo, blurred vision, muscle aches and lack of coordination, followed by skin desquamation and alopecia. Severe overdose can cause increased cerebral spinal fluid pressure, progressive drowsiness and coma.
Chronic toxicity
The most common cause of chronic vitamin A toxicity usually arises 3 months to many years after starting moderately high levels of vitamin A (generally 10 times the Recommended Dietary Allowance) – regular ingestion of over 100,000 IU daily, which is sometimes prescribed for dermatological conditions such as acne. Chronic vitamin A toxicity is marked by dry skin, cheilosis, gingivitis, muscle and joint pains, fatigue, mental dullness, depression and liver test abnormalities. Serum bilirubin is typically only mildly elevated. Serum aminotransferase and alkaline phosphatase levels are variably increased, but usually only 1 to 4 times the upper limit of normal. Serum vitamin A levels are typically, but not invariably elevated. Liver biopsy is diagnostic and shows enlarged, lipid-laden stellate cells with variable degrees of sinusoidal fibrosis. The liver may be hypoechogenic on ultrasound examination and suggest the diagnosis of nonalcoholic fatty liver disease, but the lipid-laden cells found on liver biopsy are not hepatocytes, but rather stellate cells (formerly known as Ito cells or hepatic lipocytes) which contain excess vitamin A.
Chronic, moderately high doses of vitamin A (generally over 1 to 8 years) can lead to portal hypertension with ascites and esophageal varices, even before frank cirrhosis can be shown to be present. While high doses of vitamin A are usually achieved by vitamin A supplements, hypervitaminosis A can also occur with excessive dietary intake of liver, particularly that of carnivores (bears, seals, dogs) or salt-water fish (cod liver oil).
What are the signs and symptoms of vitamin A toxicity ?
Signs and symptoms of acute vitamin A toxicity can include:
- Gastrointestinal: nausea, vomiting, loss of appetite, abdominal pain
- Neurological: dizziness, irritability, drowsiness, increased intercranial pressure due to cerebral oedema, and headache
- Dermatological: rash or desquamation (peeling skin)
- Coma and death
Signs and symptoms of chronic vitamin A toxicity can include:
- Gastrointestinal symptoms: hepatomegaly, splenomegaly
- Neurological symptoms: severe headache, pseudotumour cerebri
- Dermatological symptoms: rash, thin and coarse hair, alopecia of the eyebrows, itch, skin that is dry, rough or cracking, and dry or cracked lips
- Musculoskeletal: weakness, cortical hyperostosis of the bone, arthralgia, easy fractures
In children, signs and symptoms of vitamin A toxicity are:
- Neurological: irritability, drowsiness, delirium, coma, increased intercranial pressure, bulging fontanelles (in infants), psychiatric changes, cerebral oedema
- Ophthamological: bulging eyeballs, swelling of the cortical disc, visual disturbances
- Dermatological: skin discoloration and/or desquamation, itch
It is also important to note that vitamin A is highly teratogenic if taken during pregnancy (especially in the first 8 weeks) if intake exceeds 10,000 IU daily. Birth defects can also be caused by isotretinoin or other oral retinoids, if taken while pregnant.
Vitamin A and teratogenicity
Excessive intake of Vitamin A during pregnancy has been associated with the following birth defects, collectively known as retinoic acid syndrome:
- Encephalitis
- Microcephaly
- Craniofacial malformations (most commonly a cleft palate)
- Cardiovascular malformations (most commonly a transposition of the great vessels)
- Thymus malformation/dysfunction
How is vitamin A toxicity diagnosed ?
Diagnosis of vitamin A toxicity is based on signs and symptoms, patient history, lifestyle habits and use of supplements. There is sometimes a poor correspondence between toxicity and serum retinol levels. However, serum levels can sometimes be between 1,000 and 20,000 (with a normal range being 200 to 800 µg/L). The blood sample must be protected from light.
How is vitamin A toxicity treated ?
Vitamin a toxicity is treated by stopping the use of vitamin A supplements. Generally, signs and symptoms will resolve on their own with 1–4 weeks, depending on their severity. Birth defects caused by vitamin A toxicity during pregnancy are irreversible.
- Retinoic acid biosynthesis and metabolism. Napoli JL. FASEB J. 1996 Jul; 10(9):993-1001. https://www.ncbi.nlm.nih.gov/pubmed/8801182/[↩][↩]
- Kawaguchi R., Yu J., Honda J., Hu J., Whitelegge J., Ping P., Wiita P., Bok D., Sun H. A membrane receptor for retinol binding protein mediates cellular uptake of vitamin A. Science. 2007;315:820–825. doi: 10.1126/science.1136244.[↩]
- Harrison E.H. Mechanisms of digestion and absorption of dietary vitamin A. Annu. Rev. Nutr. 2005;25:87–103. doi: 10.1146/annurev.nutr.25.050304.092614.[↩]
- Goodman D.S. In: Plasma Retinol Binding Protein in the Retinoids. Sporn M.B., Roberts A.B., Goodman D.S., Orlando F.L., editors. Academic Press; Salt Lake, UT, USA: 1984. pp. 41–88.[↩]
- Retinoid metabolism in the skin. Roos TC, Jugert FK, Merk HF, Bickers DR. Pharmacol Rev. 1998 Jun; 50(2):315-33. http://pharmrev.aspetjournals.org/content/50/2/315.long[↩]
- Institute of Medicine, Food and Nutrition Board. Dietary Reference Intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. Washington (DC): National Academies Press; 2001.[↩]
- Joint FAO/WHO Expert Consultation. Vitamin A. In: Vitamin and mineral requirements in human nutrition. 2nd ed. Geneva (Switzerland):WHO/FAO; 2004. p. 17–44.[↩]
- Tanumihardjo SA, Russell RM, Stephensen CB, et al. Biomarkers of Nutrition for Development (BOND)—Vitamin A Review. The Journal of Nutrition. 2016;146(9):1816S-1848S. doi:10.3945/jn.115.229708. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4997277[↩]
- Retinoids and vertebrate development. Gudas LJ. J Biol Chem. 1994 Jun 3; 269(22):15399-402. http://www.jbc.org/content/269/22/15399.long[↩]
- A membrane receptor for retinol binding protein mediates cellular uptake of vitamin A. Kawaguchi R, Yu J, Honda J, Hu J, Whitelegge J, Ping P, Wiita P, Bok D, Sun H. Science. 2007 Feb 9; 315(5813):820-5. https://www.ncbi.nlm.nih.gov/pubmed/17255476/[↩]
- Ocular aldehyde dehydrogenases: protection against ultraviolet damage and maintenance of transparency for vision. Chen Y, Thompson DC, Koppaka V, Jester JV, Vasiliou V. Prog Retin Eye Res. 2013 Mar; 33():28-39. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3570594/[↩]
- Transcriptional activities of retinoic acid receptors. Lefebvre P, Martin PJ, Flajollet S, Dedieu S, Billaut X, Lefebvre B. Vitam Horm. 2005; 70():199-264. https://www.ncbi.nlm.nih.gov/pubmed/15727806/[↩]
- Mukherjee S, Date A, Patravale V, Korting HC, Roeder A, Weindl G. Retinoids in the treatment of skin aging: an overview of clinical efficacy and safety. Clinical Interventions in Aging. 2006;1(4):327-348. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2699641/[↩]
- Runne U, Orfanos CE, Gartmann H. (1973) Perorale applikation zweier derivate der vitamin A-Säure zur internen psoriasis-therapie (13-cis-beta-vitamin A-Säure und vitamin A-säure-äthylamid). Arch Dermatol Res 247:171–180.[↩]
- Colors with functions: elucidating the biochemical and molecular basis of carotenoid metabolism. von Lintig J. Annu Rev Nutr. 2010 Aug 21; 30:35-56.[↩]
- Chemistry of the retinoid (visual) cycle. Kiser PD, Golczak M, Palczewski K. Chem Rev. 2014 Jan 8; 114(1):194-232. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3858459/[↩][↩]
- Al Tanoury Z, Piskunov A, Rochette-Egly C. Vitamin A and retinoid signaling: genomic and nongenomic effects. J Lipid Res 2013;54:1761–75. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3679380/[↩]
- Overview of retinoid metabolism and function. Blomhoff R, Blomhoff HK. J Neurobiol. 2006 Jun; 66(7):606-30. https://www.ncbi.nlm.nih.gov/pubmed/16688755/[↩]
- Suharno D, West CE, Muhilal, Karyadi D, Hautvast JG. Supplementation with vitamin A and iron for nutritional anaemia in pregnant women in West Java, Indonesia. Lancet 1993;342:1325–8.[↩]
- Stephensen CB. Vitamin A, infection, and immune function. Annu Rev Nutr 2001;21:167–92.[↩][↩][↩]
- Pino-Lagos K, Benson MJ, Noelle RJ. Retinoic acid in the immune system. Ann N Y Acad Sci 2008;1143:170–87. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3826166/[↩]
- Raverdeau M, Mills KHG. Modulation of T cell and innate immune responses by retinoic acid. J Immunol 2014;192:2953–8.[↩]
- Dawson HD, Collins G, Pyle R, Key M, Weeraratna A, Deep-Dixit V, Nadal CN, Taub DD. Direct and indirect effects of retinoic acid on human Th2 cytokine and chemokine expression by human T lymphocytes. BMC Immunol 2006;7:27.[↩]
- Tang JE, Wang RJ, Zhong H, Yu B, Chen Y. Vitamin A and risk of bladder cancer: a meta-analysis of epidemiological studies. World J Surg Oncol 2014;12:130.[↩]
- Fulan H, Changxing J, Baina WY, Wencui Z, Chunqing L, Fan W, Dandan L, Dianjun S, Tong W, Da P, et al. Retinol, vitamins A, C, and E and breast cancer risk: a meta-analysis and meta-regression. Cancer Causes Control 2011;22:1383–96.[↩]
- Zhang X, Dai B, Zhang B, Wang Z. Vitamin A and risk of cervical cancer: a meta-analysis. Gynecol Oncol 2012;124:366–73.[↩]
- Wu Y, Ye Y, Shi Y, Li P, Xu J, Chen K, Xu E, Yang J. Association between vitamin A, retinol intake and blood retinol level and gastric cancer risk: a meta-analysis. Clin Nutr 2015;34:620–6.[↩]
- Altucci L, Gronemeyer H. The promise of retinoids to fight against cancer. Nat Rev Cancer 2001;1:181–93.[↩]
- Siddikuzzaman GC, Berlin Grace VM. All trans retinoic acid and cancer. Immunopharmacol Immunotoxicol 2011;33:241–9.[↩]
- Wang Z-Y, Chen Z. Acute promyelocytic leukemia: from highly fatal to highly curable. Blood 2008;111:2505–15.[↩]
- Goodman G.E., Alberts D.S., Meyskens F.L. Retinol, vitamins, and cancer prevention: 25 Years of learning and relearning. J. Clin. Oncol. 2008;26:5495–5496. doi: 10.1200/JCO.2008.19.0884. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2661498/[↩]
- Goodman G.E., Alberts D.S., Ernest D.L., Meyskens F.L. Phase I trial of retinol in cancer patients. J. Clin. Oncol. 1983;1:394–399. https://www.ncbi.nlm.nih.gov/pubmed/6668507[↩]
- Lee I.-M., Cook N.R., Manson J.E., Buring J.E., Hennekens C.H. B carotene supplementation and incidence of cancer and cardiovascular disease: The Women’s Health Study. J. Natl. Cancer Inst. 1999;91:2102–2106. doi: 10.1093/jnci/91.24.2102. https://www.ncbi.nlm.nih.gov/pubmed/10601381[↩]
- Peto R., Doll R., Buckley J.D., Sporn M.B. Can dietary β-carotene materially reduce human cancer rates? Nature. 1981;290:201–208. doi: 10.1038/290201a0. https://www.ncbi.nlm.nih.gov/pubmed/7010181[↩]
- Leitzmann M.F., Chatterjee N., Peters U., Chatterjee N., Wang Y., Albanes D., Gelmann E.P., Friesen M.D., Riboli E., Hayes R.B. Serum lycopene, other carotenoids, and prostate cancer risk: A nested case-control study in the prostate, lung, colorectal, and ovarian cancer screening trial. Cancer Epidemiol. Biomark. Prev. 2007;16:962–968. doi: 10.1158/1055-9965.EPI-06-0861. http://cebp.aacrjournals.org/content/16/5/962.long[↩]
- Martinoli L, Di Felice M, Seghieri G, Ciuti M, De Giorgio LA, Fazzini A, Gori R, Anichini R, Franconi F. Plasma retinol and alpha-tocopherol concentrations in insulin-dependent diabetes mellitus: their relationship to microvascular complications. Int J Vitam Nutr Res 1993;63:87–92.[↩]
- Iqbal S, Naseem I. Role of vitamin A in type 2 diabetes mellitus biology: effects of intervention therapy in a deficient state. Nutrition 2015;31:901–7.[↩]
- Chen W, Chen G. The roles of vitamin A in the regulation of carbohydrate, lipid, and protein metabolism. J Clin Med 2014;3:453–79[↩]
- Zhao S, Li R, Li Y, Chen W, Zhang Y, Chen G. Roles of vitamin A status and retinoids in glucose and fatty acid metabolism. Biochem Cell Biol 2012;90:142–52.[↩]
- Brun P-J, Yang KJ, Lee S-A, Yuen JJ, Blaner WS. Retinoids: potent regulators of metabolism. Biofactors 2013;39:151–63.[↩]
- Graham TE, Kahn BB. Tissue-specific alterations of glucose transport and molecular mechanisms of intertissue communication in obesity and type 2 diabetes. Horm Metab Res 2007;39:717–21.[↩]
- Norseen J, Hosooka T, Hammarstedt A, Yore MM, Kant S, Aryal P, Kiernan UA, Phillips DA, Maruyama H, Kraus BJ, et al. Retinol-binding protein 4 inhibits insulin signaling in adipocytes by inducing proinflammatory cytokines in macrophages through a c-Jun N-terminal kinase- and Toll-like receptor 4-dependent and retinol-independent mechanism. Mol Cell Biol 2012;32:2010–9.[↩]
- Ziouzenkova O, Orasanu G, Sharlach M, Akiyama TE, Berger JP, Viereck J, Hamilton JA, Tang G, Dolnikowski GG, Vogel S, et al. Retinaldehyde represses adipogenesis and diet-induced obesity. Nat Med 2007;13:695–702.[↩][↩]
- Shabrova E, Hoyos B, Vinogradov V, Kim YK, Wassef L, Leitges M, Quadro L, Hammerling U. Retinol as a cofactor for PKCδ-mediated impairment of insulin sensitivity in a mouse model of diet-induced obesity. FASEB J 2016;30:1339–55.[↩]
- WHO. Guideline: vitamin A supplementation in pregnant women. Geneva (Switzerland): WHO; 2011. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3648723[↩]
- WHO. Guideline: vitamin A supplementation in pregnancy for reducing the risk of mother-to-child transmission of HIV. Geneva (Switzerland): WHO; 2011. https://www.ncbi.nlm.nih.gov/books/NBK185128/[↩]
- Wiysonge CS, Shey M, Kongnyuy EJ, Sterne JA, Brocklehurst P. Vitamin A supplementation for reducing the risk of mother-to-child transmission of HIV infection. Cochrane Database Syst Rev 2011;1:CD003648.[↩]
- Siegfried N, Irlam JH, Visser ME, Rollins NN. Micronutrient supplementation in pregnant women with HIV infection. Cochrane Database Syst Rev 2012;3:CD009755.[↩]
- Kuhn L, Coutsoudis A, Trabattoni D, Archary D, Rossi T, Segat L, Clerici M, Crovella S. Synergy between mannose-binding lectin gene polymorphisms and supplementation with vitamin A influences susceptibility to HIV infection in infants born to HIV-positive mothers. Am J Clin Nutr 2006;84:610–5.[↩]
- WHO. Measles Factsheet, 2016. http://www.who.int/mediacentre/factsheets/fs286/en/[↩]
- Mayo-Wilson E, Imdad A, Herzer K, Yakoob MY, Bhutta ZA. Vitamin A supplements for preventing mortality, illness, and blindness in children aged under 5: systematic review and meta-analysis. BMJ 2011;343:d5094. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3162042/ [↩]
- The use and interpretation of serum retinol distributions in evaluating the public health impact of vitamin A programmes. Palmer AC, West KP Jr, Dalmiya N, Schultink W. Public Health Nutr. 2012 Jul; 15(7):1201-15.[↩]
- WHO. Serum retinol concentrations for determining the prevalence of vitamin A deficiency in populations [Internet]. Geneva (Switzerland); WHO; 2011. http://apps.who.int/iris/bitstream/10665/85859/4/WHO_NMH_NHD_MNM_11.3_eng.pdf[↩][↩]
- de Pee S, Dary O. Biochemical indicators of vitamin A deficiency: serum retinol and serum retinol binding protein. J Nutr 2002;132(9, Suppl):2895S–901S.[↩][↩][↩]
- Stephensen CB, Gildengorin G. Serum retinol, the acute phase response, and the apparent misclassification of vitamin A status in the third National Health and Nutrition Examination Survey. Am J Clin Nutr 2000;72:1170–8.[↩][↩]
- Smith JE, Brown ED, Smith JC Jr. The effect of zinc deficiency on the metabolism of retinol binding protein in the rat. J Lab Clin Med 1974;84:692–7.[↩]
- Suri DJ, Tanumihardjo JP, Gannon BM, Pinkaew S, Kaliwile C, Chileshe J, Tanumihardjo SA. Serum retinol concentrations demonstrate high specificity after correcting for inflammation but questionable sensitivity compared with liver stores calculated from isotope dilution in determining vitamin A deficiency in Thai and Zambian children. Am J Clin Nutr 2015;102:1259–65.[↩][↩][↩]
- Green MH, Green JB. Vitamin A intake and status influence retinol balance, utilization and dynamics in rats. J Nutr 1994;124:2477–85.[↩]
- Tanumihardjo SA. Vitamin A fortification efforts require accurate monitoring of population vitamin A status to prevent excessive intakes. Procedia Chem 2015;14:398–407.[↩]
- Raiten DJ, Sakr Ashour FA, Ross AC, Meydani SN, Dawson HD, Stephensen CB, Brabin BJ, Suchdev P, van Ommen B.; INSPIRE Consultative Group. Inflammation and nutritional science for programs/policies and interpretation of research evidence (INSPIRE). J Nutr 2015;145(Suppl):1039S–108S. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4448820[↩]
- Thurnham D, McCabe GP. Influence of infection and inflammation on biomarkers of nutritional status with an emphasis on vitamin A and iron. WHO report: priorities in the assessment of vitamin A and iron status in populations. Geneva (Switzerland): WHO;2012. http://www.who.int/nutrition/publications/micronutrients/background_paper4_report_assessment_vitAandIron_status.pdf[↩]
- Bissett DL. Topical retinyl priopionate – clinical comparison: a low irritation retinoid for diminishing wrinkles and 5. hyperpigmentation in photoaged human skin. Presented at: The 60th Annual Meeting of the American Academy of Dermatology; New Orleans, LA; February 22-27, 2002.[↩][↩]
- Sorg O, Antille C, Kaya G, et al. Retinoids in cosmeceuticals. Dermatol Ther 19(5):289-96;2006 Sep-Oct[↩][↩]
- Piérard-Franchimont C, Castelli D, Van Cromphaut IV, et al. Tensile properties and contours of aging facial skin. A controlled double blind comparative study of the effects of retinol, melibiose-lactose and their association. Skin Res Technol 4:237-43,1998.[↩]
- Kafi R, Kwak HS, Schumacher WE, et al. Improvement of naturally aged skin with vitamin A (retinol). Arch Dermatol 143(5):606-12;2007 May.[↩]
- Oblong JE, Bissett DL. Retinoids. In: Draelos ZD, ed. Cosmeceuticals. Philadelphia: Elsevier Saunders pp 36-42;2005.[↩]
- Creidi P, Vienne MP, Onchonisky S, et al. Profilometric evaluation of photodamage after topical retinaldehyde and retinoic acid treatment. J Am Acad Dermatol 39(6):960-5; 1998 Dec.[↩]
- Mukherjee S, Date A, Patravale V, et al. Retinoides in the treatment of skin aging: an overview of clinical efficacy and safety. Clin Interv Aging 1(4):327-48;2006[↩]