Contents
What is septic arthritis
Septic arthritis (also called infectious arthritis) is painful infection of a joint due to a bacterial, viral or fungal infection 1. The Septic arthritis infection can come from germs that travel through your bloodstream from another part of your body. Septic arthritis can also occur when a penetrating injury delivers germs directly into the joint.
The general estimated incidence of septic arthritis (infectious arthritis) in industrialized countries is about 6 -29 cases per 100,000 persons per year, with the highest rates being found in those under 15 and over 55 years old 2.
The most common route of entry into the joint is hematogenous (bloodstream) spread during bacteremia (presence of bacteria in the blood) 3. Pathogens may also enter through direct inoculation (e.g., arthrocentesis, arthroscopy, trauma) or contiguous spread from local infections (e.g., osteomyelitis, septic bursitis, abscess) 4. Once in the joint, microorganisms are deposited in the synovial membrane, causing an acute inflammatory response 5. Inflammatory mediators and pressure from large effusions lead to the destruction of joint cartilage and bone loss 6.
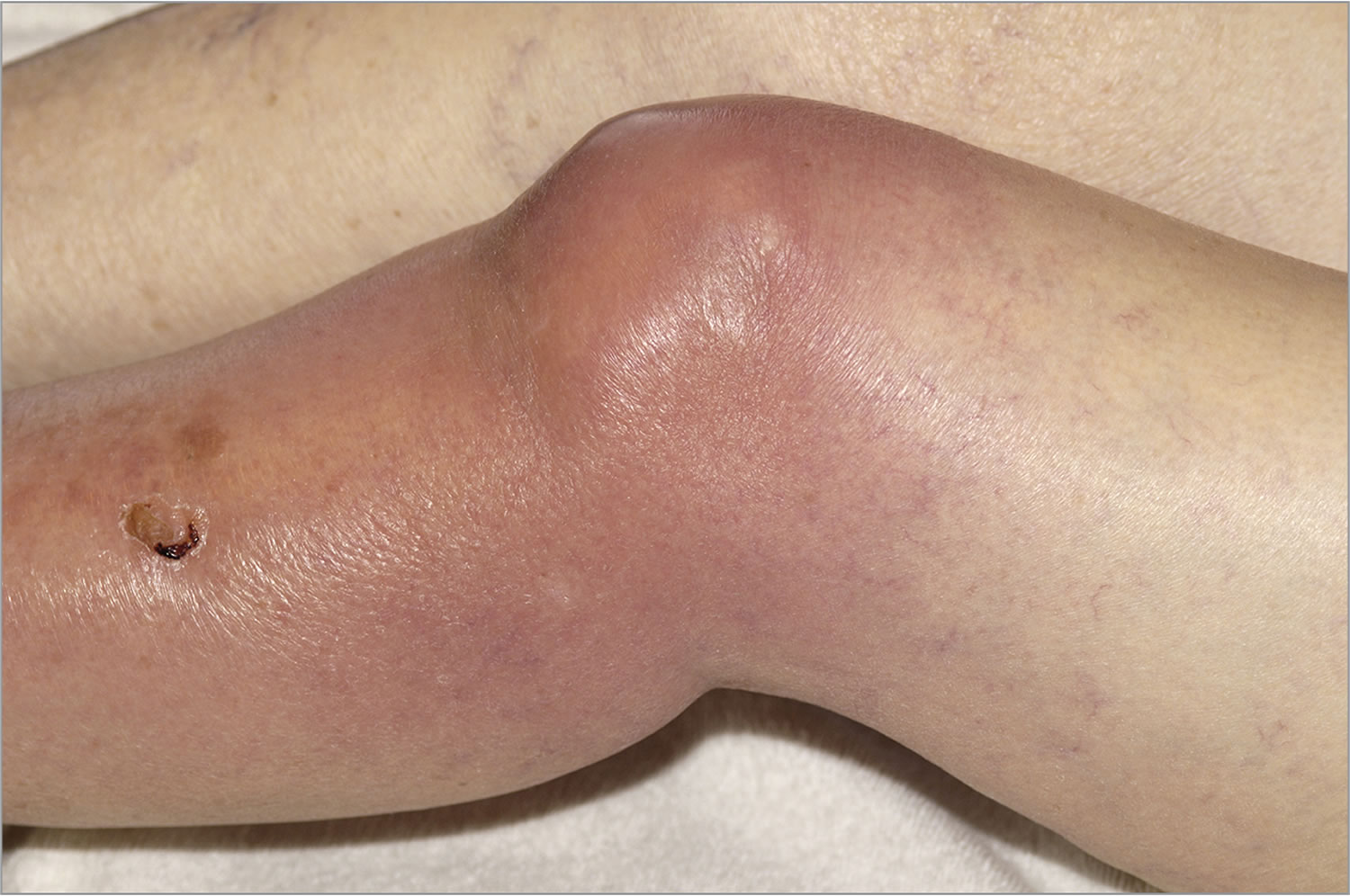
Infants and older adults are most likely to develop septic arthritis. Knees are most commonly affected, but septic arthritis also can affect hips, shoulders and other joints. The infection can quickly and severely damage the cartilage and bone within the joint, so prompt treatment is crucial.
Treatment involves draining the joint with a needle or surgically. Antibiotics also are usually needed to treat the infection.
- Failure to initiate appropriate antibiotic therapy within the first 24 to 48 hours of onset can cause subchondral bone loss and permanent joint dysfunction 7.
Another type of infectious arthritis (septic arthritis) is reactive arthritis. The reaction is to an infection somewhere else in your body. The joint is usually the knee, ankle or toe. Sometimes, reactive arthritis is set off by an infection in the bladder, or in the urethra, which carries urine out of the body. In women, an infection in the vagina can cause the reaction. For both men and women, it can start with bacteria passed on during sex. Another form of reactive arthritis starts with eating food or handling something that has bacteria on it.
Prosthetic Joint Infections
Prosthetic joint infections occur in 0.86 to 1.1 percent of knee arthroplasties 8 and in 0.3 to 1.7 percent of hip arthroplasties 9. These infections may result in failure of the joint replacement. In prosthetic joint infections, bacterial adherence to prosthetic surfaces forms biofilms, which can lead to increased resistance to the host’s immune system and to antimicrobials 10.
Prosthetic joint infections are usually caused by gram-positive cocci, including coagulase-negative staphylococci and Staph aureus 11. Other organisms, such as gram-negative bacilli 12 and mycobacteria 13, have also been implicated. Polymicrobial infections after hip and knee arthroplasties have been reported in up to 20 percent of patients 14. Risk factors for the development of prosthetic joint infections include previous fracture, seropositive rheumatoid arthritis, high body mass index, revision arthroplasty, and surgical site infections 15.
A key distinction in the diagnostic workup of patients with a prosthetic joint infection is that the intraarticular WBC (white blood cell) cutoff values for infection may be as low as 1,100 per mm3 (1.10 × 109 per L), with a neutrophil differential of greater than 64 percent 16. This low WBC count, combined with occasionally more indolent clinical presentations, can make diagnosis problematic. Diagnostic imaging modalities include fluorodeoxyglucose positron emission tomography and combined leukocyte-marrow imaging 17. Antimicrobial treatment must be effective against surface-adhering, biofilm-producing bacteria 18. Debridement, exchange, or permanent removal of the prosthesis may be necessary, depending on the clinical scenario 19. In some persons, long-term suppressive antimicrobial therapy may be warranted 18. Research is being performed on the development of antibiofilm technology to reduce the incidence of prosthetic joint infections 10.
The use of prophylactic antibiotics for invasive dental, genitourinary, gastrointestinal, and other invasive procedures within the first two years after prosthetic joint implantation is controversial. The American Academy of Orthopaedic Surgeons recommends that physicians strongly consider antibiotic prophylaxis in patients with one or more risk factors (i.e., comorbidities, immunosuppression, and previous infection) 20. However, a more recent prospective case-control study does not support this recommendation, showing no change in the incidence of prosthetic joint infections with antibiotic prophylaxis 21. The decision to use prophylactic antibiotics should be made on a case-by-case basis with input from the orthopedic surgeon and with consideration of underlying comorbidities.
Outlook (Prognosis) of septic arthritis
Before antibiotics were available, two-thirds of patients died from septic arthritis 22. Current mortality rates of bacterial arthritis range from 10 to 20 percent, depending on the presence of comorbid conditions, such as older age, coexisting renal or cardiac disease, and concurrent immunosuppression 23. Factors associated with death include age 65 years or older, and infection in the shoulder, elbow, or at multiple sites 23.
After completing antimicrobial therapy, patients with Staph aureus septic arthritis regain 46 to 50 percent of their baseline joint function 24. In contrast, adults with pneumococcal septic arthritis who survive infection (the mortality rate is approximately 20 percent) will return to 95 percent of their baseline joint function after completing antimicrobial therapy 4. Morbidity (e.g., amputation, arthrodesis, prosthetic surgery, severe functional deterioration) occurs in one-third of patients with bacterial arthritis, usually affecting older patients, those with preexisting joint disease, and those with synthetic intraarticular material 23.
Septic arthritis causes
Septic arthritis develops when bacteria or other tiny disease-causing organisms (virus & fungi) spread through the blood to a joint. Septic arthritis can develop when an infection, such as a skin infection or urinary tract infection, spreads through your bloodstream to a joint. Less commonly, a puncture wound, drug injection, or surgery in or near a joint can give the microorganisms entry into the joint space. Joints that are commonly affected are the knee and hip.
The lining of your joints (synovium) has little ability to protect itself from infection. Your body’s reaction to the infection — including inflammation that can increase pressure and reduce blood flow within the joint — contributes to the damage.
Clinical presentations can be broadly grouped into three categories: nongonococcal, gonococcal, and other (e.g., Lyme disease, mycobacterial, fungal).
- Most cases of acute septic arthritis are caused by staphylococcus (40 percent) or streptococcus bacteria (28 percent) 25.
- Bacterial infection with Staphylococcus aureus (staph) is the most common cause. Staph commonly lives on even healthy skin.
- Other microorganisms include Gram-negative bacilli (19 percent), mycobacteria (8 percent), gram-negative cocci (3 percent), gram-positive bacilli (1 percent), and anaerobes (1 percent) 25.
- The most common causative organism in both children and adult septic arthritis is Staphylococcus aureus 26. Staphylococcus aureus (staph) is the primary cause of bacterial arthritis in 40% of cases from England and Wales, 56% of cases from France and 37% of cases from tropical Australia 27.
- Streptococci from groups A, B, C and G are also commonly isolated from septic arthritis in immunocompromised hosts or in patients with severe gastrointestinal or genitourinary infections 28. Streptococcus pneumoniae, Escherichia coli, Proteus sp., Salmonella sp., Serratia marcescens, and Neisseria sp. have also been reported as causal agents of septic arthritis 2. It has been estimated that no causative agent is identified in around half of the patients because the severity of this pathology requires a prompt therapy, even before the isolation of the infectious agent.
Septic arthritis may be seen at any age. In children, it occurs most often in those younger than 3 years. The hip is often the site of infection in infants. Most cases are caused by the bacteria group B streptococcus. Another common cause is Haemophilus influenza, especially if the child has not been vaccinated for this bacteria.
- Chronic septic arthritis (which is less common) is caused by organisms including Mycobacterium tuberculosis and Candida albicans.
Septic arthritis can also be due to the bacteria that cause gonorrhea [a common sexually transmitted infection 29], which is caused by the bacteria Neisseria gonorrhoeae. Gonococcal arthritis is a complication of gonorrhea. Gonococcal arthritis affects women more often than men. It is most common among sexually active teen girls. Any type of sex can spread gonorrhea. You can get it through contact with the mouth, vagina, penis, or anus. Gonorrhea is the second most commonly reported communicable disease. Approximately 330,000 cases occur in the US each year.
The following conditions increase your risk for septic arthritis:
- Artificial joint implants
- Existing joint problems. Chronic diseases and conditions that affect your joints — such as osteoarthritis, gout, rheumatoid arthritis or lupus — can increase your risk of septic arthritis, as can an artificial joint, previous joint surgery and joint injury.
- Taking medications for rheumatoid arthritis. People with rheumatoid arthritis have a further increase in risk because of medications they take that can suppress the immune system, making infections more likely to occur. Diagnosing septic arthritis in people with rheumatoid arthritis is difficult because many of the signs and symptoms are similar.
- Bacterial infection somewhere else in your body
- Presence of bacteria in your blood
- Chronic illness or disease (such as diabetes, rheumatoid arthritis, and sickle cell disease)
- Intravenous (IV) or injection drug use
- Medicines that suppress your immune system
- Recent joint injury
- Recent joint arthroscopy or other surgery
- Skin fragility. Skin that breaks easily and heals poorly can give bacteria access to your body. Skin conditions such as psoriasis and eczema increase your risk of septic arthritis, as do infected skin wounds. People who regularly inject drugs also have a higher risk of infection at the site of injection.
- Weak immune system. People with a weak immune system are at greater risk of septic arthritis. This includes people with diabetes, kidney and liver problems, and those taking drugs that suppress their immune systems.
- Joint trauma. Animal bites, puncture woods or cuts over a joint can put you at risk of septic arthritis.
Risk Factors for Septic Arthritis
Contiguous spread |
Skin infection, cutaneous ulcers |
Direct inoculation |
Previous intraarticular injection |
Prosthetic joint: early and delayed |
Recent joint surgery |
Hematogenous spread |
Diabetes mellitus |
Human immunodeficiency virus infection |
Hematogenous spread |
Immunosuppressive medication |
Intravenous drug abuse |
Osteoarthritis |
Other cause of sepsis |
Prosthetic joint: late |
Rheumatoid arthritis |
Sexual activity (specifically for gonococcal arthritis) |
Other factors |
Age older than 80 years |
The most important risk factor for septic arthritis is preexisting joint pathologies, especially rheumatoid arthritis or prosthetic joint surgery. In these patients, septic arthritis incidence increases to 70 per 100,000 persons 30. Septic arthritis is generally considered a secondary infection, that is, the bacterium escapes from the bloodstream and enters the surrounding tissues. A number of strategies such as endothelial attachment, transcytosis, paracytosis and bacterial transportation by professional phagocytes have been described as putative mechanisms that allow the infectious agent to disseminate from the blood to the joint or other tissues 31.
Having a combination of these condition puts you at greater risk of getting septic arthritis than having just one risk factor does.
Nongonococcal arthritis
Septic arthritis is caused by nongonococcal pathogens in more than 80 percent of patients 5. Nongonococcal arthritis often affects older persons, is acute in nature, and is monoarticular in more than 80 percent of patients 32. Synovial fluid cultures are positive in more than 90 percent of patients with nongonococcal arthritis, as opposed to blood cultures, which are positive in only 50 percent of patients 25.
Gram-positive staphylococci and streptococci are the causative agents in the majority of bacterial arthritis cases in which an organism is identified 4 and are associated with drug abuse, cellulitis, abscesses, endocarditis, and chronic osteomyelitis 33. Staphylococcus aureus is the organism most commonly found in patients with septic arthritis in the United States and other developed countries 34 and Streptococcus species is the next most common 35. The presence of methicillin-resistant S. aureus (MRSA) is an emerging clinical problem. Although data are mostly limited to case reports, the incidence of methicillin-resistant S. aureus (MRSA) ranges between 5 and 25 percent of bacterial arthritis cases, and tends to affect older persons, involve the shoulder, and the health care–associated methicillin-resistant S. aureus (MRSA) strain 36.
Gram-negative bacilli represent approximately 14 to 19 percent of septic arthritis cases12,18 and are associated with invasive urinary tract infections, intravenous drug use, older age, compromised immune system, and skin infections 33. The two most common gram-negative organisms detected in adults are Pseudomonas aeruginosa and Escherichia coli 37. Historically, Haemophilus influenzae infection has occurred more often in children 38, although this may be tempered by widespread H. influenzae type b vaccination 39.
Gonococcal arthritis
People with disseminated Neisseria gonorrhoeae infection are usually young, healthy, and sexually active 25. Disseminated gonococcal infection may have various clinical musculoskeletal presentations, with or without associated dermatitis. Patients typically display a migratory pattern of arthralgias, tenosynovial inflammation, or nonerosive arthritis 25. Blood cultures are seldom positive and synovial fluid cultures are variable, with a positive result in only 25 to 70 percent of patients with gonococcal arthritis 40. When a disseminated gonococcal infection is suspected, cultures should be taken from potentially infected mucosal sites (e.g., urethra, rectum, pharynx, cervix) 41. PCR testing has a sensitivity of 76 percent and a specificity of 96 percent for N. gonorrhoeae, and may be useful in patients with culture-negative disease if the clinical scenario is unclear or similar to a reactive arthritis 42.
Other Infections
Fungal arthritis usually has an insidious onset and indolent course 43. Synovial fluid cultures or biopsy can confirm the diagnosis.
Mycobacterial infectious arthritis is also indolent, which can cause a considerable delay in diagnosis, although joint damage does not occur as rapidly as it does in bacterial infections 44. Articular Mycobacterium tuberculosis infection typically affects the hip or knee; is usually caused by reactivation from past dissemination; and may occur without other manifestations of active tuberculosis 44. Synovial fluid culture is positive in 80 percent of patients with M. tuberculosis infection.34 Acid-fast smears are not helpful and are often negative 44. Histology is not specific because it may mimic other granulomatous diseases, although synovial biopsy will be positive for M. tuberculosis in about 95 percent of cases 45.
Borrelia burgdorferi infection initially causes viral-like migratory arthralgias of Lyme disease. Late disease is characterized by an intermittent oligoarthritis that usually involves the knee or other large joints 46. The diagnosis of Lyme arthritis can be made with a two-step serologic testing process involving enzyme-linked immunosorbent assay, followed by confirmation with a Western blot or immunoblot test 47. B. burgdorferi cannot be cultured from synovial fluid 48; however, PCR testing is positive in 85 percent of patients with Lyme arthritis, making it a confirmatory test 49. It should be noted that PCR testing cannot distinguish live from dead organisms 48.
Complications of septic arthritis
If treatment is delayed, septic arthritis can lead to joint degeneration and permanent damage.
Septic arthritis symptoms
Symptoms usually come on quickly. Septic arthritis typically causes extreme joint pain and difficulty using the affected joint (usually in just one joint), which gets worse with movement. The joint could be swollen, red and warm, and you might have a fever.
Symptoms in newborns or infants:
- Crying when infected joint is moved (for example, during diaper changes)
- Fever
- Not able to move the limb with the infected joint (pseudoparalysis)
- Fussiness
Symptoms in children and adults:
- Not able to move the limb with the infected joint (pseudoparalysis)
- Severe joint pain
- Joint swelling
- Joint redness
- Fever
Chills may occur, but are uncommon.
Diagnosis of septic arthritis
The health care provider will examine the joint and ask about the symptoms.
The following tests typically help diagnose septic arthritis:
- Joint fluid analysis. Infections can alter the color, consistency, volume and makeup of the fluid within your joints. A sample of this fluid can be withdrawn from your affected joint with a needle. Laboratory tests can determine what organism is causing your infection, so your doctor will know which medications to prescribe.
- Blood tests. These can determine if there are signs of infection in your blood. A sample of your blood is removed from a vein with a needle.
- Imaging tests. X-rays and other imaging tests of the affected joint can assess damage to the joint.
There are no data on imaging studies that are pathognomonic for acute septic arthritis. Plain films establish a baseline and may detect fractures, chondrocalcinosis, or inflammatory arthritis. Ultrasonography is more sensitive for detecting effusions, particularly in difficult-to-examine joints, such as the hip 50. Magnetic resonance imaging findings that suggest an acute intraarticular infection include the combination of bone erosions with marrow edema 51. Imaging may allow guided arthrocentesis, particularly in difficult-to-examine joints (e.g., hip, sacroiliac, costochondral).
Septic arthritis treatment
Antibiotics are used to treat the infection.
Removing the infected joint fluid is crucial. Drainage methods include:
- Needle. In some cases, your doctor can withdraw the infected fluid with a needle inserted into the joint space.
- Scope procedure. In arthroscopy, a flexible tube with a video camera at its tip is placed in your joint through a small incision. Suction and drainage tubes are then inserted through small incisions around your joint.
- Open surgery. Some joints, such as the hip, are more difficult to drain with a needle or arthroscopy, so an open surgical procedure might be necessary.
Resting, raising the joint above heart level, and using cool compresses may help relieve pain. After the joint starts to heal, exercising it can help speed recovery.
Antibiotics
To select the most effective medication, your doctor must identify the microbe causing your infection. Antibiotics are usually given through a vein in your arm at first. Later, you may be able to switch to oral antibiotics.
Typically, treatment lasts from two to six weeks. Antibiotics carry a risk of side effects, including nausea, vomiting and diarrhea. Allergic reactions also can occur. Ask your doctor about what side effects to expect from your medication.
- Septic arthritis. https://medlineplus.gov/ency/article/000430.htm[↩]
- Nade S: Septic arthritis. Best Pract Res Clin Rheumatol 2003, 17(2):183–200. 10.1016/S1521-6942(02)00106-7[↩][↩]
- Margaretten ME, Kohlwes J, Moore D, Bent S. Does this adult patient have septic arthritis? JAMA. 2007;297(13):1478–1488.[↩]
- Ross JJ, Saltzman CL, Carling P, Shapiro DS. Pneumococcal septic arthritis: review of 190 cases. Clin Infect Dis. 2003;36(3):319–327.[↩][↩][↩]
- Goldenberg DL. Septic arthritis. Lancet. 1998;351(9097):197–202.[↩][↩]
- Smith JW, Piercy EA. Infectious arthritis. Clin Infect Dis. 1995;20(2):225–230.[↩]
- Mathews CJ, Weston VC, Jones A, Field M, Coakley G. Bacterial septic arthritis in adults. Lancet. 2010;375(9717):846–855.[↩]
- Phillips JE, Crane TP, Noy M, Elliott TS, Grimer RJ. The incidence of deep prosthetic infections in a specialist orthopaedic hospital: a 15-year prospective survey. J Bone Joint Surg Br. 2006;88(7):943–948.[↩]
- Choong PF, Dowsey MM, Carr D, Daffy J, Stanley P. Risk factors associated with acute hip prosthetic joint infections and outcome of treatment with a rifampin-based regimen. Acta Orthop. 2007;78(6):755–765.[↩]
- Trampuz A, Osmon DR, Hanssen AD, Steckelberg JM, Patel R. Molecular and antibiofilm approaches to prosthetic joint infection. Clin Orthop Relat Res. . 2003;(414):69–88.[↩][↩]
- Peersman G, Laskin R, Davis J, Peterson M. Infection in total knee replacement: a retrospective review of 6489 total knee replacements. Clin Orthop Relat Res. 2001;(392):15–23.[↩]
- Berbari EF, Hanssen AD, Duffy MC, et al. Risk factors for prosthetic joint infection: case-control study. Clin Infect Dis. 1998;27(5):1247–1254.[↩]
- Eid AJ, Berbari EF, Sia IG, Wengenack NL, Osmon DR, Razonable RR. Prosthetic joint infection due to rapidly growing mycobacteria: report of 8 cases and review of the literature. Clin Infect Dis. 2007;45(6):687–694.[↩]
- Marculescu CE, Cantey JR. Polymicrobial prosthetic joint infections: risk factors and outcome. Clin Orthop Relat Res. 2008;466(6):1397–1404.[↩]
- Jämsen E, Huhtala H, Puolakka T, Moilanen T. Risk factors for infection after knee arthroplasty. A register-based analysis of 43,149 cases. J Bone Joint Surg Am. 2009;91(1):38–47.[↩]
- Ghanem E, Parvizi J, Burnett RS, et al. Cell count and differential of aspirated fluid in the diagnosis of infection at the site of total knee arthroplasty. J Bone Joint Surg Am. 2008;90(8):1637–1643.[↩]
- Love C, Marwin SE, Palestro CJ. Nuclear medicine and the infected joint replacement. Semin Nucl Med. 2009;39(1):66–78.[↩]
- Zimmerli W, Trampuz A, Ochsner PE. Prosthetic-joint infections. N Engl J Med. 2004;351(16):1645–1654.[↩][↩]
- Zimmerli W. Infection and musculoskeletal conditions: prosthetic-joint-associated infections. Best Pract Res Clin Rheumatol. 2006;20(6):1045–1063.[↩]
- American Academy of Orthopaedic Surgeons. Information statement: antibiotic prophylaxis for bacteremia in patients with joint replacements. http://www.aaos.org/about/papers/advistmt/1033.asp. Accessed December 5, 2010.[↩]
- Berbari EF, Osmon DR, Carr A, et al. Dental procedures as risk factors for prosthetic hip or knee infection: a hospital-based prospective case-control study [published correction appears in Clin Infect Dis. 2010;50(6):944]. Clin Infect Dis. 2010;50(1):8–16.[↩]
- Dickie AS. Current concepts in the management of infections in bones and joints. Drugs. 1986;32(5):458–475.[↩]
- Kaandorp CJ, Krijnen P, Moens HJ, Habbema JD, van Schaardenburg D. The outcome of bacterial arthritis: a prospective community-based study. Arthritis Rheum. 1997;40(5):884–892.[↩][↩][↩]
- Weston VC, Jones AC, Bradbury N, Fawthrop F, Doherty M. Clinical features and outcome of septic arthritis in a single UK Health District 1982–1991. Ann Rheum Dis. 1999;58(4):214–219.[↩]
- Ryan MJ, Kavanagh R, Wall PG, Hazleman BL. Bacterial joint infections in England and Wales: analysis of bacterial isolates over a four year period. Br J Rheumatol. 1997;36(3):370–373.[↩][↩][↩][↩][↩]
- Howard-Jones AR, Isaacs D, Gibbons PJ: Twelve-month outcome following septic arthritis in children. J Pediatr Orthop B 2013, 22(5):486–490. 10.1097/BPB.0b013e32836027ca[↩]
- Ryan MJ, Kavanagh R, Wall PG, Hazleman BL: Bacterial joint infections in England and Wales: analysis of bacterial isolates over a four year period. Br J Rheumatol 1997, 36(3):370–373. 10.1093/rheumatology/36.3.370[↩]
- Goldenberg DL: Septic arthritis. Lancet 1998, 351(9097):197–202. 10.1016/S0140-6736(97)09522-6[↩]
- Gonorrhea. https://medlineplus.gov/ency/article/007267.htm[↩]
- Kaandorp CJ, Van Schaardenburg D, Krijnen P, Habbema JD, van de Laar MA: Risk factors for septic arthritis in patients with joint disease. A prospective study. Arthritis Rheum 1995, 38(12):1819–1825. 10.1002/art.1780381215[↩]
- Edwards AM, Massey RC: How does Staphylococcus aureus escape the bloodstream? Trends Microbiol 2011, 19(4):184–910. 10.1016/j.tim.2010.12.005[↩]
- Shirtliff ME, Mader JT. Acute septic arthritis. Clin Microbiol Rev. 2002;15(4):527–544.[↩]
- Deesomchok U, Tumrasvin T. Clinical study of culture-proven cases of non-gonococcal arthritis. J Med Assoc Thai. 1990;73(11):615–623.[↩][↩]
- Cunningham R, Cockayne A, Humphreys H. Clinical and molecular aspects of the pathogenesis of Staphylococcus aureus bone and joint infections. J Med Microbiol. 1996;44(3):157–164.[↩]
- Schattner A, Vosti KL. Bacterial arthritis due to beta-hemolytic streptococci of serogroups A, B, C, F, and G. Analysis of 23 cases and a review of the literature. Medicine (Baltimore). 1998;77(2):122–139.[↩]
- Ross JJ, Davidson L. Methicillin-resistant Staphylococcus aureus septic arthritis: an emerging clinical syndrome. Rheumatology (Oxford). 2005;44(9):1197–1198.[↩]
- Morgan DS, Fisher D, Merianos A, Currie BJ. An 18 year clinical review of septic arthritis from tropical Australia. Epidemiol Infect. 1996;117(3):423–428[↩]
- Goldenberg DL, Cohen AS. Acute infectious arthritis. A review of patients with nongonococcal joint infections (with emphasis on therapy and prognosis). Am J Med. 1976;60(3):369–377.[↩]
- Howard AW, Viskontas D, Sabbagh C. Reduction in osteomyelitis and septic arthritis related to Haemophilus influenzae type B vaccination. J Pediatr Orthop. 1999;19(6):705–709.[↩]
- Goldenberg DL. Bacterial arthritis. In: Ruddy S, Harris ED, Sledge CB, Kelley WN, eds. Kelley’s Textbook of Rheumatology. 6th ed. Philadelphia, Pa.: Saunders; 2001:1469–1483.[↩]
- Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines 2006. http://www.cdc.gov/std/treatment/2006/urethritis-and-cervicitis.htm[↩]
- Liebling MR, Arkfeld DG, Michelini GA, et al. Identification of Neisseria gonorrhoeae in synovial fluid using the polymerase chain reaction. Arthritis Rheum. 1994;37(5):702–709.[↩]
- Ytterberg SR. Infectious disorders: mycobacterial, fungal, and parasitic arthritis. In: Klippel JH, Stone JH, Crofford LJ, White PH, eds. Primer on the Rheumatic Diseases. 13th ed. New York, NY: Springer; 2008: 290–295.[↩]
- Gardam M, Lim S. Mycobacterial osteomyelitis and arthritis. Infect Dis Clin North Am. 2005;19(4):819–830.[↩][↩][↩]
- Harrington JT. Mycobacterial and fungal infections. In: Ruddy S, Harris ED, Sledge CB, Kelley WN, eds. Kelley’s Textbook of Rheumatology. 6th ed. Philadelphia, Pa.: Saunders; 2001:1493–1505.[↩]
- Bacon RM, Kugeler K, Mead PS; Centers for Disease Control and Prevention (CDC). Surveillance for Lyme disease—United States, 1992–2006. MMWR Surveill Summ. 2008;57(10):1–9.[↩]
- Centers for Disease Control and Prevention (CDC). Recommendations for test performance and interpretation from the Second National Conference on Serologic Diagnosis of Lyme Disease. MMWR Morb Mortal Wkly Rep. 1995;44(31):590–591.[↩]
- Hu L. Lyme arthritis. Infect Dis Clin North Am. 2005;19(4):947–961.[↩][↩]
- Steere A. Borrelia burgdorferi (Lyme disease, Lyme borreliosis). In: Mandell GL, Douglas RG, Bennett JE, Dolin R, eds. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. 7th ed. Philadelphia, Pa.: Churchill, Livingstone/Elsevier; 2010:3071–3081.[↩]
- Zieger MM, Dörr U, Schulz RD. Ultrasonography of hip joint effusions. Skeletal Radiol. 1987;16(8):607–611.[↩]
- Graif M, Schweitzer ME, Deely D, Matteucci T. The septic versus non-septic inflamed joint: MRI characteristics. Skeletal Radiol. 1999;28(11):616–620.[↩]