Contents
What is silent thyroiditis
Silent thyroiditis also known as painless thyroiditis, transient painless thyroiditis, painless thyroiditis with transient hyperthyroidism, painless subacute thyroiditis, sporadic thyroiditis, destructive thyroiditis, spontaneously resolving lymphocytic thyroiditis, transient thyrotoxicosis with lymphocytic thyroiditis, is a painless inflammation of your thyroid gland (painless thyroiditis) characterized by lymphocytic infiltration and can lead to thyrotoxicosis (overactive thyroid) and hypothyroidism (underactive thyroid) 1, 2, 3, 4, 5, 6, 7, 8, 9, 10. Silent thyroiditis is considered a variant of Hashimoto’s thyroiditis (Hashimoto’s disease) or chronic autoimmune thyroiditis, forming part of the spectrum of autoimmune disorder with thyroid involvement 11, 12. A significant percentage of patients with silent thyroiditis present with elevated antibodies against thyroid peroxidase (anti-TPO) and antithyroglobulin antibodies (anti-Tg) 11.
Silent thyroiditis is most likely to occur in women during the months following pregnancy known as postpartum thyroiditis 13. Postpartum thyroiditis is considered to be a form of silent thyroiditis occurring after delivery 14. Whether postpartum thyroiditis and silent thyroiditis are linked or interconnected is not clear 13. For many people with silent thyroiditis, the thyroid function returns to normal after the first episode. Silent thyroiditis can recur, though. If it does, over time people with silent thyroiditis may develop long-term hypothyroidism also known as underactive thyroid (low thyroid hormone levels in the blood).
The exact cause of silent thyroiditis is unknown. Silent thyroiditis is a type of lymphocytic thyroiditis or an autoimmune thyroiditis 1, 5, 3, 4, 8, 9, 10, 2, 15, 16. Autoimmune thyroiditis refers to a group of conditions in which there is inflammation of the thyroid gland secondary to thyroid autoantibodies. Many patients with silent thyroiditis have a personal or a family history of other autoimmune diseases, thereby indirectly supporting the concept that it is an autoimmune thyroiditis 17. There is no significant association with viral infections 7. There is a significant association with HLA genotype DR3. Silent thyroiditis can also be caused by medicines such as interferon and amiodarone, and some types of chemotherapy, which affect the immune system 18, 19, 20, 21, 22. Recently, silent thyroiditis has been caused by durvalumab 23, lithium 24 and following vaccination against COVID-19 25.
Silent thyroiditis is not infectious. People cannot catch the disease from you. Silent thyroiditis also is not inherited within families like some other thyroid conditions.
Silent thyroiditis patients present with abrupt onset of thyrotoxicosis that can be associated with the development of a goiter or enlargement of a preexisting goiter 26. Repeated episodes may occur in the same individual 7. In a review on 112 patients, 68 were female and the age at onset was 32.4 +/- 18.5 in females and 24.9 +/- 8.2 years in males 3. None of the patients presented with thyroid pain. The duration of the thyrotoxic phase was variable, but for the most part, it lasted less than one year. The mean duration was 3.6 months (range 1 to 12.5). Symptoms began 2.5 – 2.2 months preceding the initial evaluation. This period is shorter than is usually seen with Graves’ disease and much shorter than in patients with toxic multinodular goiter. Bulging eyes (exophthalmos) and pretibial myxedema were absent 26. The thyroid gland is typically firm in consistency. Forty three percent of patients had an enlarged thyroid (goiter), which was generally symmetrical and enlargement was in most instances mild 26.
Doctors diagnose silent thyroiditis based on:
- Your medical history and physical exam. Your doctor will start by taking a medical history and performing a physical exam. In addition to asking about your symptoms, your doctor will check your neck for a goiter, which some people with silent thyroiditis can develop.
- Blood tests. Your doctor will order one or more blood tests to check for hypothyroidism and its causes. Examples include tests for:
- Thyroid-stimulating hormone (TSH). Thyroid stimulating hormone (TSH) is produced by your anterior pituitary gland. When the pituitary detects low thyroid hormones in your blood, it sends thyroid-stimulating hormone (TSH) to the thyroid to prompt an increase in thyroid hormone production. High TSH levels in the blood indicates hypothyroidism.
- Thyroid hormones T4 (thyroxine) and T3 (triiodothyronine). The main thyroid hormone is thyroxine (T4). A low blood level of thyroxine (T4) confirms the findings of a thyroid-stimulating hormone (TSH) test and indicates the problem is within the thyroid itself.
- Thyroid peroxidase antibodies (anti-TPO or TPOAb). Usually in Hashimoto’s disease, the immune system produces an antibody to thyroid peroxidase (anti-TPO or TPOAb), a protein that plays an important part in thyroid hormone production. Most people with Hashimoto’s disease will have thyroid peroxidase (TPO) antibodies in their blood. Lab tests for other antibodies associated with Hashimoto’s disease may need to be done.
- Radioactive iodine uptake (RAIU). Radioactive iodine uptake (RAIU) tests thyroid function. It measures how much radioactive iodine is taken up by your thyroid gland in a certain time period.
- You are given a pill that contains a tiny amount of radioactive iodine. After swallowing it, you wait as the iodine collects in the thyroid.
- The first uptake is usually done 4 to 6 hours after you take the iodine pill. Another uptake is usually done 24 hours later. During the uptake, you lie on your back on a table. A device called a gamma probe is moved back and forth over the area of your neck where the thyroid gland is located.
- The probe detects the location and intensity of the rays given off by the radioactive material. A computer displays how much of the tracer is taken up by the thyroid gland.
- Erythrocyte sedimentation rate (ESR). Erythrocyte sedimentation rate (ESR) is commonly called a “sed rate”. Erythrocyte sedimentation rate (ESR) is a test that indirectly measures the level of certain proteins in the blood. This measurement correlates with the amount of inflammation in your body.
- C-reactive protein (CRP). C-reactive protein (CRP) is produced by the liver. The level of CRP rises when there is inflammation in the body. It is one of a group of proteins, called acute phase reactants that go up in response to inflammation. The levels of acute phase reactants increase in response to certain inflammatory proteins called cytokines. These proteins are produced by white blood cells during inflammation.
During the first phase of silent thyroiditis, discharge of thyroid hormone from the inflamed thyroid results in increases in serum T4, T3 and a decrease in serum TSH 26. During this phase, there is no uptake of radioactive iodine in the thyroid. If thyrotoxicosis factitia is considered in the differential diagnosis, measurement of serum thyroglobulin levels is useful 26. During ingestion of levothyroxine, little or no thyroglobulin is present whereas serum thyroglobulin levels are elevated in silent thyroiditis 26. In 17 out of 71 patients with silent thyroiditis, moderate elevations of antithyroglobulin antibodies (anti-TPO or TPOAb) were present 3. Antimicrosomal antibodies were examined in 53 patients using the complement fixation test or by microsomal fluorescence. Using the former technique, 22 patients had positive antibodies, and by the latter 4 out of 7 were positive 3. In a small series of 7 patients with silent thyroiditis evaluated with a more sensitive radioimmuno assay (RIA) for human antithyroglobulin antibodies (anti-TPO or TPOAb), all were positive 5. The white blood cell count is generally normal. In 53 episodes, 34 had elevated erythrocyte sedimentation rate (ESR), but it was greater than 40 mm/hr in only 8 3. This contrasts with the typical marked elevation of the ESR in patients with subacute thyroiditis and helps to differentiate the two conditions. T4 and T3 reach subnormal levels in the hypothyroid range in 40% of patients 3. After the hypothyroid phase, patients gradually enter the euthyroid (normal) phase, heralded by an increase in thyroid hormone levels and resumption of thyroidal radioactive iodine uptake. The hypothyroid phase may last several months. In 26 episodes, patients became euthyroid after a mean period of 62 months after the onset of the hyperthyroid symptoms. TSH levels may increase during the recovery phase, and can remain elevated for many months. The delayed increase of TSH is due to its suppression during the thyrotoxic phase. Permanent hypothyroidism occurs in about 7% of patients with silent thyroiditis, but a subset of patients may ultimately become permanently hypothyroid. The echogenicity is decreased and a correlation between the decrease in the echo signal at the onset and nadir of the T3 level has been suggested 27.
Silent thyroiditis treatment is based on your symptoms. As too much thyroid hormone in your body (thyrotoxicosis) is usually mild in silent thyroiditis, there is often no need for any treatment 26. In some patients when the disease is severe, it is symptomatically treated with beta-blockers during the thyrotoxic phase relieve rapid heart rate and excessive sweating. In patients with more severe thyrotoxicosis, administration of nonsteroidal anti-inflammatory drugs (NSAIDs) and prednisone may be of benefit 28. Antithyroid medication is not effective in preventing hormone release from the affected gland. After the thyrotoxic phase, many patients become temporarily hypothyroid (underactive thyroid) and therapy with levothyroxine should be initiated in symptomatic patients or if hypothyroid phase is prolonged 11. After a few months, levothyroxine therapy should be gradually withdrawn in order to assess whether the hypothyroidism is transient or permanent 26. Only a small proportion of patients remain permanently hypothyroid. Some patients, who initially recovered, may ultimately develop permanent thyroid failure 29. In a series of 54 patients, Nikolai et al. 29 reported that about half of the patients developed permanent hypothyroidism. This is in contrast with subacute thyroiditis where permanent hypothyroidism is less common.
In patients with unusual repeated episodes of recurrent silent thyroiditis more definitive treatment may be warranted. Radioactive iodine therapy was reported to be effective in some cases of recurrent silent thyroiditis 30, 31, 32, 33. Mittra and McDougall 30 reported 4 cases of recurrent silent thyroiditis wherein three of four patients who had 3, 4, and 9 episodes of thyrotoxicosis received radioablative iodine therapy with no further recurrence after the therapy; however, all of three patients became hypothyroid that required levothyroxine supplement. There have been reports of recurrent silent thyroiditis successfully treated with thyroidectomy as well 34.
Silent thyroiditis are normally self-limited. Silent thyroiditis often goes away on its own within 1 year. The acute phase usually ends within 3 months. Some people develop hypothyroidism over time. They need to be treated for a while with a medicine that replaces thyroid hormone. Regular follow-ups with a doctor are recommended.
Thyroid gland
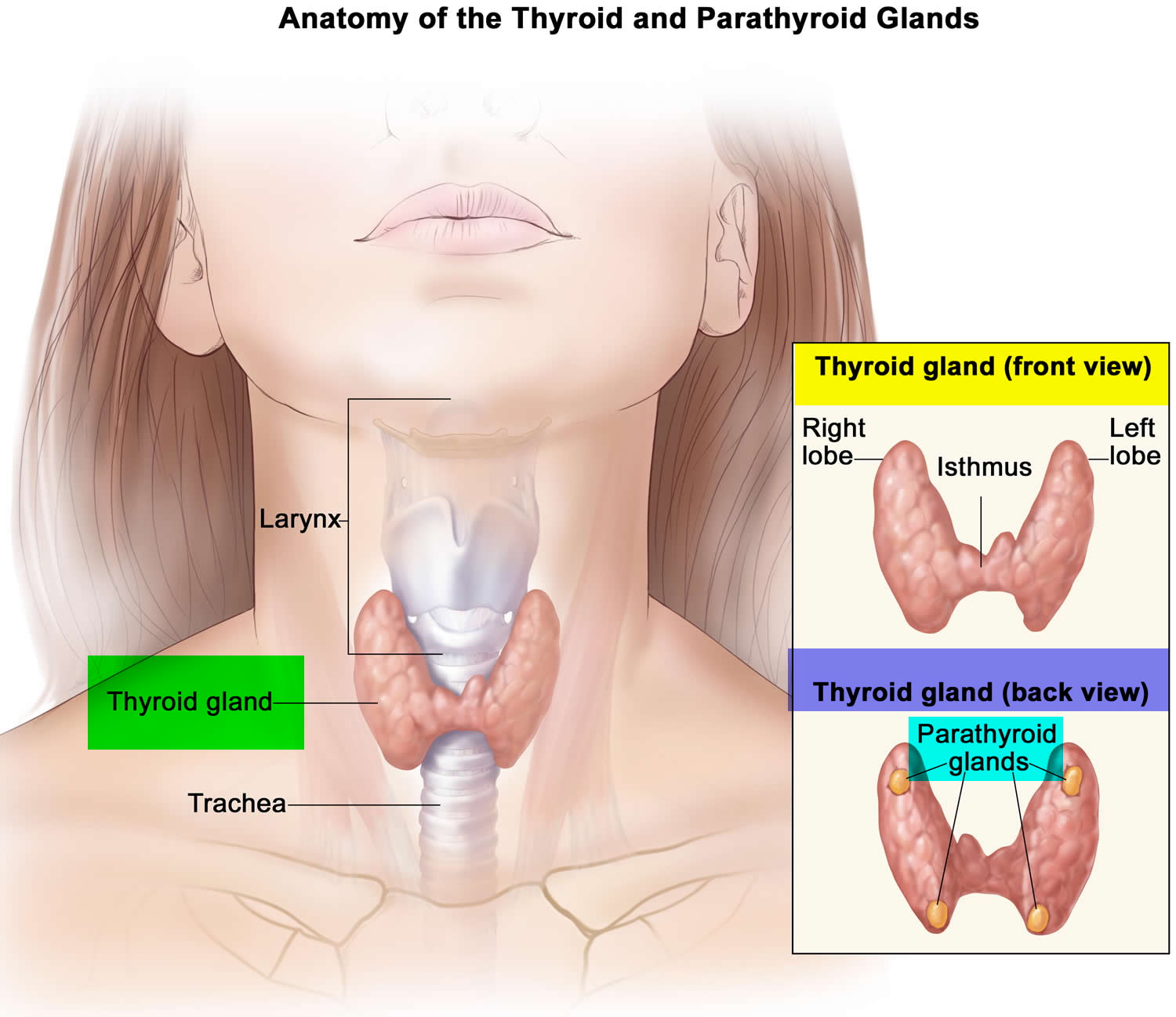
The thyroid gland is the largest adult gland to have a purely endocrine function, weighing about 25-30 g. The thyroid gland is a small butterfly shaped gland with 2 lobes, the right lobe and the left lobe joined by a narrow piece of the thyroid gland called the isthmus, that is located in front of your neck near the base of your throat, beneath the larynx (voice box or Adam’s apple). About 50% of thyroid glands have a small third lobe, called the pyramidal lobe. It extends superiorly from the isthmus. The thyroid gland makes and releases hormones. You can’t usually feel a thyroid gland that is normal.
The thyroid gland has 2 main types of cells:
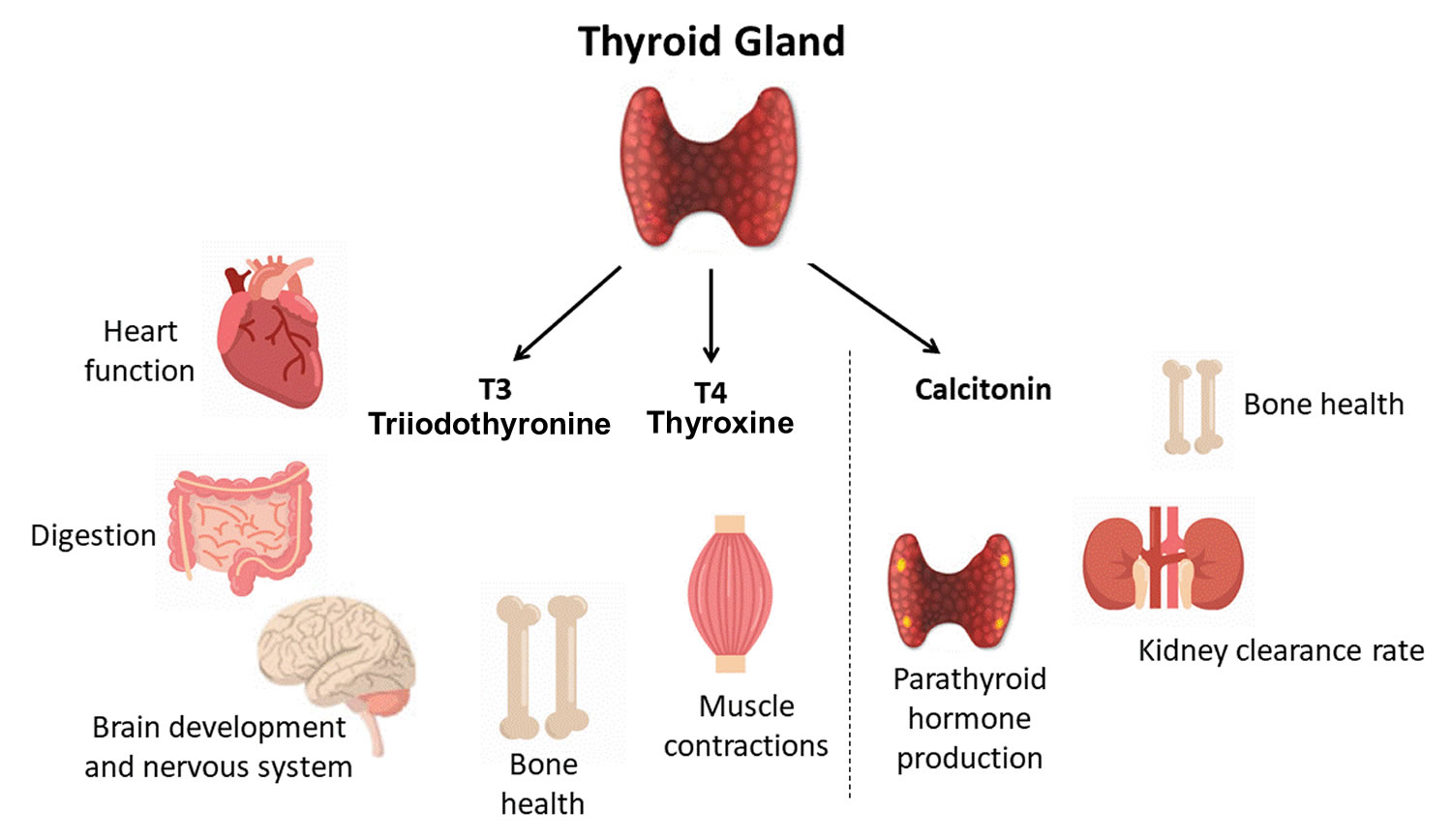
- Follicular cells use iodine from the blood to make thyroid hormones, which help regulate a person’s metabolism. Having too much thyroid hormone (hyperthyroidism) can cause a fast or irregular heartbeat, trouble sleeping, nervousness, hunger, weight loss, and a feeling of being too warm. Having too little thyroid hormone (hypothyroidism) causes a person to slow down, feel tired, and gain weight. The amount of thyroid hormone released by the thyroid gland is regulated by the pituitary gland at the base of the brain, which makes a substance called thyroid-stimulating hormone (TSH) (see Figure 5).
- C cells also called parafollicular cells at the periphery of the follicles that make calcitonin, a hormone that helps control how your body uses calcium. The parafollicular cells (C cells) respond to rising levels of blood calcium by secreting the hormone calcitonin. Calcitonin antagonizes (blocks) parathyroid hormone (PTH) and stimulates osteoblast activity, thus promoting calcium deposition and bone formation. It is important mainly in children, having relatively little effect in adults. Parathyroid hormone is made by parathyroid glands. These sit behind and are attached to the thyroid gland (see Figure 1).
Other, less common cells in the thyroid gland include immune system cells (lymphocytes) and supportive (stromal) cells.
Thyroid hormone is secreted or inhibited in response to fluctuations in metabolic rate. The brain monitors the body’s metabolic rate and stimulates thyroid hormone secretion through the action of thyrotropin-releasing hormone (TRH) and thyroid stimulating hormone (TSH) as depicted in figure 5.
The primary effect of thyroid hormone (TH) is to increase one’s metabolic rate. As a result, it raises oxygen consumption and has a calorigenic effect—it increases heat production. To ensure an adequate blood and oxygen supply to meet this increased metabolic demand, thyroid hormone also raises the breathing (respiratory) rate, heart rate, and strength of the heartbeat. It stimulates the appetite and accelerates the breakdown of carbohydrates, fats, and protein for fuel. Thyroid hormone also promotes alertness and quicker reflexes; growth hormone secretion; growth of the bones, skin, hair, nails, and teeth; and development of the fetal nervous system.
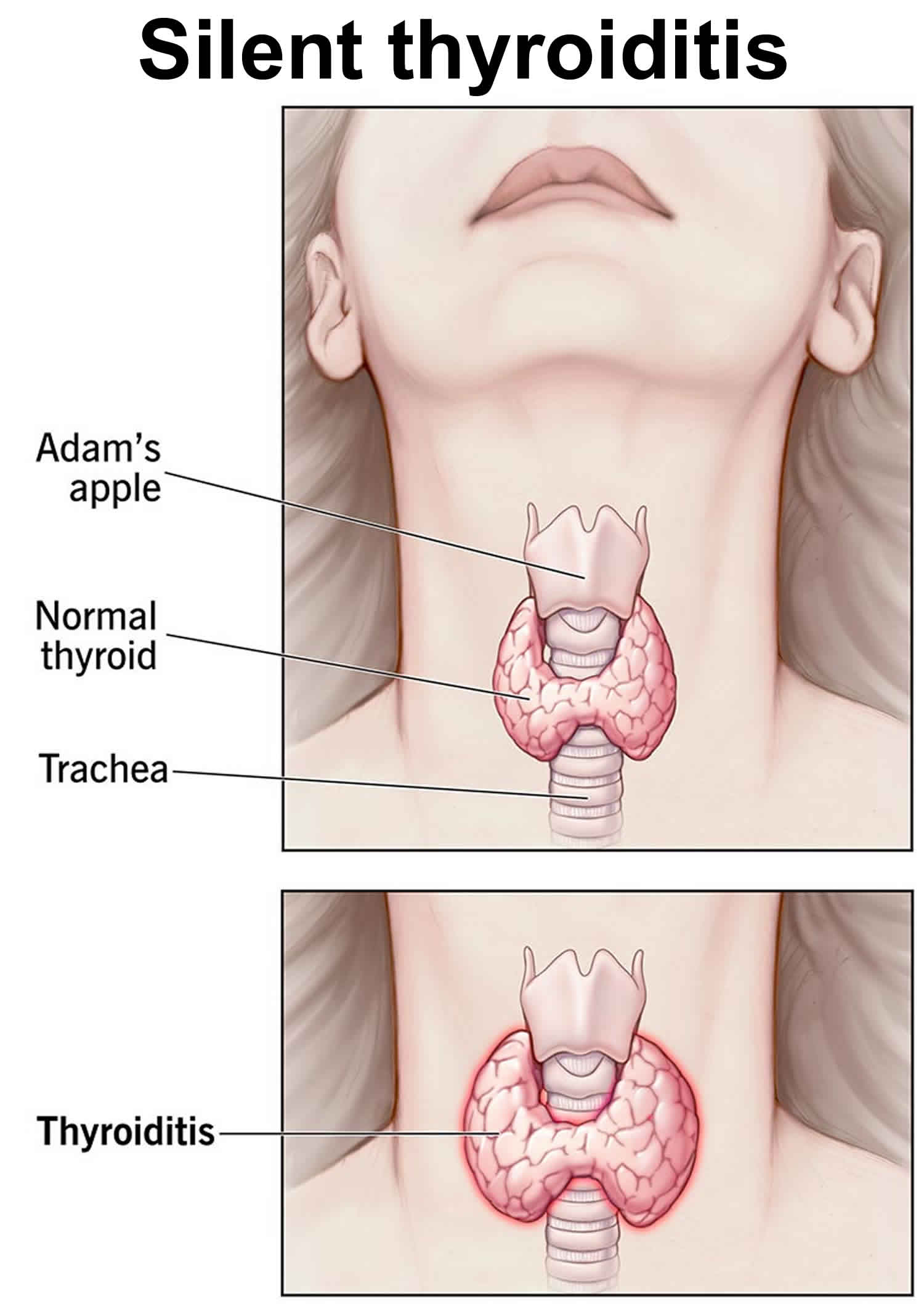
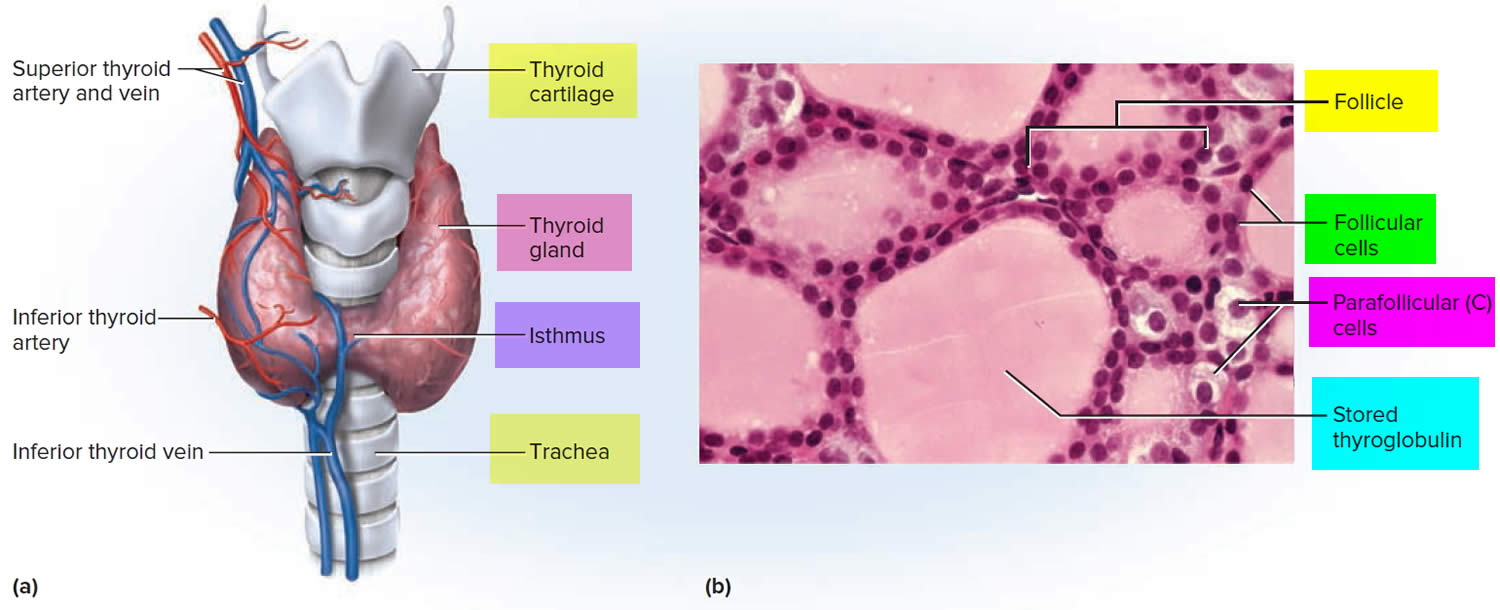
Figure 1. Thyroid gland location and parathyroid gland
Footnotes: Anatomy of the thyroid and parathyroid glands. The thyroid gland lies at the base of the throat near the trachea. It is shaped like a butterfly, with the right lobe and left lobe connected by a thin piece of tissue called the isthmus. The parathyroid glands are four pea-sized organs found in the neck near the thyroid. The thyroid and parathyroid glands make hormones.
Figure 2. Thyroid gland location
Figure 3. Thyroid gland anatomy
Footnote: (a) Gross anatomy, anterior view. (b) Histology, showing the saccular thyroid follicles (the source of thyroid hormone) and nests of C cells (the source of calcitonin).
What does the thyroid gland do?
Formation, storage, and release of thyroid hormones
The thyroid gland is the only endocrine gland that stores its secretory product in large quantities—normally about a 100-day supply. Synthesis and secretion of triiodothyronine (T3) and thyroxine or tetraiodothyronine (T4) occurs as follows:
- Iodide trapping. Thyroid follicular cells trap iodide ions (I −) by actively transporting them from the blood into the cytosol. As a result, the thyroid gland normally contains most of the iodide in the body.
- Synthesis of thyroglobulin. While the follicular cells are trapping I −, they are also synthesizing thyroglobulin (TGB), a large glycoprotein that is produced in the rough endoplasmic reticulum, modified in the Golgi complex, and packaged into secretory vesicles. The vesicles then undergo exocytosis, which releases thyroglobulin into the lumen of the follicle.
- Oxidation of iodide. Some of the amino acids in thyroglobulin are tyrosines that will become iodinated. However, negatively charged iodide (I −) ions cannot bind to tyrosine until they undergo oxidation (removal of electrons) to iodine: I −→ I. As the iodide ions are being oxidized, they pass through the membrane into the lumen of the follicle.
- Iodination of tyrosine. As iodine atoms (I) form, they react with tyrosines that are part of thyroglobulin molecules. Binding of one iodine atom yields monoiodotyrosine (T1), and a second iodination produces diiodotyrosine (T2). The thyroglobulin with attached iodine atoms, a sticky material that accumulates and is stored in the lumen of the thyroid follicle, is termed colloid.
- Coupling of monoiodotyrosine (T1) and diiodotyrosine (T2). During the last step in the synthesis of thyroid hormone, two diiodotyrosine (T2) molecules join to form thyroxine (T4) or one T1 and one T2 join to form triiodothyronine (T3).
- Pinocytosis and digestion of colloid. Droplets of colloid reenter follicular cells by pinocytosis and merge with lysosomes. Digestive enzymes in the lysosomes break down thyroglobulin, cleaving off molecules of triiodothyronine (T3) and thyroxine (T4).
- Secretion of thyroid hormones. Because T3 and T4 are lipid soluble, they diffuse through the plasma membrane into interstitial fluid and then into the blood. T4 normally is secreted in greater quantity than T3, but T3 is several times more potent. Moreover, after T4 enters a body cell, most of it is converted to T3 by removal of one iodine.
- Transport thyroid hormones in the blood. More than 99% of both the T3 and the T4 combine with transport proteins in the blood, mainly thyroxine binding globulin (TBG).
Figure 4. Thyroid hormones
Actions of thyroid hormones
Because most body cells have receptors for thyroid hormones, triiodothyronine (T3) and thyroxine (T4) affect tissues throughout the body. Thyroid hormones act on their target cells mainly by inducing gene transcription and protein synthesis. The newly formed proteins in turn carry out the cellular response.
Functions of thyroid hormones include the following:
- Increase basal metabolic rate. Thyroid hormones raise the basal metabolic rate (BMR), the rate of energy expenditure under standard or basal conditions (awake, at rest, and fasting). When basal metabolic rate increases, cellular metabolism of carbohydrates, lipids, and proteins increases. Thyroid hormones increase BMR in several ways: (1) They stimulate synthesis of additional Na+/K+ ATPases, which use large amounts of ATP to continually eject sodium ions (Na+) from cytosol into extracellular fluid and potassium ions (K+) from extracellular fluid into cytosol; (2) they increase the concentrations of enzymes involved in cellular respiration, which increases the breakdown of organic fuels and ATP production; and (3) they increase the number and activity of mitochondria in cells, which also increases ATP production. As cells produce and use more ATP, basal metabolic rate increases, more heat is given off and body temperature rises, a phenomenon called the calorigenic effect. In this way, thyroid hormones play an important role in the maintenance of normal body temperature. Normal mammals can survive in freezing temperatures, but those whose thyroid glands have been removed cannot.
- Enhance actions of catechlolamines. Thyroid hormones have permissive effects on the catecholamines (epinephrine and norepinephrine) because they up-regulate β-adrenergic receptors. Catecholamines bind to β-adrenergic receptors, promoting sympathetic responses. Therefore, symptoms of excess levels of thyroid hormone include increased heart rate, more forceful heartbeats, and increased blood pressure.
- Regulate development and growth of nervous tissue and bones. Thyroid hormones are necessary for the development of the nervous system: They promote synapse formation, myelin production, and growth of dendrites. Thyroid hormones are also required for growth of the skeletal system: They promote formation of ossification centers in developing bones, synthesis of many bone proteins, and secretion of growth hormone (GH) and insulin-like growth factors (IGFs). Deficiency of thyroid hormones during fetal development, infancy, or childhood causes severe mental retardation and stunted bone growth.
Control of thyroid hormone secretion
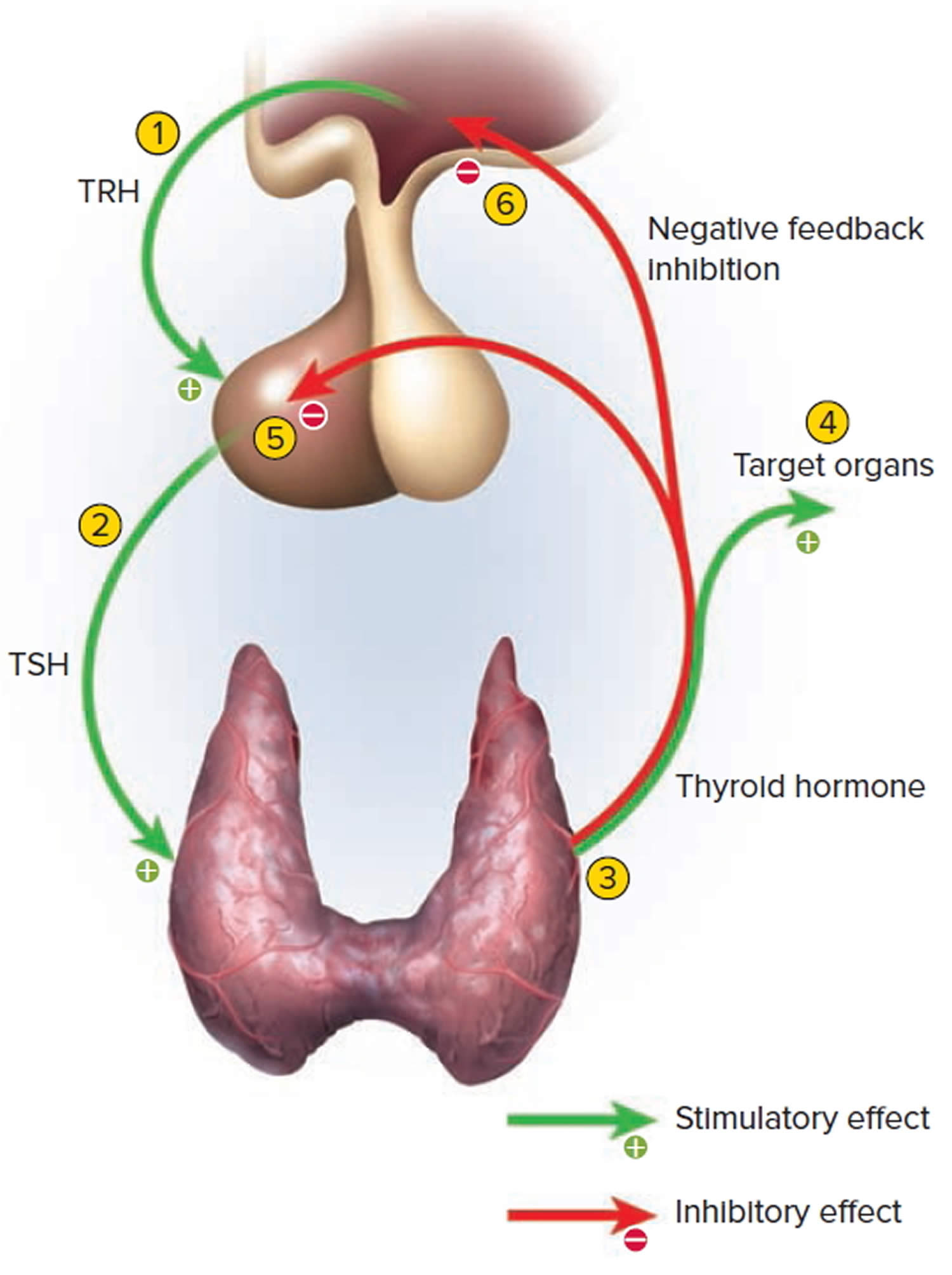
Thyrotropin-releasing hormone (TRH) from the hypothalamus and thyroid-stimulating hormone (TSH) from the anterior pituitary stimulate secretion of thyroid hormones, as shown in Figure 5:
- Low blood levels of T3 and T4 or low metabolic rate stimulate the hypothalamus to secrete thyrotropin-releasing hormone (TRH).
- Thyrotropin-releasing hormone (TRH) enters the hypothalamic–hypophyseal portal system and flows to the anterior pituitary, where it stimulates thyrotrophs to secrete thyroid stimulating hormone (TSH).
- Thyroid stimulating hormone (TSH) stimulates virtually all aspects of thyroid follicular cell activity, including iodide trapping, hormone synthesis and secretion, and growth of the follicular cells.
- The thyroid follicular cells release T3 and T4 into the blood until the metabolic rate returns to normal.
- An elevated level of T3 inhibits release of TRH and TSH (negative feedback inhibition).
Conditions that increase ATP demand—a cold environment, hypoglycemia, high altitude, and pregnancy—increase the secretion of the thyroid hormones.
Figure 5. Control of thyroid hormone secretion
Footnote: Negative Feedback Inhibition of the Anterior Pituitary Gland by the Thyroid Gland
Control of calcium balance
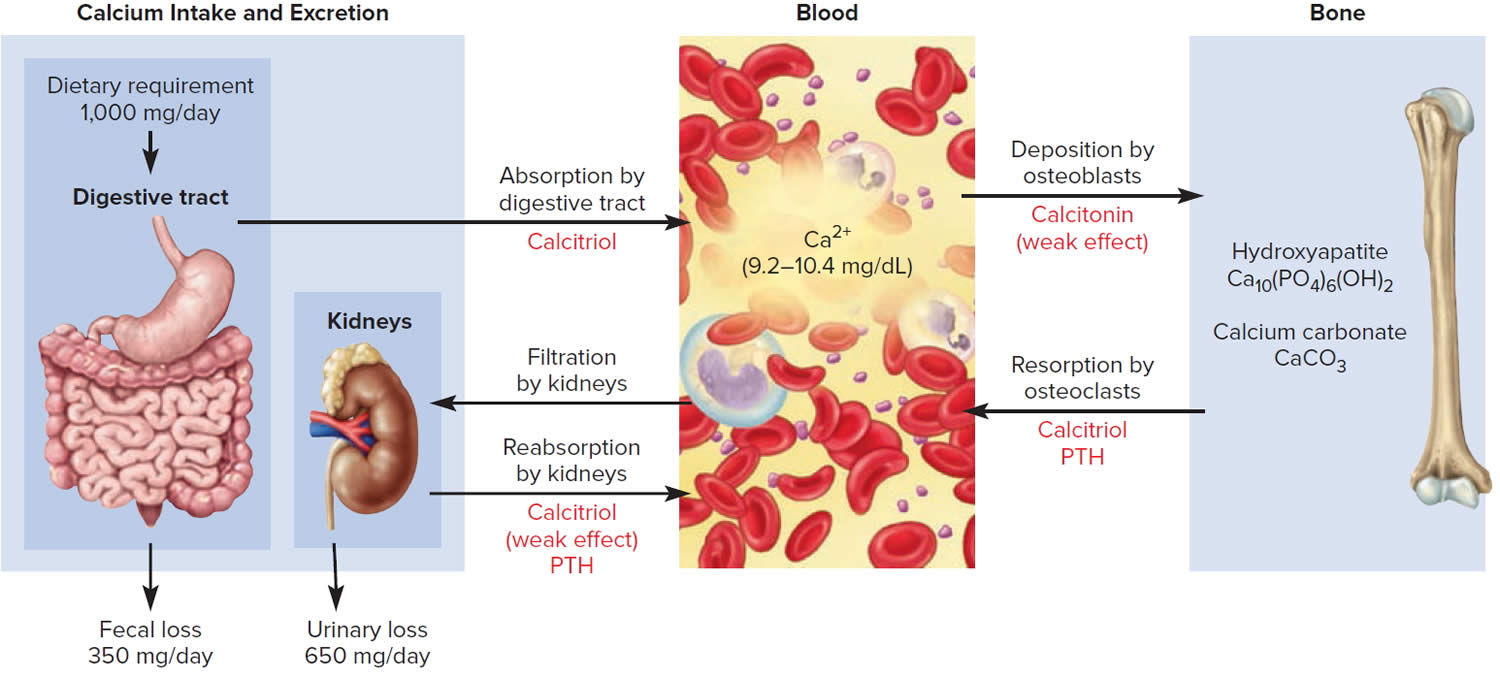
The hormone produced by the parafollicular cells of the thyroid gland is calcitonin. Calcitonin can decrease the level of calcium in the blood by inhibiting the action of osteoclasts, the cells that break down bone extracellular matrix. The secretion of calcitonin is controlled by a negative feedback system (see Figure 7).
Calcitonin is produced by C cells (clear cells) of the thyroid gland. It is secreted when the blood calcium concentration rises too high, and it lowers the concentration by two principal mechanisms:
- Osteoclast inhibition. Within 15 minutes after it is secreted, calcitonin reduces osteoclast activity by as much as 70%, so osteoclasts liberate less calcium from the skeleton.
- Osteoblast stimulation. Within an hour, calcitonin increases the number and activity of osteoblasts, which deposit calcium into the skeleton.
Calcitonin plays an important role in children but has only a weak effect in most adults. The osteoclasts of children are highly active in skeletal remodeling and release 5 g or more of calcium into the blood each day. By inhibiting this activity, calcitonin can significantly lower the blood calcium level in children. In adults, however, the osteoclasts release only about 0.8 g of calcium per day. Calcitonin cannot change adult blood calcium very much by suppressing this lesser contribution. Calcitonin deficiency is not known to cause any adult disease. Calcitonin may, however, inhibit bone loss in pregnant and lactating women. Miacalcin, a calcitonin extract derived from salmon that is 10 times more potent than human calcitonin, is prescribed to treat osteoporosis.
Figure 6. Hormonal control of calcium balance
Footnote: The central panel represents the blood reservoir of calcium and shows its normal (safe) range. Calcitriol and Parathyroid Hormone (PTH) regulate calcium exchanges between the blood and the small intestine and kidneys (left). Calcitonin, calcitriol, and Parathyroid Hormone (PTH) regulate calcium exchanges between blood and bone (right).
Thyroid antibodies
Antibodies are proteins produced by your immune system in response to invasion by foreign proteins (antigens) — such as bacteria, fungi, parasites, viruses and toxins — and remove them from your body. Antibodies help protect you from viruses and bacteria. Thyroid antibodies develop when your immune system mistakenly attacks your thyroid cells, tissues or proteins. This leads to thyroid inflammation (thyroiditis), thyroid tissue damage and/or disruption of thyroid function. These antibodies attacking your body are also known as autoantibodies or antithyroid antibodies. These autoantibodies or antithyroid antibodies cause autoimmune thyroid disorders (a disease in which your body’s immune system mistakenly attacks healthy cells in your thyroid), such as Graves’ disease and Hashimoto’s thyroiditis.
If your initial thyroid test results show signs of a thyroid problem, and if there is a suspicion of autoimmune thyroid disease, your doctor may order one or more thyroid antibody tests (see Table 1). Antibody tests are used to confirm the diagnosis of autoimmune thyroid diseases. Some people will test positive for more than one type of thyroid antibody.
In people with subclinical thyroid disease, the presence of antibodies can indicate the person may go on to develop full-blown thyroid disease in the future, but that treatment is not yet required. Positive antibodies can also be present in people without thyroid disease.
Understanding the underlying cause of thyroid dysfunction can help your doctor plan the best course of treatment.
Table 1. Thyroid antibodies
| Antibodies | What do they indicate? |
|---|---|
| Thyroid peroxidase antibodies (TPOAb) | Raised in Hashimoto’s thyroiditis (or autoimmune thyroiditis) and sometimes raised in Graves’ disease |
| Thyroglobulin antibodies (Tg Ab) – antibodies directed against the thyroglobulin (a protein present in the thyroid gland), from which thyroid hormones are produced | May be measured as part of the monitoring of people previously treated for thyroid cancer. Also sometimes raised in Hashimoto’s thyroiditis |
| Thyroid stimulating hormone receptor antibodies (TRAb). TRAb is potentially stimulatory and blocking to the thyroid gland. | Raised in Graves’ disease |
| Thyroid Stimulating Immunoglobulin (TSI) antibody specific to Graves’ disease. It is a stimulatory antibody and the one that causes an overactive thyroid gland. | May be raised in Graves’ disease. This is not routinely tested and used mainly as a research tool. |
Footnote: Thyroid antibody tests are used to diagnose autoimmune thyroid disease. Thyroid antibody tests may also be used to determine whether your mild hypothyroidism (underactive thyroid) is likely to get worse over time, to evaluate the risk to the fetus in pregnant individuals with thyroid disease, to estimate the risk of relapse after treatment for Graves’ disease and to monitor if you have been previously treated for thyroid cancer 35.
[Source 36 ]Silent thyroiditis causes
The exact cause of silent thyroiditis is unknown. Silent thyroiditis is a type of lymphocytic thyroiditis or an autoimmune thyroiditis 1, 5, 3, 4, 8, 9, 10, 2, 15, 16. Autoimmune thyroiditis refers to a group of conditions in which there is inflammation of the thyroid gland secondary to thyroid autoantibodies. Many patients with silent thyroiditis have a personal or a family history of other autoimmune diseases, thereby indirectly supporting the concept that it is an autoimmune thyroiditis 17. There is no significant association with viral infections 7. There is a significant association with HLA genotype DR3. Silent thyroiditis can also be caused by medicines such as interferon and amiodarone, and some types of chemotherapy, which affect the immune system 18, 19, 20, 21, 22. Recently, silent thyroiditis has been caused by durvalumab 23, lithium 24 and following vaccination against COVID-19 25 and COVID-19 infection 37, 38, 39, 40.
On histological examination, thyroid follicles are disrupted and infiltrated by lymphocytes and plasma cells 41, 7. The infiltration is diffuse and/or focal, sometimes with the formation of lymphoid follicles. The follicular cells are heterogeneous in appearance. They can be cuboidal or columnar when stimulated by TSH. Some of the hypertrophic follicular cells have an oxyphilic cytoplasm (Hürthle or Ashkenazy cells). Thyroid tissue obtained during the hypothyroid or early recovery phase may show regenerating follicles with little colloid. In some patients, persistent mild lymphocytic thyroiditis is seen. Fibrosis is usually minimal, but can be extensive in some cases. Occasionally multinucleated giant cells, which are characteristic of subacute thyroiditis, are observed. The histological picture of postpartum thyroiditis is identical 26.
Silent thyroiditis symptoms
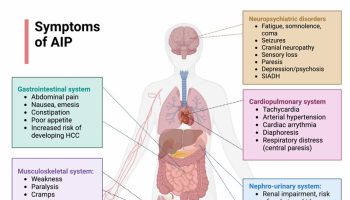
Silent thyroiditis symptoms may include abrupt onset of too much thyroid hormone in your body (thyrotoxicosis) that can be associated with the development of an enlargement of your thyroid gland (a goiter) or enlargement of a preexisting goiter 26. These symptoms may last for up to 3 months.
Signs and symptoms of mild and moderate thyrotoxicosis sometimes looks like other health problems. That can make it hard to diagnose. It can cause many symptoms, including:
- Losing weight without trying.
- Having an irregular heartbeat, also called arrhythmia.
- Pounding of the heart, sometimes called heart palpitations.
- Having a rapid heartbeat (tachycardia) — usually a heart rate higher than 100 beats per minute.
- Experiencing muscle weakness.
- Increased hunger.
- Feeling shaky, usually a small trembling in the hands and fingers.
- Feeling nervous, anxious and/or irritable.
- Experiencing increased sensitivity to heat.
- Sweating.
- Experiencing changes in your menstrual cycles (period).
- Changes in bowel patterns, especially more-frequent bowel movements.
- Enlarged thyroid gland, sometimes called a goiter, which may appear as a swelling at the base of the neck.
- Tiredness.
- Sleep problems.
- Warm, moist skin.
- Thinning skin.
- Fine, brittle hair.
Older adults are more likely to have symptoms that are hard to notice. These symptoms may include an irregular heartbeat, weight loss, depression, and feeling weak or tired during ordinary activities.
A severe case of thyrotoxicosis (too much thyroid hormone in your body) is called thyroid storm, or thyroid crisis. This condition is rare and requires immediate medical attention, as it can be life-threatening.
Symptoms of thyroid storm (severe thyrotoxicosis) include:
- Having a very rapid heartbeat.
- Having a high fever.
- Feeling very agitated and confused.
- Having nausea, vomiting and diarrhea.
- Dehydration.
- Confusion.
- Delirium.
- Experiencing a loss of consciousness.
Eventually, the decline in thyroid hormone production can result in hypothyroidism (underactive thyroid) signs and symptoms:
- Fatigue and sluggishness
- Increased sensitivity to cold
- Increased sleepiness
- Dry skin
- Constipation
- Muscle weakness
- Muscle aches, tenderness and stiffness
- Joint pain and stiffness
- Irregular or excessive menstrual bleeding
- Depression
- Problems with memory or concentration
- Swelling of the thyroid (goiter)
- A puffy face
- Brittle nails
- Hair loss
- Enlargement of the tongue
Because hypothyroidism develops slowly, you may not notice symptoms of the disease for months. Many of these symptoms, especially fatigue and weight gain, are common and do not necessarily mean you have a thyroid problem.
Hypothyroidism symptoms can persist until the thyroid recovers normal function. The recovery of the thyroid can take many months in some people. Some people only notice the hypothyroid symptoms and do not have symptoms of hyperthyroidism to begin with.
Silent thyroiditis diagnosis
Silent thyroiditis is diagnosed with a medical history, physical exam and blood tests. Depending on the results of the blood tests, you may need other tests too.
Medical history and physical exam
During the exam, your doctor may check for:
- Slight tremor in your fingers and hands.
- Overactive reflexes.
- Rapid or irregular pulse.
- Eye changes.
- Warm, moist skin.
Your doctor also examines your thyroid gland as you swallow to see if it’s larger than usual, bumpy or tender.
Blood tests
Your doctor will order one or more blood tests to check for hypothyroidism and its causes. Examples include tests for:
- Thyroid-stimulating hormone (TSH). Thyroid stimulating hormone (TSH) is produced by your anterior pituitary gland. When the pituitary detects low thyroid hormones in your blood, it sends thyroid-stimulating hormone (TSH) to the thyroid to prompt an increase in thyroid hormone production. High TSH levels in the blood indicates hypothyroidism.
- Thyroid hormones T4 (thyroxine) and T3 (triiodothyronine). The main thyroid hormone is thyroxine (T4). A low blood level of thyroxine (T4) confirms the findings of a thyroid-stimulating hormone (TSH) test and indicates the problem is within the thyroid itself.
- Thyroid peroxidase antibodies (anti-TPO or TPOAb). Usually in Hashimoto’s disease, the immune system produces an antibody to thyroid peroxidase (anti-TPO or TPOAb), a protein that plays an important part in thyroid hormone production. Most people with Hashimoto’s disease will have thyroid peroxidase (TPO) antibodies in their blood. Lab tests for other antibodies associated with Hashimoto’s disease may need to be done.
- Radioactive iodine uptake (RAIU). Radioactive iodine uptake (RAIU) tests thyroid function. It measures how much radioactive iodine is taken up by your thyroid gland in a certain time period.
- You are given a pill that contains a tiny amount of radioactive iodine. After swallowing it, you wait as the iodine collects in the thyroid.
- The first uptake is usually done 4 to 6 hours after you take the iodine pill. Another uptake is usually done 24 hours later. During the uptake, you lie on your back on a table. A device called a gamma probe is moved back and forth over the area of your neck where the thyroid gland is located.
- The probe detects the location and intensity of the rays given off by the radioactive material. A computer displays how much of the tracer is taken up by the thyroid gland.
- Higher-than-normal uptake may be due to an overactive thyroid gland. The most common cause is Graves disease. Other conditions can cause some areas of higher-than-normal uptake in the thyroid gland. These include:
- An enlarged thyroid gland that contains nodules producing too much thyroid hormone (toxic nodular goiter)
- A single thyroid nodule that is producing too much thyroid hormone (toxic adenoma). These conditions often result in normal uptake, but the uptake is concentrated into a few (hot) areas while the rest of the thyroid gland does not take up any iodine (cold areas). This can only be determined if the scan is done along with the uptake test.
- Lower-than-normal uptake may be due to:
- Factitious hyperthyroidism (taking too much thyroid hormone medicine or supplements)
- Iodine overload
- Subacute thyroiditis (swelling or inflammation of the thyroid gland)
- Silent (or painless) thyroiditis
- Amiodarone (medicine to treat some kinds of heart disease)
- Erythrocyte sedimentation rate (ESR). Erythrocyte sedimentation rate (ESR) is commonly called a “sed rate”. Erythrocyte sedimentation rate (ESR) is a test that indirectly measures the level of certain proteins in the blood. This measurement correlates with the amount of inflammation in your body.
- C-reactive protein (CRP). C-reactive protein (CRP) is produced by the liver. The level of CRP rises when there is inflammation in the body. It is one of a group of proteins, called acute phase reactants that go up in response to inflammation. The levels of acute phase reactants increase in response to certain inflammatory proteins called cytokines. These proteins are produced by white blood cells during inflammation.
During the first phase of silent thyroiditis, discharge of thyroid hormone from the inflamed thyroid results in increases in serum T4, T3 and a decrease in serum TSH 26. During this phase, there is no uptake of radioactive iodine in the thyroid. If thyrotoxicosis factitia is considered in the differential diagnosis, measurement of serum thyroglobulin levels is useful 26. During ingestion of levothyroxine, little or no thyroglobulin is present whereas serum thyroglobulin levels are elevated in silent thyroiditis 26. In 17 out of 71 patients with silent thyroiditis, moderate elevations of antithyroglobulin antibodies (anti-TPO or TPOAb) were present 3. Antimicrosomal antibodies were examined in 53 patients using the complement fixation test or by microsomal fluorescence. Using the former technique, 22 patients had positive antibodies, and by the latter 4 out of 7 were positive 3. In a small series of 7 patients with silent thyroiditis evaluated with a more sensitive radioimmuno assay (RIA) for human antithyroglobulin antibodies (anti-TPO or TPOAb), all were positive 5. The white blood cell count is generally normal. In 53 episodes, 34 had elevated erythrocyte sedimentation rate (ESR), but it was greater than 40 mm/hr in only 8 3. This contrasts with the typical marked elevation of the ESR in patients with subacute thyroiditis and helps to differentiate the two conditions. T4 and T3 reach subnormal levels in the hypothyroid range in 40% of patients 3. After the hypothyroid phase, patients gradually enter the euthyroid (normal) phase, heralded by an increase in thyroid hormone levels and resumption of thyroidal radioactive iodine uptake. The hypothyroid phase may last several months. In 26 episodes, patients became euthyroid after a mean period of 62 months after the onset of the hyperthyroid symptoms. TSH levels may increase during the recovery phase, and can remain elevated for many months. The delayed increase of TSH is due to its suppression during the thyrotoxic phase. Permanent hypothyroidism occurs in about 7% of patients with silent thyroiditis, but a subset of patients may ultimately become permanently hypothyroid. The echogenicity is decreased and a correlation between the decrease in the echo signal at the onset and nadir of the T3 level has been suggested 27.
Silent thyroiditis treatment
Silent thyroiditis treatment is based on your symptoms. As too much thyroid hormone in your body (thyrotoxicosis) is usually mild in silent thyroiditis, there is often no need for any treatment 26. In some patients, therapy with a beta-blocker can be considered during the thyrotoxic phase relieve rapid heart rate and excessive sweating. In patients with more severe thyrotoxicosis, administration of nonsteroidal anti-inflammatory drugs (NSAIDs) and prednisone may be of benefit 28. After the thyrotoxic phase, many patients become temporarily hypothyroid (underactive thyroid) and therapy with levothyroxine should be initiated in symptomatic patients. After a few months, levothyroxine therapy should be gradually withdrawn in order to assess whether the hypothyroidism is transient or permanent 26. Only a small proportion of patients remain permanently hypothyroid. Some patients, who initially recovered, may ultimately develop permanent thyroid failure 29. In a series of 54 patients, Nikolai et al. 29 reported that about half of the patients developed permanent hypothyroidism. This is in contrast with subacute thyroiditis where permanent hypothyroidism is less common.
Silent thyroiditis prognosis
Silent thyroiditis are normally self-limited. Silent thyroiditis often goes away on its own within 1 year. The acute phase usually ends within 3 months. Some people develop hypothyroidism over time. They need to be treated for a while with a medicine that replaces thyroid hormone. Regular follow-ups with a doctor are recommended.
The clinical course of silent thyroiditis consists usually of an initial hyperthyroid phase, followed by a hypothyroid phase, and subsequent restoration of a normal thyroid gland function (euthyroid) metabolic state 26. 57 out of 112 patients became euthyroid and did not develop clinical hypothyroidism 26. After a brief period of normal thyroid gland function (euthyroidism), transient biochemical hypothyroidism developed in 17 patients. In 32 patients clinical hypothyroidism was present 3. Nikolai et al. 28 reported silent thyroiditis recurrence rate of 10%, and Emerson and Farwell 42 reported less than 5%. Yamamoto et al. 43 have reported a patient with 7 episodes of silent thyroiditis within 4-year period, between 2 episodes of postpartum thyroiditis. The largest number of silent thyroiditis recurrences noted in a single patient is nine 30.
Development of Graves’ disease, after painless thyroiditis has been documented and TSH receptor antibodies have been found in these patients 15.
- Lazarus J: Sporadic and postpartum thyroiditis. In: Braverman L, Utiger R eds. The Thyroid. Philadelphia: Lippincott-Raven; 524-535, 2005.[↩][↩][↩]
- Woolf PD, Daly R. Thyrotoxicosis with painless thyroiditis. Am J Med. 1976 Jan;60(1):73-9. doi: 10.1016/0002-9343(76)90535-0[↩][↩][↩]
- Woolf PD. Transient painless thyroiditis with hyperthyroidism: a variant of lymphocytic thyroiditis? Endocr Rev. 1980 Fall;1(4):411-20. doi: 10.1210/edrv-1-4-411[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Gegick CG, Herring WB. Painless subacute thyroiditis: a report of two cases. N C Med J. 1977 Jul;38(7):387-9.[↩][↩][↩]
- Dorfman SG, Cooperman MT, Nelson RL, Depuy H, Peake RL, Young RL. Painless thyroiditis and transient hyperthyroidism without goiter. Ann Intern Med. 1977 Jan;86(1):24-8. doi: 10.7326/0003-4819-86-1-24[↩][↩][↩][↩][↩]
- Jackson IM. Editorial: “Hyper-thyroiditis” – a diagnostic pitfall. N Engl J Med. 1975 Sep 25;293(13):661-2. doi: 10.1056/NEJM197509252931311[↩]
- Nikolai TF, Brosseau J, Kettrick MA, Roberts R, Beltaos E. Lymphocytic thyroiditis with spontaneously resolving hyperthyroidism (silent thyroiditis). Arch Intern Med. 1980 Apr;140(4):478-82.[↩][↩][↩][↩][↩]
- Hamburger JI. Occult subacute thyroiditis–diagnostic challenge. Mich Med. 1971 Dec;70(31):1125-7.[↩][↩][↩]
- Morrison J, Caplan RH. Typical and atypical (‘silent’) subacute thyroiditis in a wife and husband. Arch Intern Med. 1978 Jan;138(1):45-8.[↩][↩][↩]
- Gorman CA, Duick DS, Woolner LB, Wahner HW. Transient hyperthyroidism in patients with lymphocytic thyroiditis. Mayo Clin Proc. 1978 Jun;53(6):359-65.[↩][↩][↩]
- Pearce EN, Farwell AP, Braverman LE. Thyroiditis. N Engl J Med. 2003 Jun 26;348(26):2646-55. doi: 10.1056/NEJMra021194. Erratum in: N Engl J Med. 2003 Aug 7;349(6):620.[↩][↩][↩]
- Miller, K.K. and Daniels, G.H. (2001), Association between lithium use and thyrotoxicosis caused by silent thyroiditis. Clinical Endocrinology, 55: 501-508. https://doi.org/10.1046/j.1365-2265.2001.01381.x[↩]
- Hanseree P, Salvador VB, Sachmechi I, Kim P. Recurrent silent thyroiditis as a sequela of postpartum thyroiditis. Case Rep Endocrinol. 2014;2014:286373. doi: 10.1155/2014/286373[↩][↩]
- Muller AF, Drexhage HA, Berghout A. Postpartum thyroiditis and autoimmune thyroiditis in women of childbearing age: recent insights and consequences for antenatal and postnatal care. Endocr Rev. 2001 Oct;22(5):605-30. doi: 10.1210/edrv.22.5.0441[↩]
- Iitaka M, Morgenthaler NG, Momotani N, Nagata A, Ishikawa N, Ito K, Katayama S, Ito K. Stimulation of thyroid-stimulating hormone (TSH) receptor antibody production following painless thyroiditis. Clin Endocrinol (Oxf). 2004 Jan;60(1):49-53. doi: 10.1111/j.1365-2265.2004.01939.x[↩][↩][↩]
- Tokuda Y, Kasagi K, Iida Y, Yamamoto K, Hatabu H, Hidaka A, Konishi J, Ishii Y. Sonography of subacute thyroiditis: changes in the findings during the course of the disease. J Clin Ultrasound. 1990 Jan;18(1):21-6. doi: 10.1002/jcu.1870180106[↩][↩]
- Parker M, Klein I, Fishman LM, Levey GS. Silent thyrotoxic thyroiditis in association with chronic adrenocortical insufficiency. Arch Intern Med. 1980 Aug;140(8):1108-9.[↩][↩]
- Hollenberg A, Wiersinga WM. Hyperthyroid disorders. In: Melmed S, Auchus RJ, Goldfine AB, Koenig RJ, Rosen CJ, eds. Williams Textbook of Endocrinology. 14th ed. Philadelphia, PA: Elsevier; 2020:chap 12[↩][↩]
- Brent GA, Weetman AP. Hypothyroidism and thyroiditis. In: Melmed S, Auchus RJ, Goldfine AB, Koenig RJ, Rosen CJ, eds. Williams Textbook of Endocrinology. 14th ed. Philadelphia, PA: Elsevier; 2020:chap 13.[↩][↩]
- Pearce EN, Hollenberg AN. Thyroid. In: Goldman L, Cooney KA, eds. Goldman-Cecil Medicine. 27th ed. Philadelphia, PA: Elsevier; 2024:chap 207.[↩][↩]
- Ramos-Levi AM, Marazuela M. Thyroiditis. In: Robertson RP, ed. DeGroot’s Endocrinology. 8th ed. Philadelphia, PA: Elsevier; 2023:chap 73.[↩][↩]
- Smith JR, Wassner AJ. Thyroiditis. In: Kliegman RM, St. Geme JW, Blum NJ, Shah SS, Tasker RC, Wilson KM, eds. Nelson Textbook of Pediatrics. 21st ed. Philadelphia, PA: Elsevier; 2020:chap 582.[↩][↩]
- Mengíbar JL, Capel I, Bonfill T, Mazarico I, Espuña LC, Caixàs A, Rigla M. Simultaneous onset of type 1 diabetes mellitus and silent thyroiditis under durvalumab treatment. Endocrinol Diabetes Metab Case Rep. 2019 Jul 15;2019(1):19-0045. doi: 10.1530/EDM-19-0045[↩][↩]
- Dawahir W, Gilden J, Moid A, Trendafilova V. ODP448 A Case of Lithium-Induced Silent Thyroiditis. J Endocr Soc. 2022 Nov 1;6(Suppl 1):A751–2. doi: 10.1210/jendso/bvac150.1551[↩][↩]
- Capezzone M, Tosti-Balducci M, Morabito EM, Caldarelli GP, Sagnella A, Cantara S, Alessandri M, Castagna MG. Silent thyroiditis following vaccination against COVID-19: report of two cases. J Endocrinol Invest. 2022 May;45(5):1079-1083. doi: 10.1007/s40618-021-01725-y[↩][↩]
- Kopp P. Thyrotoxicosis of other Etiologies. [Updated 2010 Dec 1]. In: Feingold KR, Anawalt B, Blackman MR, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK285562[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Miyakawa M, Tsushima T, Onoda N, Etoh M, Isozaki O, Arai M, Shizume K, Demura H. Thyroid ultrasonography related to clinical and laboratory findings in patients with silent thyroiditis. J Endocrinol Invest. 1992 Apr;15(4):289-95. doi: 10.1007/BF03348732[↩][↩]
- Nikolai TF, Coombs GJ, McKenzie AK, Miller RW, Weir GJ Jr. Treatment of lymphocytic thyroiditis with spontaneously resolving hyperthyroidism (silent thyroiditis). Arch Intern Med. 1982 Dec;142(13):2281-3. doi: 10.1001/archinte.142.13.2281[↩][↩][↩]
- Nikolai TF, Coombs GJ, McKenzie AK. Lymphocytic Thyroiditis With Spontaneously Resolving Hyperthyroidism and Subacute Thyroiditis: Long-term Follow-up. Arch Intern Med. 1981;141(11):1455–1458. doi:10.1001/archinte.1981.00340120063014[↩][↩][↩][↩]
- Mittra ES, McDougall IR. Recurrent silent thyroiditis: a report of four patients and review of the literature. Thyroid. 2007 Jul;17(7):671-5. doi: 10.1089/thy.2006.0335[↩][↩][↩]
- Ohye H. Recurrent severe painless thyroiditis requiring multiple treatments with radioactive iodine. Thyroid. 2008 Nov;18(11):1231-2. doi: 10.1089/thy.2007.0273[↩]
- Park HM. Painless thyroiditis and radioactive iodine therapy. Thyroid. 2009 Sep;19(9):1013. doi: 10.1089/thy.2008.0427[↩]
- Choe W, McDougall IR. Ablation of thyroid function with radioactive iodine after recurrent episodes of silent thyroiditis. Thyroid. 1993 Winter;3(4):311-3. doi: 10.1089/thy.1993.3.311[↩]
- Ishii H, Takei M, Sato Y, Ito T, Ito K, Sakai Y, Yumita W, Suzuki S, Komatsu M. A case of severe and recurrent painless thyroiditis requiring thyroidectomy. Med Princ Pract. 2013;22(4):408-10. doi: 10.1159/000345212[↩]
- Thyroid Antibodies Test. https://www.testing.com/tests/thyroid-antibodies[↩]
- Thyroid antibodies explained. https://www.btf-thyroid.org/thyroid-antibodies-explained[↩]
- Muller I, Cannavaro D, Dazzi D, et al. SARS‐CoV‐2‐related atypical thyroiditis. Lancet Diabetes Endocrinol. 2020;8(9):739‐741. doi: 10.1016/S2213-8587(20)30266-7[↩]
- Asfuroglu Kalkan E, Ates I. A case of subacute thyroiditis associated with Covid‐19 infection. J Endocrinol Invest. 2020;43(8):1173‐1174. doi: 10.1007/s40618-020-01316-3[↩]
- Mattar SAM, Koh SJQ, Rama Chandran S, Cherng BPZ. Subacute thyroiditis associated with COVID‐19. BMJ Case Rep. 2020;13(8):e237336. doi: 10.1136/bcr-2020-237336[↩]
- Quaytman J, Gollamudi U, Bass N, Suresh S. Reactive arthritis and silent thyroiditis following SARS-CoV-2 infection: Case report and review of the literature. Clin Case Rep. 2022 Feb 7;10(2):e05430. doi: 10.1002/ccr3.5430[↩]
- Mizukami Y, Michigishi T, Hashimoto T, Tonami N, Hisada K, Matsubara F, Takazakura E. Silent thyroiditis: a histologic and immunohistochemical study. Hum Pathol. 1988 Apr;19(4):423-31. doi: 10.1016/s0046-8177(88)80492-1[↩]
- Emerson C, Farwell AP. Sporadic silent thyroidits, postpartum thyroiditis, and subacute thyroiditis. In: Braverman LE, Utiger RD, editors. Werner and Ingbar’s the Thyroid: A Fundamental and Clinical Text. Philadelphia, Pa, USA: Lippincott, Williams and Wilkins; 2000. pp. 578–589.[↩]
- Yamamoto M, Shibuya N, Chen LC, Ogata E. Seasonal recurrence of transient hypothyroidism in a patient with autoimmune thyroiditis. Endocrinol Jpn. 1988 Feb;35(1):135-42. doi: 10.1507/endocrj1954.35.135[↩]