Contents
What is the thyroid gland
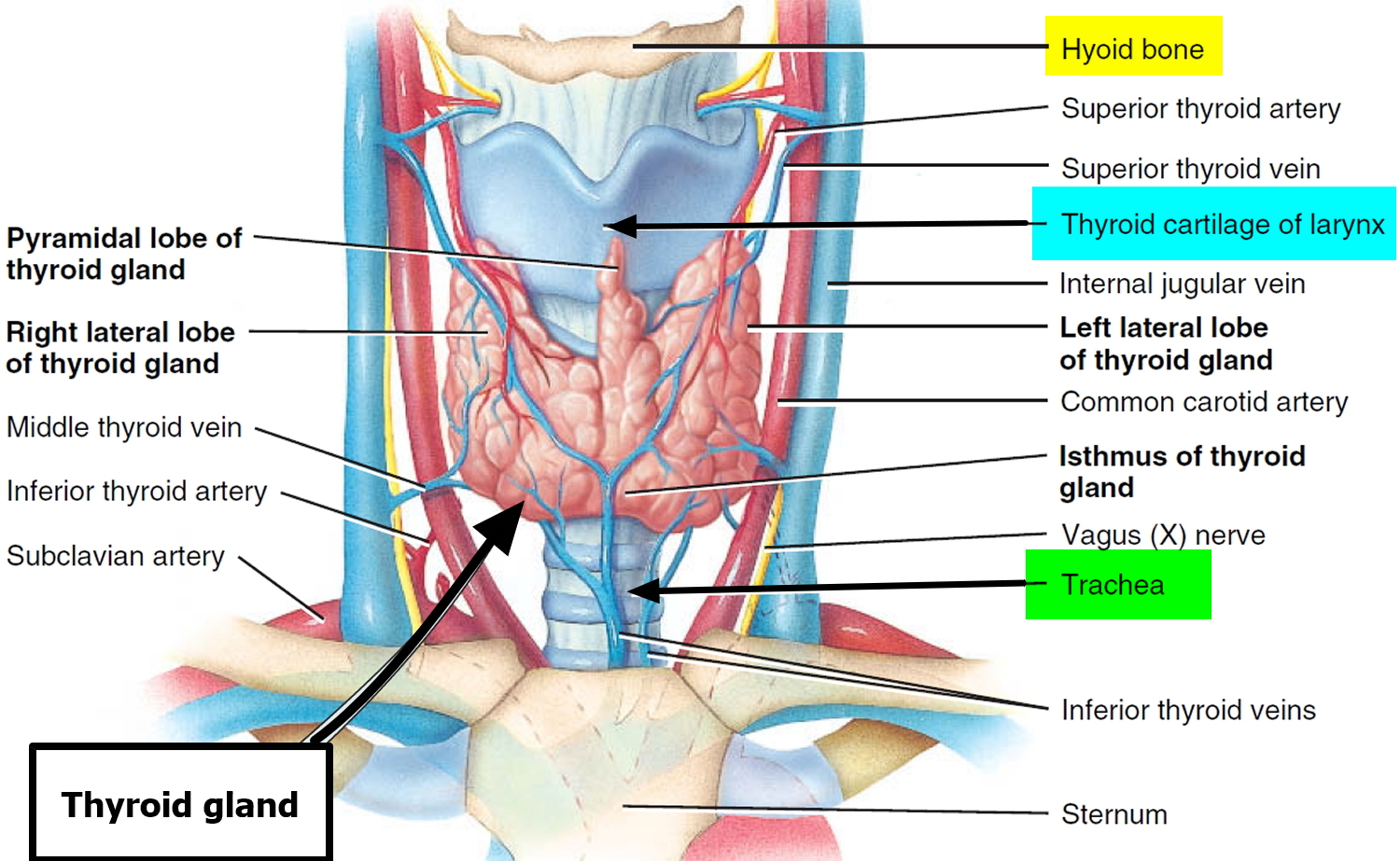
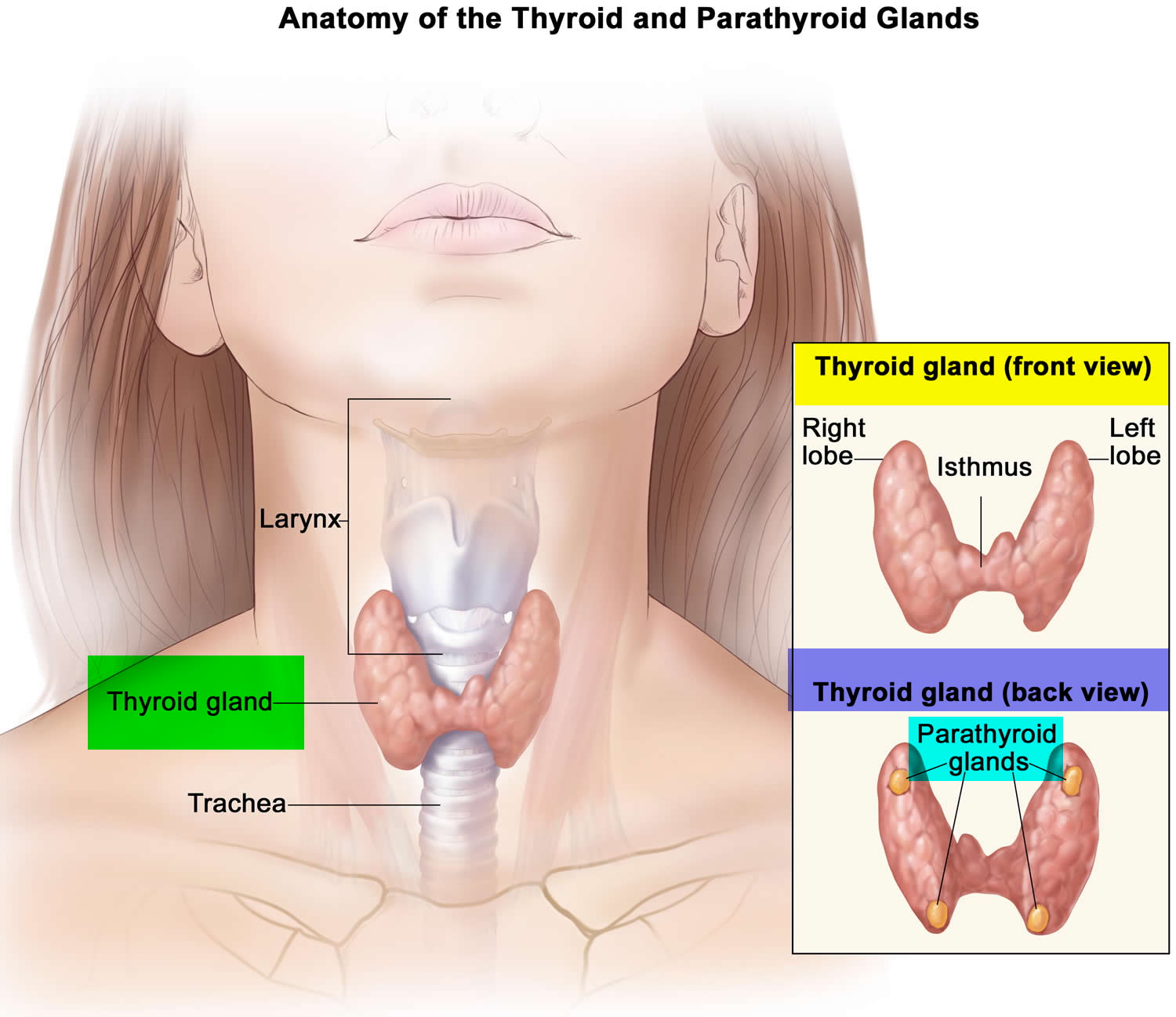
The thyroid gland is the largest adult gland to have a purely endocrine function, weighing about 25-30 g. The butterfly-shaped thyroid gland is located adjacent to the trachea just inferior to the larynx (voice box) and is named for the nearby shield like thyroid cartilage of the larynx.
The thyroid gland is composed of right and left lateral lobes, one on either side of the trachea, that are connected by an isthmus in front of the trachea. About 50% of thyroid glands have a small third lobe, called the pyramidal lobe. It extends superiorly from the isthmus.
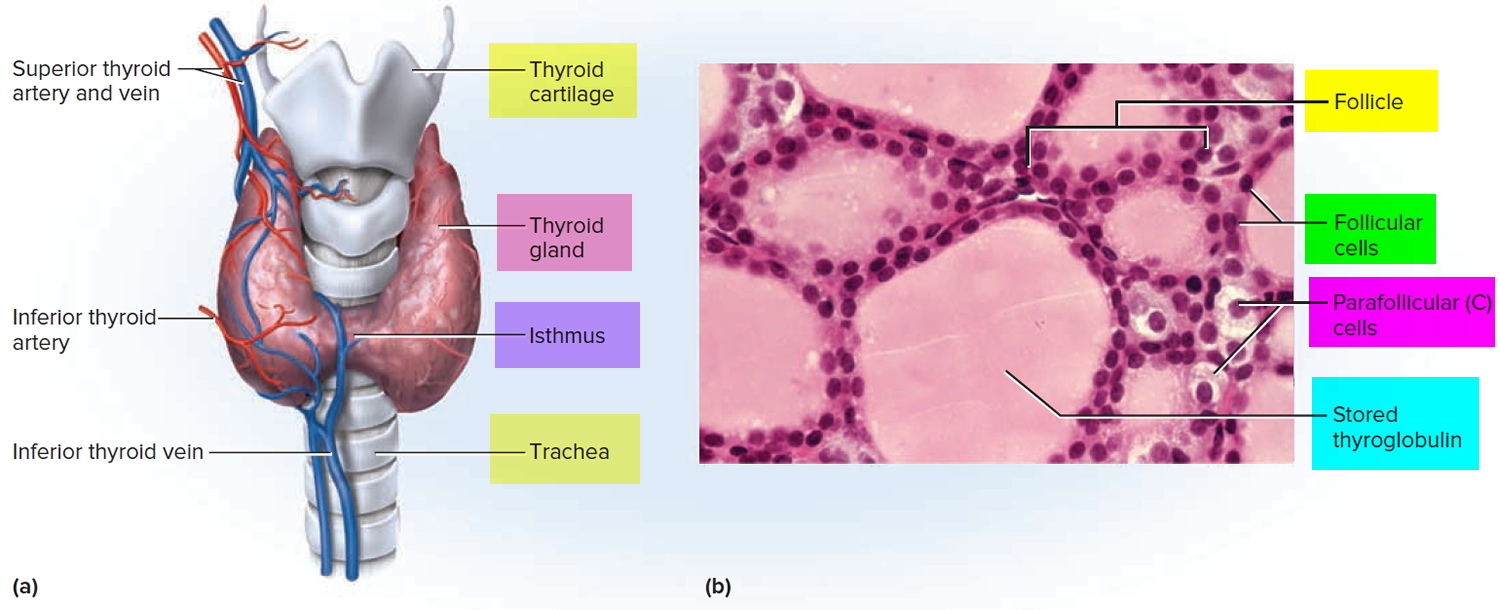
Microscopic spherical sacs called thyroid follicles make up most of the thyroid gland. The wall of each follicle consists primarily of cells called follicular cells, most of which extend to the lumen (internal space) of the follicle. A basement membrane surrounds each follicle. When the follicular cells are inactive, their shape is low cuboidal to squamous, but under the influence of thyroid stimulating hormone (TSH) they become active in secretion and range from cuboidal to low columnar in shape. The follicular cells produce two hormones: thyroxine, which is also called tetraiodothyronine (T4) because it contains four atoms of iodine and triiodothyronine (T3), which contains three atoms of iodine. T3 and T4 together are also known as thyroid hormones.
A few cells called parafollicular cells or C cells lie between follicles. They produce the hormone calcitonin, which helps regulate calcium homeostasis.
Like other endocrine glands, the thyroid releases these hormones directly into the bloodstream. Each follicle is surrounded by a basket like network of capillaries, the globular clusters of blood vessels. These are supplied by the superior and inferior thyroid arteries. The thyroid receives one of the body’s highest rates of blood flow per gram of tissue and consequently has a dark reddish brown color.
Thyroid hormone is secreted or inhibited in response to fluctuations in metabolic rate. The brain monitors the body’s metabolic rate and stimulates thyroid hormone (TH) secretion through the action of thyrotropin-releasing hormone (TRH) and thyroid stimulating hormone (TSH) as depicted in figure 3.
The primary effect of thyroid hormone (TH) is to increase one’s metabolic rate. As a result, it raises oxygen consumption and has a calorigenic effect—it increases heat production. To ensure an adequate blood and oxygen supply to meet this increased metabolic demand, thyroid hormone also raises the respiratory rate, heart rate, and strength of the heartbeat. It stimulates the appetite and accelerates the breakdown of carbohydrates, fats, and protein for fuel. Thyroid hormone also promotes alertness and quicker reflexes; growth hormone secretion; growth of the bones, skin, hair, nails, and teeth; and development of the fetal nervous system.
The thyroid gland also contains nests of parafollicular cells, also called clear (C) cells, at the periphery of the follicles. They respond to rising levels of blood calcium by secreting the hormone calcitonin. Calcitonin antagonizes parathyroid hormone and stimulates osteoblast activity, thus promoting calcium deposition and bone formation. It is important mainly in children, having relatively little effect in adults.
Figure 1. Thyroid gland location
Figure 2. Thyroid gland location
Figure 3. Thyroid gland
Thyroid gland function
Formation, Storage, and Release of Thyroid Hormones
The thyroid gland is the only endocrine gland that stores its secretory product in large quantities—normally about a 100-day supply. Synthesis and secretion of triiodothyronine (T3) and tetraiodothyronine (T4) occurs as follows:
- Iodide trapping. Thyroid follicular cells trap iodide ions (I −) by actively transporting them from the blood into the cytosol. As a result, the thyroid gland normally contains most of the iodide in the body.
- Synthesis of thyroglobulin. While the follicular cells are trapping I −, they are also synthesizing thyroglobulin (TGB), a large glycoprotein that is produced in the rough endoplasmic reticulum, modified in the Golgi complex, and packaged into secretory vesicles. The vesicles then undergo exocytosis, which releases thyroglobulin into the lumen of the follicle.
- Oxidation of iodide. Some of the amino acids in thyroglobulin are tyrosines that will become iodinated. However, negatively charged iodide (I −) ions cannot bind to tyrosine until they undergo oxidation (removal of electrons) to iodine: I −→ I. As the iodide ions are being oxidized, they pass through the membrane into the lumen of the follicle.
- Iodination of tyrosine. As iodine atoms (I) form, they react with tyrosines that are part of thyroglobulin molecules. Binding of one iodine atom yields monoiodotyrosine (T1), and a second iodination produces diiodotyrosine (T2). The thyroglobulin with attached iodine atoms, a sticky material that accumulates and is stored in the lumen of the thyroid follicle, is termed colloid.
- Coupling of monoiodotyrosine (T1) and diiodotyrosine (T2). During the last step in the synthesis of thyroid hormone, two diiodotyrosine (T2) molecules join to form tetraiodothyronine (T4) or one T1 and one T2 join to form triiodothyronine (T3).
- Pinocytosis and digestion of colloid. Droplets of colloid reenter follicular cells by pinocytosis and merge with lysosomes. Digestive enzymes in the lysosomes break down thyroglobulin, cleaving off molecules of T3 and T4.
- Secretion of thyroid hormones. Because T3 and T4 are lipid soluble, they diffuse through the plasma membrane into interstitial fluid and then into the blood. T4 normally is secreted in greater quantity than T3, but T3 is several times more potent. Moreover, after T4 enters a body cell, most of it is converted to T3 by removal of one iodine.
- Transport in the blood. More than 99% of both the T3 and the T4 combine with transport proteins in the blood, mainly thyroxine binding globulin (TBG).
Figure 4. Thyroid gland anatomy
Note: (a) Gross anatomy, anterior view. (b) Histology, showing the saccular thyroid follicles (the source of thyroid hormone) and nests of C cells (the source of calcitonin).
Actions of Thyroid Hormones
Because most body cells have receptors for thyroid hormones, T3 and T4 affect tissues throughout the body. Thyroid hormones act on their target cells mainly by inducing gene transcription and protein synthesis. The newly formed proteins in turn carry out the cellular response.
Functions of thyroid hormones include the following:
- Increase basal metabolic rate. Thyroid hormones raise the basal metabolic rate (BMR), the rate of energy expenditure under standard or basal conditions (awake, at rest, and fasting). When basal metabolic rate increases, cellular metabolism of carbohydrates, lipids, and proteins increases. Thyroid hormones increase BMR in several ways: (1) They stimulate synthesis of additional Na+/K+ ATPases, which use large amounts of ATP to continually eject sodium ions (Na+) from cytosol into extracellular fluid and potassium ions (K+) from extracellular fluid into cytosol; (2) they increase the concentrations of enzymes involved in cellular respiration, which increases the breakdown of organic fuels and ATP production; and (3) they increase the number and activity of mitochondria in cells, which also increases ATP production. As cells produce and use more ATP, basal metabolic rate increases, more heat is given off and body temperature rises, a phenomenon called the calorigenic effect. In this way, thyroid hormones play an important role in the maintenance of normal body temperature. Normal mammals can survive in freezing temperatures, but those whose thyroid glands have been removed cannot.
- Enhance actions of catechlolamines. Thyroid hormones have permissive effects on the catecholamines (epinephrine and norepinephrine) because they up-regulate β-adrenergic receptors. Catecholamines bind to β-adrenergic receptors, promoting sympathetic responses. Therefore, symptoms of excess levels of thyroid hormone include increased heart rate, more forceful heartbeats, and increased blood pressure.
- Regulate development and growth of nervous tissue and bones. Thyroid hormones are necessary for the development of the nervous system: They promote synapse formation, myelin production, and growth of dendrites. Thyroid hormones are also required for growth of the skeletal system: They promote formation of ossification centers in developing bones, synthesis of many bone proteins, and secretion of growth hormone (GH) and insulin-like growth factors (IGFs). Deficiency of thyroid hormones during fetal development, infancy, or childhood causes severe mental retardation and stunted bone growth.
Control of Thyroid Hormone Secretion
Thyrotropin-releasing hormone (TRH) from the hypothalamus and thyroid-stimulating hormone (TSH) from the anterior pituitary stimulate secretion of thyroid hormones, as shown in Figure 5:
- Low blood levels of T3 and T4 or low metabolic rate stimulate the hypothalamus to secrete thyrotropin-releasing hormone (TRH).
- Thyrotropin-releasing hormone (TRH) enters the hypothalamic–hypophyseal portal system and flows to the anterior pituitary, where it stimulates thyrotrophs to secrete thyroid stimulating hormone (TSH).
- Thyroid stimulating hormone (TSH) stimulates virtually all aspects of thyroid follicular cell activity, including iodide trapping, hormone synthesis and secretion, and growth of the follicular cells.
- The thyroid follicular cells release T3 and T4 into the blood until the metabolic rate returns to normal.
- An elevated level of T3 inhibits release of TRH and TSH (negative feedback inhibition).
Conditions that increase ATP demand—a cold environment, hypoglycemia, high altitude, and pregnancy—increase the secretion of the thyroid hormones.
Figure 5. Control of Thyroid Hormone Secretion
Note: Negative Feedback Inhibition of the Anterior Pituitary Gland by the Thyroid Gland
Control of calcium balance
The hormone produced by the parafollicular cells of the thyroid gland is calcitonin. Calcitonin can decrease the level of calcium in the blood by inhibiting the action of osteoclasts, the cells that break down bone extracellular matrix. The secretion of calcitonin is controlled by a negative feedback system (see Figure 7).
Calcitonin is produced by C cells (clear cells) of the thyroid gland. It is secreted when the blood calcium concentration rises too high, and it lowers the concentration by two principal mechanisms:
- Osteoclast inhibition. Within 15 minutes after it is secreted, calcitonin reduces osteoclast activity by as much as 70%, so osteoclasts liberate less calcium from the skeleton.
- Osteoblast stimulation. Within an hour, calcitonin increases the number and activity of osteoblasts, which deposit calcium into the skeleton.
Calcitonin plays an important role in children but has only a weak effect in most adults. The osteoclasts of children are highly active in skeletal remodeling and release 5 g or more of calcium into the blood each day. By inhibiting this activity, calcitonin can significantly lower the blood calcium level in children. In adults, however, the osteoclasts release only about 0.8 g of calcium per day. Calcitonin cannot change adult blood calcium very much by suppressing this lesser contribution. Calcitonin deficiency is not known to cause any adult disease. Calcitonin may, however, inhibit bone loss in pregnant and lactating women. Miacalcin, a calcitonin extract derived from salmon that is 10 times more potent than human calcitonin, is prescribed to treat osteoporosis.
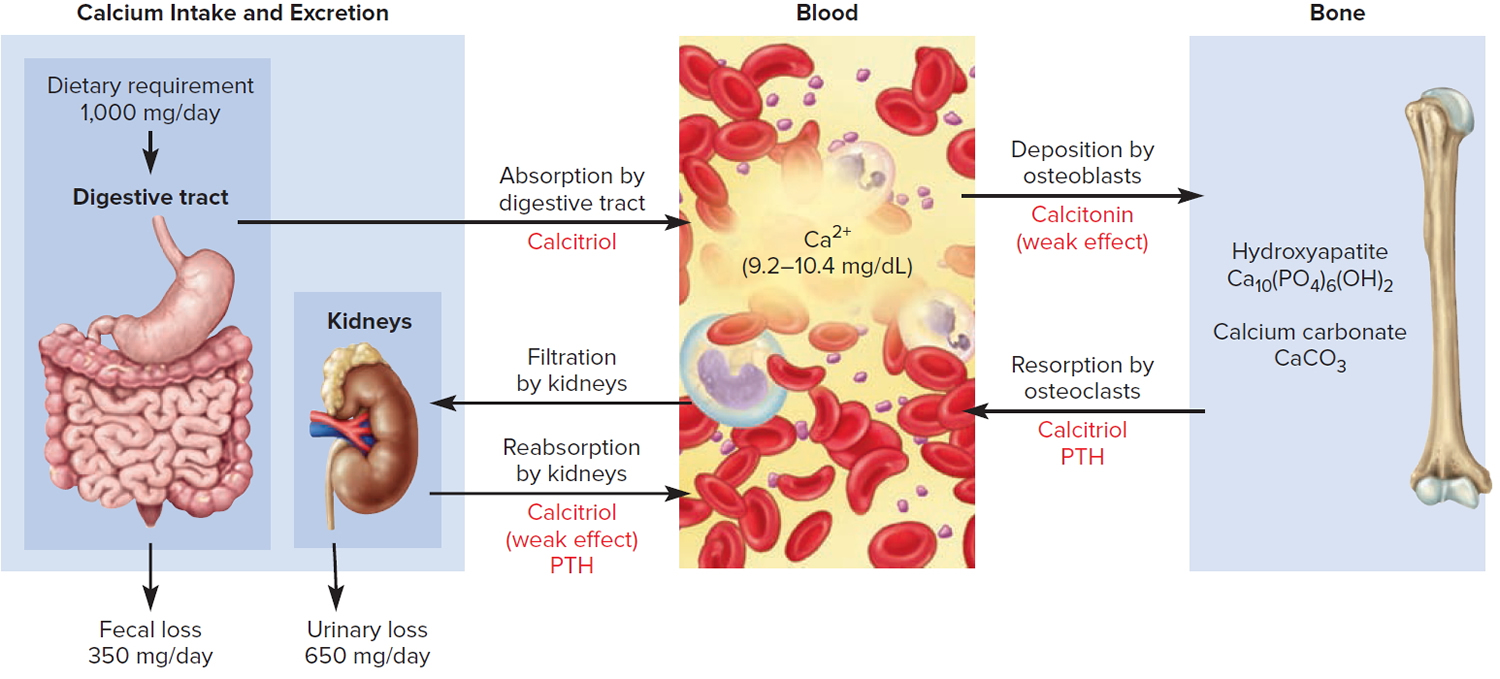
Figure 6. Hormonal control of calcium balance
Note: The central panel represents the blood reservoir of calcium and shows its normal (safe) range. Calcitriol and Parathyroid Hormone (PTH) regulate calcium exchanges between the blood and the small intestine and kidneys (left). Calcitonin, calcitriol, and Parathyroid Hormone (PTH) regulate calcium exchanges between blood and bone (right).
Figure 7. Correction of hypercalcemia (high blood calcium)
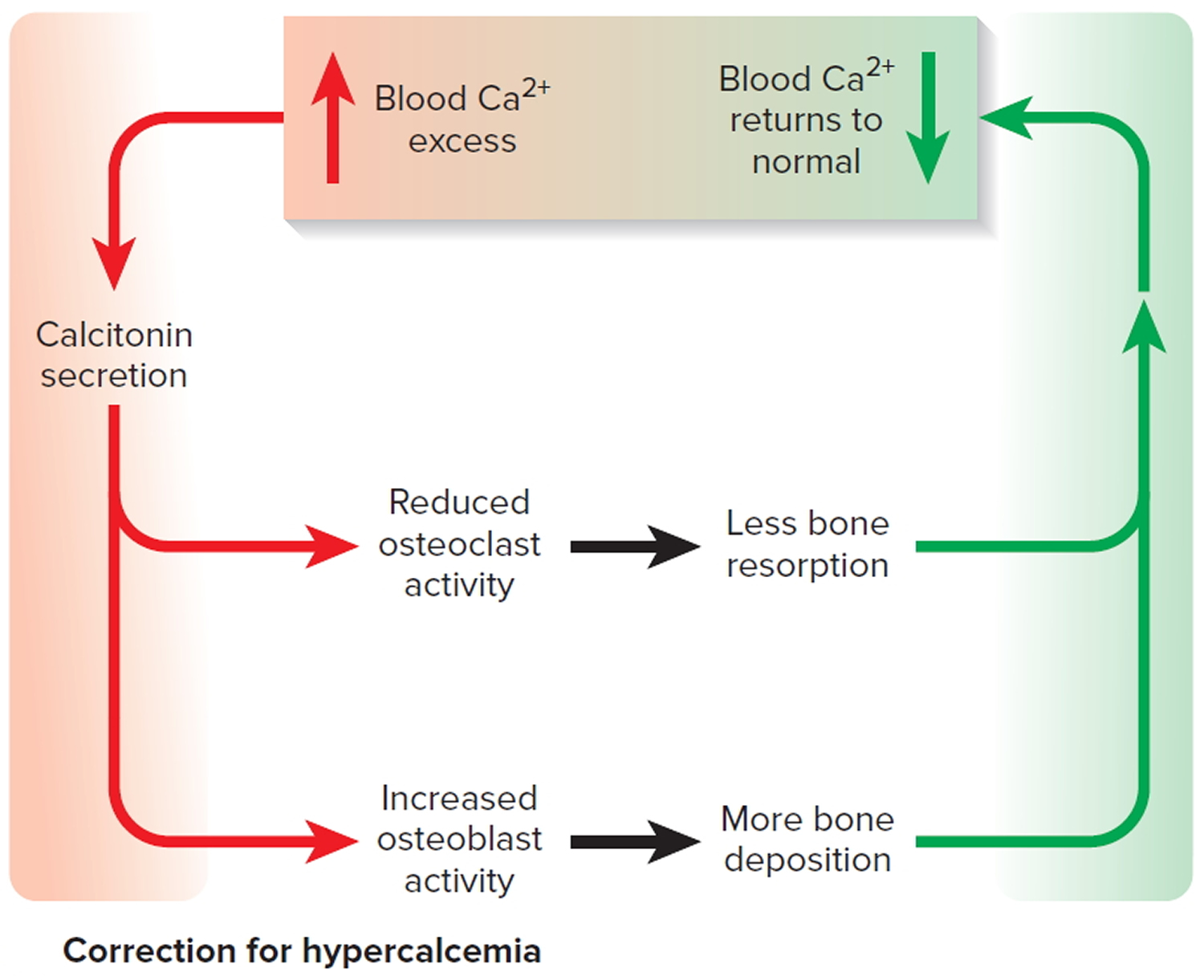
Note: Correction of high blood calcium by calcitonin
Thyroid gland problems
Thyroid hormones (T3 and T4) regulate how the body breaks down food and either uses that energy immediately or stores it for the future. In other words, your thyroid hormones regulate your body’s metabolism.
Another gland, called the anterior pituitary gland, actually controls how well the thyroid works. The anterior pituitary gland is located at the base of the brain and produces thyroid-stimulating hormone (TSH). The bloodstream carries TSH to the thyroid gland, where it tells the thyroid to produce more thyroid hormones, as needed.
Thyroid hormones influence virtually every organ system in the body. They tell organs how fast or slow they should work. Thyroid hormones also regulate the consumption of oxygen and the production of heat.
When outside influences such as disease, damage to the thyroid or certain medicines break down communication between your thyroid and the anterior pituitary gland and the hypothalamus, your thyroid might not produce enough hormone. This would slow down all of your body’s functions, a condition known as hypothyroidism or underactive thyroid. Your thyroid could also produce too much hormone sending your systems into overdrive, a condition known as hyperthyroidism or overactive thyroid. These two conditions are most often features of an underlying thyroid disease.
Thyroid problems include:
- Goiter – enlargement of the thyroid gland
- Hyperthyroidism – when your thyroid gland makes more thyroid hormones than your body needs
- Hypothyroidism – when your thyroid gland does not make enough thyroid hormones
- Thyroid cancer
- Thyroid nodules – lumps in the thyroid gland
- Thyroiditis – swelling of the thyroid
To diagnose thyroid diseases, doctors use a medical history, physical exam, and thyroid tests. They sometimes also use a biopsy. Treatment depends on the problem, but may include medicines, radioiodine therapy, or thyroid surgery.
What is Goiter ?
A simple goiter is an enlargement of the thyroid gland 1. It is usually not a tumor or cancer.
Simple goiters are more common in:
- People over age 40
- People with a family history of goiter
- Women
Causes of goiter
The thyroid gland is an important organ of the endocrine system. It is located at the front of the neck just above where your collarbones meet. The gland makes the hormones that control the way every cell in the body uses energy. This process is called metabolism.
Iodine deficiency is the most common cause of goiter. The body needs iodine to produce thyroid hormone. If you do not have enough iodine in your diet, the thyroid gets larger to try and capture all the iodine it can, so it can make the right amount of thyroid hormone. So, a goiter can be a sign the thyroid is not able to make enough thyroid hormone. The use of iodized salt in the United States prevents a lack of iodine in the diet.
Other causes of goiter include:
- The body’s immune system attacking the thyroid gland (autoimmune problem) e.g. Graves’ disease
- Certain medicines (lithium, amiodarone)
- Infections
- Cigarette smoking
- Certain foods (soy, peanuts, vegetables in the broccoli and cabbage family)
- Toxic nodular goiter, an enlarged thyroid gland that has a small, rounded growth or many growths called nodules, which produce too much thyroid hormone.
Symptoms of goiter
The main symptom is an enlarged thyroid gland. The size may range from a single small nodule to a large mass at the front of the neck.
Some people with a simple goiter may have symptoms of an underactive thyroid gland.
In rare cases, an enlarged thyroid can put pressure on the windpipe (trachea) and food tube (esophagus). This can lead to:
- Breathing difficulties (with very large goiters), especially when lying flat on the back
- Cough
- Hoarseness
- Swallowing difficulties, especially with solid food
- Pain in the area of the thyroid
How is goiter diagnosed ?
The health care provider will do a physical exam. This involves feeling your neck as you swallow. Swelling in the area of the thyroid may be felt.
If you have a very large goiter, you may have pressure on your neck veins. As a result, when the provider asks you to raise your arms above your head, you may feel dizzy.
Blood tests may be ordered to measure thyroid function:
- Free thyroxine (T4)
- Thyroid stimulating hormone (TSH)
Tests to look for abnormal and possibly cancerous areas in the thyroid gland include:
- Thyroid scan and uptake
- Ultrasound of the thyroid
If nodules are found on an ultrasound, a biopsy may be needed to check for thyroid cancer.
Treatment of goiter
A goiter only needs to be treated if it is causing symptoms.
Treatments for an enlarged thyroid include:
- Thyroid hormone replacement pills if the goiter is due to an underactive thyroid
- Small doses of Lugol’s iodine or potassium iodine solution if the goiter is due to a lack of iodine
- Radioactive iodine to shrink the gland, especially if the thyroid is producing too much thyroid hormone
- Surgery (thyroidectomy) to remove all or part of the gland
Outlook (Prognosis) of goiter
A simple goiter may disappear on its own, or may become larger. Over time, the thyroid gland may stop making enough thyroid hormone. This condition is called hypothyroidism.
In some cases, a goiter becomes toxic and produces thyroid hormone on its own. This can cause high levels of thyroid hormone, a condition called hyperthyroidism.
When to see a Medical Professional
Call your provider if you experience any swelling in the front of your neck or any other symptoms of goiter.
Prevention of goiter
Using iodized table salt prevents most simple goiters.
What is Hashimoto’s thyroiditis ?
Hashimoto’s thyroiditis (also called autoimmune or chronic lymphocytic thyroiditis) is the most common thyroid disease in the United States 2. It is an inherited condition that affects over 10 million Americans and is about seven times more common in women than in men.
Hashimoto’s thyroiditis is characterized by the production of immune cells and autoantibodies by the body’s immune system that can damage thyroid cells and compromise their ability to make thyroid hormone. Hypothyroidism occurs if the amount of thyroid hormone which can be produced is not enough for the body’s needs. The thyroid gland may also enlarge, forming a goiter.
Causes of Hashimoto’s thyroiditis
Hashimoto’s thyroiditis results from a malfunction in the immune system. When working properly, the immune system is designed to protect the body against invaders such as bacteria, viruses and other foreign substances. The immune system of someone with Hashimoto’s thyroiditis mistakenly recognizes normal thyroid cells as foreign tissue, and it produces antibodies that may destroy these cells. Although various environmental factors have been studied, none have been positively proven to be the cause of Hashimoto’s thyroiditis.
Signs & Symptoms of Hashimoto’s thyroiditis
Hashimoto’s thyroiditis may not cause symptoms for many years and may remain undiagnosed until an enlarged thyroid gland or abnormal blood tests are discovered as part of a routine examination. When symptoms do develop, they are either related to local pressure in the neck caused by the goiter itself or to the low levels of thyroid hormone.
The first sign of this disease may be painless swelling in the lower front of the neck. This enlargement may eventually become easily visible. It may be associated with an uncomfortable pressure sensation in the lower neck, and this pressure on surrounding structures may cause additional symptoms, including difficulty swallowing.
Although many of the symptoms associated with thyroid hormone deficiency occur commonly in patients without thyroid disease, patients with Hashimoto’s thyroiditis who develop hypothyroidism are more likely to experience the following:
- Fatigue
- Drowsiness
- Forgetfulness
- Difficulty with learning
- Dry, brittle hair and nails
- Dry, itchy skin
- Puffy face
- Constipation
- Sore muscles
- Weight gain
- Heavy menstrual flow
- Increased sensitivity to many medications
The thyroid enlargement and/or hypothyroidism caused by Hashimoto’s thyroiditis progresses in many patients, causing a slow worsening of symptoms. Therefore, patients should be recognized and adequately treated with thyroid hormone. Optimal treatment with thyroid hormone will eliminate any symptoms due to thyroid hormone deficiency, usually prevent further thyroid enlargement, and may sometimes cause shrinkage of an enlarged thyroid gland.
How is Hashimoto’s thyroiditis diagnosed ?
A physician experienced in the diagnosis and treatment of thyroid disease can detect a goiter due to Hashimoto’s thyroiditis by performing a physical examination and can recognize hypothyroidism by identifying characteristic symptoms, finding typical physical signs and performing appropriate laboratory tests.
Tests
THYROID ANTIBODIES: Testing for increased thyroid antibodies provides the most specific laboratory evidence of Hashimoto’s thyroiditis, but the antibodies are not present in all cases.
TSH (THYROID-STIMULATING HORMONE OR THYROTROPIN): Increased TSH level in the blood is the most accurate indicator of hypothyroidism. TSH is produced by another gland, the pituitary, which is located behind the nose at the base of the brain. The level of TSH rises dramatically when the thyroid gland even slightly under produces thyroid hormone. So, in patients with normal pituitary function, a normal level of TSH reliably excludes hypothyroidism.
FREE THYROXINE ESTIMATE: The active portion of all of the thyroxine circulating in the blood. A low level of free thyroxine is consistent with thyroid hormone deficiency. However, free thyroxine values in the “normal range” may actually represent thyroid hormone deficiency in a particular patient, since a high level of TSH stimulation may keep the free thyroxine levels “within normal limits” for many years.
FINE-NEEDLE ASPIRATION OF THE THYROID: Usually not necessary for most patients with Hashimoto’s thyroiditis, but a good way to diagnose difficult cases and a necessary procedure if a thyroid nodule is also present.
How is Hashimoto’s thyroiditis treated ?
For patients with thyroid enlargement (goiter) or hypothyroidism, thyroid hormone therapy is clearly needed, since proper dosage corrects any symptoms due to thyroid hormone deficiency and may decrease the goiter’s size. Treatment generally consists of taking a single daily tablet of levothyroxine. Older patients who may have underlying heart disease are usually started on a low dose and gradually increased, while younger, healthy patients can be started on full replacement doses at once.
While you may improve in many ways within a week, the full impact of thyroid medicine may take quite some time. For example, skin changes may take up to 3-6 months to resolve. Because of the generally permanent and often progressive nature of Hashimoto’s thyroiditis, it is usually necessary to treat it throughout one’s lifetime and to realize that dosage of medicine required may have to be adjusted from time to time.
Optimal adjustment of thyroid hormone dosage, guided by laboratory tests rather than symptoms alone, is critical, since the body is very sensitive to even small changes in thyroid hormone levels. Levothyroxine tablets come in 12 different strengths, and it is essential to take them in a consistent manner every day. If the dose is not adequate, the thyroid gland may continue to enlarge and symptoms of hypothyroidism will persist. This may be associated with increased serum cholesterol levels, possibly increasing the risk for atherosclerosis and heart disease. If the dose is too strong, it can cause symptoms of hyperthyroidism, creating excessive strain on the heart and an increased risk of developing osteoporosis.
What is hyperthyroidism ?
Hyperthyroidism develops when the body is exposed to excessive amounts of thyroid hormone. This disorder occurs in almost one percent of all Americans and affects women five to 10 times more often than men 3. In its mildest form, hyperthyroidism may not cause recognizable symptoms. More often, however, the symptoms are discomforting, disabling or even life-threatening.
What are the causes of hyperthyroidism ?
Graves’ disease: Graves’ disease (named after Irish physician Robert Graves) is an autoimmune disorder that frequently results in thyroid enlargement and hyperthyroidism. In some patients, swelling of the muscles and other tissues around the eyes may develop, causing eye prominence, discomfort or double vision. Like other autoimmune diseases, this condition tends to affect multiple family members. It is much more common in women than in men and tends to occur in younger patients.
Postpartum thyroiditis: Five to 10 percent of women develop mild to moderate hyperthyroidism within several months of giving birth. Hyperthyroidism in this condition usually lasts for approximately one to two months. It is often followed by several months of hypothyroidism, but most women will eventually recover normal thyroid function. In some cases, however, the thyroid gland does not heal, so the hypothyroidism becomes permanent and requires lifelong thyroid hormone replacement. This condition may occur again with subsequent pregnancies.
Silent thyroiditis: Transient (temporary) hyperthyroidism can be caused by silent thyroiditis, a condition which appears to be the same as postpartum thyroiditis, but is not related to pregnancy. It is not accompanied by a painful thyroid gland.
Subacute thyroiditis: This condition may follow a viral infection and is characterized by painful thyroid gland enlargement and inflammation, which results in the release of large amounts of thyroid hormones into the blood. Fortunately, this condition usually resolves spontaneously. The thyroid usually heals itself over several months, but often not before a temporary period of low thyroid hormone production (hypothyroidism) occurs.
Toxic multinodular goiter: Multiple nodules in the thyroid can produce excessive thyroid hormone, causing hyperthyroidism. Typically diagnosed in patients over the age of 50, this disorder is more likely to affect heart rhythm. In many cases, the person has had the goiter for many years before it becomes overactive.
Toxic nodule: A single nodule or lump in the thyroid can also produce more thyroid hormone than the body requires and lead to hyperthyroidism. This disorder is not familial.
Excessive iodine ingestion: Various sources of high iodine concentrations, such as kelp tablets, some expectorants, amiodarone (Cordarone, Pacerone – medications used to treat certain problems with heart rhythms) and x-ray dyes may occasionally cause hyperthyroidism in patients who are prone to it.
Overmedication with thyroid hormone: Patients who receive excessive thyroxine replacement treatment can develop hyperthyroidism. They should have their thyroid hormone dosage evaluated by a physician at least once each year and should NEVER give themselves “extra” doses.
What are the signs and symptoms of hyperthyroidism ?
When hyperthyroidism develops, a goiter (enlargement of the thyroid) is usually present and may be associated with some or many of the following features:
- Fast heart rate, often more than 100 beats per minute
- Becoming anxious, irritable, argumentative
- Trembling hands
- Weight loss, despite eating the same amount or even more than usual
- Intolerance of warm temperatures and increased likelihood to perspire
- Loss of scalp hair
- Tendency of fingernails to separate from the nail bed
- Muscle weakness, especially of the upper arms and thighs
- Loose and frequent bowel movements
- Smooth skin
- Change in menstrual pattern
- Increased likelihood for miscarriage
- Prominent “stare” of the eyes
- Protrusion of the eyes, with or without double vision (in patients with Graves’ disease)
- Irregular heart rhythm, especially in patients older than 60 years of age
- Accelerated loss of calcium from bones, which increases the risk of osteoporosis and fractures
How is hyperthyroidism diagnosed ?
Sometimes a general physician can diagnose and treat the cause of hyperthyroidism, but assistance is often needed from an endocrinologist, a physician who specializes in managing thyroid disease. Characteristic symptoms and physical signs of the disease can be detected by a trained physician. In addition, tests can be used to confirm the diagnosis and to determine the cause.
Tests
TSH (THYROID-STIMULATING HORMONE OR THYROTROPIN): A low TSH level in the blood is the most accurate indicator of hyperthyroidism. The body shuts off production of this pituitary hormone when the thyroid gland even slightly overproduces thyroid hormone. If the TSH level is low, it is very important to also check thyroid hormone levels to confirm the diagnosis of hyperthyroidism.
ESTIMATES OF FREE THYROXINE AND FREE TRIIODOTHYRONINE: When hyperthyroidism develops, free thyroxine and free triiodothyronine levels rise above previous values in that specific patient (although they may still fall within the normal range for the general population) and are often considerably elevated.
TSI (THYROID-STIMULATING IMMUNOGLOBULIN): A substance often found in the blood when Graves’ disease is the cause of hyperthyroidism.
RADIOACTIVE IODINE UPTAKE (RAIU): The amount of iodine the thyroid gland can collect, and a thyroid scan, which shows how the iodine is distributed throughout the thyroid gland.
THYROID SCAN: This information can be useful in determining the cause of hyperthyroidism and, ultimately, its treatment.
How is hyperthyroidism treated ?
Appropriate management of hyperthyroidism requires careful evaluation and ongoing care by a physician experienced in the treatment of this complex condition.
Before the development of current treatment options, the death rate from severe hyperthyroidism was as high as 50 percent. Now several effective treatments are available and, with proper management, death from hyperthyroidism is rare. Deciding which treatment is best depends on what caused the hyperthyroidism, its severity and other conditions present.
- Antithyroid Drugs
In the United States, two drugs are available for treating hyperthyroidism: propylthiouracil (PTU) and methimazole (Tapazole). Except for early pregnancy, methimazole is preferred because PTU can cause fatal liver damage, although rarely. These medications control hyperthyroidism by slowing thyroid hormone production. They may take several months to normalize thyroid hormone levels. Some patients with hyperthyroidism caused by Graves’ disease experience a spontaneous or natural remission of hyperthyroidism after a 12- to 18-month course of treatment with these drugs and may sometimes avoid permanent underactivity of the thyroid (hypothyroidism), which often occurs as a result of using the other methods of treating hyperthyroidism. Unfortunately, the remission is frequently only temporary, with the hyperthyroidism recurring after several months or years off medication and requiring additional treatment, so relatively few patients are treated solely with antithyroid medication in the United States.
Antithyroid drugs may cause an allergic reaction in about five percent of patients who use them. This usually occurs during the first six weeks of drug treatment. Such a reaction may include rash or hives; but after discontinuing use of the drug, the symptoms resolve within one to two weeks and there is no permanent damage.
A more serious side effect, but occurring in only about one in 250-500 patients during the first four to eight weeks of treatment, is a rapid decrease of white blood cells in the bloodstream. This could increase susceptibility to serious infection. Symptoms such as a sore throat, infection or fever should be reported promptly to your physician, and a white blood cell count should be done immediately. In nearly every case, when a person stops using the medication, the white blood cell count returns to normal. Very rarely, antithyroid drugs may cause severe liver problems, which can be detected by monitoring blood tests or joint problems characterized by joint pain and/or swelling. Your physician should be contacted if there is yellowing of the skin (jaundice), fever, loss of appetite or abdominal pain.
- Radioactive Iodine Treatment
Iodine is an essential ingredient in the production of thyroid hormone. Each molecule of thyroid hormone contains either four (T4) or three (T3) molecules of iodine. Since most overactive thyroid glands are quite hungry for iodine, it was discovered in the 1940s that the thyroid could be “tricked” into destroying itself by simply feeding it radioactive iodine. The radioactive iodine is given by mouth, usually in capsule form, and is quickly absorbed from the bowel. It then enters the thyroid cells from the bloodstream and gradually destroys them. Maximal benefit is usually noted within three to six months.
It is not possible to eliminate “just the right amount” of the diseased thyroid gland, since radioiodine eventually damages all thyroid cells. Therefore, most endocrinologists strive to completely destroy the diseased thyroid gland with a single dose of radioiodine. This results in the intentional development of an underactive thyroid state (hypothyroidism), which is easily, predictably and inexpensively corrected by lifelong daily use of oral thyroid hormone replacement therapy. Although every effort is made to calculate the correct dose of radioiodine for each patient, not every treatment will successfully correct the hyperthyroidism, particularly if the goiter is quite large and a second dose of radioactive iodine is occasionally needed.
Thousands of patients have received radioiodine treatment, including former President of the United States George H. W. Bush and his wife, Barbara. The treatment is a very safe, simple and reliably effective one. Because of this, it is considered by most thyroid specialists in the United States to be the treatment of choice for hyperthyroidism cases caused by overproduction of thyroid hormone.
- Surgical Removal of the Thyroid
Although seldom used now as the preferred treatment for hyperthyroidism, operating to remove most of the thyroid gland may occasionally be recommended in certain situations, such as a pregnant woman with severe uncontrolled disease in whom radioiodine would not be safe for the baby. Surgery usually leads to permanent hypothyroidism and lifelong thyroid hormone replacement therapy.
- Other Treatments
A drug from the class of beta-adrenergic blocking agents (which decrease the effects of excess thyroid hormone) may be used temporarily to control hyperthyroid symptoms until other therapies take effect. In cases where hyperthyroidism is caused by thyroiditis or excessive ingestion of either iodine or thyroid hormone, this may be the only type of treatment required. Iodine drops are prescribed when hyperthyroidism is severe or prior to undergoing surgery for Graves’ disease.
What is hypothyroidism ?
An underactive thyroid, or hypothyroidism, occurs when the thyroid gland produces less than the normal amount of thyroid hormone. The result is the “slowing down” of many bodily functions. Although hypothyroidism may be temporary, it usually is a permanent condition. Of the nearly 30 million people estimated to be suffering from thyroid dysfunction, most have hypothyroidism.
Causes of hypothyroidism
Autoimmune thyroiditis: The body’s immune system may produce a reaction in the thyroid gland that results in hypothyroidism and, most often, a goiter (enlargement of the thyroid). Other autoimmune diseases may be associated with this disorder, and additional family members may also be affected.
Central or pituitary hypothyroidism: TSH (Thyroid-stimulating hormone) is produced by the pituitary gland, which is located behind the nose at the base of the brain. Any destructive disease of the pituitary gland or hypothalamus, which sits just above the pituitary gland, may cause damage to the cells that secrete TSH, which stimulates the thyroid to produce normal amounts of thyroid hormone. This is a very rare cause of hypothyroidism.
Congenital hypothyroidism: An infant may be born with an inadequate amount of thyroid tissue or an enzyme defect that does not allow normal thyroid hormone production. If this condition is not treated promptly, physical stunting and/or mental damage (cretinism) may develop.
Medications: Lithium, high doses of iodine and amiodarone — an antiarrhythmic agent used for various types of cardiac dysrhythmias — can cause hypothyroidism.
Postpartum thyroiditis: Five percent to 10 percent of women develop mild to moderate hyperthyroidism within several months of giving birth. Hyperthyroidism in this condition usually lasts for approximately one to two months. It is often followed by several months of hypothyroidism, but most women will eventually recover normal thyroid function. In some cases, however, the thyroid gland does not heal, so the hypothyroidism becomes permanent and requires lifelong thyroid hormone replacement. This condition may occur again with subsequent pregnancies.
Radioactive iodine treatment: Hypothyroidism frequently develops as a desired therapeutic goal after the use of radioactive iodine treatment for hyperthyroidism.
Silent Thyroiditis: Transient (temporary) hyperthyroidism can be caused by silent thyroiditis, a condition which appears to be the same as postpartum thyroiditis but not related to pregnancy. It is not accompanied by a painful thyroid gland.
Subacute thyroiditis: This condition may follow a viral infection and is characterized by painful thyroid gland enlargement and inflammation, which results in the release of large amounts of thyroid hormone into the blood. Fortunately, this condition usually resolves spontaneously. The thyroid usually heals itself over several months, but often not before a temporary period of hypothyroidism occurs.
Thyroid surgery: Hypothyroidism may be related to surgery on the thyroid gland, especially if most of the thyroid has been removed.
What are the signs and symptoms of hypothyroidism ?
In its earliest stage, hypothyroidism may cause few symptoms, since the body has the ability to partially compensate for a failing thyroid gland by increasing the stimulation to it, much like pressing down on the accelerator when climbing a hill to keep the car going the same speed. As thyroid hormone production decreases and the body’s metabolism slows, a variety of features may result.
- Pervasive fatigue
- Drowsiness
- Forgetfulness
- Difficulty with learning
- Dry, brittle hair and nails
- Dry, itchy skin
- Puffy face
- Constipation
- Sore muscles
- Weight gain and fluid retention
- Heavy and/or irregular menstrual flow
- Increased frequency of miscarriages
- Increased sensitivity to many medications
How is hypothyroidism diagnosed ?
Characteristic symptoms and physical signs, which can be detected by a physician, can signal hypothyroidism. However, the condition may develop so slowly that many patients do not realize that their body has changed, so it is critically important to perform diagnostic laboratory tests to confirm the diagnosis and to determine the cause of hypothyroidism. A primary care physician may make the diagnosis of hypothyroidism, but assistance is often needed from an endocrinologist, a physician who is a specialist in thyroid diseases.
Tests
TSH (THYROID-STIMULATING HORMONE OR THYROTROPIN): An increased TSH level in the blood is the most accurate indicator of primary (non-pituitary) hypothyroidism. Production of this pituitary hormone is increased when the thyroid gland even slightly underproduces thyroid hormone.
ESTIMATES OF FREE THYROXINE: The active thyroid hormone in the blood. It is important to note that there is a range of free thyroxine levels in the blood of normal people, similar to the range for height, and that a value of free thyroxine that is “within normal limits” for the general population may not be appropriate for a particular individual.
THYROID ANTIBODIES: Indicates the likelihood of auto-immune thyroiditis being the cause of hypothyroidism.
How is hypothyroidism treated ?
Hypothyroidism is generally treated with a single daily dose of levothyroxine, given as a tablet. An experienced physician can prescribe the correct form and dosage to return the thyroid balance to normal. Older patients who may have underlying heart disease are usually started at a low dose and gradually increased while younger healthy patients can be started on full replacement doses at once. Thyroid hormone acts very slowly in some parts of the body, so it may take several months after treatment for some features to improve.
Levothyroxine tablets come in 12 different strengths, and it is essential to take them in a consistent manner every day. A dose of thyroid hormone that is too low may fail to prevent enlargement of the thyroid gland, allow symptoms of hypothyroidism to persist, and be associated with increased serum cholesterol levels, which may increase the risk for atherosclerosis and heart disease. A dose that is too high can cause symptoms of hyperthyroidism, create excessive strain on the heart, and lead to an increased risk of developing osteoporosis.
It is extremely important that women planning to become pregnant are kept well adjusted, since hypothyroidism can affect the development of the baby. During pregnancy, thyroid hormone replacement requirements often change, so more frequent monitoring is necessary. Various medications and supplements (particularly iron) may affect the absorption of thyroid hormone; therefore, the levels may need more frequent monitoring during illness or change in medication and supplements.
Thyroid hormone is critical for normal brain development in babies. Infants requiring thyroid hormone therapy should NOT be treated with purchased liquid suspensions, since the active hormone may deteriorate once dissolved and the baby could receive less thyroid hormone than necessary. Instead, infants with hypothyroidism should receive their thyroid hormone by crushing a single tablet daily of the correct dose and suspending it in one teaspoon of liquid and administering it properly.
Since most cases of hypothyroidism are permanent and often progressive, it is usually necessary to treat this condition throughout one’s lifetime. Periodic monitoring of TSH levels and clinical status are necessary to ensure that the proper dose is being given, since medication doses may have to be adjusted from time to time. Optimal adjustment of thyroid hormone dosage is critical, since the body is very sensitive to even small changes in thyroid hormone levels.
Appropriate management of hypothyroidism requires continued care by a physician experienced in the treatment of this condition.
Thyroid Cancer
The thyroid gland is located in the lower front of the neck, above the collarbones and below the voice box (larynx). Thyroid cancer (carcinoma) usually appears as a painless lump in this area 4. In most cases, the lump is only on one side, and the results of thyroid function tests (blood tests) are usually normal.
There are four main types of thyroid cancer (papillary, follicular, medullary and anaplastic), but the vast majority of cases are either papillary or follicular 4.
- Papillary thyroid cancer is the most common type of thyroid cancer, accounting for 70 to 80 percent of all cases. It is most commonly diagnosed in women 30-40 years old and most frequently spreads to cervical (neck) lymph nodes.
- Follicular thyroid cancer is the second most common type of thyroid cancer, accounting for 10 to 15 percent of cases. Although it usually does not spread, when it does it goes to the lungs and bones through the bloodstream.
- Most common types of thyroid cancer are “sporadic” or isolated, and not inherited. However, an uncommon type of thyroid cancer, medullary cancer, which makes up about five percent of all thyroid cancers, can be familial, or run in families. When medullary cancer is inherited as a familial disease, it can be detected by a genetic blood test. Unless the disease is inherited, your children will not be affected.
- Anaplastic thyroid cancer accounts for less than five percent of thyroid cancer patients. It is the most aggressive form of thyroid cancer and treatment is rarely effective.
Because the most common thyroid cancers, papillary and follicular, tend to grow slowly, usually do not spread beyond the neck and respond to treatment, most patients with thyroid cancers have excellent prognoses. For example, the 20-year survival of the most common type, papillary thyroid cancer, is almost 95 percent.
The estimated number of newly diagnosed thyroid cancer patients has continued an upward trend for more than 15 years! This represents an alarming and rapid percentage increase for any form of cancer, especially since most all other cancers are either stable or declining in their incidence rates. Fortunately, virtually the entire rate of increasing thyroid cancer patients annually is due to newly diagnosed papillary cancer, rather than other types of more aggressive thyroid cancer. The exact cause (or causes) is not clear; but, this rise in the incidence of papillary thyroid cancer has been attributed to better and earlier diagnostic imaging with ultrasound. However, other background environmental causes are difficult to exclude and there are continuing efforts to analyze this incidence trend.
What are the causes of thyroid cancer ?
As with many types of cancer, the specific reason for developing thyroid cancer remains a mystery in the vast majority of patients. Some major risk factors are:
- External radiation to the head or neck, especially during childhood
- Genetic predisposition (the influence of heredity), particularly for the medullary type of thyroid cancer
What are the signs and symptoms of thyroid cancer ?
Many patients with thyroid cancer have no symptoms and are found by chance to have a lump in the thyroid gland during a routine physical exam, or an imaging study of the neck done for unrelated reasons such as a carotid ultrasound, CT or MRI scan of the spine or chest. Other patients with thyroid cancer become aware of a gradually enlarging lump in the front portion of the neck, which usually moves with swallowing. Occasionally, the lump may cause a feeling of pressure. Obviously, finding a lump in the neck should be brought to the attention of your physician, even in the absence of other symptoms.
How is thyroid cancer diagnosed ?
First, your physician takes a detailed history and performs a careful physical examination, especially of the thyroid gland. The best diagnostic approach for a specific patient will be determined by your physician after careful consideration of all the facts. The tests available to your physician for evaluation of the thyroid lump include, but are not limited to, the following:
- Fine-needle aspiration biopsy: This is usually done first and, if positive, significantly reduces the need for more elaborate and expensive testing
- Ultrasonography – this may be required for guidance of the fine needle biopsy if the nodule is difficult to feel
- Thyroid scan – this can be done to see if the mass is capable of concentrating radioiodine, particularly in those patients with low TSH levels, who are likely to have hot nodules, which are almost always benign.
- Blood studies.
How is thyroid cancer treated ?
The great majority of patients with thyroid cancer have a disease that can be successfully treated. In order to ensure your chances for a successful outcome, it is important to receive treatment and follow-up care from those with a great deal of experience in the diagnosis and treatment of thyroid cancer. This is usually an endocrinologist, a doctor who specializes in hormone-related disorders.
What treatment will you require ?
Treatment depends on the type and extent of cancer. Treatment options include surgery, radioactive iodine, external radiation (see below), and chemotherapy. All patients require thyroid surgery and many receive radioiodine after surgery (see below).
What kind of surgery ?
Removal of part or all of the thyroid gland (thyroidectomy) is the first step in management. Lymph nodes with cancer in them are also removed. A surgeon who has experience with thyroid cancer is the best choice for performing your surgery.
You may be thinking, “Shouldn’t I be seeing an oncologist?” The answer is usually no. An endocrinologist is the physician who deals primarily with the diagnosis, treatment and follow-up of most patients with thyroid cancer. However, if/when standard therapy fails to control the progression of thyroid cancer and chemotherapy is being considered, then consultation with an oncologist is appropriate.
Will you require radiation ? If so, what at type ?
Conventional radiation therapy, the type that is generally used for cancer, is not used very often to treat thyroid cancer 4. It is reserved to treat thyroid cancer that cannot be removed surgically or eliminated with radioactive iodine. Fortunately, it is only required to treat a small minority of thyroid cancer cases. This type of radiation treatment is often referred to as external radiation therapy because the source of the radiation comes from outside the body.
Most often patients with thyroid cancer who require radiation treatment receive radioactive iodine. This type of radiation works internally once it enters your body. It is administered by either swallowing a capsule or drinking a radioactive liquid containing a radioactive form of iodine.
What are Thyroid Nodules ?
A thyroid nodule is a lump in or on the thyroid gland 5. Thyroid nodules are common, but are usually not diagnosed. They are detected in about six percent of women and one to two percent of men. They are 10 times as common in older individuals than in younger ones. Sometimes several nodules will develop in the same person. Any time a lump is discovered in thyroid tissue, the possibility of malignancy (cancer) must be considered. Fortunately, the vast majority of thyroid nodules are benign (not cancerous) 5.
What causes a thyroid nodule ?
Nodules can be caused by a simple overgrowth of “normal” thyroid tissue, fluid-filled cysts, inflammation (thyroiditis), or a tumor (either benign or cancerous).
What are the signs and symptoms of a thyroid nodule ?
Most patients with thyroid nodules have no symptoms whatsoever. Many are found by chance to have a lump in the thyroid gland on a routine physical exam or an imaging study of the neck done for unrelated reasons (for example, a CT or MRI scan of the spine or chest, a carotid ultrasound, etc.). In addition, a substantial number of nodules are first noticed by patients or those they know who see a lump in the front portion of the neck, which may or may not cause symptoms, such as a vague pressure sensation or discomfort when swallowing.
Obviously, finding a lump in the neck should be brought to the attention of your physician, even in the absence of symptoms.
How is a thyroid nodule diagnosed ?
THYROID SCAN: A thyroid scan is a picture of the thyroid gland taken after a small dose of a radioactive isotope normally concentrated by thyroid cells has been injected or swallowed. The scan tells whether the nodule is hyperfunctioning (a “hot” nodule), or taking up more radioactivity than normal thyroid tissue does; taking up the same amount as normal tissue (a “warm” nodule); or taking up less (a “cold” nodule). Because cancer is rarely found in hot nodules, a scan showing a hot nodule eliminates the need for fine needle biopsy. If a hot nodule causes hyperthyroidism, it can be treated with radioiodine or surgery.
THYROID NEEDLE BIOPSY: A thyroid fine needle biopsy employs a very thin needle, usually smaller than one used to draw blood and is a simple procedure that can be performed in the physician’s office. Many physicians numb the skin over the nodule prior to the biopsy, but it is not necessary to be put to sleep, and patients can usually return to work or home afterward with no ill effects. This test provides specific information about a particular patient’s nodule, information that no other test can offer short of surgery. Although the test is not perfect, a thyroid needle biopsy will provide sufficient information on which to base a treatment decision more than 75 percent of the time, eliminating the need for additional diagnostic studies.
Use of fine needle biopsy has drastically reduced the number of patients who have undergone unnecessary operations for benign nodules. However, about 10 to 20 percent of biopsy specimens are interpreted as inconclusive or inadequate; that is, the pathologist cannot be certain whether the nodule is cancerous or benign. This situation is particularly common with cystic (fluid-filled) nodules, which contain very few thyroid cells to examine, and with those nodules composed of clusters of thyroid or follicular cells that cannot be conclusively determined to be either benign or malignant. In such cases, a physician who is experienced with thyroid disease can use other criteria to make a decision about whether or not to operate. The fine needle biopsy can be repeated in those patients whose initial attempt failed to yield enough material to make a diagnosis. Many physicians use thyroid ultrasonography (ultrasound) to guide the needle’s placement.
THYROID ULTRASONOGRAPHY: Thyroid ultrasonography is a procedure for obtaining pictures of the thyroid gland by using high-frequency sound waves that pass through the skin and are reflected back to the machine to create detailed images of the thyroid. It can visualize nodules as small as two to three millimeters. Ultrasound distinguishes thyroid cysts (fluid-filled nodules) from solid nodules. Many nodules have both solid and cystic components, and very few purely cystic nodules occur. Recent advances in ultrasonography help physicians identify nodules that are more likely to be cancerous.
Thyroid ultrasonography is also utilized for guidance of a fine needle for aspirating thyroid nodules. Ultrasound guidance enables physicians to biopsy the nodule to obtain an adequate amount of material for interpretation. Such guidance allows the biopsy sample to be obtained from the solid portion of those nodules that are both solid and cystic, and it avoids getting a specimen from the surrounding normal thyroid tissue if the nodule is small. Even when a thyroid biopsy sample is reported as benign, the size of the nodule should be monitored. A thyroid ultrasound examination provides an objective and precise method for detection of a change in the size of the nodule. A nodule with a benign biopsy that is stable or decreasing in size is unlikely to be malignant or require surgical treatment.
How is a thyroid nodule treated ?
Your endocrinologist will use the tests mentioned above to arrive at a recommendation for optimal management of your nodule. Most patients who appear to have benign nodules require no specific treatment and can simply be followed by their physician. Some physicians prescribe levothyroxine with hopes of preventing nodule growth or reducing the size of cold nodules, while radioiodine may be used to treat hot nodules.
If cancer is suspected, surgical treatment will be recommended. The primary goal of therapy is to remove all thyroid nodules that are cancerous and, if malignancy is confirmed, remove the rest of the thyroid gland along with any abnormal lymph nodes. If surgery is not recommended, it is important to have regular follow-up of the nodule by a physician experienced in such an evaluation.
- Goiter – simple. Medline Plus. https://medlineplus.gov/ency/article/001178.htm[↩]
- What is Hashimoto’s thyroiditis ? American Association of Clinical Endocrinologists. https://www.empoweryourhealth.org/endocrine-conditions/thyroid/about_hashimotos_thyroiditis[↩]
- What is hyperthyroidism ? American Association of Clinical Endocrinologists. https://www.empoweryourhealth.org/thyroid/about_hyperthyroidism[↩]
- About Thyroid Cancer. American Association of Clinical Endocrinologists. https://www.empoweryourhealth.org/endocrine-conditions/thyroid/about_thyroid_cancer[↩][↩][↩]
- About Thyroid Nodules. American Association of Clinical Endocrinologists. https://www.empoweryourhealth.org/endocrine-conditions/thyroid/about_thyroid_nodules[↩][↩]