Contents
What is valley fever
Valley fever, also called coccidioidomycosis, is an infection caused by the fungus (or mold) Coccidioides. Valley fever, also known as “San Joaquin Valley Fever” or “Desert Rheumatism”, is the oldest of the major systemic fungal infection, considered to be one of the most infectious fungal diseases 1. The term “Valley fever” usually refers to Coccidioides infection in the lungs, but the infection can spread to other parts of the body in severe cases (this is called “disseminated coccidioidomycosis”). The Coccidioides fungus is known to live in the soil in the southwestern United States and parts of Mexico and Central and South America. The Coccidioides fungus was also recently found in south-central Washington. People can get Valley fever by breathing in the microscopic fungal spores from the air, although most people who breathe in the spores don’t get sick. Usually, people who get sick with Valley fever will get better on their own within weeks to months, but some people will need antifungal medication. Certain groups of people are at higher risk for becoming severely ill. It’s difficult to prevent exposure to Coccidioides in areas where it’s common in the environment, but people who are at higher risk for severe Valley fever should try to avoid breathing in large amounts of dust if they’re in these areas.
Two different species of Coccidioides have been recognized as causative agents of Valley fever: Coccidioides immitis and Coccidioides posadasii 2. The former appears to be limited to Southern California and Northern Mexico, while the latter occurs in all other endemic areas 1. Valley fever is almost exclusively prevalent in the Western Hemisphere, mostly in semiarid to arid life zones of Southwestern United States, north of Mexico, and some places in Central and South America 3. This fungal infection can cause a broad clinical spectrum: it can be asymptomatic, or it can range from a mild febrile illness to severe pulmonary manifestations or disseminated disease 4. In symptomatic patients, the most common manifestation is pulmonary disease, but many other organs can be affected. Cutaneous involvement of coccidioidomycosis entails a great significance because of its wide heterogeneity of clinical manifestations that can confuse the clinician.
It is estimated that 150,000 new Valley fever infections occur annually in areas of the Southwestern United States. Coccidioidomycosis is not a nationally reportable disease (it is only reported in the states of California and Arizona), therefore, the exact national incidence is unknown 1.
The incidence of Coccidioides spp. infection has increased in recent years. In a recent summary by the California Department of Health, it rose from 4.3 to 11.6 per 100,000 population from 2001 to 2010 5. In Arizona, it rose from 12 to 58.2 per 100,000 population over the same time period 6. Overall, it is estimated that 70% of all Coccidioidomycosis cases in USA occur in Arizona, and 25% in California 7.
In Latin America there are no reliable statistics. In Mexico, Valley fever is not a reportable disease and its true incidence is unknown, though most clinical case reports originate in the northern region of the country. Regional skin test surveys with coccidioidin in Mexico have shown positive skin reactions between 10% (in Tijuana and Baja California, 1991) and 93% (in Coahuila, 2005) 8. Historically, Argentina is of great interest, since it was the place where the first case of Coccidioidomycosis was reported in 1892. Even though the magnitude of infection in this country is unknown, a prevalence from 14.8 to 16% has been estimated according to some coccidioidin surveys 1.
The exponential rise in Valley fever, particularly in endemic places, has been attributed to many factors, including a growing population, migration of susceptible people to endemic areas, an increase in individuals of advanced age or with immunosuppression, soil disturbances caused by construction and businesses, and climate changes 9.
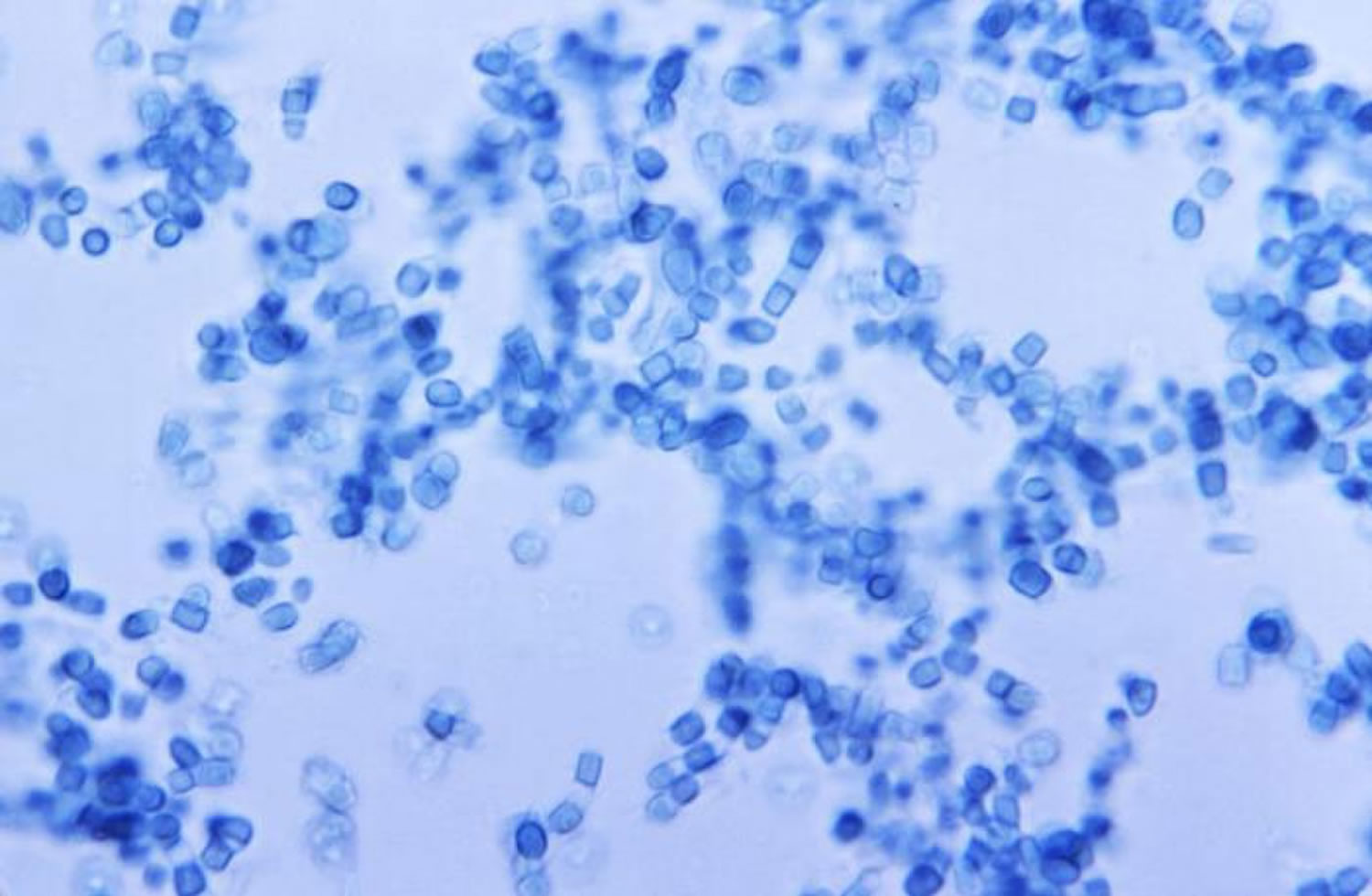
Figure 1. Valley fever Coccidioides fungus
Coccidioidomycosis is endemic in the Western hemisphere, particularly between the 40º latitudes north and south. There are two predominant regions, located in the Southwestern United States and the north of Mexico (Baja California, Chihuahua, Coahuila, Nuevo León and Tamaulipas, all of them corresponding to the Sonoran Desert zone) 10. Other areas have been described in central states of Mexico, as well as in Central and South America 11.
Coccidioides spp. are found in alkaline, sandy soils from semi-desert regions with hot summers, gentle winters, and annual rainfall between 10 and 50 cm. These fungi are usually found 10 to 30 cm beneath the surface 4.
Different types of cactus and bushes like Larrea tridentata (“creosote bush”) have been identified as common vegetation in the type of soil where this fungus lives 12.
Is Valley fever contagious?
No. The fungus that causes Valley fever, Coccidioides, can’t spread from the lungs between people or between people and animals. However, in extremely rare instances, a wound infection with Coccidioides can spread Valley fever to someone else,8 or the infection can be spread through an organ transplant with an infected organ 13.
Can my pets get Valley fever?
Yes. Pets, particularly dogs, can get valley fever, but it is not contagious between animals and people. Valley fever in dogs is similar to valley fever in humans.10 Like humans, many dogs that are exposed to Coccidioides never get sick. Dogs that do develop symptoms often have symptoms that include coughing, lack of energy, and weight loss. If you’re concerned about your pet’s risk of getting Valley fever or if you think that your pet has Valley fever, please talk to a veterinarian.
How can I prevent Valley fever?
It’s very difficult to avoid breathing in the fungus Coccidioides in areas where it’s common in the environment. People who live in these areas can try to avoid spending time in dusty places as much as possible. People who are at risk for severe Valley fever (such as people who have weakened immune systems, pregnant women, people who have diabetes, or people who are Black or Filipino) may be able to lower their chances of developing the infection by trying to avoid breathing in the fungal spores.
The following are some common-sense methods that may be helpful to avoid getting Valley fever. It’s important to know that although these steps are recommended, they haven’t been proven to prevent Valley fever.
- Try to avoid areas with a lot of dust like construction or excavation sites. If you can’t avoid these areas, wear an N95 respirator (a type of face mask) while you’re there.
- Stay inside during dust storms and close your windows.
- Avoid activities that involve close contact to dirt or dust, including yard work, gardening, and digging.
- Use air filtration measures indoors.
- Clean skin injuries well with soap and water to reduce the chances of developing a skin infection, especially if the wound was exposed to dirt or dust.
- Take preventive antifungal medication if your healthcare provider says you need it.
Is there a vaccine for Valley fever?
No. Currently, there is no vaccine to prevent Valley fever, but scientists have been trying to make one since the 1960s. Because people who’ve had Valley fever are usually protected from getting it again, a vaccine could make the body’s immune system think that it’s already had Valley fever, which would likely prevent a person from being able to get the infection.
Scientists have tried several different ways to make a Valley fever vaccine. When one version of the vaccine was tested on humans in the 1980s, it didn’t provide good protection, and it also caused people to develop side effects such as swelling at the injection site.12 Since then, scientists have been looking at ways to make a vaccine with different ingredients that will provide better protection against Valley fever and won’t cause side effects. Studies of these new vaccines are ongoing, so it’s possible that a vaccine to prevent Valley fever could become available in the future 14.
I’ve had Valley fever before; could I get it again?
Usually not. If you’ve already had Valley fever, your immune system will most likely protect you from getting it again. Some people can have the infection come back again (a relapse) after getting better the first time, but this is very rare.
Valley fever complications
Some people, especially pregnant women, people with weakened immune systems — such as those living with HIV/AIDS — and those of Filipino, Hispanic, African or Native American descent are at risk of developing a more severe form of coccidioidomycosis.
Complications of coccidioidomycosis may include:
- Severe pneumonia. Most people recover from coccidioidomycosis-related pneumonia without complications. Others, mainly Filipinos, Hispanics, blacks, Native Americans and those with weakened immune systems, may become seriously ill.
- Ruptured lung nodules. A small percentage of people develop thin-walled nodules (cavities) in their lungs. Many of these eventually disappear without causing any problems, but some may rupture, causing chest pain and difficulty breathing. A ruptured lung nodule might require the placement of a tube into the space around the lungs to remove the air or surgery to repair the damage.
- Disseminated disease. This is the most serious complication of coccidioidomycosis. If the fungus spreads (disseminates) throughout the body, it can cause problems ranging from skin ulcers and abscesses to bone lesions, severe joint pain, heart inflammation, urinary tract problems and meningitis — a potentially fatal infection of the membranes and fluid covering the brain and spinal cord.
What causes valley fever
Valley fever or coccidioidomycosis is caused by two distinct Coccidiodies species of soil fungus, Coccidioides immitis and Coccidioides posadaii. The former seems to be restricted to California, Arizona, and Baja California (Mexico), while the latter is found in the remaining endemic regions. Both species differ genetically, but they are clinically and microbiologically indistinguishable 10.
Like many other fungi, coccidioides species have a complex life cycle. In the soil, they grow as a mold with long filaments that break off into airborne spores when the soil is disturbed.
The spores are extremely small and can be carried hundreds of miles by the wind. Once inside the lungs, the spores reproduce, perpetuating the cycle of the disease.
The infection occurs through:
- Inhalation of spores during the course of outdoor activities (most common)
- Puncture wounds with infected objects
- Organ transplantation or sexual transmission from an infected individual (rare)
Risk factors for Valley fever
- Dust exposure in endemic areas. Anyone who inhales the spores that cause valley fever is at risk of infection. People who have jobs that expose them to dust are most at risk — construction, road and agricultural workers, ranchers, archeologists, and military personnel on field exercises.
- Race. For reasons that aren’t well-understood, Filipinos, Hispanics, blacks and Native Americans are more susceptible to developing serious infection with coccidioidomycosis than are whites. Certain groups are considered more vulnerable to coccidioidal infection. Racial predilection for Filipino or African American patients has been reported 15. These groups have higher rates for infection and dissemination of disease, ranging from 10 to 175 times higher than other ethnicities 16.
- Pregnancy. Pregnant women are also considered a high risk group for severe or disseminated disease, especially during later stages of pregnancy (usually during the third trimester), presumably associated with immunologic and hormonal factors 17. Immunosuppressed patients, predominantly those with alteration of T cell function, are at higher risk of infection and dissemination of disease. This group includes patients with human immunodeficiency virus (HIV), hematologic malignancies such as non-Hodgkin lymphoma (NHL) or chronic lymphocytic leukemia (CLL) 18, inflammatory arthritis, and diabetes 19. Organ transplantation and subsequent use of immunosuppressive agents have also been described as important risk factors for the disease.1
- Weakened immune system. Anyone with a weakened immune system is at increased risk of serious complications. This includes people living with AIDS or those being treated with steroids, chemotherapy and anti-rejection drugs after transplant surgery. People with certain autoimmune diseases, such as rheumatoid arthritis or Crohn’s disease, who are being treated with anti-tumor necrosis factor (TNF) drugs also have an increased risk of infection.
- Age. Older adults are more likely to develop valley fever. This may be because their immune systems are less robust or because they have other medical conditions that affect their overall health. Higher incidence rates have been documented among adults, ranging from 40-49 years in California, to >65 years in Arizona 6. It has a predilection for men over women (with an estimated incidence of 54% versus 46% of cases in Arizona, respectively) 20. Gender disparities are thought to be mostly related to occupational factors (since men tend to participate more in high risk, outdoor activities); however, males also have greater risk for dissemination, suggesting a hormonal or genetic component 21.
Valley fever symptoms
Symptoms of Valley fever may appear between 1 and 3 weeks after a person breathes in the fungal spores. Valley fever is the initial form of coccidioidomycosis infection. This initial, acute illness can develop into a more serious disease, including chronic and disseminated coccidioidomycosis.
Many people who are exposed to the fungus Coccidioides never have symptoms 22. Other people may have flu-like symptoms that go usually away on their own after weeks to months. If your symptoms last for more than a week, contact your healthcare provider.
Symptomatic persons (40% of cases) usually present 1 to 3 weeks after exposure with fatigue, cough, dyspnea, headache, night sweats, myalgias, and rash 23. Primary pulmonary disease is often self-limiting, but some patients fail to recover and develop complications or chronic pulmonary disease (approximately 5 to 10% of cases). Disseminated disease occurs in an estimated 1% of cases (higher rates of dissemination are observed in certain risk groups), with bones/joints, soft tissues, and meninges most commonly affected.
Approximately 5 to 10% of people who get Valley fever will develop serious or long-term problems in their lungs 24. In an even smaller percent of people (about 1%), the infection spreads from the lungs to other parts of the body, such as the central nervous system (brain and spinal cord), skin, or bones and joints 24.
Acute coccidioidomycosis (valley fever)
Primary disease generally affects the lungs, which is by far the most common site involved. In this case, it is acquired by direct inhalation of fungal spores.
The initial, or acute, form of coccidioidomycosis is often mild, with few, if any, symptoms. When signs and symptoms do occur, they appear one to three weeks after exposure. They tend to resemble those of the flu, and can range from minor to severe, including:
- Fatigue (tiredness)
- Cough
- Fever
- Shortness of breath
- Headache
- Night sweats
- Muscle aches or joint pain
- Red spotty rash on upper body or legs
Primary pulmonary coccidioidomycosis is the most common infection. Up to 60% of the affected individuals are usually asymptomatic, and the remaining 40% can present with pulmonary and systemic manifestations that usually appear one to three weeks after inhalation of fungal spores 25. Most symptomatic patients can develop a flu-like syndrome (fever, headache, profound fatigue, cough, pleuritic pain, and arthralgias), commonly known as “The Valley Fever of San Joaquin” 26. Cough and fever appear in 75% of patients; the skin is affected in up to 50% of cases during this acute phase, most commonly in the form of a non-pruritic papular rash, erythema nodosum, and erythema multiforme.1 Radiographically, it may be similar to other forms of pneumonia, showing lobar segmental or subsegmental infiltrates. Hilar adenopathy and pleural effusions may also be observed 27.
In extremely rare cases, the fungal spores can enter the skin through a cut, wound, or splinter and cause a skin infection 28.
The rash that sometimes accompanies valley fever is made up of painful red bumps that may later turn brown. The rash mainly appears on your lower legs, but sometimes on your chest, arms and back. Others may have a raised red rash with blisters or eruptions that look like pimples.
If you don’t become ill from valley fever, you may only find out you’ve been infected when you later have a positive skin or blood test or when small areas of residual infection (nodules) in the lungs show up on a routine chest X-ray. Although the nodules typically don’t cause problems, they can look like cancer on X-rays.
If you do develop symptoms, especially severe ones, the course of the disease is highly variable. It can take months to fully recover, and fatigue and joint aches can last even longer. The severity of the disease depends on several factors, including your overall health and the number of fungus spores you inhale.
Cutaneous Manifestations of coccidioidomycosis
Skin manifestations of Valley fever can be seen in three different scenarios of coccidioidal infection:
- They can be a part of the acute pulmonary infection and present as an “acute pulmonary exanthema”;
- They can reflect the presence of a disseminated infection (secondary cutaneous infection); or, in rare occasions,
- They can represent a primary infection due to direct inoculation (primary cutaneous infection).
Despite their variability and heterogeneity, cutaneous manifestations of Coccidioides immitis differ between these three forms of disease 29.
Cutaneous manifestations can be further categorized as reactive and organism-specific 30. Reactive manifestations do not contain visible microorganisms, and occur during acute primary pulmonary infection in up to 50% of patients 31. They include erythema nodosum, erythema multiforme, acute generalized exanthema, reactive interstitial granulomatous dermatitis and Sweet’s syndrome 32. Organism-specific manifestations present with lesions that contain the organism, which can be identified in cultures or histopathology; they result from hematogenous spread of a primary pulmonary infection (secondary cutaneous disease) or, very rarely, as a result of direct trauma to the skin (primary cutaneous disease) 31.
Sweet’s Syndrome, also called “acute febrile neutrophilic dermatosis” has been reported as a very rare feature of coccidioidomycosis, with few cases reported in the literature 33. It is characterized by an acute eruption, consisting of painful erythematous, well-demarcated papules and plaques, in association with fever and neutrophilic leukocytosis. In some cases, vesicles and pustules can be seen. The common topography involves the arms, face and neck, and the biopsy specimen shows a diffuse inflammatory infiltrate in the dermis, with neutrophils and leukocytoclastic debris. Even though the mainstay of treatment for Sweet’s syndrome are corticosteroids, they are not recommended in the setting of coccidioidal infection, since they could exacerbate the condition 33.
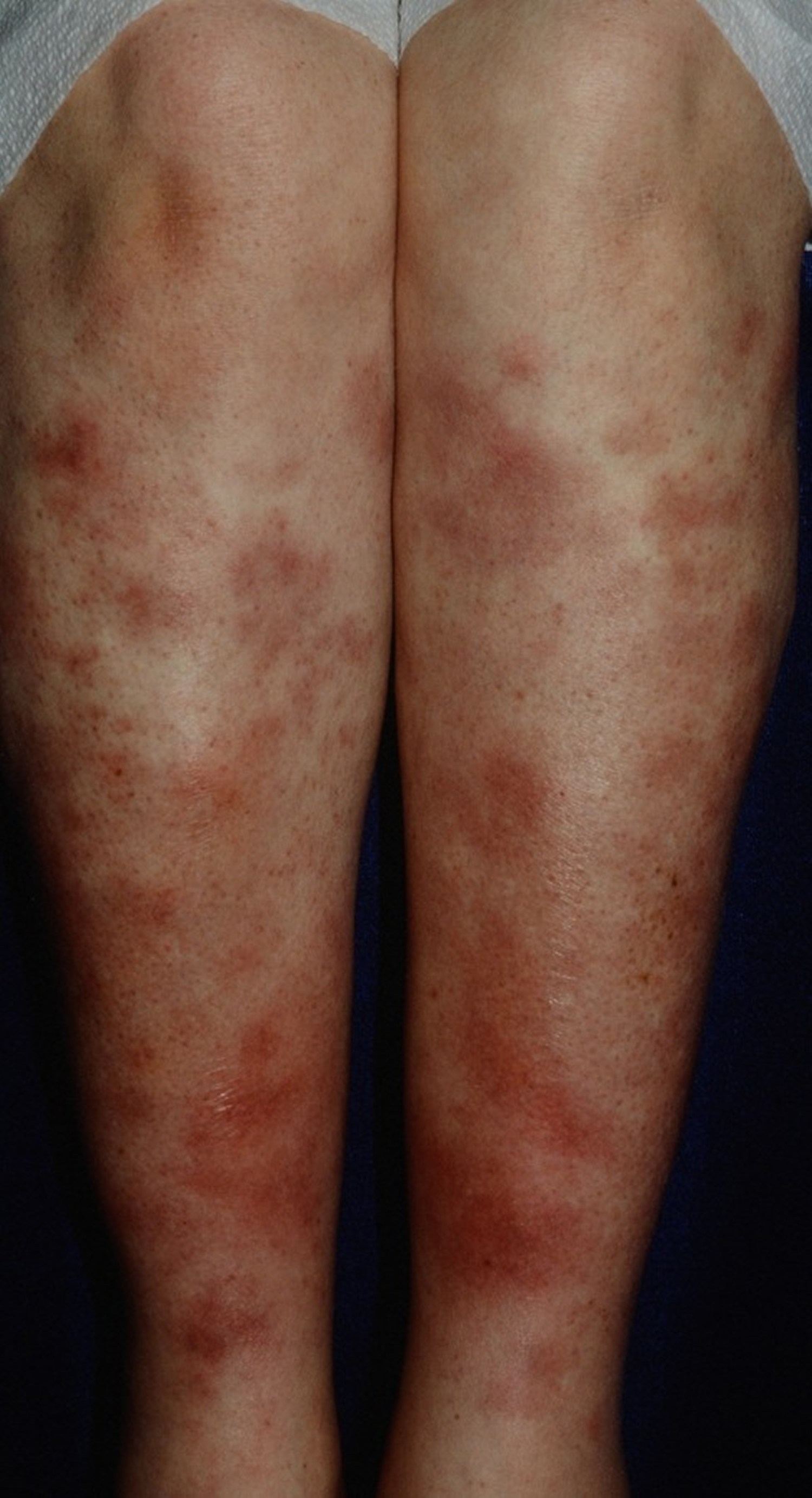
Figure 2. Valley fever rash – Erythema nodosum, considered the most frequent reactive manifestation and commonly involving the lower extremities
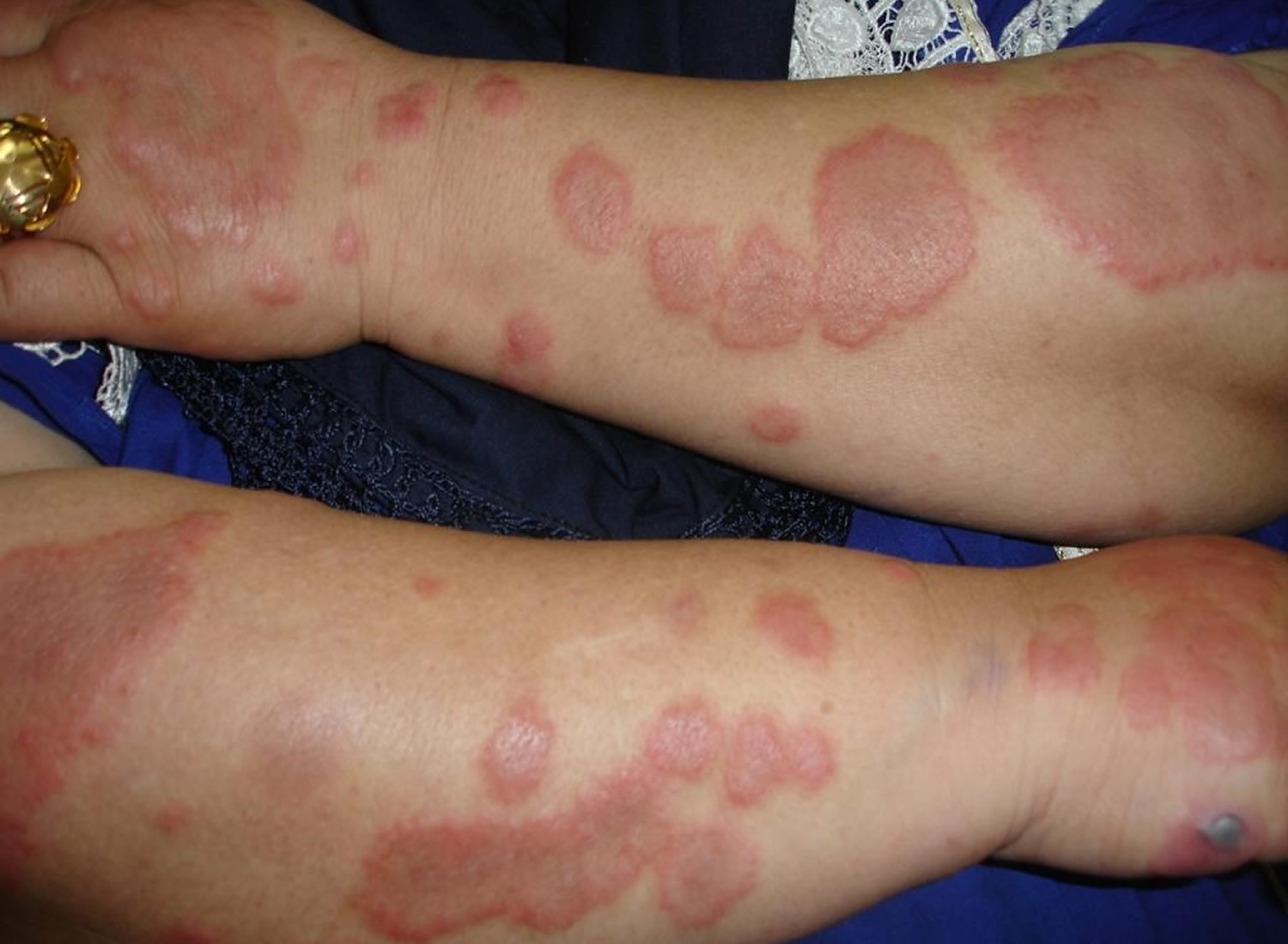
Figure 3. Valley fever rash – Erythema multiforme, another common manifestation of coccidioidomycosis, characterized by target-like lesions
Chronic coccidioidomycosis
If the initial coccidioidomycosis infection doesn’t completely resolve, it may progress to a chronic form of pneumonia. This complication is most common in people with weakened immune systems.
Chronic and progressive pneumonia may occur in a small percentage of cases. The patients present symptoms that last for more than 3 months (persistent cough, hemoptysis, and weight loss). Radiographically, cavitations and solitary pulmonary nodules are detected 34.
Signs and symptoms include:
- Low-grade fever
- Weight loss
- Cough
- Chest pain
- Blood-tinged sputum (matter discharged during coughing)
- Nodules in the lungs.
Disseminated coccidioidomycosis
The most serious form of the disease, disseminated coccidioidomycosis, occurs when the infection spreads (disseminates) beyond the lungs to other parts of the body. Most often these parts include the skin, bones, liver, brain, heart, and the membranes that protect the brain and spinal cord (meninges).
Disseminated disease occurs in 1% to 5% of all patients affected by coccidioidomycosis, and it usually is clinically manifested within 2 years of exposure. Dissemination can affect any organ, with the skin, central nervous system and musculoskeletal system being reported as the most prevalent 35. Osteoarticular infection is usually monoarticular (affecting the knee) or it can affect the vertebrae. These can be difficult to treat. Central nervous system involvement most commonly presents as meningeal infection, which can be fatal if left untreated (95% of patients die within 2 years of diagnosis), and requires life-long therapy 36.
Some factors that entail a higher risk for dissemination of coccidioidomycosis are Filipino and African patients, HIV/AIDS, organ transplantation, use of immunosuppressive medications, older age, diabetes and late-stage pregnancy 19.
The signs and symptoms of disseminated disease depend on which parts of your body are affected and may include:
- Nodules, ulcers and skin lesions that are more serious than the rash that sometimes occurs with other forms of the disease
- Painful lesions in the skull, spine or other bones
- Painful, swollen joints, especially in the knees or ankles
- Meningitis — an infection of the membranes and fluid surrounding the brain and spinal cord.
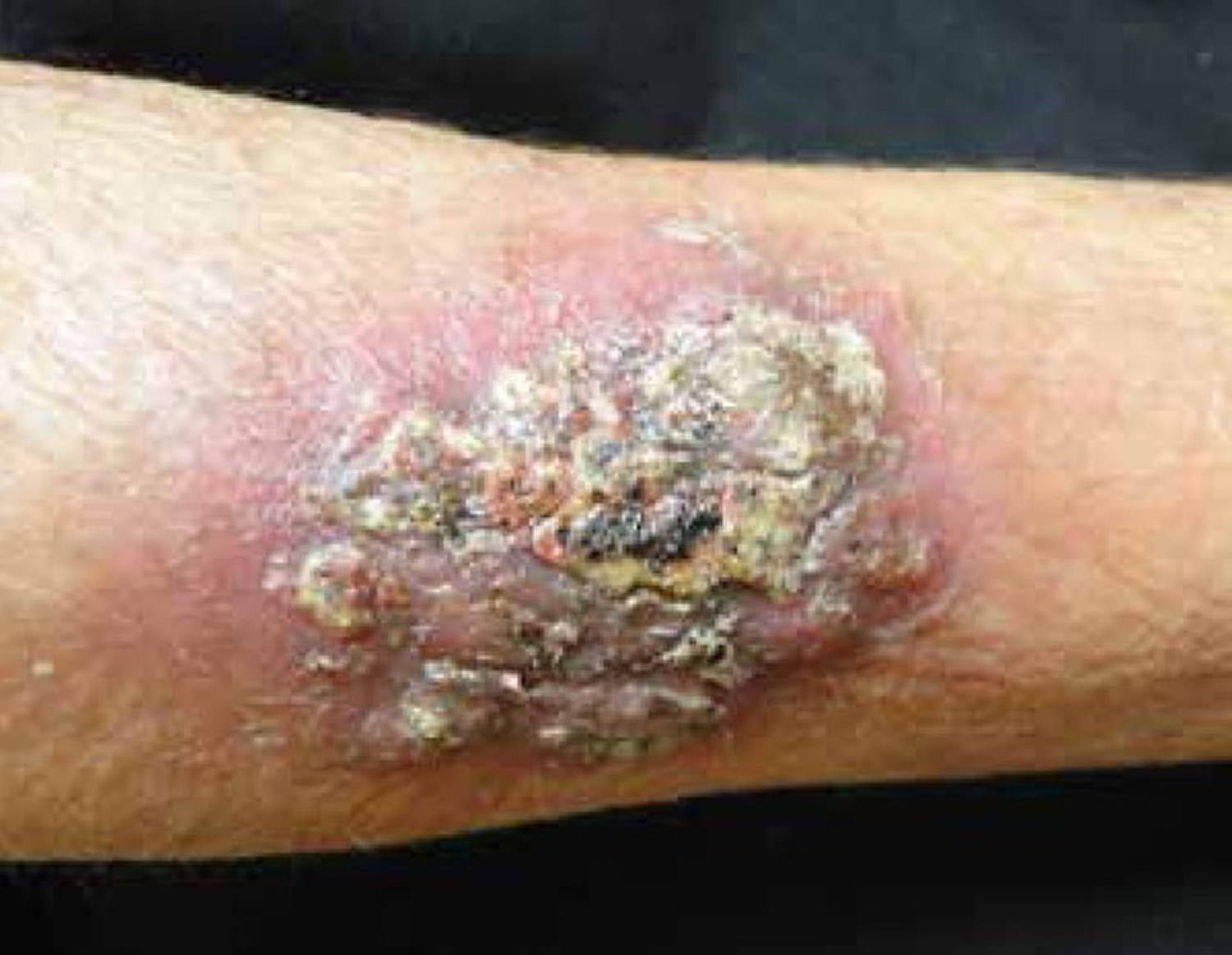
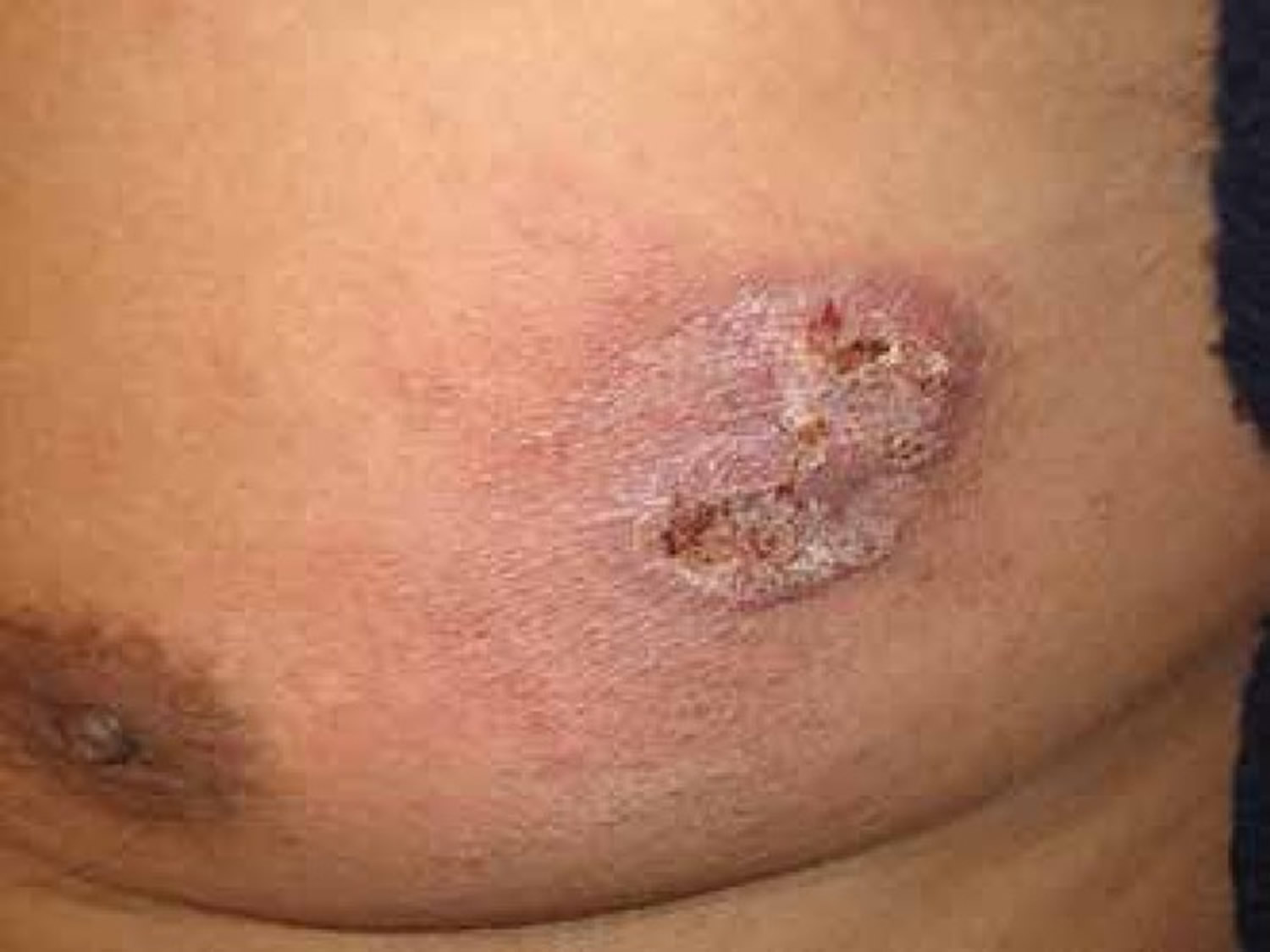
Figure 4. Disseminated coccidioidomycosis presenting as an erythematous, crusted plaque in the upper extremity
Figure 5. Disseminated coccidioidomycosis presenting as an erythemato-squamous plaque on the upper chest
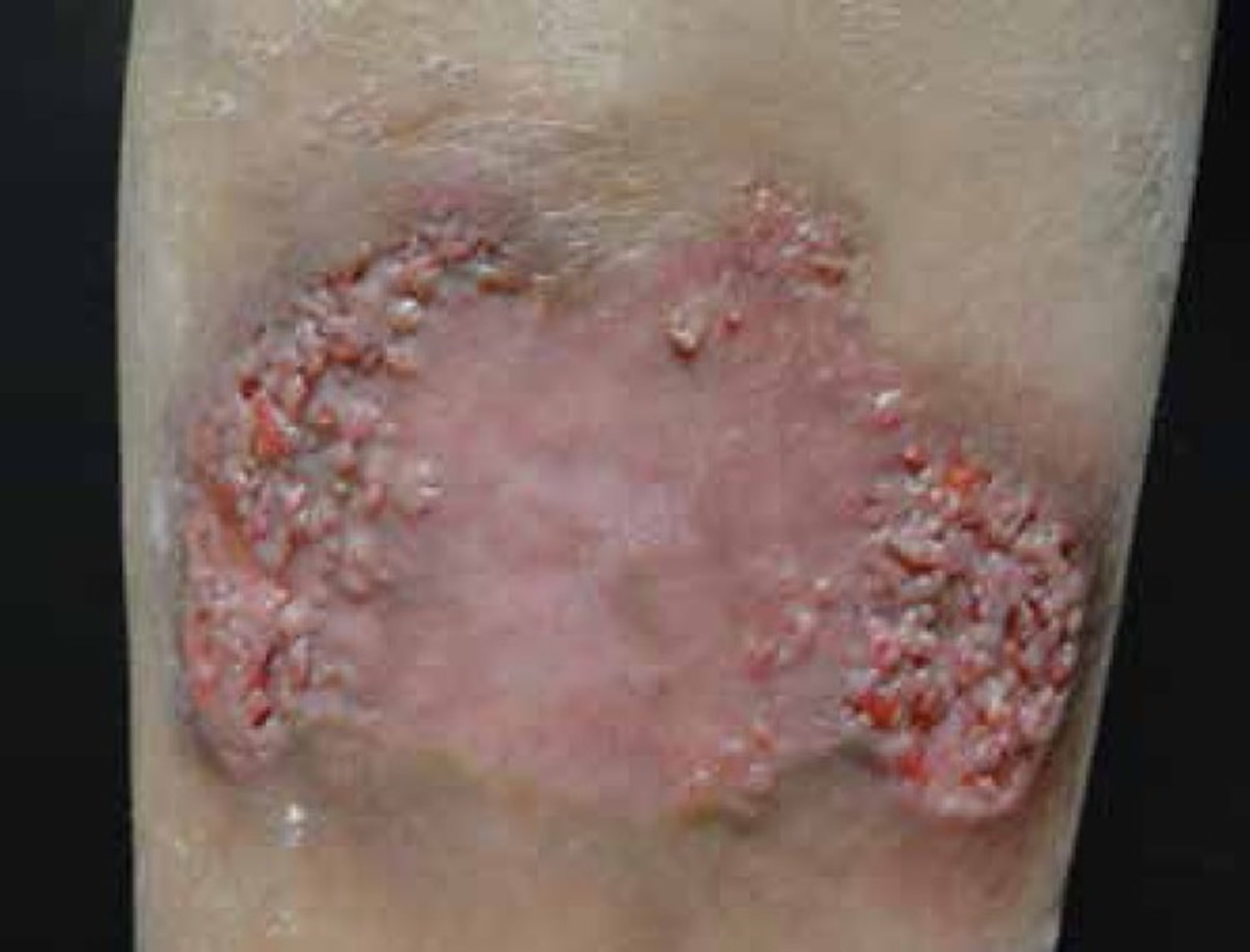
Figure 6. Verrucous plaques are also a common manifestation of disseminated coccidioidomycosis
Valley fever diagnosis
Valley fever is difficult to diagnose solely on the basis of signs and symptoms, because they’re usually vague and overlap with symptoms that occur in other illnesses. Even a chest X-ray can’t distinguish valley fever from other lung diseases.
A definitive diagnosis depends on finding evidence of coccidioides organisms in tissue, blood or other body secretions. For that reason, you’re likely to have one or more of the following tests:
- Sputum smear or culture. These tests check a sample of the matter that’s discharged while coughing (sputum) for the presence of coccidioides organisms.
- Blood tests. Through a blood test, your doctor can check for antibodies against the fungus that causes valley fever.
- Serologic tests to detect IgM and IgG antibodies are most often used to diagnose coccidioidomycosis 37.
Serology:
- Enzyme immunoassay (EIA): A very sensitive and commonly used method for diagnosing coccidioidomycosis. Two Coccidioides EIAs are currently available
- Premier ® Coccidioides EIA – Meridian Bioscience, Inc.
- Coccidioides Antibody Enzyme Immunoassay – Immuno Mycologics, Inc. (IMMY)
- Immunodiffusion (ID): detects IgM antibodies; positive early in the course of infection.
- Complement Fixation (CF): detects IgG antibodies and allows for assessment of disease severity.
Culture: Can be performed on tissue and respiratory specimens; however, sputum can be difficult to obtain for culture since patients’ coughs are often non-productive.
Microscopy: for detection of spherules in tissue or respiratory secretions; low sensitivity.
Urinary antigen detection: Not widely used, but may have some utility in diagnosing coccidioidomycosis in immunocompromised patients with severe forms of the disease 37.
Polymerase Chain Reaction (PCR): PCR for detection of Coccidioides directly from clinical specimens is still experimental, but promising 38.
Valley fever treatment
For many people, the symptoms of Valley fever will go away within a few months without any treatment. Healthcare providers choose to prescribe antifungal medication for some people to try to reduce the severity of symptoms or prevent the infection from getting worse. Antifungal medication is typically given to people who are at higher risk for developing severe Valley fever.
Certain groups of people may be at higher risk for developing the severe forms of Valley fever, such as:
- People who have weakened immune systems, for example, people who:
- Pregnant women 42
- People who have diabetes 43
- People who are Black 44 or Filipino 45
The treatment is usually 3 to 6 months of fluconazole or another type of antifungal medication. There are no over-the-counter medications to treat Valley fever. If you have Valley fever, you should talk to your healthcare provider about whether you need treatment. The healthcare provider who diagnoses you with Valley fever may suggest that you see other healthcare providers who specialize in treating Valley fever.
People who have severe lung infections or infections that have spread to other parts of the body always need antifungal treatment and may need to stay in the hospital. For these types of infections, the course of treatment is usually longer than 6 months. Valley fever that develops into meningitis is fatal if it’s not treated, so lifelong antifungal treatment is necessary for those cases.
Rest
Most people with acute valley fever don’t require treatment. Even when symptoms are severe, the best therapy for otherwise healthy adults is often bed rest and fluids — the same approach used for colds and the flu. Still, doctors carefully monitor people with valley fever.
Antifungal medications
If symptoms don’t improve or become worse or if you are at increased risk of complications, your doctor may prescribe an antifungal medication, such as fluconazole. Antifungal medications are also used for people with chronic or disseminated disease.
In general, the antifungal drugs fluconazole (Diflucan) or itraconazole (Sporanox, Onmel) are used for all but the most serious forms of coccidioidomycosis disease.
All antifungals can have serious side effects. However, these side effects usually go away once the medication is stopped. Possible side effects of fluconazole and itraconazole are nausea, vomiting, abdominal pain and diarrhea.
More serious infection may be treated initially with an intravenous antifungal medication such as amphotericin B (Abelcet, Amphotec, others).
Two newer medications — voriconazole and posaconazole (Noxafil) — may also be used to treat more serious infections.
Antifungals control the fungus, but sometimes don’t destroy it, and relapses may occur. For many people, a single bout of valley fever results in lifelong immunity, but the disease can be reactivated, or you can be reinfected if your immune system is significantly weakened.
- Laniado-Laborín R, Alcandar-Schramm JM, Cazares-Adame R. Coccidioidomycosis: An Update. Curr Fungal Infect Rep. 2012;6:113–120.[↩][↩][↩][↩]
- Garcia SCG, Alanis JCS, Flores MG, Gonzalez SEG, Cabrera LV, Candiani JO. Coccidioidomycosis and the skin: a comprehensive review. Anais Brasileiros de Dermatologia. 2015;90(5):610-619. doi:10.1590/abd1806-4841.20153805. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4631225/[↩]
- Sharma S, Thompson III GR. How I Treat Coccidioidomycosis. Curr Fungal Infect Rep. 2012;7:29–35.[↩]
- Brown J, Benedict K, Park BJ, Thompson 3rd GR. Coccidioidomycosis: epidemiology. Clin Epidemiol. 2013;5:185–197. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3702223/[↩][↩]
- Centers for Disease Control and Prevention Increase in Coccidioidomycosis – California, 2000-2007. MMWR Morb Mortal Wkly Rep. 2009;58:105–109. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5805a1.htm[↩]
- Sunenshine RH, Anderson S, Erhart L, Vossbrink A, Kelly PC, Engelthaler D, et al. Public health surveillance for coccidioidomycosis in Arizona. Ann N Y Acad Sci. 2007;1111:96–102. https://www.ncbi.nlm.nih.gov/pubmed/17513465[↩][↩]
- Valley Fever (Coccidioidomycosis) Statistics. https://www.cdc.gov/fungal/diseases/coccidioidomycosis/statistics.html[↩]
- Mondragón-González R, Méndez-Tovar LJ, Bernal-Vázquez E, Hernández-Hernández F, López-Martínez R, Manzano-Gayosso P, et al. Detección de infección por Coccidioides immitis en zonas del estado de Coahuila, México. Rev Argent Microbiol. 2005;37:135–138. https://www.ncbi.nlm.nih.gov/pubmed/16323661[↩]
- Hector RF, Laniado-Laborin R. Coccidioidomycosis–a fungal disease of the Americas. PLoS Med. 2005;2 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC545195/[↩]
- Murthy MH, Blair JE. Coccidioidomycosis. Curr Fungal Infect Rep. 2009;3:7–14.[↩][↩]
- Laniado-Laborín R. Coccidioidomycosis and other endemic mycoses in Mexico. Rev Iberoam Micol. 2007;24:249–258. https://www.ncbi.nlm.nih.gov/pubmed/18095755[↩]
- Welsh O, Vera-Cabrera L, Rendon A, Gonzalez G, Bonifaz A. Coccidioidomycosis. Clin Dermatol. 2012;30:573–591. https://www.ncbi.nlm.nih.gov/pubmed/23068145[↩]
- Dierberg KL, Marr KA, Subramanian A, Nace H, Desai N, Locke JE, et al. Donor-derived organ transplant transmission of coccidioidomycosis. Transpl Infect Dis. 2012 Jun;14(3):300-4.[↩]
- Cole GT, Hurtgen BJ, Hung CY. Progress Toward a Human Vaccine Against Coccidioidomycosis. Curr Fungal Infect Rep. 2012 Dec 1;6(4):235-44.[↩]
- Ruddy BE, Mayer AP, Ko MG, Labonte HR, Borovansky JA, Boroff ES, et al. Coccidioidomycosis in African Americans. Mayo Clin Proc. 2011;86:63–69. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3012635/[↩]
- Rosenstein NE, Emery KW, Werner SB, Kao A, Johnson R, Rogers D, et al. Risk factors for severe pulmonary and disseminated coccidioidomycosis: Kern County, California, 1995-1996. Clin Infect Dis. 2001;32:708–715. https://www.ncbi.nlm.nih.gov/pubmed/11229838[↩]
- Bercovitch RS, Catanzaro A, Schwartz BS, Pappagianis D, Watts DH, Ampel NM. Coccidioidomycosis during pregnancy: a review and recommendations for management. Clin Infect Dis. 2011;53:363–368. https://www.ncbi.nlm.nih.gov/pubmed/21810749[↩]
- Blair JE, Smilack JD, Caples SM. Coccidioidomycosis in patients with hematologic malignancies. Arch Intern Med. 2005;165:113–117. https://www.ncbi.nlm.nih.gov/pubmed/15642886[↩]
- Santelli AC, Blair JE, Roust LR. Coccidioidomycosis in patients with diabetes mellitus. Am J Med. 2006;119:964–969. https://www.ncbi.nlm.nih.gov/pubmed/17071165[↩][↩]
- Tsang CA, Anderson SM, Imholte SB, Erhart LM, Chen S, Park BJ, et al. Enhanced surveillance of coccidioidomycosis, Arizona, USA, 2007-2008. Emerg Infect Dis. 2010;16:1738–1744. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3294516/[↩]
- Drutz DJ, Huppert M, Sun SH, McGuire WL. Human sex hormones stimulate the growth and maturation of Coccidioides immitis. Infect Immun. 1981;32:897–907. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC351527/[↩]
- Smith CE, Whiting EG, et al. The use of coccidioidin. American review of tuberculosis 1948;57:330-60.[↩]
- Thompson GR, 3rd. Pulmonary coccidioidomycosis. Semin Respir Crit Care Med 2011;32:754-63.[↩]
- Thompson GR, 3rd. Pulmonary coccidioidomycosis. Seminars in respiratory and critical care medicine 2011;32:754-63.[↩][↩]
- Parish JM, Blair JE. Coccidioidomycosis. Mayo Clin Proc. 2008;83:343–348. https://www.ncbi.nlm.nih.gov/pubmed/18316002[↩]
- Crum NF, Lederman ER, Stafford CM, Parrish JS, Wallace MR. Coccidioidomycosis: a descriptive survey of a reemerging disease. Clinical characteristics and current controversies. Medicine (Baltimore) 2004;83:149–175. https://www.ncbi.nlm.nih.gov/pubmed/15118543[↩]
- Batra P. Pulmonary coccidioidomycosis. J Thorac Imaging. 1992;7:29–38. https://www.ncbi.nlm.nih.gov/pubmed/1404543[↩]
- Chang A, Tung RC, McGillis TS, Bergfeld WF, Taylor JS. Primary cutaneous coccidioidomycosis. J Am Acad Dermatol. 2003 Nov;49(5):944-9.[↩]
- Arenas R. Micología médica ilustrada. 3. ed. Mexico: McGraw Hill; 2008.[↩]
- DiCaudo DJ, Connolly SM. Interstitial granulomatous dermatitis associated with pulmonary coccidioidomycosis. J Am Acad Dermatol. 2001;45:840–845. https://www.ncbi.nlm.nih.gov/pubmed/11712027[↩]
- Blair JE. State-of-the-art treatment of coccidioidomycosis: skin and soft-tissue infections. Ann N Y Acad Sci. 2007;1111:411–421. https://www.ncbi.nlm.nih.gov/pubmed/17332079[↩][↩]
- DiCaudo DJ. Coccidioidomycosis: a review and update. J Am Acad Dermatol. 2006;55:929–942. https://www.ncbi.nlm.nih.gov/pubmed/17110216[↩]
- DiCaudo DJ, Ortiz KJ, Mengden SJ, Lim KK. Sweet syndrome (acute febrile neutrophilic dermatosis) associated with pulmonary coccidioidomycosis. Arch Dermatol. 2005;141:881–884. https://www.ncbi.nlm.nih.gov/pubmed/16027305[↩][↩]
- Chiller TM, Galgiani JN, Stevens DA. Coccidioidomycosis. Infect Dis Clin North Am. 2003 Mar;17(1):41–57. viii–viii. https://www.ncbi.nlm.nih.gov/pubmed/12751260[↩]
- Parish JM, Blair JE. Coccidioidomycosis. Mayo Clin Proc. 2008;83:343–348 https://www.ncbi.nlm.nih.gov/pubmed/18316002[↩]
- Dewsnup DH, Galgiani JN, Graybill JR, Diaz M, Rendon A, Cloud GA, et al. Is it ever safe to stop azole therapy for Coccidioides immitis meningitis. Ann Intern Med. 1996;124:305–310. https://www.ncbi.nlm.nih.gov/pubmed/8554225[↩]
- Durkin M, P C, T K, R M, BM K, D B, et al. Diagnosis of Coccidioidomycosis with Use of the Coccidioides Antigen Enzyme Immunoassay. Clin Infect Dis. 2008;47:e69-73.[↩][↩]
- Sheff KW, York ER, Driebe EM, et al. Development of a rapid, cost-effective TaqMan Real-Time PCR Assay for identification and differentiation of Coccidioides immitis and Coccidioides posadasii. Med Mycol 2009;48:466-469.[↩]
- Woods CW, McRill C, Plikaytis BD, Rosenstein NE, Mosley D, Boyd D, et al. Coccidioidomycosis in human immunodeficiency virus-infected persons in Arizona, 1994-1997: incidence, risk factors, and prevention. J Infect Dis. 2000 Apr;181(4):1428-34.[↩]
- Blair JE, Logan JL. Coccidioidomycosis in solid organ transplantation. Clin Infect Dis. 2001 Nov 1;33(9):1536-44.[↩]
- Bergstrom L, Yocum DE, Ampel NM, Villanueva I, Lisse J, Gluck O, et al. Increased risk of coccidioidomycosis in patients treated with tumor necrosis factor alpha antagonists. Arthritis and rheumatism. 2004 Jun;50(6):1959-66.[↩]
- Bercovitch RS, Catanzaro A, Schwartz BS, Pappagianis D, Watts DH, Ampel NM. Coccidioidomycosis during pregnancy: a review and recommendations for management. Clin Infect Dis. 2011 Aug;53(4):363-8.[↩]
- Rosenstein NE, Emery KW, Werner SB, Kao A, Johnson R, Rogers D, et al. Risk factors for severe pulmonary and disseminated coccidioidomycosis: Kern County, California, 1995-1996. Clin Infect Dis. 2001 Mar 1;32(5):708-15.[↩]
- Durry E, Pappagianis D, Werner SB, Hutwagner L, Sun RK, Maurer M, et al. Coccidioidomycosis in Tulare County, California, 1991: reemergence of an endemic disease. J Med Vet Mycol. 1997 Sep-Oct;35(5):321-6.[↩]
- Crum NF, Lederman ER, Stafford CM, Parrish JS, Wallace MR. Coccidioidomycosis: a descriptive survey of a reemerging disease. Clinical characteristics and current controversies. Medicine. 2004 May;83(3):149-75.[↩]