Contents
- What is Osteoporosis
What is Osteoporosis
Osteoporosis literally means “porous bone.” Having osteoporosis raises the risk of experiencing fractures 1, 2. This disease is characterized by too little bone formation, excessive bone loss, or a combination of both, leading to bone fragility and an increased risk of fractures of the hip, spine and wrist 3.
Bone is continually being formed and resorbed. Normally, bone formation and resorption are closely balanced. Osteoblasts (cells that make the organic matrix of bone and then mineralize bone) and osteoclasts (cells that resorb bone) are regulated by parathyroid hormone (PTH), calcitonin, estrogen, vitamin D, various cytokines, and other local factors such as prostaglandins 4.
What is Bone
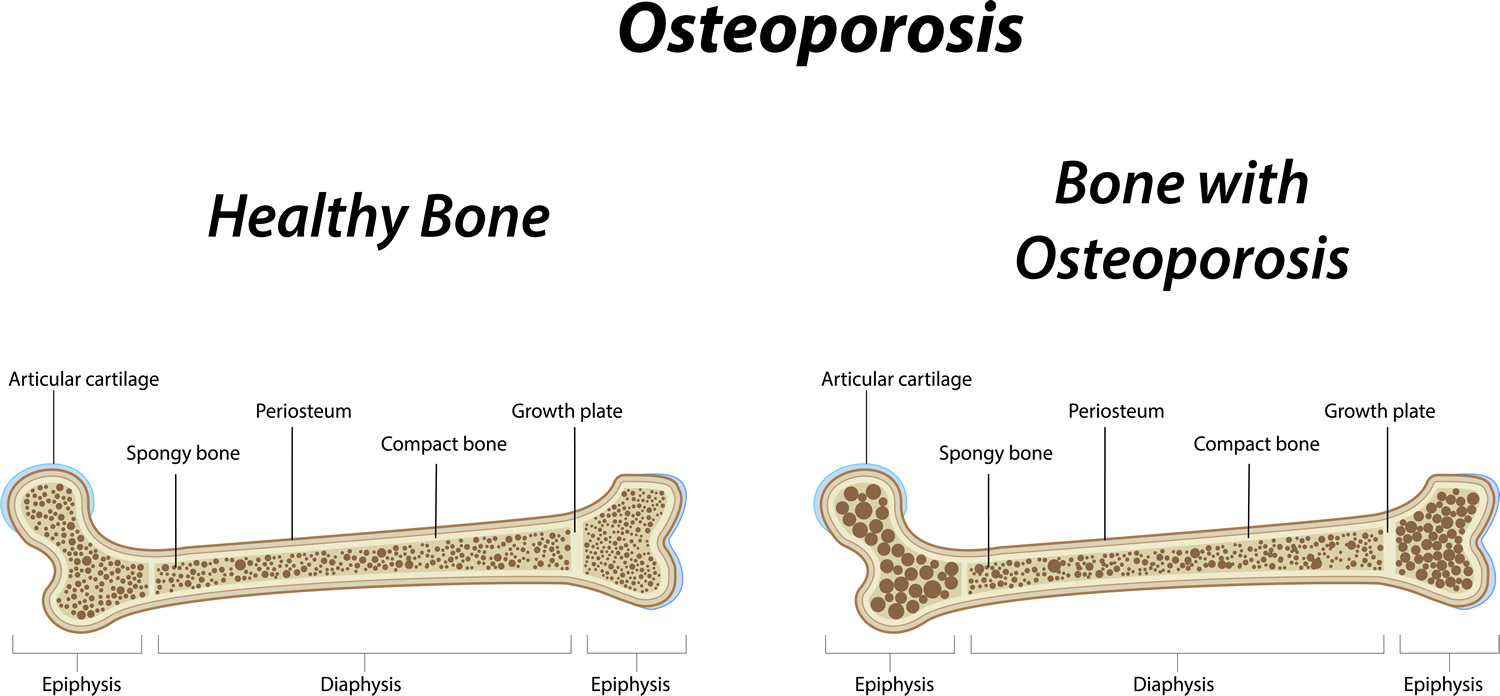
To understand osteoporosis, it is important to learn about bone. Made mostly of collagen, bone is living, growing tissue. Collagen is a protein that provides a soft framework, and calcium phosphate is a mineral that adds strength and hardens the framework. This combination of collagen and calcium makes bone strong and flexible enough to withstand stress. More than 99 percent of the body’s calcium is contained in the bones and teeth. The remaining 1 percent is found in the blood 5.
Two types of bone found in the body—cortical and trabecular. Cortical bone is dense and compact. It forms the outer layer of the bone. Trabecular bone makes up the inner layer of the bone and has a spongy, honeycomb-like structure.
Bone Remodeling: throughout life, bone is constantly renewed through a two-part process called remodeling. This process consists of resorption and formation. During resorption, special cells called osteoclasts break down and remove old bone tissue. During bone formation, new bone tissue is laid down to replace the old. Several hormones including calcitonin, parathyroid hormone, vitamin D, estrogen (in women), and testosterone (in men), among others, regulate osteoclast and osteoblast function 5.
Think of bone as a bank account where you “deposit” and “withdraw” bone tissue. During childhood and the teenage years, new bone is added to the skeleton faster than old bone is removed. As a result, bones become larger, heavier, and denser. For most people, bone formation continues at a faster pace than removal until bone mass peaks during the third decade of life.
After age 25, bone “withdrawals” can begin to exceed “deposits.” For many people, this bone loss can be prevented by continuing to get calcium, vitamin D, and exercise and by avoiding tobacco and excessive alcohol use. Osteoporosis develops when bone removal occurs too quickly, replacement occurs too slowly, or both. You are more likely to develop osteoporosis if you did not reach your maximum peak bone mass during your bone-building years.
Bone mass in older adults equals the peak bone mass achieved by age 18–25 minus the amount of bone subsequently lost. Peak bone mass is determined largely by genetic factors, with contributions from nutrition, endocrine status, physical activity, and health during growth 6. Blacks reach higher peak bone mass than whites and Asians, whereas Hispanics have intermediate values. Men have higher bone mass than women. After achieving peak, bone mass plateaus for about 10 yr, during which time bone formation approximately equals bone resorption. After this, bone loss occurs at a rate of about 0.3 to 0.5%/yr. Beginning with menopause, bone loss accelerates in women to about 3 to 5%/yr for about 5 to 7 yr and then the rate of loss decelerates 4.
Women are more likely than men to develop osteoporosis. This is because women generally have smaller, thinner bones than men have and because women can lose bone tissue rapidly in the first 4 to 8 years after menopause because of the sharp decline in production of the hormone estrogen. Produced by the ovaries, estrogen has been shown to have a protective effect on bone. Women usually go through menopause between age 45 and 55. After menopause, bone loss in women greatly exceeds that in men. However, by age 65, women and men tend to lose bone tissue at the same rate. Although men do not undergo the equivalent of menopause, production of the male hormone testosterone may decrease, and this can lead to increased bone loss and a greater risk of developing osteoporosis.
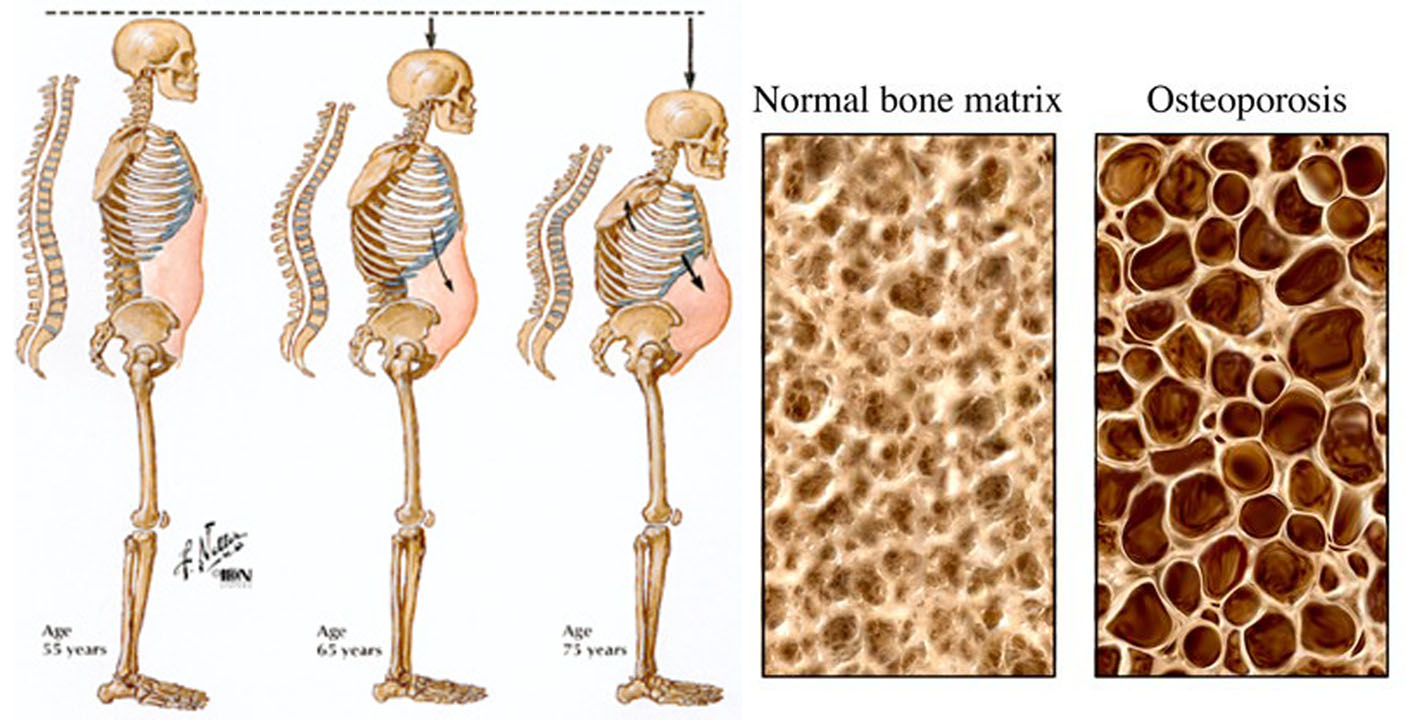
Osteoporotic bone loss affects cortical and trabecular (cancellous) bone. Cortical thickness and the number and size of trabeculae decrease, resulting in increased porosity. Trabeculae may be disrupted or entirely absent. Trabecular bone loss occurs more rapidly than cortical bone loss because trabecular bone is more porous and bone turnover is higher. However, loss of both types contributes to skeletal fragility 4.
The process of bone remodeling that maintains a healthy skeleton may be considered a preventive maintenance program, continually removing older bone and replacing it with new bone. Bone loss occurs when this balance is altered, resulting in greater bone removal than replacement. The imbalance occurs with menopause and advancing age. With the onset of menopause, the rate of bone remodeling increases, magnifying the impact of the remodeling imbalance. The loss of bone tissue leads to disordered skeletal architecture and an increase in fracture risk 7.
The U.S. Preventive Services Task Force, an independent panel of experts in primary care and prevention, recommends that all women age 65 and older be screened for osteoporosis. The task force also recommends screening for women under the age of 65 who are at high risk for fractures. Men over the age 65 who are at high risk for fractures should talk to their doctor about screening. If you are over 50 and have broken a bone, you may have osteoporosis or be at increased risk for the disease. You should also ask your doctor about osteoporosis if you notice that you have lost height or your posture has become stooped or hunched, or if you experience sudden back pain. You may also want to be evaluated for osteoporosis and fracture risk if you have a chronic disease or eating disorder known to increase the risk of osteoporosis, are taking one or more medications known to cause bone loss, or have multiple risk factors for osteoporosis and osteoporosis-related fractures 8.
Having weak bones that easily break is a sign of osteoporosis. It is normal for your bones to become less dense as you grow older, but osteoporosis speeds up this process. This condition can particularly lead to problems in older age because broken bones do not heal as easily in older people as they do in young people, and the consequences are more serious. In general, osteoporosis is more common in women, and they often develop it at a younger age.
Osteoporosis does not affect everyone to the same degree. Women, especially older women, are much more likely to get the disease than are men. In fact, women over age 50 accounted for over 75 percent (7.8 million) of the total cases of osteoporosis at the hip in 2002 9. Women are more susceptible than men to osteoporosis because they begin with less bone mass and lose it at a somewhat faster rate.
Treatment of Osteoporosis = Prevention of Fractures
Individuals with osteoporosis are at high risk of suffering one or more fractures, injuries that can often be physically debilitating and potentially lead to a downward spiral in physical and mental health 10. Generalized osteoporosis is the most common form of the disease, affecting most of the skeleton. Osteoporosis can also occur in localized parts of the skeleton as a result of injury or conditions that reduce muscle forces on the bone, such as limb paralysis. There are a variety of different types of osteoporosis. The most common form of osteoporosis is known as “primary osteoporosis”—that is, osteoporosis that is not caused by some other specific disorder. Bone loss caused by specific diseases or medications (see below) is referred to as “secondary osteoporosis.”
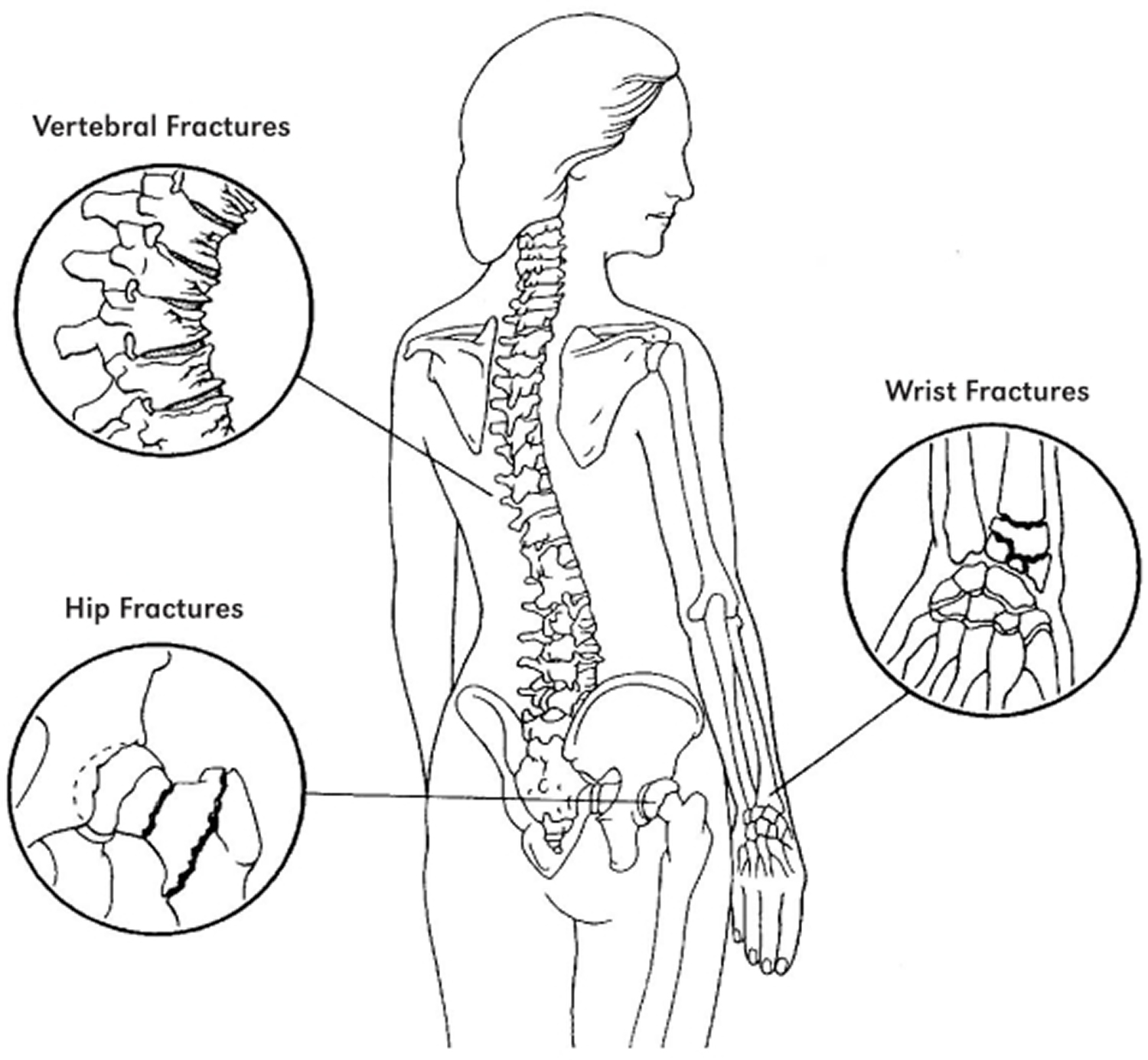
Osteoporosis affects all bones in the body. However, breaks are most common in the hip, wrist, and spine, also called vertebrae . Vertebrae support your body, helping you to stand and sit up. Osteoporosis in the vertebrae can cause serious problems for women. A fracture in this area occurs from day-to-day activities like climbing stairs, lifting objects, or bending forward. Signs of osteoporosis 11:
- Sloping shoulders
- Curve in the back
- Height loss
- Back pain
- Hunched posture
- Protruding abdomen
Getting older does not mean that you will automatically develop osteoporosis, but the risk does increase with age. People over the age of 70 are more likely to have low bone density. Plus, the risk of falling increases in old age, which then also makes fractures more likely.
But there are several things you can do to protect and strengthen your bones — even if you are already older.
- For access to the free Fracture Risk Assessment Tool (FRAX tool) go here 12.
What is Osteopenia vs Osteoporosis
Differentiating Osteopenia, Osteoporosis and Osteomalacia
Osteopenia is a term to define bone density that is not normal (lower-than-normal bone mass and bone mineral density), but also not as low as osteoporosis 13. By definition from the World Health Organization osteopenia is defined by bone densitometry as a T score −1 to −2.5 SD 14.
Two metabolic bone diseases decrease bone mass: osteoporosis and osteomalacia.
- In osteoporosis, bone mass decreases, but the ratio of bone mineral to bone matrix is normal.
- In osteomalacia, the ratio of bone mineral to bone matrix is low.
Osteoporosis results from a combination of low peak bone mass, increased bone resorption, and impaired bone formation. Osteomalacia is due to impaired mineralization, usually because of severe vitamin D deficiency or abnormal vitamin D metabolism (see Vitamin D). Osteomalacia can be caused by disorders that interfere with vitamin D absorption (eg, celiac disease) and by certain drugs (eg, phenytoin, phenobarbital). Osteoporosis is much more common than osteomalacia in the US. The two disorders may coexist, and their clinical expression is similar; moreover, mild to moderate vitamin D deficiency can occur in osteoporosis 4.
Osteomalacia should be suspected if the vitamin D level is consistently very low. To definitively differentiate between the two disorders, clinicians can do a tetracycline-labeled bone biopsy 4.
There are many causes for osteopenia including calcium and vitamin D deficiency and inactivity. Osteopenia frequently develops in people taking antiretroviral drugs for HIV; however, the association between antiretroviral drugs drugs and osteopenia is unclear 15. Genetics plays an important role in a person’s bone mineral density and often Caucasian women with a thin body habitus who are premenopausal are found to have osteopenia. Correction of calcium and vitamin D deficiency and walking 3 to 5 miles a week can often improve bone density in the hip and spine. There are a variety of pharmaceutical agents that have been recommended for the treatment of osteopenia and osteoporosis including hormone replacement therapy, selective estrogen receptor modulator therapy, anti-resorptive therapy. In addition patients with osteoporosis who have failed anti-resorptive therapy can have a significant improvement in their bone density with anabolic therapy 13.
While pharmacological treatment of patients with prevalent osteoporotic fractures is universally accepted, the treatment of patients at increased risk of fracture due to low bone mass is more controversial. Our ability to detect patients at increased risk has improved significantly after the widespread availability of Dual Energy X-ray Absorptiometry (DXA), which provides a precise assessment of the amount of mineralized bone in the skeleton. According to the WHO criteria for assessment of DXA measurements, patients are considered having low bone mass (osteopenic), when their bone mineral density (BMD) t-score of the spine or hip lies between −1 and −2,5. Although fracture risk increases with decreases in bone mineral density, the vast majority of osteoporotic fractures occur in osteopenic patients. This is due to the fact that even though the risk of fracture is lower in the osteopenia than in osteoporosis, the number of subjects at risk is much higher in the osteopenic range due to the Gaussian distribution of bone mineral density values in the population (see below Fig. 1). In an analysis of self reported fractures from the National Osteoporosis Risk Assessment study Siris et al. 16 reported that 82% of postmenopausal women with fractures had T scores better than −2.5. The study comprised 149,524 white postmenopausal women aged 50 to 104 years (mean age, 64.5 years). New fractures were reported by 2,259 women, including 393 hip fractures; but only 6.4% exhibited baseline T scores of −2.5 or less. Although fracture rates were highest in women with a t-score <−2,5, only 18% of the osteoporotic fractures and 26% of hip fractures occurred in this group 16.
Figure 1. Distribution of fracture rates and number of women with fractures according to BMD t-scores from the The National Osteoporosis Risk Assessment (NORA) study, (NORA), which comprised 149,524 white postmenopausal women aged 50 to 104 years (mean age, 64.5 years). Bone mineral density (BMD) was assessed by peripheral bone densitometry at the heel, finger, or forearm. Although fracture rates were highest in women with the lowest t-scores (open bars), the largest absolute number of fractures (black bars) was seen in the osteopenic range of t-score (−1 to −2,5). (Source 16).
Identification of osteopenic patients at increased risk of fracture
Bone mineral density is related to bone strength and low bone mineral density is a major risk factor for fractures. However, most patients presenting with a fracture do not have bone mineral density based osteoporosis, defined according to the World Health Organization (WHO) definition as a T score of −2.5 or below. The most poignant example is hip fracture, where only half the patients exhibit t-scores below −2.5 17. In addition, and independent of bone-related risks, extraskeletal risk factors such as falls contribute to fracture risk and are present in the majority of patients older than 50 years presenting with a clinical fracture, and falls are the dominant event leading to forearm and hip fracture 18.
Summary: An ever increasing array of effective treatments is at our disposal, to protect patients with osteopenia against fractures. While there is general consensus on treating osteopenic individuals with prevalent low energy fractures, the treatment of osteopenia without fracture is still debatable 19. However, current evidence indicates that specific drug therapy should be instituted if an osteopenic patients has prevalent fractures or suffers new fractures, be it clinical or asymptomatic. Moreover, a significant accumulation of several significant risk factors, for example as indicated by the Fracture Risk Assessment Tool (FRAX tool) may constitute an indication for medical treatment by means of drugs. Patients without such risk factors should be counselled on a “bone friendly” lifestyle with nutritional modifications, regular exercise, moderation in alcohol use and If possible smoking cessation. In patients with low vitamin D levels, Calcium plus vitamin D supplementation may also be indicated 19.
- For access to the free Fracture Risk Assessment Tool (FRAX tool) go here 12.
How Common is Osteoporosis
In the US, data from the National Health and Nutrition Examination Survey, 2005–2008, 4 percent of men 50 years of age and over have osteoporosis of the femur neck or lumbar spine and 16 percent of women 50 years of age and over with osteoporosis of the femur neck or lumbar spine 20.
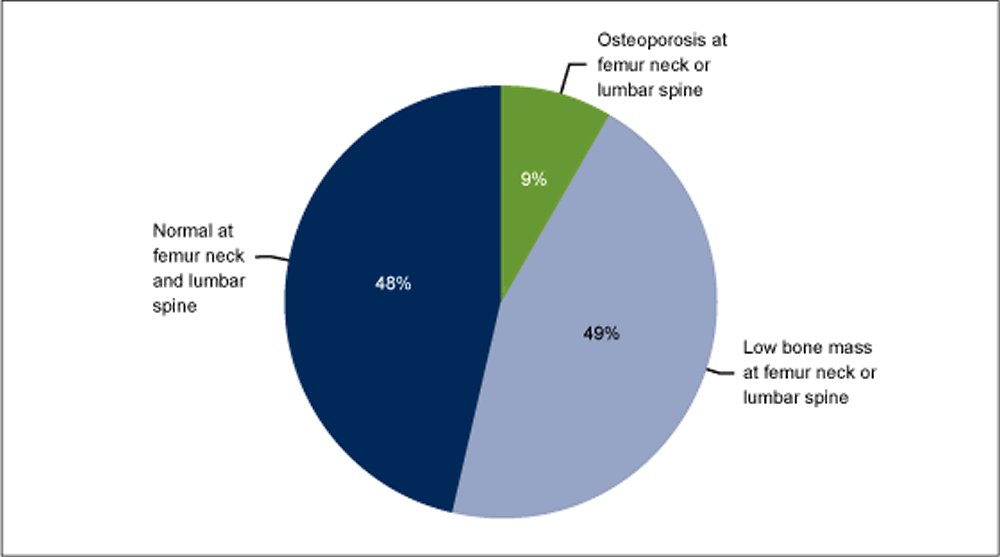
- 9% of adults aged 50 years and over had osteoporosis, as defined by the World Health Organization, at either the femur neck or lumbar spine. About one-half had low bone mass at either site, while 48% had normal bone mass at both sites.
- Estimates of poor skeletal status at the femur neck or lumbar spine when considered alone were not the same as estimates based on the two skeletal sites together because some individuals had the condition at one site but not the other.
- The prevalence of osteoporosis or low bone mass at either the femur neck or lumbar spine differed by age, sex, and race and ethnicity. The prevalence was higher in women and increased with age. Differences between racial and ethnic groups varied by sex and skeletal status category.
This data brief presents the most recent national data on osteoporosis or low bone mass at either the femur neck or lumbar spine among older adults in the United States population based on these WHO categories. Results are presented by age, sex, and race and ethnicity.
Footnote: The percentages shown will not add up to 100% due to double counting among those with osteoporosis at either skeletal site or low bone mass at either skeletal site.
[Source 21]The prevalence of osteoporosis or low bone mass at either the femur neck or lumbar spine is higher in women than men in each decade or when compared overall for aged 50 years and over after adjusting for age differences between the two sexes. The age-adjusted prevalence of osteoporosis at either skeletal site was 16% in women compared with 4% in men. The age-adjusted prevalence of low bone mass at either skeletal site was 61% in women compared with 38% in men.
The prevalence of osteoporosis or low bone mass at either skeletal site differ by race and ethnicity in men after adjusting for age differences between the racial and ethnic groups. The age-adjusted prevalence of osteoporosis at either skeletal site in men of other races (9%) was higher than the prevalence in non-Hispanic white men (4%). The age-adjusted prevalence of low bone mass at either skeletal site was lower in non-Hispanic black men (24%) compared with non-Hispanic white men (39%).
The prevalence of osteoporosis or low bone mass at either the femur neck or lumbar spine differ by race and ethnicity in women after adjusting for age differences between the racial and ethnic groups. When compared with the age-adjusted prevalence of osteoporosis in non-Hispanic white women (15%), the age-adjusted prevalence of osteoporosis at either skeletal site is higher in Mexican-American women (26%) and lower in non-Hispanic black women (9%). When compared with the age-adjusted prevalence in non-Hispanic white women (62%), the age-adjusted prevalence of low bone mass at either skeletal site is higher in women of other races (72%) and lower in non-Hispanic black women (44%).
According to the Robert Koch Institute, 8% of men and 13% of women in Germany between the ages of 60 and 69 report being diagnosed with osteoporosis. But it is not known exactly how many people in Germany actually have osteoporosis or how many bone fractures are caused by osteoporosis. Also, not everyone with osteoporosis ends up with a bone fracture 3.
Hip fractures in particular can have serious consequences. This is a problem that almost only affects older people: In Germany, 6 to 7 out of 1,000 people over the age of 65 break their hip bone (femoral neck) every year. Most of them are over the age of 75.
- For access to the free Fracture Risk Assessment Tool (FRAX tool) go here 12.
Causes of Osteoporosis
Inside bones there is a supporting structure with interconnecting bony plates and rods called trabeculae. This structure is called trabecular or spongy bone because it looks a bit like a sponge or honeycomb.
Osteoporosis develops when a large amount of the spongy bone tissue breaks down, leaving bigger spaces. The bone becomes more porous as a result. This affects the fine structure of the bones, and they become brittle. Some people’s bones become so fragile that even tripping over something or lifting a heavy shopping bag is enough to cause vertebrae to break.
Bone density starts to gradually decrease once you turn 30, even if you do not have osteoporosis. Around the age of 50 this process speeds up, especially in women. Before menopause, the female sex hormone estrogen helps protect the bones by slowing down bone loss. So after menopause, when this hormone level drops, bone tissue is lost more quickly.
If no cause for the bone loss can be found, it is called primary osteoporosis. Secondary osteoporosis is when bones have become fragile due to something else, like another condition or long-term corticosteroid use.
Osteoporosis can develop as a primary disorder or secondarily due to some other factor. The sites of fracture are similar in primary and secondary osteoporosis.
- Primary osteoporosis
More than 95% of osteoporosis in women and about 80% in men is primary. Most cases occur in postmenopausal women and older men. Gonadal insufficiency is an important factor in both men and women. Other factors that may accelerate bone loss in patients with primary osteoporosis include decreased calcium intake, low vitamin D levels, certain drugs, and hyperparathyroidism. Some patients have an inadequate intake of calcium during the bone growth years of adolescence and thus never achieve peak bone mass 4.
The major mechanism of bone loss is increased bone resorption, resulting in decreased bone mass and microarchitectural deterioration, but sometimes bone formation is impaired. The mechanisms of bone loss may involve the following:
- Local changes in the production of bone-resorbing cytokines, such as increases in cytokines that stimulate bone resorption
- Impaired formation response during bone remodeling (probably caused by age-related decline in the number and activity of osteoblasts)
- Other factors such as a decline in local and systemic growth factors
Fragility fractures rarely occur in children, adolescents, premenopausal women, or men < 50 yr with normal gonadal function and no detectable secondary cause, even in those with low bone mass (low Z-scores on dual-energy x-ray absorptiometry [DXA]). Such uncommon cases are considered idiopathic osteoporosis.
- Secondary osteoporosis
Secondary osteoporosis accounts for < 5% of osteoporosis in women and about 20% in men. The causes (see Causes of Secondary Osteoporosis) may also further accelerate bone loss and increase fracture risk in patients with primary osteoporosis 4.
Patients with chronic kidney disease may have several reasons for low bone mass, including secondary hyperparathyroidism, elevated serum phosphate, calcitriol deficiency, abnormalities of serum calcium and vitamin D, osteomalacia, and low-turnover bone disorders (adynamic bone disease).
Causes of Secondary Osteoporosis
- Cancer (eg, multiple myeloma)
- Chronic obstructive pulmonary disease (COPD) (due to the disorder itself, as well as tobacco use and/or treatment with glucocorticoids). Chronic obstructive pulmonary disease is airflow limitation caused by an inflammatory response to inhaled toxins, often cigarette smoke. Alpha-1 antitrypsin deficiency and various occupational exposures are less common causes in nonsmokers.
- Chronic kidney disease
- Drugs (eg, glucocorticoids, anticonvulsants, medroxyprogesterone, aromatase inhibitors, rosiglitazone, pioglitazone, thyroid replacement therapy, heparin, ethanol, tobacco)
- Endocrine disease (eg, glucocorticoid excess, hyperparathyroidism, hyperthyroidism, hypogonadism, hyperprolactinemia, diabetes mellitus)
- Hypercalciuria
- Hypervitaminosis A
- Hypophosphatasia
- Hypovitaminosis D
- Immobilization
- Liver disease
- Malabsorption syndromes
- Prolonged weightlessness (as occurs in space flight)
- Rheumatoid arthritis
Risk Factors for Osteoporosis
There are a number of factors that can increase someone’s risk of developing osteoporosis. Some can be influenced, whereas others cannot. The main risk factors for osteoporosis include:
Factors that you can’t control:
- Being female. Women develop osteoporosis more often than men, and they are also more likely to have bone fractures.
- Getting older. As we get older, our bone density decreases and the risk of developing osteoporosis increases. Men over the age of 65 and post-menopausal women are at the greatest risk.
- Sex hormone deficiencies: The most common manifestation of estrogen deficiency in premenopausal women is amenorrhea, the abnormal absence of menstrual periods. Missed or irregular periods can be caused by various factors, including hormonal disorders as well as extreme levels of physical activity combined with restricted calorie intake—for example, in female marathon runners, ballet dancers, and women who spend a great deal of time and energy working out at the gym. Low estrogen levels in women after menopause and low testosterone levels in men also increase the risk of osteoporosis. Lower than normal estrogen levels in men may also play a role. Low testosterone and estrogen levels are often a cause of osteoporosis in men being treated with certain medications for prostate cancer.
- Having a small, thin body (under 127 pounds)
- Having a family history of osteoporosis. Women whose mother or father broke their hip because of osteoporosis are at greater risk of developing osteoporosis themselves.
- Being white or Asian, but African American women and Latinas are also at risk
- Not getting your period (if you should be getting it)
- Having a disorder that increases your risk of getting osteoporosis, (such as rheumatoid arthritis, type 1 diabetes, premature menopause, anorexia nervosa)
- Not getting enough exercise
- Long-term use of certain medicines, including:
- Glucocorticoids — medicines used to treat many illnesses, including arthritis, asthma, and lupus
- Some antiseizure medicines
- Gonadotropin-releasing hormone — used to treat endometriosis
- Antacids with aluminum — the aluminum blocks calcium absorption
- Some cancer treatments
- Too much replacement thyroid hormone
Factors that you can control:
- Smoking
- Drinking too much alcohol. Experts recommend no more than 1 drink a day for women.
- A diet low in dairy products or other sources of calcium and vitamin D
- Not getting enough exercise
- Low body weight (compared to body size). Anorexia nervosa, for example, is an eating disorder that leads to abnormally low body weight, malnutrition, amenorrhea, and other effects on the body that adversely affect bone health. Late onset of puberty and early menopause reduce lifetime estrogen exposure in women and also increase the risk of osteoporosis.
- Diet low in calcium
- Vitamin D deficiency
- Long-term steroid use
- Use of other medications, such as some antidepressants (SSRIs), diabetes medicines (glitazones), glucocorticoids and some anticonvulsants lead to bone loss and increased risk of osteoporosis. Other drugs that may lead to bone loss include anticlotting drugs, such as heparin; drugs that suppress the immune system, such as cyclosporine; and drugs used to treat prostate cancer.
Signs and Symptoms of Osteoporosis
Patients with osteoporosis are asymptomatic unless a fracture has occurred. Sometimes there are obvious signs that a person has osteoporosis – for example they may ”shrink” a little and develop a stooped posture. But often the first sign that someone has osteoporosis is when they break a bone, sometimes without knowing how or why it happened. This kind of break is called a “spontaneous fracture.” When bone mass is lost the risk of breaking a bone (fractures) is higher. Osteoporosis that has already caused a fracture is referred to as “established” osteoporosis.
The bones of the spinal column (vertebrae) are the most likely to break or “collapse” in someone who has osteoporosis. Sometimes this will cause back pain, but most people do not notice anything.
Broken vertebrae are one reason why many older people stoop over and develop what is often called a “dowager’s hump” at the top of their spine.
Osteoporosis also commonly affects the wrist, the upper arm and the femur (thigh bone).
Nonvertebral fractures are typically symptomatic, but about two thirds of vertebral compression fractures are asymptomatic (although patients may have underlying chronic back pain due to other causes such as osteoarthritis). A vertebral compression fracture that is symptomatic begins with acute onset of pain that usually does not radiate, is aggravated by weight bearing, may be accompanied by point spinal tenderness, and typically begins to subside in 1 wk. However, residual pain may last for months or be constant.
Multiple thoracic compression fractures eventually cause dorsal kyphosis, with exaggerated cervical lordosis (dowager’s hump). Abnormal stress on the spinal muscles and ligaments may cause chronic, dull, aching pain, particularly in the lower back. Patients may have shortness of breath due to the reduced intrathoracic volume and/or abdominal discomfort due to the compression of the abdominal cavity as the rib cage approaches the pelvis.
How you can find out if you have Osteoporosis
There are tests you can get to find out your bone density. This is related to how strong or fragile your bones are. One test is called dual-energy X-ray absorptiometry (DXA or dexa). A DXA scan takes X-rays of your bones. Screening tools also can be used to predict the risk of having low bone density or breaking a bone. Talk with your doctor or nurse about this test or tools to assess risk.
- DXA Scan
Bone density should be measured using dual-energy X-ray absorptiometry (DXA) to screen people at risk, to provide a quantitative measure of bone loss, and to monitor those undergoing treatment 22.
A DXA scan is recommended for the following patients:
- All women ≥ 65 yr
- Women between menopause and age 65 who have risk factors, including a family history of osteoporosis, a low body mass index (eg, previously defined as body weight < 127 lb), and use of tobacco and/or drugs with a high risk of bone loss (eg, glucocorticoids)
- Patients (men and women) of any age who have had fragility fractures
- Patients with evidence on imaging studies of decreased bone density or asymptomatic vertebral compression fractures incidentally noted on imaging studies
- Patients at risk of secondary osteoporosis
Although low bone density (and the associated increased risk of fracture) can be suggested by plain x-rays, it should be confirmed by a bone density measurement.
Dual-energy X-ray absorptiometry (DXA) is used to measure bone mineral density (g/cm2); it defines osteopenia or osteoporosis (in the absence of osteomalacia), predicts the risk of fracture, and can be used to follow treatment response. Bone density of the lumbar spine, hip, distal radius, or the entire body can be measured. (Quantitative CT scanning can produce similar measurements of the spine or hip but is currently not widely available.) Bone density is ideally measured at two sites, including the lumbar spine and one hip; however, at some centers, measurements are taken of the spine and both hips.
If the spine or a hip is not available for scanning (eg, because of hardware from prior total hip arthroplasty), the distal radius can be scanned (called “1/3 radius” on the DXA scan report). The distal radius should also be scanned in a patient with hyperparathyroidism because this is the most common site of bone loss in hyperparathyroidism.
DXA results are reported as T-scores and Z-scores. The T-score corresponds to the number of standard deviations that the patient’s bone density differs from the peak bone mass of a healthy, young person of the same sex and ethnicity. The WHO establishes cutoff values for T-scores that define osteopenia and osteoporosis. A T-score < -1.0 and > -2.5 defines osteopenia. A T-score ≤ -2.5 defines osteoporosis.
The Z-score corresponds to the number of standard deviations that the patient’s bone mineral density differs from that of a person of the same age and sex and should be used for children, premenopausal women, or men < 50 yr. If the Z-score is ≤ -2.0, bone density is low for the patient’s age and secondary causes of bone loss should be considered.
Current central DXA systems can also assess vertebral deformities in the lower thoracic and lumbar spine, a procedure termed vertebral fracture analysis (VFA). Vertebral deformities, even those clinically silent, are diagnostic of osteoporosis and are predictive of an increased risk of future fractures. VFA is more likely to be useful in patients with height loss ≥ 3 cm.
The need for drug therapy is based on the probability of fracture, which depends on DXA results as well as other factors. The fracture risk assessment (FRAX) score (WHO Fracture Risk Assessment Tool 12) predicts the 10-yr probability of a major osteoporotic (hip, spine, forearm, or humerus) or hip fracture in untreated patients. The score accounts for significant risk factors for bone loss and fracture. If the FRAX score is above certain thresholds (in the US, a ≥ 20% probability of major osteoporotic fracture or 3% probability of hip fracture), drug therapy should be recommended.
- For access to the free Fracture Risk Assessment Tool (FRAX tool) go here 12.
Monitoring for ongoing bone loss or the response to treatment with serial DXA scans should be done using the same DXA machine, and the comparison should use actual bone mineral density (g/cm2) rather than T-score. In patients with osteopenia, DXA should be repeated periodically to determine whether there is ongoing bone loss or development of frank osteoporosis requiring treatment. The frequency for follow-up DXA varies from patient to patient. DXA is often done every 2 to 3 yr but can sometimes be done less frequently, for example, if bone density is normal and fracture risk is low. In patients being treated for osteoporosis, DXA should be repeated, usually about every 2 to 3 yr, but sometimes more frequently in patients taking glucocorticoids. A stable or improved bone mineral density predicts a lower fracture risk. Monitoring bone density with a repeat DXA scan may help identify patients at higher risk of fractures due to a suboptimal response to osteoporosis treatment 22. Patients being treated for osteoporosis who have a significantly decreased bone mineral density on serial DXA examinations should be evaluated for drug adherence and secondary causes of bone loss.
When you should get a bone density (DXA) test
If you are age 65 or older, you should get a bone density test to screen for osteoporosis. If you are younger than 65 and have risk factors for osteoporosis, ask your doctor or nurse if you need a bone density test before age 65. Bone density testing is recommended for older women whose risk of breaking a bone is the same or greater than that of a 65‑year‑old white woman with no risk factors other than age. To find out your fracture risk and whether you need early bone density testing, your doctor will consider factors such as:
- Your age and whether you have reached menopause
- Your height and weight
- Whether you smoke
- Your daily alcohol use
- Whether your mother or father has broken a hip
- Medicines you use
- Whether you have a disorder that increases your risk of getting osteoporosis
Plain x-rays
Bones show decreased radiodensity and loss of trabecular structure, but not until about 30% of bone has been lost. However, plain x-rays are important for documenting fractures resulting from bone loss. Loss of vertebral body height and increased biconcavity characterize vertebral compression fractures. Thoracic vertebral fractures may cause anterior wedging of the bone. In long bones, although the cortices may be thin, the periosteal surface remains smooth. Vertebral fractures at T4 or above raise concern of cancer rather than osteoporosis. Plain x-rays of the spine should be considered in older patients with severe back pain and localized vertebral spinous tenderness 4.
Glucocorticoid-induced osteoporosis is likely to cause rib fractures as well as fractures at other sites where osteoporotic fractures are common. Hyperparathyroidism can be differentiated when it causes subperiosteal resorption or cystic bone lesions (rarely). Osteomalacia may cause abnormalities on imaging tests similar to those of osteoporosis (see What is Osteopenia vs Osteoporosis : Differentiating Osteopenia, Osteoporosis and Osteomalacia).
Other testing
An evaluation for secondary causes of bone loss should be considered in a patient with a Z-score ≤ -2.0 or if a cause of secondary bone loss is clinically suspected. Laboratory testing 4 should usually include the following:
- Serum calcium, magnesium, and phosphorus
- 25-Hydroxy vitamin D level
- Liver function tests, including an alkaline phosphatase (hypophosphatasia)
- Intact PTH level (hyperparathyroidism)
- Serum testosterone in men (hypogonadism)
- 24-h urine for calcium and creatinine (hypercalciuria)
Other tests such as thyroid-stimulating hormone or free thyroxine to check for hyperthyroidism, measurements of urinary free cortisol, and blood counts and other tests to rule out cancer, especially myeloma (eg, serum and urine protein electrophoresis), should be considered depending on the clinical presentation.
Patients with weight loss should be screened for GI disorders (eg, malabsorption, celiac disease, inflammatory bowel disease) as well as cancer. Bone biopsy is reserved for unusual cases (eg, young patients with fragility fractures and no apparent cause, patients with chronic kidney disease who may have other bone disorders, patients with persistently very low vitamin D levels suspected of having osteomalacia).
Levels of fasting serum C-telopeptide cross-links (CTX) or urine N-telopeptide cross-links reflect increased bone resorption 4. Although reliability varies for routine clinical use, C-telopeptide cross-links and N-telopeptide cross-links may be helpful in monitoring response to therapy or with the timing of a drug holiday.
Bone Fracture Areas in Osteoporosis
(Source 10).
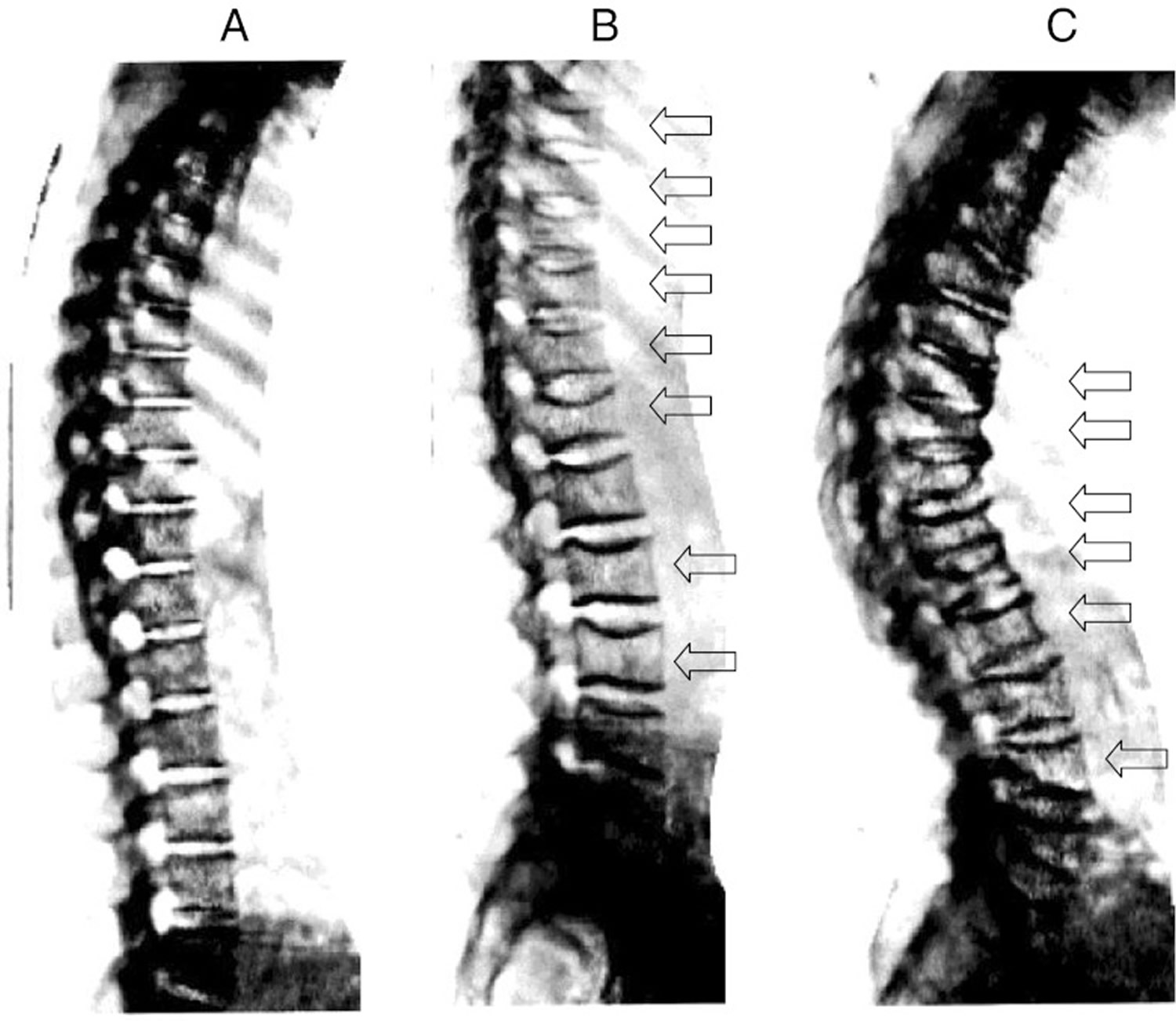
Spine x-ray imaging as obtained from on DXA scanner. Normal spine (a); young woman with vertebral deformities (arrows) after post partum osteoporosis (b); severe osteoporosis in 68 year old woman with multiple moderate and severe compression fractures (c) (Source 19).
How to Prevent Osteoporosis
The best way to prevent weak bones is to work on building strong ones. No matter how old you are, it is never too late to start. Building strong bones during childhood and the teen years is one of the best ways to keep from getting osteoporosis later. As you get older, your bones don’t make new bone fast enough to keep up with the bone loss. And after menopause, bone loss happens more quickly. But there are steps you can take to slow the natural bone loss with aging and to prevent your bones from becoming weak and brittle.
There are several things you can do on your own to protect your bones and lower your individual risk of developing osteoporosis. These include eating a diet rich in calcium, getting regular exercise and quitting smoking. Making sure your body has enough vitamin D is also important. Dietary supplements can be used as an additional source of calcium and vitamin D if you are not getting enough in your diet.
Femoral neck fractures are especially likely to have serious consequences because most people do not fully recover afterwards. Although osteoporosis can increase the likelihood of this kind of fracture, falls are the greatest risk factor for hip fractures. So it is especially important for older people to try to prevent falls. Getting exercise can help here: Physical activity can make you feel more confident and improve your coordination. It is also important to take care of hazards around your home that could increase your chances of slipping or stumbling. These may include things like loose cables, rugs and door thresholds 23.
Lifestyle Approaches to Promote Bone Health
There is much that individuals can do to promote their own bone health throughout life. This section outlines recommendations for diet, physical activity, and other lifestyle practices that can help to achieve that goal. Moreover, the activities and practices suggested in this section contribute not only to bone health, but to overall health and vitality. In fact, bone-specific recommendations fit well within an overall program of good nutrition and physical activity that should be followed in order to prevent the onset of many of the major chronic diseases affecting Americans.
Calcium
Bones contain a lot of calcium. It is important to get enough calcium in your diet. You can get calcium through foods and/or calcium pills, which you can get at the grocery store or drug store. Getting calcium through food is definitely better since the food provides other nutrients that keep you healthy. Talk with your doctor or nurse before taking calcium pills to see which kind is best for you. Taking more calcium pills than recommended doesn’t improve your bone health. So, try to reach these goals through a combination of food and supplements.
Lifelong adequate calcium intake is necessary for the acquisition of peak bone mass and subsequent maintenance of bone health. The skeleton contains 99 % of the body’s calcium stores; when the exogenous supply is inadequate, bone tissue is resorbed from the skeleton to maintain serum calcium at a constant level.
Americans obtain most of their calcium from dairy products. Most Americans above age 9 on average do not consume recommended levels of calcium 24. In fact, approximately three 8-ounce glasses of milk each day, combined with the calcium from the rest of a normal diet, is enough to meet the recommended daily requirements for most adults.
For postmenopausal women, the recommended total daily calcium intake is 1,200 mg per day in two or more doses. These levels of intake can be achieved through dietary sources of calcium, including both dairy and non-dairy products. In addition, calcium supplements (e.g., calcium carbonate, calcium citrate, other calcium salts) are available in the form of pills, chewable tablets, and liquids 25.
Calcium Supplements
The two main forms of calcium in supplements are carbonate and citrate. Calcium carbonate is more commonly available and is both inexpensive and convenient 26. Due to its dependence on stomach acid for absorption, calcium carbonate is absorbed most efficiently when taken with food, whereas calcium citrate is absorbed equally well when taken with or without food 27. Calcium citrate is also useful for people with achlorhydria, inflammatory bowel disease, or absorption disorders 28. Other calcium forms in supplements or fortified foods include gluconate, lactate, and phosphate. Calcium citrate malate is a well-absorbed form of calcium found in some fortified juices 29.
Calcium supplements contain varying amounts of elemental calcium. For example, calcium carbonate is 40% calcium by weight, whereas calcium citrate is 21% calcium. Fortunately, elemental calcium is listed in the Supplement Facts panel, so consumers do not need to calculate the amount of calcium supplied by various forms of calcium supplements.
The percentage of calcium absorbed depends on the total amount of elemental calcium consumed at one time; as the amount increases, the percentage absorption decreases. Absorption is highest in doses ≤500 mg 28. So, for example, one who takes 1,000 mg/day of calcium from supplements might split the dose and take 500 mg at two separate times during the day.
Some individuals who take calcium supplements might experience gastrointestinal side effects including gas, bloating, constipation, or a combination of these symptoms. Calcium carbonate appears to cause more of these side effects than calcium citrate 28, so consideration of the form of calcium supplement is warranted if these side effects are reported. Other strategies to alleviate symptoms include spreading out the calcium dose throughout the day and/or taking the supplement with meals.
Here’s how much calcium you need each day 30 :
| Ages | Milligrams(mg) per day |
|---|---|
| 9-18 | 1,300 |
| 19-50 | 1,000 |
| 51 and older | 1,200 |
Pregnant or nursing women need the same amount of calcium as other women of the same age.
There is no evidence that calcium intake in excess of these amounts confers additional bone strength. Intakes in excess of 1200 to 1500 mg/day may increase the risk of developing kidney stones, cardiovascular disease, and stroke. The scientific literature is highly controversial in this area 31, 32, 33, 34.
The total daily calcium intake should not exceed 2,500 mg 35.
In analysing data from 5 trials which included 742 patients found that after two years of treatment, the bone mineral density of the lumbar spine and forearm of patients taking calcium and vitamin D therapy improved more than patients who had no treatment. There was no difference in the number of fractures or laboratory measures of bone density between the two groups. It was found that calcium and vitamin D is effective at preventing and treating corticosteroid-induced bone loss at the lumbar spine and forearm. The treatment appears to be safe 36 lists the nutrient content of many foods with calcium arranged by nutrient content 37 and by food name 38.
Selected food sources of calcium are listed in Table 2. Check the food labels for more information.
| Food | Milligrams (mg) per serving | Percent DV* |
|---|---|---|
| Yogurt, plain, low fat, 8 ounces | 415 | 42 |
| Mozzarella, part skim, 1.5 ounces | 333 | 33 |
| Sardines, canned in oil, with bones, 3 ounces | 325 | 33 |
| Yogurt, fruit, low fat, 8 ounces | 313–384 | 31–38 |
| Cheddar cheese, 1.5 ounces | 307 | 31 |
| Milk, nonfat, 8 ounces** | 299 | 30 |
| Soymilk, calcium-fortified, 8 ounces | 299 | 30 |
| Milk, reduced-fat (2% milk fat), 8 ounces | 293 | 29 |
| Milk, buttermilk, lowfat, 8 ounces | 284 | 28 |
| Milk, whole (3.25% milk fat), 8 ounces | 276 | 28 |
| Orange juice, calcium-fortified, 6 ounces | 261 | 26 |
| Tofu, firm, made with calcium sulfate, ½ cup*** | 253 | 25 |
| Salmon, pink, canned, solids with bone, 3 ounces | 181 | 18 |
| Cottage cheese, 1% milk fat, 1 cup | 138 | 14 |
| Tofu, soft, made with calcium sulfate, ½ cup*** | 138 | 14 |
| Ready-to-eat cereal, calcium-fortified, 1 cup | 100–1,000 | 10–100 |
| Frozen yogurt, vanilla, soft serve, ½ cup | 103 | 10 |
| Turnip greens, fresh, boiled, ½ cup | 99 | 10 |
| Kale, fresh, cooked, 1 cup | 94 | 9 |
| Ice cream, vanilla, ½ cup | 84 | 8 |
| Chinese cabbage, bok choi, raw, shredded, 1 cup | 74 | 7 |
| Bread, white, 1 slice | 73 | 7 |
| Pudding, chocolate, ready to eat, refrigerated, 4 ounces | 55 | 6 |
| Tortilla, corn, ready-to-bake/fry, one 6” diameter | 46 | 5 |
| Tortilla, flour, ready-to-bake/fry, one 6” diameter | 32 | 3 |
| Sour cream, reduced fat, cultured, 2 tablespoons | 31 | 3 |
| Bread, whole-wheat, 1 slice | 30 | 3 |
| Kale, raw, chopped, 1 cup | 24 | 2 |
| Broccoli, raw, ½ cup | 21 | 2 |
| Cheese, cream, regular, 1 tablespoon | 14 | 1 |
* DV = Daily Value. DVs were developed by the U.S. Food and Drug Administration to help consumers compare the nutrient contents among products within the context of a total daily diet. The DV for calcium is 1,000 mg for adults and children aged 4 years and older. Foods providing 20% of more of the DV are considered to be high sources of a nutrient, but foods providing lower percentages of the DV also contribute to a healthful diet.
Vitamin D
Vitamin D plays a major role in calcium absorption, health of bone, bone mineralization (hardening), muscle performance, balance and risk of falling. Vitamin D is produced in your skin when it is exposed to sunlight. You need 10 to 15 minutes of sunlight to the hands, arms, and face, two to three times a week to make enough vitamin D. The amount of time depends on how sensitive your skin is to light. It also depends on your use of sunscreen, your skin color, and the amount of pollution in the air. You can also get vitamin D by eating foods, such as milk, or by taking vitamin pills. Vitamin D taken in the diet by food or pills is measured in international units (IU). Look at the pill bottle or food label for the IU amount.
The National Osteoporosis Foundation recommends an intake of 800 to 1000 international units (IU) of vitamin D per day for adults age 50 and older. Vitamin D is synthesized in the skin through sunlight exposure, or it may be taken as a supplement. However, the skin of older individuals does not synthesize vitamin D as well as the skin of younger individuals, and in some parts of the country, the winter sun does not produce vitamin D in the skin of all individuals. In addition, vitamin D is not available in many foods other than fortified milk, which contains 100 IU (international units) per cup. Thus, many individuals will need to take a supplement, especially those who avoid sun exposure, use sun block, or do not drink milk. The recommended dose of vitamin D is 200 to 600 IU daily, with the dose dependent on age, as shown in the table below 35. However, many experts are recommending more vitamin D for the frail elderly 40.
Institute of Medicine Dietary Reference Intakes for vitamin D are 600 IU/day until age 70 and 800 IU/day for adults age 71 years and older.
Here’s how much vitamin D you need each day:
| Ages | IU per day |
|---|---|
| 19-70 | 600 |
| 71+ | 800 |
The total daily vitamin D intake of persons who are not vitamin D deficient should not exceed 2,000 IU 41. Many calcium supplements contain vitamin D. Most multivitamins contain 400 IU of vitamin D. Vitamin D supplements can be taken on their own, or with calcium or food.
Adults who are vitamin D deficient require treatment with higher doses of vitamin D, may be treated with 50,000 IU of vitamin D2 or vitamin D3 once a week or the equivalent daily dose (7000 IU vitamin D2 or vitamin D3) for 8–12 weeks to achieve a 25(OH)D blood level of approximately 30 ng/ml. This regimen should be followed by maintenance therapy of 1500–2000 IU/day or whatever dose is needed to maintain the target blood level 42, 43.
Vitamin D deficiency can lead to secondary hyperparathyroidism with normal levels of blood calcium. It should be noted that the optimal range for 25-hydroxyvitamin D is higher than the “normal” ranges reported from clinical laboratories, since these ranges are obtained from a population that includes individuals with sub-optimal levels. Patients can be treated with vitamin D supplementation of 50,000 IU once a week for up to 3 months with follow-up blood tests of vitamin D, calcium, and PTH (parathyroid hormone) levels. Some patients may require longer courses of treatment 44.
Table 4: Adequate Intakes (Al) or Recommended Daily Allowances (RDA) and Tolerable Upper Intake Levels (UL) for Calcium, Vitamin D, Phosphorus, and Magnesium by Life-Stage Group for United States and Canada
| Life-stage group | Calcium (mg/day) | Vitamin D (IU/day) | Phosphorous (mg/day) | Magnesium (mg/day) | |||||
|---|---|---|---|---|---|---|---|---|---|
| Al | UL | Al | UL | RDA | UL | RDA | UL† | ||
| Male | Female | ||||||||
| 0–6 months | 210 | ND* | 200 | 1000 | 100 | ND* | 30 | 30 | ND* |
| 7–12 months | 270 | ND* | 200 | 1000 | 275 | ND* | 75 | 75 | ND* |
| 1–3 years | 500 | 2500 | 200 | 2000 | 460 | 3000 | 80 | 80 | 65 |
| 4–8 years | 800 | 2500 | 200 | 2000 | 500 | 3000 | 130 | 130 | 110 |
| 9–13 years | 1300 | 2500 | 200 | 2000 | 1250 | 4000 | 240 | 240 | 350 |
| 14–18 years | 1300 | 2500 | 200 | 2000 | 1250 | 4000 | 410 | 360 | 350 |
| 19–30 years | 1000 | 2500 | 200 | 2000 | 700 | 4000 | 400 | 310 | 350 |
| 31–50 years | 1000 | 2500 | 200 | 2000 | 700 | 4000 | 420 | 320 | 350 |
| 51–70 years | 1200 | 2500 | 400 | 2000 | 700 | 4000 | 420 | 320 | 350 |
| > 70 years | 1200 | 2500 | 600 | 2000 | 700 | 3000 | 420 | 320 | 350 |
| Pregnancy: | |||||||||
| <18 years | 1300 | 2500 | 200 | 2000 | 1250 | 3500 | 400 | 350 | |
| 19–30 years | 1000 | 2500 | 200 | 2000 | 700 | 3500 | 350 | 350 | |
| 31–50 years | 1000 | 2500 | 200 | 2000 | 700 | 3500 | 360 | 350 | |
| Lactation: | |||||||||
| <18 years | 1300 | 2500 | 200 | 2000 | 1250 | 4000 | 360 | 350 | |
| 19–3 years | 1000 | 2500 | 200 | 2000 | 700 | 4000 | 310 | 350 | |
| 31–50 years | 1000 | 2500 | 200 | 2000 | 700 | 4000 | 320 | 350 | |
Footnote: Represents intake from pharmacological agents only, does not include intake from food and water.
AI = Adequate Intakes
UL = Tolerable Upper Intake Levels (represents intake from pharmacological agents only, does not include intake from food and water.)
RDA = Recommended Daily Allowances
ND = Not Determinable
[Source: IOM 1997 45]What foods provide vitamin D ?
Very few foods naturally have vitamin D.
The flesh of fatty fish (such as salmon, tuna, and mackerel) and fish liver oils are among the best sources 46, 47. Small amounts of vitamin D are found in beef liver, cheese, and egg yolks. Vitamin D in these foods is primarily in the form of vitamin D3 and its metabolite 25(OH)D3 48. Some mushrooms provide vitamin D2 in variable amounts 49, 50. Mushrooms with enhanced levels of vitamin D2 from being exposed to ultraviolet light under controlled conditions are also available.
The U.S. Department of Agriculture’s (USDA’s) Nutrient Database website 51 lists the nutrient content of many foods with vitamin D arranged by nutrient content 52 and by food name 53.
Fortified foods provide most of the vitamin D in the American diet 46, 50. For example, almost all of the U.S. milk supply is voluntarily fortified with 100 IU/cup 46. (In Canada, milk is fortified by law with 35–40 IU/100 mL, as is margarine at ≥530 IU/100 g.) In the 1930s, a milk fortification program was implemented in the United States to combat rickets, then a major public health problem 46. Other dairy products made from milk, such as cheese and ice cream, are generally not fortified. Ready-to-eat breakfast cereals often contain added vitamin D, as do some brands of orange juice, yogurt, margarine and other food products.
Both the United States and Canada mandate the fortification of infant formula with vitamin D: 40–100 IU/100 kcal in the United States and 40–80 IU/100 kcal in Canada 46.
Fortified foods provide most of the vitamin D in American diets.
- Fatty fish such as salmon, tuna, and mackerel are among the best sources.
- Beef liver, cheese, and egg yolks provide small amounts.
- Mushrooms provide some vitamin D. In some mushrooms that are newly available in stores, the vitamin D content is being boosted by exposing these mushrooms to ultraviolet light.
- Almost all of the U.S. milk supply is fortified with 400 IU of vitamin D per quart. But foods made from milk, like cheese and ice cream, are usually not fortified.
- Vitamin D is added to many breakfast cereals and to some brands of orange juice, yogurt, margarine, and soy beverages; check the labels.
| Food | IUs per serving* | Percent DV** |
|---|---|---|
| Cod liver oil, 1 tablespoon | 1,360 | 340 |
| Swordfish, cooked, 3 ounces | 566 | 142 |
| Salmon (sockeye), cooked, 3 ounces | 447 | 112 |
| Tuna fish, canned in water, drained, 3 ounces | 154 | 39 |
| Orange juice fortified with vitamin D, 1 cup (check product labels, as amount of added vitamin D varies) | 137 | 34 |
| Milk, nonfat, reduced fat, and whole, vitamin D-fortified, 1 cup | 115-124 | 29-31 |
| Yogurt, fortified with 20% of the DV for vitamin D, 6 ounces (more heavily fortified yogurts provide more of the DV) | 80 | 20 |
| Margarine, fortified, 1 tablespoon | 60 | 15 |
| Sardines, canned in oil, drained, 2 sardines | 46 | 12 |
| Liver, beef, cooked, 3 ounces | 42 | 11 |
| Egg, 1 large (vitamin D is found in yolk) | 41 | 10 |
| Ready-to-eat cereal, fortified with 10% of the DV for vitamin D, 0.75-1 cup (more heavily fortified cereals might provide more of the DV) | 40 | 10 |
| Cheese, Swiss, 1 ounce | 6 | 2 |
* IUs = International Units.
** DV = Daily Value. DVs were developed by the U.S. Food and Drug Administration to help consumers compare the nutrient contents among products within the context of a total daily diet. The DV for vitamin D is currently set at 400 IU for adults and children age 4 and older. Food labels, however, are not required to list vitamin D content unless a food has been fortified with this nutrient. Foods providing 20% or more of the DV are considered to be high sources of a nutrient, but foods providing lower percentages of the DV also contribute to a healthful diet.
Animal-based foods can provide some vitamin D in the form of 25(OH)D, which appears to be approximately five times more potent than the parent vitamin in raising serum 25(OH)D concentrations 55. One study finds that taking into account the serum 25(OH)D content of beef, pork, chicken, turkey, and eggs can increase the estimated levels of vitamin D in the food from two to 18 times, depending upon the food 55. At the present time, the USDA’s Nutrient Database does not include 25(OH)D when reporting the vitamin D content of foods. Actual vitamin D intakes in the U.S. population may be underestimated for this reason.
Other Nutrients Important to Bone
The Institute of Medicine 45 recently provided recommended intakes for other bone-related nutrients, including phosphorus and magnesium (see Table 4: Adequate Intakes (Al) or Recommended Daily Allowances (RDA) and Tolerable Upper Intake Levels (UL) for Calcium, Vitamin D, Phosphorus, and Magnesium by Life-Stage Group for United States and Canada). Most Americans consume adequate quantities of phosphorus through their regular intake of meats, cereals, milk, and processed foods. While some beverages such as soft drinks also contain phosphorus, they are not a preferred source of phosphorus because they may displace calcium-rich beverages like milk 56.
Magnesium intakes may be suboptimal in those who do not eat enough green leafy vegetables, whole grains, nuts, and dairy products. Fortunately, most diets contain adequate levels of other bone-related micronutrients, such as vitamins K and C, copper, manganese, zinc, and iron, to promote bone health.
Some dietary components may potentially have negative effects on bone health, especially if calcium intakes are not adequate. For example, high levels of sodium or caffeine intake can increase calcium excretion in the urine. The effects of these factors can be overcome by increasing the amount of calcium in the diet 57. Studies have linked excessive amounts of phosphorus to altered calcium metabolism, but it appears that the typical level of phosphorus consumed by most individuals in the United States should not negatively affect bone health 45. Excessive amounts of preformed vitamin A (e.g., retinol) can also have negative effects on bone, so individuals should not consume more than the recommended dietary allowance for this vitamin 58. The vitamin A precursor (beta carotene) found in many fruits and vegetables does not have negative effects on bone, however.
Vitamin K and Osteoporosis
The effect of vitamin K intakes and status on bone health and osteoporosis has been a focus of scientific research 59, 60, 61. Matrix Gla-protein, a vitamin K-dependent protein present in vascular smooth muscle, bone, and cartilage, is the focus of considerable scientific research because it might help reduce abnormal calcification 62.
Vitamin K is a cofactor for the gamma-carboxylation of many proteins, including osteocalcin, one of the main proteins in bone 63. Some research indicates that high serum levels of undercarboxylated osteocalcin are associated with lower bone mineral density 64, 63. Some, but not all, studies also link higher vitamin K intakes with higher bone mineral density and/or lower hip fracture incidence 65, 66, 67, 68, 69, 70.
Although vitamin K is involved in the carboxylation of osteocalcin, it is unclear whether supplementation with any form of vitamin K reduces the risk of osteoporosis. In 2006, Cockayne and colleagues conducted a systematic review and meta-analysis of randomized controlled trials that examined the effects of vitamin K supplementation on bone mineral density and bone fracture 71. Most of the trials were conducted in Japan and involved postmenopausal women; trial duration ranged from 6 to 36 months. Thirteen trials were included in the systematic review, and 12 showed that supplementation with either phytonadione or MK-4 improved bone mineral density. Seven of the 13 trials also had fracture data that were combined in a meta-analysis. All of these trials used MK-4 at either 15 mg/day (1 trial) or 45 mg/day (6 trials). MK-4 supplementation significantly reduced rates of hip fractures, vertebral fractures, and all nonvertebral fractures.
A subsequent clinical trial found that MK-7 supplementation (180 mcg/day for 3 years) improved bone strength and decreased the loss in vertebral height in the lower thoracic region of the vertebrae in postmenopausal women 72. Other randomized clinical trials since the 2006 review by Cockayne et al. have found that vitamin K supplementation has no effect on bone mineral density in elderly men or women 73, 74. In one of these studies, 381 postmenopausal women received either 1 mg phylloquinone, 45 mg MK-4, or placebo daily for 12 months 74. All participants also received daily supplements containing 630 mg calcium and 400 IU vitamin D3. At the end of the study, participants receiving either phylloquinone or MK-4 had significantly lower levels of undercarboxylated osteocalcin compared to those receiving placebo. However, there were no significant differences in bone mineral density of the lumbar spine or proximal femur among any of the treatment groups. The authors noted the importance of considering the effect of vitamin D on bone health when comparing the results of vitamin K supplementation studies, especially if both vitamin K and vitamin D (and/or calcium) are administered to the treatment group but not the placebo group. The administration of vitamin D and/or calcium along with vitamin K could partly explain why some studies have found that vitamin K supplementation improves bone health while others have not.
In Japan and other parts of Asia, a pharmacological dose of MK-4 (45 mg) is used as a treatment for osteoporosis 64. The European Food Safety Authority has approved a health claim for vitamin K, noting that “a cause and effect relationship has been established between the dietary intake of vitamin K and the maintenance of normal bone” 75. The FDA has not authorized a health claim for vitamin K in the United States.
Physical Activity
Weight-bearing, strength, and balance-training exercises are also an important part of any osteoporosis prevention and treatment program, regardless of age. They can help increase or preserve bone mass and may also help reduce the risk of falling. All types of physical activity can contribute to bone health. Activities that are weight bearing or involve impact are most useful for increasing or maintaining bone mass. Since continued physical activity provides a positive stimulus for bone, muscle, and other aspects of health, a lifelong commitment to physical activity and exercise is critical. Ending a physical activity regimen will result in bone mass returning to the level that existed before the activity began. Since repetitive programs of physical activity may be discontinued due to lack of motivation or interest, variety and creativity are important if physical activity is to be continued over the long term.
Physical activity will only affect bone at the skeletal sites that are stressed (or loaded) by the activity. In other words, physical activity programs do not necessarily benefit the whole skeleton, although any type of activity provides more benefit to bone than does no activity at all. For bone gain to occur, the stimulus must be greater than that which the bone usually experiences. Static loads applied continuously (such as standing) do not promote increased bone mass.
Complete lack of activity, such as periods of immobility, causes bone loss. When it is not possible to avoid immobility (e.g., bed rest during sickness), even brief daily weight-bearing movements can help to reduce bone loss. General physical activity every day and some weight-bearing, strength-building, and balance-enhancing activities 2 or more times a week are generally effective for promoting bone health for most persons.
Any activity that imparts impact (such as jumping or skipping) may increase bone mass more than will low- and moderate-intensity, endurance-type activities, such as brisk walking. However, endurance activities may still play an important role in skeletal health by increasing muscle mass and strength, balance, and coordination, and they may also help prevent falls in the elderly. Endurance activity is also very important for other aspects of health, such as helping to prevent obesity, diabetes, or cardiovascular disease.
Load-bearing physical activities such as jumping need not be engaged in for long periods of time to provide benefits to skeletal health. In fact, 5–10 minutes daily may suffice. Most adults should begin with weight-bearing exercise and gradually add some skipping and jumping activity. Longer periods (30–45 minutes) may be needed for weight training or walking/jogging. Those who have been inactive should work up to this amount of time gradually using a progressive program, e.g., start with shorter times and easier activities (light weights or walking) and then increase time or intensity slowly (by no more than 10 percent each week) in order to avoid injury.
Activities that are not weight bearing or are low impact may help improve balance and coordination and maintain muscle mass, which can help prevent falls. To encourage increased levels of physical activity among all age groups, “Physical Activity and Health: A Surgeon General’s Report” 76 recommends a “minimum of 30 minutes of physical activity of moderate intensity (such as brisk walking) on most, if not all, days of the week”. Since the skeleton responds preferentially to strength training and short bouts of high-load impact activity (such as skipping or jumping), the same report recommends that adults supplement their cardiorespiratory endurance activity with strength-developing exercise at least two times per week.
For those who cannot engage in regular physical activity due to disability, mechanical stimulation of the skeleton might prove beneficial. Recent, small studies found that use of vibrating platforms increased BMD (bone mineral density) and slowed bone loss 77, 78, 79. This may provide another way to reduce fracture risk both in the elderly and in younger individuals with disabling conditions that limit their ability to exercise. However, the long-term safety and efficacy of such approaches remain to be determined, and therefore specific rehabilitation and exercise programs aimed at increasing activity and function remain critically important in the frail elderly and in younger individuals with neuromuscular disabilities.
As noted earlier, the evidence does not lead to a specific set of exercises or practices but rather a set of principles that can be applied and varied according to the age and current physical condition of an individual. Many of these principles have been reviewed by expert panels of the American College of Sports Medicine 80 and they lead to the following suggestions for the frequency, intensity, length, and type of physical activity regimens to benefit bone health for individuals of all ages:
- Physical activities that include a variety of loading patterns (such as strength training or aerobic classes) may promote increased bone mass more than do activities that involve normal or regular loading patterns (such as running).
- General physical activity every day and some weight-bearing, strength-building, and balance-enhancing activities 2 or more times a week are generally effective for promoting bone health for most persons.
- Any activity that imparts impact (such as jumping or skipping) may increase bone mass more than will low- and moderate-intensity, endurance-type activities, such as brisk walking. However, endurance activities may still play an important role in skeletal health by increasing muscle mass and strength, balance, and coordination, and they may also help prevent falls in the elderly. Endurance activity is also very important for other aspects of health, such as helping to prevent obesity, diabetes, or cardiovascular disease.
- Load-bearing physical activities such as jumping need not be engaged in for long periods of time to provide benefits to skeletal health. In fact, 5–10 minutes daily may suffice. Most adults should begin with weight-bearing exercise and gradually add some skipping and jumping activity. Longer periods (30–45 minutes) may be needed for weight training or walking/jogging. Those who have been inactive should work up to this amount of time gradually using a progressive program, e.g., start with shorter times and easier activities (light weights or walking) and then increase time or intensity slowly (by no more than 10 percent each week) in order to avoid injury.
- Physical activities that include a variety of loading patterns (such as strength training or aerobic classes) may promote increased bone mass more than do activities that involve normal or regular loading patterns (such as running).
Physical Activity for Older Adults
Most elderly individuals should strongly consider engaging in regular physical activity. Physical activity is the only single therapy that can simultaneously improve muscle mass, muscle strength, balance, and bone strength. As a result, it may decrease the risk of fractures, in part by reducing the risk of falling. In fact, fall-risk reduction may be the biggest benefit of physical activity for the elderly.
This summary of a Cochrane review 81 presents what we know from research about the effect of exercise on bone mass in postmenopausal women. The review shows that for postmenopausal women the results suggest a relatively small statistically significant, but possibly important, effect of exercise on bone density compared with control groups. Exercise has the potential to be a safe and effective way to avert bone loss in postmenopausal women.
- Exercise will improve bone mineral density slightly.
- Exercise will reduce the chances of having a fracture slightly.
The following guidelines should be used to maximize the potential fall prevention benefits of physical activity in the elderly:
- Physical activity needs to be of sufficient intensity to improve muscle strength, since poor muscle strength is a known risk factor for falls. Strength or resistance training is best for building muscle, but even aerobic endurance activity can yield some improvements in muscle strength.
- Improving balance can be an important component of any physical activity program designed to decrease falls. This program may include balance training exercises or a movement activity such as Tai Chi. Any activity that requires weight bearing and challenges the postural system can improve balance and potentially help reduce falls.
- Physical activity must be performed on average 3 times per week for 30–45 minutes per session for at least three months for strength and balance benefits to be realized, and it must be continued if benefits are to be maintained.
- Those who suffer a fall that requires a visit to a health care provider or an emergency room should ask for a fall risk assessment that includes a program of physical activity.
Fall Prevention
Falls are not just the result of getting older and falls represent perhaps the biggest threat to the bone health and the functional independence of older individuals. Falls are common and frequently are the precipitating event that leads to a fracture or fractures in an individual. Thus, fall prevention offers another important opportunity to protect the bones throughout life, but particularly in those over age 60. Falls occur for a variety of reasons, with multiple factors often contributing to a single fall. These factors include problems with balance, mobility, vision, lower extremity weakness, and/or blood pressure or circulation. Often these problems are compounded by an acute illness (e.g., infection, fever, dehydration, arrhythmia), a new medication, or an environmental stress (e.g., standing or walking on an unsafe surface, poor lighting) that leads to the fall. To reduce the risk of falls, a variety of fall prevention measures should be encouraged for frail, elderly individuals. These include regular vision checks; elimination (where possible) of medications and/or dosages that may cause dizziness, low blood pressure, or confusion; and addressing environmental problems or obstacles that can lead to falls, including removing throw rugs, installing night lights, installing railings on stairs and grab bars in showers, encouraging use of rubber-soled shoes and slippers, and attaching phone cords and other wires to the baseboard of the wall. Hip protectors or hip pads might also be useful in reducing the impact of those falls that do occur.
Many falls can be prevented. Falls are usually caused by a number of things. By changing some of these things, you can lower your chances of falling:
- Begin a regular exercise program: Exercise is one of the most important ways to reduce your chances of falling. It makes you stronger and helps you feel better. Exercises that improve balance and coordination (like tai chi) are the most helpful. Lack of exercise leads to weakness and increases your chances of falling. Ask your doctor or health care worker about the best type of exercise program for you.
- Have your eyes checked by an eye doctor. You may be wearing the wrong glasses or have a condition such as glaucoma or cataracts that limits your vision. Poor vision can increase your chances of falling.
- Make your home safer: About half of all falls happen at home. To make your home safer:
- Remove things you can trip over (such as papers, books, clothes, and shoes) from stairs and places where you walk.
- Remove small throw rugs or use double-sided tape to keep the rugs from slipping.
- Keep items you use often in cabinets you can reach easily without using a stepstool.
- Have grab bars put in next to your toilet and in the tub or shower.
- Use nonslip mats in the bathtub and on shower floors.
- Improve the lighting in your home. As you get older, you need brighter lights to see well. Lamp shades or frosted bulbs can reduce glare.
- Have handrails and lights put in on all staircases.
- Wear shoes that give good support and have thin nonslip soles. Avoid wearing slippers and athletic shoes with deep treads.
Nutrition
Nutrition has been shown to be important during recovery from hip fracture. Supplementation with calcium, vitamin D, and protein (20 grams per day) have been reported to improve hospital and rehabilitation courses and to increase bone mineral density a year after the fracture 82.
- Healthy nutrition, since many nutrients are important for bone health, it is important to eat a well-balanced diet containing a variety of foods. Following the Dietary Guidelines for Americans (2015-2020) 83. From childhood into old age, a diet low in calcium and vitamin D can increase your risk of osteoporosis and fractures. Excessive dieting or inadequate caloric intake can also be bad for bone health. People who are very thin and do not have much body fat to cushion falls have an increased risk of fracture.
In addition to having a healthy diet, sufficient physical activity, and avoiding falls, there are some other bone-healthy behaviors that can help protect the skeleton throughout life:
- Maintain a healthy body weight.
- Avoid smoking. Most studies indicate that smoking is a risk factor for osteoporosis and fracture, although the exact reasons for the harmful effects of tobacco use on bone health are unclear.
- If you drink alcoholic beverages, do so in moderation (i.e., one drink per day for women and two drinks per day for men). People who drink heavily are more prone to bone loss and fractures because of poor nutrition and harmful effects on calcium balance and hormonal factors. Drinking too much also increases the risk of falling, which is likely to increase fracture risk 84.
- For women, see a health care provider if menstrual periods stop for three months.
- For those who have a medical condition or who use medications that can affect the skeleton, talk to a health care provider about ways to safeguard your skeleton.
- An inactive lifestyle or extended bed rest: Low levels of physical activity and prolonged periods of inactivity can contribute to an increased rate of bone loss. They also leave you in poor physical condition, which can increase your risk of falling and breaking a bone.
How is Osteoporosis Diagnosed
Many current clinical guidelines recommend that assessment of osteoporosis or low bone mass, as defined by the World Health Organization 1, be based on bone mineral density 85 at either the femur neck region of the proximal femur (hip) or the lumbar spine 2, 86.
Mineral is what gives hardness to bones, and the density of mineral in the bones is an important determinant of bone strength. Bone mineral density testing can be used to definitively diagnose osteoporosis, detect low bone mass before osteoporosis develops, and help predict your risk of future fractures 8. If it is thought that someone might have osteoporosis, their bone density can be measured. Here the amount of minerals in the bones is measured, which can help to estimate how high the risk of a bone fracture is. This test is also done to help plan treatment or see how well it is working.
In general, the lower your bone density, the higher your risk for fracture. The results of a bone density test will help guide decisions about starting therapy to prevent or treat osteoporosis. Bone mineral density testing may also be used to monitor the effectiveness of ongoing therapy.
The most widely recognized test for measuring bone mineral density is a quick, painless, noninvasive technology known as central dual-energy x-ray absorptiometry (DXA). This technique, which uses low levels of x rays, involves passing a scanner over your body while you are lying on a cushioned table. DXA can be used to determine bone mineral density of the entire skeleton and at various sites that are prone to fracture, such as the hip, spine, or wrist. Bone density measurement by DXA at the hip and spine is generally considered the most reliable way to diagnose osteoporosis and predict fracture risk.
The results of bone mineral density tests are reported as T-scores. According to current definitions,
- a T-score above -1 is normal,
- a T-score between -1 and -2.5 is below normal, and
- a T-score of -2.5 or less means you have osteoporosis.
A bone mineral density test on its own can only partially predict a person’s risk of bone fractures. There are other factors besides bone density that influence the health of your bones. These include the quality of the bone tissue and your skeletal structure. Measurements of bone mineral density are not 100% accurate either, and it is also important to take into account things like a person’s general health, other conditions and any risk factors.
Other types of tests may be used too – for example, a blood test could be used to rule out other conditions that could be causing the loss of bone mass.
Treatment of Osteoporosis
One of the primary goals in the treatment of osteoporosis and other bone diseases is to maintain bone health by preventing bone loss and perhaps even by building new bone. Another goal is to minimize the risk and/or impact of falls, since they are typically the precursor to the most devastating consequence of bone disease: fractures. The best way to realize these goals is to employ a combination of various prevention and treatment strategies.
- The first step in the prevention and treatment of osteoporosis and the prevention of fractures is to build a foundation of nutrition and lifestyle measures that maximize bone health. The diet should not only be adequate in calcium and vitamin D, but should have a healthy balance of other nutrients. A weight-bearing exercise program should be developed. Cigarette smoking and excessive alcohol use must be avoided. In the older individual, at high risk for fractures, the changes in lifestyle would include a plan not only to maximize physical activity, but also to minimize the risk of falls. The use of hip protectors can be considered in some high-risk patients. Diseases that increase the risk of falls by causing visual impairment, postural hypotension (a drop in blood pressure on standing, which leads to dizziness), or poor balance should be treated. Drugs that cause bone loss or increase the risk of falls should be avoided or given at the lowest effective dose.
- The Second Step: The next step is to identify and treat diseases that produce secondary osteoporosis or aggravate primary osteoporosis. These measures are the foundation upon which specific pharmacotherapy is built and should never be forgotten.
- The Third Step: If there is sufficiently high risk of fracture to warrant pharmacotherapy, the patient is usually started on antiresorptives. Anabolic agents are used in individuals in whom antiresorptive therapy is not adequate to prevent bone loss or fractures 87.
Medications for Treatment of Osteoporosis
The U.S. Food and Drug Administration (FDA) has approved several medications for prevention or treatment of osteoporosis, based on their ability to reduce fractures.
All medicines can have side effects. Some medicines and side effects are mentioned in this publication. Some side effects may be more severe than others. You should review the package insert that comes with your medicine and ask your health care provider or pharmacist if you have any questions about the possible side effects.
- Bisphosphonates
Bisphosphonates, such as etidronate, alendronate, ibandronate, are first-line drug therapy and are commonly used drugs for treatment of postmenopausal osteoporosis. Several bisphosphonates are approved for the prevention or treatment of osteoporosis. These medications slows down the cells that break down the old bone. This means the cells that grow new bone have a chance to catch up and strengthen the bone.
By inhibiting bone resorption, bisphosphonates preserve bone mass and can decrease vertebral and hip fractures by up to 50%. Bone turnover is reduced after 3 mo of bisphosphonate therapy and fracture risk reduction is evident as early as 1 yr after beginning therapy. DXA scanning, when done serially to monitor response to treatment, need not normally be done at intervals < 2 yr. Bisphosphonates can be given orally or intravenously (IV) 4.
Overall, a review 88 supports the use of bisphosphonates to reduce the risk of spinal fractures and in the prevention and treatment of steroid-induced bone loss at the lumbar spine and femoral neck.
For preventing non-spinal fractures, the authors found that there was little or no difference whether patients used bisphosphonates or not, although this evidence was low-certainty because the methods used to assess non-spinal fractures were subject to bias.
Bisphosphonates include the following:
- Alendronate (10 mg once/day or 70 mg po once/wk)
- Risedronate (5 mg po once/day, 35 mg po once/wk, or 150 mg po once/mo)
- Zoledronic acid (5 mg IV once/yr)
- Ibandronate po (150 mg once/mo) or IV (3 mg once every 3 mo)
Oral bisphosphonates must be taken on an empty stomach with a full (8-oz, 250 mL) glass of water, and the patient must remain upright for at least 30 min (60 min for ibandronate) and not take anything else by mouth during this time period. These drugs are safe to use in patients with a creatinine clearance > 35 mL/min. Bisphosphonates can cause esophageal irritation 4. Esophageal disorders that delay transit time and symptoms of upper GI disorders are relative contraindications to oral bisphosphonates. IV bisphosphonates are indicated if a patient is unable to tolerate or is nonadherent with oral bisphosphonates 4.
Osteonecrosis of the jaw has been associated with use of bisphosphonates; however, this condition is rare in patients taking oral bisphosphonates 89. Current screening and diagnostic tests are unreliable for predicting a patient’s risk of developing the condition 89. Osteonecrosis of the jaw may occur spontaneously or after dental extraction or trauma, radiation therapy to the head and neck (osteoradionecrosis), or high-dose IV bisphosphonate therapy (eg, for cancer treatment). Osteonecrosis of the jaw may be a refractory osteomyelitis rather than true osteonecrosis, particularly when associated with bisphosphonate use. There is no evidence that routine use of oral bisphosphonates for treatment or prevention of osteoporosis increases risk of ONJ. Currently, otherwise appropriate bisphosphonate use should not be discouraged. However, it seems reasonable to do any necessary oral surgery before beginning bisphosphonate therapy and to encourage good oral hygiene while patients are taking bisphosphonates 89. The benefits of reduction of osteoporosis-related fractures far outweigh this small risk.
Long-term bisphosphonate use may also increase the risk of atypical femoral fractures 4. These fractures occur in the mid-shaft of the femur with minimal or no trauma and may be preceded by weeks or months of thigh pain. The fractures may also be bilateral. To minimize fracture incidence, consideration should be given to stopping bisphosphonates (a bisphosphonate holiday) 4 after about:
- 3 to 5 yr of use in patients with osteoporosis (by DXA scan) but few or no other risk factors for bone loss (3 yr for IV zoledronic acid and 5 yr for oral bisphosphonates)
- 5 to 10 yr of use in patients with osteoporosis (by DXA scan) and more risk factors
Patients on a bisphosphonate holiday should be closely monitored for a new fracture or accelerated bone loss evident on a DXA scan. During therapy with an antiresorptive drug, such as a bisphosphonate, bone turnover is suppressed as evidenced by low fasting N-telopeptide cross-links (< 40 nmol/L) or C-telopeptide cross-links. These markers may remain low for ≥ 2 yr of a drug holiday.
In untreated patients, an increase in levels of bone turnover markers indicates an increased risk of fracture. However, it is not clear whether levels of bone turnover markers should be used as criteria for when to start or end a drug holiday. The decision to begin or end a drug holiday is complex and should take into account the patient’s risk factors.
Intranasal salmon calcitonin should not regularly be used for treating osteoporosis 4. Salmon calcitonin may provide short-term analgesia after an acute fracture, such as a painful vertebral fracture, due to an endorphin effect. It has not been shown to reduce fractures 4.
- Bisphosphonates in Children and Adolescents with secondary osteoporosis
This evidence 90 does not support bisphosphonates as standard therapy for children and adolescents with secondary osteoporosis. Short-term (3 years or less) bisphosphonate use appears to be well-tolerated. An accepted criterion for osteoporosis in children, a standardized approach to bone mineral density reporting, and examining functional bone health outcomes (e.g., fracture rates) will allow for appropriate comparisons across studies.
- Parathyroid hormone
A form of human parathyroid hormone (PTH) is approved for postmenopausal women and men with osteoporosis who are at high risk for having a fracture. It helps the body build up new bone faster than the old bone is broken down 4.
Parathyroid hormone which stimulates new bone formation, is generally indicated in patients who have the following characteristics:
- Cannot tolerate antiresorptive drugs or have contraindications to their use
- Fail to respond (ie, develop new fractures or lose bone mineral density) to antiresorptive drugs, as well as calcium, vitamin D, and exercise
- Possibly have severe osteoporosis (eg, T-score < -3.5) or multiple vertebral fragility fractures
- Have glucocorticoid-induced osteoporosis
When given daily by injection for an average of 20 mo, synthetic PTH (PTH 1-34; teriparatide) 4. Teriparatide is an injectable form of human parathyroid hormone. It increases bone mass and reduces risk of fractures. Patients taking teriparatide should have a creatinine clearance > 35 mL/min. Use of the drug for more than 2 years is not recommended 4.
- RANK ligand (RANKL) inhibitor
A RANK ligand (RANKL) inhibitor is approved for postmenopausal women with osteoporosis and men who are at high risk for fracture. Denosumab is a monoclonal antibody against RANKL (receptor activator of nuclear factor kappa-B ligand) and reduces bone resorption by osteoclasts 4. Denosumab may be helpful in patients not tolerant of or unresponsive to other therapies or in patients with impaired renal function. Denosumab is contraindicated in patients with hypocalcemia and in pregnant women 4.
- Estrogen agonists/antagonists (SERM)
An estrogen agonist/antagonist (also called a selective estrogen receptor modulator or SERM) is approved for the prevention and treatment of osteoporosis in postmenopausal women. SERMs are not estrogens, but they have estrogen-like effects on some tissues and estrogen-blocking effects on other tissues.
Raloxifene is a selective estrogen receptor modulator (SERM) that may be appropriate for treatment of osteoporosis in women who cannot take bisphosphonates 4. It reduces vertebral fractures by about 50% but has not been shown to reduce hip fractures 4. Raloxifene does not stimulate the uterus and antagonizes estrogen effects in the breast. It has been shown to reduce the risk of invasive breast cancer. Raloxifene has been associated with an increased risk of thromboembolism 4.
- Calcitonin
Calcitonin is approved for the treatment of osteoporosis in women who are at least 5 years beyond menopause. Calcitonin is a hormone involved in calcium regulation and bone metabolism.
The results 91 showed that calcitonin prevents bone loss at the spine and forearm by about 3% after the first year of therapy. There was no effect on bone loss at the hip. Calcitonin was not statistically different from placebo at preventing fractures of the spine and long bones, such as hip fractures. Efficacy of calcitonin for fracture prevention in steroid-induced osteoporosis remains to be established. Calcitonin was associated with four times the side effects of placebo, and these were mostly nausea and facial flushing.
- Estrogen and hormone therapy
Estrogen can preserve bone density and prevent fractures 4. Most effective if started within 4 to 6 yr of menopause, estrogen may slow bone loss and possibly reduce fractures even when started much later 4. Use of estrogen increases the risk of thromboembolism and endometrial cancer and may increase the risk of breast cancer. The risk of endometrial cancer can be reduced in women with an intact uterus by taking a progestin with estrogen 4. However, taking a combination of a progestin and estrogen increases the risk of breast cancer, coronary artery disease, stroke, and biliary disease. Because of these risks and the availability of other treatments for osteoporosis, the potential harms of estrogen treatment for osteoporosis treatment outweigh its potential benefits for most women; when treatment is initiated, a short course with close monitoring should be considered.
Estrogen and combined estrogen and progestin (hormone therapy) are approved for the prevention of postmenopausal osteoporosis as well as the treatment of moderate to severe hot flashes and vaginal dryness that may accompany menopause. Estrogen without an added progestin is recommended only for women who have had a hysterectomy (surgery to remove the uterus), because estrogen increases the risk of developing cancer of the uterine lining and progestin reduces that risk.
The Food and Drug Administration has recommended that women use hormone therapy at the lowest dose and for the shortest time, and carefully consider and discuss with their doctor other approved osteoporosis treatments.
No drug should be considered indefinite in duration. All non-bisphosphonate medications produce temporary effects that wane upon discontinuation. If these treatments are stopped, benefits rapidly disappear. In contrast, bisphosphonates may allow residual effects even after treatment discontinuation. Therefore, it may be possible to discontinue bisphosphonates and retain residual benefits against fracture at least for several years.Since there is no extensive evidence base to guide treatment duration decisions, duration decisions need to be individualized 92. After the initial 3- to 5-year treatment period, a comprehensive risk assessment should be performed. This should include interval clinical history, particularly with respect to intercurrent fracture history and new chronic diseases or medications, as well as height measurement, BMD testing, and vertebral imaging if there has been any documented height loss during the treatment period. It is reasonable to discontinue bisphosphonates after 3 to 5 years in people who appear to be at modest risk of fracture after the initial treatment period. In contrast, for those who appear to be at high risk for fracture, continued treatment with a bisphosphonate or an alternative therapy should be considered 93.
The idea of combining an anabolic agent with an anti-resorptive therapy has been around for more than a decade. With the advent of human parathyroid hormone, combination studies have been proposed to evaluate the impact of human parathyroid hormone therapy with antiresorptive therapies such as estrogen and alendronate. Parathyroid hormone plus estrogen has been shown to have a greater effect on spine and hip bone mineral density than estrogen alone, but there are no trials comparing that combination to human parathyroid hormone alone, nor are there any studies that evaluate differences in the impact on fracture rates 94. Two studies examining the effects of simultaneous PTH and alendronate treatment suggest that there may actually be smaller bone mineral density increases with combination therapy than with parathyroid hormone alone 95, 96.
- Chinese herbal medicines for osteoporosis
A review of 108 studies with 10,655 people 97 who took Chinese herbal medicines for their osteoporosis. The review found that the beneficial effect of Chinese herbal medicines in improving BMD is still uncertain and more rigorous studies are warranted.
- Fluoride
Although fluoride has an ability to increase bone mineral density at lumbar spine, it does not result in a reduction of vertebral fractures. In increasing the dose of fluoride, one increases the risk of non-vertebral fracture and gastrointestinal side effects without any effect on the vertebral fracture rate 98.
Treatment of Osteoporotic Fractures
For all osteoporotic fractures, the consistent goal is for patients to regain their pre-fracture level of function. All patients with low-trauma fractures should be evaluated for other bone diseases, secondary causes of bone loss and they should also be evaluated with respect to the need for additional preventive measures (calcium, vitamin D, exercise, fall prevention) and for drug therapy. What follows is a review of the various available treatments for specific types of osteoporotic-related fractures, including fractures of the hip, spine, and wrist.
- Hip Fractures
Surgery is the most common treatment for individuals who suffer a hip fracture. Virtually all intertrochanteric fractures (those in the major part of the hip) and most femoral neck fractures (those in the neck section of the hip) are surgically stabilized with the use of internal metal devices. A large percentage of displaced (unconnected) femoral neck fractures are treated with partial or total replacement of the hip because of the significant risk of healing complications 99.
- Spine Fractures
Spine fractures usually occur in the middle or lower section of the back as a result of minor strain, such as lifting a grocery bag. Some patients develop fractures without any identifiable trauma. Spine fractures due to osteoporosis result in the progressive collapse of bones in these areas, which typically cause increasing levels of spinal deformity and pain. However, about two-thirds of spine fractures go undiagnosed because there is little or no pain, or the pain is attributed to one of the many other causes of back pain 100. Similarly, other signs of a spine fracture, including deformities and height loss, are often accepted as a normal part of aging and thus not investigated further.
It is not unusual for patients to have prolonged pain and disability following spine fractures. Treatment of spine fractures typically focuses on pain control and progressive increases in levels of mobilization. Back braces are of limited benefit. More recently, procedures have been developed to treat patients who have prolonged pain. Vertebroplasty is a technique in which acrylic cement (or orthopedic cement mixture) is injected into the spine bone for the purpose of stabilizing the fracture 101. Kyphoplasty involves using a balloon to re-expand the collapsed bone and then filling the cavity with bone cement. This procedure has the potential to stabilize the fracture, prevent further collapse, and restore some degree of height to the bone 102. Both vertebroplasty and kyphoplasty have been shown to provide effective pain relief and stabilization of the fracture 101, 102. Although complications from these procedures have been infrequent, they can be significant if the bone cement leaks out into the blood stream or into the spinal canal, causing nerve damage. Unfortunately, the potential benefit of these two procedures has not yet been accurately assessed in randomised double blind clinical trials, where they might be compared to each other and to nonsurgical management.
- Wrist Fractures
Wrist fractures commonly occur as a result of osteoporosis. They include fractures of the radius and/or ulna (the two long bones in the forearm), as well as of the small bones of the wrist. The term Colles’ fractures refers to fractures of the end of the radius, which has a large amount of trabecular bone. Wrist fractures are usually treated by either surgical repositioning and casting or placement of an external fixation device to prevent further fracture. Depending on the type of fracture, one of the following will be used to immobilize the wrist until an x-ray shows evidence of healing (usually in 4–8 weeks): a brace or splint, cast, external fixation, internal fixation, or combined external and internal fixation. Although most patients return to an adequate level of functioning, many do experience some loss of range of motion of the wrist.
- Other Fractures
Osteoporotic fractures occur in other areas of the body, including the upper arm, thigh, shin, collar bone, and ribs. These fractures are treated by a variety of surgical and non-surgical measures.
Exercises for Those With Osteoporosis
Individuals who have already experienced osteoporotic fractures should avoid certain types of physical activities and exercises. For example, those who have had vertebral fractures may need to avoid activities that flex the spine.
Spine Fractures: Walking should be encouraged even in frail individuals. Exercising in a way that safely challenges balance is also important for rehabilitation of spinal fracture patients, but resistance/strengthening exercises should not be initiated or resumed until the fracture has healed (in approximately 8 to 12 weeks). Since the risk of another spine fracture is high in patients who have had fractures, patients should be instructed to avoid exercises and activities that put high loads on the bones of the spine, such as flexing or rotating the spine (sit ups, toe touches). Exercises and activities done with good spine alignment and low to moderate amounts of weight should be gradually increased, with the goals of regaining muscle strength and promoting maintenance of bone mass. Abdominal strengthening (by tightening the muscles in the abdomen or belly without moving the back) is safe and important to reducing loads on the low back. Spinal extension exercise (i.e., stretching backwards) within a moderate range is safe and can improve hyperkyphosis (a spine that is bent excessively forward) and may help prevent new spine fractures 103.
Wrist Fractures: Rehabilitation of the wrist after the cast, brace, or surgical metal is removed requires about 3 months. During healing of a wrist fracture, all of the following are important: arm elevation; early mobilization of the hand, elbow, and shoulder; and control of swelling. Progressive exercises, taught by either a physical or occupational therapist, typically include active and passive range of motion and resistance and grip strengthening, such as squeezing a ball 104. A small number of patients suffer from sympathetic dystrophy (complex regional pain syndrome) after a wrist fracture, resulting in swelling, weakness, and chronic pain in the wrist.
The foundation of a good physical activity regimen involves at least 30 minutes (adults) or 60 minutes (children) of moderate physical activity every day. This regimen can and should involve a variety of activities. Some can be routine activities like walking or gardening. Others may occur more infrequently and differ from day to day and week to week, such as dancing, aerobic classes, biking, swimming, tennis, golf, or hiking. However, it is clear from the evidence presented in Chapter 6 that physical activity to specifically benefit bone health should involve loading (stressing) the skeleton. As a result, weight-bearing activities such as walking should be included in an optimal physical activity regimen to benefit the musculoskeletal system. Moreover, the evidence suggests that the most beneficial physical activity regimens for bone health include strength-training or resistance-training activities. These activities place levels of loading on bone that are beyond those seen in everyday activities; examples include jumping for the lower limbs and weight lifting or resistance training for the lower and upper skeleton. Finally, while a focus on activities that build or maintain bone strength is appropriate and necessary, many older individuals will remain at high risk of fracture. For these individuals, balance training can provide the added benefit of helping to prevent potentially injurious falls.
Summary
Osteoporosis is a progressive metabolic bone disease that decreases bone density (bone mass per unit volume), with deterioration of bone structure. Nine percent of adults over age 50 years had osteoporosis at either the femur neck or lumbar spine and roughly one-half had low bone mass at either of these two skeletal sites. Having osteoporosis raises the risk of experiencing fractures. Skeletal weakness leads to fractures with minor or inapparent trauma, particularly in the thoracic and lumbar spine, wrist, and hip (called fragility fractures). Diagnosis is by dual-energy x-ray absorptiometry (DXA scan) or by confirmation of a fragility fracture.
The prevalence of osteoporosis or low bone mass at either the femur neck or lumbar spine was not the same as the prevalence of these conditions when the two skeletal sites were considered separately because some individuals had these conditions at one of the skeletal sites but not the other. The prevalence of osteoporosis or low bone mass differed by age, sex, and race and ethnicity. The prevalence was higher in women and increased with age. Differences by race and ethnicity varied depending on sex and skeletal status category, but when compared with non-Hispanic white persons, Mexican-American persons, and persons of other races tended to be at higher risk, and non-Hispanic black persons tended to be at lower risk of either osteoporosis or low bone mass at the femur neck or lumbar spine.
Prevention and treatment involve risk factor modification, calcium and vitamin D supplements, exercises to maximize bone and muscle strength, improve balance, and minimize the risk of falls, and drug therapy to preserve bone mass or stimulate new bone formation.
Recommendations
- Diet must include adequate amounts of total calcium intake (1000 mg/day for men 50–70; 1200 mg/day for women 51 and older and men 71 and older), incorporating dietary supplements if diet is insufficient.
- Vitamin D intake (800–1000 IU/day), including supplements if necessary for individuals age 50 and older.
- Regular weight-bearing and muscle-strengthening exercise to improve agility, strength, posture, and balance; maintain or improve bone strength; and reduce the risk of falls and fractures. Since continued physical activity provides a positive stimulus for bone, muscle, and other aspects of health, a lifelong commitment to physical activity and exercise is critical.
- Assess risk factors for falls and offer appropriate modifications (e.g., home safety assessment, balance training exercises, correction of vitamin D insufficiency, avoidance of central nervous system depressant medications, careful monitoring of antihypertensive medication, and visual correction when needed).
- Cessation of tobacco smoking and avoidance of excessive alcohol intake.
There is good evidence that proper nutrition and lifestyle can promote bone health and that medications can slow bone loss or even build new bone. However, there is still no “cure” for osteoporosis.
- Kanis JA, McCloskey EV, Johansson H, et al. A reference standard for the description of osteoporosis. Bone 42(4) 67–75. 2008.[↩][↩]
- National Osteoporosis Foundation. Clinician’s Guide to Prevention and Treatment of Osteoporosis. National Osteoporosis Foundation, Washington, DC. 1–36. 2008.[↩][↩]
- National Center for Biotechnology Information, U.S. National Library of Medicine. – Osteoporosis – https://www.ncbi.nlm.nih.gov/pubmedhealth/PMHT0024680/[↩][↩]
- Merck Sharp & Dohme Corp., Merck Manual. Osteoporosis. https://www.merckmanuals.com/professional/musculoskeletal-and-connective-tissue-disorders/osteoporosis/osteoporosis[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- National Institute of Arthritis and Musculoskeletal and Skin Diseases. Bone Basics. https://www.niams.nih.gov/Health_Info/Bone/Bone_Health/default.asp[↩][↩]
- Khosla S, Riggs BL. Pathophysiology of age-related bone loss and osteoporosis. Endocrinol Metab Clin N Am. 2005;34:1015–1030. doi: 10.1016/j.ecl.2005.07.009. https://www.ncbi.nlm.nih.gov/pubmed/16310636[↩]
- Osteoporos Int. 2014; 25(10): 2359–2381. – Clinician’s Guide to Prevention and Treatment of Osteoporosis – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4176573/[↩]
- National Institute of Health, Osteoporosis and Related Bone Diseases ~ National Resource Center. Osteoporosis Basics . https://www.niams.nih.gov/Health_Info/Bone/Osteoporosis/osteoporosis_hoh.asp[↩][↩]
- National Center for Biotechnology Information, U.S. National Library of Medicine. – Bone Health and Osteoporosis: A Report of the Surgeon General. – https://www.ncbi.nlm.nih.gov/books/NBK45515/[↩]
- National Osteoporosis Foundation. Osteoporosis: What is it?. Washington, DC: National Osteoporosis Foundation; [Cited 2004 Mar 1]. Available from: http://www.nof.org/osteoporosis/index.htm.[↩][↩]
- Office on Women’s Health, U.S. Department of Health and Human Services. Osteoporosis. https://www.womenshealth.gov/a-z-topics/osteoporosis[↩]
- Centre for Metabolic Bone Diseases, University of Sheffield, UK. Fracture Risk Assessment Tool. https://www.sheffield.ac.uk/FRAX/tool.jsp[↩][↩][↩][↩][↩]
- Reviews in Endocrine and Metabolic Disorders, December 2010, Volume 11, Issue 4, pp 237–251. Diagnosis and treatment of osteopenia. https://link.springer.com/article/10.1007%2Fs11154-010-9154-0[↩][↩]
- WHO Scientific Group on Assessment of Osteoporosis at Primary Health Care Level 2004. http://www.who.int/chp/topics/Osteoporosis.pdf[↩]
- AIDSInfo, U.S. Department of Health and Human Services. https://aidsinfo.nih.gov/understanding-hiv-aids/glossary/537/osteopenia [↩]
- Bone mineral density thresholds for pharmacological intervention to prevent fractures. Siris ES, Chen YT, Abbott TA, Barrett-Connor E, Miller PD, Wehren LE, Berger ML. Arch Intern Med. 2004 May 24; 164(10):1108-12. https://www.ncbi.nlm.nih.gov/pubmed/15159268/[↩][↩][↩]
- Zoledronic acid and clinical fractures and mortality after hip fracture. Lyles KW, Colón-Emeric CS, Magaziner JS, Adachi JD, Pieper CF, Mautalen C, Hyldstrup L, Recknor C, Nordsletten L, Moore KA, Lavecchia C, Zhang J, Mesenbrink P, Hodgson PK, Abrams K, Orloff JJ, Horowitz Z, Eriksen EF, Boonen S, HORIZON Recurrent Fracture Trial. N Engl J Med. 2007 Nov 1; 357(18):1799-809. https://www.ncbi.nlm.nih.gov/pubmed/17878149/[↩]
- Bone and fall-related fracture risks in women and men with a recent clinical fracture. van Helden S, van Geel AC, Geusens PP, Kessels A, Nieuwenhuijzen Kruseman AC, Brink PR. J Bone Joint Surg Am. 2008 Feb; 90(2):241-8. https://www.ncbi.nlm.nih.gov/pubmed/18245581/[↩]
- Rev Endocr Metab Disord. 2012 Sep; 13(3): 209–223. Published online 2011 Jun 28. doi: 10.1007/s11154-011-9187-z. Treatment of osteopenia. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3411311/[↩][↩][↩]
- Centers for Disease Control and Prevention. – Osteoporosis or Low Bone Mass at the Femur Neck or Lumbar Spine in Older Adults: United States, 2005–2008 – https://www.cdc.gov/nchs/data/databriefs/db93.htm[↩]
- CDC/NCHS, National Health and Nutrition Examination Survey, 2005–2008.[↩]
- Leslie WD, Majumdar SR, Morin SN, Lix LM: Change in bone mineral density is an indicator of treatment-related antifracture effect in routine clinical practice: A registry-based cohort study. Ann Intern Med 165(7):465–472, 2016. doi: 10.7326/M15-2937. http://annals.org/aim/article/2534408/change-bone-mineral-density-indicator-treatment-related-antifracture-effect-routine[↩][↩]
- National Center for Biotechnology Information, U.S. National Library of Medicine. – Osteoporosis: Overview – https://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0072713/[↩]
- National Center for Biotechnology Information, U.S. National Library of Medicine – Bone Health and Osteoporosis: A Report of the Surgeon General.- https://www.ncbi.nlm.nih.gov/books/NBK45523/[↩]
- National Center for Biotechnology Information, U.S. National Library of Medicine. – Prevention and Treatment for Those Who Have Bone Diseases – https://www.ncbi.nlm.nih.gov/books/NBK45501/[↩]
- The National Institute of Health, Office of Dietary Supplements. Calcium. https://ods.od.nih.gov/factsheets/Calcium-HealthProfessional/ [↩]
- Straub DA. Calcium supplementation in clinical practice: a review of forms, doses, and indications. Nutr Clin Pract. 2007;22:286-96. https://www.ncbi.nlm.nih.gov/pubmed/17507729?dopt=Abstract[↩]
- Committee to Review Dietary Reference Intakes for Vitamin D and Calcium, Food and Nutrition Board, Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D. Washington, DC: National Academy Press, 2010.[↩][↩][↩]
- Andon MB, Peacock M, Kanerva RL, De Castro JAS. Calcium absorption from apple and orange juice fortified with calcium citrate malate (CCM). J Am Coll Nutr 1996;15:313-6. https://www.ncbi.nlm.nih.gov/pubmed/8935449?dopt=Abstract[↩]
- Office on Women’s Health, the U.S. Department of Health and Human Services. Osteoporosis. https://www.womenshealth.gov/a-z-topics/osteoporosis[↩]
- Prentice RL, Pettinger MB, Jackson RD, et al. Health risks and benefits from calcium and vitamin D supplementation: Women’s Health Initiative clinical trial and cohort study. Osteoporos Int. 2013;24(2):567–580. doi: 10.1007/s00198-012-2224-2. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3557387/[↩]
- Reid IR, Bolland MJ. Calcium supplements: bad for the heart? Heart. 2012;98(12):895–896. doi: 10.1136/heartjnl-2012-301904. https://www.ncbi.nlm.nih.gov/pubmed/22626897[↩]
- Bolland MJ, Grey A, Avenell A, Gamble GD, Reid IR. Calcium supplements with or without vitamin D and risk of cardiovascular events: reanalysis of the Women’s Health Initiative limited access dataset and meta-analysis. BMJ. 2011;19:342. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3079822/[↩]
- Moyer VA, U.S. Preventive Services Task Force Vitamin D and calcium supplementation to prevent fractures in adults: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2013;158(9):691–696. https://www.ncbi.nlm.nih.gov/pubmed/23440163[↩]
- Institute of Medicine. Dietary reference intakes for calcium, phosphorus, magnesium, vitamin D, and fluoride. Washington (DC): National Academy Press; 1997.[↩][↩]
- Cochrane Review 27 April 1998. – Calcium and vitamin D for treating osteoporosis caused by the use of steroids – http://www.cochrane.org/CD000952/MUSKEL_calcium-and-vitamin-d-for-treating-osteoporosis-caused-by-the-use-of-steroids.
Calcium Rich Foods
Milk, yogurt, and cheese are rich natural sources of calcium and are the major food contributors of this nutrient to people in the United States [1]. Nondairy sources include vegetables, such as Chinese cabbage, kale, and broccoli. Spinach provides calcium, but its bioavailability is poor. Most grains do not have high amounts of calcium unless they are fortified; however, they contribute calcium to the diet because they contain small amounts of calcium and people consume them frequently. Foods fortified with calcium include many fruit juices and drinks, tofu, and cereals.
The U.S. Department of Agriculture’s (USDA’s) Nutrient Database website ((The USDA Food Composition Databases. https://ndb.nal.usda.gov/ndb/[↩]
- The USDA Food Composition Databases. Calcium Content. https://ods.od.nih.gov/pubs/usdandb/Calcium-Content.pdf[↩]
- The USDA Food Composition Databases. Foods Calcium Content. https://ods.od.nih.gov/pubs/usdandb/Calcium-Food.pdf[↩]
- U.S. Department of Agriculture, Agricultural Research Service. 2015. USDA National Nutrient Database for Standard Reference, Release 28. Nutrient Data Laboratory Home Page. https://ndb.nal.usda.gov/ndb/[↩]
- Heaney RP, Weaver CM. Calcium and vitamin D. Endocrinol Metab Clin North Am. 2003 Mar;32(1):181–94. vii–viii. Review.[↩]
- Institute of Medicine. Dietary reference intakes for calcium, phosphorus, magnesium, vitamin D, and fluoride. Washington, DC: National Academy Press; 1997. p. 432.[↩]
- Looker AC, Pfeiffer CM, Lacher DA, Schleicher RL, Picciano MF, Yetley EA. Serum 25-hydroxyvitamin D status of the US population: 1988–1994 compared to 2000–2004. Am J Clin Nutr. 2008;88(6):1519–1527. doi: 10.3945/ajcn.2008.26182. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2745830/[↩]
- Wortsman J, Matsuoka LY, Chen TC, Lu Z, Holick MF. Decreased bioavailability of vitamin D in obesity. Am J Clin Nutr. 2000;72(3):690–693. https://www.ncbi.nlm.nih.gov/pubmed/10966885[↩]
- Pettifor JM. Nutritional and drug-induced rickets and osteomalacia. In: Favus MJ, editor. Primer on the metabolic bone diseases and disorders of mineral metabolism. 5. Washington, DC: American Society for Bone and Mineral Research; 2003. pp. 399–407. [↩]
- Institute of Medicine. Dietary reference intakes for calcium, phosphorus, magnesium, vitamin D, and fluoride. Washington, DC: National Academy Press; 1997. p. 432. [↩][↩][↩]
- Institute of Medicine, Food and Nutrition Board. Dietary Reference Intakes for Calcium and Vitamin D. Washington, DC: National Academy Press, 2010[↩][↩][↩][↩][↩]
- U.S. Department of Agriculture, Agricultural Research Service. 2011. USDA National Nutrient Database for Standard Reference, Release 24. Nutrient Data Laboratory Home Page. https://www.ars.usda.gov/northeast-area/beltsville-md/beltsville-human-nutrition-research-center/nutrient-data-laboratory/[↩]
- Ovesen L, Brot C, Jakobsen J. Food contents and biological activity of 25-hydroxyvitamin D: a vitamin D metabolite to be reckoned with? Ann Nutr Metab 2003;47:107-13. https://www.ncbi.nlm.nih.gov/pubmed/12743460?dopt=Abstract[↩]
- Mattila PH, Piironen VI, Uusi-Rauva EJ, Koivistoinen PE. Vitamin D contents in edible mushrooms. J Agric Food Chem 1994;42:2449-53.[↩]
- Calvo MS, Whiting SJ, Barton CN. Vitamin D fortification in the United States and Canada: current status and data needs. Am J Clin Nutr 2004;80:1710S-6S. https://www.ncbi.nlm.nih.gov/pubmed/15585792?dopt=Abstract[↩][↩]
- The USDA Food Composition Databases. https://ndb.nal.usda.gov/ndb/[↩]
- The USDA Food Composition Databases. Vitamin D Content. https://ods.od.nih.gov/pubs/usdandb/VitaminD-Content.pdf[↩]
- The USDA Food Composition Databases. Foods Vitamin D Content. https://ods.od.nih.gov/pubs/usdandb/VitaminD-Food.pdf[↩]
- U.S. Department of Agriculture, Agricultural Research Service. 2011. USDA National Nutrient Database for Standard Reference, Release 24. Nutrient Data Laboratory Home Page, https://www.ars.usda.gov/northeast-area/beltsville-md/beltsville-human-nutrition-research-center/nutrient-data-laboratory/[↩][↩]
- Taylor CL, Patterson KY, Roseland JM, Wise SA, Merkel JM, Pehrsson PR, Yetley EA. Including food 25-hydroxyvitamin D in intake estimates may reduce the discrepancy between dietary and serum measures of vitamin D status. J Nutr 2014;144:654-9. https://www.ncbi.nlm.nih.gov/pubmed/24623845?dopt=Abstract[↩][↩]
- Whiting SJ, Healey A, Psiuk S, Mirwald R, Kowalski K, Bailey DA. Relationship between carbonated and other low nutrient dense beverages and bone mineral content of adolescents. Nutr Res. 2001 Aug;21 (8):1107–15.[↩]
- Fitzpatrick L, Heaney RP. Got soda. J Bone Miner Res. 2003 Sep;18(9):1570–2. https://www.ncbi.nlm.nih.gov/pubmed/12968665[↩]
- Institute of Medicine. Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. Washington, DC: National Academy Press; 2000. p. 800.[↩]
- Booth SL, Tucker KL, Chen H, et al. Dietary vitamin K intakes are associated with hip fracture but not with bone mineral density in elderly men and women. Am J Clin Nutr. 2000; 71:1201–8. https://www.ncbi.nlm.nih.gov/pubmed/10799384?dopt=Citation[↩]
- Booth SL, Broe KE, Gagnon DR, et al. Vitamin K intake and bone mineral density in women and men. Am J Clin Nutr. 2003; 77:512–6. https://www.ncbi.nlm.nih.gov/pubmed/12540415?dopt=Citation[↩]
- Feskanich D, Weber P, Willett WC, Rockett H, Booth SL, Colditz GA. Vitamin K intake and hip fractures in women: a prospective study. Am J Clin Nutr. 1999; 69:74–9. https://www.ncbi.nlm.nih.gov/pubmed/9925126?dopt=Citation[↩]
- Schurgers LJ. Vitamin K: key vitamin in controlling vascular calcification in chronic kidney disease. Kidney Int2013;83:782-4. https://www.ncbi.nlm.nih.gov/pubmed/23633049?dopt=Abstract[↩]
- Gundberg CM, Lian JB, Booth SL. Vitamin K-dependent carboxylation of osteocalcin: friend or foe? Adv Nutr 2012;3:149-57. https://www.ncbi.nlm.nih.gov/pubmed/22516722?dopt=Abstract[↩][↩]
- Suttie JW. Vitamin K. In: Coates PM, Betz JM, Blackman MR, et al., eds. Encyclopedia of Dietary Supplements. 2nd ed. London and New York: Informa Healthcare; 2010:851-60.[↩][↩]
- Yaegashi Y, Onoda T, Tanno K, Kuribayashi T, Sakata K, Orimo H. Association of hip fracture incidence and intake of calcium, magnesium, vitamin D, and vitamin K. Eur J Epidemiol 2008;23:219-25. https://www.ncbi.nlm.nih.gov/pubmed/18214692?dopt=Abstract[↩]
- Rejnmark L, Vestergaard P, Charles P, Hermann AP, Brot C, Eiken P, et al. No effect of vitamin K1 intake on bone mineral density and fracture risk in perimenopausal women. Osteoporos Int 2006;17:1122-32. https://www.ncbi.nlm.nih.gov/pubmed/16683180?dopt=Abstract[↩]
- Feskanich D, Weber P, Willett WC, Rockett H, Booth SL, Colditz GA. Vitamin K intake and hip fractures in women: a prospective study. Am J Clin Nutr 1999;69:74-9. https://www.ncbi.nlm.nih.gov/pubmed/9925126?dopt=Abstract[↩]
- Booth SL, Broe KE, Gagnon DR, Tucker KL, Hannan MT, McLean RR, et al. Vitamin K intake and bone mineral density in women and men. Am J Clin Nutr 2003;77:512-6. https://www.ncbi.nlm.nih.gov/pubmed/12540415?dopt=Abstract[↩]
- Booth SL, Tucker KL, Chen H, Hannan MT, Gagnon DR, Cupples LA, et al. Dietary vitamin K intakes are associated with hip fracture but not with bone mineral density in elderly men and women. Am J Clin Nutr 2000;71:1201-8. https://www.ncbi.nlm.nih.gov/pubmed/10799384?dopt=Abstract[↩]
- Chan R, Leung J, Woo J. No association between dietary vitamin K intake and fracture risk in chinese community-dwelling older men and women: a prospective study. Calcif Tissue Int 2012;90:396-403. https://www.ncbi.nlm.nih.gov/pubmed/22451220?dopt=Abstract[↩]
- Cockayne S, Adamson J, Lanham-New S, Shearer MJ, Gilbody S, Torgerson DJ. Vitamin K and the prevention of fractures: systematic review and meta-analysis of randomized controlled trials. Arch Intern Med 2006;166:1256-61. https://www.ncbi.nlm.nih.gov/pubmed/16801507?dopt=Abstract[↩]
- Knapen MH, Drummen NE, Smit E, Vermeer C, Theuwissen E. Three-year low-dose menaquinone-7 supplementation helps decrease bone loss in healthy postmenopausal women. Osteoporos Int 2013;24:2499-507. https://www.ncbi.nlm.nih.gov/pubmed/23525894?dopt=Abstract[↩]
- Booth SL, Dallal G, Shea MK, Gundberg C, Peterson JW, Dawson-Hughes B. Effect of vitamin K supplementation on bone loss in elderly men and women. J Clin Endocrinol Metab 2008;93:1217-23. https://www.ncbi.nlm.nih.gov/pubmed/18252784?dopt=Abstract[↩]
- Binkley N, Harke J, Krueger D, Engelke J, Vallarta-Ast N, Gemar D, et al. Vitamin K treatment reduces undercarboxylated osteocalcin but does not alter bone turnover, density, or geometry in healthy postmenopausal North American women. J Bone Miner Res 2009;24:983-91. https://www.ncbi.nlm.nih.gov/pubmed/19113922?dopt=Abstract[↩][↩]
- European Food Safety Authority. Scientific opinion on the substantiation of health claims related to vitamin K and maintenance of bone pursuant to Article 13(1) of Regulation (EC) No 1924/2006. The EFSA Journal 2009;7:1228.[↩]
- US Department of Health and Human Services. Physical activity and health: A report of the Surgeon General. Atlanta (GA): U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, President’s Council on Physical Fitness and Sports; 1996. p. 300.[↩]
- Rubin C, Recker R, Cullen D, Ryaby J, McCabe J, Mcleod K. Prevention of postmenopausal bone loss by a low-magnitude, high-frequency mechanical stimuli: A clinical trial assessing compliance, efficacy, and safety. J Bone Miner Res. 2004 Mar;19(3):343–51. https://www.ncbi.nlm.nih.gov/pubmed/15040821[↩]
- Verschueren SM, Roelants M, Delecluse C, Swinnen S, Vanderschueren D, Boonen S. Effect of 6-month whole body vibration training on hip density, muscle strength, and postural control in postmenopausal women: A randomized controlled pilot study. J Bone Miner Res. 2004 Mar;19(3):352–9. https://www.ncbi.nlm.nih.gov/pubmed/15040822[↩]
- Ward K, Alsop C, Caulton J, Rubin C, Adams J, Mughal Z. Low magnitude mechanical loading is osteogenic in children with disabling conditions. J Bone Miner Res. 2004 Mar;19(3):360–9. https://www.ncbi.nlm.nih.gov/pubmed/15040823[↩]
- Kraemer et al. 2002, ACSM 1998a, ACSM 1998b[↩]
- Cochrane Review 6 July 2011. – Exercise for preventing and treating osteoporosis in postmenopausal women – http://www.cochrane.org/CD000333/MUSKEL_exercise-for-preventing-and-treating-osteoporosis-in-postmenopausal-women[↩]
- Schurch MA, Rizzoli R, Slosman D, Vadas L, Vergnaud P, Bonjour JP. Protein supplements increase serum insulin-like growth factor-I levels and attenuate proximal femur bone loss in patients with recent hip fracture. A randomized, double-blind, placebo-controlled trial. Ann Intern Med. 1998 May 15;128(10):801–9.[↩]
- 2015-2020 Dietary Guidelines for Americans 8th Edition. https://www.cnpp.usda.gov/dietary-guidelines[↩]
- The National Institutes of Health, Osteoporosis and Related Bone Diseases, National Resource Center. – Osteoporosis Basics – https://www.niams.nih.gov/Health_Info/Bone/Osteoporosis/osteoporosis_hoh.asp[↩]
- National Center for Biotechnology Information, U.S. National Library of Medicine. Bone Mineral Density (BMD). – https://www.ncbi.nlm.nih.gov/pubmedhealth/PMHT0024682/[↩]
- Baim S, Binkley N, Bilezikian JP, Kendler DL, et al. Official positions of the International Society for Clinical Densitometry and executive summary of the 2007 ISCD Position Development Conference. J Clin Densitom 11:75–91. 2008.[↩]
- National Center for Biotechnology Information, U.S. National Library of Medicine. – The Osteoporosis Pyramid for Prevention and Treatment – https://www.ncbi.nlm.nih.gov/books/NBK45501/figure/ch9.f1/?report=objectonly[↩]
- Cochrane Review 5 October 2016. – Bisphosphonates for treating osteoporosis caused by the use of steroids – http://www.cochrane.org/CD001347/MUSKEL_bisphosphonates-treating-osteoporosis-caused-use-steroids[↩]
- Edwards BJ, Hellstein JW, Jacobsen PL, et al: Updated recommendations for managing the care of patients receiving oral bisphosphonate therapy: An advisory statement from the American Dental Association Council on Scientific Affairs. J Am Dent Assoc 139:1674–1677, 2008. https://www.ncbi.nlm.nih.gov/pubmed/19047674[↩][↩][↩]
- Cochrane Review 17 October 2007. – Bisphosphonate therapy for children and adolescents with secondary osteoporosis. – http://www.cochrane.org/CD005324/MUSKEL_bisphosphonate-therapy-for-children-and-adolescents-with-secondary-osteoporosis.[↩]
- Cochrane Review 24 January 2000. – Calcitonin for preventing and treating corticosteroid-induced osteoporosis – http://www.cochrane.org/CD001983/MUSKEL_calcitonin-for-preventing-and-treating-corticosteroid-induced-osteoporosis[↩]
- Boonen S, Ferrari S, Miller PD. Postmenopausal osteoporosis treatment with antiresorptives: effects of discontinuation or long-term continuation on bone turnover and fracture risk—a perspective. J Bone Miner Res. 2012;27(5):963–974. doi: 10.1002/jbmr.1570. https://www.ncbi.nlm.nih.gov/pubmed/22467094[↩]
- Black DM, Bauer DC, Schwartz AV, Cummings SR, Rosen CJ. Continuing bisphosphonate treatment for osteoporosis—for whom and for how long? N Engl J Med. 2012;366(22):2051–2053. doi: 10.1056/NEJMp1202623. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4015798/[↩]
- Cosman F, Nieves J, Woelfert L, Formica C, Gordon S, Shen V, Lindsay R. Parathyroid hormone added to established hormone therapy: Effects on vertebral fracture and maintenance of bone mass after parathyroid hormone withdrawal. J Bone Miner Res. 2001 May;16(5):925–31. https://www.ncbi.nlm.nih.gov/pubmed/11341338[↩]
- Black DM, Greenspan SL, Ensrud KE, Palermo L, McGowan JA, Lang TF, Garnero P, Bouxsein ML, Bilezikian JP, Rosen CJ. PaTH Study Investigators. The effects of parathyroid hormone and alendronate alone or in combination in postmenopausal osteoporosis. N Engl J Med. 2003 Sep 25;349(13):1207–15. https://www.ncbi.nlm.nih.gov/pubmed/14500804[↩]
- Finkelstein JS, Hayes A, Hunzelman JL, Wyland JJ, Lee H, Neer RM. The effects of parathyroid hormone, alendronate, or both in men with osteoporosis. N Engl J Med. 2003 Sep 25;349(13):1216–26. https://www.ncbi.nlm.nih.gov/pubmed/14500805[↩]
- Cochrane Review 6 March 2014. – Chinese herbal medicines for osteoporosis -http://www.cochrane.org/CD005467/MUSKEL_chinese-herbal-medicines-for-osteoporosis[↩]
- Cochrane Review 23 October 2000. – Fluoride can increase bone mineral density at the lumbar spine, it does not reduce vertebral fractures. – http://www.cochrane.org/CD002825/MUSKEL_fluoride-can-increase-bone-mineral-density-at-the-lumbar-spine-it-does-not-reduce-vertebral-fractures.[↩]
- Zuckerman JD. Hip fracture. N Engl J Med. 1996 Jun 6;334(23):1519–25.[↩]
- Johnell O, Scheek WH, Lu Y, Reginster JY, Need AG, Seeman E. Additive effects of raloxifene and alendronate on bone density and biochemical markers of bone remodeling in postmenopausal women with osteoporosis. J Clin Endocrinol Metab. 2002 Mar;87(3):985–92.[↩]
- Evans A, Jensen M, Kip K, DeNardo AJ, Lawler GJ, Negin GA, Remley KB, Boutin SM, Dunnagan SA. Vertebral compression fractures: Pain reduction and improvement in functional mobility after percutaneous polymethylmethacrylate vertebroplasty—retrospective report of 245 cases. Radiology. 2003 Feb;226(2):366–72.[↩][↩]
- Lieberman IH, Dudeney S, Reinhardt MK, Bell G. Initial outcome and efficacy of “kyphoplasty” in the treatment of painful osteoporotic vertebral compression fractures. Spine. 2001 Jul 15;26(14):1631–8.[↩][↩]
- Sinaki M, Itoi E, Wahner HW, Wollan P, Gelzcer R, Mullan BP, Collins DA, Hodgson SF. Stronger back muscles reduce the incidence of vertebral fractures: a prospective 10-year follow-up of postmenopausal women. Bone. 2002 Jun;30(6):836–41. https://www.ncbi.nlm.nih.gov/pubmed/12052450[↩]
- Bonner FJ Jr, Sinaki M, Grabois M, Shipp KM, Lane JM, Lindsay R, Gold DT, Cosman F, Bouxsein ML, Weinstein JN, et al. Health professional’s guide to rehabilitation of the patient with osteoporosis. Osteoporos Int. 2003;14(Suppl 2):S1–22. https://www.ncbi.nlm.nih.gov/pubmed/12759719[↩]