Contents
- Azathioprine
- Azathioprine special precautions
- How does azathioprine work?
- Azathioprine uses
- Azathioprine dosage
- Adult dose for kidney transplant
- Adult dose for rheumatoid arthritis
- Adult dose for Acute Crohn’s disease
- Adult dose for Maintenance Crohn’s disease
- Adult dose for Ulcerative Colitis
- Adult dose for Chronic Inflammatory Demyelinating Polyradiculoneuropathy
- Adult dose for Atopic Dermatitis
- Adult dose for Sarcoidosis
- Adult dose for Uveitis
- Adult dose for Multiple Sclerosis
- Adult dose for Systemic Lupus Erythematosus (SLE)
- Adult dose for Chronic Active Hepatitis
- Adult dose for Takayasu’s Arteritis
- Children dose for Atopic Dermatitis
- Children dose for Organ Transplant (Rejection Prevention)
- Children dose for Eczema
- Children dose for Systemic Lupus Erythematosus (SLE)
- What should I do if I forget a dose?
- Azathioprine side effects
- Azathioprine overdose
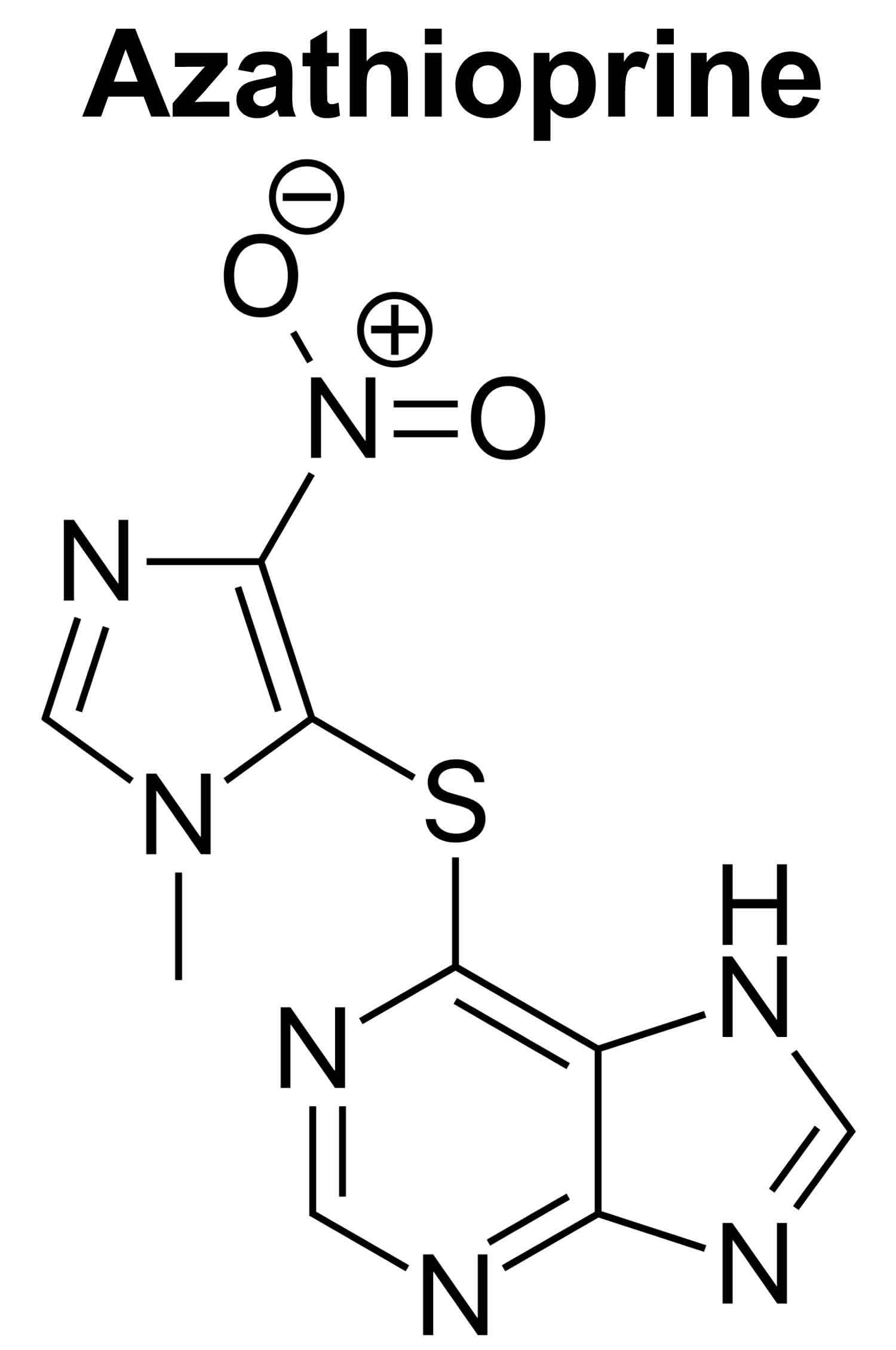
Azathioprine
Azathioprine also known as Imuran or Azasan is an immunosuppressant or a drug that weakens your body’s immune system in order to help keep it from “rejecting” a transplanted organ such as a kidney 1. Organ rejection happens when your immune system treats the new organ as an invader and attacks it. Azathioprine is used with other medications to prevent transplant rejection (attack of the transplanted organ by your immune system) in people who received kidney transplants. Azathioprine is also used as a disease-modifying antirheumatic drug (DMARD) or immunomodulator to treat autoimmune conditions such as severe rheumatoid arthritis (a condition in which the body attacks its own joints, causing pain, swelling, and loss of function), Crohn’s disease, ulcerative colitis and autoimmune hepatitis when other medications and treatments have not helped 1, 2.
Azathioprine was introduced into use in the early 1960s and represented a major breakthrough in the development of organ rejection therapy, allowing for more widespread and successful use of kidney, heart and liver transplantation. Azathioprine was approved for use in the United States in 1968 and is still widely used in organ transplantation and in treatment of autoimmune conditions.
Azathioprine comes as a tablet 50, 75 and 100 mg to be taken by mouth; a parenteral formulation is also available. Azathioprine is usually taken once or twice a day after meals. The usual Azathioprine dose is 1 to 3 mg/kg of body weight or 50 to 150 mg daily and it is typically given long term. Take azathioprine at around the same time(s) every day. Follow the directions on your prescription label carefully, and ask your doctor or pharmacist to explain any part you do not understand. Take azathioprine exactly as directed. Do not take more or less of it or take it more often than prescribed by your doctor.
If you are taking azathioprine to treat rheumatoid arthritis, your doctor may start you on a low dose and gradually increase your dose after 6-8 weeks and then not more than once every 4 weeks. Your doctor may gradually decrease your dose when your condition is controlled. If you are taking azathioprine to prevent kidney transplant rejection, your doctor may start you on a high dose and decrease your dose gradually as your body adjusts to the transplant.
Azathioprine controls rheumatoid arthritis but does not cure it. It may take up to 12 weeks before you feel the full benefit of azathioprine. Azathioprine prevents transplant rejection only as long as you are taking the medication. Continue to take azathioprine even if you feel well. Do not stop taking azathioprine without talking to your doctor.
Azathioprine usually requires 6 to 8 weeks to work. The recommendation is to consider stopping Azathioprine if there is no improvement in 3 months 3. Checking thiopurine S-methyltransferase (TPMT) activity is suggested before starting azathioprine. Thiopurine S-methyltransferase (TPMT) deficiency is a condition characterized by significantly reduced activity of thiopurine S-methyltransferase (TPMT) enzyme that helps the body process drugs called thiopurines, which include 6-thioguanine, 6-mercaptopurine, and azathioprine 4. A potential complication of treatment with thiopurine drugs is damage to the bone marrow (hematopoietic toxicity). Although this complication can occur in anyone who takes these drugs, people with thiopurine S-methyltransferase (TPMT) deficiency are at highest risk. Bone marrow normally makes several types of blood cells, including red blood cells, which carry oxygen; white blood cells, which help protect the body from infection; and platelets, which are involved in blood clotting. Damage to the bone marrow results in myelosuppression, a condition in which the bone marrow is unable to make enough of these cells. A shortage of red blood cells (anemia) can cause pale skin (pallor), weakness, shortness of breath, and extreme tiredness (fatigue). Low numbers of white blood cells (neutropenia) can lead to frequent and potentially life-threatening infections. A shortage of platelets (thrombocytopenia) can cause easy bruising and bleeding.
Many doctors recommend that patients’ TPMT activity levels be tested before Azathioprine is prescribed. In people who are found to have reduced TPMT enzyme activity, thiopurine drugs may be given at a significantly lower dose or different medications can be used to reduce the risk of hematopoietic toxicity. Misclassification of TMPT phenotype can occur by prior blood transfusion 5.
TPMT deficiency does not appear to cause any health problems other than those associated with thiopurine drug treatment 4.
Azathioprine common side effects include nausea, abdominal upset, rash, aphthous ulcers and dose related bone marrow suppression. Long term use of azathioprine is associated with an increased risk of certain types of cancer, especially skin cancer and lymphoma and possibly liver carcinoma.
Azathioprine may increase your risk of developing certain types of cancer, especially skin cancer and lymphoma (cancer that begins in the cells that fight infection). If you have had a kidney transplant, there may be a higher risk that you will develop cancer even if you do not take azathioprine. Tell your doctor if you have or have ever had cancer and if you are taking or have ever taken alkylating agents such as chlorambucil (Leukeran), cyclophosphamide (Cytoxan), or melphalan (Alkeran) for cancer. To decrease the risk that you will develop skin cancer, avoid prolonged or unnecessary exposure to sunlight and wear protective clothing, sunglasses, and sunscreen. Tell your doctor immediately if you notice any changes in your skin or any lumps or masses anywhere in your body.
Some teenage and young adult males who took azathioprine alone or with another medication called a tumor necrosis factor (TNF) blocker to treat Crohn’s disease (a condition in which the body attacks the lining of the digestive tract causing pain, diarrhea, weight loss, and fever) or ulcerative colitis (a condition which causes swelling and sores in the lining of the colon [large intestine] and rectum) developed hepatosplenic T-cell lymphoma (HSTCL). HSTCL is a very serious type of cancer that often causes death within a short period of time. Azathioprine has not been approved by the Food and Drug Administration (FDA) for the treatment of Crohn’s disease or ulcerative colitis, but doctors may sometimes prescribe azathioprine to treat these conditions. If you develop any of these symptoms during your treatment, call your doctor immediately: stomach pain; fever; unexplained weight loss; night sweats or easy bruising or bleeding.
Azathioprine can cause a decrease in the number of blood cells in your bone marrow, which may cause serious or life-threatening infections. The risk that the number of blood cells that you have will decrease is highest if you have a genetic (inherited) risk factor. Your doctor may order a test before or during your treatment to see if you have this risk factor. Taking certain medications may also increase the risk that your blood cells will decrease, so tell your doctor if you are taking any of the following: angiotensin converting enzyme (ACE) inhibitors such as benazepril (Lotensin), captopril, enalapril (Vasotec), fosinopril, lisinopril (Prinivil, Zestril), moexipril (Univasc), perindopril (Aceon), quinapril (Accupril), Ramipril (Altace), or trandolapril (Mavik); trimethoprim and sulfamethoxazole (Bactrim, Septra); and ribavirin (Copegus, Rebetol, Virazole). If you experience any of the following symptoms, call your doctor immediately: unusual bleeding or bruising; excessive tiredness; pale skin; headache; confusion; dizziness; fast heartbeat; difficulty sleeping; weakness; shortness of breath; and sore throat, fever, chills, and other signs of infection. Your doctor will order tests before, during, and after your treatment to see if your blood cells are affected by this medication.
Talk to your doctor about the possible risks of using azathioprine for your condition.
Azathioprine special precautions
Before taking azathioprine:
- tell your doctor and pharmacist if you are allergic to azathioprine, any other medications, or any of the ingredients in azathioprine tablets. Ask your doctor or pharmacist for a list of the ingredients.
- tell your doctor and pharmacist what prescription and nonprescription medications, vitamins, nutritional supplements, and herbal products you are taking or plan to take. Be sure to mention the medications mentioned in the IMPORTANT WARNING section and any of the following: allopurinol (Zyloprim); aminosalicylates such as mesalamine (Apriso, Asacol, Pentasa, others), olsalazine (Dipentum), and sulfasalazine (Azulfidine); and anticoagulants (‘blood thinners’) such as warfarin (Coumadin). Your doctor may need to change the doses of your medications or monitor you carefully for side effects.
- tell your doctor if you have any type of infection, or if you have or have ever had kidney disease.
- tell your doctor if you are pregnant, plan to become pregnant, or are breast-feeding. You should use birth control to be sure you or your partner will not become pregnant while you are taking azathioprine. Call your doctor if you or your partner become pregnant while you are taking azathioprine. Azathioprine may harm the fetus.
- if you are having surgery, including dental surgery, tell the doctor or dentist that you are taking azathioprine.
- do not have any vaccinations during or after your treatment without talking to your doctor.
Test patient for hepatitis B and C and Tuberculin Skin Test (Mantoux Test) for diagnosis of silent (latent) tuberculosis (TB) infection. A pregnancy test before azathioprine treatment initiation is also a recommendation 6.
Azathioprine contraindications
Azathioprine contraindications include 7, 8, 9, 3, 6:
- Hypersensitivity
- Pregnancy or plan for pregnancy: Contraception recommended. azathioprine can increase the risk of spontaneous miscarriage, low birth weight, and preterm delivery. Although data in systemic lupus erythematosus (SLE) and renal transplant patients showed safety in pregnancy. In some specific conditions like SLE and antiphospholipid antibody syndrome, the benefits of taking immunosuppressive medications are more than harms in keeping the mother safe.
- Breastfeeding as mercaptopurine (6-MP) was present in breast-milk of women who take azathioprine
- Unknown thiopurine S-methyltransferase (TPMT) status or deficient thiopurine S-methyltransferase (TPMT) activity due to the high risk of myelosuppression 4
- Known malignancy
- Clinically active infection
Azathioprine relative contraindications 6:
- Allopurinol intake concomitantly with azathioprine due to severe myelosuppression
- Cyclophosphamide or chlorambucil treatment in the past.
Azathioprine drug interactions
Tell your doctor about all your current medicines. Many drugs can affect azathioprine, especially:
- Allopurinol
- Febuxostat
- Ribavirin.
This list is not complete and many other drugs may affect azathioprine. This includes prescription and over-the-counter medicines, vitamins, and herbal products.
How does azathioprine work?
Azathioprine is a purine analog and prodrug (a pharmacologically inactive medication that metabolizes inside the body and converts into a pharmacologically active drug compound) that converts to its active metabolites, mercaptopurine (6-MP) and thioguanine (6-TGN), by the action of hypoxanthine-guanine phosphoribosyltransferase (HPRT) and thiopurine methyltransferase (TPMT) enzymes 1. Azathioprine then blocks purine synthesis thus inhibiting DNA, RNA and subsequent protein synthesis 10. Azathioprine metabolites are incorporated into the replicating DNA and halt cell division. Azathioprine metabolites inhibit the maturation of T cells (T lymphocytes) and blocks delayed hypersensitivity reactions; it also has antiinflammatory activity.
Azathioprine works by decreasing the activity of the body’s immune system so it will not attack the transplanted organ or the joints.
Azathioprine is absorbed rapidly through the gastrointestinal system and does not penetrate the blood-brain barrier. Azathioprine undergoes metabolism in the liver, and excretion is via the kidneys, which increases its toxicity in kidney failure 3.
Azathioprine uses
Azathioprine (Imuran or Azasan) is approved by the U.S. Food and Drug Administration (FDA) for the symptomatic treatment of active rheumatoid arthritis as a corticosteroid sparing agent. Azathioprine also has approval as add-on therapy for the prevention of kidney transplant rejection 11, 3.
Azathioprine (Imuran or Azasan) is also used off-label for the treatment of inflammatory bowel disease (Crohn’s disease & ulcerative colitis) 12, Churg-Strauss syndrome, autoimmune hepatitis (for maintenance treatment along with steroids) 13, 14, chronic immune thrombocytopenic purpura (rare autoimmune disorder that causes you to have low platelet levels) as second-line agent) 15, lupus glomerulonephritis 11, fibrotic connective tissue disease-associated interstitial lung disease 16, multiple sclerosis, severe myasthenia gravis, recurrent pericarditis 17, psoriasis, non-infectious uveitis 18, relapsing polychondritis (a very rare autoimmune disease characterized by a relapsing inflammation of the cartilaginous tissues such as joints, ears, nose, intervertebral discs, larynx, trachea and cartilaginous bronchi) 19, dermatomyositis/polymyositis, erythema multiforme, severe and refractory atopic dermatitis, chronic actinic dermatitis, pyoderma gangrenosum, Behcet disease, cutaneous vasculitis, pityriasis rubra pilaris, lichen planus, bullous pemphigoid, and pemphigus vulgaris 3, 20. Azathioprine or mercaptopurine (6-MP) are treatment options in children with Crohn’s disease as a maintenance treatment 5. Azathioprine is also used to treat ulcerative colitis (a condition which causes swelling and sores in the lining of the colon [large intestine] and rectum) and Crohn’s disease.
Azathioprine dosage
The starting dose for azathioprine is 2 to 2.5 mg/kg of body weight per day, except for patients with TPMT or NUDT15 gene mutation, in which the starting dose is lower than normal 21. Azathioprine dose adjustments are necessary for liver and kidney disease 20.
Azathioprine tablets may be administered after meals to decrease gastrointestinal adverse effects. Administration can be by intravenous (IV) push over 5 minutes, at a concentration not exceeding 5 mg/ml. Azathioprine can be further diluted with normal saline or dextrose 5% in water and administered by intermittent infusion over 30 to 60 minutes. However, it may also be infused over 5 minutes up to over 8 hours.
Adult dose for kidney transplant
Use: Adjunctive therapy for prevention of rejection in renal homotransplantation.
- Initial dose: Azathioprine 3 to 5 mg/kg orally or IV once a day, beginning at the time of transplant
- Maintenance dose: Azathioprine 1 to 3 mg/kg orally or IV once a day
Comments:
- In a minority of cases, Azathioprine therapy has been started 1 to 3 days before transplantation
- Azathioprine dose should not be increased to toxic levels because of threatened rejection
Adult dose for rheumatoid arthritis
- Initial dose: 1 mg/kg (50 to 100 mg) orally or IV per day given in 1 to 2 divided doses
- Maintenance dose: Lowest effective dose
- Maximum dose: 2.5 mg/kg orally or IV per day
- Duration: At least 12 weeks
Comments:
- Azathioprine dose may be increased by 0.5 mg/kg/day (or approximately 25 mg/day), after 6 to 8 weeks of starting treatment and thereafter at 4 week intervals if necessary.
- Gradual dose reduction is recommended to reduce the risk of toxicities.
- Therapeutic response occurs after several weeks of therapy, usually 6 to 8 weeks. Patients not improved after 12 weeks can be considered refractory.
- Azathioprine may be continued long-term in patients with clinical response.
Adult dose for Acute Crohn’s disease
- Azathioprine dose 1.5 to 4 mg/kg per day for 10 days up to 52 weeks
Adult dose for Maintenance Crohn’s disease
- Azathioprine dose 1.5 to 4 mg/kg per day for 10 days up to 52 weeks
Adult dose for Ulcerative Colitis
Study (n=9)
- IV: Azathioprine dose 20 to 40 mg/kg via IV infusion over 36 hours or 40 mg/kg as three 8-hour infusions over 3 days followed by oral azathioprine
- Oral: Azathioprine dose 2 mg/kg orally per day beginning the day after completion of the IV loading dose
Study (n=12)
- Azathioprine dose 50 mg per day for 2 weeks, then 2 to 2.5 mg/kg per day plus mesalamine 500 mg orally 3 times a day; these drugs were started immediately after signs of remission was achieved (mean: 14.5 days) with cyclosporine IV (4 mg/kg/day)
Adult dose for Chronic Inflammatory Demyelinating Polyradiculoneuropathy
- Study (n=14): Azathioprine dose 2 to 3 mg/kg orally once a day for 9 months
Adult dose for Atopic Dermatitis
- Study (n=37): Azathioprine dose 2.5 mg/kg orally once a day, in the morning, for 3 months
Adult dose for Sarcoidosis
Study (n=11)
- Initial dose: Azathioprine dose 2 mg/kg per day in combination with prednisolone 0.6 to 0.8 mg/kg per day, with prednisolone reduced to 0.1 mg/kg within 2 to 3 months
- Maintenance dose: Azathioprine dose 2 mg/kg per day with prednisolone 0.1 mg/kg per day for 21 to 22 months
Adult dose for Uveitis
Study (n=14)
- Treatment of choroidal neovascularization: Azathioprine dose 1 to 1.5 mg/kg orally per day, in combination with prednisolone and cyclosporine
Adult dose for Multiple Sclerosis
Study (n=6)
Patients refractory to interferon beta-1b:
- Initial dose: Azathioprine should be titrated up to 1.5 mg/kg per day over 1 month, followed by 50 mg increments in 6-month intervals, concomitantly with 8 million international units subcutaneous interferon beta-1b on alternate days
- Maintenance dose: Azathioprine dose 2 mg/kg per day
Adult dose for Systemic Lupus Erythematosus (SLE)
Studies: Azathioprine dose 1 to 3 mg/kg actual body weight/day orally or IV once a day
Study (n=55)
- Diffuse proliferative lupus glomerulonephritis: Sequential therapy starting with prednisone (1 mg/kg/day) for 8 to 10 weeks, gradually tapering to maintenance dosage of 5 to 10 mg/day, together with oral cyclophosphamide (1 to 2 mg/kg/day) for 6 to 9 months followed by azathioprine 50 to 100 mg/day
Adult dose for Chronic Active Hepatitis
Study (n=72)
- Autoimmune hepatitis: Azathioprine dose 1 to 2 mg/kg per day, concomitantly with prednisolone (5 to 15 mg/day) for a minimum of 1 year (average 5 years)
Adult dose for Takayasu’s Arteritis
Study (n=15): Azathioprine dose2 mg/kg actual body weight/day for 1 year in combination with prednisolone taper
Children dose for Atopic Dermatitis
Study (n=37)
- Greater than 17 years: Azathioprine dose 2.5 mg/kg orally once a day, in the morning, for 3 months
Children dose for Organ Transplant (Rejection Prevention)
- Initial dose: Azathioprine dose 3 to 5 mg/kg orally or IV once a day, beginning at the time of transplant
- Maintenance dose: Azathioprine dose 1 to 3 mg/kg orally or IV once a day
Children dose for Eczema
Study (n=91)
- Greater than 6 years: Azathioprine dose 2.5 to 3.5 mg/kg per day in patients with normal levels of thiopurine methyltransferase
Children dose for Systemic Lupus Erythematosus (SLE)
Case Study (n=67)
Lupus Nephritis:
- Greater than 5 years: Azathioprine dose 2 to 3 mg/kg per day (maximum dose: 150 mg/day)
- Dose should be titrated to maintain a total white blood cell count between 3 and 4 x 10(3) cells/mL.
What should I do if I forget a dose?
Take the missed dose as soon as you remember it. However, if it is almost time for the next dose, skip the missed dose and continue your regular dosing schedule. Do not take a double dose to make up for a missed one.
Azathioprine side effects
Azathioprine may cause side effects. Tell your doctor if any of these symptoms are severe or do not go away.
Azathioprine common side effects include the following 22, 23, 10, 13, 24, 25, 26, 27, 28, 29, 30, 31:
- Nausea; is the most frequent side effect
- Dose-dependent.
- Early-onset nausea usually resolves without dose alteration
- Fever
- Fatigue
- Joint aches and pain (arthralgias) and muscle aches & pain (myalgia)
- Bone marrow suppression causing pancytopenia, thrombocytopenia, leukopenia – there are reports of dose-dependent, life-threatening cases.
- This complication correlates with the thioguanine (6-TGN) level
- There is a higher risk of myelosuppression in patients who take allopurinol or angiotensin-converting enzyme inhibitors (ACEIs) and in kidney disease
- An increase in the mean corpuscular volume of the red blood cells is also an expected side effect
- Rash
- Liver injury: Liver injury correlates with a 6-mercaptopurine (6-MP) level of more than 5700 pmol/8 x 108 red blood cell.
- Liver injury categorizes into two groups.
- Acute idiosyncratic liver injury happens in the early course and resolves with stopping the medication.
- Nodular regenerative hyperplasia occurs in inflammatory bowel disease and organ transplant patients several years after therapy.
- Liver injury categorizes into two groups.
- Infections (7.4%): Concomitant use of azathioprine and steroids will increase the risk of Pneumocystis pneumonia (PCP, a serious infection caused by the fungus Pneumocystis jirovecii) in leukopenic patients.
- Hypersensitivity: symptoms including fever, chills, arthralgia/myalgia, liver abnormalities, erythema nodosum
- Kidney damage
Some side effects can be serious. If you experience any of the following symptoms or those listed in the IMPORTANT WARNING section, see your doctor immediately.
- rash
- fever
- weakness
- muscle pain
Complete blood count (CBC) and liver function test (LFT) monitoring weekly are recommended initially for the first 4 to 8 weeks. Complete blood count (CBC) and liver function test (LFT) should get checked every three months for the rest of the treatment once the maintenance dose is achieved. However, it is advisable to check CBC and LFT more frequently in patients with kidney or liver diseases or elderly patients on high dosages of azathioprine or low TPMT activity. If labs show leukopenia (WBC less than 3 x 109/L), thrombocytopenia (platelet less than 120 x 109/L), or transaminitis (liver biochemistry more than half of the normal upper limit), the medication should be stopped 28.
If patients have abdominal pain or severe nausea/vomiting, serum amylase is required to rule out pancreatitis. Lymph node and skin examination should be performed biannually 6. If a generalized wart occurs, the azathioprine dose should be reduced or switched to another agent.
Some studies suggested monitoring the level of azathioprine metabolites (e.g., 6-TGN and 6-MP) to avoid specific complications 13.
Azathioprine rare side effects include 32, 33, 34, 28, 35, 22, 20, 31:
- Diarrhea
- Carcer: cutaneous hyperkeratosis and nonmelanoma skin cancer (squamous cell carcinoma [SCC]) in myasthenia gravis (most likely due to increased risk of photosensitivity), solid-organ transplant and IBD patients/ lymphoma in transplant and inflammatory bowel disease (IBD) patients
- Pancreatitis (3.3%): more in females with Crohn disease
- Dose-dependent
- Usually happens in the first six weeks
- In the case of pancreatitis, discontinue azathioprine
- Alopecia including telogen effluvium, anagen effluvium, and plica neuropathica
- Macrocytic anemia
- Sweet syndrome (acute febrile neutrophilic dermatosis)
- Pneumonitis: in inflammatory bowel disease (IBD) and kidney transplant patients
- Upper airway edema
- Tremor: in organ transplant and Crohn patient: dose-dependent
In cases of hepatic sinusoidal obstruction syndrome, azathioprine must be discontinued permanently. If severely leukopenic, thrombocytopenic, or infected, azathioprine treatment should stop.
Azathioprine may cause other side effects. Call your doctor if you have any unusual problems while you are taking azathioprine.
Azathioprine overdose
Azathioprine overdose symptoms may include the following:
- nausea
- vomiting
- diarrhea
- sore throat, fever, chills, and other signs of infection
Azathioprine overdose symptoms include gastrointestinal symptoms, bradycardia, hepatotoxicity and bone marrow suppression (myelosuppression) 36. Azathioprine overdose usually happens when more than 1.5 times of daily dose is taken by the patient.
In the acute azathioprine overdose setting, activated charcoal may help with decreasing the symptoms within two hours of ingestion 37. No specific antidote is known for azathioprine. In severe cases of azathioprine overdose, dialysis is permissible as azathioprine is dialysable.
- Mohammadi O, Kassim TA. Azathioprine. [Updated 2023 May 1]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK542190[↩][↩][↩]
- LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Azathioprine. [Updated 2017 Aug 21]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK548332[↩]
- Anstey AV, Wakelin S, Reynolds NJ; British Association of Dermatologists Therapy, Guidelines and Audit Subcommittee. Guidelines for prescribing azathioprine in dermatology. Br J Dermatol. 2004 Dec;151(6):1123-32. doi: 10.1111/j.1365-2133.2004.06323.x[↩][↩][↩][↩][↩]
- Thiopurine S-methyltransferase deficiency. https://medlineplus.gov/genetics/condition/thiopurine-s-methyltransferase-deficiency[↩][↩][↩]
- Ruemmele FM, Veres G, Kolho KL, Griffiths A, Levine A, Escher JC, Amil Dias J, Barabino A, Braegger CP, Bronsky J, Buderus S, Martín-de-Carpi J, De Ridder L, Fagerberg UL, Hugot JP, Kierkus J, Kolacek S, Koletzko S, Lionetti P, Miele E, Navas López VM, Paerregaard A, Russell RK, Serban DE, Shaoul R, Van Rheenen P, Veereman G, Weiss B, Wilson D, Dignass A, Eliakim A, Winter H, Turner D; European Crohn’s and Colitis Organisation; European Society of Pediatric Gastroenterology, Hepatology and Nutrition. Consensus guidelines of ECCO/ESPGHAN on the medical management of pediatric Crohn’s disease. J Crohns Colitis. 2014 Oct;8(10):1179-207. doi: 10.1016/j.crohns.2014.04.005[↩][↩]
- Menter A, Korman NJ, Elmets CA, Feldman SR, Gelfand JM, Gordon KB, Gottlieb AB, Koo JY, Lebwohl M, Lim HW, Van Voorhees AS, Beutner KR, Bhushan R. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 4. Guidelines of care for the management and treatment of psoriasis with traditional systemic agents. J Am Acad Dermatol. 2009 Sep;61(3):451-85. doi: 10.1016/j.jaad.2009.03.027[↩][↩][↩][↩]
- van der Woude CJ, Ardizzone S, Bengtson MB, Fiorino G, Fraser G, Katsanos K, Kolacek S, Juillerat P, Mulders AG, Pedersen N, Selinger C, Sebastian S, Sturm A, Zelinkova Z, Magro F; European Crohn’s and Colitis Organization. The second European evidenced-based consensus on reproduction and pregnancy in inflammatory bowel disease. J Crohns Colitis. 2015 Feb;9(2):107-24. doi: 10.1093/ecco-jcc/jju006[↩]
- Provan D, Stasi R, Newland AC, Blanchette VS, Bolton-Maggs P, Bussel JB, Chong BH, Cines DB, Gernsheimer TB, Godeau B, Grainger J, Greer I, Hunt BJ, Imbach PA, Lyons G, McMillan R, Rodeghiero F, Sanz MA, Tarantino M, Watson S, Young J, Kuter DJ. International consensus report on the investigation and management of primary immune thrombocytopenia. Blood. 2010 Jan 14;115(2):168-86. doi: 10.1182/blood-2009-06-225565[↩]
- Petri M. Immunosuppressive drug use in pregnancy. Autoimmunity. 2003 Feb;36(1):51-6. doi: 10.1080/0891693031000067296[↩]
- Wee JS, Marinaki A, Smith CH. Life threatening myelotoxicity secondary to azathioprine in a patient with atopic eczema and normal thiopurine methyltransferase activity. BMJ. 2011 Mar 25;342:d1417. doi: 10.1136/bmj.d1417[↩][↩]
- Ladrière M. Indications actuelles de l’azathioprine en néphrologie [Current indications of azathioprine in nephrology]. Nephrol Ther. 2013 Feb;9(1):8-12. French. doi: 10.1016/j.nephro.2012.08.002[↩][↩]
- Schmidt C, Herrlinger K, Siegmund B, Bokemeyer B, Schreiber S, Stallmach A; Kompetenznetz Darmerkrankungen. Azathioprin in der Therapie des Morbus Crohn–eine Standortbestimmung vor dem Hintergrund aktueller Studien [Azathioprine in Crohn’s disease therapy–guidance against the background of recent studies]. Z Gastroenterol. 2014 Dec;52(12):1423-30. German. doi: 10.1055/s-0034-1385650[↩]
- Sheiko MA, Sundaram SS, Capocelli KE, Pan Z, McCoy AM, Mack CL. Outcomes in Pediatric Autoimmune Hepatitis and Significance of Azathioprine Metabolites. J Pediatr Gastroenterol Nutr. 2017 Jul;65(1):80-85. doi: 10.1097/MPG.0000000000001563[↩][↩][↩]
- Aljumah AA, Al Jarallah B, Albenmousa A, Al Khathlan A, Al Zanbagi A, Al Quaiz M, Al-Judaibi B, Nabrawi K, Al Hamoudi W, Alghamdi M, Fallatah H. The Saudi association for the study of liver diseases and transplantation clinical practice guidelines for management of autoimmune hepatitis. Saudi J Gastroenterol. 2018 Nov;24(7 Suppl):S1-S20. doi: 10.4103/sjg.SJG_159_18[↩]
- Cooper, N. (2017), State of the art – how I manage immune thrombocytopenia. Br J Haematol, 177: 39-54. https://doi.org/10.1111/bjh.14515[↩]
- Oldham JM, Lee C, Valenzi E, Witt LJ, Adegunsoye A, Hsu S, Chen L, Montner S, Chung JH, Noth I, Vij R, Strek ME. Azathioprine response in patients with fibrotic connective tissue disease-associated interstitial lung disease. Respir Med. 2016 Dec;121:117-122. doi: 10.1016/j.rmed.2016.11.007[↩]
- Imazio M, Lazaros G, Brucato A, Gaita F. Recurrent pericarditis: new and emerging therapeutic options. Nat Rev Cardiol. 2016 Feb;13(2):99-105. doi: 10.1038/nrcardio.2015.115[↩]
- Touhami S, Diwo E, Sève P, Trad S, Bielefeld P, Sène D, Abad S, Brézin A, Quartier P, Koné Paut I, Weber M, Chiquet C, Errera MH, Sellam J, Cacoub P, Kaplanski G, Kodjikian L, Bodaghi B, Saadoun D. Expert opinion on the use of biological therapy in non-infectious uveitis. Expert Opin Biol Ther. 2019 May;19(5):477-490. doi: 10.1080/14712598.2019.1595578[↩]
- Mathian A, Miyara M, Cohen-Aubart F, Haroche J, Hie M, Pha M, Grenier P, Amoura Z. Relapsing polychondritis: A 2016 update on clinical features, diagnostic tools, treatment and biological drug use. Best Pract Res Clin Rheumatol. 2016 Apr;30(2):316-333. doi: 10.1016/j.berh.2016.08.001[↩]
- Meggitt SJ, Anstey AV, Mohd Mustapa MF, Reynolds NJ, Wakelin S. British Association of Dermatologists’ guidelines for the safe and effective prescribing of azathioprine 2011. Br J Dermatol. 2011 Oct;165(4):711-34. doi: 10.1111/j.1365-2133.2011.10575.x[↩][↩][↩]
- Williet N, Roblin X. Trend towards dose reduction of azathioprine as monotherapy in inflammatory bowel disease patients: what about for combination therapy? Therap Adv Gastroenterol. 2017 Jan;10(1):5-10. doi: 10.1177/1756283X16670074[↩]
- Joshi R, Singh S. Plica Neuropathica (Plica polonica) Following Azathioprine-induced Pancytopenia. Int J Trichology. 2010 Jul;2(2):110-2. doi: 10.4103/0974-7753.77523[↩][↩]
- Steponaitiene R, Kupcinskas J, Survilaite S, Varkalaite G, Jonaitis L, Kiudelis G, Denapiene G, Valantinas J, Skieceviciene J, Kupcinskas L. TPMT and ITPA genetic variants in Lithuanian inflammatory bowel disease patients: Prevalence and azathioprine-related side effects. Adv Med Sci. 2016 Mar;61(1):135-40. doi: 10.1016/j.advms.2015.09.008[↩]
- Guidelines for monitoring drug therapy in rheumatoid arthritis. American College of Rheumatology Ad Hoc Committee on Clinical Guidelines. Arthritis Rheum. 1996 May;39(5):723-31. https://doi.org/10.1002/art.1780390503[↩]
- Melzer N, Ruck T, Fuhr P, Gold R, Hohlfeld R, Marx A, Melms A, Tackenberg B, Schalke B, Schneider-Gold C, Zimprich F, Meuth SG, Wiendl H. Clinical features, pathogenesis, and treatment of myasthenia gravis: a supplement to the Guidelines of the German Neurological Society. J Neurol. 2016 Aug;263(8):1473-94. doi: 10.1007/s00415-016-8045-z[↩]
- Broekman MMTJ, Coenen MJH, van Marrewijk CJ, Wanten GJA, Wong DR, Verbeek ALM, Klungel OH, Hooymans PM, Guchelaar HJ, Scheffer H, Derijks LJJ, de Jong DJ; TOPIC Recruitment Team. More Dose-dependent Side Effects with Mercaptopurine over Azathioprine in IBD Treatment Due to Relatively Higher Dosing. Inflamm Bowel Dis. 2017 Oct;23(10):1873-1881. doi: 10.1097/MIB.0000000000001163[↩]
- Weinshilboum RM, Sladek SL. Mercaptopurine pharmacogenetics: monogenic inheritance of erythrocyte thiopurine methyltransferase activity. Am J Hum Genet. 1980 Sep;32(5):651-62. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1686086/pdf/ajhg00191-0015.pdf[↩]
- Nielsen OH, Vainer B, Rask-Madsen J. Review article: the treatment of inflammatory bowel disease with 6-mercaptopurine or azathioprine. Aliment Pharmacol Ther. 2001 Nov;15(11):1699-708. doi: 10.1046/j.1365-2036.2001.01102.x[↩][↩][↩]
- Saway PA, Heck LW, Bonner JR, Kirklin JK. Azathioprine hypersensitivity. Case report and review of the literature. Am J Med. 1988 May;84(5):960-4. doi: 10.1016/0002-9343(88)90079-4[↩]
- González-Olivares M, Khedaoui R, Martínez-Morán C, Borbujo J. Azathioprine-Induced Hypersensitivity Reaction Presenting as Erythema Nodosum. Actas Dermosifiliogr. 2017 Jul-Aug;108(6):591-593. English, Spanish. doi: 10.1016/j.ad.2016.12.006[↩]
- Karaahmet F, Akinci H, Ayte R, Hamamci M, Coskun Y, Yuksel I. Tremor as dose dependent side-effect of azathioprine in remission patient with ileal Crohn’s disease. J Crohns Colitis. 2013 Oct;7(9):e404. doi: 10.1016/j.crohns.2013.04.010[↩][↩]
- Fuggle NR, Bragoli W, Mahto A, Glover M, Martinez AE, Kinsler VA. The adverse effect profile of oral azathioprine in pediatric atopic dermatitis, and recommendations for monitoring. J Am Acad Dermatol. 2015 Jan;72(1):108-14. doi: 10.1016/j.jaad.2014.08.048[↩]
- Trivedi CD, Pitchumoni CS. Drug-induced pancreatitis: an update. J Clin Gastroenterol. 2005 Sep;39(8):709-16. doi: 10.1097/01.mcg.0000173929.60115.b4[↩]
- van Geenen EJ, de Boer NK, Stassen P, Linskens RK, Bruno MJ, Mulder CJ, Stegeman CA, van Bodegraven AA. Azathioprine or mercaptopurine-induced acute pancreatitis is not a disease-specific phenomenon. Aliment Pharmacol Ther. 2010 Jun;31(12):1322-9. doi: 10.1111/j.1365-2036.2010.04287.x[↩]
- Gupta P, Shaffrali F. An unusual side effect of azathioprine. Clin Exp Dermatol. 2015 Dec;40(8):929-30. doi: 10.1111/ced.12639[↩]
- Jack KL, Koopman WJ, Hulley D, Nicolle MW. A Review of Azathioprine-Associated Hepatotoxicity and Myelosuppression in Myasthenia Gravis. J Clin Neuromuscul Dis. 2016 Sep;18(1):12-20. doi: 10.1097/CND.0000000000000133[↩]
- Gregoriano C, Ceschi A, Rauber-Lüthy C, Kupferschmidt H, Banner NR, Krähenbühl S, Taegtmeyer AB. Acute thiopurine overdose: analysis of reports to a National Poison Centre 1995-2013. PLoS One. 2014 Jan 29;9(1):e86390. doi: 10.1371/journal.pone.0086390[↩]