Contents
What is cytomegalovirus
Cytomegalovirus (CMV), a type 5 herpesvirus, is a common virus found around the world that infects people of all ages. Between 50 percent and 80 percent of adults in the United States have had a cytomegalovirus infection by age 40. In the United States, nearly one in three children are already infected with cytomegalovirus by age 5 years. Although young children with cytomegalovirus infection generally have no symptoms, cytomegalovirus can be present in their body fluids for months after they first become infected. Regular hand washing, especially after contact with body fluids of young children, is commonly recommended to avoid spread of infections, including cytomegalovirus. Once cytomegalovirus is in a person’s body, it stays there for life and can reactivate. A person can also be reinfected with a different strain (variety) of the cytomegalovirus.
Most people infected with cytomegalovirus show no signs or symptoms. That’s because a healthy person’s immune system usually keeps the cytomegalovirus from causing illness. However, cytomegalovirus infection can cause serious health problems for people with weakened immune systems, as well as babies infected with the cytomegalovirus before they are born (congenital cytomegalovirus).
For most people, cytomegalovirus infection is not a serious health problem. However, certain groups are at high risk for serious complications from cytomegalovirus infection:
- Infants infected in utero (congenital cytomegalovirus infection)
- Very low birth weight and premature infants
- People with compromised immune systems, such as from organ and bone marrow transplants, and people infected with human immunodeficiency virus (HIV) or AIDS
Infections with cytomegalovirus can include: cytomegalovirus pneumonia, cytomegalovirus gastroenteritis, cytomegalovirus retinitis, and cytomegalovirus encephalitis (brain infection).
Cytomegalovirus transmission
Cytomegalovirus is spread through close contact with body fluids. People with cytomegalovirus may shed (pass) the virus in body fluids, such as urine, saliva, blood, tears, semen, and breast milk. cytomegalovirus is spread from an infected person in the following ways:
- From direct contact with urine or saliva, especially from babies and young children
- Through sexual contact
- From breast milk
- Through transplanted organs and blood transfusions
A woman who is infected with cytomegalovirus can pass the virus to her developing baby during pregnancy. Women may be able to lessen their risk of getting cytomegalovirus by reducing contact with saliva and urine from babies and young children. Some ways do this are: kissing children on the cheek or head rather than the lips, and washing hands after changing diapers. These cannot eliminate your risk of getting cytomegalovirus, but may lessen your chances of getting it. If a woman gets cytomegalovirus when she is pregnant, she can pass it on to her baby. Usually the babies do not have health problems. But some babies can develop lifelong disabilities.
Cytomegalovirus can also be transmitted to infants through contact with maternal genital secretions during delivery or through breast milk. Healthy infants and children who acquire cytomegalovirus after birth generally have few, if any, symptoms or complications from the infection. Women who are infected with cytomegalovirus can still breastfeed infants born full-term.
Cytomegalovirus can also be transmitted through transplanted organs and blood transfusions.
Although the virus is not highly contagious, it has been shown to spread among household members and young children in daycare centers.
A blood test can tell whether a person has ever been infected with cytomegalovirus. Most people with cytomegalovirus don’t need treatment. If you have a weakened immune system, your doctor may prescribe antiviral medicine. Good hygiene, including proper hand washing, may help prevent infections.
Contact with young children’s saliva and urine is a common way cytomegalovirus is passed to adults
Cytomegalovirus is common in children and can be found in especially high amounts in young children’s saliva and urine. Avoiding contact with saliva and urine from young children might reduce the risk of cytomegalovirus infection, although research studies don’t provide a clear answer. Some examples of how to avoid contact include kissing children on the cheek or head rather than the lips and washing hands after changing diapers. These cannot eliminate a woman’s risk of catching cytomegalovirus, but may lessen her chances of getting it. Reducing contact with saliva and urine will not prevent a previous cytomegalovirus infection from becoming active again.
Child care workers have a higher risk of cytomegalovirus infection because they work closely with many young children and should be counseled about cytomegalovirus and its risks.
Characteristics of cytomegalovirus
Cytomegalovirus is a herpesvirus. This group of viruses include herpes simplex virus types 1 and 2, varicella-zoster virus, and Epstein-Barr virus. These viruses share a characteristic ability to establish lifelong latency. After initial infection, which may cause few symptoms, cytomegalovirus becomes latent, residing in cells without causing detectable damage or clinical illness.
People who are infected with cytomegalovirus sometimes shed the virus in body fluids, such as urine, saliva, blood, tears, semen, and breast milk. The shedding of virus may occur intermittently, without any detectable signs, and without causing symptoms. However, in people who are severely immunocompromised by medication or disease, viral reactivation may lead to symptomatic disease.
Cytomegalovirus prognosis
Cytomegalovirus infection in the immunocompromised host can be life-threatening, and the severity of the disease is dependent on the strength of the individual’s immune system. Individuals who have undergone bone marrow transplant have been shown to have the highest mortality risk.
About 7% of affected infants develop severe generalized disease, in which the brain is almost always involved. Otherwise milder effects are more common.
Cytomegalovirus complications
Complications of cytomegalovirus infection vary, depending on your overall health and when you were infected.
Healthy adults
Rarely, cytomegalovirus causes a healthy adult to develop mononucleosis. Other rare complications for healthy adults include problems with the digestive system, liver, brain and nervous system.
People with weakened immunity
Disseminated fatal infection with widespread visceral involvement may occur in the immunocompromised and may cause encephalitis, retinitis, pneumonitis and diffuse involvement of the gastrointestinal tract.
Complications of cytomegalovirus infection can include:
- Vision loss, due to inflammation of the light-sensing layer of the eye (cytomegalovirus retinitis)
- Digestive system problems, including inflammation of the colon (colitis), esophagus (esophagitis) and liver (hepatitis)
- Nervous system problems, including brain inflammation (encephalitis)
- Pneumonia
Infants with congenital cytomegalovirus
Complications are more likely to develop if the infant’s mother had a primary cytomegalovirus infection during pregnancy, rather than a reactivated infection. Complications for the baby can include:
- Hearing loss
- Intellectual disability
- Vision problems
- Seizures
- Lack of coordination
- Weakness or problems using muscles.
Cytomegalovirus pregnancy
In the United States, nearly half of women have already been infected with cytomegalovirus before their first pregnancy. Of women who have never had a cytomegalovirus infection, it is estimated that 1-4% of them will have a primary infection during pregnancy.
A woman who has a primary cytomegalovirus infection during pregnancy is more likely to pass cytomegalovirus to her fetus than a women who is reinfected or has a reactivation of the latent virus during pregnancy. However, in the United States, 50-75% of congenital cytomegalovirus infections occur among infants born to mothers already infected with cytomegalovirus, who either had a reinfection or a reactivation during pregnancy.
Most babies born with a cytomegalovirus infection will be healthy 1):
- About 4 out of 5 babies born with cytomegalovirus infection will not have health problems.
- However, about 1 out of 5 (20%) babies with cytomegalovirus infection at birth have long-term health problems. That means of the 20,000 or so babies born with cytomegalovirus infection each year, most will never have health problems. About 4,000 babies will have long-term health problems.
Routine screening for primary cytomegalovirus infection during pregnancy is not recommended in the United States. Most laboratory tests currently available cannot conclusively detect if a primary cytomegalovirus infection occurred during pregnancy. This makes it difficult to counsel pregnant women about the risk to their fetuses. The lack of a proven treatment to prevent or treat infection of the fetus reduces the potential benefits of prenatal screening.
Very low birth weight and premature infants
There are no recommendations against breastfeeding by mothers who are cytomegalovirus-seropositive. However, premature infants (born <30 weeks gestational age and <1500g) who acquire cytomegalovirus from breast milk may be at risk of developing a late-onset sepsis-like syndrome. The potential benefits of human milk versus the risk of cytomegalovirus transmission should be considered when making a decision about breastfeeding of very low birth weight infants (birth weight <1500 g) by mothers known to be cytomegalovirus-seropositive. Freezing and pasteurization of breast milk can decrease the risk of transmission; however, freezing does not eliminate the risk of transmission.
Congenital cytomegalovirus
When a baby is born with cytomegalovirus infection, it is called congenital cytomegalovirus infection. About one out of every 200 babies are born with congenital cytomegalovirus infection. However, only about one in five babies with congenital cytomegalovirus infection will be sick from the virus or will have long-term health problems.
Transmission
Women can pass cytomegalovirus to their baby during pregnancy. The virus in the woman’s blood can cross through the placenta and infect the baby. This can happen when a pregnant woman experiences a first-time infection, a reinfection with a different cytomegalovirus strain (variety), or a reactivation of a previous infection during pregnancy.
Congenital cytomegalovirus signs and symptoms
Most babies with congenital cytomegalovirus infection never show signs or have health problems. However, some babies may have health problems that are apparent at birth or may develop later during infancy or childhood. Although not fully understood, it is possible for cytomegalovirus to cause the death of a baby during pregnancy (pregnancy loss).
Some babies may have signs of congenital cytomegalovirus infection at birth. These signs include:
- Premature birth,
- Liver, lung and spleen problems,
- Small size at birth (Low birth weight),
- Small head size (microcephaly) and
- Seizures.
About 1 in 10 (10%) babies with cytomegalovirus infection at birth will have noticeable signs, such as a small head (microcephaly), jaundice, or an enlarged liver or spleen. These babies may also have long-term health problems such as hearing loss, developmental delay, and vision loss.
About 40-60% of infants born with signs of congenital cytomegalovirus infection at birth will have long-term health problems, such as:
- Hearing loss,
- Vision loss,
- Intellectual disability[2 pages],
- Small head size (microcephaly),
- Lack of coordination,
- Weakness or problems using muscles, and
- Seizures.
About 1 in 7 (15%) babies born with cytomegalovirus infection may develop hearing loss, from birth up to age 5 years, even though they have no noticeable signs at birth. Treatment with antivirals for these babies is not currently recommended, but is being evaluated to assess safety and possible benefit. Early detection and interventions such as hearing aids and speech therapy can help their development. These babies do not appear to have other long-term health problems. Hearing loss may be present at birth or may develop later in babies who passed their newborn hearing test. About 10-20% of infants with congenital cytomegalovirus infection who have no signs at birth will have, or later develop, hearing loss.
Babies with signs of cytomegalovirus infection at birth who are diagnosed and treated early may have better health outcomes 2):
Because the signs of cytomegalovirus infection at birth are similar to other medical conditions, the diagnosis must be confirmed by laboratory testing within two to three weeks after birth. Babies who are diagnosed with cytomegalovirus infection and have signs can be treated with medications called antivirals, which may improve hearing and developmental outcomes.
Congenital cytomegalovirus diagnosis
Congenital cytomegalovirus infection can be diagnosed by testing a newborn baby’s saliva, urine, or blood. Such specimens must be collected for testing within two to three weeks after the baby is born in order to confirm a diagnosis of congenital cytomegalovirus infection.
Only tests that detect cytomegalovirus live virus (through viral culture) or cytomegalovirus DNA (through polymerase chain reaction (PCR)) can be used to diagnose congenital cytomegalovirus infection. Congenital cytomegalovirus infection cannot be diagnosed using samples collected more than two to three weeks after birth because testing after this time cannot distinguish between congenital infection and an infection acquired during and after delivery.
Serological tests are not recommended for diagnosing congenital cytomegalovirus infection. Maternal IgG antibodies pass through the placenta during pregnancy; thus, cytomegalovirus IgG testing of infants may reflect maternal antibody status, and does not necessarily indicate infection in the infant. Maternal IgM antibodies do not cross the placenta and, thus, cytomegalovirus IgM in the newborn would indicate congenital infection, but is only present in 25-40% of newborns with congenital infection.
Congenital cytomegalovirus treatment
- Antiviral medications, such as ganciclovir or valganciclovir, may improve hearing and developmental outcomes in infants with symptoms of congenital cytomegalovirus disease. Ganciclovir can have serious side effects, and has only been studied in infants with symptomatic congenital cytomegalovirus disease. There is limited data on the effectiveness of ganciclovir or valganciclovir to treat infants with isolated hearing loss.
- Any infant diagnosed with congenital cytomegalovirus infection should have his or her hearing and vision tested regularly. Most infants with congenital cytomegalovirus grow up healthy. However, if the child has delayed onset of hearing or vision problems, early detection may improve outcomes.
Use of antivirals for treating babies with congenital cytomegalovirus infection who have no signs at birth is not currently recommended.
Babies with congenital cytomegalovirus infection, with or without signs at birth, should have regular hearing checks.
Regularly follow-up with your baby’s doctor to discuss the care and additional services your child may need.
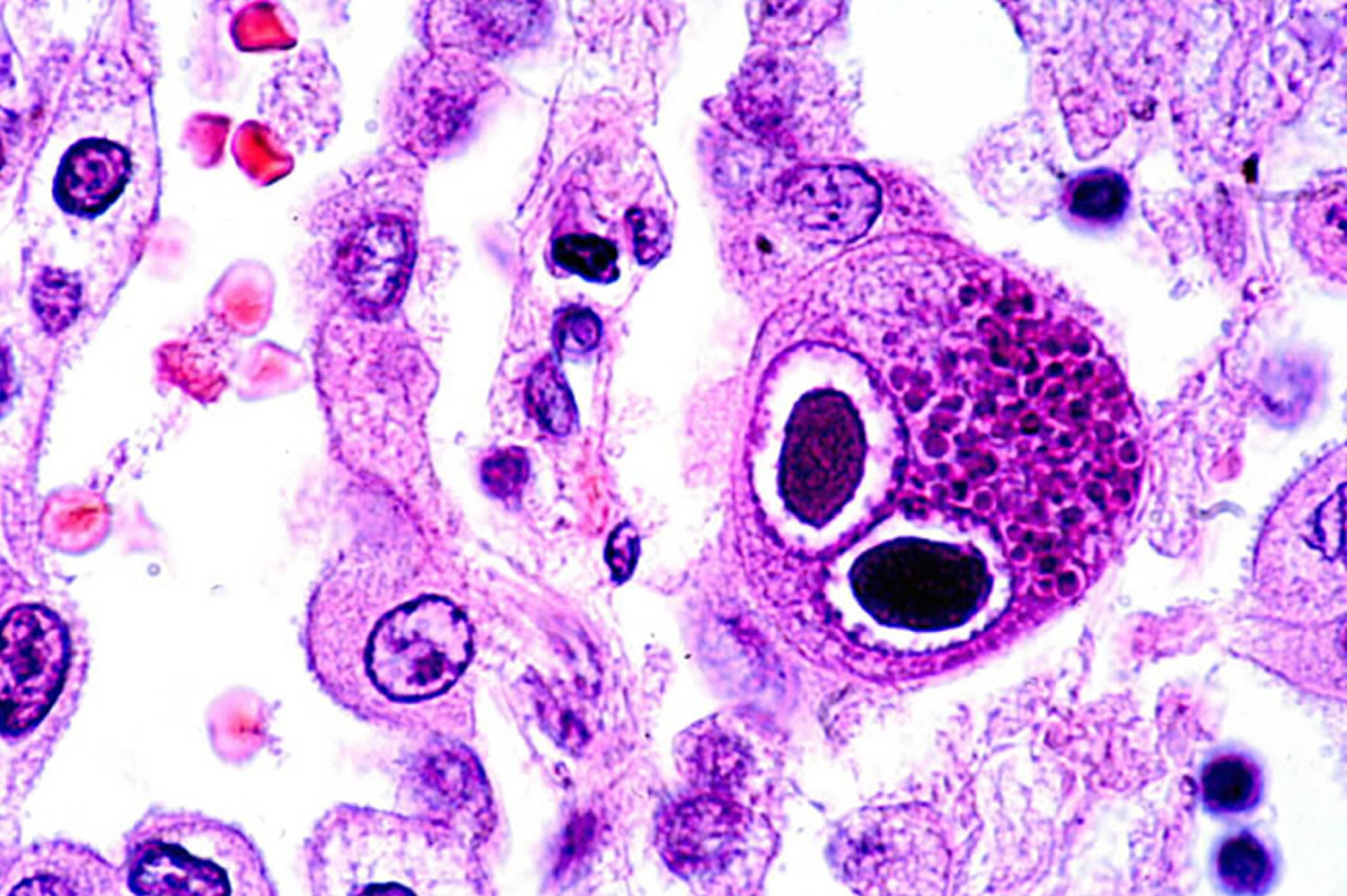
Cytomegalovirus retinitis
Cytomegalovirus remains an important cause of opportunistic infection in severe immunocompromised states secondary to depression of cell-mediated immunity (with CD4 counts <50/μL) more commonly due to acquired immune deficiency syndrome (AIDS), followed by immunosuppressive drugs, blood transfusion and postallogeneic hematopoietic stem cell transplantation and lymphoma 3).
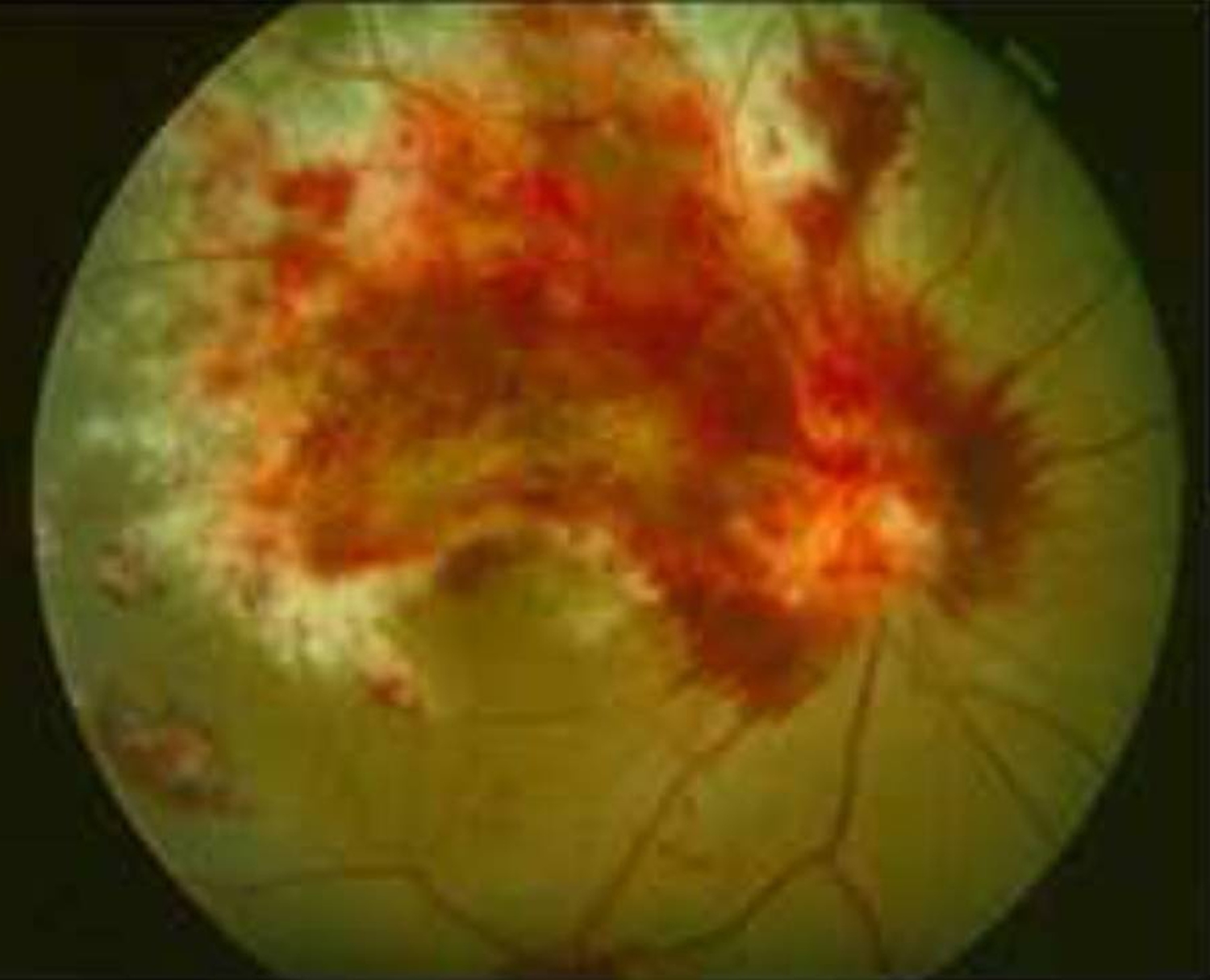
Prior to the advent of the acquired immune deficiency syndrome (AIDS), cytomegalovirus retinitis (Figure 1) was a rare disease seen primarily among transplant patients, but also among those with congenital cytomegalovirus infection and other forms of immune compromise 4). Cytomegalovirus retinitis was estimated to affect ~1% of patients undergoing renal transplant and ~0.5% of those undergoing bone marrow transplant 5). Cytomegalovirus retinitis became evident early in the AIDS epidemic that cytomegalovirus retinitis was a frequent opportunistic infection among patients with AIDS, and that it typically occurred in patients with CD4+ T cells (helper T cells) <50 cells/μL 6).
With the advent of highly active antiretroviral therapy (HAART), the incidence of cytomegalovirus retinitis has decreased by 80-90%, although that decrease has leveled off and cytomegalovirus retinitis continues to occur 7). HIV infection now is a chronic disease with survival after infection with HIV estimated at >14 years 8). However, because of late testing among many high-risk patients, approximately one-third of patients with newly-diagnosed HIV infection in the United States still will progress to AIDS within one year of the diagnosis of HIV infection 9), resulting in an ongoing population at risk for cytomegalovirus retinitis.
Cytomegalovirus retinitis symptoms
Some people with cytomegalovirus retinitis have no symptoms.
If there are symptoms, they may include:
- Blind spots
- Blurred vision and other vision problems
- Floaters
Cytomegalovirus retinitis usually begins in 1 eye, but often progresses to the other eye. Without treatment, damage to the retina can lead to blindness in 4 to 6 months or less.
The primary cause of visual loss in patients with cytomegalovirus retinitis is necrotic damage to the fovea or optic nerve from cytomegalovirus infection (Table 1) 10). In the era before HAART, the second leading cause of visual loss was retinal detachment, whereas in the HAART era it was cataract, in part due to the reduced incidence of retinal detachment in the HAART era 11). Macular edema was uncommon in the pre-HAART era, but is a more common cause of visual impairment in the HAART era, particularly among patients with immune recovery uveitis 12).
Figure 1. Cytomegalovirus retinitis
Table 1. Causes of Visual Loss from Cytomegalovirus Retinitis
| Pre-HAARTa era | HAART era | ||
|---|---|---|---|
| Cause (% of eyes) | 20/200 or worse | 20/50 or worse | 20/200 or worse |
| Macula or optic nerve disease | 54-84 | 63.1 | 69.8 |
| Cataract | No data | 22.0 | 28.6 |
| Retinal detachment | 36-63 | 12.9 | 16.7 |
| Macular edema | <5 | 12.4 | 11.1 |
| Immune recovery uveitis | Not applicable | 4.3 | 3.2 |
| Epiretinal membrane | <5 | 3.3 | 4.1 |
Cytomegalovirus retinitis prognosis
Even with treatment, the disease can worsen to blindness. This progression may be because the virus becomes resistant to the antiviral drugs so that the drugs are no longer effective, or because the person’s immune system has deteriorated further.
Cytomegalovirus retinitis may also lead to retinal detachment, in which the retina detaches from the back of the eye, causing blindness.
Cytomegalovirus retinitis diagnosis
Cytomegalovirus retinitis is diagnosed through an ophthalmologic exam. Dilation of the pupils and ophthalmoscopy will show signs of cytomegalovirus retinitis.
Cytomegalovirus infection can be diagnosed with blood or urine tests that look for substances specific to the infection. A tissue biopsy can detect the viral infection and presence of cytomegalovirus virus particles, but this is rarely done.
Cytomegalovirus retinitis treatment
Treatment of cytomegalovirus retinitis is influenced by the fact that all available treatments are virostatic and do not eliminate the virus. A histopathologic study of treated cytomegalovirus retinitis demonstrated the presence of viral DNA at the border of the lesion, but ineffective assembly of intact virions 14). With cessation of therapy viral assembly resumes, as does clinical cytomegalovirus retinitis. Therefore, unless there is immune recovery, chronic, life-long therapy to prevent relapse is required for patients with AIDS. Three United States Food and Drug Administration (FDA)-approved drugs are available for the treatment of cytomegalovirus retinitis: ganciclovir (Cytovene, Roche Pharmaceuticals, Nutley, NJ), foscarnet (Foscavir, AstraZeneca Pharmaceuticals LP, Wilmington, DE), and cidofovir (Vistide, Gilead, Foster City, CA). A fourth drug, fomivirsen (Vitravene, Isis Pharmaceuticals, Inc., Carlsbad, CA), an anti-sense aptamer, was FDA-approved and then withdrawn from the market when the incidence of cytomegalovirus retinitis declined after the introduction of HAART. Ganciclovir can be administered by IV infusion, surgical placement of a sustained-release implant into the vitreous (Vitrasert, Bausch & Lomb Pharmaceuticals, Inc., Tampa, FL), which typically lasts at least six months, or orally with the pro-drug valganciclovir (Valcyte, Roche Pharmaceuticals). An oral formulation of ganciclovir, with poor bioavailability, largely has been replaced by valganciclovir. Foscarnet and cidofovir are administered intravenously. Ganciclovir and foscarnet also are administered off-label as intravitreal injections 15). Cidofovir was investigated as an intravitreal drug, but its use largely has been discontinued due to a narrow therapeutic-toxic window and problems with uveitis and hypotony 16). The treatment of cytomegalovirus retinitis has been the subject of numerous clinical trials, sponsored both by the pharmaceutical industry and by the National Institutes of Health (NIH).
Lastly, given the limitations on access to more expensive treatments, there is the potential for substantial visual loss due to cytomegalovirus retinitis 17). In a publication from a hospital in Chiang Mai, Thailand in 2007, cytomegalovirus retinitis retinitis was the second leading cause of blindness (34.6%) behind cataract (36.3%) and ahead of glaucoma (3.0%), diabetic retinopathy (2.4%), and age-related macular degeneration (0.8%) 18).
Cytomegalovirus causes
Cytomegalovirus is related to the viruses that cause chickenpox, herpes simplex and mononucleosis. cytomegalovirus may cycle through periods when it lies dormant and then reactivates. If you’re healthy, cytomegalovirus mainly stays dormant.
During activation you can pass the virus to other people. Casual contact doesn’t transmit cytomegalovirus. The virus is spread through body fluids — including blood, urine, saliva, breast milk, tears, semen and vaginal fluids.
Transmission might occur through:
- Touching your eyes or the inside of your nose or mouth after coming into contact with the body fluids of an infected person.
- Sexual contact with an infected person.
- The breast milk of an infected mother.
- Organ transplantation or blood transfusions.
- Birth. An infected mother can pass the virus to her baby before or during birth. The risk of virus transmission to your baby is higher if it’s the first time you’ve had the infection rather than a reactivated infection.
Risk factors for cytomegalovirus
Cytomegalovirus is a widespread and common virus that can infect almost anyone. Most healthy children and adults who contract the virus have few if any symptoms, so cytomegalovirus often goes undiagnosed.
Cytomegalovirus is most commonly an opportunistic infection in the immunocompromised.
For Example:
- recipients of bone marrow transplant transplants.
- solid-organ transplants.
- in patients with AIDS
- cytomegalovirus infection can be a primary infection, as well as a re-activation. Congenital cytomegalovirus infection can occur in both cases.
Cytomegalovirus prevention
cytomegalovirus is common in children and can be found in especially high amounts in young children’s saliva and urine. Avoiding contact with saliva and urine from young children might reduce the risk of cytomegalovirus infection. Some examples of how to avoid contact include kissing children on the cheek or head rather than the lips and washing hands after changing diapers. These cannot eliminate a person’s risk of catching cytomegalovirus, but may lessen her chances of getting it.
Careful hygiene is the best prevention against cytomegalovirus. You can take these precautions:
- Wash your hands often. Use soap and water for 15 to 20 seconds, especially if you have contact with young children or their diapers, drool or other oral secretions. This is especially important if the children attend child care.
- Avoid contact with tears and saliva when you kiss a child. Instead of kissing a child on the lips, for instance, kiss on the forehead. This is especially important if you’re pregnant.
- Avoid sharing food or drinking out of the same glass as others. Sharing glasses and kitchen utensils can spread the cytomegalovirus virus.
- Be careful with disposable items. When disposing of diapers, tissues and other items that have been contaminated with bodily fluids, be careful not to touch your hands to your face until after thoroughly washing your hands.
- Clean toys and countertops. Clean any surfaces that come in contact with children’s urine or saliva.
- Practice safe sex. Wear a condom during sexual contact to prevent spreading the cytomegalovirus virus through semen and vaginal fluids.
If you have a compromised immune system, you may benefit from taking antiviral medication to prevent cytomegalovirus disease.
Experimental vaccines are being tested for women of childbearing age. These vaccines may be useful in preventing cytomegalovirus infection in mothers and infants, and reducing the chance that babies born to women who are infected while pregnant will develop disabilities.
Cytomegalovirus signs and symptoms
For most healthy people who acquire cytomegalovirus (cytomegalovirus) after birth, there are few symptoms and aren’t aware that they have been infected and there’s no long-term health consequences. Occasionally, some people who acquire cytomegalovirus infection may experience a mononucleosis-like syndrome with prolonged fever and hepatitis.
In some cytomegalovirus cases, infection in healthy people can cause mild illness that may include
- Fever,
- Sore throat,
- Muscle aches,
- Fatigue, and
- Swollen glands.
Cytomegalovirus mononucleosis is less likely than infectious mononucleosis to cause enlarged lymph nodes and spleen.
Once a person becomes infected, the virus establishes lifelong latency and may reactivate intermittently. Disease from reactivation of cytomegalovirus infection rarely occurs unless the person’s immune system is suppressed due to therapeutic drugs or disease.
People with weakened immune systems who get cytomegalovirus can have more serious symptoms affecting the eyes, lungs, liver, esophagus, stomach, and intestines. Babies born with cytomegalovirus can have brain, liver, spleen, lung, and growth problems. Hearing loss is the most common health problem in babies born with congenital cytomegalovirus infection, which may be detected soon after birth or may develop later in childhood.
If your immune system is weakened, you might experience more-serious signs and symptoms affecting your:
- Eyes
- Lungs
- Liver
- Esophagus
- Stomach
- Intestines
- Brain
Babies
Most babies with congenital cytomegalovirus appear healthy at birth.
A few babies with congenital cytomegalovirus who appear healthy at birth can develop signs over time — sometimes not for months or years after birth. The most common of these late-occurring signs are hearing loss and developmental delay. A small number of babies may also develop vision problems.
Babies with congenital cytomegalovirus who are sick at birth tend to have significant signs and symptoms, including:
- Premature birth
- Low birth weight
- Yellow skin and eyes (jaundice)
- Enlarged and poorly functioning liver
- Purple skin splotches or a rash or both
- Abnormally small head (microencephaly)
- Enlarged spleen
- Pneumonia
- Seizures
Cytomegalovirus diagnosis
Primary cytomegalovirus infections usually go unrecognized because most people are asymptomatic or do not have specific symptoms. Primary cytomegalovirus infection should be suspected if a woman:
- Has symptoms of infectious mononucleosis but has negative test results for Epstein-Barr virus, or
- Shows signs of hepatitis, but has negative test results for hepatitis A, hepatitis B, and hepatitis C.
Cytomegalovirus may be detected by viral culture or polymerase chain reaction (PCR) of infected blood, urine, saliva, cervical secretions, or breast milk.
Cytomegalovirus infection is usually diagnosed using serologic testing. Serum samples collected one to three months apart can be used to diagnose primary infection. Seroconversion (1st sample IgG negative, 2nd sample IgG positive) is clear evidence for recent primary infection. However, diagnosis of cytomegalovirus infection between birth and one year can be complicated by the presence of maternal cytomegalovirus IgG.
Interpretation of Laboratory Tests
Laboratory Diagnosis of cytomegalovirus Infection for Persons ≥12 Months of Age
Serologic tests that detect cytomegalovirus antibodies (IgM and IgG antibody to cytomegalovirus) are widely available from commercial laboratories. The enzyme-linked immunosorbent assay (ELISA) is the most common serologic test for measuring antibody to cytomegalovirus.
- A positive test for cytomegalovirus IgG indicates that a person was infected with cytomegalovirus at some time during their life, but does not indicate when a person was infected. This applies for persons ≥12 months of age when maternal antibodies are no longer present.
- Measurement of cytomegalovirus IgG in paired samples taken one to three months apart can be used to diagnose primary infection; seroconversion (1st sample IgG negative, 2nd sample IgG positive) is clear evidence for recent primary infection.
- The presence of cytomegalovirus IgM cannot be used by itself to diagnose primary cytomegalovirus infection because IgM can also be present during secondary cytomegalovirus infection, which includes reinfection with a different strain or reactivation of latent cytomegalovirus acquired in the past. IgM positive results in combination with low IgG avidity results are considered reliable evidence for primary infection, with limitations of avidity testing described below.
- IgG avidity testing may provide useful information regarding timing of infection. IgG antibodies produced following primary cytomegalovirus infection have low avidity (low binding strength). Two to four months following infection, IgG antibodies mature to high-avidity (high binding strength). Therefore, avidity assays can be used to assess low avidity (which indicates recent infection) versus high avidity (past infection). However, not all avidity assays have been validated and, therefore, should be interpreted with caution. The review by Prince and Lapé-Nixon 19) lists commercial tests for cytomegalovirus IgG avidity that can be purchased in the United States, and describes challenges with interpreting cytomegalovirus IgG avidity testing results.
- Cytomegalovirus IgG avidity testing is now a proven, valuable laboratory tool for diagnosing primary cytomegalovirus infection during pregnancy. Low avidity indicates primary infection within the preceding 3 to 4 months, with an increased risk of intrauterine transmission to the fetus/newborn. High avidity during the first trimester excludes postconception primary infection and indicates a low risk of intrauterine transmission. Although not quite as powerful as a high-avidity result, an intermediate-avidity result during the first trimester also indicates a low risk of intrauterine transmission. In contrast, an intermediate-avidity or high-avidity result during the second or third trimester does not rule out postconception primary infection and is associated with increased risk of transmission. The vast majority of patients with primary cytomegalovirus infection exhibit both low cytomegalovirus IgG avidity and detectable cytomegalovirus IgM, but rarely is only one of these abnormal results present; thus, maximum detection of primary cytomegalovirus infections, particularly when the first sample is collected after the first trimester, requires that both cytomegalovirus IgG avidity and cytomegalovirus IgM avidity testing be performed on cytomegalovirus IgG-positive samples. Several diagnostic companies now offer cytomegalovirus IgG avidity kits/assays, but many are not yet available for purchase worldwide. Manufacturers are encouraged to seek solutions to distribution and regulatory issues that currently block the widespread availability of their products, thus enabling the global application of cytomegalovirus IgG avidity testing as a tool for assessing cytomegalovirus transmission risk during pregnancy.
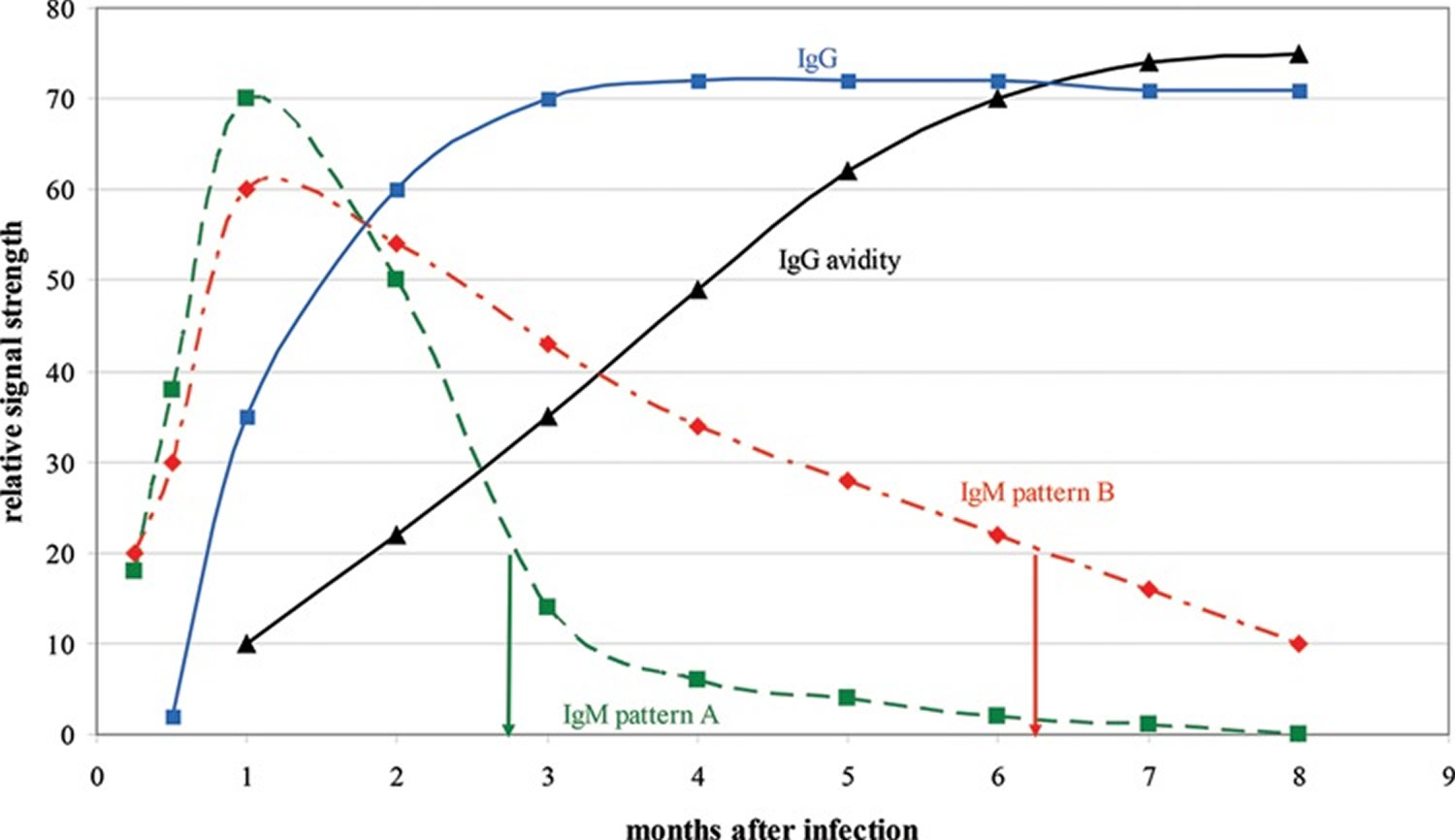
Figure 2. Relative changes in cytomegalovirus IgM, IgG, and IgG avidity levels over time following primary cytomegalovirus infection
Note: IgM pattern A represents the typical IgM response pattern, whereas IgM pattern B represents long-term IgM persistence. In a CMV IgG-positive individual, an IgM-positive result of 20 indicates infection around 3 months previously if the individual exhibits IgM pattern A but around 6 months previously if the individual exhibits IgM pattern B. By employing CMV IgG avidity testing, the correct time since infection can be determined; a low-avidity result (expected to be about 30 based on this figure) indicates primary infection about 3 months previously, whereas a high-avidity result (expected to be about 70) indicates primary infection more than 6 months previously.
[Source 20)]Table 2. Commercially available CMV IgG avidity kits
| Manufacturer (test name) | Method | Dissociating agent | Low-avidity scoreb | High-avidity scoreb | Can be purchased in USA? |
|---|---|---|---|---|---|
| Bio-Rad | ELISA | Urea | <40 | >55 | No |
| Diesse | ELISA | Urea | <30 | >40 | Yes |
| Euroimmun | ELISA | Urea | <40 | >60 | Yes |
| Radim | ELISA | Urea | <35 | >45 | Yes |
| Technogenetics | ELISA | Potassium thiocyanate | <25 | >45 | No |
| Vidia | ELISA | Urea | <40 | >60 | Yes |
| Virion/Serion | ELISA | Urea | <45 | >55 | Yes |
| Mikrogen | IBL | Urea | NA | NA | Yes |
| bioMérieux (Vidas) | ELFA | Urea | <40 | >65 | No |
| DiaSorin (Liaison) | CIA | Urea | <20 | >30 | No |
| Roche (Elecsys) | V-CIA | Guanidine chloride | <45 | >55 | No |
| Abbott (Architect) | CMIA | Nonec | <50 | >60 | No |
Laboratory Diagnosis of Congenital cytomegalovirus Infection in Newborns
The standard laboratory test for diagnosing congenital cytomegalovirus infection is polymerase chain reaction (PCR) on saliva, with urine usually collected and tested for confirmation. The reason for the confirmatory test on urine is because most cytomegalovirus seropositive mothers shed cytomegalovirus virus in their breast milk. This can cause a false-positive cytomegalovirus result on saliva collected shortly after the baby has breast fed.
Specific steps for appropriate collection of saliva samples from a baby are as follows:
- Saliva specimen should be collected more than one hour after breastfeeding and within 3 weeks of birth because detection of cytomegalovirus after 3 weeks could be the result of post-partum infection.
- Insert a sterile cotton or polyester swab into the baby’s mouth between the gum and cheek and swirl for several seconds.
- Remove the swab and place into buffer formulated for PCR diagnostic testing (several are available). If cytomegalovirus is present, it will leach from swab to the liquid.
- The liquid is processed according to manufacturer’s instructions, and PCR testing is performed according to the protocol in the laboratory.
- Specific procedures and interpretation of tests vary according to laboratory.
Currently, testing of newborns for cytomegalovirus is not routinely performed. Though, some states perform targeted cytomegalovirus testing of newborns who fail hearing screening.
The Centers for Disease Control and Prevention (CDC) is currently studying whether dried blood spots (DBS), which are already collected on almost all newborns, can identify the majority of children most likely to suffer long-term health problems from congenital cytomegalovirus.
Cytomegalovirus treatment
Healthy people who are infected with cytomegalovirus usually do not require medical treatment.
Antiviral medications are available to treat cytomegalovirus infection in people who have weakened immune systems who have either sight-related (cytomegalovirus retinitis) or life-threatening illnesses due to cytomegalovirus infection and babies who show symptoms of congenital cytomegalovirus infection. They can slow reproduction of the virus, but can’t eliminate it.
- In immunocompromised patients, ganciclovir (5mg/kg daily for 14-21 days) reduces retinitis and gastrointestinal damage and can eliminate cytomegalovirus from blood, urine and respiratory secretions.
- Foscarnet and cidofovir can be used where there is drug resistance to ganciclovir in some AIDS patients, however, as they are nephrotoxic, their use should be restricted to severe cases of the disease.
Researchers are studying new medications and vaccines to treat and prevent cytomegalovirus.
References [ + ]