Contents
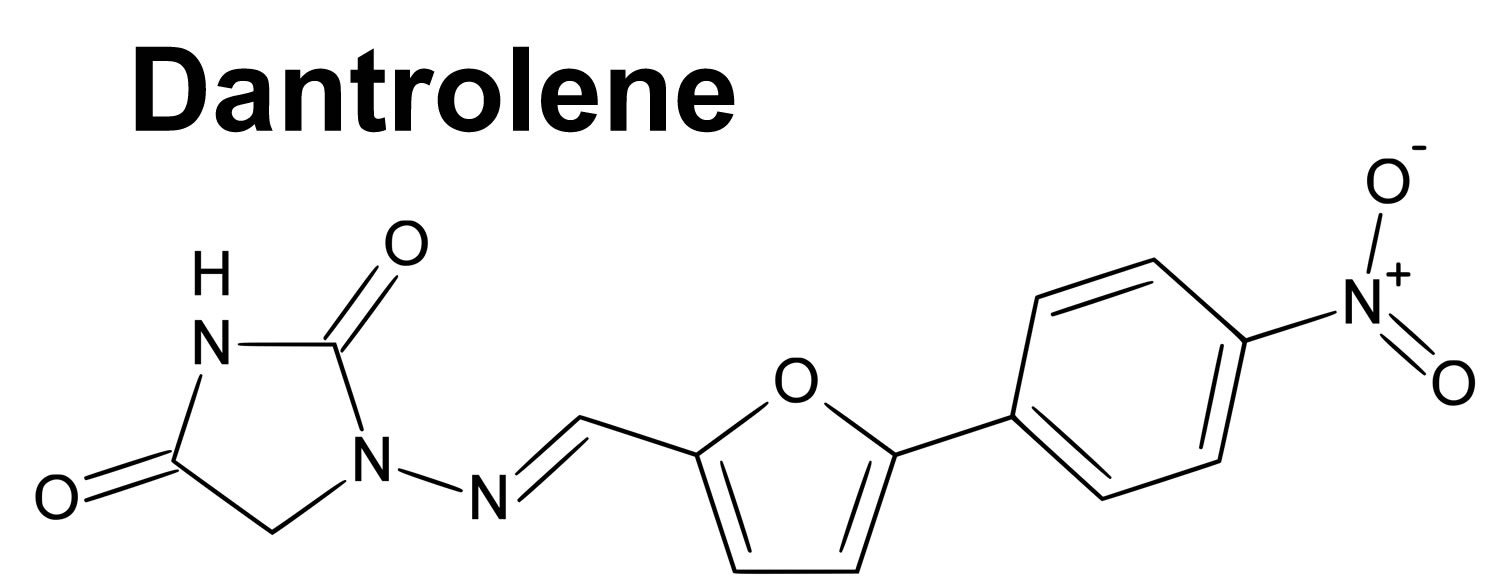
Dantrolene
Dantrolene (Dantrium, Revonto, Ryanodex) is a postsynaptic muscle relaxant that is used to treat muscle spasticity (muscle stiffness and tightness) or muscle spasms caused by conditions such as spinal cord injuries, stroke, multiple sclerosis, or cerebral palsy. Dantrolene is also used to prevent, treat or reduce the risk of malignant hyperthermia, a serious reaction to particular anesthetic drugs that causes a fast rise in body temperature (hyperthermia), muscle contractions, abnormally fast breathing, breakdown of muscle fibers (rhabdomyolysis), and increased acid levels in the blood and other tissues (acidosis) 1. Dantrolene is currently the only specific medication used for treating a malignant hyperthermia crisis. Dantrolene differs from other commonly used muscle relaxants in acting peripherally on muscle by blocking postsynaptic acetylcholine receptors, rather than centrally on the spinal cord or brain.
Dantrolene comes as capsules of 25, 50 and 100 mg to be taken by mouth. When used to treat muscle spasticity, in adults dantrolene initial dose is 25 mg taken once a day for 7 days and then increased gradually every 7 days to a dose of 25 to 100 mg three or four times a day. Your doctor will probably start you on a low dose of dantrolene for muscle spasticity (muscle stiffness and tightness) and gradually increase your dose, not more than once every 7 days. If your symptoms do not improve or get worse within 45 days of taking dantrolene, see your doctor.
Dantrolene is also available in parenteral formulations for therapy of acute episodes of malignant hyperthermia; the recommended initial dose being 1 mg/kg intravenously.
When used to prevent malignant hyperthermia, Dantrolene is usually given orally in doses of 4 to 8 mg/kg daily, starting 1 or 2 days before surgery. When used after a malignant hyperthermia crisis, Dantrolene is usually given in 4 divided doses for 1 to 3 days . Follow the directions on your prescription label carefully, and ask your doctor or pharmacist to explain any part you do not understand. Take dantrolene exactly as directed. Do not take more or less of it or take it more often than prescribed by your doctor.
Dantrolene common side effects include weakness, nausea, drowsiness, fatigue and dizziness. The intravenous administration of dantrolene in healthy volunteers has resulted in skeletal muscle weakness, shortness of breath (dyspnea), breathing muscle weakness, and decreased inspiratory capacity 2. These are expected symptoms given the mechanism of action of Dantrolene. Oral dantrolene carries a black box warning for the potential for life-threatening liver damage, including overt hepatitis. Liver function test should be performed in patient before administering the oral capsule Dantrolene and requires monitoring throughout the course of treatment. Dantrolene should stop immediately if liver function becomes impaired 3.
Dantrolene can cause life-threatening liver damage 4. Do not use dantrolene for conditions other than those recommended by your doctor. Do not take dantrolene at a time when you need muscle tone for safe balance and movement during certain activities. Do not take more than the recommended amount prescribed by your doctor. Do not take dantrolene if you have liver disease. If you experience any of the following symptoms of liver problems, see your doctor immediately: yellowing of the skin or eyes, dark urine, black tarry stools, nausea, vomiting, loss of appetite, pain in the upper right part of the stomach, or unusual bruising or bleeding.
Keep all appointments with your doctor and the laboratory. Your doctor will order certain lab tests before and during your treatment to check your response to dantrolene.
Dantrolene special precautions
Before taking dantrolene:
- tell your doctor and pharmacist if you are allergic to dantrolene, any other medications, or any of the ingredients in dantrolene capsules. Ask your pharmacist for a list of the ingredients.
- tell your doctor and pharmacist what other prescription and nonprescription medications, vitamins, nutritional supplements, and herbal products you are taking or plan to take. Be sure to mention any of the following: antidepressants; medications for anxiety; calcium channel blockers such as amlodipine (Norvasc), diltiazem (Cardizem, Dilacor, Tiazac, others), felodipine (Plendil), isradipine (DynaCirc), nicardipine (Cardene), nifedipine (Adalat, Procardia), nimodipine (Nimotop), nisoldipine (Sular), and verapamil (Calan, Verelan); estrogen-containing contraceptives (birth control pills, patches, rings, and injections); or estrogen replacement therapy; medications for mental illness; medications for seizures; sedatives; sleeping pills; or tranquilizers. Your doctor may need to change the doses of your medications or monitor you carefully for side effects.
- tell your doctor if you have or have ever had muscle spasms from a rheumatic disorder, or heart or lung disease.
- tell your doctor if you are pregnant, plan to become pregnant, or are breast-feeding. If you become pregnant while taking dantrolene, call your doctor immediately.
- if you are having surgery, including dental surgery, tell the doctor or dentist that you are taking dantrolene.
- you should know that dantrolene may make you drowsy. Do not drive a car, avoid hazardous activity or operate machinery until you know how dantrolene will affect you. Dizziness or drowsiness can cause falls, accidents, or severe injuries.
- ask your doctor about the safe use of alcoholic beverages while you are taking dantrolene. Alcohol can make the side effects from dantrolene worse.
- you should plan to avoid unnecessary or prolonged exposure to sunlight or tanning beds and wear protective clothing, sunglasses, and sunscreen (SPF 30+ or higher). Dantrolene may make your skin sensitive to sunlight.
Pregnancy
Dantrolene is an FDA pregnancy category C drug. For pregnant patients, it is permissible to use dantrolene in instances of malignant hyperthermia; in other instances, clinicians should consider alternatives, even though fetal harm is not expected based on limited human data.
Breastfeeding
Because no information is available on the long-term use of dantrolene during breastfeeding, an alternate drug may be preferred, especially while nursing a newborn or preterm infant. After short-term use, Dantrolene would be expected to be eliminated from milk in 1 to 2 days 5.
Administration of several intravenous dantrolene doses were given over 3 days to a postpartum mother in divided doses. Total daily dosages were 720 mg on day 1, 320 mg on day 2, and 80 mg on day 3. A milk level of 1.2 mg/L was measured on day 2 after a total of 640 mg had been given. The milk level was 50 mcg/L on day 4 about 35 hours after the last dose. Dantrolene half-life in milk was 9.2 hours 6.
Dantrolene drug interactions
Using dantrolene with other drugs that make you sleepy or slow your breathing can cause dangerous side effects or death. Ask your doctor before using opioid medication, a sleeping pill, a muscle relaxer, or medicine for anxiety or seizures.
Tell your doctor about all your other medicines, especially:
- heart or blood pressure medication;
- hormone replacement therapy; or
- hormonal birth control (pills, injections, implants, skin patches, or vaginal rings).
This list is not complete. Other drugs may affect dantrolene, including prescription and over-the-counter medicines, vitamins, and herbal products. Not all possible drug interactions are listed here.
Dantrolene and liver damage
The use of dantrolene is contraindicated in patients with active liver disease such as hepatitis and cirrhosis. Symptomatic hepatitis, occasionally fatal, has been reported at various dosage levels of dantrolene. However, the risk at dosages up to 400 mg/day appears to be much lower than that at 800 mg/day or more. Other risk factors may include female gender, age over 35 years, and use of other medications (e.g., estrogens). Both asymptomatic liver enzyme elevations as well as overt hepatitis have occurred following dantrolene exposures of varying duration, although the latter most frequently has been observed between the third and twelfth month of therapy. All patients treated with dantrolene should have liver function tests (ALT, AST, alkaline phosphatase, total bilirubin) performed at baseline and regularly thereafter. Discontinuation of therapy should be considered if liver enzymes are significantly elevated. The drug should be withdrawn promptly, however, if laboratory abnormalities are accompanied by symptoms of hepatic injury such as fever, rash, anorexia, nausea, vomiting, fatigue, right upper quadrant pain, dark urine, and jaundice. Following complete recovery, reinstitution of dantrolene therapy is generally not recommended except in those patients who clearly benefit from the medication. Any attempt to restart treatment should occur in the hospital with low initial dosages, gradual increases, and frequent monitoring.
Dantrolene contraindications
No contraindications exist to the use of IV dantrolene to treat malignant hyperthermia 7. Oral dantrolene is contraindicated in patients with underlying liver disease, including cirrhosis, non-alcoholic steatohepatitis, and hepatitis B or C infections 7.
Dantrolene mechanism of action
The precise mechanism of action of Dantrolene remains controversial – direct action on the calcium release channel RyR1 skeletal ryanodine receptor versus indirect via other molecular components of the sarcoplasmic reticulum are both proposed mechanisms 8, 9, 10, 11. Dantrolene is a lipid soluble diphenylhydantoin analogue that inhibits muscle contractions by decreasing the release of calcium from the sarcoplasmic reticulum in muscle tissue. Dantrolene acts intracellularly in skeletal muscle to lessen the excitation-contraction coupling interaction between actin and myosin within the individual sarcomere. This function occurs by antagonizing ryanodine receptors within the sarcoplasmic reticulum, which inhibits the release of calcium ions vital to the contraction process 12.
By decreasing the amount of calcium within each sarcomere, calcium cannot bind to the troponin on actin filaments; this prevents the uncovering of the myosin-binding site on the actin, preventing the actin and myosin cross-bridging from occurring, thus decreasing the contractibility and energy expenditure of the muscle cells 7.
Dantrolene is metabolized in the liver at least in part by the CYP450 enzyme system and is excreted in the urine. It has a half-life of between 4 and 8 hours.
Dantrolene uses
Dantrolene is in a class of medications called skeletal muscle relaxants. Dantrolene acts on the spinal cord nerves to treat muscle spasticity (muscle stiffness and tightness) caused by conditions such as a spinal cord injury, stroke, cerebral palsy, or multiple sclerosis.
Dantrolene is also used to treat or prevent muscle stiffness and spasms caused by malignant hyperthermia (a rapid rise in body temperature and severe muscle contractions), based upon its ability to block calcium release, which is the initiating event in malignant hyperthermia that can occur during surgery with certain types of anesthesia.
Dantrolene and malignant hyperthermia
Malignant hyperthermia is a serious reaction to the volatile halogenated anesthetics or depolarizing muscle relaxants used for anesthesia, causing sustained muscle contraction or spasms, dangerously high body temperature (hyperthermia), abnormally fast breathing, breakdown of muscle fibers (rhabdomyolysis), and increased acid levels in the blood and other tissues (acidosis), potentially leading to intraoperative patient death 13, 1. These symptoms develop as a response to the anesthetic agents acting on defective ryanodine receptors (calcium channels in muscle cell sarcoplasmic reticulum) 7. Without prompt treatment, the complications caused by malignant hyperthermia can be fatal. Treatments for malignant hyperthermia include the medication dantrolene, ice packs and other measures to cool body temperature, as well as supportive care.
In most cases, the gene that puts you at risk of malignant hyperthermia is inherited, usually from one parent who also has it. The most commonly affected gene is RyR1 14. These mutations render RyR1 leaky and more sensitive to its ligands. Thus, volatile anesthetics trigger abnormal calcium release in the resting muscle, which can be inhibited by dantrolene 15, 16. More rarely affected genes include CACNA1S and STAC3. Less often, the affected gene is not inherited and is the result of a random genetic change. Genetic testing can reveal whether you have an affected gene. This genetic disorder is called malignant hyperthermia susceptibility (MHS) 17.
Your risk of having the malignant hyperthermia susceptibility (MHS) is higher if someone in your family has it:
- You only need to inherit one changed gene from a parent to be affected by this disorder (autosomal dominant inheritance pattern). If one of your parents has the gene change that causes malignant hyperthermia susceptibility (MHS), you have a 50% chance of having malignant hyperthermia susceptibility (MHS) too.
- If you have other relatives with malignant hyperthermia susceptibility (MHS), your chance of having it is also increased.
Your risk of having malignant hyperthermia is also higher if you or a close relative has:
- A history of an event that is suspected to be malignant hyperthermia during anesthesia
- A history of muscle tissue breakdown called rhabdomyolysis, which can be triggered by exercise in extreme heat and humidity or when taking a statin drug
- Certain muscle diseases and disorders caused by inherited gene changes.
If you have a family history of malignant hyperthermia or a relative who has problems with anesthesia or malignant hyperthermia susceptibility (MHS), tell your health care provider or anesthesiologist before surgery or any procedure that requires anesthesia. Evaluating your risk of malignant hyperthermia allows your anesthesiologist to avoid certain anesthesia drugs that do not trigger malignant hyperthermia may be used as part of your anesthesia.
All inhalation anesthetics except nitrous oxide are triggers for malignant hyperthermia 18. The muscle relaxant succinylcholine is also a trigger for malignant hyperthermia. No other anesthetic drugs appear to be triggers, including propofol and ketamine 18. Neither are catecholamines, nondepolarizing muscle relaxants, catechol congeners, digitalis or similar agents 19.
In most malignant hyperthermia cases, no signs or symptoms of susceptibility to malignant hyperthermia exist until you’re exposed to certain drugs used for anesthesia.
Signs and symptoms of malignant hyperthermia may vary and can occur during anesthesia or during recovery shortly after surgery. They can include:
- Severe muscle rigidity or spasms
- Rapid, shallow breathing and problems with low oxygen and high carbon dioxide
- Rapid heart rate
- Irregular heart rhythm
- Dangerously high body temperature
- Excessive sweating
- Patchy, irregular skin color (mottled skin)
In rare cases, people at risk of malignant hyperthermia have shown signs of a reaction after intense physical activity during excessive heat or humidity, during a viral illness, or when taking statin medication used to lower cholesterol.
If you’re at risk of malignant hyperthermia and you don’t have a serious reaction during your first exposure to certain anesthesia drugs, you’re still at risk if you receive these drugs in the future. Other anesthesia drugs that don’t trigger a reaction can be used instead.
Malignant hyperthermia is diagnosed based on signs and symptoms, monitoring during and immediately after anesthesia, and lab tests to identify complications.
- Susceptibility testing. Testing to find out if you’re at increased risk of malignant hyperthermia (susceptibility testing) may be recommended if you have risk factors. Testing may include genetic testing or a muscle biopsy test.
- Genetic testing. The gene change (mutation) that makes you susceptible to malignant hyperthermia is identified using genetic testing. A sample of your blood is collected and sent to a lab for analysis. Genetic testing can identify the gene change that shows you have the genetic disorder called malignant hyperthermia susceptibility (MHS).
- Muscle biopsy (contracture test). In some cases, your health care provider may recommend a muscle biopsy if you’re at risk of malignant hyperthermia. During this test, a small piece of muscle tissue is surgically removed for lab analysis. In the lab, the specimen is exposed to triggering chemicals for malignant hyperthermia to determine how the muscle contracts. Because this test must be done on muscle tissue immediately after removing it, travel to a specialized muscle biopsy center is necessary.
Immediate treatment of malignant hyperthermia includes:
- Dantrolene (Dantrium, Revonto, Ryanodex) is used to treat the reaction by stopping the release of calcium into muscles. Administer dantrolene 2.5 mg/kg until the reaction subsides up to a maximum of 10 mg/kg 13, 20. Other medications may be given to correct problems with a balance of the body’s chemicals (metabolic imbalance) and treat complications.
- Oxygen. You may have oxygen through a face mask. In most cases, oxygen is given through a tube placed in the windpipe (trachea).
- Body cooling. Ice packs, cooling blankets, a fan with cool mist and chilled intravenous (IV) fluids may be used to help reduce body temperature.
- Extra fluids. You may also get extra fluids through an intravenous (IV) line.
- Supportive care. You may need to stay in the hospital in intensive care for a day or two to monitor your temperature, blood pressure, heart rate, breathing and response to treatment. Several lab tests will be done frequently to check the extent of any muscle breakdown and possible kidney damage. A stay in the hospital is usually needed until lab test results start to return to a standard range.
With treatment, malignant hyperthermia usually resolves within a few days. Complete recovery can occur if the signs and symptoms of malignant hyperthermia are recognized early, rapid treatment with dantrolene, and prevention of the rapid rise in core temperature by using cooling measures 21. Multiple organ failure and death can still occur, however, even with prompt treatment. The mortality rate is less than 5% 13.
The complications of malignant hyperthermia include 13:
- Brain damage
- Cardiac arrest
- Death
- Heart failure
- Internal bleeding
- Kidney failure
- Pulmonary edema
- Skeletal muscle degeneration
After the patient has been stabilized, they must be taken to the intensive care unit (ICU) for at least 24 hours for monitoring and to watch for signs of recrudescence. Patients at the highest risk for recurrence are those with a large muscle mass or who have undergone at least 150 minutes of anesthetic exposure before triggering 13.
Follow-up care
If you’ve experienced malignant hyperthermia due to certain anesthesia drugs, exercising during excessive heat and humidity could trigger another reaction. Talk to your health care provider about any precautions you should take.
Also, check with your health care provider to see if you should have genetic testing to determine if you have a genetic disorder that puts you at risk of malignant hyperthermia. Ask if close family members should also consider genetic testing.
If you have the genetic disorder called malignant hyperthermia susceptibility (MHS) that puts you at risk of malignant hyperthermia, wear a medical alert bracelet or necklace. This lets health care providers know of your risk, especially in an emergency, when you may not be able to talk.
Dantrolene dosage
Dantrolene is available as an intravenous injection and an oral capsule. The choice of the method of administration is dependent on the intended use of Dantrolene.
It is important to note that multiple formulations exist for IV dantrolene. Dantrolene formulations differ in the amount of sterile water required to reconstitute each vial and the dantrolene concentration within each vial. One formulation is available in 20 mg vials that must be reconstituted with 60 mL of sterile water per vial, while the other is available in 250 mg vials that must be reconstituted with 5 mL of sterile water per vial. Regardless of which formulation of dantrolene is administered, a dose of 2.5 mg/kg is recommended to treat a malignant hyperthermia episode. Dantrolene is a highly lipophilic drug with low solubility in water, making the reconstitution of the drug in sterile water challenging. The newer formulation that combines nanosuspension technology with a lyophilized formulation resulting in a much faster reconstitution of the drug, which can greatly improve the time to treatment and may be especially useful in locations where only one provider is available to treat the patient.
Keep in mind that additional doses of dantrolene might be necessary to treat malignant hyperthermia triggering events adequately; a dose of 1 mg/kg every 4 to 6 hours is recommended for the first 24 to 48 hours after an episode of malignant hyperthermia.
Dantrolene should not be combined with verapamil, as it may lead to hyperkalemia and hypotension 18.
All facilities where malignant hyperthermia triggering anesthetics are administered are recommended to stock an adequate amount of dantrolene and other medications and rescue equipment needed to treat a malignant hyperthermia crisis.
Malignant hyperthermia treatment
When symptoms of malignant hyperthermia present, 2.5 mg/kg dantrolene via intravenous push should be administered immediately. If the signs and symptoms of malignant hyperthermia persist, additional intravenous boluses of 1 to 2.5 mg/kg are indicated to a maximum cumulative dose of 10 mg/kg. Following the successful treatment of the initial reaction, 1 mg/kg of IV dantrolene should be provided every 6 hours for 24 hours since the last observed symptom of malignant hyperthermia to prevent a recurrence 22. At any point during the treatment of malignant hyperthermia, contacting the Malignant Hyperthermia Association of the United States is appropriate for dantrolene dosing recommendations and ongoing treatment 23.
Malignant hyperthermia prevention
Dantrolene 2.5 mg/kg intravenously over one minute, approximately 75 minutes before surgery. Repeat doses can be given during anesthesia and surgery in prolonged cases. Although this is an approved indication for using dantrolene, it is unnecessary if patients avoid triggering agents when they are susceptible to malignant hyperthermia. The recommended weight-based dose is the same for adults and children.
Chronic Muscle Spasms
Generally, Dantrolene 100 mg orally three or four times daily. Start oral dantrolene at 25 mg daily for 7 days is the initial dosing, and the dose should be titrated to the maximum individual effect. Doses are typically increased by 25 mg at a time and require monitoring for seven days before further advancement. The maximum Dantrolene dose is 400 mg/day. For children 5 years of age and older, Dantrolene divided into three or four doses; start at 0.5 mg/kg/dose orally each day for seven days and increase to 0.5 mg/kg/dose orally three times a day for seven days, then 1 mg/kg/dose three times daily for seven days, and then 2 mg/kg/dose orally three times daily. The maximum dose is 100 mg orally four times daily, but clinicians need to use the lowest effective dose.
What should I do if I forget a dose?
Take the missed dose as soon as you remember it. However, if it is almost time for the next dose, skip the missed dose and continue your regular dosing schedule. Do not take a double dose to make up for the missed one.
Dantrolene side effects
Dantrolene may cause side effects. Tell your doctor if any of these symptoms are severe or do not go away:
- muscle weakness
- dizziness
- diarrhea
- fatigue
In addition to the symptoms mentioned in the IMPORTANT WARNING section, if you experience and of the following allergic reaction symptoms, see your doctor immediately:
- seizures
- hives
- swelling of your face, lips, tongue, or throat
- difficulty breathing or slow, shallow breathing.
See your doctor right away if you have signs of liver problems such as nausea, upper stomach pain, itching, tired feeling, loss of appetite, dark urine, clay-colored stools, or jaundice (yellowing of the skin or eyes). These side effects may be most likely to occur between 3 and 12 months of your treatment with dantrolene.
Dantrolene overdose
Dantrolene overdose symptoms may include:
- vomiting
- diarrhea
- muscle weakness
- extreme tiredness
- coma
No reversal drug or antidote exists for IV or oral dantrolene. For liver damage induced by oral dantrolene, discontinue the medication, and initiate symptomatic therapy.
- Malignant hyperthermia. https://medlineplus.gov/genetics/condition/malignant-hyperthermia[↩][↩]
- Ward A, Chaffman MO, Sorkin EM. Dantrolene. A review of its pharmacodynamic and pharmacokinetic properties and therapeutic use in malignant hyperthermia, the neuroleptic malignant syndrome and an update of its use in muscle spasticity. Drugs. 1986 Aug;32(2):130-68. doi: 10.2165/00003495-198632020-00003[↩]
- Amano T, Fukami T, Ogiso T, Hirose D, Jones JP, Taniguchi T, Nakajima M. Identification of enzymes responsible for dantrolene metabolism in the human liver: A clue to uncover the cause of liver injury. Biochem Pharmacol. 2018 May;151:69-78. doi: 10.1016/j.bcp.2018.03.002[↩]
- LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Dantrolene. [Updated 2017 Jan 30]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK548144[↩]
- Drugs and Lactation Database (LactMed®) [Internet]. Bethesda (MD): National Institute of Child Health and Human Development; 2006-. Dantrolene. [Updated 2021 Mar 17]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK501309[↩]
- Fricker RM, Hoerauf KH, Drewe J, Kress HG. Secretion of dantrolene into breast milk after acute therapy of a suspected malignant hyperthermia crisis during cesarean section. Anesthesiology. 1998 Oct;89(4):1023-5. doi: 10.1097/00000542-199810000-00028[↩]
- Ratto D, Joyner RW. Dantrolene. [Updated 2023 Apr 3]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK535398[↩][↩][↩][↩]
- Szentandrássy N., Magyar Z.É., Hevesi J., Bányász T., Nánási P.P., Almássy J. Therapeutic approaches of ryanodine receptor-associated heart diseases. Int. J. Mol. Sci. 2022;23:4435. doi: 10.3390/ijms23084435[↩]
- Saljic A., Heijman J., Dobrev D. Emerging antiarrhythmic drugs for atrial fibrillation. Int. J. Mol. Sci. 2022;23:4096. doi: 10.3390/ijms23084096[↩]
- Ellis KO, Castellion AW, Honkomp LJ, Wessels FL, Carpenter JE, Halliday RP. Dantrolene, a direct acting skeletal muscle relaxant. J Pharm Sci. 1973 Jun;62(6):948-51. doi: 10.1002/jps.2600620619[↩]
- Hopkins PM, Gupta PK, Bilmen JG. Malignant hyperthermia. Handb Clin Neurol. 2018;157:645-661. doi: 10.1016/B978-0-444-64074-1.00038-0[↩]
- Zucchi R, Ronca-Testoni S. The sarcoplasmic reticulum Ca2+ channel/ryanodine receptor: modulation by endogenous effectors, drugs and disease states. Pharmacol Rev. 1997 Mar;49(1):1-51. https://pharmrev.aspetjournals.org/content/49/1/1.long[↩]
- Watt S, McAllister RK. Malignant Hyperthermia. [Updated 2023 Aug 17]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430828[↩][↩][↩][↩][↩]
- Riazi S., Kraeva N., Hopkins P.M. Malignant Hyperthermia in the Post-Genomics Era: New Perspectives on an Old Concept. Anesthesiology. 2018;128:168–180. doi: 10.1097/ALN.0000000000001878[↩]
- MacLennan D.H., Phillips M.S. Malignant hyperthermia. Science. 1992;256:789–794. doi: 10.1126/science.256.5058.789[↩]
- Yuchi Z., Lau K., Van Petegem F. Disease mutations in the ryanodine receptor central region: Crystal structures of a phosphorylation hot spot domain. Structure. 2012;20:1201–1211. doi: 10.1016/j.str.2012.04.015[↩]
- Rosenberg H, Sambuughin N, Riazi S, et al. Malignant Hyperthermia Susceptibility. 2003 Dec 19 [Updated 2020 Jan 16]. In: Adam MP, Feldman J, Mirzaa GM, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1146[↩]
- Rosenberg H, Pollock N, Schiemann A, Bulger T, Stowell K. Malignant hyperthermia: a review. Orphanet J Rare Dis. 2015 Aug 4;10:93. doi: 10.1186/s13023-015-0310-1[↩][↩][↩]
- Hopkins PM. Malignant hyperthermia: advances in clinical management and diagnosis. Br J Anaesth. 2000 Jul;85(1):118-28. Erratum in: Br J Anaesth 2001 Apr;86(4):605. https://doi.org/10.1093/bja/85.1.118[↩]
- Rosenberg H, Sambuughin N, Riazi S, et al. Malignant Hyperthermia Susceptibility. 2003 Dec 19 [Updated 2020 Jan 16]. In: Adam MP, Feldman J, Mirzaa GM, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2023. Figure 2. [MHAUS treatment guide for malignant hyperthermia]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1146/figure/mhs.F2[↩]
- Larach MG, Brandom BW, Allen GC, Gronert GA, Lehman EB. Malignant hyperthermia deaths related to inadequate temperature monitoring, 2007-2012: a report from the North American malignant hyperthermia registry of the malignant hyperthermia association of the United States. Anesth Analg. 2014 Dec;119(6):1359-66. doi: 10.1213/ANE.0000000000000421[↩]
- Burkman JM, Posner KL, Domino KB. Analysis of the clinical variables associated with recrudescence after malignant hyperthermia reactions. Anesthesiology. 2007 May;106(5):901-6; quiz 1077-8. doi: 10.1097/01.anes.0000265148.86566.68[↩]
- Riazi S, Kraeva N, Hopkins PM. Updated guide for the management of malignant hyperthermia. Can J Anaesth. 2018 Jun;65(6):709-721. English. doi: 10.1007/s12630-018-1108-0[↩]