Contents
- Lung cancer
- Types of lung cancer
- Chest anatomy
- Human lungs
- What are Lung Nodules?
- Lung cancer signs and symptoms
- Lung cancer causes
- Lifetime chance of getting lung cancer
- Lung cancer prevention
- Lung cancer screening
- Lung cancer complications
- Lung cancer diagnosis
- Lung cancer staging
- Lung cancer treatment
- Lung cancer prognosis
- Non-Small Cell Lung Cancer
- Types of non-small cell lung cancer
- Histopathology of non-small cell lung cancer
- Non-small cell lung cancer causes
- Non-small cell lung cancer prevention
- Non-small cell lung cancer symptoms and signs
- Non-small cell lung cancer diagnosis
- Non-Small Cell Lung Cancer Stages
- Non-Small Cell Lung Cancer Survival Rates by Stage
- Non-small cell lung cancer treatment
- Surgery
- Radiation therapy
- Chemotherapy
- Targeted therapy drugs
- Drugs that target tumor blood vessel growth (Angiogenesis inhibitors)
- Drugs that target cells with EGFR changes (EGFR inhibitors)
- Drugs that target cells with ALK gene changes (ALK inhibitors)
- Drugs that target cells with ROS1 gene changes (ROS1 inhibitors)
- Drugs that target cells with BRAF gene changes (BRAF inhibitors)
- Drugs that target cells with RET gene changes (RET inhibitors)
- Drugs that target cells with MET gene changes (MET inhibitors)
- Drugs that target cells with HER2 gene changes (HER2 inhibitors)
- Drugs that target cells with TRK gene changes (TRK inhibitors)
- Radiofrequency Ablation
- Tumor Treating Fields Therapy
- Immunotherapy for Non-Small Cell Lung Cancer
- Maintenance therapy
- Palliative Procedures for Non-Small Cell Lung Cancer
- Small cell lung cancer
- Small Cell Lung Cancer causes
- Small Cell Lung Cancer Risk Factors
- Small Cell Lung Cancer prevention
- Small Cell Lung Cancer signs and symptoms
- Small Cell Lung Cancer Diagnosis
- Small Cell Lung Cancer Stages
- Small Cell Lung Cancer Survival Rates by Stage
- Small Cell Lung Cancer Treatment
- Small cell lung cancer prognosis
Lung cancer
Lung cancer is a type of cancer that starts in the windpipe (trachea), the main airway (bronchus) or the lung tissue. Most lung cancer are either small cell lung cancer (SCLC) or non-small cell lung cancer (NSCLC). In general, about 13% of all lung cancers are small cell lung cancer (SCLC) and about 87% are non-small cell lung cancer (NSCLC) 1. Lung cancer is one of the most common cancers in the world. Lung cancer is the second most common cancer (not counting skin cancer) and the leading cause of cancer death in the United States 2. In men, prostate cancer is more common, while in women breast cancer is more common 3. About 14% of all new cancers are lung cancers. The most important risk factor and cause for lung cancer is smoking, which results in approximately 85% of all U.S. lung cancer cases 4. The more cigarettes you smoke per day and the earlier you started smoking, the greater your risk of lung cancer. Although the prevalence of smoking has decreased, approximately 37% of U.S. adults are current or former smokers 4. The incidence of lung cancer increases with age and occurs most commonly in persons aged 55 years or older. Increasing age and cumulative exposure to tobacco smoke are the 2 most common risk factors for lung cancer. High levels of pollution, radiation and asbestos exposure may also increase risk.
Lung cancers typically start in the cells lining the bronchi and parts of the lung such as the bronchioles or alveoli. Lung cancer has a poor prognosis, and nearly 90% of persons with lung cancer die of the disease.
There are 2 main types of lung cancer and they are treated very differently:
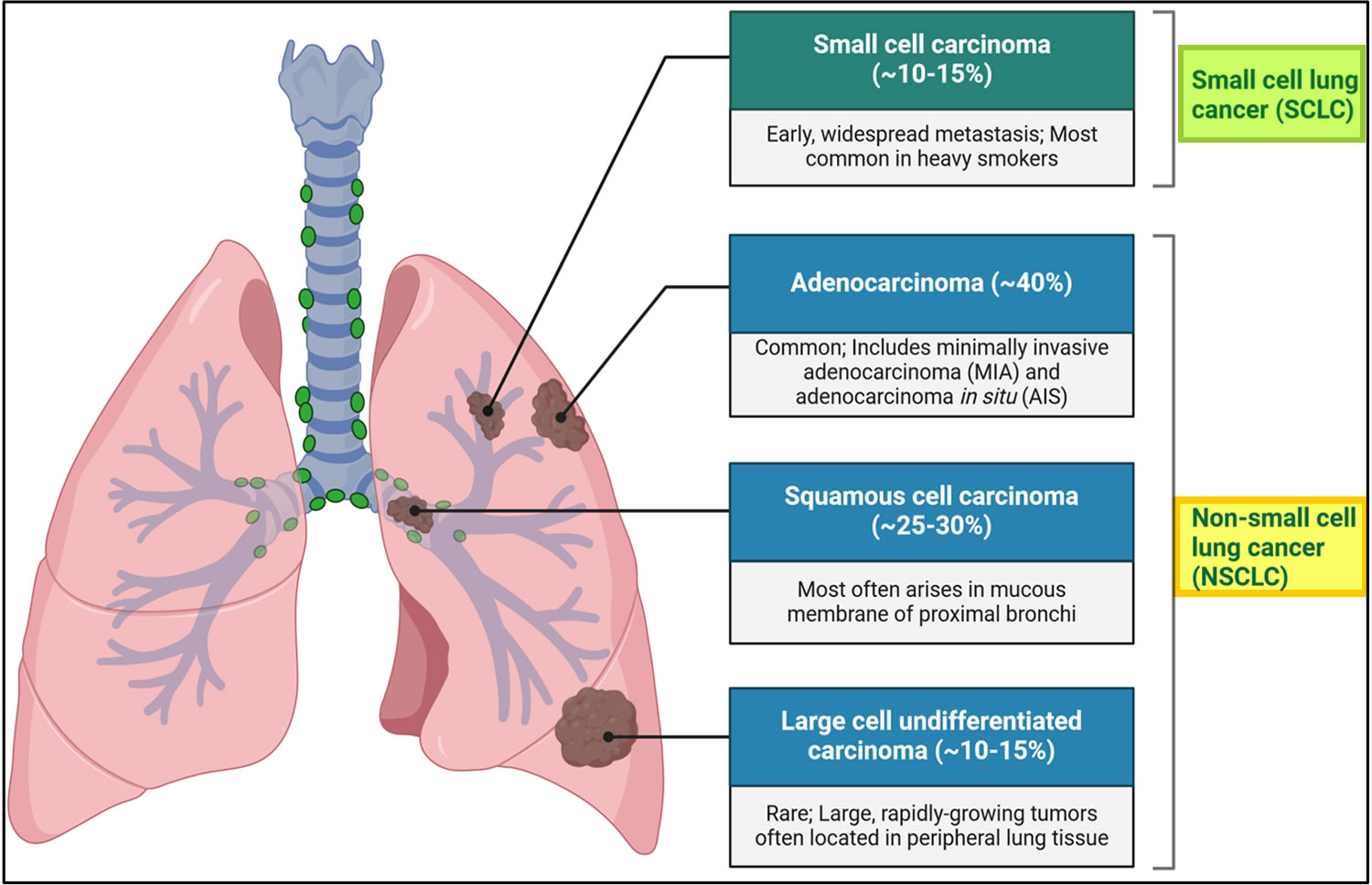
- Non small cell lung cancer (NSCLC). Non-small cell lung cancer is an umbrella term for several types of lung cancers. About 80% to 85% of lung cancers are non small cell lung cancer (NSCLC). The main subtypes of non small cell lung cancer are adenocarcinoma, squamous cell carcinoma, and large cell carcinoma. These subtypes, which start from different types of lung cells are grouped together as non small cell lung cancer because their treatment and prognoses (outlook) are often similar.
- Small cell lung cancer (SCLC) also called oat cell cancer. About 10% to 15% of all lung cancers are small cell lung cancer (SCLC). Small cell lung cancer occurs almost exclusively in heavy smokers. Small cell lung cancer tends to grow and spread faster than non small cell lung cancer. About 70% of people with small cell lung cancer will have cancer that has already spread at the time they are diagnosed. Since this cancer grows quickly, it tends to respond well to chemotherapy and radiation therapy. Unfortunately, for most people, the cancer will return at some point.
- Other types of lung tumors. Along with the main types of lung cancer, other tumors can occur in the lungs.
- Lung carcinoid tumors also known as lung carcinoids: Carcinoid tumors of the lung account for fewer than 5% of lung tumors. Most of these grow slowly. Lung carcinoid tumors start in neuroendocrine cells, a special kind of cell found in the lungs. Neuroendocrine cells are also found in other areas of the body, but only cancers that form from neuroendocrine cells in the lungs are called lung carcinoid tumors.
- There are 2 types of lung carcinoid tumors 5:
- Typical carcinoids tend to grow slowly and rarely spread beyond the lungs. About 9 out of 10 lung carcinoids are typical carcinoids. They also do not seem to be linked with smoking.
- Atypical carcinoids grow a little faster and are somewhat more likely to spread to other organs. They have more cells that are dividing and look more like a fast-growing tumor. They are much less common than typical carcinoids and may be found more often in people who smoke.
- There are 2 types of lung carcinoid tumors 5:
- Other types of lung cancer such as adenoid cystic carcinomas, lymphomas, and sarcomas, as well as benign lung tumors such as hamartomas are rare.
- Cancers that spread to the lungs also known as secondary lung cancer: Cancers that start in other organs (such as the breast, pancreas, kidney, or skin) can sometimes spread (metastasize) to the lungs, but these are not lung cancers. For example, cancer that starts in the breast and spreads to the lungs is still breast cancer, not lung cancer. Treatment for metastatic cancer to the lungs is based on where it started (the primary cancer site).
- Lung carcinoid tumors also known as lung carcinoids: Carcinoid tumors of the lung account for fewer than 5% of lung tumors. Most of these grow slowly. Lung carcinoid tumors start in neuroendocrine cells, a special kind of cell found in the lungs. Neuroendocrine cells are also found in other areas of the body, but only cancers that form from neuroendocrine cells in the lungs are called lung carcinoid tumors.
Cancer that starts in the lung is called primary lung cancer. If cancer spreads to your lungs from somewhere else in your body, this is secondary lung cancer.
Figure 1. Lung cancer types
Footnotes: Overview of non-small cell lung cancer types of cancers that develop from the lung’s epithelial cells, with three main subtypes: adenocarcinoma, squamous cell carcinoma, and large cell carcinoma.
[Source 6 ]People who smoke have the greatest risk of lung cancer 7, though lung cancer can also occur in people who have never smoked. The risk of lung cancer increases with the length of time and number of cigarettes you’ve smoked. Smoking is estimated to account for about 90% of all lung cancer cases 8, with a relative risk of lung cancer approximately 20-fold higher in smokers than in nonsmokers 7. If you quit smoking, even after smoking for many years, you can significantly reduce your chances of developing lung cancer.
Estimated new cases and deaths from lung cancer (non–small cell lung cancer and small cell lung cancer combined) in the United States in 2025 8, 8, 1:
- New cases: About 226,650 new cases of lung cancer (110,680 in men and 115,970 in women). The number of new lung cancer cases continues to decrease, partly because people are quitting smoking 9. However, in developing countries, non-small cell lung cancer cases are rising due to higher smoking rates and growing industrialization 6.
- Deaths: About 124,730 deaths from lung cancer (64,190 in men and 60,540 in women). Death rates for lung cancer are higher among the middle-aged and older populations. Lung and bronchus cancer is the first leading cause of cancer death in the United States. The death rate was 32.4 per 100,000 men and women per year based on 2018–2022 deaths, age-adjusted.
- 5-Year Relative Survival: 26.7%. Relative survival is an estimate of the percentage of patients who would be expected to survive the effects of their cancer. It excludes the risk of dying from other causes. Because survival statistics are based on large groups of people, they cannot be used to predict exactly what will happen to an individual patient. No two patients are entirely alike, and treatment and responses to treatment can vary greatly.
- Percentage of All Cancer Deaths: 20.4%. Lung cancer is by far the leading cause of cancer death, making up almost 20.4% of all cancer deaths. Each year, more people die of lung cancer than of colon, breast, and prostate cancers combined.
- The percent of lung and bronchus cancer deaths is highest among people aged 65–74. With the Median Age At Death 72 years of age.
- Rate of New Lung Cancer Cases and Deaths per 100,000: The rate of new cases of lung and bronchus cancer was 49 per 100,000 men and women per year. The death rate was 32.4 per 100,000 men and women per year. These rates are age-adjusted and based on 2017–2021 cases and 2018–2022 deaths.
- Lifetime Risk of Developing Lung Cancer: Approximately 5.7 percent of men and women will be diagnosed with lung and bronchus cancer at some point during their lifetime, based on 2018–2021 data.
- Prevalence of Lung Cancer: In 2021, there were an estimated 610,816 people living with lung and bronchus cancer in the United States.
Lung cancer mainly occurs in older people. About 2 out of 3 people diagnosed with lung cancer are 65 or older, while less than 2% are younger than 45. The average age at the time of diagnosis is about 70 3.
The 5-year relative survival rate from 2014 to 2020 for patients with lung cancer was 26.7% 8. The 5-year relative survival rate for patients with local-stage (63.7%), regional-stage (35.9%), and distant-stage (8.9%) disease varies markedly, depending on the stage at diagnosis 8. However, early-stage non–small cell lung cancer (NSCLC) has a better prognosis and can be treated with surgical resection.
The type of lung cancer you have tells you the type of cell that the cancer started in. Knowing this helps your doctor decide which treatment you need.
Lung cancer typically doesn’t cause signs and symptoms in its earliest stages. Signs and symptoms of lung cancer typically occur when the disease is advanced. Common symptoms of lung cancer may include 10:
- A cough that doesn’t go away and gets worse over time
- Constant chest pain
- Coughing up blood, even a small amount
- Shortness of breath or wheezing
- Hoarseness of voice
- Repeated problems with pneumonia or bronchitis
- Frequent chest infections
- Swelling of the neck and face
- Difficulty swallowing
- Loss of appetite
- Weight loss without trying
- Fatigue
- Bone pain
- Headache
- Some people have swollen fingers and nails called finger clubbing. They may also have pain and swelling in their joints. This condition is called hypertrophic pulmonary osteoarthropathy (HPOA).
You should see your doctor right away if you have new symptoms that concern you, especially those listed above. This is particularly important if you have risk factors for lung cancer such as a history of lung disease, a family history of lung cancer or you are a smoker.
Doctors diagnose lung cancer using a physical exam, imaging, and lab tests. Treatment depends on the type, stage, and how advanced it is. Treatments include surgery, chemotherapy, radiation therapy, and targeted therapy. Targeted therapy uses substances that attack cancer cells without harming normal cells. Together, you and your medical team can create a cancer treatment plan that contains the goals of your cancer treatment and the steps that these involve.
Treatment for lung cancer usually begins with surgery to remove the cancer. If your lung cancer is very large or has spread to other parts of your body, surgery may not be possible. Treatment might start with medicine and radiation instead. Your medical team considers many factors when creating a treatment plan. These factors may include your overall health, the type and stage of your cancer, and your preferences.
Some people with lung cancer choose not to have treatment. For instance, you may feel that the side effects of treatment will outweigh the potential benefits. When that’s the case, your doctor may suggest comfort care to treat only the symptoms the cancer is causing.
The prognosis of lung cancer depends on how advanced it was at diagnosis (staging), the effectiveness of cancer treatment and your general health. Your doctor can discuss realistic expectations for your own prognosis.
Types of lung cancer
There are 2 main types of lung cancer 11. Knowing which type you have is important because it affects your treatment options and your outlook (prognosis). If you aren’t sure which type of lung cancer you have, ask your doctor so you can get the right information.
- Non small cell lung cancer (NSCLC). About 80% to 85% of lung cancers are non-small cell lung cancer (NSCLC). Non-small cell lung cancer is an umbrella term for several types of lung cancers. The main subtypes of non-small cell lung cancer (NSCLC) are adenocarcinoma, squamous cell carcinoma, and large cell carcinoma. These subtypes, which start from different types of lung cells are grouped together as non-small cell lung cancer (NSCLC) because their treatment and prognoses (outlook) are often similar.
- Small cell lung cancer (SCLC) also known as oat cell cancer. About 10% to 15% are small cell lung cancer (SCLC). Small cell lung cancers are also classed as neuroendocrine tumors. Neuroendocrine tumors (NETs) are rare tumors that develop in cells of the neuroendocrine system. In small cell lung cancer, the tumor starts in the neuroendocrine cells of the lung. Small cell lung cancer (SCLC) tends to grow and spread faster than non-small cell lung cancer (NSCLC). About 70% of people with small cell lung cancer (SCLC) will have cancer that has already spread at the time they are diagnosed. Since this cancer grows quickly, it tends to respond well to chemotherapy and radiation therapy. Unfortunately, for most people, the cancer will return at some point.
Along with the 2 main types of lung cancer (non-small cell lung cancer and small cell lung cancer), other tumors can occur in the lungs.
- Lung carcinoid tumors also known as lung carcinoids: Carcinoid tumors of the lung account for fewer than 5% of lung tumors. Most of these grow slowly. Lung carcinoid tumors start in neuroendocrine cells, a special kind of cell found in the lungs. Neuroendocrine cells are also found in other areas of the body, but only cancers that form from neuroendocrine cells in the lungs are called lung carcinoid tumors.
- There are 2 types of lung carcinoid tumors 5:
- Typical carcinoids tend to grow slowly and rarely spread beyond the lungs. About 9 out of 10 lung carcinoids are typical carcinoids. They also do not seem to be linked with smoking.
- Atypical carcinoids grow a little faster and are somewhat more likely to spread to other organs. They have more cells that are dividing and look more like a fast-growing tumor. They are much less common than typical carcinoids and may be found more often in people who smoke.
- There are 2 types of lung carcinoid tumors 5:
- Other types of lung cancer such as adenoid cystic carcinomas, lymphomas, and sarcomas, as well as benign lung tumors such as hamartomas are rare. These are treated differently from the more common lung cancers.
- Cancers that spread to the lungs: Cancers that start in other organs (such as the breast, pancreas, kidney, or skin) can sometimes spread (metastasize) to the lungs, but these are not lung cancers. For example, cancer that starts in the breast and spreads to the lungs is still breast cancer, not lung cancer. Treatment for metastatic cancer to the lungs is based on where it started (the primary cancer site).
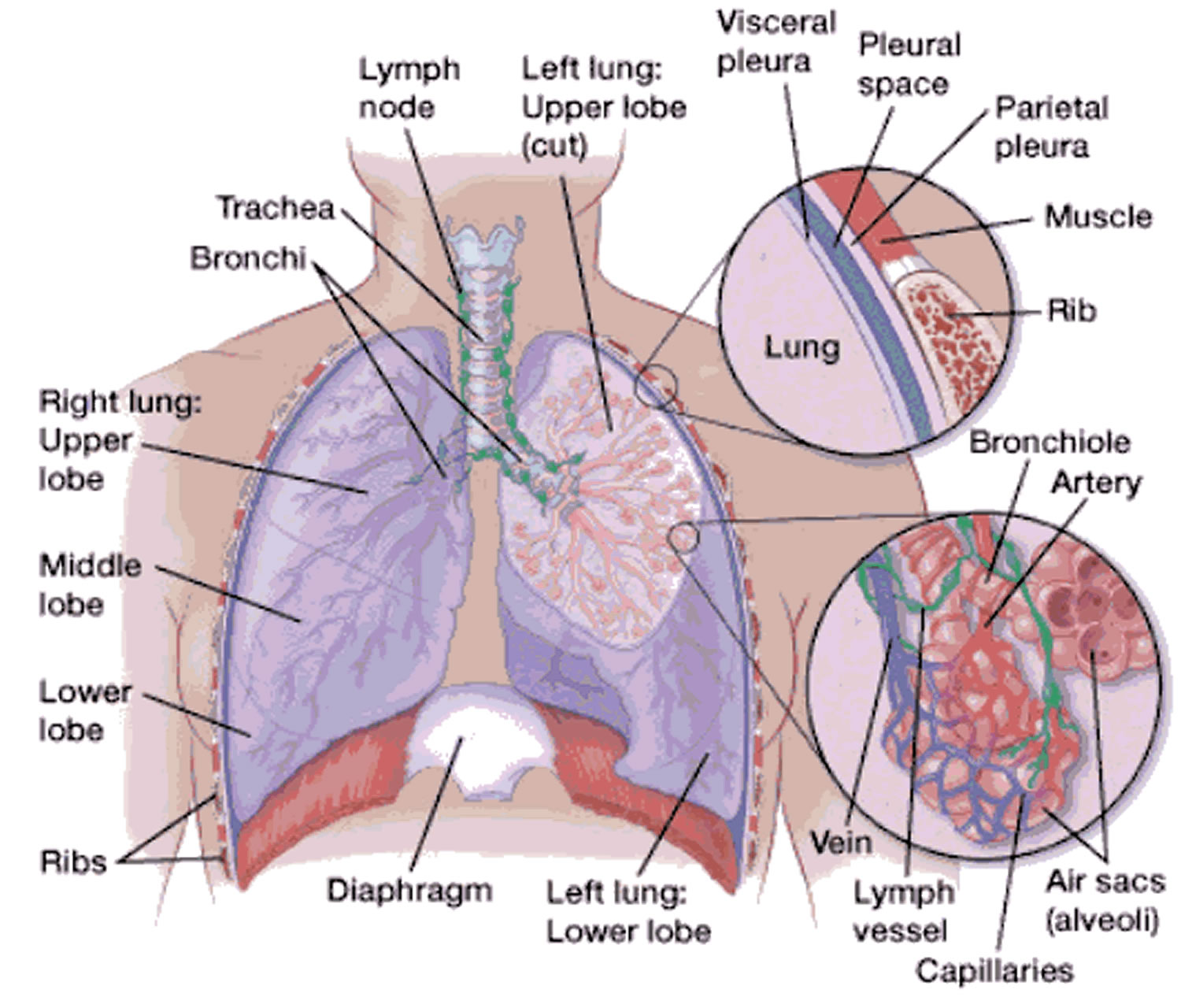
Chest anatomy
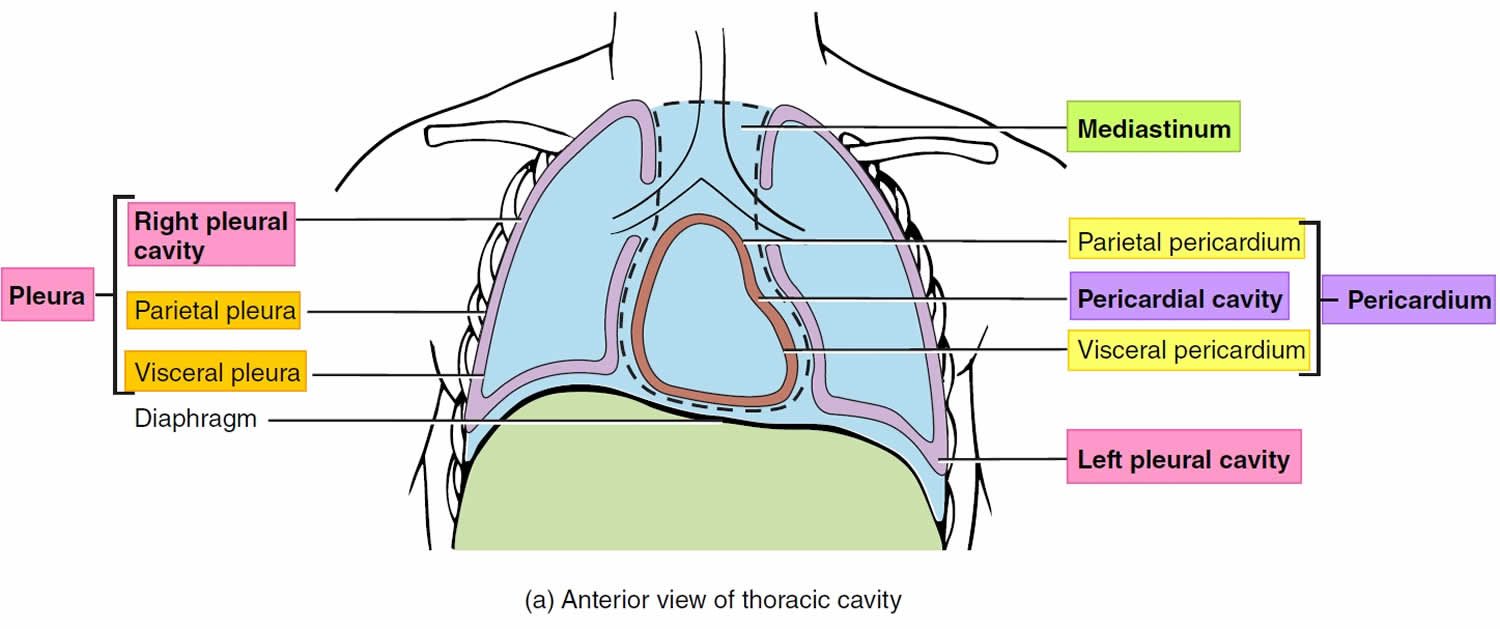
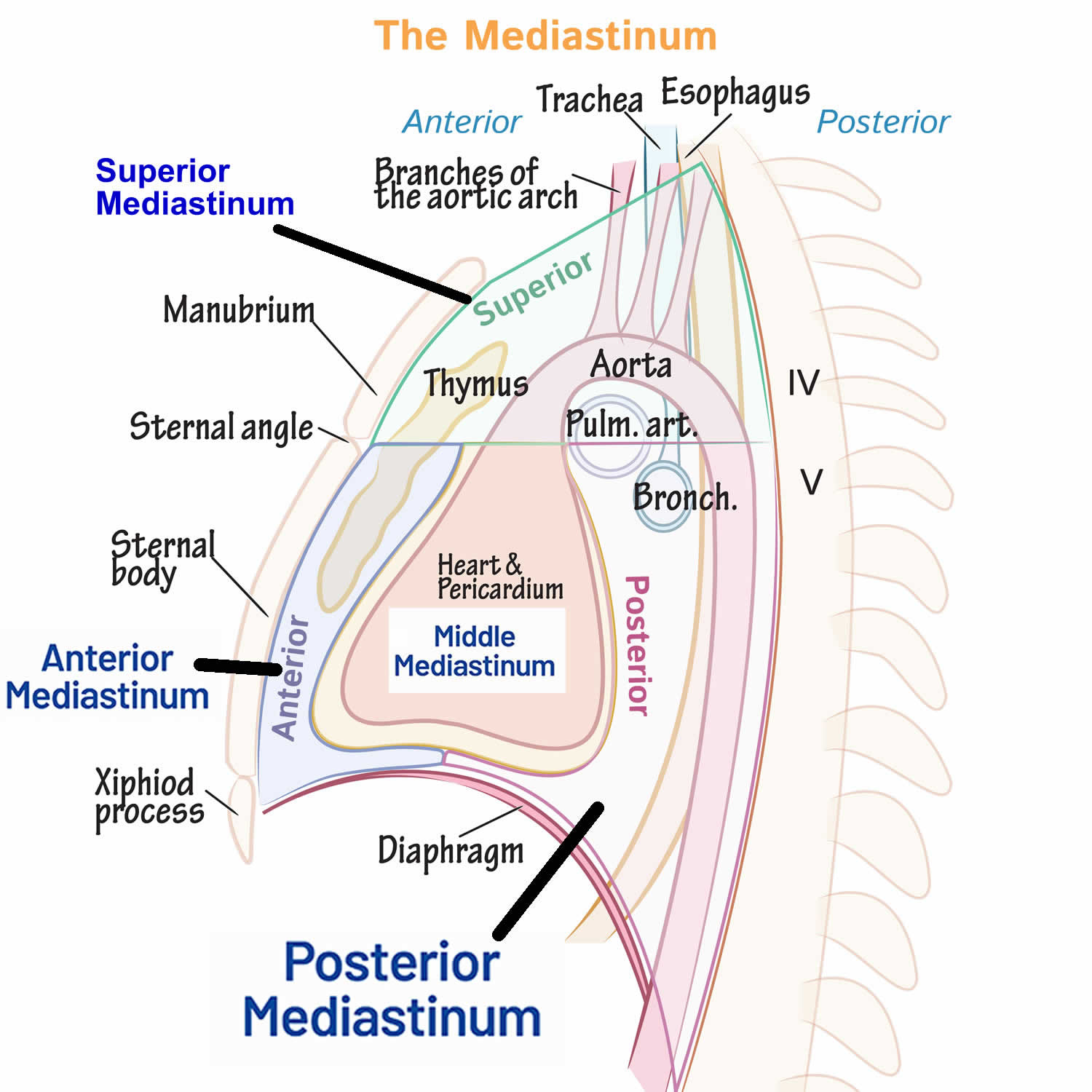
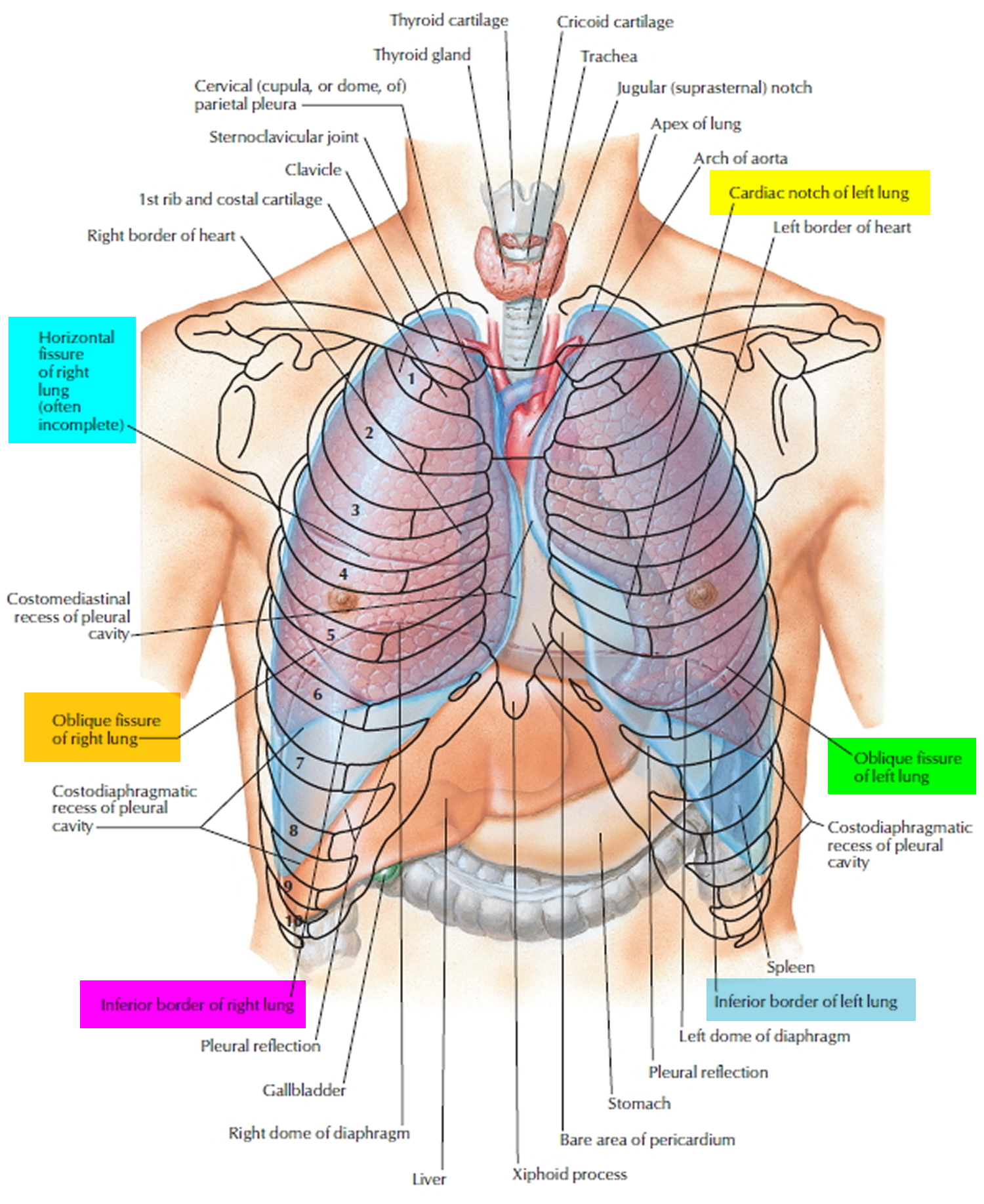
Your chest cavity also called the thoracic cavity is formed by the ribs, the muscles of the chest, the sternum (breastbone), and the thoracic portion of the vertebral column. Within your thoracic cavity are 3 smaller cavities: (a) 2 pleural cavities (fluid-filled spaces one around each lung), your left pleural cavity (holds your left lung) and your right pleural cavity (holds your right lung) and (b) a central portion of your thoracic cavity between your lungs called the mediastinum (media- = middle; -stinum = partition). The mediastinum is the central portion of your thoracic cavity between your lungs, extending from the base of your neck (from your first rib and sternum) to the diaphragm. The mediastinum contains your heart (pericardial cavity, peri- = around; -cardial = heart, a fluid-filled space that surrounds your heart), the major blood vessels connected to your heart and lungs, the trachea (windpipe) and bronchi, the esophagus (foodpipe), the thymus, and lymph nodes but not your lungs. Your right and left lungs are on either side of the mediastinum. The diaphragm is a dome-shaped muscle that separates the thoracic cavity from the abdominopelvic cavity.
Your mediastinum is divided into several parts, which researchers call compartments. The traditional or classical model divides your mediastinum into four parts:
- Superior mediastinum: The top part, located superior to (above) your heart.
- Anterior mediastinum: The part anterior to (in front of) your heart, between your heart and your sternum (breastbone).
- Middle mediastinum: The part that contains your heart.
- Posterior mediastinum: The part posterior to (behind) your heart.
A membrane is a thin, pliable tissue that covers, lines, partitions, or connects internal organs (viscera). One example is a slippery, double-layered membrane associated with body cavities that does not open directly to the exterior called a serous membrane. Serous membrane covers your internal organs (viscera) within the thoracic and abdominal cavities and also lines the walls of the thorax and abdomen. The parts of a serous membrane are (1) the parietal layer (outer layer), a thin epithelium that lines the walls of the cavities, and (2) the visceral layer (inner layer), a thin epithelium that covers and adheres to the viscera within the cavities. Between the two layers is a potential space that contains a small amount of lubricating fluid (serous fluid). The fluid allows the internal organs (viscera) to slide somewhat during movements, such as when the lungs inflate and deflate during breathing.
Within the right and left sides of your thoracic cavity (chest cavity), the compartments that contain your lungs, on either side of the mediastinum, are lined with a membrane called the parietal pleura (outer serous membrane) lining the inside of your rib cage (parietal pleura lines the chest wall) and covering the superior surface of the diaphragm. A similar membrane, called the visceral pleura (inner serous membrane), clings to the surface of your lungs forming the external surface of your lung. The visceral (inner) and parietal (outer) pleural membranes are separated only by a thin film of watery fluid called serous fluid, which is secreted by the parietal and visceral pleural membranes. Although no actual space normally exists between the parietal (outer) and visceral (inner) pleural membranes, the potential space between them is called the pleural cavity. The parietal pleura (outer membrane) and visceral pleura (inner membrane) slide with little friction across the cavity walls as your lungs move, expand and collapse during respiration.
Figure 1. Chest cavity
Footnote: The black dashed lines indicate the borders of the mediastinum.
Figure 2. Mediastinum
Human lungs
The lungs are soft, spongy, cone-shaped organs in the thoracic (chest) cavity. The lungs consist largely of air tubes and spaces. The balance of the lung tissue, its stroma, is a framework of connective tissue containing many elastic fibers. As a result, the lungs are light, soft, spongy, elastic organs that each weigh only about 0.6 kg (1.25 pounds). The elasticity of healthy lungs helps to reduce the effort of breathing.
The left and right lungs are situated in the left and right pleural cavities inside the thoracic cavity. They are separated from each other by the heart and other structures of the mediastinum, which divides the thoracic cavity into two anatomically distinct chambers. As a result, if trauma causes one lung to collapse, the other may remain expanded. Below the lungs, a thin, dome-shaped muscle called the diaphragm separates the chest from the abdomen. When you breathe, the diaphragm moves up and down, forcing air in and out of the lungs. The thoracic cage encloses the rest of the lungs.
Each lung occupies most of the space on its side of the thoracic cavity. A bronchus and some large blood vessels suspend each lung in the cavity. These tubular structures enter the lung on its medial surface.
Parietal refers to a membrane attached to the wall of a cavity; visceral refers to a membrane that is deeper—toward the interior—and covers an internal organ, such as a lung. Within the thoracic (chest) cavity, the compartments that contain the lungs, on either side of the mediastinum, are lined with a membrane called the parietal pleura. A similar membrane, called the visceral pleura, covers each lung.
The parietal and visceral pleural membranes are separated only by a thin film of watery fluid (serous fluid), which they secrete. Although no actual space normally exists between these membranes, the potential space between them is called the pleural cavity.
A thin lining layer called the pleura surrounds the lungs. The pleura protects your lungs and helps them slide back and forth against the chest wall as they expand and contract during breathing. A layer of serous membrane, the visceral pleura, firmly attaches to each lung surface and folds back to become the parietal pleura. The parietal pleura, in turn, borders part of the mediastinum and lines the inner wall of the thoracic cavity and the superior surface of the diaphragm.
In certain conditions, the pleural cavities may fill with air (pneumothorax), blood (hemothorax), or pus. Air in the pleural cavities, most commonly introduced in a surgical opening of the chest or as a result of a stab or gunshot wound, may cause the lungs to collapse. This collapse of a part of a lung, or rarely an entire lung, is called atelectasis. The goal of treatment is the evacuation of air (or blood) from the pleural space, which allows the lung to reinflate. A small pneumothorax may resolve on its own, but it is oft en necessary to insert a chest tube to assist in evacuation.
The thoracic (chest) cavity is divided by a thick wall called the mediastinum. This is the region between the lungs, extending from the base of the neck to the diaphragm. It is occupied by the heart, the major blood vessels connected to it, the esophagus, the trachea and bronchi, and a gland called the thymus.
Each lung is a blunt cone with the tip, or apex, pointing superiorly. The apex on each side extends into the base of the neck, superior to the first rib. The broad concave inferior portion, or base, of each lung rests on the superior surface of the diaphragm.
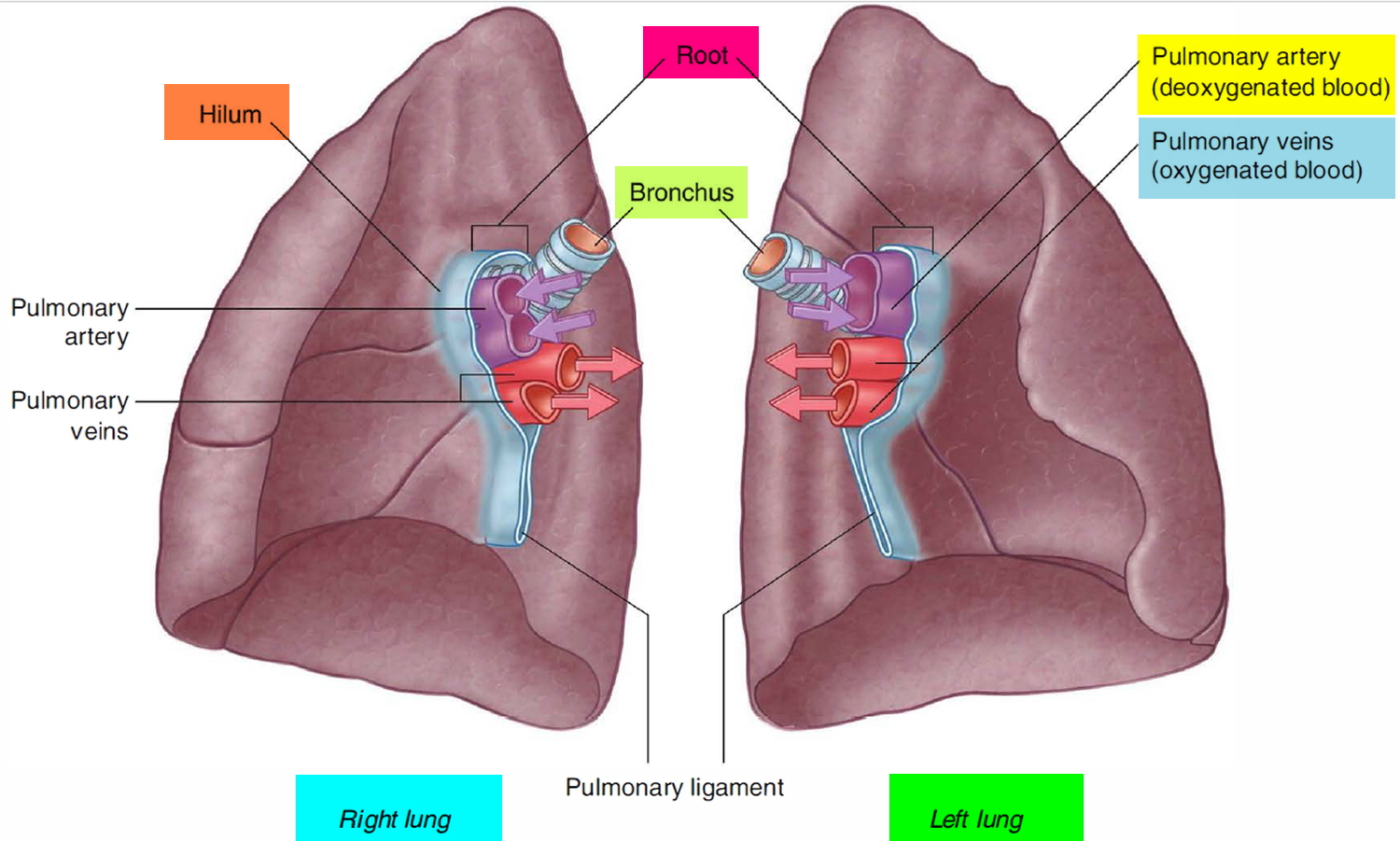
On the medial (mediastinal) surface of each lung is an indentation, the hilum, through which blood vessels, bronchi, lymphatic vessels, and nerves enter and exit the lung. Collectively, these structures attach the lung to the mediastinum and are called the root of the lung. The largest components of this root are the pulmonary artery and veins and the main (primary) bronchus. Because the heart is tilted slightly to the left of the median plane of the thorax, the left and right lungs differ slightly in shape and size.
Within each root and located in the hilum are:
- a pulmonary artery,
- two pulmonary veins,
- a main bronchus,
- bronchial vessels,
- nerves, and
- lymphatics.
Generally, the pulmonary artery is superior at the hilum, the pulmonary veins are inferior, and the bronchi are somewhat posterior in position. On the right side, the lobar bronchus to the superior lobe branches from the main bronchus in the root, unlike on the left where it branches within the lung itself, and is superior to the pulmonary artery.
Figure 3. Lungs anatomy
Figure 4. Hilum (roots) of the lungs
Several deep fissures divide the two lungs into different patterns of lobes.
- The left lung is divided into two lobes, the superior lobe and the inferior lobe, by the oblique fissure. The left lung is somewhat smaller than the right and has a cardiac notch, a deviation in its anterior border that accommodates the heart.
- The right lung is partitioned into three lobes, the superior, middle, and inferior lobes, by the oblique and horizontal fissures.
Each lung lobe is served by a lobar (secondary) bronchus and its branches. Each of the lobes, in turn, contains a number of bronchopulmonary segments separated from one another by thin partitions of dense connective tissue. Each segment receives air from an individual segmental (tertiary) bronchus. There are approximately ten bronchopulmonary segments arranged in similar, but not identical, patterns in each of the two lungs.
The bronchopulmonary segments have clinical significance in that they limit the spread of some diseases within the lung, because infections do not easily cross the connective tissue partitions between them. Furthermore, because only small veins span these partitions, surgeons can neatly remove segments without cutting any major blood vessels.
The smallest subdivision of the lung that can be seen with the naked eye is the lobule. Appearing on the lung surface as hexagons ranging from the size of a pencil eraser to the size of a penny, each lobule is served by a bronchiole and its branches. In most city dwellers and in smokers, the connective tissue that separates the individual lobules is blackened with carbon.
Each lung has a half-cone shape, with a base, apex, two surfaces, and three borders.
- The base sits on the diaphragm.
- The apex projects above rib I and into the root of the neck.
- The two surfaces-the costal surface lies immediately adjacent to the ribs and intercostal spaces of the thoracic wall. The mediastinal surface lies against the mediastinum anteriorly and the vertebral column posteriorly and contains the comma-shaped hilum of the lung, through which structures enter and leave.
- The three borders-the inferior border of the lung is sharp and separates the base from the costal surface. The anterior and posterior borders separate the costal surface from the medial surface. Unlike the anterior and inferior borders, which are sharp, the posterior border is smooth and rounded.
Right lung
The right lung has three lobes and two fissures. Normally, the lobes are freely movable against each other because they are separated, almost to the hilum, by invaginations of visceral pleura. These invaginations form the fissures:
- The oblique fissure separates the inferior lobe (lower lobe) from the superior lobe and the middle lobe of the right lung.
- The horizontal fissure separates the superior lobe (upper lobe) from the middle lobe.
The approximate position of the oblique fissure on a patient, in quiet respiration, can be marked by a curved line on the thoracic wall that begins roughly at the spinous process of the vertebra TIV level of the spine, crosses the fifth interspace laterally, and then follows the contour of rib VI anteriorly.
The horizontal fissure follows the fourth intercostal space from the sternum until it meets the oblique fissure as it crosses rib V.
The orientations of the oblique and horizontal fissures determine where clinicians should listen for lung sounds from each lobe. The largest surface of the superior lobe is in contact with the upper part of the anterolateral wall and the apex of this lobe proj ects into the root of the neck. The surface of the middle lobe lies mainly adjacent to the lower anterior and lateral wall. The costal surface of the inferior lobe is in contact with the posterior and inferior walls.
The medial surface of the right lung lies adjacent to a number of important structures in the mediastinum and the root of the neck. These include the:
- heart,
- inferior vena cava,
- superior vena cava,
- azygos vein, and
- esophagus.
The right subclavian artery and vein arch over and are related to the superior lobe of the right lung as they pass over the dome of the cervical pleura and into the axilla.
Left lung
The left Iung is smaller than the right lung and has two lobes separated by an oblique fissure. The oblique fissure of the left lung is slightly more oblique than the corresponding fissure of the right lung. During quiet respiration, the approximate position of the left oblique fissure can be marked by a curved line on the thoracic wall that begins between the spinous processes of thoracic vertebrae 3 (T3) and thoracic vertebrae 4 (TIV), crosses the fifth interspace laterally, and follows the contour of 6th rib anteriorly.
As with the right lung, the orientation of the oblique fissure determines where to listen for lung sounds from each lobe. The largest surface of the superior lobe is in contact with the upper part of the anterolateral wall, and the apex of this lobe proj ects into the root of the neck. The costal surface of the inferior lobe is in contact with the posterior and inferior walls.
The inferior portion o f the medial surface of the left lung, unlike the right lung, is notched because of the heart’s projection into the left pleural cavity from the middle mediastinum. From the anterior border of the lower part of the superior lobe a tongue-like extension (the lingula of the left lung) projects over the heart bulge.
The medial surface of the left lung lies adjacent to a number of important structures in the mediastinum and root of the neck. These include the:
- heart,
- aortic arch,
- thoracic aorta, and
- esophagus.
The left subclavian artery and vein arch over and are related to the superior lobe of the left lung as they pass over the dome of the cervical pleura and into the axilla.
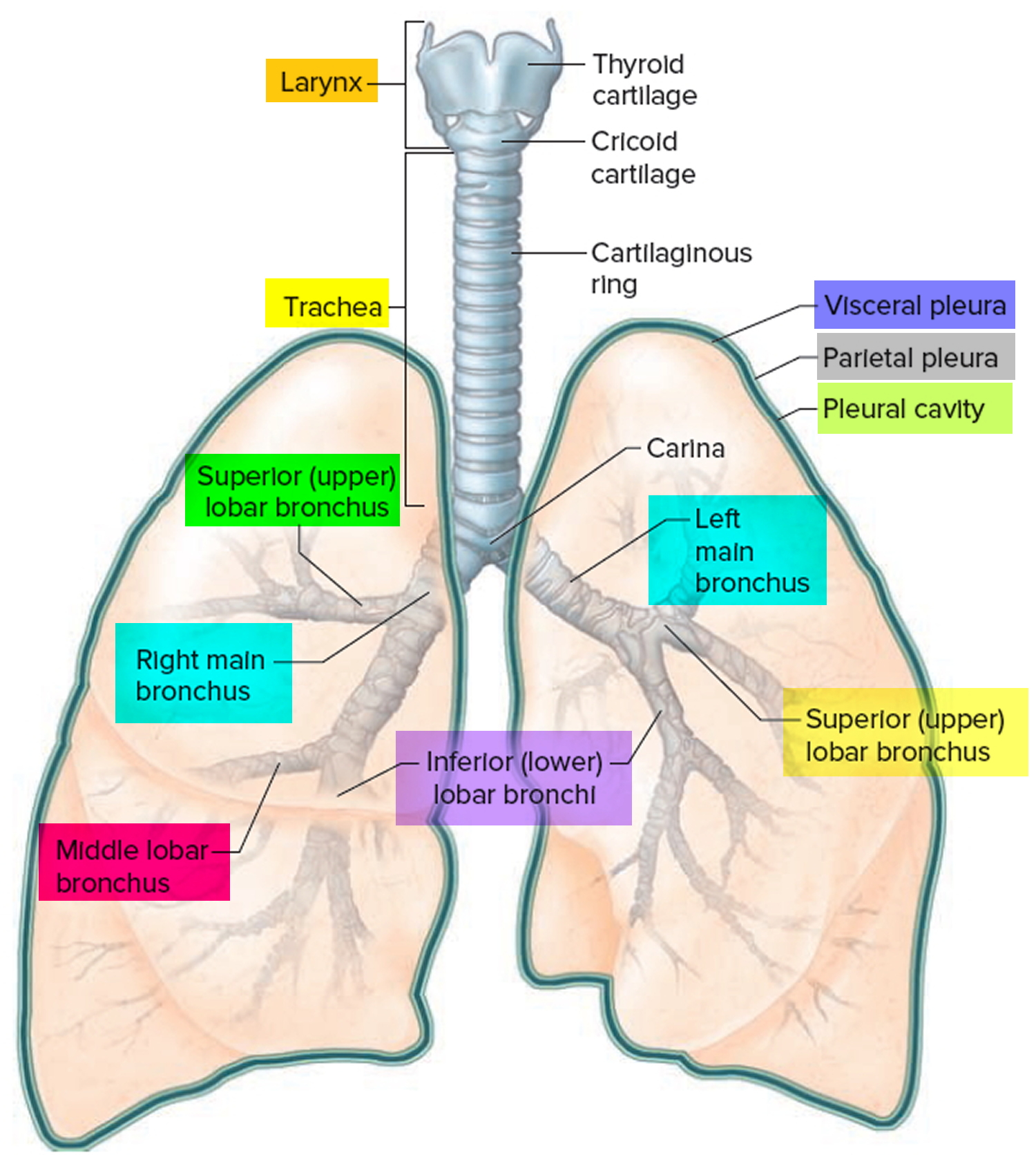
Bronchial tree
The trachea is a flexible tube that extends from cervical spine C6 (vertebral level C VI) in the lower neck to thoracic spine T4-T5 (vertebral level T4 to T5) in the mediastinum where it bifurcates into a right and a left main bronchus. The trachea is held open by C-shaped transverse cartilage rings embedded in its wall the open part of the C facing posteriorly. The lowest tracheal ring has a hook-shaped structure, the carina, that projects backwards in the midline between the origins of the two main bronchi. The posterior wall of the trachea is composed mainly of smooth muscle. Each main bronchus enters the root of a lung and passes through the hilum into the lung itself. The right main bronchus is wider and takes a more vertical course through the root and hilum than the left main bronchus. Therefore, inhaled foreign bodies tend to lodge more frequently on the right side than on the left.
The bronchial tree consists of branched airways leading from the trachea to the microscopic air sacs in the lungs. Its branches begin with the right and left main (primary) bronchi, which arise from the trachea at the level of the fifth thoracic vertebra. Each bronchus enters its respective lung. A short distance from its origin, each main bronchus divides into lobar (secondary) bronchi. The lobar bronchi branch into segmental (tertiary) bronchi, which supply bronchopulmonary segments. Within each bronchopulmonary segment, the segmental bronchi give rise to multiple generations of divisions of increasingly finer tubes and, ultimately, to bronchioles , which further subdivide to terminal bronchioles, respiratory bronchioles, and finally to very thin tubes called alveolar ducts. These ducts lead to thin-walled outpouchings called alveolar sacs. Alveolar sacs lead to smaller, microscopic air sacs called alveoli (singular, alveolus), which lie within capillary networks (Figure 6). The alveoli are the sites of gas exchange between the inhaled air and the bloodstream.
The structure of a bronchus is similar to that of the trachea, but the tubes that branch from it have less cartilage in their walls, and the bronchioles lack cartilage. As the cartilage diminishes, a layer of smooth muscle surrounding the tube becomes more prominent. This muscular layer persists even in the smallest bronchioles, but only a few muscle cells are associated with the alveolar ducts.
The absence of cartilage in the bronchioles allows their diameters to change in response to contraction of the smooth muscle in their walls, similar to what happens with arterioles of the cardiovascular system. Part of the “fight-or-flight” response, triggered by the sympathetic nervous system, is bronchodilation, in which the smooth muscle relaxes and the airways become wider and allow more airflow. The opposite, bronchoconstriction, occurs when the smooth muscle contracts and it becomes difficult to move air in and out of the lungs. Bronchoconstriction can occur with allergies. Asthma is an extreme example of bronchoconstriction.
The mucous membranes of the bronchial tree continue to filter the incoming air, and the many branches of the tree distribute the air to alveoli throughout the lungs. The alveoli, in turn, provide a large surface area of thin simple squamous epithelial cells through which gases are easily exchanged. Oxygen diffuses from the alveoli into the blood in nearby capillaries, and carbon dioxide diffuses from the blood into the alveoli.
Figure 5. Bronchial tree of the lungs
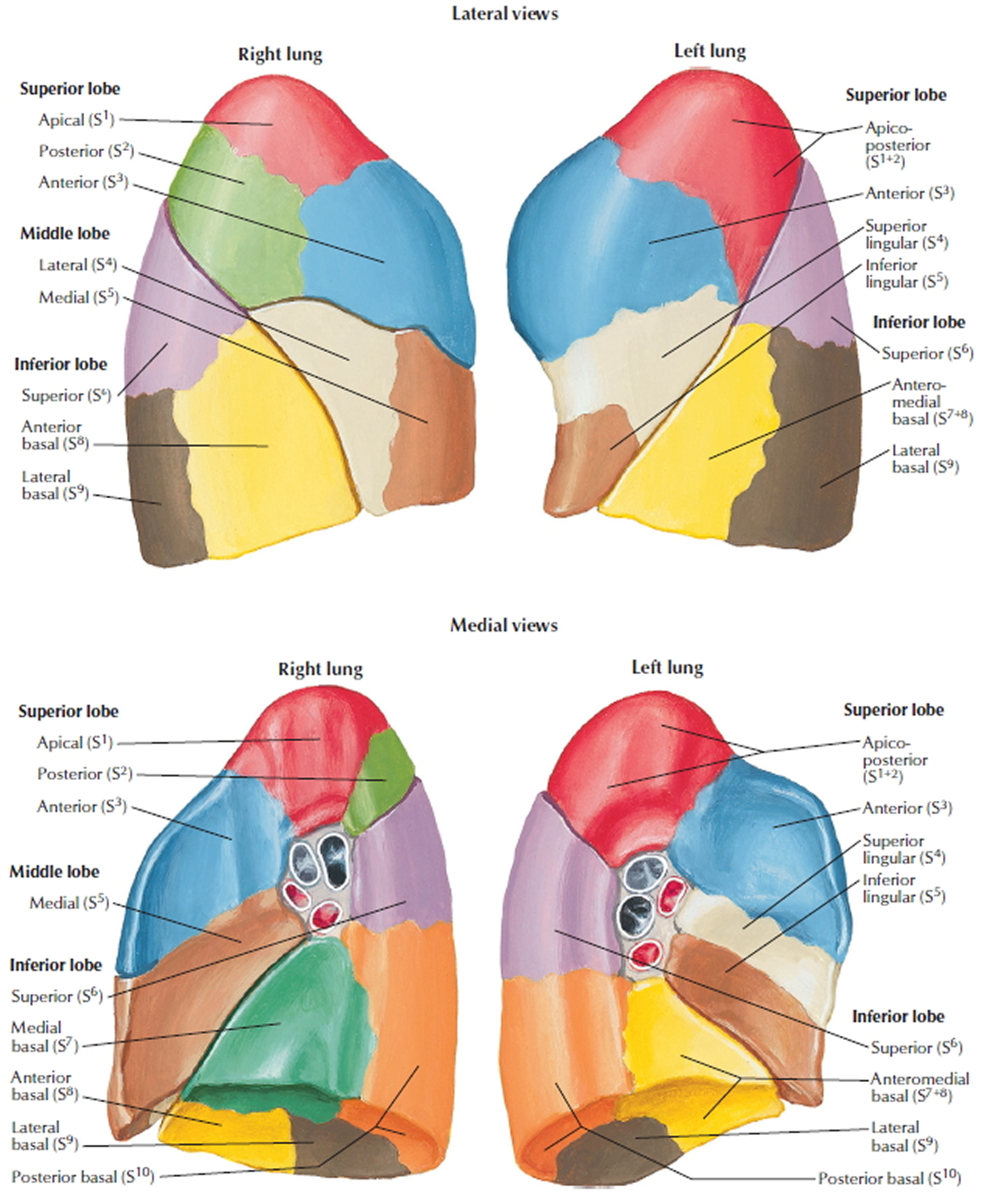
Bronchopulmonary segments
A bronchopulmonary segment is the area of lung supplied by a segmental bronchus and its accompanying pulmonary artery branch. Tributaries of the pulmonary vein tend to pass intersegmentally between and around the margins of segments. Each bronchopulmonary segment is shaped like an irregular cone, with the apex at the origin of the segmental bronchus and the base projected peripherally onto the surface of the lung.
A bronchopulmonary segment is the smallest functionally independent region of a lung and the smallest area of lung that can be isolated and removed without affecting adjacent regions.
There are ten bronchopulmonary segments in each lung; some of them fuse in the left lung.
Figure 6. Bronchopulmonary segments
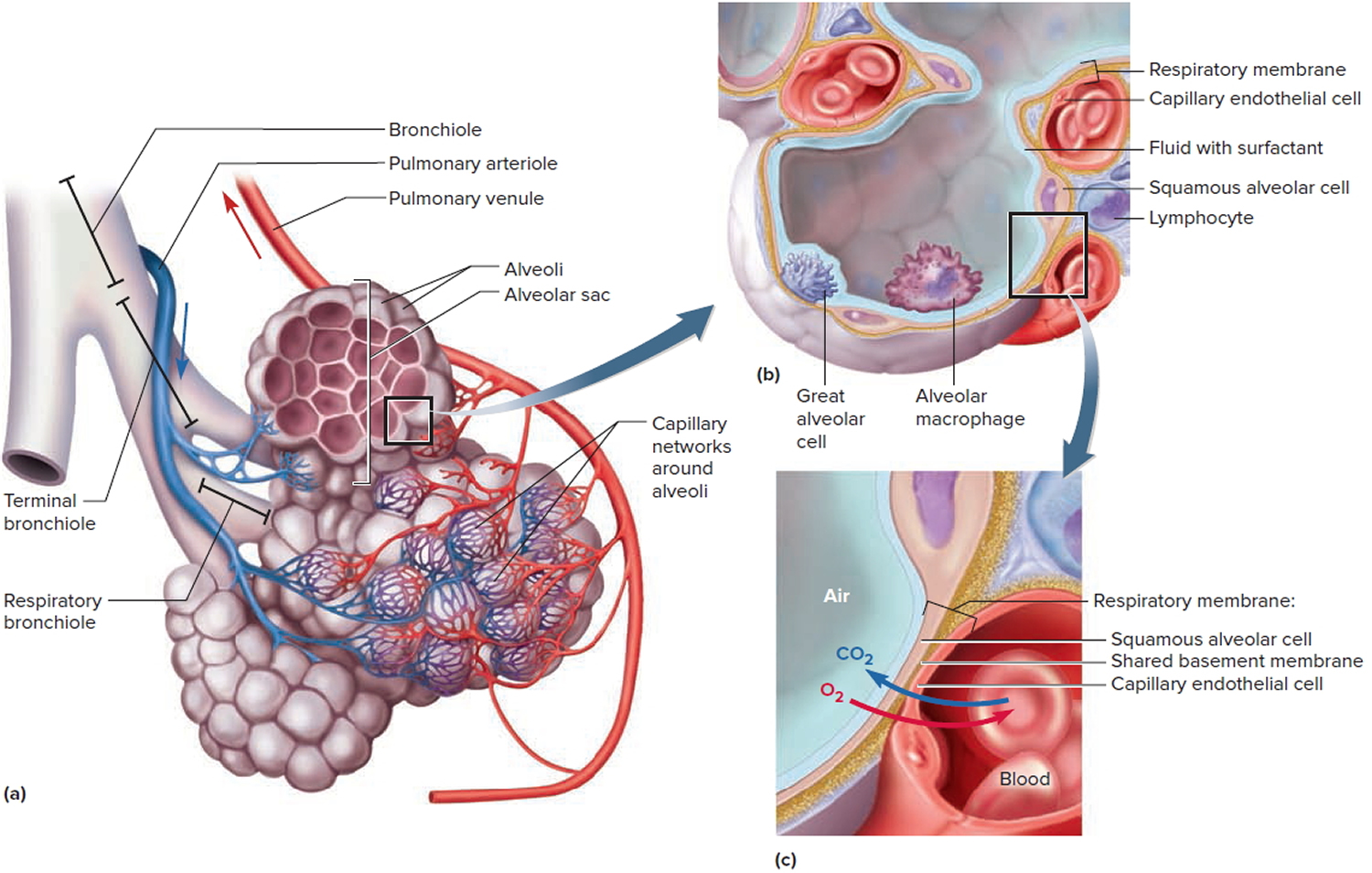
Lung Alveoli
Each human lung is a spongy mass composed of 150 million little sacs, the alveoli. These provide about 70 m², per lung, of gas-exchange surface—about equal to the floor area of a handball court or a room about 8.4 m (25 ft) square.
An alveolus is a pouch about 0.2 to 0.5 mm in diameter. Thin, broad cells called squamous (type I) alveolar cells cover about 95% of the alveolar surface area. Their thinness allows for rapid gas diffusion between the air and blood. The other 5% is covered by round to cuboidal great (type II) alveolar cells. Even though they cover less surface area, these considerably outnumber the squamous alveolar cells.
Great (type II) alveolar cells have two functions:
- They repair the alveolar epithelium when the squamous cells are damaged; and
- They secrete pulmonary surfactant, a mixture of phospholipids and protein that coats the alveoli and smallest bronchioles and prevents the bronchioles from collapsing when one exhales.
The most numerous of all cells in the lung are alveolar macrophages (dust cells), which wander the lumens of the alveoli and the connective tissue between them. These cells keep the alveoli free of debris by phagocytizing dust particles that escape entrapment by mucus in the higher parts of the respiratory tract. In lungs that are infected or bleeding, the macrophages also phagocytize bacteria and loose blood cells. As many as 100 million alveolar macrophages perish each day as they ride up the mucociliary escalator to be swallowed and digested, thus ridding the lungs of their load of debris.
Each alveolus is surrounded by a web of blood capillaries supplied by small branches of the pulmonary artery. The barrier between the alveolar air and blood, called the respiratory membrane, consists only of the squamous alveolar cell, the squamous endothelial cell of the capillary, and their shared basement membrane. These have a total thickness of only 0.5 μm, just 1/15 the diameter of a single red blood cell.
It is very important to prevent fluid from accumulating in the alveoli, because gases diffuse too slowly through liquid to sufficiently aerate the blood. Except for a thin film of moisture on the alveolar wall, the alveoli are kept dry by the absorption of excess liquid by the blood capillaries. The mean blood pressure in these capillaries is only 10 mm Hg compared to 30 mm Hg at the arterial end of the average capillary elsewhere. This low blood pressure is greatly overridden by the oncotic pressure that retains fluid in the capillaries, so the osmotic uptake of water overrides filtration and keeps the alveoli free of fluid. The lungs also have a more extensive lymphatic drainage than any other organ in the body. The low capillary blood pressure also prevents rupture of the delicate respiratory membrane.
Figure 7. Lungs alveoli
Note: (a) Clusters of alveoli and their blood supply. (b) Structure of an alveolus. (c) Structure of the respiratory membrane.
How your lungs work
Your lungs have a system of tubes that carry oxygen in and out as you breathe. The windpipe divides into two tubes, the right bronchus and left bronchus. These split into smaller tubes called secondary bronchi. They split again to make smaller tubes called bronchioles. The bronchioles have small air sacs at the end called alveoli.
In the air sacs, oxygen passes into your bloodstream from the air breathed in. Your bloodstream carries oxygen to all the cells in your body. At the same time carbon dioxide passes from your bloodstream into the air sacs. This waste gas is removed from the body as you breathe out.
What are Lung Nodules?
A lung nodule (or mass) is a small abnormal area that is sometimes found during a CT scan of the chest. Most lung nodules seen on CT scans are not cancer. They are more often the result of old infections, scar tissue, or other causes. The risk of developing cancer in very small lung nodules (<5 mm) is very low 12. But tests are often needed to be sure a lung nodule is not cancer.
Most often the next step is to get a repeat CT scan of the chest to see if the lung nodule is growing over time. The time between scans might range anywhere from a few months to a year, depending on how likely your doctor thinks that the lung nodule could be cancer. This is based on the size, shape, and location of the lung nodule, as well as whether it appears to be solid or filled with fluid. If a repeat CT scan of the chest shows that the lung nodule has grown, your doctor might also want to get another type of imaging test called a positron emission tomography (PET) scan, which can often help tell if it is cancer.
If later scans show that the lung nodule has grown, or if the nodule has other concerning features, your doctor will want to get a sample of it to check it for cancer cells. This is called a biopsy. This can be done in different ways:
- The doctor might pass a long, thin tube called a bronchoscope, down your throat and into the airways of your lung to reach the nodule. A small tweezer on the end of the bronchoscope can be used to get a sample of the lung nodule.
- If the lung nodule is in the outer part of the lung, the doctor might pass a thin, hollow needle through the skin of the chest wall with the guidance of a CT scan and into the nodule to get a sample.
- If there is a higher chance that the nodule is cancer or if the nodule can’t be reached with a needle or bronchoscope, surgery might be done to remove the nodule and some surrounding lung tissue. Sometimes larger parts of the lung might be removed as well.
After a biopsy is done, the tissue sample will be looked at closely in the lab by a doctor called a pathologist. The pathologist will check the biopsy for cancer, infection, scar tissue, and other lung problems. If cancer is found, then special tests will be done to find out what kind of cancer it is. If something other than cancer is found, the next step will depend on the diagnosis. Some lung nodules will be followed with a repeat CT scan in 6-12 months for a few years to make sure it does not change. If the lung nodule biopsy shows an infection, you might be sent to a specialist called an infectious disease doctor, for further testing. Your doctor will decide on the next step, depending on the results of the biopsy.
Lung cancer signs and symptoms
Most lung cancers do not cause any symptoms until they have spread, but some people with early lung cancer do have symptoms. If you go to your doctor when you first notice symptoms, your cancer might be diagnosed at an earlier stage, when treatment is more likely to be effective.
Most of these symptoms are more likely to be caused by something other than lung cancer. Still, if you have any of these problems, it’s important to see your doctor right away so the cause can be found and treated, if needed.
The most common symptoms of lung cancer are:
- A cough that does not go away or gets worse
- Coughing up blood or rust-colored sputum (spit or phlegm)
- Chest pain that is often worse with deep breathing, coughing, or laughing
- Hoarseness of voice
- Loss of appetite
- Unexplained weight loss
- Shortness of breath
- Feeling tired or weak
- Infections such as bronchitis and pneumonia that don’t go away or keep coming back
- New onset of wheezing
- Some people have swollen fingers and nails called finger clubbing. They may also have pain and swelling in their joints. This condition is called hypertrophic pulmonary osteoarthropathy (HPOA).
If lung cancer spreads to other parts of the body, it may cause:
- Bone pain (like pain in the back or hips)
- Nervous system changes (such as headache, weakness or numbness of an arm or leg, dizziness, balance problems, or seizures), from cancer spread to the brain
- Yellowing of the skin and eyes (jaundice), from cancer spread to the liver
- Swelling of lymph nodes (collection of immune system cells) such as those in the neck or above the collarbone
Some lung cancers can cause syndromes, which are groups of specific symptoms.
Horner syndrome
Cancers of the upper part of the lungs are sometimes called Pancoast tumors. These tumors are more likely to be non-small cell lung cancer (NSCLC) than small cell lung cancer (SCLC).
Pancoast tumors can affect certain nerves to the eye and part of the face, causing a group of symptoms called Horner syndrome:
- Drooping or weakness of one upper eyelid (ptosis)
- A smaller pupil (dark part in the center of the eye) in the same eye (miosis)
- Little or no sweating on the same side of the face (anhidrosis)
Pancoast tumors can also sometimes cause severe shoulder pain.
Superior vena cava syndrome
The superior vena cava (SVC) is a large vein that carries blood from the head and arms down to the heart. It passes next to the upper part of the right lung and the lymph nodes inside the chest. Tumors in this area can press on the superior vena cava, which can cause the blood to back up in the veins. This can lead to swelling in the face, neck, arms, and upper chest (sometimes with a bluish-red skin color). It can also cause headaches, dizziness, and a change in consciousness if it affects the brain. While superior vena cava syndrome can develop gradually over time, in some cases it can become life-threatening, and needs to be treated right away.
Paraneoplastic syndromes
Some lung cancers make hormone-like substances that enter the bloodstream and cause problems with distant tissues and organs, even though the cancer has not spread to those places. These problems are called paraneoplastic syndromes. Sometimes paraneoplastic syndromes may be the first symptoms of lung cancer. Because the symptoms affect other organs, a disease other than lung cancer may first be suspected as causing them.
Paraneoplastic syndromes can happen with any lung cancer but are more often associated with small cell lung cancer (SCLC). Some common syndromes include:
- SIADH (syndrome of inappropriate anti-diuretic hormone): In this condition, the cancer cells make ADH (anti-diuretic hormone), a hormone that causes the kidneys to hold water. This lowers salt levels in the blood. Symptoms of syndrome of inappropriate anti-diuretic hormone (SIADH) can include fatigue, loss of appetite, muscle weakness or cramps, nausea, vomiting, restlessness, and confusion. Without treatment, severe cases may lead to seizures and coma.
- Cushing syndrome: In this condition, the cancer cells make ACTH, a hormone that causes the adrenal glands to make cortisol. This can lead to symptoms such as weight gain, easy bruising, weakness, drowsiness, and fluid retention. Cushing syndrome can also cause high blood pressure, high blood sugar levels, or even diabetes.
- Nervous system problems: Small cell lung cancer (SCLC) can sometimes cause the body’s immune system to attack parts of the nervous system, which can lead to problems. One example is a muscle disorder called Lambert-Eaton syndrome. In this syndrome, muscles around the hips become weak. One of the first signs may be trouble getting up from a sitting position. Later, muscles around the shoulder may become weak. A less common problem is paraneoplastic cerebellar degeneration, which can cause loss of balance and unsteadiness in arm and leg movement, as well as trouble speaking or swallowing. Small cell lung cancer can also cause other nervous system problems, such as muscle weakness, sensation changes, vision problems, or even changes in behavior.
- High levels of calcium in the blood (hypercalcemia), which can cause frequent urination, thirst, constipation, nausea, vomiting, belly pain, weakness, fatigue, dizziness, and confusion
- Blood clots
Again, many of these symptoms are more likely to be caused by something other than lung cancer. Still, if you have any of these problems, it’s important to see your doctor right away so the cause can be found and treated, if needed.
Lung cancer causes
Smoking causes the majority of lung cancers — both in smokers and in people exposed to secondhand smoke. About 80% of lung cancer deaths are caused by smoking, and many others are caused by exposure to secondhand smoke. But lung cancer also occurs in people who never smoked and in those who never had prolonged exposure to secondhand smoke. In these cases, there may be no clear cause of lung cancer. People who smoke and are exposed to other known risk factors such as radon and asbestos are at an even higher risk.
Doctors believe smoking causes lung cancer by damaging the cells that line the lungs. When you inhale cigarette smoke, which is full of cancer-causing substances (carcinogens), changes in the lung tissue begin almost immediately. At first your body may be able to repair this damage. But with each repeated exposure, normal cells that line your lungs are increasingly damaged. Over time, the damage causes cells to act abnormally and eventually cancer may develop.
Workplace exposures to asbestos, diesel exhaust or certain other chemicals can also cause lung cancers in some people who don’t smoke.
A small portion of lung cancers develop in people with no known risk factors for the disease. Some of these might just be random events that don’t have an outside cause, but others might be due to factors that scientists don’t yet know about.
Lung cancers in people who don’t smoke are often different from those that occur in people who do. They tend to develop in younger people and often have certain gene changes that are different from those in tumors found in people who smoke. In some cases, these gene changes can be used to guide treatment.
Inherited gene changes
Some people inherit DNA mutations (changes) from their parents that greatly increase their risk for developing certain cancers. But inherited mutations alone are not thought to cause very many lung cancers. Still, genes do seem to play a role in some families with a history of lung cancer. For example, people who inherit certain DNA changes in a particular chromosome (chromosome 6) are more likely to develop lung cancer, even if they don’t smoke or only smoke a little.
Some people seem to inherit a reduced ability to break down or get rid of certain types of cancer-causing chemicals in the body, such as those found in tobacco smoke. This could put them at higher risk for lung cancer.
Other people inherit faulty DNA repair mechanisms that make it more likely they will end up with DNA changes. People with DNA repair enzymes that don’t work normally might be especially vulnerable to cancer-causing chemicals and radiation.
Some non-small cell lung cancers (NSCLCs) make too much EGFR protein (which comes from an abnormal EGFR gene). This specific gene change is seen more often with adenocarcinoma of the lung in young, non-smoking, Asian women, but the excess EGFR protein has also been seen in more than 60% of metastatic non-small cell lung cancers.
Researchers are developing tests that may help identify such people, but these tests are not yet used routinely. For now, doctors recommend that all people avoid tobacco smoke and other exposures that might increase their cancer risk.
Acquired gene changes
Gene changes related to lung cancer are usually acquired during a person’s lifetime rather than inherited. Acquired mutations in lung cells often result from exposure to factors in the environment, such as cancer-causing chemicals in tobacco smoke. But some gene changes may just be random events that sometimes happen inside a cell, without having an outside cause.
Acquired changes in certain genes, such as the RB1 tumor suppressor gene, are thought to be important in the development of small cell lung cancer (SCLC). Acquired changes in genes such as the p16 tumor suppressor gene and the K-RAS oncogene, are thought to be important in the development of non-small cell lung cancer. Changes in the TP53 tumor suppression gene and to chromosome 3 can be seen in both non-small cell lung cancer and small cell lung cancer. Not all lung cancers share the same gene changes, so there are undoubtedly changes in other genes that have not yet been found.
Risk factors for lung cancer
A number of factors may increase your risk of lung cancer. Some risk factors can be controlled, for instance, by quitting smoking. And other factors can’t be controlled, such as your family history.
Risk factors for lung cancer include:
- Smoking. Your risk of lung cancer increases with the number of cigarettes you smoke each day and the number of years you have smoked. Cigar smoking and pipe smoking are almost as likely to cause lung cancer as cigarette smoking. Smoking low-tar or “light” cigarettes increases lung cancer risk as much as regular cigarettes. Smoking menthol cigarettes might increase the risk even more since the menthol may allow people to inhale more deeply. Quitting at any age can significantly lower your risk of developing lung cancer.
- Exposure to secondhand smoke. Even if you don’t smoke, your risk of lung cancer increases if you’re exposed to secondhand smoke. Secondhand smoke is thought to cause more than 7,000 deaths from lung cancer each year.
- Previous radiation therapy. If you’ve undergone radiation therapy to the chest for another type of cancer, you may have an increased risk of developing lung cancer.
- Exposure to radon gas. Radon is produced by the natural breakdown of uranium in soil, rock and water that eventually becomes part of the air you breathe. Unsafe levels of radon can accumulate in any building, including homes. Homes and other buildings in nearly any part of the United States can have high indoor radon levels (especially in basements). According to the US Environmental Protection Agency (EPA), radon is the second leading cause of lung cancer in this country, and is the leading cause among people who don’t smoke.
- Exposure to asbestos and other carcinogens. Workplace exposure to asbestos and other substances known to cause cancer — such as uranium, beryllium, cadmium, silica, vinyl chloride, nickel compounds, chromium compounds, coal products, mustard gas, chloromethyl ethers and diesel exhaust— can increase your risk of developing lung cancer, especially if you’re a smoker. Government and industry have taken steps in recent years to help protect workers from many of these exposures. But the dangers are still there, so if you work around these agents, be careful to limit your exposure whenever possible.
- Taking certain dietary supplements. Studies looking at the possible role of vitamin supplements in reducing lung cancer risk have had disappointing results. In fact, 2 large studies found that people who smoked who took beta carotene supplements actually had an increased risk of lung cancer. The results of these studies suggest that people who smoke should avoid taking beta carotene supplements.
- Arsenic in drinking water. Studies of people in parts of Southeast Asia and South America with high levels of arsenic in their drinking water have found a higher risk of lung cancer. In most of these studies, the levels of arsenic in the water were many times higher than those typically seen in the United States, even areas where arsenic levels are above normal. For most Americans who are on public water systems, drinking water is not a major source of arsenic.
- Air pollution. In cities, air pollution (especially near heavily trafficked roads) appears to raise the risk of lung cancer slightly. This risk is far less than the risk caused by smoking, but some researchers estimate that worldwide about 5% of all deaths from lung cancer may be due to outdoor air pollution.
- Family history of lung cancer. Brothers, sisters, and children of people who have had lung cancer may have a slightly higher risk of lung cancer themselves, especially if the relative was diagnosed at a younger age. It’s not clear how much of this risk might be due to shared genes among family members and how much might be from shared household exposures (such as tobacco smoke or radon). Researchers have found that genetics seems to play a role in some families with a strong history of lung cancer.
Lifetime chance of getting lung cancer
Overall, the chance that a man will develop lung cancer in his lifetime is about 1 in 17; for a woman, the risk is about 1 in 18 3. These numbers include both smokers and non-smokers. For smokers the risk is much higher, while for non-smokers the risk is lower.
Black men are about 12% more likely to develop lung cancer than White men 3. The rate is about 16% lower in Black women than in White women 3. Both black and white women have lower rates than men, but the gap is closing. The lung cancer rate has been dropping among men over the past few decades, but only for about the last decade in women 3.
Statistics on survival in people with lung cancer vary depending on the type of lung cancer, the stage (extent) of the cancer when it is diagnosed, and other factors.. For survival statistics based on the stage of the cancer, see Non-Small Cell Lung Cancer Survival Rates By Stage.
Despite the very serious prognosis (outlook) of lung cancer, some people with earlier stage cancers are cured. More than 430,000 people alive today have been diagnosed with lung cancer at some point 3.
Lung cancer prevention
You can reduce your risk of lung cancer if you:
- Don’t smoke. If you’ve never smoked, don’t start. Talk to your children about not smoking so that they can understand how to avoid this major risk factor for lung cancer. Begin conversations about the dangers of smoking with your children early so that they know how to react to peer pressure.
- Stop smoking. Stop smoking now. Quitting reduces your risk of lung cancer, even if you’ve smoked for years. Talk to your healthcare team about strategies and aids that can help you quit. Options include nicotine replacement products, medicines and support groups.
- Avoid secondhand smoke. If you live or work with a person who smokes, urge them to quit. At the very least, ask them to smoke outside. Avoid areas where people smoke, such as bars. Seek out smoke-free options.
- Avoid carcinogens at work. Take precautions to protect yourself from exposure to toxic chemicals at work. Follow your employer’s precautions. For instance, if you’re given a face mask for protection, always wear it. Ask your healthcare professional what more you can do to protect yourself at work. Your risk of lung damage from workplace carcinogens increases if you smoke.
- Eat a diet full of fruits and vegetables. Choose a healthy diet with a variety of fruits and vegetables. Food sources of vitamins and nutrients are best. Avoid taking large doses of vitamins in pill form, as they may be harmful. For instance, researchers hoping to reduce the risk of lung cancer in people who smoked heavily gave them beta carotene supplements. Results showed the supplements increased the risk of cancer in people who smoke.
- Exercise most days of the week. If you don’t exercise regularly, start out slowly. Try to exercise most days of the week.
Tobacco smoke
Prevention offers the greatest opportunity to fight lung cancer. Although decades have passed since the link between smoking and lung cancers became clear, smoking is still responsible for most lung cancer deaths.
Quitting reduces your risk of lung cancer, even if you’ve smoked for years. Talk to your doctor about strategies and stop-smoking aids that can help you quit. Options include nicotine replacement products, medications and support groups.
Avoid secondhand smoke. If you live or work with a smoker, urge him or her to quit. At the very least, ask him or her to smoke outside. Avoid areas where people smoke, such as bars and restaurants, and seek out smoke-free options.
Environmental causes
Researchers also continue to look into some of the other causes of lung cancer, such as exposure to radon and diesel exhaust. Finding new ways to limit these exposures could potentially save many more lives.
Test your home for radon. Have the radon levels in your home checked, especially if you live in an area where radon is known to be a problem. High radon levels can be remedied to make your home safer. For information on radon testing, contact your local department of public health or a local chapter of the American Lung Association.
Avoid carcinogens at work. Take precautions to protect yourself from exposure to toxic chemicals at work. Follow your employer’s precautions. For instance, if you’re given a face mask for protection, always wear it. Ask your doctor what more you can do to protect yourself at work. Your risk of lung damage from workplace carcinogens increases if you smoke.
Healthy Diet, Nutrition, and Lifestyle
Researchers are looking for ways to use vitamins or medicines to prevent lung cancer in people at high risk, but so far none have been shown to clearly reduce risk.
Some studies have suggested that a diet high in fruits and vegetables may offer some protection, but more research is needed to confirm this. While any protective effect of fruits and vegetables on lung cancer risk is likely to be much smaller than the increased risk from smoking, following the American Cancer Society dietary recommendations (such as staying at a healthy weight and eating a diet high in fruits, vegetables, and whole grains) may still be helpful.
Avoid taking large doses of vitamins in pill form, as they may be harmful. For instance, researchers hoping to reduce the risk of lung cancer in heavy smokers gave them beta carotene supplements. Results showed beta-carotene supplements actually increased the risk of lung cancer in smokers. The Carotene and Retinol Efficacy Trial 13 included 18,314 male and female current and former smokers (with at least a 20 pack-year history [equivalent to smoking 1 pack per day for 20 years or 2 packs per day for 10 years, for example] of cigarette smoking), as well as some men occupationally exposed to asbestos (who also have a higher risk of lung cancer), all aged 45–74 years. The study randomized participants to take supplements containing 30 mg beta-carotene plus 25,000 IU (7,500 mcg RAE) retinyl palmitate or a placebo daily for about 6 years to evaluate the potential effects on lung cancer risk 13. The trial was ended prematurely after a mean of 4 years, partly because the supplements were unexpectedly found to have increased lung cancer risk by 28% and death from lung cancer by 46%; the supplements also increased the risk of all-cause mortality by 17%. A subsequent study followed the Carotene and Retinol Efficacy Trial participants for an additional 6 years after they stopped taking the study supplements 14. During this time, the differences in lung cancer risk between the intervention and placebo groups were no longer statistically significant, with one exception: women in the intervention group had a 33% higher risk of lung cancer 14.
Exercise most days of the week. A meta-analysis of leisure-time physical activity and lung cancer risk revealed that higher levels of physical activity protect against lung cancer 15. The overall evidence for physical activity has been mixed, but several studies have reported that individuals who are more physically active have a lower risk of lung cancer than those who are more sedentary, even after adjustment for cigarette smoking 16, 17. The American Institute for Cancer Research evidence review rated the inverse association between physical activity and lung cancer as limited suggestive evidence 18.
Early detection
As mentioned in lung cancer screening, screening with low dose spiral CT (LDCT) scans in people at high risk of lung cancer (due to smoking history) lowers the risk of death from lung cancer, when compared to chest x-rays 19. Lung cancer screening is generally offered to older adults who have smoked heavily for many years or who have quit in the past 15 years.
Another approach now being studied uses newer, sensitive tests to look for cancer cells in sputum samples 20. Researchers have found several changes often seen in the DNA of lung cancer cells. Studies are looking at tests that can spot these DNA changes to see if they can find lung cancers at an earlier stage.
Lung cancer screening
Screening is meant to find cancer in people who do not have symptoms of the disease. The U.S. Preventive Services Task Force (USPSTF) makes recommendations about the effectiveness of specific preventive care services for patients without related signs or symptoms 21.
It U.S. Preventive Services Task Force bases its recommendations on the evidence of both the benefits and harms of the service and an assessment of the balance. The U.S. Preventive Services Task Force does not consider the costs of providing a service in this assessment.
In 2021, the USPSTF recommends annual screening for lung cancer with low-dose computed tomography (LDCT) in adults aged 50 to 80 years who have a 20 pack-year smoking history and currently smoke or have quit within the past 15 years 22. Screening should be discontinued once a person has not smoked for 15 years or develops a health problem that substantially limits life expectancy or the ability or willingness to have curative lung surgery 22.
For low-dose computed tomography (LDCT), you lie on a thin, flat table that slides back and forth inside the hole in the middle of the CT scanner, which is a large, doughnut-shaped machine. As the table moves into the opening, an x-ray tube rotates within the scanner, sending out many tiny x-ray beams at precise angles. These beams quickly pass through your body and are detected on the other side of the scanner. A computer then converts these results into detailed images of the lungs.
The main benefit of screening is finding the cancer earlier and thus, lowering the chance of dying from lung cancer.
As with any type of screening, it’s important to be aware that, not everyone who gets screened will benefit. Screening with LDCT will not find all lung cancers. Not all of the cancers that are found will be found at an early stage. Some people with lung cancer that is found by screening will still die from that cancer.
Low-dose computed tomography (LDCT) scans can also find things that turn out not to be cancer, but that still have to be checked out with more tests to know what they are. You might need more CT scans, or less often, invasive tests such as a lung biopsy, in which a piece of lung tissue is removed with a needle or during surgery. These tests may lead to serious complications, but they rarely do.
Low-dose computed tomography (LDCT) scans also expose people to a small amount of radiation with each test. It is less than the dose from a standard CT, but it is more than the dose from a chest x-ray. Some people who are screened may end up needing further CT scans, which means more radiation exposure.
American Cancer Society’s guidelines for lung cancer screening
The American Cancer Society has thoroughly reviewed the subject of lung cancer screening and issued guidelines that are aimed at doctors and other health care providers 23:
The American Cancer Society recommends yearly screening for lung cancer with a low-dose CT (LDCT) scan for people ages 50 to 80 years who 24:
- Smoke or used to smoke
- AND
- Have at least a 20 pack-year history of smoking
A pack-year is equal to smoking 1 pack (or about 20 cigarettes) per day for a year. For example, a person could have a 20 pack-year history by smoking 1 pack a day for 20 years, or by smoking 2 packs a day for 10 years.
Before deciding to be screened, people should have a discussion with a health care professional about the purpose of screening and how it is done, as well as the benefits, limits, and possible harms of screening.
People who still smoke should be counseled about quitting and offered interventions and resources to help them.
People should not be screened if they have serious health problems that will likely limit how long they will live, or if they won’t be able to or won’t want to get treatment if lung cancer is found.
To get the most benefit from screening, patients need to be in good health. For example, they need to be able to have surgery and other treatments to try to cure lung cancer if it is found. Patients who need home oxygen therapy probably couldn’t withstand having part of a lung removed, and so are not candidates for screening. Patients with other serious medical problems that would shorten their lives or keep them from having surgery might not benefit enough from screening for it to be worth the risks, and so should also not be screened.
Metal implants in the chest (like pacemakers) or back (like rods in the spine) can interfere with x-rays and lead to poor quality CT images of the lungs. People with these types of implants were also kept out of the National Lung Screening Trial, and so should not be screened with CT scans for lung cancer according to the American Cancer Society guidelines.
Doctors should talk to these patients about the benefits, limitations, and potential harms of lung cancer screening. Screening should only be done at facilities that have the right type of CT scanner and that have a lot of experience using low-dose CT scans for lung cancer screening. The facility should also have a team of specialists that can provide the appropriate care and follow-up of patients with abnormal results on the scans.
If you and your doctor decide that you should be screened, you should get a low-dose CT scan every year until you reach the age of 74, as long as you are still in good health.
Lung cancer complications
Some people with lung cancer will develop symptoms, such as shortness of breath, a cough and/or chest pain, because of how the cancer affects the lung’s function. As it advances, lung cancer may cause other complications. These can include fluid build-up in the space around your lung (pleural effusion). Lung cancer can also affect your appetite and you may lose weight. You may feel very fatigued and/or have difficulty sleeping.
You may also experience these symptoms and others as side effects of lung-cancer treatments.
Your medical team has a lot of experience in treating symptoms and complications of lung cancer and can give you advice and support to manage them.
Even if you are receiving cancer treatment, there is still a chance that your cancer can spread to another part of your body (metastasis). If this happens, you and your medical team may adjust your cancer treatment plan.
Lung cancer can cause complications, such as:
- Shortness of breath. People with lung cancer can experience shortness of breath if cancer grows to block the major airways. Lung cancer can also cause fluid to accumulate around the lungs, making it harder for the affected lung to expand fully when you inhale.
- Coughing up blood. Lung cancer can cause bleeding in the airway, which can cause you to cough up blood (hemoptysis). Sometimes bleeding can become severe. Treatments are available to control bleeding.
- Pain. Advanced lung cancer that spreads to the lining of a lung or to another area of the body, such as a bone, can cause pain. Tell your doctor if you experience pain, as many treatments are available to control pain.
- Fluid in the chest (pleural effusion). Lung cancer can cause fluid to accumulate in the space that surrounds the affected lung in the chest cavity (pleural space). Fluid accumulating in the chest can cause shortness of breath. Treatments are available to drain the fluid from your chest and reduce the risk that pleural effusion will occur again.
- Cancer that spreads to other parts of the body (metastasis). Lung cancer often spreads (metastasizes) to other parts of the body, such as the brain and the bones. Cancer that spreads can cause pain, nausea, headaches, or other signs and symptoms depending on what organ is affected. Once lung cancer has spread beyond the lungs, it’s generally not curable. Treatments are available to decrease signs and symptoms and to help you live longer.
Lung cancer diagnosis
If there’s reason to think that you may have lung cancer, your doctor can order a number of tests to look for cancerous cells and to rule out other conditions.
Tests may include:
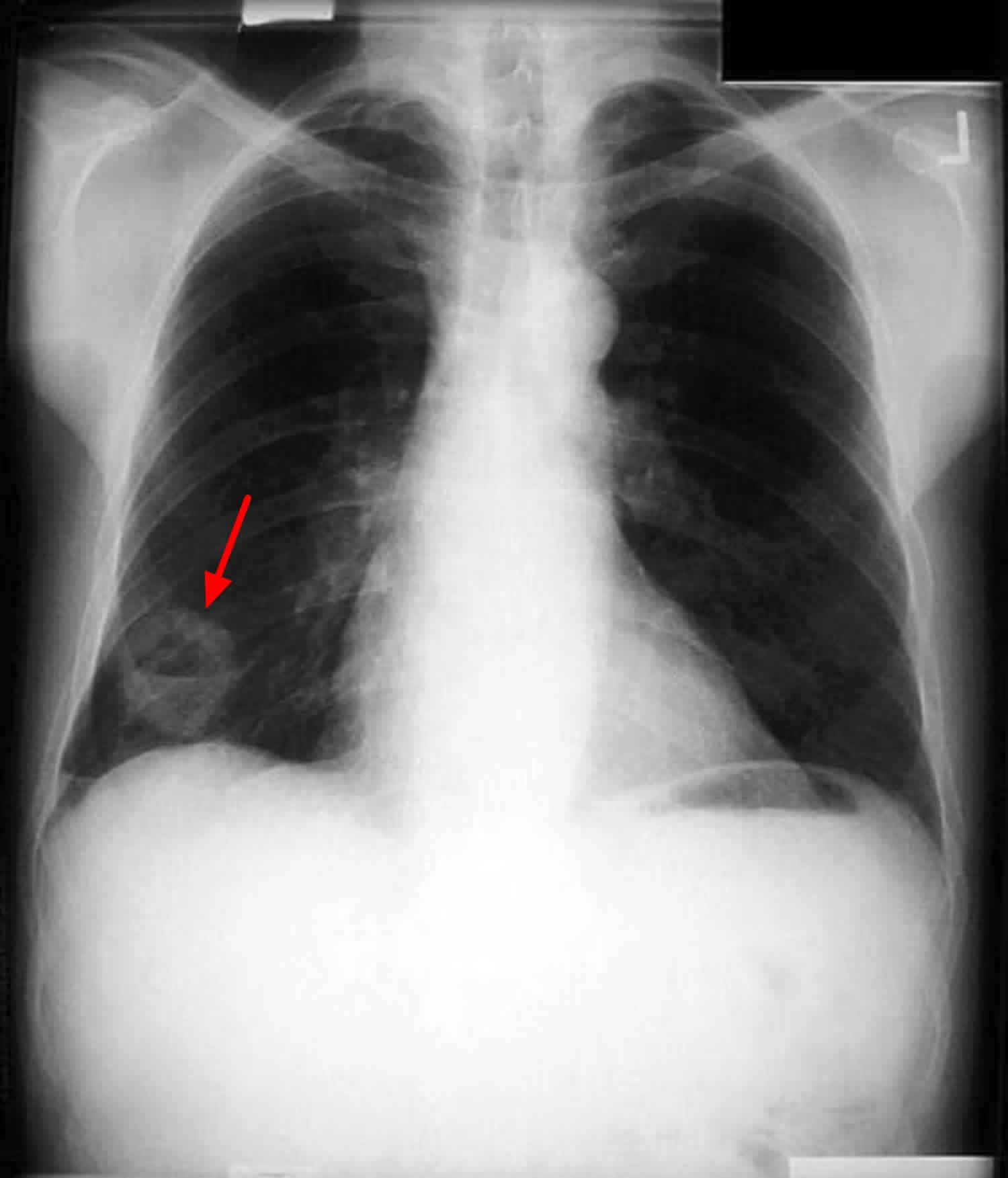
- Imaging tests. An X-ray image of your lungs may reveal an abnormal mass or nodule. A CT scan can reveal small lesions in your lungs that might not be detected on an X-ray. MRI scans are most often used to look for possible spread of lung cancer to the brain or spinal cord. Positron emission tomography (PET) scan a slightly radioactive form of sugar (known as FDG) is injected into the blood and collects mainly in cancer cells.
- Sputum cytology. If you have a cough and are producing sputum, looking at the sputum under the microscope can sometimes reveal the presence of lung cancer cells. The best way to do this is to get early morning samples 3 days in a row. This test is more likely to help find cancers that start in the major airways of the lung, such as squamous cell lung cancers. It might not be as helpful for finding other types of lung cancer. If your doctor suspects lung cancer, further testing will be done even if no cancer cells are found in the sputum.
- Tissue sample (biopsy). A sample of abnormal cells may be removed in a procedure called a biopsy. Your doctor can perform a biopsy in a number of ways, including bronchoscopy, in which your doctor examines abnormal areas of your lungs using a lighted tube that’s passed down your throat and into your lungs. Mediastinoscopy, in which an incision is made at the base of your neck and surgical tools are inserted behind your breastbone to take tissue samples from lymph nodes is also an option. Another option is needle biopsy, in which your doctor uses X-ray or CT images to guide a needle through your chest wall and into the lung tissue to collect suspicious cells. A biopsy sample may also be taken from lymph nodes or other areas where cancer has spread, such as your liver.
Careful analysis of your cancer cells in a lab will reveal what type of lung cancer you have. Results of sophisticated testing can tell your doctor the specific characteristics of your cells that can help determine your prognosis and guide your treatment.
Molecular tests for gene changes
In some cases, especially for non-small cell lung cancer (NSCLC), doctors may test for specific gene changes in the cancer cells that could mean certain targeted drugs might help treat the cancer. For example:
- About 20% to 25% of non-small cell lung cancer (NSCLC) have changes in the KRAS gene that cause them to make an abnormal KRAS protein, which helps the cancer cells grow and spread. Non-small cell lung cancer (NSCLC) with this mutation are often adenocarcinomas, resistant to other drugs such as EGFR inhibitors, and are most often found in people with a smoking history.
- EGFR (epidermal growth factor receptor) is a protein that appears in high amounts on the surface of 10% to 20% of NSCLC cells and helps them grow. Some drugs that target EGFR can be used to treat NSCLC with changes in the EGFR gene, which are more common in certain groups, such as those who don’t smoke, women, and Asians. But these drugs don’t seem to be as helpful in patients whose cancer cells have changes in the KRAS gene.
- About 5% of non-small cell lung cancer (NSCLC) have a change in the ALK gene. This change is most often seen in people who don’t smoke (or who smoke lightly) and have the adenocarcinoma subtype of non-small cell lung cancer (NSCLC). Doctors may test cancers for changes in the ALK gene to see if drugs that target this change may help them.
- About 1% to 2% of non-small cell lung cancer (NSCLC) have a rearrangement in the ROS1 gene, which might make the tumor respond to certain targeted drugs.
- A small percentage of non-small cell lung cancer (NSCLC) have changes in the RET gene. Certain drugs that target cells with RET gene changes might be options for treating these tumors.
- About 5% of non-small cell lung cancer (NSCLC) have changes in the BRAF gene. Certain drugs that target cells with BRAF gene changes might be an option for treating these tumors.
- A small percentage of non-small cell lung cancer (NSCLC) have certain changes in the MET gene that make them more likely to respond to some targeted drugs.
- In a small percentage of non-small cell lung cancer (NSCLC), the cancer cells have certain changes in the HER2 gene that make them more likely to respond to a targeted drug.
- A small number of non-small cell lung cancer (NSCLC) have changes in one of the NTRK genes that make them more likely to respond to some targeted drugs.
These genetic tests can be done on tissue taken during a biopsy or surgery for lung cancer. If the biopsy sample is too small and all the studies cannot be done, the testing may also be done on blood that is taken from a vein just like a regular blood draw. This blood contains the DNA from dead tumor cells found in the bloodstream of people with advanced lung cancer. Obtaining the tumor DNA through a blood draw is called a “liquid biopsy”. Liquid biopsies are done in cases where a tissue biopsy is not possible or if a tissue biopsy is felt to be too dangerous for the patient.
Tests for certain proteins on tumor cells
Lab tests might also be done to look for certain proteins on the cancer cells. For example, non-small cell lung cancer (NSCLC) cells might be tested for the PD-L1 (program death ligand 1) protein, which can show if the cancer is more likely to respond to treatment with certain immunotherapy drugs.
Lung cancer grades
Grading is a way of dividing cancer cells into groups based on how the cells look under a microscope. This gives your doctors an idea of how quickly or slowly the cancer might grow and whether it is likely to spread.
For most lung cancers, there isn’t a specific grading system doctors use. But generally:
- Grade 1: The cells look very like normal cells. They tend to be slow growing and are less likely to spread than higher grade cancer cells. They are called low grade.
- Grade 2: The cells look more abnormal and are more likely to spread. This grade is also called moderately well differentiated or moderate grade.
- Grades 3 and 4: The cells look very abnormal and not like normal cells. They tend to grow quickly and are more likely to spread. They are called poorly differentiated or high grade.
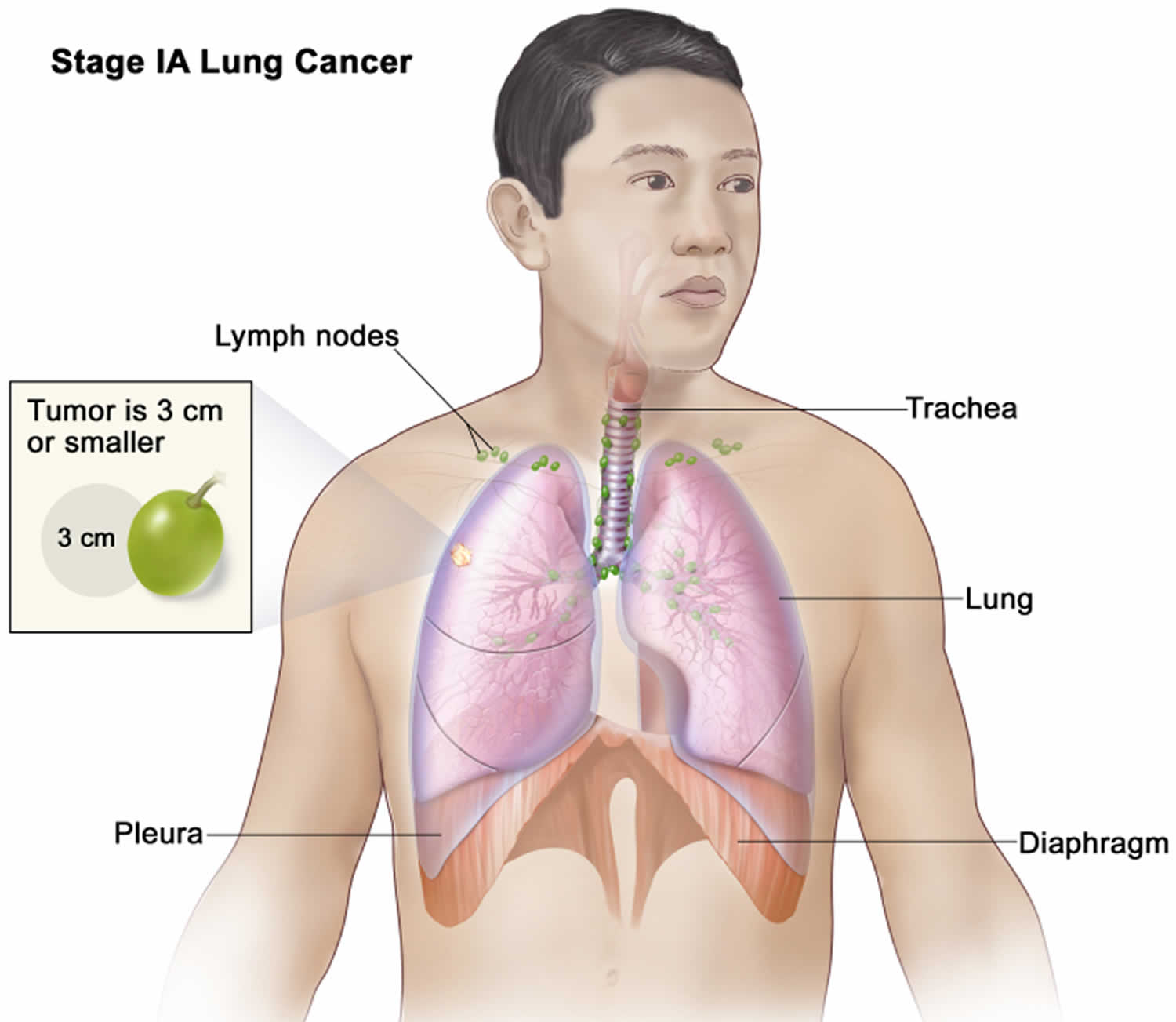
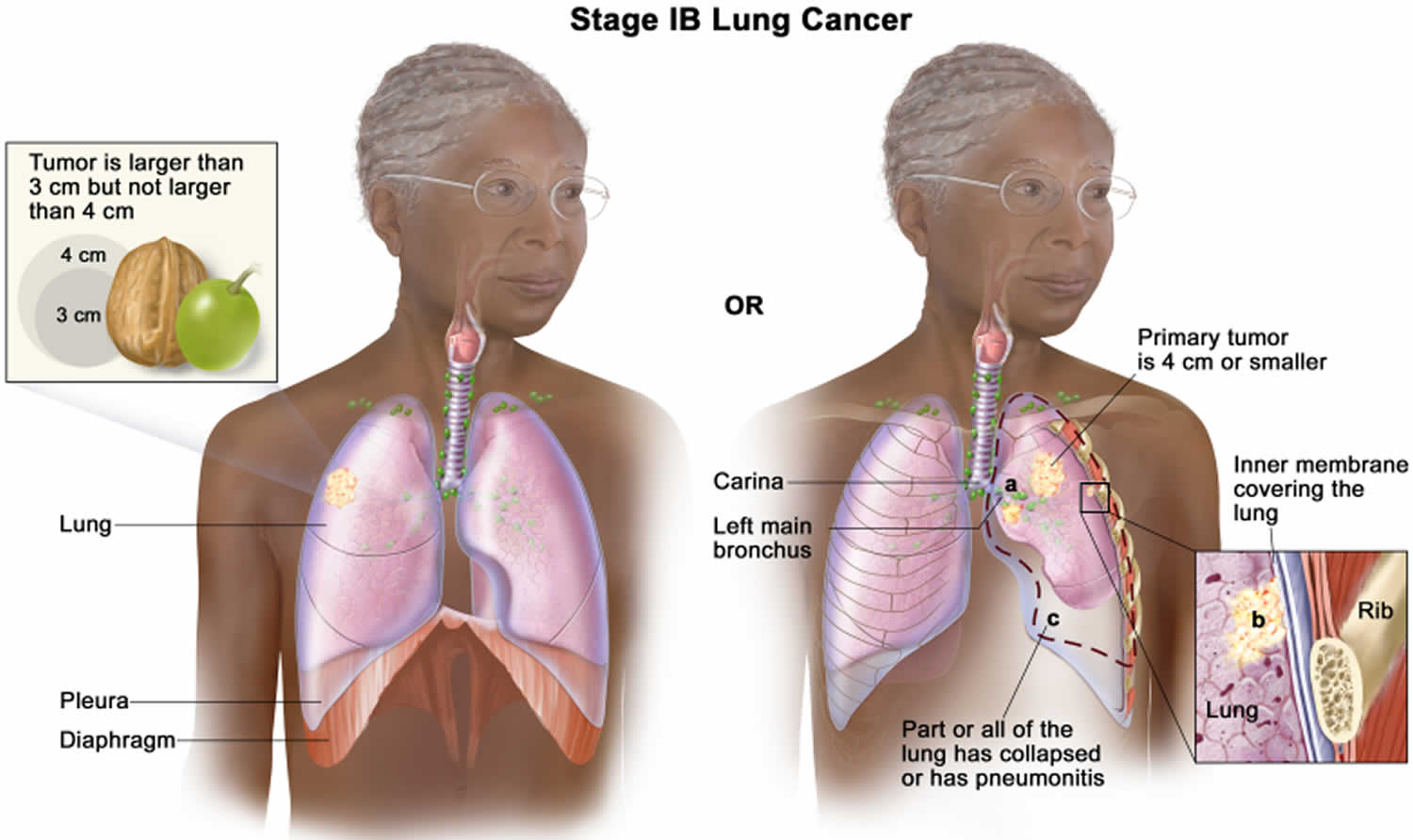
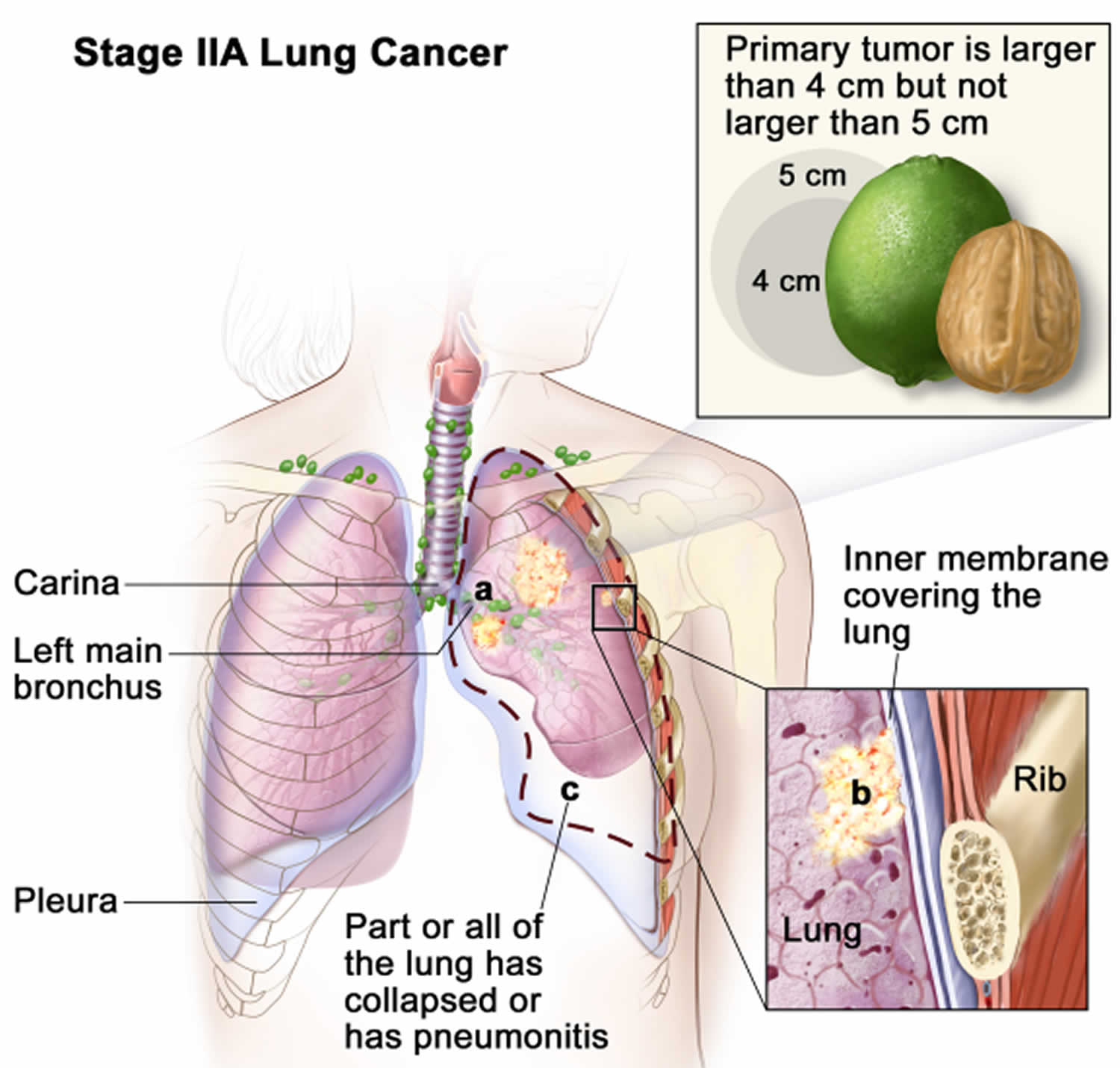
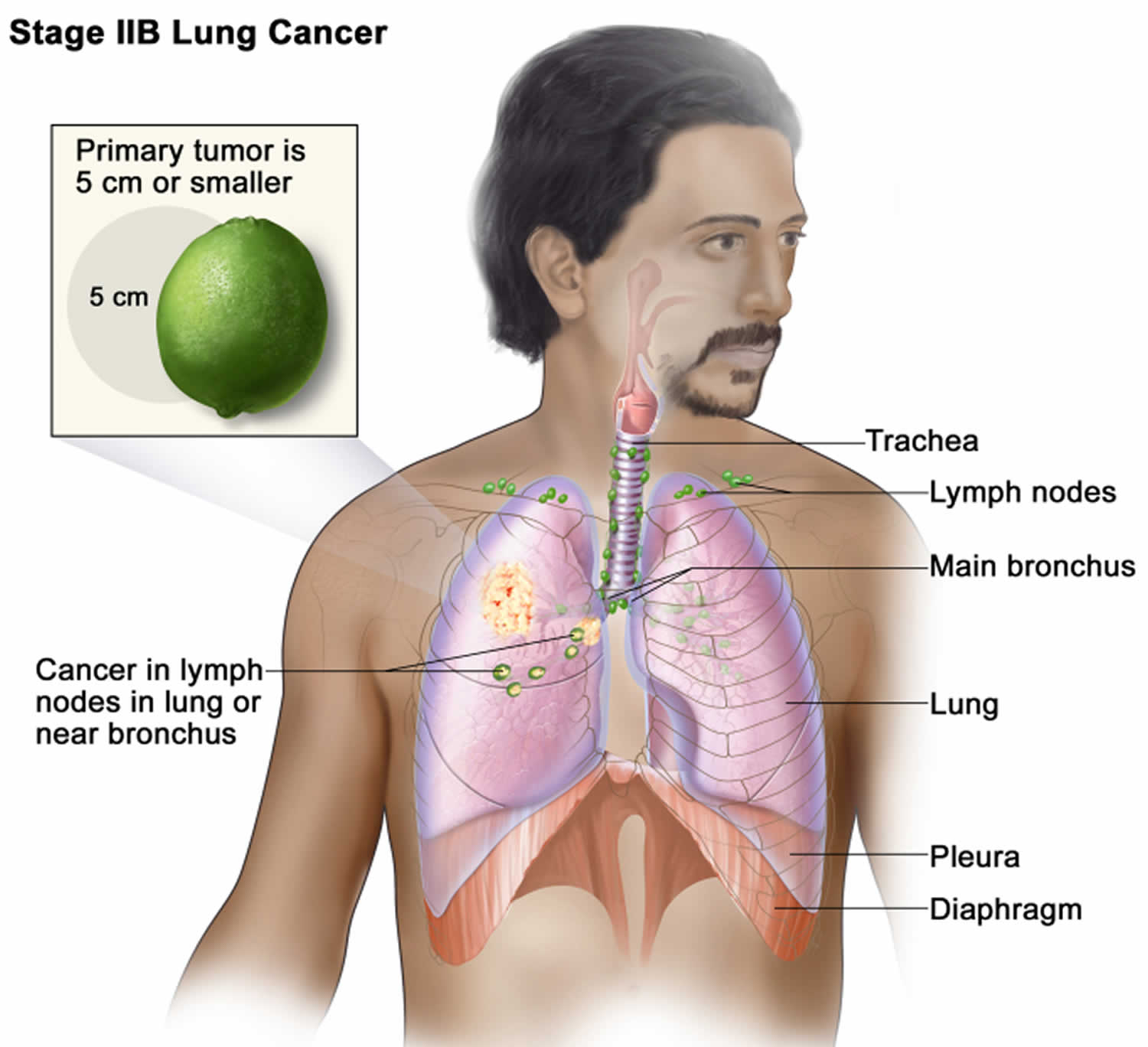
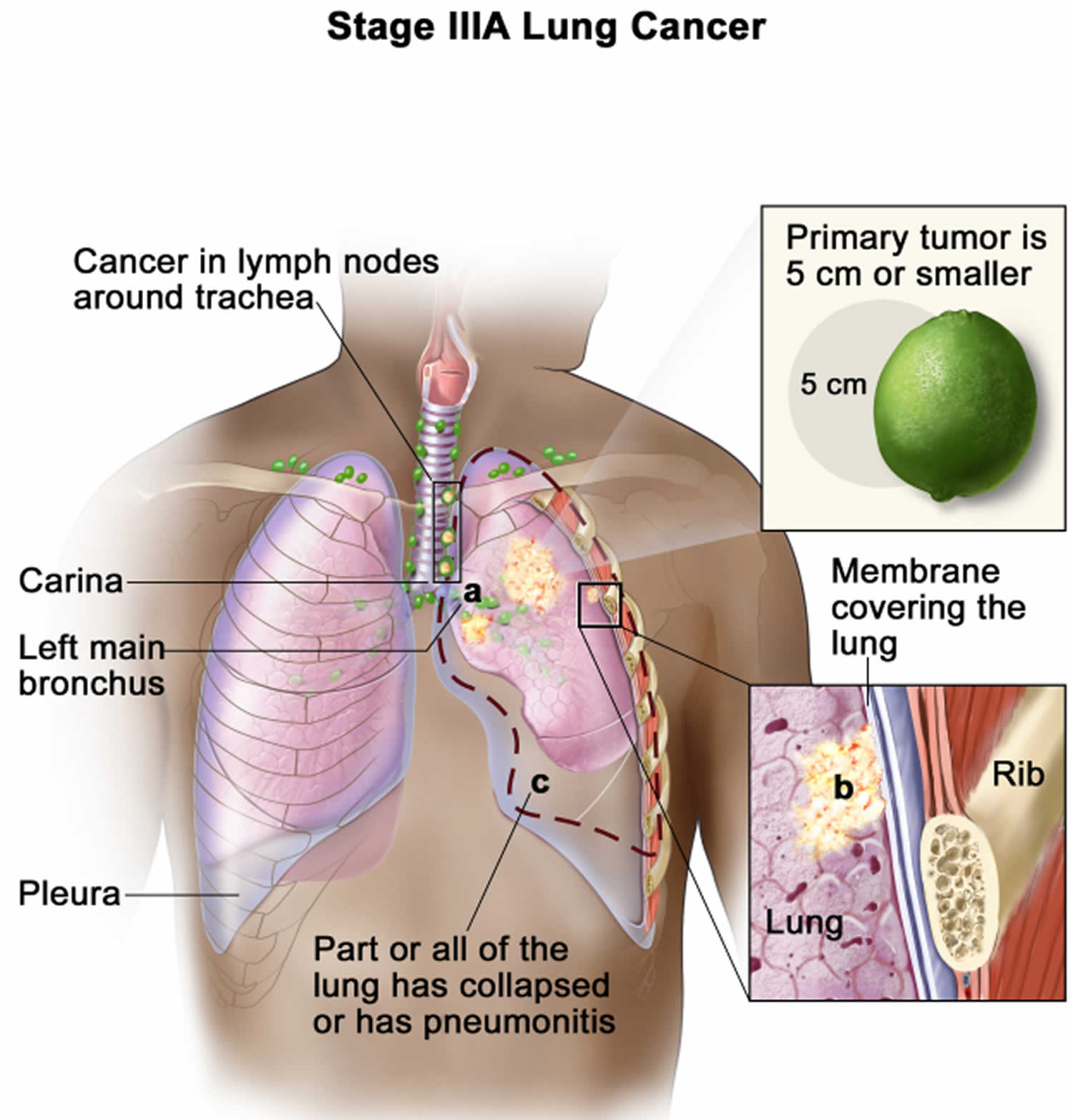
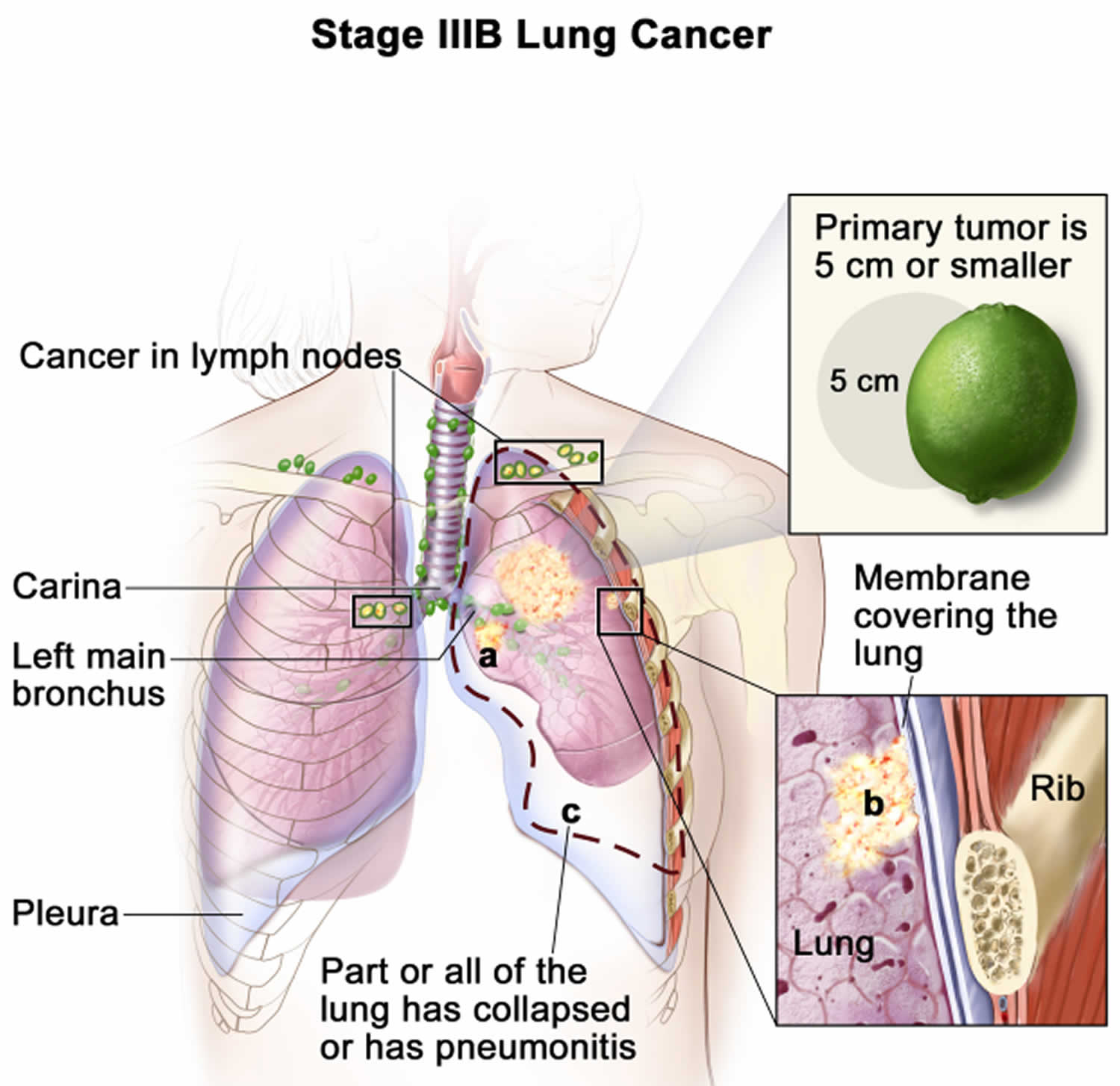
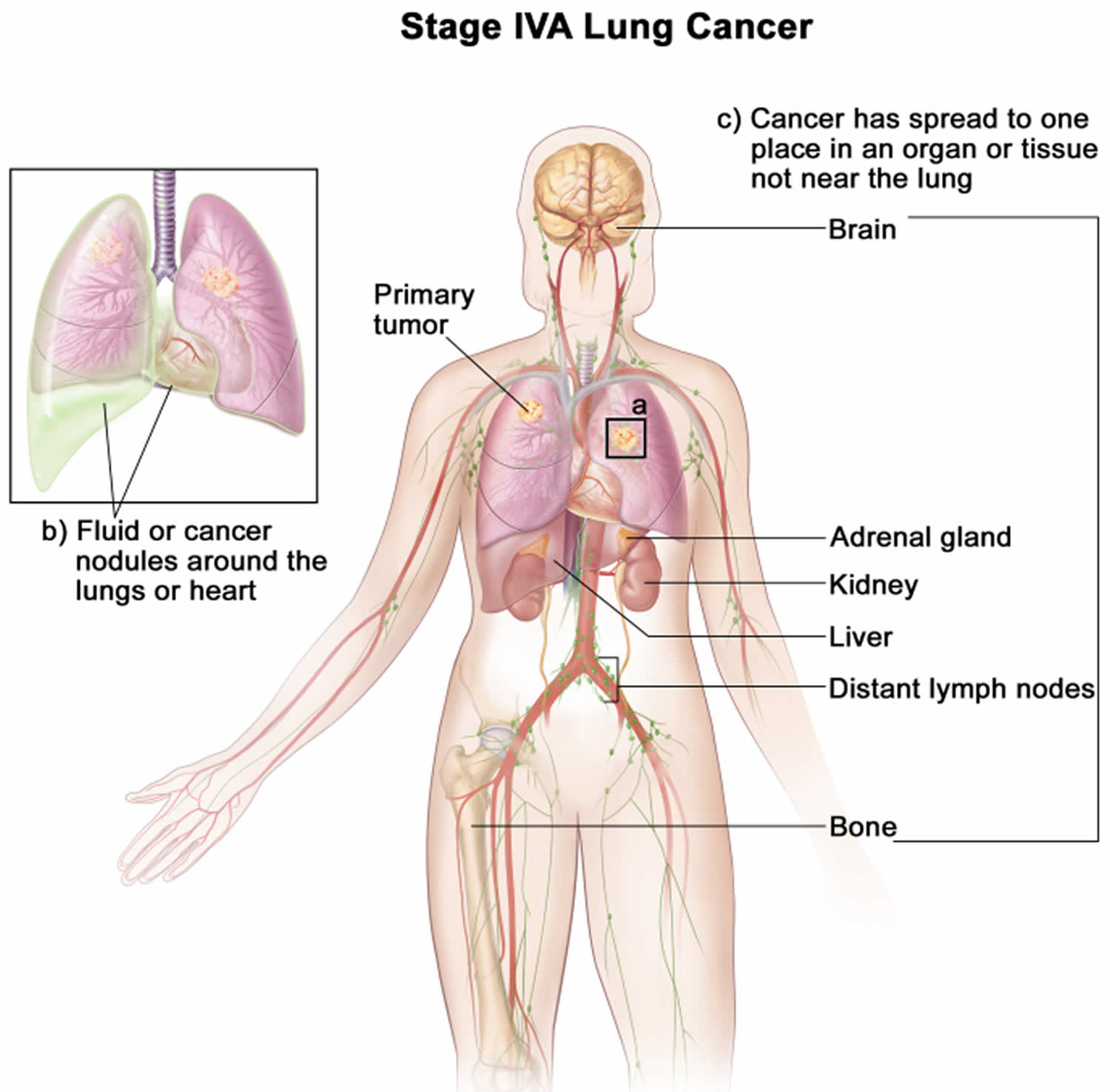
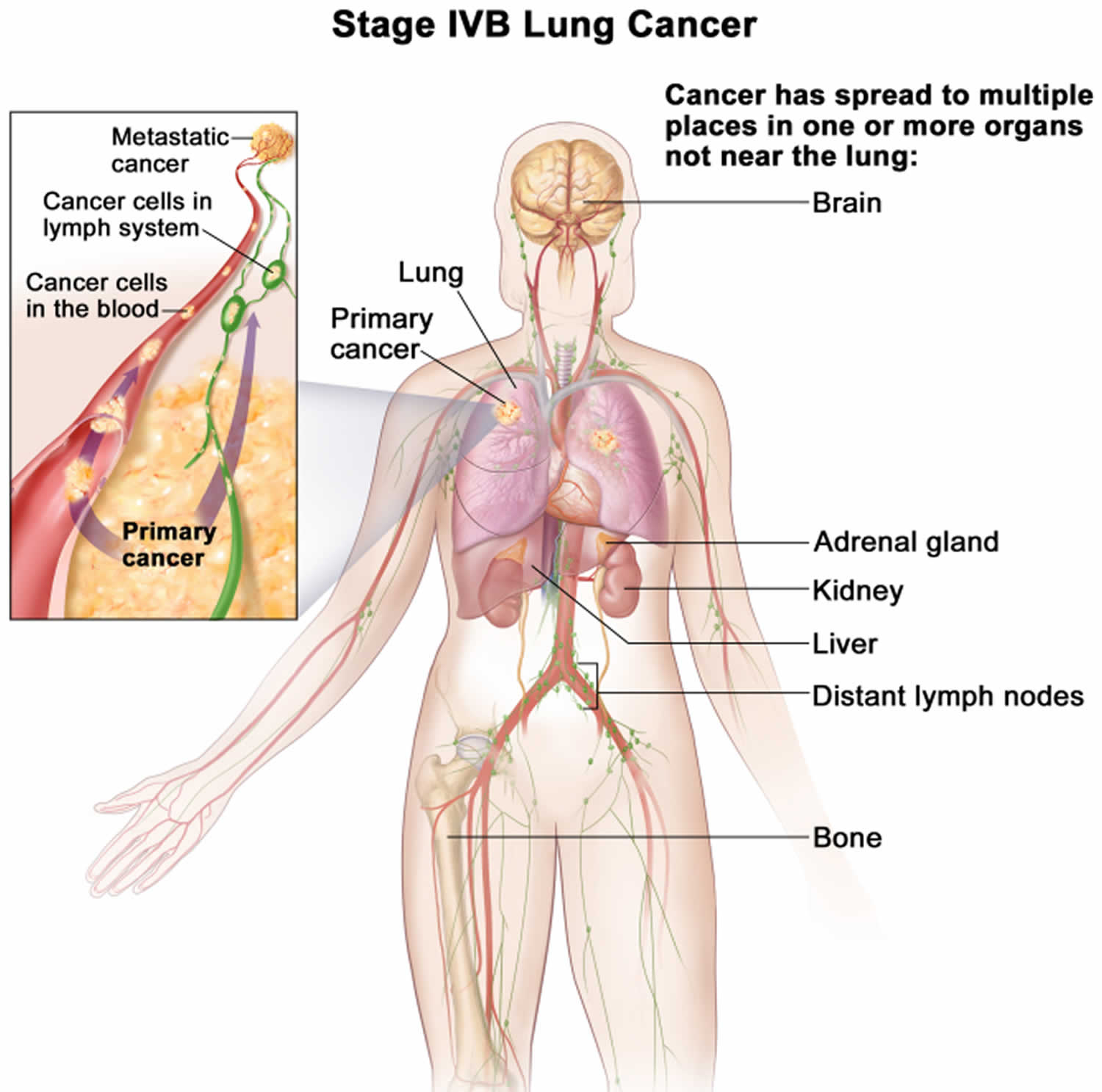
Lung cancer staging
Once your lung cancer has been diagnosed, your doctor will work to determine the stage (extent) of your cancer. The stage of a cancer tells you how big it is and whether it has spread. Your cancer’s stage helps you and your doctor decide what treatment is most appropriate. For example, the best treatment for an early-stage cancer may be surgery or radiation, while a more advanced-stage cancer may need treatments that reach all parts of the body, such as chemotherapy, targeted drug therapy, or immunotherapy.
Staging tests may include imaging procedures that allow your doctor to look for evidence that cancer has spread beyond your lungs. These tests include CT, MRI, positron emission tomography (PET) and bone scans. Sometimes it’s not possible to be certain about the stage of a cancer until after surgery. Not every test is appropriate for every person, so talk with your doctor about which procedures are right for you.
When a cancer is staged again after the initial staging, it is sometimes referred to as restaging. Often the same tests that were done when the cancer was first diagnosed (such as physical exams, imaging tests, endoscopy exams, biopsies, and maybe surgery) are done again.
With any type of restaging, the new stage classification is added to the original stage, but it doesn’t replace it. The stage assigned at diagnosis is still the one that is most important when discussing statistics like survival rates.
A more formal system to describe the growth and spread of lung cancer is the American Joint Committee on Cancer (AJCC) TNM staging system, which is based on 3 key pieces of information:
- The size and extent of the main tumor (T): How large is the tumor? Has it grown into nearby structures or organs?
- The spread to nearby (regional) lymph nodes (N): Has the cancer spread to nearby lymph nodes?
- The spread (metastasis) (M) to other organs of the body: Has the cancer spread to distant organs such as the brain, bones, adrenal glands, liver, or the other lung?
Numbers or letters appear after T, N, and M provide more details about each of these factors. Higher numbers mean the cancer is more advanced. Once the T, N, and M categories have been determined, this information is combined in a process called stage grouping, to assign an overall stage.
The following explains what the letters and numbers mean:
Primary tumor (T)
- TX: Main tumor cannot be measured.
- T0: Main tumor cannot be found.
- T1, T2, T3, T4: Refers to the size and/or extent of the main tumor. The higher the number after the T, the larger the tumor or the more it has grown into nearby tissues. T’s may be further divided to provide more detail, such as T3a and T3b.
Regional lymph nodes (N)
- NX: Cancer in nearby lymph nodes cannot be measured.
- N0: There is no cancer in nearby lymph nodes.
- N1, N2, N3: Refers to the number and location of lymph nodes that contain cancer. The higher the number after the N, the more lymph nodes that contain cancer.
Distant metastasis (M)
- MX: Metastasis cannot be measured.
- M0: Cancer has not spread to other parts of the body.
- M1: Cancer has spread to other parts of the body.
Other ways to describe lung cancer stage
The TNM system helps describe cancer in great detail and the TNM combinations are often grouped into five less-detailed stages. When talking about your cancer, your doctor may describe it as one of these stages:
- Stage 0: The earliest stage (pre-cancer) where abnormal cells are present but have not spread to nearby tissue. Also called carcinoma in situ (CIS) or in-situ carcinoma. Carcinoma in situ (CIS) is not cancer, but it may become cancer. In carcinoma in situ (CIS) the carcinoma cells are only in the top layer of cells of the bronchi, bronchioles, or alveoli, without growing into the deeper layers below.
- Stage 1, Stage 2 and Stage 3: Cancer is present. The higher the number, the larger the cancer tumor and the more it has spread into nearby tissues.
- Stage 4: The cancer has spread (metastasize) outside the lung to lymph nodes and other parts of your body.
As a rule, the lower the stage number, the less the cancer has spread. A higher number, such as stage IV (4), means the cancer is considered advanced and has spread to other areas of the body. And within a stage, an earlier letter (or number) means a lower stage.
The same TNM staging system is used for both small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC), although it’s generally not as important for small cell lung cancer (SCLC).
Another staging system that is used for all types of cancer groups the cancer into one of five main categories. This staging system is more often used by cancer registries than by doctors. But, you may still hear your doctor describe your cancer in one of the following ways:
- In situ:Abnormal cells are present but have not spread to nearby tissue.
- Localized: Cancer is limited to the place where it started, with no sign that it has spread.
- Regional: Cancer has spread to nearby lymph nodes, tissues, or organs.
- Distant: Cancer has spread to distant parts of the body.
- Unknown: There is not enough information to figure out the stage.
Stage 1 lung cancer
Stage 1 is part of the number staging system and means your cancer is small. It hasn’t spread to your lymph nodes or other distant organs.
Stage 1 can be divided into 1A and 1B.
Here is a simplified description:
- Stage 1A means the cancer is 3 cm or smaller.
- Stage 1B means the cancer is between 3 cm and 4 cm.
It might also be growing into structures such as:
- the main airway of the lung (main bronchus)
- the membrane covering the lung (visceral pleura)
- Or the cancer is making the lung partly or completely collapse by blocking the airway.
In the TNM staging system, stage 1A is the same as T1a-c, N0, M0.
Stage 1B is the same as T2a, N0, M0
Stage 1A lung cancer
Footnote: Stage 1A lung cancer. The tumor is in the lung only and is 3 centimeters or smaller. Cancer has not spread to the lymph nodes.
[Source 25 ]Stage 1B lung cancer
Footnote: Stage 1B lung cancer. The tumor is larger than 3 centimeters but not larger than 4 centimeters. Cancer has not spread to the lymph nodes; OR the tumor is 4 centimeters or smaller. Cancer has not spread to the lymph nodes and one or more of the following is found: (a) cancer has spread to the main bronchus, but has not spread to the carina; and/or (b) cancer has spread to the inner membrane that covers the lung; and/or (c) part of the lung or the whole lung has collapsed or has pneumonitis (inflammation of the lung).
[Source 26 ]Stage 2 lung cancer
Stage 2 is part of the number staging system. It can be divided into stage 2A and 2B. Part of the affected lung might have collapsed.
Stage 2 non small cell lung cancer is sometimes called early stage non small cell lung cancer.
- Stage 2A means that the cancer is between 4 cm and 5 cm in size but there are no cancer cells in any lymph nodes.
- Stage 2B means that the cancer is up to 5 cm in size and there are cancer cells in the lymph nodes close to the affected lung.
- Or the cancer is between 5 cm and 7 cm but there are no cancer cells in any lymph nodes.
- Or the cancer is not in any lymph nodes but has spread into one or more of the following areas:
- the chest wall (ribs, muscle or skin)
- the nerve close to the lung (the phrenic nerve)
- the layers that cover the heart (mediastinal pleura and parietal pericardium)
- Or the cancer is less than 7 cm but there is more than one tumor in the same lobe of the lung.
In the TNM staging system stage 2A is the same as T2b, N0, M0
Stage 2B is the same as one of the following:
- T1a-c, N1, M0
- T2a-b, N1, M0
- T3, N0, M0
Stage 2A lung cancer
Footnote: Stage 2A lung cancer. The tumor is larger than 4 centimeters but not larger than 5 centimeters. Cancer has not spread to the lymph nodes and one or more of the following may be found: (a) cancer has spread to the main bronchus, but has not spread to the carina; and/or (b) cancer has spread to the inner membrane that covers the lung; and/or (c) part of the lung or the whole lung has collapsed or has pneumonitis (inflammation of the lung).
[Source 27 ]Stage 2B lung cancer
Footnote: Stage 2B lung cancer. The primary tumor is 5 centimeters or smaller and cancer has spread to the lymph nodes on the same side of the chest as the primary tumor. The lymph nodes with cancer are in the lung or near the bronchus.
[Source 28 ]Stage 3 lung cancer
Stage 3 is part of the number staging system. Stage 3 non small cell lung cancer is sometimes called locally advanced cancer.
Stage 3 can be divided into 3A, 3B and 3C.
- Stage 3A can mean different things.
- Your cancer is up to 5 cm in size and has spread to the lymph nodes in the center of the chest on the same side as the tumor.
- Or the cancer is between 5 cm and 7 cm and there is more than one tumor in the same lobe of the lung.
- Or the cancer has spread into one or more of the following areas just outside the lung:
- the chest wall (ribs, muscle or skin)
- the nerve close to the lung (the phrenic nerve)
- the layers that cover the heart (mediastinal pleura and parietal pericardium)
- lymph nodes in the lung or close to the lung
- Or your cancer is larger than 7 cm. It hasn’t spread into lymph nodes but has spread into one or more of the following areas:
- the muscle under the lung (diaphragm)
- the center area of the chest (mediastinum)
- the heart
- a main blood vessel
- the wind pipe (trachea)
- the nerve that goes to the voice box (larynx)
- the food pipe (oesophagus)
- a spinal bone
- the area where the wind pipe divides (the carina)
- Or your cancer is in more than one lobe of the same lung and there might also be cancer cells in lymph nodes close to the affected lung.
- Stage 3B can also mean different things.
- Your cancer is less than 5cm and has spread into lymph nodes in one of these places:
- the opposite side of the chest from the affected lung
- the neck
- above the collarbone
- Or your cancer is between 5cm to 7 cm and has spread into lymph nodes in the center of the chest.
- Or the cancer is any size, has spread into lymph nodes in the center of the chest, and has spread into one or more of the following areas:
- the chest wall
- the muscle under the lung (diaphragm)
- the layers that cover the heart (mediastinal pleura and parietal pericardium)
- Or stage 3B means that your cancer has spread into the lymph nodes in the center of the chest. The lung tumor is more than 7 cm or it has spread into a major structure in your chest such as:
- the heart
- the wind pipe (trachea)
- the food pipe (oesophagus)
- a main blood vessel
- Your cancer is less than 5cm and has spread into lymph nodes in one of these places:
- Stage 3C can also mean different things.
- Your cancer is between 5 cm and 7 cm in size or has spread into one or more of the following:
- the nerve close to the lung (phrenic nerve)
- the covering of the heart (parietal pericardium)
- and it has spread into lymph nodes:
- in the center of the chest on the opposite side from the affected lung or
- at the top of the lung on the same side or opposite side or
- above the collar bone
- Or there is more than one tumour in a different lobe of the same lung.
- Or stage 3C can mean the cancer is bigger than 7 cm or it has spread into one of the following:
- the muscle under the lung (the diaphragm)
- the center of the chest (mediastinum)
- the heart
- a major blood vessel
- the wind pipe (trachea)
- the nerve going to the voice box (the recurrent laryngeal nerve)
- the food pipe (esophagus)
- a spinal bone
- the area where the windpipe divides (the carina)
- and it has spread into lymph nodes:
- in the center of the chest on the opposite side from the affected lung or
- at the top of the lung on the same side or opposite side or
- above the collar bone
- Or there are tumors in more than one lobe of the lung.
- Your cancer is between 5 cm and 7 cm in size or has spread into one or more of the following:
In the TNM staging system stage 3A is the same as one of the following:
- T1a-c, N2, M0
- T2a-b, N2, M0
- T3, N1, M0
- T4, N0, M0
- T4, N1, M0
Stage 3B is the same as one of the following:
- T1a-c, N3, M0
- T2a-b, N3, M0
- T3, N2, M0
- T4, N2, M0
Stage 3C is the same as:
- T3, N3, M0
- T4, N3, M0
Stage 3A lung cancer
Footnote: Stage 3A lung cancer. The tumor is 5 centimeters or smaller and cancer has spread to lymph nodes on the same side of the chest as the primary tumor. The lymph nodes with cancer are around the trachea or aorta (not shown), or where the trachea divides into the bronchi. Also, one or more of the following may be found: (a) cancer has spread to the main bronchus, but has not spread to the carina; and/or (b) cancer has spread to the inner membrane that covers the lung; and/or (c) part of the lung or the whole lung has collapsed or has pneumonitis (inflammation of the lung).
[Source 29 ]Stage 3B lung cancer
Footnote: Stage 3B lung cancer. The primary tumor is 5 centimeters or smaller and cancer has spread to lymph nodes above the collarbone on the same side of the chest as the primary tumor or to any lymph nodes on the opposite side of the chest as the primary tumor. Also, one or more of the following may be found: (a) cancer has spread to the main bronchus, but has not spread to the carina; and/or (b) cancer has spread to the inner membrane that covers the lung; and/or (c) part of the lung or the whole lung has collapsed or has pneumonitis (inflammation of the lung).
[Source 30 ]Stage 4 lung cancer
Stage 4 lung cancer also called advanced lung cancer, which means the cancer has spread. Stage 4 is divided into stage 4A and 4B.
- Stage 4A can mean any of the following:
- there is cancer in both lungs
- the cancer is in the covering of the lung (the pleura) or the covering of the heart (pericardium)
- or there is fluid around the lungs or the heart that contains cancer cells
- Or it can mean that there is a single area of cancer that has spread outside the chest to a lymph node or to an organ such as the liver or bone.
- Stage 4B means that the cancer has spread to several areas in one or more organs.
In the TNM staging system, stage 4 is the same as one of the following:
- Any T, Any N, M1a
- Any T, Any N, M1b
- Any T, Any N, M1c
Stage 4A lung cancer
Footnote: Stage 4A lung cancer. The tumor may be any size and cancer may have spread to the lymph nodes. One or more of the following is found: (a) there are one or more tumors in the lung that does not have the primary tumor; and/or (b) cancer is found in fluid around the lungs or heart or there are cancer nodules in the lining around the lungs or the sac around the heart; and/or (c) cancer has spread to one place in an organ or tissue not near the lung, such as the brain, adrenal gland, kidney, liver, or bone, or to a lymph node that is not near the lung.
[Source 31 ]Stage 4B lung cancer
Footnote: Stage 4B lung cancer. The cancer has spread to multiple places in one or more organs that are not near the lung, such as the brain, adrenal gland, kidney, liver, distant lymph nodes, or bone.
[Source 32 ]Lung cancer treatment
You and your doctor choose a cancer treatment plan based on a number of factors, such as your overall health, the type and stage of your cancer, the goal of treatment and your preferences. The most suitable treatment will also depend on whether the cancer started in your lung (primary) or spread from another part of your body (metastasis). In the case of a metastasis to the lung, the treatment is usually chosen based on the location of the primary cancer.
Types of lung cancer treatments can include:
- Surgery, to cut out the cancer — this includes a lobectomy where one lobe of a lung is removed, a pneumonectomy, where one whole lung is removed, or a wedge resection where only part of a lung is removed
- Radiotherapy, which damages cancer cells and stops them dividing or spreading
- Chemotherapy, which uses strong medicines to kill cancer cells
- Targeted therapy, which uses medicine that attacks specific features of a cancer — for example, certain genetic mutations
- Immunotherapy, which helps your immune system see cancer cells and kill them
Your medical team may recommend one or a combination of these treatments, depending on your situation.
Cancer treatment may be given with the aim of removing all signs of cancer (curative). In other cases, the goal may be to relieve symptoms and maximise quality of life (palliative care) without necessarily curing the cancer completely.
In some cases, you may choose not to undergo treatment. For instance, you may feel that the side effects of treatment will outweigh the potential benefits. When that’s the case, your doctor may suggest comfort care to treat only the symptoms the cancer is causing, such as pain or shortness of breath.
Surgery
During surgery, your surgeon works to remove the lung cancer and a margin of healthy tissue. Procedures to remove lung cancer include:
- Wedge resection to remove a small section of lung that contains the tumor along with a margin of healthy tissue
- Segmental resection to remove a larger portion of lung, but not an entire lobe
- Lobectomy to remove the entire lobe of one lung
- Pneumonectomy to remove an entire lung
If you undergo surgery, your surgeon may also remove lymph nodes from your chest in order to check them for signs of cancer.
Surgery may be an option if your cancer is confined to the lungs. If you have a larger lung cancer, your doctor may recommend chemotherapy or radiation therapy before surgery in order to shrink the cancer. If there’s a risk that cancer cells were left behind after surgery or that your cancer may recur, your doctor may recommend chemotherapy or radiation therapy after surgery.
Radiation therapy
Radiation therapy uses high-powered energy beams from sources such as X-rays and protons to kill cancer cells. During radiation therapy, you lie on a table while a machine moves around you, directing radiation to precise points on your body.
For people with locally advanced lung cancer, radiation may be used before surgery or after surgery. It’s often combined with chemotherapy treatments. If surgery isn’t an option, combined chemotherapy and radiation therapy may be your primary treatment.
For advanced lung cancers and those that have spread to other areas of the body, radiation therapy may help relieve symptoms, such as pain.
Chemotherapy
Chemotherapy uses drugs to kill cancer cells. One or more chemotherapy drugs may be given through a vein in your arm (intravenously) or taken orally. A combination of drugs usually is given in a series of treatments over a period of weeks or months, with breaks in between so that you can recover.
Chemotherapy is often used after surgery to kill any cancer cells that may remain. It can be used alone or combined with radiation therapy. Chemotherapy may also be used before surgery to shrink cancers and make them easier to remove.
In people with advanced lung cancer, chemotherapy can be used to relieve pain and other symptoms.
Stereotactic body radiotherapy
Stereotactic body radiotherapy, also known as radiosurgery, is an intense radiation treatment that aims many beams of radiation from many angles at the cancer. Stereotactic body radiotherapy treatment is typically completed in one or a few treatments.
Stereotactic body radiotherapy may be an option for people with small lung cancers who can’t undergo surgery. It may also be used to treat lung cancer that spreads to other parts of the body, including the brain.
Targeted drug therapy
Targeted drug treatments focus on specific abnormalities present within cancer cells. By blocking these abnormalities, targeted drug treatments can cause cancer cells to die.
Many targeted therapy drugs are used to treat lung cancer, though most are reserved for people with advanced or recurrent cancer.
Some targeted therapies only work in people whose cancer cells have certain genetic mutations. Your cancer cells may be tested in a laboratory to see if these drugs might help you.
Immunotherapy
Immunotherapy uses your immune system to fight cancer. Your body’s disease-fighting immune system may not attack your cancer because the cancer cells produce proteins that help them hide from the immune system cells. Immunotherapy works by interfering with that process.
Immunotherapy treatments are generally reserved for people with locally advanced lung cancers and cancers that have spread to other parts of the body.
Alternative medicine
Complementary and alternative lung cancer treatments can’t cure your cancer. But complementary and alternative treatments can often be combined with your doctor’s care to help relieve signs and symptoms.
The American College of Chest Physicians suggests people with lung cancer may find comfort in:
- Acupuncture. During an acupuncture session, a trained practitioner inserts small needles into precise points on your body. Acupuncture may relieve pain and ease cancer treatment side effects, such as nausea and vomiting, but there’s no evidence that acupuncture has any effect on your cancer.
- Hypnosis. Hypnosis is typically done by a therapist who leads you through relaxation exercises and asks you to think pleasing and positive thoughts. Hypnosis may reduce anxiety, nausea and pain in people with cancer.
- Massage. During a massage, a massage therapist uses his or her hands to apply pressure to your skin and muscles. Massage can help relieve anxiety and pain in people with cancer. Some massage therapists are specially trained to work with people who have cancer.
- Meditation. Meditation is a time of quiet reflection in which you focus on something, such as an idea, image or sound. Meditation may reduce stress and improve quality of life in people with cancer.
- Yoga. Yoga combines gentle stretching movements with deep breathing and meditation. Yoga may help people with cancer sleep better.
Palliative care
People with lung cancer often experience signs and symptoms of the cancer, as well as side effects of treatment. Supportive care, also known as palliative care, is a specialty area of medicine that involves working with a doctor to minimize your signs and symptoms. The palliative care team’s goal is to improve quality of life for you and your family.
Your doctor may recommend that you meet with a palliative care team soon after your diagnosis to ensure that you’re comfortable during and after your cancer treatment.
Palliative care specialists work with you, your family and your care team. They provide an extra layer of support while you have cancer treatment. You can have palliative care at the same time you’re getting strong cancer treatments, such as surgery, chemotherapy or radiation therapy.
In one study, people with advanced non-small cell lung cancer who began receiving supportive care soon after their diagnosis lived longer than those who continued with treatments, such as chemotherapy and radiation. Those receiving supportive care reported improved mood and quality of life. They survived, on average, almost three months longer than did those receiving standard care.
Lung cancer prognosis
The expected prognosis (outcome or outlook) of your lung cancer will depend on its type, its stage and how well it responds to treatment.
Non-small cell lung cancer prognosis
The tumor (T), spread to nearby lymph nodes (N) and spread to distant sites (M) also called the TNM stage, at presentation in patients with non small cell lung cancer (NSCLC) has the most significant impact on prognosis. A higher TNM stage correlates with advanced disease and poor outcomes. Other factors indicative of poor prognosis include performance status at the time of diagnosis, anorexia, weight loss, and the presence of liver or skin metastases 33. Molecular studies have revealed that patients with activating mutations of EGFR in patients with adenocarcinoma have a better prognosis than those without EGFR mutations 34.
Table 1. 5-year relative survival rates for non-small cell lung cancer
| National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) database stage | 5-year relative survival rate |
|---|---|
| Localized | 64% |
| Regional | 37% |
| Distant | 8% |
| All SEER stages combined | 26% |
Footnotes: The SEER database tracks 5-year relative survival rates for non-small cell lung cancer (NSCLC) in the United States, based on how far the cancer has spread. The SEER database, however, does not group cancers by American Joint Committee on Cancer (AJCC) TNM stages (stage 1, stage 2, stage 3, etc.). Instead, the SEER database groups cancers into localized, regional, and distant stages:
- Localized: There is no sign that the cancer has spread outside of the lung.
- Regional: The cancer has spread outside the lung to nearby structures or lymph nodes.
- Distant: The cancer has spread to distant parts of the body, such as the brain, bones, liver, or the other lung.
Understanding the numbers
- These numbers apply only to the stage of the cancer when it is first diagnosed. They do not apply later on if the cancer grows, spreads, or comes back after treatment.
- These numbers don’t take everything into account. Survival rates are grouped based on how far the cancer has spread. But other factors, such as the subtype of non-small cell lung cancer (NSCLC), gene changes in the cancer cells, your age and overall health, and how well the cancer responds to treatment, can also affect your outlook.
Small cell lung cancer prognosis
The extent of disease and the stage at presentation is the most important prognostic factor for small cell lung cancer (SCLC). Patients with the localized-stage disease have a five-year survival rate of 29%, whereas patients with the extensive-stage disease have a five-year survival rate of 3% 36. Similar to non small cell lung cancer, performance status and weight loss are independent prognostic factors for small cell lung cancer, where patients with poor performance status and/or weight loss at the time of diagnosis have a decreased survival rate.
Table 2. 5-year relative survival rates for small cell lung cancer
| National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) database stage | 5-year relative survival rate |
|---|---|
| Localized | 29% |
| Regional | 18% |
| Distant | 3% |
| All SEER stages combined | 7% |
Footnotes: The SEER database tracks 5-year relative survival rates for small cell lung cancer (SCLC) in the United States, based on how far the cancer has spread. The SEER database, however, does not group cancers by American Joint Committee on Cancer (AJCC) TNM stages (stage 1, stage 2, stage 3, etc.). Instead, the SEER database groups cancers into localized, regional, and distant stages:
- Localized: There is no sign that the cancer has spread outside of the lung.
- Regional: The cancer has spread outside the lung to nearby structures or lymph nodes.
- Distant: The cancer has spread to distant parts of the body, such as the brain, bones, liver, or the other lung.
Understanding the numbers
- These numbers apply only to the stage of the cancer when it is first diagnosed. They do not apply later on if the cancer grows, spreads, or comes back after treatment.
- These numbers don’t take everything into account. Survival rates are grouped based on how far the cancer has spread. But other factors, such as gene changes in the cancer cells, your age and overall health, and how well the cancer responds to treatment, can also affect your outlook.
- People now being diagnosed with small cell lung cancer (SCLC) may have a better outlook than these numbers show. Treatments have improved over time, and these numbers are based on people who were diagnosed and treated at least five years earlier.
Non-Small Cell Lung Cancer
Non-small cell lung cancer (NSCLC) is an umbrella term for several types of lung cancers 6, 37. Non-small cell lung cancer (NSCLC) is any type of epithelial lung cancer other than small cell lung cancer (SCLC). Non-small cell lung cancer arises from the epithelial cells of the lung of the central bronchi to terminal alveoli 37. Non-small cell lung cancer (NSCLC) is the most common type of lung cancer accounting for about 80% to 85% of all lung cancer cases, and it’s characterized by slower growth and spread compared to small cell lung cancer (SCLC) 38, 39. Squamous cell carcinoma, adenocarcinoma, and large cell carcinoma are all subtypes of non-small cell lung cancer (Figure 1), but there are several other types that occur less frequently and all types can occur in unusual histologic variants. The histological type of non-small cell lung cancer (NSCLC) correlates with site of origin, reflecting the variation in respiratory tract epithelium of the bronchi to alveoli. Squamous cell carcinoma usually starts near a central bronchus. Adenocarcinoma and bronchioloalveolar carcinoma usually originate in peripheral lung tissue. Adenocarcinoma is the most common and often occurs in the outer parts of the lung, frequently affecting non-smokers, and younger people. Squamous cell carcinoma usually develops in the central parts of the lungs and is closely linked to smoking. Large cell carcinoma is rarer, more aggressive, and tends to spread quickly 40. Each subtype has unique features that help in diagnosing and treating the non-small cell lung cancer complex 41.
Non-small cell lung cancers are grouped together because they behave in a similar way and respond to treatment in a similar way. As a class, non-small cell lung cancers (NSCLCs) are relatively insensitive to chemotherapy and radiation therapy compared with small cell lung cancer (SCLC). Patients with resectable disease may be cured by surgery or surgery followed by chemotherapy. Local control can be achieved with radiation therapy in a large number of patients with unresectable disease, but cure is seen only in a small number of patients. Patients with locally advanced unresectable disease may achieve long-term survival with radiation therapy combined with chemotherapy. Patients with advanced metastatic disease may achieve improved survival and palliation of symptoms with chemotherapy, targeted agents, and other supportive measures 42.
The 5-year relative survival rate from 2014 to 2020 for patients with lung cancer was 26.7% 8. The 5-year relative survival rate for patients with local-stage (63.7%), regional-stage (35.9%), and distant-stage (8.9%) disease varies markedly, depending on the stage at diagnosis 8. However, early-stage non–small cell lung cancer (NSCLC) has a better prognosis and can be treated with surgical resection.
Smoking is the main cause of non-small cell lung cancer (NSCLC), accounting for about 85% of lung cancer cases 6. Tobacco smoke contains many carcinogens that damage DNA, causing genetic mutations and cancer 6. The risk of non-small cell lung cancer is closely linked to how long and how much a person smokes, and it remains high even years after quitting. Additionally, exposure to secondhand smoke significantly raises the risk of lung cancer in non-smokers 43.
Although non-small cell lung cancers are associated with cigarette smoke, adenocarcinomas may be found in patients who have never smoked 44. Environmental factors are also a major risk factor for non-small cell lung cancer. Exposure to harmful substances at work, such as asbestos, arsenic, chromium, and diesel exhaust, increases the risk of lung cancer 45:S31–S49. doi: 10.21037/tlcr.2019.03.05)). Radon gas, a naturally occurring radioactive gas found in soil and building materials, is the second leading cause of lung cancer after smoking 6. Air pollution, particularly fine particulate matter (PM2.5), is also linked to higher rates of lung cancer, especially in urban areas with heavy traffic and industrial activity 46.
Genetic factors significantly impact the likelihood of developing non-small cell lung cancer. Having a family history of lung cancer increases the risk, indicating a hereditary aspect. Certain genetic variations and mutations, like those in the p53 tumor suppressor gene, are associated with a higher risk of non-small cell lung cancer 6. The development of non-small cell lung cancer involves several genetic mutations and changes in cell growth, differentiation, and death pathways. Additionally, specific mutations such as EGFR, ALK, ROS1, and KRAS genes, which promote the uncontrolled growth and spread of non-small cell lung cancer cells but also serve as targets for personalized treatments 47, 48. Understanding these molecular mechanisms is crucial for creating targeted treatments that block these pathways and improve patient outcomes 49. Ongoing research aims to identify genetic risk factors for lung cancer to find high-risk individuals and develop preventive strategies 50.
Early-stage non-small cell lung cancer usually shows no symptoms or has vague symptoms like a persistent cough, chest pain, shortness of breath, and unexplained weight loss, which can delay diagnosis 6. Diagnostic tools such as chest X-rays, CT scans, and PET scans are vital for detecting and determining the stage of the non-small cell lung cancer 51. Biopsy and histopathological examination are essential for confirming the diagnosis and identifying the subtype. The treatment for non-small cell lung cancer has changed a lot in the past decade. Traditional treatments like surgery, radiation, and chemotherapy are still important for managing the disease. However, new targeted therapies and immunotherapies have significantly improved non-small cell lung cancer treatment 52.
Targeted therapies, such as tyrosine kinase inhibitors (TKIs) for specific genetic mutations like EGFR and ALK, offer a personalized approach that boosts survival and quality of life. Immunotherapies, including immune checkpoint inhibitors like pembrolizumab and nivolumab, have proven effective in strengthening the body’s immune response against cancer cells, resulting in lasting benefits for some patients 53. Despite these treatment advances, there are still challenges in treating non-small cell lung cancer. Issues such as treatment resistance, the diversity of the disease, and limited access to advanced therapies remain significant hurdles 54. Research is ongoing to better understand resistance mechanisms, find new treatment targets, and develop combination therapies to address these challenges. Additionally, early detection through better screening methods, such as low-dose CT (LDCT) scans for high-risk individuals, and the use of molecular diagnostics like liquid biopsies, are crucial for improving patient outcomes and survival rates 55.
If you have non-small cell lung cancer (NSCLC) or are close to someone who does, knowing what to expect can help you cope. Here you can find out all about non-small cell lung cancer (NSCLC), including risk factors, symptoms, how it is found, and how it is treated.
If you have non-small cell lung cancer or are close to someone who does, knowing what to expect can help you cope. Here you can find out all about non-small cell lung cancer, including risk factors, symptoms, how it is found, and how it is treated.
Figure 9. Non-small cell lung cancer
Footnote: Non–small cell lung cancer. A cavitating right lower lobe squamous cell carcinoma.
[Source 56 ]Types of non-small cell lung cancer
There are subtypes of non-small cell lung cancer, which start from different types of lung cells. But they are grouped together as non-small cell lung cancer (NSCLC) because the approach to treatment and prognosis (outlook) are often similar.
Adenocarcinoma
About 40% of lung cancers are adenocarcinomas. Adenocarcinomas in the cells that would normally secrete substances such as mucus.
This type of lung cancer occurs mainly in people who currently smoke or former smokers, but it is also the most common type of lung cancer seen in people who don’t smoke. It is more common in women than in men, and it is more likely to occur in younger people than other types of lung cancer.
Adenocarcinoma is usually found in outer parts of the lung. Though it tends to grow slower than other types of lung cancer and is more likely to be found before it has spread, this varies from patient to patient.
The following variants of adenocarcinoma are recognized in the International Association for the Study of Lung Cancer classification:
- Well-differentiated fetal adenocarcinoma.
- Mucinous (colloid) adenocarcinoma.
- Mucinous cystadenocarcinoma.
- Signet ring adenocarcinoma.
- Clear cell adenocarcinoma.
People with a type of adenocarcinoma called adenocarcinoma in situ (previously called bronchioloalveolar carcinoma) tend to have a better outlook than those with other types of lung cancer.
Squamous cell (epidermoid) carcinoma
About 25% to 30% of all lung cancers are squamous cell carcinomas. Squamous cell carcinomas start in early versions of squamous cells, which are flat cells that line the inside of the airways in the lungs. Squamous cell carcinomas are often linked to a history of smoking and tend to be found in the central part of the lungs, near a main airway (bronchus). The incidence of squamous cell carcinoma of the lung has been decreasing in recent years.
Large cell (undifferentiated) carcinoma
Large cell (undifferentiated) carcinoma accounts for about 10% to 15% of lung cancers. Large cell carcinoma can appear in any part of the lung. The cancer cells appear large and round under the microscope. Large cell carcinoma tends to grow and spread quickly, which can make it harder to treat. A subtype of large cell carcinoma, known as large cell neuroendocrine carcinoma (LCNEC), is a fast-growing cancer that is very similar to small cell lung cancer.
Several uncommon large cell carcinoma variants are recognized in the International Association for the Study of Lung Cancer classification, including the following:
- Large cell neuroendocrine carcinoma (LCNEC).
- Basaloid carcinoma.
- Lymphoepithelioma-like carcinoma.
- Clear cell carcinoma.
- Large cell carcinoma with rhabdoid phenotype.
Basaloid carcinoma is a recognized as a variant of squamous cell carcinoma, and rarely, adenocarcinomas may have a basaloid pattern; however, in tumors without either of these features, they are regarded as a variant of large cell carcinoma.
Large cell neuroendocrine carcinoma (LCNEC) is recognized as a histologically high-grade non-small cell carcinoma. It has a very poor prognosis similar to that of small cell lung cancer (SCLC). Atypical carcinoid is recognized as an intermediate-grade neuroendocrine tumor with a prognosis that falls between typical carcinoid and high-grade small cell lung cancer and large cell neuroendocrine carcinoma (LCNEC).
Other subtypes
A few other subtypes of non-small cell lung cancer (NSCLC), such as adenosquamous carcinoma and sarcomatoid carcinoma, are much less common.
Spindle cell carcinomas and giant cell carcinomas comprise only 0.4% of all lung cancers, and carcinosarcomas comprise only 0.1% of all lung malignancies. In addition, this group of tumors reflects a continuum in histologic heterogeneity, as well as epithelial and mesenchymal differentiation. On the basis of clinical and molecular data, biphasic pulmonary blastoma is regarded as part of the spectrum of carcinomas with pleomorphic, sarcomatoid, or sarcomatous elements.
Histopathology of non-small cell lung cancer
Non-small cell lung cancer is a heterogeneous aggregate of histologies. The most common histologies include the following:
- Epidermoid or squamous cell carcinoma.
- Adenocarcinoma.
- Large cell carcinoma.
The histological type of non-small cell lung cancer correlates with site of origin, reflecting the variation in respiratory tract epithelium of the bronchi to alveoli. Squamous cell carcinoma usually starts near a central bronchus. Adenocarcinoma and bronchioloalveolar carcinoma usually originate in peripheral lung tissue. These histologies are often classified together because approaches to diagnosis, staging, prognosis, and treatment are similar.
Non-small cell lung cancer causes
The most well-known cause for non-small cell lung cancer is tobacco smoking 57, 58, 9, 6, 37. Other causes of lung cancer include alcohol use, environmental exposure to secondhand smoke, asbestos, radon, arsenic, chromium, nickel, as well as exposure to ionizing radiation, and polycyclic aromatic hydrocarbons 59. Radiation therapy can also cause primary lung cancer when it is utilized for the treatment of other malignancies such as breast cancer 60 and Hodgkin lymphoma 61.
Patients with pulmonary fibrosis have been found to have an approximately sevenfold increase in the risk of developing lung cancer, and this has been shown to be independent of tobacco use 62. The incidence of lung cancer in patients with human immunodeficiency virus (HIV) has also been found to be increased compared to the uninfected population, and this has been shown to be independent of smoking status or antiretroviral therapy use in the HIV population 63.
Smoking-related lung carcinogenesis is a multistep process. Squamous cell carcinoma and adenocarcinoma have defined premalignant precursor lesions. Before becoming invasive, lung epithelium may undergo morphological changes that include the following:
- Hyperplasia: the enlargement of an organ or tissue caused by an increase in the reproduction rate of its cells, as an initial stage in the development of cancer.
- Metaplasia: abnormal change in the nature of a tissue.
- Dysplasia: the abnormal growth or development of cells of a tissue or organ.
- Carcinoma in situ: is a group of abnormal cells that are found only in the place where they first formed in the body.
Dysplasia and carcinoma in situ are considered the principal premalignant lesions because they are more likely to progress to invasive cancer and less likely to spontaneously regress.
In addition, after resection of a lung cancer, there is a 1% to 2% risk per patient per year that a second lung cancer will occur 64.
Risk Factors for non-small cell lung cancer
Increasing age is the most important risk factor for most cancers. Other risk factors for lung cancer include:
- Current or history of tobacco use: cigarettes, pipes, and cigars 65.
- Exposure to cancer-causing substances in secondhand smoke 66, 67.
- Occupational exposure to asbestos, arsenic, chromium, beryllium, nickel, and other agents 68.
- Radiation exposure from any of the following:
- Living in an area with air pollution 73, 74, 75.
- Family history of lung cancer 76.
- Human immunodeficiency virus infection (HIV) 77.
- Beta carotene supplements in heavy smokers 78, 79.
The single most important risk factor for the development of lung cancer is smoking. For smokers, the risk for lung cancer is on average tenfold higher than in lifetime nonsmokers (defined as a person who has smoked <100 cigarettes in his or her lifetime). The risk increases with the quantity of cigarettes, duration of smoking, and starting age.
Smoking cessation results in a decrease in precancerous lesions and a reduction in the risk of developing lung cancer. Former smokers continue to have an elevated risk for lung cancer for years after quitting. Asbestos exposure may exert a synergistic effect of cigarette smoking on the lung cancer risk 80.
Genetic and Molecular Alterations in Non-Small Cell Lung Cancer
Non-small cell lung cancer is defined by many genetic and molecular changes that cause its development. These changes impact different pathways within cells that control growth, survival, and how cells mature. Common mutations in non-small cell lung cancer involve genes like EGFR, KRAS, and ALK, which are crucial for cell communication and growth. There are also changes in genes like PIK3CA, BRAF, and MET that add to the formation and progression of tumors 81. Epigenetic changes, like DNA methylation and histone modifications, also control how genes are expressed and how tumors behave. The TP53 and RB1 tumor suppressor genes are commonly turned off, which lets cells grow uncontrollably and causes genetic instability 82. Advances in next-generation sequencing and other molecular diagnostic techniques have enabled the identification of these alterations, allowing for personalized treatment strategies that target specific genetic abnormalities 83. Therefore, knowing the intricate genetic makeup of non-small cell lung cancer is vital for creating new targeted therapies and making better outcomes for patients.
Epidermal Growth Factor Receptor (EGFR)
A notable portion of non-small cell lung cancer, especially in adenocarcinoma and non-smokers, has activating mutations in the EGFR gene. These mutations are most common in specific parts (exons 18–21) of the EGFR gene, causing continuous activation of EGFR tyrosine kinase 84. This leads to uncontrolled cell growth and survival. To counter this, targeted therapies like erlotinib and gefitinib, known as tyrosine kinase inhibitors (TKIs), have been created to block EGFR activity. These treatments have proven highly effective in patients with non-small cell lung cancer that have EGFR mutations 85:S24–S31. doi: 10.1038/onc.2009.198)).
Anaplastic Lymphoma Kinase (ALK)
A small percentage of non-small cell lung cancer cases have rearrangements in the ALK gene, like the EML4-ALK fusion, mainly seen in non-smokers and younger patients. These rearrangements create an ALK tyrosine kinase that is always active 86. This constant activity triggers pathways that drive cell growth and survival. ALK inhibitors such as crizotinib and alectinib have shown significant benefits for patients with non-small cell lung cancer that have ALK rearrangements 87.
KRAS
KRAS mutations are quite common in non-small cell lung cancer, especially in adenocarcinomas and smokers. These mutations often occur in specific parts (codons 12, 13, and 61) of the KRAS gene. They activate pathways like MAPK and PI3K-AKT, promoting cell growth, survival, and resistance to cell death 88. Developing effective targeted therapies for KRAS mutations has been challenging, but progress has been made with KRAS G12C inhibitors like sotorasib and adagrasib, showing promise in clinical trials and providing new hope for patients with this mutation 89.
ROS1
Rearrangements in the ROS1 gene, like those in ALK, create fusion proteins that are always active and promote cancerous signaling. Treating ROS1-rearranged non-small cell lung cancer with ROS1 inhibitors like crizotinib has been effective. Newer ROS1 inhibitors such as entrectinib and lorlatinib are also in development to tackle resistance mechanisms and enhance outcomes for patients 90.
BRAF
BRAF mutations, especially V600E are present in a subset of non-small cell lung cancer cases. Drugs called BRAF inhibitors, such as vemurafenib and dabrafenib, target these mutations and have proven effective in treating BRAF-mutant non-small cell lung cancer. Combining BRAF inhibitors with MEK inhibitors, like trametinib, has also improved treatment outcomes by blocking multiple points in the signaling pathway 91.
MET
MET gene amplifications and exon 14 skipping mutations play a role in the development of non-small cell lung cancer by activating the MET receptor tyrosine kinase. Drugs known as MET inhibitors, such as crizotinib and capmatinib, are used to target these changes. Newer treatments like tepotinib have shown significant benefits in clinical trials and are now being included in treatment plans for patients with MET-driven non-small cell lung cancer 92.
Non-small cell lung cancer prevention
A significant number of patients cured of their smoking-related lung cancer may develop a second malignancy. In the Lung Cancer Study Group trial of 907 patients with stage T1, N0 resected tumors, the rate was 1.8% per year for nonpulmonary second cancers and 1.6% per year for new lung cancers 93. Other studies have reported even higher risks of second tumors in long-term survivors, including rates of 10% for second lung cancers and 20% for all second cancers 94.
Because of the persistent risk of developing second lung cancers in former smokers, various chemoprevention strategies have been evaluated in randomized control trials. None of the phase III trials with the agents beta carotene, retinol, 13-cis-retinoic acid, [alpha]-tocopherol, N-acetylcysteine, or acetylsalicylic acid has demonstrated beneficial, reproducible results 79, 95, 96, 97, 98. Chemoprevention of second primary cancers of the upper aerodigestive tract is undergoing clinical evaluation in patients with early-stage lung cancer.
Can Non-Small Cell Lung Cancer Be Found Early?
Usually symptoms of lung cancer do not appear until the disease is already at an advanced stage 23. Even when lung cancer does cause symptoms, many people may mistake them for other problems, such as an infection or long-term effects from smoking. This may delay the diagnosis.
Some lung cancers are found early by accident as a result of tests for other medical conditions. For example, lung cancer may be found by tests done for other reasons in people with heart disease, pneumonia, or other lung conditions. A small portion of these people do very well and may be cured of lung cancer.
Screening is the use of tests or exams to find a disease in people who don’t have symptoms. Doctors have looked for many years for a good screening test for lung cancer, but only in recent years has a study shown that a test known as a low-dose CT (low dose helical CT) scan can help lower the risk of dying from this disease.
The National Lung Screening Trial was a large clinical trial that looked at using low-dose CT (low dose helical CT) of the chest to screen for lung cancer. CT scans of the chest provide more detailed pictures than chest x-rays and are better at finding small abnormal areas in the lungs. Low-dose CT of the chest uses lower amounts of radiation than a standard chest CT and does not require the use of intravenous (IV) contrast dye.
The National Lung Screening Trial compared low-dose CT of the chest to chest x-rays in people at high risk of lung cancer to see if these scans could help lower the risk of dying from lung cancer. The study included more than 50,000 people ages 55 to 74 who were current or former smokers and were in fairly good health. To be in the study, they had to have at least a 30 pack-year history of smoking.
A pack-year is the number of cigarette packs smoked each day multiplied by the number of years a person has smoked. Someone who smoked a pack of cigarettes per day for 30 years has a 30 pack-year smoking history, as does someone who smoked 2 packs a day for 15 years.
Former smokers could enter the study if they had quit within the past 15 years. The study did not include people if they had a history of lung cancer or lung cancer symptoms, if they had part of a lung removed, if they needed to be on oxygen at home to help them breathe, or if they had other serious medical problems.
People in the study got either 3 low-dose CT scans or 3 chest x-rays, each a year apart, to look for abnormal areas in the lungs that might be cancer. After several years, the study found that people who got low-dose CT had a 20% lower chance of dying from lung cancer than those who got chest x-rays. They were also 7% less likely to die overall (from any cause) than those who got chest x-rays.
Screening with low-dose CT scan was also shown to have some downsides that need to be considered. One drawback of this test is that it also finds a lot of abnormalities that have to be checked out with more tests, but that turn out not to be cancer. About 1 out of 4 people in the National Lung Screening Trial had such a finding. This may lead to additional tests such as other CT scans or more invasive tests such as needle biopsies or even surgery to remove a portion of lung in some people. These tests can sometimes lead to complications (like a collapsed lung) or rarely, death, even in people who do not have cancer (or who have very early stage cancer).
Low-dose CT scans also expose people to a small amount of radiation with each test. It is less than the dose from a standard CT, but it is more than the dose from a chest x-ray. Some people who are screened may end up needing further CT scans, which means more radiation exposure. When done in tens of thousands of people, this radiation may cause a few people to develop breast, lung, or thyroid cancers later on.
The National Lung Screening Trial was a large study, but it left some questions that still need to be answered. For example, it’s not clear if screening with low-dose CT scans would have the same effect if different people were allowed in the study, such as those who smoke less (or not at all), or people younger than age 55 or older than 74. Also, in the National Lung Screening Trial, patients got 3 scans over 2 years. It’s not yet clear what the effect would be if people were screened for longer than 2 years.
These factors, and others, need to be taken into account by people and their doctors who are considering whether or not screening with low-dose CT scans is right for them.
Non-small cell lung cancer symptoms and signs
Most lung cancers do not cause any symptoms until they have spread, but some people with early lung cancer do have symptoms 99.
Lung cancer may present with symptoms or be found incidentally on chest imaging. Symptoms and signs may result from the location of the primary local invasion or compression of adjacent thoracic structures, distant metastases, or paraneoplastic phenomena.
The most common symptoms of lung cancer are 99:
- A cough that does not go away or gets worse
- Coughing up blood or rust-colored sputum (spit or phlegm)
- Chest pain that is often worse with deep breathing, coughing, or laughing
- Hoarseness
- Weight loss and loss of appetite
- Shortness of breath
- Feeling tired or weak
- Infections such as bronchitis and pneumonia that don’t go away or keep coming back
- New onset of wheezing.
Symptoms may result from local invasion or compression of adjacent thoracic structures such as compression involving the esophagus causing dysphagia, compression involving the laryngeal nerves causing hoarseness, or compression involving the superior vena cava causing facial edema and distension of the superficial veins of the head and neck. Symptoms from distant metastases may also be present and include neurological defect or personality change from brain metastases or pain from bone metastases. Infrequently, patients may present with symptoms and signs of paraneoplastic diseases such as hypertrophic osteoarthropathy with digital clubbing or hypercalcemia from parathyroid hormone-related protein. Physical examination may identify enlarged supraclavicular lymphadenopathy, pleural effusion or lobar collapse, unresolved pneumonia, or signs of associated disease such as chronic obstructive pulmonary disease or pulmonary fibrosis.
When lung cancer spreads to distant organs, it may cause:
- Bone pain (like pain in the back or hips)
- Nervous system changes (such as headache, weakness or numbness of an arm or leg, dizziness, balance problems, or seizures), from cancer spread to the brain or spinal cord
- Yellowing of the skin and eyes (jaundice), from cancer spread to the liver
- Lumps near the surface of the body, due to cancer spreading to the skin or to lymph nodes (collections of immune system cells), such as those in the neck or above the collarbone
Most of these symptoms are more likely to be caused by something other than lung cancer. Still, if you have any of these problems, it’s important to see your doctor right away so the cause can be found and treated, if needed.
Some lung cancers can cause syndromes, which are groups of very specific symptoms.
Horner syndrome
Cancers of the top part of the lungs (sometimes called Pancoast tumors) sometimes can affect certain nerves to the eye and part of the face, causing a group of symptoms called Horner syndrome:
- Drooping or weakness of one eyelid
- A smaller pupil (dark part in the center of the eye) in the same eye
- Reduced or absent sweating on the same side of the face
Pancoast tumors can also sometimes cause severe shoulder pain.
Superior vena cava syndrome
The superior vena cava (SVC) is a large vein that carries blood from the head and arms back to the heart. It passes next to the upper part of the right lung and the lymph nodes inside the chest. Tumors in this area can press on the superior vena cava, which can cause the blood to back up in the veins. This can lead to swelling in the face, neck, arms, and upper chest (sometimes with a bluish-red skin color). It can also cause headaches, dizziness, and a change in consciousness if it affects the brain. While superior vena cava syndrome can develop gradually over time, in some cases it can become life-threatening, and needs to be treated right away.
Paraneoplastic syndromes
Some lung cancers can make hormone-like substances that enter the bloodstream and cause problems with distant tissues and organs, even though the cancer has not spread to those tissues or organs. These problems are called paraneoplastic syndromes. Sometimes these syndromes can be the first symptoms of lung cancer. Because the symptoms affect organs besides the lungs, patients and their doctors may suspect at first that a disease other than lung cancer is causing them.
Some of the more common paraneoplastic syndromes that can be caused by non-small cell lung cancer include:
- High blood calcium levels (hypercalcemia), which can cause frequent urination, thirst, constipation, nausea, vomiting, belly pain, weakness, fatigue, dizziness, confusion, and other nervous system problems
- Excess growth/thickening of certain bones, especially those in the finger tips, which is often painful
- Blood clots
- Excess breast growth in men (gynecomastia)
Again, many of these symptoms are more likely to be caused by something other than lung cancer. Still, if you have any of these problems, it’s important to see your doctor right away so the cause can be found and treated, if needed.
Non-small cell lung cancer diagnosis
Investigations of patients with suspected non-small cell lung cancer focus on confirming the diagnosis and determining the extent of the disease. Treatment options for patients are determined by histology, stage, and general health and comorbidities of the patient.
The procedures used to determine the presence of cancer include the following:
- History.
- Physical examination.
- Routine laboratory evaluations.
- Chest x-ray.
- Chest CT scan with infusion of contrast material.
- Biopsy.
Before a patient begins lung cancer treatment, an experienced lung cancer pathologist must review the pathologic material. This is critical because small cell lung cancer (SCLC), which responds well to chemotherapy and is generally not treated surgically, can be confused on microscopic examination with non-small cell lung cancer 100. Immunohistochemistry and electron microscopy are invaluable techniques for diagnosis and subclassification, but most lung tumors can be classified by light microscopic criteria.
Imaging tests
Imaging tests use x-rays, magnetic fields, sound waves, or radioactive substances to create pictures of the inside of your body. Imaging tests may be done for a number of reasons both before and after a diagnosis of lung cancer, including:
- To look at suspicious areas that might be cancer
- To learn how far cancer may have spread
- To help determine if treatment is working
- To look for possible signs of cancer coming back after treatment
Chest x-ray
This is often the first test your doctor will do to look for any abnormal areas in the lungs. Plain x-rays of your chest can be done at imaging centers, hospitals, and even in some doctors’ offices. If something suspicious is seen, your doctor may order more tests.
Computed tomography (CT) scan
A CT scan uses x-rays to make detailed cross-sectional images of your body. Instead of taking one picture, like a regular x-ray, a CT scanner takes many pictures as it rotates around you while you lie on a table. A computer then combines these pictures into images of slices of the part of your body being studied.
A CT scan is more likely to show lung tumors than routine chest x-rays. It can also show the size, shape, and position of any lung tumors and can help find enlarged lymph nodes that might contain cancer that has spread from the lung. This test can also be used to look for masses in the adrenal glands, liver, brain, and other internal organs that might be due to the spread of lung cancer.
CT-guided needle biopsy: If a suspected area of cancer is deep within your body, a CT scan can be used to guide a biopsy needle into the suspected area.
Real-time tumor imaging
Researchers are looking to use new imaging techniques, such as four-dimensional computed tomography (4DCT), to help improve treatment. In this technique, the CT machine scans the chest continuously for about 30 seconds. It shows where the tumor is in relation to other structures as a person breathes, as opposed to just giving a ‘snapshot’ of a point in time, like a standard CT does.
Four-dimensional computed tomography (4DCT) can be used to determine exactly where the tumor is during each part of the breathing cycle, which can help doctors deliver radiation to a tumor more precisely. This technique might also be used to help show if a tumor is attached to or invading important structures in the chest, which could help doctors determine if a patient might be eligible for surgery.
Magnetic resonance imaging (MRI) scan
Like CT scans, MRI scans provide detailed images of soft tissues. But MRI scans use radio waves and strong magnets instead of x-rays. A contrast material called gadolinium is often injected into a vein before the scan to better see details.
MRI scans are most often used to look for possible spread of lung cancer to the brain or spinal cord. Rarely, MRI of the chest may be done to see if the cancer has grown into central structures in the chest.
Positron emission tomography (PET) scan
For this test, a form of radioactive sugar (known as FDG) is injected into the blood. Because cancer cells in the body are growing quickly, they absorb more of the radioactive sugar. This radioactivity can be seen with a special camera.
PET/CT scan: Often a PET scan is combined with a CT scan using a special machine that can do both at the same time. This lets the doctor compare areas of higher radioactivity on the PET scan with the more detailed appearance of that area on the CT scan. This is the type of PET scan most often used in patients with lung cancer.
If you appear to have early stage lung cancer, your doctor can use this test to help see if the cancer has spread to nearby lymph nodes or other areas, which can help determine if surgery may be an option for you. This test can also be helpful in getting a better idea if an abnormal area on another imaging test might be cancer.
PET/CT scans can also be useful if your doctor thinks the cancer might have spread but doesn’t know where. They can show spread of cancer to the liver, bones, adrenal glands, or some other organs. They are not as useful for looking at the brain, since all brain cells use a lot of glucose.
PET/CT scans are often helpful in diagnosing lung cancer, but their role in checking whether treatment is working is unproven. Most doctors do not recommend PET/CT scans for routine follow up of patients with lung cancer after treatment.
Bone scan
For this test, a small amount of low-level radioactive material is injected into the blood. The substance settles in areas of bone changes throughout the entire skeleton. This radioactivity can be seen with a special camera.
A bone scan can help show if a cancer has spread to the bones. But this test isn’t needed very often because PET scans, which are often done in patients with non-small cell lung cancer, can usually show if cancer has spread to the bones. Bone scans are done mainly when there is reason to think the cancer may have spread to the bones (because of symptoms such as bone pain) and other test results aren’t clear.
Tests for diagnosing lung cancer
Symptoms and the results of certain tests may strongly suggest that a person has lung cancer, but the actual diagnosis is made by looking at lung cells with a microscope.
The cells can be taken from lung secretions (sputum or phlegm), fluid removed from the area around the lung (thoracentesis), or from a suspicious area using a needle or surgery (known as a biopsy). The choice of which test(s) to use depends on the situation.
Sputum cytology
A sample of mucus you cough up from the lungs (sputum) is looked at under a microscope to see if it has cancer cells. The best way to do this is to get early morning samples from you 3 days in a row. This test is more likely to help find cancers that start in the major airways of the lung, such as squamous cell lung cancers. It may not be as helpful for finding other types of non-small cell lung cancer. If your doctor suspects lung cancer, further testing will be done even if no cancer cells are found in the sputum.
Thoracentesis
If there is a buildup of fluid around the lungs (called a pleural effusion), doctors can perform thoracentesis to find out if it is caused by cancer spreading to the lining of the lungs (pleura). The buildup might also be caused by other conditions, such as heart failure or an infection.
For this procedure, the skin is numbed and a hollow needle is inserted between the ribs to drain the fluid. (In a similar test called pericardiocentesis, fluid is removed from within the sac around the heart.) The fluid is checked under a microscope for cancer cells. Chemical tests of the fluid are also sometimes useful in telling a malignant (cancerous) pleural effusion from one that is not.
If a malignant pleural effusion has been diagnosed, thoracentesis may be repeated to remove more fluid. Fluid buildup can keep the lungs from filling with air, so thoracentesis can help a person breathe better.
Needle biopsy
Doctors can often use a hollow needle to get a small sample from a suspicious area (mass).
In a fine needle aspiration (FNA) biopsy, the doctor uses a syringe with a very thin, hollow needle to withdraw (aspirate) cells and small fragments of tissue.
In a core biopsy, a larger needle is used to remove one or more small cores of tissue. Samples from core biopsies are larger than fine needle aspiration biopsies, so they are often preferred.
An advantage of needle biopsies is that they don’t require a surgical incision. The drawback is that they remove only a small amount of tissue. In some cases (particularly with fine needle aspiration biopsies), the amount removed might not be enough to both make a diagnosis and to classify DNA changes in the cancer cells that can help doctors choose anticancer drugs.
Transthoracic needle biopsy: If the suspected tumor is in the outer part of the lungs, the biopsy needle can be inserted through the skin on the chest wall. The area where the needle is to be inserted may be numbed with local anesthesia first. The doctor then guides the needle into the area while looking at the lungs with either fluoroscopy (which is like an x-ray, but creates a moving image on a screen rather than a single picture on film) or CT scans.
If CT is used, the needle is inserted toward the mass (tumor), a CT image is taken, and the direction of the needle is guided based on the image. This is repeated a few times until the needle is within the mass.
A possible complication of this procedure is that air may leak out of the lung at the biopsy site and into the space between the lung and the chest wall. This is called a pneumothorax. It can cause part of the lung to collapse and possibly trouble breathing. If the air leak is small, it often gets better without any treatment. Large air leaks are treated by putting a small tube into the chest space and sucking out the air over a day or two, after which it usually heals on its own.
Other approaches to needle biopsies: An fine needle aspiration biopsy may also be done to check for cancer in the lymph nodes between the lungs:
Transtracheal fine needle aspiration or transbronchial fine needle aspiration is done by passing the needle through the wall of the trachea (windpipe) or bronchi (the large airways leading into the lungs) during bronchoscopy or endobronchial ultrasound (described below).
In some patients an fine needle aspiration biopsy is done during endoscopic esophageal ultrasound (described below) by passing the needle through the wall of the esophagus.
Bronchoscopy
Bronchoscopy can help the doctor find some tumors or blockages in the larger airways of the lungs, which can often be biopsied during the procedure.
For this exam, a lighted, flexible fiber-optic tube (called a bronchoscope) is passed through the mouth or nose and down into the windpipe and bronchi. The mouth and throat are sprayed first with a numbing medicine. You may also be given medicine through an intravenous (IV) line to make you feel relaxed.
Small instruments can be passed down the bronchoscope to take biopsy samples. The doctor can also sample cells from the lining of the airways with a small brush (bronchial brushing) or by rinsing the airways with sterile saltwater (bronchial washing). These tissue and cell samples are then looked at under a microscope.
Tests to find lung cancer spread in the chest
If lung cancer has been found, it’s often important to know if it has spread to the lymph nodes in the space between the lungs (mediastinum) or other nearby areas. This can affect a person’s treatment options. Several types of tests can be used to look for this cancer spread.
Endobronchial ultrasound
Ultrasound is a type of imaging test that uses sound waves to create pictures of the inside of your body. For this test, a small, microphone-like instrument called a transducer gives off sound waves and picks up the echoes as they bounce off body tissues. The echoes are converted by a computer into an image on a computer screen.
For endobronchial ultrasound, a bronchoscope is fitted with an ultrasound transducer at its tip and is passed down into the windpipe. This is done with numbing medicine (local anesthesia) and light sedation.
The transducer can be pointed in different directions to look at lymph nodes and other structures in the mediastinum (the area between the lungs). If suspicious areas such as enlarged lymph nodes are seen on the ultrasound, a hollow needle can be passed through the bronchoscope and guided into these areas to obtain a biopsy. The samples are then sent to a lab to be looked at under a microscope.
Endoscopic esophageal ultrasound
This test is like endobronchial ultrasound, except the doctor passes an endoscope (a lighted, flexible scope) down the throat and into the esophagus (the tube connecting the throat to the stomach). This is done with numbing medicine (local anesthesia) and light sedation.
The esophagus is just behind the windpipe and is close to some lymph nodes inside the chest to which lung cancer may spread. As with endobronchial ultrasound, the transducer can be pointed in different directions to look at lymph nodes and other structures inside the chest that might contain lung cancer. If enlarged lymph nodes are seen on the ultrasound, a hollow needle can be passed through the endoscope to get biopsy samples of them. The samples are then sent to a lab to be looked at under a microscope.
Mediastinoscopy and mediastinotomy
These procedures may be done to look more directly at and get samples from the structures in the mediastinum (the area between the lungs). They are done in an operating room by a surgeon while you are under general anesthesia (in a deep sleep). The main difference between the two is in the location and size of the incision.
- Mediastinoscopy: A small cut is made in the front of the neck and a thin, hollow, lighted tube is inserted behind the sternum (breast bone) and in front of the windpipe to look at the area. Instruments can be passed through this tube to take tissue samples from the lymph nodes along the windpipe and the major bronchial tube areas. Looking at the samples under a microscope can show if they have cancer cells.
- Mediastinotomy: The surgeon makes a slightly larger incision (usually about 2 inches long) between the left second and third ribs next to the breast bone. This lets the surgeon reach some lymph nodes that can’t be reached by mediastinoscopy.
Thoracoscopy
Thoracoscopy can be done to find out if cancer has spread to the spaces between the lungs and the chest wall, or to the linings of these spaces. It can also be used to sample tumors on the outer parts of the lungs as well as nearby lymph nodes and fluid, and to assess whether a tumor is growing into nearby tissues or organs. This procedure is not often done just to diagnose lung cancer, unless other tests such as needle biopsies are unable to get enough samples for the diagnosis.
Thoracoscopy is done in the operating room while you are under general anesthesia (in a deep sleep). A small cut (incision) is made in the side of the chest wall. (Sometimes more than one cut is made.) The doctor then puts a thin, lighted tube with a small video camera on the end through the incision to view the space between the lungs and the chest wall. Using this, the doctor can see possible cancer deposits on the lining of the lung or chest wall and remove small pieces of tissue for examination. When certain areas can’t be reached with thoracoscopy, the surgeon may need to make a larger incision in the chest wall, known as a thoracotomy.
Thoracoscopy can also be used as part of the treatment to remove part of a lung in some early-stage lung cancers. This type of operation, known as video-assisted thoracic surgery (VATS), is described below.
Lab tests of biopsy and other samples
Samples that have been collected during biopsies or other tests are sent to a pathology lab. A pathologist, a doctor who uses lab tests to diagnose diseases such as cancer, will look at the samples with a microscope and may do other special tests to help better classify the cancer. Cancers from other organs can spread to the lungs. It’s very important to find out where the cancer started, because treatment is different depending on the type of cancer.
The results of these tests are described in a pathology report, which is usually available within about a week. If you have any questions about your pathology results or any diagnostic tests, talk to your doctor. If needed, you can get a second opinion of your pathology report by having your tissue samples sent to a pathologist at another lab.
Immunohistochemical tests
For this test, very thin slices of the samples are attached to glass microscope slides. The samples are then treated with special proteins (antibodies) that attach only to a specific substance found in certain cancer cells. If the cancer cells have that substance, the antibody will attach to the cells. Chemicals are then added so that antibodies change color. The doctor who looks at the sample under a microscope can see this color change.
Molecular tests
In some cases, doctors may look for specific gene changes in the cancer cells that could mean certain targeted drugs might help treat the cancer. For example:
The epidermal growth factor receptor (EGFR) is a protein that sometimes appears in high amounts on the surface of cancer cells and helps them grow. Some drugs that target EGFR seem to work best against lung cancers with certain changes in the EGFR gene, which are more common in certain groups, such as non-smokers, women, and Asians. But these drugs don’t seem to be as helpful in patients whose cancer cells have changes in the KRAS gene. Many doctors now test for changes in genes such as EGFR and KRAS to determine if these newer treatments are likely to be helpful.
About 5% of non-small cell lung cancers (NSCLCs) have a change in a gene called ALK. This change is most often seen in non-smokers (or light smokers) who have the adenocarcinoma subtype of non-small cell lung cancer. Doctors may test cancers for changes in the ALK gene to see if drugs that target this change may help them.
About 1% to 2% of non-small cell lung cancers have a rearrangement in the ROS1 gene, which might make the tumor respond to certain targeted drugs. A similar percentage have a rearrangement in the RET gene. Certain drugs that target cells with RET gene changes might be options for treating these tumors.
Some non-small cell lung cancers have changes in the BRAF gene. Certain drugs that target cells with BRAF gene changes might be option for treating these tumors.
Newer lab tests for certain other genes or proteins may also help guide the choice of treatment.
Molecular Features
The identification of mutations in lung cancer has led to the development of molecularly targeted therapy to improve the survival of subsets of patients with metastatic disease 101. In particular, subsets of adenocarcinoma now can be defined by specific mutations in genes encoding components of the epidermal growth factor receptor (EGFR) and downstream mitogen-activated protein kinases (MAPK) and phosphatidylinositol 3-kinases (PI3K) signaling pathways. These mutations may define mechanisms of drug sensitivity and primary or acquired resistance to kinase inhibitors.
Other genetic abnormalities of potential relevance to treatment decisions include translocations involving the anaplastic lymphoma kinase (ALK)-tyrosine kinase receptor, which are sensitive to ALK inhibitors, and amplification of MET (mesenchymal epithelial transition factor), which encodes the hepatocyte growth factor receptor. MET amplification has been associated with secondary resistance to EGFR tyrosine kinase inhibitors.
Blood tests
Blood tests are not used to diagnose lung cancer, but they can help to get a sense of a person’s overall health. For example, they can be used to help determine if a person is healthy enough to have surgery.
A complete blood count (CBC) looks at whether your blood has normal numbers of different types of blood cells. For example, it can show if you are anemic (have a low number of red blood cells), if you could have trouble with bleeding (due to a low number of blood platelets), or if you are at increased risk for infections (because of a low number of white blood cells). This test will be repeated regularly if you are treated with chemotherapy, because these drugs can affect blood-forming cells of the bone marrow.
Blood chemistry tests can help spot abnormalities in some of your organs, such as the liver or kidneys. For example, if cancer has spread to the liver and bones, it may cause abnormal levels of certain chemicals in the blood, such as a high level of lactate dehydrogenase (LDH).
Pulmonary function tests
Pulmonary function tests are often done after lung cancer is diagnosed to see how well your lungs are working (for example, how much emphysema or chronic bronchitis is present). This is especially important if surgery might be an option in treating the cancer. Surgery to remove lung cancer may mean removing part or all of a lung, so it’s important to know how well the lungs are working beforehand. Some people with poor lung function (like those with lung damage from smoking) don’t have enough lung reserve to withstand removing even part of a lung. These tests can give the surgeon an idea of whether surgery is a good option, and if so, how much lung can safely be removed.
There are different types of pulmonary function tests, but they all basically have you breathe in and out through a tube that is connected to a machine that measures airflow.
Sometimes pulmonary function tests are coupled with a test called an arterial blood gas. In this test, blood is removed from an artery (instead of from a vein, like most other blood tests) to measure the amount of oxygen and carbon dioxide that it contains.
Fluorescence bronchoscopy
Also known as autofluorescence bronchoscopy, this technique might help doctors find some lung cancers earlier, when they are likely to be easier to treat. For this test, the doctor inserts a bronchoscope through the mouth or nose and into the lungs. The end of the bronchoscope has a special fluorescent light on it, instead of a normal (white) light.
The fluorescent light causes abnormal areas in the airways to show up in a different color than healthy parts of the airway. Some of these areas might not be visible under white light, so the color difference can help doctors find these areas sooner. Some cancer centers now use this technique to look for early lung cancers, especially if there are no obvious tumors seen with normal bronchoscopy.
Virtual bronchoscopy
This imaging test uses a chest CT scan to create a detailed 3-dimensional picture of the airways in the lungs. The images can be viewed as if the doctor were actually using a bronchoscope.
Virtual bronchoscopy has some possible advantages over standard bronchoscopy. First, it is non-invasive and doesn’t require anesthesia. It also helps doctors view some airways that they might not able to get to with standard bronchoscopy, such as those being blocked by a tumor. But this test has some drawbacks as well. For example, it doesn’t show color changes in the airways that might indicate a problem. It also doesn’t let a doctor take samples of suspicious areas like bronchoscopy does. Still, it can be a useful tool in some situations, such as in people who might be too sick to get a standard bronchoscopy.
This test will probably become more available as the technology improves.
Lung tumors near the center of the chest can be biopsied during bronchoscopy, but bronchoscopes have trouble reaching the outer parts of the lungs, so tumors in these areas often need to be biopsied using a needle passed through the skin.
This newer approach can help a doctor use a bronchoscope to biopsy a tumor in the outer part of the lung. First, CT scans are used to create a virtual bronchoscopy. The abnormal area is identified, and a computer helps guide a bronchoscope to the area so that it can be biopsied. The bronchoscope used has some special attachments that allow it to reach further than a regular bronchoscope.
This takes special equipment and training, and it is not widely available at this time.
Prognostic Factors
Multiple studies have attempted to identify the prognostic importance of a variety of clinicopathologic factors 102, 103, 104.
Factors that have correlated with adverse prognosis include the following:
- Presence of pulmonary symptoms.
- Large tumor size (>3 cm).
- Nonsquamous histology.
- Metastases to multiple lymph nodes within a TNM-defined nodal station 105.
- Vascular invasion 106.
For patients with inoperable disease, prognosis is adversely affected by poor performance status and weight loss of more than 10%. These patients have been excluded from clinical trials evaluating aggressive multimodality interventions.
In multiple retrospective analyses of clinical trial data, advanced age alone has not been shown to influence response or survival with therapy 107.
Because treatment is not satisfactory for almost all patients with non-small cell lung cancer, eligible patients should consider taking part in clinical trials. If you would like to learn more about clinical trials that might be right for you, start by asking your doctor if your clinic or hospital conducts clinical trials.
Non-Small Cell Lung Cancer Stages
The stage of a cancer describes how far it has spread. Your treatment and prognosis (outlook) depend, to a large extent, on the cancer’s stage.
There are actually 2 types of staging descriptions for non-small cell lung cancer 108:
- The clinical stage is based on the results of physical exams, biopsies, imaging tests (CT scan, chest x-ray, PET scan, etc.), and other tests.
- If you have surgery, your doctor can also determine the pathologic stage, which is based on the same factors as the clinical stage, plus what is found as a result of the surgery.
The clinical and pathologic stages might be different in some cases. For example, during surgery the doctor may find cancer in an area that did not show up on imaging tests, which might give the cancer a more advanced pathologic stage.
Because many people with non-small cell lung cancer do not have surgery, the clinical stage is often used when describing the extent of this cancer. But when it is available, the pathologic stage is likely to be more accurate than the clinical stage, as it uses the additional information obtained at surgery.
Understanding the stage of your non-small cell lung cancer
The system used most often to stage non-small cell lung cancer is the American Joint Committee on Cancer TNM system, which is based on:
- The size of the main (primary) tumor (T) and whether it has grown into nearby areas.
- Whether the cancer has spread to nearby (regional) lymph nodes (N). Lymph nodes are small bean-shaped collections of immune system cells to which cancers often spread before going to other parts of the body.
- Whether the cancer has spread (metastasized; M) to other organs of the body. The most common sites are the brain, bones, adrenal glands, liver, kidneys, and the other lung.
Numbers or letters appear after T, N, and M to provide more details about each of these factors. Higher numbers mean the cancer is more advanced. Once the T, N, and M categories have been determined, this information is combined in a process called stage grouping, and an overall stage is assigned.
TNM staging system
The TNM staging system is complex and can be hard for patients (and even some doctors) to understand. If you have any questions about the stage of your cancer, ask your doctor to explain it to you.
T categories for lung cancer
- TX: The main (primary) tumor can’t be assessed, or cancer cells were seen on sputum cytology or bronchial washing but no tumor can be found.
- T0: There is no evidence of a primary tumor.
- Tis: The cancer is found only in the top layers of cells lining the air passages. It has not invaded into deeper lung tissues. This is also known as carcinoma in situ.
- T1: The tumor is no larger than 3 centimeters (cm)—slightly less than 1¼ inches—across, has not reached the membranes that surround the lungs (visceral pleura), and does not affect the main branches of the bronchi.
- If the tumor is 2 cm (about 4/5 of an inch) or less across, it is called T1a. If the tumor is larger than 2 cm but not larger than 3 cm across, it is called T1b.
- T2: The tumor has 1 or more of the following features:
- It is larger than 3 cm across but not larger than 7 cm.
- It involves a main bronchus, but is not closer than 2 cm (about ¾ inch) to the carina (the point where the windpipe splits into the left and right main bronchi).
- It has grown into the membranes that surround the lungs (visceral pleura).
- The tumor partially clogs the airways, but this has not caused the entire lung to collapse or develop pneumonia.
- If the tumor is 5 cm or less across, it is called T2a. If the tumor is larger than 5 cm across (but not larger than 7 cm), it is called T2b.
- T3: The tumor has 1 or more of the following features:
- It is larger than 7 cm across.
- It has grown into the chest wall, the breathing muscle that separates the chest from the abdomen (diaphragm), the membranes surrounding the space between the two lungs (mediastinal pleura), or membranes of the sac surrounding the heart (parietal pericardium).
- It has grown into a main bronchus and is closer than 2 cm (about ¾ inch) to the carina, but it does not involve the carina itself.
- It has grown into the airways enough to cause an entire lung to collapse or to cause pneumonia in the entire lung.
- Two or more separate tumor nodules are present in the same lobe of a lung.
- T4: The cancer has 1 or more of the following features:
- A tumor of any size has grown into the space between the lungs (mediastinum), the heart, the large blood vessels near the heart (such as the aorta), the windpipe (trachea), the tube connecting the throat to the stomach (esophagus), the backbone, or the carina.
- Two or more separate tumor nodules are present in different lobes of the same lung.
N categories for lung cancer
- NX: Nearby lymph nodes cannot be assessed.
- N0: There is no spread to nearby lymph nodes.
- N1: The cancer has spread to lymph nodes within the lung and/or around the area where the bronchus enters the lung (hilar lymph nodes). Affected lymph nodes are on the same side as the primary tumor.
- N2: The cancer has spread to lymph nodes around the carina (the point where the windpipe splits into the left and right bronchi) or in the space between the lungs (mediastinum). Affected lymph nodes are on the same side as the primary tumor.
- N3: The cancer has spread to lymph nodes near the collarbone on either side, and/or spread to hilar or mediastinal lymph nodes on the side opposite the primary tumor.
M categories for lung cancer
- M0: No spread to distant organs or areas. This includes the other lung, lymph nodes further away than those mentioned in the N stages above, and other organs or tissues such as the liver, bones, or brain.
- M1a: Any of the following:
- The cancer has spread to the other lung.
- The cancer has spread as nodules (small lumps) in the pleura (the lining of the lung).
- Cancer cells are found in the fluid around the lung (called a malignant pleural effusion).
- Cancer cells are found in the fluid around the heart (called a malignant pericardial effusion).
- M1b: The cancer has spread to distant lymph nodes or to other organs such as the liver, bones, or brain.
Stage grouping for lung cancer
Once the T, N, and M categories have been assigned, this information is combined to assign an overall stage of 0, I, II, III, or IV. This process is called stage grouping. Some stages are subdivided into A and B. The stages identify cancers that have a similar outlook (prognosis) and thus are treated in a similar way. Patients with lower stage numbers tend to have a better outlook.
TX, N0, M0: Cancer cells are seen in a sample of sputum or other lung fluids, but the cancer isn’t found with other tests, so its location can’t be determined.
Stage 0
Tis, N0, M0: The cancer is found only in the top layers of cells lining the air passages. It has not invaded deeper into other lung tissues and has not spread to lymph nodes or distant sites. Stage 0 non-small cell lung cancer (NSCLC) frequently progresses to invasive cancer 109. Patients may be offered surveillance bronchoscopies and, if lesions are detected, potentially curative therapies.
Stage 1A
T1a/T1b, N0, M0: The cancer is no larger than 3 cm across, has not reached the membranes that surround the lungs, and does not affect the main branches of the bronchi. It has not spread to lymph nodes or distant sites.
Stage 1B
T2a, N0, M0: The cancer has 1 or more of the following features:
- The main tumor is larger than 3 cm across but not larger than 5 cm.
- The tumor has grown into a main bronchus, but is not within 2 cm of the carina (and it is not larger than 5 cm).
- The tumor has grown into the visceral pleura (the membranes surrounding the lungs) and is not larger than 5 cm.
- The tumor is partially clogging the airways (and is not larger than 5 cm).
The cancer has not spread to lymph nodes or distant sites.
Stage 2A
Three main combinations of categories make up this stage.
T1a/T1b, N1, M0: The cancer is no larger than 3 cm across, has not grown into the membranes that surround the lungs, and does not affect the main branches of the bronchi. It has spread to lymph nodes within the lung and/or around the area where the bronchus enters the lung (hilar lymph nodes). These lymph nodes are on the same side as the cancer. It has not spread to distant sites.
OR
T2a, N1, M0: The cancer has 1 or more of the following features:
- The main tumor is larger than 3 cm across but not larger than 5 cm.
- The tumor has grown into a main bronchus, but is not within 2 cm of the carina (and it is not larger than 5 cm).
- The tumor has grown into the visceral pleura (the membranes surrounding the lungs) and is not larger than 5 cm.
- The tumor is partially clogging the airways (and is not larger than 5 cm).
The cancer has also spread to lymph nodes within the lung and/or around the area where the bronchus enters the lung (hilar lymph nodes). These lymph nodes are on the same side as the cancer. It has not spread to distant sites.
OR
T2b, N0, M0: The cancer has 1 or more of the following features:
- The main tumor is larger than 5 cm across but not larger than 7 cm.
- The tumor has grown into a main bronchus, but is not within 2 cm of the carina (and it is between 5 and 7 cm across).
- The tumor has grown into the visceral pleura (the membranes surrounding the lungs) and is between 5 and 7 cm across.
- The tumor is partially clogging the airways (and is between 5 and 7 cm across).
The cancer has not spread to lymph nodes or distant sites.
Stage 2B
Two combinations of categories make up this stage.
T2b, N1, M0: The cancer has 1 or more of the following features:
- The main tumor is larger than 5 cm across but not larger than 7 cm.
- The tumor has grown into a main bronchus, but is not within 2 cm of the carina (and it is between 5 and 7 cm across).
- The tumor has grown into the visceral pleura (the membranes surrounding the lungs) and is between 5 and 7 cm across.
- The cancer is partially clogging the airways (and is between 5 and 7 cm across).
It has also spread to lymph nodes within the lung and/or around the area where the bronchus enters the lung (hilar lymph nodes). These lymph nodes are on the same side as the cancer. It has not spread to distant sites.
OR
T3, N0, M0: The main tumor has 1 or more of the following features:
- It is larger than 7 cm across.
- It has grown into the chest wall, the breathing muscle that separates the chest from the abdomen (diaphragm), the membranes surrounding the space between the lungs (mediastinal pleura), or membranes of the sac surrounding the heart (parietal pericardium).
- It invades a main bronchus and is closer than 2 cm (about ¾ inch) to the carina, but it does not involve the carina itself.
- It has grown into the airways enough to cause an entire lung to collapse or to cause pneumonia in the entire lung.
- Two or more separate tumor nodules are present in the same lobe of a lung.
The cancer has not spread to lymph nodes or distant sites.
Stage 3A
Three main combinations of categories make up this stage.
T1 to T3, N2, M0: The main tumor can be any size. It has not grown into the space between the lungs (mediastinum), the heart, the large blood vessels near the heart (such as the aorta), the windpipe (trachea), the tube connecting the throat to the stomach (esophagus), the backbone, or the carina. It has not spread to different lobes of the same lung.
The cancer has spread to lymph nodes around the carina (the point where the windpipe splits into the left and right bronchi) or in the space between the lungs (mediastinum). These lymph nodes are on the same side as the main lung tumor. The cancer has not spread to distant sites.
OR
T3, N1, M0: The cancer has 1 or more of the following features:
- It is larger than 7 cm across.
- It has grown into the chest wall, the breathing muscle that separates the chest from the abdomen (diaphragm), the membranes surrounding the space between the lungs (mediastinal pleura), or membranes of the sac surrounding the heart (parietal pericardium).
- It invades a main bronchus and is closer than 2 cm to the carina, but it does not involve the carina itself.
- Two or more separate tumor nodules are present in the same lobe of a lung.
- It has grown into the airways enough to cause an entire lung to collapse or to cause pneumonia in the entire lung.
It has also spread to lymph nodes within the lung and/or around the area where the bronchus enters the lung (hilar lymph nodes). These lymph nodes are on the same side as the cancer. It has not spread to distant sites.
OR
T4, N0 or N1, M0: The cancer has 1 or more of the following features:
- A tumor of any size has grown into the space between the lungs (mediastinum), the heart, the large blood vessels near the heart (such as the aorta), the windpipe (trachea), the tube connecting the throat to the stomach (esophagus), the backbone, or the carina.
- Two or more separate tumor nodules are present in different lobes of the same lung.
It may or may not have spread to lymph nodes within the lung and/or around the area where the bronchus enters the lung (hilar lymph nodes). Any affected lymph nodes are on the same side as the cancer. It has not spread to distant sites.
Stage 3B
Two combinations of categories make up this stage.
Any T, N3, M0: The cancer can be of any size. It may or may not have grown into nearby structures or caused pneumonia or lung collapse. It has spread to lymph nodes near the collarbone on either side, and/or has spread to hilar or mediastinal lymph nodes on the side opposite the primary tumor. The cancer has not spread to distant sites.
OR
T4, N2, M0: The cancer has 1 or more of the following features:
- A tumor of any size has grown into the space between the lungs (mediastinum), the heart, the large blood vessels near the heart (such as the aorta), the windpipe (trachea), the tube connecting the throat to the stomach (esophagus), the backbone, or the carina.
- Two or more separate tumor nodules are present in different lobes of the same lung.
The cancer has also spread to lymph nodes around the carina (the point where the windpipe splits into the left and right bronchi) or in the space between the lungs (mediastinum). Affected lymph nodes are on the same side as the main lung tumor. It has not spread to distant sites.
Stage 4
Stage 4 lung cancer also called advanced lung cancer, which means the cancer has spread. Stage 4 is divided into stage 4A and 4B.
- Stage 4A can mean any of the following:
- there is cancer in both lungs
- the cancer is in the covering of the lung (the pleura) or the covering of the heart (pericardium)
- or there is fluid around the lungs or the heart that contains cancer cells
- Or it can mean that there is a single area of cancer that has spread outside the chest to a lymph node or to an organ such as the liver or bone.
- Stage 4B means that the cancer has spread to several areas in one or more organs.
In the TNM staging system, stage 4 is the same as one of the following:
- Any T, Any N, M1a
- Any T, Any N, M1b
- Any T, Any N, M1c
Non-Small Cell Lung Cancer Survival Rates by Stage
Survival rates tell you what portion of people with the same type and stage of cancer are still alive a certain amount of time (usually 5 years) after they were diagnosed. These numbers can’t tell you how long you will live, but they may help give you a better understanding about how likely it is that your treatment will be successful.
What is a 5-year survival rate?
Statistics on the outlook for a certain type and stage of cancer are often given as 5-year survival rates, but many people live longer – often much longer – than 5 years. The 5-year survival rate is the percentage of people who live at least 5 years after being diagnosed with cancer. For example, a 5-year survival rate of 80% means that an estimated 80 out of 100 people who have that cancer are still alive 5 years after being diagnosed. Keep in mind, however, that many of these people live much longer than 5 years after diagnosis.
But remember, the 5-year survival rates are estimates – your outlook can vary based on a number of factors specific to you. Survival rates don’t tell the whole story
Survival rates are often based on previous outcomes of large numbers of people who had the disease, but they can’t predict what will happen in any particular person’s case. There are a number of limitations to keep in mind:
- The numbers below are among the most current available. But to get 5-year survival rates, doctors have to look at people who were treated at least 5 years ago. As treatments are improving over time, people who are now being diagnosed with non-small cell lung cancer may have a better outlook than these statistics show.
- These statistics are based on the stage of the cancer when it was first diagnosed. They do not apply to cancers that later come back or spread, for example.
- The outlook for people with non-small cell lung cancer varies by the stage (extent) of the cancer – in general, the survival rates are higher for people with earlier stage cancers.
- But many other factors can affect a person’s outlook, such as the subtype of non-small cell lung cancer, gene changes in the cancer cells, the person’s age and overall health, and how well the cancer responds to treatment. The outlook for each person is specific to his or her circumstances.
Your doctor can tell you how these numbers may apply to you, as he or she is familiar with your particular situation.
The numbers below are calculated from the National Cancer Institute’s SEER database, based on people who were diagnosed with non-small cell lung cancer between 1998 and 2000. Although they are based on people diagnosed several years ago, they are the most recent rates published for the current AJCC staging system.
These survival rates include people who die from causes other than cancer.
- The 5-year survival rate for people with stage 1A non-small cell lung cancer is about 49%.
- For people with stage 1B non-small cell lung cancer the 5-year survival rate is about 45%.
- For stage 2A cancer, the 5-year survival rate is about 30%.
- For stage 2B cancer, the survival rate is about 31%.
- The 5-year survival rate for stage 3A non-small cell lung cancer is about 14%.
- For stage 3B cancers the survival rate is about 5%.
- Non-small cell lung cancer that has spread to other parts of the body is often hard to treat. Metastatic, or stage 4 non-small cell lung cancer, has a 5-year survival rate of about 1%. Still, there are often many treatment options available for people with this stage of cancer.
Remember, these survival rates are only estimates – they can’t predict what will happen to any individual person. These statistics can be confusing and may lead you to have more questions. Talk to your doctor to better understand your specific situation.
Non-small cell lung cancer treatment
Depending on the stage of the cancer and other factors, treatment options for people with non-small cell lung cancer can include:
- Surgery
- Radiofrequency ablation (RFA)
- Radiation therapy
- Chemotherapy
- Targeted therapies
- Immunotherapy
Palliative treatments can also be used to help with symptoms.
In many cases, more than one of type of treatment is used.
Which doctors treat non-small cell lung cancer?
You may have different types of doctors on your treatment team, depending on the stage of your cancer and your treatment options. These doctors could include:
- A thoracic surgeon: a doctor who treats diseases of the lungs and chest with surgery
- A radiation oncologist: a doctor who treats cancer with radiation therapy
- A medical oncologist: a doctor who treats cancer with medicines such as chemotherapy, targeted therapy, and immunotherapy
- A pulmonologist: a doctor who specializes in medical treatment of diseases of the lungs
You might have many other specialists on your treatment team as well, including physician assistants, nurse practitioners, nurses, respiratory therapists, nutrition specialists, social workers, and other health professionals. See Health Professionals Associated With Cancer Care for more on this.
Making treatment decisions
It’s important to discuss all of your treatment options, including their goals and possible side effects, with your doctors to help make the decision that best fits your needs. It’s also very important to ask questions if there is anything you’re not sure about.
Getting a second opinion
You may also want to get a second opinion. This can give you more information and help you feel more certain about the treatment plan you choose. If you aren’t sure where to go for a second opinion, ask your doctor for help.
Thinking about taking part in a clinical trial
Clinical trials are carefully controlled research studies that are done to get a closer look at promising new treatments or procedures. Clinical trials are one way to get state-of-the art cancer treatment. In some cases they may be the only way to get access to newer treatments. They are also the best way for doctors to learn better methods to treat cancer. Still, they are not right for everyone.
If you would like to learn more about clinical trials that might be right for you, start by asking your doctor if your clinic or hospital conducts clinical trials.
Considering complementary and alternative methods
You may hear about complementary or alternative methods that your doctor hasn’t mentioned to treat your cancer or relieve symptoms. These methods can include vitamins, herbs, and special diets, or other methods such as acupuncture or massage, to name a few.
Complementary methods refer to treatments that are used along with your regular medical care. Alternative treatments are used instead of a doctor’s medical treatment. Although some of these methods might be helpful in relieving symptoms or helping you feel better, many have not been proven to work. Some might even be dangerous.
As you consider your options, look for “red flags” that might suggest fraud. Does the method promise to cure all or most cancers? Are you told not to have regular medical treatments? Is the treatment a “secret” that requires you to visit certain providers or travel to another country?
Be sure to talk to your cancer care team about any method you are thinking about using. They can help you learn what is known (or not known) about the method, which can help you make an informed decision.
Choosing to stop treatment or choosing no treatment at all
For some people, when treatments have been tried and are no longer controlling the cancer, it could be time to weigh the benefits and risks of continuing to try new treatments. Whether or not you continue treatment, there are still things you can do to help maintain or improve your quality of life. Learn more in If Cancer Treatments Stop Working.
Some people, especially if the cancer is advanced, might not want to be treated at all. There are many reasons you might decide not to get cancer treatment, but it’s important to talk this through with your doctors before you make this decision. Remember that even if you choose not to treat the cancer, you can still get supportive care to help with pain or other symptoms.
Help getting through treatment
Your cancer care team will be your first source of information and support, but there are other resources for help when you need it. Hospital- or clinic-based support services are an important part of your care. These might include nursing or social work services, financial aid, nutritional advice, rehab, or spiritual help.
Surgery
Surgery to remove the cancer (often along with other treatments) may be an option for early stage non-small cell lung cancer. If surgery can be done, it provides the best chance to cure non-small cell lung cancer. Lung cancer surgery is a complex operation that can have serious consequences, so it should be done by a surgeon who has a lot of experience operating on lung cancers.
Doctors now use video-assisted thoracic surgery (VATS) to treat some small lung tumors. This procedure lets doctors remove parts of the lung through smaller incisions, which can result in shorter hospital stays and less pain for patients. Doctors are now studying if video-assisted thoracic surgery (VATS) can be used for larger lung tumors.
In a newer approach to this type of operation, the surgeon sits at a specially designed control panel inside the operating room to maneuver long surgical instruments using robotic arms. This approach, known as robotic-assisted surgery, is now being used in some larger cancer centers.
Types of lung surgery
Different operations can be used to treat (and possibly cure) non-small cell lung cancer:
- Pneumonectomy: This surgery removes an entire lung. This might be needed if the tumor is close to the center of the chest.
- Lobectomy: The lungs are made up of 5 lobes (3 on the right and 2 on the left). In this surgery, the entire lobe containing the tumor(s) is removed. This is often the preferred type of operation for non-small cell lung cancer if it can be done.
- Segmentectomy or wedge resection: In these surgeries, only part of a lobe is removed. This approach might be used, for example, if a person doesn’t have enough lung function to withstand removing the whole lobe.
- Sleeve resection: This operation may be used to treat some cancers in large airways in the lungs. If you think of the large airway with a tumor as similar to the sleeve of a shirt with a stain a couple of inches above the wrist, the sleeve resection would be like cutting across the sleeve above and below the stain and then sewing the cuff back onto the shortened sleeve. A surgeon may be able to do this operation instead of a pneumonectomy to preserve more lung function.
With any of these operations, nearby lymph nodes are also removed to look for possible spread of the cancer. These operations require general anesthesia (where you are in a deep sleep) and are usually done through a surgical incision between the ribs in the side of the chest (called a thoracotomy).
The type of operation your doctor recommends depends on the size and location of the tumor and on how well your lungs are functioning. Doctors often prefer to do a more extensive operation (for example, a lobectomy instead of a segmentectomy) if a person’s lungs are healthy enough, as it may provide a better chance to cure the cancer.
When you wake up from surgery, you will have a tube (or tubes) coming out of your chest and attached to a special canister to allow excess fluid and air to drain out. The tube(s) will be removed once the fluid drainage and air leak subside. Generally, you will need to spend 5 to 7 days in the hospital after the surgery.
Possible risks and side effects of lung surgery
Surgery for lung cancer is a major operation and can have serious side effects, which is why surgery isn’t a good idea for everyone. While all surgeries carry some risks, these depend to some degree on the extent of the surgery and the person’s health beforehand.
Possible complications during and soon after surgery can include reactions to anesthesia, excess bleeding, blood clots in the legs or lungs, wound infections, and pneumonia. While it is rare, some people may not survive the surgery.
Recovering from lung cancer surgery typically takes weeks to months. If the surgery is done through a thoracotomy (a long incision in the chest), the surgeon must spread ribs to get to the lung, so the area near the incision will hurt for some time after surgery. Your activity might be limited for at least a month or two. People who have video-assisted thoracic surgery instead of thoracotomy tend to have less pain after surgery and to recover more quickly.
If your lungs are in good condition (other than the presence of the cancer) you can usually return to normal activities after some time if a lobe or even an entire lung has been removed. If you also have another lung disease such as emphysema or chronic bronchitis (which are common among long-time smokers), you might become short of breath with certain levels of activity after surgery.
Surgery for lung cancers with limited spread to other organs
If the lung cancer has spread to your brain or to an adrenal gland and there is only one tumor, you may benefit from having the tumor removed. This surgery should be considered only if the tumor in the lung can also be removed completely. Even then, not all lung cancer experts agree with this approach, especially if the tumor is in the adrenal gland.
For tumors in the brain, this is done by surgery through a hole in the skull (called a craniotomy). It should only be done if the tumor can be removed without damaging vital areas of the brain.
Radiation therapy
Depending on the stage of the non-small cell lung cancer and other factors, radiation therapy might be used:
- As the main treatment (sometimes along with chemotherapy), especially if the lung tumor can’t be removed because of its size or location, if a person isn’t healthy enough for surgery, or if a person doesn’t want surgery.
- After surgery (alone or along with chemotherapy) to try to kill any small areas of cancer that surgery might have missed.
- Before surgery (usually along with chemotherapy) to try to shrink a lung tumor to make it easier to operate on.
- To treat a single area of cancer spread, such as a tumor in the brain or an adrenal gland. (This might be done along with surgery to treat the main lung tumor.)
- To relieve (palliate) symptoms of advanced NSCLC such as pain, bleeding, trouble swallowing, cough, or problems caused by spread to other organs such as the brain. For example, brachytherapy is most often used to help relieve blockage of large airways by cancer.
Types of radiation therapy
There are 3 main types of radiation therapy:
- External beam radiation therapy
- Brachytherapy (internal radiation therapy)
- Proton therapy
External beam radiation therapy
External beam radiation therapy focuses radiation from outside the body on the cancer. This is the type of radiation therapy most often used to treat non-small cell lung cancer or its spread to other organs.
Before your treatments start, the radiation team will take careful measurements to determine the correct angles for aiming the radiation beams and the proper dose of radiation. This planning session, called simulation, usually includes getting imaging tests such as CT scans.
Treatment is much like getting an x-ray, but the radiation dose is stronger. The procedure itself is painless. Each treatment lasts only a few minutes, although the setup time – getting you into place for treatment – usually takes longer. Most often, radiation treatments to the lungs are given 5 days a week for 5 to 7 weeks, but this can vary based on the type of external beam radiation therapy and the reason it’s being given.
In recent years, newer external beam radiation therapy techniques have been shown to help doctors treat lung cancers more accurately while lowering the radiation exposure to nearby healthy tissues. These include:
- Three-dimensional conformal radiation therapy (3D-CRT): 3D-CRT uses special computers to precisely map the tumor’s location. Radiation beams are then shaped and aimed at the tumor(s) from several directions, which makes it less likely to damage normal tissues.
- Intensity modulated radiation therapy (IMRT): IMRT is an advanced form of 3D therapy. It uses a computer-driven machine that moves around you as it delivers radiation. Along with shaping the beams and aiming them at the tumor from several angles, the intensity (strength) of the beams can be adjusted to limit the dose reaching nearby normal tissues. This technique is used most often if tumors are near important structures such as the spinal cord. Many cancer centers now use IMRT.
- A variation of IMRT is called volumetric modulated arc therapy (VMAT). It uses a machine that delivers radiation quickly as it rotates once around the body. This allows each treatment to be given over just a few minutes.
- Stereotactic body radiation therapy (SBRT): SBRT, also known as stereotactic ablative radiotherapy (SABR), is sometimes used to treat very early-stage lung cancers when surgery isn’t an option due to a person’s health or in people who don’t want surgery. Instead of giving a small dose of radiation each day for several weeks, SBRT uses very focused beams of high-dose radiation given in fewer (usually 1 to 5) treatments. Several beams are aimed at the tumor from different angles. To target the radiation precisely, you are put in a specially designed body frame for each treatment. This reduces the movement of the lung tumor during breathing. Like other forms of external radiation, the treatment itself is painless. Early results with SBRT for smaller lung tumors have been very promising, and it seems to have a low risk of complications. It is also being studied for tumors that have spread to other parts of the body, such as the bones or liver.
- Stereotactic radiosurgery (SRS): SRS is a type of stereotactic radiation therapy that is given in only one session. It can sometimes be used instead of or along with surgery for single tumors that have spread to the brain. In one version of this treatment, a machine called a Gamma Knife® focuses about 200 beams of radiation on the tumor from different angles over a few minutes to hours. Your head is kept in the same position with a rigid frame. In another version, a linear accelerator (a machine that creates radiation) that is controlled by a computer moves around your head to deliver radiation to the tumor from many different angles. These treatments can be repeated if needed.
Brachytherapy (internal radiation therapy)
In people with non-small cell lung cancer, brachytherapy is sometimes used to shrink tumors in the airway to relieve symptoms. For this type of treatment, the doctor places a small source of radioactive material (often in the form of small pellets) directly into the cancer or into the airway next to the cancer. This is usually done through a bronchoscope, but it may also be done during surgery. The radiation travels only a short distance from the source, limiting the effects on surrounding healthy tissues. The radiation source is usually removed after a short time. Less often, small radioactive “seeds” are left in place permanently, and the radiation gets weaker over several weeks.
Several newer methods of giving radiation therapy have become available in recent years, For example, some newer radiation therapy machines have imaging scanners built into them. This advance, known as image guided radiation therapy (IGRT), lets the doctor take pictures of the lung and make minor adjustments in aiming just before giving the radiation. This may help deliver the radiation more precisely, which might result in fewer side effects.
Proton therapy
In people with non-small cell lung cancer, especially Stage 3, proton therapy may be an option. Proton therapy is a type of radiation that uses protons rather than x-rays. A proton is a positively charged particle, which can be targeted specifically to the tumor. Compared to x-rays, proton therapy beams are less likely to damage surrounding organs, such as the heart and esophagus (tube we use to swallow). This form of radiation therapy continues to be studied, and is offered in most specialized lung cancer treatment centers.
Possible side effects of radiation therapy
If you are going to get radiation therapy, it’s important to ask your doctor beforehand about the possible side effects so you know what to expect. Common side effects depend on where the radiation is aimed and can include:
- Fatigue
- Nausea and vomiting
- Loss of appetite and weight loss
- Skin changes in the area being treated, which can range from mild redness to blistering and peeling
- Hair loss where the radiation enters the body
Often these go away after treatment. When radiation is given with chemotherapy, the side effects are often worse.
Radiation therapy to the chest may damage your lungs and cause a cough, problems breathing, and shortness of breath. These usually improve after treatment is over, although sometimes they may not go away completely.
Your esophagus, which is in the middle of your chest, may be exposed to radiation, which could cause a sore throat and trouble swallowing during treatment. This might make it hard to eat anything other than soft foods or liquids for a while. This also improves after completion of treatment.
Radiation therapy to large areas of the brain can sometimes cause memory loss, headaches, trouble thinking, or reduced sexual desire. Usually these symptoms are minor compared with those caused by a brain tumor, but they can affect your quality of life.
Chemotherapy
Chemotherapy (chemo) is treatment with anti-cancer drugs injected into a vein or taken by mouth. These drugs enter the bloodstream and go throughout the body, making this treatment useful for cancer anywhere in the body.
Depending on the stage of non-small cell lung cancer and other factors, chemo may be used in different situations:
- Before surgery (sometimes along with radiation therapy) to try to shrink a tumor. This is known as neoadjuvant therapy.
- After surgery (sometimes along with radiation therapy) to try to kill any cancer cells that might have been left behind. This is known as adjuvant therapy.
- For locally advanced non-small cell lung cancer: Sometimes, chemo along with radiation therapy is given as the main treatment for more advanced cancers that have grown into nearby structures if surgery is not an option or for people who aren’t healthy enough for surgery.
- For metastatic (stage 4) non-small cell lung cancer: Chemo may be given for lung cancer that has spread to areas outside the lung, such as the bones, liver, or adrenal gland.
Chemo is often not recommended for patients in poor health, but advanced age by itself is not a barrier to getting chemo.
Drugs used to treat non-small cell lung cancer
The chemo drugs most often used for non-small cell lung cancer include:
- Cisplatin
- Carboplatin
- Paclitaxel (Taxol)
- Albumin-bound paclitaxel (nab-paclitaxel, Abraxane)
- Docetaxel (Taxotere)
- Gemcitabine (Gemzar)
- Vinorelbine (Navelbine)
- Etoposide (VP-16)
- Pemetrexed (Alimta)
Most often, treatment for non-small cell lung cancer uses a combination of 2 chemo drugs. Studies have shown that adding a third chemo drug doesn’t add much benefit and is likely to cause more side effects. Single-drug chemo is sometimes used for people who might not tolerate combination chemotherapy well, such as those in poor overall health or who are elderly.
If a combination is used, it often includes cisplatin or carboplatin plus one other drug. Sometimes combinations that do not include these drugs, such as gemcitabine with vinorelbine or paclitaxel, may be used.
For people with advanced lung cancers who meet certain criteria, a targeted therapy drug such as bevacizumab (Avastin), ramucirumab (Cyramza), or necitumumab (Portrazza) may be added to treatment as well.
Doctors give chemo in cycles, with a period of treatment (usually 1 to 3 days) followed by a rest period to allow the body time to recover. Some chemo drugs, though, are given every day. Chemo cycles generally last about 3 to 4 weeks. The schedule varies depending on the drugs used. For example, with some drugs, the chemo is given only on the first day of the cycle. With others, it is given for a few days in a row, or once a week. Then, at the end of the cycle, the chemo schedule repeats to start the next cycle.
For advanced cancers, the initial chemo combination is often given for 4 to 6 cycles. Some doctors now recommend giving treatment beyond this with a single chemo or targeted drug, even in people who have had a good response to their initial chemotherapy. Some studies have found that this continuing treatment, known as maintenance therapy, might help keep the cancer in check and help some people live longer.
Adjuvant and neoadjuvant chemo is often given for 3 to 4 months, depending on the drugs used. The length of treatment for advanced lung cancer is based on how well it is working and what side effects you have.
Lung Cancer Research
If the initial chemo treatment for advanced lung cancer is no longer working, the doctor may recommend second-line treatment with a single chemo drug such as docetaxel or pemetrexed, or with a targeted therapy or immunotherapy drug. Again, advanced age is no barrier to receiving these drugs as long as the person is in good general health.
New combinations: Many clinical trials are looking at newer combinations of chemotherapy drugs to determine which are the safest and most effective. This is especially important in patients who are older and have other health problems. Doctors are also studying better ways to combine chemotherapy with radiation therapy and other treatments.
Lab tests to help predict if chemo will be helpful: Doctors know that adjuvant chemotherapy after surgery may be more helpful for some people with early (stage I or II) cancers than for others, but figuring out which patients to give it to is not easy. In early studies, newer lab tests that look at patterns of certain genes in the cancer cells have shown promise in telling which people might benefit most. Larger studies of these tests are now trying to confirm their usefulness.
Other lab tests may help predict whether a lung cancer will respond to particular chemo drugs. For example, studies have found that tumors with high levels of the excision repair cross-complementation group 1 (ERCC1) gene protein are less likely to respond to chemo that includes cisplatin or carboplatin, while tumors with high levels of the RRM1 protein (Ribonucleotide Reductase Catalytic Subunit M1) seem less likely to respond to chemo with gemcitabine. Doctors are now looking to see if tests for these markers can help guide the choice of treatment, so these are not a part of standard treatment.
Possible side effects of chemotherapy
Chemo drugs attack cells that are dividing quickly, which is why they work against cancer cells. But other cells in the body, such as those in the bone marrow (where new blood cells are made), the lining of the mouth and intestines, and the hair follicles, also divide quickly. These cells are also likely to be affected by chemo, which can lead to certain side effects.
The side effects of chemo depend on the type and dose of drugs given and how long they are taken. Some common side effects include:
- Hair loss
- Mouth sores
- Loss of appetite
- Nausea and vomiting
- Diarrhea or constipation
- Increased chance of infections (from having too few white blood cells)
- Easy bruising or bleeding (from having too few blood platelets)
- Fatigue (from having too few red blood cells)
These side effects usually go away after treatment is finished. There are often ways to lessen these side effects. For example, drugs can be given to help prevent or reduce nausea and vomiting.
Some drugs can have specific side effects. For example, drugs such as cisplatin, vinorelbine, docetaxel, or paclitaxel can cause nerve damage (peripheral neuropathy). This can sometimes lead to symptoms (mainly in the hands and feet) such as pain, burning or tingling sensations, sensitivity to cold or heat, or weakness. In most people this goes away or gets better once treatment is stopped, but it may last a long time in some people.
Be sure to report any side effects you notice while getting chemo to your medical team so that they can be treated promptly. In some cases, the doses of the chemo drugs may need to be reduced or treatment may need to be delayed or stopped to prevent the effects from getting worse.
Targeted therapy drugs
Researchers are learning more about the inner workings of lung cancer cells that control their growth and spread. This is being used to develop new targeted therapy drugs. Many of these are already being used to treat non-small cell lung cancer. Others are now being tested in clinical trials to see if they can help people with advanced lung cancer live longer or relieve their symptoms. Newer targeted drugs being studied include ganetespib, nintedanib, selumetinib, dacomitinib, and custirsen.
Researchers are also working on lab tests to help predict which patients might be helped by which drugs. Studies have found that some patients do not benefit from certain targeted therapies, whereas others are more likely to have their tumors shrink. For example, a test can find changes in the epidermal growth factor receptor (EGFR) gene that make it much more likely that a person’s lung cancer will respond to treatment with a targeted drug called an EGFR inhibitor. Similar gene tests for other targeted treatments are now being studied. Predicting who might benefit could save some people from trying treatments that are unlikely to work for them and would probably cause unneeded side effects.
Drugs that target tumor blood vessel growth (Angiogenesis inhibitors)
For tumors to grow, they need to form new blood vessels to keep them nourished. This process is called angiogenesis. Some targeted drugs, called angiogenesis inhibitors, block this new blood vessel growth. Angiogenesis inhibitors help stop the formation of new blood vessels. Angiogenesis inhibitors are generally monoclonal antibodies (lab-made versions of a specific immune system protein) that target vascular the endothelial growth factor (VEGF), a protein that helps new blood vessels to form, called VEGF inhibitors.
Examples of Vascular Endothelial Growth Factor (VEGF) inhibitors:
- Bevacizumab (Avastin) is used in combination with chemotherapy, immunotherapy, or the targeted drug erlotinib for management of advanced or metastatic non-small cell lung cancer.
- Ramucirumab (Cyramza) is used in combination with the targeted drug erlotinib or chemotherapy for management of advanced or metastatic non-small cell lung cancer.
Side effects of angiogenesis inhibitors:
- Common side effects: Nosebleed, headache, high blood pressure, protein in urine, change in taste, dry skin, back pain, dry skin, excessive tearing
- Rare, but serious side effects: Severe bleeding, formation of holes (perforations) in the intestine, issues with wound healing, clots in the heart and/or brain, and brain damage called reversible posterior leukoencephalopathy syndrome. (RPLS), very high blood pressure, kidney damage, allergic reaction to the drug (infusion reaction), thyroid damage, ovarian failure, and fetal harm.
Because of the risks of bleeding, these drugs often aren’t used in people who are coughing up blood or who are taking drugs called blood thinners. The risk of serious bleeding in the lungs is higher in patients with the squamous cell type of non-small cell lung cancer, which is why current guidelines do not recommend using bevacizumab in people with this type of lung cancer.
Drugs that target cells with EGFR changes (EGFR inhibitors)
Epidermal growth factor receptor (EGFR) is a protein on the surface of cells. It normally helps the cells grow and divide. Some non-small cell lung cancer cells have too much EGFR, which makes them grow faster. Drugs called EGFR inhibitors can block the signal from EGFR that tells the cells to grow. Some of these drugs can be used to treat non-small cell lung cancer.
Examples of epidermal growth factor receptor (EGFR) inhibitors:
- EGFR inhibitors that target cells with either an exon 19 or exon 21 mutation
- Drugs such Afatinib (Gilotrif), Erlotinib (Tarceva), Dacomitinib (Vizimpro), Gefitinib (Iressa), and Osimertinib (Tagresso) can be given alone.
- Lazertinib (Lazcluze) in combination with amivantamab (Rybrevant).
- Osimertinib in combination with chemotherapy (cisplatin or carboplatin with pemetrexed).
- Erlotinib in combination with a VEGF inhibitor (ramucirumab or bevacizumab).
- Amivantamab in combination with chemo (carboplatin and pemetrexed).
- EGFR inhibitors that target cells with S768I, L861Q and/or G719X mutations
- Drugs such as afatinib (Gilotrif), osimertinib (Tagrisso), erlotinib (Tarceva), dacomitinib (Vizimpro), and gefitinib (Iressa) can be given alone.
- Amivantamab in combination with chemo (carboplatin and pemetrexed).
- EGFR inhibitors that target cells with an exon 20 mutation
- Amivantamab (Rybrevant) in combination with chemo (carboplatin and pemetrexed). Amivantamab is a monoclonal antibody (a lab-made version of a specific immune system protein) that targets two proteins that help cancer cells grow: EGFR and MET. Because it binds to two proteins, it’s called a bispecific antibody.
- Amivantamab given alone is also an option after chemotherapy has been tried.
Side effects of EGFR inhibitors:
- Common side effects: Skin problems, nail changes, fatigue, loss of appetite, nausea, vomiting, and diarrhea.
- Rare, but serious side effects: A lung condition called interstitial lung disease or pneumonitis; damage to the heart muscle; heart rhythm changes; other organ damage including kidney, gut and liver; harm to a fetus; and severe skin rashes.
Drugs that target cells with ALK gene changes (ALK inhibitors)
About 5% of non-small cell lung cancers have a rearrangement in a gene called ALK. This change is most often seen in non-smokers (or light smokers) who have the adenocarcinoma subtype of non-small cell lung cancer. The ALK gene rearrangement produces an abnormal ALK protein that causes the cells to grow and spread.
Drugs that target the abnormal ALK protein include:
- Third-generation ALK inhibitor [Lorlatinib (Lorbrena)] and second-generation ALK inhibitors [alectinib (Alecensa), brigatinib (Alunbrig), ceritinib (Zykadia), and ensartinib (Ensacove)] are often the preferred first treatment for advanced NSCLC with the ALK rearrangement mutation. These later generation ALK inhibitors tend to get into the brain better than crizotinib, a first-generation ALK inhibitor.
- Alectinib (Alecensa) can be given to patients with early stage ALK-positive NSCLC in the adjuvant setting (after the lung tumor has been removed).
- First- generation ALK inhibitor: Crizotinib (Xalkori) was the first ALK inhibitor approved for advanced non-small cell lung cancer with an ALK rearrangement. It is still given in certain situations.
Side effects of ALK inhibitors:
- Common side effects: Vision changes, nausea, vomiting, diarrhea, swelling in hands and/or feet, tiredness, muscle soreness, rash, cough, nerve damage (peripheral neuropathy), and changes in lab values.
- Rare, but serious side effects: Liver damage; a lung condition called interstitial lung disease or pneumonitis; changes in heart rhythm; and harm to a fetus.
Drugs that target cells with ROS1 gene changes (ROS1 inhibitors)
About 1% to 2% of non-small cell lung cancers have a rearrangement in a gene called ROS1. This change is most often seen in people who have the adenocarcinoma subtype of non-small cell lung cancer and whose tumors are also negative for ALK, KRAS and EGFR mutations. The ROS1 gene rearrangement is similar to the ALK gene rearrangement, and some drugs can work on cells with either ALK or ROS1 gene changes. These drugs, called ROS1 inhibitors, can often shrink tumors in people whose advanced lung cancers have a ROS1 gene change. They are taken as a pill.
Examples of ROS1 inhibitors:
- Entrectinib (Rozlytrek), crizotinib (Xalkori) and ceritinib (Zykadia) are often the first treatment options for advanced non-small cell lung cancer with a ROS1 rearrangement.
- Lorlatinib (Lorbrena) is an option for patients with ROS1-mutated advanced non-small cell lung cancer who have already been treated with the either entrectinib, crizotinib, or ceritinib.
- Repotrectinib (Augtyro) is an option for patients with ROS1-mutated advanced non-small cell lung cancer who have or who have not been treated with a ROS1 inhibitor.
Side effects of ROS1 inhibitors:
- Common side effects: Vision changes, muscle soreness, swelling in hands and/or feet, nerve damage (called peripheral neuropathy), diarrhea, fatigue, and changes in lab tests.
- Rare, but serious side effects: Liver damage; changes in heart rhythm and heart function; lung condition called interstitial lung disease or pneumonitis; high blood pressure; changes in mood or mental status; and harm to a fetus.
Drugs that target cells with BRAF gene changes (BRAF inhibitors)
In some non-small cell lung cancers, the cells have changes in the BRAF gene. Cells with these changes make an altered BRAF protein that helps them grow.
Examples of BRAF inhibitors:
- Combination treatment: A combination of BRAF inhibitor and a MEK inhibitor is often given together as the first or later treatment for advanced NSCLC with the BRAF V600E mutation. Examples include:
- Dabrafenib (Tafinlar), a BRAF inhibitor, with trametinib (Mekinist), a MEK inhibitor
Encorafenib (Braftovi), a BRAF inhibitor, with binimetinib (Mektovi), a MEK inhibitor
- Dabrafenib (Tafinlar), a BRAF inhibitor, with trametinib (Mekinist), a MEK inhibitor
- Single drug treatment: Vemurafenib (Zelboraf), another BRAF inhibitor, or dabrafenib can be taken alone if the patient can’t take the combination treatment.
These drugs can be used together to treat metastatic non-small cell lung cancer if it has a certain type of BRAF gene change.
These drugs are taken as pills or capsules each day.
Side effects of BRAF inhibitors:
- Common side effects: Skin thickening, rash, itching, sensitivity to the sun, headache, fever, joint pain, tiredness, hair loss, nausea, and diarrhea.
- Rare, but serious side effects: Bleeding, heart rhythm problems, liver or kidney problems, lung problems, severe allergic reactions, severe skin or eye problems, increased blood sugar levels, and squamous cell skin cancer.
Drugs that target cells with RET gene changes (RET inhibitors)
In a small percentage of non-small cell lung cancers, the tumor cells have rearrangement in the RET gene that cause them to make an abnormal form of the RET protein. This abnormal protein helps the tumor cells grow. Drugs known as RET inhibitors can be used to treat advanced non-small cell lung cancer with the RET rearrangement.
Examples of RET inhibitors:
- Selpercatinib (Retevmo) or pralsetinib (Gayreto) is often the preferred first treatment for metastatic non-small cell lung cancer with the RET rearrangement.
- Cabozantinib (Cometriq, Cabometyx) has activity against RET, ROS1, MET, and VEGF. It can be used to treat RET-mutated non-small cell lung cancer in certain situations.
These drugs are taken by mouth as capsules, typically once or twice a day.
Side effects of RET inhibitors:
- Common side effects: Dry mouth, diarrhea or constipation, high blood pressure, tiredness, swelling in hands and/or feet, skin rash, muscle and joint pain, and low blood cell counts or changes in other blood tests.
- Rare, but serious side effects: Liver damage, lung damage, allergic reactions, changes in heart rhythm, bleeding easily, and problems with wound healing.
Drugs that target cells with MET gene changes (MET inhibitors)
In some non-small cell lung cancers, cancer cells have changes in the MET gene, called a MET exon 14 skipping mutation, that cause them to make an abnormal form of the MET protein. This abnormal protein helps the cancer cells grow and spread. Drugs called MET inhibitors can be used to treat metastatic non-small cell lung cancer if the cancer cells have certain types of MET gene changes, by attacking the MET protein.
Examples of MET inhibitors:
- Capmatinib (Tabrecta) or tepotinib (Tepmetko) is often the preferred first treatment for metastatic non-small cell lung cancer with the MET exon 14 skipping mutation.
- Crizotinib (Xalkori) has activity against MET, ALK, and ROS1. It can be used to treat MET-mutated non-small cell lung cancer in certain situations.
These drugs are taken as a pill once or twice a day.
Side effects of MET inhibitors:
- Common side effects: Nausea, vomiting, diarrhea, tiredness, swelling in hands and/or feet, muscle and joint pain, low blood cell counts, or changes in other blood tests.
- Rare, but serious side effects: A lung condition called interstitial lung disease or pneumonitis; liver damage; harm to a fetus; and increased sensitivity to sunlight (photosensitivity).
Drugs that target cells with HER2 gene changes (HER2 inhibitors)
In a small percentage of non-small cell lung cancers, the cancer cells have certain changes in the HER2 (ERBB2) gene that help them grow. HER2-directed drugs can be used to treat metastatic non-small cell lung cancer if the cancer cells have certain types of HER2 gene changes. These drugs are infused into a vein (IV). They are typically given once every few weeks.
Examples of HER2 inhibitors:
- Fam-trastuzumab deruxtecan-nxki (Enhertu) is an antibody-drug conjugate (ADC). It’s made up of a lab-made antibody that targets the HER2 protein, which is linked to a chemotherapy drug. The antibody acts like a homing signal by attaching to the HER2 protein on cancer cells, bringing the chemo directly to them. It can be used to treat non-small cell lung cancer with HER2 mutations if you’ve already had at least one other type of drug treatment.
- Ado-trastuzumab emtansine (Kadcyla) is also a HER2-targeted antibody-drug conjugate. It can be used to treat HER2-mutated non-small cell lung cancer in certain situations.
- Zenocutuzumab-zbco (Bizengri) is a bispecific antibody that binds to HER2 and HER3 and prevents a protein called neuregulin 1 (NRG1) from binding to HER3. It can be used to treat non-small cell lung cancer with a NRG1 gene fusion mutation, if you’ve already had at least one other type of drug treatment.
Side effects of HER2 inhibitors:
- Common side effects: Low white blood cell, red blood cell, and platelet counts; tiredness; hair loss; nausea and vomiting; muscle and joint pain; and changes in other blood tests.
- Rare, but serious side effects, specific to fam-trastuzumab deruxtecan: Changes in heart function and a severely low white blood cell count, which increases your risk of infection.
Drugs that target cells with TRK gene changes (TRK inhibitors)
A very small number of non-small cell lung cancers have changes in one of the NTRK genes, called NTRK gene fusions. Cells with these gene changes make abnormal TRK proteins, which can lead to abnormal cell growth and cancer. TRK inhibitors target and disable the proteins made by the NTRK genes.
Examples of TRK inhibitors:
- Larotrectinib (Vitrakyi) or entrectinib (Rozlytrek) is often the preferred first treatment for metastatic non-small cell lung cancer with an NTRK gene fusion mutation.
These drugs are taken as pills, once or twice daily.
Side effects of NTRK inhibitors:
- Common side effects: Abnormal liver tests; decreased white blood cell and red blood cells; muscle and joint pain; tiredness; diarrhea or constipation; nausea and vomiting; and stomach pain.
- Rare, but serious side effects: Mental changes, such as confusion, changes in mood, changes in sleep; liver damage; changes in heart rhythm and/or function; vision changes; and harm to a fetus.
Radiofrequency Ablation
This treatment might be an option for some people some small lung tumors that are near the outer edge of the lungs, especially if they can’t tolerate surgery.
Radiofrequency Ablation uses high-energy radio waves to heat the tumor. A thin, needle-like probe is put through the skin and moved in until the tip is in the tumor. Placement of the probe is guided by CT scans. Once the tip is in place, an electric current is passed through the probe, which heats the tumor and destroys the cancer cells.
Radiofrequency Ablation is usually done as an outpatient procedure, using local anesthesia (numbing medicine) where the probe is inserted. You may be given medicine to help you relax as well.
You might have some pain where the needle was inserted for a few days after the procedure. Major complications are uncommon, but they can include the partial collapse of a lung (which often goes away on its own) or bleeding into the lung.
Types of ablation for lung cancer:
- Radiofrequency ablation (RFA) uses high-energy radio waves to heat and destroy the tumor cells.
- Cryoablation uses liquid nitrogen or argon gas to freeze and destroy the tumor cells.
- Microwave ablation uses electromagnetic energy to heat and destroy the tumor cells.
Ways to do ablation for lung cancer:
- Ablation using percutaneous approach: A thin, needle-like probe is put through the skin and moved in until the tip is in the tumor. Placement of the probe is guided by CT scans. This is usually done as an outpatient procedure, using local anesthesia (numbing medicine) where the probe is inserted. You may be given medicine to help you relax.
- Ablation using bronchoscopy: You will be given anesthesia for the bronchoscopy. Ablation will be done through the bronchoscopy tube and your airway, rather than from the outside through the skin. This is still quite a new way to give ablative treatment.
Tumor Treating Fields Therapy
Researchers have found that exposing some types of cancer cells to alternating electric fields, also known as tumor treating fields (TTFields), can interfere with the cells’ ability to grow and spread. A wearable device known as Optune Lua is a tumor treating fields (TTFields) treatment that creates such electric fields. It is an option to help treat some people with non-small cell lung cancer (NSCLC). Optune Lua can be used along with either an immune checkpoint inhibitor or with the chemotherapy drug docetaxel to treat some people with metastatic NSCLC that has grown during or after treatment with chemotherapy.
How is Tumor Treating Fields (TTF) therapy given?
For tumor treating fields (TTFields) treatment, the chest and/or back is shaved (if needed), and pads containing sets of electrodes are placed on the skin, usually 2 on the chest and 2 on the back. The electrodes are attached to a battery pack (kept in a backpack) and are worn for most of the day – typically at least 18 hours. They create mild electric currents that are thought to affect cancer cells more than normal cells.
Possible side effects of Tumor Treating Fields (TTF) therapy
Side effects of the device are usually limited to the electrode sites. They can include:
- Skin irritation
- Allergic reactions
- Local warmth and tingling sensations
- Muscle twitching
- Infections
- Breakdown of the skin (ulcers)
Immunotherapy for Non-Small Cell Lung Cancer
Researchers are developing immunotherapy drugs that can help the body’s immune system fight the cancer. An important part of your immune system is its ability to keep itself from attacking normal cells in the body. To do this, it uses “checkpoint” proteins on immune cells, which act like switches that need to be turned on (or off) to start an immune response. Cancer cells sometimes use these checkpoints to avoid being attacked by the immune system. Drugs that target these checkpoints called checkpoint inhibitors can be used to treat some people with non-small cell lung cancer (NSCLC).
PD-1/PD-L1 inhibitors
Nivolumab (Opdivo), pembrolizumab (Keytruda), and cemiplimab (Libtayo) target PD-1, a protein on certain immune cells (called T cells) that normally helps keep these cells from attacking other cells in the body. By blocking PD-1, these drugs boost the immune response against cancer cells. This can shrink some tumors or slow their growth.
Atezolizumab (Tecentriq) and durvalumab (Imfinzi) target PD-L1, a protein related to PD-1 that is found on some tumor cells and immune cells. Blocking this protein can help boost the immune response against cancer cells. This can shrink some tumors or slow their growth.
These drugs can be used in different situations to treat non-small cell lung cancer. In some cases, before one of these drugs can be used, lab tests might need to be done on the cancer cells to show they have at least a certain amount of the PD-L1 protein (which would mean these drugs are more likely to work) and if the cancer cells have “driver mutations.” Depending on the results of those lab tests and the stage of lung cancer, the PD-1/PD-L1 inhibitors can be used in different ways. They can be given with or without chemotherapy, with or without a CTLA-4 inhibitor (see below), before or after surgery for early-stage lung cancer, or for a long period of time for advanced-stage lung cancers.
All of these drugs can be given as intravenous (IV) infusions. Atezolizumab (as Tecentriq Hybreza) and nivolumab (as Opdivo Qvantig) can also be given as an injection under the skin (subcutaneously) over several minutes. Depending on the drug, they might be given every 2, 3, 4, or 6 weeks.
Possible side effects
- Common side effects of these drugs can include fatigue, cough, nausea, itching, skin rash, loss of appetite, constipation, joint pain, and diarrhea.
- Other, more serious side effects occur less often.
- Infusion reactions: Some people might have an infusion reaction while getting these drugs. This is like an allergic reaction, and can include fever, chills, flushing of the face, rash, itchy skin, feeling dizzy, wheezing, and trouble breathing. It’s important to tell your cancer care team right away if you have any of these symptoms while getting these drugs.
- Autoimmune reactions: These drugs work by removing one of the safeguards on the body’s immune system. Sometimes the immune system starts attacking other parts of the body, which can cause serious or even life-threatening problems in the lungs, intestines, liver, hormone-making glands, kidneys, or other organs.
It’s very important to report any new side effects to your cancer care team as soon as possible. If serious side effects do occur, treatment may need to be stopped and you may get high doses of corticosteroids to suppress your immune system.
CTLA-4 inhibitors
Ipilimumab (Yervoy) and tremelimumab (Imjudo) are also drugs that boost the immune response, but they block CTLA-4, another protein on T cells that normally helps keep them in check. These drugs are used along with a PD-1 inhibitor (ipilimumab with nivolumab, and tremelimumab with durvalumab); they are not used alone. They might be an option as part of the first treatment for certain types of advanced non-small cell lung cancer, most often along with chemo as well.
These drugs are given by intravenous (IV) infusion, usually once every 3 or 6 weeks.
Possible side effects
- The most common side effects of these drugs include fatigue, diarrhea, skin rash, itching, muscle or bone pain, and belly pain.
- Serious side effects seem to happen more often with CTLA-4 inhibitors than with the PD-1 and PD-L1 inhibitors.
- Infusion reactions: Some people might have an infusion reaction while getting one of these drugs. This is like an allergic reaction, and can include fever, chills, flushing of the face, rash, itchy skin, feeling dizzy, wheezing, and trouble breathing. It’s important to tell your doctor or nurse right away if you have any of these symptoms while getting an infusion. It’s very important to report any new side effects during or after treatment with any of these drugs to your cancer care team promptly. If serious side effects do occur, you may need to stop treatment and take high doses of corticosteroids to suppress your immune system.
- Autoimmune reactions: These drugs can sometimes cause the immune system to attack other parts of the body, which can lead to serious problems in the intestines, liver, hormone-making glands, nerves, skin, eyes, or other organs. In some people, these side effects can be life-threatening.
Maintenance therapy
For people with advanced lung cancers who get chemotherapy, combinations of 2 chemo drugs (sometimes along with a targeted drug) are typically given for about 4 to 6 cycles. Some studies have found that with cancers that have not progressed, continuing treatment beyond the 4 to 6 cycles with a single chemo drug such as pemetrexed or with a targeted drug may help some people live longer. This is known as maintenance therapy. A possible downside to this continued treatment is that people may not get a break from treatment side effects. Some doctors now recommend maintenance therapy, while others await further research on this topic.
Palliative Procedures for Non-Small Cell Lung Cancer
Palliative, or supportive care, is aimed at relieving symptoms and improving a person’s quality of life.
People with lung cancer often benefit from procedures to help with problems caused by the cancer. For example, people with advanced lung cancer can have shortness of breath. This can be caused by a number of things, including fluid around the lung or an airway that is blocked by a tumor. Although treating the cancer with chemotherapy or other drugs may help with this, other treatments may be needed as well.
Treating fluid buildup in the area around the lungs
Sometimes fluid can build up in the chest outside of the lungs. This is called a pleural effusion. It can press on the lungs and cause trouble breathing.
Thoracentesis
This is done to drain the fluid. For this procedure, the doctor will numb an area in the chest, and then place a hollow needle into the space between the lungs and the ribs to drain the fluid. This is often done using ultrasound to guide the needle into the fluid.
Pleurodesis
This procedure might be done to remove the fluid and keep it from coming back.
One way to do this is to make a small cut in the skin of the chest wall, and place a hollow tube (called a chest tube) into the chest to remove the fluid. Then a substance is instilled into the chest through the tube that causes the linings of the lung (visceral pleura) and chest wall (parietal pleura) to stick together, sealing the space and limiting further fluid buildup. A number of substances can be used for this, such as talc, the antibiotic doxycycline, or a chemotherapy drug like bleomycin. The tube is often left in for a couple of days to drain any new fluid that might collect.
Another way to do this is to blow talc into the space around the lungs during an operation. This is done through a small incision using thoracoscopy.
Catheter placement
This is another way to control the buildup of fluid. One end of the catheter (a thin, flexible tube) is placed in the chest through a small cut in the skin, and the other end is left outside the body. This is done in a doctor’s office or hospital. Once in place, the catheter can be attached to a special bottle or other device to allow the fluid to drain out on a regular basis.
Treating fluid buildup around the heart
Lung cancer can sometimes spread to the area around the heart. This can lead to fluid buildup inside the sac around the heart (called a pericardial effusion). The fluid can press on the heart, affecting how well it works.
Pericardiocentesis
In this procedure, the fluid is drained with a needle placed into the space around the heart. This is usually done using an ultrasound of the heart (echocardiogram) to guide the needle.
Creating a pericardial window
This procedure can be done to keep the fluid from building up again. During surgery, a piece of the sac around the heart (the pericardium) is removed to allow the fluid to drain into the chest or belly.
Treating an airway blocked by a tumor
If the cancer is growing into an airway in the lung, it can block the airway and cause problems like pneumonia or shortness of breath. Treatments can often relieve the blockage in the airway.
Photodynamic therapy (PDT)
This type of treatment can be used to treat very early-stage lung cancers that are only in the outer layers of the lung airways, when other treatments aren’t appropriate. It can also be used to help open up airways blocked by tumors to help people breathe better.
For this technique, a light-activated drug called porfimer sodium (Photofrin) is injected into a vein. This drug collects more in cancer cells than in normal cells. After a couple of days (to give the drug time to build up in the cancer cells), a bronchoscope is passed down the throat and into the lung. This may be done with either local anesthesia (where the throat is numbed) and sedation, or with general anesthesia (where you are in a deep sleep). A special laser light on the end of the bronchoscope is aimed at the tumor, which activates the drug and causes the cells to die. The dead cells are then removed a few days later during a bronchoscopy. This process can be repeated if needed.
PDT can cause swelling in the airway for a few days, which may lead to some shortness of breath, as well as coughing up blood or thick mucus. Some of this drug also collects in normal cells in the body, such as skin and eye cells. This can make you very sensitive to sunlight or strong indoor lights. Too much exposure can cause serious skin reactions (like a severe sunburn), so doctors recommend staying out of any strong light for several weeks after the injection.
Laser therapy
Lasers can sometimes be used to treat very small tumors in the linings of airways. They can also be used to help open up airways blocked by larger tumors to help people breathe better.
You are usually asleep (under general anesthesia) for this type of treatment. The laser is on the end of a bronchoscope, which is passed down the throat and next to the tumor. The doctor then aims the laser beam at the tumor to burn it away. This treatment can usually be repeated, if needed.
Stent placement
If a lung tumor has grown into an airway and is causing problems, sometimes a hard silicone or metal tube called a stent is placed in the airway to help keep it open using a bronchoscope. This is often done after other treatments such as PDT or laser therapy.
Small cell lung cancer
Small cell lung cancer (SCLC) also called oat cell cancer, is a type of fast-growing, aggressive lung cancer that usually starts in the central airways and spreads quickly, often before symptoms appear 110, 111, 112, 113, 114, 115, 116, 117, 118. Small cell lung cancer (SCLC) typically starts in the bronchi, the main airways in your chest. Almost all cases of small cell lung cancer (SCLC) are linked to smoking 119, 120, 121. Small cell lung cancer (SCLC) accounts for about 10% to 15% of lung cancers. The cancer cells are small, but they usually grow very quickly and create large tumors. Small cell lung cancers are grade 3 cancers also known as high-grade or poorly differentiated cancer, which mean that the cancer cells look very abnormal and are likely to grow and spread quickly. Small cell lung cancer is more responsive to chemotherapy and radiation therapy than other cell types of lung cancer 122, 123, 124. Platinum–etoposide-based chemotherapy combined with immunotherapy constitutes the standard treatment for patients with small-cell lung cancer (SCLC) 113. However, a cure is difficult to achieve because small cell lung cancer has a greater tendency to spread rapidly (metastasize) to other parts of your body, including your brain, liver, and bone by the time of diagnosis 125, 123, 126. The median survival rate seldom exceeds 1 year 118, 127. The overall 5 year survival rate for small cell lung cancer (SCLC) patients continues to be under 7% 125, 128. Each year, around 250,000 patients are diagnosed with small-cell lung cancer (SCLC) worldwide, with approximately 200,000 deaths resulting from small-cell lung cancer 118, 122, 127.
Approximately 60-70% of patients with small cell lung cancer (SCLC) have clinically disseminated or extensive disease at presentation 129. Extensive-stage small cell lung cancer (SCLC) is incurable. When given combination chemotherapy, patients with extensive-stage small cell lung cancer (SCLC) have a complete response rate of more than 20% and a median survival longer than 7 months; however, only 2% are alive at 5 years 130. For individuals with limited-stage small cell lung cancer (SCLC) that is treated with combination chemotherapy plus chest radiation, a complete response rate of 80% and survival of 17 months have been reported; 12-15% of patients are alive at 5 years 131.
Indicators of poor prognosis include the following 129:
- Relapsed disease
- Weight loss of greater than 10% of baseline body weight
- Poor performance status
- Hyponatremia 132
- Genome-wide association studies have identified single-nucleotide polymorphisms (eg, within the promoter region of YAP1 on chromosome 11q22) that may affect survival in patients with small cell lung cancer (SCLC) 133, 134.
There are currently 2 types of small cell lung cancer (SCLC) 135:
- Small cell carcinoma (SCLC): This is the most common form of small cell lung cancer.
- Combined small cell carcinoma (C-SCLC): Combined small cell carcinoma (C-SCLC) represents about 2% to 5% of all small cell carcinomas. Combined small cell carcinoma (C-SCLC) is a combination of small cell lung cancer cells with additional components from any of the non-small cell lung carcinoma (NSCLC) histological types. These usually include adenocarcinoma (ADC), squamous cell carcinoma (SCC), large-cell neuroendocrine carcinoma (LCNEC) and less commonly spindle cell carcinoma or giant cell carcinoma 136. No minimum percentage of the additional component is required for a combined small cell carcinoma (C-SCLC) diagnosis with the exception of mixed large-cell neuroendocrine carcinoma (LCNEC) and small cell carcinoma (SCLC), where a minimum of 10% LCNEC component is required given the frequent presence of scattered large cells in surgically resected SCLC 136.
Patients diagnosed with combined small cell carcinoma (C-SCLC) are currently recommended to receive the same treatment as small cell carcinoma (SCLC) in the absence of clear evidence suggesting different strategies 137, 138.
Small cell lung cancers are also classified as neuroendocrine tumors 139, 140. You might also hear them called lung neuroendocrine neoplasms (NENs). This means the same as lung neuroendocrine cancer. Neuroendocrine tumors (NETs) are rare tumors that develop in cells of the neuroendocrine system. In small cell lung cancer, the tumor starts in the neuroendocrine cells of the lung. Neuroendocrine cells are specialized cells that act like both nerve cells and endocrine cells, receiving signals from the nervous system (like nerve cells) and responding by releasing hormones (like endocrine cells), such as serotonin (5HT), gastrin-releasing peptide (GRP), neuron-specific enolase (NSE), and bombesin 125, 141. It is worth noting that calcitonin gene-related peptide (CGRP), neural cell adhesion molecule 1 (NCAM1), and mammalian achaete-scute complex homolog 1 (MASH1/ASCL1) have significant functions in neuronal differentiation, and are prominently expressed in pulmonary neuroendocrine cells (PNECs) 125, 142.
There are 2 key groups of lung neuroendocrine cancer:
- Lung neuroendocrine tumors (NETs): Neuroendocrine tumors (NETs) include the following:
- Low-grade typical carcinoid. Typical carcinoids are grade 1 cancers. Typical carcinoids are also called carcinoid tumors. They are slow growing and less likely to spread outside the lungs. The cancer cells look very like normal cells. Doctors also call these well differentiated cancers. Around 2 in every 100 lung cancers (around 2%) diagnosed in the US every year are typical carcinoids.
- Intermediate-grade atypical carcinoid. Atypical carcinoids are grade 2 cancers. Atypical carcinoids are also called carcinoid tumors. Doctors also call these moderately differentiated cancers. They behave somewhere between grade 1 and grade 3 cancers. They usually grow faster than typical carcinoids. And they are more likely to spread. The cancer cells look more abnormal. Fewer than 1 in every 100 lung cancers (1%) diagnosed in the US every year are atypical carcinoids.
- High-grade neuroendocrine tumors, including large-cell neuroendocrine carcinoma (LCNEC) and small cell lung cancer (SCLC).
- Lung neuroendocrine carcinomas (NECs) – these include small cell neuroendocrine carcinomas and large cell neuroendocrine carcinomas (LCNEC). Both neuroendocrine carcinomas are categorized as high-grade neoplasms, in contrast to typical carcinoid and atypical carcinoid neoplasms, which are low-grade and intermediate grade, respectively 139, 140.
- Small cell lung cancer arising from neuroendocrine cells forms one extreme of the spectrum of neuroendocrine carcinomas (NECs) of the lung.
- Large cell neuroendocrine carcinomas (LCNEC) are grade 3 cancers. They tend to grow quickly and are more likely to spread. The cancer cells look very abnormal. Doctors also call these cancers poorly differentiated cancers. Around 3 out of every 100 lung cancers (3%) diagnosed in the US every year are this type. They are linked to smoking.
Neuroendocrine tumors (NETs) and neuroendocrine carcinomas (NECs) are very different. So it is important to know which one you have. Talk to your doctor or cancer specialist if you are not sure.
Small cell lung cancer (SCLC) primarily originates from lung neuroendocrine cells (PNECs), but can also develop from lung epithelial cells, including basal or club cells and cuboidal alveolar type 2 (AT2) cells in particular instances 113. Researchers have identified Tuft cells as potential progenitor cells in a specific subtype of small-cell lung cancer (SCLC) 125, 143. Tuft cells, also known as brush cells, are chemosensory cells that reside in the epithelial lining of the lungs 125, 144. The development of small-cell lung cancer (SCLC) is associated with loss-of-function mutations in genes such as tumor protein 53 (TP53) and retinoblastoma 1 (RB1) in lung neuroendocrine cells (PNECs), tuft, club, or AT2 cells 113.
Small cell lung cancer (SCLC) was initially classified as a lymphosarcoma in 1879 by Harting and Hesse 145, 140; however, it was reclassified as tiny ‘oat cell’ carcinomas of the lung in 1926 140, 146. Since then, small cell lung cancer (SCLC) has been widely recognized as oat cell carcinoma 140, 147, 148. According to the World Health Organization (WHO) Classification of Tumors, “small cell lung cancer is defined by light microscopy as a tumor with cells that have a small size, a round-to-fusiform shape, scant cytoplasm, finely granular nuclear chromatin and absent or inconspicuous nucleoli” 111. Necrosis may be extensive, and cells manifest a high mitotic rate. small cell lung cancers originate from epithelial cells, and up to 90% will express thyroid transcription factor-1 (TTF1). Epithelial cell markers including cytokeratin can be used to distinguish small cell lung cancer from lymphoma and other neuroendocrine tumors.
Because of differences in clinical behavior, therapy, and epidemiology, small cell lung cancers are classified separately in the World Health Organization (WHO) revised classification 139, 149. Small cell lung cancer is now divided into small cell lung cancer (SCLC) and combined small-cell lung cancer (C-SCLC) 150. No minimum percentage of the additional component is required for a combined small cell lung cancer diagnosis with the exception of mixed large cell neuroendocrine carcinoma (LCNEC) and small cell lung cancer, in which a 10% minimum large cell neuroendocrine carcinoma (LCNEC) component is required 151.
Figure 10. Small-cell lung cancer subtypes
Footnotes: Small cell lung cancer (SCLC) can be categorized into specific subtypes defined by the varying expression of 4 essential transcriptional regulators: achaete-scute homolog 1 (ASCL1; also referred to as ASH1) (SCLC-A subtype), neurogenic differentiation factor 1 (NEUROD1) (SCLC-N subtype), yes-associated protein 1 (YAP1) (SCLC-Y subtype), and POU class 2 homeobox 3 (POU2F3) (SCLC-P subtype). The most recent nomenclature designates a subtype known as ‘inflamed’ small-cell lung cancer (SCLC-I) 128, 152, 153, 122, 125, 154, 155. The relationship among these subtype-specific transcription factors encompasses varying levels of neuroendocrine differentiation. In a comprehensive multiomic evaluation of 437 small cell lung cancers, primarily metastatic, the overall distribution of samples was as follows: 35.7% coming from the A subgroup, 17.6% coming from the N subgroup, 6.4% coming from the P subgroup, 21.1% coming from the Y subgroup, and 19.2% coming from mixed subgroups 121, 156. The A and N subtypes are typically associated with elevated expression of neuroendocrine markers, while the P and Y subtypes exhibit reduced levels of these markers 121.
[Source 113 ]Small Cell Lung Cancer causes
Scientists don’t know what causes each case of lung cancer. But they do know many of the risk factors for these cancers and how some of them can cause cells to become cancerous.
Smoking
Tobacco smoking is by far the leading cause of small cell lung cancer. Most small cell lung cancer deaths are caused by smoking or exposure to secondhand smoke, with only about 2% of cases arising in never-smokers 157. Patients with small cell lung cancer should be encouraged to stop smoking, as smoking cessation is associated with improved survival 158.
Smoking is clearly the strongest risk factor for lung cancer, but it often interacts with other factors. Smokers exposed to other known risk factors such as radon and asbestos are at even higher risk. Not everyone who smokes gets lung cancer, so other factors like genetics probably play a role as well.
Lung cancer in non-smokers
It is rare for someone who has never smoked to be diagnosed with small cell lung cancer, but it can happen. Lung cancer in non-smokers can be caused by exposure to radon, secondhand smoke, air pollution, or other factors. Workplace exposures to asbestos, diesel exhaust, or certain other chemicals can also cause lung cancers in some people who don’t smoke.
A small portion of lung cancers occur in people with no known risk factors for the disease. Some of these might just be random events that don’t have an outside cause, but others might be due to factors that we don’t yet know about.
Gene changes that may lead to lung cancer
Some of the risk factors for lung cancer can cause certain changes in the DNA of lung cells. These changes can lead to abnormal cell growth and, sometimes, cancer. DNA is the chemical in each of our cells that makes up our genes, which control how our cells function. We usually look like our parents because they are the source of our DNA. But DNA also can influence our risk for developing certain diseases, such as some kinds of cancer.
Some genes help control when cells grow, divide into new cells, and die:
- Genes that help cells grow, divide, or stay alive are called oncogenes.
- Genes that help control cell division or cause cells to die at the right time are called tumor suppressor genes.
Cancers can be caused by DNA changes that turn on oncogenes or turn off tumor suppressor genes.
Inherited gene changes
Some people inherit DNA mutations (changes) from their parents that greatly increase their risk for developing certain cancers. But inherited mutations alone are not thought to cause very many lung cancers.
Still, genes do seem to play a role in some families with a history of lung cancer. For example, some people seem to inherit a reduced ability to break down or get rid of certain types of cancer-causing chemicals in the body, such as those found in tobacco smoke. This could put them at higher risk for lung cancer.
Other people may inherit faulty DNA repair mechanisms that make it more likely they will end up with DNA changes. People with DNA repair enzymes that don’t work normally might be especially vulnerable to cancer-causing chemicals and radiation.
Researchers are developing tests that may help identify such people, but these tests are not yet used routinely. For now, doctors recommend that all people avoid tobacco smoke and other exposures that might increase their cancer risk.
Acquired gene changes
Gene changes related to SCLC are usually acquired during life rather than inherited. Acquired mutations in lung cells often result from exposure to factors in the environment, such as cancer-causing chemicals in tobacco smoke. But some gene changes may just be random events that sometimes happen inside a cell, without having an outside cause.
Acquired changes in certain genes, such as the TP53 and RB1 tumor suppressor genes, are thought to be important in the development of small cell lung cancer. Changes in these and other genes may also make some lung cancers more likely to grow and spread than others. Not all lung cancers share the same gene changes, so there are undoubtedly changes in other genes that have not yet been found.
Small Cell Lung Cancer Risk Factors
A risk factor is anything that affects your chance of getting a disease such as cancer. Different cancers have different risk factors. Some risk factors, like smoking, can be changed. Others, like a person’s age or family history, can’t be changed.
But having a risk factor, or even several, does not mean that you will get the disease. And some people who get the disease may have few or no known risk factors.
Several risk factors can make you more likely to develop lung cancer. These factors are related to the risk of lung cancer in general, so it’s possible that some of these might not apply to small cell lung cancer.
Tobacco smoke
Smoking is by far the leading risk factor for lung cancer. About 80% of all lung cancer deaths are thought to result from smoking, and this number is probably even higher for small cell lung cancer. It’s very rare for someone who has never smoked to have small cell lung cancer. The risk for lung cancer among smokers is many times higher than among non-smokers. The longer you smoke and the more packs per day you smoke, the greater your risk.
Cigar smoking and pipe smoking are almost as likely to cause lung cancer as cigarette smoking. Smoking low-tar or “light” cigarettes increases lung cancer risk as much as regular cigarettes. Smoking menthol cigarettes might increase the risk even more, as the menthol may allow smokers to inhale more deeply.
Secondhand smoke: If you don’t smoke, breathing in the smoke of others (called secondhand smoke or environmental tobacco smoke) can increase your risk of developing lung cancer. Secondhand smoke is thought to cause more than 7,000 deaths from lung cancer each year.
Exposure to radon
Radon is a radioactive gas that occurs naturally when uranium in soil and rocks breaks down. It cannot be seen, tasted, or smelled. According to the US Environmental Protection Agency (EPA), radon is the second leading cause of lung cancer, and is the leading cause among non-smokers.
Outdoors, there is so little radon that it is not likely to be dangerous. But indoors, radon can become more concentrated. Breathing it in exposes your lungs to small amounts of radiation. This might increase your risk of lung cancer.
Homes and other buildings in nearly any part of the United States can have high indoor radon levels (especially in basements).
Exposure to asbestos
People who work with asbestos (such as in some mines, mills, textile plants, places that use insulation, and shipyards) are several times more likely to die of lung cancer. Lung cancer risk is much greater in workers exposed to asbestos who also smoke. It’s not clear how much low-level or short-term exposure to asbestos might raise lung cancer risk.
People exposed to large amounts of asbestos also have a greater risk of developing mesothelioma, a type of cancer that starts in the pleura (the lining surrounding the lungs).
In recent years, government regulations have greatly reduced the use of asbestos in commercial and industrial products. It’s still present in many homes and other older buildings, but it’s not usually considered harmful as long as it’s not released into the air by deterioration, demolition, or renovation.
Other cancer-causing substances in the workplace
Other carcinogens (cancer-causing substances) found in some workplaces that can increase lung cancer risk include:
- Radioactive ores such as uranium
- Inhaled chemicals such as arsenic, beryllium, cadmium, silica, vinyl chloride, nickel compounds, chromium compounds, coal products, mustard gas, and chloromethyl ethers
- Diesel exhaust
The government and industry have taken steps in recent years to help protect workers from many of these exposures. But the dangers are still there, so if you work around these products, be careful to limit your exposure whenever possible.
Air pollution
In cities, air pollution (especially near heavily trafficked roads) appears to raise the risk of lung cancer slightly. This risk is far less than the risk caused by smoking, but some researchers estimate that worldwide about 5% of all deaths from lung cancer may be due to outdoor air pollution.
Arsenic in drinking water
Studies of people in parts of Southeast Asia and South America with high levels of arsenic in their drinking water have found a higher risk of lung cancer. In most of these studies, the levels of arsenic in the water were many times higher than those typically seen in the United States, even in areas where arsenic levels are above normal. For most Americans who are on public water systems, drinking water is not a major source of arsenic.
Radiation therapy to the lungs
People who have had radiation therapy to the chest for other cancers are at higher risk for lung cancer, particularly if they smoke. Examples include people who have been treated for Hodgkin disease or women who get chest radiation after a mastectomy for breast cancer. Women who receive radiation therapy to the breast after a lumpectomy do not appear to have a higher than expected risk of lung cancer.
Personal or family history of lung cancer
If you have had lung cancer, you have a higher risk of developing another lung cancer.
Brothers, sisters, and children of those who have had lung cancer may have a slightly higher risk of lung cancer themselves, especially if the relative was diagnosed at a younger age. It’s not clear how much of this risk might be due to shared genes among family members and how much might be from shared household exposures (such as tobacco smoke or radon).
Researchers have found that genetics does seem to play a role in some families with a strong history of lung cancer. Research is ongoing in this area.
Certain dietary supplements
Studies looking at the possible role of vitamin supplements in reducing lung cancer risk have not been promising so far. In fact, 2 large studies found that smokers who took beta carotene supplements actually had an increased risk of lung cancer. The results of these studies suggest that smokers should avoid taking beta carotene supplements.
Factors with uncertain or unproven effects on lung cancer risk
Marijuana smoke
There are some reasons to think that smoking marijuana might increase lung cancer risk:
- Marijuana smoke contains tar and many of same the cancer-causing substances that are in tobacco smoke. (Tar is the sticky, solid material that remains after burning, which is thought to contain most of the harmful substances in smoke.)
- Marijuana cigarettes (joints) are typically smoked all the way to the end, where tar content is the highest.
- Marijuana is inhaled very deeply and the smoke is held in the lungs for a long time, which gives any cancer-causing substances more opportunity to deposit in the lungs.
- Because marijuana is still illegal in many places, it may not be possible to control what other substances it might contain.
Those who use marijuana tend to smoke fewer marijuana cigarettes in a day or week than the amount of tobacco consumed by cigarette smokers. The lesser amount smoked would make it harder to see an impact on lung cancer risk.
It’s been hard to study whether there is a link between marijuana and lung cancer because marijuana has been illegal in many places for so long, and it’s not easy to gather information about the use of illegal drugs.
Also, in studies that have looked at past marijuana use in people who had lung cancer, most of the marijuana smokers also smoked cigarettes. This can make it hard to know how much any increase in risk is from tobacco and how much might be from marijuana. More research is needed to know the cancer risks from smoking marijuana.
Talc and talcum powder
Talc is a mineral that in its natural form may contain asbestos. Some studies have suggested that talc miners and millers might have a higher risk of lung cancer and other respiratory diseases because of their exposure to industrial grade talc. But other studies have not found an increase in lung cancer rate.
Talcum powder is made from talc. By law since the 1970s, all home-use talcum products (baby, body, and facial powders) in the United States have been asbestos-free. The use of cosmetic talcum powder has not been found to increase lung cancer risk.
Small Cell Lung Cancer prevention
Not all lung cancers can be prevented. But there are things you can do that might lower your risk, such as changing the risk factors that you can control.
Stay away from tobacco
The best way to reduce your risk of lung cancer is not to smoke and to avoid breathing in other people’s smoke.
If you stop smoking before a cancer develops, your damaged lung tissue gradually starts to repair itself. No matter what your age or how long you’ve smoked, quitting may lower your risk of lung cancer and help you live longer.
Avoid radon
Radon is an important cause of lung cancer. You can reduce your exposure to radon by having your home tested and treated, if needed.
Avoid or limit exposure to cancer-causing chemicals
Avoiding exposure to known cancer-causing chemicals, in the workplace and elsewhere, might also be helpful. People working where these exposures are common should try to keep exposure to a minimum when possible.
Eat a healthy diet
A healthy diet with lots of fruits and vegetables may also help reduce your risk of lung cancer. Some evidence suggests that a diet high in fruits and vegetables may help protect against lung cancer in both smokers and non-smokers. But any positive effect of fruits and vegetables on lung cancer risk would be much less than the increased risk from smoking.
Trying to reduce the risk of lung cancer in current or former smokers by giving them high doses of vitamins or vitamin-like drugs has not been successful so far. In fact, some studies have found that supplements of beta-carotene, a nutrient related to vitamin A, appear to increase the rate of lung cancer in these people.
Some people who get lung cancer do not have any clear risk factors. Although we know how to prevent most lung cancers, at this time we don’t know how to prevent all of them.
Small Cell Lung Cancer signs and symptoms
Most lung cancers do not cause any symptoms until they have spread, but some people with early lung cancer do have symptoms. If you go to your doctor when you first notice symptoms, your cancer might be diagnosed at an earlier stage, when treatment is more likely to be effective.
Most of these symptoms are more likely to be caused by something other than lung cancer. Still, if you have any of these problems, it’s important to see your doctor right away so the cause can be found and treated, if needed. The most common symptoms of lung cancer are:
- A cough that does not go away or gets worse
- Coughing up blood or rust-colored sputum (spit or phlegm)
- Chest pain that is often worse with deep breathing, coughing, or laughing
- Hoarseness
- Weight loss and loss of appetite
- Shortness of breath
- Feeling tired or weak
- Infections such as bronchitis and pneumonia that don’t go away or keep coming back
- New onset of wheezing
When lung cancer spreads to other parts of the body, it may cause:
- Bone pain (like pain in the back or hips)
- Nervous system changes (such as headache, weakness or numbness of an arm or leg, dizziness, balance problems, or seizures), from cancer spread to the brain
- Yellowing of the skin and eyes (jaundice), from cancer spread to the liver
- Lumps near the surface of the body, due to cancer spreading to the skin or to lymph nodes (collection of immune system cells) such as those in the neck or above the collarbone
Some lung cancers can cause syndromes, which are groups of specific symptoms.
Horner syndrome
Cancers of the upper part of the lungs are sometimes called Pancoast tumors. These tumors are more likely to be non-small cell lung cancer (NSCLC) than small cell lung cancer (SCLC).
Pancoast tumors can affect certain nerves to the eye and part of the face, causing a group of symptoms called Horner syndrome:
- Drooping or weakness of one eyelid
- A smaller pupil (dark part in the center of the eye) in the same eye
- Reduced or absent sweating on the same side of the face
Pancoast tumors can also sometimes cause severe shoulder pain.
Superior vena cava syndrome
The superior vena cava is a large vein that carries blood from the head and arms back to the heart. It passes next to the upper part of the right lung and the lymph nodes inside the chest. Tumors in this area can press on the superior vena cava, which can cause the blood to back up in the veins. This can lead to swelling in the face, neck, arms, and upper chest (sometimes with a bluish-red skin color). It can also cause headaches, dizziness, and a change in consciousness if it affects the brain. While superior vena cava syndrome can develop gradually over time, in some cases it can become life-threatening, and needs to be treated right away.
Paraneoplastic syndromes
Some lung cancers make hormone-like substances that enter the bloodstream and cause problems with distant tissues and organs, even though the cancer has not spread to those tissues or organs. These problems are called paraneoplastic syndromes. Sometimes these syndromes may be the first symptoms of lung cancer. Because the symptoms affect other organs, patients and their doctors may first suspect that a disease other than lung cancer is causing them.
Some of the more common paraneoplastic syndromes associated with small cell lung cancer are:
- SIADH (syndrome of inappropriate anti-diuretic hormone): In this condition, the cancer cells make a hormone (ADH) that causes the kidneys to retain (hold) water. This lowers salt levels in the blood. Symptoms of SIADH can include fatigue, loss of appetite, muscle weakness or cramps, nausea, vomiting, restlessness, and confusion. Without treatment, severe cases may lead to seizures and coma.
- Cushing syndrome: In this condition, the cancer cells make ACTH, a hormone that makes the adrenal glands secrete cortisol. This can lead to symptoms such as weight gain, easy bruising, weakness, drowsiness, and fluid retention. Cushing syndrome can also cause high blood pressure and high blood sugar levels, or even diabetes.
- Nervous system problems: Small cell lung cancer can sometimes cause the body’s immune system to attack parts of the nervous system, which can lead to problems. One example is a muscle disorder called Lambert-Eaton syndrome. In this syndrome, muscles around the hips become weak. One of the first signs may be trouble getting up from a sitting position. Later, muscles around the shoulder may become weak. A rarer problem is paraneoplastic cerebellar degeneration, which can cause loss of balance and unsteadiness in arm and leg movement, as well as trouble speaking or swallowing. Small cell lung cancer can also cause other nervous system problems, such as muscle weakness, sensation changes, vision problems, or even changes in behavior.
Again, many of these symptoms can also be caused by something other than lung cancer. Still, if you have any of these problems, it’s important to see your doctor right away so the cause can be found and treated, if needed.
Small Cell Lung Cancer Diagnosis
Screening can find some lung cancers, but most lung cancers are found because they are causing problems. If you have possible signs or symptoms of lung cancer, see your doctor, who will examine you and may order some tests. The actual diagnosis of lung cancer is made after looking at a sample of your lung cells under a microscope.
Medical history and physical exam
- Your doctor will ask about your medical history to learn about your symptoms and possible risk factors. You will also be examined for signs of lung cancer or other health problems.
If the results of your history and physical exam suggest you might have lung cancer, you will have tests to look for it. These could include imaging tests and/or biopsies of lung tissue.
Imaging tests to look for lung cancer
Imaging tests use x-rays, magnetic fields, sound waves, or radioactive substances to create pictures of the inside of your body. Imaging tests might be done for a number of reasons both before and after a diagnosis of lung cancer, including:
- To look at suspicious areas that might be cancer
- To learn if and how far cancer has spread
- To help determine if treatment is working
- To look for possible signs of cancer coming back after treatment
Chest x-ray
This is often the first test your doctor will do to look for any abnormal areas in the lungs. Plain x-rays of your chest can be done at imaging centers, hospitals, and even in some doctors’ offices. If the x-ray result is normal, you probably don’t have lung cancer (although some lung cancers may not show up on an x-ray). If something suspicious is seen, your doctor will likely order more tests.
Computed tomography (CT) scan
A CT scan combines many x-rays to make detailed cross-sectional images of your body.
A CT scan is more likely to show lung tumors than a routine chest x-ray. It can also show the size, shape, and position of any lung tumors and can help find enlarged lymph nodes that might contain cancer that has spread from the lung. Most people with small cell lung cancer (SCLC) will get a CT of the chest and abdomen to look at the lungs and lymph nodes, and to look for abnormal areas in the adrenal glands, liver, and other organs that might be from the spread of lung cancer. Some people will get a CT of the brain to look for cancer spread, but an MRI is more likely to be used when looking at the brain.
CT guided needle biopsy: If a suspected area of cancer is deep within your body, a CT scan can be used to guide a biopsy needle precisely into the suspected area.
Magnetic resonance imaging (MRI) scan
Like CT scans, MRI scans show detailed images of soft tissues in the body. But MRI scans use radio waves and strong magnets instead of x-rays.
Most patients with SCLC will have an MRI scan of the brain to look for possible cancer spread, although a CT scan may be used instead. MRI may also be used to look for possible spread to the spinal cord if the patients have certain symptoms.
Positron emission tomography (PET) scan
For a PET scan, you are injected with a slightly radioactive form of sugar, which collects mainly in cancer cells. A special camera is then used to create a picture of areas of radioactivity in the body.
A PET scan can be a very important test if you appear to have early stage (or limited) SCLC. Your doctor can use this test to see if the cancer has spread to lymph nodes or other organs, which can help determine your treatment options. A PET scan can also give a better idea whether an abnormal area on a chest x-ray or CT scan might be cancer. PET scans are also useful if your doctor thinks the cancer may have spread but doesn’t know where.
PET/CT scan: Some machines can do both a PET scan and a CT scan at the same time. This lets the doctor compare areas of higher radioactivity on the PET scan with the more detailed appearance of that area on the CT scan. For people with SCLC, PET/CT scans are used more often than PET scans alone.
Bone scan
A bone scan can help show if a cancer has spread to the bones. This test is done mainly when there is reason to think the cancer may have spread to the bones (because of symptoms such as bone pain) and other test results aren’t clear.
For this test, you are injected with a slightly radioactive chemical that collects mainly in abnormal areas of bone. A special camera is then used to create a picture of areas of radioactivity in the body..
PET scans can also usually show if the cancer has spread to the bones, so you usually won’t need a bone scan if a PET scan has already been done.
Tests to diagnose lung cancer
Symptoms and the results of imaging tests might suggest that a person has lung cancer, but the actual diagnosis is made by looking at cells from your lung with a microscope.
The cells can be taken from lung secretions (sputum or phlegm), fluid removed from the area around the lung (thoracentesis), or from a suspicious area (biopsy). The choice of which test(s) to use depends on the situation.
Sputum cytology
For this test, a sample of sputum (mucus you cough up from the lungs) is looked at under a microscope to see if it has cancer cells. The best way to do this is to get early morning samples from you 3 days in a row. This test is more likely to help find cancers that start in the major airways of the lung, such as most small cell lung cancers and squamous cell lung cancers. It may not be as helpful for finding other types of lung cancer.
Thoracentesis
If fluid has built up around your lungs (called a pleural effusion), doctors can use thoracentesis to relieve symptoms and to see if it is caused by cancer spreading to the lining of the lungs (pleura). The buildup might also be caused by other conditions, such as heart failure or an infection.
For this procedure, the skin is numbed and a hollow needle is inserted between the ribs to drain the fluid. (In a similar test called pericardiocentesis, fluid is removed from within the sac around the heart.) A microscope is used to check the fluid for cancer cells. Chemical tests of the fluid are also sometimes useful in telling a malignant (cancerous) pleural effusion from one that is not.
If a malignant pleural effusion has been diagnosed, thoracentesis may be repeated to remove more fluid. Fluid buildup can keep the lungs from filling with air, so thoracentesis can help a person breathe better.
Needle biopsy
Doctors can often use a hollow needle to get a small sample from a suspicious area (mass).
- In a fine needle aspiration (FNA) biopsy, the doctor uses a syringe with a very thin, hollow needle to withdraw (aspirate) cells and small fragments of tissue.
- In a core biopsy, a larger needle is used to remove one or more small cores of tissue. Samples from core biopsies are larger than FNA biopsies, so they are often preferred.
An advantage of needle biopsies is that they don’t require a surgical incision, but in some cases they might not provide enough of a sample to make a diagnosis.
Transthoracic needle biopsy: If the suspected tumor is in the outer part of the lungs, the biopsy needle can be inserted through the skin on the chest wall. The area where the needle is to be inserted may be numbed with local anesthesia first. The doctor then guides the needle into the area while looking at the lungs with either fluoroscopy (which is like an x-ray, but the image is shown on a screen rather than on film) or CT scans. Unlike fluoroscopy, CT doesn’t give a constant picture, so the needle is inserted toward the mass, a CT image is taken, and the direction of the needle is guided based on the image. This is repeated a few times until the needle is within the mass.
A possible complication of this procedure is that air may leak out of the lung at the biopsy site and into the space between the lung and the chest wall. This is called a pneumothorax. It can cause part of the lung to collapse and could cause trouble breathing. If the air leak is small, it often gets better without any treatment. Larger air leaks are treated by putting a small tube into the chest space and sucking out the air over a day or two, after which it usually heals on its own.
Other approaches to needle biopsies: An FNA biopsy may also be done to check for cancer in the lymph nodes between the lungs:
- Transtracheal FNA or transbronchial FNA is done by passing the needle through the wall of the trachea (windpipe) or bronchi (the large airways leading into the lungs) during bronchoscopy or endobronchial ultrasound (described below).
- Some patients have an FNA biopsy done during endoscopic esophageal ultrasound (described below) by passing the needle through the wall of the esophagus.
Bronchoscopy
Bronchoscopy can help the doctor find some tumors or blockages in the larger airways of the lungs. It may be used to find a lung tumor or to take a sample of a tumor to see if it is cancer.
For this exam, a lighted, flexible fiber-optic tube (called a bronchoscope) is passed through the mouth or nose and down into the windpipe and bronchi. The mouth and throat are sprayed first with a numbing medicine. You may also be given medicine through an intravenous (IV) line to make you feel relaxed.
Small instruments can be passed down the bronchoscope to take biopsy samples. The doctor can also sample cells that line the airways by using a small brush (bronchial brushing) or by rinsing the airways with sterile saltwater (bronchial washing). These tissue and cell samples are then looked at under a microscope.
Tests to find lung cancer spread
If lung cancer has been found, it’s often important to know if it has spread to the lymph nodes in the space between the lungs (mediastinum) or other nearby areas. This can affect a person’s treatment options.
Several types of tests might be done to look for cancer spread if surgery could be an option for treatment, but this is not often the case for small cell lung cancer. These tests are used more often for non-small cell lung cancer.
Endobronchial ultrasound
Ultrasound is a type of imaging test that uses sound waves to create pictures of the inside of your body. For this test, a small, microphone-like instrument called a transducer gives off sound waves and picks up the echoes as they bounce off body tissues. The echoes are converted by a computer into an image on a computer screen.
For endobronchial ultrasound, a bronchoscope is fitted with an ultrasound transducer at its tip and is passed down into the windpipe. This is done with numbing medicine (local anesthesia) and light sedation.
The transducer can be pointed in different directions to look at lymph nodes and other structures in the mediastinum (the area between the lungs). If suspicious areas such as enlarged lymph nodes are seen on the ultrasound, a hollow needle can be passed through the bronchoscope to get biopsy samples of them. The samples are then sent to a lab to be looked at with a microscope.
Endoscopic esophageal ultrasound
This test is like endobronchial ultrasound, except the doctor passes an endoscope (a lighted, flexible scope) down the throat and into the esophagus (the tube connecting the throat to the stomach). This is done with numbing medicine (local anesthesia) and light sedation.
The esophagus is just behind the windpipe and is close to some lymph nodes inside the chest to which lung cancer may spread. As with endobronchial ultrasound, the transducer can be pointed in different directions to look at lymph nodes and other structures inside the chest that might contain lung cancer. If enlarged lymph nodes are seen on the ultrasound, a hollow needle can be passed through the endoscope to get biopsy samples of them. The samples are then sent to a lab to be looked at under a microscope.
Mediastinoscopy and mediastinotomy
These procedures may be done to look more directly at and get samples from the structures in the mediastinum (the area between the lungs). They are done in an operating room by a surgeon while you are under general anesthesia (in a deep sleep). The main difference between the two is in the location and size of the incision.
Mediastinoscopy: A small cut is made in the front of the neck and a thin, hollow, lighted tube is inserted behind the sternum (breast bone) and in front of the windpipe to look at the area. Instruments can be passed through this tube to take tissue samples from the lymph nodes along the windpipe and the major bronchial tube areas. Looking at the samples under a microscope can show if they contain cancer cells.
Mediastinotomy: The surgeon makes a slightly larger incision (usually about 2 inches long) between the second and third ribs next to the breast bone. This lets the surgeon reach some lymph nodes that cannot be reached by mediastinoscopy.
Thoracoscopy
This procedure can be done to find out if cancer has spread to the spaces between the lungs and the chest wall, or to the linings of these spaces (called pleura). It can also be used to sample tumors on the outer parts of the lungs as well as nearby lymph nodes and fluid, and to assess whether a tumor is growing into nearby tissues or organs. This procedure is not often done just to diagnose lung cancer, unless other tests such as needle biopsies are unable to get enough samples for the diagnosis.
Thoracoscopy is done in an operating room while you are under general anesthesia (in a deep sleep). A small cut (incision) is made in the side of the chest wall. (Sometimes more than one cut is made.) The doctor then puts a thin, lighted tube with a small video camera on the end through the incision to view the space between the lungs and the chest wall. Using this, the doctor can see possible cancer deposits on the lining of the lung or chest wall and remove small pieces of the tissue to be looked at under the microscope. (When certain areas can’t be reached with thoracoscopy, the surgeon may need to make a larger incision in the chest wall, known as a thoracotomy.)
Thoracoscopy can also be used as part of the treatment to remove part of a lung in some early-stage lung cancers. This type of operation, known as video-assisted thoracic surgery (VATS), is described in more detail in Surgery for Small Cell Lung Cancer.
Bone marrow aspiration and biopsy
These tests look for spread of the cancer into the bone marrow. Bone marrow is the soft, inner part of certain bones where new blood cells are made.
The two tests are usually done at the same time. The samples are most often taken from the back of the pelvic (hip) bone.
In bone marrow aspiration, you lie on a table (either on your side or on your belly). The skin over the hip is cleaned. Then the skin and the surface of the bone are numbed with local anesthetic, which may cause a brief stinging or burning sensation. A thin, hollow needle is then inserted into the bone, and a syringe is used to suck out a small amount of liquid bone marrow. Even with the anesthetic, most people still have some brief pain when the marrow is removed.
A bone marrow biopsy is usually done just after the aspiration. A small piece of bone and marrow is removed with a slightly larger needle that is pushed down into the bone. The biopsy will likely also cause some brief pain.
Bone marrow aspiration and biopsy are sometimes done in patients thought to have early (limited) stage SCLC but who have blood test results suggesting the cancer might have reached the bone marrow. In recent years, PET scans have been used more often for staging, so these tests are now rarely done for SCLC.
Lab tests of biopsy and other samples
Samples that have been collected during biopsies or other tests are sent to a pathology lab. A pathologist, a doctor who uses lab tests to diagnose diseases such as cancer, will look at the samples under a microscope and may do other special tests to help better classify the cancer. Cancers from other organs can spread to the lungs. It’s very important to find out where the cancer started, because treatment is different depending on the type of cancer.
The results of these tests are described in a pathology report, which is usually available within about a week. If you have any questions about your pathology results or any diagnostic tests, talk to your doctor.
Blood tests
Blood tests are not used to diagnose lung cancer, but they can help to get a sense of a person’s overall health. For example, they can be used to help tell if a person is healthy enough to have surgery.
A complete blood count (CBC) determines whether your blood has normal numbers of different types of blood cells. For example, it can show if you are anemic (have a low number of red blood cells), if you could have trouble with bleeding (due to a low number of blood platelets), or if you are at increased risk for infections (due to a low number of white blood cells). This test will be repeated regularly if you are treated with chemotherapy, because these drugs can affect blood-forming cells of the bone marrow.
Blood chemistry tests can help spot abnormalities in some of your organs, such as the liver or kidneys. For example, if cancer has spread to the bones, it may cause higher than normal levels of calcium and alkaline phosphatase.
Lung function tests
Lung (or pulmonary) function tests (PFTs) may be done after lung cancer is diagnosed to see how well your lungs are working. They are generally only needed if surgery might be an option in treating the cancer, which is rare in small cell lung cancer. Surgery to remove lung cancer requires removing part or all of a lung, so it’s important to know how well the lungs are working beforehand.
There are different types of PFTs, but they all basically have you breathe in and out through a tube that is connected to a machine that measures airflow.
Small Cell Lung Cancer Stages
The stage of a cancer describes how far it has spread. The stage is one of the most important factors in deciding how to treat the cancer and determining how successful treatment might be.
For treatment purposes, most doctors use a 2-stage system that divides SCLC into limited stage and extensive stage. For limited stage cancer, a person might benefit from more aggressive treatments such as chemotherapy combined with radiation therapy to try to cure the cancer. For extensive stage disease, chemotherapy alone is likely to be a better option to control (not cure) the cancer.
Limited stage and extensive stage
For treatment purposes, most doctors use a 2-stage system that divides small cell lung cancer into limited stage and extensive stage 159. This helps determine if a person might benefit from more aggressive treatments such as chemotherapy combined with radiation therapy to try to cure the cancer (for limited stage cancer), or whether chemotherapy alone is likely to be a better option (for extensive stage cancer).
Limited stage
This means that the cancer is only on one side of the chest and can be treated with a single radiation field. This generally includes cancers that are only in one lung (unless tumors are widespread throughout the lung), and that might have also reached the lymph nodes on the same side of the chest.
This generally means your cancer:
- is only in one lung (unless tumors are widespread throughout the lung)
- and that might also have reached the lymph nodes on the same side of the chest – for example, in the center of the chest or above the collar bone
Cancer in lymph nodes above the collarbone (called supraclavicular nodes) might still be considered limited stage as long as they are on the same side of the chest as the cancer. Some doctors also include lymph nodes at the center of the chest (mediastinal lymph nodes) even when they are closer to the other side of the chest.
What is important is that the cancer is confined to an area that is small enough to be treated with radiation therapy in one “port” or one treatment area. Only about 1 out of 3 people with small cell lung cancer have limited stage cancer when it is first found.
If you have limited disease you are likely to have chemotherapy as well as radiotherapy treatment. This is sometimes called chemoradiotherapy.
Some people also have surgery.
Extensive stage
Extensive disease means that the cancer has spread widely throughout the lung, to the other lung, to lymph nodes on the other side of the chest, or to other parts of the body (including the bone marrow). Many doctors consider small cell lung cancer that has spread to the fluid around the lung (a malignant pleural effusion) to be extensive stage as well. About 2 out of 3 people with small cell lung cancer have extensive disease when their cancer is first found.
Extensive disease small cell lung cancer might have spread:
- within the chest (either to the other lung or to lymph nodes further away from the cancer)
- or to other parts of your body
Or there may be cancer cells in the fluid around the lung (a malignant pleural effusion).
The main treatment for extensive stage disease is chemotherapy.
The TNM staging system
A more formal system to describe the growth and spread of lung cancer is the American Joint Committee on Cancer TNM staging system, which is based on:
- The size of the main (primary) tumor (T) and whether it has grown into nearby areas.
- Whether the cancer has spread to nearby (regional) lymph nodes (N). Lymph nodes are small bean-shaped collections of immune cells to which cancers often spread before going to other parts of the body.
- Whether the cancer has spread (metastasized) (M) to other organs of the body. (The most common sites are the brain, bones, adrenal glands, liver, kidneys, and the other lung.)
Numbers or letters appear after T, N, and M to provide more details about each of these factors. Higher numbers mean the cancer is more advanced. Once the T, N, and M categories have been determined, this information is combined in a process called stage grouping, to assign an overall stage.
Details of the TNM staging system
The TNM staging system is complex and can be hard for patients (and even some doctors) to understand. If you have any questions about the stage of your cancer, ask your doctor to explain it to you.
T categories for lung cancer
- TX: The main (primary) tumor can’t be assessed, or cancer cells were seen on sputum cytology or bronchial washing but no tumor can be found.
- T0: There is no evidence of a primary tumor.
- Tis: Cancer is found only in the top layers of cells lining the air passages. It has not grown into deeper lung tissues. This is also known as carcinoma in situ.
- T1: The tumor is no larger than 3 centimeters (cm)—slightly less than 1¼ inches—across, has not reached the membranes that surround the lungs (visceral pleura), and does not affect the main branches of the bronchi.
- If the tumor is 2 cm (about 4/5 of an inch) or less across, it is called T1a. If the tumor is larger than 2 cm but not larger than 3 cm across, it is called T1b.
- T2: The tumor has 1 or more of the following features:
- It is larger than 3 cm across but not larger than 7 cm.
- It involves a main bronchus, but is not closer than 2 cm (about ¾ inch) to the carina (the point where the windpipe splits into the left and right main bronchi).
- It has grown into the membranes that surround the lungs (visceral pleura).
- The tumor partially clogs the airways, but this has not caused the entire lung to collapse or develop pneumonia.
- If the tumor is 5 cm or less across, it is called T2a. If the tumor is larger than 5 cm across (but not larger than 7 cm), it is called T2b.
- T3: The tumor has 1 or more of the following features:
- It is larger than 7 cm across.
- It has grown into the chest wall, the breathing muscle that separates the chest from the abdomen (diaphragm), the membranes surrounding the space between the two lungs (mediastinal pleura), or membranes of the sac surrounding the heart (parietal pericardium).
- It has grown into a main bronchus and is closer than 2 cm (about ¾ inch) to the carina, but it does not involve the carina itself.
- It has grown into the airways enough to cause an entire lung to collapse or to cause pneumonia in the entire lung.
- Two or more separate tumor nodules are present in the same lobe of a lung
- T4: The cancer has 1 or more of the following features:
- A tumor of any size has grown into the space between the lungs (mediastinum), the heart, the large blood vessels near the heart (such as the aorta), the windpipe (trachea), the tube connecting the throat to the stomach (esophagus), the backbone, or the carina.
- Two or more separate tumor nodules are present in different lobes of the same lung.
N categories for lung cancer
- NX: Nearby lymph nodes cannot be assessed.
- N0: There is no spread to nearby lymph nodes.
- N1: The cancer has spread to lymph nodes within the lung and/or around the area where the bronchus enters the lung (hilar lymph nodes). Affected lymph nodes are on the same side as the primary tumor.
- N2: The cancer has spread to lymph nodes around the carina (the point where the windpipe splits into the left and right bronchi) or in the space between the lungs (mediastinum). Affected lymph nodes are on the same side as the primary tumor.
- N3: The cancer has spread to lymph nodes near the collarbone on either side, and/or spread to hilar or mediastinal lymph nodes on the side opposite the primary tumor.
M categories for lung cancer
- M0: No spread to distant organs or areas. This includes the other lung, lymph nodes further away than those mentioned in the N stages above, and other organs or tissues such as the liver, bones, or brain.
- M1a: Any of the following:
- The cancer has spread to the other lung.
- The cancer has spread as nodules (small lumps) in the pleura (the lining of the lung).
- Cancer cells are found in the fluid around the lung (called a malignant pleural effusion).
- Cancer cells are found in the fluid around the heart (called a malignant pericardial effusion).
- M1b: The cancer has spread to distant lymph nodes or to other organs such as the liver, bones, or brain.
Stage grouping for lung cancer
Once the T, N, and M categories have been assigned, this information is combined to assign an overall stage of 0, I, II, III, or IV. This process is called stage grouping. Some stages are subdivided into A and B. The stages identify cancers that have a similar outlook (prognosis). Patients with lower stage numbers tend to have a better outlook.
TX, N0, M0: Cancer cells are seen in a sample of sputum or other lung fluids, but the cancer isn’t found with other tests, so its location can’t be determined.
Stage 0
Tis, N0, M0: The cancer is found only in the top layers of cells lining the air passages. It has not invaded deeper into other lung tissues and has not spread to lymph nodes or distant sites.
Stage 1A
T1a/T1b, N0, M0: The cancer is no larger than 3 cm across, has not reached the membranes that surround the lungs, and does not affect the main branches of the bronchi. It has not spread to lymph nodes or distant sites.
Stage 1B
T2a, N0, M0: The cancer has 1 or more of the following features:
- The main tumor is between larger than 3 cm across but not larger than 5 cm.
- The tumor has grown into a main bronchus, but is not within 2 cm of the carina (and it is not larger than 5 cm).
- The tumor has grown into the visceral pleura (the membranes surrounding the lungs) and is not larger than 5 cm.
- The tumor is partially clogging the airways (and is not larger than 5 cm).
- The cancer has not spread to lymph nodes or distant sites.
Stage 2A
There are 3 main combinations of categories that make up this stage.
- T1a/T1b, N1, M0: The cancer is no larger than 3 cm across, has not grown into the membranes that surround the lungs, and does not affect the main branches of the bronchi. It has spread to lymph nodes within the lung and/or around the area where the bronchus enters the lung (hilar lymph nodes). These lymph nodes are on the same side as the cancer. It has not spread to distant sites.
- OR
- T2a, N1, M0: The cancer has 1 or more of the following features:
- The main tumor is larger than 3 cm across but not larger than 5 cm.
- The tumor has grown into a main bronchus, but is not within 2 cm of the carina (and it is not larger than 5 cm).
- The tumor has grown into the visceral pleura (the membranes surrounding the lungs) and is not larger than 5 cm.
- The tumor is partially clogging the airways (and is not larger than 5 cm).
- The cancer has also spread to lymph nodes within the lung and/or around the area where the bronchus enters the lung (hilar lymph nodes). These lymph nodes are on the same side as the cancer. It has not spread to distant sites.
- OR
- T2b, N0, M0: The cancer has 1 or more of the following features:
- The main tumor is larger than 5 cm across but not larger than 7 cm.
- The tumor has grown into a main bronchus, but is not within 2 cm of the carina (and it is between 5 and 7 cm across).
- The tumor has grown into the visceral pleura (the membranes surrounding the lungs) and is between 5 and 7 cm across.
- The tumor is partially clogging the airways (and is between 5 and 7 cm across).
- The cancer has not spread to lymph nodes or distant sites.
Stage 2B
There are 2 combinations of categories that make up this stage.
T2b, N1, M0: The cancer has 1 or more of the following features:
- The main tumor is larger than 5 cm across but not larger than 7 cm.
- The tumor has grown into a main bronchus, but is not within 2 cm of the carina (and it is between 5 and 7 cm across).
- The tumor has grown into the visceral pleura (the membranes surrounding the lungs) and is between 5 and 7 cm across.
- The cancer is partially clogging the airways (and is between 5 and 7 cm across).
It has also spread to lymph nodes within the lung and/or around the area where the bronchus enters the lung (hilar lymph nodes). These lymph nodes are on the same side as the cancer. It has not spread to distant sites.
OR
T3, N0, M0: The main tumor has 1 or more of the following features:
- It is larger than 7 cm across.
- It has grown into the chest wall, the breathing muscle that separates the chest from the abdomen (diaphragm), the membranes surrounding the space between the lungs (mediastinal pleura), or membranes of the sac surrounding the heart (parietal pericardium).
- It invades a main bronchus and is closer than 2 cm (about ¾ inch) to the carina, but it does not involve the carina itself.
- It has grown into the airways enough to cause an entire lung to collapse or to cause pneumonia in the entire lung.
- Two or more separate tumor nodules are present in the same lobe of a lung.
The cancer has not spread to lymph nodes or distant sites.
Stage 3A
Three main category combinations make up this stage.
T1 to T3, N2, M0: The main tumor can be any size. It has not grown into the space between the lungs (mediastinum), the heart, the large blood vessels near the heart (such as the aorta), the windpipe (trachea), the tube connecting the throat to the stomach (esophagus), the backbone, or the carina. It has not spread to different lobes of the same lung.
The cancer has spread to lymph nodes around the carina (the point where the windpipe splits into the left and right bronchi) or in the space between the lungs (mediastinum). These lymph nodes are on the same side as the main lung tumor. The cancer has not spread to distant sites.
OR
T3, N1, M0: The cancer has 1 or more of the following features:
- It is larger than 7 cm across.
- It has grown into the chest wall, the breathing muscle that separates the chest from the abdomen (diaphragm), the membranes surrounding the space between the lungs (mediastinal pleura), or membranes of the sac surrounding the heart (parietal pericardium).
- It invades a main bronchus and is closer than 2 cm to the carina, but it does not involve the carina itself.
- Two or more separate tumor nodules are present in the same lobe of a lung
- It has grown into the airways enough to cause an entire lung to collapse or to cause pneumonia in the entire lung.
The cancer has also spread to lymph nodes within the lung and/or around the area where the bronchus enters the lung (hilar lymph nodes). These lymph nodes are on the same side as the cancer. It has not spread to distant sites.
OR
T4, N0 or N1, M0: The cancer has 1 or more of the following features:
- A tumor of any size has grown into the space between the lungs (mediastinum), the heart, the large blood vessels near the heart (such as the aorta), the windpipe (trachea), the tube connecting the throat to the stomach (esophagus), the backbone, or the carina.
- Two or more separate tumor nodules are present in different lobes of the same lung.
It may or may not have spread to lymph nodes within the lung and/or around the area where the bronchus enters the lung (hilar lymph nodes). Any affected lymph nodes are on the same side as the cancer. It has not spread to distant sites.
Stage 3B
Two category combinations make up this stage.
Any T, N3, M0: The cancer can be of any size. It may or may not have grown into nearby structures or caused pneumonia or lung collapse. It has spread to lymph nodes near the collarbone on either side, and/or has spread to hilar or mediastinal lymph nodes on the side opposite the primary tumor. The cancer has not spread to distant sites.
OR
T4, N2, M0: The cancer has 1 or more of the following features:
- A tumor of any size has grown into the space between the lungs (mediastinum), the heart, the large blood vessels near the heart (such as the aorta), the windpipe (trachea), the tube connecting the throat to the stomach (esophagus), the backbone, or the carina.
- Two or more separate tumor nodules are present in different lobes of the same lung.
The cancer has also spread to lymph nodes around the carina (the point where the windpipe splits into the left and right bronchi) or in the space between the lungs (mediastinum). Affected lymph nodes are on the same side as the main lung tumor. It has not spread to distant sites.
Stage 4
Stage 4 lung cancer also called advanced lung cancer, which means the cancer has spread. Stage 4 is divided into stage 4A and 4B.
- Stage 4A can mean any of the following:
- there is cancer in both lungs
- the cancer is in the covering of the lung (the pleura) or the covering of the heart (pericardium)
- or there is fluid around the lungs or the heart that contains cancer cells
- Or it can mean that there is a single area of cancer that has spread outside the chest to a lymph node or to an organ such as the liver or bone.
- Stage 4B means that the cancer has spread to several areas in one or more organs.
In the TNM staging system, stage 4 is the same as one of the following:
- Any T, Any N, M1a
- Any T, Any N, M1b
- Any T, Any N, M1c
Small Cell Lung Cancer Survival Rates by Stage
Survival rates tell you what portion of people with the same type and stage of cancer are still alive a certain amount of time (usually 5 years) after they were diagnosed. These numbers can’t tell you how long you will live, but they may help give you a better understanding about how likely it is that your treatment will be successful. Some people will want to know the survival rates for their cancer type and stage, and some people won’t. If you don’t want to know, you don’t have to.
What is a 5-year survival rate?
Statistics on the outlook for a certain type and stage of cancer are often given as 5-year survival rates, but many people live longer – often much longer – than 5 years. The 5-year survival rate is the percentage of people who live at least 5 years after being diagnosed with cancer. For example, a 5-year survival rate of 50% means that an estimated 50 out of 100 people who have that cancer are still alive 5 years after being diagnosed. Keep in mind, however, that many of these people live much longer than 5 years after diagnosis.
Relative survival rates are a more accurate way to estimate the effect of cancer on survival. These rates compare people with cancer to people in the overall population. For example, if the 5-year relative survival rate for a specific type and stage of cancer is 50%, it means that people who have that cancer are, on average, about 50% as likely as people who don’t have that cancer to live for at least 5 years after being diagnosed.
But remember, survival rates are estimates – your outlook can vary based on a number of factors specific to you.
Survival rates don’t tell the whole story
Survival rates are often based on previous outcomes of large numbers of people who had the disease, but they can’t predict what will happen in any particular person’s case. There are a number of limitations to keep in mind:
- The numbers below are among the most current available. But to get 5-year survival rates, doctors have to look at people who were treated at least 5 years ago. As treatments are improving over time, people who are now being diagnosed with small cell lung cancer might have a better outlook than these statistics show.
- These statistics are based on the stage of the cancer when it was first diagnosed. They do not apply to cancers that later come back or spread, for example.
- The outlook for people with small cell lung cancer varies by the stage (extent) of the cancer – in general, the survival rates are higher for people with earlier stage cancers. But other factors can affect a person’s outlook, such as a person’s age and overall health, and how well the cancer responds to treatment. The outlook for each person is specific to his or her circumstances.
Your doctor can tell you how these numbers may apply to you, as he or she is familiar with your particular situation.
The numbers below are relative survival rates calculated from the National Cancer Institute’s SEER database, based on people who were diagnosed with small cell lung cancer between 1988 and 2001.
These survival rates are based on the TNM staging system in use at the time, which has since been modified slightly for the latest version. Because of this, the survival numbers may be slightly different for the latest staging system.
- The 5-year relative survival rate for people with stage 1 small cell lung cancer is about 31%.
- For stage 2 small cell lung cancer, the 5-year relative survival rate is about 19%.
- The 5-year relative survival rate for stage 3 small cell lung cancer is about 8%.
- Small cell lung cancer that has spread to other parts of the body is often hard to treat. Stage 4 small cell lung cancer has a relative 5-year survival rate of about 2%. Still, there are often treatment options available for people with this stage of cancer.
Remember, these survival rates are only estimates – they can’t predict what will happen to any individual person. These statistics can be confusing and may lead you to have more questions. Talk to your doctor to better understand your situation.
Small Cell Lung Cancer Treatment
If you’ve been diagnosed with small cell lung cancer, your cancer care team will discuss your treatment options with you. It’s important that you think carefully about your choices. You will want to weigh the benefits of each treatment option against the possible risks and side effects.
Depending on the stage of the cancer and other factors, the main treatment options for people with small cell lung cancer include:
- Chemotherapy
- Radiation therapy
- Surgery
Palliative treatments can also be used to help with symptoms.
Sometimes, more than one of type of treatment is used. If you have small cell lung cancer, you will probably get chemotherapy if you are healthy enough. If you have limited stage disease, radiation therapy and – rarely – surgery may be options as well.
What types of doctors treat small cell lung cancer?
You may have different types of doctors on your treatment team, depending on the stage of your cancer and your treatment options. These doctors could include:
- A medical oncologist: a doctor who treats cancer with medicines such as chemotherapy
- A pulmonologist: a doctor who specializes in medical treatment of diseases of the lungs
- A radiation oncologist: a doctor who treats cancer with radiation therapy
- A thoracic surgeon: a doctor who treats diseases in the lungs and chest with surgery
You might have many other specialists on your treatment team as well, including physician assistants, nurse practitioners, nurses, respiratory therapists, nutrition specialists, social workers, and other health professionals.
Making treatment decisions
It’s important to discuss all of your treatment options, including their goals and possible side effects, with your doctors to help make the decision that best fits your needs. It’s also very important to ask questions if there is anything you’re not sure about. See What should you ask your doctor about small cell lung cancer? for ideas.
Getting a second opinion
You may also want to get a second opinion. This can give you more information and help you feel more certain about the treatment plan you choose. If you aren’t sure where to go for a second opinion, ask your doctor for help.
Thinking about taking part in a clinical trial
Clinical trials are carefully controlled research studies that are done to get a closer look at promising new treatments or procedures. Clinical trials are one way to get state-of-the art cancer treatment. Sometimes they may be the only way to get access to newer treatments. They are also the best way for doctors to learn better methods to treat cancer. Still, they are not right for everyone.
If you would like to learn more about clinical trials that might be right for you, start by asking your doctor if your clinic or hospital conducts clinical trials.
Considering complementary and alternative methods
You may hear about complementary or alternative methods that your doctor hasn’t mentioned to treat your cancer or relieve symptoms. These methods can include vitamins, herbs, and special diets, or other methods such as acupuncture or massage, to name a few.
Complementary methods refer to treatments that are used along with your regular medical care. Alternative treatments are used instead of a doctor’s medical treatment. Although some of these methods might be helpful in relieving symptoms or helping you feel better, many have not been proven to work. Some might even be dangerous.
As you consider your options, look for “red flags” that might suggest fraud. Does the method promise to cure all or most cancers? Are you told not to have regular medical treatments? Is the treatment a “secret” that requires you to visit certain providers or travel to another country?
Be sure to talk to your cancer care team about any method you are thinking about using. They can help you learn what is known (or not known) about the method, which can help you make an informed decision.
Choosing to stop treatment or choosing no treatment at all
For some people, when treatments have been tried and are no longer controlling the cancer, it could be time to weigh the benefits and risks of continuing to try new treatments. Whether or not you continue treatment, there are still things you can do to help maintain or improve your quality of life.
Some people, especially if the cancer is advanced, might not want to be treated at all. There are many reasons you might decide not to get cancer treatment, but it’s important to talk this through with your doctors before you make this decision. Remember that even if you choose not to treat the cancer, you can still get supportive care to help with pain or other symptoms.
Help getting through treatment
Your cancer care team will be your first source of information and support, but there are other resources for help when you need it. Hospital- or clinic-based support services are an important part of your care. These might include nursing or social work services, financial aid, nutritional advice, rehab, or spiritual help.
Chemotherapy for Small Cell Lung Cancer
Chemotherapy (chemo) is treatment with anti-cancer drugs injected into a vein or taken by mouth. These drugs enter the bloodstream and go throughout the body, making this treatment useful for cancer anywhere in the body.
Chemo is typically part of the treatment for small cell lung cancer. This is because small cell lung cancer has usually already spread by the time it is found (even if the spread can’t be seen on imaging tests), so other treatments such as surgery or radiation therapy would not reach all areas of cancer.
- For people with limited stage small cell lung cancer, chemo is often given along with radiation therapy. This is known as chemoradiation.
- For people with extensive stage small cell lung cancer, chemo with or without immunotherapy. Sometimes radiation therapy is given as well.
Some patients in poor health might not be able to tolerate intense doses of chemo. But older age by itself is not a reason to not get chemo.
Drugs used to treat small cell lung cancer
Generally, small cell lung cancer is first treated with combinations of chemo drugs. The combinations used most often are:
- Cisplatin and etoposide
- Carboplatin and etoposide
If the small cell lung cancer worsens or comes back after treatment with the above combination of chemo drugs, other chemo drugs may then be given. These drugs are usually given by themselves:
- Topotecan (Hycamtin)
- Lurbinectedin (Zepzelca)
- Docetaxel (Taxotere)
- Paclitaxel (Taxol)
- Gemcitabine (Gemzar)
- Irinotecan (Camptosar)
- Temozolomide (Temodar)
- Vinorelbine (Navelbine)
Doctors give chemo in cycles. Each cycle includes the period of treatment followed by a rest period to give you time to recover from the effects of the drugs. Cycles are most often 3 or 4 weeks long, and initial treatment is typically 4 to 6 cycles. The schedule varies depending on the drugs used. For example, some drugs are given only on the first day of the chemo cycle. Others are given for a few days in a row, or once a week. Then, at the end of the cycle, the chemo schedule repeats to start the next cycle.
For advanced cancers, the initial chemo combination is often given for 4 to 6 cycles, sometimes in combination with an immunotherapy drug. Beyond this, doctors might also recommend extending treatment with a single immunotherapy drug, for people who have had a good response to their initial chemotherapy or have had no worsening of their cancer.
If the cancer progresses (gets worse) during treatment or returns after treatment is finished, other chemo drugs may be tried. The choice of drugs depends to some extent on how soon the cancer begins to grow again. The longer it takes for the cancer to return, the more likely it is to respond to further treatment.
- If cancer returns more than 6 months after treatment, it might respond again to the same chemo drugs that were given the first time.
- If the cancer comes back sooner, or if it keeps growing during treatment, further treatment with the same drugs isn’t likely to be helpful. If further chemo is given, most doctors prefer treatment with a single, different drug to help limit side effects. small cell lung cancer that progresses or comes back can be hard to treat, so taking part in a clinical trial of newer treatments might be a good option for some people.
Possible side effects of chemotherapy
Chemo drugs can cause side effects. These depend on the type and dose of drugs given and how long they are taken. Some of the more common side effects of chemo include:
- Hair loss
- Mouth sores
- Loss of appetite
- Nausea and vomiting
- Diarrhea or constipation
- Increased chance of infections (from having too few white blood cells)
- Easy bruising or bleeding (from having too few blood platelets)
- Fatigue (from having too few red blood cells)
These side effects usually go away after treatment. There are often ways to lessen these side effects. For example, drugs can help prevent or reduce nausea and vomiting.
Some drugs can have specific side effects. For example:
- Drugs such as cisplatin and carboplatin can damage nerve endings. This is called peripheral neuropathy. It can sometimes lead to symptoms (mainly in the hands and feet) such as pain, burning or tingling sensations, sensitivity to cold or heat, or weakness. In most people this goes away or gets better after treatment is stopped, but it may last a long time in some people.
- Cisplatin can also cause kidney damage. To help prevent this, doctors give lots of IV fluids before and after each dose of the drug is given.
Immunotherapy for Small Cell Lung Cancer
Immunotherapy is the use of medicines to help your immune system to recognize and destroy cancer cells more effectively. An important part of your immune system is its ability to keep itself from attacking normal cells in the body. To do this, it uses “checkpoint” proteins on immune cells, which act like switches that need to be turned on (or off) to start an immune response. Cancer cells sometimes use these checkpoints to avoid being attacked by the immune system.
Drugs that target these checkpoints called checkpoint inhibitors can be used to treat some people with small cell lung cancer (SCLC).
PD-L1 inhibitors
Atezolizumab (Tecentriq) and durvalumab (Imfinzi) target PD-L1, a checkpoint protein found on some tumor cells and immune cells. Blocking this protein can help boost the immune response against cancer cells. These drugs can be used as part of the first treatment for advanced small cell lung cancer (SCLC), along with etoposide and a platinum chemo drug (like carboplatin or cisplatin). Either drug can then be continued alone as maintenance therapy after the chemo is done.
Durvalumab can be given for treatment of early stage small cell lung cancer (SCLC), after chemo (ie. cisplatin and etoposide) with radiation has been given.
These drugs are given as an intravenous (IV) infusion, typically every 2, 3, or 4 weeks. Atezolizumab can also be given (as Tecentriq Hybreza) as an injection under the skin (subcutaneously) over several minutes, typically once every 3 weeks.
Possible side effects of checkpoint inhibitors include:
- Fatigue
- Cough
- Nausea
- Skin rash
- Loss of appetite
- Constipation
- Joint pain
- Diarrhea
Other, more serious side effects occur less often:
- Infusion reactions: Some people might have an infusion reaction while getting these drugs. This is like an allergic reaction, and can include fever, chills, flushing of the face, rash, itchy skin, feeling dizzy, wheezing, and trouble breathing. It’s important to tell your doctor or nurse right away if you have any of these symptoms while getting these drugs.
- Autoimmune reactions: These drugs remove one of the safeguards on the body’s immune system. Sometimes the immune system responds by attacking other parts of the body, which can cause serious or even life-threatening problems in the lungs, intestines, liver, hormone-making glands, kidneys, or other organs.
It’s very important to report any new side effects to someone on your cancer care team as soon as possible. If serious side effects do occur, treatment may need to be stopped and you might be given high doses of corticosteroids to suppress your immune system.
Bispecific T-cell engager (BiTE)
Tarlatamab (Imdelltra) is a type of immunotherapy know as a bispecific T-cell engager (BiTE). Once it’s injected into the body, one part of the drug attaches to immune cells called T cells, while another part attaches to the DLL3 protein on small cell lung cancer cells. This brings the two together, which helps the immune system attack the cancer cells.
Tarlatamab can be used to treat advanced (extensive-stage) small cell lung cancer that is no longer being helped by chemo that included a platinum drug such as cisplatin or carboplatin.
This drug is given as an IV infusion, typically once a week at first, and then once every 2 weeks. Because of the risk of serious side effects such as cytokine release syndrome (CRS) and nervous system problems (see below), your doctor will want to watch you closely in a healthcare setting for about 24 hours after the first 2 infusions, and for at least several hours after later treatments.
Possible side effects of tarlatamab include:
- Fever
- Feeling very tired
- Nausea
- Loss of appetite
- A bad or metallic taste in your mouth
- Muscle or bone pain
- Constipation
- Abnormal blood test results
Tarlatamab can also cause more serious side effects:
- Cytokine release syndrome (CRS): This is a serious side effect that can occur when T cells in the body release chemicals (cytokines) that ramp up the immune system. This happens most often within the first day after the infusion, and it can be life-threatening. Symptoms can include:
- Fever and chills
- Severe nausea and vomiting
- Trouble breathing
- Feeling very tired
- Fast heartbeat
- Feeling dizzy, lightheaded, or confused
- Headache
- Problems with balance and movement, such as trouble walking
- Your cancer cacre team will watch you closely for possible signs of cytokine release syndrome (CRS), especially during and after the first few infusions. Be sure to contact your cancer care team right away if you have any symptoms that might be from cytokine release syndrome (CRS).
- Nervous system problems: This drug can affect the nervous system, which could lead to serious or even life-threatening side effects that can occur days to weeks after treatment. Symptoms can include:
- Headaches
- Weakness, numbness, or tingling in the hands or feet
- Feeling dizzy or confused
- Trouble speaking or understanding things
- Memory loss
- Trouble sleeping
- Fainting
- Tremors
- Seizures
- Serious infections: Some people might get a serious infection while getting this drug. Tell your cancer care team right away if you have a fever, cough, chest pain, shortness of breath, sore throat, rash, or pain when urinating.
- Low blood cell counts: This drug might lower your blood cell counts, which can increase your risk of infections or bleeding and may make you feel tired or short of breath. Your doctor will check your blood cell counts regularly during your treatment.
- Liver problems: This drug can affect your liver, which might show up on lab tests. It might also cause symptoms such as feeling tired, loss of appetite, pain in the upper right part of your belly, dark colored urine, or yellowing of your skin or the white parts of your eyes (jaundice).
Radiation Therapy for Small Cell Lung Cancer
Radiation therapy uses high-energy rays (such as x-rays) or particles to kill cancer cells.
Depending on the stage of small cell lung cancer and other factors, radiation therapy might be used in several situations:
- In limited stage small cell lung cancer, radiation therapy can be given at the same time as chemotherapy (chemo) to treat the tumor and lymph nodes in the chest. Giving chemo and radiation together is called concurrent chemoradiation. The radiation may be started with the first or second cycle of chemo.
- Radiation can also be given after the chemo is finished. This is sometimes done for patients with extensive stage disease, or it can be used for people with limited stage disease who have trouble getting chemotherapy and radiation at the same time (as an alternative to chemoradiation).
- Small cell lung cancer often spreads to the brain. Radiation can be given to the brain to help lower the chances of problems from cancer spread there. This is called prophylactic cranial irradiation. This is most often used to treat people with limited stage small cell lung cancer, but it can also help some people with extensive stage small cell lung cancer.
- Radiation can be used to shrink tumors to relieve (palliate) symptoms of lung cancer such as pain, bleeding, trouble swallowing, cough, shortness of breath, and problems caused by spread to other organs such as the brain.
Types of radiation therapy
The type of radiation therapy most often used to treat small cell lung cancer is called external beam radiation therapy (EBRT). It delivers radiation from outside the body and focuses it on the cancer.
Before treatments start, your radiation team will take careful measurements to find the correct angles for aiming the radiation beams and the proper dose of radiation. This planning session, called simulation, usually includes getting imaging tests such as CT scans.
Treatment is much like getting an x-ray, but the radiation is more intense. The procedure itself is painless. Each treatment lasts only a few minutes, although the setup time – getting you into place for treatment – usually takes longer.
Most often, radiation as part of the initial treatment for small cell lung cancer is given once or twice daily, 5 days a week, for 3 to 7 weeks. Radiation to relieve symptoms and prophylactic cranial radiation are given for shorter periods of time, typically less than 3 weeks.
In recent years, newer EBRT techniques have been shown to help doctors treat lung cancers more accurately while lowering the radiation exposure to nearby healthy tissues. These include:
- Three-dimensional conformal radiation therapy (3D-CRT): 3D-CRT uses special computer programs to precisely map the location of the tumor(s). Radiation beams are shaped and aimed at the tumor(s) from several directions, which makes it less likely to damage normal tissues.
- Intensity modulated radiation therapy (IMRT): IMRT is an advanced form of 3D therapy. It uses a computer-driven machine that moves around the patient as it delivers radiation. Along with shaping the beams and aiming them at the tumor from several angles, the intensity (strength) of the beams can be adjusted to limit the dose reaching nearby normal tissues. This technique is used most often if tumors are near important structures such as the spinal cord. Many major cancer centers now use IMRT.
- A variation of IMRT is called volumetric modulated arc therapy (VMAT). It uses a machine that delivers radiation quickly as it rotates once around the body. Each treatment is given over just a few minutes.
- Four-dimensional conformal radiation therapy (4DCT) shows where the tumor is in relation to other structures during each part of the breathing cycle, as opposed to just giving a “snapshot” of a point in time, like a standard CT does. This technique might also be used to help show if a tumor is attached to or invading important structures in the chest, which could help doctors determine if a person might be eligible for surgery.
- Stereotactic body radiation therapy (SBRT), also known as stereotactic ablative radiotherapy (SABR), is most often used to treat early-stage SCLC when surgery isn’t an option due to a person’s health or in people who don’t want surgery. It might also be considered for tumors that have limited spread to other parts of the body, such as the brain or adrenal glands. Instead of giving a small dose of radiation each day for several weeks, SBRT uses very focused beams of high-dose radiation given in fewer (usually 1 to 5) treatments. Several beams are aimed at the tumor from different angles. To target the radiation precisely, you are put in a specially designed body frame for each treatment. This reduces the movement of the lung tumor during breathing.
- Stereotactic radiosurgery (SRS) isn’t really surgery, but a type of stereotactic radiation therapy that is given in only 1 session. It can sometimes be used instead of or along with surgery for single spots tumor.
Possible side effects of radiation therapy
If you are going to get radiation therapy, it’s important to ask your doctor beforehand about the possible side effects so that you know what to expect. Common side effects of radiation therapy can include:
- Skin changes in the area being treated, which can range from mild redness to blistering and peeling
- Hair loss (in the area where the radiation enters the body)
- Fatigue (tiredness)
- Nausea and vomiting
- Loss of appetite and weight loss
Most of these side effects go away after treatment, but some can last a long time. When chemotherapy is given with radiation, the side effects are often worse.
Radiation therapy to the chest may damage your lungs, which might cause a cough, problems breathing, and shortness of breath. These usually improve after treatment is over, although sometimes they may not go away completely.
Your esophagus, which is in the middle of your chest, may be exposed to radiation, which could cause a sore throat and trouble swallowing during or shortly after treatment. This might make it hard to eat anything other than soft foods or liquids for a while.
Radiation therapy to large areas of the brain can sometimes cause memory loss, fatigue, headaches, trouble thinking, or reduced sexual desire. Usually these symptoms are minor compared with those caused by cancer that has spread to the brain, but they can affect your quality of life.
Surgery for Small Cell Lung Cancer
Surgery is rarely used as part of the main treatment for small cell lung cancer, as the cancer has usually already spread by the time it is found.
Occasionally (in fewer than 1 out of 20 patients), the cancer is found as only a single lung tumor, with no spread to lymph nodes or other organs. Surgery may be an option for these early-stage cancers, usually followed by additional treatment such as chemotherapy and/or immunotherapy.
If your doctor thinks the lung cancer can be treated with surgery, pulmonary function tests will be done first to see if you would still have enough healthy lung tissue left after surgery. Other tests will check the function of your heart and other organs to be sure you’re healthy enough for surgery.
Because surgery isn’t helpful for more advanced stage lung cancers, your doctor will also want to make sure the cancer hasn’t already spread to the lymph nodes between the lungs. This is often done just before surgery with mediastinoscopy or with some of the other techniques. If cancer cells are in the lymph nodes, then surgery is not likely to be helpful.
Types of lung surgery
Different operations can be used to treat small cell lung cancer:
- Pneumonectomy: An entire lung is removed in this surgery. This might be needed if the tumor is close to the center of the chest.
- Lobectomy: The lungs have 5 lobes (3 in the right lung and 2 in the left). In this surgery, the entire lobe containing the tumor is removed. If it can be done, this is often the preferred type of operation for small cell lung cancer.
- Segmentectomy or wedge resection: In these operations, only the part of the lobe with the tumor is removed. This approach might be used if a person doesn’t have enough normal lung function to withstand removing the whole lobe.
- Sleeve resection: This operation may be used to treat some cancers in large airways in the lungs. If you think of the large airway with a tumor as similar to the sleeve of a shirt with a stain a few inches above the wrist, the sleeve resection would be like cutting across the sleeve (airway) above and below the stain (tumor) and then sewing the cuff back onto the shortened sleeve. A surgeon may be able to do this operation instead of a pneumonectomy to preserve more lung function.
In general, lobectomy is the preferred operation for small cell lung cancer if it can be done, because it offers a better chance of removing all of the cancer than segmentectomy or wedge resection.
With any of these operations, nearby lymph nodes are also removed to look for possible spread of the cancer. These operations require general anesthesia (where you are in a deep sleep) and are usually done through a surgical incision between the ribs in the side of the chest (called a thoracotomy).
When you wake up from surgery, you will have a tube (or tubes) coming out of your chest and attached to a special canister to allow excess fluid and air to drain out. The tube(s) will be removed once the fluid drainage and air leak stop. Most people will spend about a week in the hospital after the surgery.
Possible risks and side effects of lung surgery
Surgery for lung cancer is a major operation and can have serious side effects, which is why surgery isn’t a good idea for everyone. While all surgeries carry some risks, they depend to some degree on the extent of the surgery and a person’s health beforehand.
Possible complications during and soon after surgery can include reactions to anesthesia, excess bleeding, blood clots in the legs or lungs, wound infections, and pneumonia. While it is rare, in some cases people may not survive the surgery.
Recovering from lung cancer surgery typically takes weeks to months. When the surgery is done through a thoracotomy, the surgeon must spread the ribs to get to the lung, so the area near the incision will hurt for some time after surgery. Your activity will be limited for at least a month.
If your lungs are in good condition (other than the presence of the cancer) you can usually return to normal activities after some time if a lobe or even an entire lung has been removed.
If you also have another lung disease such as emphysema or chronic bronchitis (which are common among heavy smokers), you might become short of breath with activity after surgery.
Palliative Procedures for Small Cell Lung Cancer
Palliative, or supportive care, is aimed at relieving symptoms and improving a person’s quality of life.
People with small cell lung cancer often benefit from procedures to help with problems caused by the cancer. For example, people with advanced lung cancer can be short of breath. This can be caused by many things, including fluid around the lung or an airway that is blocked by a tumor. Although treating the cancer with chemotherapy or other drugs may help with this over time, other treatments may be needed as well.
Treating an airway blocked by a tumor
Tumors can sometimes grow into the lung airways, blocking them and causing problems such as pneumonia or shortness of breath. Sometimes this is treated with radiation therapy, but other techniques can also be used.
Photodynamic therapy (PDT)
Photodynamic therapy is sometimes used to help open up airways blocked by tumors to help people breathe better.
For this technique, a light-activated drug called porfimer sodium (Photofrin) is injected into a vein. This drug collects more in cancer cells than in normal cells. After a couple of days (to give the drug time to build up in the cancer cells), a bronchoscope is passed down the throat and into the lung. This can be done with either local anesthesia (numbing the throat) and sedation, or with general anesthesia (which puts you in a deep sleep). A special laser light on the end of the bronchoscope is aimed at the tumor, which activates the drug and kills the cells. The dead cells are then removed a few days later during a bronchoscopy. This process can be repeated if needed.
Photodynamic therapy can cause swelling in the airway for a few days, which may lead to some shortness of breath, as well as coughing up blood or thick mucus. Some of this drug also collects in normal cells in the body, such as skin and eye cells. This can make you very sensitive to sunlight or strong indoor lights. Too much exposure can cause serious skin reactions (like a severe sunburn), so doctors recommend staying out of any strong light for several weeks after the injection.
Laser therapy
Lasers can sometimes be used to help open up airways blocked by tumors to help people breathe better.
You are usually asleep (under general anesthesia) for this type of treatment. The laser is on the end of a bronchoscope, which is passed down the throat and next to the tumor. The doctor then aims the laser beam at the tumor to burn it away. This treatment can usually be repeated, if needed.
Stent placement
If a lung tumor has grown into an airway and is causing problems, sometimes a bronchoscope is used to put a hard silicone or metal tube called a stent in the airway to help keep it open. This is often done after other treatments such as photodynamic therapy or laser therapy.
Treating fluid buildup in the area around the lung
Sometimes fluid can build up in the chest outside of the lungs. This is called a pleural effusion. It can press on the lungs and cause trouble breathing.
Thoracentesis
This is done to drain the fluid. For this procedure, the doctor will numb an area in the chest, and then place a hollow needle into the space between the lungs and the ribs to drain the fluid. This is often done using ultrasound to guide the needle into the fluid.
Pleurodesis
This procedure might be done to remove the fluid and keep it from coming back.
One way to do this is to make a small cut in the skin of chest wall, and place a hollow tube (called a chest tube) into the chest to remove the fluid. Then a substance is instilled into the chest through the tube that causes the linings of the lung (visceral pleura) and chest wall (parietal pleura) to stick together, sealing the space and limiting further fluid buildup. A number of substances can be used for this, such as talc, the antibiotic doxycycline, or a chemotherapy drug like bleomycin. The tube is often left in for a couple of days to drain any new fluid that might collect.
Another way to do this is to blow talc into the space around the lungs during an operation. This is done through a small incision using thoracoscopy
Catheter placement
This is another way to control the buildup of fluid. One end of the catheter (a thin, flexible tube) is placed in the chest through a small cut in the skin, and the other end is left outside the body. This is done in a doctor’s office or hospital. Once in place, the catheter can be attached to a special bottle or other device to allow the fluid to drain out on a regular basis.
Treating fluid buildup around the heart
Lung cancer can sometimes spread to the area around the heart. This can lead to fluid buildup inside the sac around the heart (called a pericardial effusion), which can press on the heart and affect how well it works.
Pericardiocentesis
In this procedure, the fluid is drained with a needle placed into the space around the heart. This is usually done using an echocardiogram (an ultrasound of the heart) to guide the needle.
Creating a pericardial window
This procedure can be done to keep the fluid from building up again. During surgery, a piece of the sac around the heart (the pericardium) is removed to allow the fluid to drain into the chest or belly.
Small cell lung cancer prognosis
The extent of disease and the stage at presentation is the most important prognostic factor for small cell lung cancer (SCLC). Patients with the localized-stage disease have a five-year survival rate of 29%, whereas patients with the extensive-stage disease have a five-year survival rate of 3% 36. In patients with extensive-stage disease, the median survival is 6 to 12 months with currently available therapy, but long-term disease-free survival is rare. Similar to non small cell lung cancer, performance status and weight loss are independent prognostic factors for small cell lung cancer, where patients with poor performance status and/or weight loss at the time of diagnosis have a decreased survival rate.
Prophylactic cranial radiation prevents central nervous system recurrence and can improve survival in patients with good performance status who have had a complete response or a very good partial response to chemoradiation in limited-stage disease or chemotherapy in extensive-stage disease 160, 161.
Thoracic radiation may also improve long-term outcomes for these patients 162
All patients with small cell lung cancer (SCLC) may appropriately be considered for inclusion in clinical trials at the time of diagnosis.
Regardless of stage, the prognosis for patients with small cell lung cancer is poor despite improvements in diagnosis and therapy during the past 25 years 110. Without treatment, small cell lung cancer has the most aggressive clinical course of any type of lung tumor, with a median survival from diagnosis of only 2 to 4 months. About 10% of people with small cell lung cancer remain free of disease during the 2 years from the start of therapy, which is the time period during which most relapses occur. However, even these patients are at risk of dying of lung cancer (both small and non-small cell types) 163. The overall survival rate at 5 years is 5% to 10% 163, 164, 165, 166.
An important prognostic factor for small cell lung cancer is the extent of disease. Patients with limited-stage disease have a better prognosis than patients with extensive-stage disease. For patients with limited-stage disease, the median survival is 16 to 24 months and the 5-year survival rates is 14% with current forms of treatment 166, 164, 167, 131. Patients diagnosed with limited-stage disease who smoke should be encouraged to stop smoking before undergoing combined-modality therapy because continued smoking may compromise survival 168.
Patients with limited-stage disease have improved long-term survival with combined-modality therapy 131, 169. Although long-term survivors have been reported among patients who received either surgery or chemotherapy alone, chemotherapy combined with thoracic radiation therapy is considered the standard of care 170. Adding thoracic radiation therapy increases absolute survival by approximately 5% over chemotherapy alone 169, 171. Multiple trials and meta-analyses have evaluated the optimal timing of thoracic radiation therapy relative to chemotherapy, with the weight of evidence suggesting a small benefit to early thoracic radiation therapy 166, 172, 173.
- Key Statistics for Lung Cancer. https://www.cancer.org/cancer/types/lung-cancer/about/key-statistics.html[↩][↩]
- American Cancer Society: Cancer Facts and Figures 2017. Atlanta, Ga: American Cancer Society, 2017. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2017/cancer-facts-and-figures-2017.pdf[↩]
- Key Statistics for Lung Cancer. American Cancer Society. https://www.cancer.org/cancer/non-small-cell-lung-cancer/about/key-statistics.html[↩][↩][↩][↩][↩][↩][↩]
- Humphrey L, Deffebach M, Pappas M, Baumann C, Artis K, Priest Mitchell JP, et al. Screening for Lung Cancer: Systematic Review to Update the U.S. Preventive Services Task Force Recommendation Statement. Evidence Synthesis No. 105. AHRQ Publication No. 13-05196-EF-1. Rockville, MD: Agency for Healthcare Research and Quality; 2013.[↩][↩]
- What Are Lung Carcinoid Tumors? https://www.cancer.org/cancer/lung-carcinoid-tumor/about/what-is-lung-carcinoid-tumor.html[↩][↩]
- Garg P, Singhal S, Kulkarni P, Horne D, Malhotra J, Salgia R, Singhal SS. Advances in Non-Small Cell Lung Cancer: Current Insights and Future Directions. J Clin Med. 2024 Jul 18;13(14):4189. doi: 10.3390/jcm13144189[↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Alberg AJ, Brock MV, Ford JG, et al. Epidemiology of lung cancer: diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013;143(5 suppl):e1S-e29S.[↩][↩]
- Lung and Bronchus Cancer — Cancer Stat Facts. https://seer.cancer.gov/statfacts/html/lungb.html[↩][↩][↩][↩][↩][↩][↩]
- Ji X., Chen J., Ye J., Xu S., Lin B., Hou K. Epidemiological analysis of global and regional lung cancer mortality: Based on 30-year data analysis of global burden disease database. Healthcare. 2023;11:2920. doi: 10.3390/healthcare11222920[↩][↩]
- Lung Cancer. Medline Plus. https://medlineplus.gov/lungcancer.html[↩]
- Lung Cancer. American Cancer Society. https://www.cancer.org/cancer/lung-cancer.html[↩]
- Johnson DB, Powers MA, Wu S, Huang YT. Follow-up recommendations for chest CT scan reports of incidental pulmonary nodules. Chest. 2012 Jan;141(1):280-281. doi: 10.1378/chest.11-2157[↩]
- Omenn GS, Goodman GE, Thornquist MD, Balmes J, Cullen MR, Glass A, Keogh JP, Meyskens FL, Valanis B, Williams JH, Barnhart S, Hammar S. Effects of a combination of beta carotene and vitamin A on lung cancer and cardiovascular disease. N Engl J Med. 1996 May 2;334(18):1150-5. doi: 10.1056/NEJM199605023341802[↩][↩]
- Goodman GE, Thornquist MD, Balmes J, Cullen MR, Meyskens FL Jr, Omenn GS, Valanis B, Williams JH Jr. The Beta-Carotene and Retinol Efficacy Trial: incidence of lung cancer and cardiovascular disease mortality during 6-year follow-up after stopping beta-carotene and retinol supplements. J Natl Cancer Inst. 2004 Dec 1;96(23):1743-50. doi: 10.1093/jnci/djh320[↩][↩]
- Tardon, A., Lee, W. J., Delgado-Rodriguez, M., Dosemeci, M., Albanes, D., Hoover, R., & Blair, A. (2005). Leisure-time physical activity and lung cancer: a meta-analysis. Cancer causes & control : CCC, 16(4), 389–397. https://doi.org/10.1007/s10552-004-5026-9[↩]
- Lee IM, Sesso HD, Paffenbarger RS Jr. Physical activity and risk of lung cancer. Int J Epidemiol. 1999 Aug;28(4):620-5. doi: 10.1093/ije/28.4.620[↩]
- Mao Y, Pan S, Wen SW, Johnson KC; Canadian Cancer Registries Epidemiology Research Group. Physical activity and the risk of lung cancer in Canada. Am J Epidemiol. 2003 Sep 15;158(6):564-75. doi: 10.1093/aje/kwg186[↩]
- Wiseman M. The second World Cancer Research Fund/American Institute for Cancer Research expert report. Food, nutrition, physical activity, and the prevention of cancer: a global perspective. Proc Nutr Soc. 2008 Aug;67(3):253-6. doi: 10.1017/S002966510800712X[↩]
- Henschke CI, McCauley DI, Yankelevitz DF, Naidich DP, McGuinness G, Miettinen OS, Libby DM, Pasmantier MW, Koizumi J, Altorki NK, Smith JP. Early Lung Cancer Action Project: overall design and findings from baseline screening. Lancet. 1999 Jul 10;354(9173):99-105. doi: 10.1016/S0140-6736(99)06093-6[↩]
- What’s New in Non-Small Cell Lung Cancer Research ? American Cancer Society. https://www.cancer.org/cancer/non-small-cell-lung-cancer/about/new-research.html[↩]
- The U.S. Preventive Services Task Force. https://www.uspreventiveservicestaskforce.org/[↩]
- Lung Cancer: Screening. Final Recommendation Statement March 09, 2021. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/lung-cancer-screening[↩][↩]
- Can Non-Small Cell Lung Cancer Be Found Early ? American Cancer Society. https://www.cancer.org/cancer/non-small-cell-lung-cancer/detection-diagnosis-staging/detection.html[↩][↩]
- Can Lung Cancer Be Found Early? https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/detection.html[↩]
- PDQ Adult Treatment Editorial Board. Non-Small Cell Lung Cancer Treatment (PDQ®): Patient Version. 2021 Aug 27. In: PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute (US); 2002-. [Figure, Stage IA lung cancer. The…] Available from: https://www.ncbi.nlm.nih.gov/books/NBK65917/figure/CDR0000062956__534[↩]
- PDQ Adult Treatment Editorial Board. Non-Small Cell Lung Cancer Treatment (PDQ®): Patient Version. 2021 Aug 27. In: PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute (US); 2002-. [Figure, Stage IB lung cancer. The…] Available from: https://www.ncbi.nlm.nih.gov/books/NBK65917/figure/CDR0000062956__535[↩]
- PDQ Adult Treatment Editorial Board. Non-Small Cell Lung Cancer Treatment (PDQ®): Patient Version. 2021 Aug 27. In: PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute (US); 2002-. [Figure, Stage IIA lung cancer. The…] Available from: https://www.ncbi.nlm.nih.gov/books/NBK65917/figure/CDR0000062956__536[↩]
- PDQ Adult Treatment Editorial Board. Non-Small Cell Lung Cancer Treatment (PDQ®): Patient Version. 2021 Aug 27. In: PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute (US); 2002-. [Figure, Stage IIB lung cancer (1)…] Available from: https://www.ncbi.nlm.nih.gov/books/NBK65917/figure/CDR0000062956__537[↩]
- PDQ Adult Treatment Editorial Board. Non-Small Cell Lung Cancer Treatment (PDQ®): Patient Version. 2021 Aug 27. In: PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute (US); 2002-. [Figure, Stage IIIA lung cancer (1)…] Available from: https://www.ncbi.nlm.nih.gov/books/NBK65917/figure/CDR0000062956__539[↩]
- PDQ Adult Treatment Editorial Board. Non-Small Cell Lung Cancer Treatment (PDQ®): Patient Version. 2021 Aug 27. In: PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute (US); 2002-. [Figure, Stage IIIB lung cancer (1)…] Available from: https://www.ncbi.nlm.nih.gov/books/NBK65917/figure/CDR0000062956__542[↩]
- PDQ Adult Treatment Editorial Board. Non-Small Cell Lung Cancer Treatment (PDQ®): Patient Version. 2021 Aug 27. In: PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute (US); 2002-. [Figure, Stage IVA lung cancer. The…] Available from: https://www.ncbi.nlm.nih.gov/books/NBK65917/figure/CDR0000062956__545[↩]
- PDQ Adult Treatment Editorial Board. Non-Small Cell Lung Cancer Treatment (PDQ®): Patient Version. 2021 Aug 27. In: PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute (US); 2002-. [Figure, Stage IVB lung cancer. The…] Available from: https://www.ncbi.nlm.nih.gov/books/NBK65917/figure/CDR0000062956__546[↩]
- Hoang T, Xu R, Schiller JH, Bonomi P, Johnson DH. Clinical model to predict survival in chemonaive patients with advanced non-small-cell lung cancer treated with third-generation chemotherapy regimens based on eastern cooperative oncology group data. J Clin Oncol. 2005 Jan 1;23(1):175-83. doi: 10.1200/JCO.2005.04.177[↩]
- D’Angelo SP, Janjigian YY, Ahye N, Riely GJ, Chaft JE, Sima CS, Shen R, Zheng J, Dycoco J, Kris MG, Zakowski MF, Ladanyi M, Rusch V, Azzoli CG. Distinct clinical course of EGFR-mutant resected lung cancers: results of testing of 1118 surgical specimens and effects of adjuvant gefitinib and erlotinib. J Thorac Oncol. 2012 Dec;7(12):1815-1822. doi: 10.1097/JTO.0b013e31826bb7b2[↩]
- Lung Cancer Survival Rates. https://www.cancer.org/cancer/lung-cancer/detection-diagnosis-staging/survival-rates.html[↩][↩]
- Sculier JP, Chansky K, Crowley JJ, Van Meerbeeck J, Goldstraw P; International Staging Committee and Participating Institutions. The impact of additional prognostic factors on survival and their relationship with the anatomical extent of disease expressed by the 6th Edition of the TNM Classification of Malignant Tumors and the proposals for the 7th Edition. J Thorac Oncol. 2008 May;3(5):457-66. doi: 10.1097/JTO.0b013e31816de2b8[↩][↩]
- Non-Small Cell Lung Cancer Treatment (PDQ®)–Health Professional Version. https://www.cancer.gov/types/lung/hp/non-small-cell-lung-treatment-pdq[↩][↩][↩]
- Non-Small Cell Lung Cancer. American Cancer Society. https://www.cancer.org/cancer/non-small-cell-lung-cancer.html[↩]
- Molina J.R., Yang P., Cassivi S.D., Schild S.E., Adjei A.A. Non-small cell lung cancer: Epidemiology, risk factors, treatment, and survivorship. Mayo Clin. Proc. 2008;83:584–594. doi: 10.1016/S0025-6196(11)60735-0[↩]
- Dela Cruz C.S., Tanoue L.T., Matthay R.A. Lung cancer: Epidemiology, etiology, and prevention. Clin. Chest Med. 2011;32:605–644. doi: 10.1016/j.ccm.2011.09.001[↩]
- Chen Z., Fillmore C.M., Hammerman P.S., Kim C.F., Wong K.K. Non-small-cell lung cancers: A heterogeneous set of diseases. Nat. Rev. Cancer. 2014;14:535–546. doi: 10.1038/nrc3775[↩]
- PDQ Adult Treatment Editorial Board. Non-Small Cell Lung Cancer Treatment (PDQ®): Health Professional Version. 2022 Mar 17. In: PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute (US); 2002-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK65865[↩]
- Flor L.S., Anderson J.A., Ahmad N., Aravkin A., Carr S., Dai X., Gil G.F., Hay S.I., Malloy M.J., McLaughlin S.A., et al. Health effects associated with exposure to secondhand smoke: A burden of proof study. Nat. Med. 2024;30:149–167. doi: 10.1038/s41591-023-02743-4[↩]
- Non-Small Cell Lung Cancer Treatment. National Cancer Institute. https://www.cancer.gov/types/lung/hp/non-small-cell-lung-treatment-pdq[↩]
- Shankar A., Dubey A., Saini D., Singh M., Prasad C.P., Roy S., Bharati S.J., Rinki M., Singh N., Seth T., et al. Environmental and occupational determinants of lung cancer. Transl. Lung Cancer Res. 2019;8((Suppl. S1[↩]
- Thangavel P., Park D., Lee Y.C. Recent insights into particulate matter (PM2.5)-mediated toxicity in humans: An overview. Int. J. Environ. Res. Public Health. 2022;19:7511. doi: 10.3390/ijerph19127511[↩]
- Lee J.Y., Bhandare R.R., Boddu S.H.S., Shaik A.B., Saktivel L.P., Gupta G., Negi P., Barakat M., Singh S.K., Dua K., et al. Molecular mechanisms underlying the regulation of tumor suppressor genes in lung cancer. Biomed. Pharmacother. 2024;173:116275. doi: 10.1016/j.biopha.2024.116275[↩]
- Grodzka A., Knopik-Skrocka A., Kowalska K., Kurzawa P., Krzyzaniak M., Stencel K., Bryl M. Molecular alterations of driver genes in non-small cell lung cancer: From diagnostics to targeted therapy. EXCLI J. 2023;22:415–432. doi: 10.17179/excli2023-6122[↩]
- Min H.Y., Lee H.Y. Molecular targeted therapy for anticancer treatment. Exp. Mol. Med. 2022;54:1670–1694. doi: 10.1038/s12276-022-00864-3[↩]
- Jung CY. Biopsy and mutation detection strategies in non-small cell lung cancer. Tuberc Respir Dis (Seoul). 2013 Nov;75(5):181-7. doi: 10.4046/trd.2013.75.5.181[↩]
- Biswas B., Talwar D., Meshram P., Julka P.K., Mehta A., Somashekhar S.P., Chilukuri S., Bansal A. Navigating patient journey in early diagnosis of lung cancer in India. Lung India. 2023;40:48–58. doi: 10.4103/lungindia.lungindia_144_22[↩]
- Araghi M., Mannani R., Heidarnejad Maleki A., Hamidi A., Rostami S., Safa S.H., Faramarzi F., Khorasani S., Alimohammadi M., Tahmasebi S., et al. Recent advances in non-small cell lung cancer targeted therapy; an update review. Cancer Cell Int. 2023;23:162. doi: 10.1186/s12935-023-02990-y[↩]
- Mitchell C.L., Zhang A.L., Bruno D.S., Almeida F.A. NSCLC in the Era of Targeted and Immunotherapy: What Every Pulmonologist Must Know. Diagnostics. 2023;13:1117. doi: 10.3390/diagnostics13061117[↩]
- Rivera-Concepcion J., Uprety D., Adjei A.A. Challenges in the use of targeted therapies in non-small cell lung cancer. Cancer Res. Treat. 2022;54:315–329. doi: 10.4143/crt.2022.078[↩]
- Ashrafi A., Akter Z., Modareszadeh P., Modareszadeh P., Berisha E., Alemi P.S., Chacon Castro M.d.C., Deese A.R., Zhang L. Current landscape of therapeutic resistance in lung cancer and promising strategies to overcome resistance. Cancers. 2022;14:4562. doi: 10.3390/cancers14194562[↩]
- Non-Small Cell Lung Cancer (NSCLC). https://emedicine.medscape.com/article/279960-overview[↩]
- Larsen J.E., Minna J.D. Molecular biology of lung cancer: Clinical implications. Clin. Chest Med. 2011;32:703–740. doi: 10.1016/j.ccm.2011.08.003[↩]
- Xu J., Tian L., Qi W., Lv Q., Wang T. Advancements in NSCLC: From pathophysiological insights to targeted treatments. Am. J. Clin. Oncol. 2024;47:291–303. doi: 10.1097/COC.0000000000001088[↩]
- Alberg AJ, Samet JM. Epidemiology of lung cancer. Chest. 2003 Jan;123(1 Suppl):21S-49S. doi: 10.1378/chest.123.1_suppl.21s[↩]
- Huang YJ, Huang TW, Lin FH, Chung CH, Tsao CH, Chien WC. Radiation Therapy for Invasive Breast Cancer Increases the Risk of Second Primary Lung Cancer: A Nationwide Population-Based Cohort Analysis. J Thorac Oncol. 2017 May;12(5):782-790. doi: 10.1016/j.jtho.2017.01.021[↩]
- Lorigan P, Radford J, Howell A, Thatcher N. Lung cancer after treatment for Hodgkin’s lymphoma: a systematic review. Lancet Oncol. 2005 Oct;6(10):773-9. doi: 10.1016/S1470-2045(05)70387-9[↩]
- Hubbard R, Venn A, Lewis S, Britton J. Lung cancer and cryptogenic fibrosing alveolitis. A population-based cohort study. Am J Respir Crit Care Med. 2000 Jan;161(1):5-8. doi: 10.1164/ajrccm.161.1.9906062[↩]
- Grulich AE, van Leeuwen MT, Falster MO, Vajdic CM. Incidence of cancers in people with HIV/AIDS compared with immunosuppressed transplant recipients: a meta-analysis. Lancet. 2007 Jul 7;370(9581):59-67. doi: 10.1016/S0140-6736(07)61050-2[↩]
- Johnson BE: Second lung cancers in patients after treatment for an initial lung cancer. J Natl Cancer Inst 90 (18): 1335-45, 1998. https://www.ncbi.nlm.nih.gov/pubmed/9747865[↩]
- Alberg AJ, Ford JG, Samet JM, et al.: Epidemiology of lung cancer: ACCP evidence-based clinical practice guidelines (2nd edition). Chest 132 (3 Suppl): 29S-55S, 2007. https://www.ncbi.nlm.nih.gov/pubmed/17873159[↩]
- Tulunay OE, Hecht SS, Carmella SG, et al.: Urinary metabolites of a tobacco-specific lung carcinogen in nonsmoking hospitality workers. Cancer Epidemiol Biomarkers Prev 14 (5): 1283-6, 2005. http://cebp.aacrjournals.org/content/14/5/1283.long[↩]
- Anderson KE, Kliris J, Murphy L, et al.: Metabolites of a tobacco-specific lung carcinogen in nonsmoking casino patrons. Cancer Epidemiol Biomarkers Prev 12 (12): 1544-6, 2003. http://cebp.aacrjournals.org/content/12/12/1544.long[↩]
- Straif K, Benbrahim-Tallaa L, Baan R, et al.: A review of human carcinogens–part C: metals, arsenic, dusts, and fibres. Lancet Oncol 10 (5): 453-4, 2009. https://www.ncbi.nlm.nih.gov/pubmed/19418618[↩]
- Friedman DL, Whitton J, Leisenring W, et al.: Subsequent neoplasms in 5-year survivors of childhood cancer: the Childhood Cancer Survivor Study. J Natl Cancer Inst 102 (14): 1083-95, 2010. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2907408/[↩]
- Gray A, Read S, McGale P, et al.: Lung cancer deaths from indoor radon and the cost effectiveness and potential of policies to reduce them. BMJ 338: a3110, 2009. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2769068/[↩]
- Berrington de González A, Kim KP, Berg CD: Low-dose lung computed tomography screening before age 55: estimates of the mortality reduction required to outweigh the radiation-induced cancer risk. J Med Screen 15 (3): 153-8, 2008. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2782431/[↩]
- Shimizu Y, Kato H, Schull WJ: Studies of the mortality of A-bomb survivors. 9. Mortality, 1950-1985: Part 2. Cancer mortality based on the recently revised doses (DS86). Radiat Res 121 (2): 120-41, 1990. https://www.ncbi.nlm.nih.gov/pubmed/2305030[↩]
- Katanoda K, Sobue T, Satoh H, et al.: An association between long-term exposure to ambient air pollution and mortality from lung cancer and respiratory diseases in Japan. J Epidemiol 21 (2): 132-43, 2011. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3899505/[↩]
- Cao J, Yang C, Li J, et al.: Association between long-term exposure to outdoor air pollution and mortality in China: a cohort study. J Hazard Mater 186 (2-3): 1594-600, 2011. https://www.ncbi.nlm.nih.gov/pubmed/21194838[↩]
- Hales S, Blakely T, Woodward A: Air pollution and mortality in New Zealand: cohort study. J Epidemiol Community Health 66 (5): 468-73, 2012. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3318234/[↩]
- Lissowska J, Foretova L, Dabek J, et al.: Family history and lung cancer risk: international multicentre case-control study in Eastern and Central Europe and meta-analyses. Cancer Causes Control 21 (7): 1091-104, 2010. https://www.ncbi.nlm.nih.gov/pubmed/20306329[↩]
- Shiels MS, Cole SR, Kirk GD, et al.: A meta-analysis of the incidence of non-AIDS cancers in HIV-infected individuals. J Acquir Immune Defic Syndr 52 (5): 611-22, 2009. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2790038/[↩]
- The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. The Alpha-Tocopherol, Beta Carotene Cancer Prevention Study Group. N Engl J Med 330 (15): 1029-35, 1994. http://www.nejm.org/doi/full/10.1056/NEJM199404143301501[↩]
- Omenn GS, Goodman GE, Thornquist MD, et al.: Effects of a combination of beta carotene and vitamin A on lung cancer and cardiovascular disease. N Engl J Med 334 (18): 1150-5, 1996. http://www.nejm.org/doi/full/10.1056/NEJM199605023341802[↩][↩]
- Wingo PA, Ries LA, Giovino GA, et al.: Annual report to the nation on the status of cancer, 1973-1996, with a special section on lung cancer and tobacco smoking. J Natl Cancer Inst 91 (8): 675-90, 1999. https://www.ncbi.nlm.nih.gov/pubmed/10218505[↩]
- Suster D.I., Mino-Kenudson M. Molecular pathology of primary non-small cell lung cancer. Arch. Med. Res. 2020;51:784–798. doi: 10.1016/j.arcmed.2020.08.004[↩]
- Friedlaender A., Perol M., Banna G.L., Parikh K., Addeo A. Oncogenic alterations in advanced NSCLC: A molecular super-highway. Biomark. Res. 2024;12:24. doi: 10.1186/s40364-024-00566-0[↩]
- Friedlaender A., Banna G., Malapelle U., Pisapia P., Addeo A. Next generation sequencing and genetic alterations in squamous cell lung carcinoma: Where are we today? Front. Oncol. 2019;9:166. doi: 10.3389/fonc.2019.00166[↩]
- Bethune G, Bethune D, Ridgway N, Xu Z. Epidermal growth factor receptor (EGFR) in lung cancer: an overview and update. J Thorac Dis. 2010 Mar;2(1):48-51. https://pmc.ncbi.nlm.nih.gov/articles/PMC3256436[↩]
- Gazdar A.F. Activating and resistance mutations of EGFR in non-small-cell lung cancer: Role in clinical response to EGFR tyrosine kinase inhibitors. Oncogene. 2009;28((Suppl. S1[↩]
- Arbour K.C., Riely G.J. Diagnosis and treatment of anaplastic lymphoma kinase-positive non-small cell lung cancer. Hematol. Oncol. Clin. N. Am. 2017;31:101–111. doi: 10.1016/j.hoc.2016.08.012[↩]
- Shackelford R.E., Vora M., Mayhall K., Cotelingam J. ALK-rearrangements and testing methods in non-small cell lung cancer: A review. Genes Cancer. 2014;5:1–14. doi: 10.18632/genesandcancer.3[↩]
- Reita D., Pabst L., Pencreach E., Guerin E., Dano L., Rimelen V., Voegeli A.-C., Vallat L., Mascaux C., Beau-Faller M. Direct targeting KRAS mutation in non-small cell lung cancer: Focus on resistance. Cancers. 2022;14:1321. doi: 10.3390/cancers14051321[↩]
- Santarpia M., Ciappina G., Spagnolo C.C., Squeri A., Passalacqua M.I., Aguilar A., Gonzalez-Cao M., Giovannetti E., Silvestris N., Rosell R. Targeted therapies for KRAS-mutant non-small cell lung cancer: From preclinical studies to clinical development-a narrative review. Transl. Lung Cancer Res. 2023;12:346–368. doi: 10.21037/tlcr-22-639[↩]
- D’Angelo A., Sobhani N., Chapman R., Bagby S., Bortoletti C., Traversini M., Ferrari K., Voltolini L., Darlow J., Roviello G. Focus on ROS1-positive non-small cell lung cancer (NSCLC): Crizotinib, resistance mechanisms and the newer generation of targeted therapies. Cancers. 2020;12:3293. doi: 10.3390/cancers12113293[↩]
- Yan N., Guo S., Zhang H., Zhang Z., Shen S., Li X. BRAF-mutated non-small cell lung cancer: Current treatment status and future perspective. Front. Oncol. 2022;12:863043. doi: 10.3389/fonc.2022.863043[↩]
- Spitaleri G., Trillo Aliaga P., Attili I., Signore E.D., Corvaja C., Corti C., Uliano J., Passaro A., de Marinis F. MET in non-small-cell lung cancer (NSCLC): Cross ‘a long and winding road’ looking for a target. Cancers. 2023;15:4779. doi: 10.3390/cancers15194779[↩]
- Thomas P, Rubinstein L: Cancer recurrence after resection: T1 N0 non-small cell lung cancer. Lung Cancer Study Group. Ann Thorac Surg 49 (2): 242-6; discussion 246-7, 1990. https://www.ncbi.nlm.nih.gov/pubmed/2154958[↩]
- Martini N, Bains MS, Burt ME, et al.: Incidence of local recurrence and second primary tumors in resected stage I lung cancer. J Thorac Cardiovasc Surg 109 (1): 120-9, 1995. https://www.ncbi.nlm.nih.gov/pubmed/7815787[↩]
- van Boxem AJ, Westerga J, Venmans BJ, et al.: Photodynamic therapy, Nd-YAG laser and electrocautery for treating early-stage intraluminal cancer: which to choose? Lung Cancer 31 (1): 31-6, 2001. https://www.ncbi.nlm.nih.gov/pubmed/11162864[↩]
- Blumberg J, Block G: The Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study in Finland. Nutr Rev 52 (7): 242-5, 1994. https://www.ncbi.nlm.nih.gov/pubmed/8090376[↩]
- Lippman SM, Lee JJ, Karp DD, et al.: Randomized phase III intergroup trial of isotretinoin to prevent second primary tumors in stage I non-small-cell lung cancer. J Natl Cancer Inst 93 (8): 605-18, 2001. https://www.ncbi.nlm.nih.gov/pubmed/11309437[↩]
- van Zandwijk N, Dalesio O, Pastorino U, et al.: EUROSCAN, a randomized trial of vitamin A and N-acetylcysteine in patients with head and neck cancer or lung cancer. For the EUropean Organization for Research and Treatment of Cancer Head and Neck and Lung Cancer Cooperative Groups. J Natl Cancer Inst 92 (12): 977-86, 2000. https://www.ncbi.nlm.nih.gov/pubmed/10861309[↩]
- Non-Small Cell Lung Cancer Signs and Symptoms. American Cancer Society. https://www.cancer.org/cancer/non-small-cell-lung-cancer/detection-diagnosis-staging/signs-symptoms.html[↩][↩]
- Travis WD, Colby TV, Corrin B, et al.: Histological typing of lung and pleural tumours. 3rd ed. Berlin: Springer-Verlag, 1999.[↩]
- Pao W, Girard N: New driver mutations in non-small-cell lung cancer. Lancet Oncol 12 (2): 175-80, 2011. https://www.ncbi.nlm.nih.gov/pubmed/21277552[↩]
- Martini N, Bains MS, Burt ME, et al.: Incidence of local recurrence and second primary tumors in resected stage I lung cancer. J Thorac Cardiovasc Surg 109 (1): 120-9, 1995. http://www.jtcvsonline.org/article/S0022-5223(95)70427-2/fulltext[↩]
- Ichinose Y, Yano T, Asoh H, et al.: Prognostic factors obtained by a pathologic examination in completely resected non-small-cell lung cancer. An analysis in each pathologic stage. J Thorac Cardiovasc Surg 110 (3): 601-5, 1995. https://www.ncbi.nlm.nih.gov/pubmed/7564425[↩]
- Fontanini G, Bigini D, Vignati S, et al.: Microvessel count predicts metastatic disease and survival in non-small cell lung cancer. J Pathol 177 (1): 57-63, 1995. https://www.ncbi.nlm.nih.gov/pubmed/7472781[↩]
- Naruke T, Goya T, Tsuchiya R, et al.: Prognosis and survival in resected lung carcinoma based on the new international staging system. J Thorac Cardiovasc Surg 96 (3): 440-7, 1988. https://www.ncbi.nlm.nih.gov/pubmed/2842549[↩]
- Khan OA, Fitzgerald JJ, Field ML, et al.: Histological determinants of survival in completely resected T1-2N1M0 nonsmall cell cancer of the lung. Ann Thorac Surg 77 (4): 1173-8, 2004. https://www.ncbi.nlm.nih.gov/pubmed/15063229[↩]
- Earle CC, Tsai JS, Gelber RD, et al.: Effectiveness of chemotherapy for advanced lung cancer in the elderly: instrumental variable and propensity analysis. J Clin Oncol 19 (4): 1064-70, 2001. https://www.ncbi.nlm.nih.gov/pubmed/11181670[↩]
- Non-Small Cell Lung Cancer Stages. American Cancer Society. https://www.cancer.org/cancer/non-small-cell-lung-cancer/detection-diagnosis-staging/staging.html[↩]
- Jeremy George, P., Banerjee, A. K., Read, C. A., O’Sullivan, C., Falzon, M., Pezzella, F., Nicholson, A. G., Shaw, P., Laurent, G., & Rabbitts, P. H. (2007). Surveillance for the detection of early lung cancer in patients with bronchial dysplasia. Thorax, 62(1), 43–50. https://doi.org/10.1136/thx.2005.052191[↩]
- Small Cell Lung Cancer Treatment (PDQ®)–Health Professional Version. https://www.cancer.gov/types/lung/hp/small-cell-lung-treatment-pdq[↩][↩]
- Basumallik N, Agarwal M. Small Cell Lung Cancer. [Updated 2023 Jul 10]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK482458[↩][↩]
- Small Cell Lung Cancer (SCLC). https://emedicine.medscape.com/article/280104-overview[↩]
- Das S, Samaddar S. Recent Advances in the Clinical Translation of Small-Cell Lung Cancer Therapeutics. Cancers (Basel). 2025 Jan 14;17(2):255. doi: 10.3390/cancers17020255[↩][↩][↩][↩][↩]
- Sen T, Takahashi N, Chakraborty S, Takebe N, Nassar AH, Karim NA, Puri S, Naqash AR. Emerging advances in defining the molecular and therapeutic landscape of small-cell lung cancer. Nat Rev Clin Oncol. 2024 Aug;21(8):610-627. doi: 10.1038/s41571-024-00914-x[↩]
- Yu T, Lok BH. Strategies to Target Chemoradiotherapy Resistance in Small Cell Lung Cancer. Cancers (Basel). 2024 Oct 10;16(20):3438. doi: 10.3390/cancers16203438[↩]
- Pangua C, Rogado J, Serrano-Montero G, Belda-Sanchís J, Álvarez Rodríguez B, Torrado L, Rodríguez De Dios N, Mielgo-Rubio X, Trujillo JC, Couñago F. New perspectives in the management of small cell lung cancer. World J Clin Oncol. 2022 Jun 24;13(6):429-447. doi: 10.5306/wjco.v13.i6.429[↩]
- Couñago F, de la Pinta C, Gonzalo S, Fernández C, Almendros P, Calvo P, Taboada B, Gómez-Caamaño A, Guerra JLL, Chust M, González Ferreira JA, Álvarez González A, Casas F. GOECP/SEOR radiotherapy guidelines for small-cell lung cancer. World J Clin Oncol. 2021 Mar 24;12(3):115-143. doi: 10.5306/wjco.v12.i3.115[↩]
- Yuan M., Zhao Y., Arkenau H.-T., Lao T., Chu L., Xu Q. Signal Pathways and Precision Therapy of Small-Cell Lung Cancer. Signal. Transduct. Target Ther. 2022;7:187. doi: 10.1038/s41392-022-01013-y[↩][↩][↩]
- Wang S., Tang J., Sun T., Zheng X., Li J., Sun H., Zhou X., Zhou C., Zhang H., Cheng Z., et al. Survival Changes in Patients with Small Cell Lung Cancer and Disparities between Different Sexes, Socioeconomic Statuses and Ages. Sci. Rep. 2017;7:1339. doi: 10.1038/s41598-017-01571-0[↩]
- Yang W., Wang W., Li Z., Wu J., Xu X., Chen C., Ye X. Differences between Advanced Large Cell Neuroendocrine Carcinoma and Advanced Small Cell Lung Cancer: A Propensity Score Matching Analysis. J. Cancer. 2023;14:1541–1552. doi: 10.7150/jca.84600[↩]
- Sen T., Takahashi N., Chakraborty S., Takebe N., Nassar A.H., Karim N.A., Puri S., Naqash A.R. Emerging Advances in Defining the Molecular and Therapeutic Landscape of Small-Cell Lung Cancer. Nat. Rev. Clin. Oncol. 2024;21:610–627. doi: 10.1038/s41571-024-00914-x[↩][↩][↩]
- Rudin C.M., Brambilla E., Faivre-Finn C., Sage J. Small-Cell Lung Cancer. Nat. Rev. Dis. Prim. 2021;7:3. doi: 10.1038/s41572-020-00235-0[↩][↩][↩]
- Ying Q., Fan R., Shen Y., Chen B., Zhang J., Li Q., Shi X. Small Cell Lung Cancer—An Update on Chemotherapy Resistance. Curr. Treat. Options Oncol. 2024;25:1112–1123. doi: 10.1007/s11864-024-01245-w[↩][↩]
- Wang Q., Gümüş Z.H., Colarossi C., Memeo L., Wang X., Kong C.Y., Boffetta P. SCLC: Epidemiology, Risk Factors, Genetic Susceptibility, Molecular Pathology, Screening, and Early Detection. J. Thorac. Oncol. 2023;18:31–46. doi: 10.1016/j.jtho.2022.10.002[↩]
- Solta A., Ernhofer B., Boettiger K., Megyesfalvi Z., Heeke S., Hoda M.A., Lang C., Aigner C., Hirsch F.R., Schelch K., et al. Small Cells—Big Issues: Biological Implications and Preclinical Advancements in Small Cell Lung Cancer. Mol. Cancer. 2024;23:41. doi: 10.1186/s12943-024-01953-9[↩][↩][↩][↩][↩][↩][↩]
- Rudin C.M., Poirier J.T. Shining Light on Novel Targets and Therapies. Nat. Rev. Clin. Oncol. 2017;14:75–76. doi: 10.1038/nrclinonc.2016.203[↩]
- Davies A.M., Lara P.N., Lau D.H., Gandara D.R. Treatment of Extensive Small Cell Lung Cancer. Hematol. Oncol. Clin. N. Am. 2004;18:373–385. doi: 10.1016/j.hoc.2003.12.012[↩][↩]
- Schwendenwein A., Megyesfalvi Z., Barany N., Valko Z., Bugyik E., Lang C., Ferencz B., Paku S., Lantos A., Fillinger J., et al. Molecular Profiles of Small Cell Lung Cancer Subtypes: Therapeutic Implications. Mol. Ther. Oncolytics. 2021;20:470–483. doi: 10.1016/j.omto.2021.02.004[↩][↩]
- Small Cell Lung Cancer (SCLC). https://emedicine.medscape.com/article/280104-overview#a6[↩][↩]
- Jackman DM, Johnson BE. Small-cell lung cancer. Lancet. 2005 Oct 15-21;366(9494):1385-96. doi: 10.1016/S0140-6736(05)67569-1[↩]
- Jänne PA, Freidlin B, Saxman S, Johnson DH, Livingston RB, Shepherd FA, Johnson BE. Twenty-five years of clinical research for patients with limited-stage small cell lung carcinoma in North America. Cancer. 2002 Oct 1;95(7):1528-38. doi: 10.1002/cncr.10841[↩][↩][↩]
- Hermes A, Waschki B, Reck M. Hyponatremia as prognostic factor in small cell lung cancer–a retrospective single institution analysis. Respir Med. 2012 Jun;106(6):900-4. doi: 10.1016/j.rmed.2012.02.010[↩]
- Wu C, Xu B, Yuan P, Miao X, Liu Y, Guan Y, Yu D, Xu J, Zhang T, Shen H, Wu T, Lin D. Genome-wide interrogation identifies YAP1 variants associated with survival of small-cell lung cancer patients. Cancer Res. 2010 Dec 1;70(23):9721-9. doi: 10.1158/0008-5472.CAN-10-1493[↩]
- Xun WW, Brennan P, Tjonneland A, et al. Single-nucleotide polymorphisms (5p15.33, 15q25.1, 6p22.1, 6q27 and 7p15.3) and lung cancer survival in the European Prospective Investigation into Cancer and Nutrition (EPIC). Mutagenesis. 2011 Sep;26(5):657-66. doi: 10.1093/mutage/ger030[↩]
- Travis WD, Colby TV, Corrin B, et al.: Histological typing of lung and pleural tumours. 3rd ed. Springer-Verlag, 1999.[↩]
- Travis W.D., Brambilla E., Burke A., et al. International Agency for Research on Cancer; Lyon: 2021. WHO Classification of Tumours. Thoracic Tumours.[↩][↩]
- Dingemans A.C., Fruh M., Ardizzoni A., et al. Small-cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2021;32:839–853. doi: 10.1016/j.annonc.2021.03.207[↩]
- National Comprehensive Cancer Network Small Cell Lung Cancer (version 3.2021) https://www.nccn.org/professionals/physician_gls/pdf/sclc.pdf[↩]
- Travis W.D., Brambilla E., Nicholson A.G., Yatabe Y., Austin J.H.M., Beasley M.B., Chirieac L.R., Dacic S., Duhig E., Flieder D.B., et al. The 2015 World Health Organization Classification of Lung Tumors. J. Thorac. Oncol. 2015;10:1243–1260. doi: 10.1097/JTO.0000000000000630[↩][↩][↩]
- Krpina K., Vranić S., Tomić K., Samaržija M., Batičić L. Small Cell Lung Carcinoma: Current Diagnosis, Biomarkers, and Treatment Options with Future Perspectives. Biomedicines. 2023;11:1982. doi: 10.3390/biomedicines11071982[↩][↩][↩][↩][↩]
- Cutz E., Yeger H., Pan J. Pulmonary Neuroendocrine Cell System in Pediatric Lung Disease—Recent Advances. Pediatr. Dev. Pathol. 2007;10:419–435. doi: 10.2350/07-04-0267.1[↩]
- Linnoila R.I. Functional Facets of the Pulmonary Neuroendocrine System. Lab. Investig. 2006;86:425–444. doi: 10.1038/labinvest.3700412[↩]
- Huang Y.-H., Klingbeil O., He X.-Y., Wu X.S., Arun G., Lu B., Somerville T.D.D., Milazzo J.P., Wilkinson J.E., Demerdash O.E., et al. POU2F3 Is a Master Regulator of a Tuft Cell-like Variant of Small Cell Lung Cancer. Genes Dev. 2018;32:915–928. doi: 10.1101/gad.314815.118[↩]
- Rhodin J., Dalhamn T. Electron Microscopy of the Tracheal Ciliated Mucosa in Rat. Zellforsch. Mikrosk. Anat. 1956;44:345–412. doi: 10.1007/BF00345847[↩]
- Meuwissen R., Linn S.C., Linnoila R.I., Zevenhoven J., Mooi W.J., Berns A. Induction of Small Cell Lung Cancer by Somatic Inactivation of Both Trp53 and Rb1 in a Conditional Mouse Model. Cancer Cell. 2003;4:181–189. doi: 10.1016/S1535-6108(03)00220-4[↩]
- Maleki Z. Diagnostic Issues with Cytopathologic Interpretation of Lung Neoplasms Displaying High-grade Basaloid or Neuroendocrine Morphology. Diagn. Cytopathol. 2011;39:159–167. doi: 10.1002/dc.21351[↩]
- Nicholson S.A., Beasley M.B., Brambilla E., Hasleton P.S., Colby T.V., Sheppard M.N., Falk R., Travis W.D. Small Cell Lung Carcinoma (SCLC) Am. J. Surg. Pathol. 2002;26:1184–1197. doi: 10.1097/00000478-200209000-00009[↩]
- Kontogianni K., Nicholson A.G., Butcher D., Sheppard M.N. CD56: A Useful Tool for the Diagnosis of Small Cell Lung Carcinomas on Biopsies with Extensive Crush Artefact. J. Clin. Pathol. 2005;58:978–980. doi: 10.1136/jcp.2004.023044[↩]
- Travis W.D., Brambilla E., Burke A.P., Marx A., Nicholson A.G. Introduction to the 2015 World Health Organization Classification of Tumors of the Lung, Pleura, Thymus, and Heart. J. Thorac. Oncol. 2015;10:1240–1242. doi: 10.1097/JTO.0000000000000663[↩]
- Lei Y, Feng H, Qiang H, Shang Z, Chang Q, Qian J, Zhang Y, Zhong R, Fan X, Chu T. Clinical characteristics and prognostic factors of surgically resected combined small cell lung cancer: a retrospective study. Lung Cancer. 2020 Aug;146:244-251. doi: 10.1016/j.lungcan.2020.06.021[↩]
- Simbolo M, Centonze G, Ali G, Garzone G, Taormina S, Sabella G, Ciaparrone C, Mafficini A, Grillo F, Mangogna A, Volante M, Mastracci L, Fontanini G, Pilotto S, Bria E, Infante M, Capella C, Rolli L, Pastorino U, Milella M, Milione M, Scarpa A. Integrative molecular analysis of combined small-cell lung carcinomas identifies major subtypes with different therapeutic opportunities. ESMO Open. 2022 Feb;7(1):100308. doi: 10.1016/j.esmoop.2021.100308[↩]
- Rudin C.M., Poirier J.T., Byers L.A., Dive C., Dowlati A., George J., Heymach J.V., Johnson J.E., Lehman J.M., MacPherson D., et al. Molecular Subtypes of Small Cell Lung Cancer: A Synthesis of Human and Mouse Model Data. Nat. Rev. Cancer. 2019;19:289–297. doi: 10.1038/s41568-019-0133-9[↩]
- Baine M.K., Hsieh M.-S., Lai W.V., Egger J.V., Jungbluth A.A., Daneshbod Y., Beras A., Spencer R., Lopardo J., Bodd F., et al. SCLC Subtypes Defined by ASCL1, NEUROD1, POU2F3, and YAP1: A Comprehensive Immunohistochemical and Histopathologic Characterization. J. Thorac. Oncol. 2020;15:1823–1835. doi: 10.1016/j.jtho.2020.09.009[↩]
- Megyesfalvi Z., Gay C.M., Popper H., Pirker R., Ostoros G., Heeke S., Lang C., Hoetzenecker K., Schwendenwein A., Boettiger K., et al. Clinical Insights into Small Cell Lung Cancer: Tumor Heterogeneity, Diagnosis, Therapy, and Future Directions. CA Cancer J. Clin. 2023;73:620–652. doi: 10.3322/caac.21785[↩]
- Simpson K.L., Stoney R., Frese K.K., Simms N., Rowe W., Pearce S.P., Humphrey S., Booth L., Morgan D., Dynowski M., et al. A Biobank of Small Cell Lung Cancer CDX Models Elucidates Inter- and Intratumoral Phenotypic Heterogeneity. Nat. Cancer. 2020;1:437–451. doi: 10.1038/s43018-020-0046-2[↩]
- Puri S., Naqash A.R., Elliott A., Kerrigan K.C., Patel S.B., Seeber A., Kocher F., UPRETY D., Mamdani H., Kulkarni A., et al. Real-World Multiomic Characterization of Small Cell Lung Cancer Subtypes to Reveal Differential Expression of Clinically Relevant Biomarkers. J. Clin. Oncol. 2021;39:8508. doi: 10.1200/JCO.2021.39.15_suppl.8508[↩]
- Pesch B., Kendzia B., Gustavsson P., Jöckel K.-H., Johnen G., Pohlabeln H., Olsson A., Ahrens W., Gross I.M., Brüske I., et al. Cigarette Smoking and Lung Cancer—Relative Risk Estimates for the Major Histological Types from a Pooled Analysis of Case–Control Studies. Int. J. Cancer. 2012;131:1210–1219. doi: 10.1002/ijc.27339[↩]
- Parsons A, Daley A, Begh R, Aveyard P. Influence of smoking cessation after diagnosis of early stage lung cancer on prognosis: systematic review of observational studies with meta-analysis. BMJ. 2010 Jan 21;340:b5569. doi: 10.1136/bmj.b5569[↩]
- Stahel R.A., Ginsberg R., Havemann K., Hirsch F.R., Ihde D.C., Jassem J., Karrer K., Herbert Maurer L., Osterlind K., Van Houtte P. Staging and Prognostic Factors in Small Cell Lung Cancer: A Consensus Report. Lung Cancer. 1989;5:119–126. doi: 10.1016/0169-5002(89)90156-6[↩]
- Slotman B, Faivre-Finn C, Kramer G, Rankin E, Snee M, Hatton M, Postmus P, Collette L, Musat E, Senan S; EORTC Radiation Oncology Group and Lung Cancer Group. Prophylactic cranial irradiation in extensive small-cell lung cancer. N Engl J Med. 2007 Aug 16;357(7):664-72. doi: 10.1056/NEJMoa071780[↩]
- Aupérin A, Arriagada R, Pignon JP, Le Péchoux C, Gregor A, Stephens RJ, Kristjansen PE, Johnson BE, Ueoka H, Wagner H, Aisner J. Prophylactic cranial irradiation for patients with small-cell lung cancer in complete remission. Prophylactic Cranial Irradiation Overview Collaborative Group. N Engl J Med. 1999 Aug 12;341(7):476-84. doi: 10.1056/NEJM199908123410703[↩]
- Slotman BJ, van Tinteren H, Praag JO, Knegjens JL, El Sharouni SY, Hatton M, Keijser A, Faivre-Finn C, Senan S. Use of thoracic radiotherapy for extensive stage small-cell lung cancer: a phase 3 randomised controlled trial. Lancet. 2015 Jan 3;385(9962):36-42. doi: 10.1016/S0140-6736(14)61085-0. Erratum in: Lancet. 2015 Jan 3;385(9962):28.[↩]
- Johnson BE, Grayson J, Makuch RW, Linnoila RI, Anderson MJ, Cohen MH, Glatstein E, Minna JD, Ihde DC. Ten-year survival of patients with small-cell lung cancer treated with combination chemotherapy with or without irradiation. J Clin Oncol. 1990 Mar;8(3):396-401. doi: 10.1200/JCO.1990.8.3.396[↩][↩]
- Fry WA, Menck HR, Winchester DP. The National Cancer Data Base report on lung cancer. Cancer. 1996 May 1;77(9):1947-55. doi: 10.1002/(SICI)1097-0142(19960501)77:9<1947::AID-CNCR27>3.0.CO;2-Z[↩][↩]
- Lassen U, Osterlind K, Hansen M, Dombernowsky P, Bergman B, Hansen HH. Long-term survival in small-cell lung cancer: posttreatment characteristics in patients surviving 5 to 18+ years–an analysis of 1,714 consecutive patients. J Clin Oncol. 1995 May;13(5):1215-20. doi: 10.1200/JCO.1995.13.5.1215[↩]
- Murray N, Coy P, Pater JL, Hodson I, Arnold A, Zee BC, Payne D, Kostashuk EC, Evans WK, Dixon P, et al. Importance of timing for thoracic irradiation in the combined modality treatment of limited-stage small-cell lung cancer. The National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol. 1993 Feb;11(2):336-44. doi: 10.1200/JCO.1993.11.2.336[↩][↩][↩]
- Turrisi AT 3rd, Kim K, Blum R, Sause WT, Livingston RB, Komaki R, Wagner H, Aisner S, Johnson DH. Twice-daily compared with once-daily thoracic radiotherapy in limited small-cell lung cancer treated concurrently with cisplatin and etoposide. N Engl J Med. 1999 Jan 28;340(4):265-71. doi: 10.1056/NEJM199901283400403[↩]
- Videtic GM, Stitt LW, Dar AR, Kocha WI, Tomiak AT, Truong PT, Vincent MD, Yu EW. Continued cigarette smoking by patients receiving concurrent chemoradiotherapy for limited-stage small-cell lung cancer is associated with decreased survival. J Clin Oncol. 2003 Apr 15;21(8):1544-9. doi: 10.1200/JCO.2003.10.089[↩]
- Pignon JP, Arriagada R, Ihde DC, Johnson DH, Perry MC, Souhami RL, Brodin O, Joss RA, Kies MS, Lebeau B, et al. A meta-analysis of thoracic radiotherapy for small-cell lung cancer. N Engl J Med. 1992 Dec 3;327(23):1618-24. doi: 10.1056/NEJM199212033272302[↩][↩]
- Chandra V, Allen MS, Nichols FC 3rd, Deschamps C, Cassivi SD, Pairolero PC. The role of pulmonary resection in small cell lung cancer. Mayo Clin Proc. 2006 May;81(5):619-24. doi: 10.4065/81.5.619[↩]
- Warde P, Payne D. Does thoracic irradiation improve survival and local control in limited-stage small-cell carcinoma of the lung? A meta-analysis. J Clin Oncol. 1992 Jun;10(6):890-5. doi: 10.1200/JCO.1992.10.6.890[↩]
- Perry MC, Eaton WL, Propert KJ, Ware JH, Zimmer B, Chahinian AP, Skarin A, Carey RW, Kreisman H, Faulkner C, et al. Chemotherapy with or without radiation therapy in limited small-cell carcinoma of the lung. N Engl J Med. 1987 Apr 9;316(15):912-8. doi: 10.1056/NEJM198704093161504[↩]
- Takada M, Fukuoka M, Kawahara M, Sugiura T, Yokoyama A, Yokota S, Nishiwaki Y, Watanabe K, Noda K, Tamura T, Fukuda H, Saijo N. Phase III study of concurrent versus sequential thoracic radiotherapy in combination with cisplatin and etoposide for limited-stage small-cell lung cancer: results of the Japan Clinical Oncology Group Study 9104. J Clin Oncol. 2002 Jul 15;20(14):3054-60. doi: 10.1200/JCO.2002.12.071[↩]