Contents
Mycophenolate mofetil
Mycophenolate mofetil also known as CellCept, MMF, Myfortic or mycophenolate is an immunosupressant or a drug that weakens your body’s immune system in order to help keep it from “rejecting” a transplanted organ also known as graft-versus-host disease (GVHD) that is used along with other medications to prevent organ rejection in adults and children at least 3 months of age and older who have received a kidney, heart, or liver transplant 1. Mycophenolate mofetil is also being studied in the prevention of graft-versus-host disease (GVHD) after stem cell transplants for cancer, and in the treatment of some autoimmune disorders. Mycophenolate mofetil should be prescribed only by physicians with experience in immunosuppression and management of its complications.
Mycophenolate mofetil was approved for use in the United States in 1995 and its current indications are for prevention of organ rejection after transplantation. It is also used off label in therapy of active and refractory forms of autoimmune diseases.
Mycophenolate mofetil comes as capsules of 250 mg and tablets of 500 mg and a oral suspension (200 mg/mL) called CellCept and as mycophenolic acid in 180 and 360 mg delayed-release tablets (releases the medication in your intestine) called Myfortic to be taken by mouth. Mycophenolate mofetil is also available as a sterile powder in a vial for injection, which contains the equivalent of 500 mg mycophenolate mofetil as the hydrochloride salt for intravenous administration.
Mycophenolate mofetil is usually taken twice a day on an empty stomach (1 hour before or 2 hours after eating or drinking), unless your doctor tells you otherwise. Take mycophenolate mofetil at about the same times every day, and try to space your doses about 12 hours apart. Follow the directions on your prescription label carefully, and ask your doctor or pharmacist to explain any part you do not understand. Take mycophenolate mofetil exactly as directed. Do not take more or less of it or take it more often than prescribed by your doctor.
In adults, the usual dose of mycophenolate mofetil ranges from 1.25 to 2 g day. The typical maintenance dose is 1 to 1.5 grams twice daily. In children, it is administered as a 600 mg/m² dose every 12 hours up to a maximum daily dose of 2 g. Dose reduction should be considered in patients with severe kidney impairment 2, 3.
Enteric-coated mycophenolate sodium is available as delayed-release tablets containing either 180 or 360 mg of Myfortic (mycophenolic acid). It can be used as an alternative in the patients with adverse mycophenolate mofetil-induced gastrointestinal effects 4.
Mycophenolate mofetil in the delayed-release tablet (Myfortic) is absorbed differently by your body than Mycophenolate mofetil in the oral suspension, tablet, and capsule (CellCept). These products cannot be substituted for each other. Each time you have your prescription filled, make sure that you have received the right product. If you think you received the wrong medication, talk to your doctor and pharmacist right away.
Swallow the tablets, delayed-release tablets, and capsules whole; do not split, chew, or crush them. Do not open the capsules. If a capsule is accidentally opened, avoid contact with your skin or eyes and avoid breathing in the powder. If any contact occurs, wash the affected area well with soap and water; rinse eyes with water.
Do not mix mycophenolate mofetil suspension with any other medication.
Be careful not to spill the suspension or to splash it onto your skin. If you do get the suspension on your skin, wash the area well with soap and water. If you get the suspension in your eyes, rinse with plain water. Use wet paper towels to wipe up any spilled liquids.
Mycophenolate mofetil helps prevent organ transplant rejection only as long as you are taking the medication. Continue to take mycophenolate mofetil even if you feel well. Do not stop taking mycophenolate mofetil without talking to your doctor.
Mycophenolate mofetil most common dose-related side effects are gastrointestinal and genitourinary. Gastrointestinal symptoms include gastrointestinal upset, nausea, diarrhea, abdominal cramps, constipation, vomiting and anorexia while genitourinary symptoms include urgency, frequency, dysuria, hematuria and sterile pyuria 2. Gastrointestinal symptoms are usually managed either by dose reduction or by splitting the total dose into three or four doses per day. Mycophenolate mofetil other side effects are headache, fatigue and dizziness. Uncommon but potentially severe adverse events include blood dyscrasias, gastrointestinal bleeding or ulceration, increased risk for lymphoma or other malignancies, increased risk for serious infections, and embryo-fetal toxicity.
Being an immunosuppressant, mycophenolate mofetil increases the risk of bacterial, fungal and viral infections and has a long-term risk of carcinogenicity like lymphomas and skin cancers.
Other reported adverse events include hematological (i.e., anemia, neutropenia, thrombocytopenia), neurologic (i.e., headache, tinnitus, insomnia), cutaneous (i.e., exanthematous eruptions, onycholysis), cardiorespiratory (i.e., dyspnea, chest pain, palpitations) and metabolic (i.e., hypercholesterolemia, hyperglycemia, hypophosphatemia and hypo/hyperkalemia).
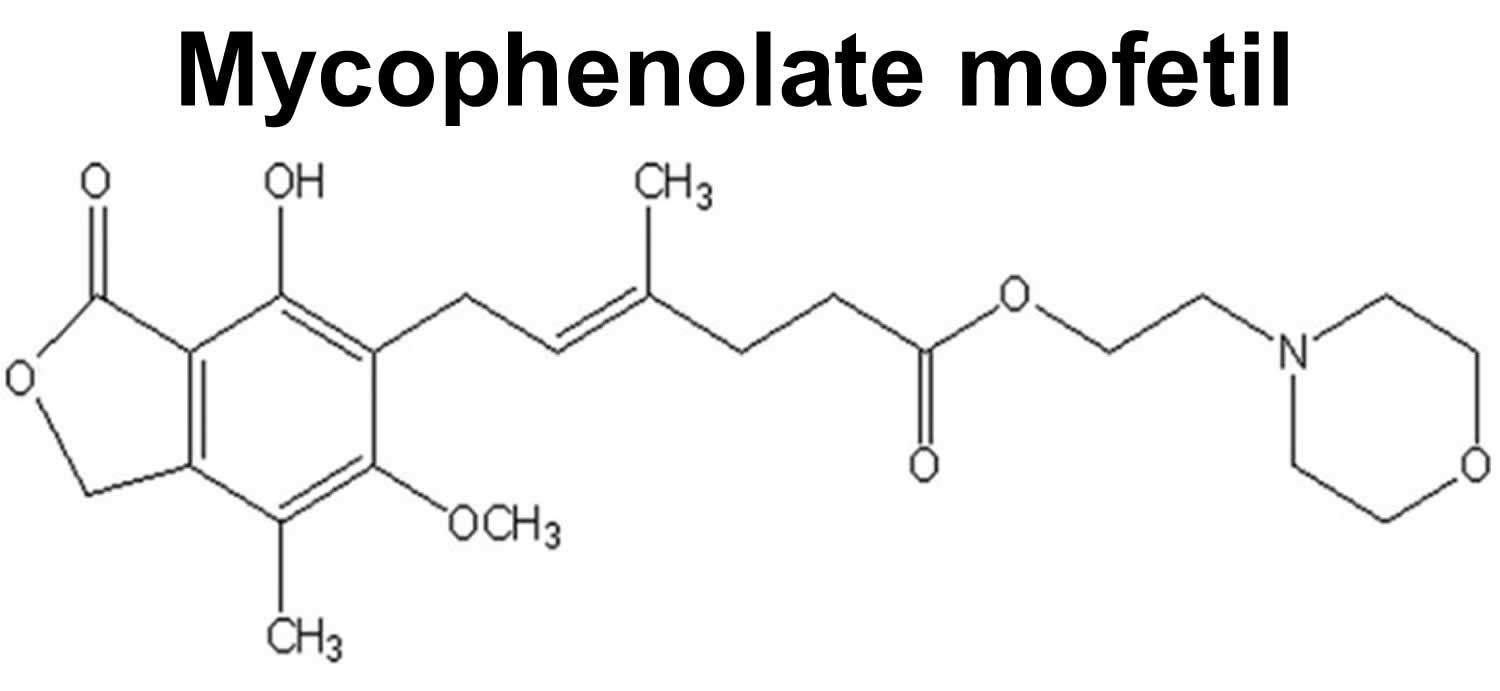
Figure 1. Mycophenolate mofetil chemical structure
Risk of birth defects:
Mycophenolate mofetil must not be taken by women who are pregnant or who may become pregnant. There is a high risk that mycophenolate mofetil will cause miscarriage (loss of the pregnancy) during the first 3 months of pregnancy or will cause the baby to be born with birth defects (problems that are present at birth).
You should not take mycophenolate mofetil if you are pregnant or if you may become pregnant. You must have a negative pregnancy test before starting your treatment with mycophenolate mofetil, again 8 to 10 days later, and at routine follow-up appointments. You must use acceptable birth control during your treatment, and for 6 weeks after you stop taking mycophenolate mofetil. Your doctor will tell you which forms of birth control are acceptable for you to use. Mycophenolate may decrease the effectiveness of oral contraceptives (birth control pills), so it is especially important to use a second form of birth control along with this type of contraceptive.
If you are a male with a female partner who may become pregnant, you should use acceptable birth control during treatment and for at least 90 days after your last dose. Do not donate sperm during your treatment and for at least 90 days after your last dose.
Call your doctor right away if you think you or your partner, is pregnant or if you miss a menstrual period.
Because of the possibility that your donation may go to a female who may be or become pregnant, do not donate blood during your treatment and for at least 6 weeks after your last dose.
Risk of serious infections:
Mycophenolate mofetil weakens the body’s immune system and may decrease your ability to fight infection and increase the risk that you will get a serious infection, including severe fungal, bacterial, or viral infections that spread through the body. These infections may need to be treated in a hospital and may cause death. Tell your doctor if you have any type of infection or if you think you may have any type of infection now. This includes minor infections (such as open cuts or sores), infections that come and go (such as cold sores), and chronic infections that do not go away. Also tell your doctor if you have or have ever had human immunodeficiency virus (HIV), acquired immunodeficiency syndrome (AIDS), hepatitis B virus infection (HBV; an ongoing liver infection), hepatitis C virus infection (HCV; an ongoing liver infection), or herpes zoster (shingles; a rash that can occur in people who have had chickenpox in the past). If you experience any of the following symptoms, call your doctor immediately: fever, sore throat, chills, or cough; unusual bruising or bleeding; pain or burning during urination; frequent urination; wound or sore that is red, warm, or won’t heal; drainage from a skin wound; general weakness, extreme tiredness, or sick feeling; symptoms of the ”flu” or a ”cold”; pain or swelling in the neck, groin, or armpits; white patches in the mouth or throat; cold sores; blisters; headache or earache; or other signs of infection.
Mycophenolate mofetil may increase the risk that you will develop progressive multifocal leukoencephalopathy (PML; a rare infection of the brain that cannot be treated, prevented, or cured and that usually causes death or severe disability). Tell your doctor if you have or have ever had PML, or another condition that affects your immune system. If you experience any of the following symptoms, call your doctor immediately: weakness on one side of the body or in the legs, difficulty or inability to control your muscles, confusion or difficulty thinking clearly, or a lack of interest or concern for usual activities or things you usually care about.
Mycophenolate mofetil may increase your risk of developing certain types of cancer, including lymphoma (a type of cancer that develops in the lymph system) and skin cancer. Tell your doctor if you have or have ever had skin cancer or any other type of cancer. Avoid unnecessary or prolonged exposure to real and artificial sunlight (tanning beds, sunlamps) and light therapy and wear protective clothing, sunglasses, and sunscreen (with a SPF factor of 30 or above). This will help to decrease your risk of developing skin cancer. Call your doctor if you experience any of the following symptoms: pain or swelling in the neck, groin, or armpits; a new skin sore or bump; a change in the size or color of a mole; a brown or black skin lesion (sore) with uneven edges or one part of the lesion that does not look like the other; skin changes; sores that do not heal; unexplained fever; tiredness that does not go away; weight loss; or any other changes to your health.
Your doctor or pharmacist will give you the manufacturer’s patient information sheet (Medication Guide) when you begin treatment with mycophenolate mofetil and each time you refill your prescription. Read the information carefully and ask your doctor or pharmacist if you have any questions.
Keep all appointments with your doctor and the laboratory. Your doctor will order certain lab tests to check your body’s response to mycophenolate mofetil.
Talk to your doctor about the risks of taking mycophenolate mofetil.
Mycophenolate mofetil special precautions
Before taking mycophenolate mofetil:
- tell your doctor and pharmacist if you are allergic to mycophenolate mofetil, mycophenolic acid, any other medications, or any of the ingredients in the mycophenolate mofetil or mycophenolic acid product you are taking. If you are taking mycophenolate mofetil liquid, tell your doctor and pharmacist if you are allergic to aspartame or sorbitol. Ask your pharmacist for a list of the ingredients.
- tell your doctor and pharmacist what other prescription and nonprescription medications, vitamins, nutritional supplements, and herbal products you are taking or plan to take. Also be sure
- tell your doctor if you stop taking any of your medications. Your doctor may need to change the doses of your medications or monitor you carefully for side effects.
- if you are taking sevelamer (Renagel, Renvela), or antacids that contain magnesium or aluminum, take them 2 hours after you take mycophenolate mofetil.
- tell your doctor if you have or have ever had Lesch-Nyhan syndrome or Kelley-Seegmiller syndrome (inherited diseases that cause high levels of a certain substance in the blood, joint pain, and problems with motion and behavior); anemia (a lower than normal number of red blood cells); neutropenia (less than normal number of white blood cells); ulcers or any disease that affects your stomach, intestines, or digestive system; or kidney or liver disease.
- you should not breastfeed while using mycophenolate mofetil.
- you should know that mycophenolate mofetil may make you drowsy, confused, dizzy, lightheaded, or cause uncontrollable shaking of a part of the body. Do not drive a car or operate machinery until you know how this medication affects you.
- do not have any vaccinations without talking to your doctor.
- if you have phenylketonuria (PKU, an inherited condition in which a special diet must be followed to prevent damage to your brain that can cause severe intellectual disability), you should know that mycophenolate mofetil suspension contains aspartame, a source of phenylalanine.
Mycophenolate mofetil can cause a miscarriage or birth defects, especially during the first 3 months of pregnancy. You will need to have a negative pregnancy test before and during treatment with this medicine. If you are able to get pregnant, you must use specific forms of birth control to prevent pregnancy while using mycophenolate mofetil, and for at least 6 weeks after your last dose. Sexually active men and their partners should use effective contraception during treatment and for at least 90 days after the last dose.
Mycophenolate mofetil can make birth control pills less effective. Ask your doctor about using an additional barrier form of birth control (condom, diaphragm, cervical cap, or contraceptive sponge) to prevent pregnancy.
You do not need to use additional birth control if you use an intrauterine device (IUD), if you have had a tubal ligation, or if your sexual partner has had a vasectomy.
This medicine comes with patient instructions about the most effective non-hormonal forms of birth control to use. Follow these directions carefully. Ask your doctor if you have any questions.
If a pregnancy occurs during treatment, do not stop taking mycophenolate mofetil. See your doctor for instructions.
Mycophenolate mofetil is sometimes given to pregnant women. Your doctor will decide whether you should use this medicine if you are unable to use other needed transplant medications.
What should I avoid while using mycophenolate mofetil?
Avoid driving or hazardous activity until you know how mycophenolate mofetil will affect you. Your reactions could be impaired.
Mycophenolate mofetil can make you sunburn more easily. Avoid sunlight or tanning beds. Wear protective clothing and use sunscreen (SPF 30 or higher) when you are outdoors.
Do not receive a “live” vaccine while using mycophenolate mofetil. The vaccine may not work as well and may not fully protect you from disease.
You must not donate blood or sperm while using this medicine, and for at least 6 weeks (for blood) or 90 days (for sperm) after your last dose.
Mycophenolate mofetil contraindications
Mycophenolate mofetil is absolutely contraindicated in patients with known hypersensitivity to any component of the drug and is included in pregnancy category C 2, 5. Peptic ulcer disease, kidney disease, liver disease, lactation and cardiopulmonary disease are the relative contraindications 6
Safety and efficacy have not been established in patients younger than 3 months.
Consult WARNINGS section for additional precautions.
Mycophenolate mofetil drug interactions
Drugs such as acyclovir, ganciclovir and probenecid inhibit the tubular secretion of mycophenolate mofetil and hence, increase its level. Drugs like antacids (containing aluminum and magnesium), ferrous sulphate, metronidazole and fluoroquinolones decrease the absorption and bioavailability of mycophenolate mofetil and decrease its blood level. Cholestyramine inhibits enterohepatic recirculation of mycophenolate mofetil and decreases its level. Salicylates and furosemide compete with mycophenolate mofetil for plasma albumin binding and hence, increase the elimination of mycophenolate mofetil. Mycophenolic mofetil does not seem to interact with other immunosuppressive agents except azathioprine. A combination of mycophenolate mofetil and azathioprine should not be used as both block purine synthesis by the same pathway 2, 5
Mycophenolate mofetil mechanism of action
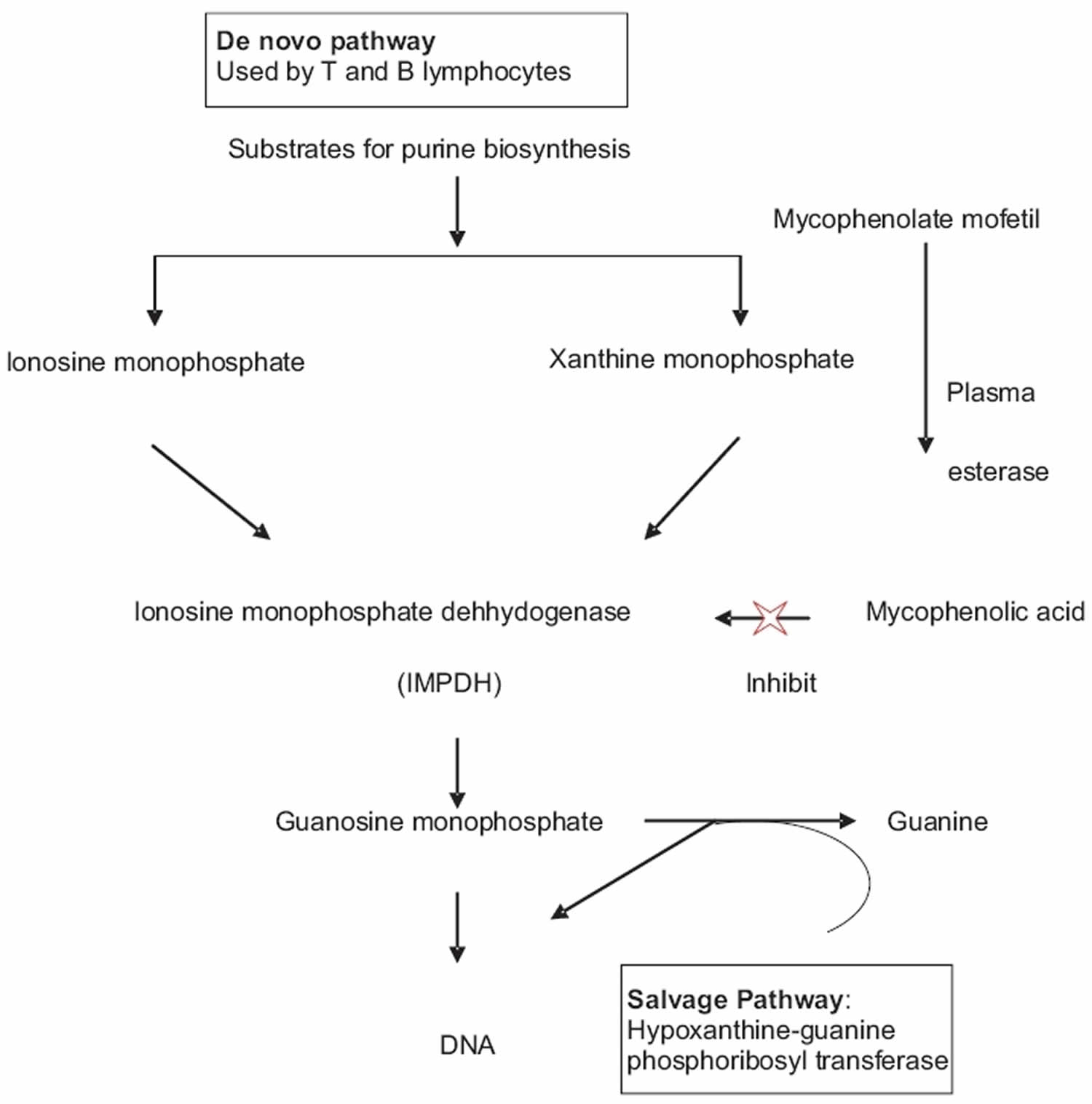
Mycophenolate mofetil is a prodrug (inactive drug) that is rapidly converted in your body by plasma esterase to active metabolite called mycophenolic acid (the active metabolite of mycophenolate mofetil) 7. Mycophenolic acid (MPA) is a potent immunosuppressive agent and acts as a selective, uncompetitive and reversible inhibitor of the enzyme, inosine monophosphate dehydrogenase (IMPDH), a key enzyme in the de novo purine biosynthesis pathway 8, 9, 10. Inosine monophosphate dehydrogenase (IMPDH) is an enzyme required for the conversion of inosine monophosphate (IMP) and xanthine monophosphate (XMP) to guanosine monophosphate, which is an important substrate for the synthesis of DNA and RNA 8, 9, 10. T and B lymphocytes are more dependent on de novo synthesis of purines for their proliferation than other cell types that can utilize the salvage pathway, i.e., hypoxanthine-guanine phosphoribosyl transferase pathway for their proliferation 11. Mycophenolic acid (MPA) has more potent cytostatic effects on T and B lymphocytes and causes decreases in the levels of immunoglobulins and delayed type hypersensitivity responses 12. Mycophenolate mofetil also prevents the glycosylation of lymphocyte and monocyte glycoproteins that are involved in adhesion to endothelial cells. Therefore, it inhibits chemotaxis, impairs antigen presentation and induces immune tolerance 13. This is the principal mechanism by which mycophenolic acid (MPA) exerts immunosuppressive effects.
There are two isoforms of inosine monophosphate dehydrogenase (IMPDH): Type 1 and type 2. The IMPDH type 1 isoform is used by nonreplicating cells while the IMPDH type 2 isoform is predominantly used by proliferating lymphocytes. Mycophenolic acid (MPA) has five times higher binding affinity for the IMPDH type 2 isoform, which is expressed in activated lymphocytes, than of the type 1 isoform of IMPDH, which is expressed in most cell types and therefore, causing depletion of guanosine nucleotides, inhibition of DNA synthesis and the arrest of replicating lymphocytes in the S phase 14. Therefore, MMF is more cytotoxic to proliferating T and B-lymphocytes.
Three other mechanisms may also contribute to the efficacy of mycophenolic acid (MPA) in preventing allograft rejection and other applications. First, mycophenolic acid can induce apoptosis of activated T-lymphocytes, which may eliminate clones of cells responding to antigenic stimulation. Second, by depleting guanosine nucleotides, mycophenolic acid suppresses glycosylation and the expression of some adhesion molecules, thereby decreasing the recruitment of lymphocytes and monocytes into sites of inflammation and graft rejection. Third, by depleting guanosine nucleotides mycophenolic acid also depletes tetrahydrobiopterin, a co-factor for the inducible form of nitric oxide synthase (iNOS). Mycophenolic acid therefore suppresses the production by inducible form of nitric oxide synthase (iNOS) of nitric oxide (NO) and consequent tissue damage mediated by peroxynitrite 8, 9, 10.
Figure 2. Mycophenolate mofetil mechanism of action
[Source 7 ]Mycophenolate mofetil uses
Mycophenolate mofetil (Cellcept or Myfortic) only U.S. Food and Drug Administration (FDA) approved indication is for the prevention of organ transplant rejection in conjunction with cyclosporine and corticosteroids 15, 16, 17. Mycophenolate mofetil is also used in the management of patients with serious autoimmune diseases.
Mycophenolate mofetil was first used in psoriasis in 1975 and since then, case reports and clinical trials document its use in various skin conditions as “off-label” indications 7:
- Psoriasis
- Vesiculobullous disorders
- Pemphigus vulgaris
- Pemphigus foliaceus
- Paraneoplastic pemphigus
- Bullous pemphigoid
- Cicatricial pemphigoid
- Linear IgA disease
- Epidermolysis bullosa acquisita
- Connective tissue disease
- Systemic lupus erythematosus (SLE)
- Subacute cutaneous lupus erythematosus
- Discoid lupus erythematosus
- Dermatomyositis
- Scleroderma
- Dermatitis
- Atopic dermatitis
- Dyshidrotic dermatitis
- Chronic actinic dermatitis
- Vasculitis
- Urticarial vasculitis
- Takayasu’s arteritis
- Microscopic polyangiitis
- Polyarteritis nodosa
- Wegener’s granulomatosis
- Others: Lichen planus, pyoderma gangrenosum, recurrent erythema multiforme, sarcoidosis, Weber-Christian disease and chronic idiopathic urticaria
Mycophenolate mofetil has been successfully used either as monotherapy or in combination with systemic steroids or as a steroid-sparing agent. Mycophenolate mofetil is best suited for individuals in whom other systemic immunotherapies are contraindicated because of hypertension, impaired renal function or liver disease (()Non-steroidal immunosuppressive drugs. In : Khopkar U, Pande S, Nischal KC. Hand book of Dermatological Drug Therapy. 1 st ed. New Delhi: Elsevier Publication: 2007. p. 127-38.).
Although early reports have shown good efficacy and tolerability of mycophenolate mofetil, randomized clinical trials with long surveillance periods are needed to document the efficacy and long-term safety of the drug in various skin diseases.
Mycophenolate mofetil (MMF) administration advice:
- The IV formulation should be administered within 24 hours following transplantation. The IV formulation can be administered for up to 14 days.
- Patients should be switched to the oral formulation as soon as they can tolerate oral medication.
- The IV formulation should only be administered by a slow IV infusion over at least 2 hours. It should never be administered by rapid or bolus IV injection.
- The IV formulation incompatible with 0.9% normal saline, Ringers and lactated Ringers solutions, and should not be mixed or given via the same catheter with other IV drugs or admixtures.
- The oral tablets should not be crushed or chewed.
- The oral capsules should not be opened.
- Doses may be taken with or without food.
- Take a missed dose as soon as possible, except if it is closer than 2 hours to the next scheduled dose.
Monitoring
Complete physical examination should be carried out at each visit or at least every 6-12 months to look for any opportunistic infections and malignancies. Complete blood counts should be performed at baseline and biweekly during the first 2-3 months and then, monthly through the first year. Liver function and renal function tests should be performed at baseline and serum transaminases should be repeated after one month and then, quarterly. During follow-up, treatment should be discontinued if the leukocyte count is less than 3500-4000 cells/mm³
Mycophenolate mofetil dosage
You must remain under the care of a doctor while you are using mycophenolate mofetil. Follow all directions on your prescription label and read all medication guides or instruction sheets. Use mycophenolate mofetil exactly as directed.
Mycophenolate mofetil injection is given as an infusion into a vein. A healthcare provider will give you this injection.
Take oral mycophenolate mofetil on an empty stomach, at least 1 hour before or 2 hours after a meal.
Swallow the capsule or tablet whole and do not crush, chew, break, or open it. Tell your doctor if you have trouble swallowing a tablet or capsule.
Read and carefully follow any Instructions for Use provided with your medicine. Ask your doctor or pharmacist if you do not understand these instructions.
Shake the oral suspension (liquid) before you measure a dose. Use only the dosing syringe provided with mycophenolate mofetil.
Mycophenolate mofetil (CellCept) and mycophenolic acid (Myfortic) are not absorbed equally in the body. Avoid medication errors by using only the brand, form, and strength of this medicine that your doctor has prescribed.
You will need frequent medical tests.
Store at room temperature away from moisture and heat. Keep the bottle tightly closed when not in use. Throw away any unused liquid that is older than 60 days.
The liquid medicine may also be stored in the refrigerator. Do not freeze.
Adult dose for Organ Transplant
- Kidney transplantation: 1 g orally or IV 2 times a day (2 gm per day); [in clinical trials, 1.5 g orally or IV 2 times a day (3 gm per day) was used effectively, however, the safety profile for 3 gm a day was lower]
- Heart transplantation: 1.5 g orally or IV 2 times a day (3 gm per day)
- Liver transplantation: 1.5 gm orally or 1 gm IV 2 times a day (3 gm per day orally or 2 gm per day IV)
Comments:
- Mycophenolate mofetil should be used concomitantly with cyclosporine and corticosteroids.
- The IV formulation should be administered over no less than 2 hours.
- IV administration is recommended in patients unable to take oral medication; oral administration should be initiated as soon as possible.
Elderly patient dose for Organ Transplant
- Kidney transplantation: 1 g orally or IV 2 times a day (2 gm per day)
- Heart transplantation: 1.5 g orally or IV 2 times a day (3 gm per day)
- Liver transplantation: 1.5 gm orally or 1 gm IV 2 times a day (3 gm per day orally or 2 gm per day IV)
Comments:
- Mycophenolate mofetil should be used concomitantly with cyclosporine and corticosteroids.
- The IV formulation should be administered over no less than 2 hours.
- IV administration is recommended in patients unable to take oral medication; oral administration should be initiated as soon as possible.
Children dose for Organ Transplant
Kidney transplantation
- 3 months to 18 years of age: Oral Suspension: 600 mg/m² orally 2 times a day up to a maximum of 2 grams per day
- Children with a body surface area of 1.25 to 1.5 m² may be dosed with the oral capsules at 750 mg orally 2 times a day (1.5 g per day)
- Children with a body surface area greater than 1.5 m² may be dosed with the oral capsules at 1 g orally 2 times a day (2 g per day)
Comments:
- Mycophenolate mofetil should be used concomitantly with cyclosporine and corticosteroids.
- The IV formulation should be administered over no less than 2 hours.
- IV administration is recommended in patients unable to take oral medication; oral administration should be initiated as soon as possible.
Dose Adjustments
- If a patient develops neutropenia (absolute neutrophil count less than 1300/mm³), dosing should be stopped or the dose reduced, and the patient should be managed appropriately.
- Close clinical monitoring is recommended if this drug is used in patients with liver disease.
- Doses greater than 2 g/day (especially in kidney dysfunction with CrCl less than 25 mL/min) are not recommended.
Higher doses may lead to more severe gastrointestinal adverse reactions and enhanced immunosuppression.
What should I do if I forget a dose?
If you are taking mycophenolate mofetil tablet, capsule, or suspension (Cellcept) take the missed dose as soon as you remember it. However, if the next dose is less than 2 hours away, skip the missed dose and continue your regular dosing schedule. Do not take a double dose to make up for a missed one.
If you are taking mycophenolate mofetil delayed release tablet (Myfortic) take the missed dose as soon as you remember it. However, if it is almost time for the next dose, skip the missed dose and continue your regular dosing schedule. Do not take a double dose to make up for a missed one.
Mycophenolate mofetil side effects
Mycophenolate mofetil may cause side effects. Tell your doctor if any of these symptoms are severe or do not go away:
- constipation
- nausea
- vomiting
- difficulty falling asleep or staying asleep
- pain, especially in the back, muscles, or joints
- headache
- gas
- prickling, tingling, or burning feeling on the skin
- swelling of the hands, arms, feet, ankles, or lower legs
- tremor
Mycophenolate mofetil affects your immune system and may increase your risk of cancer or serious infection. See your doctor right away if you have:
- fever of 100.5° F (38 °C) or higher, swollen glands, painful mouth sores, cold or flu symptoms, headache, ear pain, loss of smell or taste, stomach pain, vomiting, diarrhea, weight loss;
- weakness on one side of your body, loss of muscle control, confusion, thinking problems, loss of interest in things that normally interest you;
- pain around the transplanted kidney, pain or burning when you urinate, dark urine, jaundice (yellowing of the skin or eyes);
- tingly or painful blistering rash on one side of your body, swelling, warmth, redness, or oozing around a skin wound; or
- a new skin lesion, or a mole that has changed in size or color.
Some side effects can be serious. If you experience any of the following symptoms or those listed in the IMPORTANT WARNING section, see your doctor immediately:
- sudden severe stomach pain, stomach pain that doesn’t go away, or diarrhea
- difficulty breathing
- chest pain
- rash
- itching
- dizziness, fainting, pale skin, lack of energy, shortness of breath, or fast heartbeat
- unusual bleeding or bruising; vomiting or spitting up blood or brown material that resembles coffee grounds; bloody or black, tarry stools; or blood in urine
- fever, muscle or joint stiffness or pain.
Get emergency medical help if you have symptoms of an allergic reaction to mycophenolate mofetil such as hives, difficult breathing, swelling of your face, lips, tongue, or throat.
Mycophenolate mofetil may cause a serious brain infection that can lead to disability or death. Call your doctor right away if you have problems with speech, thought, vision, or muscle movement. These symptoms may start gradually and get worse quickly.
Mycophenolate mofetil may cause other side effects. See your doctor if you have any unusual problems while taking mycophenolate mofetil.
Mycophenolate mofetil overdose
Mycophenolate mofetil overdose symptoms may include the following:
- stomach pain
- nausea
- vomiting
- heartburn
- diarrhea
- fever, sore throat, chills, cough and other signs of infection
Mycophenolic acid (the active metabolite) and the inactive glucuronide metabolite are not removed in significant amounts by hemodialysis.
- LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Mycophenolate. [Updated 2020 Feb 3]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK548945[↩]
- Perlis C, Pan TD, McDonald CJ. Cytotoxic agents. In : Wolverton S, editor. Comprehensive dermatologic drug therapy. 2 nd ed. Philadelphia: WB Saunders; 2000. p. 197-217.[↩][↩][↩][↩]
- Assmann T, Ruzicka T. New immunosuppressive drugs in dermatology (mycophenolate mofetil, tacrolimus): Unapproved uses, dosages or indications. Clin Dermatol 2002;20:505.[↩]
- Product Monograph. Myofortic (mycophenolate sodium). Novartis Pharma: USA; February 2004.[↩]
- Non-steroidal immunosuppressive drugs. In : Khopkar U, Pande S, Nischal KC. Hand book of Dermatological Drug Therapy. 1 st ed. New Delhi: Elsevier Publication: 2007. p. 127-38.[↩][↩]
- Hoffman-La Roche Inc. Cellcept (Mycophenolate mofetil) Product Information: 1995.[↩]
- Surjushe A, Saple D G. Mycophenolate mofetil. Indian J Dermatol Venereol Leprol 2008;74:180-184. https://ijdvl.com/mycophenolate-mofetil[↩][↩][↩]
- Allison AC, Eugui EM. Mycophenolate mofetil and its mechanisms of action. Immunopharmacology. 2000 May;47(2-3):85-118. https://doi.org/10.1016/S0162-3109(00)00188-0[↩][↩][↩]
- Allison AC. Mechanisms of action of mycophenolate mofetil. Lupus. 2005;14 Suppl 1:s2-8. https://doi.org/10.1191/0961203305LU2109[↩][↩][↩]
- Allison AC. Mechanisms of action of mycophenolate mofetil in preventing chronic rejection. Transplant Proc. 2002 Nov;34(7):2863-6. https://doi.org/10.1016/S0041-1345(02)03538-8[↩][↩][↩]
- Schiff MH, Goldblum R, Rees MM. New DMARD. Mycophenolate mofetil (Myco-M) effectively treats rheumatoid arthritis (RA) patients for one year. Arthritis Rheum 1991;34:89.[↩]
- Sliverman KJE, Pomeianz MK, Pak G, Washenik K, Schupack JL. Rediscovering mycophenolic acid: A review of its mechanism, side effects and potential uses. J Am Acad Dermatol 1997;37:445.[↩]
- Mehling A, Grabbe S, Voskort M, Schwarz T, Luger TA, Beissert S. Mycophenolate mofetil impairs the maturation and function of murine dendritic cells. J Immunol 2000;165:2374.[↩]
- Carr SF, Papp E, Wu JC, Natsumeda Y. Characterization of human type 1 and type 2 IMP dehydrogenases. J Biol Chem 1993;268:272-86.[↩]
- Drugs@FDA: FDA-Approved Drugs. https://www.accessdata.fda.gov/scripts/cder/daf[↩]
- Hart A, Smith JM, Skeans MA, Gustafson SK, Stewart DE, Cherikh WS, Wainright JL, Boyle G, Snyder JJ, Kasiske BL, Israni AK. Kidney. Am J Transplant. 2016 Jan;16 Suppl 2(Suppl 2):11-46. doi: 10.1111/ajt.13666[↩]
- Ekberg H, Tedesco-Silva H, Demirbas A, Vítko S, Nashan B, Gürkan A, Margreiter R, Hugo C, Grinyó JM, Frei U, Vanrenterghem Y, Daloze P, Halloran PF; ELITE-Symphony Study. Reduced exposure to calcineurin inhibitors in renal transplantation. N Engl J Med. 2007 Dec 20;357(25):2562-75. doi: 10.1056/NEJMoa067411[↩]