Contents
What is rheumatic heart disease
Rheumatic heart disease is a chronic valvular heart disease resulting after severe or repetitive episodes of acute rheumatic fever, an autoimmune response to group A Streptococcus infection 1. Inadequately treated group A streptococcal pharyngitis (inflammation of the pharynx, causing a sore throat) can lead to development of acute rheumatic fever and subsequent rheumatic heart disease, both of which are found at high rates among children living in the South Pacific and developing countries. Chronic rheumatic heart disease remains an important public health problem in developing countries. Long-term penicillin injections are effective in preventing recurrent acute rheumatic fever attacks and subsequent development of rheumatic heart disease. Efforts to improve pharyngitis diagnosis and treatment and compliance with penicillin prophylaxis might reduce the burden of acute rheumatic fever and rheumatic heart disease among children.
Rheumatic heart disease almost always affects left-sided heart valves 2. Although right-sided valves are rarely damaged directly, they are usually affected as a consequence of malfunction of the left sided valves. The most common manifestation in the early stages is leakiness of a left-sided (mitral) valve, which allows blood to leak back from the lower chamber (ventricle) to the upper chamber (atrium). This can remain silent for several years as the heart enlarges to compensate before it finally fails. Narrowing of the mitral valve can also develop, which results in obstruction of blood flow (Figures 2 & 3). The resulting back pressure can be reflected to the right side, responsible for leakiness of the right sided valves and enlargement of the right chambers.
Rheumatic heart disease symptoms
Symptoms of rheumatic heart disease include features of heart failure such as shortness of breath, ankle swelling, fatigue, and rapid heartbeat (attributable to abnormal rhythms resulting from dilated heart chambers) 2. Stagnation of blood in enlarged chambers can result in clot formation, which can then break off and obstruct blood flow to different parts of the body, resulting for example, in a stroke. Very often, the diagnosis is made late in the course of the disease, through complications such as these or signs of heart failure.
Diagnosis by the physician is mainly dependent on identifying abnormal heart murmurs generated from the diseased valves and may be of limited accuracy, especially in early stages of the disease. Scanning the heart using ultrasound (called echocardiography) helps in earlier diagnosis and in identifying the affected valve(s), which are typically thickened and in advanced stages, structurally distorted 3.
Open-heart valve surgery or other invasive procedures (such as balloon dilatation of the narrowed valve) are usually required for advanced disease. These sometimes carry significant risk, are expensive, and are often not readily available in the developing world. Hence, prevention and early identification are valuable.
Figure 1. Rheumatic Fever
What is Rheumatic Fever
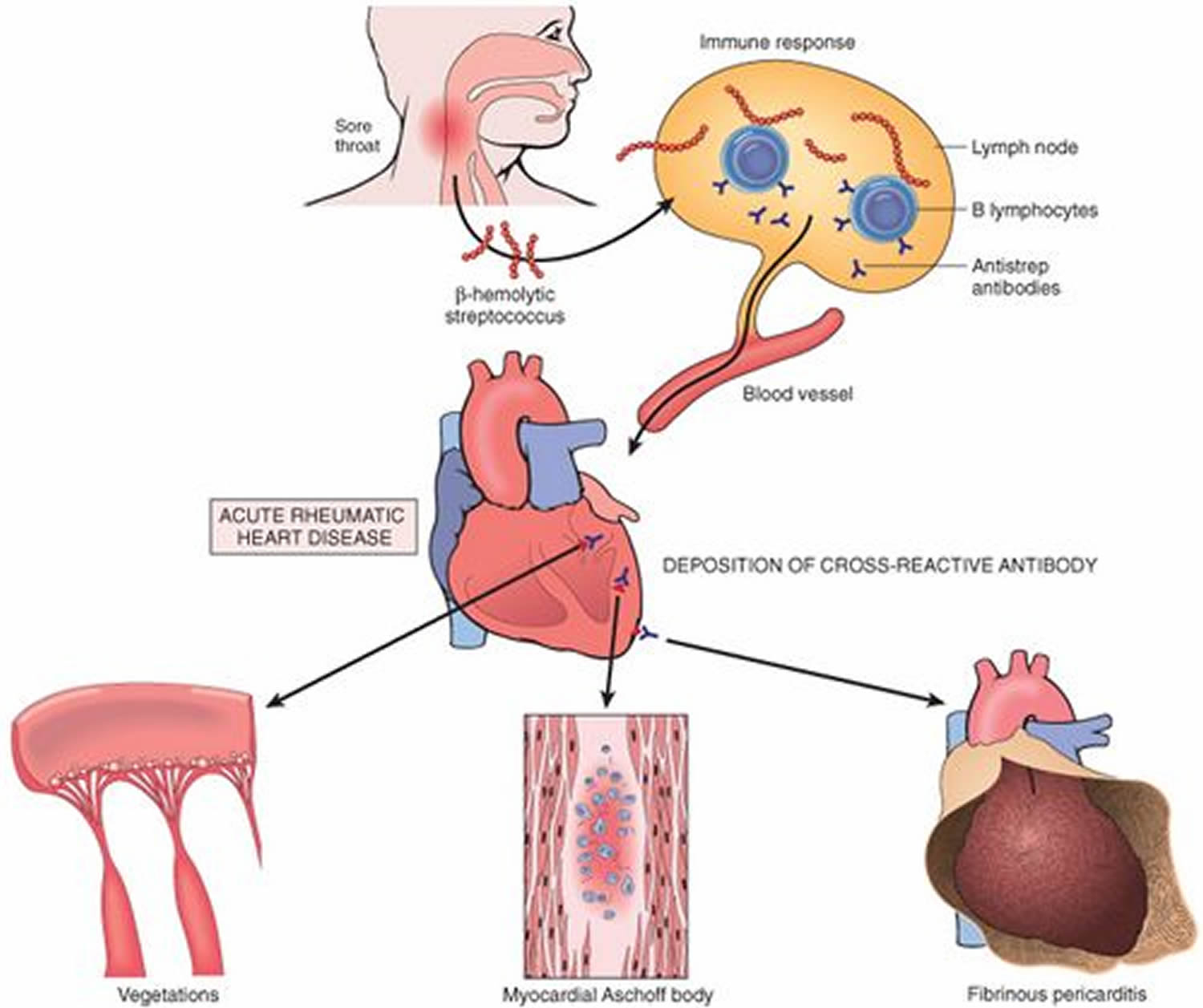
Rheumatic fever is not an infection itself, but rather the result of an untreated group A streptococcal throat infection 4. When your body senses the strep infection, it generates antibodies to help destroy the bacteria. However, because of close structural similarity between certain parts of the bacterial cell wall and heart tissue, these antibodies may also act against the person’s own heart valves (which separate the different chambers of the heart). This leads to valve damage in the form of narrowed (stenotic) or leaky (regurgitant) valves. Over time, there is progressive damage (rheumatic heart disease) that may lead to heart failure, stroke, infection of the valves (infective endocarditis), and death 3.
Rheumatic fever is an immunologically mediated inflammatory disease, that occurs as a delayed reaction to group A streptococcal throat infection, in genetically susceptible individuals 5. Group A streptococcal infection, in addition to causing pharyngitis, pyoderma (a skin infection with formation of pus) and severe invasive disease (e.g., streptococcal toxic shock syndrome and necrotizing fasciitis), group A streptococcal organisms can trigger postinfection syndromes that result from a crossreaction between patient antibodies to bacterial surface proteins and cardiac, neuronal, and synovial tissues 6. The pathogenesis of rheumatic fever in susceptible individuals is related to autoimmune humoral and cellular responses directed toward human tissues, triggered by the response to β-hemolytic group A streptococci. Streptococcal antibodies cross-react with several human tissues including heart, skin, brain, glomerular basement membrane, striated and smooth muscles 7. Rheumatic heart disease occurs in 30 to 45% of rheumatic fever patients 8. Heart involvement induces pericardium (the membrane enclosing the heart, consisting of an outer fibrous layer and an inner double layer of serous membrane), myocardium (the muscular tissue of the heart) and endocardium (the thin, smooth membrane which lines the inside of the chambers of the heart and forms the surface of the valves) inflammation, leading to permanent damage of heart valves 7. Recurrent or severe acute rheumatic fever can cause permanent heart valve damage and rheumatic heart disease, which increases the risk for cardiac conditions (e.g., infective endocarditis, stroke, and congestive heart failure) 9, 10. Mitral valve immunohistochemistry studies with acute rheumatic fever patients show the predominance of CD4+ T cells and macrophages at the site of heart lesions 11, 12.
Acute rheumatic fever, characterized primarily by carditis (inflammation of the heart), chorea (a neurological disorder characterized by jerky involuntary movements affecting especially the shoulders, hips, and face) and polyarthritis (arthritis that involves 5 or more joints simultaneously), occurs a minimum of 2–3 weeks after an episode of untreated or inadequately treated pharyngitis 13. Acute rheumatic fever does not cause lasting damage to the nervous tissue or joints. However, damage to heart valves can be irreversible and is worsened by repeat episodes of acute rheumatic fever 9, 14. Permanent valvular damage, or rheumatic heart disease, increases the risk for infective endocarditis, stroke, heart failure, and premature death, and might necessitate valve replacement surgery 15. Because pharyngitis and acute rheumatic fever are most common in children, the recurrence of acute rheumatic fever, and, thus, the risk for developing rheumatic heart disease, can continue into adolescence and young adulthood.
Antibiotics can prevent acute rheumatic fever if administered no more than 9 days after symptom onset 13. Long-term benzathine penicillin G injections are effective in preventing recurrent acute rheumatic fever attacks and are recommended to be administered every 3–4 weeks for 10 years or until age 21 years to children who receive a diagnosis of acute rheumatic fever 14.
Acute rheumatic fever is no longer a nationally notifiable disease in the United States, and its annual incidence in the continental United States declined in the late 20th century to approximately 0.04–0.06 cases per 1,000 children 16. Exceptions to these low acute rheumatic fever incidence rates in the United States include Samoan persons living in Hawaii and residents of American Samoa, an American territory in the South Pacific 17, 18. Acute rheumatic fever rates in Hawaii have been as high as nearly 0.1 cases per 1,000 children, with even higher rates among persons of Samoan and Hawaiian ethnicity 17. Acute rheumatic fever occurs most commonly among children aged 5–15 years 13.
With improved diagnosis and treatment of group A streptococcal pharyngitis, the United States and other industrialized countries have seen a steep decline in rheumatic heart disease prevalence since the mid-20th century. However, in some parts of the world, rheumatic heart disease is the most common cardiac disease of children and young adults 14. The highest rheumatic heart disease rates occur in sub-Saharan Africa, with an estimated 5.7 cases per 1,000 children aged 5–14 years, and in the Pacific region and indigenous populations of Australia and New Zealand, with 3.5 cases per 1,000 15.
The epidemiology of rheumatic fever in developed countries has changed dramatically over the past decades. In developing areas, the prevalence is still high at up to 24 per 1000 population 19. Rheumatic fever occurs most frequently among children and adolescents between 5 and 18 years, coinciding with the age distribution of the highest prevalence of streptococcal infections 20. Multiple factors influence rates of acute rheumatic fever and rheumatic heart disease, including host immune factors and lifestyle (e.g., crowding or access to health care), as well as the biologic characteristics of circulating group A streptococcal strains 9. However, opportunities for prevention exist and include improving access to medical care and using evidence-based strategies to identify and treat group A streptococcal pharyngitis early (primary prevention) and diagnose and prevent recurrent acute rheumatic fever and rheumatic heart disease (secondary prevention) 14.
Signs and symptoms of rheumatic fever
Arthritis
Arthritis is the most common manifestation, present in 60-80% of patients 21, 22. Arthritis usually affects the peripheral large joints; small joints and axial skeleton are rarely involved. Knees, ankles, elbows and wrists are the most frequently affected 5. In addition to arthralgia (painful joint), the joints are red, warm and swollen. Arthritis is characteristically asymmetrical, migratory, and very painful, although some patients may present mild joint complaints. It usually resolves spontaneously at the most in 2 or 3 weeks. Arthritis in acute rheumatic fever has an excellent response to salicylates (e.g. aspirin, indomethacin, ibuprofen) 23.
Approximately one third of patients with joint involvement reported in a series of 786 cases of rheumatic fever presented with “atypical” articular manifestations, considered by some as a separate entity, i.e., post-streptococcal reactive arthritis. It seems reasonable to include these manifestations as part of the spectrum of rheumatic fever. Secondary prophylaxis should be recommended in these situations.
Carditis
Acute carditis was present in 50% of patients in a large recent series 21. It is a pancarditis, but valvular involvement is the rule. The commonest involved valve is the mitral, frequently associated with aortic valve involvement. Right-sided heart valves are rarely affected.
On pathological examination, the valves are thickened and display rows of small vegetations along their apposing surfaces. Myocarditis is characterised by infiltration of mononuclear cells, vasculitis and degenerative changes of the interstitial connective tissue 24. The pathognomonic lesion is the Aschoff body in the proliferative stage, present in 30 to 40 per cent of biopsies of patients with acute rheumatic fever 25. It is seen mainly in the interstitial connective tissue of the myocardium, particularly perivascularly 24. Inflammation of the valves consists of oedema and mononuclear cell infiltration of the valvular tissue and the chordae tendineae in the acute phase; fibrosis and calcification occur with maintenance of the inflammatory process 24.
The clinical picture includes high pulse rate, congestive heart failure, arrhytmias and pericardial friction rubs. On the first attack, valvulitis is suspected in the presence of a new apical systolic murmur of mitral regurgitation (associated or not with an apical mid-diastolic murmur) and/or a basal diastolic murmur of aortic regurgitation 26. Cardiomegaly is noted on X-Ray and on echocardiogram. Myocarditis and/or pericarditis in the absence of valvular involvement is unlikely due to acute rheumatic fever 26. It is contentious if myocardial disfunction in acute rheumatic fever is valvular or myocardial in origin 27. In fact, in a subset of patients, the initial presentation may be quite severe, with overt heart failure, fever and toxaemia, making the differential diagnosis with infective endocarditis very difficult, in particular in patients with recurrent rheumatic heart disease 5.
The valvular lesions in rheumatic fever often result in residual damage. Nevertheless, in milder forms of rheumatic carditis patients may recover from valvulitis without sequelae 28. In the first attack, the lesions are predominantly regurgitant, due to ring dilatation, swollen cusps, chordal rupture or papillary muscle dysfunction. In the chronic phase, obstructive lesions are more frequent.
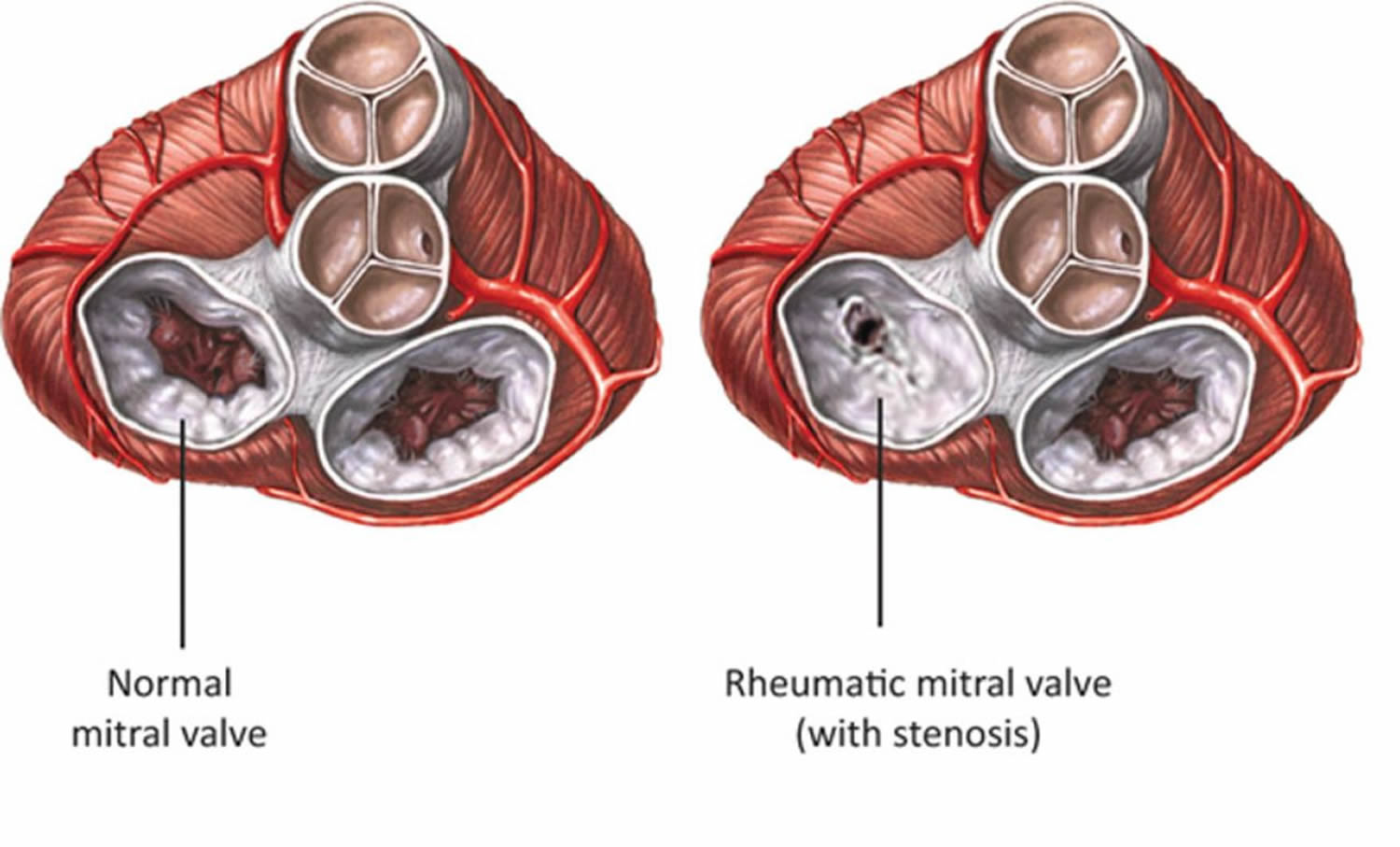
Figure 2. Example of the pathological presentation of chronic rheumatic mitral valve disease.
[Source 29]Note: Rheumatic mitral stenosis. Cross-sectional view of the base of the heart showing the 4 valves. The mitral valve (a left-sided heart valve; identified by arrows) has 2 leaflets around an orifice through which it allows blood flow from the upper chamber (atrium) to the lower chamber (ventricle). On the left-hand side is a normal mitral valve with normal leaflets, whereas on the right-hand side a mitral valve narrowed as a result of rheumatic heart disease is depicted. Note how the orifice is severely narrowed as a result of abnormal thickening and fusion of the valve leaflets. Obstruction to blood flow through the narrowed orifice results in symptoms as heart failure or stroke.
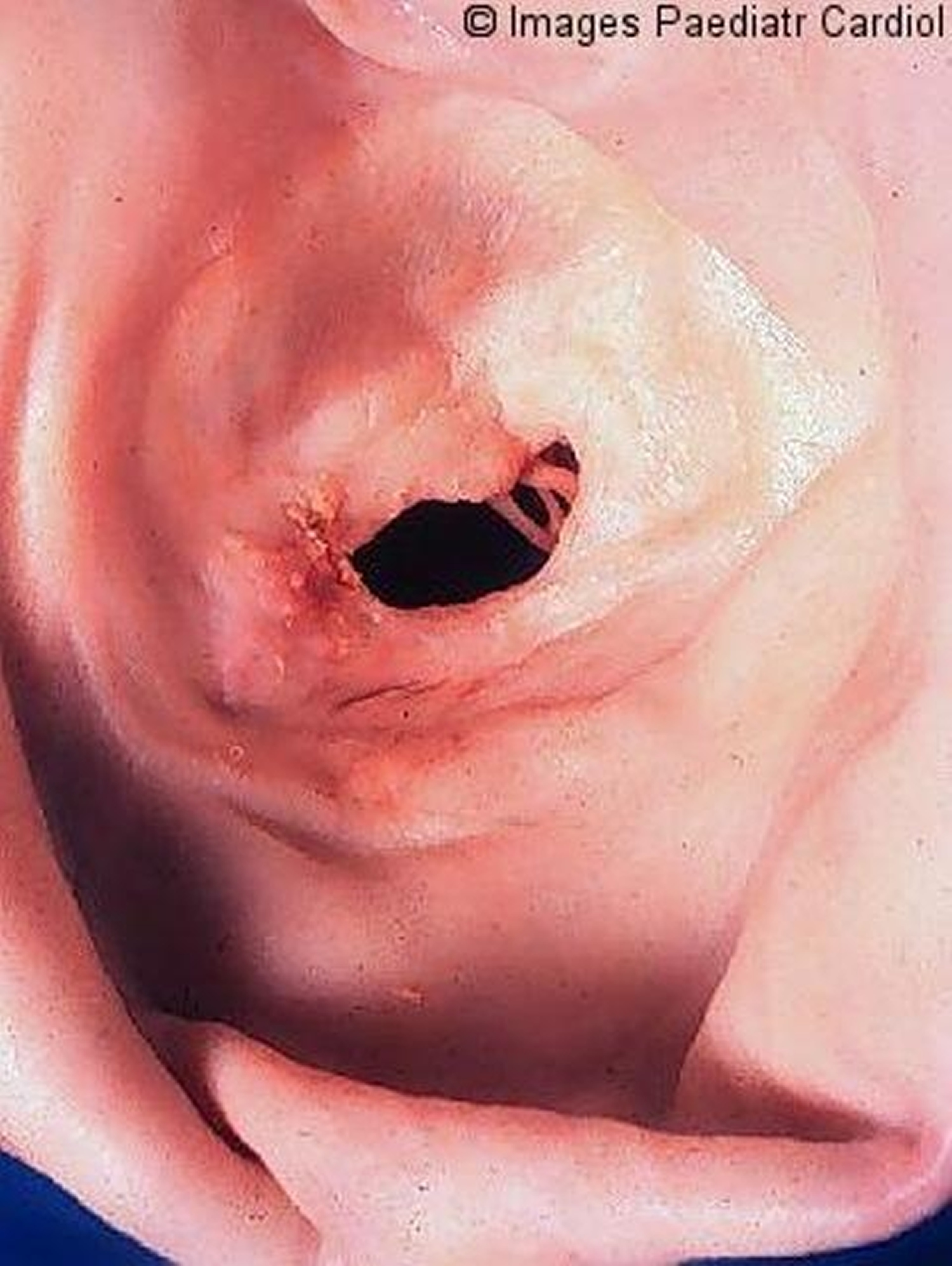
Figure 3. Photo of chronic rheumatic mitral valve disease.
Note: Rheumatic mitral stenosis. Cross-sectional view of the base of the heart showing the 4 valves. The mitral valve (a left-sided heart valve; identified by arrows) has 2 leaflets around an orifice through which it allows blood flow from the upper chamber (atrium) to the lower chamber (ventricle). On the left-hand side is a normal mitral valve with normal leaflets, whereas on the right-hand side a mitral valve narrowed as a result of rheumatic heart disease is depicted. Note how the orifice is severely narrowed as a result of abnormal thickening and fusion of the valve leaflets. Obstruction to blood flow through the narrowed orifice results in symptoms as heart failure or stroke.Both commissures are fused; the cusps are severely thickened. The left atrium is huge. The valve is both incompetent and stenotic (narrowed)
[Source 5]Sydenham’s chorea
Sydenham’s chorea is characterized by involuntary movements, specially of the face and limbs, muscle weakness, disturbances of speech and gait. Children usually exhibit concomitant psycologic dysfunction, especially obsessive-compulsive disorder, increased emotional lability, hyperactivity, irritablility and age-regressed behavior 30, 31, 32. It is usually a delayed manifestation, and is often the sole manifestation of acute rheumatic fever. However, chorea may occur in association with other major manifestations of rheumatic fever, particularly in the first attack 33. Evidence of a recent group A streptococcal infection is often difficult to document. Most of the patients experience resolution of the symptomatology after a few months 34, 26. However, a recurrence rate up to 32% has been described, despite the regular use of secondary benzathine penicillin prophylaxis. Some believe that these episodes represent exacerbations rather than distinct attacks of acute rheumatic fever 33, 35.
Subcutaneous nodules
Subcutaneous nodules are rarely seen and when present, they are usually associated with severe carditis. They are painless, firm, movable, measuring around 0.5 to 2 cm. They are usually located over extensor surfaces of the joints, particularly knees, wrists and elbows 26.
Erythema marginatum
This is an evanescent, erythematous, non-pruritic rash with pale centers and rounded or serpiginous margins. Lesions occur mainly on the trunk and proximal extremities and may be induced by application of heat 36.
Diagnosis of rheumatic fever
With the exception of Sydenham’s chorea, which has a latency period of several months, the clinical manifestations of acute rheumatic fever present after about 3 weeks following the streptococcal throat infection. It usually begins with nonspecific symptoms, such as fever, malaise and persistent pallor 34.
There is no specific clinical or laboratory test that establishes the diagnosis of rheumatic fever. Diagnosis is based on the revised Jones Criteria (Table 1) 26. Arthritis, carditis, chorea, and less frequently, subcutaneous nodules and erythema marginatum are major manifestations of RF. If supported by evidence of preceding streptococcal infection, the presence of two major manifestations or one major and two minor manifestations indicates a high probability of acute rheumatic fever. The two exceptions to this requirement are Sydenham’s chorea and indolent carditis. Other manifestations include arthralgia, serositis and involvement of the kidneys and lungs 34.
Table 1. Guidelines for the diagnosis of initial attack of rheumatic fever (Jones Criteria, 1992 Update)
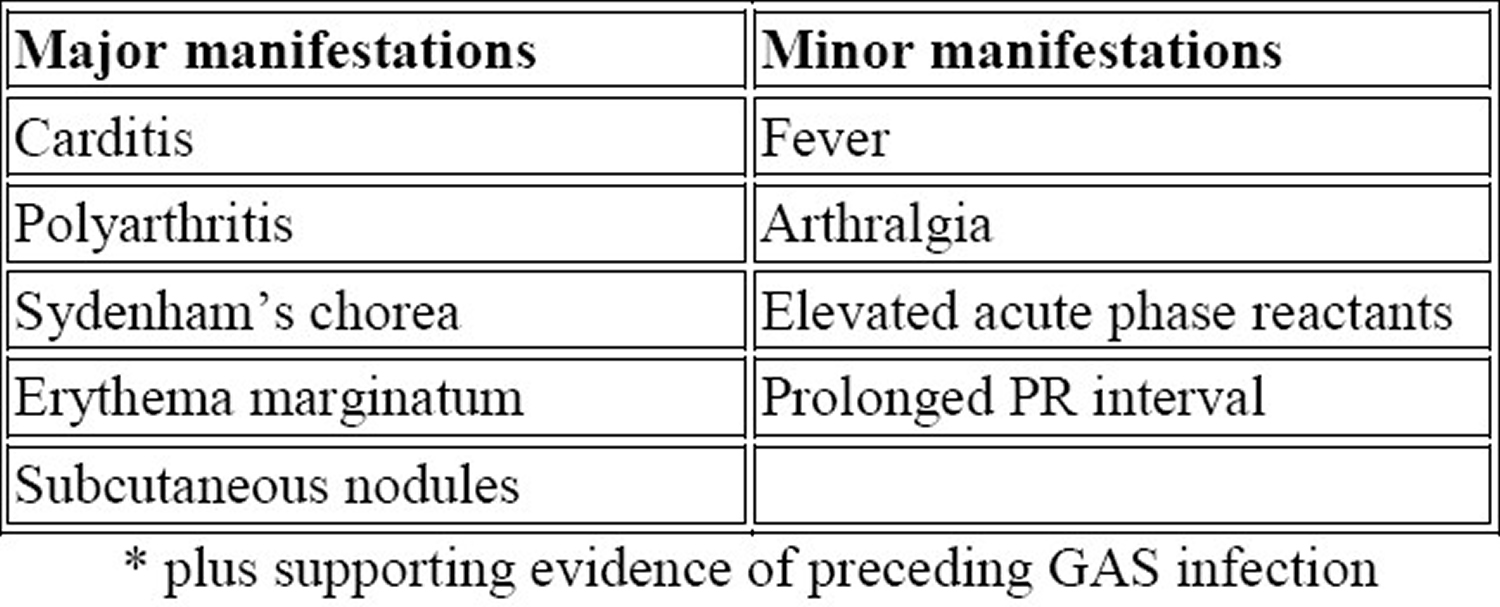
Note: GAS = Group A Streptococcus
Treatment of rheumatic fever
Prevention
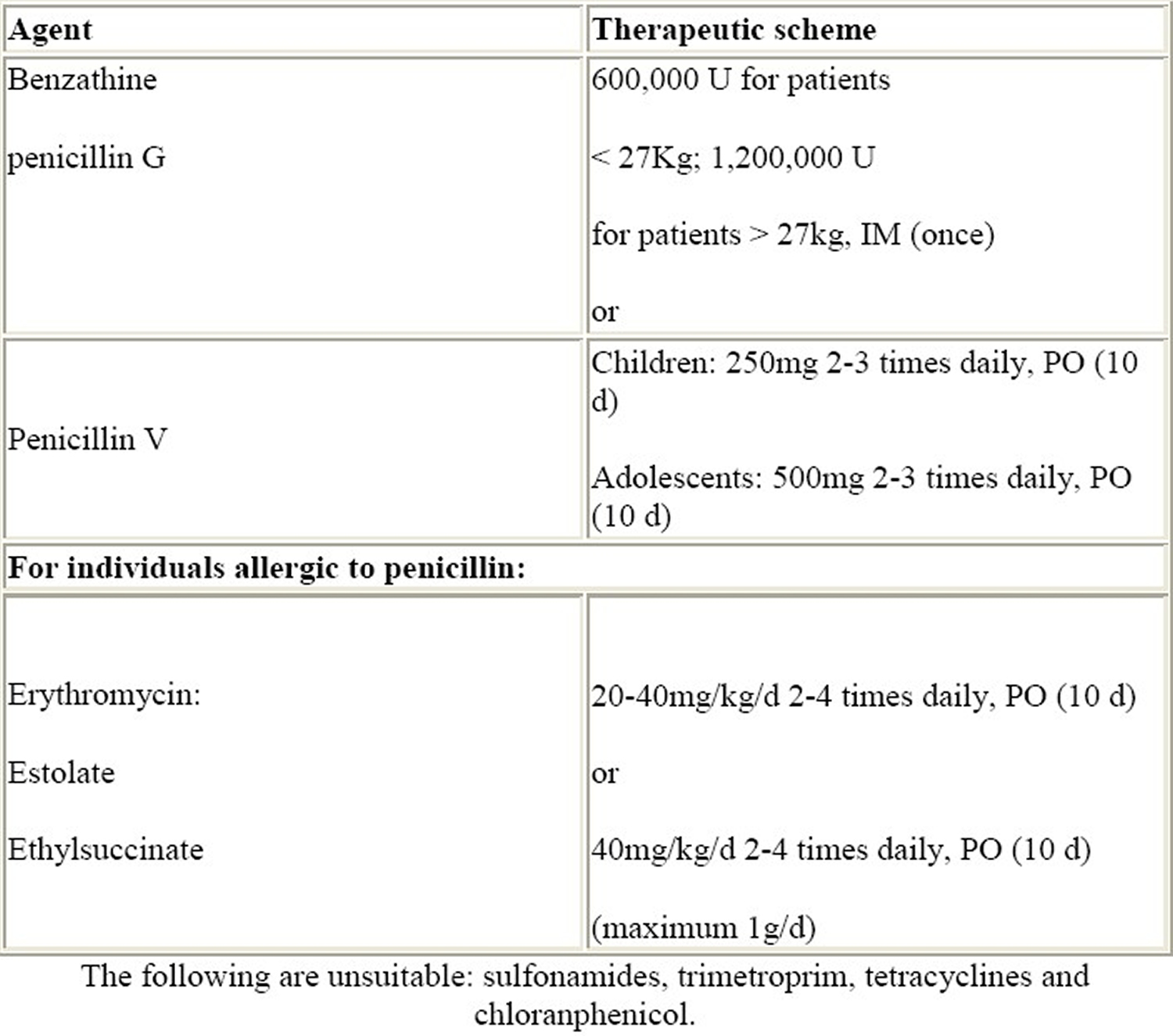
Prevention of initial attacks of rheumatic fever (primary prevention) requires the eradication of group A streptococci from the pharynx. Table 2 shows the currently recommended treatment schedules 37. Emphasis should be given to the need of eradication of group A streptococci as part of the treatment of acute rheumatic fever.
Prevention also focuses on socio-economic development as it directly impacts on hygiene, access to medical care, and living conditions (such as avoidance of overcrowding). Decline in acute rheumatic fever in the Western countries started before the antibiotic era and has been attributed to better living conditions 38.
Ideally, prevention should avoid the first acute rheumatic fever attack. Thus, primary prevention aims to achieve this by treating all Group A Streptococcus sore throat by oral or injectable penicillin (Table 2). Although not all sore throat leads to acute rheumatic fever, it is important that parents are educated to take their children to the doctor if they have symptoms of a sore throat. The doctor can identify whether the sore throat is attributable to the Group A Streptococcus bacterium by taking a swab and then treating with antibiotics. An even simpler approach where all sore throat is treated using penicillin without costly tests to identify the infecting agent could be especially relevant in resource-limited settings 39. Another potential path to primary prevention is the development of a vaccine targeting the bacterium responsible. However, lack of interest from industry and the presence of different strains of bacteria worldwide pose a major challenge.
Table 2. Primary prevention of rheumatic fever
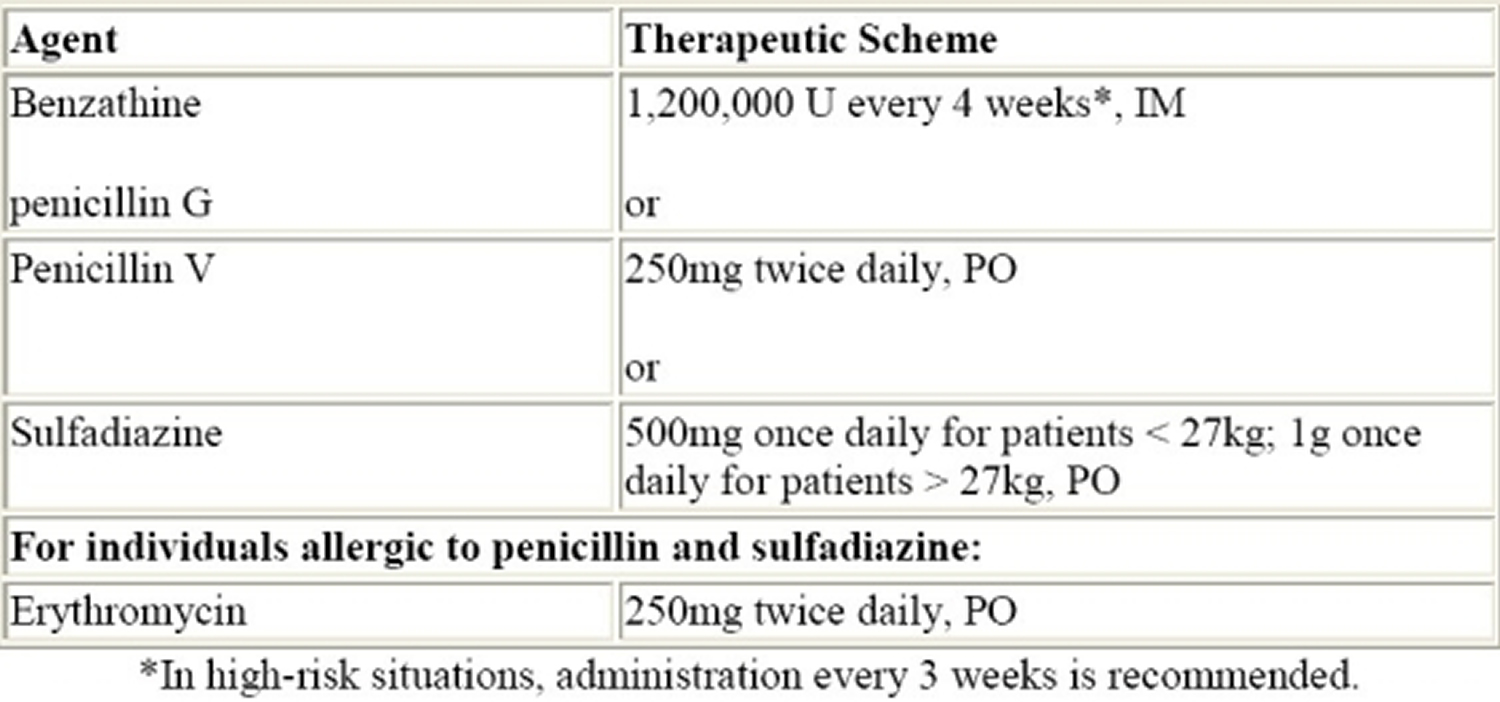
[Source 37]Antibiotic prophylaxis is the safest way to prevent recurrent attacks of acute rheumatic fever and is recommended for patients with well-documented rheumatic fever. The recommendations from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease of the Council on Cardiovascular Disease in the Young of the American Heart Association are shown on Table 3 37.
Table 3. Antibiotic prophylaxis (secondary prevention) to prevent recurrent attacks of acute rheumatic fever
[Source 37]Arthritis
Salicylates remain the first-line drugs in the treatment of arthritis. The response is usually excellent. Treatment should be started at 80 to 100 mg/kg/day (maximum, 4g daily) for 3-4 weeks. Naproxen (10-15mg/kg/day, bid) is an alternative drug, with very good response 5. Other nonsteroidal antiinflammatory drugs also can be used.
Carditis
Moderate to severe carditis is usually an indication for cortiscoteroids although efficacy in reducing sequelae has not been proven so far. Albert at al. 40 performed a meta-analysis of the literature on the treatment of rheumatic carditis, comparing corticosteroids and salicylates in preventing valvular damage. It seems clear that corticosteroids are superior to salicylates in rapidly resolving acute manifestations, but the advantage of the former in preventing a pathologic murmur at 1 year posttreatment was not statistically significant. Prednisone, 2mg/kg/day (maximum, 60mg/day) is used for two weeks and after that, the dose is gradually tapered, reducing 20 to 25% of the previous dose every week. Some advocate the concomitant use of salicylates to avoid rebound. In severe carditis, therapy may be initiated with intravenous methylprednisolone 41, 42. Intravenous immunoglobulin seems not to alter the extent and severity of carditis or decrease chronic morbitidy 43.
Heart failure usually responds to steroids. Bed rest is always recommended and should be planned on an individual basis. Diuretics and vasodilators may be used in patients with more severe haemodynamic decompensation. Digoxin should be used with caution because of the risk of toxicity in the presence of active myocarditis 44. Surgical treatment in the acute stage should be considered when clinical therapy is ineffective to control cardic failure. Valve repair, although technically more difficult, is the first choice for younger patients 45.
Chorea
Treatment with haloperidol (initial dose of 0.5 to 1mg/kg/day, maximum, 5mg/day) 46 or valproic acid (15-20 mg/kg/day) 47, 48 are helpful in decreasing the severity of involuntary movements but may not improve the behavioral symptoms. Carbamazepine has also been suggested as a first-line treatment for Sydenham’s chorea 49. Alternatively, phenobarbital also may be used, 5-7mg/kg/day, tid. Treatment is usually maintained for 8-12 weeks. Intravenous immunoglobulin therapy has been suggested 30.
- Current Cardiology Reports October 2016, 18:96. Rheumatic Heart Disease in the Twenty-First Century. https://link.springer.com/article/10.1007[↩]
- Prevention of Acute Rheumatic Fever and Rheumatic Heart Disease. Mariana Mirabel, Kumar Narayanan, Xavier Jouven and Eloi Marijon. Circulation. 2014;130:e35-e37, originally published July 28, 2014. http://circ.ahajournals.org/content/130/5/e35.full[↩][↩]
- Marijon E, Mirabel M, Celermajer DS, Jouven X. Rheumatic heart disease. Lancet. 2012;379:953–964. https://www.ncbi.nlm.nih.gov/pubmed/22405798[↩][↩]
- Texas Heart Institute. Rheumatic Fever. http://www.texasheart.org/HIC/Topics/Cond/rheufev.cfm[↩]
- Binotto M, Guilherme L, Tanaka A. Rheumatic Fever. Images in Paediatric Cardiology. 2002;4(2):12-31. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3232519/[↩][↩][↩][↩][↩]
- Martin JM, Green M. Group A Streptococcus. Semin Pediatr Infect Dis 2006;17:140–8.[↩]
- Rheumatogenic streptococci and autoimmunity. Stollerman GH. Clin Immunol Immunopathol. 1991 Nov; 61(2 Pt 1):131-42. https://www.ncbi.nlm.nih.gov/pubmed/1914256/[↩][↩]
- Guilherme L, Cury P, Demarchi LMF, et al. Rheumatic Heart Disease: Proinflammatory Cytokines Play a Role in the Progression and Maintenance of Valvular Lesions. The American Journal of Pathology. 2004;165(5):1583-1591. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1618676/[↩]
- World Health Organization. Rheumatic fever and rheumatic heart disease. World Health Organ Tech Rep Ser 2004;923:1–122.[↩][↩][↩]
- Carapetis JR, Steer AC, Mulholland EK, Weber M. The global burden of group A streptococcal diseases. Lancet Infect Dis 2005;5:685–94[↩]
- Raizada V, Williams RC, Jr, Chopra P, Gopinath N, Prakash K, Sharma KB, Cherian KM, Panday S, Arora R, Nigam M, Zabriskie JB, Husby G. Tissue distribution of lymphocytes in rheumatic heart valves as defined by monoclonal anti-T cells antibodies. Am J Med. 1983;74:90–96. https://www.ncbi.nlm.nih.gov/pubmed/6336893[↩]
- Kemeny E, Grieve T, Marcus R, Sareli P, Zabriskie JB. Identification of mononuclear cells and T cell subsets in rheumatic valvulitis. Clin Immunol Immunopathol. 1989;52:225–237. https://www.ncbi.nlm.nih.gov/pubmed/2786783[↩]
- Centers for Disease Control and Prevention. Acute Rheumatic Fever and Rheumatic Heart Disease Among Children — American Samoa, 2011–2012. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6420a5.htm[↩][↩][↩]
- Carapetis JR, Brown A, Wilson NJ, Edwards KN; Rheumatic Fever Guidelines Writing Group. An Australian guideline for rheumatic fever and rheumatic heart disease: an abridged outline. Med J Aust 2007;186:581–6.[↩][↩][↩][↩]
- Carapetis JR, Steer AC, Mulholland EK, Weber M. The global burden of group A streptococcal diseases. Lancet Infect Dis 2005;5:685–94.[↩][↩]
- Stockmann C, Ampofo K, Hersh AL, et al. Evolving epidemiologic characteristics of invasive group A streptococcal disease in Utah, 2002‒2010. Clin Infect Dis 2012;55:479–87.[↩]
- Chun LT, Reddy DV, Yim GK, Yamamoto LG. Acute rheumatic fever in Hawaii: 1966 to 1988. Hawaii Med J 1992;51:206–11.[↩][↩]
- Erdem G, Dodd A, Tuua A, et al. Acute rheumatic fever in American Samoa. Pediatr Infect Dis J 2007;26:1158–9.[↩]
- Rheumatic fever. Stollerman GH. Lancet. 1997 Mar 29; 349(9056):935-42. https://www.ncbi.nlm.nih.gov/pubmed/9093263/[↩]
- Community control of rheumatic heart disease in developing countries: 2. Strategies for prevention and control. WHO Chron. 1980 Oct; 34(10):389-95. https://www.ncbi.nlm.nih.gov/pubmed/7445489/[↩]
- da Silva CH. Rheumatic fever: a multicenter study in the state of Sao Paulo. Pediatric Committee–Sao Paulo Pediatric Rheumatology Society. Rev Hosp Clin Fac Med Sao Paulo. 1999;54:85–90. https://www.ncbi.nlm.nih.gov/pubmed/10668278[↩][↩]
- Kiss MHB. Articular involvement in rheumatic fever. Rev.Soc.Cardiol.Estado de Sao Paulo. 1993;3:26–31.[↩]
- Ayoub EM, Majeed HA. Poststreptococcal reactive arthritis. Curr Opin Rheumatol. 2000;12:306–310. https://www.ncbi.nlm.nih.gov/pubmed/10910184[↩]
- Pomerance A. Cardiac involvement in rheumatic and ‘collagen’ diseases. In: Pomerance A, Davies MJ, editors. The pathology of the heart. Oxford: Blackwell Scientific Publications; 1975. pp. 279–306.[↩][↩][↩]
- Narula J, Chopra P, Talwar KK, Reddy KS, Vasan RS, Tandon R, et al. Does endomyocardial biopsy aid in the diagnosis of active rheumatic carditis? Circulation. 1993;88:2198–2205. https://www.ncbi.nlm.nih.gov/pubmed/8222115[↩]
- Guidelines for the diagnosis of rheumatic fever. Jones Criteria, 1992 update. Special Writing Group of the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease of the Council on Cardiovascular Disease in the Young of the American Heart Association. JAMA. 1992;268:2069–2073. https://www.ncbi.nlm.nih.gov/pubmed/1404745[↩][↩][↩][↩][↩][↩]
- Essop MR, Wisenbaugh T, Sareli P. Evidence against a myocardial factor as the cause of left ventricular dilation in active rheumatic carditis. J Am Coll Cardiol. 1993;22:826–829. https://www.ncbi.nlm.nih.gov/pubmed/8354818[↩]
- Stollerman GH. Rheumatic fever. Lancet. 1997;349:935–942. https://www.ncbi.nlm.nih.gov/pubmed/9093263[↩]
- Circulation. 2014;130:e35-e37. Prevention of Acute Rheumatic Fever and Rheumatic Heart Disease. http://circ.ahajournals.org/content/130/5/e35.full[↩]
- Dajani AS, Bisno AL, Chung KJ, Durack DT, Gerber MA, Kaplan EL, et al. Prevention of rheumatic fever: a statement for health professionals by the Committee on Rheumatic Fever, Endocarditis and Kawasaki Disease of the Council on Cardiovascular Disease in the young, the American Heart Association. Pediatr Infect Dis J. 1989;8:263–266. https://www.ncbi.nlm.nih.gov/pubmed/2726321[↩][↩]
- Asbahr FR, Negrao AB, Gentil V, Zanetta DM, da Paz JA, Marques-Dias MJ, et al. Obsessive-compulsive and related symptoms in children and adolescents with rheumatic fever with and without chorea: a prospective 6-month study. Am J Psychiatry. 1998;155:1122–1124. https://www.ncbi.nlm.nih.gov/pubmed/9699708[↩]
- Mercadante MT, Busatto GF, Lombroso PJ, Prado L, Rosario-Campos MC, do VR, et al. The psychiatric symptoms of rheumatic fever. Am J Psychiatry. 2000;157:2036–2038. https://www.ncbi.nlm.nih.gov/pubmed/11097972[↩]
- Terreri MT, Roja SC, Len CA, Faustino PC, Roberto AM, Hilario MO. Sydenham’s chorea–clinical and evolutive characteristics. Sao Paulo Med J. 2002;120:16–19. https://www.ncbi.nlm.nih.gov/pubmed/11836548[↩][↩]
- da Silva NA, Pereira BA. Acute rheumatic fever. Still a challenge. Rheum Dis Clin North Am. 1997;23:545–568. https://www.ncbi.nlm.nih.gov/pubmed/9287377[↩][↩][↩]
- Berrios X, Quesney F, Morales A, Blazquez J, Bisno AL. Are all recurrences of “pure” Sydenham chorea true recurrences of acute rheumatic fever? J Pediatr. 1985;107:867–872. https://www.ncbi.nlm.nih.gov/pubmed/3906071[↩]
- Secord E, Emre U, Shah BR, Tunnessen WW., Jr Picture of the month: Erythema marginatum in acute rheumatic fever. Am.J.Dis.Child. 1992;146:637. https://www.ncbi.nlm.nih.gov/pubmed/1621672[↩]
- Dajani A, Taubert K, Ferrieri P, Peter G, Shulman S. Treatment of acute streptococcal pharyngitis and prevention of rheumatic fever: a statement for health professionals. Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease of the Council on Cardiovascular Disease in the Young, the American Heart Association. Pediatrics. 1995;96:758–764. https://www.ncbi.nlm.nih.gov/pubmed/7567345[↩][↩][↩][↩]
- Prevention of Acute Rheumatic Fever and Rheumatic Heart Disease. Mariana Mirabel, Kumar Narayanan, Xavier Jouven and Eloi Marijon. Circulation. 2014;130:e35-e37, originally published July 28, 2014. https://doi.org/10.1161/CIRCULATIONAHA.113.007855[↩]
- Marijon E, Celermajer DS, Tafflet M, El-Haou S, Jani DN, Ferreira B, Mocumbi AO, Paquet C, Sidi D, Jouven X. Rheumatic heart disease screening by echocardiography: the inadequacy of World Health Organization criteria for optimizing the diagnosis of subclinical disease. Circulation. 2009;120:663–668. http://circ.ahajournals.org/content/120/8/663[↩]
- Albert DA, Harel L, Karrison T. The treatment of rheumatic carditis: a review and meta-analysis. Medicine (Baltimore) 1995;74:1–12. https://www.ncbi.nlm.nih.gov/pubmed/7837966[↩]
- DiSciascio G, Taranta A. Rheumatic fever in children. Am Heart J. 1980;99:635–658. https://www.ncbi.nlm.nih.gov/pubmed/6989222[↩]
- Herdy GV, Pinto CA, Olivaes MC, Carvalho EA, Tchou H, Cosendey R, et al. Rheumatic carditis treated with high doses of pulsetherapy methylprednisolone. Results in children over years. Arq Bras Cardiol. 1999;72:601–606. https://www.ncbi.nlm.nih.gov/pubmed/10668230[↩]
- Voss LM, Wilson NJ, Neutze JM, Whitlock RM, Ameratunga RV, Cairns LM, et al. Intravenous immunoglobulin in acute rheumatic fever: a randomized controlled trial. Circulation. 2001;103:401–406. https://www.ncbi.nlm.nih.gov/pubmed/11157692[↩]
- Thatai D, Turi ZG. Current guidelines for the treatment of patients with rheumatic fever. Drugs. 1999;57:545–555. https://www.ncbi.nlm.nih.gov/pubmed/10235692[↩]
- Pomerantzeff PM, Brandao CM, Faber CM, Grinberg M, Cardoso LF, Tarasoutchi F, et al. Mitral valve repair in rheumatic patients. Heart Surg Forum. 2000;3:273–276. https://www.ncbi.nlm.nih.gov/pubmed/11178286[↩]
- Marques-Dias MJ, Mercadante MT, Tucker D, Lombroso P. Sydenham’s chorea. Psychiatr Clin North Am. 1997;20:809–820. https://www.ncbi.nlm.nih.gov/pubmed/9443351[↩]
- Daoud AS, Zaki M, Shakir R, al Saleh Q. Effectiveness of sodium valproate in the treatment of Sydenham’s chorea. Neurology. 1990;40:1140–1141. https://www.ncbi.nlm.nih.gov/pubmed/2113207[↩]
- Genel F, Arslanoglu S, Uran N, Saylan B. Sydenham’s chorea: clinical findings and comparison of the efficacies of sodium valproate and carbamazepine regimens. Brain Dev. 2002;24:73–76. https://www.ncbi.nlm.nih.gov/pubmed/11891095[↩]
- Harel L, Zecharia A, Straussberg R, Volovitz B, Amir J. Successful treatment of rheumatic chorea with carbamazepine. Pediatr Neurol. 2000;23:147–151.[↩]