Contents
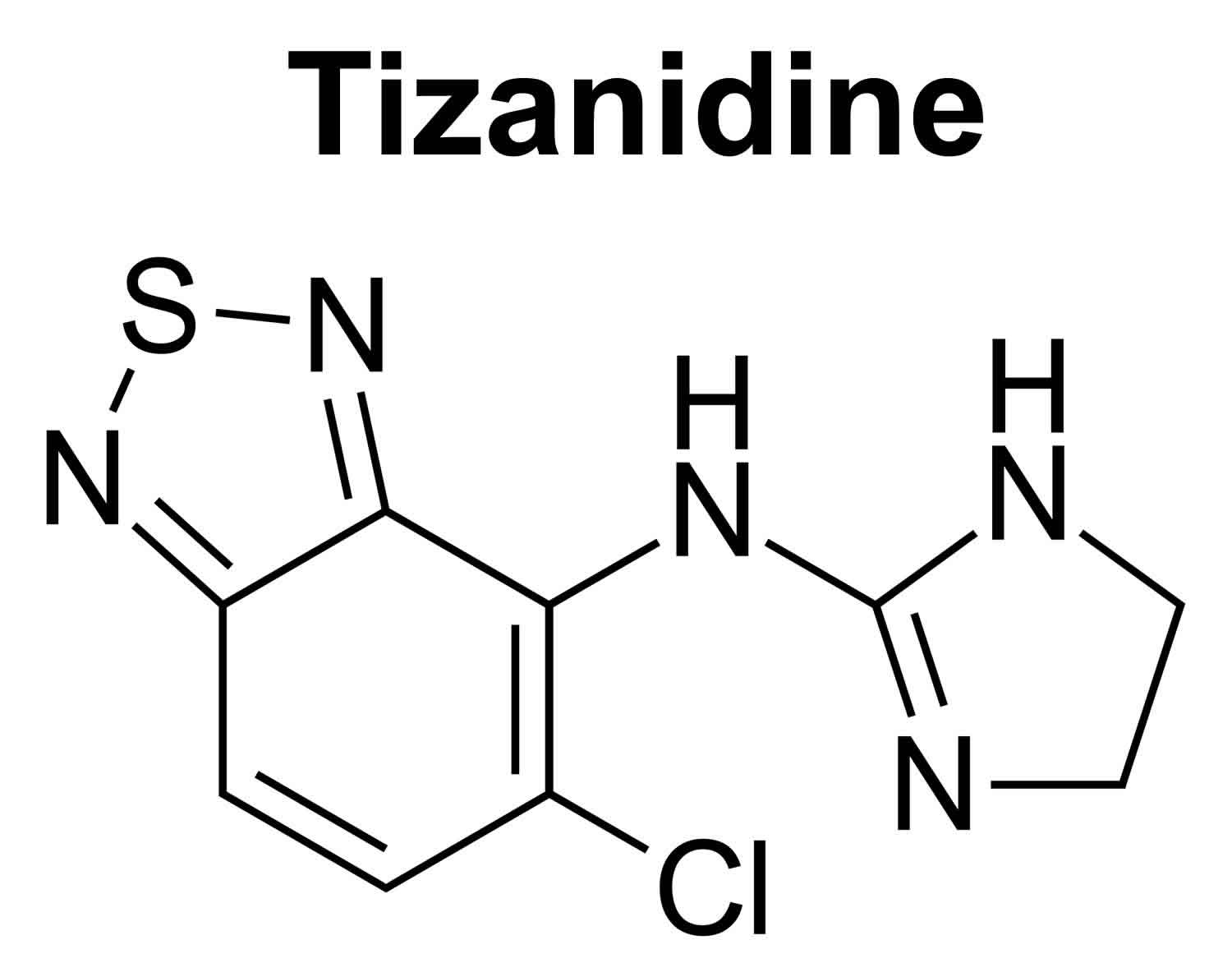
Tizanidine
Tizanidine is a short-acting muscle relaxant that is used to treat muscle spasms caused by multiple sclerosis (MS), stroke and brain or spinal injury 1, 2. Tizanidine has also been shown to be clinically effective in managing patients suffering from chronic neck and lumbosacral neuralgia with a myofascial component to their pain and regional musculoskeletal pain syndromes. It is also prescribed off-label for migraine headaches, insomnia, and as an anticonvulsant. Tizanidine can also be applied as part of a detoxification therapy regimen in patients exhibiting analgesic rebound headaches to assist with analgesic withdrawal. Tizanidine works by slowing action in your brain and nervous system to allow your muscles to relax. Tizanidine was approved for use in the United States in 1996 and currently several million prescriptions are filled yearly.
Tizanidine comes as a tablet and a capsule of 2, 4 or 6 mg to be takeb by mouth. Tizanidine is usually taken consistently either always with or always without food two or three times a day. Follow the directions on your prescription label carefully, and ask your doctor or pharmacist to explain any part you do not understand. Take tizanidine exactly as directed. Do not take more or less of it or take it more often than prescribed by your doctor.
Tizanidine capsules may be opened and sprinkled on soft foods such as applesauce. Talk to your doctor before opening the capsules because the effects of tizanidine when used in this manner may be different than when swallowing the capsule whole.
Tizanidine in the capsule is absorbed differently by the body than tizanidine in the tablet, so one product cannot be substituted for the other. Each time you have your prescription filled, look at the tablets or capsules in the bottle and make sure that you have received the right product. If you think you received the wrong medication, talk to your doctor or pharmacist right away.
Your doctor will probably start you on a low dose of tizanidine and gradually increase your dose, depending on your response to this medication. The recommended starting dose of tizanidine is 2 mg every 6 to 8 hours, up to a maximum of 3 doses in 24 hours. Do not take more than 36 mg of tizanidine in a 24-hour period 3. Too much of tizanidine can damage your liver and a few of which have been fatal 4. Cold or allergy medicine, narcotic pain medicine, sleeping pills, other muscle relaxers, and medicine for seizures, depression or anxiety can add to sleepiness caused by tizanidine.
Do not stop taking tizanidine without talking to your doctor. If you suddenly stop taking tizanidine, your heart may beat faster and you may have increased blood pressure or tightness in your muscles. Your doctor will probably decrease your dose gradually.
Tizanidine common side effects include tiredness, drowsiness, dizziness, muscular weakness, dry mouth and occasionally low blood pressure (hypotension).
How long does it take for tizanidine to work?
Tizanidine normally starts working 1 to 2 hours after taking it 5, 6. It wears off about 3 hours to 6 hours after taking it. Tizanidine can be taken up to 3 times a day to help relieve muscle spasms.
How long does tizanidine stay in my body?
Tizanidine is metabolized extensively in the liver by cytochrome P450-1A2 to inactive metabolites 1. Tizanidine has an elimination half-life of 2.5 hours, follows linear pharmacokinetics, and is excreted 60% through urine and 20% through feces 7. The maximum effect of tizanidine occurs about 1 hour to 2 hours after taking a dose. It wears off in about 3 to 6 hours after taking it. Tizanidine is typically given no more than 3 times a day. Tizanidine is usually taken up to 3 times in one day, starting at 2mg per dose. Allow 6 to 8 hours to pass between doses. Do not take more than 36 mg in a 24-hour period 3. Too much of tizanidine can damage your liver 4.
Does tizanidine lower my blood pressure?
Tizanidine is known to cause low blood pressure (hypotension). In some cases, it may cause low blood pressure that is so low that you could faint or pass out. The chances of fainting can be lowered if your doctor raises the dose of tizanidine very slowly. You may also have to be careful when you move from a sitting position to a standing position. In clinical trials, the most common side effects of tizanidine were dry mouth, sleepiness, dizziness and asthenia (defined as weakness, fatigue and/or tiredness).
Can I take tizanidine to help me sleep?
Tizanidine can cause sleepiness, but it has not been approved by the U.S. Food and Drug Administration (FDA) to treat sleep disorders. Tizanidine is a skeletal muscle relaxant. It is approved by the FDA to help relieve muscle spasms.
Sleep medicines called hypnotics can be prescribed by your doctor to help reduce the time it takes you to fall asleep. The most commonly used hypnotics are:
- Zolpidem (Ambien)
- Zaleplon (Sonata)
- Eszopiclone (Lunesta)
- Ramelteon (Rozerem)
How does tizanidine work?
Tizanidine is an imidazoline derivative and a centrally acting alpha-2 receptor agonist 1. Tizanidine inhibits the release of excitatory amino acids like glutamate and aspartate from spinal interneurons 1. Consequently, tizanidine enhances the presynaptic inhibition of motor neurons. Tizanidine has significant action on spinal polysynaptic pathways 8.
The overall effect of these actions is to reduce the facilitation of spinal motor neurons. Similarly, alpha-2 receptor-mediated inhibition of inter-neuronal activity appears to underlie tizanidine’s additional anti-nociceptive (pain reliever) and anti-convulsant activities. Spasm frequency and clonus are also reduced by tizanidine 9.
Tizanidine also has an affinity for the alpha-1 receptors but to a lesser degree, which may explain its mild and transitory effect on the cardiovascular system compared to clonidine despite their structural and biochemical similarity 10.
Tizanidine monitoring
- Creatinine and liver function tests require measurement at baseline, then one month after the maintenance dose is achieved. Periodically monitor liver function tests in patients managed with tizanidine chronically and in higher doses 4
- Monitor blood pressure and heart rate before increasing the dosage because of the risk of severe hypotension associated with the higher dose 11
- Monitor the level of spasticity by Ashworth and modified Ashworth Scales 12
- In patients with multiple sclerosis, monitor spasticity using Multiple Sclerosis Spasticity Scale (MSSS-88) 13
Tizanidine precautions
Tizanidine is a short-acting muscle relaxant that should be taken only for daily activities that require relief from muscle spasms. You should not take tizanidine if you are also taking fluvoxamine (Luvox) or ciprofloxacin (Cipro).
Do not use tizanidine at a time when you need muscle tone for safe balance and movement during certain activities. In some situations, tizanidine may endanger your physical safety to be in a state of reduced muscle tone.
Switching between tablets and capsules, or changing the way you take tizanidine with regard to eating can cause an increase in side effects or a decrease in therapeutic effect. Follow your doctor’s instructions carefully. After making any changes in how you take tizanidine, contact your doctor if you notice any change in how well the medicine works or if it causes increased side effects.
Avoid drinking alcohol. It can increase some of the side effects of tizanidine.
Before taking tizanidine:
- tell your doctor and pharmacist if you are allergic to tizanidine or any other medications.
- tell your doctor if you are taking ciprofloxacin (Cipro) or fluvoxamine. Your doctor will probably tell you not to take tizanidine if you are taking either of these medications.
- tell your doctor and pharmacist what other prescription and nonprescription medications, vitamins, nutritional supplements, and herbal products you are taking or plan to take. Be sure to mention any of the following: acyclovir (Zovirax); amiodarone (Cordarone, Pacerone); baclofen; cimetidine (Tagamet); clonidine (Catapres, Catapres-TTS); dantrolene (Dantrium); diazepam (Valium); famotidine (Pepcid, Pepcid AC); medications for anxiety, seizures, or high blood pressure; mexiletine (Mexitil); oral contraceptives (birth control pills); propafenone (Rythmol); fluoroquinolones such as gemifloxacin (Factive), levofloxacin (Levaquin), moxifloxacin (Avelox), norfloxacin (Noroxin), and ofloxacin (Floxin); ticlopidine (Ticlid); sedatives; sleeping pills; tranquilizers; verapamil (Calan, Covera, Isoptin, Verelan); and zileuton (Zyflo). Your doctor may need to change the doses of your medications or monitor you carefully for side effects. Other medications may also interact with tizanidine, so be sure to tell your doctor about all the medications you are taking, even those that do not appear on this list.
- tell your doctor if you have or have ever had kidney or liver disease.
- tell your doctor if you are pregnant, plan to become pregnant, or are breast-feeding. If you become pregnant while taking tizanidine, call your doctor.
- if you are having surgery, including dental surgery, tell the doctor or dentist that you are taking tizanidine.
- you should know that this medication may make you drowsy. Do not drive a car or operate machinery until you know how this medication affects you.
- remember that alcohol can add to the drowsiness caused by this medication.
- you should know that tizanidine may cause dizziness, lightheadedness, and fainting when you get up too quickly from a lying position. This is more common when you first start taking tizanidine. To avoid this problem, get out of bed slowly, resting your feet on the floor for a few minutes before standing up.tizanidine can decrease muscle tone, so be careful when walking or doing other activities where you rely on your muscle tone to help with your posture or balance.
Patients with Liver Impairment
The consequence of Hepatic impairment on the pharmacokinetics of tizanidine has not been evaluated. However, tizanidine is extensively metabolized by the liver; therefore, hepatic impairment would significantly influence the pharmacokinetics of tizanidine. Therefore, tizanidine should be avoided or used with extreme caution in patients with hepatic impairment.
Patients with Kidney Impairment
Tizanidine should be used cautiously in patients with renal impairment (creatinine clearance < 25 mL/min), as clearance is decreased by more than 50%. Start with a low dose, evaluate the response to therapy, and if a higher dose is needed, the dose can be increased rather than increasing the frequency of administration. The clinician should monitor the patient closely for adverse drug reactions such as dry mouth, drowsiness, asthenia, and dizziness as indicators of toxicity.
Pregnancy
The use of tizanidine in managing spinal cord injury during pregnancy has been described in case reports.[22] However, it is important to note that tizanidine is a former FDA pregnancy category-C and should be used only if indicated after careful risk-benefit evaluation.
Breastfeeding
Tizanidine is a lipid-soluble drug; hypothetically, it may be present in breast milk; its use during lactation is not advised.
Tizanidine drug interactions
Taking tizanidine with other drugs that make you sleepy or slow your breathing can cause dangerous side effects or death. Ask your doctor before using opioid medication, a sleeping pill, a muscle relaxer, or medicine for anxiety or seizures.
Tell your doctor about all your other medicines, especially:
- acyclovir
- ticlopidine
- zileuton
- birth control pills
- antibiotic – ciprofloxacin, levofloxacin, moxifloxacin, or ofloxacin
- blood pressure medicine – clonidine, guanfacine, methyldopa
- heart rhythm medicine – amiodarone, mexiletine, propafenone, verapamil
- stomach acid medicine – cimetidine, famotidine
This list is not complete. Other drugs may interact with tizanidine, including prescription and over-the-counter medicines, vitamins, and herbal products.
Tizanidine contraindications
- Hypersensitivity to tizanidine or its ingredients is a contraindication to the use of tizanidine.
- Tizanidine use requires caution in patients with hepatic impairment. Review articles on tizanidine report cases of severe hepatotoxicity, acute liver failure, and death 4
- According to product labeling, tizanidine use requires caution in patients with renal impairment (creatinine clearance < 25 mL/min). In such patients, decrease the dose. If high doses are necessary, increase the individual dosage rather than the dosage frequency.
Tizanidine uses
Tizanidine is an U.S. Food and Drug Administration (FDA) approved muscle relaxant caused by multiple sclerosis (MS), spinal cord injury, stroke, amyotrophic lateral sclerosis (ALS), and traumatic brain injury 1. Tizanidine has also been shown to be clinically effective in managing patients suffering from chronic neck and lumbosacral neuralgia with a myofascial component to their pain and regional musculoskeletal pain syndromes. It is also prescribed off-label for migraine headaches, insomnia, and as an anticonvulsant. Tizanidine can also be applied as part of a detoxification therapy regimen in patients exhibiting analgesic rebound headaches to assist with analgesic withdrawal.
Tizanidine FDA-approved uses:
- Tizanidine is indicated for spasticity management due to multiple sclerosis, spinal cord injury, stroke, amyotrophic lateral sclerosis, and traumatic brain injury 14, 15, 16.
Tizanidine Off-label Use:
- Chronic neck and lower back pain 17
- Rebound headaches due to analgesic withdrawal 18
- Chronic migraine headaches 19
- Refractory insomnia in spastic quadriplegic patients 20
- Regional musculoskeletal pain syndromes 17
Placebo-controlled studies confirm the significant efficacy of tizanidine in reducing muscle spasm in patients with spinal cord-induced spasticity. Medical literature suggests that patients with severe spasticity are more likely to benefit from tizanidine. Drug comparison studies have shown no differences in the efficacy of tizanidine compared with baclofen or diazepam. The tizanidine treatment group did not report increased weakness compared to the controls. Furthermore, patients given tizanidine experienced fewer side effects than those using controlled substances 21.
Studies exhibit that tizanidine, baclofen, and diazepam effectively decreased excessive muscle tone in patients with multiple sclerosis (MS) or cerebrovascular lesions. Muscle strength improved in all three treatment groups, but the improvement was most significant with tizanidine. Shakespeare et al. 22 also reported similar findings showing no differences in efficacy between tizanidine, baclofen, and dantrolene compared to diazepam. However, diazepam was associated with more sedation. Another study by Lataste et al. 23 showed no significant differences between tizanidine and baclofen or diazepam for muscle tone, muscle spasms, clonus, muscle strength, or overall anti-spastic effect. However, tizanidine tolerance is slightly better than diazepam and baclofen.
Groves et al. 24 report no significant differences between tizanidine, baclofen, or diazepam for spasticity by Ashworth score. However, applying global tolerability to treatment favored tizanidine compared to baclofen and diazepam.
A recent study indicates that tizanidine and other alpha-2 agonists can be used for medically supervised opioid withdrawal 25.
Animal studies have shown that tizanidine provides benefits in the perioperative period and the management of neuropathic pain, such as trigeminal neuralgia, similar to the effects of clonidine under similar circumstances.
The American Academy of Neurology guidelines reports that tizanidine should be used for generalized spasticity in cerebral palsy, for segmental/localized spasticity treatment with botulinum toxin-A is more effective 26.
Tizanidine dosage
Tizanidine is taken orally as 2 mg, 4 mg, and 6 mg capsules or as 2 mg and 4 mg tablets. Dosage starts with 2 mg orally and may repeat every 6 to 8 hours as needed. The dosage may gradually increase by 2 to 4 mg per dose for 1 to 4 days in between until there is a noticeably significant reduction of spasticity. Maximum dosing is three doses every 24 hours, up to 36 mg daily 3.
If tizanidine is used for more than nine weeks or given in high doses ranging from 20 mg to 36 mg daily, taper the dose gradually. The recommendation is to taper the dose to 2 to 4 mg daily to reduce the risk of tachycardia, rebound hypertension, and increased spasticity 27.
The patient may open the capsule and sprinkle the contents into food. Patients can take tizanidine with food or on an empty stomach. It is important to note that the extent of absorption is greater when taken with food. The tablet and capsule dosage forms are not bioequivalent when administered with food. Therefore, the clinician should counsel the patient to take tizanidine with or without food but be consistent to avoid fluctuations in concentration 28. It is also important to recognize that smoking decreases tizanidine’s plasma concentration and exposure 29.
What should I do if I forget a dose?
If your doctor has told you to take tizanidine regularly, take the missed dose as soon as you remember it. However, if it is almost time for your next dose, skip the missed dose and continue your regular dosing schedule. Do not take a double dose to make up for a missed one.
Tizanidine side effects
Tizanidine may cause side effects. Tell your doctor if any of these symptoms are severe or do not go away:
- dizziness 30
- drowsiness
- blurred vision
- weakness
- nervousness
- depression
- hallucination
- vomiting
- tingling sensation in the arms, legs, hands, and feet
- dry mouth (xerostomia) 9
- rhinitis (runny nose)
- constipation
- diarrhea
- stomach pain
- heartburn
- increased muscle spasms
- back pain
- rash
- sweating
Some tizanidine side effects can be serious. If you experience any of these symptoms, call your doctor immediately:
- nausea
- extreme tiredness
- unusual bleeding or bruising
- lack of energy
- loss of appetite
- pain in the upper right part of the stomach
- yellowing of the skin or eyes
- unexplained flu-like symptoms
- seeing things or hearing voices that do not exist
- slow heartbeat
- changes in vision
Tizanidine may cause other side effects. Call your doctor if you have any unusual problems while taking tizanidine.
Tizanidine withdrawal symptoms:
- rapid heart rate (tachycardia)
- rebound hypertension
- increased spasticity
- withdrawal symptoms are more likely to occur when discontinuing tizanidine abruptly 27
Symptoms of tizanidine overdose may include:
- drowsiness
- extreme tiredness
- confusion
- slow heartbeat (bradycardia)
- fainting
- dizziness
- slow or shallow breathing
- loss of consciousness
- coma
Management of tizanidine overdose
- There is no antidote for tizanidine toxicity.
- Tizanidine overdose management is by close monitoring of airways, administration of intravenous fluid, and vasopressors as necessary 11
- The pediatric case report described an overdose of tizanidine in spastic quadriplegia and toxicity presented with multiple organ dysfunction in the absence of sepsis 3
- A recent case report described the altered mental status and hemodynamic instability due to tizanidine overdose, and naloxone 10 mg IV administration improved Richmond Agitation-Sedation Scale (RASS) 31. The study concluded that naloxone could be used in tizanidine overdose in emergency settings; however, naloxone does not reverse the hemodynamic parameters 32.
- Ghanavatian S, Derian A. Tizanidine. [Updated 2023 Aug 28]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK519505[↩][↩][↩][↩][↩]
- Tizanidine Hydrochloride (Zanaflex®). https://www.accessdata.fda.gov/drugsatfda_docs/label/2013/021447s011_020397s026lbl.pdf[↩]
- Vila J, Morgenstern A, Vendrell L, Ortega J, Danés I. Liver, Renal, and Cardiovascular Failure After Unintentional Overdose of Tizanidine in a 2-Year-Old Child. J Pediatr Pharmacol Ther. 2021;26(6):643-646. doi: 10.5863/1551-6776-26.6.643[↩][↩][↩][↩]
- LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Tizanidine. [Updated 2017 Jan 30]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK548048[↩][↩][↩][↩]
- Emre M, Leslie GC, Muir C, Part NJ, Pokorny R, Roberts RC. Correlations between dose, plasma concentrations, and antispastic action of tizanidine (Sirdalud). J Neurol Neurosurg Psychiatry. 1994 Nov;57(11):1355-9. doi: 10.1136/jnnp.57.11.1355[↩]
- Henney HR 3rd, Runyan JD. A clinically relevant review of tizanidine hydrochloride dose relationships to pharmacokinetics, drug safety and effectiveness in healthy subjects and patients. Int J Clin Pract. 2008 Feb;62(2):314-24. doi: 10.1111/j.1742-1241.2007.01660.x[↩]
- Granfors MT, Backman JT, Laitila J, Neuvonen PJ. Tizanidine is mainly metabolized by cytochrome p450 1A2 in vitro. Br J Clin Pharmacol. 2004 Mar;57(3):349-53. doi: 10.1046/j.1365-2125.2003.02028.x[↩]
- Fuchigami T, Kakinohana O, Hefferan MP, Lukacova N, Marsala S, Platoshyn O, Sugahara K, Yaksh TL, Marsala M. Potent suppression of stretch reflex activity after systemic or spinal delivery of tizanidine in rats with spinal ischemia-induced chronic spastic paraplegia. Neuroscience. 2011 Oct 27;194:160-9. doi: 10.1016/j.neuroscience.2011.08.022[↩]
- Wagstaff AJ, Bryson HM. Tizanidine. A review of its pharmacology, clinical efficacy and tolerability in the management of spasticity associated with cerebral and spinal disorders. Drugs. 1997 Mar;53(3):435-52. doi: 10.2165/00003495-199753030-00007[↩][↩]
- Coward DM. Tizanidine: neuropharmacology and mechanism of action. Neurology. 1994 Nov;44(11 Suppl 9):S6-10; discussion S10-1.[↩]
- Spiller HA, Bosse GM, Adamson LA. Retrospective review of Tizanidine (Zanaflex) overdose. J Toxicol Clin Toxicol. 2004;42(5):593-6. doi: 10.1081/clt-200026978[↩][↩]
- Meseguer-Henarejos AB, Sánchez-Meca J, López-Pina JA, Carles-Hernández R. Inter- and intra-rater reliability of the Modified Ashworth Scale: a systematic review and meta-analysis. Eur J Phys Rehabil Med. 2018 Aug;54(4):576-590. doi: 10.23736/S1973-9087.17.04796-7[↩]
- Hui D, Argáez C. Onabotulinum Toxin A (Botox) for Spasticity Associated With Multiple Sclerosis [Internet]. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health; 2021 Mar. Available from: https://www.ncbi.nlm.nih.gov/books/NBK571918[↩]
- Kamen L, Henney HR 3rd, Runyan JD. A practical overview of tizanidine use for spasticity secondary to multiple sclerosis, stroke, and spinal cord injury. Curr Med Res Opin. 2008 Feb;24(2):425-39. doi: 10.1185/030079908×261113[↩]
- Dorst J, Ludolph AC, Huebers A. Disease-modifying and symptomatic treatment of amyotrophic lateral sclerosis. Ther Adv Neurol Disord. 2017 Oct 9;11:1756285617734734. doi: 10.1177/1756285617734734[↩]
- Chang E, Ghosh N, Yanni D, Lee S, Alexandru D, Mozaffar T. A Review of Spasticity Treatments: Pharmacological and Interventional Approaches. Crit Rev Phys Rehabil Med. 2013;25(1-2):11-22. doi: 10.1615/CritRevPhysRehabilMed.2013007945[↩]
- Malanga G, Reiter RD, Garay E. Update on tizanidine for muscle spasticity and emerging indications. Expert Opin Pharmacother. 2008 Aug;9(12):2209-15. doi: 10.1517/14656566.9.12.2209[↩][↩]
- Yancey JR, Sheridan R, Koren KG. Chronic daily headache: diagnosis and management. Am Fam Physician. 2014 Apr 15;89(8):642-8. https://www.aafp.org/pubs/afp/issues/2014/0415/p642.html[↩]
- Smith TR. Low-dose tizanidine with nonsteroidal anti-inflammatory drugs for detoxification from analgesic rebound headache. Headache. 2002 Mar;42(3):175-7. doi: 10.1046/j.1526-4610.2002.02048.x[↩]
- Tanaka H, Fukuda I, Miyamoto A, Oka R, Cho K, Fujieda K. [Effects of tizanidine for refractory sleep disturbance in disabled children with spastic quadriplegia]. No To Hattatsu. 2004 Nov;36(6):455-60. Japanese.[↩]
- Wallace JD. Summary of combined clinical analysis of controlled clinical trials with tizanidine. Neurology. 1994 Nov;44(11 Suppl 9):S60-8; discussion S68-9.[↩]
- Shakespeare D, Boggild M, Young CA. Anti‐spasticity agents for multiple sclerosis. Cochrane Database of Systematic Reviews 2003, Issue 4. Art. No.: CD001332. DOI: 10.1002/14651858.CD001332[↩]
- Lataste X, Emre M, Davis C, Groves L. Comparative profile of tizanidine in the management of spasticity. Neurology. 1994 Nov;44(11 Suppl 9):S53-9.[↩]
- Groves L, Shellenberger MK, Davis CS. Tizanidine treatment of spasticity: a meta-analysis of controlled, double-blind, comparative studies with baclofen and diazepam. Adv Ther. 1998 Jul-Aug;15(4):241-51.[↩]
- Srivastava AB, Mariani JJ, Levin FR. New directions in the treatment of opioid withdrawal. Lancet. 2020 Jun 20;395(10241):1938-1948. doi: 10.1016/S0140-6736(20)30852-7[↩]
- Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society; Delgado MR, Hirtz D, Aisen M, Ashwal S, Fehlings DL, McLaughlin J, Morrison LA, Shrader MW, Tilton A, Vargus-Adams J. Practice parameter: pharmacologic treatment of spasticity in children and adolescents with cerebral palsy (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society. Neurology. 2010 Jan 26;74(4):336-43. doi: 10.1212/WNL.0b013e3181cbcd2f[↩]
- Suárez-Lledó A, Padullés A, Lozano T, Cobo-Sacristán S, Colls M, Jódar R. Management of Tizanidine Withdrawal Syndrome: A Case Report. Clin Med Insights Case Rep. 2018 Feb 13;11:1179547618758022. doi: 10.1177/1179547618758022[↩][↩]
- Henney HR 3rd, Shah J. Relative bioavailability of tizanidine 4-mg capsule and tablet formulations after a standardized high-fat meal: a single-dose, randomized, open-label, crossover study in healthy subjects. Clin Ther. 2007 Apr;29(4):661-9. doi: 10.1016/j.clinthera.2007.04.012[↩]
- Al-Ghazawi M, Alzoubi M, Faidi B. Pharmacokinetic comparison of two 4 mg tablet formulations of tizanidine. Int J Clin Pharmacol Ther. 2013 Mar;51(3):255-62. doi: 10.5414/CP201790[↩]
- Saper JR, Lake AE 3rd, Cantrell DT, Winner PK, White JR. Chronic daily headache prophylaxis with tizanidine: a double-blind, placebo-controlled, multicenter outcome study. Headache. 2002 Jun;42(6):470-82. doi: 10.1046/j.1526-4610.2002.02122.x[↩]
- Kerson AG, DeMaria R, Mauer E, Joyce C, Gerber LM, Greenwald BM, Silver G, Traube C. Validity of the Richmond Agitation-Sedation Scale (RASS) in critically ill children. J Intensive Care. 2016 Oct 26;4:65. doi: 10.1186/s40560-016-0189-5[↩]
- Adams A, Copley C. High dose naloxone for acute tizanidine overdose in the emergency department: a case report. Clin Toxicol (Phila). 2021 Aug;59(8):764-765. doi: 10.1080/15563650.2020.1863977[↩]