Contents
- Non-seminoma testicular cancer
- Testicular cancer
- Germ cell tumor
- Difference between seminoma and non-seminoma
- Non-seminoma testicular cancer causes
- Non-seminoma testicular cancer symptoms
- Non-seminoma testicular cancer diagnosis
- Non-seminoma testicular cancer treatment
- Non-seminoma testicular cancer prognosis
- Non-seminoma testicular cancer survival rate
Non-seminoma testicular cancer
Nonseminoma is a type of cancer that begins in germ cells also called primordial germ cells that migrate during embryogenesis from the yolk sac through the mesentery to the gonads (testicles and ovaries) (see Figure 3) 1, 2. Most testicular cancers start in germ cells and are called germ cell tumors. “Germ” is short for “germinate”, which means to mature. For women germ cells eventually mature into eggs. For men germ cells mature into sperm. Germ cell tumors can generally be divided into gonadal (testicles and ovaries) and extragonadal (outside of the gonads). Germ cell tumors most often form where eggs get made (ovaries) and where sperm gets made (testicles). Less commonly, germ cell tumors form in body parts other than your ovaries and testicles, including your abdomen, chest, low back, tailbone and brain. These tumors are called extragonadal tumors.
There are different types of testicular cancer. Most testicular cancers are a type called germ cell tumors. The 2 main types of testicular germ cell tumors are:
- Seminomas. Around 55 – 60 out of every 100 testicular cancers (around 55 – 60%) are pure seminomas.
- Non-seminomas. Most other testicular germ cell tumors are non-seminomas. Non-seminomas develop from different types of cells.
- Some testicular tumors have both seminoma cells and non-seminoma cells. Doctors usually treat these in the same way as non-seminomas.
- There are also some rarer types of testicular cancer.
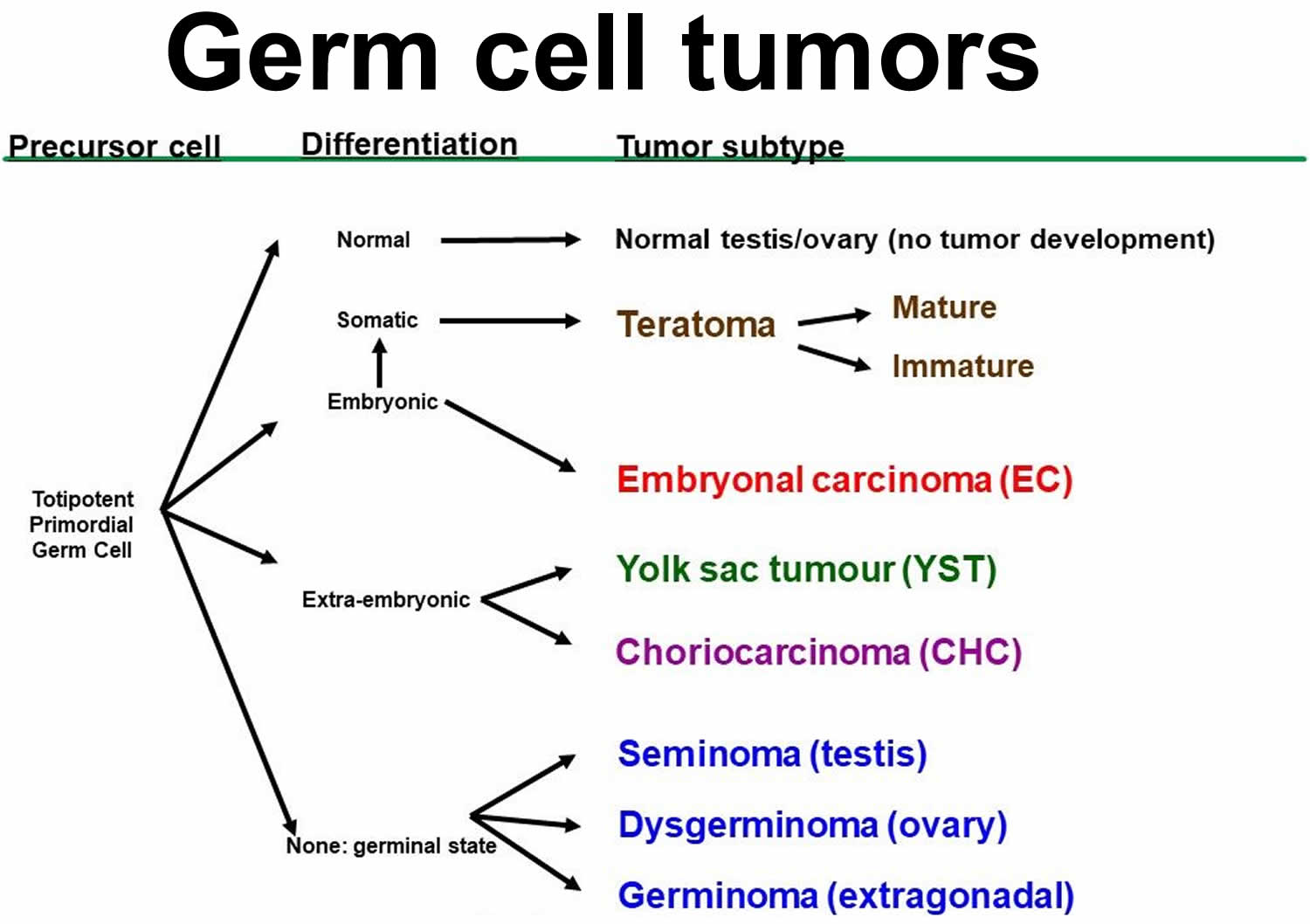
Non-seminomatous germ cell tumors are further classified into four types 3:
- Embryonal carcinoma. These cells are found in about 40% of testicular tumors, but pure embryonal carcinomas occur only 3% to 4% of the time. When seen under a microscope, embryonal carcinoma can look like tissues of very early embryos. This type of non-seminoma tends to grow rapidly and spread outside the testicle. Embryonal carcinoma can increase blood levels of a tumor marker protein called alpha-fetoprotein (AFP), as well as human chorionic gonadotropin (hCG).
- Yolk sac tumor. Yolk sac carcinoma are so named because their cells look like the yolk sac of an early human embryo. Other names for this cancer include yolk sac tumor, endodermal sinus tumor, infantile embryonal carcinoma, or orchidoblastoma. Yolk sac carcinoma is the most common form of testicular cancer in children (especially in infants), but pure yolk sac carcinomas (tumors that do not have other types of non-seminoma cells in them) are rare in adults. When they occur in children, these tumors usually are treated successfully. But they’re of more concern when they occur in adults, especially if they are pure. Yolk sac carcinomas respond very well to chemotherapy , even if they have spread. Yolk sac carcinoma almost always increases blood levels of alpha-fetoprotein (AFP).
- Choriocarcinoma. Choriocarcinoma is a very rare and fast-growing type of testicular cancer in adults. Pure choriocarcinoma is likely to spread rapidly to other parts of the body, including the lungs, bones, and brain. More often, choriocarcinoma cells are seen with other types of non-seminoma cells in a mixed germ cell tumor. These mixed tumors tend to have a somewhat better outlook than pure choriocarcinomas, although the presence of choriocarcinoma is always a worrisome finding. Choriocarcinoma increases blood levels of human chorionic gonadotropin (hCG).
- Teratoma. Teratomas are germ cell tumors with areas that, under a microscope, look like each of the 3 layers of a developing embryo: the endoderm (innermost layer), mesoderm (middle layer), and ectoderm (outer layer). Pure teratomas of the testicles are rare and do not increase alpha-fetoprotein (AFP) or human chorionic gonadotropin (hCG) levels. Most often, teratomas are seen as parts of mixed germ cell tumors. There are 3 main types of teratomas:
- Mature teratomas are tumors formed by cells a lot like the cells of adult tissues. They rarely spread. They can usually be cured with surgery, but some come back (recur) after treatment.
- Immature teratomas are less well-developed cancers with cells that look like those of an early embryo. This type is more likely than a mature teratoma to grow into (invade) nearby tissues, spread (metastasize) outside the testicle, and come back (recur) years after treatment.
- Teratomas with somatic type malignancy are very rare. These cancers have some areas that look like mature teratomas but have other areas where the cells have become a type of cancer that normally develops outside the testicle (such as a sarcoma, adenocarcinoma, or even leukemia).
Nonseminoma tumors are usually made up of more than one type of cancer cell. For example, you might have a mix of some teratoma cells and some embryonal carcinoma cells. However, it’s also possible to have pure teratomas. Although nonseminomas occur most often in the testicles or ovaries, they can occur in other tissues, such as the brain, chest, or abdomen. This happens when germ cells (primordial germ cells) that have the ability to form sperm or eggs are found in other parts of the body. These non seminoma types are all treated in the same way.
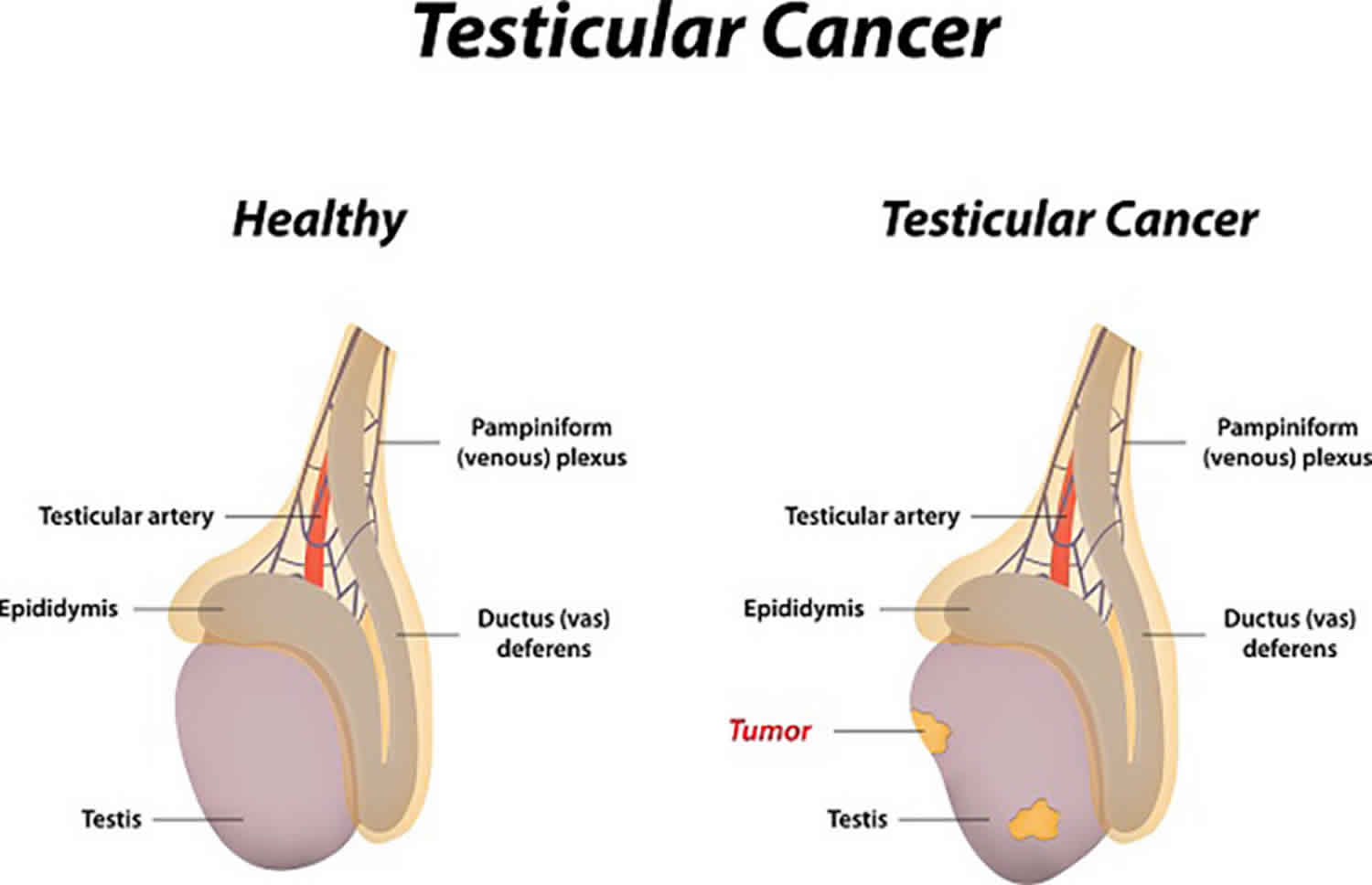
The classic presentation of a testicular cancer is as a painless testicular lump in an otherwise healthy man in the third or fourth decade of life. The presentation can vary depending on the amount of disease, clinical stage, and the presence of cancer spread (metastases). Roughly one third of patients diagnosed with non seminoma testicular cancer will present with disseminated disease 4.
Non-seminoma testicular cancer is the most likely testicular cancer to cause metastases which typically affect the lungs, liver, central nervous system, and bone in order of frequency 5. Roughly one-third of patients with non-seminoma testicular cancer will have disseminated or metastatic disease at the time of initial presentation and diagnosis 5.
Early diagnosis and immediate treatment are important since non seminoma testicular cancer has excellent cure rates due to its extreme sensitivity to cisplatin-based chemotherapy and radiation when properly combined with orchiectomy or retroperitoneal lymph node dissection.
Radical inguinal orchiectomy is the gold standard in management of suspected testicular cancers, as it has both diagnostic and therapeutic benefits. Radical orchiectomy will allow exact histopathological diagnoses and staging of the tumor. Trans-scrotal biopsy or a scrotal approach to orchiectomy should never be performed in cases of suspected testicular cancer, as it can cause contamination of the scrotum and alter patterns of lymphatic spread of cancer as well as complicate subsequent management. Radical inguinal orchiectomy surgery involves removing the testicle and spermatic cord where it exits the body to identify and likely treat the majority of cancers localized to the testis. Trans scrotal orchiectomy or biopsy of the testicular mass is contraindicated for the risk of tumor seeding of the lymphatic drainage. Spinal or general anesthesia may be used. An inguinal incision is made to expose the external and internal iliac canal. External iliac fascia is opened, revealing the spermatic cord. The spermatic cord is controlled to stop retroperitoneal lymphatic and venous drainage of tumor cells. After that, deliver the testis from the scrotum and ligate vas deferens and spermatic arteries separately. Retroperitoneal lymph node dissection is done if necessary. Reapproximate the external oblique fascia and close the skin in standard fashion. Conduct a follow-up study by staging and referring the patient for appropriate adjuvant therapies.
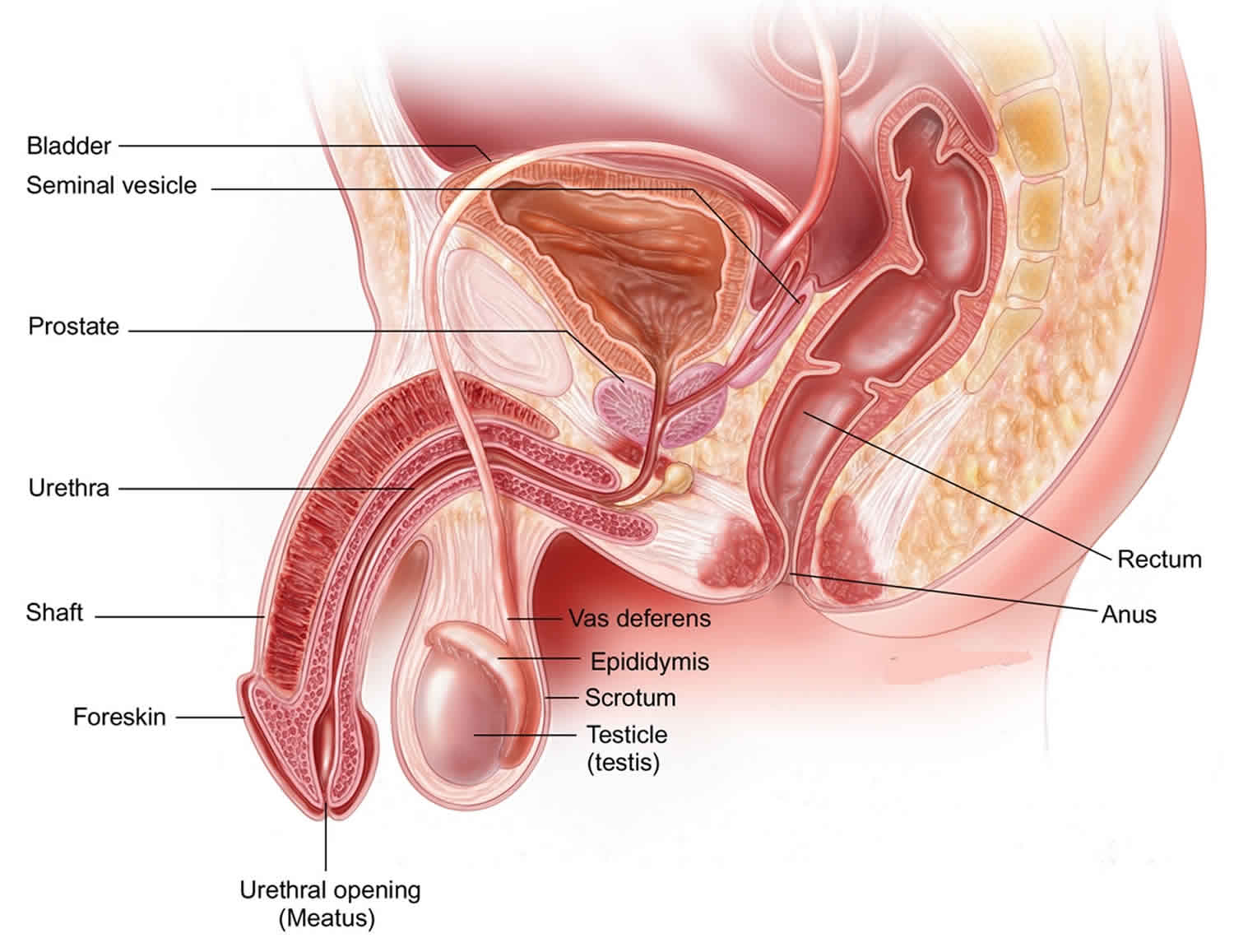
Figure 1. Male reproductive system
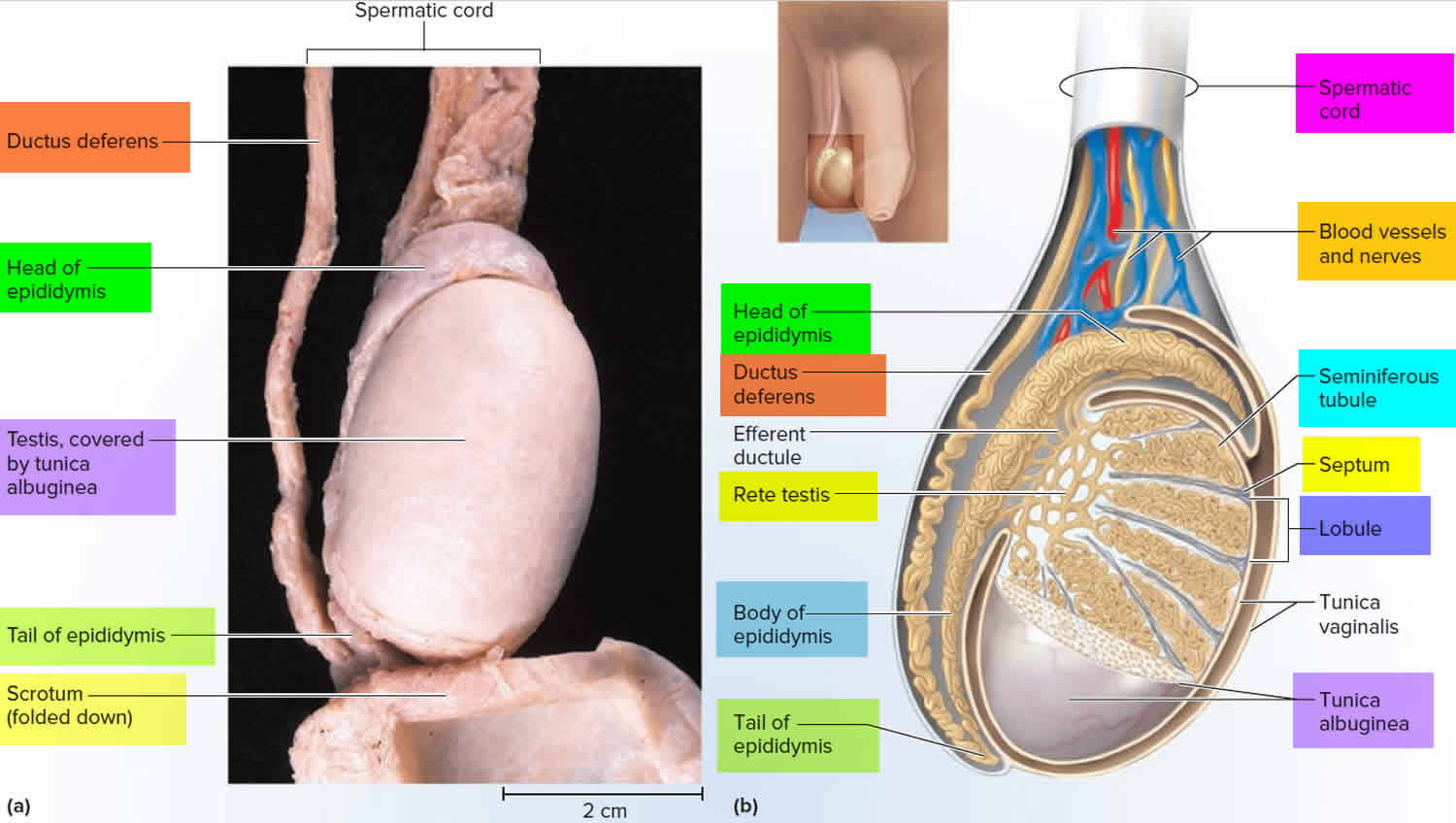
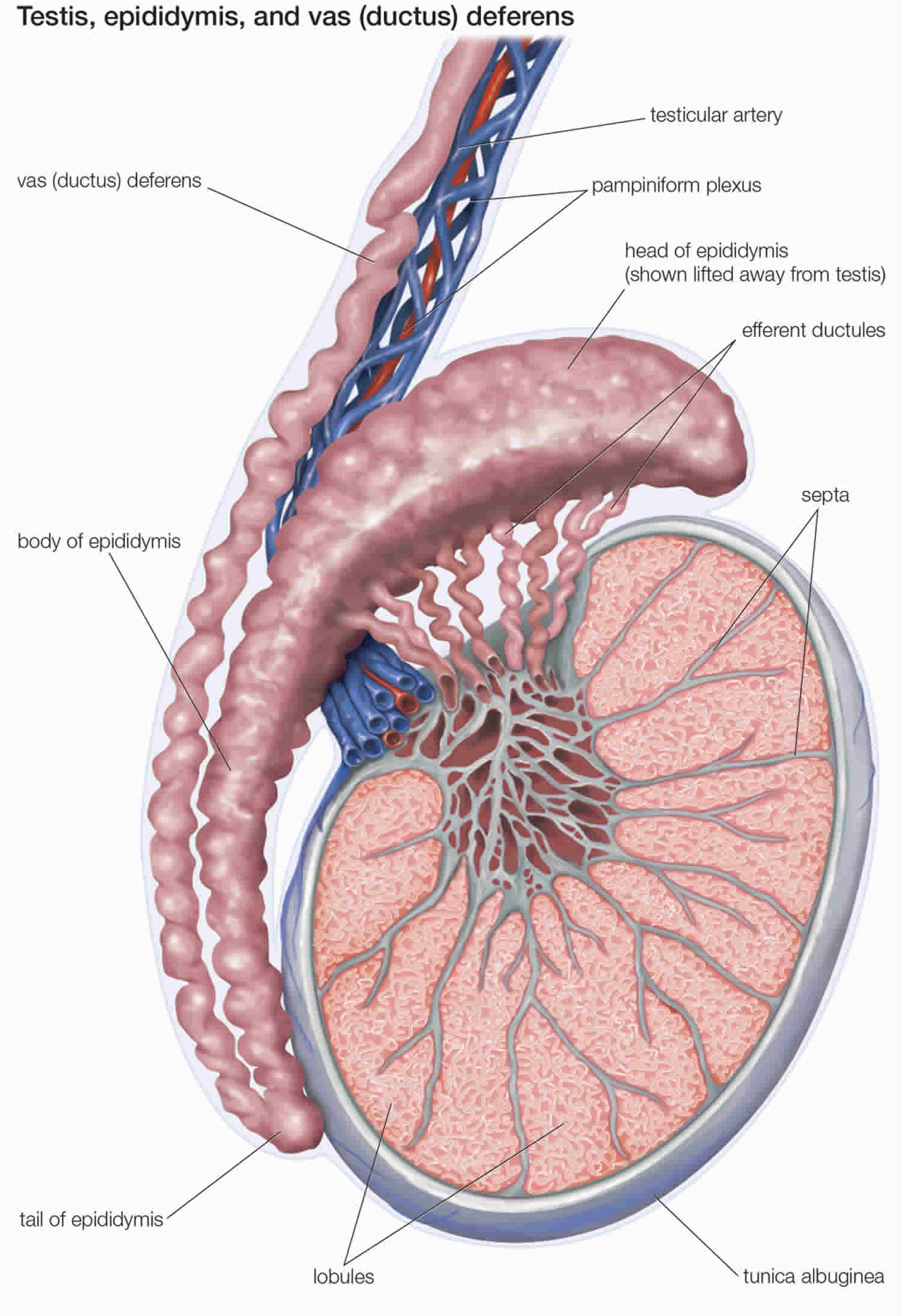
Figure 2. Testicle anatomy
Choriocarcinoma
Testicular choriocarcinoma is a very rare and very aggressive fast-growing testicular cancer in adults that is typically seen with extremely highly elevated serum human chorionic gonadotropin (hCG) levels and metastatic disease 5. Pure choriocarcinoma is likely to spread rapidly to other parts of the body, including the lungs, bones, and brain. More often, choriocarcinoma cells are seen with other types of non-seminoma cells in a mixed germ cell tumor. These mixed tumors tend to have a somewhat better outlook than pure choriocarcinomas, although the presence of choriocarcinoma is always a worrisome finding.
Pure choriocarcinomas are rare at only 1% of all testicular cancers, but they may be found as a component in up to 15% of all mixed germ cell tumors 6. Choriocarcinomas are typically poor-risk (stage 3C) at the time of diagnosis with high serum human chorionic gonadotropin (hCG) levels and non-pulmonary organ metastases 7. Unlike other non-seminomatous germ cell tumors, choriocarcinoma can spread via the blood with common sites of metastases, including the lungs, liver, and brain 7, 8.
Microscopically, testicular choriocarcinoma tumor is composed of both syncytiotrophoblasts and cytotrophoblasts, with the former staining positively for human chorionic gonadotropin (hCG) 5. The human chorionic gonadotropin (hCG) levels may become quite high, resulting in male gynecomastia (an increase in the amount of breast tissue caused by a hormone imbalance in boys or men). Testicular choriocarcinomas are highly vascular and prone to bleed, often spontaneously and sometimes immediately after chemotherapy is initiated. This hemorrhaging can be catastrophic, especially when the bleeding is significant and occurs in the lungs or brain 5. Elevated human chorionic gonadotropin (hCG) levels are usually associated only with seminomas, except for choriocarcinoma and sometimes embryonal carcinoma 5.
Embryonal Carcinoma
Embryonal carcinoma are found in about 40% of testicular tumors (about 40% of all mixed germ cell testicular cancers), but pure embryonal carcinomas occur only 3% to 4% of the time. When seen under a microscope, embryonal carcinoma can look like tissues of very early embryos. Embryonal carcinoma consists of undifferentiated malignant cells resembling primitive epithelial cells from early-stage embryos with crowded pleomorphic nuclei 9. Grossly, embryonal carcinoma is a tan to a yellow neoplasm that often exhibits large areas of hemorrhage and necrosis. The microscopic appearance of these tumors varies considerably as they may grow in solid sheets or papillary, glandular-alveolar, or tubular patterns.
Embryonal carcinoma is relatively aggressive and tends to grow rapidly and spread outside the testicle. Embryonal carcinoma can increase blood levels of a tumor marker protein called alpha-fetoprotein (AFP), as well as human chorionic gonadotropin (hCG).
Teratoma
Teratomas are germ cell tumors with areas that, under a microscope, look like each of the 3 layers of a developing embryo: the endoderm (innermost layer), mesoderm (middle layer), and ectoderm (outer layer). Teratomas contain well or poorly differentiated elements of at least two of the three germ cell layers: endoderm, mesoderm, and ectoderm. All components derived from these three germ layers are characteristically intermixed. Pure teratomas of the testicles are rare and do not increase alpha-fetoprotein (AFP) or human chorionic gonadotropin (hCG) levels.
Most often, teratomas are seen as parts of mixed germ cell tumors. There are 3 main types of teratomas:
- Mature teratomas are tumors formed by cells a lot like the cells of adult tissues. They rarely spread. They can usually be cured with surgery, but some come back (recur) after treatment.
- Immature teratomas are less well-developed cancers with cells that look like those of an early embryo. This type is more likely than a mature teratoma to grow into (invade) nearby tissues, spread (metastasize) outside the testicle, and come back (recur) years after treatment.
- Teratomas with somatic type malignancy are very rare. These cancers have some areas that look like mature teratomas but have other areas where the cells have become a type of cancer that normally develops outside the testicle (such as a sarcoma, adenocarcinoma, or even leukemia).
Mature teratomas are defined as well-differentiated tumors, whereas those that are poorly differentiated are called immature teratomas. In adolescent and adult men, histopathologists do not distinguish between the two entities, and its distinction is clinically insignificant 9.
Mature teratomas may include mature bone, cartilage, teeth, hair, and squamous epithelium. Due to these findings, teratomas are roughly called “monster tumors” in Greek. The gross appearance largely depends on the elements within it. Most of these tumors have two physical components, i.e., solid and cystic areas. Mature teratomas are typically associated with normal serum tumor markers (LDH, alpha-fetoprotein (AFP), and human chorionic gonadotropin (HCG)), but they may cause slightly elevated serum AFP levels 5. Approximately 47% of adult mixed germ cell tumors contain teratomatous elements, but pure teratomas are uncommon 5. Mature teratoma often constitutes the remnant of non-seminomatous germ cell tumors after treatment with chemo or radiation therapy, as they are relatively resistant to both. Treatment often requires surgery 5.
Yolk Sac Tumor
Yolk sac tumor are so named because their cells look like the yolk sac of an early human embryo. Pure yolk sac tumors sometimes called yolk sac carcinoma, endodermal sinus tumor, infantile embryonal carcinoma, or orchidoblastoma. Pure yolk sac carcinomas (tumors that do not have other types of non-seminoma cells in them) are rare in adults and constitute a very small percentage of adult primary testicular and retroperitoneal germ cell tumors. Yolk sac carcinoma is the most common form of testicular cancer in children (especially in infants). Yolk sac tumors are the most common testicular cancer in male children 3 years of age and younger, comprising about 30% of all testicular cancers in this age group 9. The median age at diagnosis is 18 months. Yolk sac carcinoma almost always increases blood levels of alpha-fetoprotein (AFP).
Yolk sac tumors typically grow in a glandular, papillary, or micro-cystic pattern and often make alpha-fetoprotein (AFP). Children usually have pure yolk sac tumors, while in adults, it is found as part of a mixed germ cell cancer. Most yolk sac tumors, up to 75%, will demonstrate Schiller-Duvall bodies which resemble glomeruli with a fibro-vascular core. If present, these are pathognomic for yolk sac tumors.
When yolk sac tumors occur in children, these tumors usually are treated successfully. Prognosis is usually good with treatment, especially in children 10. But they’re of more concern when they occur in adults, especially if they are pure yolk sac carcinomas (tumors that do not have other types of non-seminoma cells in them). Yolk sac carcinomas respond very well to chemotherapy , even if they have spread.
Testicular cancer
Testicular cancer is a growth of cancerous cells that starts in the testicles. The testicles also called testes, are in the scrotum. The scrotum is a loose bag of skin underneath the penis. The testicles make sperm and the hormone testosterone.
There are 3 main types of primary testicular tumor:
- Germ cell tumors. Almost all testicular cancers start in the germ cells. Germ cell tumors are classified histologically into 2 broad classes: Seminomas and Non-seminomas. These 2 types grow and spread differently and are treated differently. Around 55 – 60 out of every 100 testicular cancers (around 55 – 60%) are pure seminomas. Seminoma testicular cancers tend to happen at an older age with a peak incidence in men aged 30 to 34 years. Seminomas often grow and spread (metastasize) more slowly than nonseminomas testicular cancer 11, 12. Nonseminoma testicular cancers tend to grow and spread more quickly than seminomas. Seminomas are more sensitive to radiation. Seminomas generally have a good prognosis, while non-seminomatous tumors are much more likely to present with metastatic disease as well as mixed germ cell tumors comprising of two or more than two different germ cell tumor components. A testicular tumor that contains both seminoma and nonseminoma cells is treated as a nonseminoma.
- Sex cord-stroma tumors also called gonadal stromal tumors. Sex cord-stroma tumors are tumors that develop from the supportive tissues around the germ cells in the testicle. These tumors are rare, making up less than 5 percent of testis cancers, and have an excellent prognosis if surgically resected. There are two types of stromal tumors:
- Leydig cell tumors: Leydig cells make the male hormone testosterone. Leydig cell tumors can develop in both adults and children. These tumors often make androgens (male hormones), but sometimes they make estrogens (female sex hormones). Most Leydig cell tumors are not cancer (benign). Leydig cell tumors seldom spread beyond the testicle and are most often cured with surgery. Still, a small number of Leydig cell tumors do spread to other parts of the body. These tend to have a poor outlook because they usually don’t respond well to chemo or radiation therapy.
- Sertoli cell tumors: Sertoli cell tumors start in normal Sertoli cells, which support and nourish the sperm-making germ cells. Like the Leydig cell tumors, sertoli cell tumors are usually benign. But if they spread, they usually don’t respond well to chemo or radiation therapy.
- Secondary testicular cancers. Cancers that start in another organ and then spread (metastasize) to the testicle are called secondary testicular cancers. These are not true testicular cancers – they don’t start in the testicles. They’re named and treated based on where they started. Lymphoma is the most common secondary testicular cancer. Testicular lymphoma is more common in men older than 50 than primary testicular tumors. The outlook depends on the type and stage of lymphoma. The usual treatment is surgical removal, followed by radiation and/or chemotherapy. In boys with acute leukemia, the leukemia cells can sometimes form a tumor in the testicle. Along with chemotherapy to treat the leukemia, this might require treatment with radiation or surgery to remove the testicle. Cancers of the prostate, lung, skin (melanoma), kidney, and other organs also can spread to the testicles. The prognosis for these cancers tends to be poor because these cancers have usually spread widely to other organs as well. Treatment depends on the specific type of cancer.
Testicular cancer is a rare type of cancer but it’s one of the most common cancers in young men. Testicular cancer can happen at any age, but it happens most often in men between the ages of 15 and 35. It is estimated that up to 10,000 American men will develop testicular cancer each year.
Not all men with testicular cancer experience symptoms, but many testicular cancers present as a small bump or lump on a testicle.
Testicular cancer can grow quickly. They often spread outside the testicle to other parts of the body.
Fortunately, testicular cancer is highly treatable, even when it spreads to other parts of the body. Treatments depend on the type of testicular cancer that you have and how far it has spread. Most testicular cancers are germ cell tumors. For treatment planning, germ cell tumors are broadly divided into seminomas and nonseminomas because they have different prognosis and treatment algorithms. For patients with seminomas (all stages combined), the cure rate exceeds 90%. For patients with low-stage seminomas or nonseminomas, the cure rate approaches 100% 13, 14, 15, 16, 17.
Germ cell tumor
Germ cell tumors are tumors derived from a mass made of reproductive cells, also called primordial germ cells that migrate during embryogenesis from the yolk sac through the mesentery to the gonads (testicles and ovaries) (see Figure 3) 1, 2. “Germ” is short for “germinate”, which means to mature. For women germ cells eventually mature into eggs. For men germ cells mature into sperm. Germ cell tumors can generally be divided into gonadal (testicles and ovaries) and extragonadal (outside of the gonads). Germ cell tumors most often form where eggs get made (ovaries) and where sperm gets made (testicles). Less commonly, germ cell tumors form in body parts other than your ovaries and testicles, including your abdomen, chest, low back, tailbone and brain. These tumors are called extragonadal tumors.
Germ cell tumors may be malignant (cancerous) or benign (noncancerous). Both types of tumors can grow larger, but only cancerous (malignant) germ cell tumors can spread to other parts of your body. Cancer that’s spread (metastasized) can damage your organs and is more difficult to treat.
Germ cell tumors can also be broadly classified as teratomas, yolk sac tumors (endodermal sinus tumors), germinomas, embryonal cell carcinoma, polyembryomas, choriocarcinomas or mixed germ cell tumors.
- Teratomas are tumors that contain tissue like teeth, hair, muscle and bone. They may be mature or immature. Mature teratomas (dermoid cysts) are the most common type of ovarian germ cell tumor. They’re usually benign. Immature teratomas are typically cancerous, and they grow fast.
- Yolk sac tumors (endodermal sinus tumors) contain cells similar to those in a developing embryo. These tumors are cancerous, and they spread fast to lymph nodes and other organs. Yolk sac tumors are the most common malignant germ cell tumor diagnosed in children.
- Germinomas are cancerous tumors that may appear in your ovaries or testicles. Still, they’re most common in the brain and spinal cord (central nervous system). They’re called dysgerminoma when they’re in your ovaries and seminoma when they’re in your testicles.
- Embryonal cell carcinoma is a rare, cancerous germ cell tumor. It may exist in a pure form, but it often appears alongside other tumor types in a mixed germ cell tumor.
- Polyembryomas contain components that resemble embryos. They’re rare, fast-growing cancerous tumors often mixed with other types of germ cell tumors.
- Choriocarcinomas consist of cells that form the placenta during pregnancy. The placenta is an organ that allows the gestational parent to share nutrients with a fetus. Choriocarcinoma is a rare and cancerous germ cell tumor that can form in your ovaries or testicles but often occurs in the uterus. It can spread to both the fetus and the parent.
- Mixed germ cell tumors contain two or more types of malignant germ cell tumors. Many germ cell tumors are mixed.
Germ cell tumors in children are rare, making up only 3.5% of cancer diagnoses. Germ cell tumors are more common in adolescents and young adults, making up 13.9% of cancers diagnosed between ages 15 to 19. Germ-cell tumors affect predominantly younger males aged between 15 and 40 years, with nearly 74 500 new cases estimated globally in 2020 18, 19.
Germ cell tumors are classified histologically into 2 broad classes: Seminomas and Non-seminomas 11. Seminomas are the most common of germ cell tumors. However, both seminoma and non seminoma germ cell tumor (NSGCT) occur at about the same rate, and men can have either seminoma or nonseminomatous germ cell tumors (NSGCT) or a combination of both. Non-seminoma germ cell tumor (NSGCT) is the most likely testicular cancer to cause metastases which typically affect the lungs, liver, central nervous system, and bone in order of frequency 20.
For treatment planning, germ cell tumors are broadly divided into seminomas and nonseminomas because they have different prognostic and treatment algorithms 21. The good news is the prognosis for most germ cell tumors is excellent. For patients with seminomas (all stages combined), the cure rate exceeds 90%. For patients with low-stage seminomas or nonseminomas, the cure rate approaches 100% 22, 23, 24, 25, 26.
Figure 3. Germ cell tumors
[Source 27 ]Who is affected by germ cell tumors?
Anyone can have germ cell tumors, but infants, children, teens and young adults get them most often. Diagnoses increase and decrease based on age. For instance, diagnoses occur relatively frequently from infancy up to age 4 compared to the years immediately after. Germ cell tumor diagnoses increase as puberty approaches.
- Ovarian germ cell tumors are most common in people aged 10 to 14, but they also affect people in their 20s and 30s. Mature teratomas, which are benign, are the most common germ cell tumor diagnosis.
- Testicular germ cell tumors make up 75% of testicular cancer diagnoses in children and 90% of testicular cancers total. They’re more common in children younger than 4 and people ages 15 to 35.
- Extragonadal germ cell tumors account for 40% to 50% of germ cell tumor diagnoses from infancy to age 4. Still, children and young adults get them, too. In infants and children, these tumors most commonly appear at the base of their spine (sacrum) or tailbone (coccyx). In teens and young adults, extragonadal germ cell tumors occur most often in the center of their chest (mediastinum). Brain germ cell tumors — although rare — are more commonly diagnosed in people between 11 and 30.
Germ cell tumor symptoms
The symptoms of a germ cell tumor depend on factors like the tumor’s size and location in your body.
Ovarian germ cell tumors
Ovarian germ cell tumors don’t always cause symptoms. For instance, mature teratomas may not cause symptoms until they’ve grown large enough to create pressure in your abdomen. Often, they’re discovered on an ultrasound to determine what’s causing pelvic pain.
Ovarian germ cell tumor symptoms may include:
- Pelvic discomfort or pain.
- Painful mass in your ovary.
- Swollen belly (abdomen).
- Abdominal pain (similar to appendicitis).
- Irregular vaginal bleeding.
- Nausea.
Testicular germ cell tumors
The symptoms of testicular germ cell tumors are the same as those of testicular cancer.
Testicular germ cell tumor symptoms include:
- a lump or swelling in your testicle, but it can affect both testicles (>85% of men)
- your testicle getting bigger
- an ache or pain in your testicle or scrotum (the skin that covers the testicles) (about 20% to 30% of men experience a heavy dragging feeling or general ache)
- your scrotum feeling heavy, firm or hard
Other symptoms can include:
- a dull ache or pain in your back, groin or lower tummy
- back pain
- sudden swelling in the scrotum
- losing weight without trying
- a cough
- difficulty breathing or swallowing
- enlargement or tenderness of the breast tissue
- A small percentage present with symptoms of metastatic disease and infertility.
Difference between seminoma and non-seminoma
Nonseminoma is a type of cancer that begins in germ cells also called primordial germ cells that migrate during embryogenesis from the yolk sac through the mesentery to the gonads (testicles and ovaries) (see Figure 3) 1, 2. “Germ” is short for “germinate”, which means to mature. For women germ cells eventually mature into eggs. For men germ cells mature into sperm. Germ cell tumors can generally be divided into gonadal (testicles and ovaries) and extragonadal (outside of the gonads). Germ cell tumors most often form where eggs get made (ovaries) and where sperm gets made (testicles). Less commonly, germ cell tumors form in body parts other than your ovaries and testicles, including your abdomen, chest, low back, tailbone and brain. These tumors are called extragonadal tumors.
There are several types of nonseminoma tumors, including embryonal carcinoma, malignant teratoma, choriocarcinoma, and yolk sac tumor. These tumors are usually made up of more than one type of cancer cell. Although nonseminomas occur most often in the testicles or ovaries, they can occur in other tissues, such as the brain, chest, or abdomen. This happens when germ cells (primordial germ cells) that have the ability to form sperm or eggs are found in other parts of the body. Nonseminoma testicular cancers tend to grow and spread more quickly than seminomas 11, 12. They usually are large and cause signs and symptoms. Testicular cancers that are 100% seminoma are considered seminomas. A testicular cancer that contains both seminoma and nonseminoma cells are considered and should be treated as a nonseminoma testicular cancer 28. Testicular germ cell tumors that appear to have a seminoma histology but are accompanied by an elevated serum level of alpha-fetoprotein (AFP) should be treated as nonseminomas because seminomas do not produce alpha-fetoprotein (AFP).
Seminoma is another type cancer that begins in germ cells (malignant germ cell tumor) that involves most commonly the testicle where it’s called seminoma testicular cancer or less frequently the mediastinum (area between the lungs) where it’s called mediastinal seminoma, the retroperitoneum (the back wall of the abdomen) where it’s called retroperitoneal seminoma, or other extra-gonadal sites where it’s called extra-gonadal seminoma 29. Seminomas tend to grow and spread (metastasize) more slowly than nonseminomas testicular cancer 11, 12, although some seminomas can grow very rapidly too. Seminomas are more sensitive to radiation. Seminomas generally have a good prognosis, while non-seminomatous tumors are much more likely to present with metastatic disease as well as mixed germ cell tumors comprising of two or more than two different germ cell tumor components.
Seminomas in the testicle are common among men ages 15 to 34 years old 29. Most testicular cancers are germ cell tumors and about half of these are seminomas, which tend to affect older men, with a peak incidence in men aged 30 to 34 years 11. In the United States, testicular seminoma is the most common subtype of testicular cancer 30. A higher incidence of seminoma is seen among Whites than in African Americans, and the rate has increased in the White population over recent decades 31. In 2021, there were an estimated 299,845 men living with testicular cancer in the United States 32.
Non-seminoma testicular cancer causes
The exact cause of testicular cancer is poorly understood. Factors that may increase a man’s risk of developing testicular cancer are:
- Having an undescended testicle (cryptorchidism)
- Exposure to certain chemicals
- Family history of testicular cancer
- HIV infection
- History of testicular cancer
- History of an undescended testicle (one or both testicles fail to move into the scrotum before birth)
- Klinefelter syndrome
- Infertility
- Tobacco use
- Down syndrome
Risk factors for non-seminoma testicular cancer
Anything that increases your chance of getting a disease is called a risk factor. Factors that may increase your risk of testicular cancer include 33, 34:
- Having an undescended testicle (cryptorchidism). The testes form in the belly during fetal development. They typically descend into the scrotum before birth. If you have a testicle that never descended, your risk of testicular cancer is higher. The risk is increased even if you’ve had surgery to move the testicle to the scrotum.
- Having a family history (especially in a father or brother) of testicular cancer. If testicular cancer runs in your family, you might have an increased risk.
- Personal history of testicular cancer
- Having had abnormal development of the testicles.
- Being a young adult. Testicular cancer can happen at any age. But it’s most common in teens and young adults between 15 and 35.
- Being white. Testicular cancer is most common in white people.
The most common risk factor for testis cancer is a history of undescended testicle or cryptorchidism. Normally in the developing male fetus, the testicles form near the kidneys in the abdomen (belly). About the eighth month of pregnancy, the testicles descend, exit the body and settle in the scrotum. About 3% of boys have one or both testicles that fail to make it into the scrotum. Testicles can settle in the abdomen or in the inguinal canal or groin where the testicle exits the body wall and enters the scrotum. Most of the time, an undescended testicle will move down and settle into the scrotum within the first year of life. Sometimes surgery is required to bring down and fix the testicle to the scrotum, this surgery is called an orchiopexy.
Boys with a history of cryptorchidism have an increased risk of testicular cancer. The risk of testicular cancer is not directly related to the fact that the testicle does not descend, but it is believed that the abnormality in descent likely indicates an abnormality in the testicle that makes cancer more likely. This belief is based on the following observations: The testicular cancer usually develops in the undescended testicle (four- to sixfold increased risk of cancer), but the risk of cancer is also higher in the normal testicle (less than twofold increased risk). In addition, generally the higher the testicle, the higher the risk of testicular cancer — intra-abdominal testis have a much higher risk of cancer than those in the inguinal canal. Early surgery (orchiopexy) reduces the risk of testis cancer (two- to threefold risk if the surgery is performed prior to puberty) but does not erase the chance for that boy to develop testicular cancer later in life 35.
Testicular germ-cell tumors is associated with cryptorchidism, hypospadias and decreased fertility is often referred to as the testicular dysgenesis syndrome 36. In utero exposure to endocrine disruption chemicals might increase the likelihood of testicular dysgenesis syndrome 37. Among these chemicals, organochlorine insecticides have been demonstrated to increase the risk of testicular germ-cell tumor 38.
Furthermore, a family history of testicular cancer is another common risk factor, with an eight- to twelvefold risk if a man has a brother with testicular cancer and a two- to fourfold risk if his father has testicular cancer. While there is not a specific gene linked to testicular cancer, the cancer is highly heritable and can be passed from generation to generation. In addition, the average age at diagnosis is two to three years younger than the general population if a first-degree relative has testicular cancer. However, it should be remembered that testicular cancer is rare, and it is therefore rare for this cancer to run in families.
Men with a personal history of testicular cancer have the highest risk of developing another cancer. Fortunately, only 2% of men will develop cancer in both testicles, but that risk is twelvefold higher than men without testis cancer. In addition, men who develop testis cancer in their 20s or earlier, men with seminoma and men with germ cell neoplasia is situ formerly known as carcinoma in situ (CIS) or intratubular germ cell neoplasia (ITGCN) have a higher risk of developing a second testis cancer.
Most testis cancer arises from the precursor lesion known as germ cell neoplasia is situ (GCNIS). Germ cell neoplasia is situ (GCNIS) is present adjacent to testis cancer in 80–90% of patients. For men in whom germ cell neoplasia is situ (GCNIS) is found for other reasons, the risk of subsequent testis cancer is 50% at five years and 70% at seven years. Therefore, germ cell neoplasia is situ (GCNIS) is the last well-known risk factor for testis cancer.
Microlithiasis or small calcifications (stones) in the testicle found on ultrasound, was once believed to be a risk factor for testicular cancer. Microlithiasis is not a risk factor for testicular cancer for most men; however, if one of the other risk factors (above) exists, microlithiasis may indicate a higher risk of cancer and warrants monthly testicular self-examination and routine follow-up with a doctor.
Non-seminoma testicular cancer symptoms
Seminoma testicular cancer usually only affects one testicle, but it can affect both.
Signs and symptoms of testicular cancer include:
- a lump or swelling in your testicle, but it can affect both testicles (>85% of men)
- your testicle getting bigger
- an ache or pain in your testicle or scrotum (the skin that covers the testicles) (about 20% to 30% of men experience a heavy dragging feeling or general ache)
- your scrotum feeling heavy, firm or hard
Other symptoms can include:
- a dull ache or pain in your back, groin or lower tummy
- back pain
- sudden swelling in the scrotum
- losing weight without trying
- a cough
- difficulty breathing or swallowing
- enlargement or tenderness of the breast tissue
- A small percentage present with symptoms of metastatic disease and infertility.
Most testicular cancers present as a mass confined to the one testicle, but it can affect both. Therefore, the most common presentation is a painless testicular mass. Most of these masses can be felt and of significant size (a few to several centimeters). Small, nonpalpable lesions without pain and in the absence of distant disease have a higher likelihood of being a benign tumor. In a number of studies, upwards of 80 percent of nonpalpable, asymptomatic masses that are 2 cm or smaller will be benign tumors. Benign lesions may include testicular cysts, varicocele, small infarcts or small Leydig cell or Sertoli cell tumors. Patients with a significantly enlarged or swollen testicle are most likely to have epididymo-orchitis, especially if the testicle is red or painful. An apparent epididymo-orchitis that is painless or does not respond to antibiotics should be considered suspicious for a testicular cancer.
Patients with the localized non seminoma testicular cancer will typically present with a painless nodule, lump, mass, or swelling in one of the testes. It might also present with dull scrotal or testicular pain. Ten percent of the affected individuals will experience acute testicular pain. Testicular cancer is not associated with trauma, but the presentation in the emergency room with scrotal or testicular trauma may elicit an examination or imaging of the testes resulting in the final diagnosis. A careful testicular examination will allow the examiner to detect a firm intra-testicular lesion.
Serious, acute pain is associated with rapidly growing tumors and associated bleeding or infarction (if the tumor outgrows its blood supply). Most patients with pain complain of dull scrotal discomfort or heaviness. Rarely, trauma can lead to a diagnosis, mostly because it brings a mass or pain to the patient’s awareness.
To differentiate intra-testicular from extra-testicular lesions, each testis should be gently held and rolled between the fingers. It is important to examine the opposite testis completely. Sometimes, due to a hydrocele presence, the testis cannot be adequately examined or palpated, and the presence or absence of a testicular lesion should be confirmed with ultrasonography (which is an extension of the physical examination in such situations).
It is unusual for patients to present with symptoms or signs of disseminated disease 39. However, upwards of 30 percent of men who present with metastatic cancer, symptoms of metastases can be the presenting complaint. In patients with retroperitoneal spread, bulky retroperitoneal lymphadenopathy (enlarged lymph nodes) can lead to abdominal mass; abdominal, flank or back pain due to direct invasion or obstruction of muscles, blood vessels or the ureters; lower extremity swelling if the inferior vena cava (IVC) is compressed. Patients who are having retro-duodenal spread can present with nausea, vomiting, or gastrointestinal hemorrhage or gastrointestinal symptoms if the intestines are involved. Patients with lung metastases can present as chest pain, shortness of breath, coughing up blood (hemoptysis) and cough. Cervical or supraclavicular lymphadenopathy can be palpated in the lymphatic spread. Systemic symptoms include anorexia, malaise, and weight loss.
Male gynecomastia is found in about 5% of men with testicular cancer, presumably from elevated beta-human chorionic gonadotropin (beta-hCG) levels. Therefore, all men with gynecomastia should have a careful examination of the testicles, and patients with testicular masses should be checked for gynecomastia.
Non-seminomatous germ cell tumors is also associated with male infertility due to low sperm counts, reduced sperm motility, and increased abnormal morphology. This is thought to be due to spermatogenesis (sperm production) abnormalities and has been reported in up to 35% of patients with non seminoma testicular cancer 40, 41. Therefore, a careful examination of the testes in male infertility patients should also screen for possible testicular masses.
Non-seminoma testicular cancer diagnosis
You might find lumps, swelling or other symptoms of testicular cancer on your own. Testicular cancers can be detected during an exam by a doctor too. You’ll need other tests to see if testicular cancer is causing your symptoms.
Tests used to diagnose testicular cancer include:
- Testicular ultrasound (scrotal ultrasound). A testicular ultrasound test uses sound waves to make pictures. It can be used to make pictures of the scrotum and testicles. During an ultrasound you lie on your back with your legs spread. A doctor puts a clear gel on the scrotum. A hand-held probe is moved over the scrotum to make the pictures. Ultrasound gives your doctor more clues about any lumps around the testicle. It can help your doctor see whether the lumps look like something that isn’t cancer or if they look like cancer. An ultrasound shows whether the lumps are inside or outside the testicle. Lumps inside the testicle are more likely to be testicular cancer. Radical orchiectomy (surgery to remove the testicle) should strongly be considered for any intratesticular mass and suspicion of testis cancer.
- Serum testicular tumor markers. A blood test can detect proteins made by testicular cancer cells. This type of test is called a tumor marker test. Tumor markers for testicular cancer include beta-human chorionic gonadotropin (hCGβ), alpha-fetoprotein (AFP) and lactate dehydrogenase (LDH). Having these substances in your blood doesn’t mean you have cancer. Having levels higher than is typical is a clue your doctor uses to understand what’s going on in your body. Serum AFP, LDH, and beta-hCG should be measured before an orchiectomy. Lactate dehydrogenase (LDH) is generally a nonspecific tumor marker that is elevated in 20% to 60% of patients with non seminoma testicular cancer and is generally considered an indicator of bulky disease. Alpha-fetoprotein (AFP) has a half-life of about 6 days, while the half-life of beta-human chorionic gonadotropin (beta-hCG) is 24 to 48 hours. In practical terms, beta-human chorionic gonadotropin (beta-hCG) should begin to return to normal about 1 week after orchiectomy, while alpha-fetoprotein (AFP) will take a minimum of 5 weeks. Alpha-fetoprotein (AFP) and beta-human chorionic gonadotropin (beta-hCG) are also elevated in various lung, liver, gut, bone, and live tumors 42, 43. There are new markers to diagnose and evaluate germ cell tumors, like levels of circulating microRNAs, especially miR-371a-3p. However, their precise role in the management of testicular tumors is yet to be determined 44.
- Surgery to remove a testicle through an incision in the groin (Inguinal orchiectomy). If your doctor thinks a lump on your testicle may be cancerous, you might have surgery to remove the testicle through an incision in the groin. NOTE: The surgeon does not cut through the scrotum into the testicle to remove a sample of tissue for biopsy, because if cancer is present, this procedure could cause it to spread into the scrotum and lymph nodes. It’s important to choose a surgeon who has experience with this kind of surgery. The testicle is sent to a lab for testing. A tissue sample from the testicle is then viewed under a microscope to check for cancer cells. If cancer is found, the cell type (seminoma or nonseminoma) is determined in order to help plan treatment.
- Transscrotal biopsy is not considered appropriate because of the risk of local dissemination of tumor into the scrotum or its spread to inguinal lymph nodes 21. A retrospective analysis of reported series in which transscrotal approaches were used showed a small but statistically significant increase in local recurrence rates, compared with when the inguinal approach was used (2.9% vs. 0.4%) 45. However, distant recurrence and survival rates were indistinguishable in the two approaches 21.
- Advanced imaging. Additional imaging can be performed before or after the diagnosis of cancer is confirmed, based on the strength of suspicion for cancer. Imaging of the chest, abdomen and pelvis are typically required to evaluate for spread of testicular cancer — this process is called staging. CT, MRI and X-ray can be used for staging. PET scan or bone scan are not recommended for the routine evaluation of testicular cancer.
Testicular cancer screening
Some doctors recommend regular testicle self-exams. During a testicular self-exam you feel your testicles for any lumps or other changes. If you notice any changes that last longer than two weeks, make an appointment with your doctor.
Serum tumor markers
Alpha-fetoprotein (AFP), beta-human chorionic gonadotropin (beta-hCG), and lactate dehydrogenase (LDH) play an important role as serum tumor markers in the staging and monitoring of germ cell tumors and should be measured prior to removing the involved testicle 46. For patients with nonseminomas, one of the most significant predictors of prognosis is the degree of tumor-marker elevation after the cancerous testicle has been removed 47. Elevated levels of serum tumor markers are often the earliest sign of relapse (recurrence), making these markers useful for monitoring all stages of nonseminomas and metastatic seminomas 21.
Significant and unambiguously rising levels of alpha-fetoprotein (AFP) and/or beta-human chorionic gonadotropin (beta-hCG) signal relapsed germ cell tumor in most cases and are an indication for treatment even in the absence of radiological evidence of metastatic disease 21. However, tumor marker elevations need to be interpreted with caution. For example, false-positive beta-hCG levels can result from cross reactivity of the assay with luteinizing hormone (LH) in which case an intramuscular injection of testosterone should result in normalization of beta-hCG values 21. There are also clinical reports of marijuana use resulting in elevations of serum beta-hCG and some experts recommend querying patients about drug use and retesting beta-hCG levels after a period of abstinence from marijuana use 21. Similarly, AFP is chronically mildly elevated in some individuals for unclear reasons and can be substantially elevated by liver disease 21.
Alpha-fetoprotein (AFP)
Elevation of serum alpha-fetoprotein (AFP) is seen in 40% to 60% of men with nonseminomas. Seminomas do not produce alpha-fetoprotein (AFP). So if it’s elevated in a patient diagnosed only with seminoma, it means thatnonseminoma testicular cancer elements were missed. Men who have an elevated serum AFP have a mixed germ cell tumor (i.e., nonseminomatous germ cell tumors [NSGCT]) even if the pathology shows a pure seminoma—unless there is a more persuasive explanation for the elevated AFP, such as liver disease 21.
Beta-human chorionic gonadotropin (beta-hCG)
Elevation of beta-human chorionic gonadotropin (beta-hCG) is found in approximately 14% of patients with stage 1 pure seminomas before orchiectomy (surgery to remove the testicle) and in about one-half of patients with metastatic seminomas 48, 49, 50. Approximately 40% to 60% of men with nonseminomas have an elevated serum beta-hCG 21.
Lactate dehydrogenase (LDH)
Seminomas and nonseminomas alike may result in elevated lactate dehydrogenase (LDH) but such values are of unclear prognostic significance because LDH may be elevated in many conditions unrelated to cancer. A study evaluated the utility of lactate dehydrogenase (LDH) in 499 patients with a testicular germ cell tumor who were undergoing surveillance after orchiectomy or treatment of stage 2 or 3 disease. It found that 7.7% of patients had elevated lactate dehydrogenase (LDH) unrelated to cancer, while only 1.4% of patients had cancer-related increases in LDH 51. Among 15 patients with relapsed disease, LDH was elevated in six patients and was the first sign of relapse in one patient 51. Over 9% of the men had a persistent false-positive increase in LDH. The positive predictive value for an elevated LDH was 12.8% 51.
A second study reported that among 494 patients with stage 1 germ cell tumors who subsequently had a relapse, 125 had an elevated LDH at the time of relapse 52. Of these 125 patients, all had other evidence of relapse: 112 had a concurrent rise in AFP and/or beta-hCG, one had computed tomography (CT) evidence of relapse before the elevation in LDH, one had palpable disease on examination, and one complained of back pain that led to imaging that revealed retroperitoneal relapse 52. On one hand, measuring LDH appears to have little value for predicting relapse during surveillance of germ cell tumors. On the other hand, for patients with metastatic non-seminoma germ cell tumor (NSGCT), large studies of prognostic models have found the LDH level to be a significant independent predictor of survival 47, 22.
Testicular cancer staging
Once your doctor confirms your diagnosis, the next step is to see whether the testicular cancer has spread beyond the testicle to other parts of your body. This is called the cancer’s stage. It helps your health care team understand your prognosis and how likely your cancer is to be cured.
The following tests and procedures may be used in the testicular cancer staging process:
- Chest x-ray: An x-ray of the organs and bones inside the chest. An x-ray is a type of energy beam that can go through the body and onto film, making a picture of areas inside the body.
- Computerized tomography (CT) scan also called computed tomography, computerized tomography, or computerized axial tomography. CT scans take a series of X-ray pictures of your belly, chest and pelvis. A dye may be injected into a vein or swallowed to help the organs or tissues show up more clearly. A doctor checks the pictures for signs that cancer has spread.
- MRI (magnetic resonance imaging): A procedure that uses a magnet, radio waves, and a computer to make a series of detailed pictures of areas inside the body, such as the abdomen. This procedure is also called nuclear magnetic resonance imaging (NMRI).
- Abdominal lymph node dissection: A surgical procedure in which lymph nodes in the abdomen are removed and a sample of tissue is checked under a microscope for signs of cancer. This procedure is also called lymphadenectomy. For patients with nonseminoma, removing the lymph nodes may help stop the spread of disease. Cancer cells in the lymph nodes of seminoma patients can be treated with radiation therapy.
- Serum testicular tumor markers. Tumor marker tests such as beta-human chorionic gonadotropin (beta-hCG), alpha-fetoprotein (AFP) and lactate dehydrogenase (LDH) are often repeated after surgery to remove the testicle. The results help your doctor decide whether you might need additional treatments to kill the cancer cells. Tumor marker tests might be used during and after cancer treatment to monitor your condition. Tumor marker levels are measured again, after inguinal orchiectomy and biopsy, in order to determine the stage of the cancer. This helps to show if all of the cancer has been removed or if more treatment is needed. Tumor marker levels are also measured during follow-up as a way of checking if the cancer has come back.
Evaluation of the retroperitoneal lymph nodes, usually by CT scan, is an important aspect of staging and treatment planning in adults with testicular cancer 53, 54. Patients with a negative result have a substantial chance of having microscopic involvement of the lymph nodes. Nearly 20% of patients with seminoma and 30% of patients with nonseminoma who have normal CT scans and serum tumor markers will subsequently relapse if not given additional treatment after orchiectomy 55, 56, 57. For patients with nonseminoma, retroperitoneal lymph node dissection increases the accuracy of staging, but as many as 10% of men with normal imaging, normal tumor markers, and benign pathology at retroperitoneal lymph node dissection will still experience a relapse 58. After retroperitoneal lymph node dissection, about 25% of patients with clinical stage 1 nonseminomatous testicular cancer are restaged as pathological stage 2, and about 25% of clinical stage 2 patients are restaged as pathological stage 1 58, 59, 60. In prepubertal children, the use of serial measurements of AFP has proven sufficient for monitoring response after initial orchiectomy. Lymphangiography and para-aortic lymph node dissection do not appear to be useful or necessary in the proper staging and management of testicular cancer in prepubertal boys 61.
The stages of testicular cancer range from 0 to 3.
- Stage 0 and stage 1 testicular cancers only affect the testicle and the area around it. At these early stages, the cancer hasn’t spread to the lymph nodes or other parts of the body.
- Stage 2 testicular cancers have spread to the lymph nodes. When testicular cancer spreads to other parts of the body, it is stage 3. Not all stage 3 cancers have spread though.
- Stage 3 testicular cancers can also mean that the cancer is in the lymph nodes and the tumor marker results are very high.
Stage 0 testicular cancer (germ cell neoplasia in situ or testicular intraepithelial neoplasia)
Stage 0 testicular cancer is also called germ cell neoplasia in situ (GCNIS) or testicular intraepithelial neoplasia (TIN). In stage 0, abnormal cells are found in the tiny tubules where the sperm cells begin to develop. These abnormal cells may become cancer and spread into nearby normal tissue. All tumor marker levels are normal.
Around 5% of testicular cancer patients have germ-cell neoplasia in situ (GCNIS) in the other testis with the highest risk (∼30%) in men with testicular atrophy (volume <12 ml) and age <40 years 37. Approximately 30%-40% of patients with retroperitoneal extragonadal germ cell tumors (extragonadal germ cell tumor) harbor testicular germ-cell neoplasia in situ (intratubular germ cell neoplasia in situ) 62, 63, 64.
In most cases, testicular intraepithelial neoplasia (TIN) is diagnosed as a result of an orchiectomy that was performed to remove an invasive germ cell tumor. Generally, testicular intraepithelial neoplasia (TIN) has already been removed from the body at the time of diagnosis and requires no treatment. A more challenging situation arises if a biopsy is performed of the opposite testis and testicular intraepithelial neoplasia (TIN) is discovered. Because of the low incidence and low mortality rates associated with contralateral germ cell tumors, such biopsies are performed rarely in the United States. Therefore, testicular intraepithelial neoplasia (TIN) is almost never diagnosed in testicles that do not also have an invasive tumor 21. Consequently, a treatment decision about testicular intraepithelial neoplasia (TIN) in stage 0 testicular cancer is rarely faced in the United States 21. Treatment options for germ-cell neoplasia in situ (GCNIS) include radiation therapy, surveillance, and orchiectomy 21.
Stage 1 testicular cancer
Stage 1 testicular cancer is limited to the testis. Stage 1 testicular cancer is divided into stages 1A, 1B, and 1S.
In stage 1A, cancer is found in the testicle, including the rete testis, but has not spread to the blood vessels or lymph vessels in the testicle.
- All tumor marker levels such as beta-human chorionic gonadotropin (beta-hCG), alpha-fetoprotein (AFP) and lactate dehydrogenase (LDH) are normal.
In stage 1B, cancer:
- is found in the testicle, including the rete testis, and has spread to the blood vessels or lymph vessels in the testicle; or
- has spread into the hilar soft tissue (tissue made of fibers and fat with blood vessels and lymph vessels), the epididymis, or the outer membranes around the testicle; or
- has spread to the spermatic cord; or
- has spread to the scrotum.
- All tumor marker levels such as beta-human chorionic gonadotropin (beta-hCG), alpha-fetoprotein (AFP) and lactate dehydrogenase (LDH) are normal.
In stage 1S, cancer is found anywhere in the testicle and may have spread into the spermatic cord or scrotum. Tumor marker levels range from slightly above normal to high.
Stage 2 testicular cancer
Stage 2 testicular cancer involves the testis and the retroperitoneal or periaortic lymph nodes usually in the region of the kidney. Stage 2 testicular cancer is divided into stages 2A, 2B, and 2C.
In stage 2A, cancer is found anywhere in the testicle and may have spread into the spermatic cord or scrotum. Cancer has spread to 1 to 5 nearby lymph nodes and the lymph nodes are 2 centimeters or smaller.
- All tumor marker levels are normal or slightly above normal.
In stage 2B, cancer is found anywhere in the testicle and may have spread into the spermatic cord or scrotum. Cancer has spread to:
- 1 nearby lymph node and the lymph node is larger than 2 centimeters but not larger than 5 centimeters; or
- more than 5 nearby lymph nodes and the lymph nodes are not larger than 5 centimeters; or
- a nearby lymph node and the cancer has spread outside the lymph node.
- All tumor marker levels are normal or slightly above normal.
In stage 2C, cancer is found anywhere in the testicle and may have spread into the spermatic cord or scrotum. Cancer has spread to a nearby lymph node and the lymph node is larger than 5 centimeters.
- All tumor marker levels are normal or slightly above normal.
Stage 3 testicular cancer
Stage 3 testicular cancer has spread beyond the retroperitoneal nodes based on physical examination, imaging studies, and/or blood tests (i.e., patients with retroperitoneal adenopathy and highly elevated serum tumor markers are stage 3). Stage 3 testicular cancer is divided into stages 3A, 3B, and 3C.
In stage 3A, cancer is found anywhere in the testicle and may have spread into the spermatic cord or scrotum. Cancer may have spread to one or more nearby lymph nodes. Cancer has spread to distant lymph nodes or to the lungs.
- All tumor marker levels are normal or slightly above normal.
In stage 3B, cancer is found anywhere in the testicle and may have spread into the spermatic cord or scrotum. Cancer has spread:
- to one or more nearby lymph nodes and has not spread to other parts of the body; or
- to one or more nearby lymph nodes. Cancer has spread to distant lymph nodes or to the lungs.
- The level of one or more tumor markers is moderately above normal.
In stage 3C, cancer is found anywhere in the testicle and may have spread into the spermatic cord or scrotum. Cancer has spread:
- to one or more nearby lymph nodes and has not spread to other parts of the body; or
- to one or more nearby lymph nodes. Cancer has spread to distant lymph nodes or to the lungs.
- The level of one or more tumor markers is high.
OR
- Cancer is found anywhere in the testicle and may have spread into the spermatic cord or scrotum. Cancer has not spread to distant lymph nodes or the lung, but has spread to other parts of the body, such as the liver or bone.
- Tumor marker levels may range from normal to high.
Non-seminoma testicular cancer treatment
Testicular cancer treatment options depend on the following:
- Whether the tumor is nonseminoma or seminoma.
- The size of the tumor and where it is in the body.
- The blood levels of alpha-fetoprotein (AFP), beta-human chorionic gonadotropin (hCGβ), and lactate dehydrogenase (LDH).
- Whether the tumor has spread to other parts of the body.
- The way the tumor responds to initial treatment.
- Whether the tumor has just been diagnosed or has recurred (come back).
Testicular cancer treatment often involves surgery and chemotherapy. Which treatment options are best for you depends on the type of testicular cancer you have and its stage. Your cancer treatment team will also considers your overall health and your preferences.
Surgery
Surgery to remove the testicle (inguinal orchiectomy) and some of the lymph nodes may be done at diagnosis and staging. Radical inguinal orchiectomy with initial high ligation of the spermatic cord is the procedure of choice in diagnosing and treating a malignant testicular mass 65. Tumors that have spread to other places in the body may be partly or entirely removed by surgery.
Operations used to treat testicular cancer include:
- Surgery to remove the testicle also called a radical inguinal orchiectomy. It’s the first treatment for most testicular cancers. To remove the testicle, a surgeon makes a cut in the groin. The entire testicle is pulled out through the opening. A prosthetic, gel-filled testicle can be inserted if you choose. This might be the only treatment needed if the cancer hasn’t spread beyond the testicle.
- Surgery to remove nearby lymph nodes. If there’s concern that your cancer may have spread beyond your testicle, you might have surgery to remove some lymph nodes. To remove the lymph nodes, the surgeon makes a cut in the belly. The lymph nodes are tested in a lab to look for cancer. Surgery to remove lymph nodes is often used to treat the nonseminoma type of testicular cancer.
Radical inguinal orchiectomy is the standard procedure for diagnostic and therapeutic purposes. Radical inguinal orchiectomy surgery involves removing the testicle and spermatic cord where it exits the body to identify and likely treat the majority of cancers localized to the testis. Trans scrotal orchiectomy or biopsy of the testicular mass is contraindicated for the risk of tumor seeding of the lymphatic drainage. Spinal or general anesthesia may be used. An inguinal incision is made to expose the external and internal iliac canal. External iliac fascia is opened, revealing the spermatic cord. The spermatic cord is controlled to stop retroperitoneal lymphatic and venous drainage of tumor cells. After that, deliver the testis from the scrotum and ligate vas deferens and spermatic arteries separately. Retroperitoneal lymph node dissection is done if necessary. Reapproximate the external oblique fascia and close the skin in standard fashion. Conduct a follow-up study by staging and referring the patient for appropriate adjuvant therapies.
After the doctor removes all the cancer that can be seen at the time of the surgery, some patients may be given chemotherapy or radiation therapy after surgery to kill any cancer cells that are left. Treatment given after the surgery, to lower the risk that the cancer will come back, is called adjuvant (add-on) therapy.
Testicular cancer surgery carries a risk of bleeding and infection. If you have surgery to remove lymph nodes, there’s also a risk that a nerve might be cut. Surgeons take great care to protect the nerves. Sometimes cutting a nerve can’t be avoided. This can lead to problems with ejaculating, but it generally doesn’t affect your ability to get an erection. Ask your surgeon about options for preserving your sperm before surgery.
Ilioinguinal nerve injury can occur if the nerve is damaged during dissection of the spermatic cord. This is more common in men who underwent prior inguinal surgery (usually for an undescended testicle or hernia repair) and can occur during dissection or be inadvertently trapped in the closure of the external oblique fascia. The deficit is often decreased sensation to the medial thigh, scrotum or base of the penis. It is often transient, but can take several weeks or months to improve.
Inguinal hernia can occur if the external oblique fascia is not closed properly or if the closure breaks down. It is important to minimize strenuous activities for two to four weeks to prevent development of a hernia.
Retroperitoneal lymph node dissection (RPLND)
The retroperitoneal lymph nodes are the primary nodal sites involved in the spread of non-seminoma testicular cancer. Removal of these draining lymph nodes results in a high cure rate for testicular tumors. The cure rate for stage 1 non-seminoma testicular cancer is about 93-95% with retroperitoneal lymph node dissection (RPLND) alone. It is generally 3-4 hours of surgery with a mortality rate of less than 1%. Retroperitoneal lymph node dissection (RPLND) involves the dissection of the retroperitoneal lymph nodes with the resulting possibility of retrograde ejaculation, loss of fertility, and incontinence due to disruption of pelvic nerves during the dissection. Retroperitoneal lymph node dissection (RPLND) is still the gold standard for stage 1 non-seminoma testicular cancer for staging and therapeutic purposes. Retroperitoneal lymph node dissection may also be used for stage 2A, but for stage 2B and higher. Initial chemotherapy is the standard of care. In general, patients with residual lymph nodes 1 cm or larger after definitive chemotherapy are candidates for retroperitoneal lymph node dissection (RPLND).
Retroperitoneal lymph node dissection with adjuvant chemotherapy is the mainstay of managing low-stage non-seminomatous germ cell tumors in the United States. Retroperitoneal lymph node dissection (RPLND) is a staging, diagnostic and therapeutic procedure for patients with non-seminoma testicular cancer. One of the benefits of retroperitoneal lymph node dissection (RPLND) surgery is that it helps identify those patients who might benefit from postoperative chemotherapy. Also, men with non-seminoma testicular cancer who do not undergo retroperitoneal lymph node dissection (RPLND) will require more intensive surveillance protocols even for stage 1 disease than similar patients who may only need routine tumor markers and chest x-rays after retroperitoneal lymph node dissection (RPLND) surgery, retroperitoneal relapse rate drops to <1% 66. Some centers will tend to recommend retroperitoneal lymph node dissection (RPLND) in stage 1 nonseminoma testicular cancer over active surveillance or chemotherapy if they have histological risk factors (evidence of vascular invasion in the original testicular malignancy or a significant component of embryonal carcinoma) 5.
Retroperitoneal lymph node dissection (RPLND) is traditionally done as an open surgical procedure with a large midline abdominal incision. Removal of residual masses with retroperitoneal lymph node dissection (RPLND) after chemotherapy is a much more complicated and technically difficult procedure but provides excellent survival in high-risk/high tumor volume cases. Disease-specific survival has been reported as 81%, with 70% having no progression of their cancers 67, 68. Laparoscopic retroperitoneal lymph node dissection (RPLND) has been reported in several series with good results, even when performed after chemotherapy 67, 68.
Modern modifications include a nerve-sparing approach to save the nerves supplying the bladder neck to preserve ejaculatory function and continence 69. This approach uses a template-based right and left-sided dissection, which preserves sexual function and fertility in over 90% of patients 70. Other complications from retroperitoneal lymph node dissection (RPLND) surgery include wound infection, bowel ileus, pulmonary embolism, lymphocele, lymphedema, chylous ascites, and hydronephrosis 66.
Complications of retroperitoneal lymph node dissection (RPLND), even in experienced hands, are relatively common at 18% as the surgery is technically demanding and often complicated by patient comorbidities 71. Postoperative ejaculatory problems are common 72, 73, 74. Erectile dysfunction (the inability to get or maintain an erection long enough to have sexual intercourse) is reported in up to 15% with current nerve-sparing techniques 74, 75. Some patients may require resection of the inferior vena cava or aortic tube grafts to replace aortic segments 76, 77. Referral to tertiary care centers with surgeons experienced in retroperitoneal lymph node dissection (RPLND) surgery is recommended when this procedure is required, as in experienced hands, the outcomes are significantly better 78, 79. An experienced retroperitoneal lymph node dissection (RPLND) surgeon has been defined as one who performs 24 or more of these procedures a year.
Relapses after retroperitoneal lymph node dissection (RPLND) are typically pulmonary metastasis, with a reported incidence of 10% for those with negative nodes and 28% for patients with positive nodal disease 80. Fortunately, such recurrences are usually curable with systemic chemotherapy.
In clinical stage 1 of non-seminomatous germ cell tumors, the relapse rate is about 64%. Relapsed cases are generally considered high-risk for metastatic disease. In the United States, retroperitoneal lymph node dissection (RPLND) is offered to re-confirm the pathological stage as well as for curative treatment. However, in European countries, patients are usually offered adjuvant chemotherapy consisting of two cycles of bleomycin, etoposide and cisplatin (BEP) chemotherapy, which is generally reserved for non-seminoma testicular cancer with high-risk features, such as vascular invasion. After treatment, 95-97% of patients remain relapse-free, with a cure rate approaching 100%. Adjuvant chemotherapy with two cycles of BEP in the high-risk group can provide a long-term progression-free survival rate of 97% 81.
Chemotherapy
Chemotherapy treatment uses chemo drugs to kill cancer cells. Chemotherapy travels throughout the body. It can kill cancer cells that may have spread beyond the testicle. Chemotherapy is often used after surgery. It can help kill any cancer cells that are still in the body. When testicular cancer is very advanced, sometimes chemotherapy is used before surgery.
Non-seminomatous germ cell tumors are the most sensitive testicular cancers to cisplatin-based chemotherapy. Patients with elevated serum tumor markers are typically given 3-4 cycles of bleomycin, etoposide, and cisplatin (BEP) chemotherapy 5. After completion of chemotherapy, tumor markers are repeated to see the decline in values, along with a contrast-enhanced CT scan to evaluate for any residual mass 5. If a mass is present with normal tumor markers, this is considered an indication for retroperitoneal lymph node dissection (RPLND). In patients who developed lung fibrosis (pulmonary fibrosis) or with a history of significant pre-existing lung disease, bleomycin would be contraindicated. If the patient still has elevated markers, second-line chemotherapy, including vincristine, ifosfamide, and cisplatin (VIP), should be considered. Other second and third-line agents include gemcitabine, etoposide, paclitaxel, and oxaliplatin. Chemotherapy also may cause your body to stop making sperm. Chemotherapy causes azoospermia (a complete absence of sperm in the semen) in most patients for at least 2 to 3 years after treatment. Cisplatin and similar alkylating agents are the most injurious to sperm production (spermatogenesis), with Sertoli cells being relatively sensitive but Leydig cells being relatively resistant. Sertoli cells are also very radiosensitive. Overall, fertility is reduced by an average of 30% after chemotherapy treatment for non seminoma testicular cancer 41, 82, 83. Often, sperm production starts again as you get better after cancer treatment. But sometimes losing sperm production is permanent. Ask your doctor about your options for preserving your sperm before chemotherapy.
Side effects of chemotherapy depend on the specific medicines being used. Common side effects include fatigue, hearing loss and an increased risk of infection. Other potential side effects of chemotherapy include peripheral neuropathy, kidney failure, myelosuppression, loss of hearing, increased cardiovascular disorders, and hypogonadism (low testosterone).
Immunotherapy
Immunotherapy is treatment with medicine that helps your body’s immune system kill cancer cells. Your immune system fights off diseases by attacking germs and other cells that shouldn’t be in your body. Cancer cells survive by hiding from the immune system. Immunotherapy helps the immune system cells find and kill the cancer cells.
Immunotherapy is sometimes used for advanced testicular cancer 84. It might be an option if the cancer doesn’t respond to other treatments.
Surveillance of testicular cancer after treatment
The median time to relapse in non-seminoma testicular cancer patients is 7 months 5. The majority (90%) of non-seminoma testicular cancer patients who develop a recurrence will experience it within the first 2 years after definitive therapy 5. Therefore, the recommended surveillance schedule is relatively intense for the first 2 years. For most patients, this means a physical examination of the remaining testicle, a chest x-ray, CT scan, and serum marker levels every 2 months. Late relapses after the first 2 years are relatively uncommon at only 2% to 4% 5. When present, such recurrences are typically found in the retroperitoneum 85. However, rare instances of very late relapses have been reported up to 24 years after initial definitive therapy 86.
A more liberal monitoring plan has been suggested for low-risk patients with clinical stage 1 non-seminoma testicular cancer who may be able to avoid most of the routine surveillance CT scans, just performing them at 3 and 12 months rather than the standard scan every 2 months 87. While promising, this is not yet the standard, accepted, or recommended protocol 5.
Complete, detailed, and comprehensive reviews of the most recent recommended surveillance guidelines can be found on the NCCN website under “Guidelines for Testicular Cancer” or the American Urological Association Guidelines on Early Stage (Stage I – IIB) Testicular Cancer 78.
Stage 0 testicular cancer treatment
Treatment of stage 0 testicular cancer may include the following:
- Radiation therapy.
- Surveillance.
- Surgery to remove the testicle.
Stage 1 nonseminoma testicular cancer treatment
Stage 1A:
- Recommendations after orchiectomy include surveillance in compliant patients or use of nerve-sparing retroperitoneal lymph node dissection (RPLND) 88, 78.
- If the dissected lymph nodes are not involved with tumor, there is no need for adjuvant chemotherapy after retroperitoneal lymph node dissection (RPLND)
- If resected lymph nodes do involve the tumor, a decision must be made as to whether to give adjuvant chemotherapy based on degree of nodal involvement; chemotherapy is preferred in patients with pN2 or pN3 disease 88
- Recommended chemotherapy includes two cycles of EP (etoposide 100 mg/m2 IV on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5; every 21 day) OR one cycle of BEP (bleomycin 30 U IV on days 1, 8, and 15 plus etoposide 100 mg/m² IV on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5; every 21 day) 89
Stage 1B:
- Treatment recommendations after orchidectomy include nerve-sparing retroperitoneal lymph node dissection (RPLND) or chemotherapy
- Chemotherapy includes one or two cycles of BEP (bleomycin 30 U IV on days 1, 8, and 15 plus etoposide 100 mg/m² IV on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5; every 21 day) OR surveillance for compliant patients with T2 or T3 disease
- Adjuvant therapy after a nerve-sparing retroperitoneal lymph node dissection (RPLND) is the same as in stage 1A
Stage 1S:
- Persistent elevation of seurm tumor marker levels characterizes this stage; patients are treated with four cycles of EP (etoposide 100 mg/m² IV on days 1-5 plus cisplatin 20 mg/m2 IV on days 1-5; every 12 day ) 90 OR 3 cycles of BEP (bleomycin 30 U IV on days 1, 8, and 15 plus etoposide 100 mg/m² IV on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5; every 21 day)
Stage 2 nonseminoma testicular cancer treatment
Stage 2 nonseminoma is highly curable (>95%) 21. Men with stage 2 nonseminoma testicular cancer and persistently elevated serum tumor markers are generally treated as having stage 3 disease and receive chemotherapy. For men with normal serum tumor markers after orchiectomy, nonseminomas are divided into stages 2A, 2B, and 2C for treatment purposes. In general, stage 2A patients undergo retroperitoneal lymph node dissection (RPLND) to confirm the staging. As many as 40% of clinical stage 2A patients will have benign findings at retroperitoneal lymph node dissection (RPLND) and will be restaged as having pathological stage 1 nonseminoma testicular cancer 58. Retroperitoneal lymph node dissection (RPLND) can therefore prevent a significant number of patients with clinical stage 2A nonseminoma testicular cancer from receiving unnecessary chemotherapy.
Stage 2A
Treatment recommendations are based on serum tumor marker results after orchiectomy.
Patients with normal marker levels should be treated with nerve-sparing retroperitoneal lymph node dissection (RPLND) or chemotherapy with four cycles of EP (etoposide 100 mg/m² IV on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5; every 12 day) 90
OR
Three cycles of BEP (bleomycin 30 U IV on days 1, 8, and 15 plus etoposide 100 mg/m² IV on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5; every 21 day) 90
In patients who underwent retroperitoneal lymph node dissection (RPLND) and were found to have pN1 or pN2 disease, adjuvant chemotherapy with two cycles of EP or two cycles of BEP is preferred 90
Those with pN3 disease should receive three cycles of BEP or four cycles of EP
Stage 2B
As with stage 2A, treatment recommendations are based on serum tumor marker levels.
Treatment for patients with negative tumor markers but lymph node metastases within lymphatic drainage sites is nerve-sparing retroperitoneal lymph node dissection (RPLND) plus adjuvant therapy (as in stage 2A) or chemotherapy. Chemotherapy includes four cycles of EP (etoposide 100 mg/m² IV on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5; every 21 day) 90
OR
Three cycles of BEP (bleomycin 30 U IV on days 1, 8, and 15 plus etoposide 100 mg/m² IV on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5; every 21 day) 90
For patients with multifocal symptomatic disease or lymph node metastases within lymphatic drainage sites, recommended treatment is chemotherapy with four cycles of EP and three cycles of BEP; nerve-sparing retroperitoneal lymph node dissection (RPLND) is not recommended.
Stage 3 nonseminoma testicular cancer treatment
The International Germ Cell Cancer Consensus Group (IGCCCG) developed a risk classification for advanced nonseminoma testicular cancer based on identifying clinically independent prognostic features, including extent of disease and levels of serum tumor markers 47. In testicular cancer patients who have undergone orchiectomy, serum tumor markers are used to determine risk classification 47.
Patients with disseminated nonseminomas can be divided into good-, intermediate-, and poor-risk groups based on whether nonpulmonary visceral metastases are present, the site of the primary tumor (i.e., mediastinal vs. either gonadal or retroperitoneal), and the level of serum tumor markers 47:
- Poor-risk: Men with mediastinal primary tumors, nonpulmonary visceral metastases, or very highly elevated serum tumor markers are considered to be at poor risk. For more information, see the Stage Information for Testicular Cancer section.
- Intermediate-risk: Men with intermediate tumor markers levels are considered to be at intermediate risk.
- Good-risk: Men with good-risk disease have a testis or retroperitoneal primary tumor, metastases limited to lymph nodes and/or lungs, and tumor markers that are in the good-risk range.
In the 1997 analysis that established these risk groups, the 5-year overall survival rates were 92%, 80% and 48% in the good-, intermediate-, and poor-risk groups, respectively 21. The disease-free survival rates were 89%, 75% and 41% in the good-, intermediate-, and poor-risk groups, respectively 21. However, a 2006 pooled analysis of chemotherapy trials reported improved outcomes compared with the 1997 paper: survival rates in the good-, intermediate-, and poor-risk groups were 94%, 83%, and 71%, respectively 22.
Stage 3A (good risk)
Good risk is classified as the presence of testicular or retroperitoneal primary tumors with no nonpulmonary visceral metastases and S1 markers; 5-year progression-free survival (PFS) is 89%; 5-year survival is 92-94% 91.
- Primary chemotherapy includes four cycles of EP (etoposide 100 mg/m² IV on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5; every 21 day) 90
OR
- 3 cycles of BEP (bleomycin 30 U IV on days 1,8, and 15 plus etoposide 100 mg/m² IV on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5; every 21 day [5-day schedule]; OR bleomycin 30 U IV on days 1, 8, and 15 plus etoposide 165 mg/m² IV on days 1-3 plus cisplatin 50 mg/m2 IV on days 1-2; every 21 day [3-day schedule]) 92
Stage 3B (intermediate risk)
Intermediate risk is classified as presence of testicular or retroperitoneal primary tumors with no nonpulmonary visceral metastases and S2 markers; 5-year progression-free survival (PFS) is 75%; 5-year survival is 80-83% 91.
Patients with stage 3B intermediate risk should be treated with 4 cycles of BEP (bleomycin 30 U IV on days 1, 8, and 15 plus etoposide 100 mg/m² IV on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5; every 21 day [5-day schedule]; OR bleomycin 30 U IV on days 1, 8, and 15 plus etoposide 165 mg/m² IV on days 1-3 plus cisplatin 50 mg/m² IV on days 1-2; every 21 day [3-day schedule]) 92
Tumor markers should be measured immediately before each cycle of chemotherapy regimen and ongoing response should be assessed via continued reduction in tumor marker levels
In patients with intermediate-risk disease, chemotherapy can be followed by surveillance or retroperitoneal lymph node dissection (RPLND) for those with complete tumor response and negative markers
Residual masses in patients with normal marker levels should be resected; depending on the pathology, further chemotherapy should be considered with regimens such as VIP (etoposide 75 mg/m2 IV on days 1-5 plus ifosfamide 1.2 g/m² IV on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5 plus mesna 400 mg/m² IV infused over 30 min before ifosfamide, then 4 hour and 8 hour after the start of each ifosfamide dose (or 1200 mg/m²/24h) on days 1-5; every 21 day for four cycles) 93, 24
If marker levels are persistently abnormal after the completion of initial chemotherapy, second-line therapy should be considered.
Stage 3C (poor risk)
Poor risk is classified as the presence of a mediastinal primary tumor, nonpulmonary visceral metastases, or S3 markers; 5-year progression-free survival (PFS) is 41%; 5-year survival is 71% 91.
Patients with stage 3C poor risk should be treated with four cycles of BEP (bleomycin 30 U IV on days 1, 8, and 15 plus etoposide 100 mg/m² IV on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5; every 21 day [5-day schedule]; OR bleomycin 30 U IV on days 1, 8 and 15 plus etoposide 165 mg/m² IV on days 1-3 plus cisplatin 50 mg/m² IV on days 1-2; every 21 day [3-day schedule]) 92
In patients with brain metastases, cisplatin-based chemotherapy should be combined with surgery and radiation therapy as clinically indicated.
Tumor markers should be measured immediately before each cycle of chemotherapy, and ongoing response should be assessed via continued reduction in tumor markers.
In patients with poor-risk disease, chemotherapy can be followed by surveillance or retroperitoneal lymph node dissection (RPLND) for those with complete tumor response and negative tumor markers.
Residual masses with normal serum tumor markers should be resected; depending on the pathology, further chemotherapy should be considered with regimens such as VIP (etoposide 75 mg/m² IV on days 1-5 plus ifosfamide 1.2 g/m² IV on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5 plus mesna 400 mg/m² IV infused over 30 min before ifosfamide, then 4 hour and 8 hour after the start of each ifosfamide dose (or 1200 mg/m²/24h) on days 1-5; every 21 day for four cycles) 93, 24
If serum tumor markers are persistently abnormal after the completion of initial chemotherapy, second-line/salvage therapy should be considered 91.
Second-line treatment recommendations for metastatic disease
Favorable prognosis
Patients with a favorable prognosis defined as those with low marker levels, low tumor volume, and complete response to first-line therapy 88, should be treated with conventional doses of VeIP (vinblastine 0.11 mg/kg IV push on days 1-2 plus ifosfamide 1200 mg/m² IV on days 1-5 plus mesna 400 mg/m² IV infused over 30 min before ifosfamide, then 4 hour and 8 hour after the start of each ifosfamide dose (or 1200 mg/m²/24h) on days 1-5 plus cisplatin 20 mg/m² IV on days 1-5; every 21 day for four cycles) 94
OR
TIP (paclitaxel 250 mg/m2 IV continuous infusion over 24 hour on day 1 plus ifosfamide 1500 mg/m² IV on days 2-5 plus cisplatin 25 mg/m² IV on days 2-5 plus mesna 500 mg/m² IV infused over 30 min before ifosfamide and then 4 hour and 8 hour after each dose of ifosfamide on days 2-5; every 21 day for four cycles) 95
OR
High-dose chemotherapy with carboplatin 700 mg/m² IV plus etoposide 750 mg/m² IV; administer 5, 4, and 3 days before peripheral blood stem cell infusion for two cycles 96
OR
Paclitaxel 200 mg/m² IV over 24h on day 1 plus ifosfamide 2000 mg/m² over 4h with mesna protection on days 2-4; every 14 day for two cycles; followed by carboplatin area under the curve (AUC) 7-8 IV over 60 min on days 1-3 plus etoposide 400 mg/m² IV on days 1-3; administer with peripheral blood stem cell support every 14-21 day for three cycles 97
Unfavorable prognosis
Unfavorable prognosis is defined by incomplete response, high marker levels, high tumor volume, an extratesticular primary tumor, and late relapse 88. Treat these patients with chemotherapy; clinical trials or conventional-dose VeIP or TIP regimens are preferred; consider high-dose chemotherapy (as above). Consider surgical salvage therapy or best supportive care 88.
Persistent or recurrent disease treatment
All patients should be considered for palliative chemotherapy or radiation therapy.
Patients who have been pretreated, cisplatin-resistant disease, or have refractory germ cell tumors may be given combination chemotherapy with GEMOX (gemcitabine 1000 or 1250 mg/m² IV on days 1 and 8 plus oxaliplatin 130 mg/m² IV on day 1; every 21 day) 98, 99
OR
High-dose chemotherapy and stem-cell rescue 96, 100
Non-seminoma testicular cancer prognosis
In the past, testicular cancers were responsible for 11% of all deaths from cancer in men between 25 and 34, with an overall 5-year survival rate of only 64% 101. After excision and chemotherapy, the current overall prognosis is extremely good, with a 5-year overall survival rate of about 96% 102. Death rates have been stable over 2014–2020, with a 5-year survival rate of 95.2% 32.
The prognosis and treatment options of patients with testicular germ cell tumors is determined by the following factors:
- Stage of the cancer (whether it is in or near the testicle or has spread to other places in the body, and blood levels of AFP, beta-hCG, and LDH).
- Histology (seminoma vs. nonseminoma).
- Size of the tumor.
- Number and size of retroperitoneal lymph nodes.
- For nonseminomas, the degree to which serum tumor markers are elevated 47.
For men with disseminated seminomas, the main adverse prognostic variable is the presence of metastases to organs other than the lungs (e.g., bone, liver, or brain). For men with disseminated nonseminomas, the following variables are independently associated with poor prognosis 21:
- Metastases to organs other than the lungs.
- Highly elevated serum tumor markers.
- Tumor that originated in the mediastinum rather than the testis.
Even patients with widespread metastases at presentation, including those with brain metastases, may have curable disease and should be treated with this intent 103.
Testicular tumors are divided into 3 groups, based on how well the tumors are expected to respond to treatment 47, 104:
- Good Prognosis (disease-free survival at 5 years are roughly 90%)
- Intermediate Prognosis (disease-free survival at 5 years are roughly 75%)
- Poor Prognosis (disease-free survival at 5 years are roughly 50%)
Disease-free survival varies by stage and risk, as follows 104:
- Stage 1 nonseminoma testicular cancer is highly curable (>99%). Orchiectomy alone will cure about 70% of patients, but the remaining 30% will relapse and require additional treatment. The relapses are highly curable, and postorchiectomy surveillance is a standard treatment option, but some doctor and patients prefer to reduce the risk of relapse by having the patient undergo either a retroperitoneal lymph node dissection (RPLND) or one or two cycles of chemotherapy. Each of these three approaches has unique advantages and disadvantages, and none has been shown to result in longer survival or superior quality of life. Patients with stage 1 non-seminoma testicular cancer typically achieve a 98% disease-free survival rate at 5 years.
- Stage 2 nonseminoma is highly curable (>95%). Men with stage 2 nonseminoma testicular cancer and persistently elevated serum tumor markers are generally treated as having stage 3 non-seminoma testicular cancer and receive chemotherapy. For men with normal serum tumor markers after orchiectomy, nonseminomas are divided into stages 2A, 2B, and 2C for treatment purposes. In general, stage 2A patients undergo retroperitoneal lymph node dissection (RPLND) to confirm the staging. As many as 40% of clinical stage 2A patients will have benign findings at retroperitoneal lymph node dissection (RPLND) and will be restaged as having pathological stage 1 disease 58. Retroperitoneal lymph node dissection (RPLND) can therefore prevent a significant number of patients with clinical stage 2A disease from receiving unnecessary chemotherapy. In contrast, patients with stage 2B and 2C nonseminoma are usually treated with systemic chemotherapy for disseminated disease because these patients have a higher relapse rate after retroperitoneal lymph node dissection (RPLND). One study reported that by limiting retroperitoneal lymph node dissection (RPLND) to patients with earlier stage 2 disease and normal serum tumor markers, 5-year disease-free survival rates increased from 78% to 100% after retroperitoneal lymph node dissection, while disease-free survival rates did not change significantly among stage 2 patients receiving chemotherapy (100% vs. 98%) 105. However, the question of whether to treat patients with stage 2 nonseminoma germ cell tumors with retroperitoneal lymph node dissection or chemotherapy has never been subjected to a randomized trial. Patients with stage 2A and 2B non-seminoma testicular cancer typically achieve a 92% disease-free survival rate at 5.5 years. Patients with stage 2C non-seminoma testicular cancer can expect an approximately 92% overall survival rate at 5 years.
- Patients with stage 3 non-seminoma testicular cancer classified as low-risk have a 92% overall survival rate at 5 years; intermediate-risk patients have an 80% overall survival rate at 5 years; high-risk patients have a 48% overall survival rate at 5 years.
In a study from Norway, Mykletun et al 106 reported that, at a mean of 11 years of follow-up, men who survived testicular cancer had no clinically significant difference in quality of life compared with age-matched controls. Overall, only minimal differences were seen in quality of life among recipients of different testicular cancer treatment modalities. The apparently excellent quality-of-life results of this study may offer some reassurance regarding the potential for complications and challenges to patients facing the diagnosis and treatment of testicular cancer.
Testicular cancer can usually be cured in patients who receive adjuvant (add-on) chemotherapy or radiation therapy after their primary treatment.
Certain treatments for testicular cancer can cause infertility that may be permanent. Patients who may wish to have children should consider sperm banking before having treatment. Sperm banking is the process of freezing sperm and storing it for later use.
Good Prognosis
For nonseminoma testicular cancer, all of the following must be true:
- The tumor is found only in the testicle or in the retroperitoneum (area outside or behind the abdominal wall); and
- The tumor has not spread to organs other than the lungs; and
- The levels of all the tumor markers are slightly above normal.
- AFP < 1000 ng/mL and
- hCG < 5000 IU/L (1000ng/mL) and
- LDH < 1.5x upper limit of normal
Intermediate Prognosis
For nonseminoma testicular cancer, all of the following must be true:
- The tumor is found in one testicle only or in the retroperitoneum (area outside or behind the abdominal wall); and
- The tumor has not spread to organs other than the lungs; and
- The level of any one of the tumor markers is more than slightly above normal.
- AFP > 1000 ng/mL and
- hCG > 5000 IU/L and < 50’000 IU/L
- LDH > 1.5x and < 10x upper limit of normal
Poor Prognosis
For nonseminoma testicular cancer, at least one of the following must be true:
- The tumor is in the center of the chest between the lungs; or
- The tumor has spread to organs other than the lungs; or
- The level of any one of the tumor markers is high.
- AFP > 10’000 ng/mL or
- hCG > 50’000 IU/L (10’000 ng/mL) or
- LDH > 10x upper limit of normal
Non-seminoma testicular cancer survival rate
According to information from the Surveillance, Epidemiology, and Rend-Results (SEER) 2024 database, the 5-year relative survival rates for testicular cancer are 32:
- Because testicular cancer usually can be treated successfully, a man’s lifetime risk of dying from this cancer is very low. 5-Year Relative Survival is 95%. A relative survival rate compares people with the same type and stage of cancer to those in the overall population. For example, if the 5-year relative survival rate for a specific stage of testicular cancer is 95%, it means that people who have that cancer are, on average, about 95% as likely as people who don’t have that cancer to live for at least 5 years after being diagnosed.
- Localized disease (stage 1) 99%
- Regional disease (stage 2) 96%
- Distant disease (Stage 3) 73%
Patients who have been cured of testicular cancer have approximately a 2% cumulative risk of developing cancer in the opposite testicle during the 15 years after initial diagnosis 107, 108. Within this range, men with nonseminomatous primary tumors appear to have a lower risk of subsequent opposite testis tumors than men with seminomas 21.
Patients with testicular cancer are at an increased risk of secondary cancers because of their young age at diagnosis, high cure rate, and exposure to radiation, chemotherapy, or both 109. Because most patients with testicular cancer who receive adjuvant (add-on) chemotherapy or radiation therapy are curable, it is necessary to be aware of possible long-term effects of the various treatment modalities, such as the following:
- Fertility: Many men patients have oligospermia or sperm abnormalities before therapy, but semen analysis results generally become more normal after treatment. The impact of standard chemotherapy on fertility in patients with testicular cancer is not well defined, although it is well documented that most men can father children after treatment, often without the use of cryopreserved semen 21. In two large studies, roughly 70% of patients fathered children after treatment for testicular cancer 110, 83. The likelihood of recovering fertility is related to the type of treatment received. Children fathered by men treated for testicular cancer do not appear to have an increased risk of congenital malformations, but the data are not adequate to properly investigate this issue 111, 41. It is recommended that men wait at least 3 months after completing chemotherapy before conceiving a child (unless using cryopreserved sperm collected before chemotherapy was administered) 41. Radiation therapy, used to treat pure seminomatous testicular cancers, can cause fertility problems because of radiation scatter to the remaining testicle during radiation therapy to retroperitoneal lymph nodes 112. Depending on scatter dose, sperm counts fall after radiation therapy but may recover over the course of 1 to 2 years. Shielding techniques can be used to decrease the radiation scatter to the remaining normal testicle. Because chemotherapy, retroperitoneal lymph node dissection, and radiation therapy can each result in infertility, men can be offered the opportunity to bank sperm before undergoing any treatment for testicular cancer other than orchiectomy 21.
- Secondary leukemias: Several reports of elevated risk of secondary acute leukemia, primarily nonlymphocytic, have appeared 113, 114. An increased risk of leukemia has been associated with platinum-based chemotherapy and radiation therapy 113. Etoposide-containing regimens are also associated with a risk of secondary acute leukemias, usually in the myeloid lineage, and with a characteristic 11q23 translocation 115, 116. Etoposide-associated leukemias typically occur sooner after therapy than alkylating agent-associated leukemias and often show balanced chromosomal translocations on the long arm of chromosome 11. Standard etoposide dosages (<2 g/m² cumulative dose) are associated with a relative risk of 15 to 25, but this translates into a cumulative incidence of leukemia of less than 0.5% at 5 years. Preliminary data suggest that cumulative doses of more than 2 g/m² of etoposide may confer higher risk 21.
- Kidney function: Minor decreases in creatinine clearance occur (about a 15% decrease, on average) during platinum-based therapy, but they appear to remain stable in the long term, without significant deterioration 117.
- Lung function: A study of pulmonary function tests in 1,049 long-term survivors of testicular cancer reported a cisplatin-dose-dependent increase in the incidence of restrictive lung disease 118. Whereas men receiving up to 850 mg of cisplatin had a normal risk of restrictive lung disease, men who received over 850 mg of cisplatin had a threefold increased risk. In absolute terms, patients who received no chemotherapy had an incidence of restrictive lung disease of less than 8%, whereas the incidence of restrictive lung disease among those receiving over 850 mg of cisplatin was nearly 18%. However, only 9.5% of those with pulmonary function testing indicative of restrictive lung disease reported dyspnea. Although cisplatin was more strongly associated with decreased lung function in this study, cumulative bleomycin dose was also associated with a decline in forced vital capacity and the 1-second forced expiratory volume (FEV1) but not with restrictive lung disease 21.
- Hearing: Bilateral hearing deficits occur with cisplatin-based chemotherapy, but they generally occur at sound frequencies of 4 kHz to 8 kHz, which is outside the range of conversational tones. Therefore, hearing aids are rarely required if standard doses of cisplatin are given 117.
Although acute pulmonary toxic effects may occur with bleomycin, they are rarely fatal at total cumulative doses of less than 400 units. Because life-threatening pulmonary toxic effects can occur, the drug should be discontinued if early signs of pulmonary toxicity develop 21. Although decreases in pulmonary function are frequent, they are rarely symptomatic and are reversible after chemotherapy ends. Survivors of testis cancer who were treated with chemotherapy have been reported to be at increased risk of death from respiratory diseases, but it is unknown whether this finding is related to bleomycin exposure 119.
Men with testicular cancer who have been treated with radiation therapy and/or chemotherapy are at increased risk of cardiovascular diseases 120, 121, 122. Other studies have reported that chemotherapy for testicular cancer is associated with an increased risk of developing metabolic syndrome and hypogonadism 123, 124. Moreover, an international population-based study reported that men treated with either radiation therapy or chemotherapy were at increased risk of death from cardiovascular diseases 119.
In a retrospective series of 992 patients treated for testicular cancer between 1982 and 1992, cardiac events were increased approximately 2.5-fold in patients treated with radiation therapy and/or chemotherapy, compared with those who underwent surveillance for a median of 10.2 years. The actuarial risks of cardiac events were 7.2% for patients who received radiation therapy (92% of whom did not receive mediastinal radiation therapy), 3.4% for patients who received chemotherapy (primarily platinum-based), 4.1% for patients who received combined therapy, and 1.4% for patients who underwent surveillance management after 10 years of follow-up 121.
A population-based retrospective study of 2,339 testicular cancer survivors in the Netherlands, treated between 1965 and 1995 and followed for a median of 18.4 years, found that the overall incidence of coronary heart disease (i.e., myocardial infarction and/or angina pectoris) was increased 1.17 times compared with the general population 122. Patients who received radiation therapy to the mediastinum had a 2.5-fold increased risk of coronary heart disease, and those who also received chemotherapy had an almost threefold increased risk. Patients who were treated with infradiaphragmatic radiation therapy alone had no significantly increased risk of coronary heart disease. The older chemotherapy regimen of cisplatin, vinblastine, and bleomycin, used until the mid-1980s, was associated with a significant 1.9-fold increased risk of cardiovascular disease (i.e., heart attack, angina pectoris, and heart failure combined) 21. The newer regimen of bleomycin, etoposide, and cisplatin was associated with a borderline significant 1.5-fold increased risk of cardiovascular disease 21. Similarly, an international pooled analysis of population-based databases reported that the risk of death from circulatory disease was increased in men treated with chemotherapy (standardized mortality ratio [SMR] = 1.58) or radiation therapy (SMR = 1.70) 119.
- Dehner LP. Gonadal and extragonadal germ cell neoplasia of childhood. Hum Pathol. 1983 Jun;14(6):493-511. doi: 10.1016/s0046-8177(83)80004-5[↩][↩][↩]
- McIntyre A, Gilbert D, Goddard N, Looijenga L, Shipley J. Genes, chromosomes and the development of testicular germ cell tumors of adolescents and adults. Genes Chromosomes Cancer. 2008 Jul;47(7):547-57. doi: 10.1002/gcc.20562[↩][↩][↩]
- What Is Testicular Cancer? https://www.cancer.org/cancer/types/testicular-cancer/about/what-is-testicular-cancer.html[↩]
- Stephenson AJ, Gilligan TD. Neoplasms of the Testis. Wein AJ, Kavoussi LR, Partin AW, Peters CA, eds. Campbell-Walsh Urology. 11th ed. Philadelphia, PA: Elsevier; 2016. 784-811.[↩]
- Nauman M, Leslie SW. Nonseminomatous Testicular Tumors. [Updated 2023 Aug 14]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK568754/[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Ali TZ, Parwani AV. Benign and Malignant Neoplasms of the Testis and Paratesticular Tissue. Surg Pathol Clin. 2009 Mar;2(1):61-159. doi: 10.1016/j.path.2008.08.007[↩]
- Alvarado-Cabrero I, Hernández-Toriz N, Paner GP. Clinicopathologic analysis of choriocarcinoma as a pure or predominant component of germ cell tumor of the testis. Am J Surg Pathol. 2014 Jan;38(1):111-8. doi: 10.1097/PAS.0b013e3182a2926e[↩][↩]
- Allen MS. Presentation and management of benign mediastinal teratomas. Chest Surg Clin N Am. 2002 Nov;12(4):659-64, vi. doi: 10.1016/s1052-3359(02)00036-4[↩]
- Williamson SR, Delahunt B, Magi-Galluzzi C, Algaba F, Egevad L, Ulbright TM, Tickoo SK, Srigley JR, Epstein JI, Berney DM; Members of the ISUP Testicular Tumour Panel. The World Health Organization 2016 classification of testicular germ cell tumours: a review and update from the International Society of Urological Pathology Testis Consultation Panel. Histopathology. 2017 Feb;70(3):335-346. doi: 10.1111/his.13102[↩][↩][↩]
- Kattuoa Ml, Dunton CJ. Yolk Sac Tumors. [Updated 2023 Feb 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK563163/[↩]
- Chung P, Warde P. Testicular cancer: germ cell tumours. BMJ Clin Evid. 2016 Jan 7;2016:1807. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4704678/[↩][↩][↩][↩][↩]
- Hainsworth JD, Greco FA. Malignant Germ Cell Tumors. In: Kufe DW, Pollock RE, Weichselbaum RR, et al., editors. Holland-Frei Cancer Medicine. 6th edition. Hamilton (ON): BC Decker; 2003. Available from: https://www.ncbi.nlm.nih.gov/books/NBK12497/[↩][↩][↩]
- Ries LAG, Melbert D, Krapcho M, et al.: SEER Cancer Statistics Review, 1975-2005. National Cancer Institute, 2007.[↩]
- Krege S, Beyer J, Souchon R, et al. European consensus conference on diagnosis and treatment of germ cell cancer: a report of the second meeting of the European Germ Cell Cancer Consensus group (EGCCCG): part I. Eur Urol. 2008 Mar;53(3):478-96. doi: 10.1016/j.eururo.2007.12.024[↩]
- Groll RJ, Warde P, Jewett MA. A comprehensive systematic review of testicular germ cell tumor surveillance. Crit Rev Oncol Hematol. 2007 Dec;64(3):182-97. doi: 10.1016/j.critrevonc.2007.04.014[↩]
- Neill M, Warde P, Fleshner N. Management of low-stage testicular seminoma. Urol Clin North Am. 2007 May;34(2):127-36; abstract vii-viii. doi: 10.1016/j.ucl.2007.02.009[↩]
- Tandstad T, Dahl O, Cohn-Cedermark G, Cavallin-Stahl E, Stierner U, Solberg A, Langberg C, Bremnes RM, Laurell A, Wijkstrøm H, Klepp O. Risk-adapted treatment in clinical stage I nonseminomatous germ cell testicular cancer: the SWENOTECA management program. J Clin Oncol. 2009 May 1;27(13):2122-8. doi: 10.1200/JCO.2008.18.8953. Epub 2009 Mar 23. Erratum in: J Clin Oncol. 2009 Jul 1;27(19):3263. Erratum in: J Clin Oncol. 2010 Mar 10;28(8):1438. Dosage error in article text.[↩]
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021 May;71(3):209-249. doi: 10.3322/caac.21660[↩]
- Ghazarian AA, et al. Future of testicular germ cell tumor incidence in the United States: forecast through 2026. Cancer. 2017;123(12):2320–2328. doi: 10.1002/cncr.30597[↩]
- Nauman M, Leslie SW. Nonseminomatous Testicular Tumors. [Updated 2023 Aug 14]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK568754[↩]
- PDQ Adult Treatment Editorial Board. Testicular Cancer Treatment (PDQ®): Health Professional Version. 2024 Mar 15. In: PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute (US); 2002-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK65777/[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- van Dijk MR, Steyerberg EW, Habbema JD. Survival of non-seminomatous germ cell cancer patients according to the IGCC classification: An update based on meta-analysis. Eur J Cancer. 2006 May;42(7):820-6. doi: 10.1016/j.ejca.2005.08.043[↩][↩][↩]
- Williams SD, Birch R, Einhorn LH, Irwin L, Greco FA, Loehrer PJ. Treatment of disseminated germ-cell tumors with cisplatin, bleomycin, and either vinblastine or etoposide. N Engl J Med. 1987 Jun 4;316(23):1435-40. doi: 10.1056/NEJM198706043162302[↩]
- Nichols CR, Catalano PJ, Crawford ED, Vogelzang NJ, Einhorn LH, Loehrer PJ. Randomized comparison of cisplatin and etoposide and either bleomycin or ifosfamide in treatment of advanced disseminated germ cell tumors: an Eastern Cooperative Oncology Group, Southwest Oncology Group, and Cancer and Leukemia Group B Study. J Clin Oncol. 1998 Apr;16(4):1287-93. doi: 10.1200/JCO.1998.16.4.1287[↩][↩][↩]
- Hinton S, Catalano PJ, Einhorn LH, Nichols CR, David Crawford E, Vogelzang N, Trump D, Loehrer PJ Sr. Cisplatin, etoposide and either bleomycin or ifosfamide in the treatment of disseminated germ cell tumors: final analysis of an intergroup trial. Cancer. 2003 Apr 15;97(8):1869-75. doi: 10.1002/cncr.11271[↩]
- de Wit R, Louwerens M, de Mulder PH, Verweij J, Rodenhuis S, Schornagel J. Management of intermediate-prognosis germ-cell cancer: results of a phase I/II study of Taxol-BEP. Int J Cancer. 1999 Dec 10;83(6):831-3. doi: 10.1002/(sici)1097-0215(19991210)83:6<831::aid-ijc24>3.0.co;2-o[↩]
- Childhood Extracranial Germ Cell Tumors Treatment (PDQ®)–Health Professional Version. https://www.cancer.gov/types/extracranial-germ-cell/hp/germ-cell-treatment-pdq[↩]
- Testicular Cancer Treatment (PDQ®)–Health Professional Version. https://www.cancer.gov/types/testicular/hp/testicular-treatment-pdq#_390_toc[↩]
- Lamichhane A, Mukkamalla SKR. Seminoma. [Updated 2023 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK560513/[↩][↩]
- Ghazarian AA, Trabert B, Devesa SS, McGlynn KA. Recent trends in the incidence of testicular germ cell tumors in the United States. Andrology. 2015 Jan;3(1):13-8. doi: 10.1111/andr.288[↩]
- Nigam M, Aschebrook-Kilfoy B, Shikanov S, Eggener S. Increasing incidence of testicular cancer in the United States and Europe between 1992 and 2009. World J Urol. 2015 May;33(5):623-31. doi: 10.1007/s00345-014-1361-y[↩]
- Testicular Cancer — Cancer Stat Facts. https://seer.cancer.gov/statfacts/html/testis.html[↩][↩][↩]
- PDQ Adult Treatment Editorial Board. Testicular Cancer Treatment (PDQ®): Patient Version. 2023 May 17. In: PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute (US); 2002-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK65847/[↩]
- Lutke Holzik MF, Rapley EA, Hoekstra HJ, Sleijfer DT, Nolte IM, Sijmons RH. Genetic predisposition to testicular germ-cell tumours. Lancet Oncol. 2004 Jun;5(6):363-71. doi: 10.1016/S1470-2045(04)01493-7[↩]
- Pettersson A, Richiardi L, Nordenskjold A, Kaijser M, Akre O. Age at surgery for undescended testis and risk of testicular cancer. N Engl J Med. 2007 May 3;356(18):1835-41. doi: 10.1056/NEJMoa067588[↩]
- Skakkebaek NE, Rajpert-De Meyts E, Buck Louis GM, Toppari J, Andersson AM, Eisenberg ML, Jensen TK, Jørgensen N, Swan SH, Sapra KJ, Ziebe S, Priskorn L, Juul A. Male Reproductive Disorders and Fertility Trends: Influences of Environment and Genetic Susceptibility. Physiol Rev. 2016 Jan;96(1):55-97. doi: 10.1152/physrev.00017.2015[↩]
- Oldenburg J, Berney DM, Bokemeyer C, Climent MA, Daugaard G, Gietema JA, De Giorgi U, Haugnes HS, Huddart RA, Leão R, Sohaib A, Gillessen S, Powles T; ESMO Guidelines Committee. Electronic address: [email protected]; EURACAN. Testicular seminoma and non-seminoma: ESMO-EURACAN Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2022 Apr;33(4):362-375. doi: 10.1016/j.annonc.2022.01.002[↩][↩]
- Rajpert-De Meyts E, McGlynn KA, Okamoto K, Jewett MA, Bokemeyer C. Testicular germ cell tumours. Lancet. 2016 Apr 23;387(10029):1762-74. doi: 10.1016/S0140-6736(15)00991-5[↩]
- Bosl GJ, Vogelzang NJ, Goldman A, Fraley EE, Lange PH, Levitt SH, Kennedy BJ. Impact of delay in diagnosis on clinical stage of testicular cancer. Lancet. 1981 Oct 31;2(8253):970-3. doi: 10.1016/s0140-6736(81)91165-x[↩]
- Djaladat H, Burner E, Parikh PM, Beroukhim Kay D, Hays K. The Association Between Testis Cancer and Semen Abnormalities Before Orchiectomy: A Systematic Review. J Adolesc Young Adult Oncol. 2014 Dec 1;3(4):153-159. doi: 10.1089/jayao.2014.0012[↩]
- Spermon JR, Kiemeney LA, Meuleman EJ, Ramos L, Wetzels AM, Witjes JA. Fertility in men with testicular germ cell tumors. Fertil Steril. 2003 Jun;79 Suppl 3:1543-9. doi: 10.1016/s0015-0282(03)00335-2[↩][↩][↩][↩]
- Staples J. Alphafetoprotein, cancer, and benign conditions. Lancet. 1986 Nov 29;2(8518):1277. doi: 10.1016/s0140-6736(86)92700-5[↩]
- Hotakainen K, Ljungberg B, Paju A, Rasmuson T, Alfthan H, Stenman UH. The free beta-subunit of human chorionic gonadotropin as a prognostic factor in renal cell carcinoma. Br J Cancer. 2002 Jan 21;86(2):185-9. doi: 10.1038/sj.bjc.6600050[↩]
- Badia RR, Abe D, Wong D, Singla N, Savelyeva A, Chertack N, Woldu SL, Lotan Y, Mauck R, Ouyang D, Meng X, Lewis CM, Majmudar K, Jia L, Kapur P, Xu L, Frazier AL, Margulis V, Strand DW, Coleman N, Murray MJ, Amatruda JF, Lafin JT, Bagrodia A. Real-World Application of Pre-Orchiectomy miR-371a-3p Test in Testicular Germ Cell Tumor Management. J Urol. 2021 Jan;205(1):137-144. doi: 10.1097/JU.0000000000001337[↩]
- Capelouto CC, Clark PE, Ransil BJ, Loughlin KR. A review of scrotal violation in testicular cancer: is adjuvant local therapy necessary? J Urol. 1995 Mar;153(3 Pt 2):981-5.[↩]
- Sturgeon CM, Duffy MJ, Stenman UH, et al. National Academy of Clinical Biochemistry. National Academy of Clinical Biochemistry laboratory medicine practice guidelines for use of tumor markers in testicular, prostate, colorectal, breast, and ovarian cancers. Clin Chem. 2008 Dec;54(12):e11-79. doi: 10.1373/clinchem.2008.105601[↩]
- International Germ Cell Consensus Classification: a prognostic factor-based staging system for metastatic germ cell cancers. International Germ Cell Cancer Collaborative Group. J Clin Oncol. 1997 Feb;15(2):594-603. doi: 10.1200/JCO.1997.15.2.594[↩][↩][↩][↩][↩][↩][↩]
- Gholam D, Fizazi K, Terrier-Lacombe MJ, Jan P, Culine S, Theodore C. Advanced seminoma–treatment results and prognostic factors for survival after first-line, cisplatin-based chemotherapy and for patients with recurrent disease: a single-institution experience in 145 patients. Cancer. 2003 Aug 15;98(4):745-52. doi: 10.1002/cncr.11574[↩]
- Oliver RT, Mason MD, Mead GM, von der Maase H, Rustin GJ, Joffe JK, de Wit R, Aass N, Graham JD, Coleman R, Kirk SJ, Stenning SP; MRC TE19 collaborators and the EORTC 30982 collaborators. Radiotherapy versus single-dose carboplatin in adjuvant treatment of stage I seminoma: a randomised trial. Lancet. 2005 Jul 23-29;366(9482):293-300. doi: 10.1016/S0140-6736(05)66984-X[↩]
- Weissbach L, Bussar-Maatz R, Mann K. The value of tumor markers in testicular seminomas. Results of a prospective multicenter study. Eur Urol. 1997;32(1):16-22.[↩]
- Venkitaraman R, Johnson B, Huddart RA, Parker CC, Horwich A, Dearnaley DP. The utility of lactate dehydrogenase in the follow-up of testicular germ cell tumours. BJU Int. 2007 Jul;100(1):30-2. doi: 10.1111/j.1464-410X.2007.06905.x[↩][↩][↩]
- Ackers C, Rustin GJ. Lactate dehydrogenase is not a useful marker for relapse in patients on surveillance for stage I germ cell tumours. Br J Cancer. 2006 May 8;94(9):1231-2. doi: 10.1038/sj.bjc.6603087[↩][↩]
- Sohaib SA, Koh DM, Husband JE. The role of imaging in the diagnosis, staging, and management of testicular cancer. AJR Am J Roentgenol. 2008 Aug;191(2):387-95. doi: 10.2214/AJR.07.2758[↩]
- Leibovitch L, Foster RS, Kopecky KK, Donohue JP. Improved accuracy of computerized tomography based clinical staging in low stage nonseminomatous germ cell cancer using size criteria of retroperitoneal lymph nodes. J Urol. 1995 Nov;154(5):1759-63.[↩]
- Chung P, Warde P. Surveillance in stage I testicular seminoma. Urol Oncol. 2006 Jan-Feb;24(1):75-9. doi: 10.1016/j.urolonc.2005.07.007[↩]
- Segal R. Surveillance programs for stage I nonseminomatous germ cell tumors of the testis. Urol Oncol. 2006 Jan-Feb;24(1):68-74. doi: 10.1016/j.urolonc.2005.07.006[↩]
- Warde P, Specht L, Horwich A, Oliver T, Panzarella T, Gospodarowicz M, von der Maase H. Prognostic factors for relapse in stage I seminoma managed by surveillance: a pooled analysis. J Clin Oncol. 2002 Nov 15;20(22):4448-52. doi: 10.1200/JCO.2002.01.038[↩]
- Stephenson AJ, Bosl GJ, Motzer RJ, Kattan MW, Stasi J, Bajorin DF, Sheinfeld J. Retroperitoneal lymph node dissection for nonseminomatous germ cell testicular cancer: impact of patient selection factors on outcome. J Clin Oncol. 2005 Apr 20;23(12):2781-8. doi: 10.1200/JCO.2005.07.132[↩][↩][↩][↩]
- Choueiri TK, Stephenson AJ, Gilligan T, Klein EA. Management of clinical stage I nonseminomatous germ cell testicular cancer. Urol Clin North Am. 2007 May;34(2):137-48; abstract viii. doi: 10.1016/j.ucl.2007.02.001[↩]
- Donohue JP, Thornhill JA, Foster RS, Rowland RG, Bihrle R. Clinical stage B non-seminomatous germ cell testis cancer: the Indiana University experience (1965-1989) using routine primary retroperitoneal lymph node dissection. Eur J Cancer. 1995 Sep;31A(10):1599-604. doi: 10.1016/0959-8049(95)00330-l[↩]
- Huddart SN, Mann JR, Gornall P, Pearson D, Barrett A, Raafat F, Barnes JM, Wallendsus KR. The UK Children’s Cancer Study Group: testicular malignant germ cell tumours 1979-1988. J Pediatr Surg. 1990 Apr;25(4):406-10. doi: 10.1016/0022-3468(90)90381-i[↩]
- Gupta M, Cheaib JG, Patel HD, Sharma R, Zhang A, Bass EB, Pierorazio PM. Diagnosis and Management of Intratubular Germ Cell Neoplasia In Situ: A Systematic Review. J Urol. 2020 Jul;204(1):33-41. doi: 10.1097/JU.0000000000000758[↩]
- Kristianslund S, Fosså SD, Kjellevold K. Bilateral malignant testicular germ cell cancer. Br J Urol. 1986 Feb;58(1):60-3. doi: 10.1111/j.1464-410x.1986.tb05429.x[↩]
- Oldenburg J. Hypogonadism and fertility issues following primary treatment for testicular cancer. Urol Oncol. 2015 Sep;33(9):407-12. doi: 10.1016/j.urolonc.2015.01.014[↩]
- Leibovitch I, Baniel J, Foster RS, Donohue JP. The clinical implications of procedural deviations during orchiectomy for nonseminomatous testis cancer. J Urol. 1995 Sep;154(3):935-9.[↩]
- Albers P, Siener R, Kliesch S, Weissbach L, Krege S, Sparwasser C, Schulze H, Heidenreich A, de Riese W, Loy V, Bierhoff E, Wittekind C, Fimmers R, Hartmann M; German Testicular Cancer Study Group. Risk factors for relapse in clinical stage I nonseminomatous testicular germ cell tumors: results of the German Testicular Cancer Study Group Trial. J Clin Oncol. 2003 Apr 15;21(8):1505-12. doi: 10.1200/JCO.2003.07.169[↩][↩]
- Shayegan B, Carver BS, Stasi J, Motzer RJ, Bosl GJ, Sheinfeld J. Clinical outcome following post-chemotherapy retroperitoneal lymph node dissection in men with intermediate- and poor-risk nonseminomatous germ cell tumour. BJU Int. 2007 May;99(5):993-7. doi: 10.1111/j.1464-410X.2007.06740.x[↩][↩]
- Schirren J, Trainer S, Eberlein M, Lorch A, Beyer J, Bölükbas S. The role of residual tumor resection in the management of nonseminomatous germ cell cancer of testicular origin. Thorac Cardiovasc Surg. 2012 Sep;60(6):405-12. doi: 10.1055/s-0031-1299584[↩][↩]
- Miki T, Mizutani Y, Nakamura T, Kawauchi A, Nagahara A, Nonomura N, Okuyama A. Post-chemotherapy nerve-sparing retroperitoneal lymph node dissection for advanced germ cell tumor. Int J Urol. 2009 Apr;16(4):379-82. doi: 10.1111/j.1442-2042.2009.02251.x[↩]
- Large MC, Sheinfeld J, Eggener SE. Retroperitoneal lymph node dissection: reassessment of modified templates. BJU Int. 2009 Nov;104(9 Pt B):1369-75. doi: 10.1111/j.1464-410X.2009.08861.x[↩]
- Kollmannsberger C, Moore C, Chi KN, Murray N, Daneshmand S, Gleave M, Hayes-Lattin B, Nichols CR. Non-risk-adapted surveillance for patients with stage I nonseminomatous testicular germ-cell tumors: diminishing treatment-related morbidity while maintaining efficacy. Ann Oncol. 2010 Jun;21(6):1296-1301. doi: 10.1093/annonc/mdp473[↩]
- Arai Y, Kawakita M, Okada Y, Yoshida O. Sexuality and fertility in long-term survivors of testicular cancer. J Clin Oncol. 1997 Apr;15(4):1444-8. doi: 10.1200/JCO.1997.15.4.1444[↩]
- Jacobsen KD, Ous S, Waehre H, Trasti H, Stenwig AE, Lien HH, Aass N, Fosså SD. Ejaculation in testicular cancer patients after post-chemotherapy retroperitoneal lymph node dissection. Br J Cancer. 1999 Apr;80(1-2):249-55. doi: 10.1038/sj.bjc.6690347[↩]
- Beck SD, Bey AL, Bihrle R, Foster RS. Ejaculatory status and fertility rates after primary retroperitoneal lymph node dissection. J Urol. 2010 Nov;184(5):2078-80. doi: 10.1016/j.juro.2010.06.146[↩][↩]
- Heidenreich A, Albers P, Hartmann M, Kliesch S, Kohrmann KU, Krege S, Lossin P, Weissbach L; German Testicular Cancer Study Group. Complications of primary nerve sparing retroperitoneal lymph node dissection for clinical stage I nonseminomatous germ cell tumors of the testis: experience of the German Testicular Cancer Study Group. J Urol. 2003 May;169(5):1710-4. doi: 10.1097/01.ju.0000060960.18092.54[↩]
- Spitz A, Wilson TG, Kawachi MH, Ahlering TE, Skinner DG. Vena caval resection for bulky metastatic germ cell tumors: an 18-year experience. J Urol. 1997 Nov;158(5):1813-8. doi: 10.1016/s0022-5347(01)64137-5[↩]
- Beck SD, Lalka SG. Long-term results after inferior vena caval resection during retroperitoneal lymphadenectomy for metastatic germ cell cancer. J Vasc Surg. 1998 Nov;28(5):808-14. doi: 10.1016/s0741-5214(98)70055-2[↩]
- Stephenson A, Eggener SE, Bass EB, Chelnick DM, Daneshmand S, Feldman D, Gilligan T, Karam JA, Leibovich B, Liauw SL, Masterson TA, Meeks JJ, Pierorazio PM, Sharma R, Sheinfeld J. Diagnosis and Treatment of Early Stage Testicular Cancer: AUA Guideline. J Urol. 2019 Aug;202(2):272-281. doi: 10.1097/JU.0000000000000318[↩][↩][↩]
- Stephenson AJ, Sheinfeld J. Management of patients with low-stage nonseminomatous germ cell testicular cancer. Curr Treat Options Oncol. 2005 Sep;6(5):367-77. doi: 10.1007/s11864-005-0040-z[↩]
- Nicolai N, Miceli R, Artusi R, Piva L, Pizzocaro G, Salvioni R. A simple model for predicting nodal metastasis in patients with clinical stage I nonseminomatous germ cell testicular tumors undergoing retroperitoneal lymph node dissection only. J Urol. 2004 Jan;171(1):172-6. doi: 10.1097/01.ju.0000101513.64777.f2[↩]
- Cullen MH, Stenning SP, Parkinson MC, Fossa SD, Kaye SB, Horwich AH, Harland SJ, Williams MV, Jakes R. Short-course adjuvant chemotherapy in high-risk stage I nonseminomatous germ cell tumors of the testis: a Medical Research Council report. J Clin Oncol. 1996 Apr;14(4):1106-13. doi: 10.1200/JCO.1996.14.4.1106[↩]
- Gandini L, Sgrò P, Lombardo F, Paoli D, Culasso F, Toselli L, Tsamatropoulos P, Lenzi A. Effect of chemo- or radiotherapy on sperm parameters of testicular cancer patients. Hum Reprod. 2006 Nov;21(11):2882-9. doi: 10.1093/humrep/del167[↩]
- Huyghe E, Matsuda T, Daudin M, Chevreau C, Bachaud JM, Plante P, Bujan L, Thonneau P. Fertility after testicular cancer treatments: results of a large multicenter study. Cancer. 2004 Feb 15;100(4):732-7. doi: 10.1002/cncr.11950[↩][↩]
- Fankhauser CD, et al. Frequent PD-L1 expression in testicular germ cell tumors. Br J Cancer. 2015;113(3):411–413. doi: 10.1038/bjc.2015.244.[↩]
- Ehrlich Y, Baniel J. Late relapse of testis cancer. Urol Clin North Am. 2007 May;34(2):253-8; abstract x-xi. doi: 10.1016/j.ucl.2007.02.012[↩]
- Riedinger CB, Labbate C, Werntz RP, Eggener SE. Late Relapse of Nonseminomatous Germ Cell Tumor 24 Years Later. Urology. 2018 Dec;122:16-18. doi: 10.1016/j.urology.2018.08.014[↩]
- Rustin GJ, Mead GM, Stenning SP, Vasey PA, Aass N, Huddart RA, Sokal MP, Joffe JK, Harland SJ, Kirk SJ; National Cancer Research Institute Testis Cancer Clinical Studies Group. Randomized trial of two or five computed tomography scans in the surveillance of patients with stage I nonseminomatous germ cell tumors of the testis: Medical Research Council Trial TE08, ISRCTN56475197–the National Cancer Research Institute Testis Cancer Clinical Studies Group. J Clin Oncol. 2007 Apr 10;25(11):1310-5. doi: 10.1200/JCO.2006.08.4889[↩]
- [Guideline] National Comprehensive Cancer Network. Testicular Cancer. NCCN Clinical Practice Guidelines in Oncology. Available at https://www.nccn.org/professionals/physician_gls/pdf/testicular.pdf[↩][↩][↩][↩][↩]
- Böhlen D, Borner M, Sonntag RW, Fey MF, Studer UE. Long-term results following adjuvant chemotherapy in patients with clinical stage I testicular nonseminomatous malignant germ cell tumors with high risk factors. J Urol. 1999 Apr;161(4):1148-52.[↩]
- Xiao H, Mazumdar M, Bajorin DF, Sarosdy M, Vlamis V, Spicer J, Ferrara J, Bosl GJ, Motzer RJ. Long-term follow-up of patients with good-risk germ cell tumors treated with etoposide and cisplatin. J Clin Oncol. 1997 Jul;15(7):2553-8. doi: 10.1200/JCO.1997.15.7.2553[↩][↩][↩][↩][↩][↩][↩]
- Nonseminoma Testicular Cancer Treatment Protocols. https://emedicine.medscape.com/article/2006613-overview[↩][↩][↩][↩]
- Saxman SB, Finch D, Gonin R, Einhorn LH. Long-term follow-up of a phase III study of three versus four cycles of bleomycin, etoposide, and cisplatin in favorable-prognosis germ-cell tumors: the Indian University experience. J Clin Oncol. 1998 Feb;16(2):702-6. doi: 10.1200/JCO.1998.16.2.702[↩][↩][↩]
- Nichols CR, Saxman S. Primary salvage treatment of recurrent germ cell tumors: experience at Indiana University. Semin Oncol. 1998 Apr;25(2):210-4.[↩][↩]
- Loehrer PJ Sr, Gonin R, Nichols CR, Weathers T, Einhorn LH. Vinblastine plus ifosfamide plus cisplatin as initial salvage therapy in recurrent germ cell tumor. J Clin Oncol. 1998 Jul;16(7):2500-4. doi: 10.1200/JCO.1998.16.7.2500[↩]
- Kondagunta GV, Bacik J, Donadio A, Bajorin D, Marion S, Sheinfeld J, Bosl GJ, Motzer RJ. Combination of paclitaxel, ifosfamide, and cisplatin is an effective second-line therapy for patients with relapsed testicular germ cell tumors. J Clin Oncol. 2005 Sep 20;23(27):6549-55. doi: 10.1200/JCO.2005.19.638[↩]
- Einhorn LH, Williams SD, Chamness A, Brames MJ, Perkins SM, Abonour R. High-dose chemotherapy and stem-cell rescue for metastatic germ-cell tumors. N Engl J Med. 2007 Jul 26;357(4):340-8. doi: 10.1056/NEJMoa067749[↩][↩]
- Kondagunta GV, Bacik J, Sheinfeld J, Bajorin D, Bains M, Reich L, Deluca J, Budnick A, Ishill N, Mazumdar M, Bosl GJ, Motzer RJ. Paclitaxel plus Ifosfamide followed by high-dose carboplatin plus etoposide in previously treated germ cell tumors. J Clin Oncol. 2007 Jan 1;25(1):85-90. doi: 10.1200/JCO.2006.06.9401. Erratum in: J Clin Oncol. 2007 May 20;25(15):2149.[↩]
- De Giorgi U, Rosti G, Aieta M, Testore F, Burattini L, Fornarini G, Naglieri E, Lo Re G, Zumaglini F, Marangolo M. Phase II study of oxaliplatin and gemcitabine salvage chemotherapy in patients with cisplatin-refractory nonseminomatous germ cell tumor. Eur Urol. 2006 Nov;50(5):1032-8; discussion 1038-9. doi: 10.1016/j.eururo.2006.05.011[↩]
- Pectasides D, Pectasides M, Farmakis D, Aravantinos G, Nikolaou M, Koumpou M, Gaglia A, Kostopoulou V, Mylonakis N, Skarlos D. Gemcitabine and oxaliplatin (GEMOX) in patients with cisplatin-refractory germ cell tumors: a phase II study. Ann Oncol. 2004 Mar;15(3):493-7. doi: 10.1093/annonc/mdh103[↩]
- Papiani G, Einhorn LH. Salvage chemotherapy with high-dose carboplatin plus etoposide and autologous peripheral blood stem cell transplant in male pure choriocarcinoma: a retrospective analysis of 13 cases. Bone Marrow Transplant. 2007 Aug;40(3):235-7. doi: 10.1038/sj.bmt.1705697[↩]
- Einhorn LH. Treatment of testicular cancer: a new and improved model. J Clin Oncol. 1990 Nov;8(11):1777-81. doi: 10.1200/JCO.1990.8.11.1777[↩]
- Borni M, Kolsi F, Kammoun B, Kallel R, Souissi G, Boudawara MZ. Testicular non-seminomatous germ cell tumors (NSGCT) and brain metastases: About a rare location of the posterior fossa. Urol Case Rep. 2018 Sep 4;21:61-63. doi: 10.1016/j.eucr.2018.08.025[↩]
- Krege S, Beyer J, Souchon R, Albers P, et al. European consensus conference on diagnosis and treatment of germ cell cancer: a report of the second meeting of the European Germ Cell Cancer Consensus Group (EGCCCG): part II. Eur Urol. 2008 Mar;53(3):497-513. doi: 10.1016/j.eururo.2007.12.025[↩]
- Nonseminomatous Testicular Tumors. https://emedicine.medscape.com/article/454477-overview#a2[↩][↩]
- Stephenson AJ, Bosl GJ, Motzer RJ, Bajorin DF, Stasi JP, Sheinfeld J. Nonrandomized comparison of primary chemotherapy and retroperitoneal lymph node dissection for clinical stage IIA and IIB nonseminomatous germ cell testicular cancer. J Clin Oncol. 2007 Dec 10;25(35):5597-602. doi: 10.1200/JCO.2007.12.0808[↩]
- Mykletun A, Dahl AA, Haaland CF, Bremnes R, Dahl O, Klepp O, Wist E, Fosså SD. Side effects and cancer-related stress determine quality of life in long-term survivors of testicular cancer. J Clin Oncol. 2005 May 1;23(13):3061-8. doi: 10.1200/JCO.2005.08.048[↩]
- Fosså SD, Chen J, Schonfeld SJ, McGlynn KA, McMaster ML, Gail MH, Travis LB. Risk of contralateral testicular cancer: a population-based study of 29,515 U.S. men. J Natl Cancer Inst. 2005 Jul 20;97(14):1056-66. doi: 10.1093/jnci/dji185[↩]
- Theodore Ch, Terrier-Lacombe MJ, Laplanche A, Benoit G, Fizazi K, Stamerra O, Wibault P. Bilateral germ-cell tumours: 22-year experience at the Institut Gustave Roussy. Br J Cancer. 2004 Jan 12;90(1):55-9. doi: 10.1038/sj.bjc.6601464[↩]
- Lamichhane A, Mukkamalla SKR. Seminoma. [Updated 2023 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK560513[↩]
- Brydøy M, Fosså SD, Klepp O, Bremnes RM, Wist EA, Wentzel-Larsen T, Dahl O. Paternity following treatment for testicular cancer. J Natl Cancer Inst. 2005 Nov 2;97(21):1580-8. doi: 10.1093/jnci/dji339[↩]
- Babosa M, Baki M, Bodrogi I, Gundy S. A study of children, fathered by men treated for testicular cancer, conceived before, during, and after chemotherapy. Med Pediatr Oncol. 1994;22(1):33-8. doi: 10.1002/mpo.2950220107[↩]
- Gordon W Jr, Siegmund K, Stanisic TH, McKnight B, Harris IT, Carroll PR, Paradelo JC, Meyers FJ, Chapman RA, Meyskens FL Jr. A study of reproductive function in patients with seminoma treated with radiotherapy and orchidectomy: (SWOG-8711). Southwest Oncology Group. Int J Radiat Oncol Biol Phys. 1997 Apr 1;38(1):83-94. doi: 10.1016/s0360-3016(97)00235-6[↩]
- Travis LB, Andersson M, Gospodarowicz M, van Leeuwen FE, Bergfeldt K, Lynch CF, Curtis RE, Kohler BA, Wiklund T, Storm H, Holowaty E, Hall P, Pukkala E, Sleijfer DT, Clarke EA, Boice JD Jr, Stovall M, Gilbert E. Treatment-associated leukemia following testicular cancer. J Natl Cancer Inst. 2000 Jul 19;92(14):1165-71. doi: 10.1093/jnci/92.14.1165[↩][↩]
- van Leeuwen FE, Stiggelbout AM, van den Belt-Dusebout AW, Noyon R, Eliel MR, van Kerkhoff EH, Delemarre JF, Somers R. Second cancer risk following testicular cancer: a follow-up study of 1,909 patients. J Clin Oncol. 1993 Mar;11(3):415-24. doi: 10.1200/JCO.1993.11.3.415[↩]
- Houck W, Abonour R, Vance G, Einhorn LH. Secondary leukemias in refractory germ cell tumor patients undergoing autologous stem-cell transplantation using high-dose etoposide. J Clin Oncol. 2004 Jun 1;22(11):2155-8. doi: 10.1200/JCO.2004.11.054[↩]
- Kollmannsberger C, Hartmann JT, Kanz L, Bokemeyer C. Therapy-related malignancies following treatment of germ cell cancer. Int J Cancer. 1999 Dec 10;83(6):860-3. doi: 10.1002/(sici)1097-0215(19991210)83:6<860::aid-ijc32>3.0.co;2-l[↩]
- Osanto S, Bukman A, Van Hoek F, Sterk PJ, De Laat JA, Hermans J. Long-term effects of chemotherapy in patients with testicular cancer. J Clin Oncol. 1992 Apr;10(4):574-9. doi: 10.1200/JCO.1992.10.4.574[↩][↩]
- Haugnes HS, Aass N, Fosså SD, Dahl O, Brydøy M, Aasebø U, Wilsgaard T, Bremnes RM. Pulmonary function in long-term survivors of testicular cancer. J Clin Oncol. 2009 Jun 10;27(17):2779-86. doi: 10.1200/JCO.2008.18.5181[↩]
- Fosså SD, Gilbert E, Dores GM, Chen J, McGlynn KA, Schonfeld S, Storm H, Hall P, Holowaty E, Andersen A, Joensuu H, Andersson M, Kaijser M, Gospodarowicz M, Cohen R, Pukkala E, Travis LB. Noncancer causes of death in survivors of testicular cancer. J Natl Cancer Inst. 2007 Apr 4;99(7):533-44. doi: 10.1093/jnci/djk111[↩][↩][↩]
- Meinardi MT, Gietema JA, van der Graaf WT, van Veldhuisen DJ, Runne MA, Sluiter WJ, de Vries EG, Willemse PB, Mulder NH, van den Berg MP, Koops HS, Sleijfer DT. Cardiovascular morbidity in long-term survivors of metastatic testicular cancer. J Clin Oncol. 2000 Apr;18(8):1725-32. doi: 10.1200/JCO.2000.18.8.1725[↩]
- Huddart RA, Norman A, Shahidi M, Horwich A, Coward D, Nicholls J, Dearnaley DP. Cardiovascular disease as a long-term complication of treatment for testicular cancer. J Clin Oncol. 2003 Apr 15;21(8):1513-23. doi: 10.1200/JCO.2003.04.173[↩][↩]
- van den Belt-Dusebout AW, Nuver J, de Wit R, Gietema JA, ten Bokkel Huinink WW, Rodrigus PT, Schimmel EC, Aleman BM, van Leeuwen FE. Long-term risk of cardiovascular disease in 5-year survivors of testicular cancer. J Clin Oncol. 2006 Jan 20;24(3):467-75. doi: 10.1200/JCO.2005.02.7193[↩][↩]
- Haugnes HS, Aass N, Fosså SD, Dahl O, Klepp O, Wist EA, Svartberg J, Wilsgaard T, Bremnes RM. Components of the metabolic syndrome in long-term survivors of testicular cancer. Ann Oncol. 2007 Feb;18(2):241-8. doi: 10.1093/annonc/mdl372[↩]
- Nuver J, Smit AJ, Wolffenbuttel BH, Sluiter WJ, Hoekstra HJ, Sleijfer DT, Gietema JA. The metabolic syndrome and disturbances in hormone levels in long-term survivors of disseminated testicular cancer. J Clin Oncol. 2005 Jun 1;23(16):3718-25. doi: 10.1200/JCO.2005.02.176[↩]